1. Introduction
There have been many efforts over the last few decades to incorporate robotic capabilities for improving medical procedures such as diagnostics, surgeries, and examination. An important examination procedure consists of the palpation of tissues to detect abnormalities or differences with respect to healthy tissues. In this context, several robotic systems that can potentially help practitioners to carry out a palpation task have been proposed in the literature. For example, as early as 1998, in [
1] a tendon-actuated finger equipped with a tactile sensor is designed for minimally invasive data gathering during palpation procedures. Later, in [
2], an integration of tactile and force sensors is proposed to enhance the location of tumors by providing force reflection to the physicians in a master–slave teleoperation scheme. In the same context, in [
3], a magnified force reflection scheme is proposed to improve the user’s capabilities for performing palpation and needle insertion tasks. This same topic is studied aiming at the inclusion of force feedback in the
da Vinci Research Kit for different tasks in a master–slave teleoperation system. In [
4], the modulation of the forces applied to the tissues is investigated. The authors have found that humans tend to impose a force pattern that is independent of the non-homogeneity of the tissue. Gaussian processes are studied in [
5] to construct a stiffness map to detect tissue abnormalities, while simultaneously avoiding obstacles. A combination of image processing and a miniature force sensor is proposed in [
6] also to construct a stiffness map in a virtual reality application designed for its use in the
da Vinci Research Kit. In [
7], a recurrent neural network is employed to detect the depth of tumors from position and force measurements. A system that does not employ force sensors at the tip of the probe is proposed in [
8], using a cable-driven mechanism capable of detecting changes on the stiffness of the tissues.
The Bayesian framework has been successfully employed in the literature for tumor detection. For example, in [
9] it is employed to detect tumor inclusions at different depths by employing an industrial manipulator with a capacitive tactile sensor mounted at the tip. A similar work [
10] employs a Bayesian optimization algorithm, and it is capable of detecting the boundary of a tumor within 30 iterations as well as the depth of the inclusion, based on tactile sensor measurements. When employing images for tumor detection, there are several recently obtained results. In [
11], MRI images are processed to obtain higher order statistical features to detect brain tumor. In another work [
12], a Bayesian fuzzy clustering algorithm is combined with a neural network for tumor detection and classification based on MRI images. The tumor growth estimation is investigated in [
13] from T2-FLAIR images by employing a Bayesian inference framework. In [
14], a new capsule networks are employed in a Bayesian framework to improve the detection and classification of tumors by including the knowledge of experts neurosurgeons for the uncertain prediction cases.
In this paper, we propose to use a state observer to simultaneously estimate contact forces and robot velocities for palpation tasks by employing only position measurements and the open loop joint torques commanded to the robot actuators. Thus, the observer is based on the robot dynamic model, the control inputs and the joint position measurements. Although different force and velocity observers have been proposed in the literature in the last few years [
15,
16,
17,
18], in this work we use the extended-state finite-time observer (FTO) proposed in [
19], which has the advantage of providing finite time convergence of the forces and velocities to the real ones. Additionally, this observer has the advantage of its easy implementation and tuning. An accurate dynamic model of the manipulator is required to obtain a good approximation of the forces (this is not the case for the velocitites estimation, which is independent of the model accuracy). Nevertheless, to the best of the authors’ knowledge, there is no velocity and force observer independent of the model accuracy. The estimated velocity and force signals are, in turn, employed to estimate the biomechanical parameters of the palpated tissue, given an a priori specified model. An important feature of the proposed work is its capability of not only distinguish between tissues with different stiffness, but also providing an estimation of the tissue model parameters. These parameters can be employed in a number of applications, e.g., realistic simulation of the tissues in virtual reality systems [
20,
21] for educational/training purposes, mathematical analysis of interaction controllers, among others. A Bayesian classifier is also proposed in this paper to automatically detect the tissue which with the robot is interacting, after a training phase. Two models are proposed to model the tissue mechanics, one considering a linear elasticity and another considering a nonlinear elasticity. We have found that by combining the estimated parameters of the two models, the classification is greatly improved. In contrast with the result of [
8], instead of an ad-hoc system, a standard robotic manipulator model is employed in this work, which can be an important feature towards its implementation in a real scenario. In accordance with [
4], the modulation of forces is very important for the correct detection of tissue abnormalities. For such reason, a closed-loop force controller based on the estimated signals is proposed in this work, with a carefully selected force profile which permits to estimate the mechanical parameters accurately, while preventing any damage to the tissue. Furthermore, the shortest possible experimental duration time was also tuned to reduce the stress on the tissue.
