Enhancing Stress Detection: A Comprehensive Approach through rPPG Analysis and Deep Learning Techniques
Abstract
1. Introduction
- A novel system leveraging non-contact and physiological techniques is proposed, enabling the continuous monitoring of pervasive biomedical signals for long-term stress detection.
- Hybrid DL networks and models for rPPG signal reconstruction and Heart Rate (HR) estimation to significantly improve accuracy and efficiency in stress detection up to 95.83% with the UBFC-Phys dataset.
- Extensive experiments and empirical evaluations of Deep Learning (DL) models for stress detection provide valuable insights and comparisons.
2. Related Work
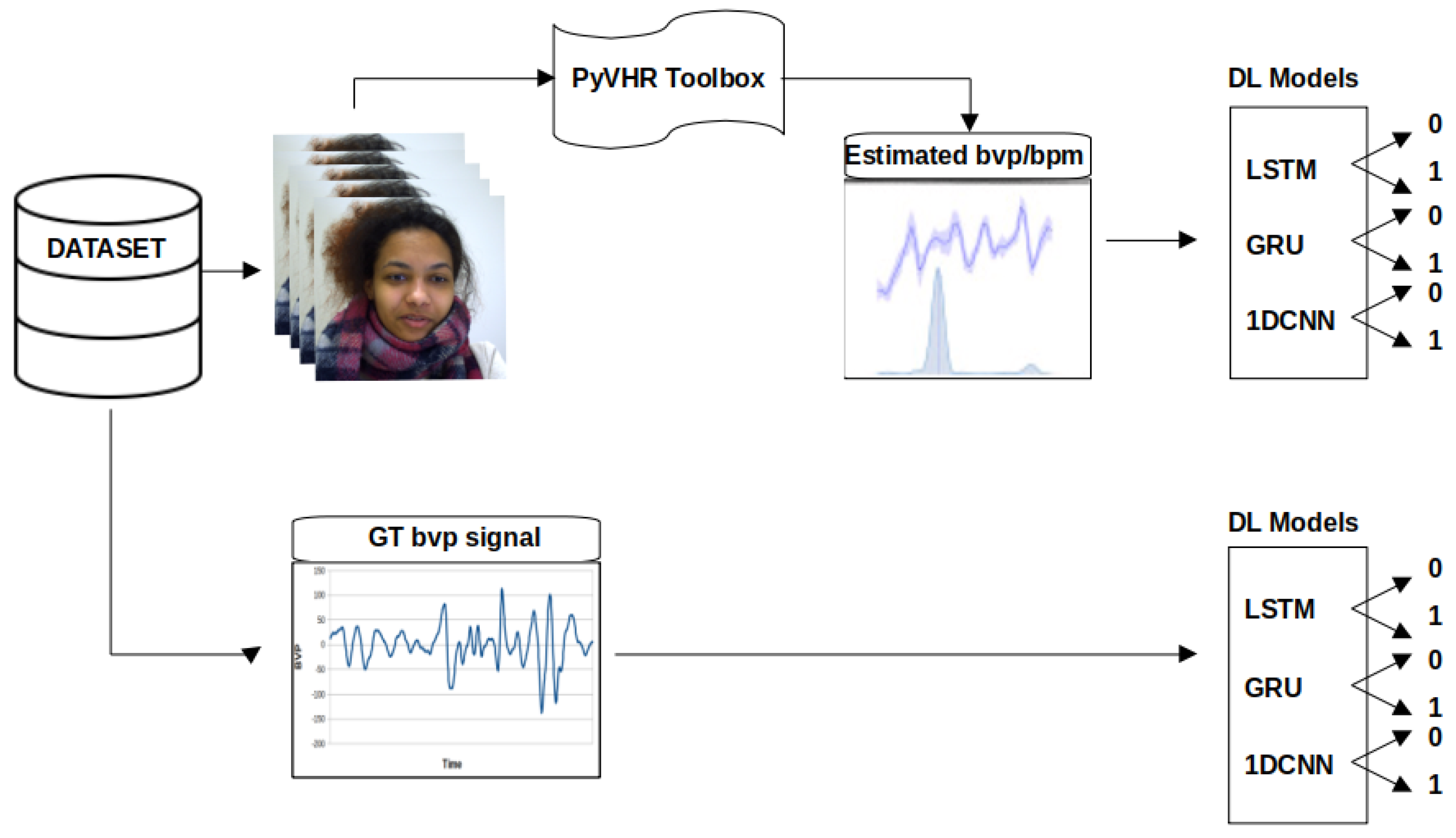
3. Method
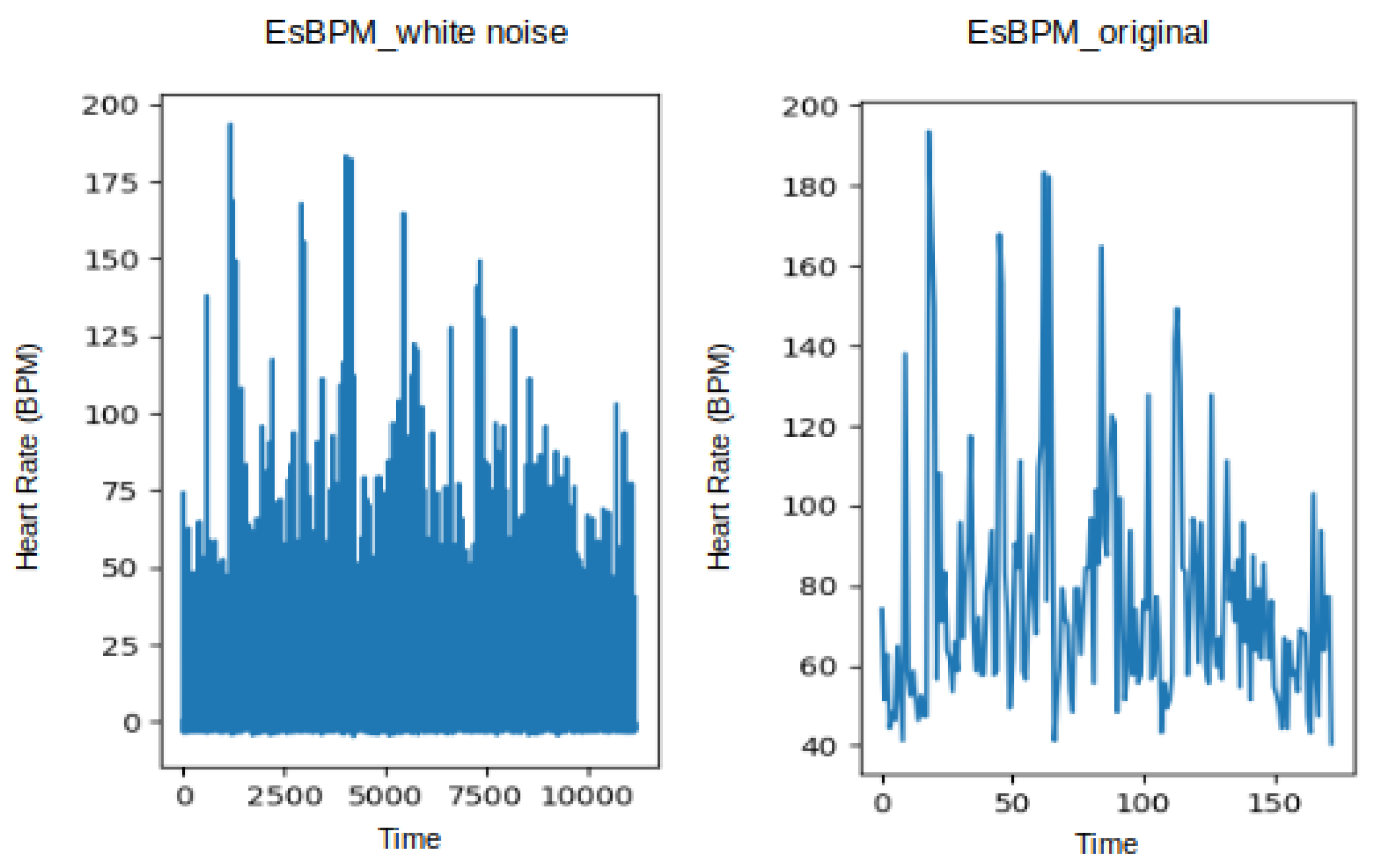
3.1. Dataset and Data Processing
3.2. Deep Learning Models
3.3. Performance Evaluation
4. Experimental Results
4.1. Classification Results
4.1.1. Performance Analysis of the DL Methods Applied to the GT Signal
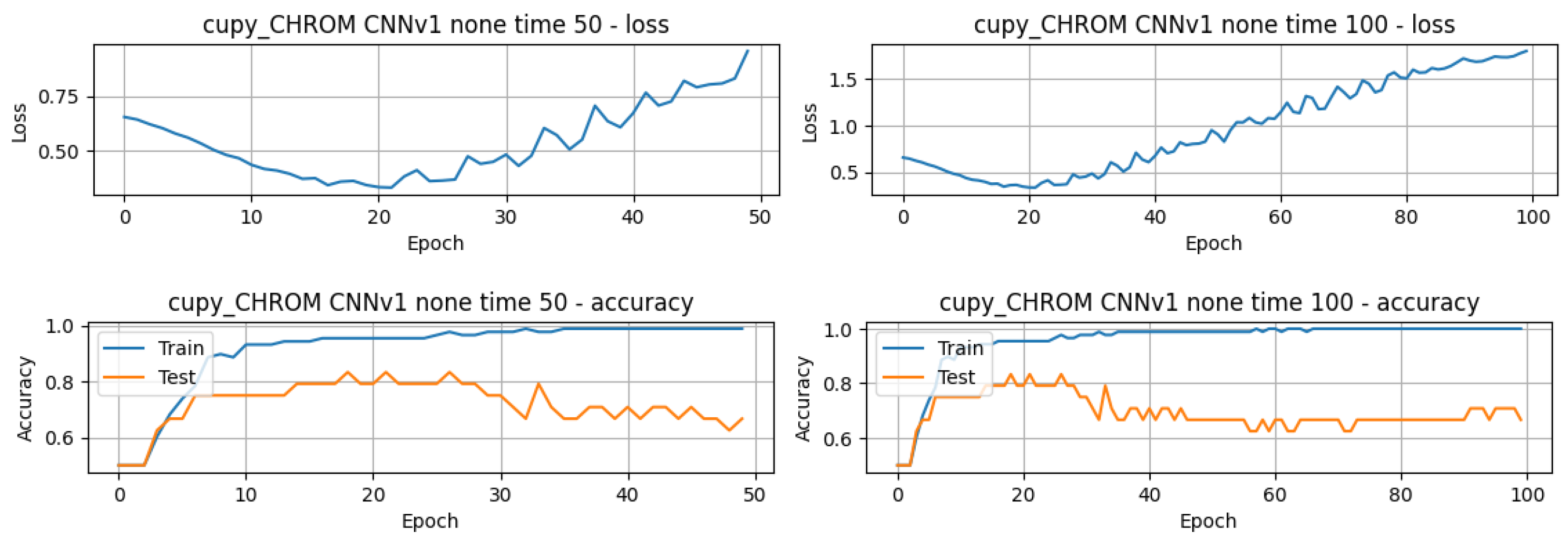
4.1.2. Performance Analysis of the DL Methods Applied to the rPPG Signal
5. Conclusions and Future Work
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Siddique, C.; D’Arcy, C. Adolescence, stress, and psychological well-being. J. Youth Adolesc. 1984, 13, 459–473. [Google Scholar] [CrossRef]
- Everly, G.S., Jr.; Lating, J.M.; Everly, G.S.; Lating, J.M. The anatomy and physiology of the human stress response. In A Clinical Guide to the Treatment of the Human Stress Response; Springer: New York, NY, USA, 2019; pp. 19–56. [Google Scholar]
- Thayer, J.F.; Åhs, F.; Fredrikson, M.; Sollers III, J.J.; Wager, T.D. A meta-analysis of heart rate variability and neuroimaging studies: Implications for heart rate variability as a marker of stress and health. Neurosci. Biobehav. Rev. 2012, 36, 747–756. [Google Scholar] [CrossRef]
- McCarty, R. The Fight-or-Flight Response: A Cornerstone of Stress Research; Elsevier: Amsterdam, The Netherlands, 2016; pp. 33–37. [Google Scholar]
- Thorsteinsson, E.B.; Brown, R.F.; Richards, C. The relationship between work-stress, psychological stress and staff health and work outcomes in office workers. Psychology 2014, 5, 1301–1311. [Google Scholar] [CrossRef]
- van Kraaij, A.W.J.; Schiavone, G.; Lutin, E.; Claes, S.; Van Hoof, C. Relationship between chronic stress and heart rate over time modulated by gender in a cohort of office workers: Cross-sectional study using wearable technologies. J. Med. Internet Res. 2020, 22, e18253. [Google Scholar] [CrossRef] [PubMed]
- McEwen, B.S. Neurobiological and systemic effects of chronic stress. Chronic Stress 2017, 1, 2470547017692328. [Google Scholar] [CrossRef] [PubMed]
- McKlveen, J.M.; Morano, R.L.; Fitzgerald, M.; Zoubovsky, S.; Cassella, S.N.; Scheimann, J.R.; Ghosal, S.; Mahbod, P.; Packard, B.A.; Myers, B.; et al. Chronic stress increases prefrontal inhibition: A mechanism for stress-induced prefrontal dysfunction. Biol. Psychiatry 2016, 80, 754–764. [Google Scholar] [CrossRef] [PubMed]
