3.6 mW Active-Electrode ECG/ETI Sensor System Using Wideband Low-Noise Instrumentation Amplifier and High Impedance Balanced Current Driver
Abstract
1. Introduction
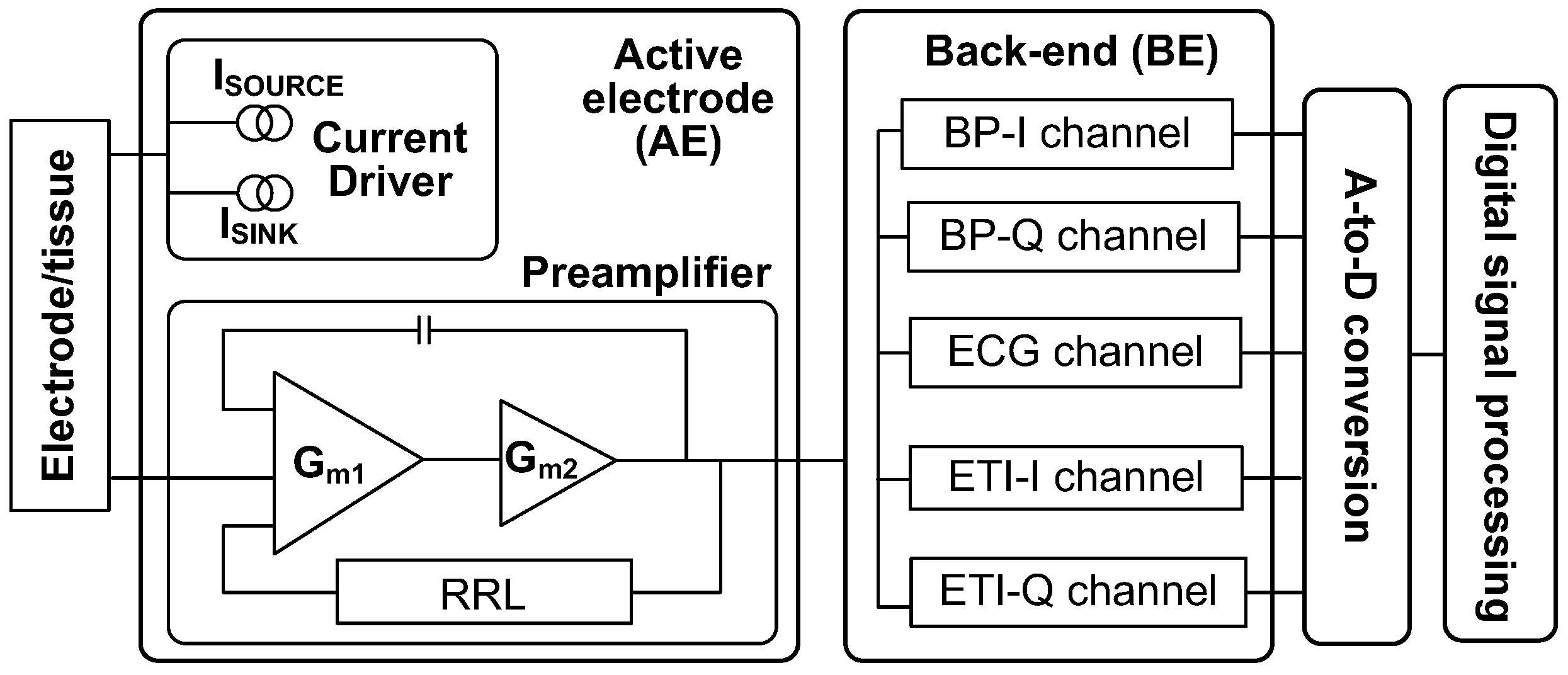
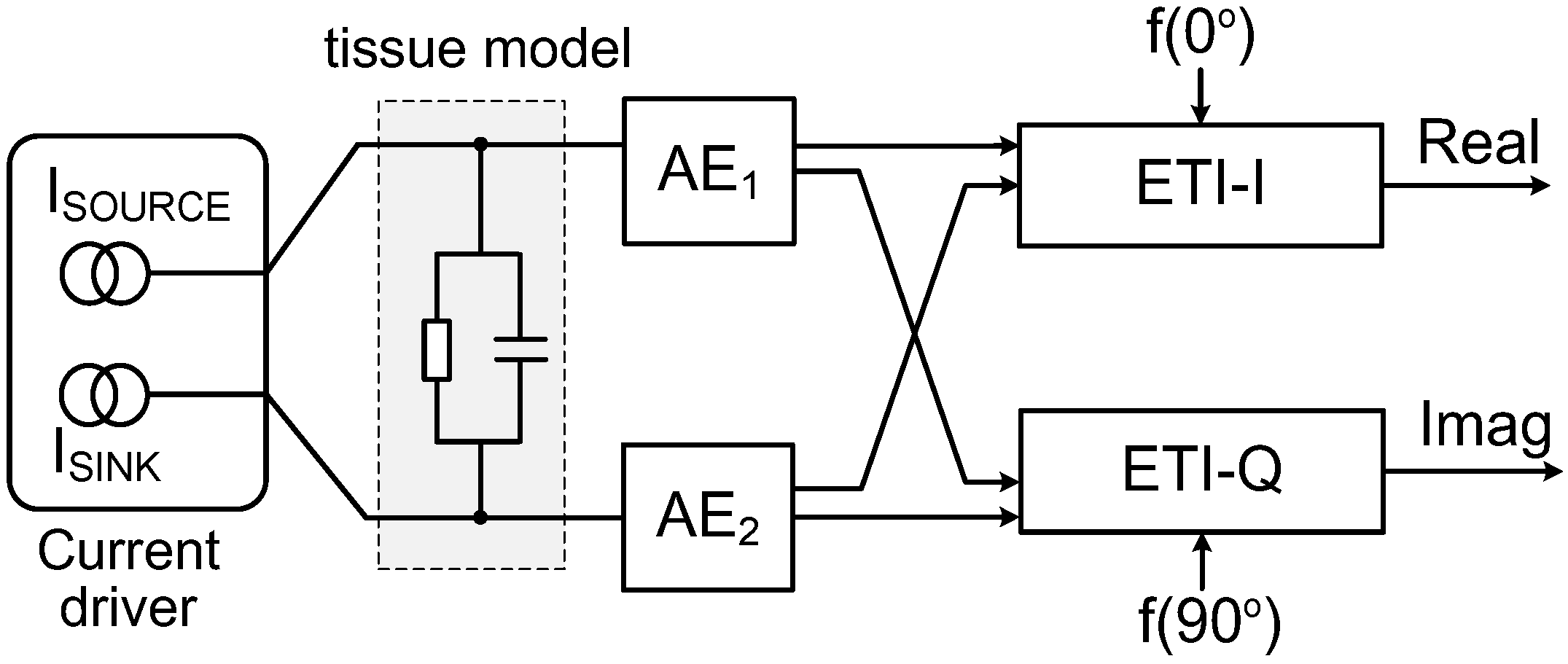
2. System Architecture
3. Active Electrode IC
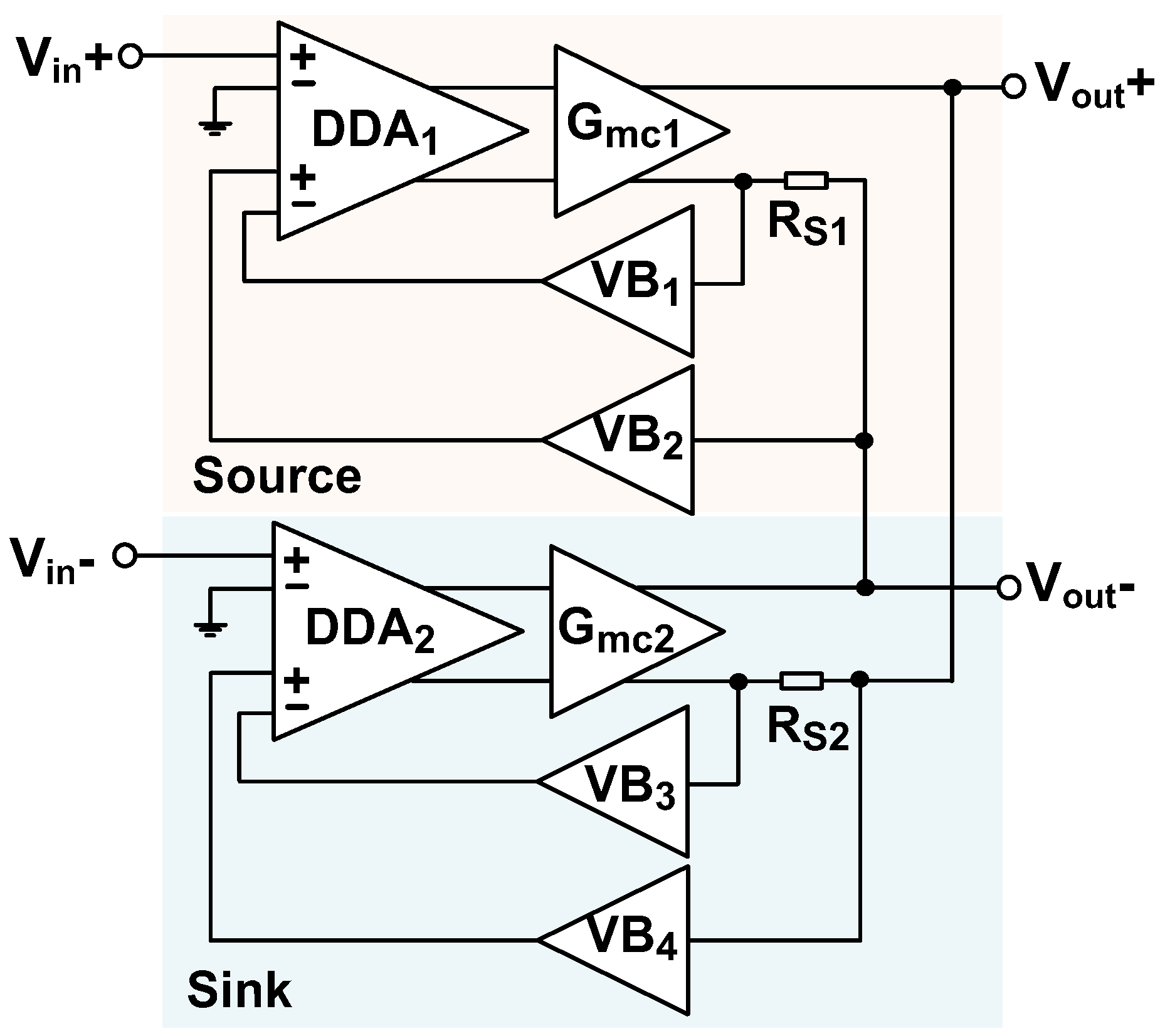
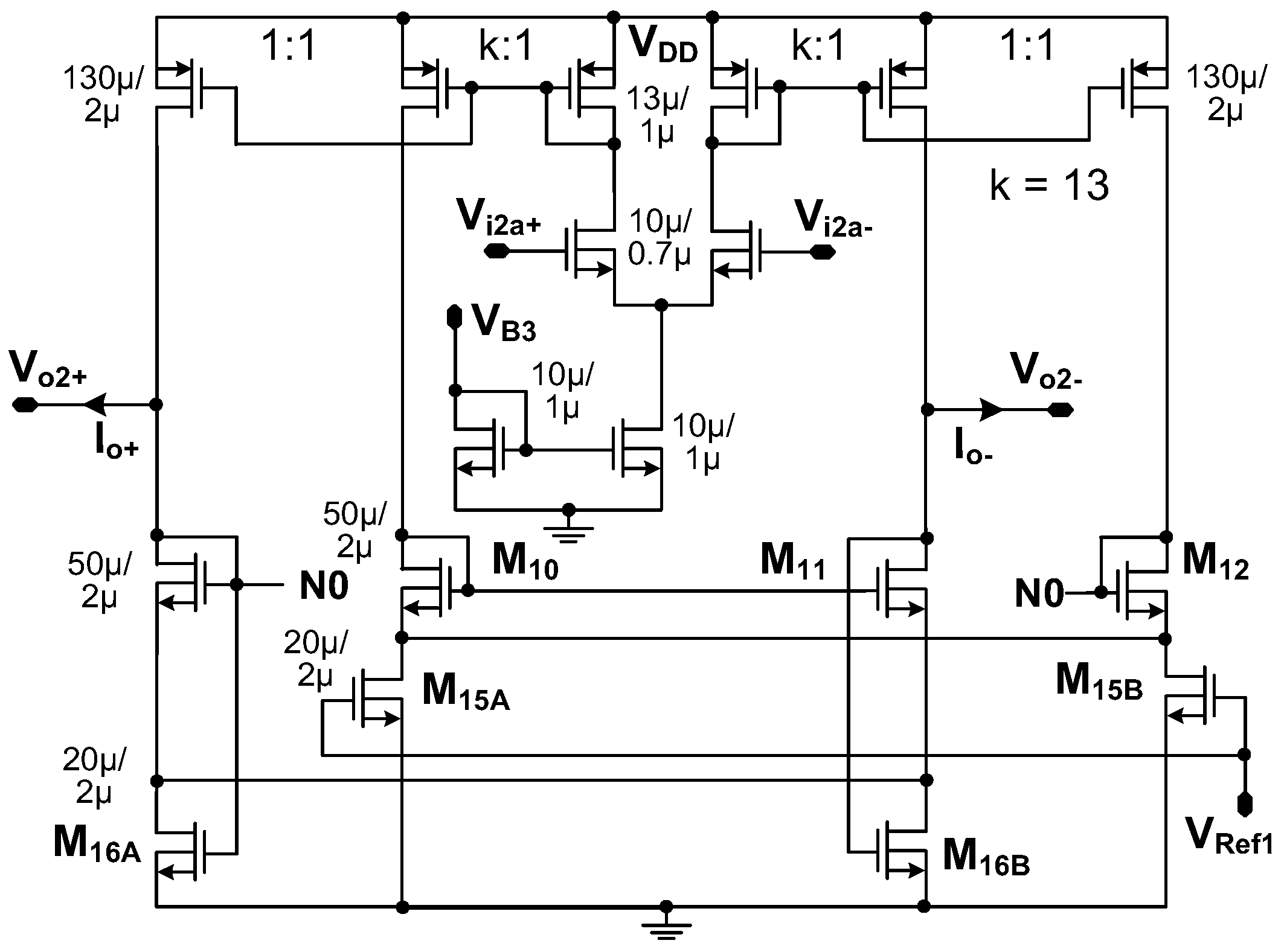
3.1. Current Driver
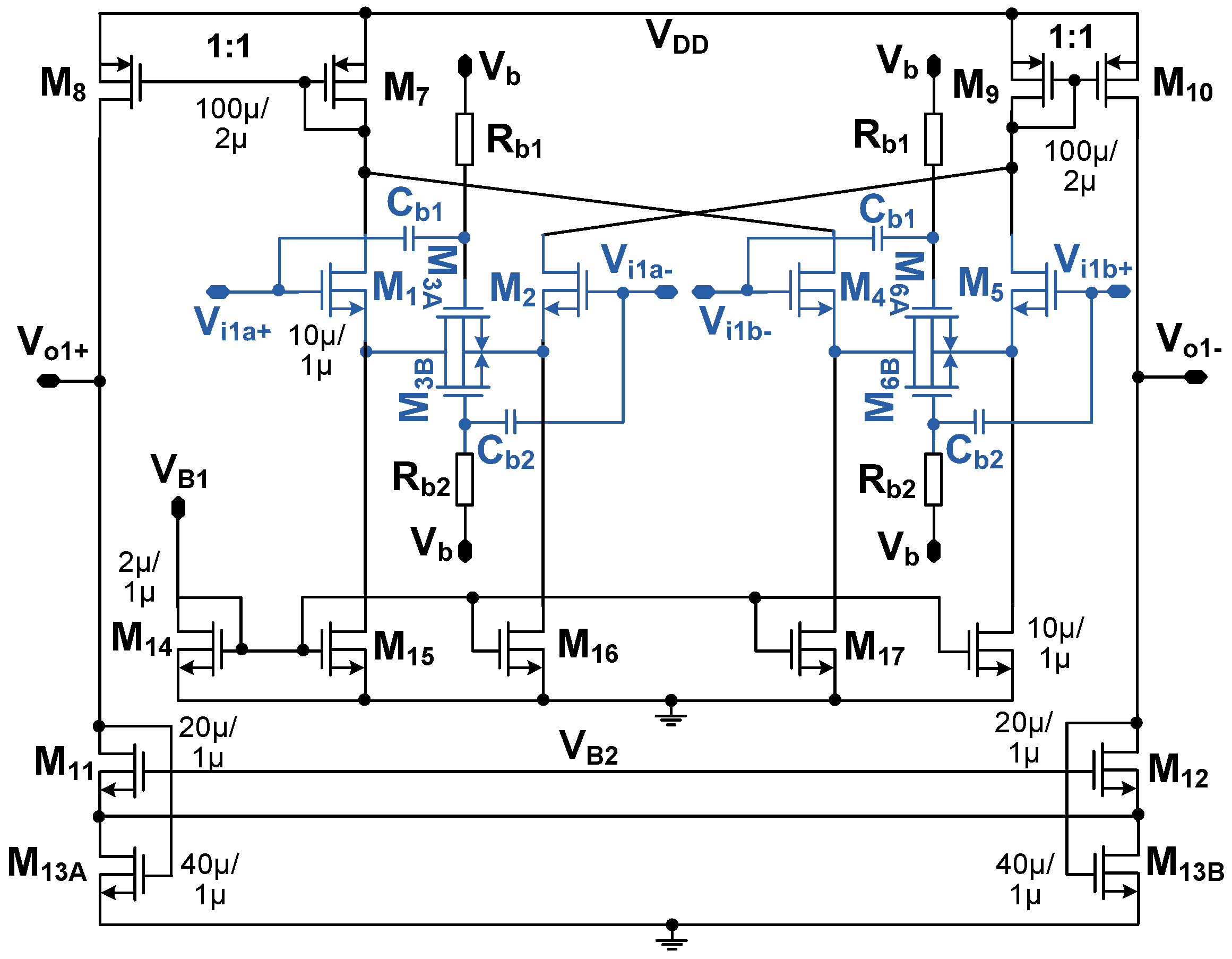
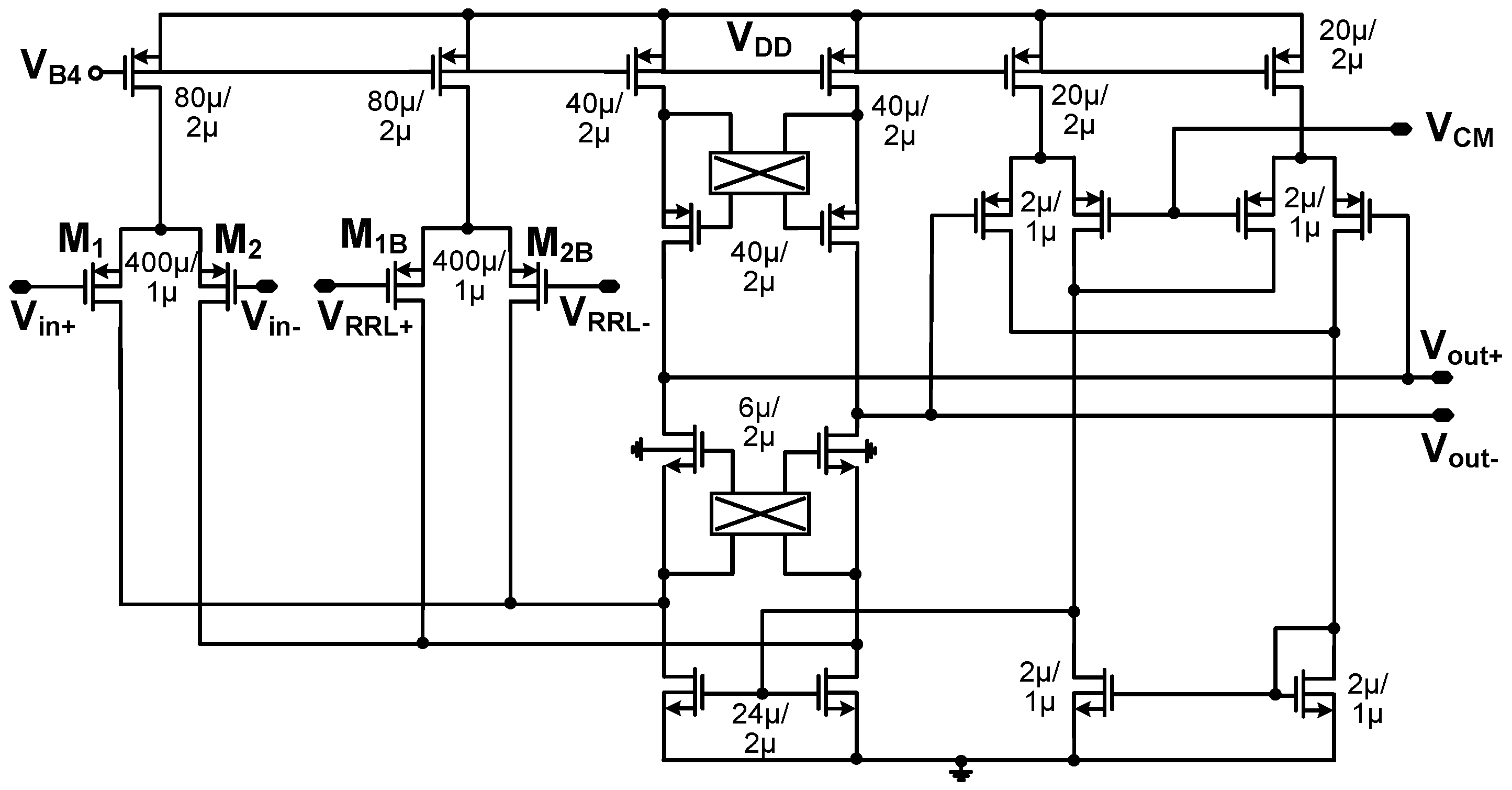
3.2. Instrumentation Amplifier
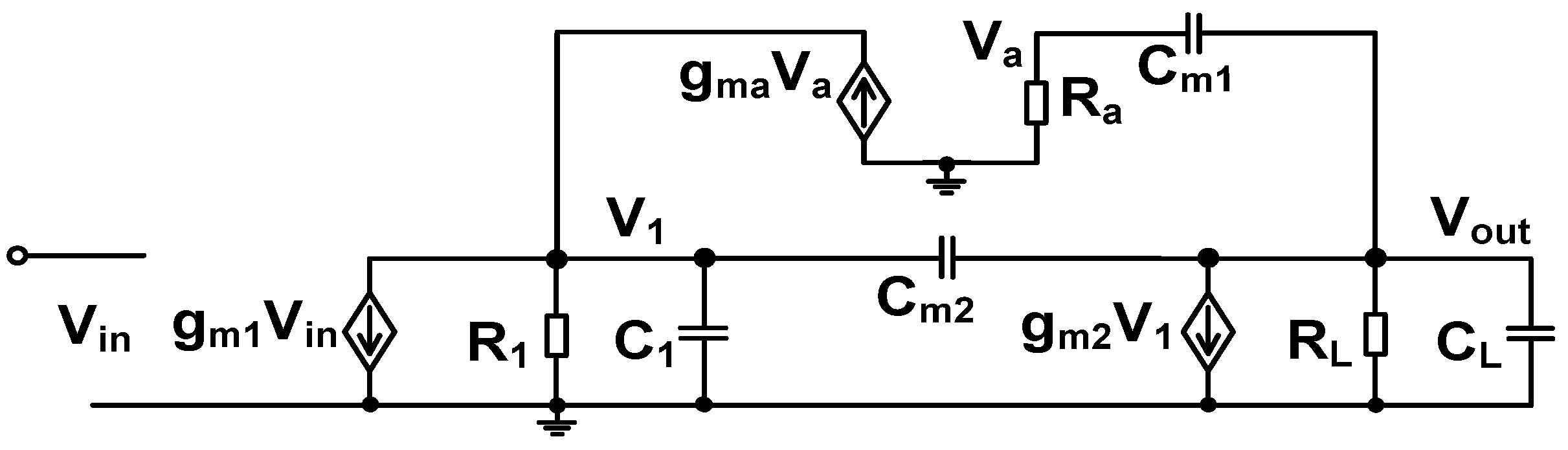
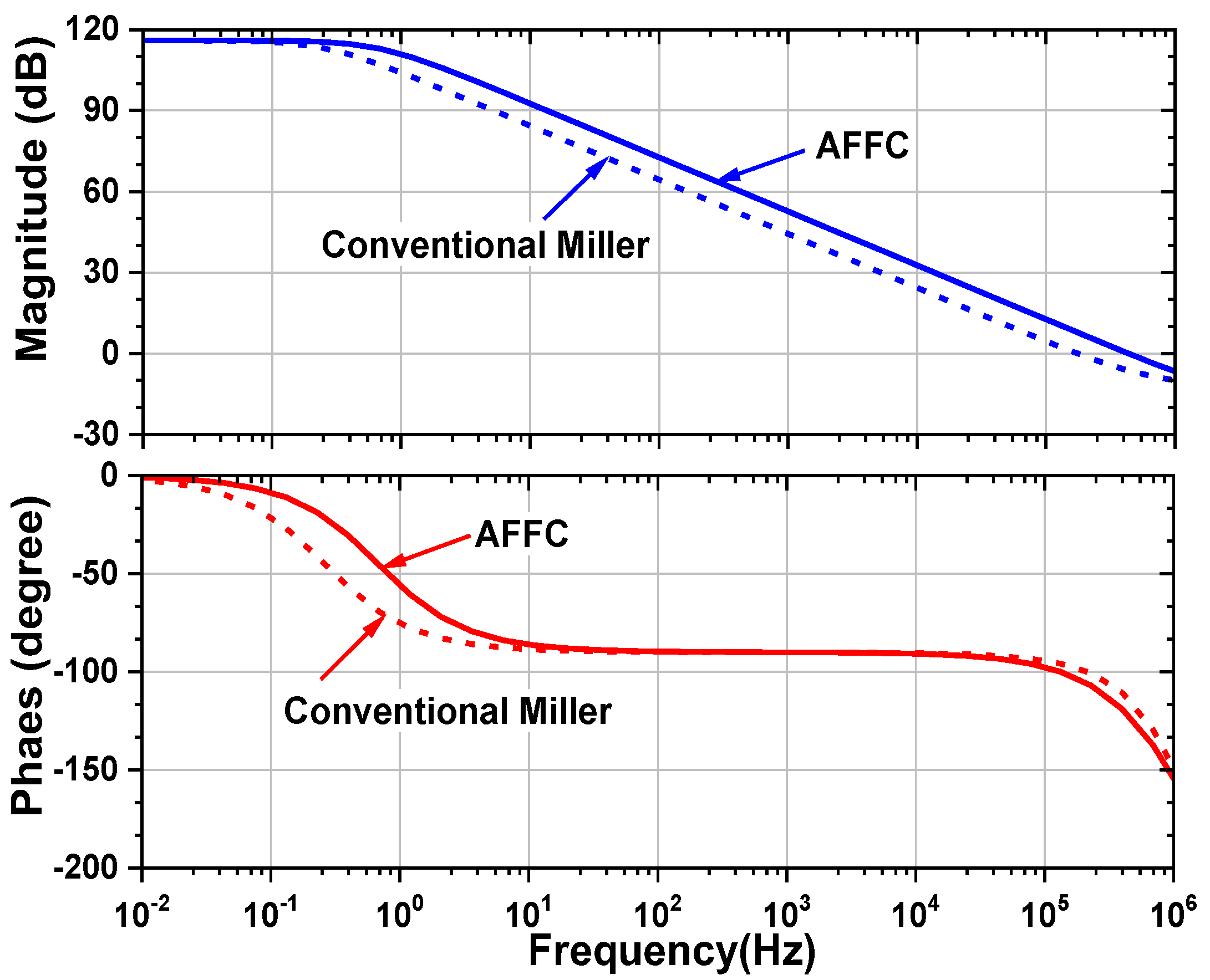
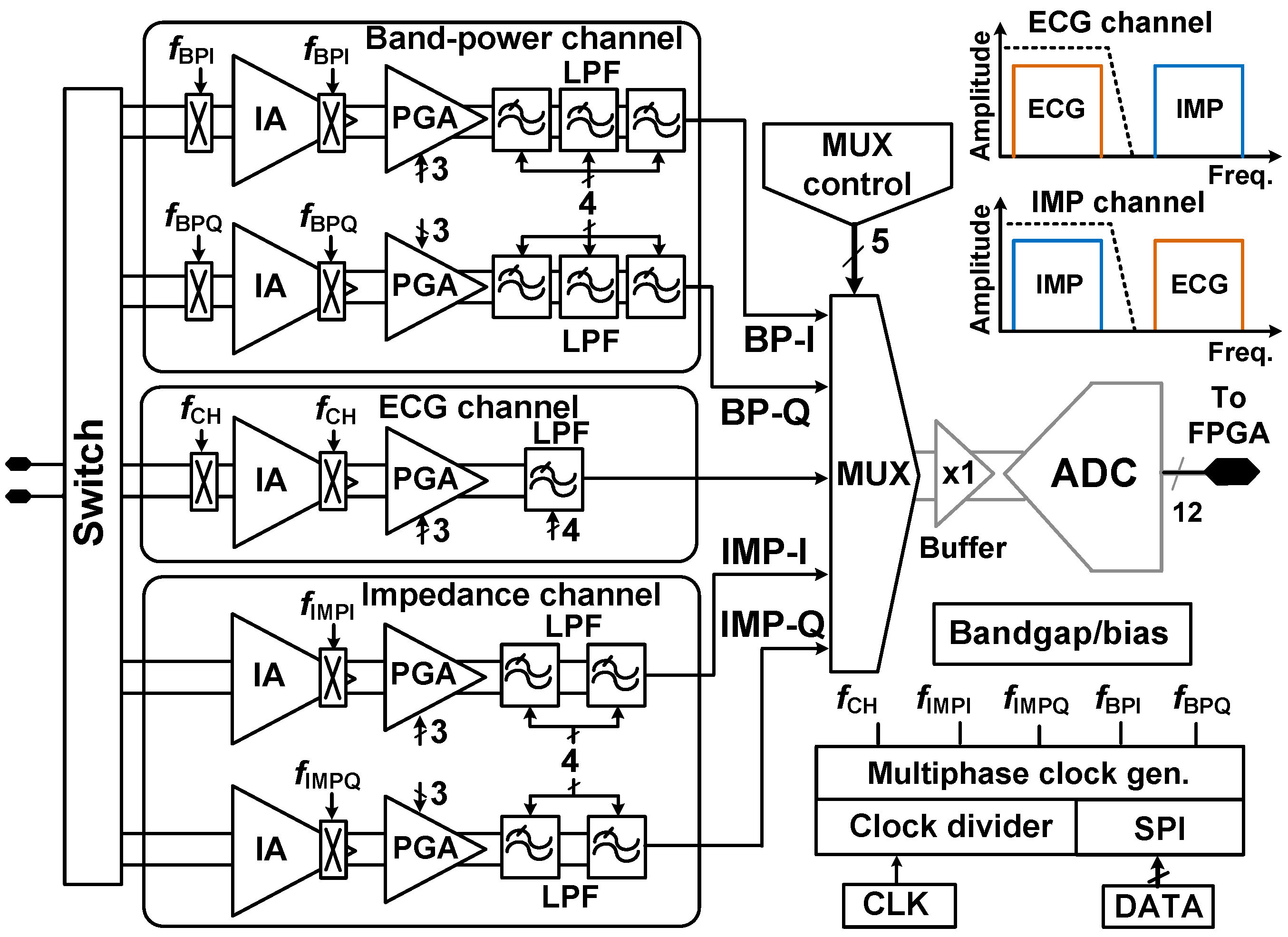
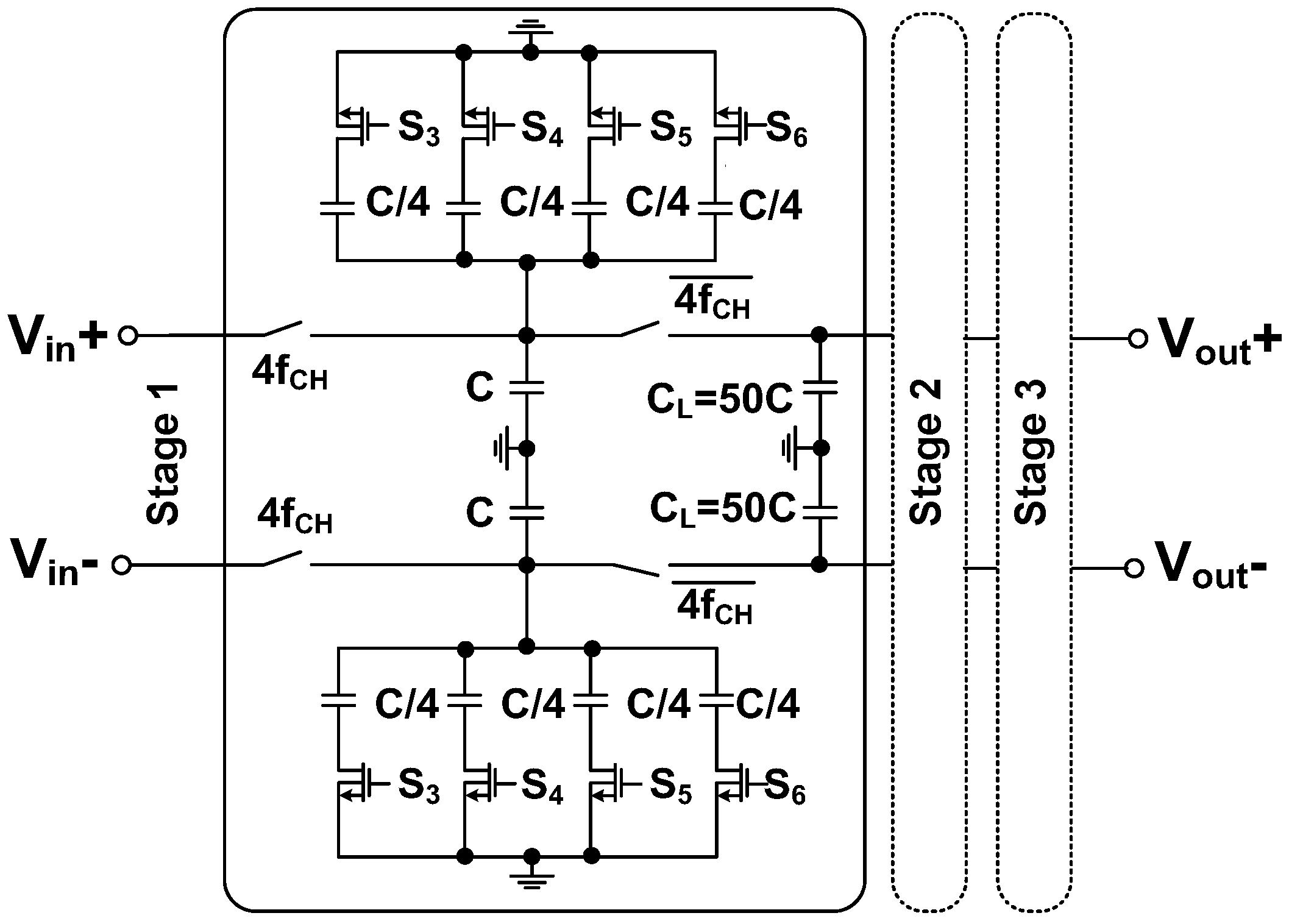
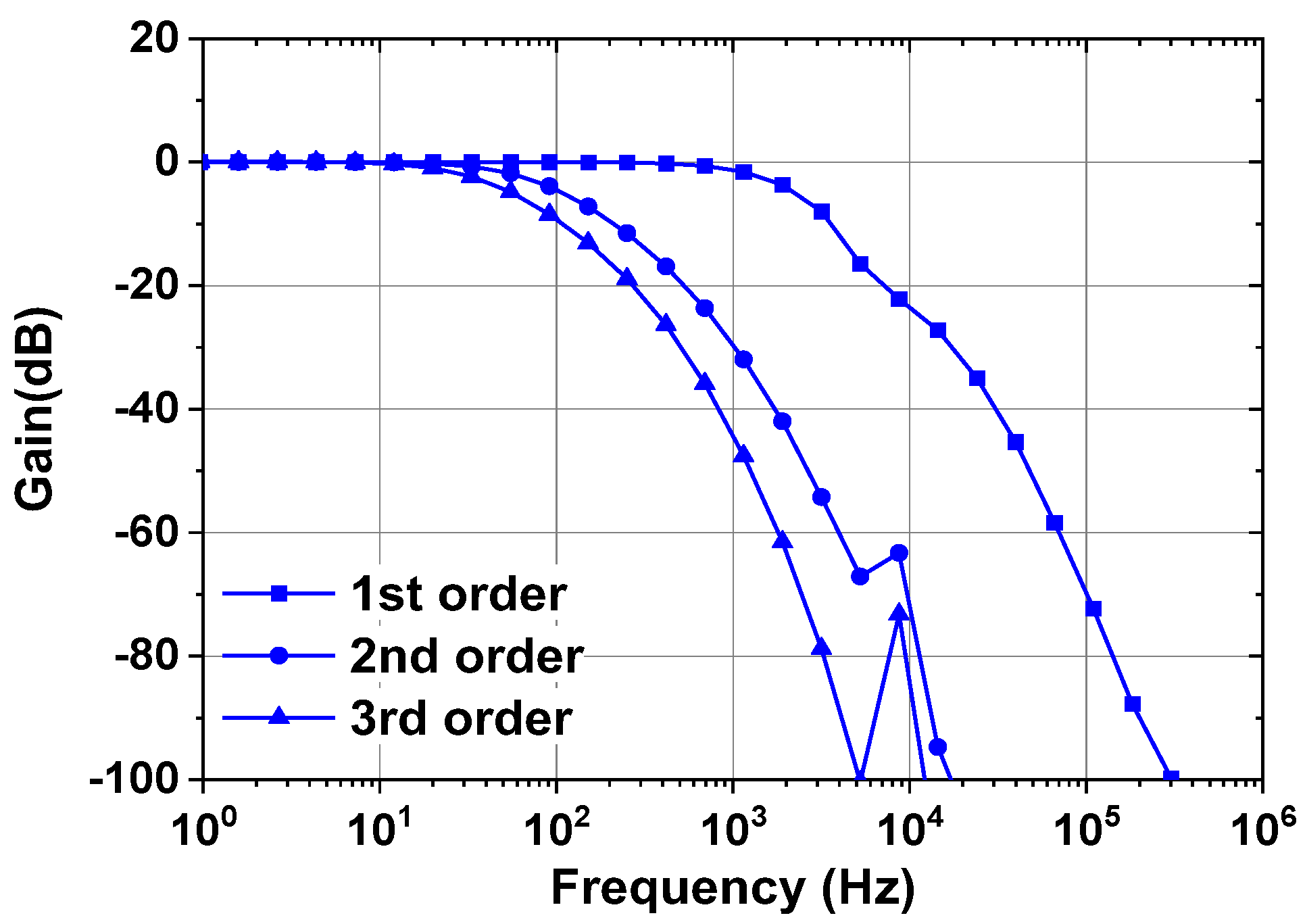
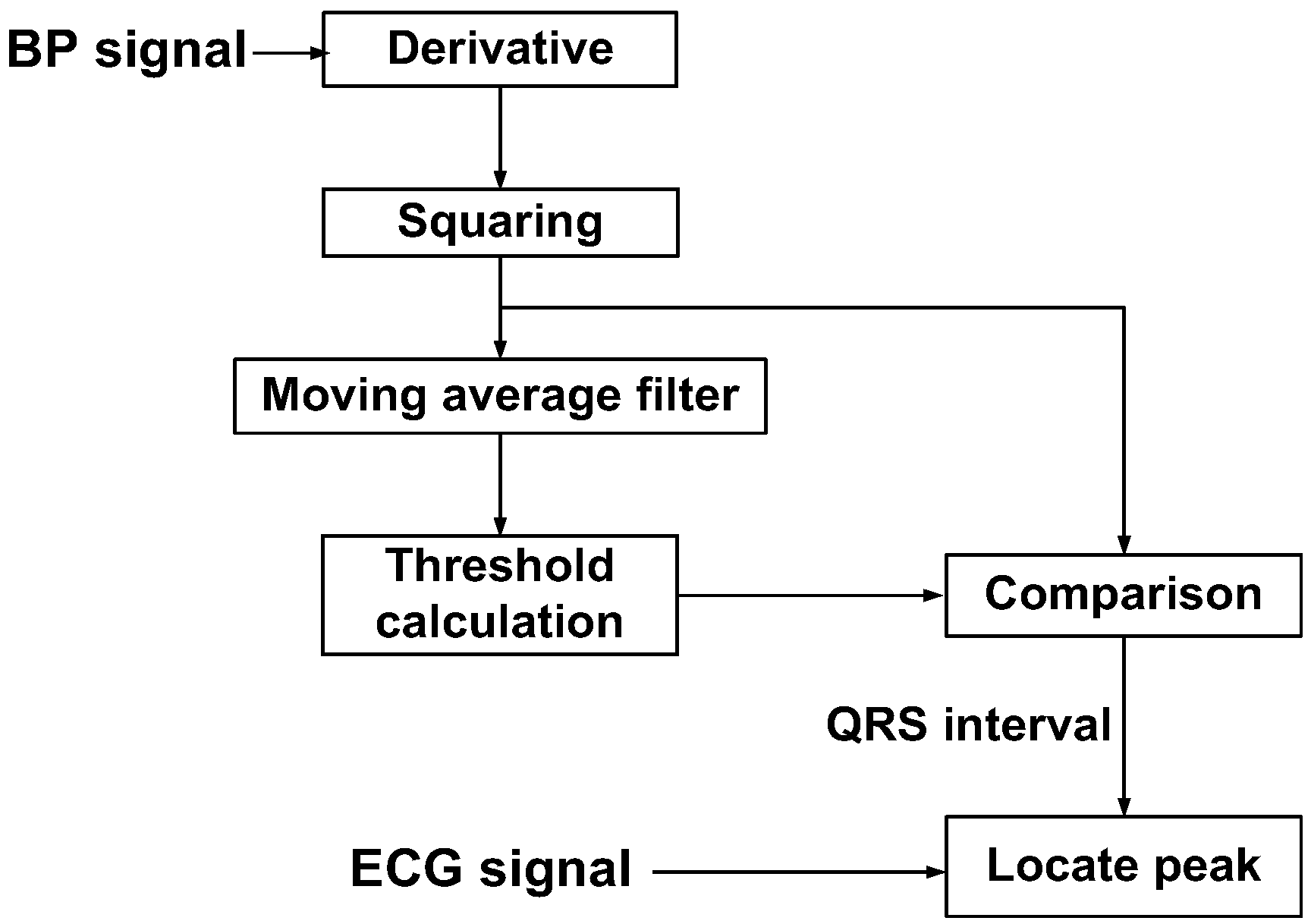
4. Back-End Signal Processing IC
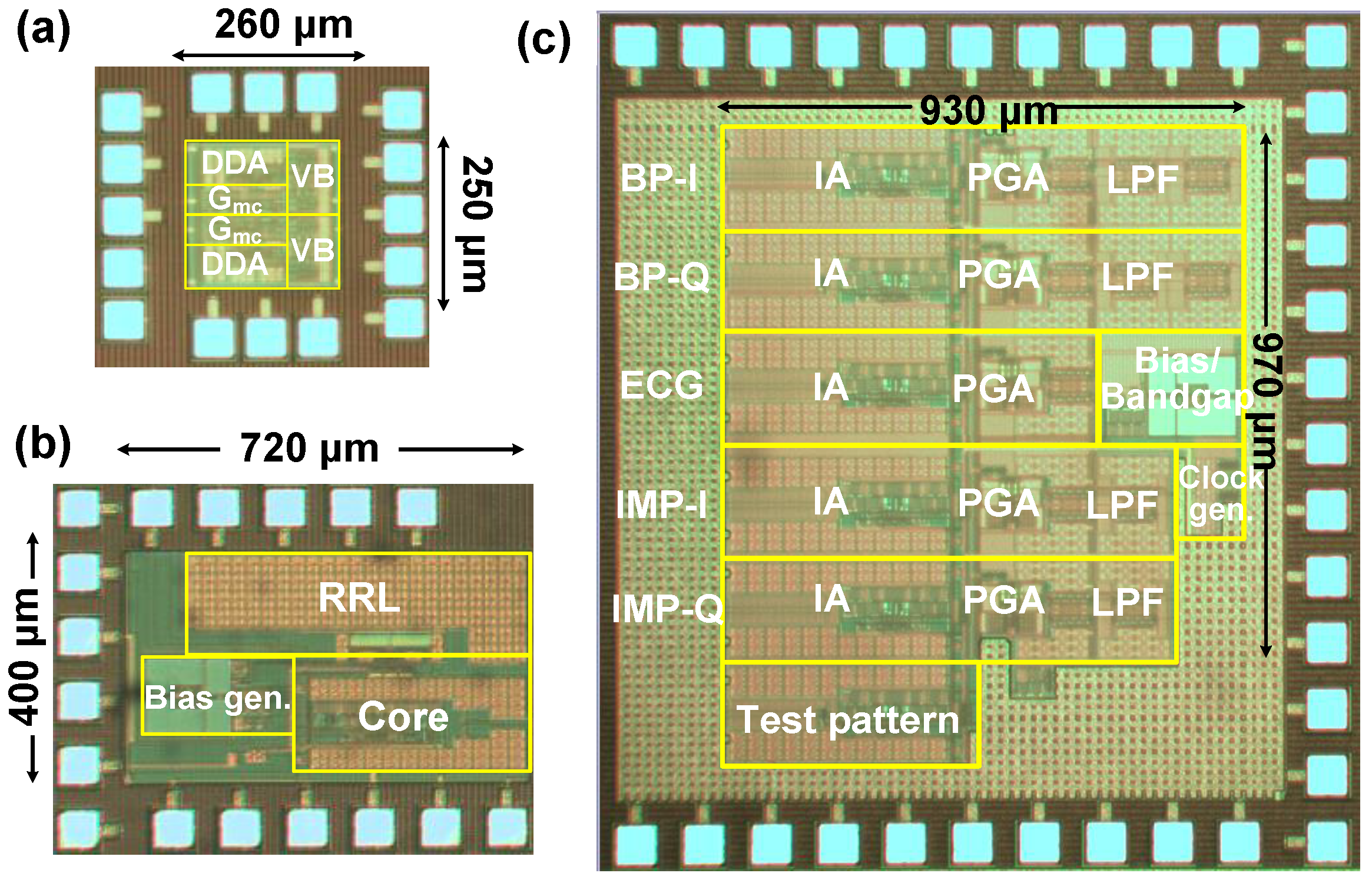
5. Measured Results
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Acknowledgments
Conflicts of Interest
Appendix A
References
- Halter, R.J.; Hartov, A.; Heaney, J.A.; Paulsen, K.D.; Schned, A.R. Electrical impedance spectroscopy of the human prostate. IEEE Trans. Biomed. Eng. 2007, 54, 1321–1327. [Google Scholar] [CrossRef] [PubMed]
- Moganti, G.L.K.; Siva Praneeth, V.N.; Vanjari, S.R.K. A hybrid bipolar active charge balancing technique with adaptive electrode tissue interface (ETI) impedance variations for facial paralysis patients. Sensors 2022, 22, 1756. [Google Scholar] [CrossRef] [PubMed]
- Marcôndes, D.W.C.; Paterno, A.S.; Bertemes-Filho, P. Parasitic effects on electrical bioimpedance systems: Critical review. Sensors 2022, 22, 8705. [Google Scholar] [CrossRef]
