Combined Use of Vis-NIR and XRF Sensors for Tropical Soil Fertility Analysis: Assessing Different Data Fusion Approaches
Abstract
1. Introduction
2. Materials and Methods
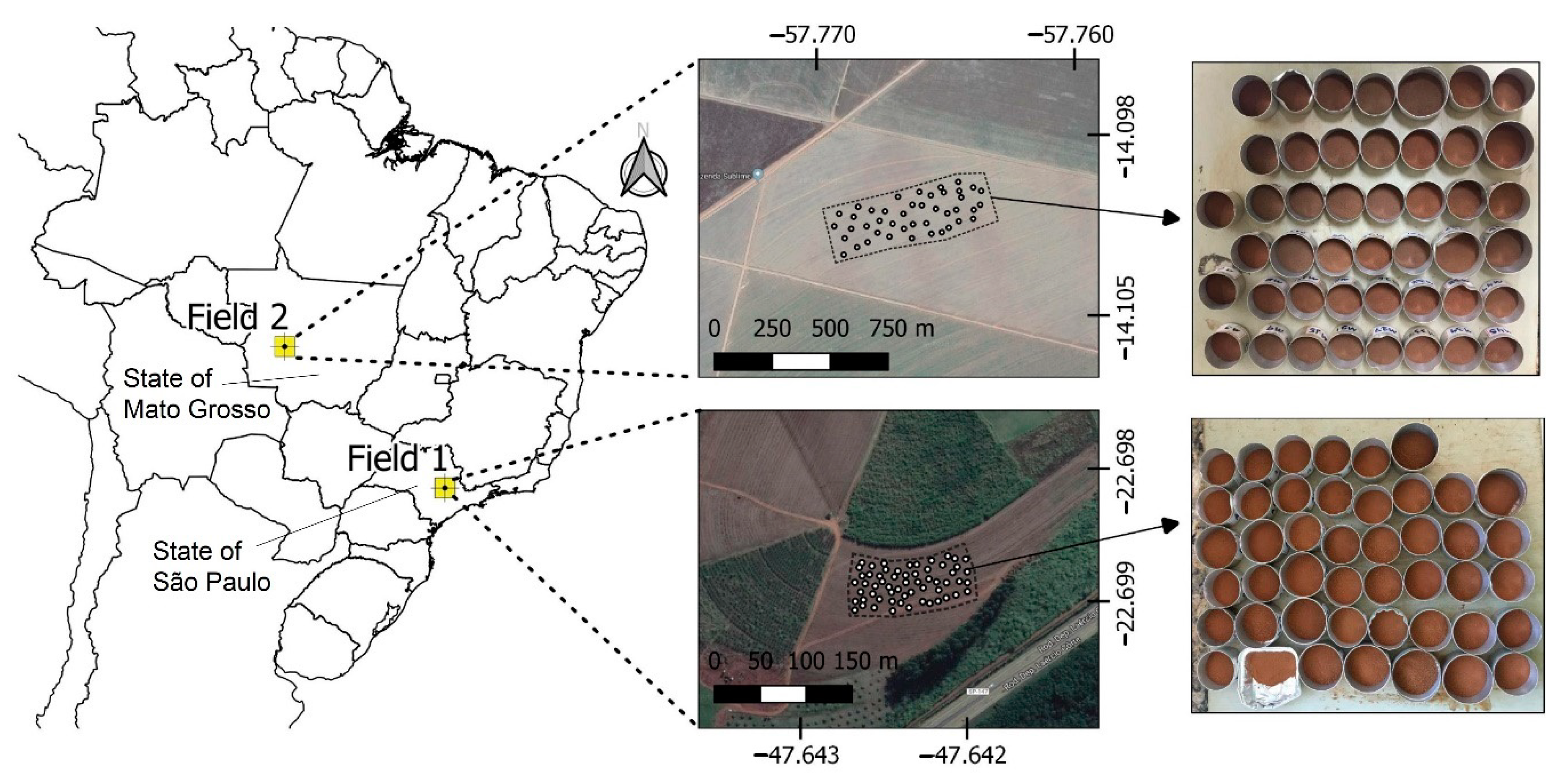
2.1. Study Sites and Soil Samples
2.2. Reference Analyses
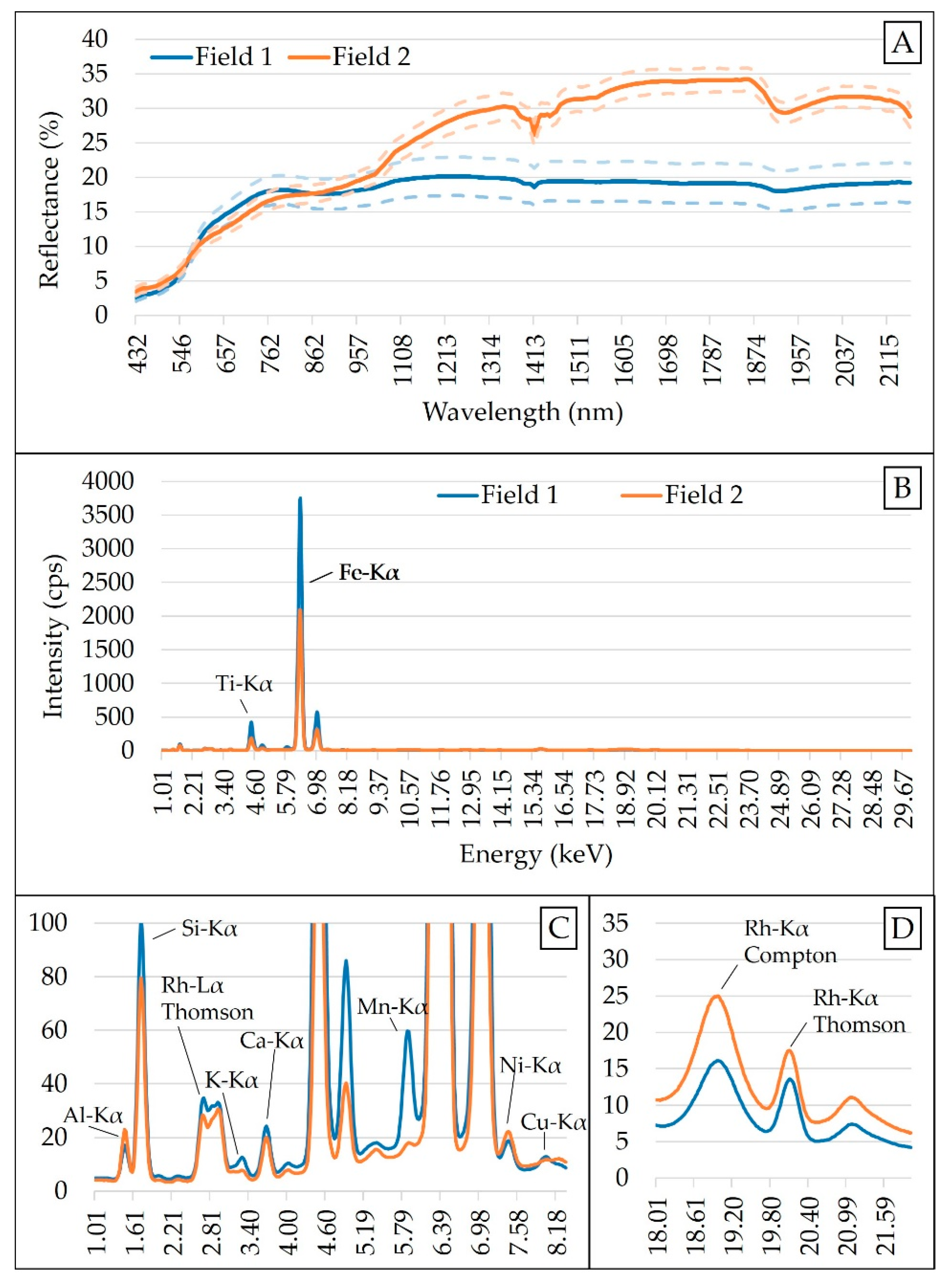
2.3. XRF Measurements and Selection of Emission Lines
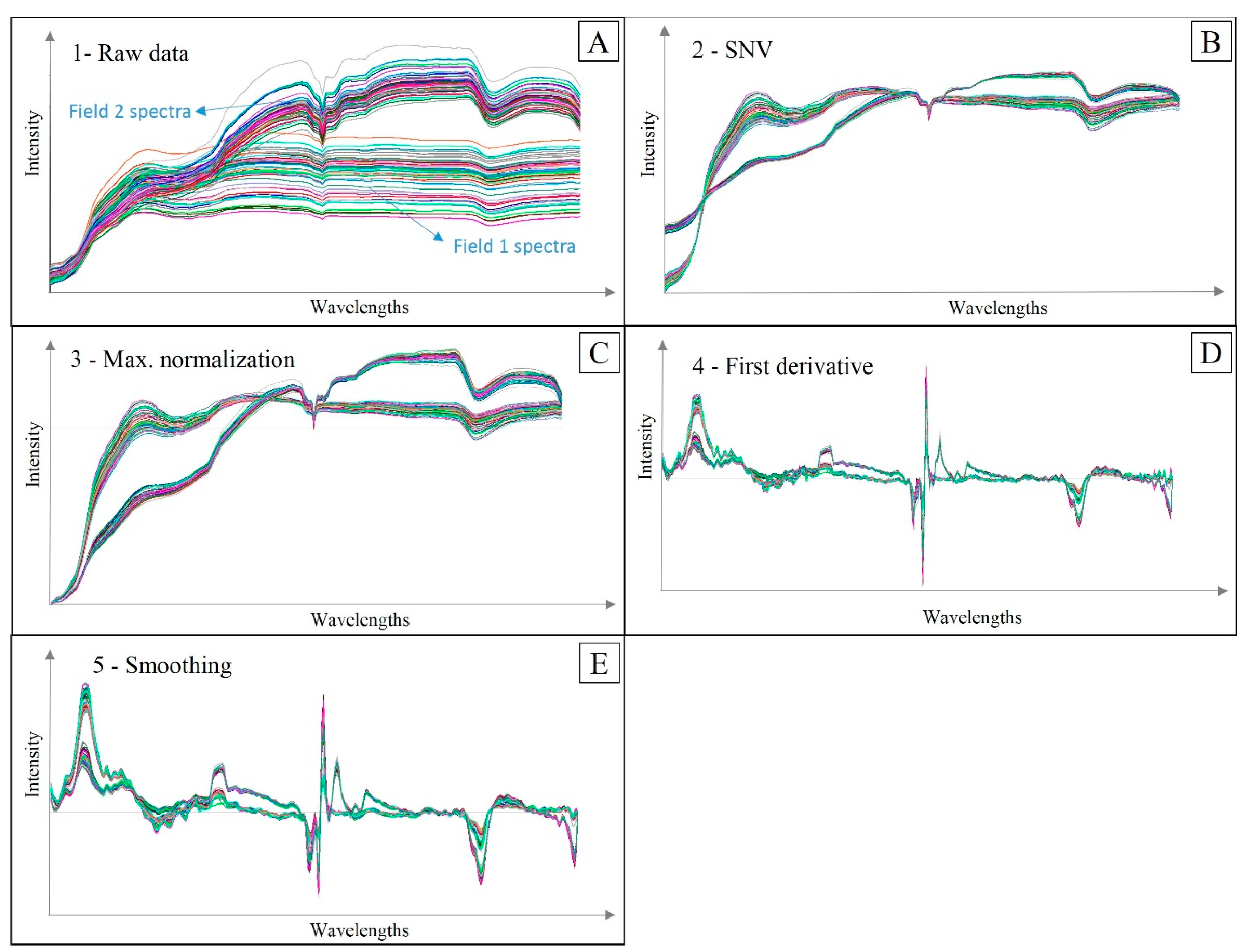
2.4. Vis-NIR Measurements and Spectra Pre-Processing
2.5. Modeling
2.5.1. Individual Models Using vis-NIR and XRF Sensors Alone
2.5.2. Data Fusion Approaches
- GR2, in which the predictions given by the vis-NIR and XRF individual models are fused according to the following Equation (1):where Y is the fused (potentially more accurate) estimation of the desired soil property; Yvis-NIR and YXRF are the corresponding predictions given by vis-NIR and XRF individual models (as described in Section 2.5.1), respectively; W0, Wvis-NIR, and WXRF are the weights of the MLR determined by minimizing the mean squared error, where the first parameter is the value of the line intercept and the others are the weights of the prediction models of both sensors;
- GR3, wherein the predictions given by the SF approach are also included in the fusion process, as described by the following Equation (2):in which YSF and WSF are, respectively, the SF predictions and their corresponding weights.
3. Results
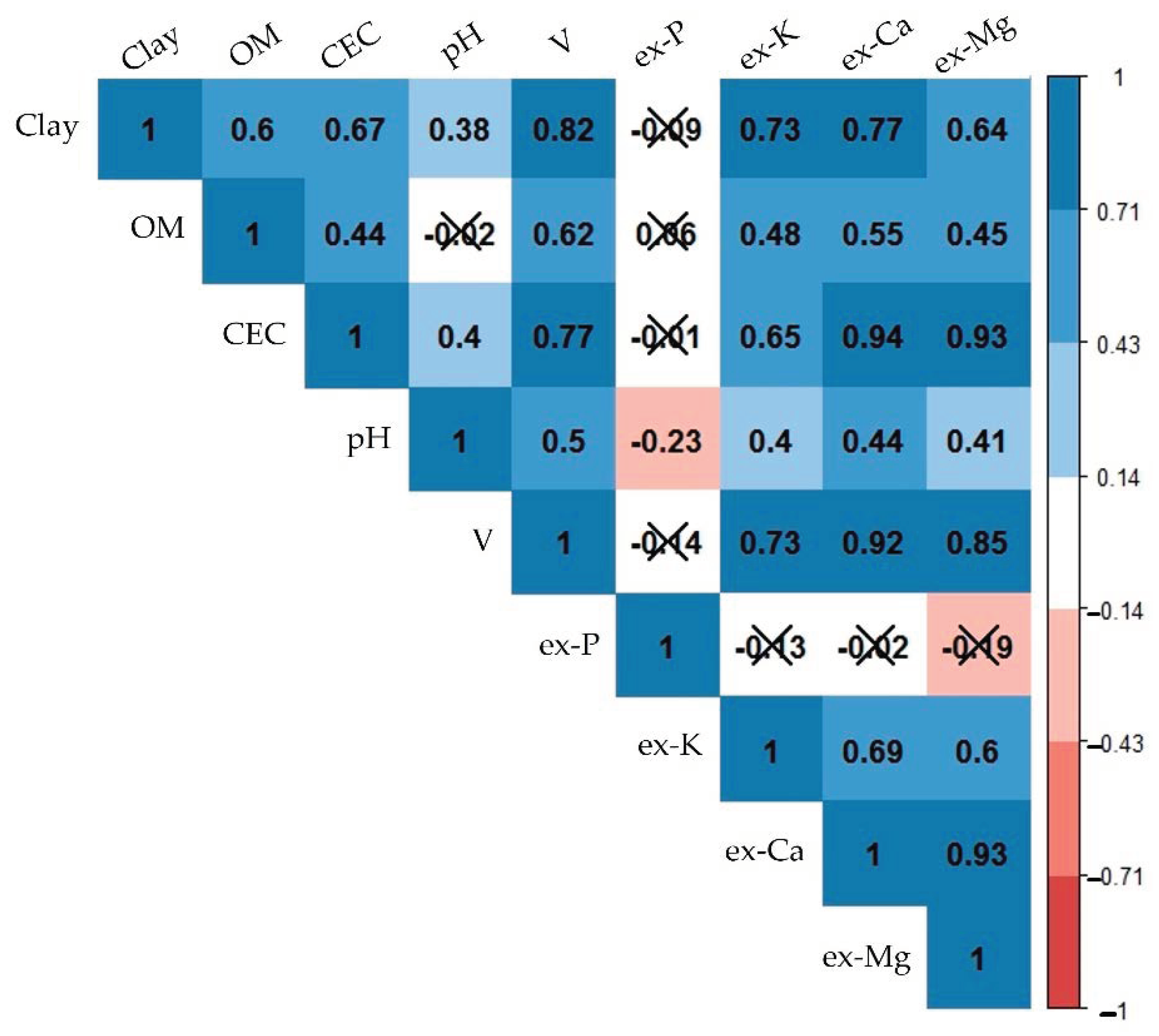
3.1. Laboratory Measured Soil Properties
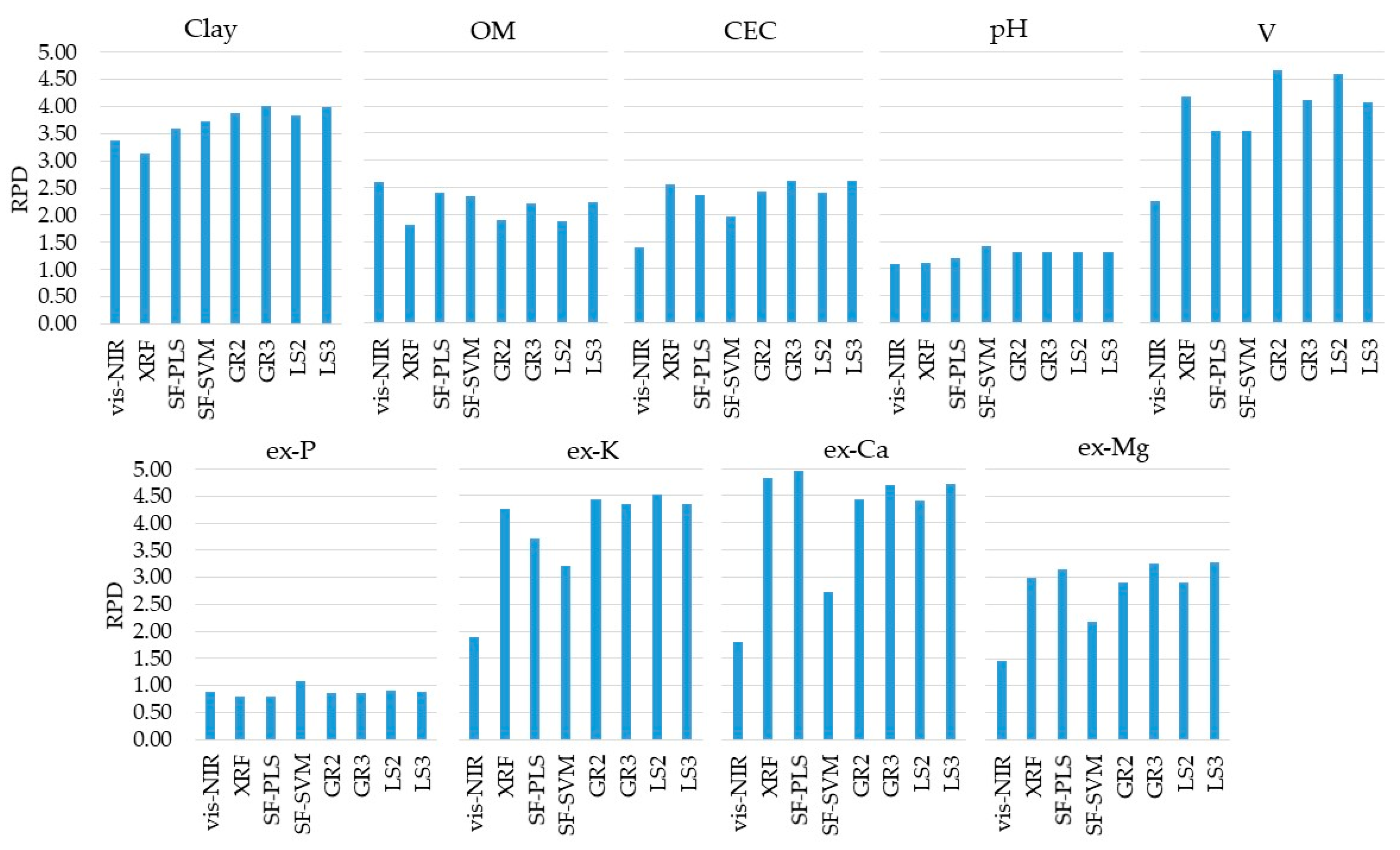
3.2. Prediction Performances of Single-Sensor and Data Fusion Models
4. Discussion
4.1. vis-NIR and XRF Individual Performance
4.2. Performance of Data Fusion Approches
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
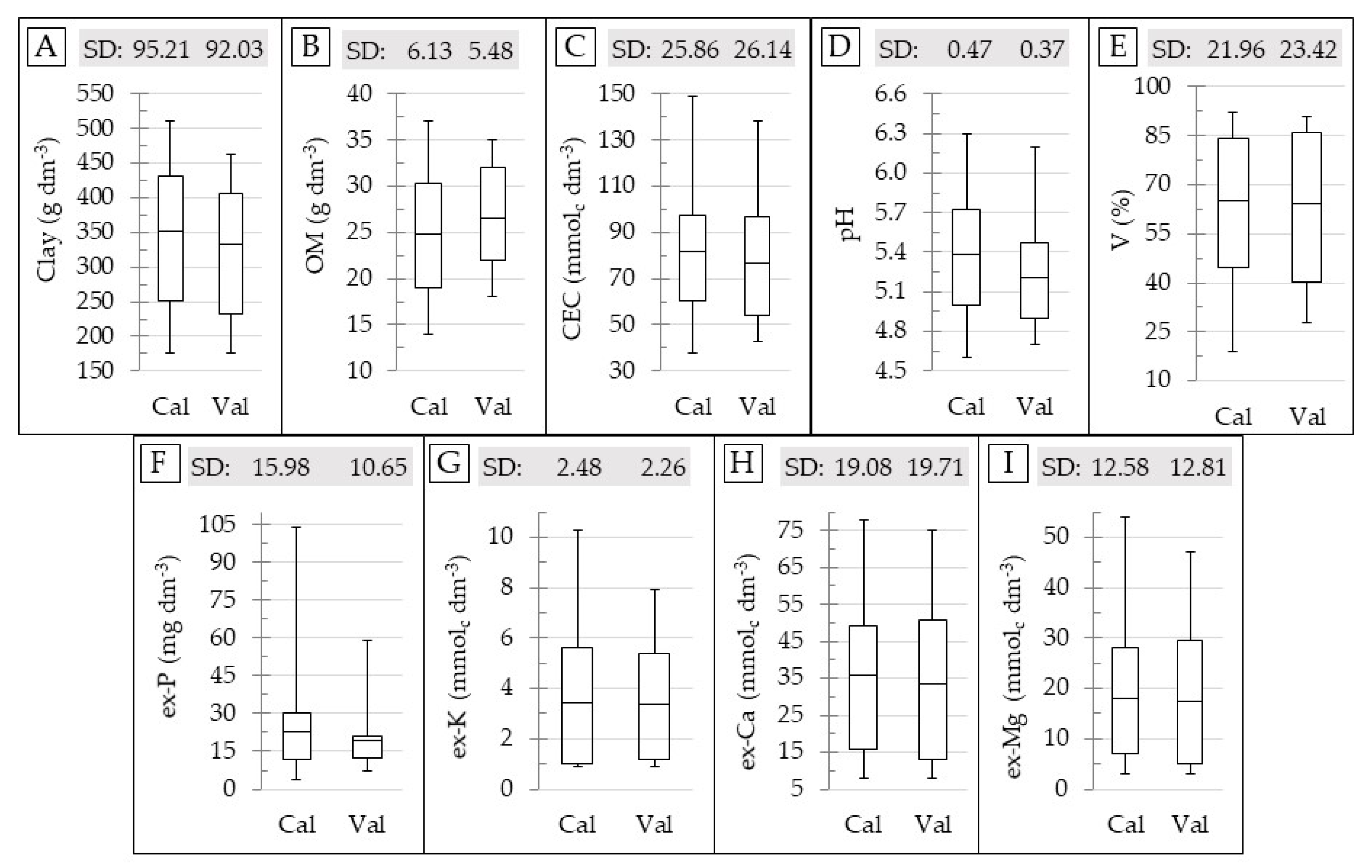
| Clay | OM 1 | CEC 2 | pH | V 3 | ex-P 4 | ex-K 4 | ex-Ca 4 | ex-Mg 4 | |
| -------------------------------------------- Calibration set (n = 68) -------------------------------------------- | |||||||||
| Skewness | −0.22 | 0.14 | 0.46 | 0.50 | −0.57 | 2.26 | 0.59 | 0.25 | 0.81 |
| Kurtosis | −1.22 | −1.10 | −0.41 | −1.02 | −1.16 | 8.75 | −0.79 | −1.01 | −0.14 |
| -------------------------------------------- Validation set (n = 34) -------------------------------------------- | |||||||||
| Skewness | −0.45 | −0.11 | 0.53 | 0.83 | −0.35 | 2.16 | 0.35 | 0.34 | 0.63 |
| Kurtosis | −1.39 | −1.45 | −0.63 | 0.26 | −1.62 | 5.78 | −1.35 | −1.11 | −0.73 |
| Clay | OM 1 | CEC 2 | pH | V 3 | ex-P 4 | ex-K 4 | ex-Ca 4 | ex-Mg 4 | |
| -------------------------------------- R2 -------------------------------------- | |||||||||
| vis-NIR | 0.93 | 0.86 | 0.51 | 0.19 | 0.80 | 0.07 | 0.74 | 0.68 | 0.52 |
| XRF | 0.92 | 0.74 | 0.88 | 0.34 | 0.95 | 0.01 | 0.95 | 0.96 | 0.89 |
| SF-PLS | 0.92 | 0.83 | 0.82 | 0.31 | 0.92 | 0.00 | 0.93 | 0.96 | 0.90 |
| SF-SVM | 0.95 | 0.85 | 0.79 | 0.49 | 0.92 | 0.14 | 0.90 | 0.88 | 0.81 |
| GR2 | 0.93 | 0.72 | 0.83 | 0.41 | 0.95 | 0.00 | 0.95 | 0.95 | 0.88 |
| GR3 | 0.94 | 0.79 | 0.85 | 0.43 | 0.94 | 0.00 | 0.95 | 0.95 | 0.91 |
| LS2 | 0.94 | 0.80 | 0.85 | 0.44 | 0.94 | 0.00 | 0.95 | 0.96 | 0.91 |
| LS3 | 0.93 | 0.72 | 0.83 | 0.42 | 0.95 | 0.00 | 0.95 | 0.95 | 0.88 |
