Telerehabilitation with ARC Intellicare to Cope with Motor and Respiratory Disabilities: Results about the Process, Usability, and Clinical Effect of the “Ricominciare” Pilot Study
Abstract
:1. Introduction
Study Design and Objectives
- Adherence to the home rehabilitation program;
- Safety of rehabilitation therapy.
- The usability and acceptability of the intervention;
- The process to provide the new care pathway;
- Clinical effectiveness: in fact, the participants will undergo pre–post-intervention monitoring of disability in basal activity of daily living (ADL), respiratory outcomes, endurance and fatigue, mood, and quality of life.
2. Materials and Methods
2.1. ARC Intellicare
2.2. Subjects
2.3. Intervention Protocol
2.4. Study Endpoints and Outcome Measures
- 1.
- Adherence-Days = Total number of days the patient accessed the platform for training versus the total number of days the exercises were prescribed (1);
- Adhthresholded (d) = 0, when the patient never tried to access into ARC device to perform one of the exercises prescribed for day d;
- Adhthresholded (d) = 1, when Nexecutions (d) ≥ 1.
- 2.
- Adherence-Repetitions = total number of repetitions performed versus total number of repetitions prescribed, considering all exercises included in the rehabilitation plan (e, from 1 to n, where n is the total number of exercises prescribed to a subject) and all days (d) of treatment (from d = first, i.e. first day of treatment to d = last, i.e. last day for which an individual rehabilitation program was prescribed) (2).
- usability and acceptability of the intervention studied through the System Usability Scale (SUS) [56] and a semi-structured ad hoc-prepared questionnaire;
- The process to provide the new care pathway, measured by the percentage of subjects resulting eligible to the study.
- Disability: modified Barthel Index (mBI) [59];
- Motor outcomes: 2MWT [61];
- Fatigue: Brief Fatigue Inventory (BFI) [62];
- Mood and anxiety: Beck Depression or Anxiety Inventory (BDI, BAI) [63];
- Quality of Life: Euro-Quality of life Questionnaire self-assessment-5 Dimension (EQ-5D) and EQ-5D-Visual Analogic Scale (EQ-5D-VAS) [64].
| Secondary Objectives | Outcome Measures | Endpoint | Ref. |
|---|---|---|---|
| Process description of the care pathway Acceptability of intervention (tech) | Percentage of subjects resulting eligible to the study Percentage of subjects who accept to undergo telerehabilitation | n.a. n.a. | n.a. n.a. |
| Usability of the intervention device (tech) | System Usability Scale (SUS) | >70% | [56] |
| Clinical |
|
|
2.5. Ethical Procedures
2.6. Statistical Analysis
3. Results
3.1. Population and Process Description
3.2. Usability and Acceptability
3.3. Adherence
3.4. Clinical Data Evolution
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cieza, A.; Causey, K.; Kamenov, K.; Hanson, S.W.; Chatterji, S.; Vos, T. Global estimates of the need for rehabilitation based on the Global Burden of Disease study 2019: A systematic analysis for the Global Burden of Disease Study 2019. Lancet 2021, 396, 2006–2017, Erratum in Lancet 2020. [Google Scholar] [CrossRef]
- Katajisto, M.; Laitinen, T. Estimating the effectiveness of pulmonary rehabilitation for COPD exacerbations: Reduction of hospital inpatient days during the following year. Int. J. Chronic Obstr. Pulm. Dis. 2017, 12, 2763–2769. [Google Scholar] [CrossRef] [PubMed]
- Thomas, E.; Lotfaliany, M.; Grace, S.L.; Oldenburg, B.; Taylor, C.B.; Hare, D.L.; Rangani, W.P.T.; Dheerasinghe, D.S.A.F.; Cadilhac, D.A.; O’Neil, E. Effect of cardiac rehabilitation on 24-month all-cause hospital readmissions: A prospective cohort study. Eur. J. Cardiovasc. Nurs. 2019, 18, 234–244. [Google Scholar] [CrossRef] [PubMed]
- Désiron, H.A.; de Rijk, A.; Van Hoof, E.; Donceel, P. Occupational therapy and return to work: A systematic literature review. BMC Public Health 2011, 11, 615. [Google Scholar] [CrossRef] [PubMed]
- Prvu Bettger, J.A.; Stineman, M.G. Effectiveness of multidisciplinary rehabilitation services in postacute care: State-of-the-science. A review. Arch. Phys. Med. Rehabil. 2007, 88, 1526–1534. [Google Scholar] [CrossRef] [PubMed]
- Sarfo, F.S.; Ulasavets, U.; Opare-Sem, O.K.; Ovbiagele, B. Tele-rehabilitation after stroke: An updated systematic review of the liter-ature. J. Stroke Cerebrovasc. Dis. 2018, 27, 2306–2318. [Google Scholar] [CrossRef]
- Stucki, G.; Bickenbach, J.; Gutenbrunner, C.; Melvin, J. Rehabilitation: The health strategy of the 21st century. J. Rehabil. Med. 2018, 50, 309–316. [Google Scholar] [CrossRef]
- Forman, C.R.; Nielsen, J.B.; Lorentzen, J. Neuroplasticity at Home: Improving Home-Based Motor Learning through Technological Solutions. A Review. Front. Rehabil. Sci. 2021, 2, 789165. [Google Scholar] [CrossRef]
- Maresca, G.; Maggio, M.G.; De Luca, R.; Manuli, A.; Tonin, P.; Pignolo, L.; Calabrò, R.S. Tele-Neuro-Rehabilitation in Italy: State of the Art and Future Perspectives. Front. Neurol. 2020, 11, 563375. [Google Scholar] [CrossRef]
- Matamala-Gomez, M.; Maisto, M.; Montana, J.I.; Mavrodiev, P.A.; Baglio, F.; Rossetto, F.; Mantovani, F.; Riva, G.; Realdon, O. The Role of Engagement in Teleneurorehabilitation: A Systematic Review. Front. Neurol. 2020, 11, 354. [Google Scholar] [CrossRef]
- Leslie, S.; Tan, J.; McRae, P.J.; O’Leary, S.P.; Adsett, J.A. The Effectiveness of Exercise Interventions Supported by Telerehabilitation for Recently Hospitalized Adult Medical Patients: A Systematic Review. Int. J. Telerehabil. 2021, 13, e6356. [Google Scholar] [CrossRef]
- Winters, J.M. Telerehabilitation Research: Emerging Opportunities. Annu. Rev. Biomed. Eng. 2002, 4, 287–320. [Google Scholar] [CrossRef] [PubMed]
- DeFre Galea, M. Telemedicine in Rehabilitation. Phys. Med. Rehabil. Clin. N. Am. 2019, 30, 473–483. [Google Scholar] [CrossRef]
- Centers for Medicare and Medicaid Services. COVID-19 Emergency Declaration Blanket Waivers for Health Care Providers 2020. Available online: https://www.cms.gov/files/document/summary-COVID-19-emergency-declaration-waivers.pdf (accessed on 23 May 2020).
