Improving Automatic Smartwatch Electrocardiogram Diagnosis of Atrial Fibrillation by Identifying Regularity within Irregularity
Abstract
:1. Introduction
2. Material and Methods
2.1. Patient Population
2.2. Data Arrangement
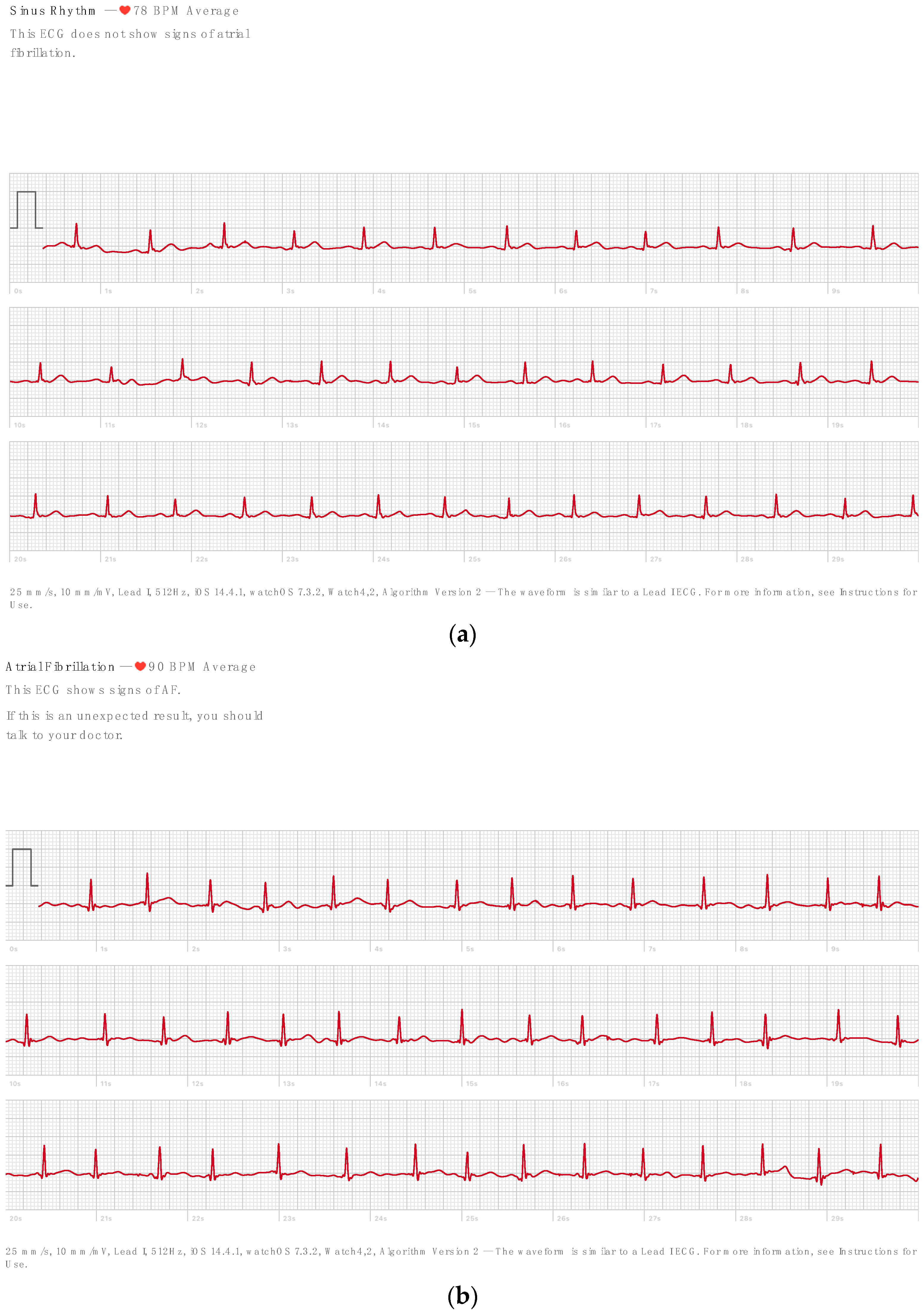
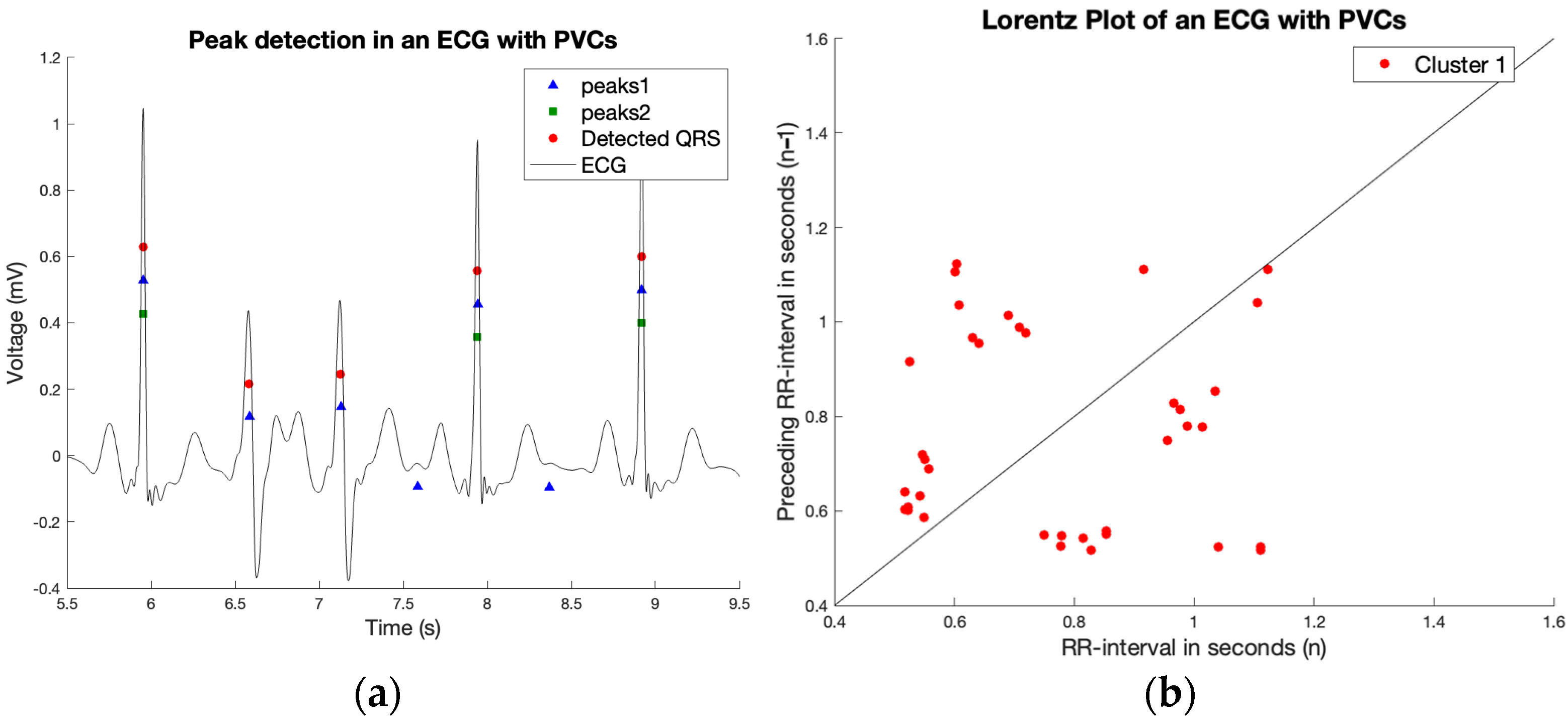
2.3. R Peak Detection
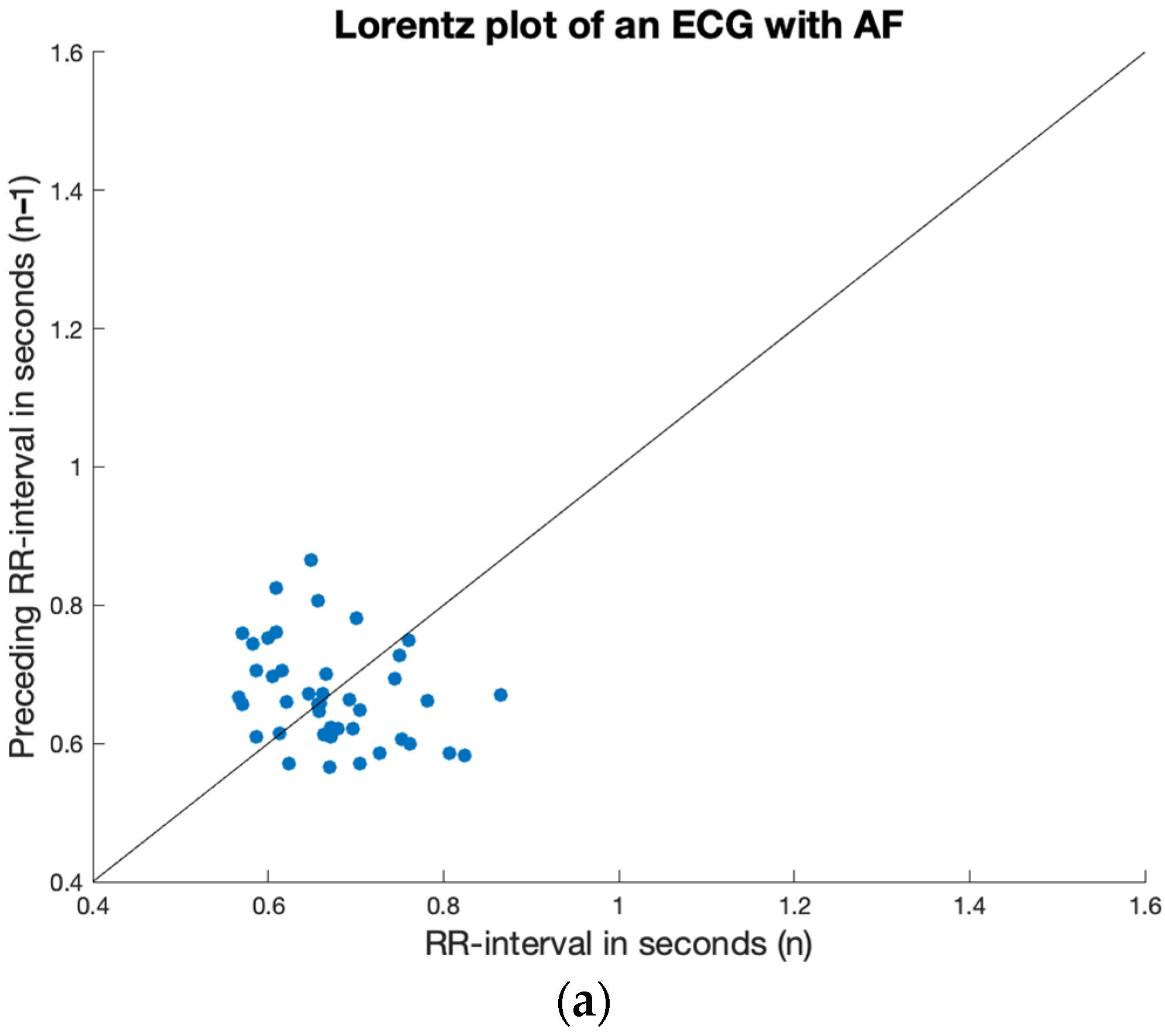