| -------------------------------------- RMSE -------------------------------------- | |||||||||
| vis-NIR | 27.32 | 2.10 | 18.66 | 0.34 | 10.38 | 12.05 | 1.20 | 10.98 | 8.85 |
| XRF | 29.40 | 3.01 | 10.19 | 0.33 | 5.60 | 13.27 | 0.53 | 4.09 | 4.28 |
| SF-PLS | 25.58 | 2.28 | 11.05 | 0.31 | 6.63 | 13.43 | 0.61 | 3.98 | 4.07 |
| SF-SVM | 24.63 | 2.34 | 13.28 | 0.26 | 6.61 | 9.89 | 0.71 | 7.26 | 5.89 |
| GR2 | 23.74 | 2.89 | 10.74 | 0.28 | 5.04 | 12.42 | 0.51 | 4.45 | 4.42 |
| GR3 | 22.93 | 2.48 | 9.99 | 0.28 | 5.70 | 12.45 | 0.52 | 4.20 | 3.94 |
| LS2 | 23.11 | 2.47 | 9.99 | 0.28 | 5.77 | 11.97 | 0.52 | 4.18 | 3.92 |
| LS3 | 24.01 | 2.92 | 10.90 | 0.28 | 5.11 | 11.70 | 0.50 | 4.46 | 4.43 |
| -------------------------------------- RMSE% -------------------------------------- | |||||||||
| vis-NIR | 9.49 | 12.37 | 19.45 | 22.42 | 16.48 | 23.16 | 17.10 | 16.39 | 20.12 |
| XRF | 10.21 | 17.73 | 10.62 | 22.15 | 8.89 | 25.51 | 7.60 | 6.11 | 9.74 |
| SF-PLS | 8.88 | 13.41 | 11.52 | 20.67 | 10.52 | 25.83 | 8.71 | 5.94 | 9.25 |
| SF-SVM | 8.55 | 13.77 | 13.85 | 17.41 | 10.50 | 19.02 | 10.11 | 10.84 | 13.39 |
| GR2 | 8.24 | 17.00 | 11.20 | 18.67 | 8.00 | 23.88 | 7.29 | 6.64 | 10.05 |
| GR3 | 7.96 | 14.59 | 10.42 | 18.67 | 9.05 | 23.94 | 7.43 | 6.27 | 8.95 |
| LS2 | 8.02 | 14.53 | 10.42 | 18.67 | 9.16 | 23.02 | 7.43 | 6.24 | 8.91 |
| LS3 | 8.34 | 17.18 | 11.37 | 18.67 | 8.11 | 22.50 | 7.14 | 6.66 | 10.07 |
| -------------------------------------- RPD -------------------------------------- | |||||||||
| vis-NIR | 3.37 | 2.61 | 1.40 | 1.10 | 2.26 | 0.88 | 1.89 | 1.79 | 1.45 |
| XRF | 3.13 | 1.82 | 2.57 | 1.11 | 4.18 | 0.80 | 4.26 | 4.82 | 2.99 |
| SF-PLS | 3.60 | 2.40 | 2.37 | 1.19 | 3.53 | 0.79 | 3.71 | 4.95 | 3.15 |
| SF-SVM | 3.74 | 2.34 | 1.97 | 1.41 | 3.54 | 1.08 | 3.20 | 2.71 | 2.17 |
| GR2 | 3.88 | 1.90 | 2.43 | 1.32 | 4.65 | 0.86 | 4.44 | 4.43 | 2.90 |
| GR3 | 4.01 | 2.21 | 2.62 | 1.32 | 4.11 | 0.86 | 4.35 | 4.69 | 3.25 |
| LS2 | 3.98 | 2.22 | 2.62 | 1.32 | 4.06 | 0.89 | 4.35 | 4.72 | 3.27 |
| LS3 | 3.83 | 1.88 | 2.40 | 1.32 | 4.58 | 0.91 | 4.53 | 4.42 | 2.89 |
| Single Sensor | Multiple Sensor | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| SF-PLS | SF-SVM | GR2 | GR3 | LS2 | LS3 | |||||||||
| RMSE | Techni. 5 | RMSE | % RI 6 | RMSE | % RI | RMSE | % RI | RMSE | % RI | RMSE | % RI | RMSE | % RI | |
| Clay | 27.32 | vis-NIR | 25.58 | 6 | 24.63 | 10 | 23.74 | 13 | 22.93 * | 16 | 24.01 | 12 | 23.11 | 15 |
| 29.40 | XRF | 13 | 16 | 19 | 22 | 18 | 21 | |||||||
| OM 1 | 2.10 * | vis-NIR | 2.28 | −8 | 2.34 | −11 | 2.89 | −37 | 2.48 | −18 | 2.92 | −39 | 2.47 | −17 |
| 3.01 | XRF | 24 | 22 | 4 | 18 | 3 | 18 | |||||||
| CEC 2 | 18.66 | vis-NIR | 11.05 | 41 | 13.28 | 29 | 10.74 | 42 | 9.99 * | 46 | 10.90 | 42 | 9.99 | 46 |
| 10.19 | XRF | −8 | −30 | −5 | 2 | −7 | 2 | |||||||
| pH | 0.34 | vis-NIR | 0.31 | 8 | 0.26 * | 22 | 0.28 | 17 | 0.28 | 17 | 0.28 | 17 | 0.28 | 17 |
| 0.33 | XRF | 7 | 21 | 16 | 16 | 16 | 16 | |||||||
| V 3 | 10.38 | vis-NIR | 6.63 | 36 | 6.61 | 36 | 5.04 * | 51 | 5.70 | 45 | 5.11 | 51 | 5.77 | 44 |
| 5.60 | XRF | −18 | −18 | 10 | −2 | 9 | −3 | |||||||
| ex-P 4 | 12.05 | vis-NIR | 13.43 | −11 | 9.89 | 18 | 12.42 | −3 | 12.45 | −3 | 11.70 * | 3 | 11.97 | 1 |
