Mobile Technologies to Promote Physical Activity during Cardiac Rehabilitation: A Scoping Review
Abstract
1. Introduction
2. Methods
2.1. Eligibility Criteria
2.2. Search Strategy
2.3. Study Selection
2.4. Data Extraction
2.5. Synthesis
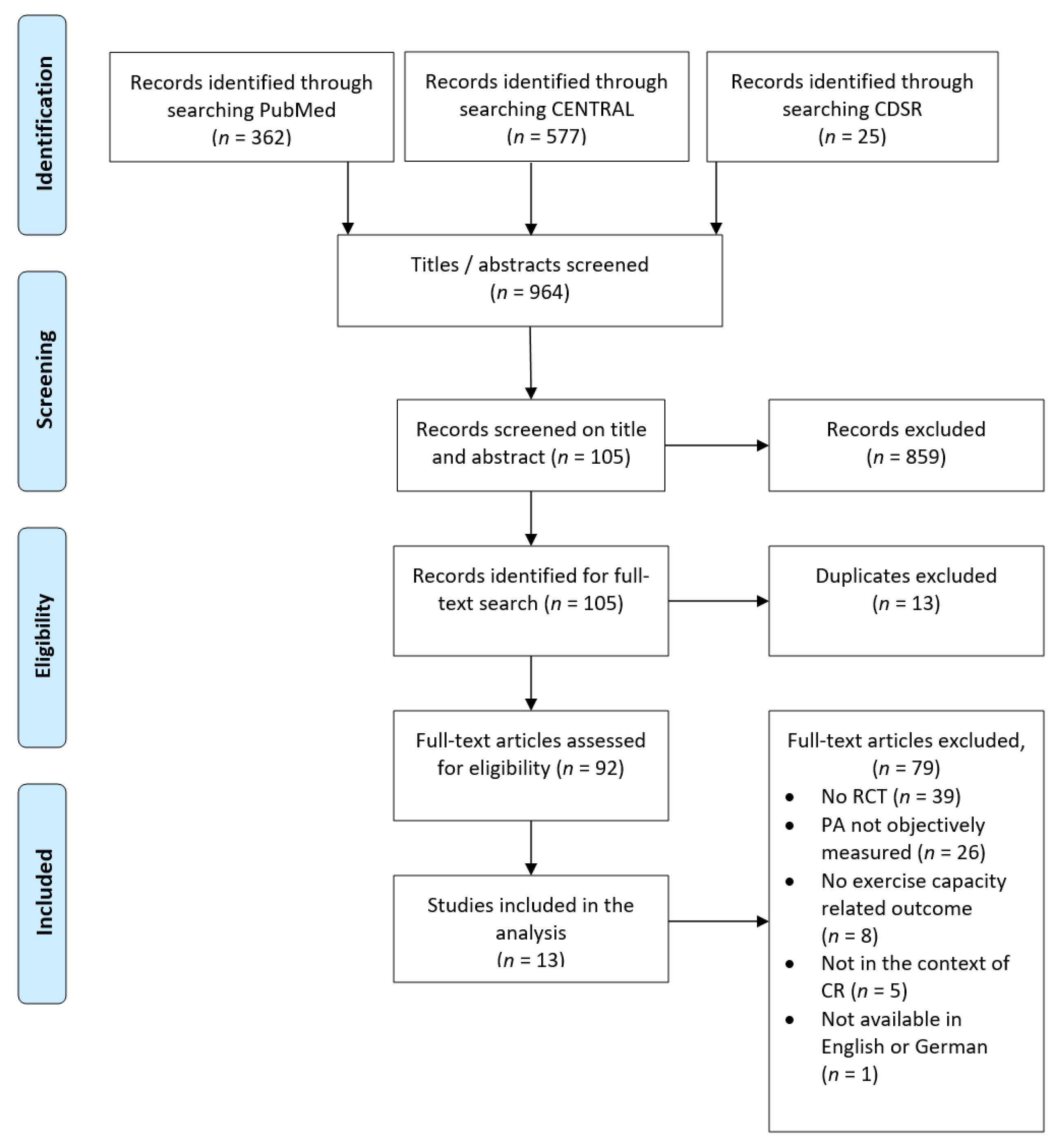
3. Results
3.1. Selected Studies
3.2. Study Objectives and Characteristics
- (a)
- Home-based CR with mTechs (IG) vs. outpatient CR without mTechs (CG) (n = 3);
- (b)
- Outpatient CR with mTechs (IG) vs. outpatient CR without mTechs (CG) (n = 2);
- (c)
- Outpatient CR followed by home-based CR with mTechs (IG) vs. outpatient CR without further CR (CG) (n = 10).