The localization of the zones of interest (e.g., tumors) is out of the scope of this paper. In turn, we assume that either the physician has already identified the zone in which they want to learn its mechanical properties, or one of the several automatic algorithms reported in the literature has been implemented in a previous step, e.g., tactile sensor-based [
2,
9,
10], force sensor (in the present work, the estimated force can be used instead of the force measured by the sensor) feedback [
3,
5,
22], preoperative images processing [
6], magnetic fields [
23], MRI scans, and CT scans. A recent review on the methods for identification of zones based on medical images analysis and artificial intelligence can be found in [
24].
The estimated contact force can be utilized for force-reflection, without further modifications, in a master–slave teleoperation system similar to the one proposed in [
2] for tumor localization, with the advantage of obviating the force sensor requirement.
In summary, the main contributions of this article are the following:
Simultaneous estimation of robot end-effector forces and velocities, using only joint position sensors and commanded torques.
Estimation of tissues’ biomechanical parameters based on the estimated forces and velocities.
A standard robot manipulator model is employed instead of an ad hoc system.
Classification of tissues is based on the estimated parameters taking into account a linear model, a nonlinear model, and the combination of both models, giving, as a result, a better classification for the last case.
The remainder of this paper is organized as follows: In
Section 2, the description of the theoretical tools to support the proposed method is presented. In
Section 3, experimental results, which serve to validate the approach, are shown. A discussion section on these experimental results is presented in
Section 4. Some concluding remarks and directions for future work are stated in
Section 5.
3. Results
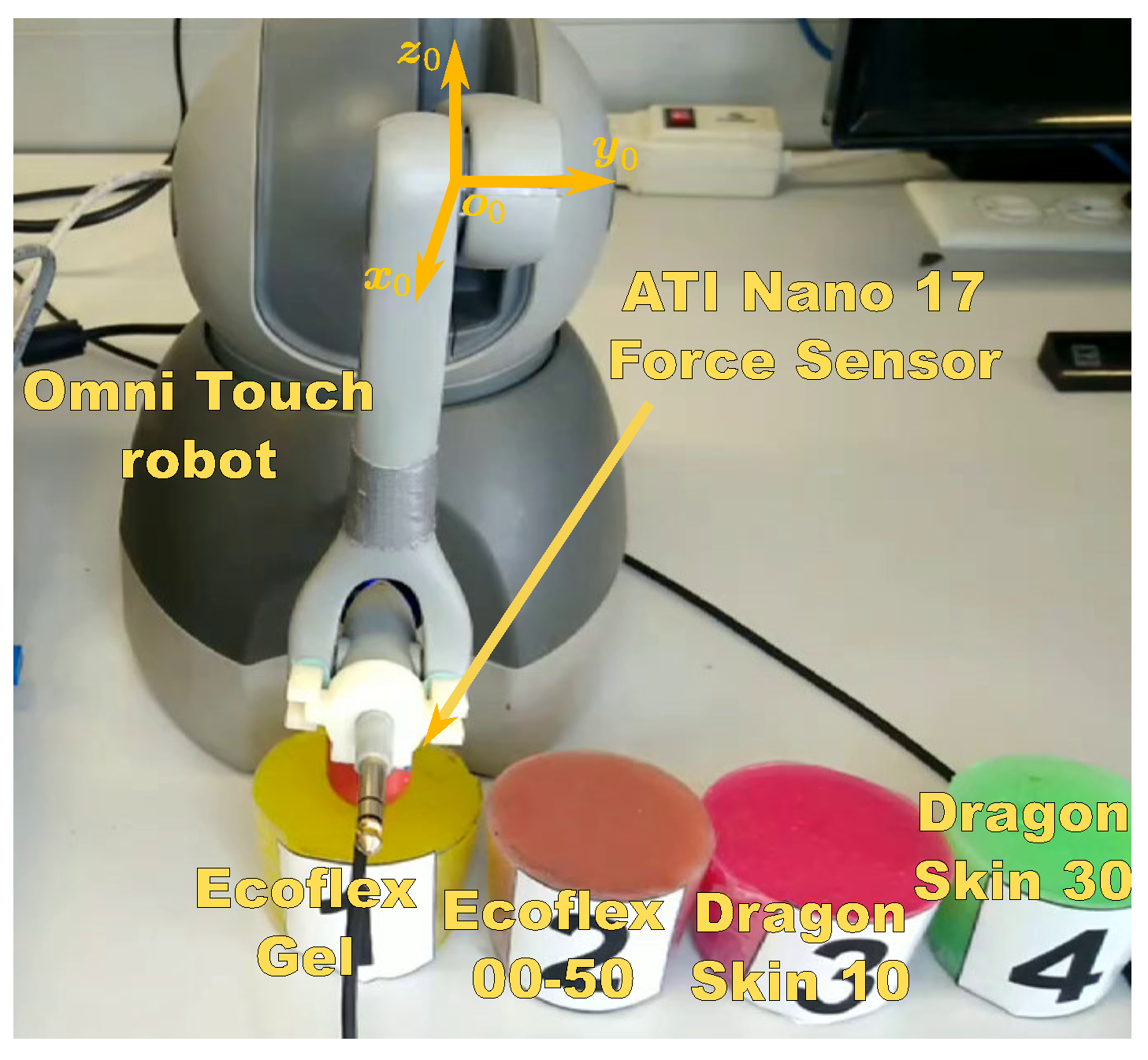
The experimental setup shown in
Figure 1 was set to validate the proposed approach of
Section 2. It consists of a
3D Systems Omni Touch manipulator of six degrees of freedom, from which only the first three are actuated, a six-axes
ATI Nano 17 force sensor (only for validation) and four silicon rubbers with different mechanical properties to represent the tissues:
Ecoflex Gel,
Ecoflex 00-50,
Dragon Skin 10, and
Dragon Skin 30.
The robot manipulator is programmed and controlled by a
Visual C++ application running on a
PC with a
Windows Operating System. The closed-loop sample-time is
, including the force sensor acquisition, robot measurement, and the observer and controller computations. The experiment consists of following a desired force profile over the tissue along the vertical axis, i.e.,
. As explained in
Section 2, the force exerted over the tissue must be carefully chosen to prevent any damage, while still having a relatively low noise-to-signal ratio. For such reasons, the desired force was chosen as
