Wearable Devices for Remote Monitoring of Heart Rate and Heart Rate Variability—What We Know and What Is Coming
Abstract
1. Introduction
2. Electrical Cardiac Signal Generation, Recording
3. Heart Rate and Heart Rate Variability
4. Analysis of Heart Rate and Heart Rate Variability
5. Development of Ambulatory and Remote Heart Rate/Rhythm Monitoring Technology
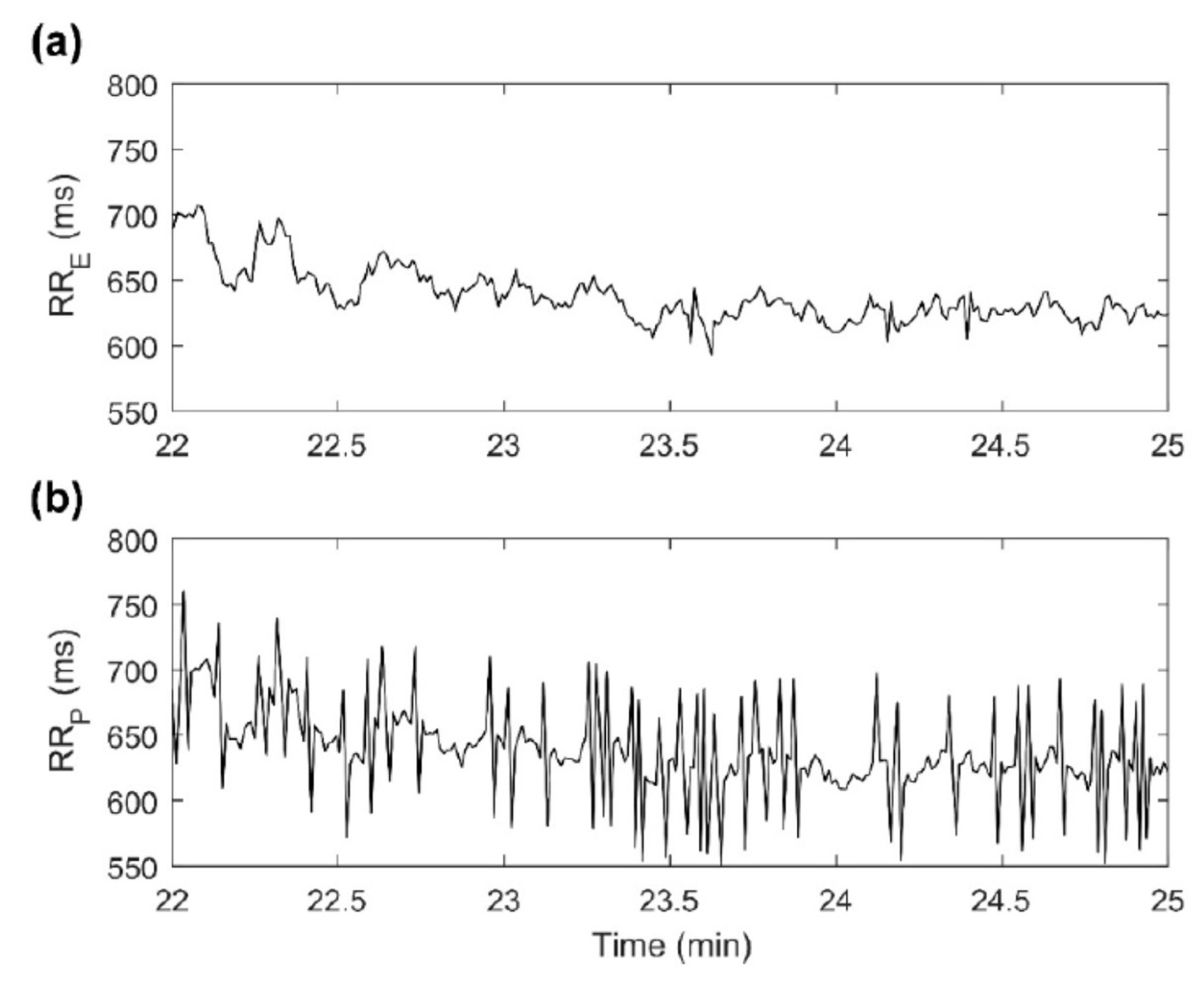
6. Sensors Used in Wearable HR/HRV Monitors
6.1. Photo Plethysmography (PPG)
6.2. ECG Based Sensors
6.3. Other Sensors
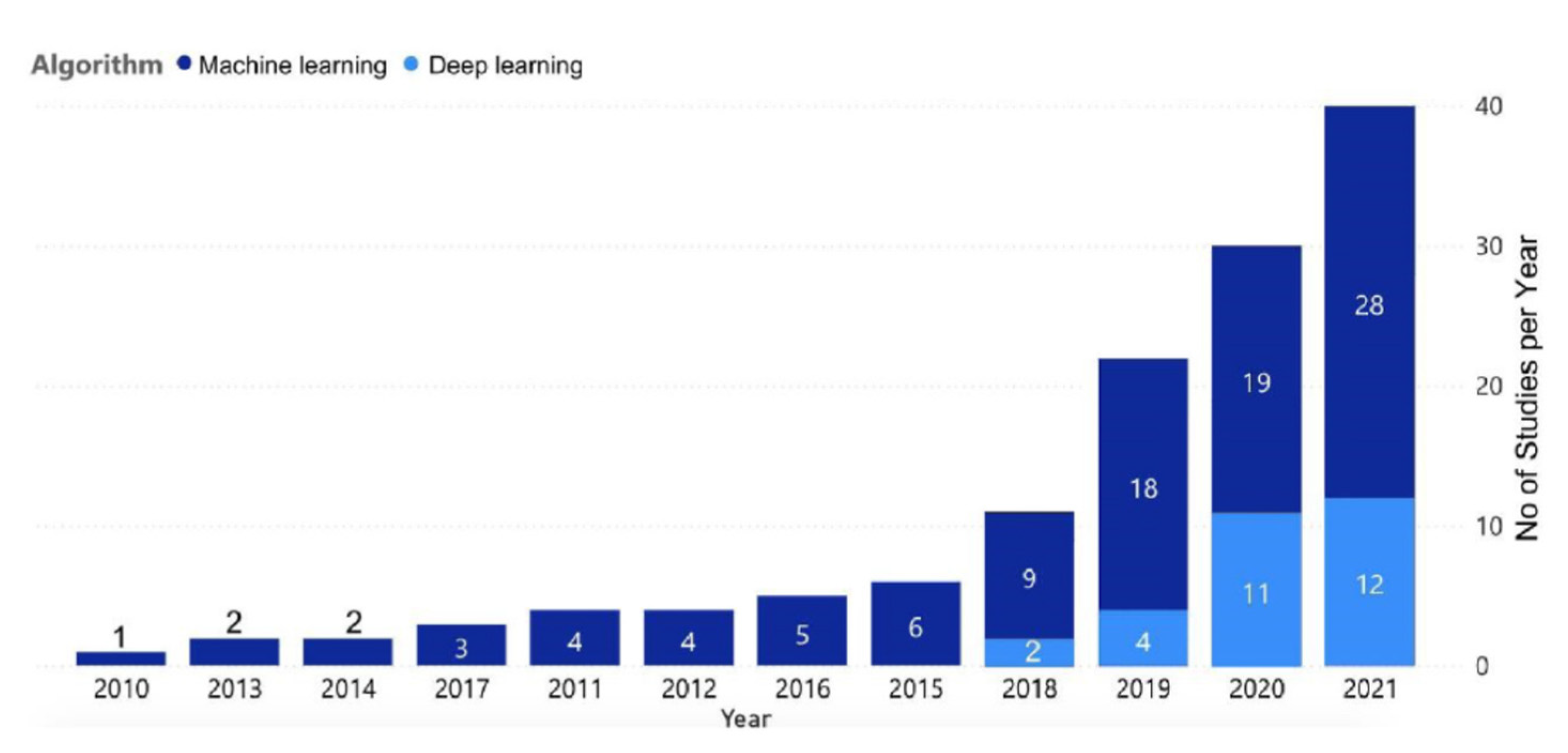
7. Data Processing and Analysis, Use of Machine Learning
8. Consumer Grade Wearable Devices for Heart Rate and Heart Rate Monitoring
9. Diagnostic Uses of Ambulatory Heart Rate and Heart Rate Variability Monitoring
9.1. Cardiology
9.2. Sleep Medicine
9.3. Diabetes Detection and Management
9.4. Other Uses
10. Challenges
11. Future Directions
12. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Bayoumy, K.; Gaber, M.; Elshafeey, A.; Mhaimeed, O.; Dineen, E.H.; Marvel, F.A.; Martin, S.S.; Muse, E.D.; Turakhia, M.P.; Tarakji, K.G.; et al. Smart Wearable Devices in Cardiovascular Care: Where We Are and How to Move Forward. Nat. Rev. Cardiol. 2021, 18, 581–599. [Google Scholar] [CrossRef]
- Soon, S.; Svavarsdottir, H.; Downey, C.; Jayne, D.G. Wearable Devices for Remote Vital Signs Monitoring in the Outpatient Setting: An Overview of the Field. BMJ Innov. 2020, 6, 55–71. [Google Scholar] [CrossRef]
- Gordan, R.; Gwathmey, J.K.; Xie, L.H. Autonomic and Endocrine Control of Cardiovascular Function. World J. Cardiol. 2015, 7, 204–214. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Wang, W.; Li, F. Association between Resting Heart Rate and Coronary Artery Disease, Stroke, Sudden Death and Noncardiovascular Diseases: A Meta-Analysis. CMAJ 2016, 188, E384–E392. [Google Scholar] [CrossRef] [PubMed]
- Singh, N.; Moneghetti, K.J.; Christle, J.W.; Hadley, D.; Plews, D.; Froelicher, V. Heart Rate Variability: An Old Metric with New Meaning in the Era of Using MHealth Technologies for Health and Exercise Training Guidance. Part One: Physiology and Methods. Arrhythm. Electrophysiol. Rev. 2018, 7, 193–198. [Google Scholar] [CrossRef] [PubMed]
- Fox, K.; Ford, I.; Steg, P.G.; Tendera, M.; Robertson, M.; Ferrari, R.; BEAUTIFUL Investigators. Heart Rate as a Prognostic Risk Factor in Patients with Coronary Artery Disease and Left-Ventricular Systolic Dysfunction (BEAUTIFUL): A Subgroup Analysis of a Randomised Controlled Trial. Lancet 2008, 372, 817–821. [Google Scholar] [CrossRef]
- Attia, Z.I.; Noseworthy, P.A.; Lopez-Jimenez, F.; Asirvatham, S.J.; Deshmukh, A.J.; Gersh, B.J.; Carter, R.E.; Yao, X.; Rabinstein, A.A.; Erickson, B.J.; et al. An Artificial Intelligence-Enabled ECG Algorithm for the Identification of Patients with Atrial Fibrillation during Sinus Rhythm: A Retrospective Analysis of Outcome Prediction. Lancet 2019, 394, 861–867. [Google Scholar] [CrossRef]
- Yan, B.P.; Lai, W.H.S.; Chan, C.K.Y.; Chan, S.C.; Chan, L.; Lam, K.; Lau, H.; Ng, C.; Tai, L.; Yip, K.; et al. Contact-Free Screening of Atrial Fibrillation by a Smartphone Using Facial Pulsatile Photoplethysmographic Signals. J. Am. Heart. Assoc. 2018, 7, e008585. [Google Scholar] [CrossRef] [PubMed]
- Shaffer, F.; Ginsberg, J.P. An Overview of Heart Rate Variability Metrics and Norms. Front. Public Health 2017, 258. [Google Scholar] [CrossRef]
- Task Force of the European Society of Cardiology the North American Society of Pacing Electrophysiology Task Force of the European Society of Cardiology and the North American Society of Pacing And. Circulation 1996, 93, 1043–1065.
