Ratiometric Strategy for Electrochemical Sensing of Carbaryl Residue in Water and Vegetable Samples
Abstract
1. Introduction
2. Materials and Methods
2.1. Apparatus
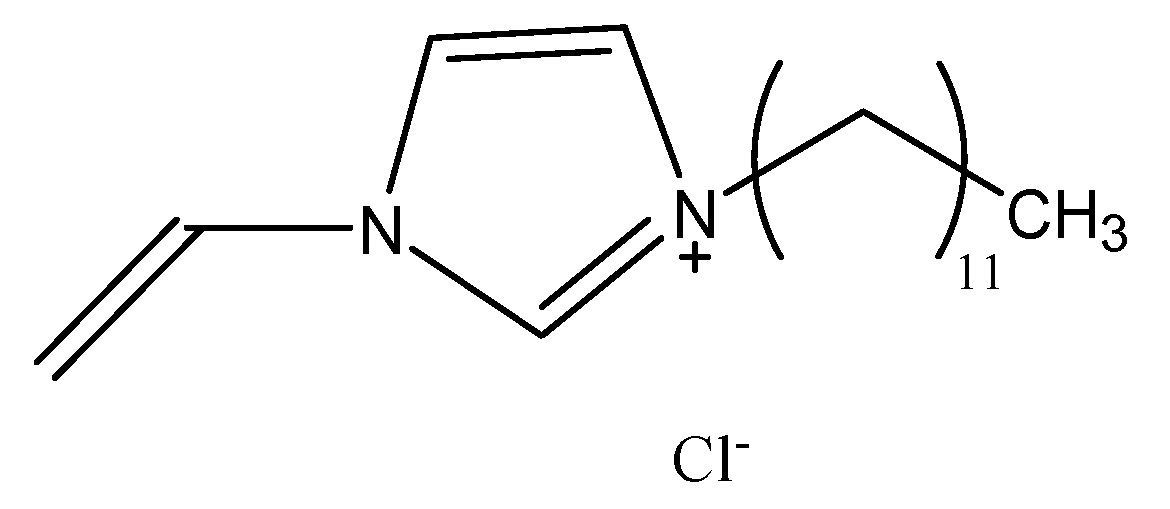
2.2. Reagents and Chemicals
2.3. The Proposed Ratiometric Strategy for Carbaryl (CBL) Determination
2.4. Preparation of Working Electrode
2.5. Electrochemical Analysis
2.6. Sample Preparation
2.7. High-Performance Liquid Chromatography (HPLC) Measurements
3. Results
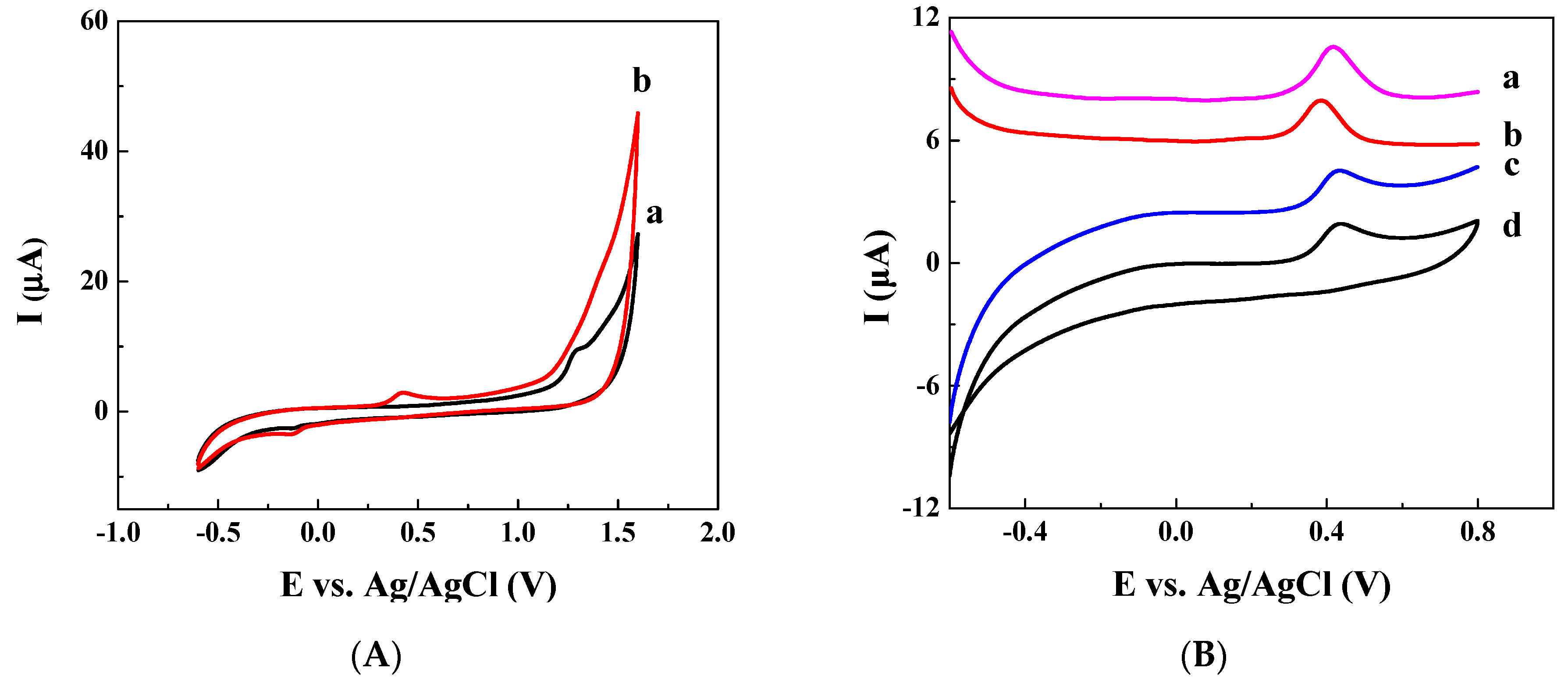
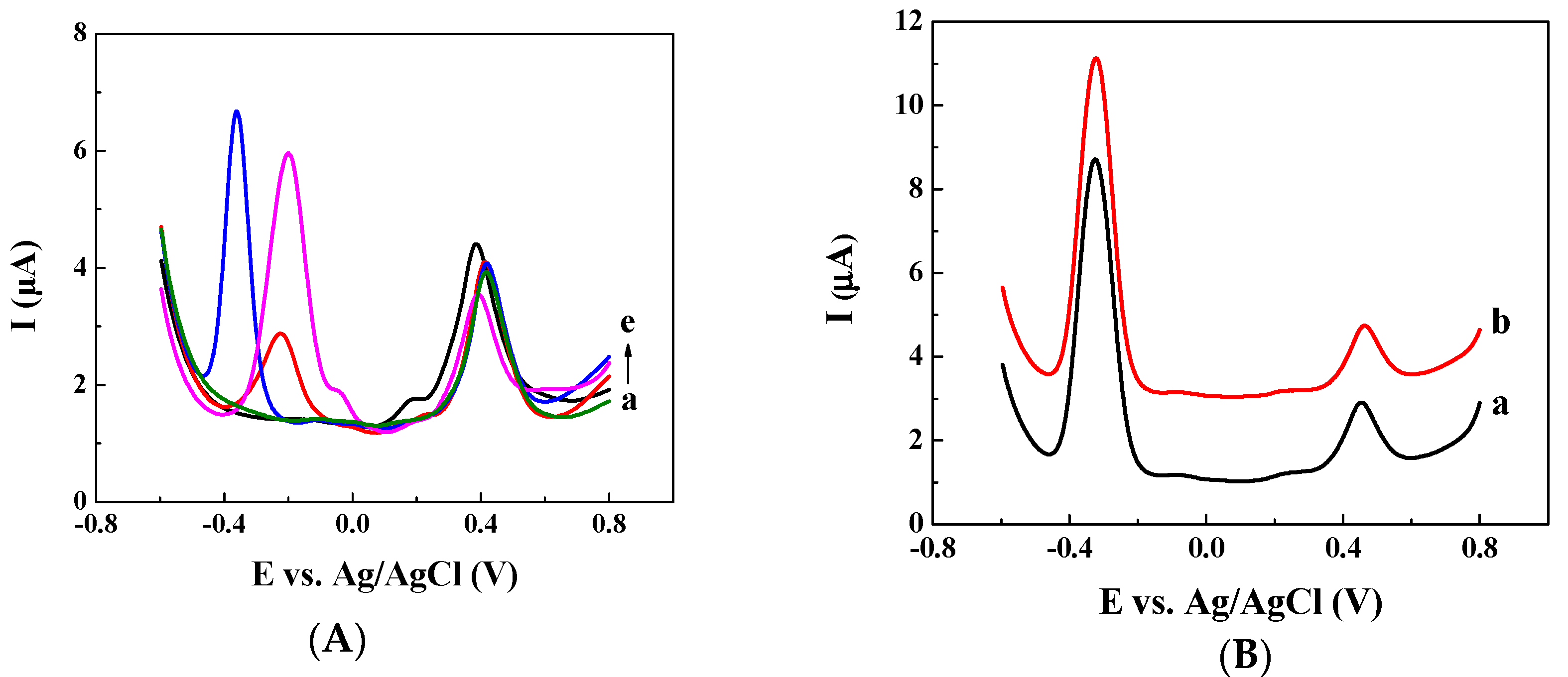
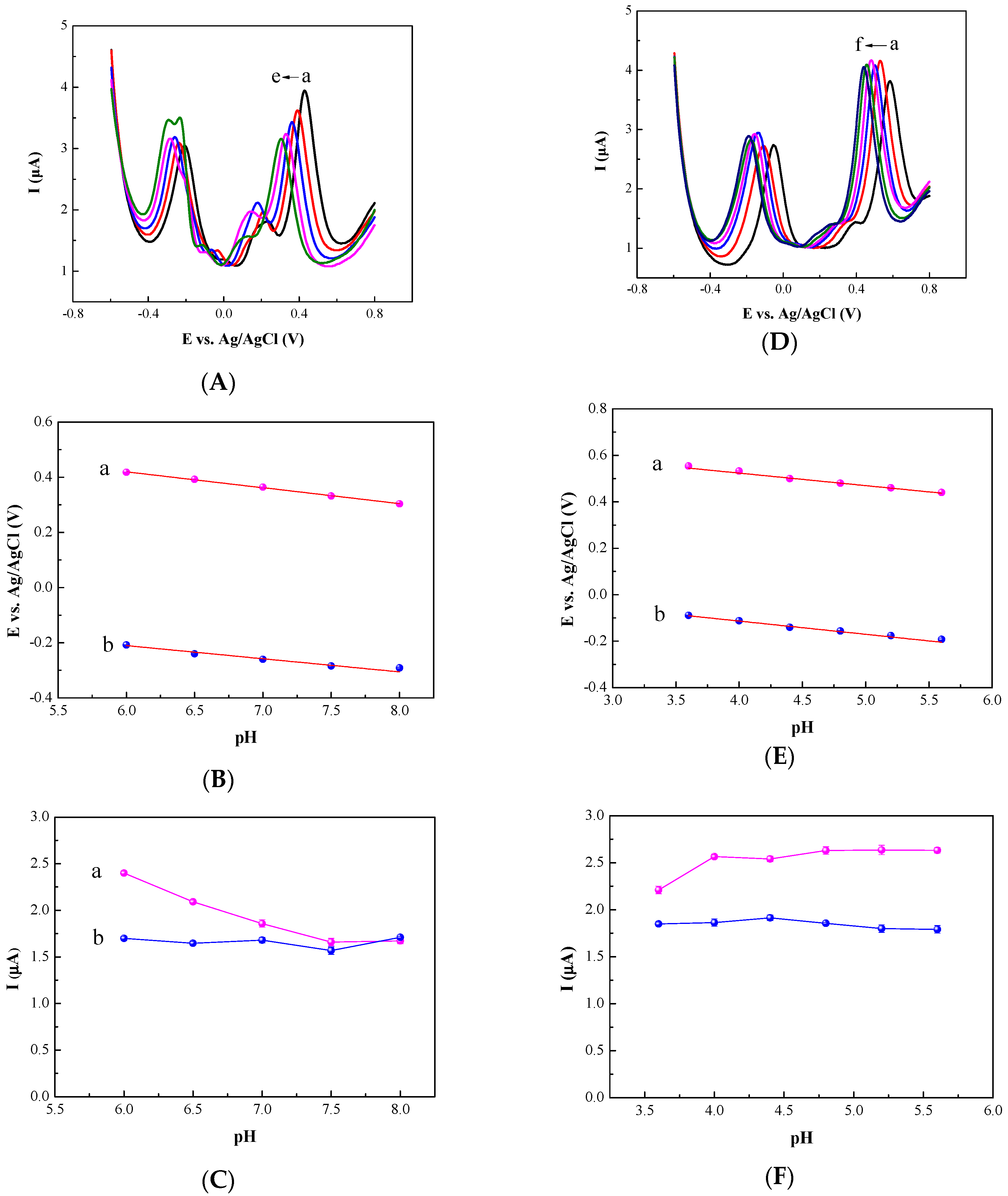
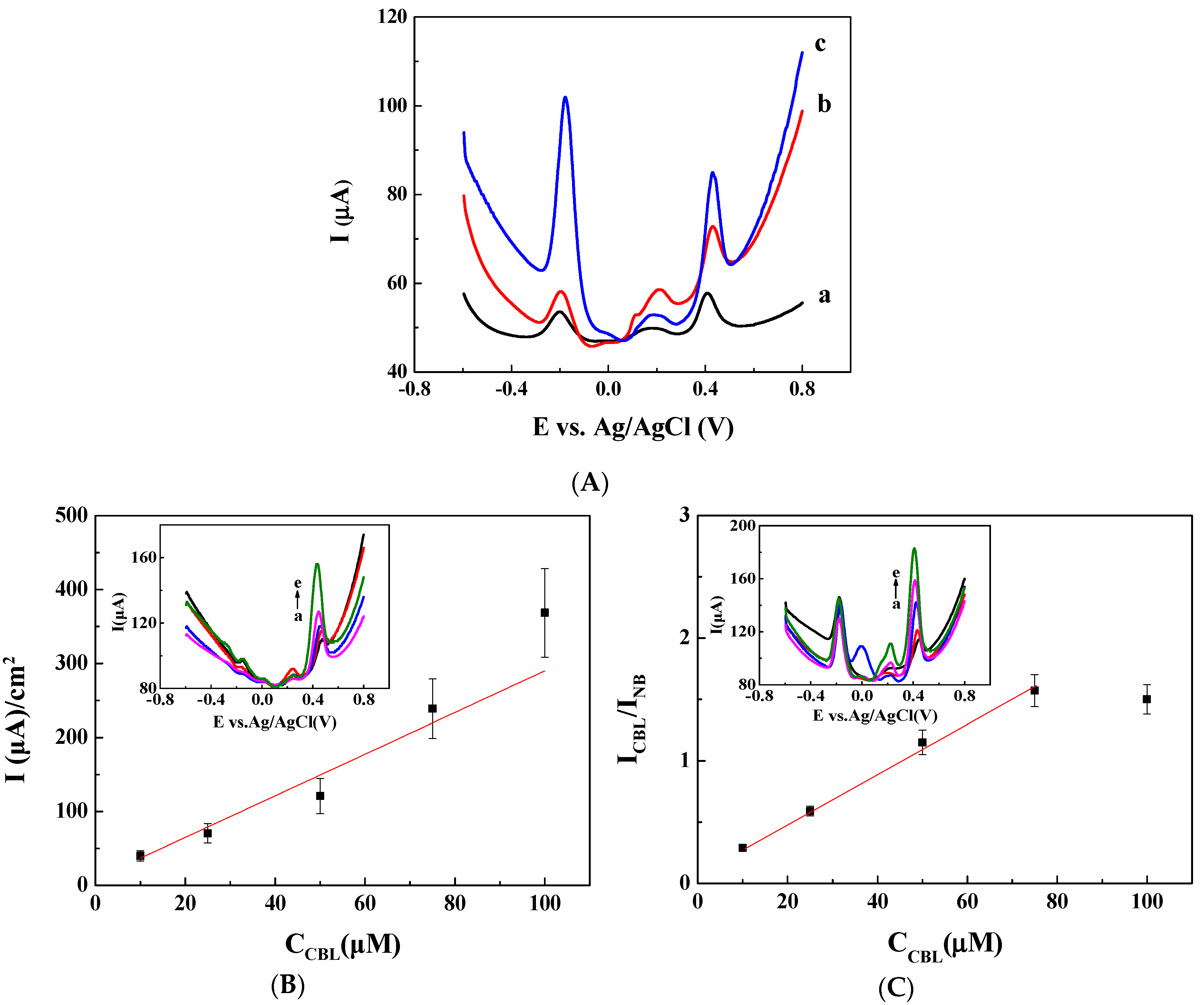
3.1. Ratiometric Strategy on Glassy Carbon Electrode (GCE)


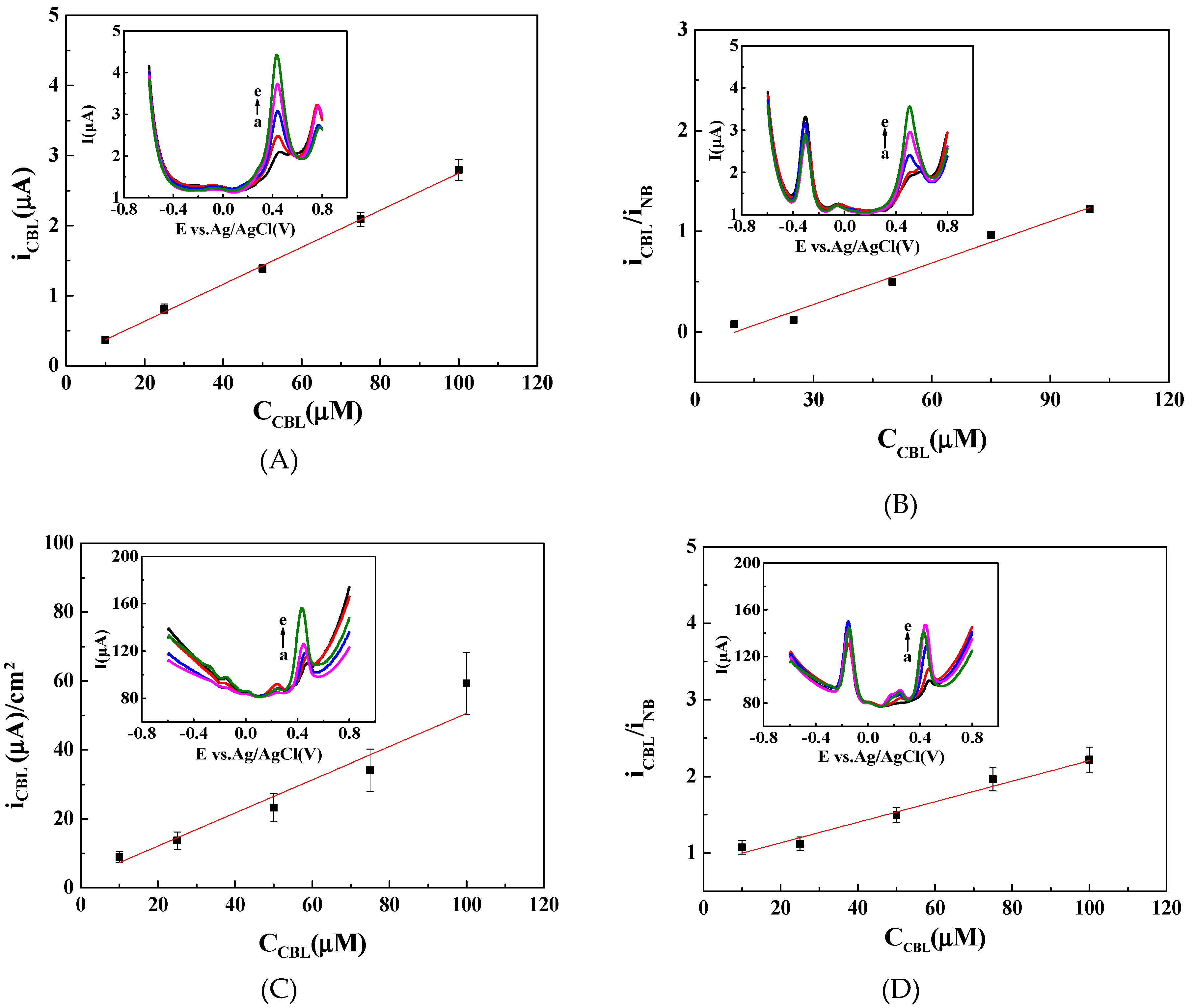
3.2. Ratiometric Strategy on Carbon Cloth (CC) Electrode
3.3. Ratiometric Strategy in Vegetable Samples
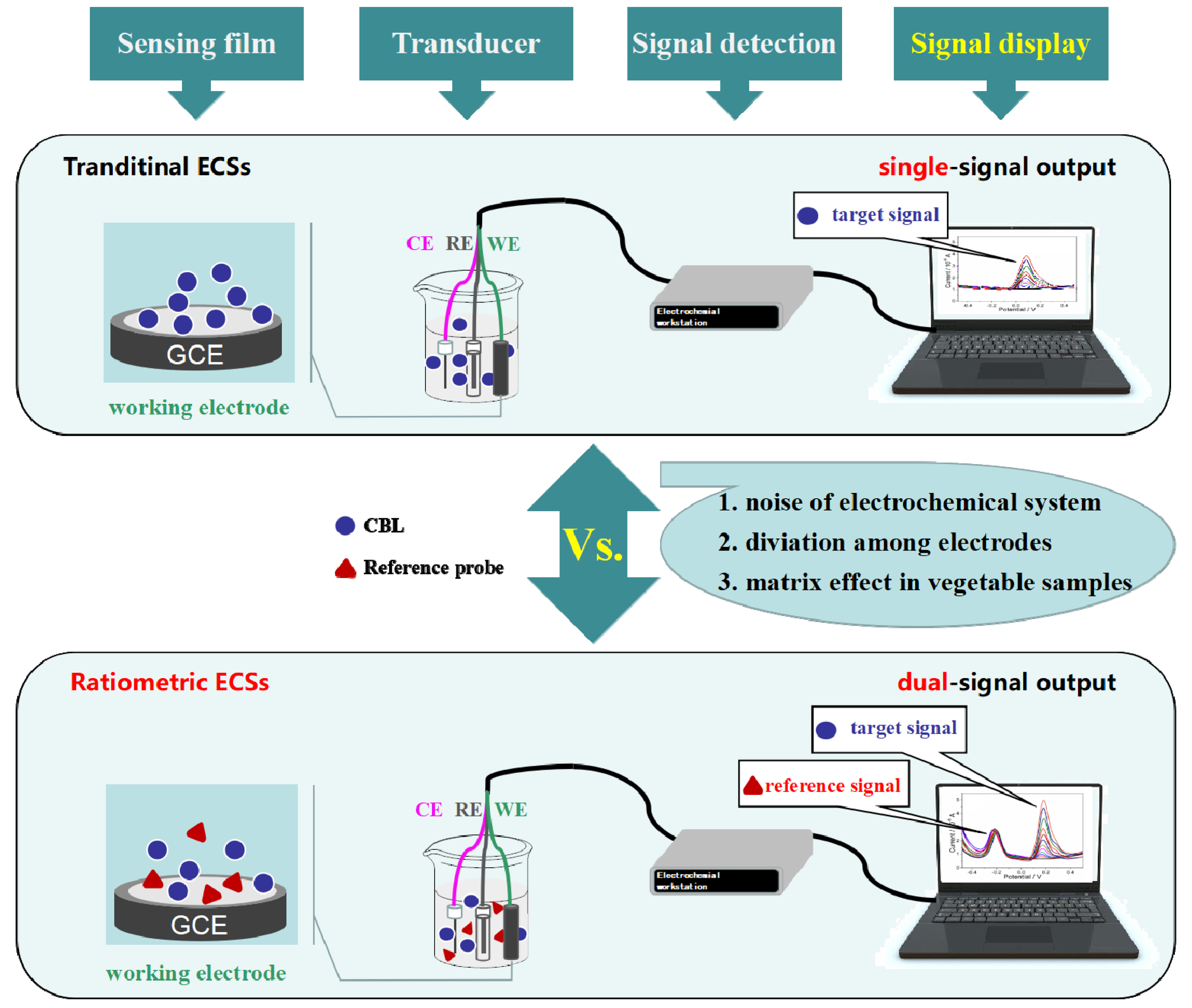
3.4. Comparison between Ratiometric Electrochemical Sensor (RECS) and Electrochemical Sensor (ECS)
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Zhang, C.; Ma, G.P.; Fang, G.Z.; Zhang, Y.; Wang, S. Development of a capillary electrophoresis-based immunoassay with laser-induced fluorescence for the detection of carbaryl in rice samples. J. Agric. Food Chem. 2008, 56, 8832–8837. [Google Scholar] [CrossRef] [PubMed]
- Dong, T.T.; Sun, J.W.; Liu, B.; Zhang, Y.; Song, Y.; Wang, S. Development of a sensitivity-improved immunoassay for the determination of carbaryl in food samples. J. Sci. Food Agric. 2010, 90, 1106–1112. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Cui, H.Y.; Cai, J.R.; Duan, Y.Q.; Liu, Y. Development of fluorescence sensing material based on CdSe/ZnS quantum dots and molecularly imprinted polymer for the detection of carbaryl in rice and Chinese cabbage. J. Agric. Food Chem. 2015, 63, 4966–4972. [Google Scholar] [CrossRef] [PubMed]
- Alsammarraie, F.K.; Lin, M.S. Using standing gold nanorod arrays as surface-enhanced raman spectroscopy (SERS) substrates for detection of carbaryl residues in fruit juice and milk. J. Agric. Food Chem. 2017, 65, 666–674. [Google Scholar] [CrossRef]