| 13.27 | XRF | −1 | 25 | 6 | 6 | 12 | 10 | |||||||
| ex-K 4 | 1.20 | vis-NIR | 0.61 | 49 | 0.71 | 41 | 0.51 | 57 | 0.52 | 57 | 0.50 * | 58 | 0.52 | 57 |
| 0.53 | XRF | −15 | −33 | 4 | 2 | 6 | 2 | |||||||
| ex-Ca 4 | 10.98 | vis-NIR | 3.98 * | 64 | 7.26 | 34 | 4.45 | 59 | 4.20 | 62 | 4.46 | 59 | 4.18 | 62 |
| 4.09 | XRF | 3 | −78 | −9 | −3 | −9 | −2 | |||||||
| ex-Mg 4 | 8.85 | vis-NIR | 4.07 | 54 | 5.89 | 33 | 4.42 | 50 | 3.94 | 55 | 4.43 | 50 | 3.92 * | 56 |
| 4.28 | XRF | 5 | −38 | −3 | 8 | −3 | 9 | |||||||
| Clay | OM 1 | CEC 2 | pH | V 3 | ex-P 4 | ex-K 4 | ex-Ca 4 | ex-Mg 4 | |
|---|---|---|---|---|---|---|---|---|---|
| K ptc | 0.81 | 0.67 | 0.58 | 0.30 | 0.80 | −0.13 | 0.90 | 0.70 | 0.58 |
| Ca ptc | 0.70 | 0.44 | 0.85 | 0.51 | 0.85 | 0.01 | 0.53 | 0.91 | 0.84 |
References
- Viscarra Rossel, R.A.; Adamchuk, V.I.; Sudduth, K.A.; Mc Kenzie, N.J.; Lobsey, C. Proximal soil sensing: An effective approach for soil measurements in space and time. Adv. Agron. 2011, 113, 237–282. [Google Scholar] [CrossRef]
- Gredilla, A.; de Vallejuelo, S.F.O.; Elejoste, N.; de Diego, A.; Madariaga, J.M. Non-destructive Spectroscopy combined with chemometrics as a tool for Green Chemical Analysis of environmental samples: A review. TrAC Trend Anal. Chem. 2016, 76, 30–39. [Google Scholar] [CrossRef]
- Viscarra Rossel, R.A.; Bouma, J. Soil sensing: A new paradigm for agriculture. Agric. Syst. 2016, 148, 71–74. [Google Scholar] [CrossRef]
- Pandey, S.; Bhatta, N.P.; Paudel, P.; Pariyar, R.; Maskey, K.H.; Khadka, J.; Panday, D. Improving fertilizer recommendations for Nepalese farmers with the help of soil-testing mobile van. J. Crop Improv. 2018, 32, 19–32. [Google Scholar] [CrossRef]
- Mouazen, A.M.; Kuang, B. On-line visible and near infrared spectroscopy for in-field phosphorous management. Soil Tillage Res. 2016, 155, 471–477. [Google Scholar] [CrossRef]
- Molin, J.P.; Faulin, G.D.C. Spatial and temporal variability of soil electrical conductivity related to soil moisture. Sci. Agric. 2013, 70, 1–5. [Google Scholar] [CrossRef]
- Demattê, J.A.; Ramirez-Lopez, L.; Marques, K.P.P.; Rodella, A.A. Chemometric soil analysis on the determination of specific bands for the detection of magnesium and potassium by spectroscopy. Geoderma 2017, 288, 8–22. [Google Scholar] [CrossRef]
- Nawar, S.; Delbecque, N.; Declercq, Y.; Smedt, P.; Finke, P.; Verdoodt, A.; Meirvenne, M.V.; Mouazen, A.M. Can spectral analyses improve measurement of key soil fertility parameters with X-ray fluorescence spectrometry? Geoderma 2019, 350, 29–39. [Google Scholar] [CrossRef]
- Kuang, B.; Mahmood, H.S.; Quraishi, M.Z.; Hoogmoed, W.B.; Mouazen, A.M.; van Henten, E.J. Sensing soil properties in the laboratory, in situ, and on-line: A review. Adv. Agron. 2012, 114, 155–223. [Google Scholar] [CrossRef]
- Nawar, S.; Corstanje, R.; Halcro, G.; Mulla, D.; Mouazen, A.M. Delineation of soil management zones for variable-rate fertilization: A review. Adv. Agron. 2017, 143, 175–245. [Google Scholar] [CrossRef]
- Molin, J.P.; Tavares, T.R. Sensor systems for mapping soil fertility attributes: Challenges, advances and perspectives in Brazilian tropical soils. Eng. Agric. 2019, 39, 126–147. [Google Scholar] [CrossRef]