- Laborde, S.; Mosley, E.; Thayer, J.F. Heart Rate Variability and Cardiac Vagal Tone in Psychophysiological Research-Recommendations for Experiment Planning, Data Analysis, and Data Reporting. Front. Psychol. 2017, 8, 213. [Google Scholar] [CrossRef] [PubMed]
- Bauer, A.; Kantelhardt, J.W.; Barthel, P.; Schneider, R.; Mäkikallio, T.; Ulm, K.; Hnatkova, K.; Schömig, A.; Huikuri, H.; Bunde, A.; et al. Deceleration Capacity of Heart Rate as a Predictor of Mortality after Myocardial Infarction: Cohort Study. Lancet 2006, 367, 1674–1681. [Google Scholar] [CrossRef]
- Schmidt, G.; Malik, M.; Barthel, P.; Schneider, R.; Ulm, K.; Rolnitzky, L.; Camm, A.J.; JT Jr, B.; Schömig, A. Heart-Rate Turbulence after Ventricular Premature Beats as a Predictor of Mortality after Acute Myocardial Infarction. Lancet 1999, 353, 1390–1396. [Google Scholar] [CrossRef]
- Grimm, W.; Schmidt, G.; Maisch, B.; Sharkova, J.; Müller, H.H.; Christ, M. Prognostic Significance of Heart Rate Turbulence Following Ventricular Premature Beats in Patients with Idiopathic Dilated Cardiomyopathy. J. Cardiovasc. Electrophysiol. 2003, 14, 819–824. [Google Scholar] [CrossRef] [PubMed]
- Holter, N.J.; Generelli, J.A. Remote recording of physiological data by radio. Rocky Mt Med. J. 1949, 46, 747–751. [Google Scholar] [PubMed]
- Holter, N.J. New Method for Heart Studies. Science 1961, 134, 1214–1220. [Google Scholar] [CrossRef] [PubMed]
- Stern, S.; Tzivoni, D. Early Detection of Silent Ischaemic Heart Disease by 24-Hour Electrocardiographic Monitoring of Active Subjects. Br. Heart J. 1974, 36, 481–486. [Google Scholar] [CrossRef]
- Mueller, W.C. Arrhythmia Detection Program for an Ambulatory ECG Monitor. Biomed Sci. Instrum. 1978, 14, 81–85. [Google Scholar]
- Sheffield, L.T. Computer-Aided Electrocardiography. J. Am. Coll. Cardiol. 1987, 10, 448–455. [Google Scholar] [CrossRef][Green Version]
- Heilbron, E.L. Advances in Modern Electrocardiographic Equipment for Long-Term Ambulatory Monitoring. Card Electrophysiol. Rev. 2002, 6, 185–189. [Google Scholar] [CrossRef]
- Trigo, J.D.; Alesanco, A.; Martínez, I.; García, J. A Review on Digital ECG Formats and the Relationships between Them. IEEE Trans. Inf. Technol. Biomed. 2012, 16, 432–444. [Google Scholar] [CrossRef] [PubMed]
- Cock, C.C. The Implantable Loop Recorder: A Tool That Is “Here to Stay.”. Indian Pacing Electrophysiol. J. 2002, 2, 15–19. [Google Scholar]
- Faust, O.; Hong, W.; Loh, H.W.; Xu, S.; Tan, R.-S.; Chakraborty, S.; Barua, P.D.; Molinari, F.; Acharya, U.R. Heart Rate Variability for Medical Decision Support Systems: A Review. Comput. Biol. Med. 2022, 145, 105407. [Google Scholar] [CrossRef]
- Bellet, S.; Eliakim, M.; Deliyiannis, S.; Figallo, E.M. Radioelectrocardiographic Changes during Strenuous Exercise in Normal Subjects. Circulation 1962, 25, 686–694. [Google Scholar] [CrossRef]
- Romero, I. Ambulatory electrocardiology. Cardiol. Rev. 2013, 21, 239–248. [Google Scholar] [CrossRef] [PubMed]
- Barr, N.L.; Voas, R.B. Telemetering physiologic responses during experimental flights. Am. J. Cardiol. 1960, 6, 54–61. [Google Scholar] [CrossRef]
- Nelson, B.W.; Allen, N.B. Accuracy of Consumer Wearable Heart Rate Measurement During an Ecologically Valid 24-Hour Period: Intraindividual Validation Study. JMIR Mhealth Uhealth 2019, 7, 10828. [Google Scholar] [CrossRef]