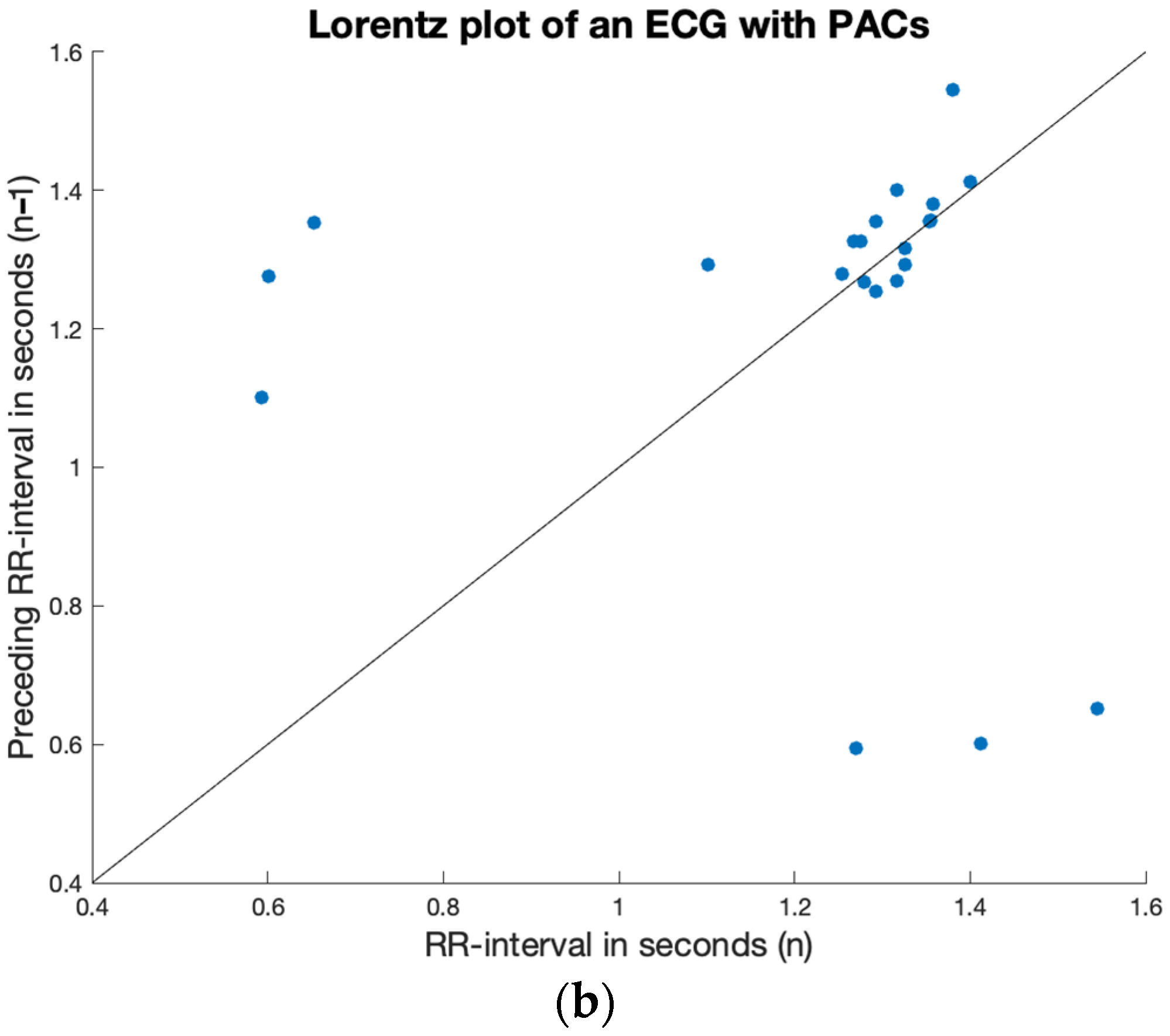
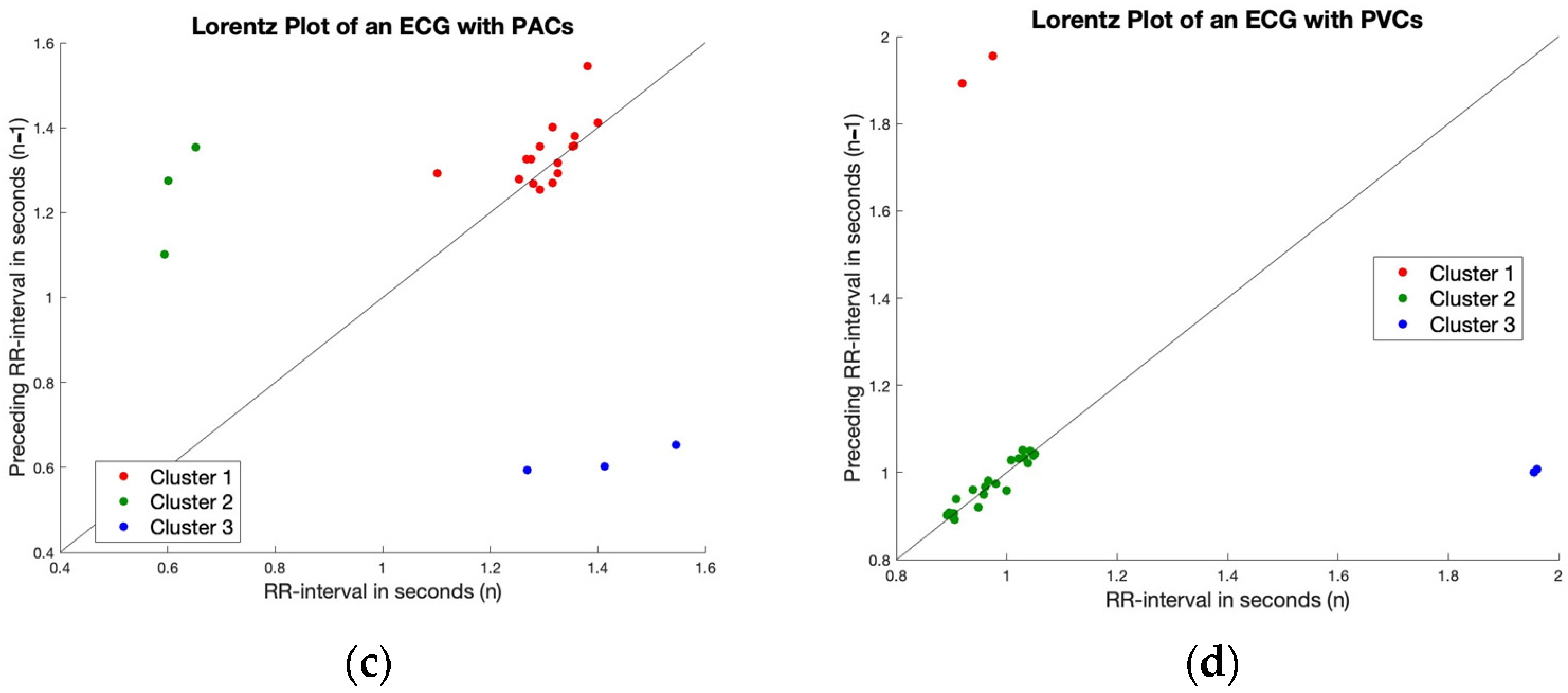
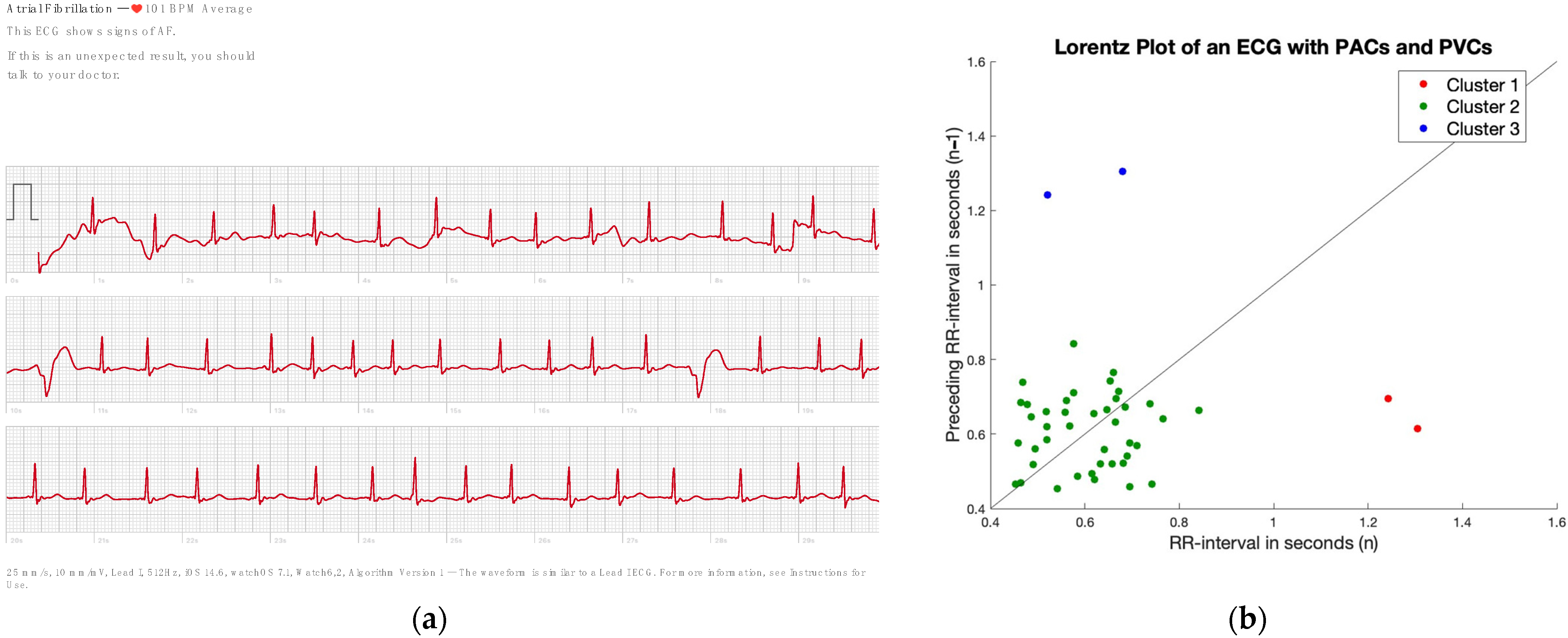
2.4. Irregularity
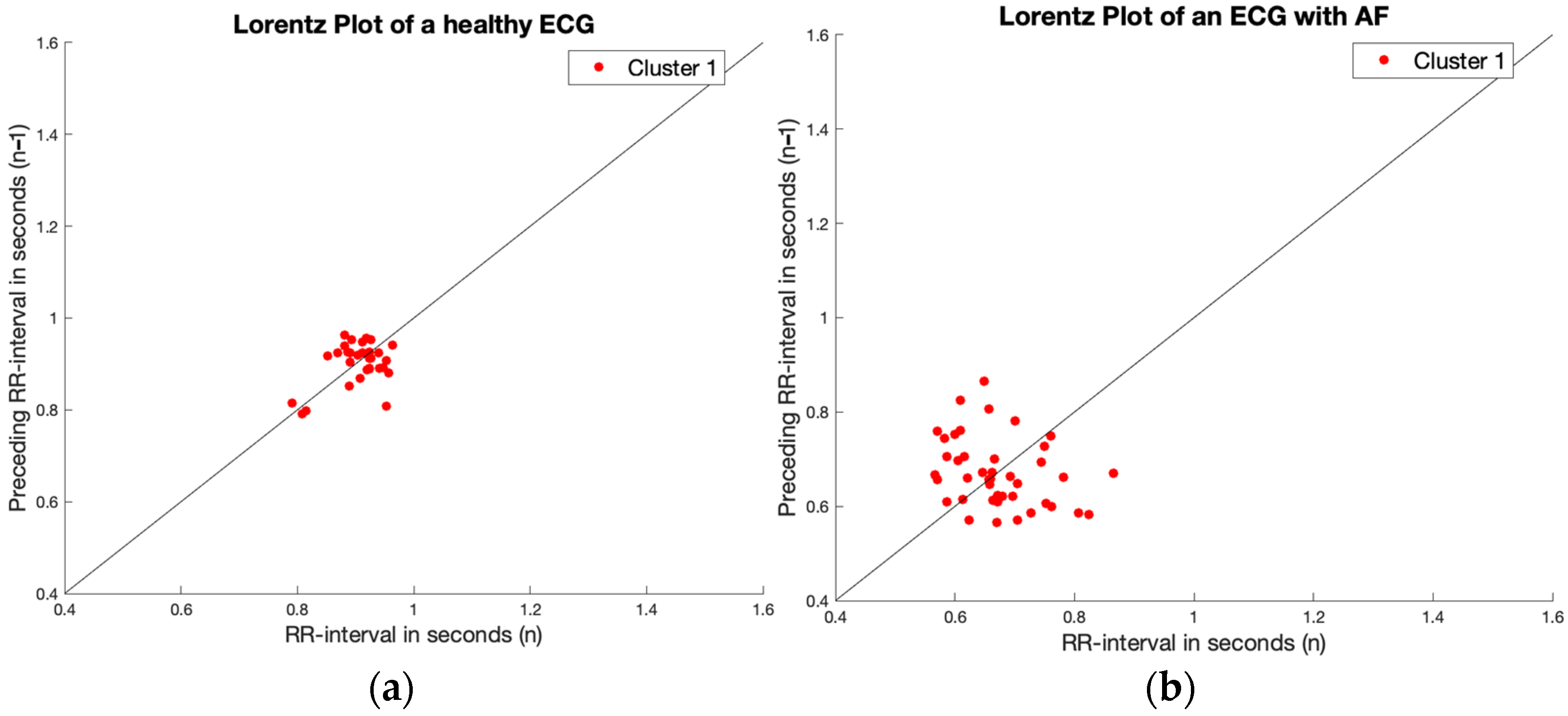
2.5. Finding Regularity in Irregularity
2.6. Validation
3. Results
3.1. Baseline Characteristics
3.2. Irregularity
3.3. Finding Regularity in Irregularity
3.4. Validation
4. Discussion
