A Systematic Review on the Use of Wearable Body Sensors for Health Monitoring: A Qualitative Synthesis
Abstract
1. Introduction
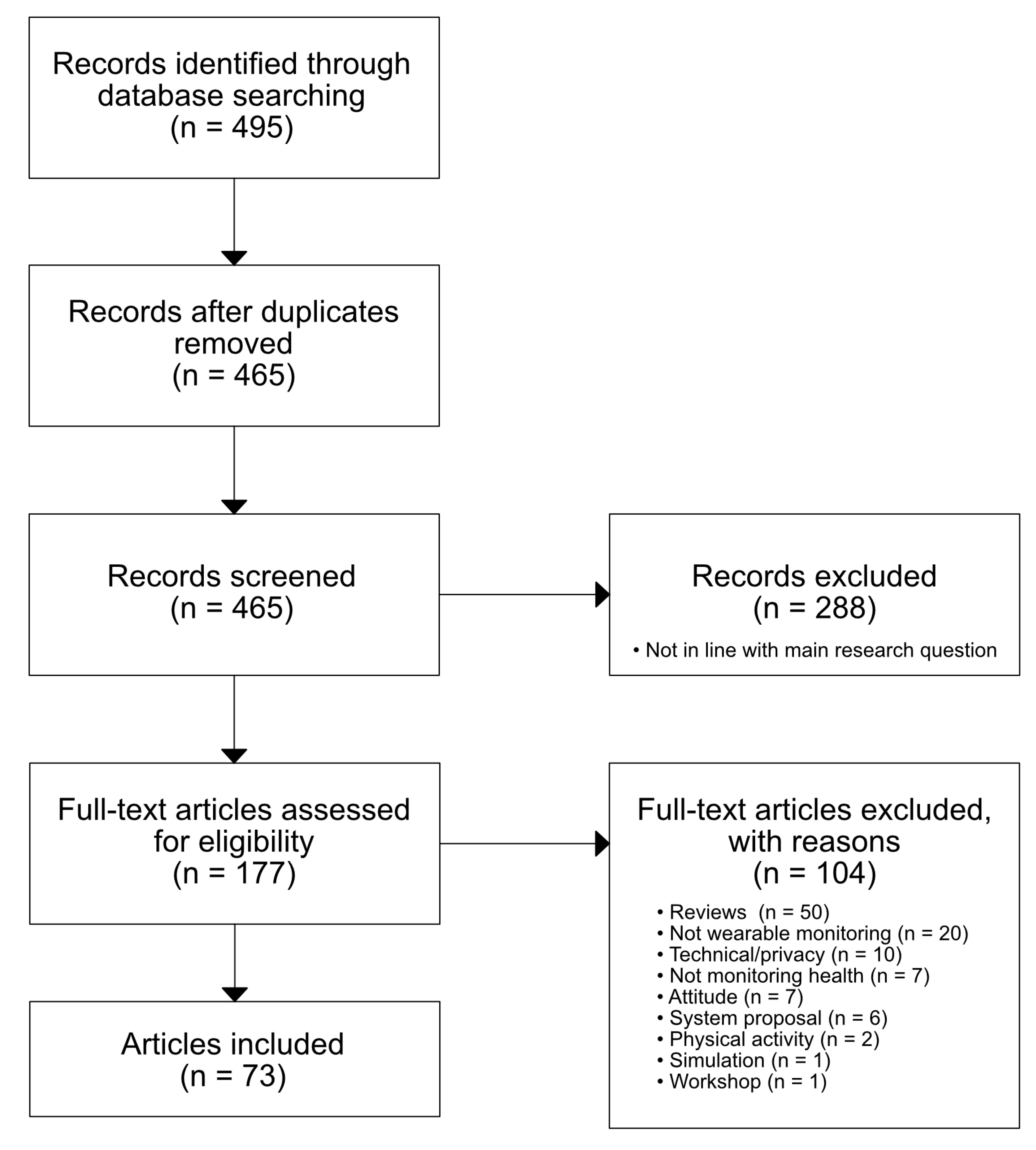
2. Methodology
Article Selection, Inclusion and Exclusion Criteria
- Articles should be published as a journal article or in conference proceedings.
- Articles should consider wearable technology and monitoring.
- Articles should present results from studies where sensor data were collected using humans. Alternatively, the articles present information on a system where the user trial is planned for but not conducted yet.
- Articles should be in English.
3. Qualitative Synthesis
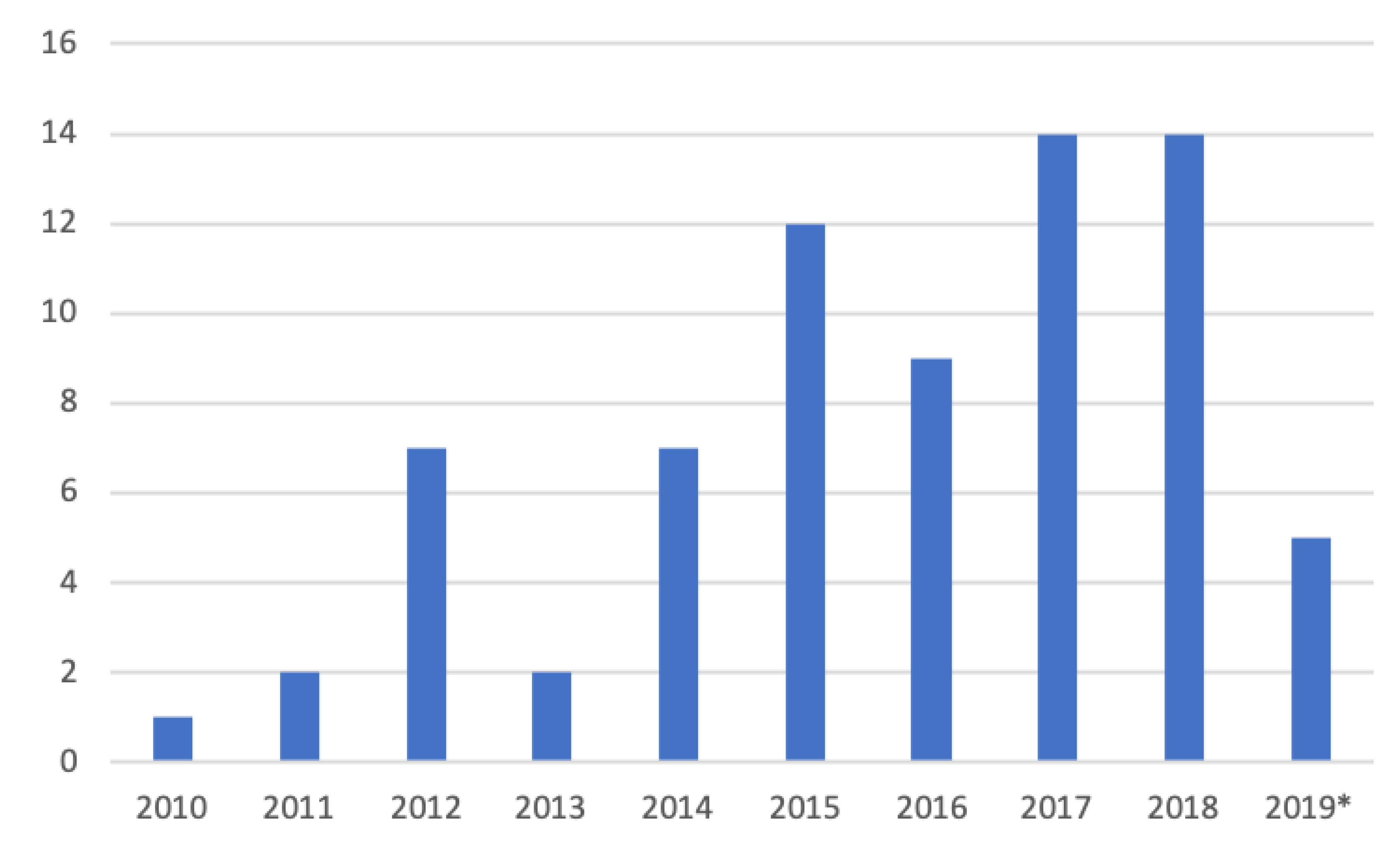
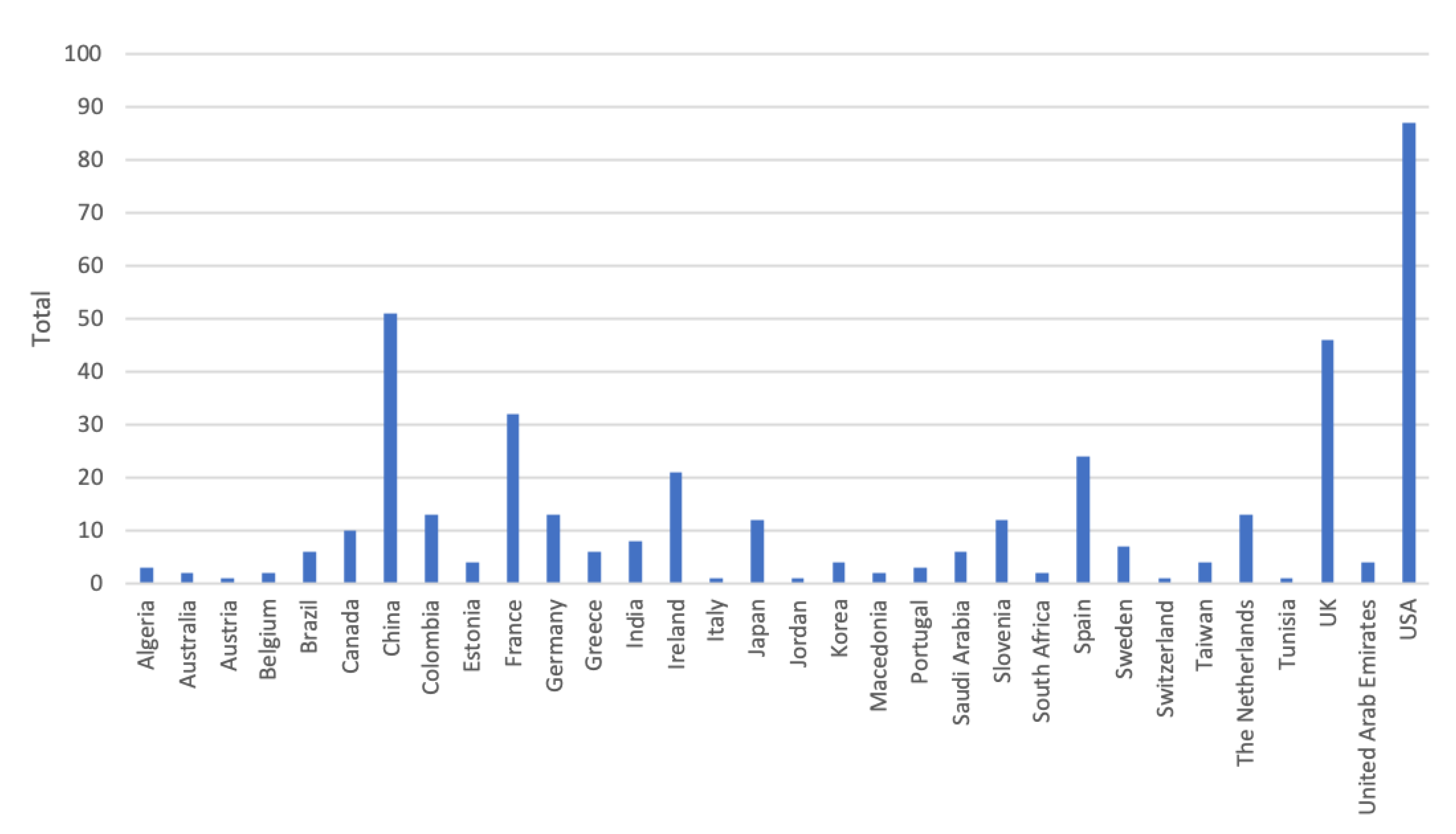
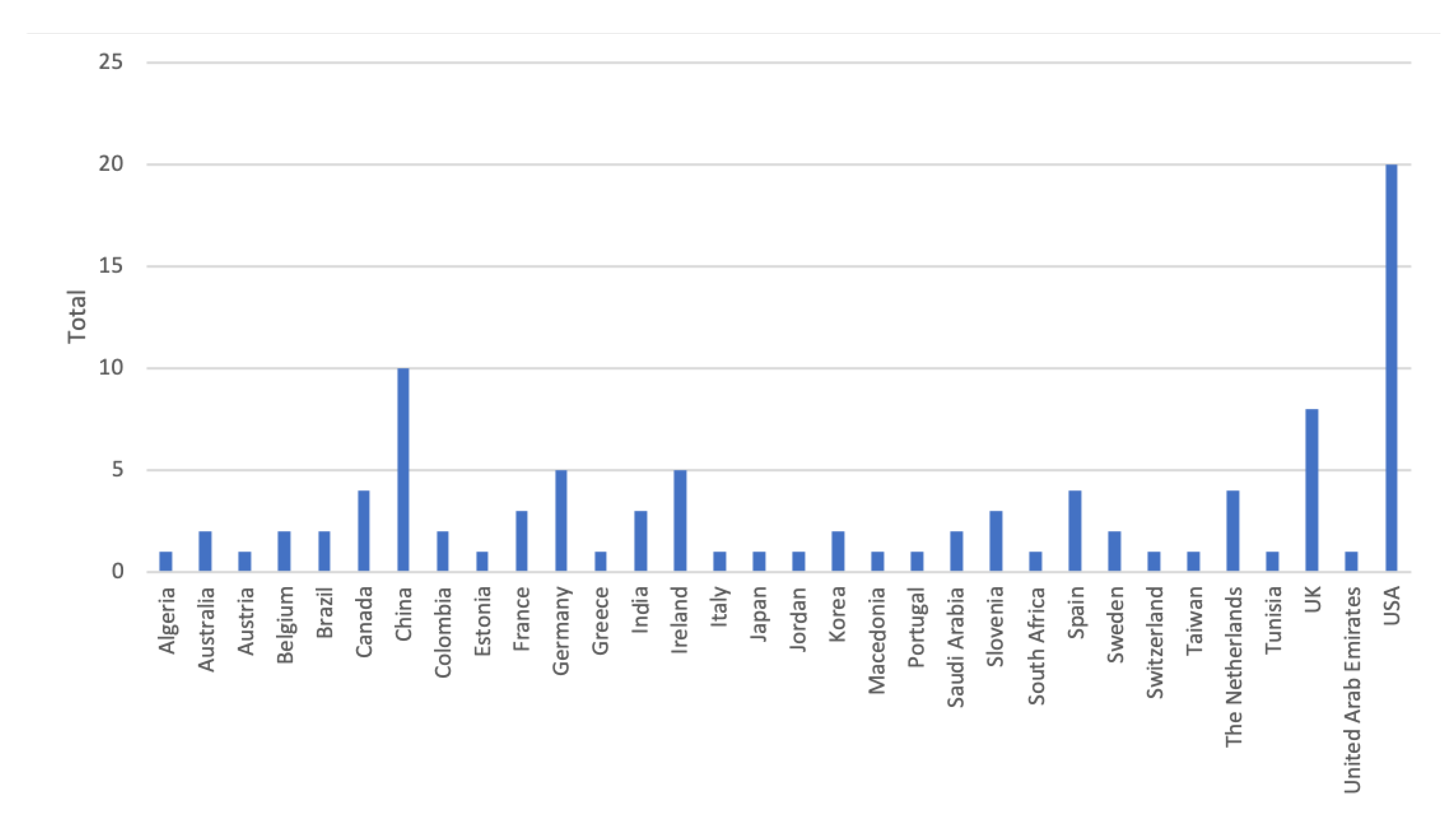
3.1. Research Methodology
3.2. Age Distribution
3.3. Gender Distribution
3.4. Tests on Patients and Healthy Users
4. Discussion and Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; the PRISMA Group. Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement. Ann. Internal Med. 2009, 151, 264–269. [Google Scholar] [CrossRef]
- Kekade, S.; Hseieh, C.H.; Islam, M.M.; Atique, S.; Mohammed Khalfan, A.; Li, Y.C.; Abdul, S.S. The usefulness and actual use of wearable devices among the elderly population. Comput. Methods Programs Biomed. 2018, 153, 137–159. [Google Scholar] [CrossRef]
- Li, Y.; Li, S.; Song, H.; Shao, B.; Yang, X.; Deng, N. Noninvasive blood pressure estimation with peak delay of different pulse waves. Int. J. Distrib. Sens. Netw. 2019, 15. [Google Scholar] [CrossRef]
- Wu, X.; Wang, Y.; Chien, C.; Pottie, G. Self-calibration of sensor misplacement based on motion signatures. In Proceedings of the 2013 IEEE International Conference on Body Sensor Networks, Cambridge, MA, USA, 6–9 May 2013; pp. 1–5. [Google Scholar] [CrossRef]
- Castro, D.; Coral, W.; Rodriguez, C.; Cabra, J.; Colorado, J. Wearable-Based Human Activity Recognition Using an IoT Approach. J. Sens. Actuator Netw. 2017, 6, 28. [Google Scholar] [CrossRef]
- Rodriguez, C.; Castro, D.M.; Coral, W.; Cabra, J.L.; Velasquez, N.; Colorado, J.; Mendez, D.; Trujillo, L.C.; ACM. IoT system for Human Activity Recognition using BioHarness 3 and Smartphone. In Proceedings of the International Conference on Future Networks and Distributed Systems, Cambridge, UK, 19–20 July 2017. [Google Scholar] [CrossRef]
- Doron, M.; Bastian, T.; Maire, A.; Dugas, J.; Perrin, E.; Gris, F.; Guillemaud, R.; Deschamps, T.; Bianchi, P.; Caritu, Y.; et al. Estimation of physical activity monitored during the day-to-day life by an autonomous wearable device (SVELTE project). In Proceedings of the 2013 35th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Osaka, Japan, 3–7 July 2013; pp. 4629–4632. [Google Scholar] [CrossRef]
- Xu, J.Y.; Chang, H.I.; Chien, C.; Kaiser, W.J.; Pottie, G.J. Context-driven, prescription-based personal activity classification: Methodology, architecture, and end-to-end implementation. IEEE J. Biomed. Health Inform. 2014, 18, 1015–1025. [Google Scholar] [CrossRef]
- Xu, J.Y.; Wang, Y.; Barrett, M.; Dobkin, B.; Pottie, G.J.; Kaiser, W.J. Personalized multilayer maily life profiling through context enabled activity classification and motion reconstruction: An integrated system approach. IEEE J. Biomed. Health Inform. 2016, 20, 177–188. [Google Scholar] [CrossRef] [PubMed]
