Prototype of Nitro Compound Vapor and Trace Detector Based on a Capacitive MIS Sensor
Abstract
1. Introduction
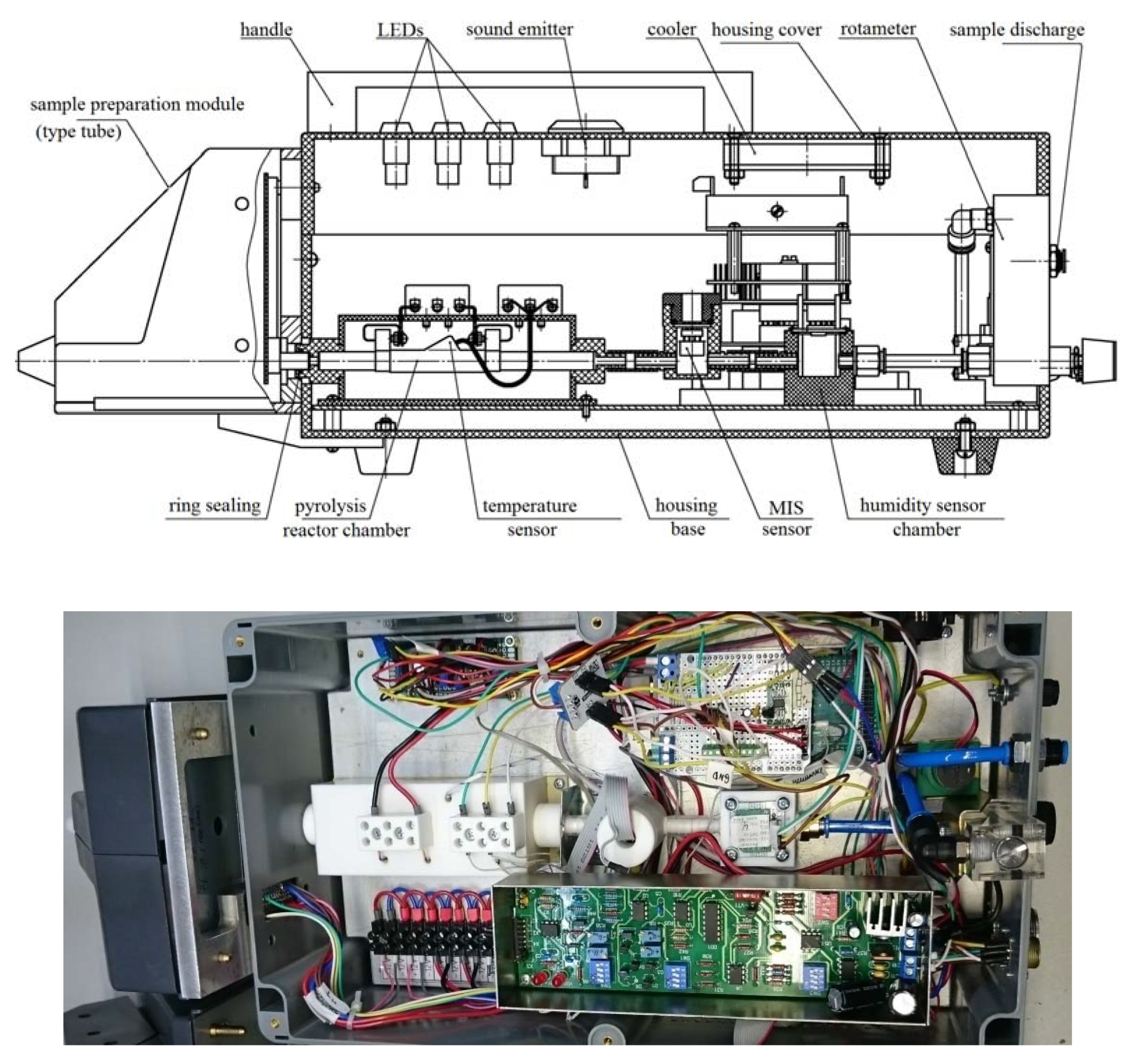
2. Design and Manufacture of Prototype Nitro Compounds Detector
- The analyzed volume of the gas path was reduced from 40 to 14 cm3;
- The length and power consumption of the pyrolysis chamber (reactor, Figure 1) were reduced from 25 to 13 cm, from 92 to 45 W, respectively;
- The design of the evaporator has been optimized, namely a constructive solution with replaceable sample preparation modules of two types has been used: for operation in the vapor registration mode (heated PTFE-tube) and in the registration mode of traces (toaster-type design, Figure 2).