4.1. Future Approaches
4.2. Study Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Lopez-Perez, A.; Sebastian, R.; Ferrero, J.M. Three-Dimensional Cardiac Computational Modelling: Methods, Features and Applications. Biomed. Eng. Online 2015, 14, 35. [Google Scholar] [CrossRef] [PubMed]
- Petryszyn, P.; Niewinski, P.; Staniak, A.; Piotrowski, P.; Well, A.; Well, M.; Jeskowiak, I.; Lip, G.; Ponikowski, P. Effectiveness of Screening for Atrial Fibrillation and Its Determinants. A Meta-Analysis. PLoS ONE 2019, 14, e0213198. [Google Scholar] [CrossRef] [PubMed]
- Pappano, A.; Wier, W. Cardiovascular Physiology, 11th ed.; Elsevier Health Sciences: Philadelphia, PA, USA, 2018. [Google Scholar]
- Dilaveris, P.E.; Kennedy, H.L. Silent Atrial Fibrillation: Epidemiology, Diagnosis, and Clinical Impact. Clin. Cardiol. 2017, 40, 413–418. [Google Scholar] [CrossRef] [PubMed]
- NHS Atrial Fibrillation—Treatment—NHS. Available online: https://www.nhs.uk/conditions/atrial-fibrillation/treatment/ (accessed on 4 May 2023).
- Raja, J.M.; Elsakr, C.; Roman, S.; Cave, B.; Pour-Ghaz, I.; Nanda, A.; Maturana, M.; Khouzam, R.N. Apple Watch, Wearables, and Heart Rhythm: Where Do We Stand? Ann. Transl. Med. 2019, 7, 417. [Google Scholar] [CrossRef] [PubMed]
- The Verge Why Apple Needed the FDA to Sign off on Its EKG but Not Its Blood Oxygen Monitor—The Verge. Available online: https://www.theverge.com/2020/10/7/21504023/apple-watch-ekg-blood-oxygen-fda-clearance (accessed on 4 May 2023).