- Velicu, O.R.; Madrid, N.M.; Seepold, R.; IEEE. Experimental sleep phases monitoring. In Proceedings of the 2016 3rd IEEE Embs International Conference on Biomedical and Health Informatics, Las Vegas, NV, USA, 24–27 February 2016; pp. 625–628. [Google Scholar]
- Seeger, C.; Buchmann, A.; Van Laerhoven, K. An Event-based BSN Middleware That Supports Seamless Switching Between Sensor Configurations. In Proceedings of the 2nd ACM SIGHIT International Health Informatics Symposium, Miami, Fl, USA, 28–30 January 2012; pp. 503–512. [Google Scholar] [CrossRef]
- Wannenburg, J.; Malekian, R. Body Sensor Network for Mobile Health Monitoring, a Diagnosis and Anticipating System. IEEE Sens. J. 2015, 15, 6839–6852. [Google Scholar] [CrossRef]
- Bonnevie, T.; Gravier, F.E.; Elkins, M.; Dupuis, J.; Prieur, G.; Combret, Y.; Viacroze, C.; Debeaumont, D.; Robleda-Quesada, A.; Quieffin, J.; et al. People undertaking pulmonary rehabilitation are willing and able to provide accurate data via a remote pulse oximetry system: a multicentre observational study. J. Physiother. 2019, 65, 28–36. [Google Scholar] [CrossRef] [PubMed]
- Caulfield, B.; Kaljo, I.; Donnelly, S. Use of a consumer market activity monitoring and feedback device improves exercise capacity and activity levels in COPD. In Proceedings of the 2014 36th Annual International Conference of the IEEE Engineering in Medicine and Biology Society, Chicago, IL, USA, 26–30 August 2014; pp. 1765–1768. [Google Scholar] [CrossRef]
- Estrada, L.; Torres, A.; Sarlabous, L.; Jané, R. Evaluating respiratory muscle activity using a wireless sensor platform. In Proceedings of the 2016 38th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Orlando, FL, USA, 16–20 August 2016; pp. 5769–5772. [Google Scholar] [CrossRef]
- Katsaras, T.; Milsis, A.; Rizikari, M.; Saoulis, N.; Varoutaki, E.; Vontetsianos, A. The use of the “Healthwear” wearable system in chronic patients’ early hospital discharge: Control randomized clinical trial. In Proceedings of the 2011 5th International Symposium on Medical Information and Communication Technology, Montreux, Switzerland, 27–30 March 2011; pp. 143–146. [Google Scholar] [CrossRef]
- Naranjo-Hernández, D.; Talaminos-Barroso, A.; Reina-Tosina, J.; Roa, L.M.; Barbarov-Rostan, G.; Cejudo-Ramos, P.; Márquez-Martín, E.; Ortega-Ruiz, F. Smart vest for respiratory rate monitoring of copd patients based on non-contact capacitive sensing. Sensors 2018, 18, 2144. [Google Scholar] [CrossRef]
- Huang, A.; Xu, W.; Li, Z.; Xie, L.; Sarrafzadeh, M.; Li, X.; Cong, J. System Light-Loading Technology for mHealth: Manifold-Learning-Based Medical Data Cleansing and Clinical Trials in WE-CARE Project. IEEE J. Biomed. Health Inform. 2014, 18, 1581–1589. [Google Scholar] [CrossRef] [PubMed]
- Huang, A.; Chen, C.; Bian, K.; Duan, X.; Chen, M.; Gao, H.; Meng, C.; Zheng, Q.; Zhang, Y.; Jiao, B.; et al. WE-CARE: An intelligent mobile telecardiology system to enable mHealth applications. IEEE J. Biomed. Health Inform. 2014, 18, 693–702. [Google Scholar] [CrossRef] [PubMed]
- Javaid, A.Q.; Chang, I.S.; Mihailidis, A. Ballistocardiogram Based Identity Recognition: Towards Zero-Effort Health Monitoring in an Internet-of-Things (IoT) Environment. In Proceedings of the 2018 40th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Honolulu, HI, USA, 18–21 July 2018; Volume 2018, pp. 3326–3329. [Google Scholar] [CrossRef]