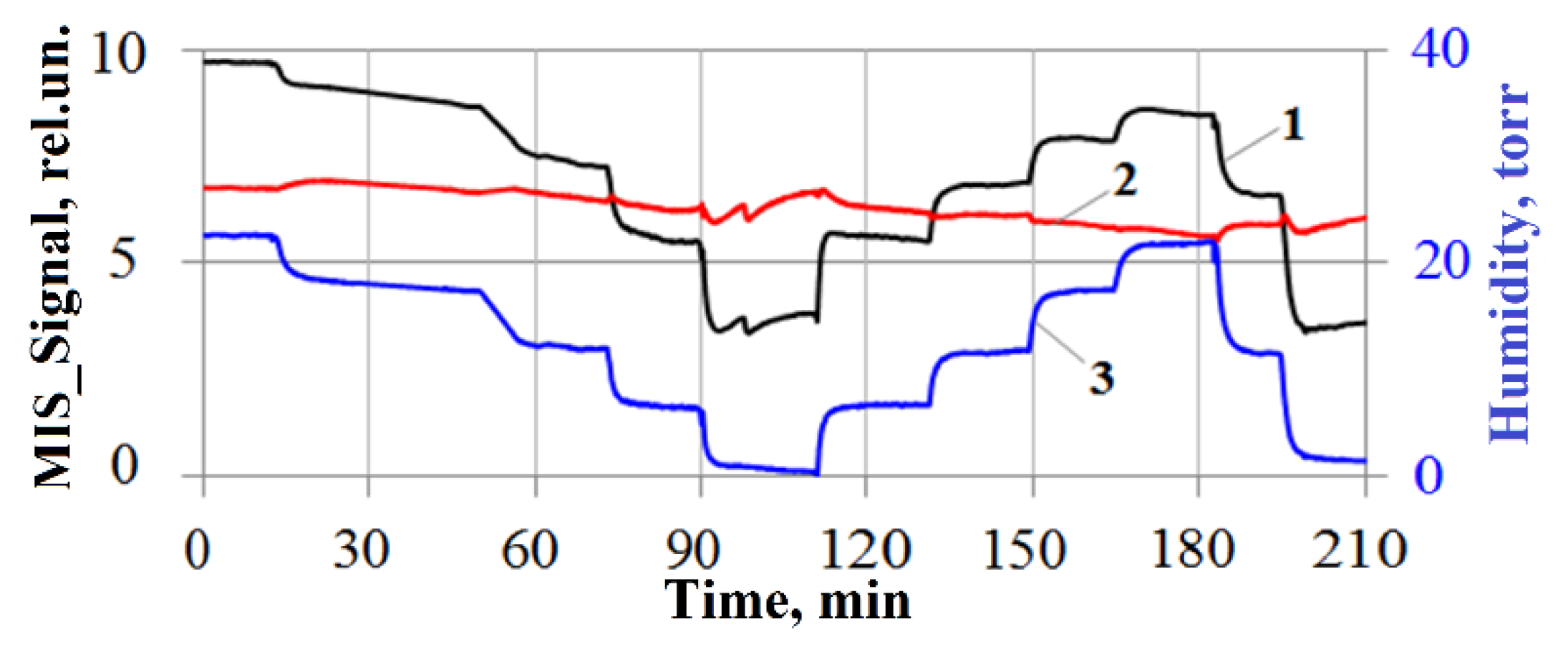
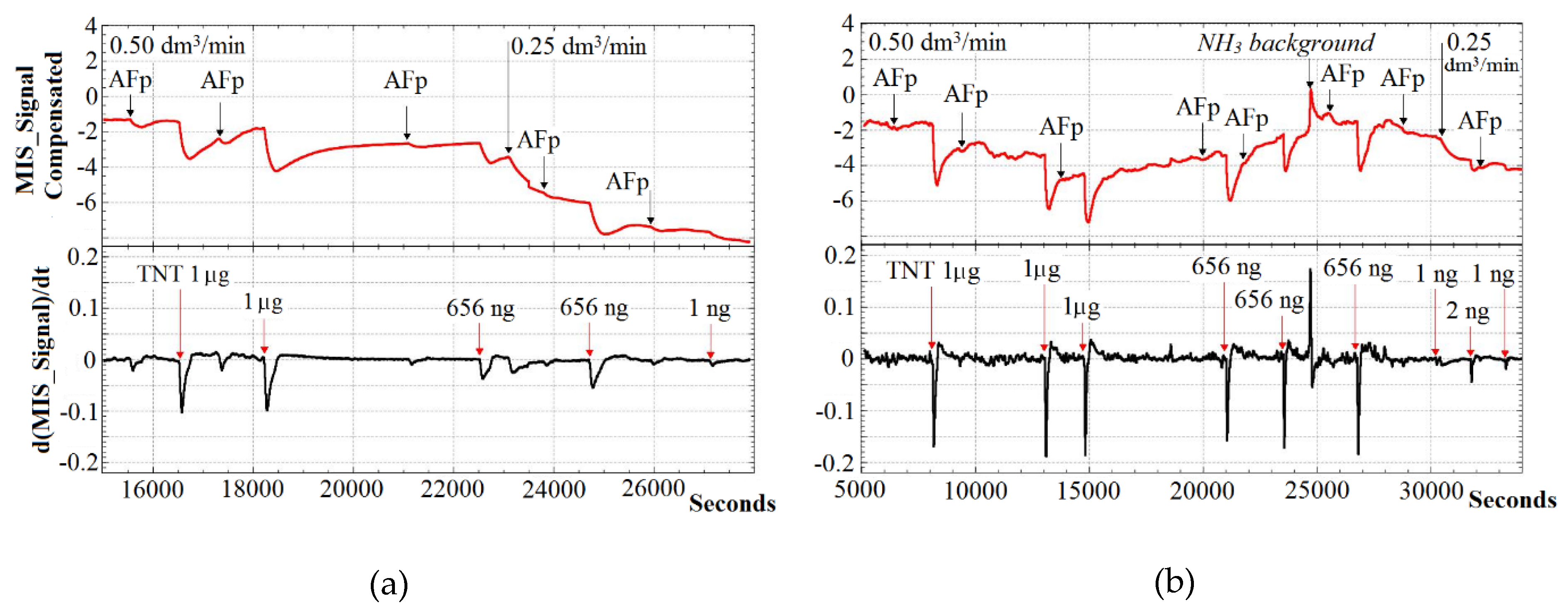
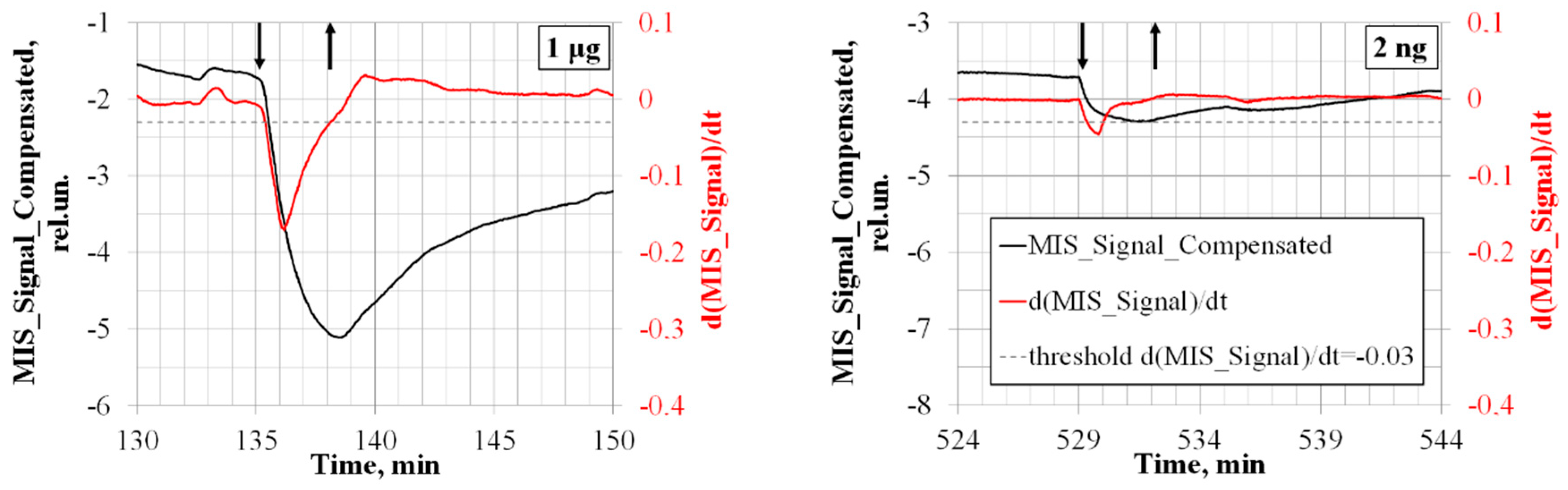