- Chi, Y.M.; Jung, T.P.; Cauwenberghs, G. Dry-contact and noncontact biopotential electrodes: Methodological review. IEEE Rev. Biomed. Eng. 2010, 3, 106–119. [Google Scholar] [CrossRef]
- Matthews, R.; Turner, P.J.; McDonald, N.J.; Ermolaev, K.; Mc Manus, T.; Shelby, R.A.; Steindorf, M. Real time workload classification from an ambulatory wireless EEG system using hybrid EEG electrodes. In Proceedings of the 2008 30th Annual International Conference of the IEEE Engineering in Medicine and Biology Society, Vancouver, BC, Canada, 20–25 August 2008; pp. 5871–5875. [Google Scholar]
- Dozio, R.; Baba, A.; Assambo, C.; Burke, M.J. Time based measurement of the impedance of the skin-electrode interface for dry electrode ECG recording. In Proceedings of the 2007 29th Annual International Conference of the IEEE Engineering in Medicine and Biology Society, Lyon, France, 22–26 August 2007; pp. 5001–5004. [Google Scholar]
- Nishimura, S.; Tomita, Y.; Horiuchi, T. Clinical application of an active electrode using an operational amplifier. IEEE Trans. Biomed. Eng. 1992, 39, 1096–1099. [Google Scholar] [CrossRef]
- Xu, J.; Mitra, S.; Matsumoto, A.; Patki, S.; van Hoof, C.; Makinwa, K.A.A.; Yazicioglu, R. A wearable 8-channel active-electrode EEG/ETI acquisition system for body area networks. IEEE J. Solid-State Circuits 2014, 49, 2005–2016. [Google Scholar] [CrossRef]
- Nuwer, M.R.; Comi, G.; Emerson, R.; Fuglsang-Frederiksen, A.; Guérit, J.-M.; Hinrichs, H.; Ikeda, A.; Luccas, J.C.; Rappelsburger, P. IFCN standards for digital recording of clinical EEG. Electroencephalogr. Clin. Neurophysiol. 1998, 106, 259–261. [Google Scholar] [CrossRef]
- Jochum, T.; Denison, T.; Wolf, P. Integrated circuit amplifiers for multi-electrode intracortical recording. J. Neural Eng. 2009, 6, 012001. [Google Scholar] [CrossRef]
