Skin Pigmentation Influence on Pulse Oximetry Accuracy: A Systematic Review and Bibliometric Analysis
Abstract
1. Introduction
- RQ1: What are the most significant publications and the ongoing research trends for prospect analysis on this topic?
- RQ2: How does skin color affect the accuracy of pulse oximeter devices incorporating photoplethysmography?
- RQ3: On which human populations have studies been conducted to verify these discrepancies and what methods have been employed to classify skin pigmentation?
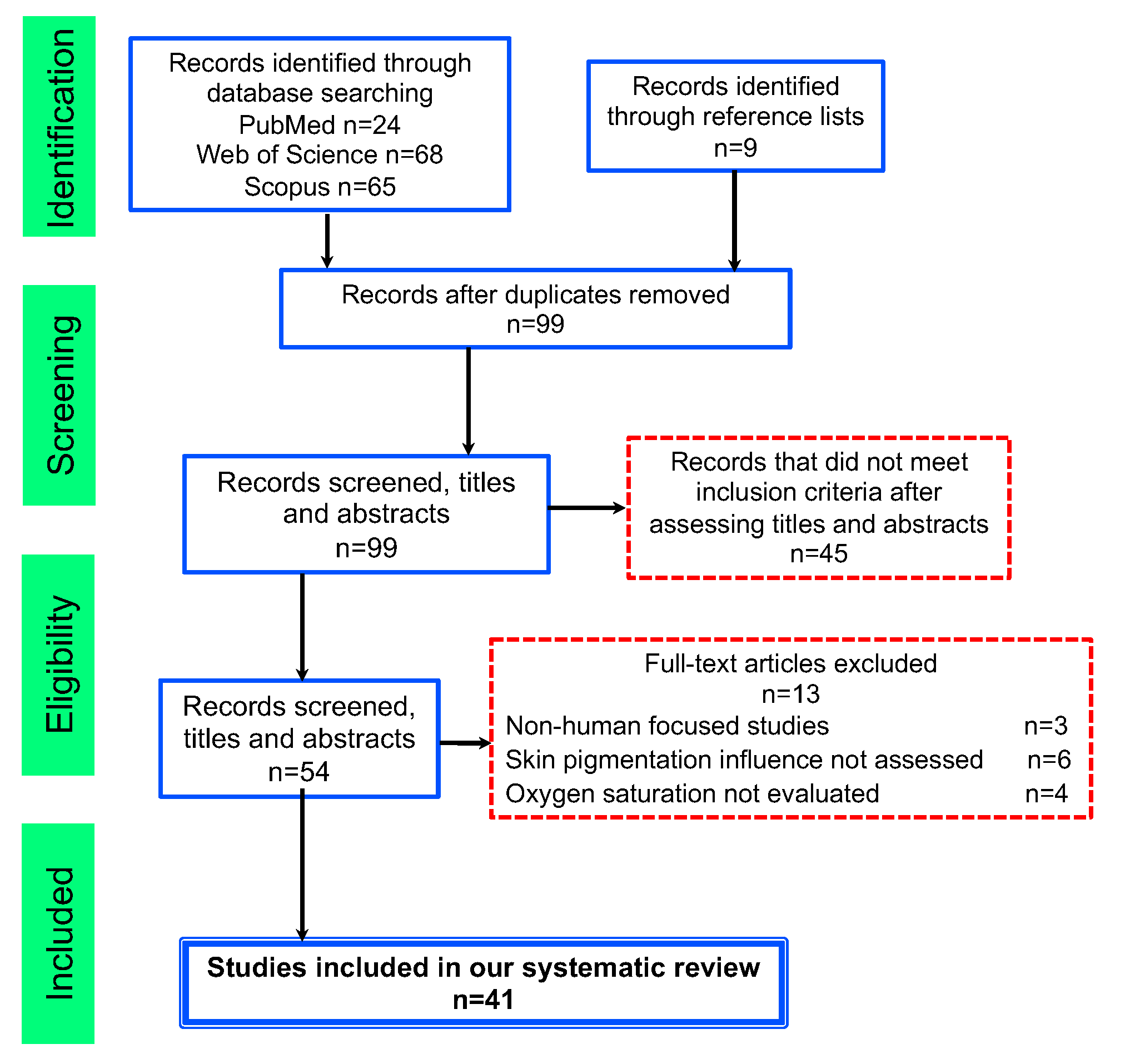
2. Methodology
2.1. Literature Search
- “oximetry” according to MeSH terms.
- “pulse oximetry” OR “oximet*” OR “oxygen saturation” to include all the relative references.
- “photoplethysmography” according to MeSH terms.
- “photoplethysmography” OR “PPG” as a generic term that refers to the optical imaging technique for detecting arterial pulsation.
- “skin” OR “pigmentation” OR “racial” OR “race” OR “ethnic*” to include all the relative references.
- “accuracy” OR “precision” OR “error” OR “reliability” OR “bias” to find all relative references.
2.2. Inclusion and Exclusion Criteria
- Non-human focused studies;
- Skin pigmentation influence not evaluated;
- References that do not focus on pulse oximetry.