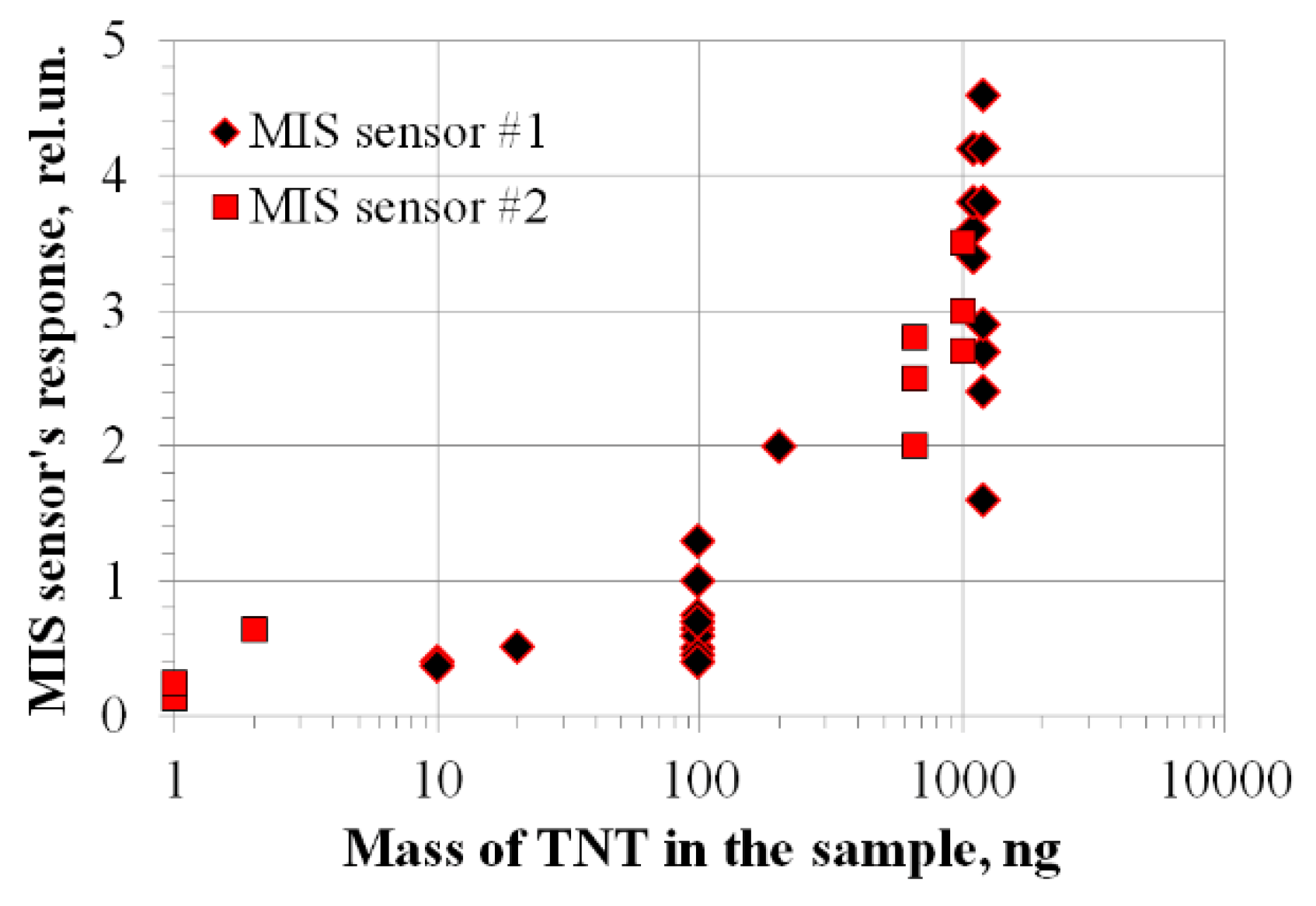
3. Nitro Compounds Detector Prototype Test Results