- Chen, W.; Liu, Y.Y.; Zhang, Y.; Fang, J.H.; Xu, P.C.; Xu, J.Q.; Li, X.X.; Liu, C.C.; Wen, W.J. Highly effective and specific way for trace analysis of carbaryl insecticides based on Au42Rh58 alloy nanocrystals. J. Mater. Chem. A. 2017, 5, 7064–7071. [Google Scholar] [CrossRef]
- Sun, B.; Wang, C.P.; Wang, Q.; Chen, L.; Dang, X.P.; Huang, J.L.; Chen, H.X. Preparation of acryloyl beta-cyclodextrin organic polymer monolithic column and its application in solid-phase microextraction and HPLC analysis for carbofuran and carbaryl in rice. Food Anal. Method 2017, 10, 3847–3855. [Google Scholar] [CrossRef]
- Sahoo, D.; Mandal, A.; Mitra, T.; Chakraborty, K.; Bardhan, M.; Dagupta, A.K. Nanosensing of pesticides by zinc oxide quantum dot: An optical and electrochemical approach for the detection of pesticides in water. J. Agric. Food Chem. 2018, 66, 414–423. [Google Scholar] [CrossRef]
- Abdalhai, M.H.; Fernandes, A.M.; Xia, X.; Musa, A.; Ji, J.; Sun, X. Electrochemical genosensor to detect pathogenic bacteria (Escherichia coli O157:H7) as applied in real food samples (fresh beef) to improve food safety and quality control. J. Agric. Food Chem. 2015, 63, 5017–5025. [Google Scholar] [CrossRef]
- Jiang, X.Q.; Yu, Y.; Chen, J.W.; Zhao, M.K.; Chen, H.; Song, X.Z.; Matzuk, A.J.; Carroll, S.L.; Tan, X.; Sizovs, A.; et al. Quantitative imaging of glutathione in live cells using a reversible reaction-based ratiometric fluorescent probe. ACS Chem. Bio. 2015, 10, 864–874. [Google Scholar] [CrossRef]
- Komatsu, K.; Urano, Y.; Kojima, H.; Nagano, T. Development of an iminocoumarin-based zinc sensor suitable or ratiometric fluorescence imaging of neuronal zinc. JACS 2007, 129, 3447–13454. [Google Scholar] [CrossRef]
- Xiong, E.; Li, Z.Z.; Zhang, X.H.; Zhou, J.W.; Yan, X.X.; Liu, Y.Q.; Chen, J.H. A triple-helix molecular switch electrochemical ratiometric biosensor for ultrasensitive detection of nucleic acids. Anal. Chem. 2017, 89, 8830–8835. [Google Scholar] [CrossRef] [PubMed]