2.3. Data Extraction and Analysis
3. Results
| Reference, Year | N | N Dark (%) | Age | Gender (Male/Female) | Type of Participants | Scale or Acenstry | Type of Oximeter |
|---|---|---|---|---|---|---|---|
| Henry N.R. et al. [53], 2022 | 26,603 | 2110 (7.93%) | 55–73 | 14,397/12,206 | ICU patients | Ancestry: Self-identified groups | High-fidelity pulse oximeter |
| Okunlola O.E. et al. [52], 2022 | 491 | 108 (22.00%) | – | – | Healthy adults under hypoxia conditions | Subjective scale: dark or light | 9 brands not especified |
| Shi C. et al. [61], 2022 | 6505 | – | 0–69 | – | Critically ill children, healthy adults, ICU patients, COVID-19 patients | Fitzpatrick phototype, Munsell scale Ancestry descriptors | – |
| Stell D. et al. [69], 2022 | 50 | 9 (18.00%) | 19–88 | – | Patients | Fitzpatrick phototype | Oxywatch MD300C19, Oxywatch MD300C29, PC- 60F Contec, CMS50D, and AM801 |
| Allado E. et al. [70], 2021 | 1045 | – | >18 | – | Pulmonary Patients | Fitzpatrick phototype | – |
| Al-Naji A. et al. [30], 2021 | 14 | – | – | 9/5 | Healthy adults, healthy babies | Subjective scale: dark or light | Rossmax, Model SA210 |
| Harskamp R.E. et al. [71], 2021 | 69 | 5 (7.25%) | 61–73 | 55/14 | ICU patients | Fitzpatrick phototype | Afac FS10D, Agptek FS10C, Anapulse ANP100, Cocobear, Contec CMS50D1, Hylogy MD-H37, F4PRO Mommed YM101, Pulox PO-200, Zacurate Pro 500DL |
| Mosooo A. et al. [72], 2021 | 25 | – | – | – | Healthy adults | Subjective scale: dark or light | – |
| Philip K.E.J. et al. [73], 2021 | 21 | – | – | – | – | – | – |
| Vesoulis Z. et al. [51], 2021 | 294 | 124 (42.18%) | 32–35 weeks | – | infants 32–35 weeks gestation | Ancestry: Asian-British, Black-British, White-British, White-Irish, White-other | Nellcor SpO2 Module with Neonatal-Adult MAX-N adhesive SpO2 sensor |
| Wiles M.D. et al. [17], 2021 | 194 | 53 (27.32%) | – | 140/154 | COVID-19 patients | Ancestry: White, Asian, Black | – |
| Sjoding M.W. et al. [50], 2020 | 10,001 | 1326 (13.26%) | – | – | ICU patients | Subjective scale: dark or light | – |
| Poets C.F. [74], 2019 | 2926 | 14 (0.48%) | – | – | Patients and infants with hypoxemia | Munsell | Nellcor N100, Nellcor N200, Nellcor Oximax, Nellcor N-600, Criticalcare 501, Kontron 7840, Radiometer OXI3, Masimo 4.0, Masimo SET, Novametrix 520, Radiometer Tosca/Masimo, Marquette Solar 8000, Masimo 7.9.1.0, Philips M1020A, Masimo 7.9.1.0, Ohmeda Biox III, Ohmeda Biox 3700, |
| Alharbi S. et al. [38], 2018 | 15 | – | 20–30 | – | Healthy adults | – | Optoelectronic patch sensor, and TempIR |
| Baek H.J. et al. [75], 2018 | 28 | 7 (25.00%) | – | – | Healthy adults | Ancestry: African American, Caucassian, Asian | Nellcor N-550 |
| Ebmeier S.J et al. [76], 2018 | 394 | 65 (16.09%) | 47–77 | 150/144 | ICU patients | Fitzpatrick phototype | Masimo, and Philips |
| Sanyal S. et al. [77], 2018 | 25 | 15 (60.00%) | 20–30 | 15/10 | Healthy adults | Fitzpatrick phototype | Biosync B-50DL |
| Foglia E.E. et al. [48], 2017 | 36 | 14 (40.00%) | 36–39 weeks | 21/15 | Infants with hypoxemia | Munsell | Masimo Radical 7, and Nellcor Oximax |
| Kumar M. et al. [78], 2015 | 12 | 4 (33.33%) | – | 7/5 | Healthy adults | Subjective scale: dark or light | Texas Instruments AFE4490SPO2EVM |
| Bensghir M. et al. [79], 2013 | 1 | 1 (100.00%) | 65 | 0/1 | with henna | – | – |
| Fallow B.A. et al. [80], 2013 | 23 | 10 (43.48%) | 19–43 | 11/12 | Healthy adults | Subjective scale: dark or light | – |
| Feiner J.R. et al. [24], 2007 | 36 | 24 (66.67%) | 19–44 | 19/17 | Healthy adults | Ancestry: African American, Hispanic Indian, Filipino, Vietnamese | Massimo Radical, Nellcor N-595, and Nonin 9700 |
| Bickler P.E. et al. [23], 2005 | 21 | 11 (52.38%) | 24–27 | – | Healthy adults | Ancestry: African American, Northern European | Nellcor N-595, Oximax-A prove, Novametrix 513, and Nonin Onyx |
| Hameedullah M.R. et al. [81], 2002 | 60 | 60 (100.00%) | 20–45 | 0/60 | Female with henna | – | Not reported |
| Wouters P.F. et al. [82], 2002 | — | – | – | – | ICU patients | – | – |
| Adler J.N. et al. [47], 1998 | 284 | 34 (11.97%) | 40-80 | 144/140 | ICU patients | Munsell scale | Nellcor D-25, and Hayward |
| Avant M.G. et al. [56], 1997 | 50 | 15 (30.00%) | – | – | Critically ill children | Subjective scale: dark or light | Nellcor oxiband, and Oximax Dura-Y |
| Bothma P.A. et al. [46], 1996 | 100 | 100 (100%) | – | – | Critically ill adults | Portable EEL reflectance spectrometer | Simed S100, Nihon Koden, and Ohmeda 3740 |
| Gaskin L. et al. [83], 1995 | 451 | – | – | – | Patients, healthy adults, athletes | – | Biox II, Biox 3700, Nellcor N200, Invivo 4500, Criticalcare 501, Ohmeda 3700, Novametrix 505, Datex, Kontron 7849, Helllige 4500, Nelccor N100, Minolta Pulse Ox7 |
| Al-Majed S.A. et al. [84], 1994 | 50 | 10 (20.00%) | – | – | Female with henna | – | – |
| Lee K.H. et al. [85], 1993 | 33 | 5 (15.15%) | 27–92 | – | ICU patients | Ancestry: Chinesse, Malay, Indian | Nellcor, Simed, and Critikon |
| Ralston A.C. et al. [86], 1991 | – | – | – | – | – | – | – |
| Zeballos R.J. et al. [87], 1991 | 33 | 33 (100.00%) | – | 33/0 | Healthy adults | Subjective scale: dark | HP 47201A, and Ohmeda Biox IIA |
