Chlorine Concentration Modelling and Supervision in Water Distribution Systems
Abstract
1. Introduction
2. Materials and Methods
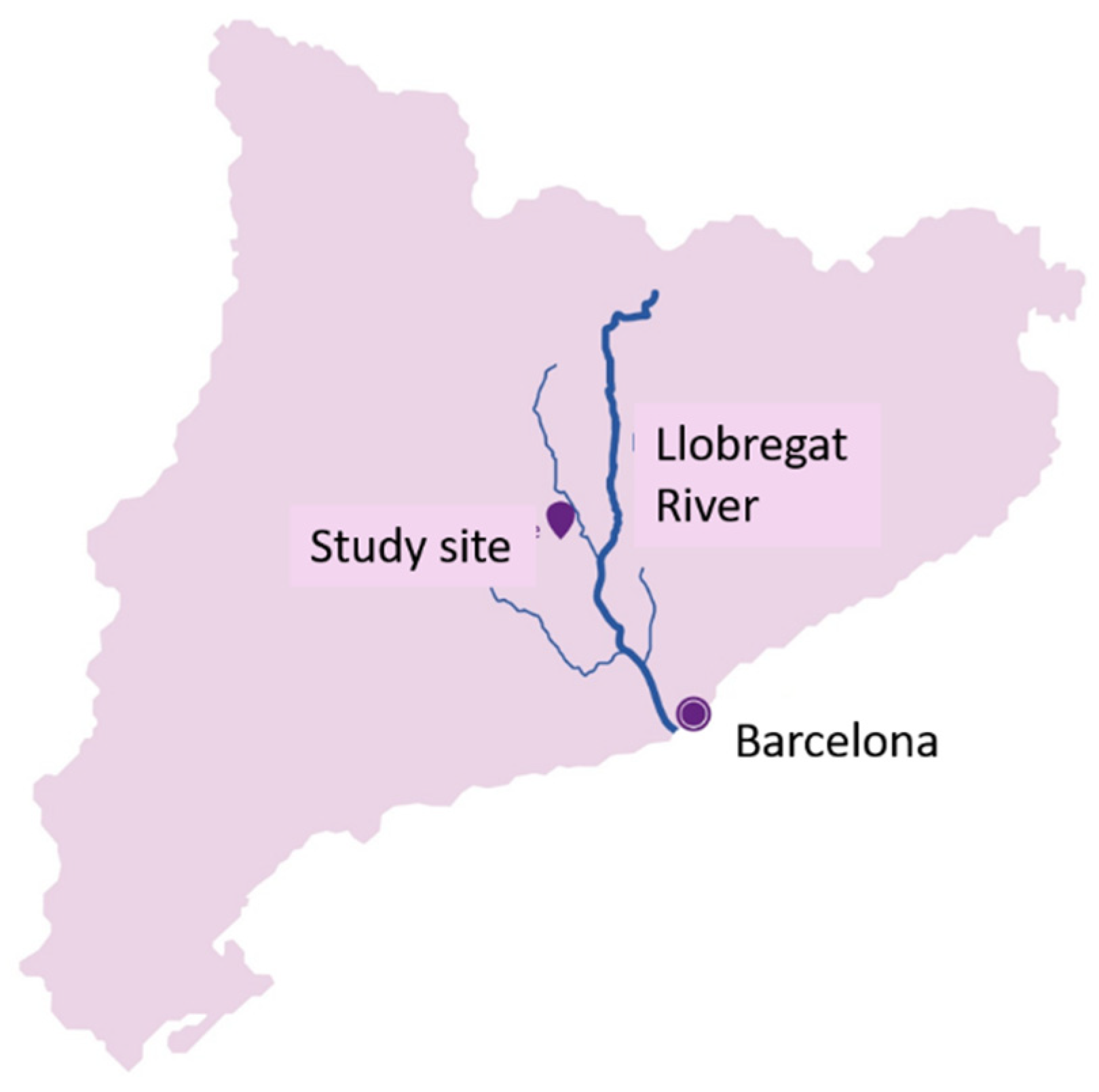
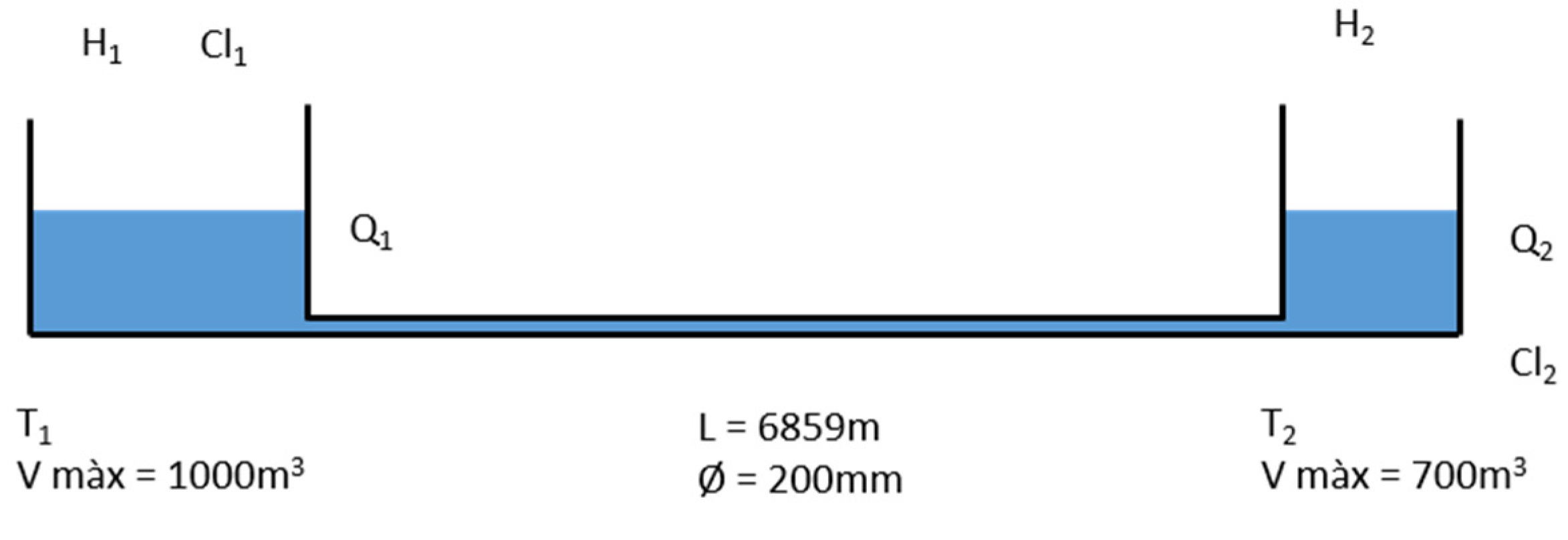
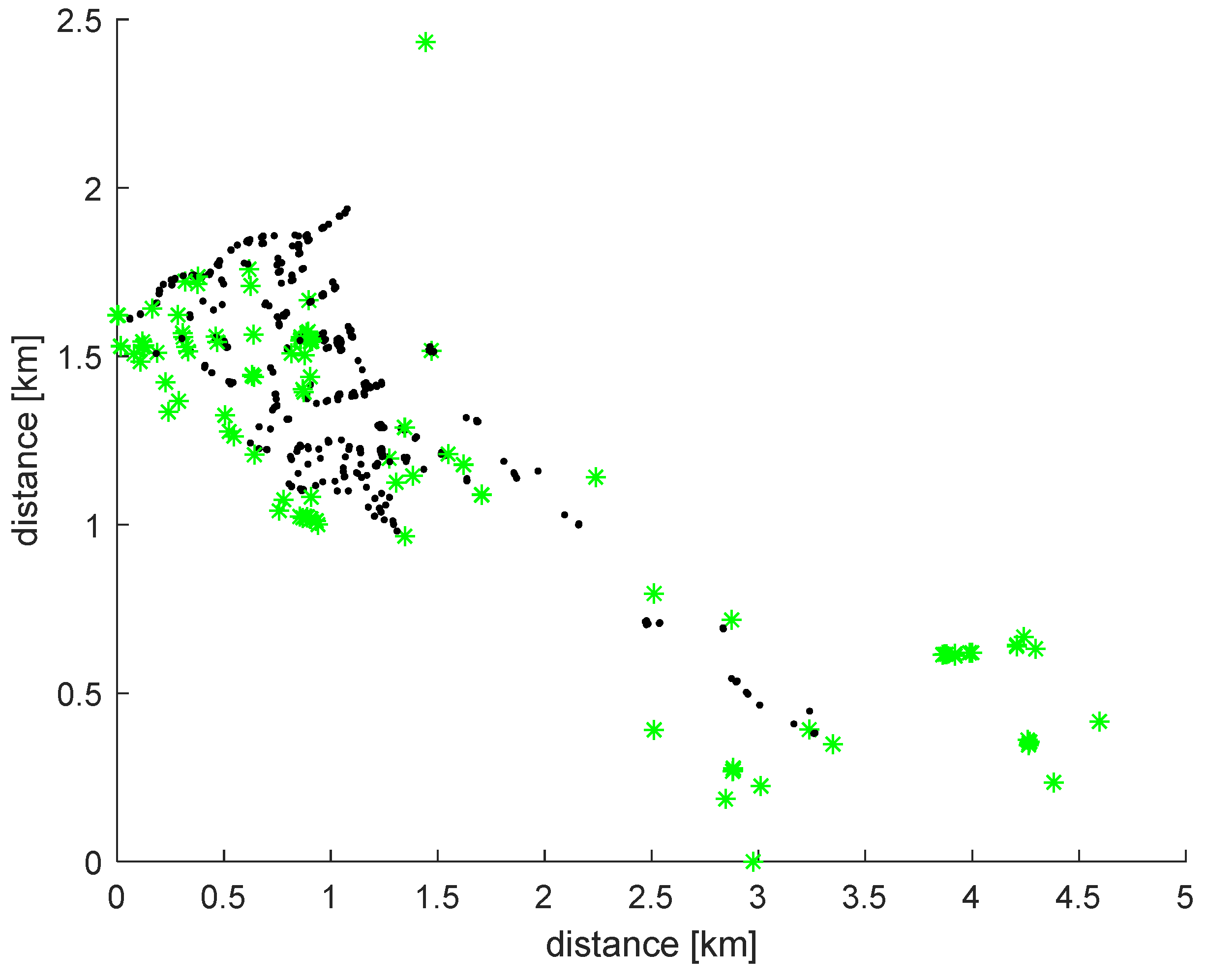
2.1. Case Study Network
2.2. On-Line Calibration
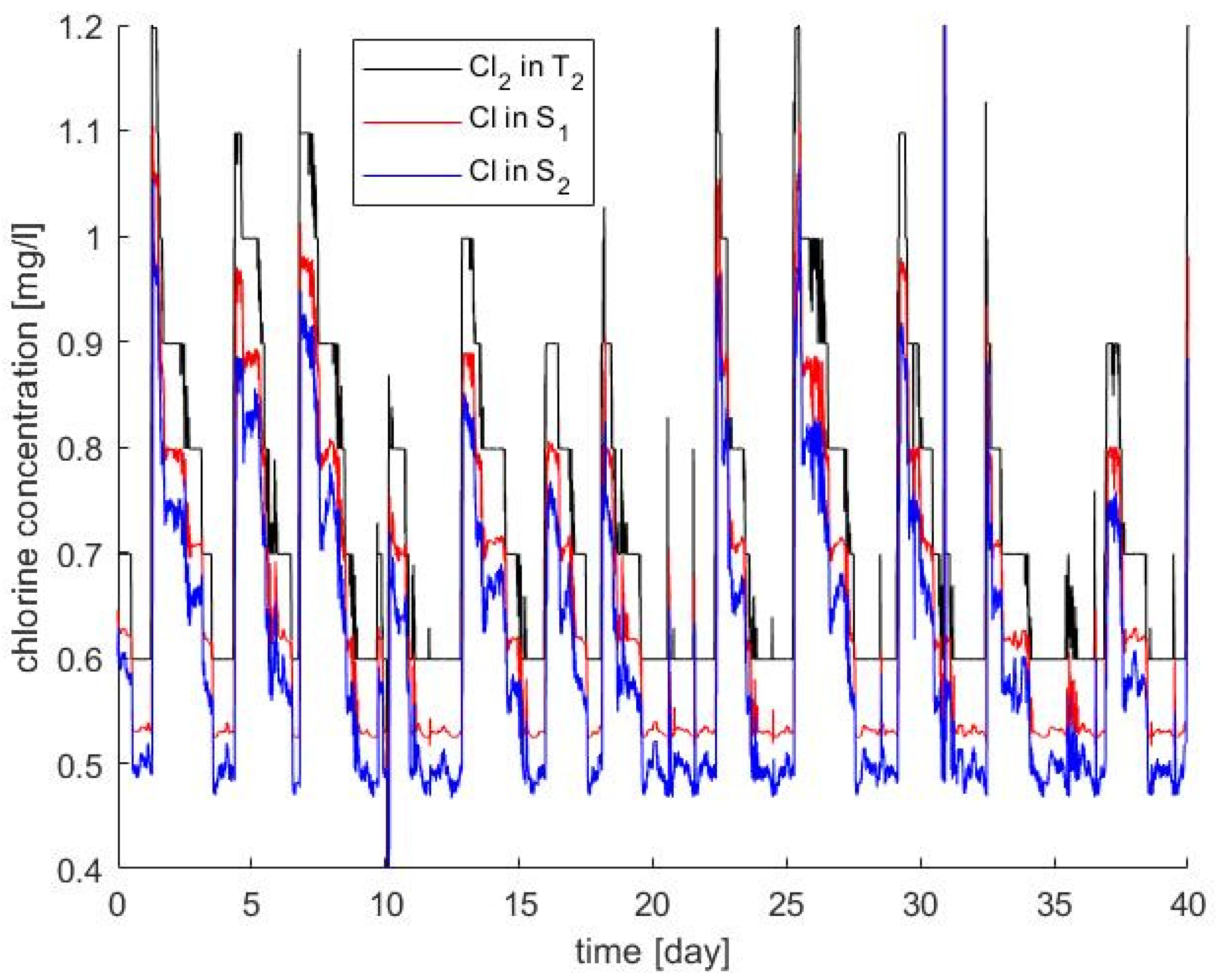
2.3. Chlorine Decay Calibration and Validation
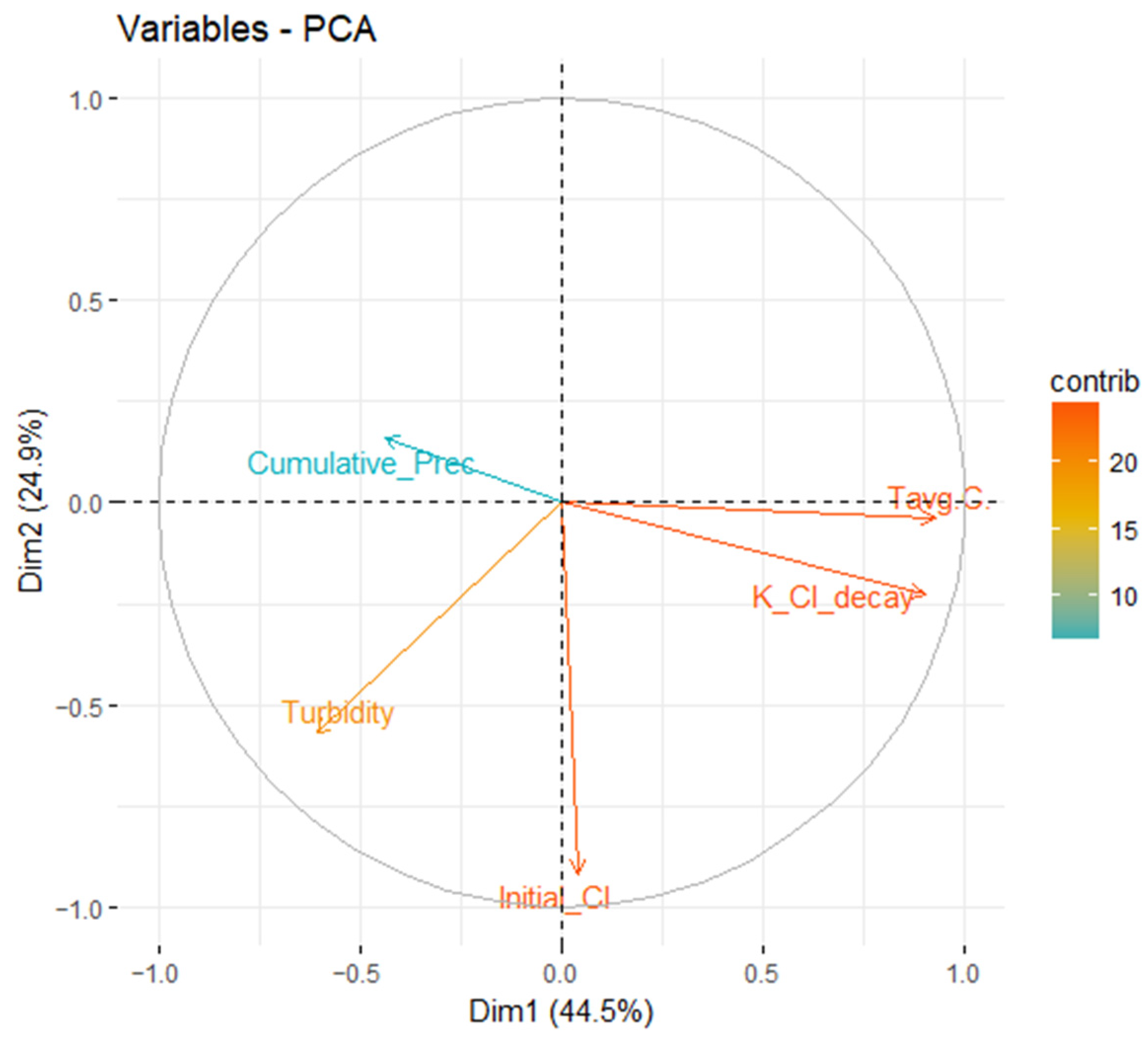
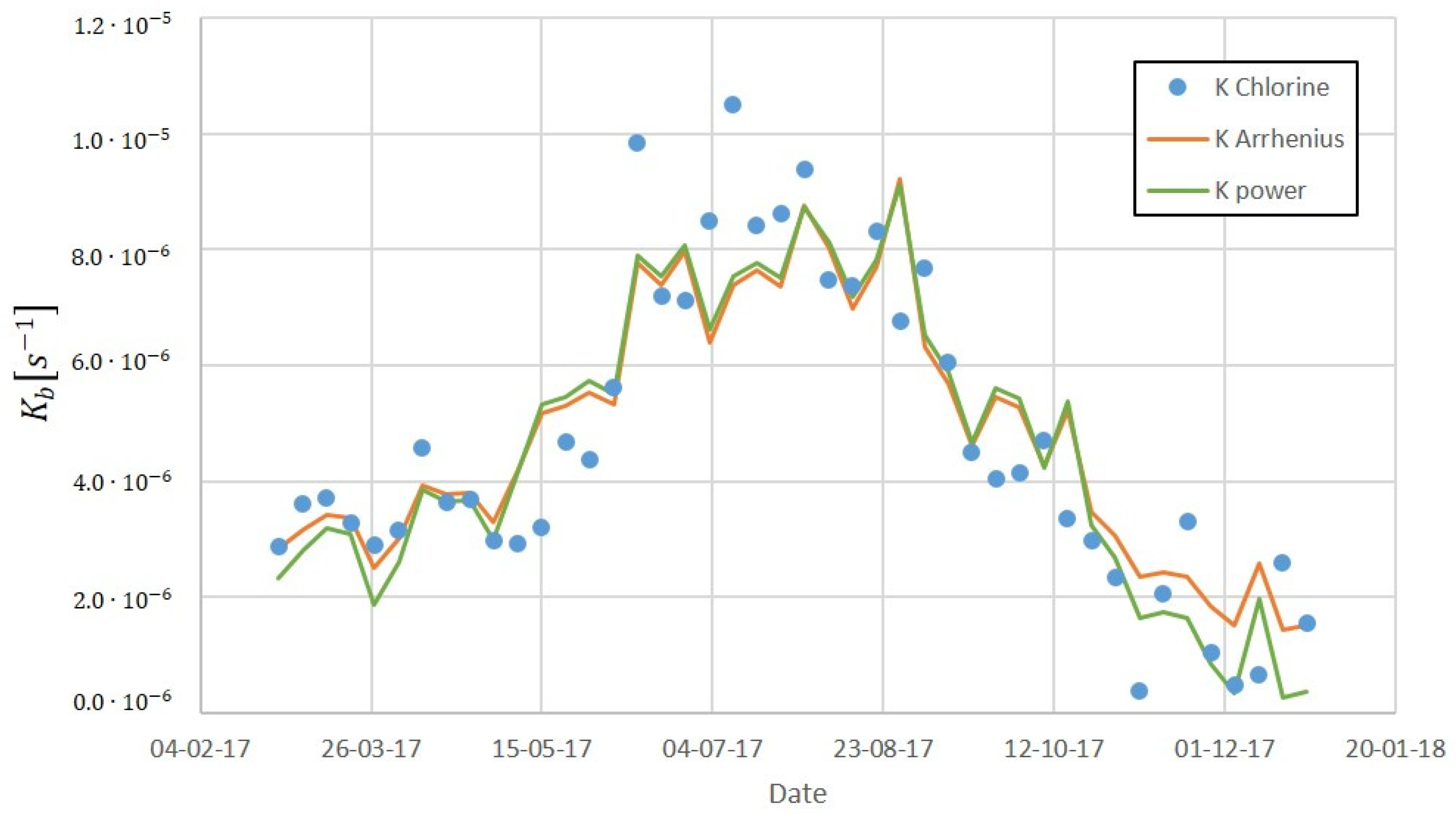
2.4. Parametrised Chlorine Decay Model
3. Results and Discussion
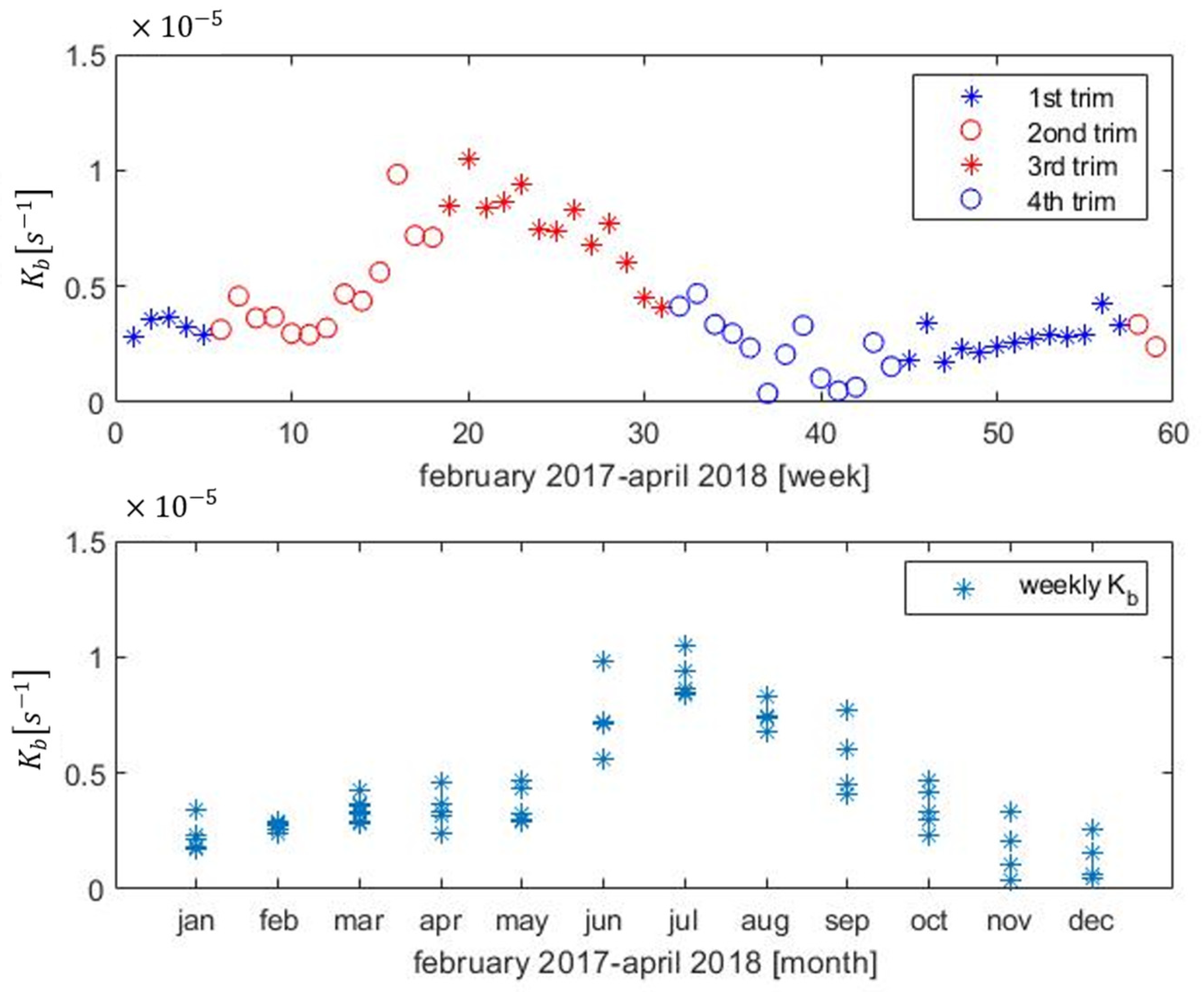
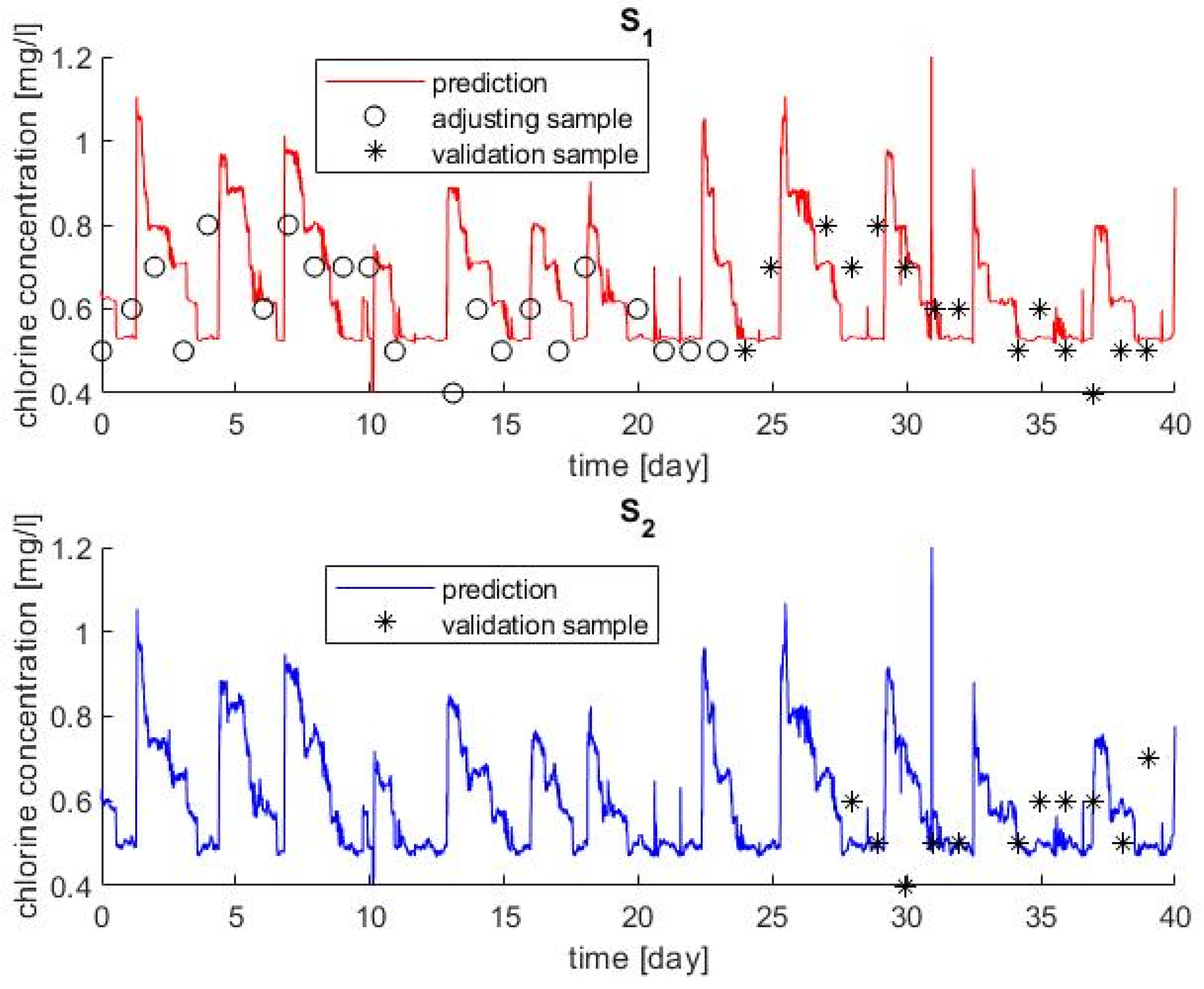
3.1. On-Line Calibration
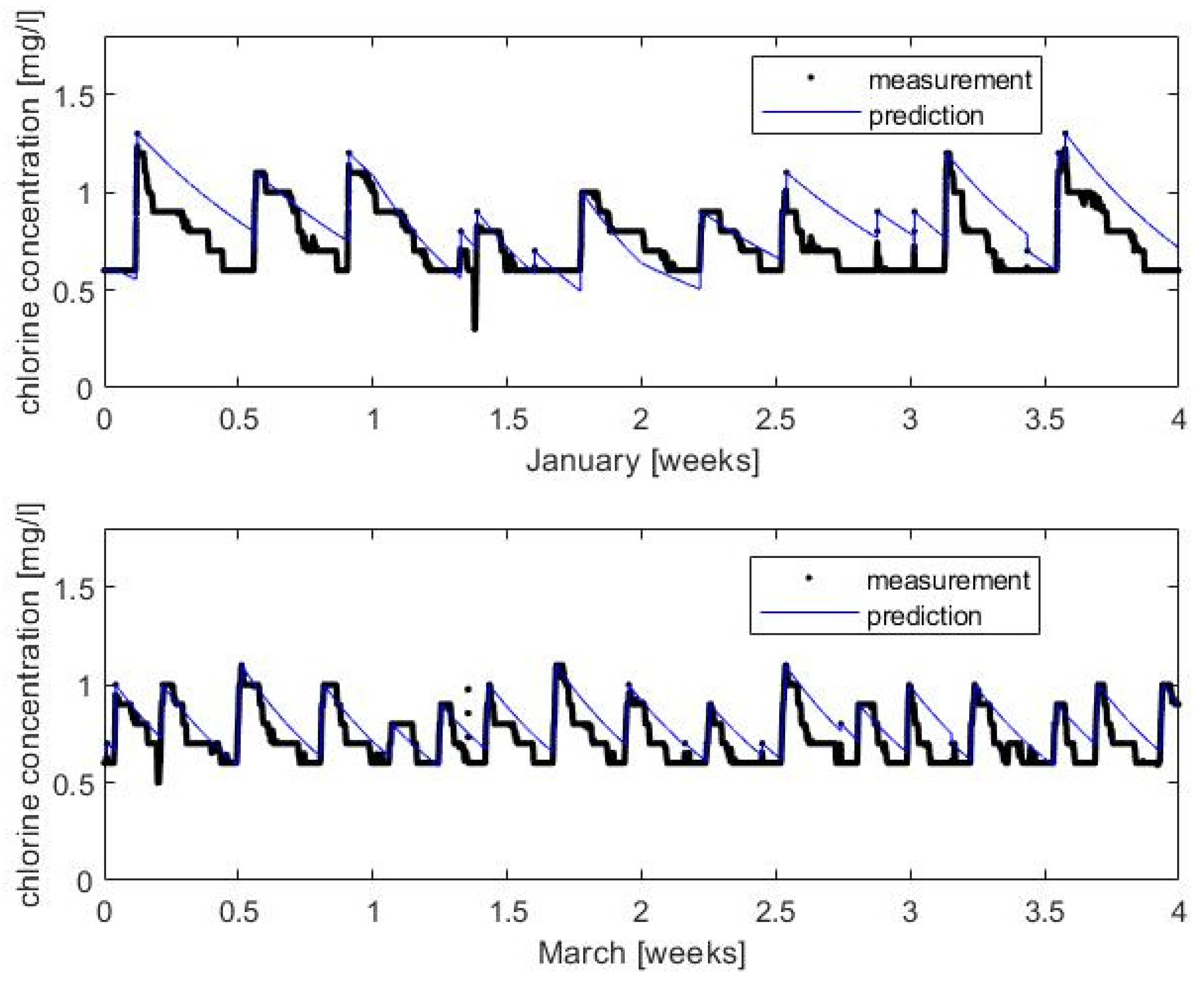
3.2. Chlorine Decay Validation
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Tsitsifli, S.; Kanakoudis, V. Disinfection Impacts to Drinking Water Safety–A Review. EWaS3 2018, 2, 603. [Google Scholar] [CrossRef]