- Samson, C.; Koh, A. Stress monitoring and recent advancements in wearable biosensors. Front. Bioeng. Biotechnol. 2020, 8, 1037. [Google Scholar] [CrossRef] [PubMed]
- Dalmeida, K.M.; Masala, G.L. HRV features as viable physiological markers for stress detection using wearable devices. Sensors 2021, 21, 2873. [Google Scholar] [CrossRef] [PubMed]
- Shaffer, F.; Ginsberg, J.P. An overview of heart rate variability metrics and norms. Front. Public Health 2017, 5, 258. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Arce, J.; Lara-Flores, L.; Portillo-Rodríguez, O.; Martínez-Méndez, R. Towards an anxiety and stress recognition system for academic environments based on physiological features. Comput. Methods Programs Biomed. 2020, 190, 105408. [Google Scholar] [CrossRef] [PubMed]
- Greco, A.; Valenza, G.; Lázaro, J.; Garzón-Rey, J.M.; Aguiló, J.; De-la Camara, C.; Bailón, R.; Scilingo, E.P. Acute stress state classification based on electrodermal activity modeling. IEEE Trans. Affect. Comput. 2021, 14, 788–799. [Google Scholar] [CrossRef]
- Pourmohammadi, S.; Maleki, A. Stress detection using ECG and EMG signals: A comprehensive study. Comput. Methods Programs Biomed. 2020, 193, 105482. [Google Scholar] [CrossRef] [PubMed]
- Marois, A.; Lafond, D.; Gagnon, J.F.; Vachon, F.; Cloutier, M.S. Predicting stress among pedestrian traffic workers using physiological and situational measures. In Proceedings of the Human Factors and Ergonomics Society Annual Meeting; SAGE Publications Sage CA: Los Angeles, CA, USA, 2018; Volume 62, pp. 1262–1266. [Google Scholar]
- Sánchez-Reolid, R.; Martínez-Rodrigo, A.; López, M.T.; Fernández-Caballero, A. Deep support vector machines for the identification of stress condition from electrodermal activity. Int. J. Neural Syst. 2020, 30, 2050031. [Google Scholar] [CrossRef]
- Tanev, G.; Saadi, D.B.; Hoppe, K.; Sorensen, H.B. Classification of acute stress using linear and non-linear heart rate variability analysis derived from sternal ECG. In Proceedings of the 2014 36th Annual International Conference of the IEEE Engineering in Medicine and Biology Society, Chicago, IL, USA, 26–30 August 2014; pp. 3386–3389. [Google Scholar]
- Garbarino, M.; Lai, M.; Bender, D.; Picard, R.W.; Tognetti, S. Empatica E3—A wearable wireless multi-sensor device for real-time computerized biofeedback and data acquisition. In Proceedings of the 2014 4th International Conference on Wireless Mobile Communication and Healthcare-Transforming Healthcare Through Innovations in Mobile and Wireless Technologies (MOBIHEALTH), Athens, Greece, 3–5 November 2014; pp. 39–42. [Google Scholar]
- Shcherbina, A.; Mattsson, C.M.; Waggott, D.; Salisbury, H.; Christle, J.W.; Hastie, T.; Wheeler, M.T.; Ashley, E.A. Accuracy in wrist-worn, sensor-based measurements of heart rate and energy expenditure in a diverse cohort. J. Pers. Med. 2017, 7, 3. [Google Scholar] [CrossRef]