- WHO Coronavirus (COVID-19) Dashboard. Available online: https://covid19.who.int/ (accessed on 20 February 2022).
- Pepa, L.; Pigliapoco, M.; Bisoglio, P.; Lambertucci, A.; Coccia, M.; Ercolani, L.; Aringolo, M.; Hibel, M.; Gastaldi, A.; Andrenelli, E.; et al. Therapeutic Exercise Protocols for People Recovering after COVID-19: A Tele-Health Approach. In Ambient Assisted Living: Italian Forum 2020; Springer International Publishing: Cham, Switzerland, 2022; pp. 355–362. [Google Scholar]
- GBD 2016 Neurology Collaborators. Global, regional, and national burden of neurological disorders, 1990–2016: A systematic analysis for the global burden of disease study 2016. Lancet Neurol. 2019, 18, 459–480. [Google Scholar] [CrossRef] [PubMed]
- Dorsey, E.R.; Bloem, B.R. The Parkinson pandemic-a call to action. JAMA Neurol. 2018, 75, 9–10. [Google Scholar] [CrossRef] [PubMed]
- GBD 2016 Parkinson’s Disease Collaborators. Global, regional, and national burden of Parkinson’s disease, 1990–2016: A sys-tematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 2018, 17, 939–953. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, M.J.; Okun, M.S. Diagnosis and Treatment of Parkinson Disease: A Review. JAMA 2020, 323, 548–560. [Google Scholar] [CrossRef] [PubMed]
- van Uem, J.M.; Marinus, J.; Canning, C.; van Lummel, R.; Dodel, R.; Liepelt-Scarfone, I.; Berg, D.; Morris, M.E.; Maetzler, W. Health-Related Quality of Life in patients with Parkinson’s disease—A systematic review based on the ICF model. Neurosci. Biobehav. Rev. 2016, 61, 26–34. [Google Scholar] [CrossRef]
- Shulman, L.M. Understanding disability in Parkinson’s disease. Mov. Disord. 2010, 25 (Suppl. S1), S131–S135. [Google Scholar] [CrossRef]
- Bloem, B.R.; Okun, M.S.; Klein, C. Parkinson’s disease. Lancet 2021, 397, 2284–2303. [Google Scholar] [CrossRef]
- Brooks, S.; Weston, D.; Greenberg, N. Social and psychological impact of the COVID-19 pandemic on people with Parkinson’s disease: A scoping review. Public Health 2021, 199, 77–86. [Google Scholar] [CrossRef] [PubMed]
- Fasano, A.; Antonini, A.; Katzenschlager, R.; Krack, P.; Odin, P.; Evans, A.H.; Foltynie, T.; Volkmann, J.; Merello, M. Man-agement of Advanced Therapies in Parkinson’s Disease Patients in Times of Humanitarian Crisis: The COVID-19 Experience. Mov. Disord. Clin. Pract. 2020, 7, 361–372. [Google Scholar] [CrossRef] [PubMed]
- Cartella, S.M.; Terranova, C.; Rizzo, V.; Quartarone, A.; Girlanda, P. COVID-19 and Parkinson’s Disease: An Overview. J. Neurol. 2021, 268, 4415–4421. [Google Scholar] [CrossRef] [PubMed]
- De Donno, A.; Acella, A.; Angrisani, C.; Gubinelli, G.; Musci, G.; Gravili, G.; Ciritella, C.; Santamato, A. Suspension of Care for Patients with Spasticity during COVID-19 Pandemic: Ethical and Medico-Legal Point of View Starting from an Italian Study. Front. Med. 2021, 8, 754456. [Google Scholar] [CrossRef] [PubMed]
- Schirinzi, T.; Di Lazzaro, G.; Salimei, C.; Cerroni, R.; Liguori, C.; Scalise, S.; Alwardat, M.; Mercuri, N.B.; Pierantozzi, M.; Stefani, A.; et al. Physical Activity Changes and Correlate Effects in Patients with Parkinson’s Disease during COVID-19 Lockdown. Mov. Disord. Clin. Pract. 2020, 7, 797–802. [Google Scholar] [CrossRef]
- Capecci, M.; Baldini, N.; Campignoli, F.; Lombardo, L.P.; Andrenelli, E.; Ceravolo, M.G. Clinical and Functional Evolution in Subjects with Parkinson’s Disease during SARS-CoV-2 Pandemic. Appl. Sci. 2023, 13, 1126. [Google Scholar] [CrossRef]
- Piroth, L.; Cottenet, J.; Mariet, A.-S.; Bonniaud, P.; Blot, M.; Tubert-Bitter, P.; Quantin, C. Comparison of the characteristics, morbidity, and mortality of COVID-19 and seasonal influenza: A nationwide, population-based retrospective cohort study. Lancet Respir. Med. 2021, 9, 251–259. [Google Scholar] [CrossRef]