- Gałuszka, A.; Migaszewski, Z.M.; Namieśnik, J. Moving your laboratories to the field—Advantages and limitations of the use of field portable instruments in environmental sample analysis. Environ. Res. 2015, 140, 593–603. [Google Scholar] [CrossRef] [PubMed]
- Horta, A.; Malone, B.; Stockmann, U.; Minasny, B.; Bishop, T.F.A.; McBratney, A.B.; Pallasser, R.; Pozza, L. Potential of integrated field spectroscopy and spatial analysis for enhanced assessment of soil contamination: A prospective review. Geoderma 2015, 241, 180–209. [Google Scholar] [CrossRef]
- Brown, D.J.; Shepherd, K.D.; Walsh, M.G.; Mays, M.D.; Reinsch, T.G. Global soil characterization with VNIR diffuse reflectance spectroscopy. Geoderma 2006, 132, 273–290. [Google Scholar] [CrossRef]
- Stenberg, B.; Viscarra Rossel, R.A.; Mouazen, A.M.; Wetterlind, J. Visible and near infrared spectroscopy in soil science. Adv. Agron. 2010, 107, 163–215. [Google Scholar] [CrossRef]
- Demattê, J.A.M.; Campos, R.C.; Alves, M.C.; Fiorio, P.R.; Nanni, M.R. Visible–NIR reflectance: A new approach on soil evaluation. Geoderma 2004, 121, 95–112. [Google Scholar] [CrossRef]
- Demattê, J.A.M.; Alves, M.R.; Gallo, B.C.; Fongaro, C.T.; Souza, A.B.; Romero, D.J.; Sato, M.V. Hyperspectral remote sensing as an alternative to estimate soil attributes. Rev. Cienc. Agron. 2015, 46, 223–232. [Google Scholar] [CrossRef]
- Tsakiridis, N.L.; Tziolas, N.V.; Theocharis, J.B.; Zalidis, G.C. A genetic algorithm-based stacking algorithm for predicting soil organic matter from vis–NIR spectral data. Eur. J. Soil Sci. 2019, 70, 578–590. [Google Scholar] [CrossRef]
- Tsakiridis, N.L.; Keramaris, K.D.; Theocharis, J.B.; Zalidis, G.C. Simultaneous prediction of soil properties from VNIR-SWIR spectra using a localized multi-channel 1-D convolutional neural network. Geoderma 2020, 367, 114208. [Google Scholar] [CrossRef]
- Ramirez-Lopez, L.; Wadoux, A.C.; Franceschini, M.H.D.; Terra, F.S.; Marques, K.P.P.; Sayão, V.M.; Demattê, J.A.M. Robust soil mapping at the farm scale with vis–NIR spectroscopy. Eur. J. Soil Sci. 2019, 70, 378–393. [Google Scholar] [CrossRef]
- Nanni, M.R.; Demattê, J.A.M. Spectral reflectance methodology in comparison to traditional soil analysis. Soil Sci. Soc. Am. J. 2006, 70, 393–407. [Google Scholar] [CrossRef]
- Terra, F.S.; Demattê, J.A.M.; Viscarra Rossel, R.A. Spectral libraries for quantitative analyses of tropical Brazilian soils: Comparing vis–NIR and mid-IR reflectance data. Geoderma 2015, 255, 81–93. [Google Scholar] [CrossRef]
- Munnaf, M.A.; Nawar, S.; Mouazen, A.M. Estimation of Secondary Soil Properties by Fusion of Laboratory and On-Line Measured Vis–NIR Spectra. Remote Sens. 2019, 11, 2819. [Google Scholar] [CrossRef]
- Tavares, T.R.; Molin, J.P.; Nunes, L.C.; Alves, E.E.N.; Melquiades, F.L.; Carvalho, H.W.P.; Mouazen, A.M. Effect of X-ray tube configuration on measurement of key soil fertility attributes with XRF. Remote Sens. 2020, 12, 963. [Google Scholar] [CrossRef]
- Tavares, T.R.; Mouazen, A.M.; Alves, E.E.N.; dos Santos, F.R.; Melquiades, F.L.; Pereira de Carvalho, H.W.; Molin, J.P. Assessing Soil Key Fertility Attributes Using a Portable X-Ray Fluorescence: A Simple Method to Overcome Matrix Effect. Agronomy 2020, 10, 787. [Google Scholar] [CrossRef]
