Rest Intervals during Virtual Reality Gaming Augments Standing Postural Sway Disturbance
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design and Participants
2.2. Virtual Reality Setup
2.3. Measures
2.3.1. Balance Measures
2.3.2. Cybersickness
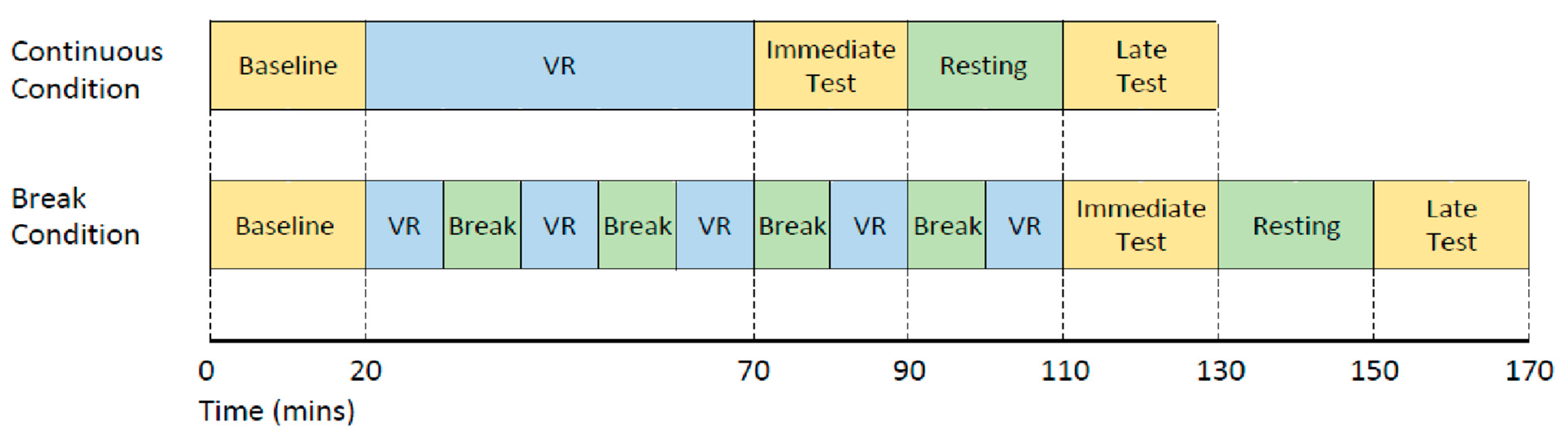
2.4. Procedure
- (1)
- Baseline: before VR exposure;
- (2)
- Immediate: immediately after completing a total of 50 min VR;
- (3)
- Late: 40 min after exiting VR exposure. The 40 min post-VR included a 20 min resting time between the immediate and late test.
2.5. Statistical Analysis
2.5.1. Primary Aim
2.5.2. Secondary Aims
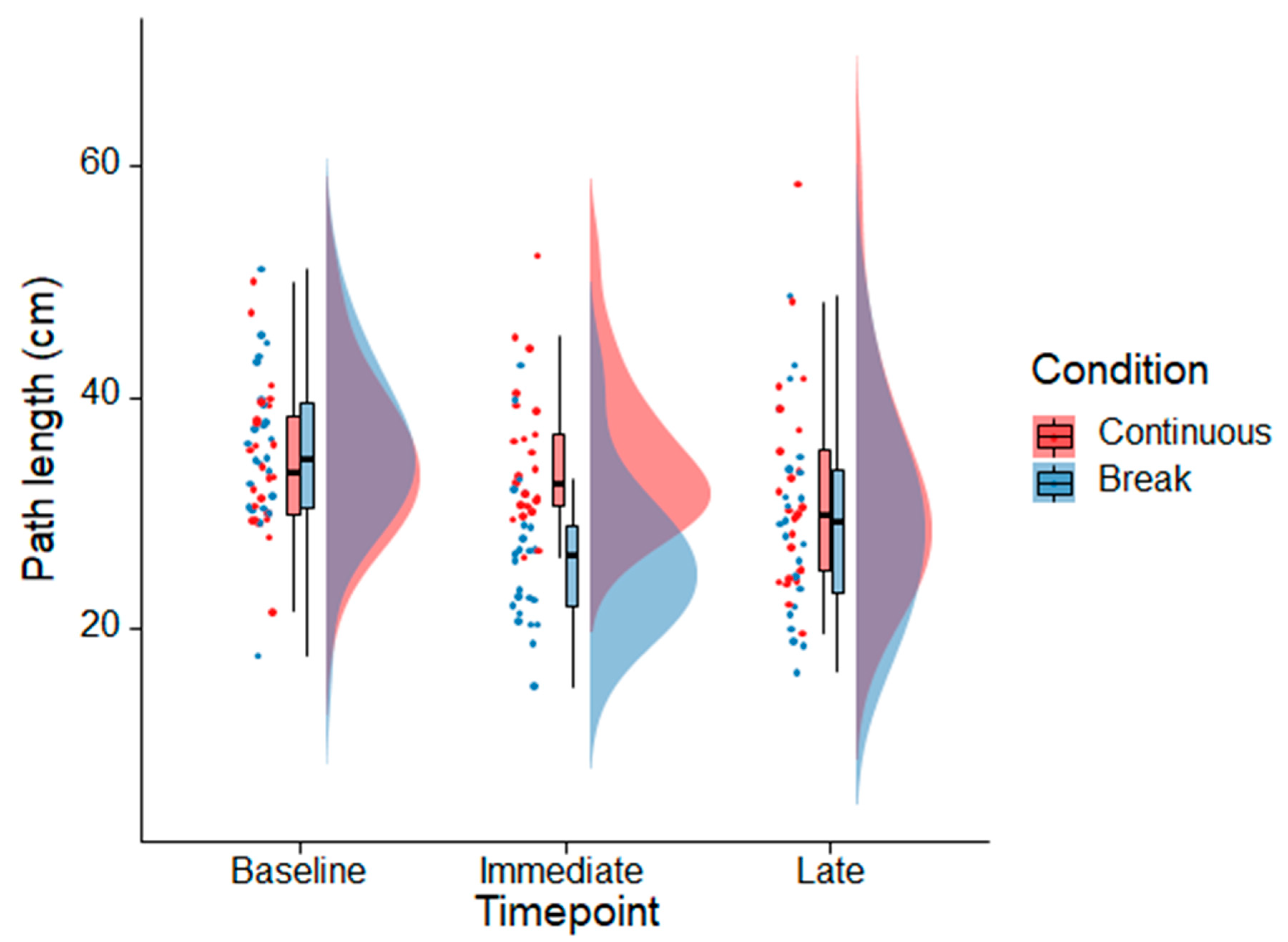
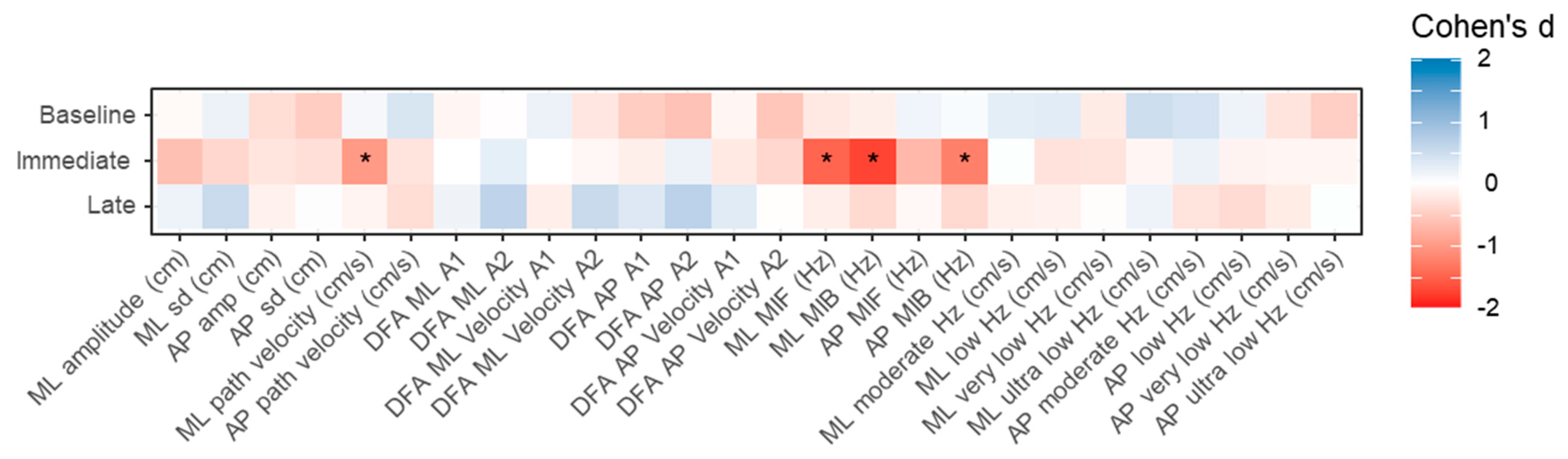
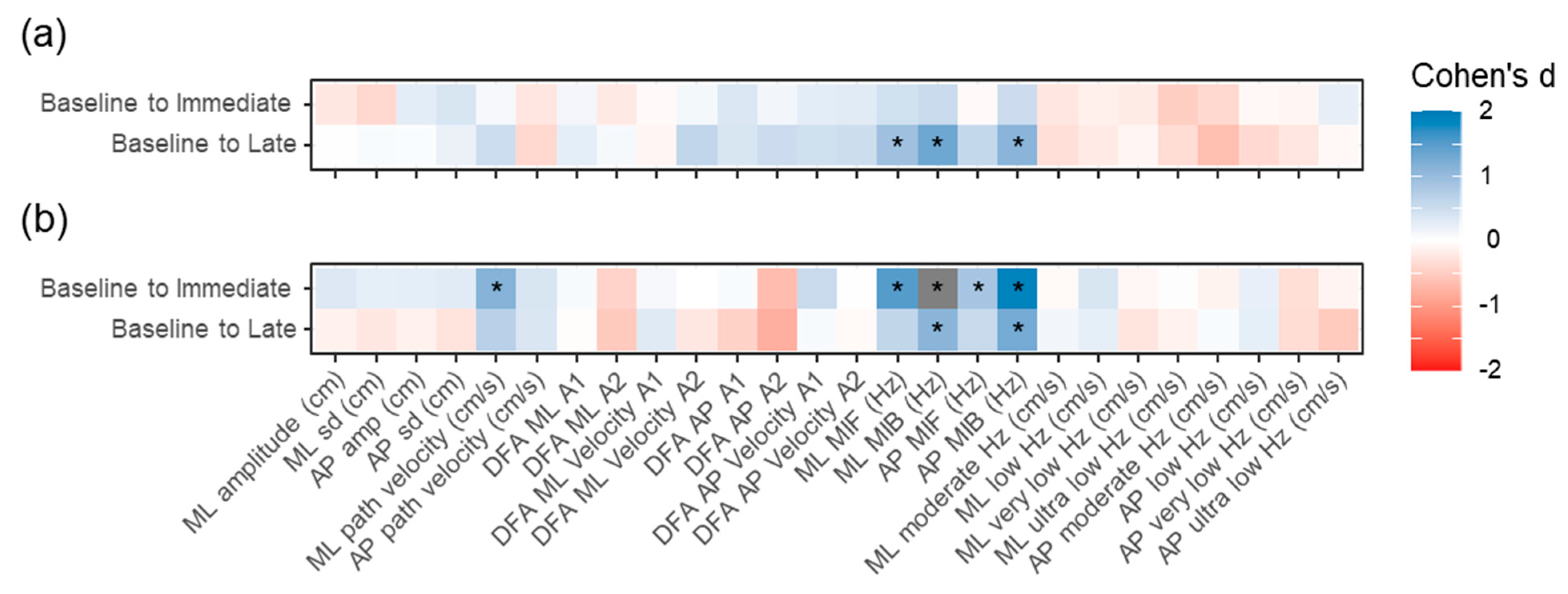
3. Results
3.1. Primary Aim
3.2. Secondary Aims
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jerald, J. The VR Book: Human-Centered Design for Virtual Reality; Association for Computing Machinery: New York, NY, USA, 2016. [Google Scholar]
