Effects of Contact Pressure in Reflectance Photoplethysmography in an In Vitro Tissue-Vessel Phantom
Abstract
1. Introduction
- How much is the degree of change in PPG signal features that an increase in sensor CP can create?
- What are the PPG signal features that are most affected?
- Is the change in a particular PPG signal feature significant enough to create misinterpretations when deriving diagnostic indices?
2. Materials and Methods
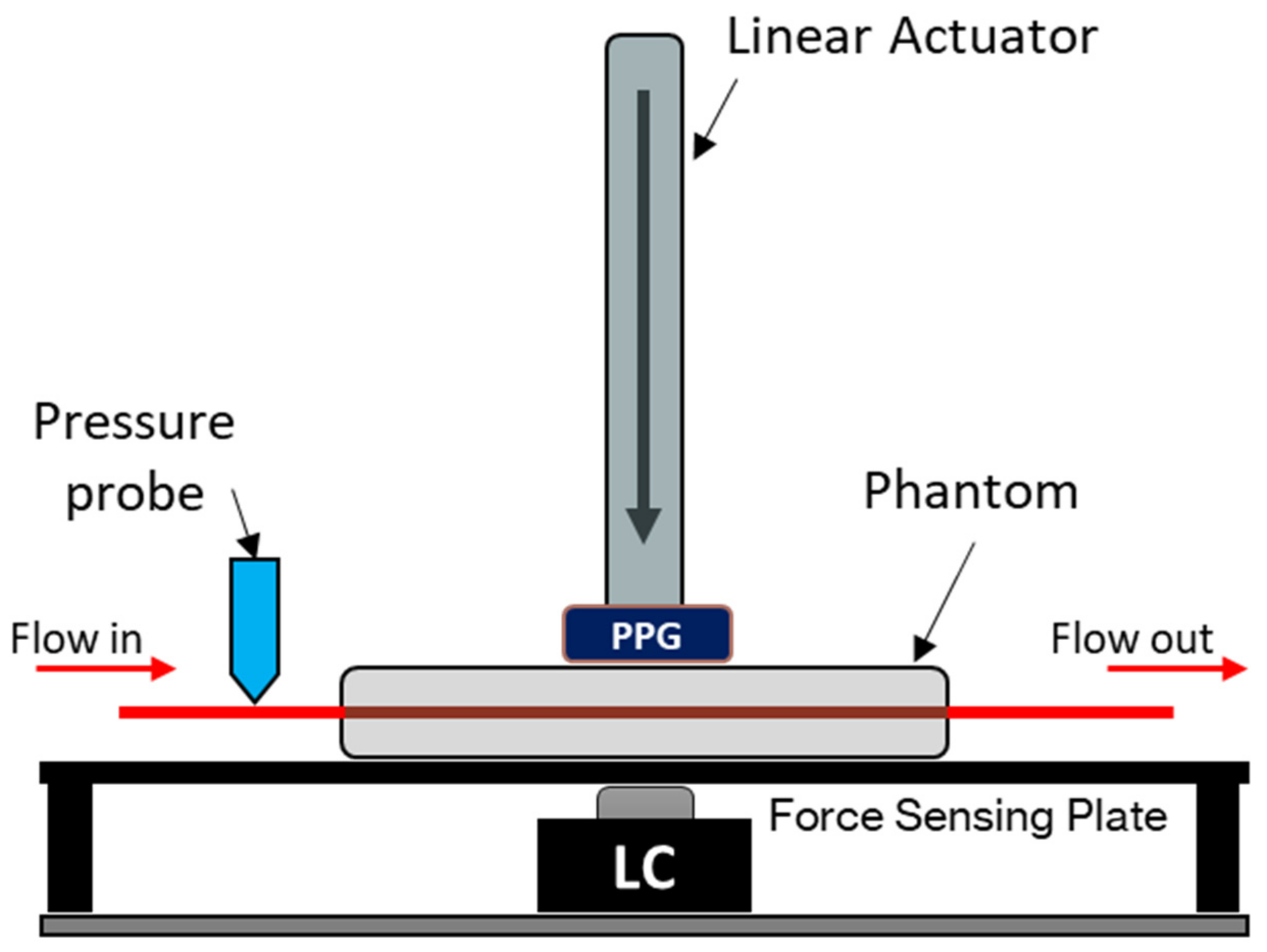
2.1. Pulsatile Pump and Tissue Phantom
2.2. Experiment Protocol
2.3. Analysis Protocol
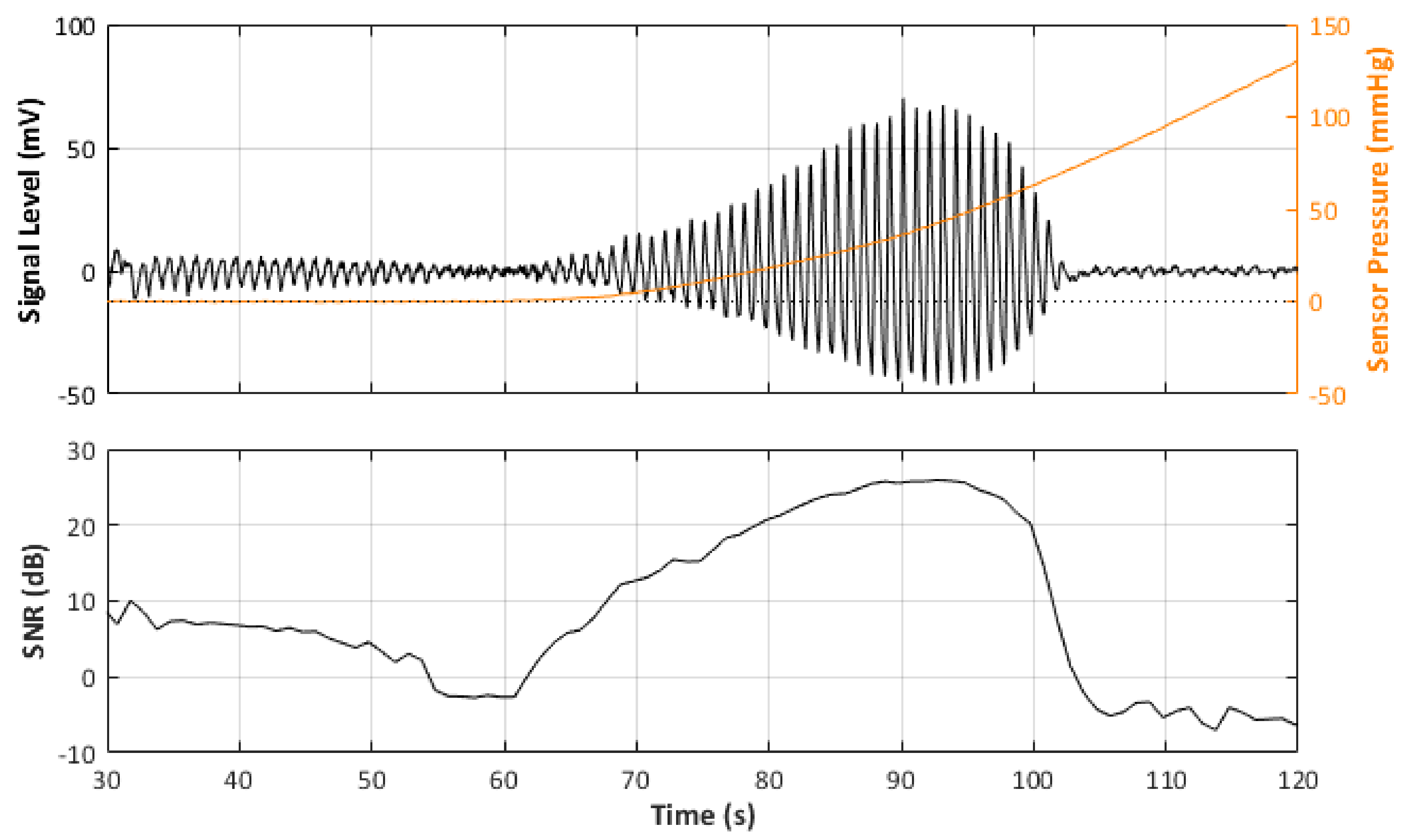
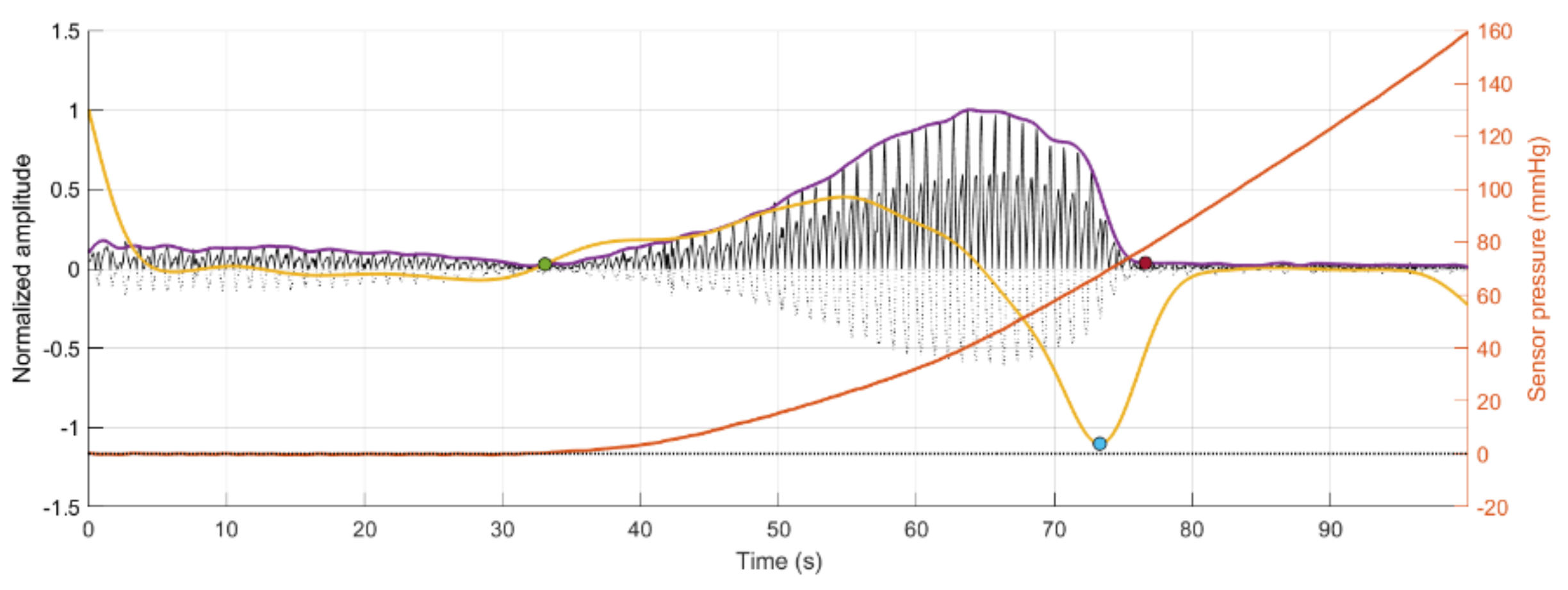
2.3.1. PPG SNR Analysis
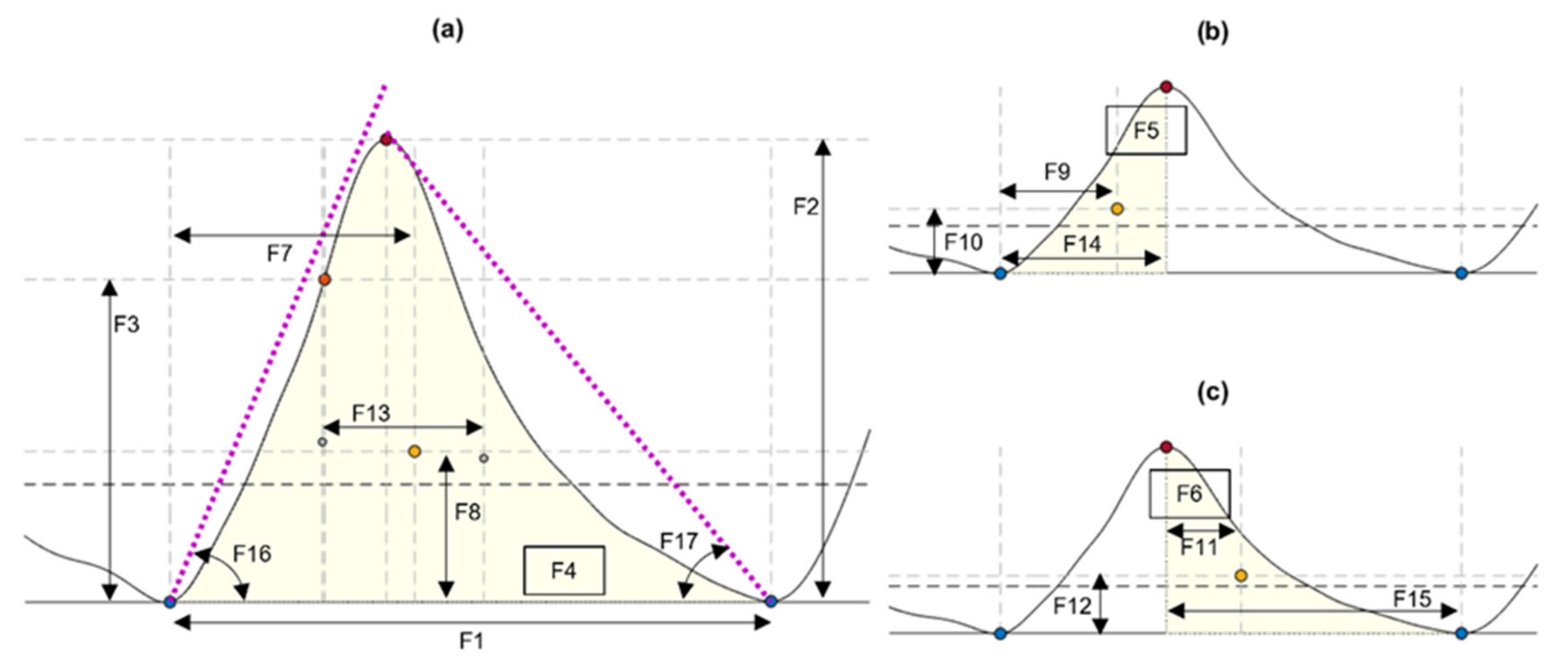
2.3.2. PPG Features Analysis
3. Results
3.1. PPG SNR vs. Contact Pressure
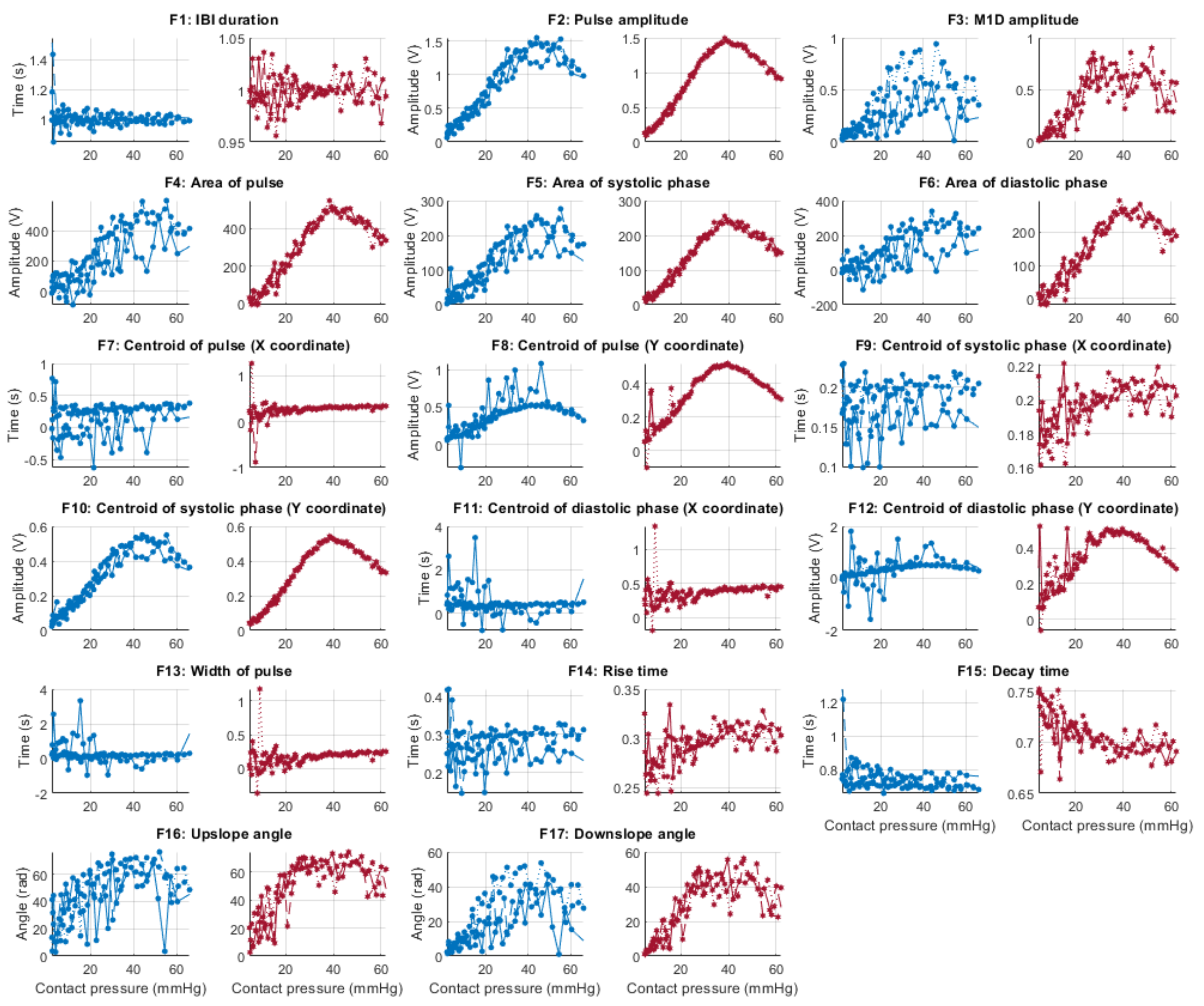
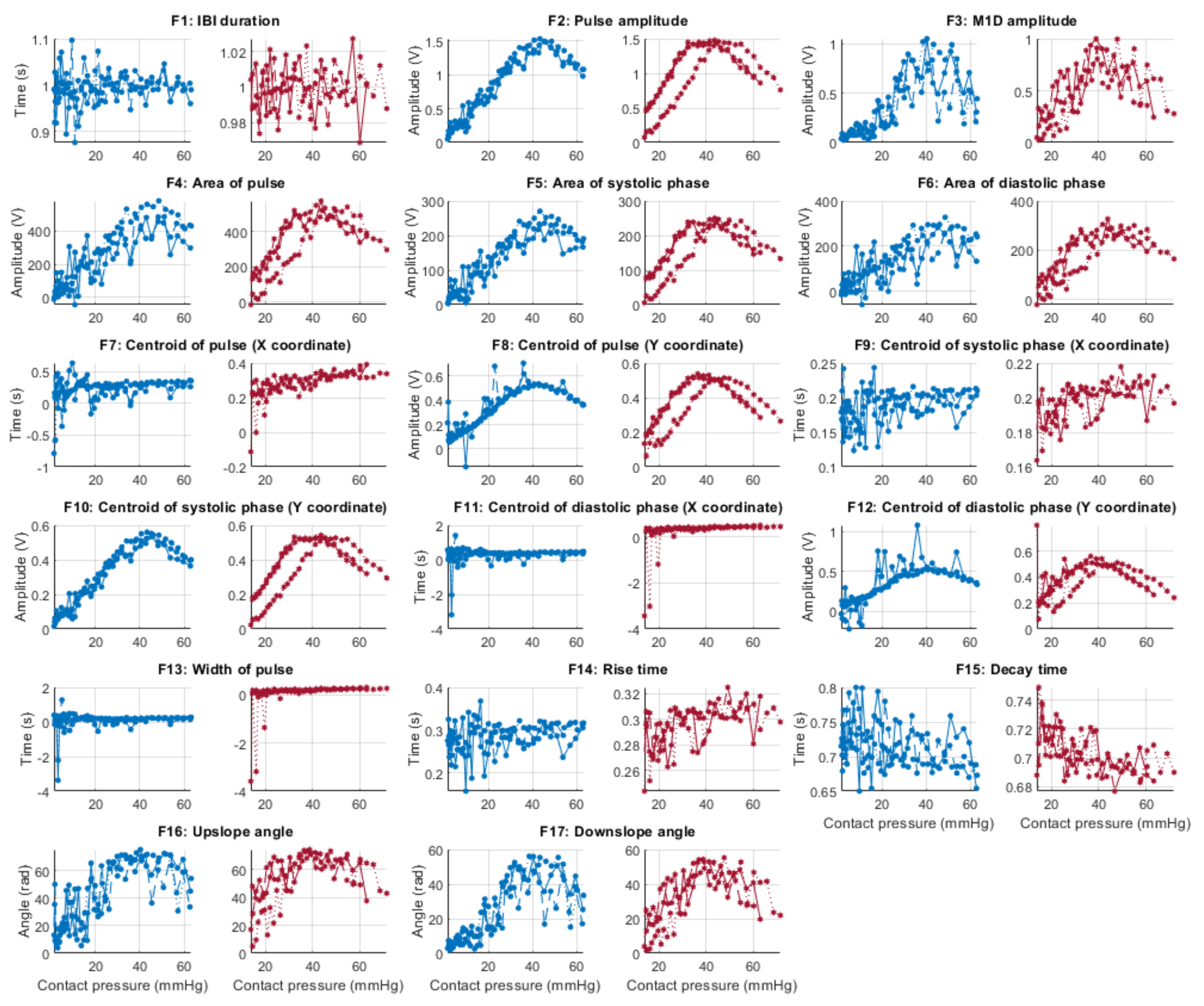
3.2. Effect of Contact Pressure on PPG Signal Features
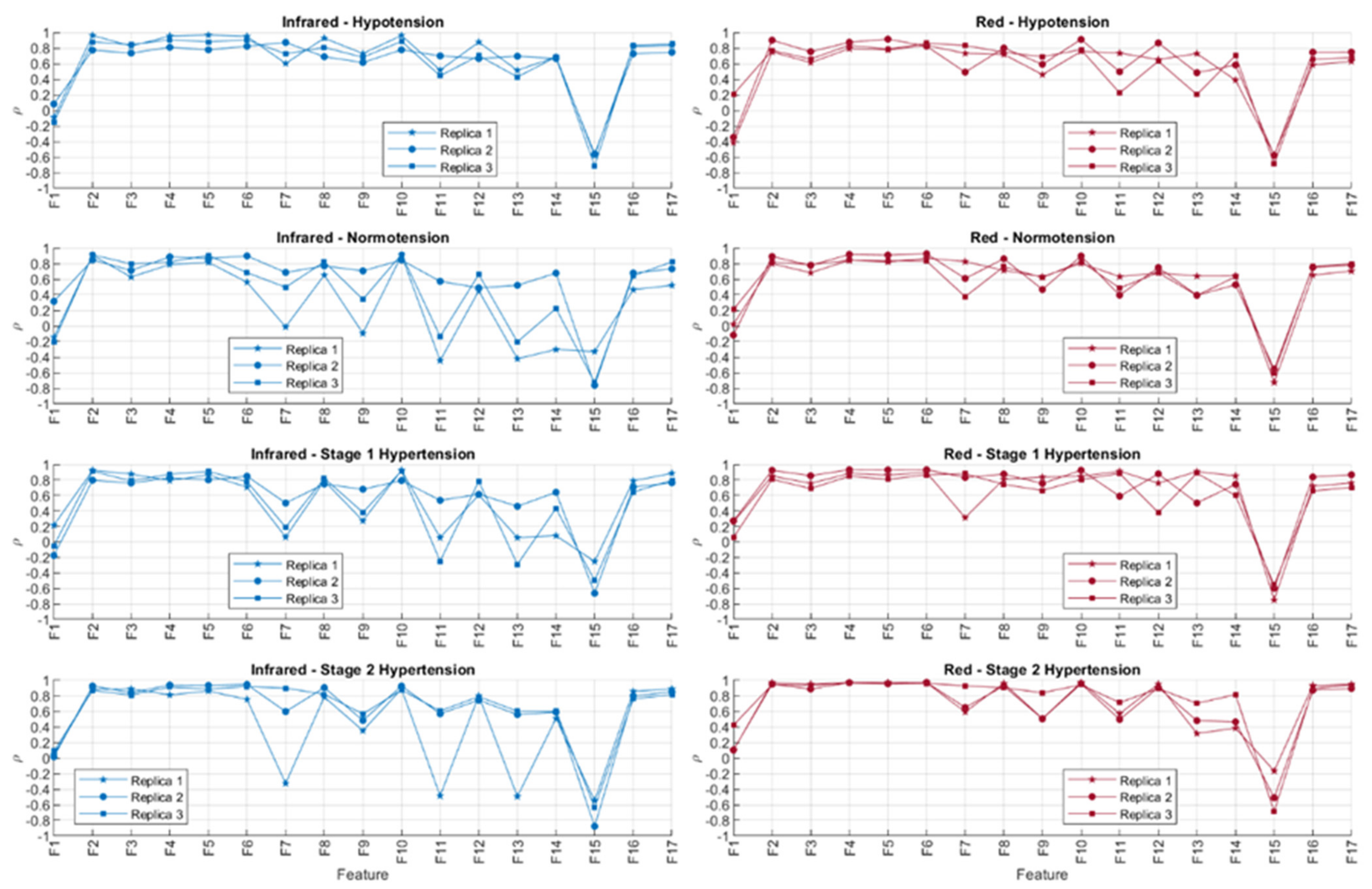
3.3. Statistical Analysis
4. Discussion
4.1. SNR Analysis
4.2. PPG Feature Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kyriacou, P.A.; Budidha, K.; Abay, T.Y. Optical techniques for blood and tissue oxygenation. Encycl. Biomed. Eng. 2019, 3, 461–472. [Google Scholar]
- Chatterjee, S.; Kyriacou, P.A. Investigating the influence of blood-vessel depth and sensor location on the light-tissue interactions using a Monte Carlo model. Int. J. Innov. Res. Phys. 2020, 2, 31–35. [Google Scholar] [CrossRef]
- Feiner, J.R.; Severinghaus, J.W.; Bickler, P.E. Dark skin decreases the accuracy of pulse oximeters at low oxygen saturation: The effects of oximeter probe type and gender. Anesth. Analg. 2007, 105 (Suppl. 6), S18–S23. [Google Scholar] [CrossRef]
- Teng, X.F.; Zhang, Y. The effect of contacting force on photoplethysmographic signals. Physiol. Meas. 2004, 25, 1323–1335. [Google Scholar] [CrossRef]