- O’Rourke, S.M.; Stockmann, U.; Holden, N.M.; McBratney, A.B.; Minasny, B. An assessment of model averaging to improve predictive power of portable vis-NIR and XRF for the determination of agronomic soil properties. Geoderma 2016, 279, 31–44. [Google Scholar] [CrossRef]
- Zhu, Y.; Weindorf, D.C.; Zhang, W. Characterizing soils using a portable X-ray fluorescence spectrometer: 1. Soil texture. Geoderma 2011, 167, 167–177. [Google Scholar] [CrossRef]
- Lima, T.M.; Weindorf, D.C.; Curi, N.; Guilherme, L.R.; Lana, R.M.; Ribeiro, B.T. Elemental analysis of Cerrado agricultural soils via portable X-ray fluorescence spectrometry: Inferences for soil fertility assessment. Geoderma 2019, 353, 264–272. [Google Scholar] [CrossRef]
- Sharma, A.; Weindorf, D.C.; Man, T.; Aldabaa, A.A.A.; Chakraborty, S. Characterizing soils via portable X-ray fluorescence spectrometer: 3, Soil reaction (pH). Geoderma 2014, 232, 141–147. [Google Scholar] [CrossRef]
- Sharma, A.; Weindorf, D.C.; Wang, D.; Chakraborty, S. Characterizing soils via portable X-ray fluorescence spectrometer: 4. Cation exchange capacity (CEC). Geoderma 2015, 239, 130–134. [Google Scholar] [CrossRef]
- Teixeira, A.F.D.S.; Weindorf, D.C.; Silva, S.H.G.; Guilherme, L.R.G.; Curi, N. Portable X-ray fluorescence (pXRF) spectrometry applied to the prediction of chemical attributes in Inceptisols under different land uses. Ciência Agrotecnol. 2018, 42, 501–512. [Google Scholar] [CrossRef]
- Santos, F.R.; Oliveira, J.F.; Bona, E.; Santos, J.V.F.; Barboza, G.M.; Melquiades, F.L. EDXRF spectral data combined with PLSR to determine some soil fertility indicators. Microchem. J. 2020, 152, 104275. [Google Scholar] [CrossRef]
- Morona, F.; dos Santos, F.R.; Brinatti, A.M.; Melquiades, F.L. Quick analysis of organic matter in soil by energy-dispersive X-ray fluorescence and multivariate analysis. Appl. Radiat. Isot. 2017, 130, 13–20. [Google Scholar] [CrossRef] [PubMed]
- Silva, S.H.G.; Teixeira, A.F.D.S.; Menezes, M.D.D.; Guilherme, L.R.G.; Moreira, F.M.D.S.; Curi, N. Multiple linear regression and random forest to predict and map soil properties using data from portable X-ray fluorescence spectrometer (pXRF). Ciência Agrotecnol. 2017, 41, 648–664. [Google Scholar] [CrossRef]
- Tavares, T.R.; Nunes, L.C.; Alves, E.E.N.; Almeida, E.; Maldaner, L.F.; Krug, F.J.; Carvalho, H.W.P.; Molin, J.P. Simplifying sample preparation for soil fertility analysis by X-ray fluorescence spectrometry. Sensors 2019, 19, 5066. [Google Scholar] [CrossRef]
- Andrade, R.; Faria, W.M.; Silva, S.H.G.; Chakraborty, S.; Weindorf, D.C.; Mesquita, L.F.; Guilherme, L.R.G.; Curi, N. Prediction of soil fertility via portable X-ray fluorescence (pXRF) spectrometry and soil texture in the Brazilian Coastal Plains. Geoderma 2020, 357, 113960. [Google Scholar] [CrossRef]
- Adamchuk, V.I.; Viscarra Rossel, R.; Sudduth, K.A.; Lammers, P.S. Sensor fusion for precision agriculture. In Sensor Fusion-Foundation and Applications; Thomas, C., Ed.; InTech: Rijeka, Croatia, 2011; pp. 27–40. [Google Scholar]