- de Sire, A.; Andrenelli, E.; Negrini, F.; Lazzarini, S.G.; Cordani, C.; Ceravolo, M.G. The International Multiprofessional Steering Committee of Cochrane Rehabilitation REH-COVER Action Rehabilitation and COVID-19: Update of the rapid living systematic review by Cochrane Rehabilitation Field as of February 28th, 2022. Eur. J. Phys. Rehabil. Med. 2022, 58, 498–501. [Google Scholar] [CrossRef]
- Tofiq, A.; Eriksson Crommert, M.; Zakrisson, A.B.; von Euler, M.; Nilsing Strid, E. Physical functioning post-COVID-19 and the recovery process: A mixed methods study. Disabil. Rehabil. 2023, 20, 1–10. [Google Scholar] [CrossRef]
- Negrini, S.; Grabljevec, K.; Boldrini, P.; Kiekens, C.; Moslavac, S.; Zampolini, M.; Christodoulou, N. Up to 2.2 million people experiencing disability suffer collateral damage each day of COVID-19 lockdown in Europe. Eur. J. Phys. Rehabil. Med. 2020, 56, 361–365. [Google Scholar] [CrossRef]
- Soriano, J.B.; Murthy, S.; Marshall, J.C.; Relan, P.; Diaz, J.V.; WHO Clinical Case Definition Working Group on Post-COVID-19 Condition. A clinical case definition of post-COVID-19 condition by a Delphi consensus. Lancet Infect. Dis. 2022, 22, e102–e107. [Google Scholar] [CrossRef] [PubMed]
- Boldrini, P.; Garcea, M.; Brichetto, G.; Reale, N.; Tonolo, S.; Falabella, V.; Fedeli, F.; Cnops, A.A.; Kiekens, C. Living with a disability during the pandemic. “Instant paper from the field” on rehabilitation answers to the COVID-19 emergency. Eur. J. Phys. Rehabil. Med. 2020, 56, 331–334. [Google Scholar] [CrossRef] [PubMed]
- Negrini, S.; Kiekens, C.; Bernetti, A.; Capecci, M.; Ceravolo, M.G.; Lavezzi, S.; Zampolini, M.; Boldrini, P. Telemedicine from research to practice during the pandemic. “Instant paper from the field” on rehabilitation answers to the COVID-19 emergency. Eur. J. Phys. Rehabil. Med. 2020, 56, 327–330. [Google Scholar] [CrossRef] [PubMed]
- Tsavourelou, A.; Stylianides, N.; Papadopoulos, A.; Dikaiakos, M.D.; Nanas, S.; Kyprianoy, T.; Tokmakidis, S.P. Telerehabilitation solution conceptual paper for community-based exercise rehabilitation of patients discharged after critical illness. Int. J. Telerehabil. 2016, 8, 61–70. [Google Scholar] [CrossRef]
- Nelson, M.; Bourke, M.; Crossley, K.; Russell, T. Telerehabilitation Versus Traditional Care Following Total Hip Replacement: A Randomized Controlled Trial Protocol. JMIR Res. Protoc. 2017, 6, e34. [Google Scholar] [CrossRef] [PubMed]
- Nix, J.; Comans, T. Home Quick—Occupational Therapy Home Visits Using mHealth, to Facilitate Discharge from Acute Admission Back to the Community. Int. J. Telerehabilitation 2017, 9, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Cottrell, M.A.; Galea, O.A.; O’Leary, S.P.; Hill, A.J.; Russell, T.G. Real-time telerehabilitation for the treatment of musculoskeletal conditions is effective and comparable to standard practice: A systematic review and meta-analysis. Clin. Rehabil. 2017, 31, 625–638. [Google Scholar] [CrossRef]
- Agostini, M.; Moja, L.; Banzi, R.; Pistotti, V.; Tonin, P.; Venneri, A.; Turolla, A. Telerehabilitation and recovery of motor function: A systematic review and meta-analysis. J. Telemed. Telecare 2015, 21, 202–213. [Google Scholar] [CrossRef]
- Grona, S.L.; Bath, B.; Busch, A.; Rotter, T.; Trask, C.; Harrison, E. Use of videoconferencing for physical therapy in people with musculoskeletal conditions: A systematic review. J. Telemed. Telecare 2017, 24, 341–355. [Google Scholar] [CrossRef]
- Sanford, J.A.; Griffiths, P.C.; Richardson, P.; Hargraves, K.; Butterfield, T.; Hoenig, H. The Effects of In-Home Rehabilitation on Task Self-Efficacy in Mobility-Impaired Adults: A Randomized Clinical Trial. J. Am. Geriatr. Soc. 2006, 54, 1641–1648. [Google Scholar] [CrossRef]
- Prvu Bettger, J.; Green, C.L.; Holmes, D.N.; Chokshi, A.; Mather, R.C., 3rd; Hoch, B.T.; de Leon, A.J.; Aluisio, F.; Seyler, T.M.; Del Gaizo, D.J.; et al. Effects of virtual exercise rehabilitation in-home therapy compared with traditional care after total knee arthroplasty: VERITAS, a randomized controlled trial. J. Bone Joint Surg. Am. 2020, 102, 101–109. [Google Scholar] [CrossRef] [PubMed]