- Manibalan, K.; Mani, V.; Chang, P.C.; Huang, C.H.; Huang, S.T.; Marchlewicz, K.; Neethirajan, S. Electrochemical latent redox ratiometric probes for real-time tracking and quantification of endogenous hydrogen sulfide production in living cells. Biosens. Bioelectron. 2017, 96, 233–238. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Kan, X. A ratiometric strategy-based electrochemical sensing interface for the sensitive and reliable detection of imidacloprid. Analyst 2018, 143, 2150–2156. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Hu, Y.; Li, Y. Molecularly imprinted polymer-decorated signal on-off ratiometric electrochemical sensor for selective and robust dopamine detection. Biosens. Bioelectron. 2019, 135, 224–230. [Google Scholar] [CrossRef] [PubMed]
- Jin, H.; Zhao, C.; Gui, R.; Gao, X.; Wang, Z. Reduced graphene oxide/nile blue/gold nanoparticles complex modified glassy carbon electrode used as a sensitive and label-free aptasensor for ratiometric electrochemical sensing of dopamine. Anal. Chim. Acta 2018, 1025, 154–162. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Gui, R.; Xu, K.Q.; Guo, H.; Jin, H.; Wang, Z. A bimetallic nanoparticle/graphene oxide/thionine composite-modified glassy carbon electrode used as a facile ratiometric electrochemical sensor for sensitive uric acid determination. New J. Chem. 2018, 42, 14796–14804. [Google Scholar] [CrossRef]
- Xu, M.; Wang, L.; Xie, Y.; Song, Y.; Wang, L. Ratiometric electrochemical sensing and biosensing based on multiple redox-active state COFDHTA-TTA. Sens. Actuators B 2019, 281, 1009–1015. [Google Scholar] [CrossRef]
- Gong, C.; Shen, Y.; Song, Y.; Wang, L. On-off ratiometric electrochemical biosensor for accurate detection of glucose. Electrochim. Acta 2017, 235, 488–494. [Google Scholar] [CrossRef]
- Wang, L.; Gong, C.; Shen, Y.; Ye, W.; Xu, M.; Song, Y. A novel ratiometric electrochemical biosensor for sensitive detection of ascorbic acid. Sens. Actuators B 2017, 242, 625–631. [Google Scholar] [CrossRef]