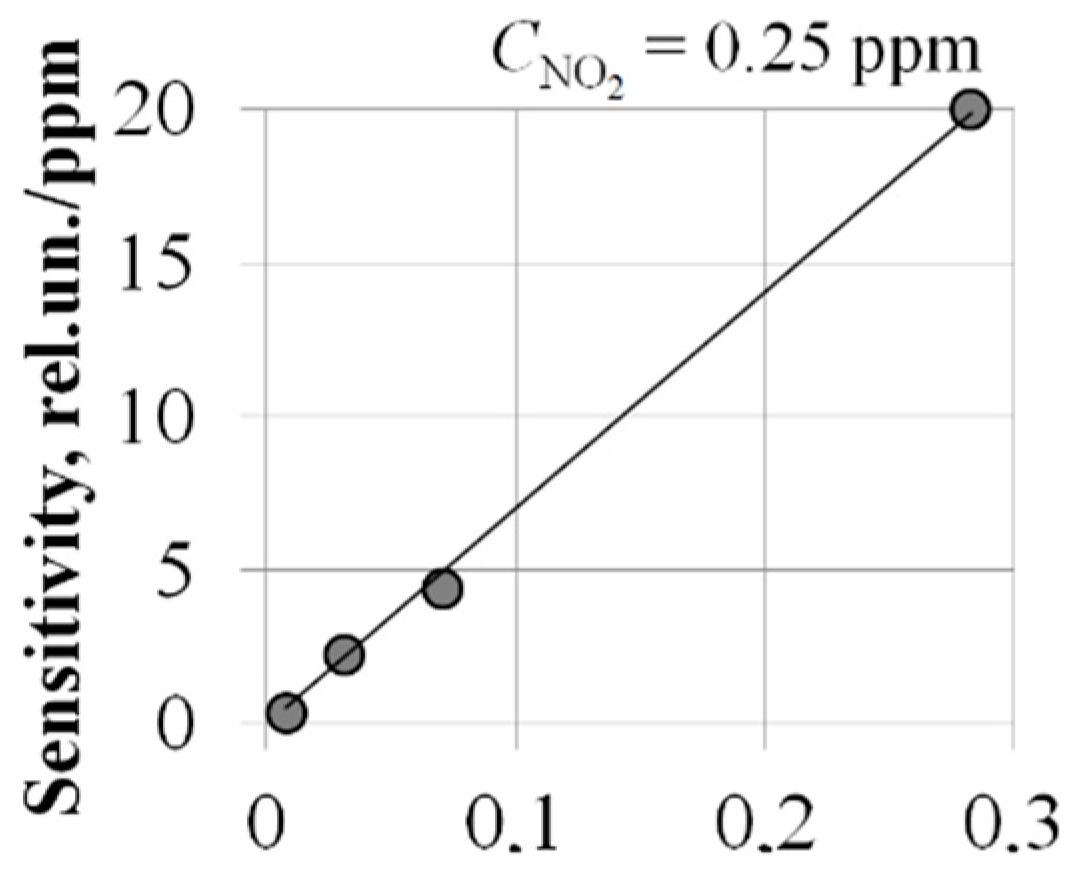
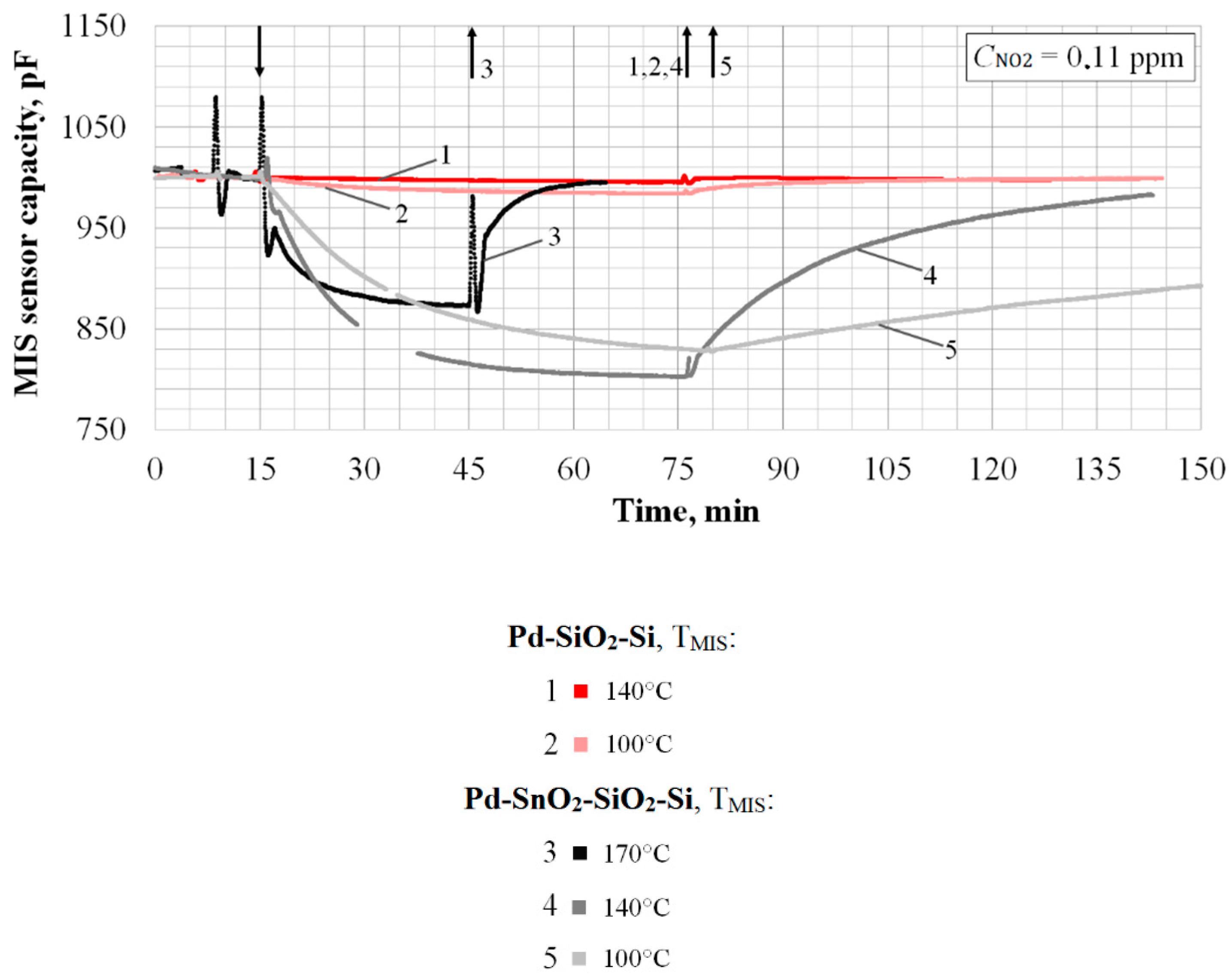
4. Investigation of the Possibility of Increasing the Sensitivity of MIS Sensors
- Nine times more for TMIS = 100 °C;
- Thirty-two times more for TMIS = 140 °C.