3.3. Effects on Exercise Capacity: Home-Based CR with mTechs vs. Outpatient CR without mTechs
3.4. Effects on Exercise Capacity: Outpatient CR with vs. without mTechs
3.5. Effects on Exercise Capacity: Outpatient CR followed by mTechs during Home-Based CR vs. Outpatient CR without Further CR
3.6. Further Reported Effects and Outcomes
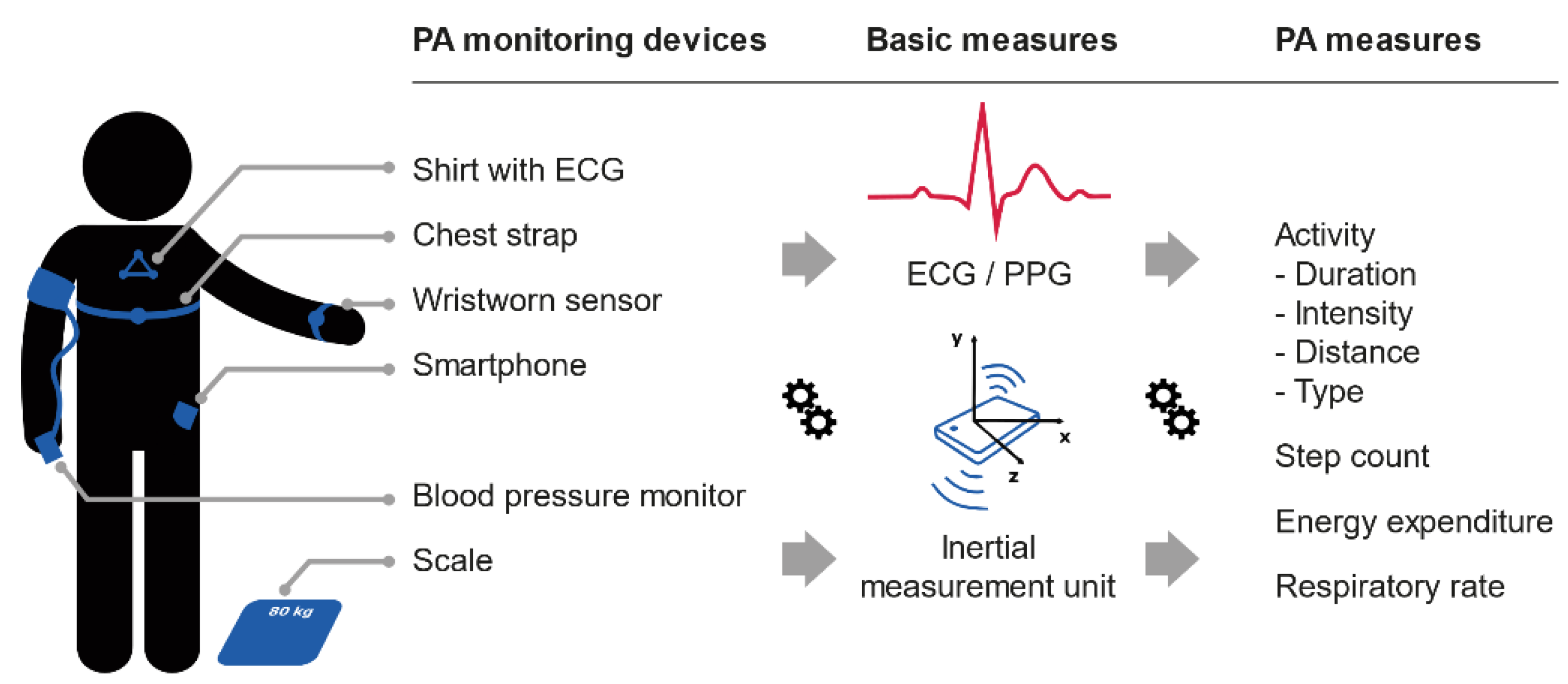
3.7. Mobile Technologies (mTechs) and Objective PA Measures
- Research-targeted devices are mainly employed in research studies, such as Actigraph’s devices.
- Custom devices are specifically developed for application in the study and are not commercially available.
- Off-the-shelf devices are targeted at sports and fitness tracking of the general public, such as Garmin, Polar and Fitbit devices.
| Study | Used Technology | Objective PA Measures | Intervention Description |
|---|---|---|---|
| mTechs during Home-Based CR vs. Outpatient CR | |||
| Avila et al. [13] | heart rate monitor (Garmin Forerunner 210 watch, Garmin International Inc., Olathe, KS, USA); web application (Garmin Connect, Garmin International Inc., Olathe, KS, USA)) | heart rate; steps; PA levels; sedentary time; active energy expenditure | feedback via phone or email once a week according to patient’s preferences; individualized aerobic exercise prescription at an individually determined target heart rate corresponding to a moderate intensity |
| Kraal et al. [20] | heart rate monitor (Garmin FR70 chest strap, Garmin International Inc., Olathe, KS, USA); web application (Garmin Connect, Garmin International Inc., Olathe, KS, USA); PA monitor worn at hip (ActiGraph wGT3X+, ActigraphCorp, Pensacalo, FL, USA) | heart rate; PA level; energy expenditure | feedback via telephone once a week by the physical therapist regarding training frequency, duration, intensity, motivation, and self-management-skills |
| Maddison et al. [21] | chest-worn wearable sensor (BioHarness 3, Zephyr Technology, Annapolis, ML, USA); custom smartphone app and web application; uniaxial accelerometer worn at waist (GT1M ActiGraph, ActigraphCorp, Pensacalo, FL, USA) | heart rate; single lead ECG; PA levels: light, moderate and vigorous activity durations; respiratory rate | physiological and geopositional data displayed in app; real-time audio coaching, feedback, and social support throughout exercise monitoring; encouragement to be active ≥ 5 days per week; individualized and progressive exercise prescriptions |
| Outpatient CR with mTechs vs. outpatient CR without mTechs | |||
| Rosario et al. [31] | STAHR (smartphone technology and heart rehabilitation) app; blood pressure monitor and weight scale, both NFC-enabled (A&D Medical, San Jose, CA, USA); own algorithm to estimate patient’s PA throughout the day | activity level (identifying walking on a level surface, walking upstairs, walking downstairs, standing, sitting, lying, movements between two stationary points) | automated feedback: daily messages based on the amount of PA performed; messages twice a week based on conditions for blood pressure, weight, questionnaire, and activity |
| Vogel et al. [32] | wrist worn activity tracker (tri-axial accelerometer, Polar Loop, Polar Electro Oy, Kempele, Finnland); web service (Polar Flow, Polar Electro Oy, Kempele, Finnland) | steps; energy expenditure; PA levels: intensity and duration | usage of smart wearables for self-monitoring PA and goal setting; questions whether problems occurred |
| Outpatient CR followed by home-based CR with mTechs vs. outpatient CR without further formal CR | |||
| Avila et al. [13] | See above. | ||
| Duscha et al. [14] | PA tracker (Fitbit Charge, Fitbit Inc., San Francisco, CA, USA) integrated into Vida’s mHealth platform: mobile technology to provide healthcare coaching (Vida Health, San Francisco, CA, USA) | PA levels; steps/day; minutes/day of exercise; distance/day in miles and floors/day | coaches called patients 1–2 times per week for 30–60 min; educational material via email; text messages to remind patients to practice healthy lifestyle habits; frequency and content of text messages not automated, but individualized |