- Caminal, P.; Sola, F.; Gomis, P.; Guasch, E.; Perera, A.; Soriano, N.; Mont, L. Validity of the Polar V800 monitor for measuring heart rate variability in mountain running route conditions. Eur. J. Appl. Physiol. 2018, 118, 669–677. [Google Scholar] [CrossRef]
- Salai, M.; Vassányi, I.; Kósa, I. Stress detection using low cost heart rate sensors. J. Healthc. Eng. 2016, 2016. [Google Scholar] [CrossRef]
- Moridani, M.; Mahabadi, Z.; Javadi, N. Heart rate variability features for different stress classification. Bratisl. Lek. Listy 2020, 121, 619–627. [Google Scholar] [CrossRef] [PubMed]
- Salahuddin, L.; Cho, J.; Jeong, M.G.; Kim, D. Ultra short term analysis of heart rate variability for monitoring mental stress in mobile settings. In Proceedings of the 2007 29th Annual International Conference of the IEEE Engineering in Medicine and Biology Society, Lyon, France, 22–26 August 2007; pp. 4656–4659. [Google Scholar]
- Schmidt, P.; Reiss, A.; Duerichen, R.; Marberger, C.; Van Laerhoven, K. Introducing wesad, a multimodal dataset for wearable stress and affect detection. In Proceedings of the 20th ACM International Conference on Multimodal Interaction, Boulder, CO, USA, 16–20 October 2018; pp. 400–408. [Google Scholar]
- Chudy, N.S. Testing of Wrist-Worn-Fitness-Tracking Devices during Cognitive Stress: A Validation Study. Bachelor’s Thesis, University of Central Florida, Orlando, FL, USA, 2017. [Google Scholar]
- Giles, D.; Draper, N.; Neil, W. Validity of the Polar V800 heart rate monitor to measure RR intervals at rest. Eur. J. Appl. Physiol. 2016, 116, 563–571. [Google Scholar] [CrossRef]
- Heikenfeld, J.; Jajack, A.; Rogers, J.; Gutruf, P.; Tian, L.; Pan, T.; Li, R.; Khine, M.; Kim, J.; Wang, J. Wearable sensors: Modalities, challenges, and prospects. Lab Chip 2018, 18, 217–248. [Google Scholar] [CrossRef]
- McDuff, D. Camera Measurement of Physiological Vital Signs. Acm Comput. Surv. 2023, 55, 1–40. [Google Scholar] [CrossRef]
- Sabour, R.M.; Benezeth, Y.; De Oliveira, P.; Chappe, J.; Yang, F. Ubfc-phys: A multimodal database for psychophysiological studies of social stress. IEEE Trans. Affect. Comput. 2021, 14, 622–636. [Google Scholar] [CrossRef]
- Cheng, Y.C.; Chou, T.I.; Indikawati, F.I.; Winiarti, S.; Dahlan, A.; Selatan, R.; Yogyakarta, D. Stress Detection from Multimodal Wearable Sensor Data. IOP Conf. Ser. Mater. Sci. Eng. 2020, 771, 012028. [Google Scholar] [CrossRef]
- Herranz Olazábal, J.; Wieringa, F.; Hermeling, E.; Van Hoof, C. Camera-Derived Photoplethysmography (rPPG) and Speckle Plethysmography (rSPG): Comparing Reflective and Transmissive Mode at Various Integration Times Using LEDs and Lasers. Sensors 2022, 22, 6059. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Wang, H.; Lu, F. Assessment of Deep Learning-Based Heart Rate Estimation Using Remote Photoplethysmography Under Different Illuminations. IEEE Trans. Hum. Mach. Syst. 2022, 52, 1236–1246. [Google Scholar] [CrossRef]