5. Discussion and Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Baili, C.; Jin, B.; Peng, R.; Zhao, F.; Yi, J.; Guan, H.; Bu, X.; Zhao, J.; Chu, S. The thermal decomposition of silver dinitramide AgN(NO2)2. J. Therm. Anal. Calorim. 2016, 126. [Google Scholar] [CrossRef]
- Oxley, J.; Smith, J.; Rogers, E.; Dong, X. Gas production from thermal decomposition of explosives: Assessing the thermal stabilities of energetic materials from gas production data. J. Energ. Mater. 2000, 18, 97–121. [Google Scholar] [CrossRef]
- Nguyen, V.B. Molecular Structure and Mechanisms of Reactions of Gas-Phase Decomposition of Anion- and Cation-Radicals of Some C-, N-, O-Nitro Compounds According to Quantum Chemical Calculations: Thesis for a Candidate of Chemical Sciences. Ph.D. Thesis, The Kazan National Research Technological University, Kazan, Russia, 2014. (In Russian). [Google Scholar]
- Oxley, J.; Smith, J.; Kirschenbaum, L.; Marimganti, S.; Vadlamannati, S. Detection of explosives in hair using ion mobility spectrometry. J. Forensic Sci. 2008, 53. [Google Scholar] [CrossRef] [PubMed]
- Wilks, A.; Hart, M.; Koehl, A.; Somerville, J.; Boyle, B.; Ruiz-Alonso, D. Characterization of a miniature, ultra-high-field, ion mobility spectrometer. Int. J. Ion Mobil. Spec. 2012, 15, 199–222. [Google Scholar] [CrossRef]
- Sabo, M.; Malásková, M.; Matejcik, S. Ion mobility spectrometry–mass spectrometry studies of ion processes in air at atmospheric pressure and their application to thermal desorption of 2,4,6-trinitrotoluene. Plasma Sources Sci. Technol. 2014, 23. [Google Scholar] [CrossRef]
- Baranova, A.; Khokhlov, K.; Chuvashov, R.; Verbitskiy, E.; Cheprakova, E.; Rusinov, G.; Charushin, V. The portable detector of nitro-explosives in vapor phase with new sensing elements on a base of pyrimidine scaffold. J. Phys. Conf. Ser. 2017, 830. [Google Scholar] [CrossRef]
- Rembelski, D.; Bordet, J.; Brouard, Q.; Minot, B.; Barthet, C.; Frénois, C. Fluorescent sensing for nitrated compounds: Study of the sensor microstructure for improvement on explosives Detection. Procedia Eng. 2015, 120, 1011–1014. [Google Scholar] [CrossRef][Green Version]
- Wang, C.; Huang, H.; Bunes, B.; Wu, N.; Xu, M.; Yang, X.; Yu, L.; Zang, L. Trace detection of RDX, HMX and PETN explosives using a fluorescence spot sensor. Sci. Rep. 2016, 6. [Google Scholar] [CrossRef]
- Martelo, L.; Neves, T.; Figueiredo, J.; Marques, L.; Fedorov, A.; Charas, A.; Berberan, S.; Burrows, H. Towards the development of a low-cost device for the detection of explosives vapors by fluorescence quenching of conjugated polymers in solid matrices. Sensors 2017, 17, 2532. [Google Scholar] [CrossRef]
- Moore, D. Recent advances in trace explosives detection instrumentation. Sens. Imaging Int. J. 2007, 8, 9–38. [Google Scholar] [CrossRef]
- Caygill, S.; Davis, F.; Higson, S. Current trends in explosive detection techniques. Talanta 2012, 88, 14–29. [Google Scholar] [CrossRef] [PubMed]
- Gruznov, V.; Baldin, M.; Makas, A.; Titov, B. Progress in methods for the identification of explosives in Russia. J. Anal. Chem. 2011, 66, 1121–1131. [Google Scholar] [CrossRef]
- Bobrovnikov, S.; Gorlov, E.; Zharkov, V. Remote detection of traces of high-energy materials on an ideal substrate using the Raman effect. Atmos. Ocean. Opt. 2017, 30, 604–608. [Google Scholar] [CrossRef]
- Chirico, R.; Almaviva, S.; Colao, F.; Fiorani, L.; Nuvoli, M.; Schweikert, W.; Schnürer, F.; Cassioli, L.; Grossi, S.; Murra, D.; et al. Proximal detection of traces of energetic materials with an eye-safe UV raman prototype developed for civil applications. Sensors 2016, 16, 8. [Google Scholar] [CrossRef]
- Zyryanov, G.V.; Kopchuk, D.S.; Kovalev, I.S.; Nosova, E.V.; Rusinov, V.L.; Chupakhin, O.N. Chemosensors for detection of nitroaromatic compounds (explosives). Russ. Chem. Rev. 2014, 83, 783–819. [Google Scholar] [CrossRef]
- Dettlaff, A.; Jakobczyk, P.; Ficek, M.; Wilk, B.; Szala, M.; Wojtas, J.; Ossowski, T.; Bogdanowicz, R. Electrochemical determination of nitroaromatic explosives at boron-doped diamond/graphene nanowall electrodes: 2,4,6-trinitrotoluene and 2,4,6-trinitroanisole in liquid effluents. J. Hazard. Mater. 2019. [Google Scholar] [CrossRef]