| Fang et al. [33] | belt strap with sensor (Ucare RG10, Micro Sensor Co., Shaanxi, China); smartphone app; web portal | heart rate; real-time ECG; activity level; energy consumption; exercise time | physiological monitoring for outdoor walking and jogging; rapid feedback by clinician; weekly telephone call; paper-based and self-study booklet |
| Frederix et al. [15] | System to provide automated feedback via email or SMS; motion sensor (Yorbody accelerometer, Yorbody, Puurs-Sint-Amand, Belgium); webservice | activity data during exercise sessions; daily number of aerobic steps (≥60 steps/min), regular steps (<60 steps/min), and total steps | weekly personalized automated feedback on PA via email or SMS; program was designed to encourage the patient to increase his/her daily amount of steps with 10% each week from baseline |
| Frederix et al. [16] | semiautomatic telecoaching system to provide feedbacks via email or SMS; motion sensor (Yorbody accelerometer, Yorbody, Puurs-Sint-Amand, Belgium); webservice | low intensity steps/day; high intensity steps/day | weekly feedback via email or SMS for encouraging to gradually achieve predefined exercise training goals; tailored dietary and smoking cessation recommendations; content of feedback messages changes over time based on how well the patients changed their prior lifestyle behavior |
| Piotrowicz et al. [12] | remote ECG device for telemonitored and telesupervised exercise training (Pro Plus Company, Warsaw, Poland); mobile phone | ECG; (heart rate used to adjust workload) | individual training sessions for each patient (defined exercise duration, breaks, and timing of ECG recording); telephone contact for psychological support |
| Piotrowicz et al. [30] | remote ECG device for telemonitored and telesupervised exercise training (Pro Plus Company, Warsaw, Poland); mobile phone for data transmission; web-based monitoring platform; blood pressure device; body-weight scale | ECG; (heart rate used to adjust workload) | individually preprogrammed training sessions for each patient (exercise duration, breaks, and timing of ECG recording); heart rate and patient’s perceived exertion used to adjust workload; telephone contact for psychological support |
| Skobel et al. [17] | own wearable sensor attached to special shirt; smartphone app; web application | heart rate; single lead ECG; activity level; respiratory rate | messages to inform about the exercise plan prescribed by the doctor; feedback on exercise sessions;tips about health and lifestyle |
| Snoek et al. [19] | Bluetooth-connected heart rate chest strap (Zephyr, Zephyr Technology, Annapolis, ML, USA): smartphone (Samsung Galaxy Ace, Samsung, Seoul, South Korea); web application | heart rate; training mode, time and intensity (determined by heart rate) | patients were contacted weekly by telephone for supportive guidance in the first month, every other week in the second month, and from then on monthly until six months; PA data discussed by using motivational interviewing; patients in CG also contacted via telephone monthly |
| Vogel et al. [32] | See above. | ||
3.8. Applied Behavioral Change Techniques (BCTs)
- Goal setting: Was goal setting in any form included?
- Self-monitoring: Was the collected data provided to the patient to allow self-monitoring?
- Feedback on exercise: Was feedback on exercise given in any form during the intervention?
- Physician/expert involved: Was a physician/expert involved in the application of the BCTs?
- Tailored prescription: Was any part of the intervention tailored to the patient?
- Real-time monitoring: Did other people than the patient monitor exercise data in real-time?