- Schneiderman, N.; Ironson, G.; Siegel, S.D. Stress and health: Psychological, behavioral, and biological determinants. Annu. Rev. Clin. Psychol. 2005, 1, 607–628. [Google Scholar] [CrossRef] [PubMed]
- Selye, H. Short letter. Nature 1936, 138, 32. [Google Scholar] [CrossRef]
- Dhama, K.; Latheef, S.K.; Dadar, M.; Samad, H.A.; Munjal, A.; Khandia, R.; Karthik, K.; Tiwari, R.; Yatoo, M.I.; Bhatt, P.; et al. Biomarkers in stress related diseases/disorders: Diagnostic, prognostic, and therapeutic values. Front. Mol. Biosci. 2019, 6, 91. [Google Scholar] [CrossRef]
- Can, Y.S.; Arnrich, B.; Ersoy, C. Stress detection in daily life scenarios using smart phones and wearable sensors: A survey. J. Biomed. Inform. 2019, 92, 103139. [Google Scholar] [CrossRef]
- Arsalan, A.; Anwar, S.M.; Majid, M. Mental stress detection using data from wearable and non-wearable sensors: A review. arXiv 2022, arXiv:2202.03033. [Google Scholar]
- Nath, R.K.; Thapliyal, H. Smart wristband-based stress detection framework for older adults with cortisol as stress biomarker. IEEE Trans. Consum. Electron. 2021, 67, 30–39. [Google Scholar] [CrossRef]
- Chan, S.F.; La Greca, A.M. Perceived stress scale (PSS). In Encyclopedia of Behavioral Medicine; Springer: Berlin/Heidelberg, Germany, 2020; pp. 1646–1648. [Google Scholar]
- Cheng, B.; Fan, C.; Fu, H.; Huang, J.; Chen, H.; Luo, X. Measuring and computing cognitive statuses of construction workers based on electroencephalogram: A critical review. IEEE Trans. Comput. Soc. Syst. 2022, 9, 1644–1659. [Google Scholar] [CrossRef]
- Wang, X.; Li, D.; Menassa, C.C.; Kamat, V.R. Investigating the effect of indoor thermal environment on occupants’ mental workload and task performance using electroencephalogram. Build. Environ. 2019, 158, 120–132. [Google Scholar] [CrossRef]
- Abellán-Huerta, J.; Prieto-Valiente, L.; Montoro-García, S.; Abellán-Alemán, J.; Soria-Arcos, F. Correlation of blood pressure variability as measured by clinic, self-measurement at home, and ambulatory blood pressure monitoring. Am. J. Hypertens. 2018, 31, 305–312. [Google Scholar] [CrossRef]
- Chen, Y.; Rao, M.; Feng, K.; Niu, G. Modified Varying Index Coefficient Autoregression Model for Representation of the Nonstationary Vibration From a Planetary Gearbox. IEEE Trans. Instrum. Meas. 2023, 72, 1–12. [Google Scholar] [CrossRef]
- Shahid, M.M.; Agada, G.E.; Kayyali, M.; Ihianle, I.K.; Machado, P. Towards Enhanced Well-Being: Monitoring Stress and Health with Smart Sensor Systems. In Proceedings of the 2023 International Conference Automatics and Informatics (ICAI), Varna, Bulgaria, 5–7 October 2023; pp. 432–437. [Google Scholar]