- Raad, M.W.; Sheltami, T.; Deriche, M. A Ubiquitous Telehealth System for the Elderly. In Internet of Things: User-Centric Iot, Pt I; Lecture Notes of the Institute for Computer Sciences Social Informatics and Telecommunications Engineering; Giaffreda, R., Vieriu, R.L., Pasher, E., Bendersky, G., Jara, A.J., Rodrigues, J., Dekel, E., Mandler, B., Eds.; Springer: Berlin, Germany, 2015; Volume 150, pp. 159–166. [Google Scholar] [CrossRef]
- Simjanoska, M.; Gjoreski, M.; Gams, M.; Bogdanova, A.M. Non-invasive blood pressure estimation from ECG using machine learning techniques. Sensors 2018, 18, 1160. [Google Scholar] [CrossRef] [PubMed]
- Susič, T.P.; Stanič, U. Penetration of the ICT technology to the health care primary sector—Ljubljana PILOT. In Proceedings of the 2016 39th International Convention on Information and Communication Technology, Electronics and Microelectronics (MIPRO), Opatija, Croatia, 30 May–3 June 2016; pp. 436–441. [Google Scholar] [CrossRef]
- Al-Taee, M.A.; Al-Nuaimy, W.; Al-Ataby, A.; Muhsin, Z.J.; Abood, S.N.; IEEE. Mobile Health Platform for Diabetes Management Based on the Internet-of-Things. In Proceedings of the 2015 IEEE Jordan Conference on Applied Electrical Engineering and Computing Technologies, New York, NY, USA, 3–5 November 2015. [Google Scholar] [CrossRef]
- Alshurafa, N.; Kalantarian, H.; Pourhomayoun, M.; Sarin, S.; Liu, J.J.; Sarrafzadeh, M. Non-invasive monitoring of eating behavior using spectrogram analysis in a wearable necklace. In Proceedings of the 2014 IEEE Healthcare Innovation Conference (HIC), Seattle, WA, USA, 8–10 October 2014; pp. 71–74. [Google Scholar] [CrossRef]
- Alshurafa, N.; Kalantarian, H.; Pourhomayoun, M.; Liu, J.J.; Sarin, S.; Shahbazi, B.; Sarrafzadeh, M. Recognition of Nutrition Intake Using Time-Frequency Decomposition in a Wearable Necklace Using a Piezoelectric Sensor. IEEE Sens. J. 2015, 15, 3909–3916. [Google Scholar] [CrossRef]
- Dong, B.; Biswas, S. Meal-time and duration monitoring using wearable sensors. Biomed. Signal Process. Control 2017, 32, 97–109. [Google Scholar] [CrossRef]
- Onoue, T.; Goto, M.; Kobayashi, T.; Tominaga, T.; Ando, M.; Honda, H.; Yoshida, Y.; Tosaki, T.; Yokoi, H.; Kato, S.; et al. Randomized controlled trial for assessment of Internet of Things system to guide intensive glucose control in diabetes outpatients: Nagoya Health Navigator Study protocol. Nagoya J. Med. Sci. 2017, 79, 323–329. [Google Scholar] [CrossRef] [PubMed]
- Atallah, L.; Wiik, A.; Jones, G.G.; Lo, B.; Cobb, J.P.; Amis, A.; Yang, G.Z. Validation of an ear-worn sensor for gait monitoring using a force-plate instrumented treadmill. Gait Posture 2012, 35, 674–676. [Google Scholar] [CrossRef]
- Godfrey, A.; Din, S.D.; Barry, G.; Mathers, J.C.; Rochester, L. Within trial validation and reliability of a single tri-axial accelerometer for gait assessment. In Proceedings of the 2014 36th Annual International Conference of the IEEE Engineering in Medicine and Biology Society, Chicago, IL, USA, 26–30 August 2014; pp. 5892–5895. [Google Scholar] [CrossRef]
- Lee, J.K.; Robinovitch, S.N.; Park, E.J. Inertial Sensing-Based Pre-Impact Detection of Falls Involving Near-Fall Scenarios. IEEE Trans. Neural Syst. Rehabil. Eng. 2015, 23, 258–266. [Google Scholar] [CrossRef]
- Liang, D.; Zhao, G.; Guo, Y.; Wang, L. Pre-impact & impact detection of falls using wireless Body Sensor Network. In Proceedings of the 2012 IEEE-EMBS International Conference on Biomedical and Health Informatics, Hong Kong, China, 5–7 January 2012; pp. 763–766. [Google Scholar] [CrossRef]