- Education: Was educational material provided in any form?
| Goal Setting | Self-Monitoring | Feedback on Exercise | Physician/Expert Involvement | Tailored Prescription | Real-Time Monitoring | Education | |
|---|---|---|---|---|---|---|---|
| mTechs during home-based CR vs. outpatient CR | |||||||
| Avila et al. [13] | √ | √ | √ | √ | √ | ||
| Kraal et al. [20] | √ | √ | √ | √ | √ | ||
| Maddison et al. [21] | √ | √ | √ | √ | √ | √ | √ |
| Outpatient CR with mTechs vs. outpatient CR without mTechs | |||||||
| Rosario et al. [31] | √ | √ | √ | ||||
| Vogel et al. [32] | √ | √ | √ | √ | |||
| Outpatient CR followed by home-based CR with mTechs vs. outpatient CR without further formal CR | |||||||
| Avila et al. [13] | √ | √ | √ | √ | √ | ||
| Duscha et al. [14] | √ | √ | √ | √ | √ | √ | |
| Fang et al. [33] | √ | √ | √ | √ | √ | √ | √ |
| Frederix et al. [15] | √ | √ | √ | √ | √ | ||
| Frederix et al. [16] | √ | √ | √ | √ | √ | √ | |
| Piotrowicz et al. [12] | √ | √ | √ | √ | √ | ||
| Piotrowicz et al. [30] | √ | √ | √ | √ | √ | √ | |
| Skobel et al. [17] | √ | √ | √ | √ | √ | √ | √ |
| Snoek et al. [19] | √ | √ | √ | √ | √ | ||
| Vogel et al. [32] | √ | √ | √ | √ | |||
4. Discussion
4.1. Study Objectives and Characteristics
4.2. Effects on Exercise Capacity
4.3. Reported Effects Different to Exercise Capacity
4.4. Applied Mobile Technologies and Objective PA Measures
4.5. Behavior Change Techniques (BCTs)
4.6. CR during and after the COVID-19 Pandemic
4.7. Limitations
4.8. Future Directions and Recommendations
- Aim at gender equality in CR research and implementation.
- Focus on technology acceptance, usability, and user experience of mTechs for CR by adopting participatory design and development methodology, especially from the so far underrepresented female perspective.
- Extend and enforce reporting guidelines for mTechs interventions.
- Research methodology for the assessment of individual elements of complex mTech CR interventions.
- Include behavioral theories and frameworks in the development of mTech CR interventions.
- Perform long-term studies to evaluate the sustainability of improved health-outcomes of home-based CR with mTechs.
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Moraga, P.; GBD. GBD 2016 Causes of Death Collaborators Global, Regional, and National Age-Sex Specific Mortality for 264 Causes of Death, 1980-2016: A Systematic Analysis for the Global Burden of Disease Study 2016. Lancet 2017, 390, 1151–1210. [Google Scholar] [CrossRef]
- Heidenreich, P.A.; Trogdon, J.G.; Khavjou, O.A.; Butler, J.; Dracup, K.; Ezekowitz, M.D.; Finkelstein, E.A.; Hong, Y.; Johnston, S.C.; Khera, A.; et al. Forecasting the Future of Cardiovascular Disease in the United States: A Policy Statement from the American Heart Association. Circulation 2011, 123, 933–944. [Google Scholar] [CrossRef]
- Lear, S.A.; Hu, W.; Rangarajan, S.; Gasevic, D.; Leong, D.; Iqbal, R.; Casanova, A.; Swaminathan, S.; Anjana, R.M.; Kumar, R.; et al. The Effect of Physical Activity on Mortality and Cardiovascular Disease in 130 000 People from 17 High-Income, Middle-Income, and Low-Income Countries: The PURE Study. Lancet 2017, 390, 2643–2654. [Google Scholar] [CrossRef]
- Pelliccia, A.; Sharma, S.; Gati, S.; Bäck, M.; Börjesson, M.; Caselli, S.; Collet, J.-P.; Corrado, D.; Drezner, J.A.; Halle, M.; et al. 2020 ESC Guidelines on Sports Cardiology and Exercise in Patients with Cardiovascular Disease: The Task Force on Sports Cardiology and Exercise in Patients with Cardiovascular Disease of the European Society of Cardiology (ESC). Eur. Heart J. 2020. [Google Scholar] [CrossRef]
- WHO. Global Recommendations on Physical Activity for Health; WHO: Genève, Switerzland, 2010; ISBN 978-92-4-159997-9. [Google Scholar]
- Anderson, L.; Sharp, G.A.; Norton, R.J.; Dalal, H.; Dean, S.G.; Jolly, K.; Cowie, A.; Zawada, A.; Taylor, R.S. Home-based versus Centre-based Cardiac Rehabilitation. Cochrane Database Syst. Rev. 2017. [Google Scholar] [CrossRef]