- Tousignant, M.; Boissy, P.; Corriveau, H.; Moffet, H. In home telerehabilitation for older adults after discharge from an acute hospital or rehabilitation unit: A proof-of-concept study and costs estimation. Disabil. Rehabil. Assist. Technol. 2006, 1, 209–216. [Google Scholar] [CrossRef]
- Bini, S.; Mahajan, J. Clinical outcomes of remote asynchronous telerehabilitation are equivalent to traditional therapy following total knee arthroplasty: A randomized control study. J. Telemed. Telecare 2017, 23, 239–247. [Google Scholar] [CrossRef]
- Yeroushalmi, S.; Maloni, H.; Costello, K.; Wallin, M.T. Telemedicine and multiple sclerosis: A comprehensive literature review. J. Telemed. Telecare 2019, 26, 400–413. [Google Scholar] [CrossRef] [PubMed]
- León-Salas, B.; González-Hernández, Y.; Infante-Ventura, D.; de Armas-Castellano, A.; García-García, J.; García-Hernández, M.; Carmona-Rodríguez, M.; Olazarán, J.; Dobato, J.L.; Rodríguez-Rodríguez, L.; et al. Telemedicine for neurological diseases: A systematic review and meta-analysis. Eur. J. Neurol. 2022, 30, 241–254. [Google Scholar] [CrossRef]
- Beange, K.H.; Chan, A.D.; Beaudette, S.M.; Graham, R.B. Concurrent validity of a wearable IMU for objective assessments of functional movement quality and control of the lumbar spine. J. Biomech. 2019, 97, 109356. [Google Scholar] [CrossRef]
- Guidolin, M.; Petrea, R.A.B.; Roberto, O.; Reggiani, M.; Menegatti, E.; Tagliapietra, L. On the accuracy of imus for human motion tracking: A comparative evaluation. In Proceedings of the 2021 IEEE International Conference on Mechatronics (ICM), Kashiwa, Japan, 7–9 March 2021; pp. 1–6. [Google Scholar] [CrossRef]
- Ceravolo, M.G.; Arienti, C.; de Sire, A.; Andrenelli, E.; Negrini, F.; Lazzarini, S.G.; Patrini, M.; Negrini, S. The International Multiprofessional Steering Committee of Cochrane Rehabilitation REH-COVER action Rehabilitation and COVID-19: The Cochrane Rehabilitation 2020 rapid living systematic review. Eur. J. Phys. Rehabil. Med. 2020, 56, 642–651. [Google Scholar] [CrossRef] [PubMed]
- Vitacca, M.; Paneroni, M.; Baiardi, P.; De Carolis, V.; Zampogna, E.; Belli, S.; Carone, M.; Spanevello, A.; Balbi, B.; Bertolotti, G. Development of a Barthel Index based on dyspnea for patients with respiratory diseases. Int. J. Chronic Obstr. Pulm. Dis. 2016, 11, 1199. [Google Scholar] [CrossRef]
- Perry, J.; Garrett, M.; Gronley, J.K.; Mulroy, S.J. Classification of Walking Handicap in the Stroke Population. Stroke 1995, 26, 982–989. [Google Scholar] [CrossRef]
- Dolgin, M.; Association, N.Y.H.; Fox, A.C.; Gorlin, R.; Levin, R.I. New York Heart Association. Criteria Committee. In Nomenclature and Criteria for Diagnosis of Diseases of the Heart and Great Vessels, 9th ed.; Lippincott Williams and Wilkins: Boston, MA, USA, 1994. [Google Scholar]
- Quinn, T.J.; Dawson, J.; Walters, M.R.; Lees, K.R. Reliability of the modified Rankin Scale: A systematic review. Stroke 2009, 40, 3393–3395. [Google Scholar] [CrossRef]
- Bangor, A.; Kortum, P.T.; Miller, J.T. An Empirical Evaluation of the System Usability Scale. Int. J. Hum. Comput. Interact. 2008, 24, 574–594. [Google Scholar] [CrossRef]
- Babatunde, F.; MacDermid, J.; MacIntyre, N. Characteristics of therapeutic alliance in musculoskeletal physiotherapy and occu-pational therapy practice: A scoping review of the literature. BMC Health Serv. Res. 2017, 17, 375. [Google Scholar]
- Palazzo, C.; Klinger, E.; Dorner, V.; Kadri, A.; Thierry, O.; Boumenir, Y.; Martin, W.; Poiraudeau, S.; Ville, I. Barriers to home-based exercise program adherence with chronic low back pain: Patient expectations regarding new technologies. Ann. Phys. Rehabil. Med. 2016, 59, 107–113. [Google Scholar] [CrossRef]
- Shah, S.; Vanclay, F.; Cooper, B. Improving the sensitivity of the Barthel Index for stroke rehabilitation. J. Clin. Epidemiol. 1989, 42, 703–709. [Google Scholar] [CrossRef] [PubMed]