- Etiwy, M.; Akhrass, Z.; Gillinov, L.; Alashi, A.; Wang, R.; Blackburn, G.; Gillinov, S.M.; Phelan, D.; Gillinov, A.M.; Houghtaling, P.L.; et al. Accuracy of Wearable Heart Rate Monitors in Cardiac Rehabilitation. Cardiovasc. Diagn. Ther. 2019, 9, 262–271. [Google Scholar] [CrossRef]
- Dagher, L.; Shi, H.; Zhao, Y.; Marrouche, N.F. Wearables in Cardiology: Here to Stay. Heart Rhythm. 2020, 17, 889–895. [Google Scholar] [CrossRef]
- Koshy, A.N.; Sajeev, J.K.; Nerlekar, N.; Brown, A.J.; Rajakariar, K.; Zureik, M.; Wong, M.C.; Roberts, L.; Street, M.; Cooke, J.; et al. Smart Watches for Heart Rate Assessment in Atrial Arrhythmias. Int. J. Cardiol. 2018, 266, 124–127. [Google Scholar] [CrossRef] [PubMed]
- Chi, Y.M.; Jung, T.P.; Cauwenberghs, G. Dry-contact and noncontact biopotential electrodes: Methodological review. IEEE Rev. Biomed. Eng. 2010, 3, 106–119. [Google Scholar] [CrossRef] [PubMed]
- Zhou, W.; Song, R.; Pan, X.; Peng, Y.; Qi, X.; Peng, J.; Hui, K.S.; Hui, K.N. Fabrication and Impedance Measurement of Novel Metal Dry Bioelectrode. Sens. Actuators A Phys. 2013, 201, 127–133. [Google Scholar] [CrossRef]
- Ha-Chul Jung; Jin-Hee Moon; Dong-Hyun Baek; Jae-Hee Lee; Yoon-Young Choi; Joung-Sook Hong; Sang-Hoon Lee CNT/PDMS Composite Flexible Dry Electrodesfor Long-Term ECG Monitoring. IEEE Trans. Biomed. Eng. 2012, 59, 1472–1479. [CrossRef]
- Lou, C.; Li, R.; Li, Z.; Liang, T.; Wei, Z.; Run, M.; Yan, X.; Liu, X. Flexible Graphene Electrodes for Prolonged Dynamic ECG Monitoring. Sensors 2016, 16, 1833. [Google Scholar] [CrossRef]
- Lee, D.H.; Lee, E.K.; Kim, C.H.; Yun, H.J.; Kim, Y.-J.; Yoo, H. Blended Polymer Dry Electrodes for Reliable Electrocardiogram and Electromyogram Measurements and Their Eco-Friendly Disposal Led by Degradability in Hot Water. Polymers 2022, 14, 2586. [Google Scholar] [CrossRef]
- Lo, L.-W.; Zhao, J.; Aono, K.; Li, W.; Wen, Z.; Pizzella, S.; Wang, Y.; Chakrabartty, S.; Wang, C. Stretchable Sponge Electrodes for Long-Term and Motion-Artifact-Tolerant Recording of High-Quality Electrophysiologic Signals. ACS Nano 2022, 16, 11792–11801. [Google Scholar] [CrossRef]
- Lee, Y.; Yim, S.-G.; Lee, G.W.; Kim, S.; Kim, H.S.; Hwang, D.Y.; An, B.-S.; Lee, J.H.; Seo, S.; Yang, S.Y. Self-Adherent Biodegradable Gelatin-Based Hydrogel Electrodes for Electrocardiography Monitoring. Sensors 2020, 20, 5737. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Liu, S.; Zhu, M.; He, Y.; Wei, Z.; Wang, Y.; Xu, Y.; Pan, H.; Huang, W.; Chen, S.; et al. Flexible Non-contact Electrodes for Wearable Biosensors System on Electrocardiogram Monitoring in Motion. Front. Neurosci. 2022, 16, 900146. [Google Scholar] [CrossRef] [PubMed]
- Samol, A.; Bischof, K.; Luani, B.; Pascut, D.; Wiemer, M.; Kaese, S. Single-Lead ECG Recordings Including Einthoven and Wilson Leads by a Smartwatch: A New Era of Patient Directed Early ECG Differential Diagnosis of Cardiac Diseases? Sensors 2019, 19, 4377. [Google Scholar] [CrossRef]
- Giovangrandi, L.; Inan, O.T.; Wiard, R.M.; Etemadi, M.; Kovacs, G.T. Ballistocardiography—A Method Worth Revisiting. In Proceedings of the 2011 Annual International Conference of the IEEE Engineering in Medicine and Biology Society, Boston, MA, USA, 30 August–3 September 2011; pp. 4279–4282. [Google Scholar]
- Yang, C.C.; Hsu, Y.L. A Review of Accelerometry-Based Wearable Motion Detectors for Physical Activity Monitoring. Sensors 2010, 10, 7772–7788. [Google Scholar] [CrossRef] [PubMed]
- Pour-Ghaz, I.; Hana, D.; Raja, J.; Ibebuogu, U.N.; Khouzam, R.N. CardioMEMS: Where We Are and Where Can We Go? Ann. Transl. Med. 2019, 7, 418. [Google Scholar] [CrossRef] [PubMed]