- Kotseva, K.; De Backer, G.; De Bacquer, D.; Rydén, L.; Hoes, A.; Grobbee, D.; Maggioni, A.; Marques-Vidal, P.; Jennings, C.; Abreu, A.; et al. Lifestyle and Impact on Cardiovascular Risk Factor Control in Coronary Patients across 27 Countries: Results from the European Society of Cardiology ESC-EORP EUROASPIRE V Registry. Eur. J. Prev. Cardiol. 2019, 26, 824–835. [Google Scholar] [CrossRef] [PubMed]
- Niebauer, J. Cardiac Rehabilitation in Austria. Wien. Med. Wochenschr. 2018, 168, 46–49. [Google Scholar] [CrossRef] [PubMed]
- Loney, T.; Standage, M.; Thompson, D.; Sebire, S.J.; Cumming, S. Self-Report vs. Objectively Assessed Physical Activity: Which Is Right for Public Health? J. Phys. Act Health 2011, 8, 62–70. [Google Scholar] [CrossRef] [PubMed]
- Sallis, J.F.; Saelens, B.E. Assessment of Physical Activity by Self-Report: Status, Limitations, and Future Directions. Res. Q. Exerc. Sport 2000, 71, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Baranowski, T.; Dworkin, R.J.; Cieslik, C.J.; Hooks, P.; Clearman, D.R.; Ray, L.; Dunn, J.K.; Nader, P.R. Reliability and Validity of Self Report of Aerobic Activity: Family Health Project. Res. Q. Exerc. Sport 1984, 55, 309–317. [Google Scholar] [CrossRef]
- Piotrowicz, E.; Zielinski, T.; Bodalski, R.; Rywik, T.; Dobraszkiewicz-Wasilewska, B.; Sobieszczanska-Malek, M.; Stepnowska, M.; Przybylski, A.; Browarek, A.; Szumowski, L.; et al. Home-Based Telemonitored Nordic Walking Training Is Well Accepted, Safe, Effective and Has High Adherence among Heart Failure Patients, Including Those with Cardiovascular Implantable Electronic Devices: A Randomised Controlled Study. Eur. J. Prev. Cardiol. 2015, 22, 1368–1377. [Google Scholar] [CrossRef] [PubMed]
- Avila, A.; Claes, J.; Goetschalckx, K.; Buys, R.; Azzawi, M.; Vanhees, L.; Cornelissen, V. Home-Based Rehabilitation With Telemonitoring Guidance for Patients With Coronary Artery Disease (Short-Term Results of the TRiCH Study): Randomized Controlled Trial. J. Med. Internet Res. 2018, 20, e225. [Google Scholar] [CrossRef] [PubMed]
- Duscha, B.D.; Piner, L.W.; Patel, M.P.; Craig, K.P.; Brady, M.; McGarrah, R.W.; Chen, C.; Kraus, W.E. Effects of a 12-Week MHealth Program on Peak VO2 and Physical Activity Patterns after Completing Cardiac Rehabilitation: A Randomized Controlled Trial. Am. Heart J. 2018, 199, 105–114. [Google Scholar] [CrossRef] [PubMed]
- Frederix, I.; Van Driessche, N.; Hansen, D.; Berger, J.; Bonne, K.; Alders, T.; Dendale, P. Increasing the Medium-Term Clinical Benefits of Hospital-Based Cardiac Rehabilitation by Physical Activity Telemonitoring in Coronary Artery Disease Patients. Eur. J. Prev. Cardiol. 2015, 22, 150–158. [Google Scholar] [CrossRef]
- Frederix, I.; Solmi, F.; Piepoli, M.F.; Dendale, P. Cardiac Telerehabilitation: A Novel Cost-Efficient Care Delivery Strategy That Can Induce Long-Term Health Benefits. Eur. J. Prev. Cardiol. 2017, 24, 1708–1717. [Google Scholar] [CrossRef]
- Skobel, E.; Knackstedt, C.; Martinez-Romero, A.; Salvi, D.; Vera-Munoz, C.; Napp, A.; Luprano, J.; Bover, R.; Glöggler, S.; Bjarnason-Wehrens, B.; et al. Internet-Based Training of Coronary Artery Patients: The Heart Cycle Trial. Heart Vessel. 2017, 32, 408–418. [Google Scholar] [CrossRef]
- Piotrowicz, E.; Piotrowicz, R.; Opolski, G.; Pencina, M.; Banach, M.; Zaręba, W. Hybrid Comprehensive Telerehabilitation in Heart Failure Patients (TELEREH-HF): A Randomized, Multicenter, Prospective, Open-Label, Parallel Group Controlled Trial-Study Design and Description of the Intervention. Am. Heart J. 2019, 217, 148–158. [Google Scholar] [CrossRef]
- Snoek, J.A.; Meindersma, E.P.; Prins, L.F.; van’t Hof, A.W.; de Boer, M.-J.; Hopman, M.T.; Eijsvogels, T.M.; de Kluiver, E.P. The Sustained Effects of Extending Cardiac Rehabilitation with a Six-Month Telemonitoring and Telecoaching Programme on Fitness, Quality of Life, Cardiovascular Risk Factors and Care Utilisation in CAD Patients: The TeleCaRe Study. J. Telemed. Telecare 2019, 1357633X1988579. [Google Scholar] [CrossRef]
- Kraal, J.J.; Van den Akker-Van Marle, M.E.; Abu-Hanna, A.; Stut, W.; Peek, N.; Kemps, H.M. Clinical and Cost-Effectiveness of Home-Based Cardiac Rehabilitation Compared to Conventional, Centre-Based Cardiac Rehabilitation: Results of the FIT@Home Study. Eur. J. Prev. Cardiol. 2017, 24, 1260–1273. [Google Scholar] [CrossRef]