- Constantinou, L.; Bayford, R.; Demosthenous, A. A wideband low-distortion CMOS current driver for tissue impedance analysis. IEEE Trans. Circuits Syst. II Express Briefs 2015, 62, 154–158. [Google Scholar] [CrossRef]
- Franco, S. Design with Operational Amplifiers and Analog Integrated Circuits; McGraw-Hill: New York, NY, USA, 2003. [Google Scholar]
- Zhang, F.; Teng, Z.; Zhong, H.; Bertemes-Filho, P.; Wilson, A.J. A comparison of modified Howland circuits as current generators with current mirror type circuits. Physiol. Meas. 2000, 21, 1–6. [Google Scholar]
- Avestruz, A.T.; Santa, W.; Carlson, D.; Jensen, R.; Stanslaski, S.; Helfenstine, A.; Denison, T. A 5 μw/channel spectral analysis IC for chronic bidirectional brain-machine interfaces. IEEE J. Solid-State Circuits 2008, 43, 3006–3024. [Google Scholar] [CrossRef]
- Lee, H.S.; Nguyen, V.N.; Pham, X.L.; Lee, J.W.; Park, H.K. A 250-μW, 18-nV/rtHz current-feedback chopper instrumentation amplifier in 180-nm CMOS for high-performance bio-potential sensing applications. Analog Int. Circuits Signal Process. 2017, 90, 137–148. [Google Scholar] [CrossRef]
- Schreier, R.; Silva, J.; Steensgaard, J.; Temes, G.C. Design-oriented estimation of thermal noise in switched-capacitor circuits. IEEE Trans. Circuits Syst. I Regul. Pap. 2005, 52, 2358–2368. [Google Scholar] [CrossRef]
- Cheng, Q.; Zhang, H.; Xue, L.; Guo, J. A 1.2-V 43.2-μW three-stage amplifier with cascode miller-compensation and Q-reduction for driving large capacitive load. In Proceedings of the 2016 IEEE International Symposium on Circuits and Systems, Montréal, QC, Canada, 22–25 July 2016; pp. 458–461. [Google Scholar]