- Mazhar, M.A.; Khan, N.A.; Ahmed, S.; Khan, A.H.; Hussain, A.; Rahisuddin; Changani, F.; Yousefi, M.; Ahmadi, S.; Vambol, V. Chlorination disinfection by-products in municipal drinking water–A review. J. Clean. Prod. 2020, 273, 123159. [Google Scholar] [CrossRef]
- Richardson, S.D.; Plewa, M.J.; Wagner, E.D.; Schoeny, R.; DeMarini, D.M. Occurrence, genotoxicity, and carcinogenicity of regulated and emerging disinfection by-products in drinking water: A review and roadmap for research. Mutat. Res. Rev. Mutat. Res. 2007, 636, 178–242. [Google Scholar] [CrossRef] [PubMed]
- European Commission. Directive (EU) 2020/2184 of the European Parlliament and of the Council of 16 December 2020 on the quality of water intended for human consumption. Off. J. Eur. Union 2020, 2019, 1–61. [Google Scholar]
- Wang, Y. Optimization of booster chlorination for water distribution system under dual uncertainties. Urban Water J. 2022, 19, 363–373. [Google Scholar] [CrossRef]
- Tiruneh, A.T.; Debessai, T.Y.; Bwembya, G.C.; Nkambule, S.J. A Mathematical Model for Variable Chlorine Decay Rates in Water Distribution Systems. Model. Simul. Eng. 2019, 2019, 5863905. [Google Scholar] [CrossRef]
- Walski, M.T.; Chase, V.D.; Savic, A.D.; Grayman, W.; Beckwith, S.; Koelle, E. Methods. In Advanced Water Distribution Modeling and Management, 1st ed.; Bentley Institute Press: Exton, PA, USA, 2007. [Google Scholar]
- Puig, V.; Ocampo-Martínez, C.; Pérez, R.; Cembrano, G.; Quevedo, J.; Escobet, T. Real-Time Monitoring and Operational Control of DrinkingWater Systems; Springer Nature: Berlin/Heidelberg, Germany, 2017. [Google Scholar]