Although a single frequency is sufficient to estimate the two coefficients in each model (
3) or (
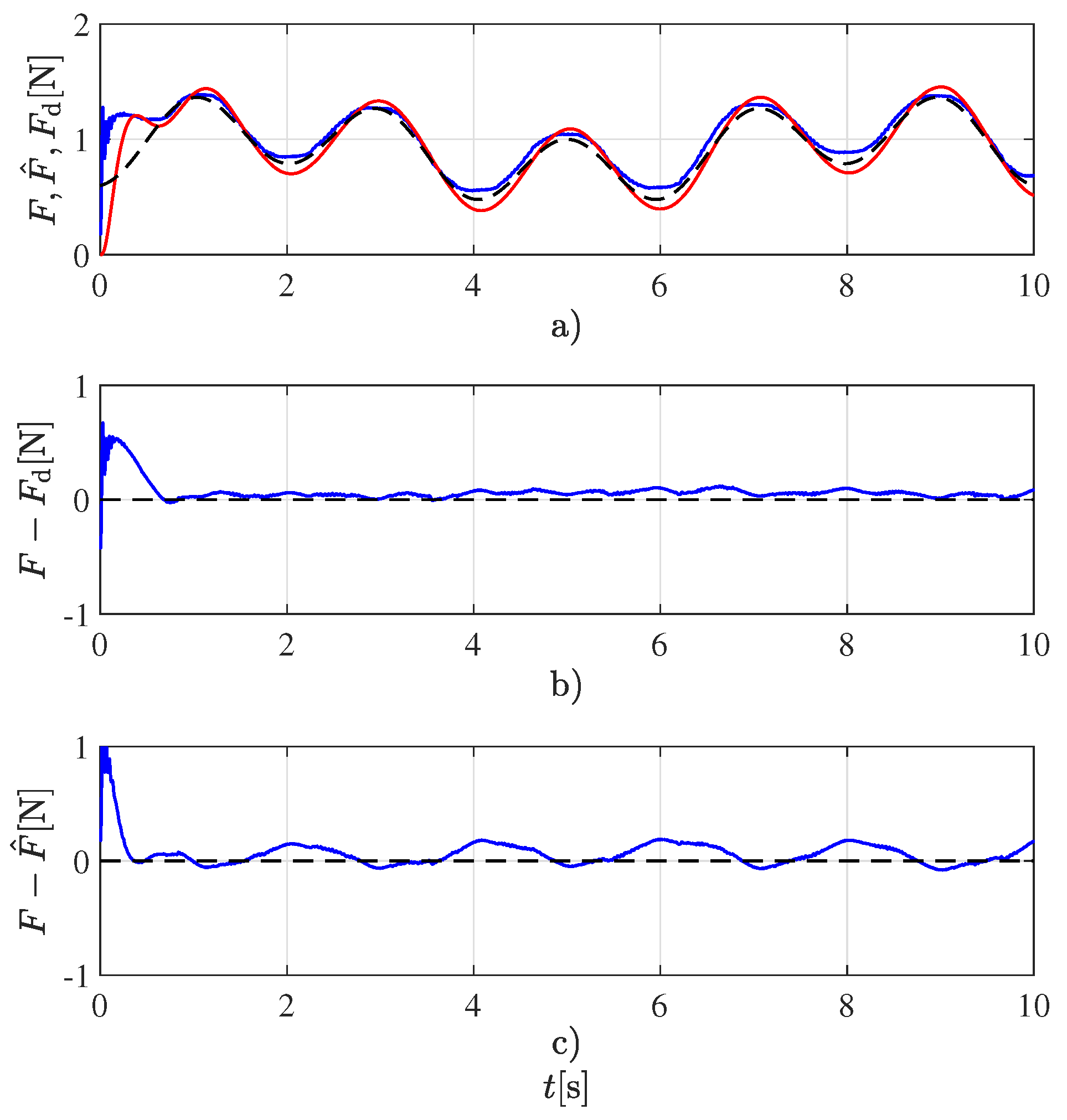
4), the more frequencies there are, the better the excitation of the system. For such reason, a signal with two frequencies was chosen instead of a single frequency signal. It was experimentally determined that increasing the number of frequencies beyond the two employed has no further advantage. This desired force profile, along with the estimated force and the measured one employed for validating the estimated one, are shown in
Figure 2. This figure also displays the force tracking error, i.e., the difference between the desired and measured forces and the force estimation error, i.e., the difference between the estimated and measured forces. Although only the experiment for the
Ecoflex 00-50 sample is shown, very similar results can be obtained for the rest of the samples.
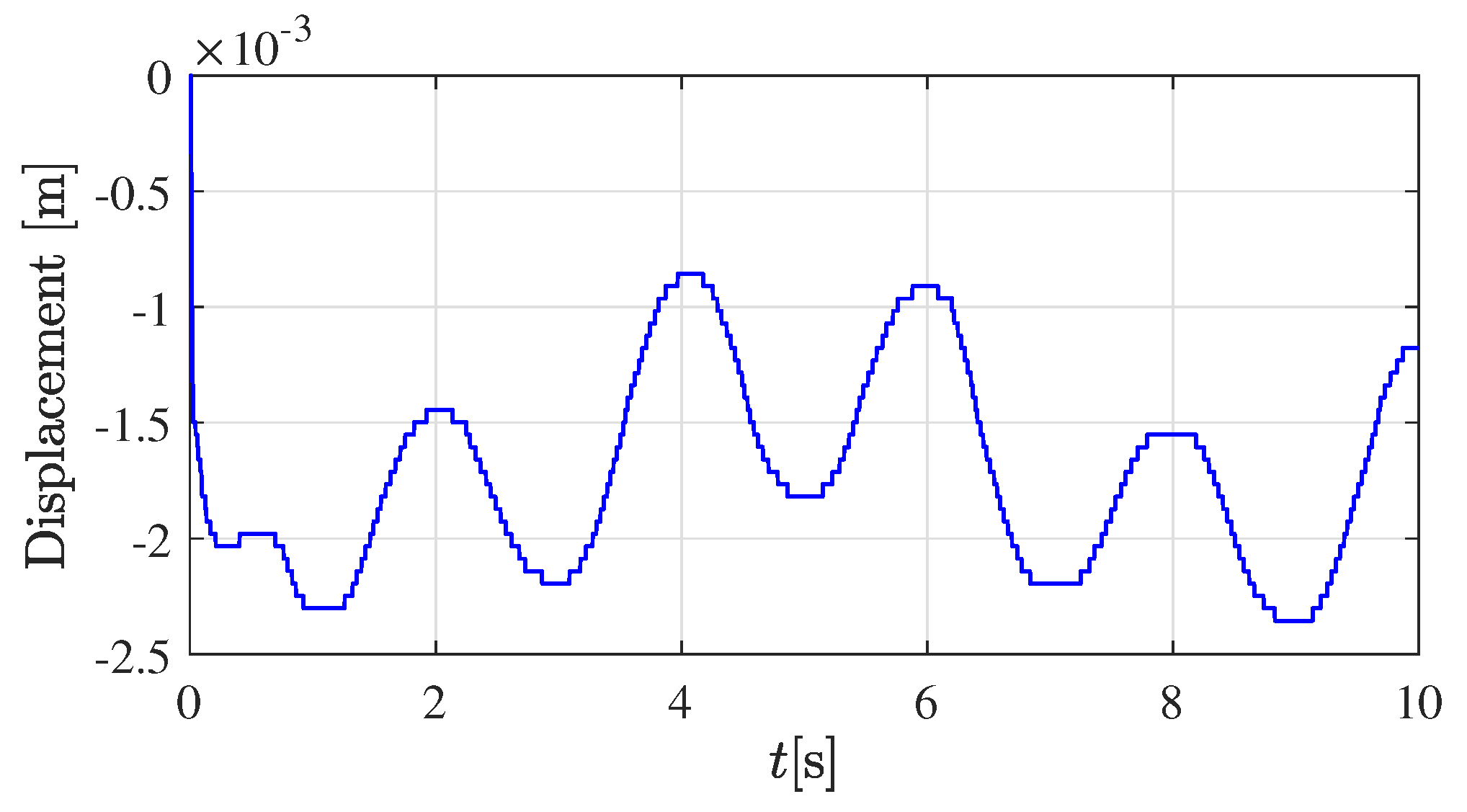
The corresponding position of the robot’s tip in the vertical direction (
-axis in robot base-coordinates) is displayed in
Figure 3. Approximately the first second of the experiment corresponds to the transient response of the closed-loop controller and observer; therefore, it is discarded for the parameters estimator (
17).
As mentioned in
Section 2, an accurate model of the employed manipulator is required to obtain reliable estimations of the robot velocity and the contact forces. The dynamic model for the three degrees of freedom configuration of the Omni Touch robot employed in this work was taken from [
33]. For the sake of completeness of this manuscript, the mentioned model can be found in
Appendix A. The robot parameters were obtained by an open-loop free-motion experiment and a standard least squares procedure. In turn, the gains chosen for the observer (
8)–(10) were
,
, and
, whereas the force controller gain in (
18) was set to
.
As mentioned in
Section 2, the experiment duration must be the shortest possible without implying a significant variation on the estimated parameter mean values. Experimentally, it was found that there is no significant difference in the mean estimation values for experiments with duration
s. Therefore, a set of 32 experiments of duration
s with the force profile given by (
22) was carried out. The mean values and the standard deviations for both models (
3) and (
4) and for each of the four rubber samples are presented in
Table 1 and
Table 2, respectively. With the aim of validating the estimated coefficients with respect to ground truth, we additionally performed the estimation of k1, b1, knl, and bnl using the real forces measured with the ATI Nano force sensor. A series of four pairwise
T-Tests applied over the estimated coefficients, comparing the ones obtained with the real versus the estimated contact forces did not reveal any significant differences for any of the four coefficients. Meaning that the proposed force-sensorless identification approach is reliable and valid.