- Apple Take an ECG with the ECG App on Apple Watch—Apple Support (IN). Available online: https://support.apple.com/en-in/HT208955 (accessed on 4 May 2023).
- Abu-Alrub, S.; Strik, M.; Ramirez, F.D.; Moussaoui, N.; Racine, H.P.; Marchand, H.; Buliard, S.; Haïssaguerre, M.; Ploux, S.; Bordachar, P. Smartwatch Electrocardiograms for Automated and Manual Diagnosis of Atrial Fibrillation: A Comparative Analysis of Three Models. Front. Cardiovasc. Med. 2022, 9, 836375. [Google Scholar] [CrossRef] [PubMed]
- Racine, H.P.; Strik, M.; van der Zande, J.; Alrub, S.A.; Caillol, T.; Haïssaguerre, M.; Ploux, S.; Bordachar, P. Role of Coexisting ECG Anomalies in the Accuracy of Smartwatch ECG Detection of Atrial Fibrillation. Can. J. Cardiol. 2022, 38, 1709–1712. [Google Scholar] [CrossRef] [PubMed]
- Kirchhof, P.; Benussi, S.; Kotecha, D.; Ahlsson, A.; Atar, D.; Casadei, B.; Castella, M.; Diener, H.-C.; Heidbuchel, H.; Hendriks, J.; et al. 2016 ESC Guidelines for the Management of Atrial Fibrillation Developed in Collaboration with EACTS. Eur. J. Cardiothorac. Surg. 2016, 50, e1–e88. [Google Scholar] [CrossRef] [PubMed]
- Isakadze, N.; Martin, S.S. How Useful Is the Smartwatch ECG? Trends Cardiovasc. Med. 2020, 30, 442–448. [Google Scholar] [CrossRef] [PubMed]
- van der Zande, J.; Strik, M.; Dubois, R.; Ploux, S.; Alrub, S.A.; Caillol, T.; Nasarre, M.; Donker, D.W.; Oppersma, E.; Bordachar, P. Using a Smartwatch to Record Precordial Electrocardiograms: A Validation Study. Sensors 2023, 23, 2555. [Google Scholar] [CrossRef]
- Shorif Uddin, M.; Chand Bansal, J. Proceedings of International Joint Conference on Computational Intelligence; Springer Nature: Berlin/Heidelberg, Germany, 2020. [Google Scholar]
- Tefas, A.; Pitas, I. Principal Component Analysis. In Intelligent Systems; CRC Press: Boca Raton, FL, USA, 2018; pp. 16.1–16.9. [Google Scholar]
- Habibzadeh, F.; Habibzadeh, P.; Yadollahie, M. On Determining the Most Appropriate Test Cut-off Value: The Case of Tests with Continuous Results. Biochem. Med. 2016, 26, 297–307. [Google Scholar] [CrossRef]
- Reddy, C.; Aggarwal, C. Data Clustering: Algorithms and Applications; CRC Press: Boca Raton, FL, USA, 2018. [Google Scholar]
- Ziefle, M.; Maciaszek, L.A. Information and Communication Technologies for Ageing Well and E-Health; Springer International Publishing: Berlin/Heidelberg, Germany, 2020. [Google Scholar]
- Walker, G.; Shostak, J. Common Statistical Methods for Clinical Research with SAS Examples, 3rd ed.; SAS Institute: Cary, NC, USA, 2010; ISBN 9781607644255. [Google Scholar]
- Nattel, S.; Guasch, E.; Savelieva, I.; Cosio, F.G.; Valverde, I.; Halperin, J.L.; Conroy, J.M.; Al-Khatib, S.M.; Hess, P.L.; Kirchhof, P.; et al. Early Management of Atrial Fibrillation to Prevent Cardiovascular Complications. Eur. Heart J. 2014, 35, 1448–1456. [Google Scholar] [CrossRef]
- Withings ScanWatch—What Is Electrocardiogram (ECG)? Available online: https://support.withings.com/hc/en-us/articles/360004559098-ScanWatch-What-is-Electrocardiogram-ECG- (accessed on 9 June 2023).