- Grabovskis, A.; Marcinkevics, Z.; Rubins, U.; Kviesis-Kipge, E. Effect of probe contact pressure on the photoplethysmographic assessment of conduit artery stiffness. J. Biomed. Opt. 2013, 18, 027004. [Google Scholar] [CrossRef]
- Shimazaki, T.; Kuwahara, Y.; Kimoto, M.; Hara, S.; Yomo, H. Effect of Position and Fastening Belt Pressure on the Accuracy of PPG-Based Heart Rate Sensor. In Proceedings of the 2018 40th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Honolulu, HI, USA, 18–21 July 2018; pp. 4323–4326. [Google Scholar]
- Kasbekar, R.S.; Mendelson, Y. Evaluation of key design parameters for mitigating motion artefact in the mobile reflectance PPG signal to improve estimation of arterial oxygenation. Physiol. Meas. 2018, 39, 075008. [Google Scholar] [CrossRef]
- Lee, J.-H.; Yang, S.; Park, J.; Kim, H.C.; Kim, E.-H.; Jang, Y.-E.; Kim, J.-T.; Kim, H.-S. Time to consider the contact force during photoplethysmography measurement during pediatric anesthesia: A prospective, nonrandomized interventional study. Pediatr. Anesthesia 2018, 28, 660–667. [Google Scholar] [CrossRef]
- Scardulla, F.; D’Acquisto, L.; Colombarini, R.; Hu, S.; Pasta, S.; Bellavia, D. A Study on the Effect of Contact Pressure during Physical Activity on Photoplethysmographic Heart Rate Measurements. Sensors 2020, 20, 5052. [Google Scholar] [CrossRef]
- Looga, R. The Valsalva manoeuvre—Cardiovascular effects and performance technique: A critical review. Respir. Physiol. Neurobiol. 2005, 147, 39–49. [Google Scholar] [CrossRef]
- Pstras, L.; Thomaseth, K.; Waniewski, J.; Balzani, I.; Bellavere, F. The Valsalva manoeuvre: Physiology and clinical examples. Acta Physiol. 2016, 217, 103–119. [Google Scholar] [CrossRef]
- Nomoni, M.; May, J.M.; Kyriacou, P.A. Novel Polydimethylsiloxane (PDMS) Pulsatile Vascular Tissue Phantoms for the In-Vitro Investigation of Light Tissue Interaction in Photoplethysmography. Sensors 2020, 20, 4246. [Google Scholar] [CrossRef]