From these two tables, it can be seen that the elasticity coefficients
and
posses more information to distinguish between the different samples, i.e., these coefficients are
dominant over
and
in models (
3) and (
4), and thus can be employed for the Bayesian classifier (BC) in (
20). A set of another 32 experiments were carried out, considering the estimated linear elasticity coefficient as the input for the BC, i.e.,
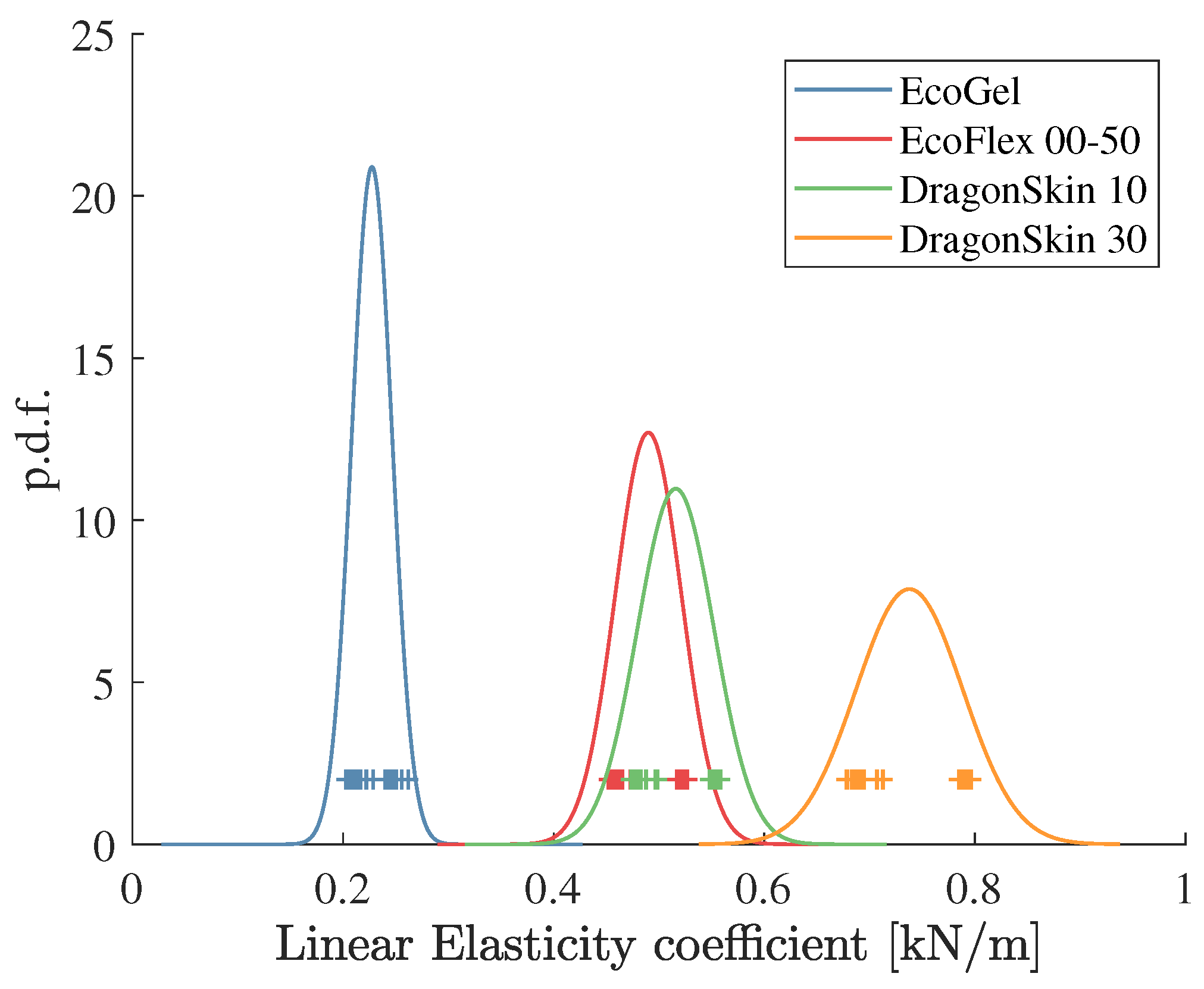
. The estimated coefficients as well as the probability density functions (pdf) obtained from
Table 1 and
Table 2 are shown in
Figure 4.
From this figure, it can be seen that the
Ecoflex Gel and the
Dragon Skin 30 samples can be easily distinguished from the other two samples. However, the pdfs of the
Ecoflex 00-50 and the
Dragon Skin 10 have an important intersection area. In fact, these two materials have a very similar stiffness when manually testing them. The percentage of correct classifications (true positives) for all the material samples is shown in
Table 3.