- Maddison, R.; Rawstorn, J.C.; Stewart, R.A.H.; Benatar, J.; Whittaker, R.; Rolleston, A.; Jiang, Y.; Gao, L.; Moodie, M.; Warren, I.; et al. Effects and Costs of Real-Time Cardiac Telerehabilitation: Randomised Controlled Non-Inferiority Trial. Heart 2019, 105, 122–129. [Google Scholar] [CrossRef]
- Driggin, E.; Madhavan, M.V.; Bikdeli, B.; Chuich, T.; Laracy, J.; Biondi-Zoccai, G.; Brown, T.S.; Der Nigoghossian, C.; Zidar, D.A.; Haythe, J.; et al. Cardiovascular Considerations for Patients, Health Care Workers, and Health Systems During the COVID-19 Pandemic. J. Am. Coll. Cardiol. 2020, 75, 2352. [Google Scholar] [CrossRef] [PubMed]
- Arksey, H.; O’Malley, L. Scoping Studies: Towards a Methodological Framework. Int. J. Soc. Res. Methodol. 2005, 8, 19–32. [Google Scholar] [CrossRef]
- Levac, D.; Colquhoun, H.; O’Brien, K.K. Scoping Studies: Advancing the Methodology. Implement. Sci. 2010, 5, 69. [Google Scholar] [CrossRef] [PubMed]
- Tricco, A.C.; Lillie, E.; Zarin, W.; O’Brien, K.K.; Colquhoun, H.; Levac, D.; Moher, D.; Peters, M.D.J.; Horsley, T.; Weeks, L.; et al. PRISMA Extension for Scoping Reviews (PRISMA-ScR): Checklist and Explanation. Ann. Intern. Med. 2018, 169, 467. [Google Scholar] [CrossRef] [PubMed]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gøtzsche, P.C.; Ioannidis, J.P.A.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA Statement for Reporting Systematic Reviews and Meta-Analyses of Studies That Evaluate Health Care Interventions: Explanation and Elaboration. PLoS Med. 2009, 6, e1000100. [Google Scholar] [CrossRef]
- Lee, D.-C.; Sui, X.; Ortega, F.B.; Kim, Y.-S.; Church, T.S.; Winett, R.A.; Ekelund, U.; Katzmarzyk, P.T.; Blair, S.N. Comparisons of Leisure-Time Physical Activity and Cardiorespiratory Fitness as Predictors of All-Cause Mortality in Men and Women. Br. J. Sports Med. 2011, 45, 504–510. [Google Scholar] [CrossRef]
- Myers, J.; Prakash, M.; Froelicher, V.; Do, D.; Partington, S.; Atwood, J.E. Exercise Capacity and Mortality among Men Referred for Exercise Testing. N. Engl. J. Med. 2002, 346, 793–801. [Google Scholar] [CrossRef]
- Michie, S.; Richardson, M.; Johnston, M.; Abraham, C.; Francis, J.; Hardeman, W.; Eccles, M.P.; Cane, J.; Wood, C.E. The Behavior Change Technique Taxonomy (v1) of 93 Hierarchically Clustered Techniques: Building an International Consensus for the Reporting of Behavior Change Interventions. Ann. Behav. Med. 2013, 46, 81–95. [Google Scholar] [CrossRef]
- Piotrowicz, E.; Pencina, M.J.; Opolski, G.; Zareba, W.; Banach, M.; Kowalik, I.; Orzechowski, P.; Szalewska, D.; Pluta, S.; Glowczynska, R.; et al. Effects of a 9-Week Hybrid Comprehensive Telerehabilitation Program on Long-Term Outcomes in Patients With Heart Failure: The Telerehabilitation in Heart Failure Patients (TELEREH-HF) Randomized Clinical Trial. JAMA Cardiol. 2019. [Google Scholar] [CrossRef]
- Rosario, M.B.D.; Lovell, N.H.; Fildes, J.; Holgate, K.; Yu, J.; Ferry, C.; Schreier, G.; Ooi, S.-Y.; Redmond, S.J. Evaluation of an MHealth-Based Adjunct to Outpatient Cardiac Rehabilitation. IEEE J. Biomed. Health Inform. 2018, 22, 1938–1948. [Google Scholar] [CrossRef]
- Vogel, J.; Auinger, A.; Riedl, R.; Kindermann, H.; Helfert, M.; Ocenasek, H. Digitally Enhanced Recovery: Investigating the Use of Digital Self-Tracking for Monitoring Leisure Time Physical Activity of Cardiovascular Disease (CVD) Patients Undergoing Cardiac Rehabilitation. PLoS ONE 2017, 12, e0186261. [Google Scholar] [CrossRef] [PubMed]
- Fang, J.; Huang, B.; Xu, D.; Li, J.; Au, W.W. Innovative Application of a Home-Based and Remote Sensing Cardiac Rehabilitation Protocol in Chinese Patients After Percutaneous Coronary Intervention. Telemed. J. E Health 2019, 25, 288–293. [Google Scholar] [CrossRef] [PubMed]
- Colella, T.J.F.; Gravely, S.; Marzolini, S.; Grace, S.L.; Francis, J.A.; Oh, P.; Scott, L.B. Sex Bias in Referral of Women to Outpatient Cardiac Rehabilitation? A Meta-Analysis. Eur. J. Prev. Cardiol. 2015, 22, 423–441. [Google Scholar] [CrossRef] [PubMed]