- Wang, L.; Ma, R.; Jiang, L.; Jia, L.; Jia, W.; Wang, H. A novel “signal-on/off” sensing platform for selective detection of thrombin based on target-induced ratiometric electrochemical biosensing and biobar-corded nanoprobe amplification strategy. Biosens. Bioelectron. 2017, 92, 390–395. [Google Scholar] [CrossRef]
- Ma, R.N.; Wang, L.L.; Wang, H.F.; Jia, L.P.; Zhang, W.; Shang, L.; Xue, Q.W.; Jia, W.L.; Liu, Q.Y.; Wang, H.S. Highly sensitive ratiometric electrochemical DNA biosensor based on homogeneous exonuclease Ⅲ-assisted target recycling amplification and one-step triggered dual-signal output. Sens. Actuators B 2018, 269, 173–179. [Google Scholar] [CrossRef]
- Yang, T.; Yu, R.; Liu, S.; Qiu, Z.; Luo, S.; Li, W.; Jiao, K. A ratiometric electrochemical deoxyribonucleic acid sensing strategy based on self-signal of highly stable reduced graphene oxide-flavin mononucleotide aqeous dispersion modified nanointerface. Sens. Actuators B 2018, 267, 519–524. [Google Scholar] [CrossRef]
- Dong, P.; Zhu, L.; Huang, J.; Ren, J.; Lei, J. Electrocatalysis of cerium metal-organic frameworks for ratiometric electrochemical detection of telomerase activity. Biosens. Bioelectron. 2019, 138, 111313–111320. [Google Scholar] [CrossRef]
- Yao, D.; Zhao, W.; Zhang, L.; Tian, Y. A ratiometric electrochemical strategy for sensitive determinaiton of furin activity based on dual signal amplification and antifouling nanosurfaces. Analyst 2017, 142, 4215–4220. [Google Scholar] [CrossRef] [PubMed]
- Xiong, E.; Wu, L.; Zhou, J.; Yu, P.; Zhang, X.; Chen, J. A ratiometric electrochemical biosensor for sensitive detection of Hg2+ based on thymine-Hg2+-thymine structure. Anal. Chim. Acta 2015, 853, 242–248. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Chang, Y.; Ma, J.; Wu, Z.; Yuan, R.; Chai, Y. Programming a target-initiated biofunctional DNAzyme nanodevice for sensitive ratiometric electrochemical biosensing. Anal. Chem. 2019, 91, 6127–6133. [Google Scholar] [CrossRef]