- Rybynok, V.; May, J.M.; Budidha, K.; Kyriacou, P.A. Design and development of a novel multi-channel photoplethysmographic research system. In Proceedings of the 2013 IEEE Point-of-Care Healthcare Technologies (PHT), Bangalore, India, 16–18 January 2013; pp. 267–270. [Google Scholar] [CrossRef]
- Budidha, K.; Rybynok, V.; Kyriacou, P.A. Design and Development of a Modular, Multichannel Photoplethysmography System. IEEE Trans. Instrum. Meas. 2018, 67, 1954–1965. [Google Scholar] [CrossRef]
- Orphanidou, C. Signal Quality Assessment in Physiological Monitoring; Springer International Publishing: Cham, Switzerland, 2017; Available online: http://hdl.handle.net/2078/ebook:124925 (accessed on 9 September 2021).
- Li, B.N.; Dong, M.C.; Vai, M.I. On an automatic delineator for arterial blood pressure waveforms. Biomed. Signal Process. Control. 2010, 5, 76–81. [Google Scholar] [CrossRef]
- Hemon, M.C.; Phillips, J.P. Comparison of foot finding methods for deriving instantaneous pulse rates from photoplethysmographic signals. J. Clin. Monit. 2015, 30, 157–168. [Google Scholar] [CrossRef]
- Mejía-Mejía, E.; May, J.M.; Torres, R.; Kyriacou, P.A. Pulse rate variability in cardiovascular health: A review on its applications and relationship with heart rate variability. Physiol. Meas. 2020, 41, 07TR01. [Google Scholar] [CrossRef] [PubMed]
- Moore, D.J.; Maher, T.; Kingston, V.; Shanik, G. Assessment of venous function using photoplethysmography. Ir. J. Med. Sci. 1982, 151, 308–312. [Google Scholar] [CrossRef] [PubMed]
- Evans, M.L.; Geddes, L.A. An assessment of blood vessel vasoactivity using photoplethysmography. Med. Instrum. 1988, 22, 29–32. [Google Scholar] [PubMed]
- Lees, T.A.; Lambert, D. Patterns of venous reflux in limbs with skin changes associated with chronic venous insufficiency. BJS 2005, 80, 725–728. [Google Scholar] [CrossRef] [PubMed]