- Wilson, R.C.; Jones, P.W. Long-term reproducibility of Borg scale estimates of breathlessness during exercise. Clin. Sci. 1991, 80, 309–312. [Google Scholar] [CrossRef] [PubMed]
- Leung, A.S.; Chan, K.K.; Sykes, K. Reliability, Validity, and Responsiveness of a 2-Min Walk Test To Assess Exercise Capacity of COPD Patients. Chest 2006, 130, 119–125. [Google Scholar] [CrossRef]
- Shuman-Paretsky, M.J.; Belser-Ehrlich, J.; Holtzer, R. Psychometric Properties of the Brief Fatigue Inventory in Community-Dwelling Older Adults. Arch. Phys. Med. Rehabil. 2014, 95, 1533–1539. [Google Scholar] [CrossRef]
- Lee, K.; Kim, D.; Cho, Y. Exploratory Factor Analysis of the Beck Anxiety Inventory and the Beck Depression Inventory-II in a Psychiatric Outpatient Population. J. Korean Med. Sci. 2018, 33, e128. [Google Scholar] [CrossRef]
- Rabin, R.; Charro, F.D. EQ-5D: A measure of health status from the EuroQol Group. Ann. Med. 2001, 33, 337–343. [Google Scholar] [CrossRef]
- Rosen, L.; Whaling, K.; Carrier, L.; Cheever, N.; Rokkum, J.; Rosen, L.; Whaling, K.; Carrier, L.; Cheever, N.; Rokkum, J. The Media and Technology Usage and Attitudes Scale: An empirical investigation. Comput. Hum. Behav. 2013, 29, 2501–2511. [Google Scholar] [CrossRef]
- Pirrotta, F.; Timpano, F.; Bonanno, L.; Nunnari, D.; Marino, S.; Bramanti, P.; Lanzafame, P. Italian Validation of Montreal Cognitive Assessment. Eur. J. Psychol. Assess. 2015, 31, 131–137. [Google Scholar] [CrossRef]
- Franceschini, M.; Rampello, A.; Agosti, M.; Massucci, M.; Bovolenta, F.; Sale, P. Walking Performance: Correlation between Energy Cost of Walking and Walking Participation. New Statistical Approach Concerning Outcome Measurement. PLoS ONE 2013, 8, e56669. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, Y.-W.; Wang, C.-H.; Wu, S.-C.; Chen, P.-C.; Sheu, C.-F.; Hsieh, C.-L. Establishing the Minimal Clinically Important Difference of the Barthel Index in Stroke Patients. Neurorehabilit. Neural Repair 2007, 21, 233–238. [Google Scholar] [CrossRef] [PubMed]
- Vitacca, M.; Malovini, A.; Balbi, B.; Aliani, M.; Cirio, S.; Spanevello, A.; Fracchia, C.; Maniscalco, M.; Corica, G.; Ambrosino, N.; et al. Minimal Clinically Important Difference in Barthel Index Dyspnea in Patients with COPD. Int. J. Chronic Obstr. Pulm. Dis. 2020, 15, 2591–2599. [Google Scholar] [CrossRef] [PubMed]
- Di Tella, S.; Isernia, S.; Pagliari, C.; Jonsdottir, J.; Castiglioni, C.; Gindri, P.; Gramigna, C.; Canobbio, S.; Salza, M.; Molteni, F.; et al. A Multidimensional Virtual Reality Neurorehabilitation Approach to Improve Functional Memory: Who Is the Ideal Candidate? Front. Neurol. 2021, 11, 618330. [Google Scholar] [CrossRef] [PubMed]
- Bohannon, R.W.; Crouch, R. Minimal clinically important difference for change in 6-minute walk test distance of adults with pathology: A systematic review. J. Eval. Clin. Pract. 2017, 23, 377–381. [Google Scholar] [CrossRef] [PubMed]
- Ries, A.L. Minimally clinically important difference for the UCSD Shortness of Breath Questionnaire, Borg Scale, and Visual Analog Scale. COPD 2005, 2, 105–110. [Google Scholar] [CrossRef] [PubMed]
- Ceravolo, M.G.; Andrenelli, E.; Arienti, C.; Côté, P.; DESire, A.; Iannicelli, V.; Lazzarini, S.G.; Negrini, F.; Patrini, M.; Negrini, S. Rehabilitation and COVID-19: Rapid living systematic review by Cochrane Rehabilitation Field—Third edition. Update as of June 30th, 2021. Eur. J. Phys. Rehabil. Med. 2021, 57, 850–857. [Google Scholar] [CrossRef]
- Rossetto, F.; Borgnis, F.; Isernia, S.; Foglia, E.; Garagiola, E.; Realdon, O.; Baglio, F. System Integrated Digital Empowering and teleRehabilitation to promote patient Activation and well-Being in chronic disabilities: A usability and acceptability study. Front. Public Health 2023, 11, 1154481. [Google Scholar] [CrossRef]
- Kim, H.-C. Acceptability Engineering: The Study of user Acceptance of Innovative Technologies. J. Appl. Res. Technol. 2015, 13, 230–237. [Google Scholar] [CrossRef]