- Lin, Y.; Jia, J.; Yang, R.; Chen, D.; Wang, J.; Luo, F.; Guo, L.; Qiu, B.; Lin, Z. Ratiometric immunosensor for GP73 detection based on the ratios of electrochemiluminescence and electrochemical signal using DNA tetrahedral nanostructure as the carrier of stable reference signal. Anal. Chem. 2019, 91, 3717–3724. [Google Scholar] [CrossRef]
- Ge, L.; Wang, W.; Li, F. Electro-grafted electrode with graphene-oxide-like DNA affinity for ratiometric homogenous electrochemical biosensing of microRNA. Anal. Chem. 2017, 89, 11560–11567. [Google Scholar] [CrossRef]
- Li, S.; Tian, Y. An electrochemical biosensor with dual signal outputs for ratiometric monitoring the levels of H2O2 and pH in the microdialysates from a rat brain. Electroanalysis 2018, 30, 1047–1053. [Google Scholar] [CrossRef]
- Yu, Y.; Wang, P.; Zhu, X.; Peng, Q.; Zhou, Y.; Yin, T.; Liang, Y.; Yin, X. Combined determination of copper ions and β-amploid peptide by a single ratiometric electrochemical biosensor. Analyst 2018, 143, 323–331. [Google Scholar] [CrossRef]
- Yu, J.B.; Jin, H.; Gui, R.J.; Wang, Z.H.; Ge, F. A general strategy to facilely design ratiometric electrochemical sensors in electrolyte solution by directly using a bare electrode for dual-signal sensing of analytes. Talanta 2017, 162, 435–439. [Google Scholar] [CrossRef]
- Zhao, C.Q.; Jin, H.; Gui, R.J.; Wang, Z.H. Facile fabrication of dual-ratiometric electrochemical sensors based on a bare electrode for dual-signal sensing of analytes in electrolyte solution. Sens. Actuators B 2017, 242, 71–78. [Google Scholar] [CrossRef]
- Meng, S.J.; Hong, Y.; Dai, Z.Y.; Huang, W.; Dong, X.C. Simultaneous detection of dihydroxybenzene isomers with ZnO nanorod/carbon cloth electrodes. ACS Appl. Mater. Inter. 2017, 9, 12453–12460. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.D.; Zheng, Y.Y.; Yuan, J.H.; Shen, J.F.; Hu, J.G.; Wang, A.J.; Wu, L.J.; Niu, L. Three-dimensional NiCo layered double hydroxide nanosheets array on carbon cloth, facile preparation and its application in highly sensitive enzymeless glucose detection. Electrochim. Acta. 2017, 224, 628–635. [Google Scholar] [CrossRef]