- Lima, F.V.; Kadiyala, V.; Huang, A.; Agusala, K.; Cho, D.; Freeman, A.M.; Druz, R. At the Crossroads! Time to Start Taking Smartwatches Seriously. Am. J. Cardiol. 2022, 9149, 654. [Google Scholar] [CrossRef]
- Patel, U.K.; Malik, P.; Patel, N.; Patel, P.; Mehta, N.; Urhoghide, E.; Aedma, S.; Chakinala, R.C.; Shah, S.; Arumaithurai, K. Newer Diagnostic and Cost-Effective Ways to Identify Asymptomatic Atrial Fibrillation for the Prevention of Stroke. Cureus 2021, 13, e12437. [Google Scholar] [CrossRef] [PubMed]
- Krittanawong, C.; Rogers, A.J.; Johnson, K.W.; Wang, Z.; Turakhia, M.P.; Halperin, J.L.; Narayan, S.M. Integration of Novel Monitoring Devices with Machine Learning Technology for Scalable Cardiovascular Management. Nat. Rev. Cardiol. 2021, 18, 75–91. [Google Scholar] [CrossRef] [PubMed]
- Khandoker, A.H.; Palaniswami, M.; Karmakar, C.K. Support Vector Machines for Automated Recognition of Obstructive Sleep Apnea Syndrome From ECG Recordings. IEEE Trans. Inf. Technol. Biomed. 2009, 13, 37–48. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.-N.; Lee, M.-Y. Bispectral Analysis and Genetic Algorithm for Congestive Heart Failure Recognition Based on Heart Rate Variability. Comput. Biol. Med. 2012, 42, 816–825. [Google Scholar] [CrossRef] [PubMed]
- Acharya, U.R.; Faust, O.; Kadri, N.A.; Suri, J.S.; Yu, W. Automated Identification of Normal and Diabetes Heart Rate Signals Using Nonlinear Measures. Comput. Biol. Med. 2013, 43, 1523–1529. [Google Scholar] [CrossRef]
- Pokaprakarn, T.; Kitzmiller, R.R.; Moorman, J.R.; Lake, D.E.; Krishnamurthy, A.K.; Kosorok, M.R. Sequence to Sequence ECG Cardiac Rhythm Classification Using Convolutional Recurrent Neural Networks. IEEE J. Biomed. Health Inform. 2022, 26, 572–580. [Google Scholar] [CrossRef]
- Iwasaki, A.; Nakayama, C.; Fujiwara, K.; Sumi, Y.; Matsuo, M.; Kano, M.; Kadotani, H. Screening of Sleep Apnea Based on Heart Rate Variability and Long Short-Term Memory. Sleep Breath. 2021, 25, 1821–1829. [Google Scholar] [CrossRef]
- Hettiarachchi, I.T.; Hanoun, S.; Nahavandi, D.; Nahavandi, S. Validation of Polar OH1 Optical Heart Rate Sensor for Moderate and High Intensity Physical Activities. PLoS ONE 2019, 14, e0217288. [Google Scholar] [CrossRef]
- Koenig, C.; Ammann, R.A.; Kuehni, C.E.; Roessler, J.; Brack, E. Continuous Recording of Vital Signs with a Wearable Device in Pediatric Patients Undergoing Chemotherapy for Cancer—An Operational Feasibility Study. Support. Care Cancer 2021, 29, 5283–5292. [Google Scholar] [CrossRef]
- Sequeira, N.; D’Souza, D.; Angaran, P.; Aves, T.; Dorian, P. Common Wearable Devices Demonstrate Variable Accuracy in Measuring Heart Rate during Supraventricular Tachycardia. Heart Rhythm. 2020, 17, 854–859. [Google Scholar] [CrossRef] [PubMed]
- Carreiro, S.; Chintha, K.K.; Shrestha, S.; Chapman, B.; Smelson, D.; Indic, P. Wearable Sensor-Based Detection of Stress and Craving in Patients during Treatment for Substance Use Disorder: A Mixed Methods Pilot Study. Drug Alcohol Depend. 2020, 209, 107929. [Google Scholar] [CrossRef]
- Hernández-Vicente, A.; Hernando, D.; Marín-Puyalto, J.; Vicente-Rodríguez, G.; Garatachea, N.; Pueyo, E.; Bailón, R. Validity of the Polar H7 Heart Rate Sensor for Heart Rate Variability Analysis during Exercise in Different Age, Body Composition and Fitness Level Groups. Sensors 2021, 21, 902. [Google Scholar] [CrossRef]
- Nazari, G.; Bobos, P.; MacDermid, J.C.; Sinden, K.E.; Richardson, J.; Tang, A. Psychometric Properties of the Zephyr Bioharness Device: A Systematic Review. BMC Sports Sci. Med. Rehabil. 2018, 10, 6. [Google Scholar] [CrossRef] [PubMed]