| Jubran A. et al. [88], 1990 | 54 | 29 (53.70%) | 17–88 | 24/30 | ICU patients | Subjective scale: dark or light | Nellcor, and Ohmeda Biox3700 |
| Ries A.L. et al. [89], 1989 | 187 | 67 (35.83%) | – | – | Pulmonary patients | Munsell scale | Ohmeda Biox III, and HP 47201A |
| Mendelson Y. et al. [21], 1988 | 7 | – | 21–29 | 5/2 | Healthy adults | Ancestry: Caucassian | HP 47201 ear oximeter, and Nellcor |
| Cecil W.T. et al. [43], 1988 | 152 | 16 (10.53%) | 46–82 | 82/70 | Patients | Subjective scale: dark or light | Nellcor N-100, Ohmeda 3700 |
| Gabrielczyk M.R. et al. [45], 1988 | 21 | 4 (19.05%) | 47-68 | – | Patients | Subjective scale: dark or light | Nellcor N-100 |
| Emery J.R. et al. [42], 1987 | – | – | – | – | Premature Infants | Subjective scale: dark or light | – |
| Wang Y.T. et al. [44], 1985 | 31 | 31 (100.00%) | – | – | Patients | Subjective scale: dark or light | Ohmeda Biox III |
| Saunders N.A. et al. [41], 1976 | 52 | 5 (9.62%) | – | – | Healthy adults | Ancestry: African | Waters XP-350, Waters 0-lJOO, HP 47201A |
3.1. Characteristics of the Selected Studies
3.2. Assessment of Risk of Bias

4. Discussion
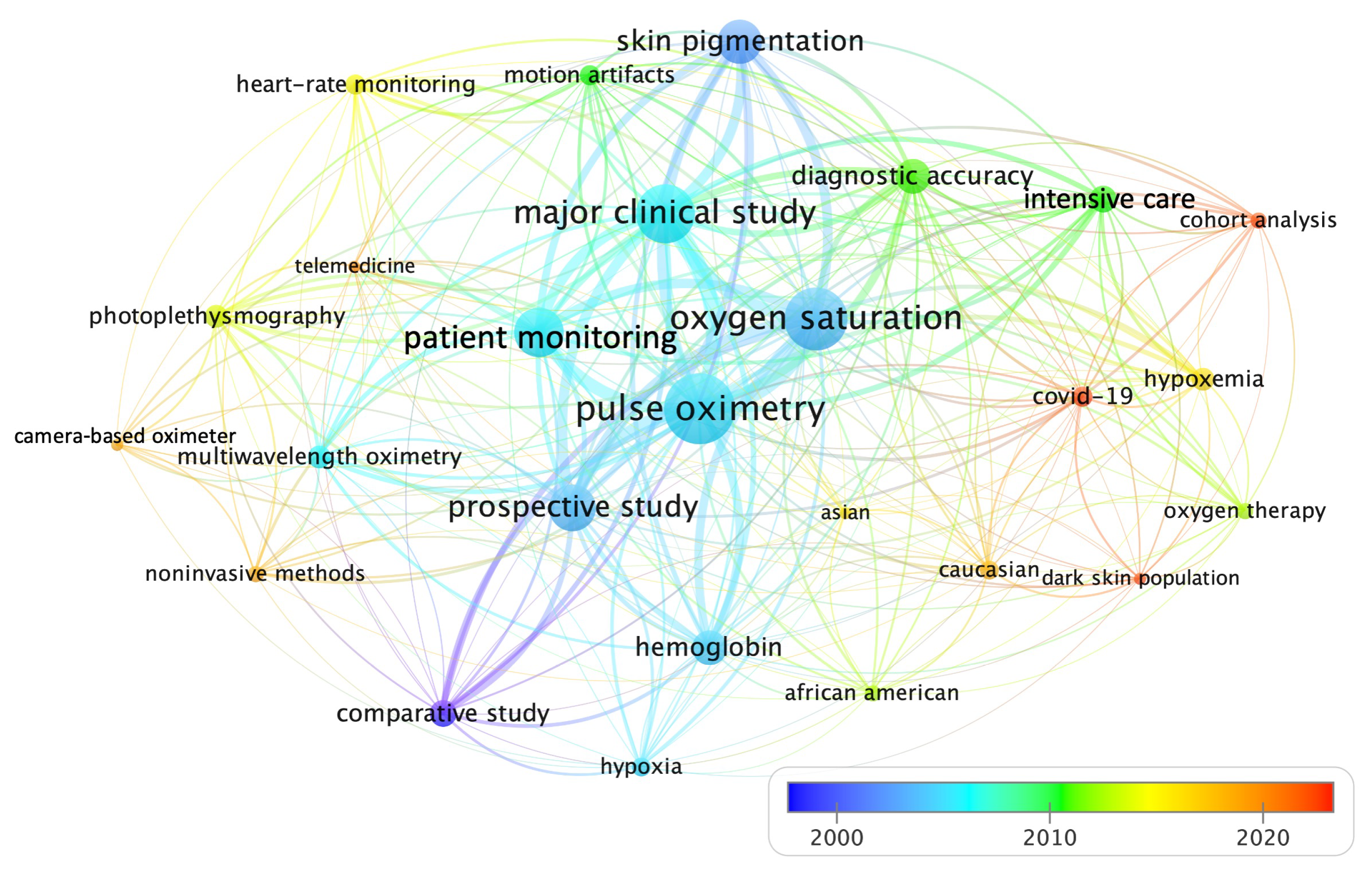
4.1. Rq1: What Are the Most Significant Publications and the Ongoing Research Trends for Prospect Analysis on This Topic?
4.2. Rq2: How Does Skin Color Affect the Accuracy of Pulse Oximeter Devices Incorporating Photoplethysmography?
4.3. Rq3: On Which Human Populations Have Studies Been Conducted to Verify These Discrepancies and What Methods Have Been Employed to Classify Skin Pigmentation?
5. Final Remarks
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Allen, J. Photoplethysmography and its application in clinical physiological measurement. Physiol. Meas. 2007, 28, R1–R39. [Google Scholar] [CrossRef] [PubMed]
- Alian, A.A.; Shelley, K.H. Best Practice & Research Clinical Anaesthesiology. Photoplethysmography 2014, 28, 395–406. [Google Scholar] [CrossRef]
- Nasseri, N.; Kleiser, S.; Wolf, U.; Wolf, M. Tissue oximetry by diffusive reflective visible light spectroscopy: Comparison of algorithms and their robustness. J. Biophoton. 2018, 11, e201700367. [Google Scholar] [CrossRef] [PubMed]
- Nitzan, M.; Romem, A.; Koppel, R. Pulse oximetry: Fundamentals and technology update. Med. Devices Evid. Res. 2014, 7, 231–239. [Google Scholar] [CrossRef]
- Jubran, A. Pulse oximetry. Crit. Care 2015, 19, 1–7. [Google Scholar] [CrossRef]
- Martín-Escudero, P.; Cabanas, A.M.; Fuentes-Ferrer, M.; Galindo-Canales, M. Oxygen Saturation Behavior by Pulse Oximetry in Female Athletes: Breaking Myths. Biosensors 2021, 11, 391. [Google Scholar] [CrossRef]
- Lee, I.; Park, N.; Lee, H.; Hwang, C.; Kim, J.H.; Park, S. Systematic Review on Human Skin-Compatible Wearable Photoplethysmography Sensors. Appl. Sci. 2021, 11, 2313. [Google Scholar] [CrossRef]
- Kim, N.H.; Yu, S.G.; Kim, S.E.; Lee, E.C. Non-Contact Oxygen Saturation Measurement Using YCgCr Color Space with an RGB Camera. Sensors 2021, 21, 6120. [Google Scholar] [CrossRef]
- Martín-Escudero, P. La oximetría en Registro Continuo en el Esfuerzo máximo en Distintas Especialidades Deportivas. Ph.D. Thesis, Universidad Complutense de Madrid, Madrid, Spain, 2003. [Google Scholar]
- Collins, J.A.; Rudenski, A.; Gibson, J.; Howard, L.; O’Driscoll, R. Relating oxygen partial pressure, saturation and content: The haemoglobin’oxygen dissociation curve. Breathe 2015, 11, 194–201. [Google Scholar] [CrossRef]
- Breuer, H.W.; Groeben, H.; Schöndeling, H.; Worth, H. Comparative analysis of arterial oxygen saturations during exercise by pulse oximetry, photometric measurements, and calculation procedures. Int. J. Sport. Med. 1990, 11, 22–25. [Google Scholar] [CrossRef]
- Scherrenberg, M.; Wilhelm, M.; Hansen, D.; Völler, H.; Cornelissen, V.; Frederix, I.; Kemps, H.; Dendale, P. The future is now: A call for action for cardiac telerehabilitation in the COVID-19 pandemic from the secondary prevention and rehabilitation section of the European Association of Preventive Cardiology. Eur. J. Prev. Cardiol. 2020, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Channa, A.; Popescu, N.; Skibinska, J.; Burget, R. The Rise of Wearable Devices during the COVID-19 Pandemic: A Systematic Review. Sensors 2021, 21, 5787. [Google Scholar] [CrossRef] [PubMed]
- Philip, K.E.J.; Bennett, B.; Fuller, S.; Lonergan, B.; McFadyen, C.; Burns, J.; Tidswell, R.; Vlachou, A. Working accuracy of pulse oximetry in COVID-19 patients stepping down from intensive care: A clinical evaluation. BMJ Open Respir. Res. 2020, 7, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Michard, F.; Shelley, K.; L’Her, E. COVID-19: Pulse oximeters in the spotlight. J. Clin. Monit. Comput. 2021, 35, 11–14. [Google Scholar] [CrossRef] [PubMed]
- England NHS. Specialty Guides for Patient Management during the Coronavirus Pandemic. Guidance for the Role and Use of Non-Invasive Respiratory Support in Adult Patients with COVID-19 (Confirmed or Suspected) 6 April 2020, Version 2. 2020. Available online: https://amhp.org.uk/app/uploads/2020/03/Guidance-Respiratory-Support.pdf (accessed on 26 March 2020).