- Guo, S.; Lee, H. Dual active-capacitive-feedback compensation for low-power large-capacitive-load three-stage amplifiers. IEEE J. Solid-State Circuits 2011, 46, 452–464. [Google Scholar] [CrossRef]
- Lee, H.; Mok, P.K.T. Active-feedback frequency-compensation technique for low-power multistage amplifiers. IEEE J. Solid-State Circuits 2003, 38, 511–520. [Google Scholar] [CrossRef]
- Vo, D.H.T.; Lee, J.W. Analysis and design of a low power regulator for a fully integrated HF-band passive RFID tag IC. Analog Integr. Circuits Signal Process. 2012, 71, 69–80. [Google Scholar] [CrossRef]
- Pham, X.T.; Duong, D.N.; Nguyen, N.T.; van Truong, N.; Lee, J.W. A 4.5 GΩ-input impedance chopper amplifier with embedded DC-servo and ripple reduction loops for impedance boosting to sub-Hz. IEEE Trans. Circuits Syst. II Express Briefs 2021, 68, 116–120. [Google Scholar] [CrossRef]
- Nguyen, C.L.; Phan, H.N.; Lee, J.W. A 12-b subranging SAR ADC using detect-and-skip switching and mismatch calibration for biopotential sensing applications. Sensors 2022, 22, 3600. [Google Scholar] [CrossRef] [PubMed]
- Razavi, B. Design of Analog CMOS Integrated Circuits; McGraw-Hill: New York, NY, USA, 2001. [Google Scholar]
- Constantinou, L.; Triantis, I.F.; Bayford, R.; Demosthenous, A. High-power CMOS current driver with accurate transconductance for electrical impedance tomography. IEEE Trans. Biomed. Circuits Syst. 2014, 8, 575–583. [Google Scholar] [CrossRef] [PubMed]
- Pan, J.; Tompkins, W.J. A real-time QRS detection algorithm. IEEE Trans. Biomed. Eng. 1985, BME-32, 230–236. [Google Scholar] [CrossRef]