- Feito, Y.; Moriarty, T.A.; Mangine, G.; Monahan, J. The Use of a Smart-Textile Garment during High-Intensity Functional Training: A Pilot Study. J. Sports Med. Phys. Fitness 2019, 59, 947–954. [Google Scholar] [CrossRef] [PubMed]
- Elliot, C.A.; Hamlin, M.J.; Lizamore, C.A. Validity and Reliability of the Hexoskin Wearable Biometric Vest During Maximal Aerobic Power Testing in Elite Cyclists. J. Strength Cond. Res. 2019, 33, 1437–1444. [Google Scholar] [CrossRef] [PubMed]
- Nageotte, M.P. Fetal Heart Rate Monitoring. Semin. Fetal Neonatal Med. 2015, 20, 144–148. [Google Scholar] [CrossRef] [PubMed]
- Chessa, M.; Butera, G.; Lanza, G.A.; Bossone, E.; Delogu, A.; de Rosa, G.; Marietti, G.; Rosti, L.; Carminati, M. Role of Heart Rate Variability in the Early Diagnosis of Diabetic Autonomic Neuropathy in Children. Herz 2002, 27, 785–790. [Google Scholar] [CrossRef]
- Bigger, J.T.; Fleiss, J.L.; Steinman, R.C.; Rolnitzky, L.M.; Kleiger, R.E.; Rottman, J.N. Frequency Domain Measures of Heart Period Variability and Mortality after Myocardial Infarction. Circulation 1992, 85, 164–171. [Google Scholar] [CrossRef]
- Steinberg, J.S.; Varma, N.; Cygankiewicz, I.; Aziz, P.; Balsam, P.; Baranchuk, A.; Cantillon, D.J.; Dilaveris, P.; Dubner, S.J.; El-Sherif, N.; et al. 2017 ISHNE-HRS Expert Consensus Statement on Ambulatory ECG and External Cardiac Monitoring/Telemetry. Heart Rhythm. 2017, 14, e55–e97. [Google Scholar] [CrossRef] [PubMed]
- Rieg, T.; Frick, J.; Baumgartl, H.; Buettner, R. Demonstration of the Potential of White-Box Machine Learning Approaches to Gain Insights from Cardiovascular Disease Electrocardiograms. PLoS ONE 2020, 15, e0243615. [Google Scholar] [CrossRef]
- Faust, O.; Kareem, M.; Ali, A.; Ciaccio, E.J.; Acharya, U.R. Automated Arrhythmia Detection Based on RR Intervals. Diagnostics 2021, 11, 1446. [Google Scholar] [CrossRef]
- Ivaturi, P.; Gadaleta, M.; Pandey, A.C.; Pazzani, M.; Steinhubl, S.R.; Quer, G. A Comprehensive Explanation Framework for Biomedical Time Series Classification. IEEE J. Biomed. Health Inform. 2021, 25, 2398–2408. [Google Scholar] [CrossRef]
- Zhang, L.; Wu, H.; Zhang, X.; Wei, X.; Hou, F.; Ma, Y. Sleep Heart Rate Variability Assists the Automatic Prediction of Long-Term Cardiovascular Outcomes. Sleep Med. 2020, 67, 217–224. [Google Scholar] [CrossRef]
- Martinez-Alanis, M.; Bojorges-Valdez, E.; Wessel, N.; Lerma, C. Prediction of Sudden Cardiac Death Risk with a Support Vector Machine Based on Heart Rate Variability and Heartprint Indices. Sensors 2020, 20, 5483. [Google Scholar] [CrossRef] [PubMed]
- Jeong, D.U.; Taye, G.T.; Hwang, H.-J.; Lim, K.M. Optimal Length of Heart Rate Variability Data and Forecasting Time for Ventricular Fibrillation Prediction Using Machine Learning. Comput Math Methods Med. 2021, 2021, 6663996. [Google Scholar] [CrossRef]
- Zhang, R.; Hua, Z.; Chen, C.; Liu, G.; Wen, W. Analysis of Autonomic Nervous Pattern in Hypertension Based on Short-Term Heart Rate Variability. Biomed. Eng./Biomed. Tech. 2021, 66, 71–79. [Google Scholar] [CrossRef] [PubMed]
- Turakhia, M.P.; Desai, M.; Hedlin, H.; Rajmane, A.; Talati, N.; Ferris, T.; Desai, S.; Nag, D.; Patel, M.; Kowey, P.; et al. Rationale and Design of a Large-Scale, App-Based Study to Identify Cardiac Arrhythmias Using a Smartwatch: The Apple Heart Study. Am. Heart J. 2019, 207, 66–75. [Google Scholar] [CrossRef] [PubMed]