- Bailenson, J. Experience on Demand: What Virtual Reality Is, How It Works, and What It Can Do; WW Norton & Company: New York, NY, USA, 2018. [Google Scholar]
- Hale, K.; Stanney, K. Handbook of Virtual Environments; CRC Press: Boca Raton, FL, USA, 2014. [Google Scholar]
- Saredakis, D.; Szpak, A.; Birckhead, B.; Keage, H.A.; Rizzo, A.; Loetscher, T. Factors Associated With Virtual Reality Sickness in Head-Mounted Displays: A Systematic Review and Meta-Analysis. Front. Hum. Neurosci. 2020, 14, 96. [Google Scholar] [CrossRef] [Green Version]
- Chang, E.; Kim, H.T.; Yoo, B. Virtual Reality Sickness: A Review of Causes and Measurements. Int. J. Hum.–Comput. Interact. 2020, 36, 1658–1682. [Google Scholar] [CrossRef]
- Muth, E.R. The challenge of uncoupled motion: Duration of cognitive and physiological aftereffects. Hum. Factors 2009, 51, 752–761. [Google Scholar] [CrossRef]
- Champney, R.K.; Stanney, K.M.; Hash, P.A.; Malone, L.C.; Kennedy, R.S.; Compton, D.E. Recovery from virtual environment exposure: Expected time course of symptoms and potential readaptation strategies. Hum. Factors 2007, 49, 491–506. [Google Scholar] [CrossRef]
- Hoffman, D.M.; Girshick, A.R.; Akeley, K.; Banks, M.S. Vergence–accommodation conflicts hinder visual performance and cause visual fatigue. J. Vis. 2008, 8, 33. [Google Scholar] [CrossRef]
- Szpak, A.; Michalski, S.C.; Saredakis, D.; Chen, C.S.; Loetscher, T. Beyond Feeling Sick: The Visual and Cognitive Aftereffects of Virtual Reality. IEEE Access 2019, 7, 130883–130892. [Google Scholar] [CrossRef]
- Hakkinen, J.; Vuori, T.; Paakka, M. Postural stability and sickness symptoms after HMD use. In Proceedings of the IEEE International Conference on Systems, Man and Cybernetics, Yasmine Hammamet, Tunisia, 6–9 October 2002. [Google Scholar]
- Kennedy, R.S.; Lilienthal, M.G. Implications of balance disturbances following exposure to virtual reality systems. In Proceedings of the IEEE Virtual Reality Annual International Symposium (VRAIS), Research Triangle Park, NC, USA, 11–15 March 1995; pp. 35–39. [Google Scholar]
- Dużmańska, N.; Strojny, P.; Strojny, A. Can simulator sickness be avoided? A review on temporal aspects of simulator sickness. Front. Psychol. 2018, 9, 2132. [Google Scholar] [CrossRef]
- Szpak, A.; Michalski, S.C.; Loetscher, T. Exergaming With Beat Saber: An Investigation of Virtual Reality Aftereffects. J. Med. Internet Res. 2020, 22, e19840. [Google Scholar] [CrossRef]
- Facebook Technologies LLC. Oculus Quest 2-Health and Safety Warnings. Available online: https://www.oculus.com/legal/health-and-safety-warnings/ (accessed on 25 May 2021).