- Liang, S.; Chu, T.; Lin, D.; Ning, Y.; Li, H.; Zhao, G. Pre-impact Alarm System for Fall Detection Using MEMS Sensors and HMM-based SVM Classifier. In Proceedings of the 2018 40th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Honolulu, HI, USA, 18–21 July 2018; pp. 4401–4405. [Google Scholar] [CrossRef]
- Paiman, C.; Lemus, D.; Short, D.; Vallery, H. Observing the State of Balance with a Single Upper-Body Sensor. Front. Robot. AI 2016, 3. [Google Scholar] [CrossRef]
- Tino, A.; Carvalho, M.; Preto, N.F.; McConville, K.M.V. Wireless vibrotactile feedback system for postural response improvement. In Proceedings of the 2011 33rd Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Boston, MA, USA, 30 August–3 September 2011; Volume 2011, pp. 5203–5206. [Google Scholar] [CrossRef]
- Williams, B.; Allen, B.; True, H.; Fell, N.; Levine, D.; Sartipi, M.; IEEE. A Real-time, Mobile Timed Up and Go System. In Proceedings of the 2015 IEEE 12th International Conference on Wearable and Implantable Body Sensor Networks (BSN), Cambridge, MA, USA, 9–12 June 2015. [Google Scholar] [CrossRef]
- Wu, Y.; Su, Y.; Feng, R.; Yu, N.; Zang, X. Wearable-sensor-based pre-impact fall detection system with a hierarchical classifier. Measurement 2019, 140, 283–292. [Google Scholar] [CrossRef]
- Zhao, G.; Mei, Z.; Liang, D.; Kamen, I.; Guo, Y.; Wang, Y.; Wang, L. Exploration and Implementation of a Pre-Impact Fall Recognition Method Based on an Inertial Body Sensor Network. Sensors 2012, 12, 15338–15355. [Google Scholar] [CrossRef]
- Zhong, R.; Rau, P.L.P.; Yan, X. Gait Assessment of Younger and Older Adults with Portable Motion-Sensing Methods: A User Study. Mob. Inf. Syst. 2019, 2019. [Google Scholar] [CrossRef]
- Giuberti, M.; Ferrari, G.; Contin, L.; Cimolin, V.; Azzaro, C.; Albani, G.; Mauro, A. Automatic UPDRS Evaluation in the Sit-to-Stand Task of Parkinsonians: Kinematic Analysis and Comparative Outlook on the Leg Agility Task. IEEE J. Biomed. Health Inform. 2015, 19, 803–814. [Google Scholar] [CrossRef] [PubMed]
- Gong, J.; Lach, J.; Qi, Y.; Goldman, M.D. Causal analysis of inertial body sensors for enhancing gait assessment separability towards multiple sclerosis diagnosis. In Proceedings of the 2015 IEEE 12th International Conference on Wearable and Implantable Body Sensor Networks (BSN), Cambridge, MA, USA, 9–12 June 2015. [Google Scholar] [CrossRef]
- Gong, J.; Qi, Y.; Goldman, M.D.; Lach, J. Causality Analysis of Inertial Body Sensors for Multiple Sclerosis Diagnostic Enhancement. IEEE J. Biomed. Health Inform. 2016, 20, 1273–1280. [Google Scholar] [CrossRef] [PubMed]
- Kuusik, A.; Alam, M.M.; Kask, T.; Gross-Paju, K. Wearable m-assessment system for neurological disease patients. In Proceedings of the 2018 IEEE 4th World Forum on Internet of Things (WF-IoT), Singapore, Singapore, 5–8 February 2018; pp. 201–206. [Google Scholar] [CrossRef]
- Sok, P.; Xiao, T.; Azeze, Y.; Jayaraman, A.; Albert, M.V. Activity recognition for incomplete spinal cord injury subjects using hidden markov models. IEEE Sens. J. 2018, 18, 6369–6374. [Google Scholar] [CrossRef]
- Stamate, C.; Magoulas, G.D.; Kueppers, S.; Nomikou, E.; Daskalopoulos, I.; Luchini, M.U.; Moussouri, T.; Roussos, G. Deep learning Parkinson’s from smartphone data. In Proceedings of the 2017 IEEE International Conference on Pervasive Computing and Communications, PerCom 2017, Kona, HI, USA, 13–17 March 2017; pp. 31–40. [Google Scholar] [CrossRef]