- Mahmood, H.S.; Hoogmoed, W.B.; van Henten, E.J. Sensor data fusion to predict multiple soil properties. Precis. Agric. 2012, 13, 628–645. [Google Scholar] [CrossRef]
- Mouazen, A.M.; Alhwaimel, S.A.; Kuang, B.; Waine, T. Multiple on-line soil sensors and data fusion approach for delineation of water holding capacity zones for site specific irrigation. Soil Tillage Res. 2014, 143, 95–105. [Google Scholar] [CrossRef]
- Castrignanò, A.; Buttafuoco, G.; Quarto, R.; Vitti, C.; Langella, G.; Terribile, F.; Venezia, A. A combined approach of sensor data fusion and multivariate geostatistics for delineation of homogeneous zones in an agricultural field. Sensors 2017, 17, 2794. [Google Scholar] [CrossRef]
- Xu, X.; Du, C.; Ma, F.; Shen, Y.; Wu, K.; Liang, D.; Zhou, J. Detection of soil organic matter from laser-induced breakdown spectroscopy (LIBS) and mid-infrared spectroscopy (FTIR-ATR) coupled with multivariate techniques. Geoderma 2019, 355, 113905. [Google Scholar] [CrossRef]
- Xu, D.; Zhao, R.; Li, S.; Chen, S.; Jiang, Q.; Zhou, L.; Shi, Z. Multi-sensor fusion for the determination of several soil properties in the Yangtze River Delta, China. Eur. J. Soil Sci. 2019, 70, 162–173. [Google Scholar] [CrossRef]
- Wang, D.; Chakraborty, S.; Weindorf, D.C.; Li, B.; Sharma, A.; Paul, S.; Ali, M.N. Synthesized use of VisNIR DRS and PXRF for soil characterization: Total carbon and total nitrogen. Geoderma 2015, 243, 157–167. [Google Scholar] [CrossRef]
- Benedet, L.; Faria, W.M.; Silva, S.H.G.; Mancini, M.; Demattê, J.A.M.; Guilherme, L.R.G.; Curi, N. Soil texture prediction using portable X-ray fluorescence spectrometry and visible near-infrared diffuse reflectance spectroscopy. Geoderma 2020, 376, 114553. [Google Scholar] [CrossRef]
- Weindorf, D.; Chakraborty, S. Portable Apparatus for Soil Chemical Characterization. Texas Tech University System. U.S. Patent US 10,107,770 B2, 23 October 2018. [Google Scholar]
- Castanedo, F. A review of data fusion techniques. Sci. World J. 2013, 2013. [Google Scholar] [CrossRef] [PubMed]
- Viscarra Rossel, R.; Walvoort, D.J.J.; McBratney, A.B.; Janik, L.J.; Skjemstad, J.O. Visible, near infrared, mid infrared or combined diffuse reflectance spectroscopy for simultaneous assessment of various soil properties. Geoderma 2006, 131, 59–75. [Google Scholar] [CrossRef]
- Xu, D.; Chen, S.; Viscarra Rossel, R.; Biswas, A.; Li, S.; Zhou, Y.; Shi, Z. X-ray fluorescence and visible near infrared sensor fusion for predicting soil chromium content. Geoderma 2019, 352, 61–69. [Google Scholar] [CrossRef]
- Granger, C.W.; Ramanathan, R. Improved methods of combining forecasts. J. Forecast. 1984, 3, 197–204. [Google Scholar] [CrossRef]
- Diks, C.G.; Vrugt, J.A. Comparison of point forecast accuracy of model averaging methods in hydrologic applications. Stoch. Environ. Res. Risk Assess. 2010, 24, 809–820. [Google Scholar] [CrossRef]
- Javadi, S.H.; Farina, A. Radar networks: A review of features and challenges. Inf. Fusion 2020, 61, 48–55. [Google Scholar] [CrossRef]
- Papoulis, A.; Pillai, S.U. Probability, Random Variables, and Stochastic Processes, 4th ed.; McGraw-Hill: New York, NY, USA, 2002. [Google Scholar]