- Davis, F.D. Perceived Usefulness, Perceived Ease of Use, and User Acceptance of Information Technology. MIS Q. 1989, 13, 319–340. [Google Scholar] [CrossRef]
- Tsertsidis, A.; Kolkowska, E.; Hedström, K. Factors influencing seniors’ acceptance of technology for ageing in place in the post-implementation stage: A literature review. Int. J. Med. Inform. 2019, 129, 324–333. [Google Scholar] [CrossRef]
- Hirani, S.P.; Rixon, L.; Beynon, M.; Cartwright, M.; Cleanthous, S.; Selva, A.; Sanders, C.; Newman, S.P. Quantifying beliefs regarding telehealth: Development of the Whole Systems Demonstrator Service User Technology Acceptability Questionnaire. J. Telemed. Telecare 2016, 23, 460–469. [Google Scholar] [CrossRef]
- Yusif, S.; Soar, J.; Hafeez-Baig, A. Older people, assistive technologies, and the barriers to adoption: A systematic review. Int. J. Med. Inform. 2016, 94, 112–116. [Google Scholar] [CrossRef] [PubMed]
- Palacholla, R.S.; Fischer, N.; Coleman, A.; Agboola, S.; Kirley, K.; Felsted, J.; Katz, C.; Lloyd, S.; Jethwani, K. Provider- and Patient-Related Barriers to and Facilitators of Digital Health Technology Adoption for Hypertension Management: Scoping Review. JMIR Cardio 2019, 3, e11951. [Google Scholar] [CrossRef] [PubMed]
- Di Tella, S.; Pagliari, C.; Blasi, V.; Mendozzi, L.; Rovaris, M.; Baglio, F. Integrated telerehabilitation approach in multiple sclerosis: A systematic review and meta-analysis. J. Telemed. Telecare 2019, 26, 385–399. [Google Scholar] [CrossRef] [PubMed]
- Isernia, S.; Di Tella, S.; Pagliari, C.; Jonsdottir, J.; Castiglioni, C.; Gindri, P.; Salza, M.; Gramigna, C.; Palumbo, G.; Molteni, F.; et al. Effects of an Innovative Telerehabilitation Intervention for People with Parkinson’s Disease on Quality of Life, Motor, and Non-motor Abilities. Front. Neurol. 2020, 11, 846. [Google Scholar] [CrossRef] [PubMed]
- Isernia, S.; Pagliari, C.; Jonsdottir, J.; Castiglioni, C.; Gindri, P.; Gramigna, C.; Palumbo, G.; Salza, M.; Molteni, F.; Baglio, F.; et al. Efficiency and Patient-Reported Outcome Measures From Clinic to Home: The Human Empowerment Aging and Disability Program for Digital-Health Rehabilitation. Front. Neurol. 2019, 10, 1206. [Google Scholar] [CrossRef] [PubMed]
- Hawley-Hague, H.; Lasrado, R.; Martinez, E.; Stanmore, E.; Tyson, S. A scoping review of the feasibility, acceptability, and effects of physiotherapy delivered remotely. Disabil. Rehabil. 2022; ahead of print. [Google Scholar] [CrossRef]
- Bally, E.L.S.; Cheng, D.; van Grieken, A.; Sanz, M.F.; Zanutto, O.; Carroll, A.; Darley, A.; Roozenbeek, B.; Dippel, D.W.J.; Raat, H. Patients’ Perspectives Regarding Digital Health Technology to Support Self-management and Improve Integrated Stroke Care: Qualitative Interview Study. J. Med. Internet Res. 2023, 25, e42556. [Google Scholar] [CrossRef]
- Martin, L.R.; Williams, S.L.; Haskard, K.B.; DiMatteo, M.R. The challenge of patient adherence. Ther. Clin. Risk Manag. 2005, 1, 189–199. [Google Scholar] [PubMed]
- Jack, K.; McLean, S.M.; Moffett, J.K.; Gardiner, E. Barriers to treatment adherence in physiotherapy outpatient clinics: A systematic review. Man. Ther. 2010, 15, 220–228. [Google Scholar] [CrossRef]
- Sluijs, E.M.; Kok, G.J.; van der Zee, J. Correlates of Exercise Compliance in Physical Therapy. Phys. Ther. 1993, 73, 771–782. [Google Scholar] [CrossRef]
- Simek, E.M.; McPhate, L.; Haines, T.P. Adherence to and efficacy of home exercise programs to prevent falls: A systematic review and meta-analysis of the impact of exercise program characteristics. Prev. Med. 2012, 55, 262–275. [Google Scholar] [CrossRef]
- Haas, B.M.; Trew, M.; Castle, P.C. Effects of Respiratory Muscle Weakness on Daily Living Function, Quality of Life, Activity Levels, and Exercise Capacity in Mild to Moderate Parkinson’s Disease. Am. J. Phys. Med. Rehabil. 2004, 83, 601–607. [Google Scholar] [CrossRef]