- Stamate, C.; Magoulas, G.D.; Kueppers, S.; Nomikou, E.; Daskalopoulos, I.; Jha, A.; Pons, J.S.; Rothwell, J.; Luchini, M.U.; Moussouri, T.; et al. The cloudUPDRS app: A medical device for the clinical assessment of Parkinson’s Disease. Pervasive Mob. Comput. 2018, 43, 146–166. [Google Scholar] [CrossRef]
- Rednic, R.; Gaura, E.; Brusey, J.; Kemp, J. Wearable posture recognition systems: Factors affecting performance. In Proceedings of the 2012 IEEE-EMBS International Conference on Biomedical and Health Informatics, Hong Kong, China, 5–7 January 2012; pp. 200–203. [Google Scholar] [CrossRef]
- Argent, R.; Slevin, P.; Bevilacqua, A.; Neligan, M.; Daly, A.; Caulfield, B. Wearable sensor-based exercise biofeedback for orthopaedic rehabilitation: A mixed methods user evaluation of a prototype system. Sensors 2019, 19, 432. [Google Scholar] [CrossRef]
- Banos, O.; Moral-Munoz, J.A.; Diaz-Reyes, I.; Arroyo-Morales, M.; Damas, M.; Herrera-Viedma, E.; Hong, C.S.; Lee, S.; Pomares, H.; Rojas, I.; et al. MDurance: A novel mobile health system to support trunk endurance assessment. Sensors 2015, 15, 13159–13183. [Google Scholar] [CrossRef]
- Lee, S.I.; Adans-Dester, C.P.; Grimaldi, M.; Dowling, A.V.; Horak, P.C.; Black-Schaffer, R.M.; Bonato, P.; Gwin, J.T. Enabling stroke rehabilitation in home and community settings: A wearable sensor-based approach for upper-limb motor training. IEEE J. Transl. Eng. Health Med. 2018, 6. [Google Scholar] [CrossRef]
- Timmermans, A.A.A.; Seelen, H.A.M.; Geers, R.P.J.; Saini, P.K.; Winter, S.; te Vrugt, J.; Kingma, H. Sensor-Based Arm Skill Training in Chronic Stroke Patients: Results on Treatment Outcome, Patient Motivation, and System Usability. IEEE Trans. Neural Syst. Rehabil. Eng. 2010, 18, 284–292. [Google Scholar] [CrossRef]
- Whelan, D.F.; O’Reilly, M.A.; Ward, T.E.; Delahunt, E.; Caulfield, B. Technology in rehabilitation: Comparing personalised and global classification methodologies in evaluating the squat exercise with wearable IMUs. Methods Inf. Med. 2017, 56, 361–369. [Google Scholar] [CrossRef]
- Xu, J.K.; Lee, U.H.; Bao, T.; Huang, Y.J.; Sienko, K.H.; Shull, P.B.; IEEE. Wearable sensing and haptic feedback research platform for gait retraining. In Proceedings of the 2017 IEEE 14th International Conference on Wearable and Implantable Body Sensor Networks, Eindhoven, The Netherlands, 9–12 May 2017; pp. 125–128. [Google Scholar]
- Lin, C.; Gamble, J.; Yang, Y.; Wang, J. Estimating the influence of chronotype and social zeitgebers on circadian rhythms using an accelerometer-based sensor network. In Proceedings of the 2012 IEEE-EMBS International Conference on Biomedical and Health Informatics, Hong Kong, China, 5–7 January 2012; pp. 285–288. [Google Scholar] [CrossRef]
- Nakamura, T.; Goverdovsky, V.; Morrell, M.J.; Mandic, D.P. Automatic Sleep Monitoring Using Ear-EEG. IEEE J. Transl. Eng. Health Med. 2017, 5, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Parnandi, A.; Gutierrez-Osuna, R. Physiological Modalities for Relaxation Skill Transfer in Biofeedback Games. IEEE J. Biomed. Health Inform. 2017, 21, 361–371. [Google Scholar] [CrossRef] [PubMed]