- Hahn, M.; Hahn, C.; Jünger, M.; Steins, A.; Zuder, D.; Klyscz, T.; Büchtemann, A.; Rassner, G.; Blazek, V. Local Cold Exposure Test with a New Arterial Photoplethysmographic Sensor in Healthy Controls and Patients with Secondary Raynaud’s Phenomenon. Microvasc. Res. 1999, 57, 187–198. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, A.A.; Latty, E.M.; Chivers, M.L.; Bailey, J.M. Measurement of Sexual Arousal in Postoperative Male-to-Female Transsexuals Using Vaginal Photoplethysmography. Arch. Sex. Behav. 2005, 34, 135–145. [Google Scholar] [CrossRef]
- Jönsson, B.; Laurent, C.; Eneling, M.; Skau, T.; Lindberg, L.-G. Automatic Ankle Pressure Measurements Using PPG in Ankle-brachial Pressure Index Determination. Eur. J. Vasc. Endovasc. Surg. 2005, 30, 395–401. [Google Scholar] [CrossRef][Green Version]
- Alian, A.A.; Shelley, K.H. Photoplethysmography. Best Pract. Res. Clin. Anaesthesiol. 2014, 28, 395–406. [Google Scholar] [CrossRef]
- O’Rourke, M. Mechanical Principles in Arterial Disease. Hypertens 1995, 26, 2–9. [Google Scholar] [CrossRef]
- Holzapfel, G.A.; Gasser, T.C.; Ogden, R.W. A New Constitutive Framework for Arterial Wall Mechanics and a Comparative Study of Material Models. J. Elast. 2000, 61, 1–48. [Google Scholar] [CrossRef]
- Dobrin, P.B. Mechanical properties of arteries. Physiol. Rev. 1978, 58, 397–460. [Google Scholar] [CrossRef] [PubMed]
- Baker, P.D.; Westenskow, D.R.; Kück, K. Theoretical analysis of non-invasive oscillometric maximum amplitude algorithm for estimating mean blood pressure. Med. Biol. Eng. Comput. 1997, 35, 271–278. [Google Scholar] [CrossRef]
- Chandrasekhar, A.; Kim, C.-S.; Naji, M.; Natarajan, K.; Hahn, J.-O.; Mukkamala, R. Smartphone-based blood pressure monitoring via the oscillometric finger-pressing method. Sci. Transl. Med. 2018, 10. [Google Scholar] [CrossRef]