- Wu, Y.; Jiang, D.; Liu, X.; Bayford, R.; Demosthenous, A. A human-machine interface using electrical impedance tomography for hand prosthesis control. IEEE Trans. Biomed. Circuits Syst. 2018, 12, 1322–1333. [Google Scholar] [CrossRef] [PubMed]
- Rao, A.J.; Murphy, E.K.; Shahghasemi, M.; Odame, K.M. Current-conveyor-based wideband current driver for electrical impedance tomography. Physiol. Meas. 2019, 40, 034005. [Google Scholar] [CrossRef]
- Jang, J.; Kim, M.; Bae, J.; Yoo, H.J. A 2.79-mW 0.5%-THD CMOS current driver IC for portable electrical impedance tomography system. In Proceedings of the 2017 IEEE Asian Solid-State Circuits Conference (A-SSCC), Seoul, South Korea, 6–8 November 2017; pp. 145–148. [Google Scholar]
- Ha, H.; Sijbers, W.; Wegberg, R.V.; Xu, J.; Konijnenburg, M.; Vis, P.; Breeschoten, A.; Song, S.; Hoof, C.V.; van Helleputte, N.A. A bio-impedance readout IC with digital-assisted baseline cancellation for two-electrode measurement. IEEE J. Solid-State Circuits 2019, 54, 2969–2979. [Google Scholar] [CrossRef]
- Hong, S.; Lee, K.; Ha, U.; Kim, H.; Lee, Y.; Kim, Y.; Yoo, H.J. A 4.9 MΩ-sensitivity mobile electrical impedance tomography IC for early breast-cancer detection system. IEEE J. Solid-State Circuits 2015, 50, 245–257. [Google Scholar] [CrossRef]
- Pan, Q.; Qu, T.; Tang, B.; Shan, F.; Hong, Z.; Xu, J. A 0.5-mΩ/rtHz dry-electrode bioimpedance interface with current mismatch cancellation and input impedance of 100 MΩ at 50 kHz. IEEE J. Solid-State Circuits 2022. early access. [Google Scholar] [CrossRef]
- Texas Instruments, AFE4300, Low-Cost, Integrated Analog Frontend for Weight-Scale and Body Composition Measurement. Available online: https://www.ti.com/lit/ds/symlink/afe4300.pdf (accessed on 9 December 2022).