- Lubitz, S.A.; Faranesh, A.Z.; Atlas, S.J.; McManus, D.D.; Singer, D.E.; Pagoto, S.; Pantelopoulos, A.; Foulkes, A.S. Rationale and Design of a Large Population Study to Validate Software for the Assessment of Atrial Fibrillation from Data Acquired by a Consumer Tracker or Smartwatch: The Fitbit Heart Study. Am. Heart J. 2021, 238, 16–26. [Google Scholar] [CrossRef] [PubMed]
- Bjerregaard, P.; Sorensen, K.E.; Molgaard, H. Predictive Value of Ventricular Premature Beats for Subsequent Ischaemic Heart Disease in Apparently Healthy Subjects. Eur. Heart J. 1991, 12, 597–601. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Haghayegh, S.; Khoshnevis, S.; Smolensky, M.H.; Diller, K.R.; Castriotta, R.J. Accuracy of Wristband Fitbit Models in Assessing Sleep: Systematic Review and Meta-Analysis. J. Med. Internet Res. 2019, 21, e16273. [Google Scholar] [CrossRef] [PubMed]
- Shen, Q.; Qin, H.; Wei, K.; Liu, G. Multiscale Deep Neural Network for Obstructive Sleep Apnea Detection Using RR Interval From Single-Lead ECG Signal. IEEE Trans. Instrum. Meas. 2021, 70, 1–13. [Google Scholar] [CrossRef]
- Martín-Montero, A.; Gutiérrez-Tobal, G.C.; Gozal, D.; Barroso-García, V.; Álvarez, D.; del Campo, F.; Kheirandish-Gozal, L.; Hornero, R. Bispectral Analysis of Heart Rate Variability to Characterize and Help Diagnose Pediatric Sleep Apnea. Entropy 2021, 23, 1016. [Google Scholar] [CrossRef]
- Rathod, S.; Phadke, L.; Chaskar, U.; Patil, C. Heart Rate Variability Measured during Rest and after Orthostatic Challenge to Detect Autonomic Dysfunction in Type 2 Diabetes Mellitus Using the Classification and Regression Tree Model. Technol. Health Care 2022, 30, 361–378. [Google Scholar] [CrossRef]
- Shashikant, R.; Chaskar, U.; Phadke, L.; Patil, C. Gaussian Process-Based Kernel as a Diagnostic Model for Prediction of Type 2 Diabetes Mellitus Risk Using Non-Linear Heart Rate Variability Features. Biomed. Eng. Lett. 2021, 11, 273–286. [Google Scholar] [CrossRef]
- Olde Bekkink, M.; Koeneman, M.; de Galan, B.E.; Bredie, S.J. Early Detection of Hypoglycemia in Type 1 Diabetes Using Heart Rate Variability Measured by a Wearable Device. Diabetes Care 2019, 42, 689–692. [Google Scholar] [CrossRef]
- Martinez, G.J.; Grover, T.; Mattingly, S.M.; Mark, G.; D’Mello, S.; Aledavood, T.; Akbar, F.; Robles-Granda, P.; Striegel, A. Alignment Between Heart Rate Variability From Fitness Trackers and Perceived Stress: Perspectives From a Large-Scale In Situ Longitudinal Study of Information Workers. JMIR Hum. Factors 2022, 9, e33754. [Google Scholar] [CrossRef] [PubMed]
- Chalmers, T.; Hickey, B.A.; Newton, P.; Lin, C.-T.; Sibbritt, D.; McLachlan, C.S.; Clifton-Bligh, R.; Morley, J.; Lal, S. Stress Watch: The Use of Heart Rate and Heart Rate Variability to Detect Stress: A Pilot Study Using Smart Watch Wearables. Sensors 2021, 22, 151. [Google Scholar] [CrossRef]
- Düking, P.; Zinner, C.; Trabelsi, K.; Reed, J.L.; Holmberg, H.-C.; Kunz, P.; Sperlich, B. Monitoring and Adapting Endurance Training on the Basis of Heart Rate Variability Monitored by Wearable Technologies: A Systematic Review with Meta-Analysis. J. Sci. Med. Sport 2021, 24, 1180–1192. [Google Scholar] [CrossRef]
- Coutts, L.V.; Plans, D.; Brown, A.W.; Collomosse, J. Deep Learning with Wearable Based Heart Rate Variability for Prediction of Mental and General Health. J. Biomed. Inform. 2020, 112, 103610. [Google Scholar] [CrossRef] [PubMed]
- Gula, L.J.; Klein, G.J.; Zurawska, U.; Massel, D.; Yee, R.; Skanes, A.C.; Krahn, A.D. Does Familiarity with Technology Predict Successful Use of an External Loop Recorder? The Loop Recorder Technology Cognition Study (LOCO). Pacing Clin. Electrophysiol. 2009, 32, 466–472. [Google Scholar] [CrossRef]