- Luan, F.; Zhang, S.; Chen, D.D.; Zheng, K.; Zhuang, X.M. CoS2-decorated ionic iquid-functionalized graphene as a novel hydrazine electrochemical sensor. Talanta 2018, 182, 529–535. [Google Scholar] [CrossRef]
- Chaiyo, S.; Mehmeti, E.; Siangproh, W.; Hoang, T.L.; Nguyen, H.P.; Chailapakul, O.; Kalcher, K. Non-enzymatic electrochemical detection of glucose with a disposable paper-based sensor using a cobalt phthalocyanine-ionic liquid-graphene composite. Biosens. Bioelectron. 2018, 102, 113–120. [Google Scholar] [CrossRef]
- Du, C.B.; Hu, X.L.; Guan, P.; Guo, L.X.; Qian, L.W.; Song, R.Y.; Wang, C.L. Water-compatible surface-imprinted microspheres for high adsorption and selective recognition of peptide drug from aqueous media. J. Mater. Chem. B 2015, 3, 3044–3053. [Google Scholar] [CrossRef]
- Yang, T.; Yu, R.Z.; Yan, Y.H.; Zeng, H.; Luo, S.Z.; Liu, N.Z.; Morrin, A.; Luo, X.L.; Li, W.H. A review of ratiomentric electrochemical sensors: From design schemes to future prospects. Sens. Actuators B 2018, 274, 501–516. [Google Scholar] [CrossRef]
- Spring, S.A.; Goggins, S.; Frost, C.G. Ratiometric electrochemical detection of β-galactosidase. Org. Biomol. Chem. 2017, 15, 7122–7126. [Google Scholar] [CrossRef]
- Goggins, S.; Naz, C.; Marsh, B.J.; Frost, C.G. Ratiometric electrochemical detection of alkaline phosphatase. Chem. Commun. 2015, 51, 561–564. [Google Scholar] [CrossRef]
- Gao, X.; Gui, R.; Guo, H.; Wang, Z.; Liu, Q. Creatinine-induced specific signal responses and enzymeless ratiometric electrochemical detection based on copper nanoparticles electrodeposited on reduced graphene oxide-based hybrids. Sens. Actuators B 2019, 285, 201–208. [Google Scholar] [CrossRef]
- Della Pelle, F.; Del Carlo, M.; Sergi, M.; Compagnone, D.; Escarpa, A. Press-transferred carbon black nanoparticles on board of microfluidic chips for rapid and sensitive amperometric determination of pheyl carbamate pesticides in environmental samples. Microchim. Acta. 2016, 183, 3143–3149. [Google Scholar] [CrossRef]