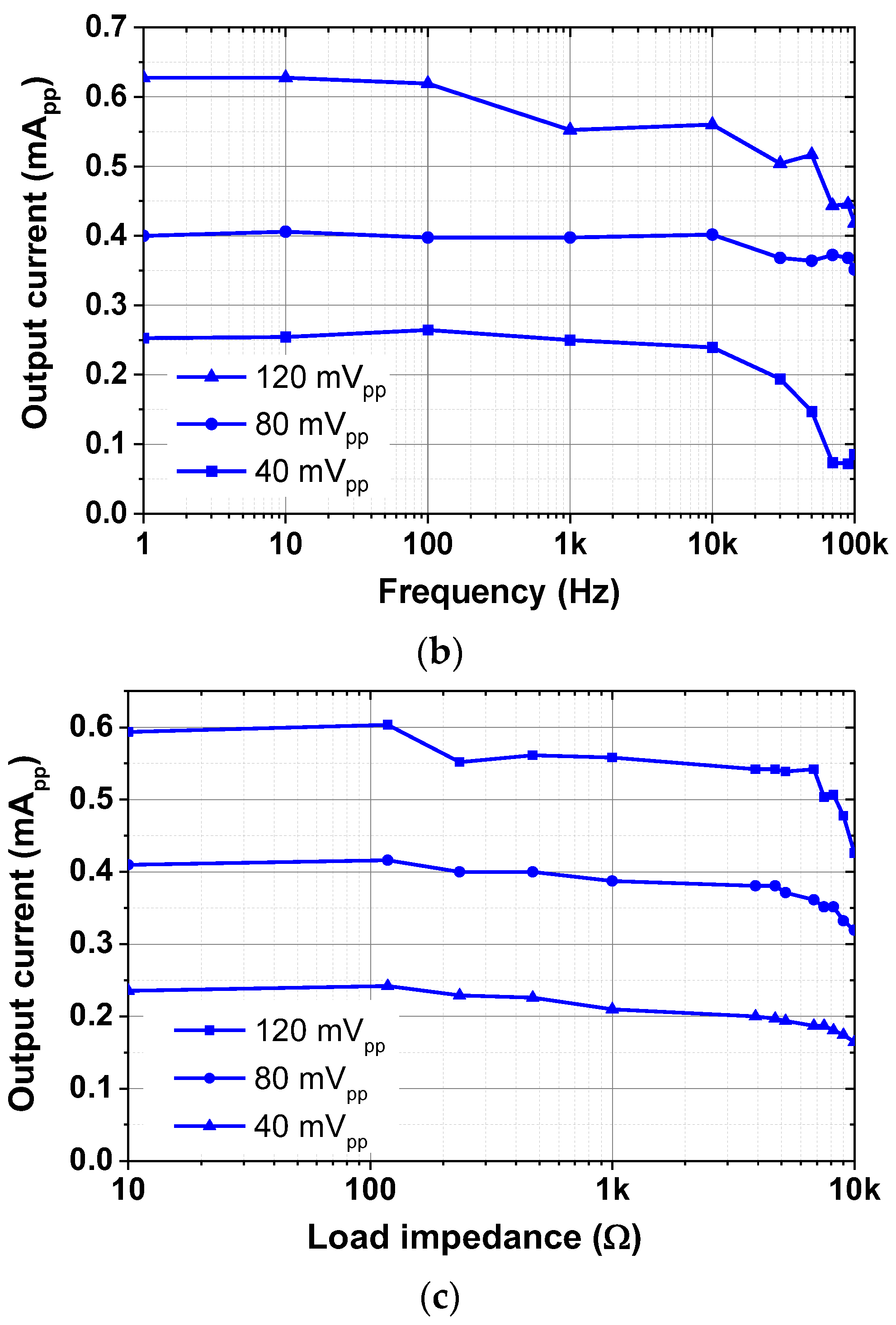
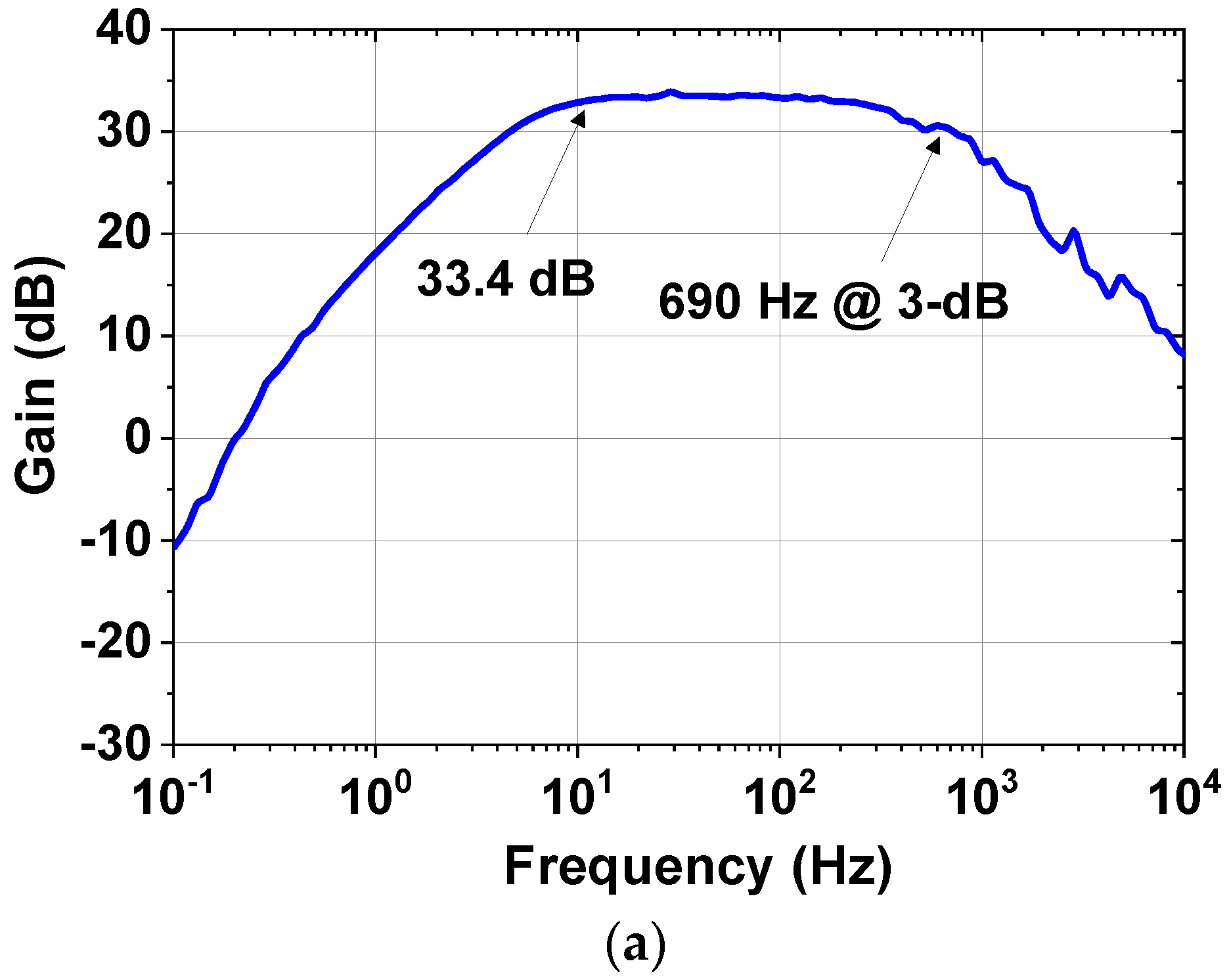
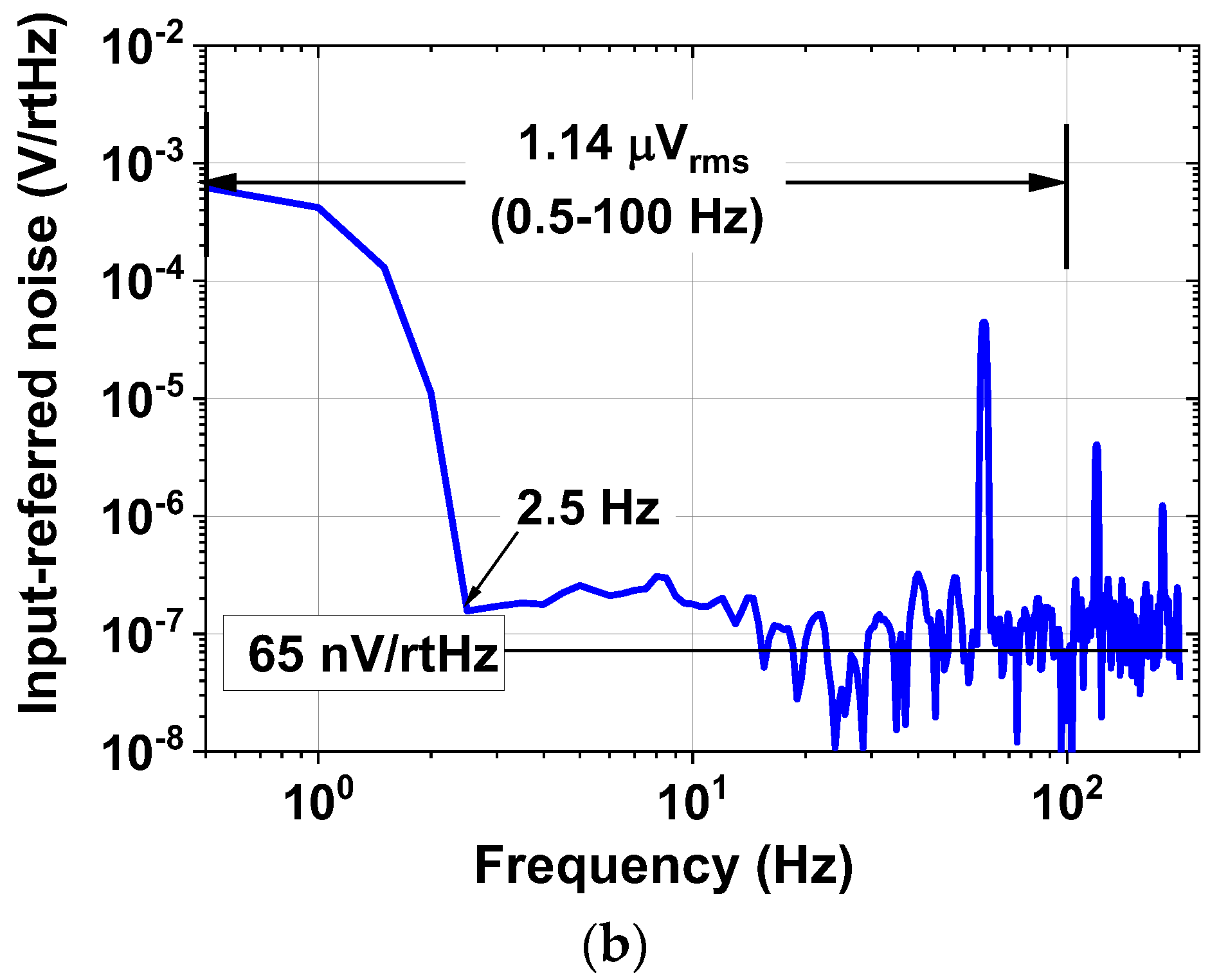

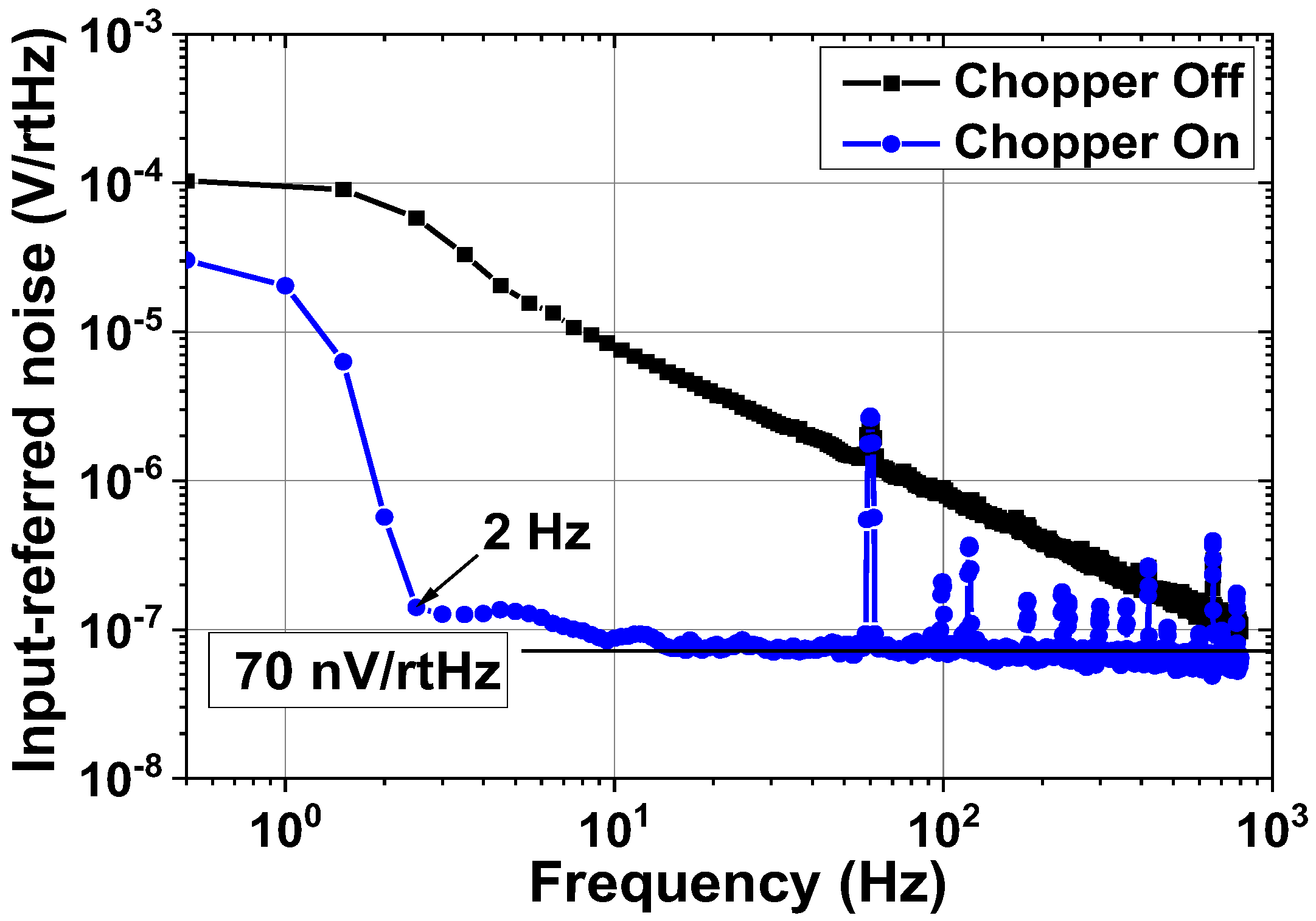

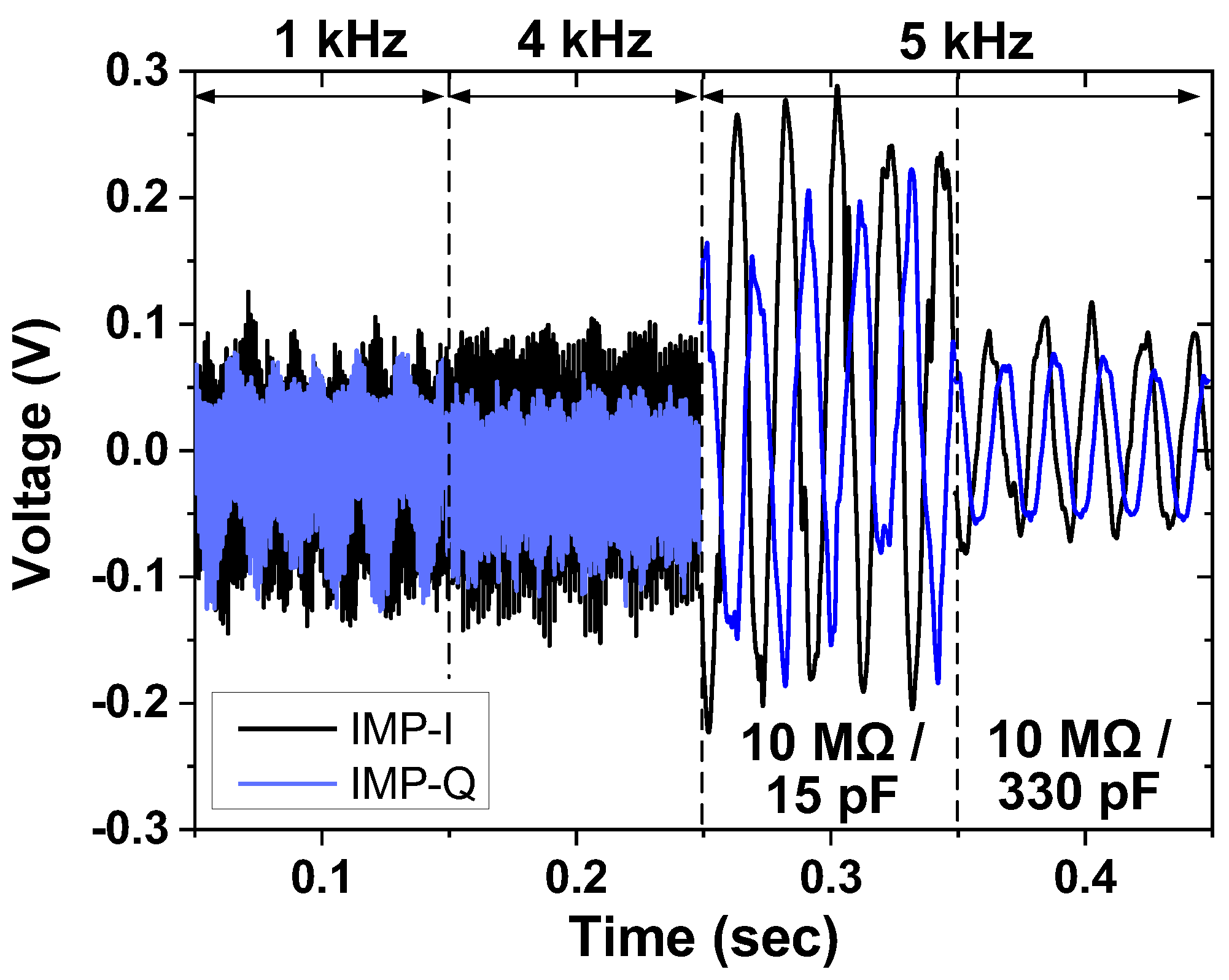
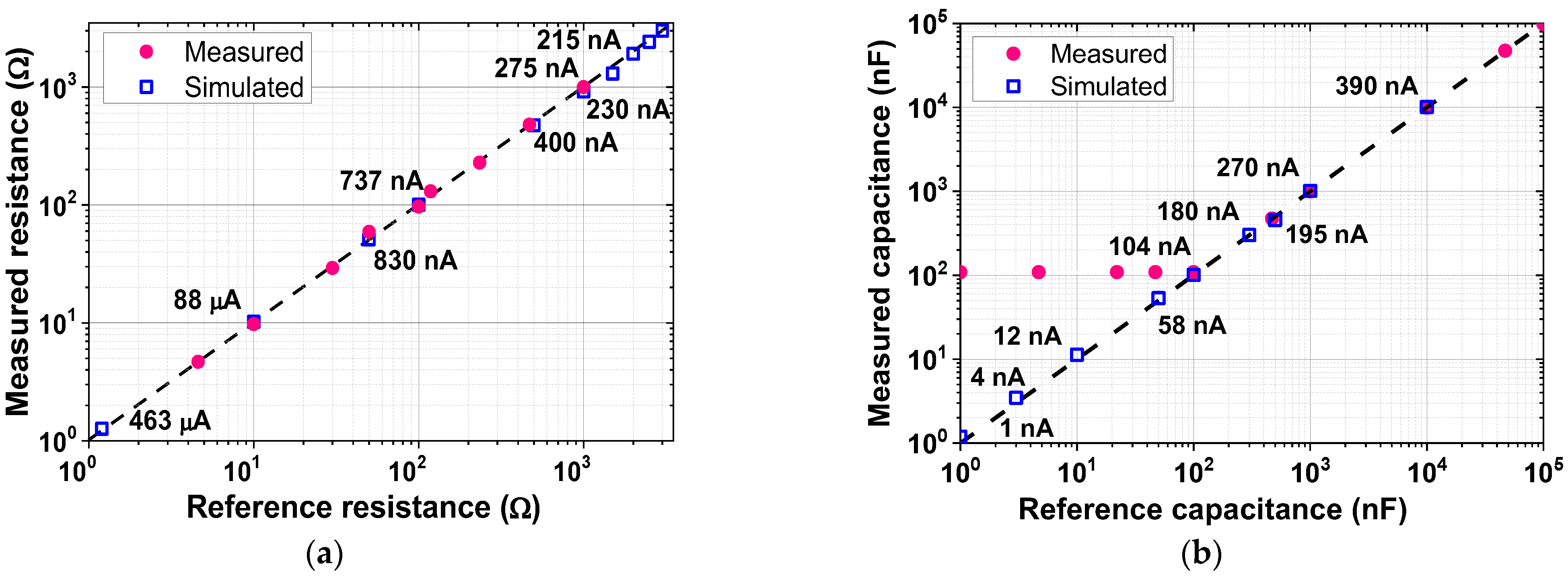
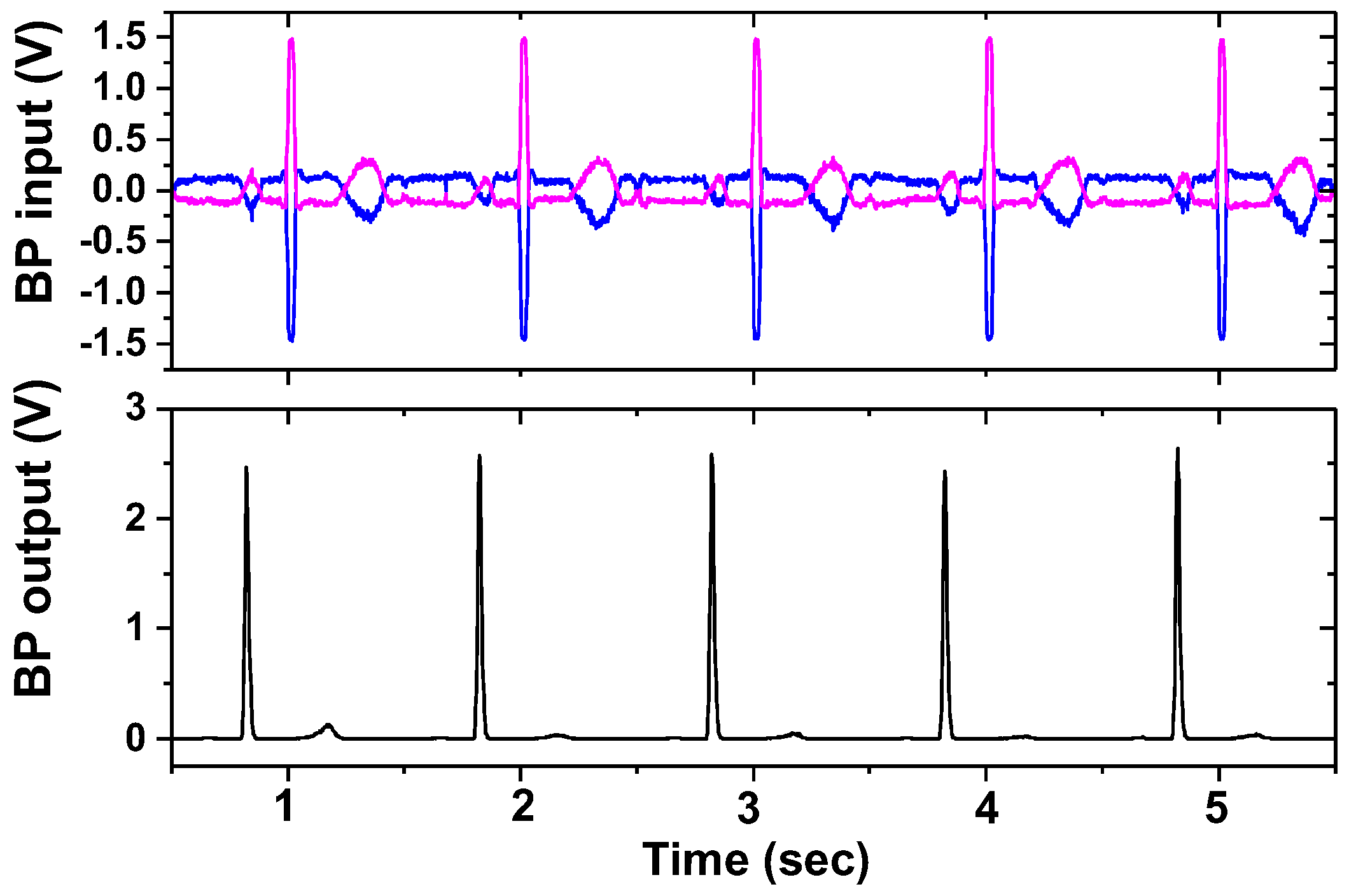
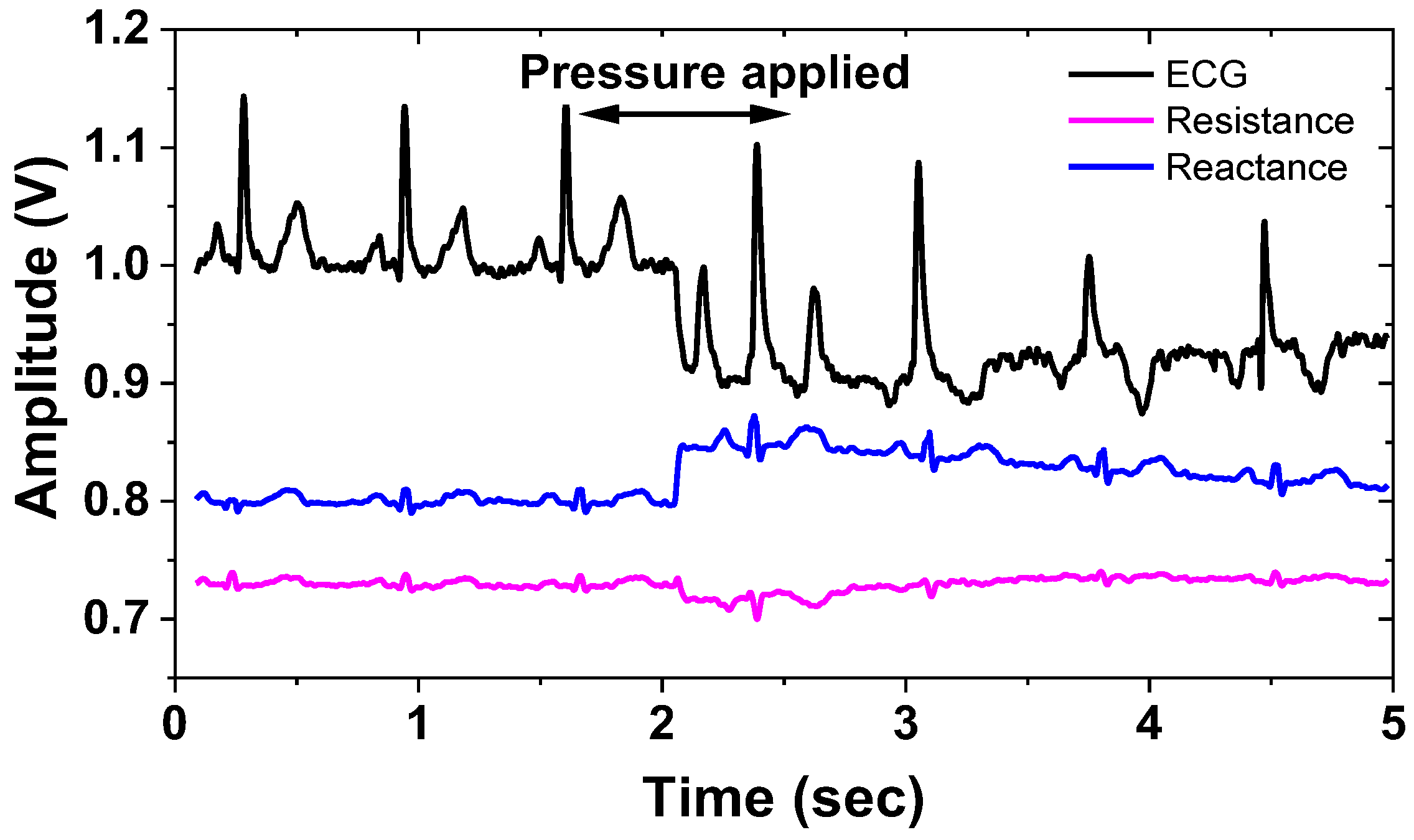

| Current driver | 1.98 mA @ 1.8 V = 3.56 mW |
| Preamplifier | 0.4 μA (core), 0.16 μA (bias) @ 1.8 V = 1 μW |
| Back-end | 3.7 μA @ 1.8 V= 6.6 μW (ECG: 0.5 μA, ETI: 1.1 μA, BP: 1.1 μA) |
| Total | 3.6 mW |
| Process | 0.18 μm CMOS | ||
|---|---|---|---|
| Active electrode | Current driver | Frequency | 1 MHz (max) |
| Amplitude | 600 μApp | ||
| Size | 0.065 mm2 | ||
| Preamplifier | Gain | 39.4 dB (differential) | |
| Input noise density | 65 nV/√Hz | ||
| Integrated noise | 1.14 μVrms (100 Hz) | ||
| Size | 0.29 mm2 | ||
| Back-end | Number of channels | 5 | |