A better classification can be obtained by considering the nonlinear elasticity coefficient
as the input for the BC. The pdfs obtained from
Table 1 and
Table 2 for this case, along with the outcomes of a new set of 32 experiments, are shown in
Figure 5.
When considering the nonlinear spring coefficient
in (
4) as the classifier input, the percentage of correct classifications improves, as displayed in
Table 4.
Finally, if instead of a single estimated parameter, a combination of
and
is used in the SBC defined in (
21), the classifier always detects the correct rubber sample, as shown in
Table 5.
In summary, the experiments presented in this section consisted of the following parts. (1) Each silicone sample is put under the robot tip. (2) The robot exerts the force profile given by (
22) by applying the force control (
18) in the vertical direction over the tissue. (3) This experiment is repeated several times for each sample to obtain the statistical parameters, i.e., mean and standard deviation. (4) Now, each sample is put randomly below the robot tip. (5) By applying the same force profile and control given by (
22) and (
18), the parameter estimator and the Bayesian classifier (
20), the algorithm returns both the estimated parameters and the tissue classification. An accompanying video where the experiment corresponding to the results of
Table 5 is shown can be found at
https://youtu.be/tAgwVmYLZp8 (accessed on 7 September 2022).
4. Discussion
The results presented in
Section 3 show that an approximate estimation of biomechanical parameters can be successfully carried out by employing the robot velocities and contact forces obtained by the Finite-Time Observer (
8)–(10) instead of directly measuring them. Although no perfect force reconstruction is possible due to the inherent model inaccuracies and unmodeled external disturbances, the estimation error, as well as the force tracking error, remained relatively low for the magnitude of the applied forces.
The parameters estimated by the method proposed in this work can be employed not only for classification purposes but for simulation of tissues, mathematical analysis of the interaction, haptic interfaces, and so on. For the sake of the proposed application, only the two models (
3) and (
4) were considered in this work, but any linearly parameterizable model can be employed instead without further modifications.
As already reported in the literature, the linear elasticity coefficient
in (
3) can be employed as a parameter for classification purposes with relatively good results when the mechanical properties of the involved tissues are very different (e.g., a healthy brain tissue versus a brain tumor, which is approximately 10 times more rigid than the healthy tissue). However, for tissues with similar stiffness the linear elasticity coefficient is no longer accurate to be used as a classifier parameter. In this work, we have found that the nonlinear elasticity coefficient
in (
4) serves as a better input to be used in a BC. Furthermore, if both estimated elasticity coefficients are combined in a SBC given in (
21), the tissue classification becomes even more accurate.
Although the parameter estimation given by (
17) is, strictly speaking, carried out
offline, since the experiment duration was carefully chosen to be the shortest possible time (
for the results presented in
Section 3), the acquired data are relatively small and thus the parameters are obtained almost instantaneously in a practical scenario. The proposed method can be directly employed in a teleoperation scheme, in which the physician would not only be able to feel the patient’s tissue due the availability of the estimated force to be employed as a feedback to the operator, but they could “ask” if a particular zone of interest has different mechanical properties with respect to a previously explored one.
Finally, the authors are aware that the experimental study presented in this work is far from being of a practical application. This is mainly due to the limitations of the robot employed for the experiments, for which only the first three of its six degrees of freedom are actuated. Notice however, that the objective of the experiments was to validate the viability of the proposed approach.
5. Conclusions
The method proposed in this work is suitable for identifying biomechanical parameters of soft tissues without the need for a force sensor nor direct velocity measurements. To avoid any damage to the tissue, a closed-loop force control is also proposed, which is in turn based on the estimated forces. The approximate parameters estimated by the proposed algorithm can be employed for a realistic simulation of the studied tissues, mathematical analysis of the interaction, and so on. In addition, the estimated parameters can be employed in a real-time procedure for the classification of tissues with different mechanical properties, which can potentially help the physicians to detect abnormalities such as tumors. Moreover, in this work, we have found that a nonlinear viscoelastic model can outperform the results obtained with the commonly employed linear model when the tissues have very similar mechanical characteristics. Finally, if these two models are combined, the classification is further improved.
As for future work, there remains the need to include the approach proposed in this work in a teleoperation scheme, aiming towards its application in a real scenario. The study of other models to represent the tissue mechanics will be explored as well. The application of the proposed scheme for other procedures such as needle insertion will be also investigated.