- Ihianle, I.K.; Machado, P.; Owa, K.; Adama, D.A.; Otuka, R.; Lotfi, A. Minimising redundancy, maximising relevance: HRV feature selection for stress classification. Expert Syst. Appl. 2024, 239, 122490. [Google Scholar] [CrossRef]
- Benezeth, Y.; Bobbia, S.; Nakamura, K.; Gomez, R.; Dubois, J. Probabilistic signal quality metric for reduced complexity unsupervised remote photoplethysmography. In Proceedings of the 2019 13th International Symposium on Medical Information and Communication Technology (ISMICT), Oslo, Norway, 8–10 May 2019; pp. 1–5. [Google Scholar]
- Hassan, M.; Malik, A.; Fofi, D.; Saad, N.; Meriaudeau, F. Novel health monitoring method using an RGB camera. Biomed. Opt. Express 2017, 8, 4838–4854. [Google Scholar] [CrossRef] [PubMed]
- Selvaraju, V.; Spicher, N.; Wang, J.; Ganapathy, N.; Warnecke, J.M.; Leonhardt, S.; Swaminathan, R.; Deserno, T.M. Continuous monitoring of vital signs using cameras: A systematic review. Sensors 2022, 22, 4097. [Google Scholar] [CrossRef] [PubMed]
- Lee, R.J.; Sivakumar, S.; Lim, K.H. Review on remote heart rate measurements using photoplethysmography. Multimed. Tools Appl. 2023, 1–30. [Google Scholar]
- Abbas, L.; Samy, S.; Ghazal, R.; Eldeib, A.M.; ElGohary, S.H. Contactless Vital Signs Monitoring for Public Health Welfare. In Proceedings of the 2021 9th International Japan-Africa Conference on Electronics, Communications, and Computations (JAC-ECC), Alexandria, Egypt, 13–14 December 2021; pp. 183–186. [Google Scholar]
- Yu, Z.; Li, X.; Zhao, G. Facial-video-based physiological signal measurement: Recent advances and affective applications. IEEE Signal Process. Mag. 2021, 38, 50–58. [Google Scholar] [CrossRef]
- Casado, C.Á.; Cañellas, M.L.; López, M.B. Depression recognition using remote photoplethysmography from facial videos. IEEE Trans. Affect. Comput. 2023, 14, 3305–3316. [Google Scholar] [CrossRef]
- Lokendra, B.; Puneet, G. AND-rPPG: A novel denoising-rPPG network for improving remote heart rate estimation. Comput. Biol. Med. 2022, 141, 105146. [Google Scholar] [CrossRef] [PubMed]
- Hasanpoor, Y.; Motaman, K.; Tarvirdizadeh, B.; Alipour, K.; Ghamari, M. Stress Detection Using PPG Signal and Combined Deep CNN-MLP Network. In Proceedings of the 2022 29th National and 7th International Iranian Conference on Biomedical Engineering (ICBME), Tehran, Iran, 21–22 December 2022; pp. 223–228. [Google Scholar]
- Kirschbaum, C.; Pirke, K.M.; Hellhammer, D.H. The ‘Trier Social Stress Test’–a tool for investigating psychobiological stress responses in a laboratory setting. Neuropsychobiology 1993, 28, 76–81. [Google Scholar] [CrossRef] [PubMed]
- Boccignone, G.; Conte, D.; Cuculo, V.; D’Amelio, A.; Grossi, G.; Lanzarotti, R.; Mortara, E. pyVHR: A Python framework for remote photoplethysmography. PeerJ Comput. Sci. 2022, 8, e929. [Google Scholar] [CrossRef] [PubMed]






| Parameters | Description |
|---|---|
| Window | The number of consecutive video frames processed to estimate the physiological signal. |