- Medina-Mirapeix, F.; Escolar-Reina, P.; Gascón-Cánovas, J.J.; Montilla-Herrador, J.; Jimeno-Serrano, F.J.; Collins, S.M. Predictive factors of adherence to frequency and duration components in home exercise programs for neck and low back pain: An observational study. BMC Musculoskelet. Disord. 2009, 10, 155. [Google Scholar] [CrossRef] [PubMed]
- Slade, S.C.; Patel, S.; Underwood, M.; Keating, J.L. What are patient beliefs and perceptions about exercise for non-specific chronic low back pain? A systematic review of qualitative studies. Clin. J. Pain 2014, 30, 995–1005. [Google Scholar] [CrossRef] [PubMed]
- Jordan, J.L.; Holden, M.A.; Mason, E.E.; Foster, N.E. Interventions to improve adherence to exercise for chronic musculoskeletal pain in adults. Cochrane Database Syst. Rev. 2010, 1, CD005956. [Google Scholar] [CrossRef]
- Petzinger, G.M.; Fisher, B.E.; McEwen, S.; Beeler, J.A.; Walsh, J.P.; Jakowec, M.W. Exercise-enhanced neuroplasticity targeting motor and cognitive circuitry in Parkinson’s disease. Lancet Neurol. 2013, 12, 716–726. [Google Scholar] [CrossRef]
- Levin, M.F.; Demers, M. Motor learning in neurological rehabilitation. Disabil. Rehabil. 2021, 43, 3445–3453. [Google Scholar] [CrossRef]
- Yen, J.M.; Lim, J.H. A Clinical Perspective on Bespoke Sensing Mechanisms for Remote Monitoring and Rehabilitation of Neurological Diseases: Scoping Review. Sensors 2023, 23, 536. [Google Scholar] [CrossRef]
- Duncan, R.P.; Combs-Miller, S.A.; McNeely, M.E.; Leddy, A.L.; Cavanaugh, J.T.; Dibble, L.E.; Ellis, T.D.; Ford, M.P.; Foreman, K.B.; Earhart, G.M. Are the average gait speeds during the 10 meter and 6 minute walk tests redundant in Parkinson disease? Gait Posture 2017, 52, 178–182. [Google Scholar] [CrossRef]
- Kataoka, H.; Sugie, K. Association between Fatigue and Hoehn-Yahr Staging in Parkinson’s Disease: Eight-Year Follow-Up Study. Neurol. Int. 2021, 13, 224–231. [Google Scholar] [CrossRef]




| Primary Objectives | Outcome Measures | Endpoint | Ref. |
|---|---|---|---|
| Adherence to home rehabilitation program | Exercise adherence (days) Exercise adherence (repetitions) | 80% 70% | [57,58] |
| Safety | Number of unanticipated serious device-related adverse effects (USADE), calculated on the total number of adverse events (AE) reported. | 0 | n.a. |
| Total | COV19 | pwPD | COV19 vs. pwPD COMPARISON | |||||
|---|---|---|---|---|---|---|---|---|
| Mean | Std. Dev. | Mean | Std. Dev. | Mean | Std. Dev. | Z Value | p Value | |
| Age | 61.1 | 10.5 | 57.3 | 13.2 | 65.4 | 3.7 | −1.7 | 0.09 |
| Gender | 7 F/14 M | 5 F/6 M | 2 F/8 M | |||||
| Education (Years) | 12.5 | 4.2 | 13.9 | 3.8 | 11 | 4.2 | −1.5 | 0.12 |
| Hospitalization length of stay (days) | 13.2 | 22.3 | 26.3 | 25.8 | n.a. | n.a. | ||
| Disease duration (years) | 9 | 2.3 | ||||||
| DAYS FROM COVID19 | 117 | 82 | ||||||
| RANKIN disability score | 1.4 | 1.0 | 1 | 0 | 1.8 | 1.3 | −1.2 | 0.24 |
| WHS (T0) | 5.3 | 1.0 | 5.5 | 0.52 | 5.2 | 1.3 | −1.9 | 0.62 |
| MTUAS | 213 | 69.6 | 235.3 | 78.5 | 188.5 | 51.6 | −1.5 | 0.14 |
| BaDI | 83.1 | 14.0 | 77.8 | 15.8 | 89 | 9.3 | −1.7 | 0.08 |
| mBI | 95.8 | 8.8 | 99.3 | 1.8 | 92 | 11.7 | −2.1 | 0.03 |
| MoCA | 25.7 | 3.2 | 26.8 | 2.1 | 24.5 | 3.8 | −1.6 | 0.1 |
| Hoehn and Yahr stage | - | - | - | - | 3 | 0.9 | n.a. | n.a. |
| UPDRS total score | - | - | - | - | 33 | 14 | n.a. | n.a. |
| Total | COV19 | pwPD | COV19 vs. pwPD COMPARISON | |||||
|---|---|---|---|---|---|---|---|---|
| Mean | Std. Dev. | Mean | Std. Dev. | Mean | Std. Dev | Z | p | |
| Adherence-Days Total | 82.2 | 17.7 | 83.7 | 14.7 | 80.4 | 21.3 | −0.07 | 0.9 |
| Adherence-Reps Total | 75.0 | 20.9 | 77.1 | 19.3 | 72.8 | 23.5 | −27 | 0.79 |
| Adherence-Days ME | 83.1 | 18.5 | 84.5 | 16.2 | 81.5 | 21.5 | −0.32 | 0.75 |
| Adherence-Days RE | 81.7 | 16.8 | 83.1 | 12.4 | 80.1 | 21.3 | −0.23 | 0.80 |
| Adherence-Reps ME | 74.7 | 21.7 | 77.1 | 20.1 | 71.9 | 24.1 | −0.18 | 0.86 |
| Adherence-Reps RE | 77.7 | 18.1 | 77.5 | 16.2 | 77.8 | 21.0 | −0.04 | 0.97 |
| T0 | T1 | Pre–Post-Treatment Wilcoxon Signed Rank Test Results | |||
|---|---|---|---|---|---|
| Mean | Std. Dev. | Mean | Std. Dev. | Z-Value; p-Value | |