- Uday, S.; Jyotsna, C.; Amudha, J.; IEEE. Detection of Stress using Wearable Sensors in IoT Platform. In Proceedings of the 2018 Second International Conference on Inventive Communication and Computational Technologies (ICICCT), Coimbatore, India, 20–21 April 2018; pp. 492–498. [Google Scholar] [CrossRef]
- Umemura, G.S.; Pinho, J.P.; Furtado, F.; Gonçalves, B.S.B.; Fomer-Cordero, A. Comparison of sleep parameters assessed by actigraphy of healthy young adults from a small town and a megalopolis in an emerging country. In Proceedings of the 2017 IEEE Healthcare Innovations and Point of Care Technologies (HI-POCT), Bethesda, MD, USA, 6–8 November 2017; pp. 200–203. [Google Scholar] [CrossRef]
- Ayzenberg, Y.; Picard, R.W. FEEL: A System for Frequent Event and Electrodermal Activity Labeling. IEEE J. Biomed. Health Inform. 2014, 18, 266–277. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Pagán, J.; Risco-Martín, J.L.; Moya, J.M.; Ayala, J.L. Grammatical Evolutionary Techniques for Prompt Migraine Prediction. In Proceedings of the Genetic and Evolutionary Computation Conference, Denver, CO, USA, 20–24 July 2016; pp. 973–980. [Google Scholar] [CrossRef]
- Rawashdeh, M.; Al-Qurishi, M.; Al-Rakhami, M.; Al-Quraishi, M.S. A multimedia cloud-based framework for constant monitoring on obese patients. In Proceedings of the 2017 IEEE International Conference on Multimedia & Expo Workshops (ICMEW), Hong Kong, China, 10–14 July 2017; pp. 139–144. [Google Scholar] [CrossRef]
- Wu, W.; Pirbhulal, S.; Sangaiah, A.K.; Mukhopadhyay, S.C.; Li, G. Optimization of signal quality over comfortability of textile electrodes for ECG monitoring in fog computing based medical applications. Future Gener. Comput. Syst. 2018, 86, 515–526. [Google Scholar] [CrossRef]
- Buonocore, C.M.; Rocchio, R.A.; Roman, A.; King, C.E.; Sarrafzadeh, M. Wireless Sensor-Dependent Ecological Momentary Assessment for Pediatric Asthma mHealth Applications. In Proceedings of the Second IEEE/ACM International Conference on Connected Health: Applications, Systems and Engineering Technologies, Philadelphia, PA, USA, 17–19 July 2017; pp. 137–146. [Google Scholar] [CrossRef]
- Depolli, M.; Avbelj, V.; Trobec, R.; Kališnik, J.M.; Tadej, K.; Susič, A.P.; Stanič, U.; Semeja, A. PCARD platform for mhealth monitoring. Informatica 2016, 40, 117–123. [Google Scholar]
- Ghazal, M.; Khalil, Y.A.; Dehbozorgi, F.J.; Alhalabi, M.T. An integrated caregiver-focused mHealth framework for elderly care. In Proceedings of the 11th IEEE International Conference on Wireless and Mobile Computing, Networking and Communications, WiMob 2015, Abu Dhabi, UAE, 19–21 October 2015; pp. 238–245. [Google Scholar] [CrossRef]
- Anupama, K.R.; Adarsh, R.; Pahwa, P.; Ramachandran, A. Machine Learning-Based Techniques for Fall Detection in Geriatric Healthcare Systems. In Proceedings of the 2018 9th International Conference on Information Technology in Medicine and Education (ITME), Hangzhou, China, 19–21 October 2018; pp. 232–237. [Google Scholar] [CrossRef]
- Boutellaa, E.; Kerdjidj, O.; Ghanem, K. Covariance matrix based fall detection from multiple wearable sensors. J. Biomed. Inform. 2019, 103189. [Google Scholar] [CrossRef]
- Memedi, M.; Tshering, G.; Fogelberg, M.; Jusufi, I.; Kolkowska, E.; Klein, G. An Interface for IoT: Feeding Back Health-Related Data to Parkinson’s Disease Patients. J. Sens. Actuator Netw. 2018, 7. [Google Scholar] [CrossRef]