| Holistic | Skin extraction technique that sets the stage for calculating the RGB trace, which is achieved by calculating the average intensity of facial skin colour for each channel separately. |
| Convexhull | A skin extractor that subtracts the eyes and mouth regions from the rest of the entire face. It offers dependable real-time face and landmark detection and tracking. |
| CuPy CHROM | A chrominance-based method used to infer the pulse signal from the RGB traces built with the CuPy Python library designed for GPU-accelerated computing with open-source arrays. |
| Torch CHROM | Built with PyTorch, which is an open-source ML framework that facilitates building, training, and deploying DL models through a dynamic computational graph. |
| Cupy POS | Plane POS is another method also used to infer the pulse signal from RGB traces, but from a projection plane that is perpendicular to the skin tone built with the CuPy library. |
| DL Method | # Layers | Layer (Type) | Output Shape | Param # | Total Params | Trainable Params | Non-Trainable Params |
|---|---|---|---|---|---|---|---|
| LSTMv1 | 3 | lstm | 11,519 × 64 | 16,896 | 22,097 | 22,097 | 0 |
| lstm | 16 | 5184 | |||||
| dense | 1 | ||||||
| LSTMv2 | 4 | lstm | 11,519 × 64 | 16,896 | 32,465 | 32,465 | 0 |
| lstm | 11,519 × 32 | 12,416 | |||||
| lstm | 16 | 3136 | |||||
| dense | 1 | 17 | |||||
| GRUv1 | 3 | gru | 11,519 × 64 | 12,864 | 16,817 | 16,817 | 0 |
| gru | 16 | 3936 | |||||
| dense | 1 | 17 | |||||
| GRUv2 | 4 | gru | 11,519 × 64 | 12,864 | 24,689 | 24,689 | 0 |
| gru | 11,519 × 32 | 9408 | |||||
| gru | 16 | 2400 | |||||
| dense | 1 | 17 | |||||
| 1D-CNNv1 | 5 | conv1d | 11,517 × 64 | 256 | 1,480,001 | 1,480,001 | 0 |
| max_pooling | 5758 × 64 | 0 | |||||
| conv1d | 5756 × 32 | 6167 | |||||
| max_pooling | 5756 × 32 | 0 | |||||
| flatten | 92,096 | 0 | |||||
| dense | 16 | 1,473,552 | |||||
| dense | 1 | 17 | |||||
| 1D-CNNv2 | 7 | conv1d | 5744 × 512 | 16896 | 2,765,441 | 2,765,441 | 1792 |
| max_pooling | 1436 × 512 | 0 | |||||
| batch_normalisation | 1436 × 512 | 2048 | |||||
| conv1d | 1429 × 256 | 1,048,832 | |||||
| max_pooling | 357 × 256 | 0 | |||||
| batch_normalisation | 357 × 356 | 1024 | |||||
| conv1d | 350 × 128 | 262,272 | |||||
| max_pooling | 87 × 128 | 0 | |||||
| batch_normalisation | 87 × 128 | 512 | |||||
| flatten | 11136 | 0 | |||||
| dense | 128 | 1,425,536 | |||||
| dropout | 128 | 0 | |||||
| dense | 64 | 8256 | |||||
| dropout | 64 | 0 | |||||
| dense | 1 | 65 | |||||
| 1D-CNNv3 | 7 | conv1d | 57,44 × 512 | 16,896 | 4,199,169 | 4,199,169 | 1792 |
| max_pooling | 1436 × 512 | 0 | |||||
| batch_normalisation | 1436 × 512 | 2048 | |||||
| conv1d | 1429 × 256 | 1,048,832 | |||||
| max_pooling | 357 × 256 | 0 | |||||
| batch_normalisation | 357 × 256 | 1024 | |||||
| conv1d | 350 × 128 | 262,272 | |||||
| max_pooling | 87 × 128 | 0 | |||||
| batch_normalisation | 87 × 128 | 512 | |||||