- Shahamat, Y.D.; Godini, K.; Khademi, S.M.R.S.; Rahmani, K.; Rahmani, A. Investigation of chlorine decay of water resource in khanbebein city, Golestan, Iran. Int. J. Environ. Health Eng. 2013, 2, 11. [Google Scholar] [CrossRef]
- Kowalska, B.; Kowalski, D.; Musz, A. Chlorine decay in water distribution systems. Env. Prot. Eng. 2006, 32, 5–16. [Google Scholar]
- Clark, R.M.; Sivaganesan, M. Predicting Chlorine Residuals in Drinking Water: Second Order Model. J. Water Resour. Plan. Manag. 2002, 128, 152–161. [Google Scholar] [CrossRef]
- Hua, P.; Vasyukova, E.; Uhl, W. A variable reaction rate model for chlorine decay in drinking water due to the reaction with dissolved organic matter. Water Res. 2015, 75, 109–122. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Reckhow, D.A.; Li, Y. A two-site chlorine decay model for the combined effects of pH, water distribution temperature and in-home heating profiles using differential evolution. Water Res. 2014, 53, 47–57. [Google Scholar] [CrossRef] [PubMed]
- Arevalo, J. Modeling Free Chlorine and Chloramine Decay in a Pilot Distribution System; University of Central Florida: Orlando, FL, USA, 2007. [Google Scholar]
- Hassan, K.Z.A.; Bower, K.C.; Miller, C.M. Iron Oxide Enhanced Chlorine Decay and Disinfection By-Product Formation. J. Environ. Eng. 2006, 132, 1609–1616. [Google Scholar] [CrossRef]
- Seyoum, A.G.; Tanyimboh, T.T. Integration of Hydraulic and Water Quality Modelling in Distribution Networks: EPANET-PMX. Water Resour. Manag. 2017, 31, 4485–4503. [Google Scholar] [CrossRef]
- Fisher, I.; Kastl, G.; Sathasivan, A. Evaluation of suitable chlorine bulk-decay models for water distribution systems. Water Res. 2011, 45, 4896–4908. [Google Scholar] [CrossRef] [PubMed]