- Salvi, D.; Ottaviano, M.; Muuraiskangas, S.; Martínez-Romero, A.; Vera-Muñoz, C.; Triantafyllidis, A.; Cabrera Umpiérrez, M.F.; Arredondo Waldmeyer, M.T.; Skobel, E.; Knackstedt, C.; et al. An M-Health System for Education and Motivation in Cardiac Rehabilitation: The Experience of HeartCycle Guided Exercise. J. Telemed. Telecare 2018, 24, 303–316. [Google Scholar] [CrossRef]
- Smith, B.; Magnani, J.W. New Technologies, New Disparities: The Intersection of Electronic Health and Digital Health Literacy. Int. J. Cardiol. 2019, 292, 280–282. [Google Scholar] [CrossRef]
- Ernsting, C.; Dombrowski, S.U.; Oedekoven, M.; O Sullivan, J.L.; Kanzler, M.; Kuhlmey, A.; Gellert, P. Using Smartphones and Health Apps to Change and Manage Health Behaviors: A Population-Based Survey. J. Med. Internet Res. 2017, 19, e101. [Google Scholar] [CrossRef]
- Bhavnani, S.P.; Parakh, K.; Atreja, A.; Druz, R.; Graham, G.N.; Hayek, S.S.; Krumholz, H.M.; Maddox, T.M.; Majmudar, M.D.; Rumsfeld, J.S.; et al. 2017 Roadmap for Innovation—ACC Health Policy Statement on Healthcare Transformation in the Era of Digital Health, Big Data, and Precision Health. J. Am. Coll. Cardiol. 2017, 70, 2696. [Google Scholar] [CrossRef]
- Triantafyllidis, A.; Kondylakis, H.; Votis, K.; Tzovaras, D.; Maglaveras, N.; Rahimi, K. Features, Outcomes, and Challenges in Mobile Health Interventions for Patients Living with Chronic Diseases: A Review of Systematic Reviews. Int. J. Med. Inf. 2019, 132, 103984. [Google Scholar] [CrossRef]
- Hoffmann, T.C.; Glasziou, P.P.; Boutron, I.; Milne, R.; Perera, R.; Moher, D.; Altman, D.G.; Barbour, V.; Macdonald, H.; Johnston, M.; et al. Better Reporting of Interventions: Template for Intervention Description and Replication (TIDieR) Checklist and Guide. BMJ 2014, 348. [Google Scholar] [CrossRef]
| Study | Objectives | Patients | Mean Age Standard Deviation | Gender Female/Total (%) |
|---|---|---|---|---|
| mTechs during home-based CR (IG) vs. outpatient CR (CG) | ||||
| Avila et al. [13] | evaluation of effects and costs of home-based exercise training with telemonitoring guidance | randomized: n = 90, IG: n = 30 (2 dropouts), CG: n = 30 (0 dropouts) | IG: 59 ± 13 CG: 62 ± 7 | IG: 4/30 (13%) CG: 3/30 (10%) |
| Kraal et al. [20] | evaluation of effects and costs of home-based exercise training with telemonitoring guidance | randomized: n = 90, IG: n = 45 (4 dropouts), CG: n = 45 (8 dropouts) | IG: 58 ± 9 CG: 61 ± 9 | IG: 5/45 (11%) CG: 5/45 (11%) |
| Maddison et al. [21] | evaluation of effects and costs of remotely monitored exercise-based cardiac telerehabilitation in adults with coronary heart disease | randomized: n = 162, IG: n = 82 (14 dropouts), CG: n = 80 (8 dropouts) | IG: 61 ± 13 CG: 62 ± 12 | IG: 13/82 (16%) CG: 10/80 (13%) |
| Outpatient CR with (IG) vs. without mTechs (CG) | ||||
| Rosario et al. [31] | evaluation of effects of mHealth-based adjunct to outpatient CR regarding completion rate and exercise capacity | randomized: n = 66, IG: n = 33 (dropouts not reported), CG: n = 33 (dropouts not reported) | IG: not reported CG: not reported | IG: 12/33 (16%) CG: 12/33 (16%) |
| Vogel et al. [32] a | evaluation of effects when smart wearables are used by patients undergoing an outpatient CR | randomized: n = 36, IG: n = 19 (6 dropouts), CG: n = 17 (1 dropout) | IG: 62 ± 9 CG: 64 ± 10 | IG: 0/19 (0%) CG: 0/17 (0%) |
| Outpatient CR followed by home-based CR with mTechs (IG) vs. outpatient CR without further formal CR (CG) | ||||
| Avila et al. [13] | evaluation of effects and costs of home-based exercise training with telemonitoring guidance | randomized: n = 90, IG: n = 30 (2 dropouts), CG: n = 30 (4 dropouts) | IG: 59 ± 13 CG: 62 ± 8 | IG: 4/30 (13%) CG: 3/30 (10%) |
| Duscha et al. [14] | evaluation of effects of a mobile health cardiovascular prevention program for patients recently graduated from CR | randomized: n = 32, IG: n = 21 (5 dropouts), CG: n = 11 (2 dropouts) | IG: 60 ± 8 CG: 67 ± 7 | IG: 3/16 (19%) CG: 3/9 (33%) |
| Fang et al. [33] | evaluation of effects of home-based cardiac telerehabilitation program in low-risk patients after percutaneous coronary | randomized: n = 80, IG: n = 33 (7 dropouts), CG: no formal CR, n = 34 (6 dropouts) | IG: 60 ± 9 CG: 61 ± 10 | IG: 12/33 (36%) CG: 13/34 (38%) |