| flatten | 11,136 | 0 | |||||
| dense | 256 | 2,851,072 | |||||
| dropout | 256 | 0 | |||||
| dense | 64 | 16,448 | |||||
| dropout | 64 | 0 | |||||
| dense | 1 | 65 |
| DL Method | Domain | Epochs | Accuracy | Precision | Recall | F1-Score | Time [s] |
|---|---|---|---|---|---|---|---|
| 1D-CNNv2 | time | 100 | 83.33% | 83.33% | 83.33% | 83.33% | 28.08 |
| LSTMv1 | time | 100 | 79.17% | 100.00% | 58.33% | 73.68% | 122.62 |
| GRUv1 | time | 50 | 79.17% | 81.82% | 75.00% | 78.26% | 60.68 |
| GRUv1 | time | 100 | 79.17% | 81.82% | 75.00% | 78.26% | 119.67 |
| 1D-CNNv3 | time | 50 | 79.17% | 81.82% | 75.00% | 78.26% | 15.21 |
| LSTMv2 | time | 50 | 75.00% | 87.50% | 58.33% | 70.00% | 92.04 |
| LSTMv2 | time | 100 | 75.00% | 87.50% | 58.33% | 70.00% | 182.08 |
| GRUv2 | time | 50 | 75.00% | 80.00% | 66.67% | 72.73% | 89.58 |
| GRUv2 | time | 100 | 75.00% | 80.00% | 66.67% | 72.73% | 175.43 |
| 1D-CNNv1 | time | 50 | 75.00% | 87.50% | 58.33% | 70.00% | 5.48 |
| 1D-CNNv1 | time | 100 | 75.00% | 87.50% | 58.33% | 70.00% | 7.62 |
| 1D-CNNv3 | time | 100 | 75.00% | 100.00% | 50.00% | 66.67% | 28.49 |
| 1D-CNNv3 | frequency | 100 | 75.00% | 100.00% | 50.00% | 66.67% | 28.74 |
| LSTMv1 | time | 50 | 70.83% | 100.00% | 41.67% | 58.82% | 62.28 |
| GRUv1 | frequency | 100 | 66.67% | 61.11% | 91.67% | 73.33% | 119.30 |
| LSTMv1 | frequency | 50 | 62.50% | 57.89% | 91.67% | 70.97% | 61.96 |
| GRUv2 | frequency | 100 | 62.50% | 57.89% | 91.67% | 70.97% | 175.16 |
| LSTMv1 | frequency | 100 | 58.33% | 55.00% | 91.67% | 68.75% | 122.32 |
| GRUv1 | frequency | 50 | 58.33% | 56.25% | 75.00% | 64.29% | 60.47 |
| 1D-CNNv1 | frequency | 100 | 54.17% | 52.63% | 83.33% | 64.52% | 7.49 |
| LSTMv2 | frequency | 100 | 50.00% | 50.00% | 100.00% | 66.67% | 181.87 |
| GRUv2 | frequency | 50 | 50.00% | 50.00% | 8.33% | 14.29% | 89.23 |
| 1D-CNNv1 | frequency | 50 | 50.00% | 50.00% | 100.00% | 66.67% | 4.24 |
| 1D-CNNv2 | time | 50 | 50.00% | 50.00% | 100.00% | 66.67% | 19.75 |
| 1D-CNNv2 | frequency | 50 | 50.00% | 50.00% | 8.33% | 14.29% | 15.42 |
| 1D-CNNv2 | frequency | 100 | 50.00% | 50.00% | 8.33% | 14.29% | 28.14 |
| 1D-CNNv3 | frequency | 50 | 50.00% | 50.00% | 8.33% | 14.29% | 15.07 |
| LSTMv2 | frequency | 50 | 41.67% | 41.67% | 41.67% | 41.67% | 92.44 |
| Work | PPG Method | ML-Method | Accuracy |
|---|---|---|---|
| This work | contact | 1D-CNN-MLP | 83.33% |
| remote | 95.83% | ||
| UBFC-Phys [29] | contact | SVM-linear kernel | 73.00% |
| remote | SVM-RBF kernel | 85.38% | |
| Stress detection using PPG signal and combined deep CNN-MLP network [56] | contact | CNN-MLP | 82.00% |
| pyVHR Method | DL-Method | Version | Aug. | Domain | Epochs | Accuracy | Precision | Recall | F1-Score | Time |
|---|---|---|---|---|---|---|---|---|---|---|
| CuPy_CHROM | LSTM | v1 | inter | freq | 50 | 83.33% | 83.33% | 83.33% | 83.33% | 59.4 |
| v2 | none | freq | 100 | 83.33% | 90.00% | 75.00% | 81.82% | 9.8 | ||