- Wiles, M.D.; El-Nayal, A.; Elton, G.; Malaj, M.; Winterbottom, J.; Gillies, C.; Moppett, I.K.; Bauchmuller, K. The effect of patient ethnicity on the accuracy of peripheral pulse oximetry in patients with COVID-19 pneumonitis: A single-centre, retrospective analysis. Anaesthesia 2021, 77, 143–152. [Google Scholar] [CrossRef]
- Shah, S.; Majmudar, K.; Stein, A.; Gupta, N.; Suppes, S.; Karamanis, M.; Capannari, J.; Sethi, S.; Patte, C. Novel Use of Home Pulse Oximetry Monitoring in COVID-19 Patients Discharged From the Emergency Department Identifies Need for Hospitalization. Acad. Emerg. Med. 2020, 27, 681–692. [Google Scholar] [CrossRef]
- Nematswerani, N.; Collie, S.; Chen, T.; Cohen, M.; Champion, J.; Feldman, C.; Richards, G.A. The impact of routine pulse oximetry use on outcomes in COVID-19-infected patients at increased risk of severe disease: A retrospective cohort analysis. S. Afr. Med. J. Suid-Afr. Tydskr. Vir Geneeskd. 2021, 111, 950–956. [Google Scholar] [CrossRef]
- World Health Organization. COVID-19 Clinical Management: Living Guidance, 25 January 2021; Technical Documents; World Health Organization: Geneva, Switzerland, 2021. [Google Scholar]
- Mendelson, Y.; Ochs, B.D. Noninvasive pulse oximetry utilizing skin reflectance photoplethysmography. IEEE Trans. Biomed. Eng. 1988, 35, 798–805. [Google Scholar] [CrossRef]
- Abay, T.Y.; Kyriacou, P.A. Reflectance Photoplethysmography as Noninvasive Monitoring of Tissue Blood Perfusion. IEEE Trans. Biomed. Eng. 2015, 62, 2187–2195. [Google Scholar] [CrossRef]
- Bickler, P.E.; Feiner, J.R.; Severinghaus, J.W. Effects of skin pigmentation on pulse oximeter accuracy at low saturation. Anesthesiology 2005, 102, 715–719. [Google Scholar] [CrossRef]
- Feiner, J.R.; Severinghaus, J.W.; Bickler, P.E. Dark skin decreases the accuracy of pulse oximeters at low oxygen saturation: The effects of oximeter probe type and gender. Anesth. Analg. 2007, 105, S18–S23. [Google Scholar] [CrossRef] [PubMed]
- Sinex, J.E. Pulse oximetry: Principles and limitations. Am. J. Emerg. Med. 1999, 17, 59–67. [Google Scholar] [CrossRef]
- Dünnwald, T.; Kienast, R.; Niederseer, D.; Burtscher, M. The Use of Pulse Oximetry in the Assessment of Acclimatization to High Altitude. Sensors 2021, 21, 1263. [Google Scholar] [CrossRef] [PubMed]
- Tannheimer, M.; Lechner, R. The correct measurement of oxygen saturation at high altitude. Sleep Breath. Schlaf. Atm. 2019, 23, 1101–1106. [Google Scholar] [CrossRef]
- Sütçü Çiçek, H.; Gümüs, S.; Deniz, Ö.; Yildiz, S.; Açikel, C.H.; Çakir, E.; Tozkoparan, E.; Uçar, E.; Bilgiç, H. Effect of nail polish and henna on oxygen saturation determined by pulse oximetry in healthy young adult females. Emerg. Med. J. EMJ 2011, 28, 783–785. [Google Scholar] [CrossRef]
- Tobin, R.M.; Pologe, J.A.; Batchelder, P.B. A characterization of motion affecting pulse oximetry in 350 patients. Anesth. Analg. 2002, 94, S54–S61. [Google Scholar]
- Al-Naji, A.; Khalid, G.A.; Mahdi, J.F.; Chahl, J. Non-Contact SpO2 Prediction System Based on a Digital Camera. Appl. Sci. 2021, 11, 4255. [Google Scholar] [CrossRef]
- De Haan, G.; Van Leest, A. Improved motion robustness of remote-PPG by using the blood volume pulse signature. Physiol. Meas. 2014, 35, 1913–1926. [Google Scholar] [CrossRef]
- Van Gastel, M.; Stuijk, S.; De Haan, G. Motion robust remote-PPG in infrared. IEEE Trans. Biomed. Eng. 2015, 62, 1425–1433. [Google Scholar] [CrossRef]
- Colvonen, P.J. Response to: Investigating sources of inaccuracy in wearable optical heart rate sensors. NPJ Digit. Med. 2021, 4, 38. [Google Scholar] [CrossRef]
- Zonios, G.; Bykowski, J.; Kollias, N. Skin melanin, hemoglobin, and light scattering properties can be quantitatively assessed in vivo using diffuse reflectance spectroscopy. J. Investig. Dermatol. 2001, 117, 1452–1457. [Google Scholar] [CrossRef] [PubMed]
- Castaneda, D.; Esparza, A.; Ghamari, M.; Soltanpur, C.; Nazeran, H. A review on wearable photoplethysmography sensors and their potential future applications in health care. Int. J. Biosens. Bioelectron. 2018, 4, 195–202. [Google Scholar] [CrossRef] [PubMed]
- Yan, L.; Hu, S.; Alzahrani, A.; Alharbi, S.; Blanos, P. A Multi-Wavelength Opto-Electronic Patch Sensor to Effectively Detect Physiological Changes against Human Skin Types. Biosensors 2017, 7, 22. [Google Scholar] [CrossRef] [PubMed]
- Wassenaar, E.B.; Van den Brand, J.G. Reliability of near-infrared spectroscopy in people with dark skin pigmentation. J. Clin. Monit. Comput. 2005, 19, 195–199. [Google Scholar] [CrossRef]
- Alharbi, S.; Hu, S.; Mulvaney, D.; Blanos, P. An applicable approach for extracting human heart rate and oxygen saturation during physical movements using a multi-wavelength illumination optoelectronic sensor system. In Progress in Biomedical Optics and Imaging—Proceedings of SPIE; Raghavachari, R., Ed.; SPIE: Bellingham, WA, USA, 2018; Volume 10486. [Google Scholar] [CrossRef]
- Costa, J.; Vieira, H.; Louro, P.; Vieira, M. Double junction photodiodes for multiwavelength photoplethysmography. In Proceedings of SPIE—The International Society for Optical Engineering; Berghmans, F.M.A.G., Ed.; SPIE: Bellingham, WA, USA, 2018; Volume 10680. [Google Scholar] [CrossRef]
- Tamura, T.; Maeda, Y.; Sekine, M.; Yoshida, M. Wearable photoplethysmographic sensors—past and present. Electronics 2014, 3, 282–302. [Google Scholar] [CrossRef]
- Saunders, N.A.; Powles, A.C.P.; Rebuck, A.S. Ear oximetry: Accuracy and tracticability in the assessment of arterial oxygenation. Am. Rev. Respir. Dis. 1976, 113, 745–749. [Google Scholar]