| Gain | 23–43 dB | ||
| Bandwidth | 0.5–1 kHz | ||
| Input noise density | 70 nV/√Hz | ||
| Size | 0.9 mm2 | ||
| [11] | [24] | [26] | [27] | [28] | This Work | |
|---|---|---|---|---|---|---|
| Bandwidth | 1 MHz | 500 kHz | 500 kHz | 10 MHz | 1 MHz | 1 MHz |
| Output impedance | 1 MΩ @ 500 kHz/ 360 kΩ @ 1 MHz | 1 MΩ @ 100 kHz/ 500 kΩ @ 500 kHz | 750 kΩ @ 500 kHz | 101 kΩ @ 1 MHz/ 19.5 kΩ @ 10 MHz | 1 MΩ @1 MHz | 1 MΩ @ 500 kHz/ 300 kΩ @ 1 MHz |
| Max. output current | 1 mApp | 5 mApp | 1 mApp | 1.2 mApp | 400 μApp | >500 μApp |
| THD | <0.1% * @ 1 mApp | 0.69% @ 5 mApp | 0.79% @ 3.97 mApp | 0.14% * @ 1.2 mApp | <0.5% @ 400 μApp | 0.29% ** @ 303 μApp |
| Supply | ±2.5 V | 18 V | ±1.65 V | 3.3 V | 1.2 V | 1.8 V |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nguyen, X.T.; Ali, M.; Lee, J.-W. 3.6 mW Active-Electrode ECG/ETI Sensor System Using Wideband Low-Noise Instrumentation Amplifier and High Impedance Balanced Current Driver. Sensors 2023, 23, 2536. https://doi.org/10.3390/s23052536
Nguyen XT, Ali M, Lee J-W. 3.6 mW Active-Electrode ECG/ETI Sensor System Using Wideband Low-Noise Instrumentation Amplifier and High Impedance Balanced Current Driver. Sensors. 2023; 23(5):2536. https://doi.org/10.3390/s23052536
Chicago/Turabian StyleNguyen, Xuan Tien, Muhammad Ali, and Jong-Wook Lee. 2023. "3.6 mW Active-Electrode ECG/ETI Sensor System Using Wideband Low-Noise Instrumentation Amplifier and High Impedance Balanced Current Driver" Sensors 23, no. 5: 2536. https://doi.org/10.3390/s23052536
APA StyleNguyen, X. T., Ali, M., & Lee, J.-W. (2023). 3.6 mW Active-Electrode ECG/ETI Sensor System Using Wideband Low-Noise Instrumentation Amplifier and High Impedance Balanced Current Driver. Sensors, 23(5), 2536. https://doi.org/10.3390/s23052536