| GRU | v2 | none | freq | 50 | 83.33% | 83.33% | 83.33% | 83.33% | 6.3 | |
| v1 | none | freq | 50 | 79.17% | 76.92% | 83.33% | 80.00% | 4.6 | ||
| 1D-CNN | v1 | wn | freq | 100 | 95.83% | 100.00% | 91.67% | 95.65% | 7.8 | |
| v2 | inter | freq | 50 | 95.83% | 100.00% | 91.67% | 95.65% | 14.5 | ||
| v3 | inter | time | 50 | 91.67% | 100.00% | 83.33% | 90.91% | 15.0 | ||
| Torch_CHROM | LSTM | v1 | inter | freq | 50 | 83.33% | 83.33% | 83.33% | 83.33% | 59.6 |
| v2 | none | freq | 50 | 83.33% | 90.00% | 75.00% | 81.82% | 6.6 | ||
| GRU | v3 | wn | freq | 100 | 83.33% | 78.57% | 91.67% | 84.62% | 114.7 | |
| v2 | none | freq | 50 | 83.33% | 83.33% | 83.33% | 83.33% | 6.5 | ||
| 1D-CNN | v3 | inter | time | 50 | 95.83% | 92.31% | 100.00% | 96.00% | 15.1 | |
| v2 | inter | freq | 100 | 91.67% | 100.00% | 83.33% | 90.91% | 27.6 | ||
| v1 | none | freq | 50 | 87.50% | 84.62% | 91.67% | 88.00% | 2.4 | ||
| CuPy_POS | LSTM | v2 | none | freq | 50 | 83.33% | 83.33% | 83.33% | 83.33% | 6.0 |
| v1 | inter | time | 50 | 79.17% | 76.92% | 83.33% | 80.00% | 59.2 | ||
| GRU | v1 | none | time | 50 | 83.33% | 83.33% | 83.33% | 83.33% | 5.3 | |
| v1 | none | time | 100 | 83.33% | 83.33% | 83.33% | 83.33% | 9.7 | ||
| 1D-CNN | v1 | inter | time | 50 | 83.33% | 83.33% | 83.33% | 83.33% | 14.8 | |
| v3 | wn | time | 50 | 83.33% | 83.33% | 83.33% | 83.33% | 14.5 | ||
| v1 | none | freq | 50 | 79.17% | 73.33% | 91.67% | 81.48% | 2.1 |
| pyVHR Method | dl_Method | Aug. | Domain | Epochs | Ac | Pr | Re | F1 | Time (s) |
|---|---|---|---|---|---|---|---|---|---|
| CuPy_CHROM | 1D-CNNv3 | inter | frequency | 50 | 1 | 1 | 1 | 1 | 14.59 |
| CuPy_CHROM | 1D-CNNv3 | inter | frequency | 100 | 1 | 1 | 1 | 1 | 27.00 |
| Torch_CHROM | 1D-CNNv2 | inter | frequency | 50 | 1 | 1 | 1 | 1 | 14.44 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fontes, L.; Machado, P.; Vinkemeier, D.; Yahaya, S.; Bird, J.J.; Ihianle, I.K. Enhancing Stress Detection: A Comprehensive Approach through rPPG Analysis and Deep Learning Techniques. Sensors 2024, 24, 1096. https://doi.org/10.3390/s24041096
Fontes L, Machado P, Vinkemeier D, Yahaya S, Bird JJ, Ihianle IK. Enhancing Stress Detection: A Comprehensive Approach through rPPG Analysis and Deep Learning Techniques. Sensors. 2024; 24(4):1096. https://doi.org/10.3390/s24041096
Chicago/Turabian StyleFontes, Laura, Pedro Machado, Doratha Vinkemeier, Salisu Yahaya, Jordan J. Bird, and Isibor Kennedy Ihianle. 2024. "Enhancing Stress Detection: A Comprehensive Approach through rPPG Analysis and Deep Learning Techniques" Sensors 24, no. 4: 1096. https://doi.org/10.3390/s24041096
APA StyleFontes, L., Machado, P., Vinkemeier, D., Yahaya, S., Bird, J. J., & Ihianle, I. K. (2024). Enhancing Stress Detection: A Comprehensive Approach through rPPG Analysis and Deep Learning Techniques. Sensors, 24(4), 1096. https://doi.org/10.3390/s24041096