- Emery, J.R. Skin pigmentation as an influence on the accuracy of pulse oximetry. J. Perinatol. Off. J. Calif. Perinat. Assoc. 1987, 7, 329–330. [Google Scholar]
- Cecil, W.T.; Thorpe, K.J.; Fibuch, E.E.; Tuohy, G.F. A clinical evaluation of the accuracy of the Nellcor N-100 and Ohmeda 3700 pulse oximeters. J. Clin. Monit. 1988, 4, 31–36. [Google Scholar] [CrossRef]
- Wang, Y.T.; Poh, S.C. Noninvasive oximetry in pigmented patients. Ann. Acad. Med. 1985, 14, 427–429. [Google Scholar]
- Gabrielczyk, M.R.; Buist, R.J. Pulse oximetry and postoperative hypothermia. Anaesthesia 1988, 43, 402–404. [Google Scholar] [CrossRef]
- Bothma, P.A.; Joynt, G.M.; Lipman, J.; Hon, H.; Mathala, B.; Scribante, J.; Kromberg, J. Accuracy of pulse oximetry in pigmented patients. S. Afr. Med. J. 1996, 86, 594–596. [Google Scholar] [PubMed]
- Adler, J.N.; Hughes, L.A.; Vivilecchia, R.; Camargo, C.A.J. Effect of skin pigmentation on pulse oximetry accuracy in the emergency department. Acad. Emerg. Med. Off. J. Soc. Acad. Emerg. Med. 1998, 5, 965–970. [Google Scholar] [CrossRef] [PubMed]
- Foglia, E.E.; Whyte, R.K.; Chaudhary, A.; Mott, A.; Chen, J.; Propert, K.J.; Schmidt, B. The Effect of Skin Pigmentation on the Accuracy of Pulse Oximetry in Infants with Hypoxemia. J. Pediatr. 2017, 182, 375–377.e2. [Google Scholar] [CrossRef] [PubMed]
- Bent, B.; Goldstein, B.A.; Kibbe, W.A.; Dunn, J.P. Investigating sources of inaccuracy in wearable optical heart rate sensors. NPJ Digit. Med. 2020, 3, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Sjoding, M.W.; Dickson, R.P.; Iwashyna, T.J.; Gay, S.E.; Valley, T.S. Racial Bias in Pulse Oximetry Measurement. N. Engl. J. Med. 2020, 383, 2477–2478. [Google Scholar] [CrossRef] [PubMed]
- Vesoulis, Z.; Tims, A.; Lodhi, H.; Lalos, N.; Whitehead, H. Racial discrepancy in pulse oximeter accuracy in preterm infants. J. Perinatol. 2021, 42, 79–85. [Google Scholar] [CrossRef]
- Okunlola, O.E.; Lipnick, M.S.; Batchelder, P.B.; Bernstein, M.; Feiner, J.R.; Bickler, P.E. Pulse Oximeter Performance, Racial Inequity, and the Work Ahead. Respir. Care 2022, 67, 252–257. [Google Scholar] [CrossRef]
- Henry, N.R.; Hanson, A.C.; Schulte, P.J.; Warner, N.S.; Manento, M.N.; Weister, T.J.; Warner, M.A. Disparities in Hypoxemia Detection by Pulse Oximetry Across Self-Identified Racial Groups and Associations with Clinical Outcomes. Crit. Care Med. 2022, 50, 204–211. [Google Scholar] [CrossRef]
- Wong, A.K.I.; Charpignon, M.; Kim, H.; Josef, C.; De Hond, A.A.; Fojas, J.J.; Tabaie, A.; Liu, X.; Mireles-Cabodevila, E.; Carvalho, L.; et al. Analysis of Discrepancies between Pulse Oximetry and Arterial Oxygen Saturation Measurements by Race and Ethnicity and Association with Organ Dysfunction and Mortality. JAMA Netw. Open 2021, 4, 1–14. [Google Scholar] [CrossRef]
- Valbuena, V.S.M.; Barbaro, R.P.; Claar, D.; Valley, T.S.; Dickson, R.P.; Gay, S.E.; Sjoding, M.W.; Iwashyna, T.J. Racial Bias in Pulse Oximetry Measurement Among Patients About to Undergo Extracorporeal Membrane Oxygenation in 2019–2020: A Retrospective Cohort Study. Chest 2021, 4, 971–978. [Google Scholar] [CrossRef]
- Avant, M. G; Lowe N.; Torres Jr., A. Comparison of accuracy and signal consistency of two reusable pulse oximeter probes in critically ill children. Respir. Care 1997, 42, 698–704. [Google Scholar]
- Warren, R.; Wyden, C.B. 2021.01.25 Letter to FDA re Bias in Pulse Oximetery Measurements; Technical Report; U.S. Food and Drug Administration: Silver Spring, MD, USA, 2021. [Google Scholar]
- NHS Race & Health Observatory. Pulse Oximetry and Racial Bias: Recommendations for National Healthcare, Regulatory and Research Bodies; Technical Report March; National Health Service England: London, UK, 2021. [Google Scholar]
- Hunasikatti, M. Racial bias in accuracy of pulse oximetry and its impact on assessments of hypopnea and T90 in clinical studies. J. Clin. Sleep Med. 2021, 17, 1145. [Google Scholar] [CrossRef] [PubMed]
- Holder, A.L.; Wong, A.-K.I. The Big Consequences of Small Discrepancies: Why Racial Differences in Pulse Oximetry Errors Matter. Crit. Care Med. 2022, 50, 335–337. [Google Scholar] [CrossRef]
- Shi, C.; Goodall, M.; Dumville, J.; Hill, J.E.; Norman, G.; Hamer, O.; Clegg, A.; Watkins, C.L.; Georgiou, G.; Alexander Hodkinson, A.; et al. Article The effects of skin pigmentation on the accuracy of pulse oximetry in measuring oxygen saturation: A systematic review and meta-analysis The effects of skin pigmentation on the accuracy of pulse and meta-analysis. PLoS Med. 2022. [Google Scholar] [CrossRef]
- Tranfield, D.; Denyer, D.; Smart, P. Towards a Methodology for Developing Evidence-Informed Management Knowledge by Means of Systematic Review* Introduction: The need for an evidence- informed approach. Br. J. Manag. 2003, 14, 207–222. [Google Scholar] [CrossRef]
- Ye, S.; Feng, S.; Huang, L.; Bian, S. Recent Progress in Wearable Biosensors: From Healthcare Monitoring to Sports Analytics. Biosensors 2020, 10, 205. [Google Scholar] [CrossRef]
- Pritišanac, E.; Urlesberger, B.; Schwaberger, B.; Pichler, G. Accuracy of pulse oximetry in the presence of fetal hemoglobin—A systematic. Children 2021, 8, 361. [Google Scholar] [CrossRef]
- Merigó, J.M.; Blanco-Mesa, F.; Gil-Lafuente, A.M.; Yager, R.R. Thirty Years of the International Journal of Intelligent Systems: A Bibliometric Review. Int. J. Intell. Syst. 2017, 32, 526–554. [Google Scholar] [CrossRef]