- Long, H.; Harley-Trochimczyk, A.; Pham, T.; Tang, Z.; Shi, T.; Zettl, A.; Carraro, C.; Worsley, M.; Maboudian, R. High surface area MoS2/Graphene hybrid aerogel for ultrasensitive NO2 detection. Adv. Funct. Mater. 2016, 26. [Google Scholar] [CrossRef]
- Donarelli, M.; Prezioso, S.; Perrozzi, F.; Bisti, F.; Nardone, M.; Giancaterini, L.; Cantalini, C.; Ottaviano, L. Response to NO2 and other gases of resistive chemically exfoliated MoS2-based gas sensors. Sens. Actuators B Chem. 2015, 207, 602–613. [Google Scholar] [CrossRef]
- Strle, D.; Stefane, B.; Zupanič, E.; Trifkovič, M.; Maček, M.; Jakša, G.; Kvasic, I.; Muševič, I. Sensitivity comparison of vapor trace detection of explosives based on chemo-mechanical sensing with optical detection and capacitive sensing with electronic detection. Sensors 2014, 14, 11467–11491. [Google Scholar] [CrossRef]
- Udrea, F.; Sunglyul, M.; Gardner, J.W.; Park, J.; Ali, S.; Choi, Y.; Guha, P.; Vieira, S.; Kim, H.; Kim, S.H.; et al. Three technologies for a smart miniaturized gas-sensor: SOI CMOS, micromachining, and CNTs—Challenges and performance. Tech. Dig. Int. Electron Devices Meet. 2008, 831–834. [Google Scholar] [CrossRef]
- Kwoka, M.; Szuber, J. Studies of NO2 gas-sensing characteristics of a novel room-temperature surface-photovoltage gas sensor device. Sensors 2020, 20, 408. [Google Scholar] [CrossRef] [PubMed]
- Yamazoe, N. Toward innovations of gas sensor technology. Sens. Actuators B Chem. 2005, 108, 2–14. [Google Scholar] [CrossRef]
- Geng, X.; Lahem, D.; Zhang, C.; Li, C.-J.; Olivier, M.-G.; Debliquy, M. Visible light enhanced black NiO sensors for ppb-level NO2 detection at room temperature. Ceram. Int. 2018, 45. [Google Scholar] [CrossRef]
- Senesac, L.; Thundat, T. Nanosensors for trace explosive detection. Mater. Today Mater Today. 2008, 11, 28–36. [Google Scholar] [CrossRef]
- Guillemot, M.; Dayber, F.; Montméat, P.; Barthet, C.; Prené, P. Detection of explosives vapours on quartz crystal microbalances: Generation of very low-concentrated vapours for sensors calibration. Procedia Chem. 2009, 1, 967–970. [Google Scholar] [CrossRef][Green Version]
- Lakkis, S.; Younes, R.; Alayli, Y.; Sawan, M. Review of recent trends in gas sensing technologies and their miniaturization potential. Sens. Rev. 2014, 34, 24–35. [Google Scholar] [CrossRef]
- Oprea, A.; Barsan, N.; Weimar, U. Work function changes in gas sensitive materials: Fundamentals and applications. Sens. Actuators B Chem. 2009, 142, 470–493. [Google Scholar] [CrossRef]
- Leidinger, M.; Sauerwald, T.; Alépée, C.; Schütze, A. Miniaturized integrated gas sensor systems combining metal oxide gas sensors and pre-concentrators. Procedia Eng. 2016, 168, 293–296. [Google Scholar] [CrossRef]
- Gradišek, A.; van Midden, M.; Koterle, M.; Prezelj, V.; Strle, D.; Stefane, B.; Brodnik, H.; Trifkovič, M.; Kvasic, I.; Zupanič, E.; et al. Improving the chemical selectivity of an electronic nose to TNT, DNT and RDX using machine learning. Sensors 2019, 19, 5207. [Google Scholar] [CrossRef]
- Patel, S.; Hobson, S.; Cemalovic, S.; Mlsna, T. Chemicapacitive microsensors for detection of explosives and TICs. Proc. SPIE Int. Soc. Opt. Eng. 2005, 5986. [Google Scholar] [CrossRef]
- Bolodurin, B.A.; Korchak, V.Y.; Litvinov, A.V.; Mikhailov, A.A.; Nozdrya, D.A.; Pomazan, Y.V.; Filipchuk, D.V.; Etrekova, M.O. Comprehensive research on the response of mis sensors of pd-sio2-si and pd-ta2o5-sio2-si structures to various gases in air. Russ. J. Gen. Chem. 2018, 88, 2732–2739. [Google Scholar] [CrossRef]
- Method and Device for Detection of Hydrogen. Application No.36892/75, 9 August 1978.
- Litvinov, A.V.; Samotaev, N.N.; Etrekova, M.O.; Klishin, Y.A.; Korolev, N.A. Cluster model of the mechanism of sensitivity of gas sensors based on MIS structures. Phys. At. Nucl. 2019, 82. [Google Scholar] [CrossRef]
- Wolkenstein, F.F. Electronic processes on semiconductor surfaces during chemisorption. M. “Nauka”. 1987. (In Russian). Available online: https://link.springer.com/content/pdf/bfm%3A978-1-4615-3656-7%2F1.pdf (accessed on 9 March 2020).