- Xie, X.; Zeng, B.; Nachabe, M. Sampling design for water distribution network chlorine decay calibration. Urban Water J. 2015, 12, 190–199. [Google Scholar] [CrossRef]
- Nejjari, F.; Puig, V.; Pérez, R.; Quevedo, J.; Cugueró, M.; Sanz, G.; Mirats, J. Chlorine Decay Model Calibration and Comparison: Application to a Real Water Network. Procedia Eng. 2014, 70, 1221–1230. [Google Scholar] [CrossRef][Green Version]
- Anderson, T.W. An Introduction to Multivariate Statistical Analysis; John Wiley & Sons: Hoboken, New Jersey, USA, 2003. [Google Scholar]
- Chowdhury, S.; Champagne, P.; McLellan, P.J. Factors Influencing Formation of Trihalomethanes in Drinking Water: Results from Multivariate Statistical Investigation of the Ontario Drinking Water Surveillance Program Database. Water Qual. Res. J. 2008, 43, 189–199. [Google Scholar] [CrossRef]
- Courtis, B.J.; West, J.R.; Bridgeman, J. Temporal and Spatial Variations in Bulk Chlorine Decay within a Water Supply System. J. Env. Eng. 2009, 135, 147–152. [Google Scholar] [CrossRef]
- Powell, J.C.; Hallam, N.B.; West, J.R.; Forster, C.F.; Simms, J. Factors which control bulk chlorine decay rates. Water Res. 2000, 34, 117–126. [Google Scholar] [CrossRef]
- Monteiro, L.; Viegas, R.; Covas, D.I.C.; Menaia, J. Modelling chlorine residual decay as influenced by temperature. Water Environ. J. 2015, 29, 331–337. [Google Scholar] [CrossRef]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pérez, R.; Martínez-Torrents, A.; Martínez, M.; Grau, S.; Vinardell, L.; Tomàs, R.; Martínez-Lladó, X.; Jubany, I. Chlorine Concentration Modelling and Supervision in Water Distribution Systems. Sensors 2022, 22, 5578. https://doi.org/10.3390/s22155578
Pérez R, Martínez-Torrents A, Martínez M, Grau S, Vinardell L, Tomàs R, Martínez-Lladó X, Jubany I. Chlorine Concentration Modelling and Supervision in Water Distribution Systems. Sensors. 2022; 22(15):5578. https://doi.org/10.3390/s22155578
Chicago/Turabian StylePérez, Ramon, Albert Martínez-Torrents, Manuel Martínez, Sergi Grau, Laura Vinardell, Ricard Tomàs, Xavier Martínez-Lladó, and Irene Jubany. 2022. "Chlorine Concentration Modelling and Supervision in Water Distribution Systems" Sensors 22, no. 15: 5578. https://doi.org/10.3390/s22155578
APA StylePérez, R., Martínez-Torrents, A., Martínez, M., Grau, S., Vinardell, L., Tomàs, R., Martínez-Lladó, X., & Jubany, I. (2022). Chlorine Concentration Modelling and Supervision in Water Distribution Systems. Sensors, 22(15), 5578. https://doi.org/10.3390/s22155578