| Study (1st Author) | Sensor Location | Optimum Pressure/Force Reported | Conversion to mmHg | Research Question | Findings |
|---|---|---|---|---|---|
| Teng 1 | Finger | 0.2–1.0 N | 21–105 mmHg A | Change in AC/DC ratio with change in contact force as an important metric when calculating blood oxygen saturation | PPG does have an optimum or “peak” value for the contact force applied. Hence careful sensor design consideration is required. |
| Grabovskis 2 | Posterior Tibial A., Femoral A., Popliteal A., | 10.9, 11.8, 15.2 kPa | 81, 88, 114 mmHg B | The effect of probe contact pressure (CP) on the PPG signal for assessing arterial stiffness | Wrong contact pressure would adversely affect the AC PPG 2nd derivative peak ratio (known as the b/a ratio), a measurement index to assess arterial function. Also, suggests an optimal contact pressure. |
| Shimazaki 3 | Forearm, Wrist | 40–50 hPa at both locations | 30–37 mmHg C | The effect of fastening or applying contact pressure in wearable devices such as wristwatches which employ PPG to measure heart rate. Motion artefact reduction during exercise, elevated respiration artefact and accuracy of heart rate prediction were the key parameters investigated | All these studies reported that CP has a significant impact (i) reducing the noise introduced by motion artefact during exercise, (ii) increasing respiration related modulations in PPG, and (iii) increasing error in heart rate calculation up to ±11 beats per minute. Additionally, studies confirmed that further optimisation of the CP is indeed needed to reliably calculate physiological parameters. |
| Kasbekar 4 | Forehead, Wrist | 12 kPa (Forehead) | 90 mmHg (Forehead) B | ||
| Lee 5 | Index Finger (Paediatric Study, mean age = 4.1 y) | 0.4–0.6 N | 5.9–8.8 mmHg D | ||
| Scardulla 6 | Wristband | 54 mmHg | NA |