| Frederix et al. [15] | evaluation of effects of a PA telemonitoring program for patients who completed phase II CR | randomized: n = 80, IG: n = 40 (6 dropouts), CG: n = 40 (8 dropouts) | IG: 58 ± 9 CG: 63 ± 10 | IG: 8/40 (20%) CG: 6/40 (15%) |
| Frederix et al. [16] | evaluation of health benefits and cost-efficacy of an additional cardiac telerehabilitation program | randomized: n = 140, IG: n = 70 (8 dropouts), CG: n = 70 (6 dropouts) | IG: 61 ± 9 CG: 61 ± 8 | IG: 10/62 (16%) CG: 13/64 (20%) |
| Piotrowicz et al. [12] | evaluation of safety, effectiveness, adherence to and acceptance of home-based telemonitored Nordic walking after cardiovascular hospitalization | randomized: n = 111, IG: n = 77 (2 dropouts), CG: n = 34 (2 dropouts) | IG: 54 ± 11 CG: 62 ± 13 | IG: 11/75 (15%) CG: 1/32 (3%) |
| Piotrowicz et al. [30] | evaluation of quality-of-life outcomes after a hybrid comprehensive cardiac telerehabilitation after cardiovascular hospitalization | randomized: n = 850, IG: n = 425 (39 dropouts), CG: no formal CR, n = 425 (30 dropouts) | IG: 63 ± 11 CG: 62 ± 10 | IG: 48/425 (11%) CG: 49/425 (12%) |
| Skobel et al. [17] b | evaluation of effects of a mobile-based CR program during phase III rehabilitation | randomized: n = 118, IG: n = 55 (36 dropouts), CG: n = 63 (21 dropouts) | IG: 60 CG: 58 | IG: 5/55 (9%) CG: 8/63 (8%) |
| Snoek et al. [19] | evaluation of acute and sustained effects of a heart-rate-based telerehabilitation program, following the completion of outpatient CR | randomized: n = 122, IG: n = 61 (1 dropout), CG: n = 61 (1 dropout) | IG: 60 ± 8 CG: 59 ± 11 | IG: 11/61 (18%) CG: 11/61 (18%) |
| Vogel et al. [32] a | evaluation of effects when smart wearables are used by patients undergoing an outpatient CR | randomized: n = 36, IG: n = 19 (6 dropouts), CG: n = 17 (1 dropout) | IG: 62 ± 9 CG: 64 ± 10 | IG: 0/19 (0%) CG: 0/17 (0%) |
| Study Characteristics | Effects a | |||||
|---|---|---|---|---|---|---|
| Duration (Weeks) | Number of Patients | Exercise Capacity | IG: Within-Group | CG: Within-Group | ∆ IG vs. ∆ CG: Between-Group | |
| mTechs during home-based CR vs. outpatient CR (n = 297) | ||||||
| Avila et al. [13] b | 6 w | n = 90 | ⩒O2peak | - | - | - |
| Kraal et al. [20] | 6 w | n = 45 | ⩒O2peak | ↑ | ↑ | - |
| Maddison et al. [21] | 6 w | n = 162 | ⩒O2peak | ⊗ | ⊗ | ⊙ |
| Outpatient CR with mTechs vs. outpatient without mTechs (n = 102) | ||||||
| Rosario et al. [31] | 6 w | n = 66 | 6MWT | - | - | - |
| Vogel et al. [32] c | 12 w | n = 36 | Peak W | ↑ | ↑ | - |
| Outpatient CR followed by home-based CR with mTechs vs. outpatient CR without further formal CR (n = 1659) | ||||||
| Avila et al. [13] d | 12 w | n = 90 | ⩒O2peak | - | - | ↑ |
| Duscha et al. [14] | 12 w | n = 32 | ⩒O2peak | ↑ | ↓ | ↑ |
| Fang et al. [33] | 6 w | n = 80 | 6MWT | ↑ | ↑ | ↑ |
| Frederix et al. [15] | 18 w | n = 80 | ⩒O2peak | ↑ | - | ↑ |
| Frederix et al. [16] e | 24 w | n = 140 | ⩒O2peak | ↑ | - | ↑ |
| Piotrowicz et al. [12] f | 8 w | n = 111 | ⩒O2peak | ↑ | - | ↑ |
| Piotrowicz et al. [30] f | 9 w | n = 850 | ⩒O2peak | ↑ | ↑ | ↑ |
| Skobel et al. [17] | 24 w | n = 118 | ⩒O2peak | - | - | ↑ |
| Snoek et al. [19] | 14 w | n = 122 | ⩒O2peak | ↑ | ↑ | - |
| Vogel et al. [32] g | 12 w | n = 36 | Peak W | ↑ | ↓ | ↑ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Meinhart, F.; Stütz, T.; Sareban, M.; Kulnik, S.T.; Niebauer, J. Mobile Technologies to Promote Physical Activity during Cardiac Rehabilitation: A Scoping Review. Sensors 2021, 21, 65. https://doi.org/10.3390/s21010065
Meinhart F, Stütz T, Sareban M, Kulnik ST, Niebauer J. Mobile Technologies to Promote Physical Activity during Cardiac Rehabilitation: A Scoping Review. Sensors. 2021; 21(1):65. https://doi.org/10.3390/s21010065
Chicago/Turabian StyleMeinhart, Florian, Thomas Stütz, Mahdi Sareban, Stefan Tino Kulnik, and Josef Niebauer. 2021. "Mobile Technologies to Promote Physical Activity during Cardiac Rehabilitation: A Scoping Review" Sensors 21, no. 1: 65. https://doi.org/10.3390/s21010065
APA StyleMeinhart, F., Stütz, T., Sareban, M., Kulnik, S. T., & Niebauer, J. (2021). Mobile Technologies to Promote Physical Activity during Cardiac Rehabilitation: A Scoping Review. Sensors, 21(1), 65. https://doi.org/10.3390/s21010065