- Zhang, Y.; Hartemink, A.E. Data fusion of vis–NIR and PXRF spectra to predict soil physical and chemical properties. Eur. J. Soil Sci. 2020, 71, 316–333. [Google Scholar] [CrossRef]
- Wan, M.; Hu, W.; Qu, M.; Li, W.; Zhang, C.; Kang, J.; Hong, Y.; Chen, Y.; Huang, B. Rapid estimation of soil cation exchange capacity through sensor data fusion of portable XRF spectrometry and Vis-NIR spectroscopy. Geoderma 2020, 363, 114163. [Google Scholar] [CrossRef]
- IUSS Working Group WRB. World reference base for soil resources 2014. In World Soil Resources Reports No. 106; Schad, P., van Huyssteen, C., Micheli, E., Eds.; FAO: Rome, Italy, 2014; 189p, ISBN 978-92-5-108369-7. [Google Scholar]
- EMBRAPA Solos. Brazilian Soil Classification System, 5th ed.; EMBRAPA: Brasília, Brazil, 2018. [Google Scholar]
- Van Raij, B.; Andrade, J.C.; Cantarela, H.; Quaggio, J.A. Análise Química Para Avaliação de Solos Tropicais; IAC: Campinas, Brazil, 2001; 285p. (In Portuguese) [Google Scholar]
- Christy, C.; Drummond, P. Mobile Soil Mapping System for Collecting Soil Reflectance Measurements. U.S. Patent 8204689B2, 19 June 2012. [Google Scholar]
- Mouazen, A.M.; Maleki, M.R.; Cockx, L.; Van Meirvenne, M.; Van Holm, L.H.J.; Merckx, R.; De Baerdemaeker, J.; Ramon, H. Optimum three-point linkage set up for improving the quality of soil spectra and the accuracy of soil phosphorus measured using an on-line visible and near infrared sensor. Soil Tillage Res. 2009, 103, 144–152. [Google Scholar] [CrossRef]
- Barnes, R.J.; Dhanoa, M.S.; Lister, S.J. Standard normal variate transformation and de-trending of near-infrared diffuse reflectance spectra. Appl. Spectrosc. 1989, 43, 772–777. [Google Scholar] [CrossRef]
- Rinnan, Å.; Van Den Berg, F.; Engelsen, S.B. Review of the most common pre-processing techniques for near-infrared spectra. TrAC Trends Anal. Chem. 2009, 28, 1201–1222. [Google Scholar] [CrossRef]
- Ben-Dor, E.; Inbar, Y.; Chen, Y. The reflectance spectra of organic matter in the visible near-infrared and short wave infrared region (400–2500 nm) during a controlled decomposition process. Remote Sens. Environ. 1997, 61, 1–15. [Google Scholar] [CrossRef]
- Nawar, S.; Mouazen, A.M. Predictive performance of mobile vis-near infrared spectroscopy for key soil properties at different geographical scales by using spiking and data mining techniques. Catena 2017, 151, 118–129. [Google Scholar] [CrossRef]
- Kennard, R.W.; Stone, L.A. Computer aided design of experiments. Technometrics 1969, 11, 137–148. [Google Scholar] [CrossRef]
- Chang, C.W.; Laird, D.A.; Mausbach, M.J.; Hurburgh, C.R. Near-infrared reflectance spectroscopy–principal components regression analyses of soil properties. Soil Sci. Soc. Am. J. 2001, 65, 480–490. [Google Scholar] [CrossRef]
- Cardelli, V.; Weindorf, D.C.; Chakraborty, S.; Li, B.; De Feudis, M.; Cocco, S.; Agnelli, A.; Choudhury, A.; Ray, D.P.; Corti, G. Non-saturated soil organic horizon characterization via advanced proximal sensors. Geoderma 2017, 288, 130–142. [Google Scholar] [CrossRef]
- Vapnik, V.N. The Nature of Statistical Learning Theory; Springer: New York, NY, USA, 1995. [Google Scholar]
- Scikit-Learn Machine Learning in Python. Available online: https://scikit-learn.org/ (accessed on 1 June 2020).