- Guerrero-Gironés, J.; Ros-Valverde, A.; Pecci-Lloret, M.P.; Rodríguez-Lozano, F.J.; Pecci-Lloret, M.R. Association between Pulpal-Periapical Pathology and Autoimmune Diseases: A Systematic Review. J. Clin. Med. 2021, 10, 4886. [Google Scholar] [CrossRef]
- Aria, M.; Cuccurullo, C. bibliometrix: An R-tool for comprehensive science mapping analysis. J. Inf. 2017, 11, 959–975. [Google Scholar] [CrossRef]
- Chen, C. CiteSpace II: Detecting and visualizing emerging trends and transient patterns in scientific literature. J. Am. Soc. Inf. Sci. Technol. 2006, 57, 359–377. [Google Scholar] [CrossRef]
- Stell, D.; Noble, J.J.; Kay, R.H.; Kwong, M.T.; Jeffryes, M.J.R.; Johnston, L.; Glover, G.; Akinluyi, E. Exploring the impact of pulse oximeter selection within the COVID-19 home-use pulse oximetry pathways. BMJ Open Respir. Res. 2022, 9, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Allado, E.; Poussel, M.; Moussu, A.; Saunier, V.; Bernard, Y.; Albuisson, E.; Chenuel, B. Innovative measurement of routine physiological variables (heart rate, respiratory rate and oxygen saturation) using a remote photoplethysmography imaging system: A prospective comparative trial protocol. BMJ Open 2021, 11, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Harskamp, R.E.; Bekker, L.; Himmelreich, J.C.L.; De Clercq, L.; Karregat, E.P.M.; Sleeswijk, M.E.; Lucassen, W.A.M. Performance of popular pulse oximeters compared with simultaneous arterial oxygen saturation or clinical-grade pulse oximetry: A cross-sectional validation study in intensive care patients. BMJ Open Respir. Res. 2021, 8, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Moço, A.; Verkruysse, W. Pulse oximetry based on photoplethysmography imaging with red and green light: Calibratability and challenges. J. Clin. Monit. Comput. 2021, 35, 123–133. [Google Scholar] [CrossRef] [PubMed]
- Philip, K.E.J.; Tidswell, R.; McFadyen, C. Racial bias in pulse oximetry: More statistical detail may help tackle the problem. BMJ 2021, 372, n298. [Google Scholar] [CrossRef]
- Poets, C.F. Noninvasive Monitoring and Assessment of Oxygenation in Infants. Clin. Perinatol. 2019, 46, 417–433. [Google Scholar] [CrossRef]
- Baek, H.J.; Shin, J.; Cho, J. The Effect of Optical Crosstalk on Accuracy of Reflectance-Type Pulse Oximeter for Mobile Healthcare. J. Healthc. Eng. 2018, 2018, 3521738. [Google Scholar] [CrossRef]
- Ebmeier, S.J.; Barker, M.; Bacon, M.; Beasley, R.C.; Bellomo, R.; Chong, C.K.; Eastwood, G.M.; Gilchrist, J.; Kagaya, H.; Pilcher, J.; et al. A Two Centre Observational Study of Simultaneous Pulse Oximetry and Arterial Oxygen Saturation Recordings in Intensive Care Unit Patients. Anaesth. Intensive Care 2018, 46, 297–303. [Google Scholar] [CrossRef]
- Sanyal, S.; Nundy, K.K. Algorithms for Monitoring Heart Rate and Respiratory Rate From the Video of a User’s Face. IEEE J. Transl. Eng. Health Med. 2018, 6, 1–11. [Google Scholar] [CrossRef]
- Kumar, M.; Veeraraghavan, A.; Sabharwal, A. DistancePPG: Robust non-contact vital signs monitoring using a camera. Biomed. Opt. Express 2015, 6, 1565–1588. [Google Scholar] [CrossRef] [PubMed]
- Bensghir, M.; Houba, A.; El Hila, J.; Ahtil, R.; Azendour, H.; Kamili, N.D. Henna dye: A cause of erroneous pulse oximetry readings. Saudi J. Anaesth. 2013, 7, 474–475. [Google Scholar] [CrossRef] [PubMed]
- Fallow, B.A.; Tarumi, T.; Tanaka, H. Influence of skin type and wavelength on light wave reflectance. J. Clin. Monit. Comput. 2013, 27, 313–317. [Google Scholar] [CrossRef] [PubMed]
- Hameedullah; Rauf, M.A.; Khan, F.A. Henna paste and pulse oximetry: Effect of different methods of application. J. Anaesthesiol. Clin. Pharmacol. 2002, 18, 193–196. [Google Scholar]
- Wouters, P.F.; Gehring, H.; Meyfroidt, G.; Ponz, L.; Gil-Rodriguez, J.; Hornberger, C.; Bonk, R.; Frankenberger, H.; Benekos, K.; Valais, J.; et al. Accuracy of pulse oximeters: The European multi-center trial. Anesth. Analg. 2002, 94, S13-6. [Google Scholar]
- Gaskin, L.; Thomas, J. Pulse Oximetry and Exercise. Physiotherapy 1995, 81, 254–261. [Google Scholar] [CrossRef]
- Al-Majed, S.A.; Harakati, M.S. The effect of henna paste on oxygen saturation reading obtained by pulse oximetry. Trop. Geogr. Med. 1994, 46, 38–39. [Google Scholar]
- Lee, K.H.; Hui, K.P.; Tan, W.C.; Lim, T.K. Factors influencing pulse oximetry as compared to functional arterial saturation in multi-ethnic Singapore. Singap. Med. J. 1993, 34, 385–387. [Google Scholar]
- Ralston, A.C.; Webb, R.K.; Runciman, W.B. Potential errors in pulse oximetry. I. Pulse oximeter evaluation. Anaesthesia 1991, 46, 202–206. [Google Scholar] [CrossRef]
- Zeballos, R.J.; Weisman, I.M. Reliability of noninvasive oximetry in black subjects during exercise and hypoxia. Am. Rev. Respir. Dis. 1991, 144, 1240–1244. [Google Scholar] [CrossRef]
- Jubran, A.; Tobin, M. Reliability of pulse oximetry in titrating supplemental oxygen therapy in ventilator-dependent patients. Chest 1990, 97, 1420–1425. [Google Scholar] [CrossRef] [PubMed]
- Ries, A.L.; Prewitt, L.M.; Johnson, J.J. Skin color and ear oximetry. Chest 1989, 96, 287–290. [Google Scholar] [CrossRef]
- Whiting, P.F.; Rutjes, A.W.S.; Westwood, M.E.; Mallett, S.; Deeks, J.J.; Reitsma, J.B.; Leeflang, M.M.G.; Sterne, J.A.C.; Bossuyt, P.M.M. QUADAS-2: A revised tool for the quality assessment of diagnostic accuracy studies. Ann. Intern. Med. 2011, 155, 529–536. [Google Scholar] [CrossRef] [PubMed]
- Whiting, D.P. Risk of Bias and Applicability Judgments. Available online: https://www.bristol.ac.uk/population-health-sciences/projects/quadas/quadas-2/ (accessed on 26 March 2020).