| In Vitro Blood Pressure (mmHg) | |||
|---|---|---|---|
| Blood Pressure State | SBP | DBP | MAP |
| Hypotensive | 93 | 50 | 64 |
| Normotensive | 112 | 72 | 85 |
| Stage 1 Hypertensive | 143 | 104 | 117 |
| Stage 2 Hypertensive | 171 | 132 | 145 |
| Feature | Description (Units of Measurement) |
|---|---|
| F1 | Cycle duration, measured from consecutive TI points (s) |
| F2 | Pulse amplitude, measured as the amplitude difference between the onset of the pulse and its systolic peak (V) |
| F3 | Amplitude of the maximum slope point, measured from the onset of the pulse (V) |
| F4 | Area of the pulse, measured from consecutive TI points (V) |
| F5 | Area of the systolic phase, measured from the onset of the pulse and the location of the systolic peak (V) |
| F6 | Area of the diastolic phase, measured from the location of the systolic peak and the onset of the next pulse (V) |
| F7 | x-coordinate of the centroid of the pulse (s) |
| F8 | y-coordinate of the centroid of the pulse (V) |
| F9 | x-coordinate of the centroid of the systolic phase of the pulse (s) |
| F10 | y-coordinate of the centroid of the systolic phase of the pulse (V) |
| F11 | x-coordinate of the centroid of the diastolic phase of the pulse (s) |
| F12 | y-coordinate of the centroid of the diastolic phase of the pulse (V) |
| F13 | Pulse width, measured as the difference between F9 and F11 (s) |
| F14 | Rise time, measured as the time between the onset of the pulse and the location of the systolic peak (s) |
| F15 | Decay time, measured as the time between the location of the systolic peak and the onset of the next pulse (s) |
| F16 | Upslope angle, measured as the inverse tangent of the triangle formed by F2 and F14 (rad) |
| F17 | Downslope angle, measured as the inverse tangent of the triangle formed by F2 and F15 (rad) |
| Hypotensive | Normotensive | Stage 1 Hypertensive | Stage 2 Hypertensive | |||||