- Nawar, S.; Mouazen, A.M. Optimal sample selection for measurement of soil organic carbon using on-line vis-NIR spectroscopy. Comput. Electron. Agric. 2018, 151, 469–477. [Google Scholar] [CrossRef]
- Mouazen, A.M.; De Baerdemaeker, J.; Ramon, H. Effect of wavelength range on the measurement accuracy of some selected soil constituents using visual-near infrared spectroscopy. J. Near Infrared Spectrosc. 2006, 14, 189–199. [Google Scholar] [CrossRef]
- Demattê, J.A.M. Characterization and discrimination of soils by their reflected electromagnetic energy. Pesqui. Agropecuária Bras. 2002, 37, 1445–1458. [Google Scholar] [CrossRef]
- Ben-Dor, E. Quantitative remote sensing of soil properties. Adv. Agron. 2002, 75, 173–244. [Google Scholar] [CrossRef]
- Lacerda, M.P.; Demattê, J.A.M.; Sato, M.V.; Fongaro, C.T.; Gallo, B.C.; Souza, A.B. Tropical texture determination by proximal sensing using a regional spectral library and its relationship with soil classification. Remote Sens. 2016, 8, 701. [Google Scholar] [CrossRef]
- Demattê, J.A.M.; Dotto, A.C.; Bedin, L.G.; Sayão, V.M.; Souza, A.B. Soil analytical quality control by traditional and spectroscopy techniques: Constructing the future of a hybrid laboratory for low environmental impact. Geoderma 2019, 337, 111–121. [Google Scholar] [CrossRef]
- Cezar, E.; Nanni, M.R.; Guerrero, C.; da Silva Junior, C.A.; Cruciol, L.G.T.; Chicati, M.L.; Silva, G.F.C. Organic matter and sand estimates by spectroradiometry: Strategies for the development of models with applicability at a local scale. Geoderma 2019, 340, 224–233. [Google Scholar] [CrossRef]
- Van Raij, B. Fertilidade do Solo e Manejo de Nutrientes; International Plant Nutrition Institute (IPNI): Piracicaba, Brazil, 2011; p. 420. (In Portuguese) [Google Scholar]
- Silva, S.; Poggere, G.; Menezes, M.; Carvalho, G.; Guilherme, L.; Curi, N. Proximal sensing and digital terrain models applied to digital soil mapping and modeling of Brazilian Latosols (Oxisols). Remote Sens. 2016, 8, 614. [Google Scholar] [CrossRef]
- Coutinho, M.A.; Alari, F.D.O.; Ferreira, M.M.; Amaral, L.R. Influence of soil sample preparation on the quantification of NPK content via spectroscopy. Geoderma 2019, 338, 401–409. [Google Scholar] [CrossRef]
- Silva, E.A.; Weindorf, D.C.; Silva, S.H.; Ribeiro, B.T.; Poggere, G.C.; Carvalho, T.S.; Goncalves, M.G.; Guilherme, L.R.; Curi, N. Advances in Tropical Soil Characterization via Portable X-Ray Fluorescence Spectrometry. Pedosphere 2019, 29, 468–482. [Google Scholar] [CrossRef]
- Gruber, A.; Dorigo, W.A.; Crow, W.; Wagner, W. Triple collocation-based merging of satellite soil moisture retrievals. IEEE Trans. Geosci. Remote Sens. 2017, 55, 6780–6792. [Google Scholar] [CrossRef]
- Element, C.A.S. Method 3051A microwave assisted acid digestion of sediments, sludges, soils, and oils. Z. Anal. Chem. 2007, 111, 362–366. [Google Scholar]

| Single Sensor | Multiple Sensor | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| SF-PLS | SF-SVM | GR2 | GR3 | LS2 | LS3 | |||||||||
| RMSE | Techni. 5 | RMSE | % RI 6 | RMSE | % RI | RMSE | % RI | RMSE | % RI | RMSE | % RI | RMSE | % RI | |
| Clay | 27.32 | vis-NIR | 25.58 | 6 | 24.63 | 10 | 23.74 | 13 | 22.93 * | 16 | 24.01 | 12 | 23.11 | 15 |
| OM 1 | 2.10 * | vis-NIR | 2.28 | −8 | 2.34 | −11 | 2.89 | −37 | 2.48 | −18 | 2.92 | −39 | 2.47 | −17 |
| CEC 2 | 10.19 | XRF | 11.05 | −8 | 13.28 | −30 | 10.74 | −5 | 9.99 * | 2 | 10.9 | −7 | 9.99 * | 2 |
| pH | 0.33 | XRF | 0.31 | 7 | 0.26 | 21 | 0.28 * | 16 | 0.28 * | 16 | 0.28 * | 16 | 0.28 * | 16 |
| V 3 | 5.6 | XRF | 6.63 | −18 | 6.61 | −18 | 5.04 * | 10 | 5.7 | −2 | 5.11 | 9 | 5.77 | −3 |
| ex-P 4 | 12.05 | vis-NIR | 13.43 | −11 | 9.89 | 18 | 12.42 | −3 | 12.45 | −3 | 11.70 * | 3 | 11.97 | 1 |
| ex-K 4 | 0.53 | XRF | 0.61 | −15 | 0.71 | −33 | 0.51 | 4 | 0.52 | 2 | 0.50 * | 6 | 0.52 | 2 |
| ex-Ca 4 | 4.09 | XRF | 3.98 * | 3 | 7.26 | −77 | 4.45 | −9 | 4.2 | −3 | 4.46 | −9 | 4.18 | −2 |
| ex-Mg 4 | 4.28 | XRF | 4.07 | 5 | 5.89 | −38 | 4.42 | −3 | 3.94 | 8 | 4.43 | −3 | 3.92 * | 9 |
| GR2 | GR3 | LS2 | LS3 | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| vis-NIR | XRF | vis-NIR | XRF | SF-PLS | vis-NIR | XRF | vis-NIR | XRF | SF-PLS | |
| Clay | 0.77 | 0.24 | 0.54 | 0.07 | 0.38 | 0.74 | 0.26 | 0.41 | 0.07 | 0.52 |
| OM 1 | 0.55 | 0.46 | 0.33 | 0.01 | 0.67 | 0.76 | 0.24 | 0.51 | -0.08 | 0.57 |
| CEC 2 | 0.18 | 0.82 | 0.07 | 0.37 | 0.59 | 0.19 | 0.81 | 0.19 | 0.45 | 0.35 |
| pH | 0.61 | 0.35 | 0.48 | 0.12 | 0.37 | 0.61 | 0.39 | 0.42 | 0.14 | 0.43 |
| V 3 | 0.37 | 0.63 | 0.30 | 0.27 | 0.44 | 0.35 | 0.65 | 0.27 | 0.07 | 0.65 |
| ex-P 4 | 0.46 | 0.62 | 0.42 | 0.48 | 0.15 | 0.55 | 0.45 | 0.50 | 0.32 | 0.18 |
| ex-K 4 | 0.13 | 0.85 | 0.10 | 0.56 | 0.34 | 0.15 | 0.85 | 0.10 | 0.52 | 0.38 |
| ex-Ca 4 | 0.10 | 0.91 | 0.08 | −0.06 | 0.98 | 0.08 | 0.92 | 0.07 | -0.06 | 1.00 |
| ex-Mg 4 | 0.26 | 0.73 | 0.18 | 0.05 | 0.78 | 0.20 | 0.80 | 0.09 | 0.05 | 0.87 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tavares, T.R.; Molin, J.P.; Javadi, S.H.; Carvalho, H.W.P.d.; Mouazen, A.M. Combined Use of Vis-NIR and XRF Sensors for Tropical Soil Fertility Analysis: Assessing Different Data Fusion Approaches. Sensors 2021, 21, 148. https://doi.org/10.3390/s21010148
Tavares TR, Molin JP, Javadi SH, Carvalho HWPd, Mouazen AM. Combined Use of Vis-NIR and XRF Sensors for Tropical Soil Fertility Analysis: Assessing Different Data Fusion Approaches. Sensors. 2021; 21(1):148. https://doi.org/10.3390/s21010148
Chicago/Turabian StyleTavares, Tiago Rodrigues, José Paulo Molin, S. Hamed Javadi, Hudson Wallace Pereira de Carvalho, and Abdul Mounem Mouazen. 2021. "Combined Use of Vis-NIR and XRF Sensors for Tropical Soil Fertility Analysis: Assessing Different Data Fusion Approaches" Sensors 21, no. 1: 148. https://doi.org/10.3390/s21010148
APA StyleTavares, T. R., Molin, J. P., Javadi, S. H., Carvalho, H. W. P. d., & Mouazen, A. M. (2021). Combined Use of Vis-NIR and XRF Sensors for Tropical Soil Fertility Analysis: Assessing Different Data Fusion Approaches. Sensors, 21(1), 148. https://doi.org/10.3390/s21010148