- McGuinness, L.A.; Higgins, J.P.T. Risk-of-bias VISualization (robvis): An R package and Shiny web app for visualizing risk-of-bias assessments. Res. Synth. Methods 2020, 12, 55–61. [Google Scholar] [CrossRef] [PubMed]
- Lee, T.Q.; Barnett, S.L.; Shanfield, S.L.; Anzel, S.H. Potential application of photoplethysmography technique in evaluating microcirculatory status of STAMP patients: Preliminary report. J. Rehabil. Res. Dev. 1990, 27, 363–368. [Google Scholar] [CrossRef] [PubMed]
- Khanam, F.T.Z.; Perera, A.G.; Al-Naji, A.; Gibson, K.; Chahl, J. Non-Contact Automatic Vital Signs Monitoring of Infants in a Neonatal Intensive Care Unit Based on Neural Networks. J. Imaging 2021, 7, 122. [Google Scholar] [CrossRef]
- Liu, H.; Ivanov, K.; Wang, Y.; Wang, L. A novel method based on two cameras for accurate estimation of arterial oxygen saturation. Biomed. Eng. Online 2015, 14, 52. [Google Scholar] [CrossRef]
- Guazzi, A.R.; Villarroel, M.; Jorge, J.; Daly, J.; Frise, M.C.; Robbins, P.A.; Tarassenko, L. Non-contact measurement of oxygen saturation with an RGB camera. Biomed. Opt. Express 2015, 6, 3320–3338. [Google Scholar] [CrossRef]
- Song, Y.; Chen, X.; Hao, T.; Liu, Z.; Lan, Z. Exploring two decades of research on classroom dialogue by using bibliometric analysis. Comput. Educ. 2019, 137, 12–31. [Google Scholar] [CrossRef]
- van Eck, N.J.; Waltman, L. Software survey: VOSviewer, a computer program for bibliometric mapping. Scientometrics 2010, 84, 523–538. [Google Scholar] [CrossRef]
- Wosik, J.; Fudim, M.; Cameron, B.; Gellad, Z.F.; Cho, A.; Phinney, D.; Curtis, S.; Roman, M.; Poon, E.G.; Ferranti, J.; et al. Telehealth transformation: COVID-19 and the rise of virtual care. J. Am. Med. Inform. Assoc. Jamia 2020, 27, 957–962. [Google Scholar] [CrossRef] [PubMed]
- van Gastel, M.; Stuijk, S.; de Haan, G. New principle for measuring arterial blood oxygenation, enabling motion-robust remote monitoring. Sci. Rep. 2016, 6, 38609. [Google Scholar] [CrossRef] [PubMed]
- Fine, J.; Branan, K.L.; Rodriguez, A.J.; Boonya-ananta, T.; Ajmal; Ramella-Roman, J.C.; McShane, M.J.; Coté, G.L. Sources of Inaccuracy in Photoplethysmography for Continuous Cardiovascular Monitoring. Biosensors 2021, 11, 126. [Google Scholar] [CrossRef] [PubMed]
- Maeda, Y.; Sekine, M.; Tamura, T. The advantages of wearable green reflected photoplethysmography. J. Med. Syst. 2011, 35, 829–834. [Google Scholar] [CrossRef]
- de Kock, J.P.; Reynolds, K.J.; Tarassenko, L.; Moyle, J.T. The effect of varying LED intensity on pulse oximeter accuracy. J. Med. Eng. Technol. 1991, 15, 111–115. [Google Scholar] [CrossRef]
- Bossuyt, P.M.; Reitsma, J.B.; Bruns, D.E.; Gatsonis, C.A.; Glasziou, P.P.; Irwig, L.; Lijmer, J.G.; Moher, D.; Rennie, D.; de Vet, H.C.W.; et al. STARD 2015: An updated list of essential items for reporting diagnostic accuracy studies. BMJ 2015, 351, h5527. [Google Scholar] [CrossRef]
- Fitzpatrick, T.B.; Breathnach, A.S. The epidermal melanin unit system. Dermatol. Wochenschr. 1963, 147, 481–489. [Google Scholar]
- Fitzpatrick, T.B. The validity and practicality of sun-reactive skin types I through VI. Arch. Dermatol. 1988, 124, 869–871. [Google Scholar] [CrossRef]
- Moreiras, H.; O’Connor, C.; Bell, M.; Tobin, D.J. Visible light and human skin pigmentation: The importance of skin phototype. Exp. Dermatol. 2021, 30, 1324–1331. [Google Scholar] [CrossRef]
- Munsell, A.H. Atlas of the Munsell Color System; Wadsworth, Howland & Co., Inc., Printers: Malden, MA, USA, 1915. [Google Scholar]
- Xiao, K.; Yates, J.M.; Zardawi, F.; Sueeprasan, S.; Liao, N.; Gill, L.; Li, C.; Wuerger, S. Characterising the variations in ethnic skin colours: A new calibrated data base for human skin. Skin Res. Technol. 2017, 23, 21–29. [Google Scholar] [CrossRef]
- Kugelman, A.; Wasserman, Y.; Mor, F.; Goldinov, L.; Geller, Y.; Bader, D. Reflectance Pulse Oximetry from Core Body in Neonates and Infants: Comparison to Arterial Blood Oxygen Saturation and to Transmission Pulse Oximetry. J. Perinatol. 2004, 24, 366–371. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hay, W.W.J.; Brockway, J.M.; Eyzaguirre, M. Neonatal pulse oximetry: Accuracy and reliability. Pediatrics 1989, 83, 717–722. [Google Scholar] [CrossRef] [PubMed]
- Luks, A.M.; Swenson, E.R. Pulse oximetry for monitoring patients with COVID-19 at home potential pitfalls and practical guidance. Ann. Am. Thorac. Soc. 2020, 17, 1040–1046. [Google Scholar] [CrossRef] [PubMed]