- Cesarino, I.; Moraes, F.C.; Lanza, M.R.V.; Machado, S.A.S. Electrochemical detection of carbamate pesticides in fruit and vegetables with a biosensor based on acetylcholinesterase immobilised on a composite of polyaniline-carbon nanotubes. Food Chem. 2012, 135, 873–879. [Google Scholar] [CrossRef]
- Salih, F.E.; Achiou, B.; Ouammou, M.; Bennazha, J.; Ouarzane, A.; Younssi, S.A.; El Rhazi, M. Electrochemical sensor based on low silica X zeolite modified carbon paste for carbaryl determination. J. Adv. Res. 2017, 669–676. [Google Scholar] [CrossRef] [PubMed]
- Pop, A.; Manea, F.; Flueras, A.; Schoonman, J. Simultaneous voltammetric detection of carbaryl and paraquat pesticides on graphene-modified boron-doped diamond electrode. Sensors 2017, 17, 2033. [Google Scholar] [CrossRef]
- Wang, M.Y.; Huang, J.R.; Wang, M.; Zhang, D.E.; Chen, J. Electrochemical nonezymatic sensor based on CoO decorated reduced graphene oxide for the simultaneous determination of carbofuran and carbaryl in fruits and vegetables. Food Chem. 2014, 151, 191–197. [Google Scholar] [CrossRef]
- Liu, B.Z.; Xiao, B.; Cui, L.Q. Electrochemical analysis of carbaryl in fruit samples on graphene oxide-ionic liquid composite modified electrode. J. Food Compos. Anal. 2015, 40, 14–18. [Google Scholar] [CrossRef]

| Electrode | Linear Range | Limit of Detection (LOD, μM) | Ref. |
|---|---|---|---|
| CB-NPs electrode | 25~125 | 12 | [42] |
| AChE/PANI/MWCNTs/GCE | 9.9~49.6 | 1.4 | [43] |
| LSX zeolite/CPE | 1~100 | 0.3 | [44] |
| BDDGR | 1~6 | 0.07 | [45] |
| CoO/rGO/GCE | 0.2~70 | 0.04 | [46] |
| GO-IL/GCE | 0.10~12.0 | 0.02 | [47] |
| Bare GCE | 5~75 | 1.0 | This work |
| IL/CC | 10~75 | 1.4 | This work |
| Sample | Amount Added (μM) | Amount Recovered (μM) | Recovery Rate (%) | Content Determined by SWV (mg kg−1) ± SD | Content Determined by HPLC (mg kg−1) ± SD |
|---|---|---|---|---|---|