- HTC Technologies Safety and Regulatory Guide. Available online: https://dl4.htc.com/vive/safty_guide/91H02887-08M%20Rev.A.PDF?_ga=1.174055779.1348312037.1468207042 (accessed on 25 May 2021).
- Deb, S.; Carruth, D.W.; Sween, R.; Strawderman, L.; Garrison, T.M. Efficacy of virtual reality in pedestrian safety research. Appl. Ergon. 2017, 65, 449–460. [Google Scholar] [CrossRef]
- Lampton, D.R.; Rodriguez, M.E.; Cotton, J.E. Simulator Sickness Symptoms during Team Training in Immersive Virtual Environments. Proc. Hum. Factors Ergon. Soc. Annu. Meet. 2000, 44, 530–533. [Google Scholar] [CrossRef]
- Moss, J.D.; Austin, J.; Salley, J.; Coats, J.; Williams, K.; Muth, E.R. The effects of display delay on simulator sickness. Displays 2011, 32, 159–168. [Google Scholar] [CrossRef]
- Clark, R.A.; Mentiplay, B.F.; Pua, Y.H.; Bower, K.J. Reliability and validity of the Wii Balance Board for assessment of standing balance: A systematic review. Gait Posture 2018, 61, 40–54. [Google Scholar] [CrossRef]
- Clark, R.A.; Pua, Y.H. SeeSway–A free web-based system for analysing and exploring standing balance data. Comput. Methods Programs Biomed. 2018, 159, 31–36. [Google Scholar] [CrossRef]
- Clark, R.A.; Howells, B.; Feller, J.; Whitehead, T.; Webster, K.E. Clinic-based assessment of weight-bearing asymmetry during squatting in people with anterior cruciate ligament reconstruction using nintendo wii balance boards. Arch. Phys. Med. Rehabil. 2014, 95, 1156–1161. [Google Scholar] [CrossRef]
- Clark, R.A.; Seah, F.T.; Chong, H.C.; Poon, C.L.; Tan, J.M.; Mentiplay, B.F.; Pua, Y.H. Standing balance post total knee arthroplasty: Sensitivity to change analysis from four to twelve weeks in 466 patients. Osteoarthr. Cartil. 2017, 25, 42–45. [Google Scholar] [CrossRef] [Green Version]
- Liang, Z.; Clark, R.; Bryant, A.; Quek, J.; Pua, Y.H. Neck musculature fatigue affects specific frequency bands of postural dynamics during quiet standing. Gait Posture 2014, 39, 397–403. [Google Scholar] [CrossRef]
- Bryant, A.L.; Pua, Y.H.; Clark, R.A. Morphology of knee extension torque-time curves following anterior cruciate ligament injury and reconstruction. J. Bone Jt. Surg.-Ser. A 2009, 91, 1424–1431. [Google Scholar] [CrossRef]
- Collins, J.J.; De Luca, C.J.; Burrows, A.; Lipsitz, L.A. Age-related changes in open-loop and closed-loop postural control mechanisms. Exp. Brain Res. 1995, 104, 480–492. [Google Scholar] [CrossRef]
- Amoud, H.; Abadi, M.; Hewson, D.J.; Michel-Pellegrino, V.; Doussot, M.; Duchêne, J. Fractal time series analysis of postural stability in elderly and control subjects. J. NeuroEng. Rehabil. 2007, 4, 12. [Google Scholar] [CrossRef] [Green Version]
- Kennedy, R.S.; Lane, N.E.; Berbaum, K.S.; Lilienthal, M.G. Simulator Sickness Questionnaire: An Enhanced Method for Quantifying Simulator Sickness. Int. J. Aviat. Psychol. 1993, 3, 203–220. [Google Scholar] [CrossRef]
- Rebenitsch, L.; Owen, C. Review on cybersickness in applications and visual displays. Virtual Real. 2016, 20, 101–125. [Google Scholar] [CrossRef]
- Allen, M.; Poggiali, D.; Whitaker, K.; Marshall, T.R.; Kievit, R.A. Raincloud plots: A multi-platform tool for robust data visualization. Wellcome Open Res. 2021, 4, 63. [Google Scholar] [CrossRef]