| BaDI WS | 83.14 | 14.03 | 92.48 | 16.36 | −3.0; 0.003 |
| BaDI COV19 | 77.82 | 15.85 | 88.91 | 22.22 | −2.1; 0.04 |
| BaDI pwPD | 89 | 9.26 | 96.4 | 3.69 | −2.4; 0.02 |
| mBI WS | 95.81 | 8.77 | 97.19 | 6.65 | −1.7; 0.09 |
| mBI COV19 | 99.27 | 1.85 | 100 | 0 | −1.3;.18 |
| mBI pwPD | 92 | 11.67 | 94.1 | 8.83 | −1.2; 0.25 |
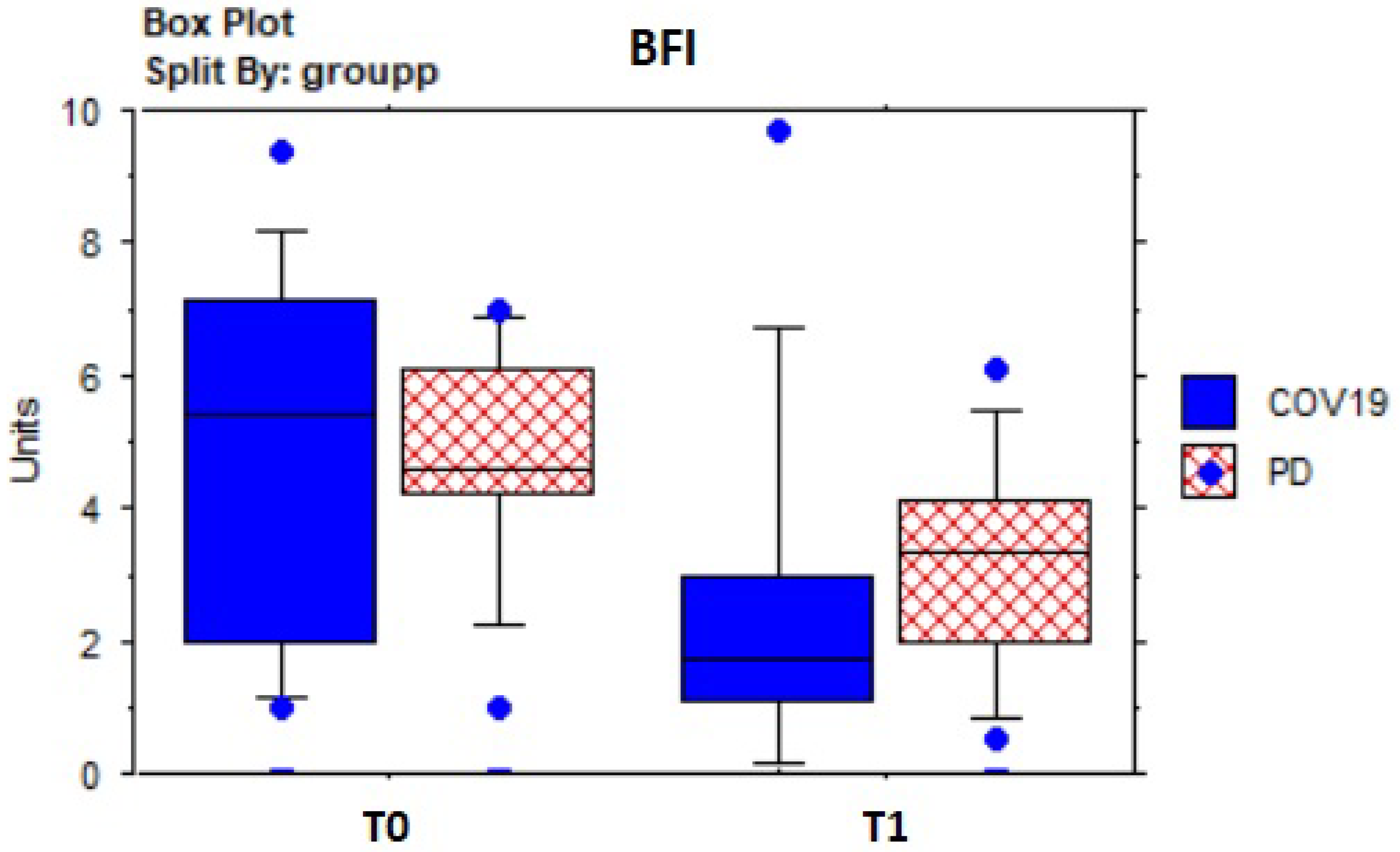
| BFI WS | 4.75 | 2.33 | 2.86 | 2.26 | −3.5; 0.0005 |
| BFI COV19 | 4.79 | 2.86 | 2.53 | 2.72 | −2.4; 0.01 |
| BFI pwPD | 4.71 | 1.73 | 3.22 | 1.67 | −2.2; 0.03 |
| HR pre 2MWT, WS | 88.48 | 11.95 | 85.29 | 15.94 | −0.88; 0.38 |
| HR pre 2MWT, COV19 | 91 | 10.17 | 85.64 | 13.79 | −0.89; 0.38 |
| HR pre 2MWT, pwPD | 85.7 | 13.64 | 84.9 | 18.8 | −0.10; 0.92 |
| SpO2 pre 2MWT, WS | 96.43 | 2.16 | 96.91 | 1.48 | −0.59; 0.55 |
| SpO2 pre 2MWT, COV19 | 96.46 | 2.42 | 97.18 | 1.47 | −0.42; 0.67 |
| SpO2 pre 2MWT, pwPD | 96.4 | 1.96 | 96.6 | 1.51 | −0.42; 0.67 |
| HR post 2MWT, WS | 96.71 | 15.13 | 101.38 | 14.09 | −0.86; 0.39 |
| HR post 2MWT, COV19 | 99.64 | 10.81 | 104.55 | 14.95 | −0.40; 0.68 |
| HR post 2MWT, pwPD | 93.5 | 18.9 | 97.9 | 12.92 | −0.89; 0.37 |
| SpO2 post 2MWT, WS | 96.38 | 1.56 | 96 | 1.70 | −0.80; 0.42 |
| SpO2 post 2MWT, COV19 | 97 | 1.61 | 96.36 | 1.29 | −0.98; 0.33 |
| SpO2 post 2MWT pwPD | 95.7 | 1.25 | 95.6 | 2.07 | −0.07; 0.94 |
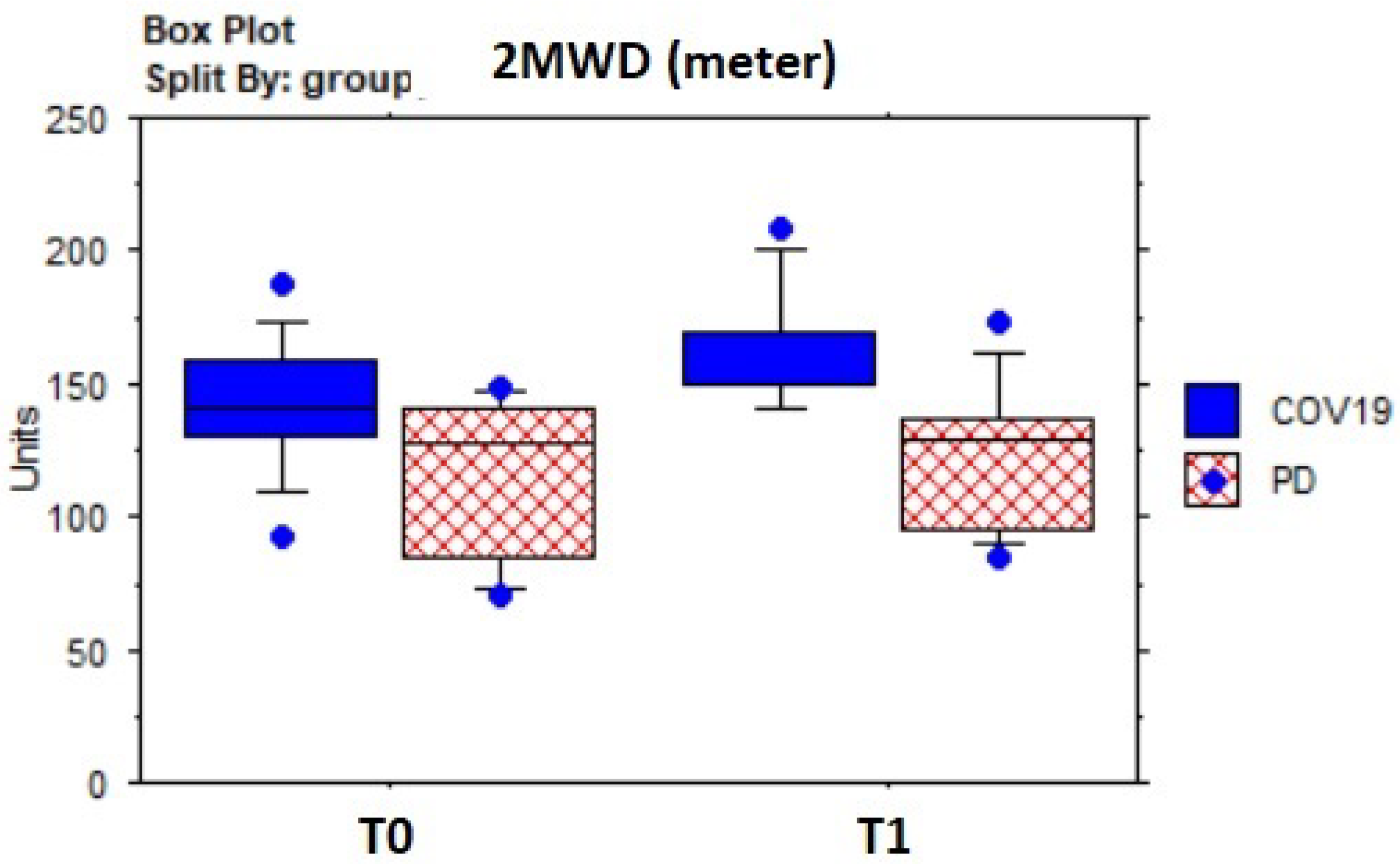
| 2MWT (m) WS | 128.95 | 29.52 | 143.24 | 30.31 | −3.3; 0.001 |
| 2MWT (m) COV19 | 140.46 | 25.23 | 160.46 | 22.3 | −2.7; 0.005 |
| 2MWT (m) pwPD | 116.3 | 29.81 | 124.3 | 27 | −0.1.8; 0.07 |
| BDI WS | 12.38 | 9.28 | 10.14 | 8.93 | −2.6; 0.01 |
| BDI COV19 | 11.91 | 9.17 | 9 | 9.61 | −2.5; 0.01 |
| BDI pwPD | 12.9 | 9.87 | 11.4 | 8.44 | −1.2; 0.23 |
| BAI WS | 13.19 | 9.62 | 9.62 | 11.93 | −3.1; 0.002 |
| BAI COV19 | 13.91 | 12.99 | 11.46 | 16.34 | −1.6; 0.09 |
| BAI pwPD | 12.4 | 4.09 | 7.6 | 3.31 | −2.8; 0.005 |
| EQ-5D WS | 8.381 | 1.884 | 7.524 | 1.83 | −2.6; 0.008 |
| EQ-5D COV19 | 8.455 | 2.067 | 7.455 | 2.21 | −2.0; 0.05 |
| EQ-5D pwPD | 8.3 | 1.77 | 7.6 | 1.43 | −1.8; 0.07 |
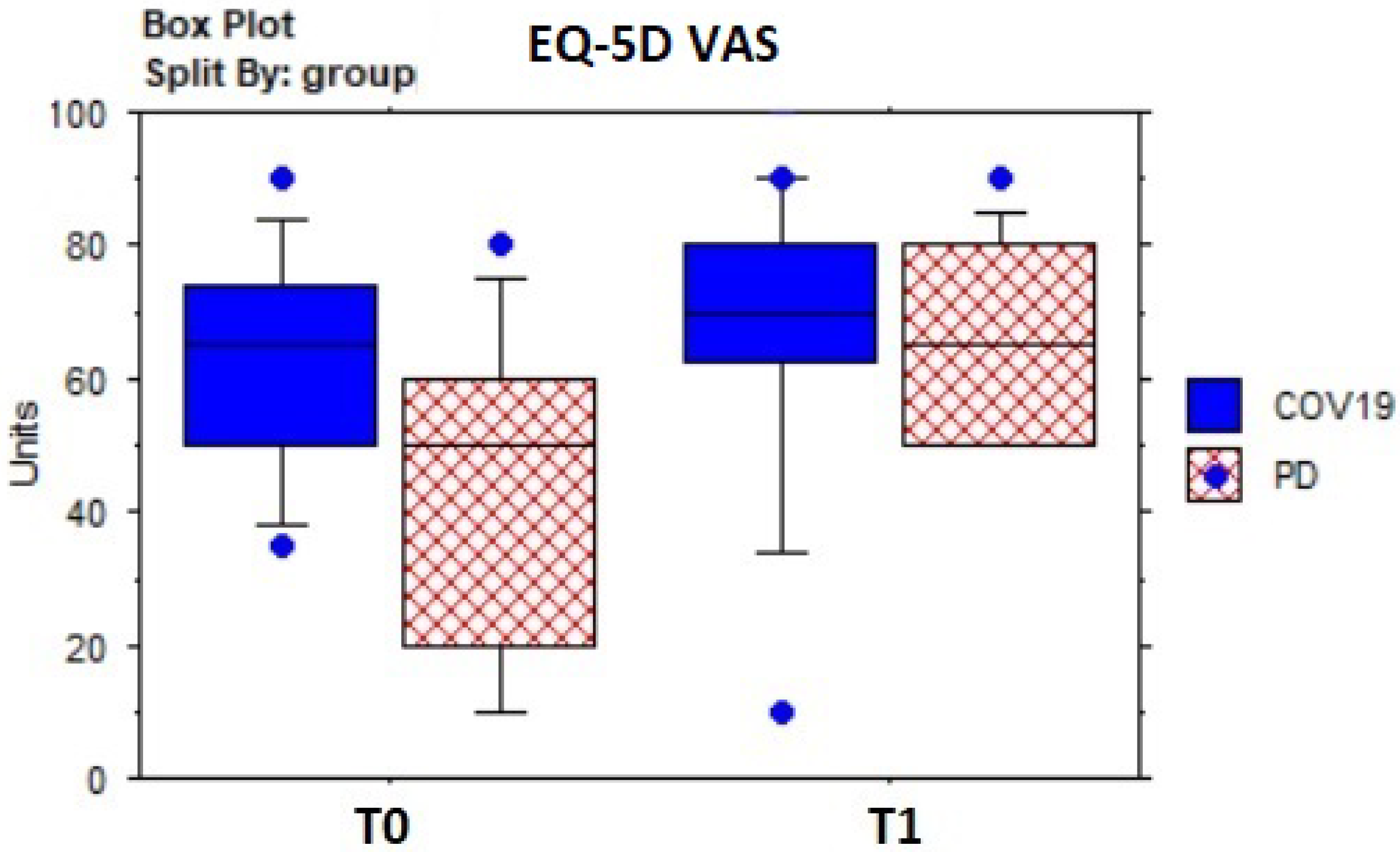
| EQ-5D VAS WS | 54.76 | 22.16 | 67.62 | 18.95 | −2.7; 0.007 |
| EQ-5D VAS COV19 | 62.73 | 17.08 | 68.18 | 22.72 | −1.5; 0.12 |
| EQ-5D VAS pwPD | 46 | 24.59 | 67 | 14.94 | −2.2; 0.03 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Capecci, M.; Cima, R.; Barbini, F.A.; Mantoan, A.; Sernissi, F.; Lai, S.; Fava, R.; Tagliapietra, L.; Ascari, L.; Izzo, R.N.; et al. Telerehabilitation with ARC Intellicare to Cope with Motor and Respiratory Disabilities: Results about the Process, Usability, and Clinical Effect of the “Ricominciare” Pilot Study. Sensors 2023, 23, 7238. https://doi.org/10.3390/s23167238
Capecci M, Cima R, Barbini FA, Mantoan A, Sernissi F, Lai S, Fava R, Tagliapietra L, Ascari L, Izzo RN, et al. Telerehabilitation with ARC Intellicare to Cope with Motor and Respiratory Disabilities: Results about the Process, Usability, and Clinical Effect of the “Ricominciare” Pilot Study. Sensors. 2023; 23(16):7238. https://doi.org/10.3390/s23167238
Chicago/Turabian StyleCapecci, Marianna, Rossella Cima, Filippo A. Barbini, Alice Mantoan, Francesca Sernissi, Stefano Lai, Riccardo Fava, Luca Tagliapietra, Luca Ascari, Roberto N. Izzo, and et al. 2023. "Telerehabilitation with ARC Intellicare to Cope with Motor and Respiratory Disabilities: Results about the Process, Usability, and Clinical Effect of the “Ricominciare” Pilot Study" Sensors 23, no. 16: 7238. https://doi.org/10.3390/s23167238
APA StyleCapecci, M., Cima, R., Barbini, F. A., Mantoan, A., Sernissi, F., Lai, S., Fava, R., Tagliapietra, L., Ascari, L., Izzo, R. N., Leombruni, M. E., Casoli, P., Hibel, M., & Ceravolo, M. G. (2023). Telerehabilitation with ARC Intellicare to Cope with Motor and Respiratory Disabilities: Results about the Process, Usability, and Clinical Effect of the “Ricominciare” Pilot Study. Sensors, 23(16), 7238. https://doi.org/10.3390/s23167238