- Dobbins, C.; Rawassizadeh, R.; Momeni, E. Detecting physical activity within lifelogs towards preventing obesity and aiding ambient assisted living. Neurocomputing 2017, 230, 110–132. [Google Scholar] [CrossRef]
- Argent, R.; Slevin, P.; Bevilacqua, A.; Neligan, M.; Daly, A.; Caulfield, B. Clinician perceptions of a prototype wearable exercise biofeedback system for orthopaedic rehabilitation: A qualitative exploration. BMJ Open 2018, 8. [Google Scholar] [CrossRef]
- Zhuang, Y.; Song, C.; Wang, A.; Lin, F.; Li, Y.; Gu, C.; Li, C.; Xu, W. SleepSense: Non-invasive sleep event recognition using an electromagnetic probe. In Proceedings of the 2015 IEEE 12th International Conference on Wearable and Implantable Body Sensor Networks (BSN), Cambridge, MA, USA, 9–12 June 2015. [Google Scholar] [CrossRef]
- Amira, T.; Dan, I.; Az-eddine, B.; Ngo, H.H.; Said, G.; Katarzyna, W. Monitoring chronic disease at home using connected devices. In Proceedings of the 2018 13th Annual Conference on System of Systems Engineering (SoSE), Paris, France, 19–22 June 2018; pp. 400–407. [Google Scholar] [CrossRef]
- Cortinas, R.; Gonzaga, J.M.; Green, A.R.; Saulenas, A.M.; BuSha, B.F. TCNJ Athlete Tracker. In Proceedings of the 2015 41st Annual Northeast Biomedical Engineering Conference (NEBEC), Troy, NY, USA, 17–19 April 2015. [Google Scholar] [CrossRef]
- Hörmann, T.; Hesse, M.; Adams, M.; Rückert, U. A Software Assistant for User-Centric Calibration of a Wireless Body Sensor. In Proceedings of the 2016 IEEE 13th International Conference on Wearable and Implantable Body Sensor Networks (BSN), San Francisco, CA, USA, 14–17 June 2016; pp. 183–188. [Google Scholar] [CrossRef]
- Warmerdam, L.; Riper, H.; Klein, M.; van den Ven, P.; Rocha, A.; Ricardo Henriques, M.; Tousset, E.; Silva, H.; Andersson, G.; Cuijpers, P. Innovative ICT solutions to improve treatment outcomes for depression: the ICT4Depression project. Stud. Health Technol. Inform. 2012, 181, 339–343. [Google Scholar]
- Rajagopalan, R.; Litvan, I.; Jung, T.P. Fall Prediction and Prevention Systems: Recent Trends, Challenges, and Future Research Directions. Sensors 2017, 17, 2509. [Google Scholar] [CrossRef]
- Tedesco, S.; Barton, J.; O’Flynn, B. A review of activity trackers for senior citizens: Research perspectives, commercial landscape and the role of the insurance industry. Sensors 2017, 17, 1277. [Google Scholar] [CrossRef]
- Mehta, L.S.; Beckie, T.M.; DeVon, H.A.; Grines, C.L.; Krumholz, H.M.; Johnson, M.N.; Lindley, K.J.; Vaccarino, V.; Wang, T.Y.; Watson, K.E.; et al. Acute Myocardial Infarction in Women A Scientific Statement From the American Heart Association. Circulation 2016, 133, 916–947. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Fonarow, G.C.; Mukamal, K.J.; Liang, L.; Schulte, P.J.; Smith, E.E.; DeVore, A.; Hernandez, A.F.; Peterson, E.D.; Bhatt, D.L. Sex and Race/Ethnicity–Related Disparities in Care and Outcomes After Hospitalization for Coronary Artery Disease Among Older Adults. Circ. Cardiovasc. Qual. Outcomes 2016, 9, 36–44. [Google Scholar] [CrossRef] [PubMed]
- Regitz-Zagrosek, V. Sex and gender differences in health. Science & Society Series on Sex and Science. EMBO Rep. 2012, 13, 596–603. [Google Scholar] [CrossRef] [PubMed]
- Smulders, E.; van Lankveld, W.; Laan, R.; Duysens, J.; Weerdesteyn, V. Does osteoporosis predispose falls? a study on obstacle avoidance and balance confidence. BMC Musculoskelet. Disord. 2011, 12, 1. [Google Scholar] [CrossRef]