- Hasselgren, A.; Kralevska, K.; Gligoroski, D.; Pedersen, S.A.; Faxvaag, A. Blockchain in Healthcare and Health Sciences-A Scoping Review. Int. J. Med. Inform. 2020, 134, 104040. [Google Scholar] [CrossRef] [PubMed]
- Patel, B.; Makaryus, A.N. Cardiac Implantable Electronic Devices and Cybersecurity. Expert Rev. Med. Devices 2021, 18, 69–77. [Google Scholar] [CrossRef] [PubMed]
- Ganeshan, R.; Enriquez, A.D.; Freeman, J.V. Remote Monitoring of Implantable Cardiac Devices: Current State and Future Directions. Curr. Opin. Cardiol. 2018, 33, 20–30. [Google Scholar] [CrossRef] [PubMed]
- Goldman, L. Telemetry or Not Telemetry: A Great Leap Forward or a Waste of Resources? Am. J. Med. 2001, 110, 67–68. [Google Scholar] [CrossRef]
- Chew, D.S.; Zarrabi, M.; You, I.; Morton, J.; Low, A.; Reyes, L.; Yuen, B.; Sumner, G.L.; Raj SR, E.; DV, W.; et al. Clinical and Economic Outcomes Associated With Remote Monitoring for Cardiac Implantable Electronic Devices: A Population-Based Analysis. Can. J. Cardiol. 2022, 38, 736–744. [Google Scholar] [CrossRef]
- Dario, C.; Delise, P.; Gubian, L.; Saccavini, C.; Brandolino, G.; Mancin, S. Large Controlled Observational Study on Remote Monitoring of Pacemakers and Implantable Cardiac Defibrillators: A Clinical, Economic, and Organizational Evaluation. Interact J. Med. Res. 2016, 5, 4. [Google Scholar] [CrossRef]
- Crossley, G.H.; Boyle, A.; Vitense, H.; Chang, Y.; Mead, R.H.; CONNECT Investigators. The CONNECT (Clinical Evaluation of Remote Notification to Reduce Time to Clinical Decision) Trial: The Value of Wireless Remote Monitoring with Automatic Clinician Alerts. J. Am. Coll. Cardiol. 2011, 57, 1181–1189. [Google Scholar] [CrossRef]
- Jo, A.; Coronel, B.D.; Coakes, C.E.; Mainous, A.G., 3rd. Is There a Benefit to Patients Using Wearable Devices Such as Fitbit or Health Apps on Mobiles? A Systematic Review. Am. J. Med. 2019, 132, 1394–1400. [Google Scholar] [CrossRef]
- Garcia, A.; Balasubramanian, V.; Lee, J.; Gardner, R.; Gummidipundi, S.; Hung, G.; Ferris, T.; Cheung, L.; Granger, C.; Kowey, P.; et al. Lessons Learned in the Apple Heart Study and Implications for the Data Management of Future Digital Clinical Trials. J. Biopharm. Stat. 2022, 32, 496–510. [Google Scholar] [CrossRef] [PubMed]
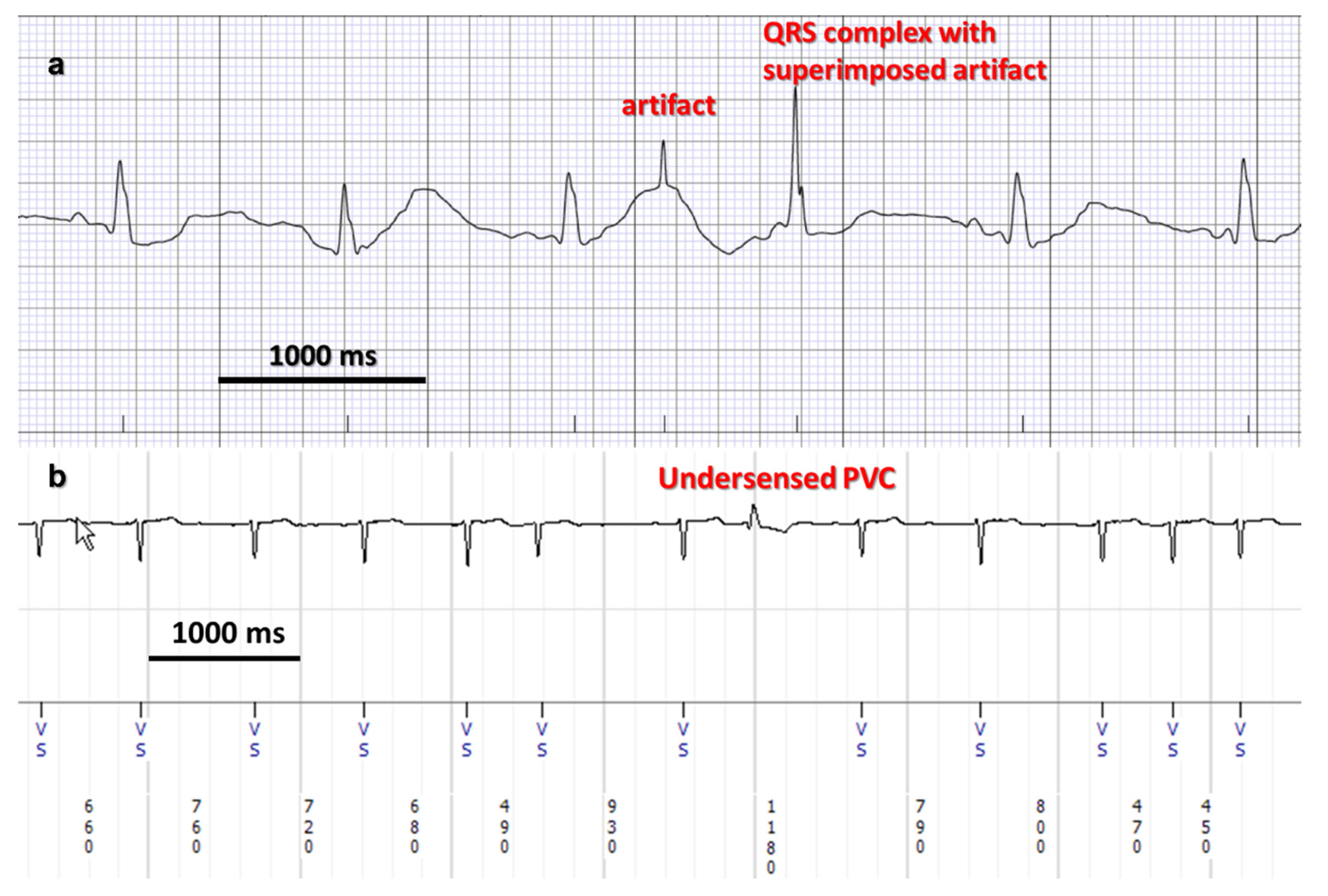

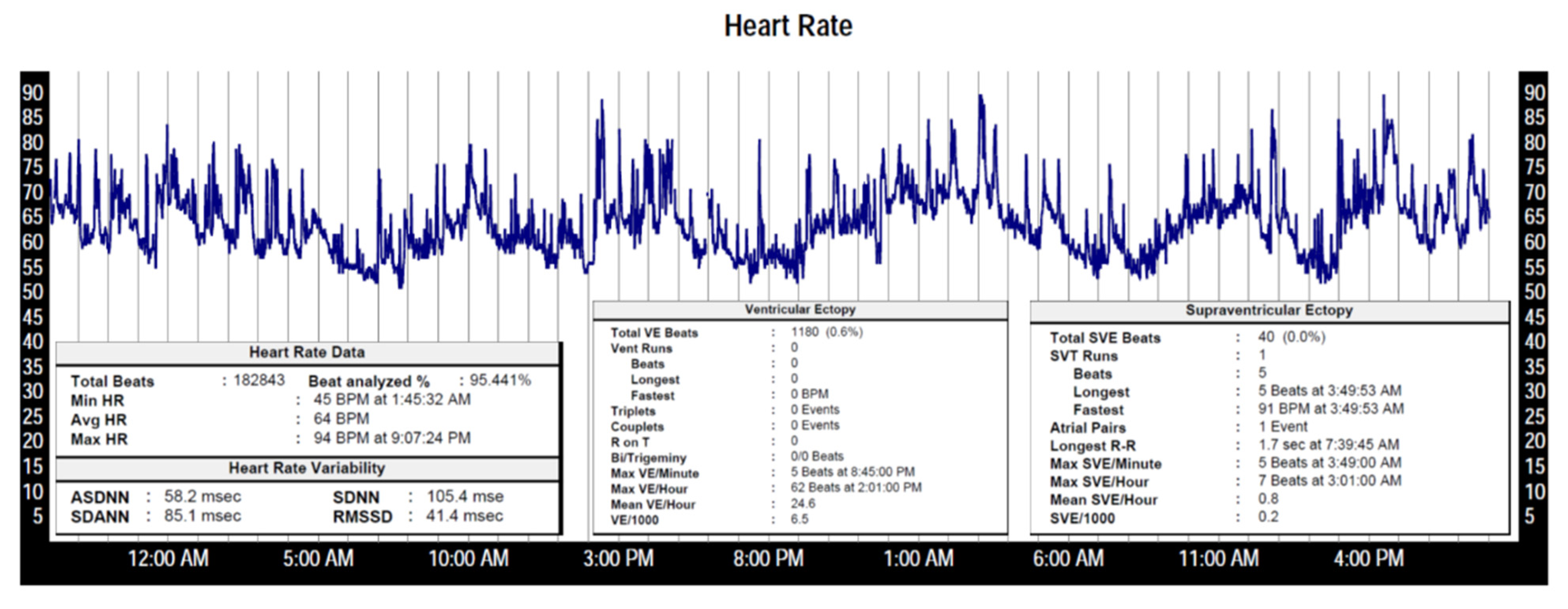
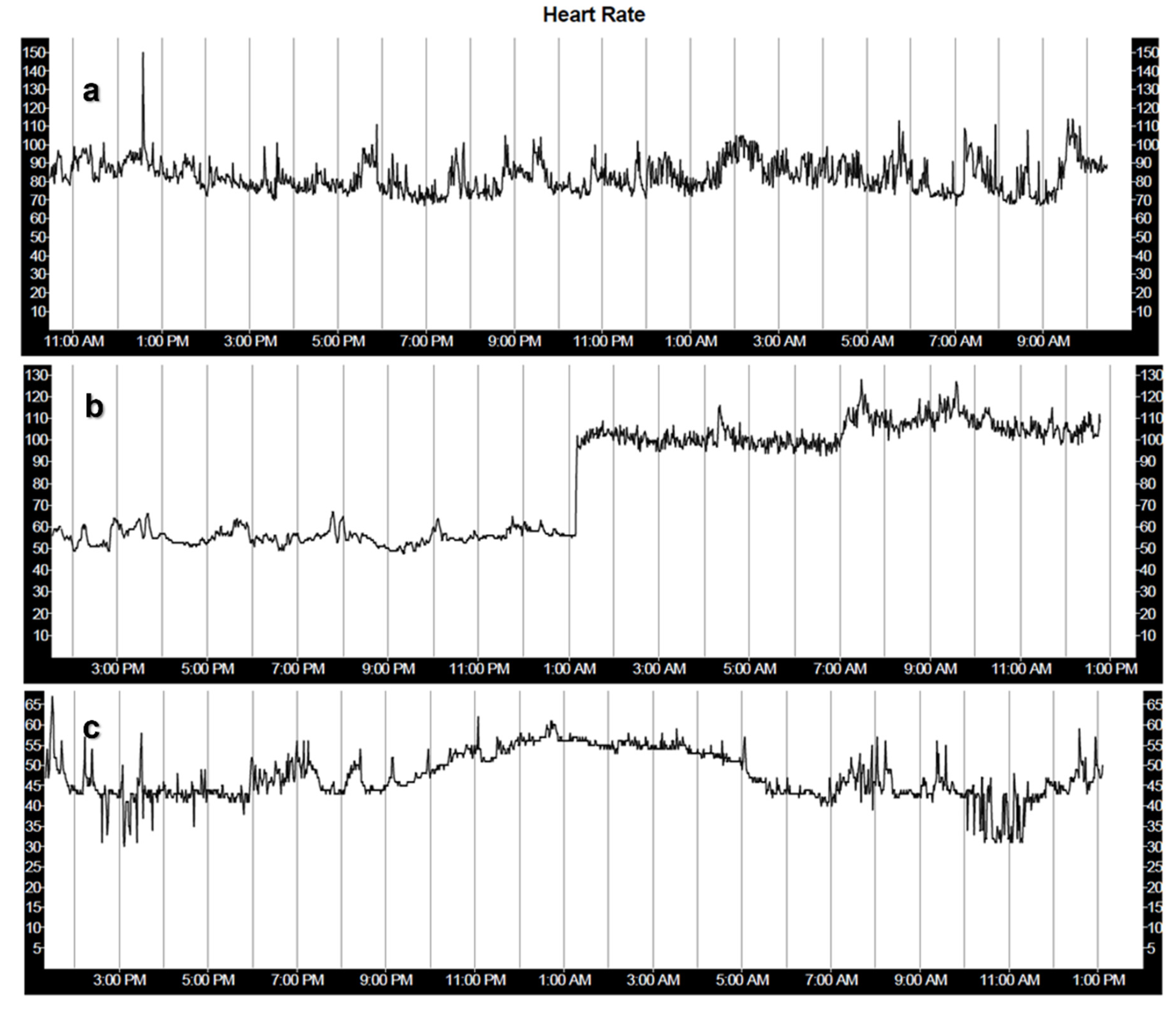

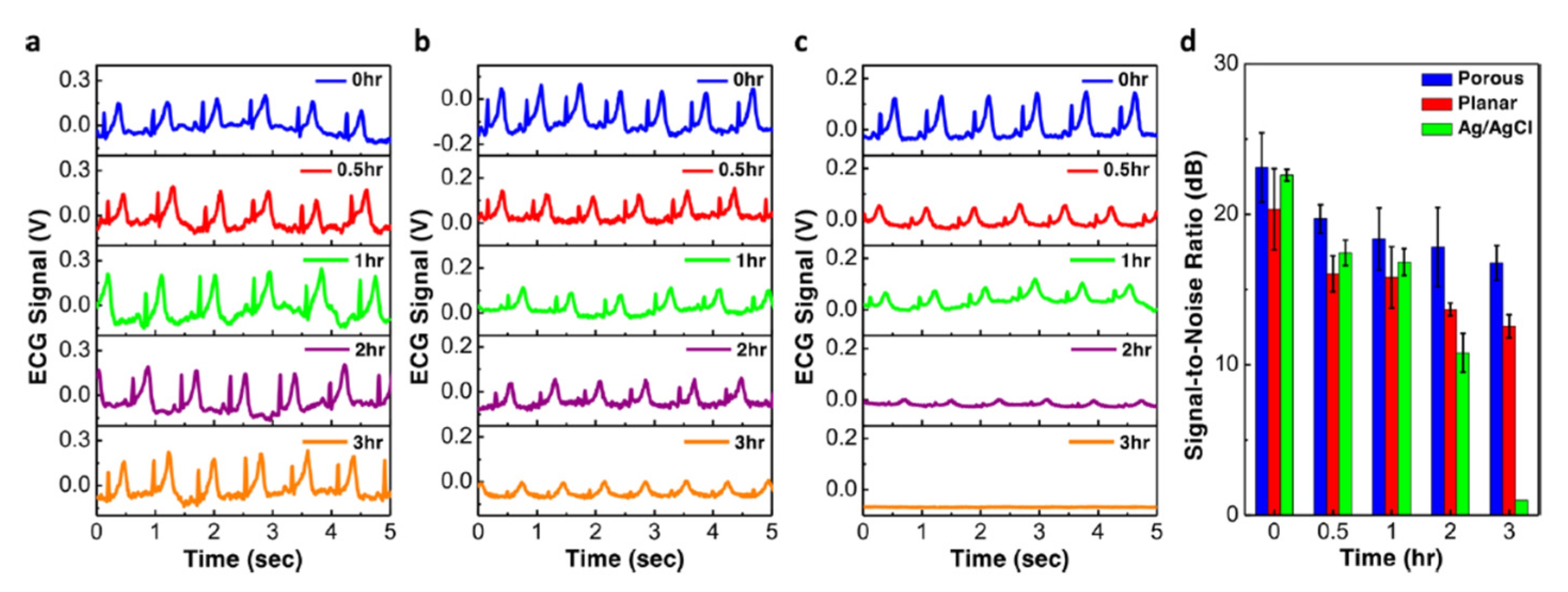

| Regulatory System | Effect on HR |
|---|---|
| Autonomic nervous system | |
| Decrease Increase |
| Endocrine system | |
| Increase Increase |
| Intrinsic cardiac factors | |
| Increase or decrease |
| Metric | Description |
|---|---|
| |
| SDNN | Standard deviation of intervals |
| SDANN | Standard deviation of the average intervals for each 5 min segment |
| RMSSD | Root mean square of successive interval differences |
| |
| Power: ULF, VLF, LF, HF | Absolute power of the ultra-low, very low, low and high-frequency bands |
| Peak: ULF, VLF, LF, HF | Peak frequency of the ultra-low, very low, low and high-frequency bands |
| LF/HF | Ratio of low-to-high frequency power |
| |
| S | Area of the ellipse which represents total heart rate variability |
| ApEn, SampEn | Approximate and sample entropy—regularity and complexity of a time series |
| DFA α1, α2 | Detrended fluctuation analysis—short- and long-term fluctuations |
| Year | Technology |
|---|---|
| 1949 | Holter and Generelli: portable apparatus for wireless transmission of biopotential signals using 50 MHz radio waves [15] |