| Indexing Terms | Items (n) |
|---|---|
| Web of Science | |
| #1 “oximet*” OR “oxygen saturation” | 68,211 |
| #2 “skin*” OR “pigmentation” OR “racial” OR “race” OR “ethnic” | 2,277,504 |
| #3 “photoplethysmography” OR PPG | 7838 |
| #4 “accuracy” OR “precision” OR “error” OR “reliability” OR “bias” | 6,062,294 |
| #5 #1 AND #2 AND #3 AND #4 | 74 |
| PubMed | |
| #1 “oximetry” [MeSH Terms] | 16,178 |
| #2 “oximet*” OR “oxygen saturation” | 44,457 |
| #3 “skin*” OR “pigmentation” OR “racial” OR “race” OR “ethnic” | 1,148,844 |
| #4 “photoplethysmography” [MeSH Terms] | 2422 |
| #5 “photoplethysmography” OR “PPG” | 7241 |
| #6 ”accuracy“ OR ”precision“ OR ”error” OR “reliability” OR “bias” | 1,835,255 |
| #7 #2 AND #3 AND #5 AND #6 | 24 |
| Scopus | |
| #1 “oximet*” OR “oxygen saturation” | 99,915 |
| #2 “skin*” OR “pigmentation” OR “racial” OR “race” OR “ethnic” | 2,038,888 |
| #3 “photoplethysmography” OR “PPG” | 14,492 |
| #4 “accuracy” OR “precision” OR “error” OR “reliability” OR “bias” | 5,911,553 |
| #5 #1 AND #2 AND #3 AND #4 | 72 |
| Description | Results |
|---|---|
| Timespan | 1976–2022 |
| Sources (Journals, Books, etc.) | 31 |
| Documents | 41 |
| Average citations per documents | 39.95 |
| Average citations per year per doc | 3.67 |
| References | 696 |
| Articles | 34 |
| Conference papers | 1 |
| Letter | 3 |
| Review | 3 |
| Keywords | 433 |
| Authors | 171 |
| Single-authored documents | 3 |
| Documents per Author | 0.23 |
| Authors per Document | 4.28 |
| Co-Authors per Documents | 4.40 |
| Items | Summary Statistics (n (%)) |
|---|---|
| Type of study (41 studies) | |
| Prospective | 32 (78.05%) |
| Retrospective | 2 (4.88%) |
| Case Report | 1 (2.44%) |
| Review | 3 (7.32%) |
| Letter | 3 (7.32%) |
| Experimental setting (35 studies) | |
| Hospital | 20 (57.14%) |
| Laboratory | 15 (42.86%) |
| Types of participants (39 studies) | |
| Infants | |
| <32 weeks gestation | 3 (7.69%) |
| infants with hypoxemia | 1 (2.56%) |
| Children | |
| Healthy babies | 1 (2.56%) |
| Critically ill | 1 (2.56%) |
| Adults | |
| Healthy adults | 13 (33.33%) |
| ICU Patients | 7 (17.95%) |
| Pulmonary patients | 2 (5.28%) |
| COVID-19 patients | 1 (2.56%) |
| Females with henna | 3 (7.69%) |
| Other patients | 6 (15.38%) |
| Athletes | 1 (2.56%) |
| Skin pigmentation classification (37 studies) | |
| Fitzpatrick phototype | 6 (16.21%) |
| Munsell scale | 4 (10.81%) |
| Ancestry | 11 (29.73%) |
| Subjective scale | 13 (35.14%) |
| Black or red henna | 2 (5.41%) |
| Portable reflectance spectrophotometer | 1 (2.70%) |
| Paper | TC | per Year | LC | LC/TC% |
|---|---|---|---|---|
| Kumar M. et al., 2015 [78] | 236 | 29.5 | 1 | 0.42 |
| Mendelson Y. et al., 1988 [21] | 181 | 5.17 | 1 | 0.55 |
| Ralston A.C. et al., 1991 [86] | 149 | 4.66 | 1 | 0.67 |
| Bickler P.E. et al., 2005 [23] | 131 | 7.28 | 10 | 7.63 |
| Feiner J.R. et al., 2007 [24] | 124 | 7.75 | 6 | 4.84 |
| Jubran A. et al., 1990 [88] | 117 | 3.55 | 5 | 4.84 |
| Sjoding M.W. et al., 2020 [50] | 111 | 37.00 | 6 | 5.41 |
| Saunders N.A. et al., 1976 [41] | 63 | 1.34 | 0 | 0.0 |
| Ries A.L. et al., 1989 [89] | 61 | 1.79 | 6 | 9.84 |
| Fallow B.A. et al., 2013 [80] | 58 | 5.80 | 0 | 0.0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cabanas, A.M.; Fuentes-Guajardo, M.; Latorre, K.; León, D.; Martín-Escudero, P. Skin Pigmentation Influence on Pulse Oximetry Accuracy: A Systematic Review and Bibliometric Analysis. Sensors 2022, 22, 3402. https://doi.org/10.3390/s22093402
Cabanas AM, Fuentes-Guajardo M, Latorre K, León D, Martín-Escudero P. Skin Pigmentation Influence on Pulse Oximetry Accuracy: A Systematic Review and Bibliometric Analysis. Sensors. 2022; 22(9):3402. https://doi.org/10.3390/s22093402
Chicago/Turabian StyleCabanas, Ana M., Macarena Fuentes-Guajardo, Katina Latorre, Dayneri León, and Pilar Martín-Escudero. 2022. "Skin Pigmentation Influence on Pulse Oximetry Accuracy: A Systematic Review and Bibliometric Analysis" Sensors 22, no. 9: 3402. https://doi.org/10.3390/s22093402
APA StyleCabanas, A. M., Fuentes-Guajardo, M., Latorre, K., León, D., & Martín-Escudero, P. (2022). Skin Pigmentation Influence on Pulse Oximetry Accuracy: A Systematic Review and Bibliometric Analysis. Sensors, 22(9), 3402. https://doi.org/10.3390/s22093402