- Feeny, A.K.; Chung, M.K.; Madabhushi, A.; Attia, Z.I.; Cikes, M.; Firouznia, M.; Friedman, P.A.; Kalscheur, M.M.; Kapa, S.; Narayan, S.M.; et al. Artificial Intelligence and Machine Learning in Arrhythmias and Cardiac Electrophysiology. Circ. Arrhythm. Electrophysiol. 2020, 13, e007952. [Google Scholar] [CrossRef] [PubMed]
- Lown, M.; Brown, M.; Brown, C.; Yue, A.M.; Shah, B.N.; Corbett, S.J.; Lewith, G.; Stuart, B.; Moore, M.; Little, P. Machine Learning Detection of Atrial Fibrillation Using Wearable Technology. PLoS ONE 2020, 15, e0227401. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Chu, Y.; Ryu, J.; Park, Y.J.; Yang, S.; Koh, S.B. Artificial Intelligence for Detection of Cardiovascular-Related Diseases from Wearable Devices: A Systematic Review and Meta-Analysis. Yonsei Med. J. 2022, 63, S93–S107. [Google Scholar] [CrossRef] [PubMed]
- Benezet-Mazuecos, J.; Rubio, J.M.; Iglesias, J.A.; de la Vieja, J.J. How to Recognize Silent Atrial Fibrillation in Pacemakers and Defibrillators—The Value of Atrial Electrograms. J. Thorac. Dis. 2018, 10, 2048–2051. [Google Scholar] [CrossRef] [PubMed]
| Variable | Disease/Diagnosis AW | n (%) |
|---|---|---|
| Cardiac Disease | No disease | 173 (24%) |
| Atrial Fibrillation | 154 (21%) | |
| Atrial Flutter/Atrial Tachycardia | 33 | |
| Ventricular Tachycardia | 3 | |
| Junctional Tachycardia | 5 | |
| Ventricular Extrasystole | 54 | |
| Atrial Extrasystole | 21 | |
| First-degree AV-block | 77 | |
| Second/third-degree AV-block | 21 | |
| Sick Sinus Syndrome/Sinus Bradycardia | 65 | |
| Pacemaker | 26 | |
| CRT | 13 | |
| Right Bundle Branch Block | 54 | |
| Left Bundle Branch Block | 47 | |
| Intermittent Bundle Branch Block | 13 | |
| Left Anterior Hemiblock | 23 | |
| Right Heart Axis | 13 | |
| Wolff-Parkinson-White Syndrome | 26 | |
| Brugada Syndrome | 13 | |
| Arrhythmogenic Right Ventricular Cardiomyopathy | 20 | |
| Hypertrophic Cardiomyopathy | 10 | |
| Long QT Syndrome | 8 | |
| Q wave | 20 | |
| ST elevation/depression | 54 | |
| Negative T | 59 | |
| AW diagnosis | Sinus Rhythm | 455 (62%) |
| Atrial Fibrillation | 137 (19%) | |
| Inconclusive | 142 (19%) |
| Disease (n) | Count RR (a) Median | SVD Ratio (b) Median |
|---|---|---|
| Atrial Fibrillation (29) | 0.059 | 7.45 |
| AV block type 1 (12) | 0.229 | 44.30 |
| AV block type 2–3 (4) | 0.267 | 48.42 |
| No abnormalities (35) | 0.269 | 64.41 |
| Premature Atrial Contractions (5) | 0.188 | 7.36 |
| Premature Ventricular Contractions (8) | 0.154 | 15.80 |
| Sick Sinus Syndrome (12) | 0.253 | 53.51 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Velraeds, A.; Strik, M.; van der Zande, J.; Fontagne, L.; Haissaguerre, M.; Ploux, S.; Wang, Y.; Bordachar, P. Improving Automatic Smartwatch Electrocardiogram Diagnosis of Atrial Fibrillation by Identifying Regularity within Irregularity. Sensors 2023, 23, 9283. https://doi.org/10.3390/s23229283
Velraeds A, Strik M, van der Zande J, Fontagne L, Haissaguerre M, Ploux S, Wang Y, Bordachar P. Improving Automatic Smartwatch Electrocardiogram Diagnosis of Atrial Fibrillation by Identifying Regularity within Irregularity. Sensors. 2023; 23(22):9283. https://doi.org/10.3390/s23229283
Chicago/Turabian StyleVelraeds, Anouk, Marc Strik, Joske van der Zande, Leslie Fontagne, Michel Haissaguerre, Sylvain Ploux, Ying Wang, and Pierre Bordachar. 2023. "Improving Automatic Smartwatch Electrocardiogram Diagnosis of Atrial Fibrillation by Identifying Regularity within Irregularity" Sensors 23, no. 22: 9283. https://doi.org/10.3390/s23229283
APA StyleVelraeds, A., Strik, M., van der Zande, J., Fontagne, L., Haissaguerre, M., Ploux, S., Wang, Y., & Bordachar, P. (2023). Improving Automatic Smartwatch Electrocardiogram Diagnosis of Atrial Fibrillation by Identifying Regularity within Irregularity. Sensors, 23(22), 9283. https://doi.org/10.3390/s23229283