- Litvinov, A.V.; Samotaev, N.N.; Etrekova, M.O.; Mikhailov, A.A. The detection of nitro compounds by using MIS sensor. IOP Conf. Ser. Mater. Sci. Eng. 2019, 498. [Google Scholar] [CrossRef]
- Etrekova, M.O.; Litvinov, A.V.; Mikhailov, A.A. The detection of nitro compounds vapor using sensors based on MIS structures manufactured by laser deposition. IOP Conf. Ser. J. Phys. 2019, 1238. [Google Scholar] [CrossRef]
- Samotaev, N.N.; Litvinov, A.V.; Podlepetskiy, B.I.; Etrekova, M.O.; Filipchuk, D.V.; Mikhailov, A.A.; Bukharov, D.G.; Demidov, V.M. Methods for measuring the output signals of gas-sensitive sensors based on MIS capacitors. Sens. Syst. 2019, 5, 47–53. (In Russian) [Google Scholar]
- Filipchuk, D.V.; Litvinov, A.V.; Mikhailov, A.A.; Etrekova, M.O. Investigation of the effect of changes in the relative humidity of the ambient air on the readings of a gas detector based on an MIS sensor. V International Conference “Laser, Plasma Research and Technology” Laplaz-2019: Collection of Scientific Papers. 2019, Volume 1, pp. 180–181. (In Russian). Available online: https://www.elibrary.ru/item.asp?id=40875625 (accessed on 9 March 2020).
- Oblov, K.Y.; Samotaev, N.N.; Etrekova, M.O.; Onishchenko, E.M. Gas Sensor. Utility Model Patent No. 192819; reg. No. 2019118497, 14 June 2019. [Google Scholar]
- Samotaev, N.; Oblov, K.; Veselov, D.; Podlepetsky, B.; Etrekova, M.; Volkov, N.; Zibilyuk, N. Technology of SMD MOX Gas Sensors Rapid Prototyping. Mater. Sci. Forum 2019, 977, 231–237. [Google Scholar] [CrossRef]
- Oblov, K.Y.; Samotaev, N.N.; Etrekova, M.O.; Gorskova, A.V. Laser micromilling technology as a key for rapid ceramic MEMS devices. Phys. At. Nucl. 2019, 82. [Google Scholar] [CrossRef]
- Kaur, J.; Roy, S.C.; Bhatnagar, M.C. Highly sensitive SnO2 thin film NO2 gas sensor operating at low temperature. Sens. Actuators B Chem. 2007, 123, 1090–1095. [Google Scholar] [CrossRef]
- Oxley, J.C.; Smith, J.L.; Luo, W.; Brady, J. Determining the vapor pressures of diacetone diperoxide (DADP), and hexamethylene triperoxide diamine (HMTD). Propellants Explos. Pyrotech. 2009, 34, 539–543. [Google Scholar] [CrossRef]




| Pd–SiO2–Si: dPd = 3 mm; Noise Level ±0.2 pF | ||||
|---|---|---|---|---|
| TMIS, °C | NO2 Sensitivity 1, pF/ppm | Response Time τ0.9, min | Recovery Time τ0.1, min | LOD 2 of NO2, ppb |
| 100 | 175 | 40 | 60 | 3.4 3 |
| 140 | 55 | - | - | 11.0 |
| Pd–SnO2–SiO2–Si: dPd = 2 mm; noise level ±0.5 pF | ||||
| 100 | 1565 | 45 | 250 | 1.0 |
| 140 | 1775 | 25 | 65 | 0.9 |
| 170 | 1180 | 15 | 15 | 1.3 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Samotaev, N.; Litvinov, A.; Etrekova, M.; Oblov, K.; Filipchuk, D.; Mikhailov, A. Prototype of Nitro Compound Vapor and Trace Detector Based on a Capacitive MIS Sensor. Sensors 2020, 20, 1514. https://doi.org/10.3390/s20051514
Samotaev N, Litvinov A, Etrekova M, Oblov K, Filipchuk D, Mikhailov A. Prototype of Nitro Compound Vapor and Trace Detector Based on a Capacitive MIS Sensor. Sensors. 2020; 20(5):1514. https://doi.org/10.3390/s20051514
Chicago/Turabian StyleSamotaev, Nikolay, Artur Litvinov, Maya Etrekova, Konstantin Oblov, Dmitrii Filipchuk, and Alexey Mikhailov. 2020. "Prototype of Nitro Compound Vapor and Trace Detector Based on a Capacitive MIS Sensor" Sensors 20, no. 5: 1514. https://doi.org/10.3390/s20051514
APA StyleSamotaev, N., Litvinov, A., Etrekova, M., Oblov, K., Filipchuk, D., & Mikhailov, A. (2020). Prototype of Nitro Compound Vapor and Trace Detector Based on a Capacitive MIS Sensor. Sensors, 20(5), 1514. https://doi.org/10.3390/s20051514