| 1961 | Holter: electrocardiorecorder—local storage of recorded data (Holter monitor) [16] |
| 1960s | Holter, Ledley, Nomura: semiautomatic electrocardiogram analysis |
| 1970s | Computer-based automated pattern recognition for ECG analysis [17] |
| 1980s | Probability density and statistical processing of electrocardiographic data Local processing of data, real time analysis (decreased need to transmit large amount of raw data for remote analysis) [18] Improved electrode technology and signal quality—evaluation of ischemia/repolarization [19] |
| 1990s | Digital storage, solid state memory with increased storage capacity; multi-channel and longer duration monitoring [20] Standardization of data formats [21] Implantable subcutaneous heart monitor [22] |
| 2000s | Miniaturization of wearable and implantable devices Minimally invasive implantable monitors—Medtronic Reveal LinQ |
| 2010s | Consumer grade remote monitoring becomes available for the general population—AliveCor Kardia (ECG), Apple Watch (heart rate) |
| 2020s | Cloud-based monitoring services Use of machine learning/artificial intelligence for signal analysis and interpretation [23] |
| Device | Sensors/Parameters |
|---|---|
| Polar OH1 | HR (PPG) |
| Everion | HR (PPG), activity, blood oxygen, temperature (more with other linked sensors) |
| Device | Sensors/Parameters |
|---|---|
| Fitbit Luxe | HR (PPG), motion, temperature |
| Fitbit Versa 3 | HR (PPG), temperature, GPS |
| Apple Watch 6 | HR (PPG), ECG, motion, blood oxygen |
| Garmin VivoSmart HR | HR (PPG), motion |
| Polar A360 | HR (PPG) |
| Device | Sensors/Parameters |
|---|---|
| Polar H7 | ECG |
| Zephyr Bioharness 3 | ECG |
| Device | Sensors/parameters |
|---|---|
| Om Bra | HR, respiration, pedometer |
| Hexoskin | ECG, blood oxygen, respiration, position and acceleration |
| Assessment | Indication |
|---|---|
| Symptom correlation with arrhythmia | Loss or near loss of consciousness, palpitations, chest pain, shortness of breath or neurological symptoms, due to unknown cause |
| Risk associated with asymptomatic arrhythmia | Patients after heart attack and decreased heart function, congestive heart failure, hypertrophic cardiomyopathy |
| Monitor antiarrhythmic management | Rate and rhythm control assessment, proarrhythmic response detection |
| Pacemaker or implanted defibrillator function | Symptoms suspected due to device malfunction and not explained by device interrogation |
| Ischemic heart disease | Transient angina pectoris, patient unable to exercise |
| Atrial fibrillation | Diagnosis of atrial fibrillation, assessment of rate and rhythm control |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alugubelli, N.; Abuissa, H.; Roka, A. Wearable Devices for Remote Monitoring of Heart Rate and Heart Rate Variability—What We Know and What Is Coming. Sensors 2022, 22, 8903. https://doi.org/10.3390/s22228903
Alugubelli N, Abuissa H, Roka A. Wearable Devices for Remote Monitoring of Heart Rate and Heart Rate Variability—What We Know and What Is Coming. Sensors. 2022; 22(22):8903. https://doi.org/10.3390/s22228903
Chicago/Turabian StyleAlugubelli, Navya, Hussam Abuissa, and Attila Roka. 2022. "Wearable Devices for Remote Monitoring of Heart Rate and Heart Rate Variability—What We Know and What Is Coming" Sensors 22, no. 22: 8903. https://doi.org/10.3390/s22228903
APA StyleAlugubelli, N., Abuissa, H., & Roka, A. (2022). Wearable Devices for Remote Monitoring of Heart Rate and Heart Rate Variability—What We Know and What Is Coming. Sensors, 22(22), 8903. https://doi.org/10.3390/s22228903