| Tomato | 10.00 | 8.11 | 81.10 | 1.63 ± 0.08 | 1.77 ± 0.05 |
| 25.00 | 21.22 | 84.88 | 4.27 ± 0.11 | 4.59 ± 0.09 | |
| 50.00 | 43.94 | 87.88 | 8.84 ± 0.29 | 9.11 ± 0.12 | |
| Cabbage | 10.00 | 8.72 | 87.20 | 1.75 ± 0.15 | 1.81 ± 0.04 |
| 25.00 | 23.19 | 92.76 | 4.66 ± 0.27 | 4.44 ± 0.15 | |
| 50.00 | 42.97 | 85.94 | 8.65 ± 0.14 | 8.99 ± 0.05 |
| Interference Factor | Electrodes | Matrix | RECS | ECS | ||||
|---|---|---|---|---|---|---|---|---|
| Linear Relationship | r | RSD (%) | Linear Relationship | r | RSD (%) | |||
| System noise | GCE | Water | iCBL/iMB = 0.007CCBL + 0.061 | 0.999 | 2.8 | iCBL = 0.029 CCBL + 0.780 | 0.947 | 7.2 |
| Electrodes deviation | IL/CC | Water | iCBL/iMB = 0.021CCBL + 0.112 | 0.999 | 6.8 | iCBL/cm2 = 3.627 CCBL − 21.161 | 0.980 | 21.0 |
| Matrix effect in vegetable samples | GCE | Tomato sample | iCBL/iMB = 0.014CCBL − 0.140 | 0.999 | 5.8 | iCBL = 0.027 CCBL + 0.105 | 0.990 | 5.9 |
| IL/CC | Tomato sample | iCBL/iMB = 0.014CCBL + 0.860 | 0.990 | 9.2 | iCBL/cm2 = 0.533 CCBL + 0.155 | 0.970 | 17.0 | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, M.; Zhang, Z.; Yang, Y.; Zhang, Y.; Wang, Y.; Chen, X. Ratiometric Strategy for Electrochemical Sensing of Carbaryl Residue in Water and Vegetable Samples. Sensors 2020, 20, 1524. https://doi.org/10.3390/s20051524
Zhang M, Zhang Z, Yang Y, Zhang Y, Wang Y, Chen X. Ratiometric Strategy for Electrochemical Sensing of Carbaryl Residue in Water and Vegetable Samples. Sensors. 2020; 20(5):1524. https://doi.org/10.3390/s20051524
Chicago/Turabian StyleZhang, Min, Zeyuan Zhang, Yanjing Yang, Yi Zhang, Yufei Wang, and Xinyi Chen. 2020. "Ratiometric Strategy for Electrochemical Sensing of Carbaryl Residue in Water and Vegetable Samples" Sensors 20, no. 5: 1524. https://doi.org/10.3390/s20051524
APA StyleZhang, M., Zhang, Z., Yang, Y., Zhang, Y., Wang, Y., & Chen, X. (2020). Ratiometric Strategy for Electrochemical Sensing of Carbaryl Residue in Water and Vegetable Samples. Sensors, 20(5), 1524. https://doi.org/10.3390/s20051524