|---|---|---|---|---|---|---|---|---|
| SNR (dB) | Pressure (mmHg) | SNR (dB) | Pressure (mmHg) | SNR (dB) | Pressure (mmHg) | SNR (dB) | Pressure (mmHg) | |
| RED | 26.7 (<1) | 38.6 (1.9) | 26.0 (<1) | 39.3 (<1) | 25.9 (<1) | 35.1 (2.0) | 26.9 (<1) | 48.2 (1.9) |
| IR | 27.0 (<1) | 44.1 (3.5) | 27.3 (<1) | 43.4 (1.7) | 26.0 (<1) | 39.0 (2.3) | 25.8 (<1) | 41.8 (1.8) |
| Hypotension | Normotension | Stage 1 Hypertension | Stage 2 Hypertension | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| R1 | R2 | R3 | R1 | R2 | R3 | R1 | R2 | R3 | R1 | R2 | R3 |
| 1 | 1 | 1 | 7 | 11 | 1 | 13 | 1 | 1 | 1 | 1 | 1 |
| 13 | 13 | 13 | 9 | 1 | 13 | 11 | 7 | 13 | 7 | 9 | 14 |
| 11 | 11 | 7 | 1 | 13 | 11 | 7 | 11 | 11 | 9 | 14 | 13 |
| 15 | 9 | 11 | 14 | 14 | 9 | 14 | 13 | 15 | 11 | 13 | 11 |
| 7 | 14 | 15 | 15 | 9 | 14 | 1 | 9 | 14 | 13 | 11 | 9 |
| 14 | 12 | 14 | 13 | 7 | 15 | 15 | 14 | 9 | 14 | 15 | 15 |
| 9 | 15 | 9 | 11 | 16 | 7 | 9 | 15 | 7 | 15 | 16 | 7 |
| 16 | 7 | 16 | 12 | 12 | 12 | 12 | 16 | 16 | 6 | 12 | 16 |
| 3 | 8 | 17 | 16 | 6 | 16 | 6 | 6 | 3 | 8 | 3 | 3 |
| 17 | 16 | 3 | 17 | 15 | 3 | 16 | 12 | 17 | 12 | 17 | 17 |
| 12 | 3 | 8 | 6 | 3 | 17 | 8 | 3 | 8 | 4 | 8 | 12 |
| 8 | 17 | 6 | 3 | 4 | 8 | 4 | 17 | 12 | 16 | 2 | 8 |
| 6 | 5 | 12 | 8 | 17 | 2 | 5 | 8 | 2 | 5 | 10 | 2 |
| 4 | 2 | 4 | 4 | 8 | 10 | 3 | 4 | 10 | 2 | 5 | 5 |
| 10 | 10 | 2 | 5 | 5 | 5 | 17 | 5 | 5 | 3 | 7 | 10 |
| 2 | 6 | 10 | 2 | 2 | 4 | 2 | 2 | 6 | 17 | 4 | 4 |
| 5 | 4 | 5 | 10 | 10 | 6 | 10 | 10 | 4 | 10 | 6 | 6 |
| Hypotension | Normotension | Stage 1 Hypertension | Stage 2 Hypertension | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| R1 | R2 | R3 | R1 | R2 | R3 | R1 | R2 | R3 | R1 | R2 | R3 |
| 1 | 14 | 13 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| 15 | 1 | 1 | 12 | 9 | 7 | 13 | 7 | 12 | 9 | 15 | 15 |
| 9 | 9 | 11 | 13 | 11 | 13 | 7 | 16 | 15 | 13 | 13 | 13 |