- Mentiplay, B.F.; Williams, G.; Tan, D.; Adair, B.; Pua, Y.H.; Bok, C.W.; Bower, K.J.; Cole, M.H.; Ng, Y.S.; Lim, L.S.; et al. Gait Velocity and Joint Power Generation after Stroke: Contribution of Strength and Balance. Am. J. Phys. Med. Rehabil. 2019, 98, 841–849. [Google Scholar] [CrossRef]
- Mentiplay, B.F.; Clark, R.A.; Bower, K.J.; Williams, G.; Pua, Y.H. Five times sit-to-stand following stroke: Relationship with strength and balance. Gait Posture 2020, 78, 35–39. [Google Scholar] [CrossRef]
- Dennison, M.S.; D’Zmura, M. Cybersickness without the wobble: Experimental results speak against postural instability theory. Appl. Ergon. 2017, 58, 215–223. [Google Scholar] [CrossRef]
- Riccio, G.E.; Stoffregen, T.A. An ecological Theory of Motion Sickness and Postural Instability. Ecol. Psychol. 1991, 3, 195–240. [Google Scholar] [CrossRef]
- Risi, D.; Palmisano, S. Effects of postural stability, active control, exposure duration and repeated exposures on HMD induced cybersickness. Displays 2019, 60, 9–17. [Google Scholar] [CrossRef]
- Chardonnet, J.-R.; Mirzaei, M.A.; Mérienne, F. Features of the Postural Sway Signal as Indicators to Estimate and Predict Visually Induced Motion Sickness in Virtual Reality. Int. J. Hum.–Comput. Interact. 2017, 33, 771–785. [Google Scholar] [CrossRef] [Green Version]
- Jeter, P.E.; Wang, J.; Gu, J.; Barry, M.P.; Roach, C.; Corson, M.; Yang, L.; Dagnelie, G. Intra-session test-retest reliability of magnitude and structure of center of pressure from the Nintendo Wii Balance Board™ for a visually impaired and normally sighted population. Gait Posture 2015, 41, 482–487. [Google Scholar] [CrossRef] [Green Version]
- Kim, A.; Darakjian, N.; Finley, J.M. Walking in fully immersive virtual environments: An evaluation of potential adverse effects in older adults and individuals with Parkinson’s disease. J. NeuroEng. Rehabil. 2017, 14, 16. [Google Scholar] [CrossRef] [Green Version]
- Widdowson, C.; Becerra, I.; Merrill, C.; Wang, R.F.; LaValle, S. Assessing Postural Instability and Cybersickness Through Linear and Angular Displacement. Hum. Factors 2019, 63, 296–311. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Clark, R.A.; Szpak, A.; Michalski, S.C.; Loetscher, T. Rest Intervals during Virtual Reality Gaming Augments Standing Postural Sway Disturbance. Sensors 2021, 21, 6817. https://doi.org/10.3390/s21206817
Clark RA, Szpak A, Michalski SC, Loetscher T. Rest Intervals during Virtual Reality Gaming Augments Standing Postural Sway Disturbance. Sensors. 2021; 21(20):6817. https://doi.org/10.3390/s21206817
Chicago/Turabian StyleClark, Ross Allan, Ancret Szpak, Stefan Carlo Michalski, and Tobias Loetscher. 2021. "Rest Intervals during Virtual Reality Gaming Augments Standing Postural Sway Disturbance" Sensors 21, no. 20: 6817. https://doi.org/10.3390/s21206817
APA StyleClark, R. A., Szpak, A., Michalski, S. C., & Loetscher, T. (2021). Rest Intervals during Virtual Reality Gaming Augments Standing Postural Sway Disturbance. Sensors, 21(20), 6817. https://doi.org/10.3390/s21206817