| 14 | 15 | 12 | 11 | 13 | 11 | 11 | 15 | 14 | 11 | 14 | 11 |
| 12 | 16 | 16 | 14 | 14 | 15 | 12 | 3 | 16 | 14 | 9 | 14 |
| 8 | 3 | 3 | 16 | 16 | 9 | 14 | 12 | 9 | 7 | 11 | 9 |
| 13 | 17 | 17 | 7 | 12 | 14 | 15 | 17 | 3 | 12 | 7 | 12 |
| 11 | 12 | 9 | 9 | 3 | 12 | 9 | 8 | 17 | 16 | 16 | 16 |
| 16 | 8 | 15 | 3 | 17 | 8 | 16 | 9 | 8 | 3 | 17 | 8 |
| 3 | 13 | 14 | 17 | 8 | 16 | 8 | 10 | 10 | 17 | 3 | 7 |
| 17 | 7 | 8 | 15 | 15 | 3 | 3 | 2 | 2 | 15 | 12 | 3 |
| 2 | 11 | 2 | 8 | 2 | 17 | 17 | 14 | 5 | 8 | 8 | 17 |
| 10 | 2 | 10 | 10 | 10 | 2 | 10 | 5 | 4 | 10 | 6 | 10 |
| 5 | 10 | 5 | 2 | 5 | 10 | 2 | 4 | 6 | 2 | 2 | 2 |
| 4 | 5 | 7 | 5 | 7 | 6 | 5 | 6 | 7 | 5 | 4 | 5 |
| 6 | 4 | 4 | 4 | 4 | 5 | 4 | 13 | 11 | 4 | 5 | 6 |
| 7 | 6 | 6 | 6 | 6 | 4 | 6 | 11 | 13 | 6 | 10 | 4 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
May, J.M.; Mejía-Mejía, E.; Nomoni, M.; Budidha, K.; Choi, C.; Kyriacou, P.A. Effects of Contact Pressure in Reflectance Photoplethysmography in an In Vitro Tissue-Vessel Phantom. Sensors 2021, 21, 8421. https://doi.org/10.3390/s21248421
May JM, Mejía-Mejía E, Nomoni M, Budidha K, Choi C, Kyriacou PA. Effects of Contact Pressure in Reflectance Photoplethysmography in an In Vitro Tissue-Vessel Phantom. Sensors. 2021; 21(24):8421. https://doi.org/10.3390/s21248421
Chicago/Turabian StyleMay, James M., Elisa Mejía-Mejía, Michelle Nomoni, Karthik Budidha, Changmok Choi, and Panicos A. Kyriacou. 2021. "Effects of Contact Pressure in Reflectance Photoplethysmography in an In Vitro Tissue-Vessel Phantom" Sensors 21, no. 24: 8421. https://doi.org/10.3390/s21248421
APA StyleMay, J. M., Mejía-Mejía, E., Nomoni, M., Budidha, K., Choi, C., & Kyriacou, P. A. (2021). Effects of Contact Pressure in Reflectance Photoplethysmography in an In Vitro Tissue-Vessel Phantom. Sensors, 21(24), 8421. https://doi.org/10.3390/s21248421