- Scheffer, A.; Schuurmans, M.; van Dijk, N.; van der Hooft, T.; de Rooij, S. Fear of falling: measurement strategy, prevalence, risk factors and consequences among older persons. Age Ageing 2008, 37, 19–24. [Google Scholar] [CrossRef]
- Delbaere, K.; Crombez, G.; Vanderstraeten, G.; Willems, T.; Cambier, D. Fear-related avoidance of activities, falls and physical frailty. A prospective community-based cohort study. Age Ageing 2004, 33, 368–373. [Google Scholar] [CrossRef]
- Arnold, D.; Busch, A.; Schachter, C.; Harrison, L.; Olsynski, W. The relationship of intrinsic fall risk factors to a recent history of falling in older women with osteoporosis. J. Orthop. Sports Phys. Ther. 2005, 35, 452–460. [Google Scholar] [CrossRef]
- Liu-Ambrose, T.; Khan, K.; Donaldson, M.; Eng, J.; Lord, S.; McKay, H. Falls-related self-efficacy is independently associated with balance and mobility in older women with low bone mass. J. Gerontol. Ser. A 2006, 51, 832–838. [Google Scholar] [CrossRef]
- A Patient’s Guide to Adult Kyphosis. Available online: https://www.umms.org/ummc/health-services/orthopedics/services/spine/patient-guides/adult-kyphosis (accessed on 16 January 2020).
| Database | Search Phrase | Number of Articles |
|---|---|---|
| Web of Science Core Collection | ALL FIELDS: ((“body sensor" or “wireless body sensor” or “wireless wearable technology” or “biomedical sensor” or “IoT”) and (“Ecare” or “mHealth” or “eHealth’) and (“Social impact” or “Compliance” or “Acceptance” or “Clinical trial” or ‘Pilot test” or ‘Human input” or “Feedback” or “Pilot application” or “Human in the loop”)) | 7 |
| Web of Science Core Collection | ALL FIELDS:((“body sensor” or “wireless body sensor” or “wireless wearable technology” or “biomedical sensor" or “IoT”) and (“care" or “Health”) and (“Social impact” or “Compliance” or “Acceptance” or “Clinical trial" or “Pilot test” or “Human input” or “Feedback” or “Pilot application" or “Human in the loop”)) | 142 |
| MEDLINE (Web of Science) | TOPIC: ((((((“body sensor”) OR “wireless body sensor”) OR “wireless wearable technology”) OR “biomedical sensor”) OR “IoT”) AND (“care”) OR “Health”)) AND ((((((((“Social impact”) OR “Compliance”) OR “Acceptance”) OR “Clinical trial”) OR “Pilot test”) OR “Human input”) OR “Feedback”) OR “Pilot application") OR “Human in the loop")) Timespan: All years. Indexes: MEDLINE. | 25 |
| Scopus | ALL(body sensor OR wireless body sensor OR wireless wearable technology OR biomedical sensor) AND (ecare OR mhealth OR ehealth) AND ( Social impact OR compliance OR acceptance OR Clinical trial OR Pilot test ) Limiting to English | 187 |
| ScienceDirect | Title, abstract, keywords: “wearable sensors” and health and impact. Limited to review articles, research articles, conference abstracts, case reports. | 13 |
| ScienceDirect | Title, abstract, keywords: “body sensor” and health and impact. Limited to review articles, research articles, conference abstracts, case reports. | 5 |
| Academic Search Elite | Free text search: “body sensor” and health and impact English. | 8 |
| Academic Search Elite | Free text search: “body sensor” and health and acceptance | 3 |
| ACM Digital Library | (+“body sensor” +and +health +and +impact) | 12 |
| IEEE Xplore | “body sensor” and health and impact | 81 |
| IEEE Xplore | “body sensor” and health and trial | 12 |
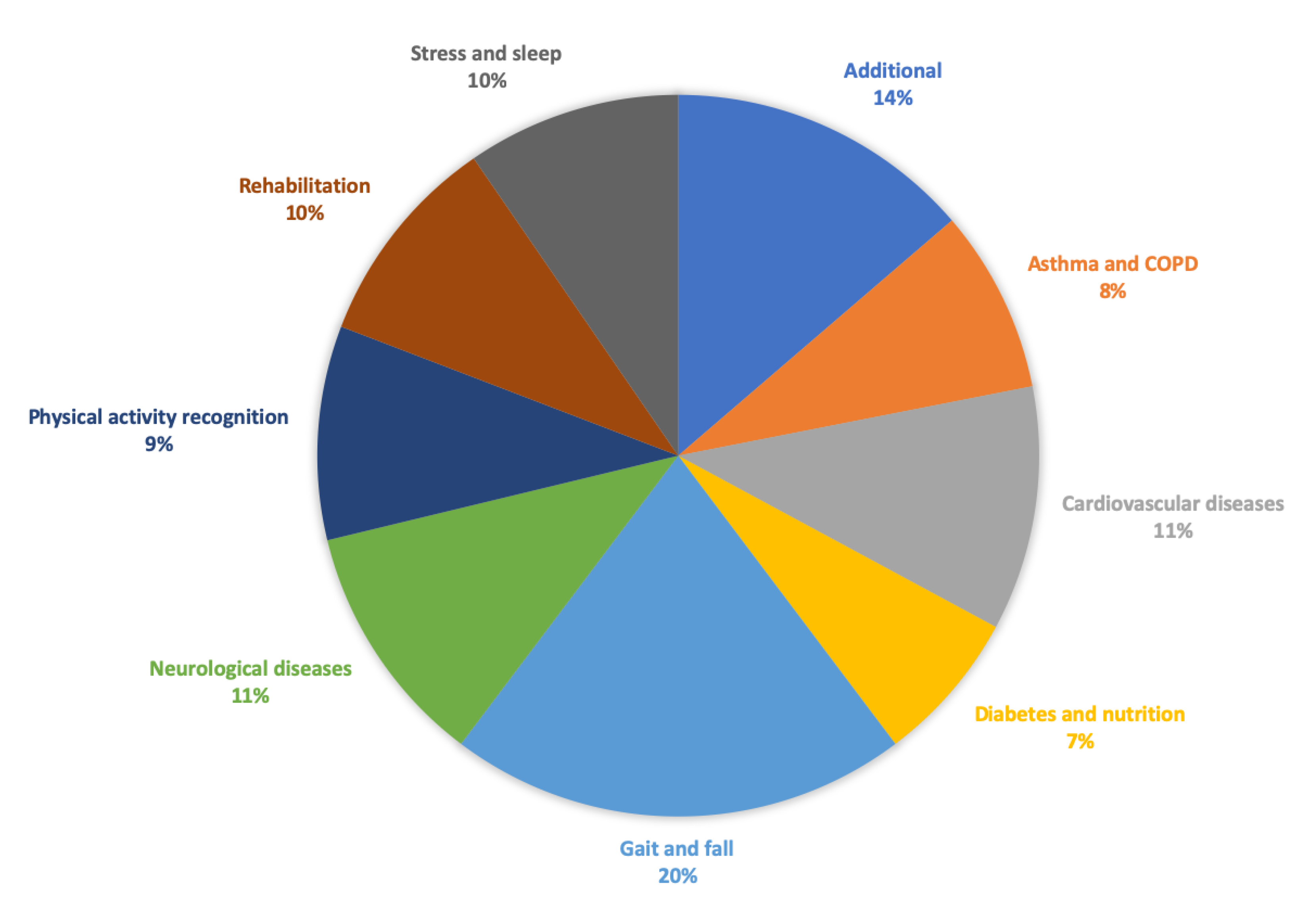
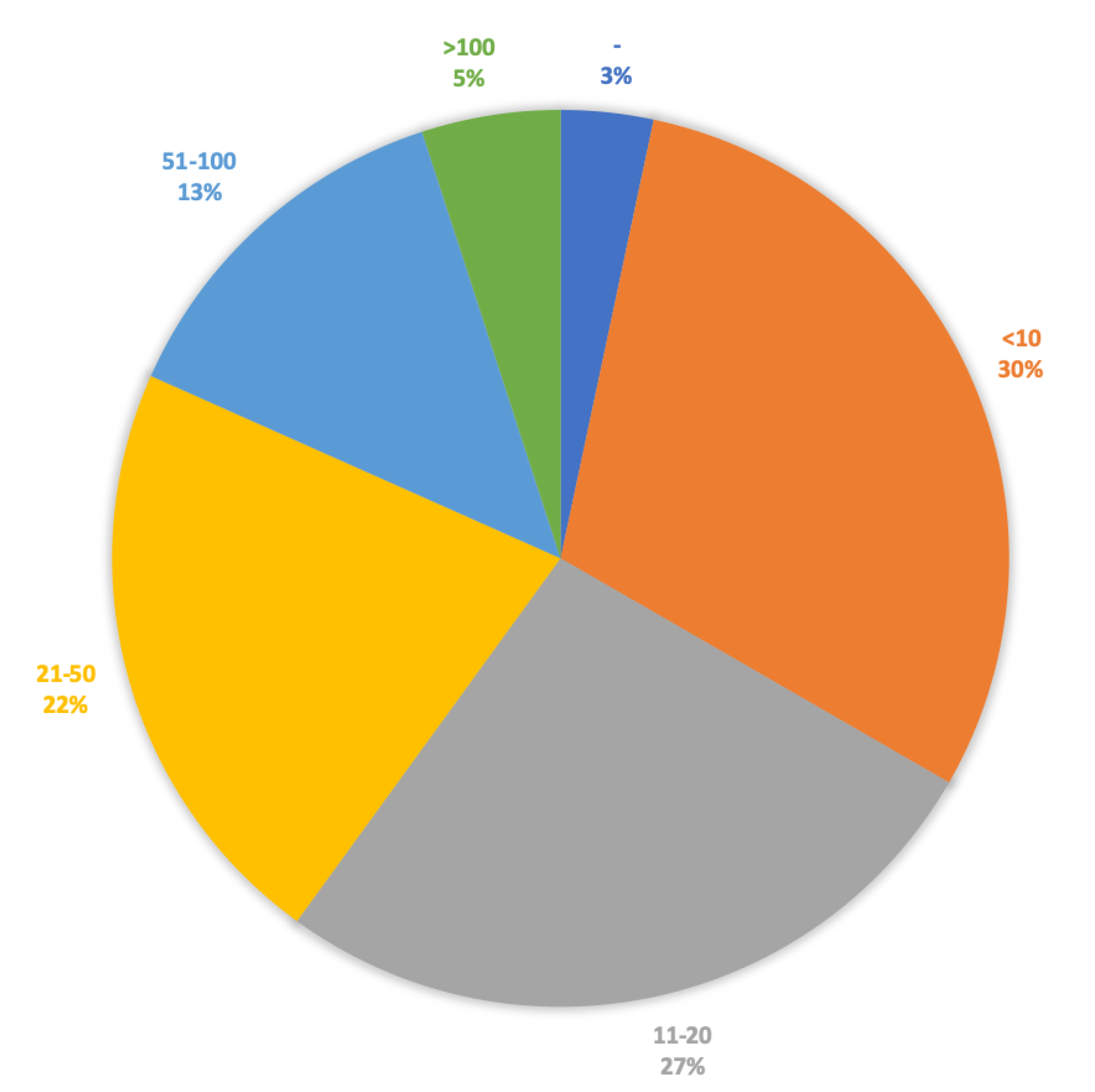
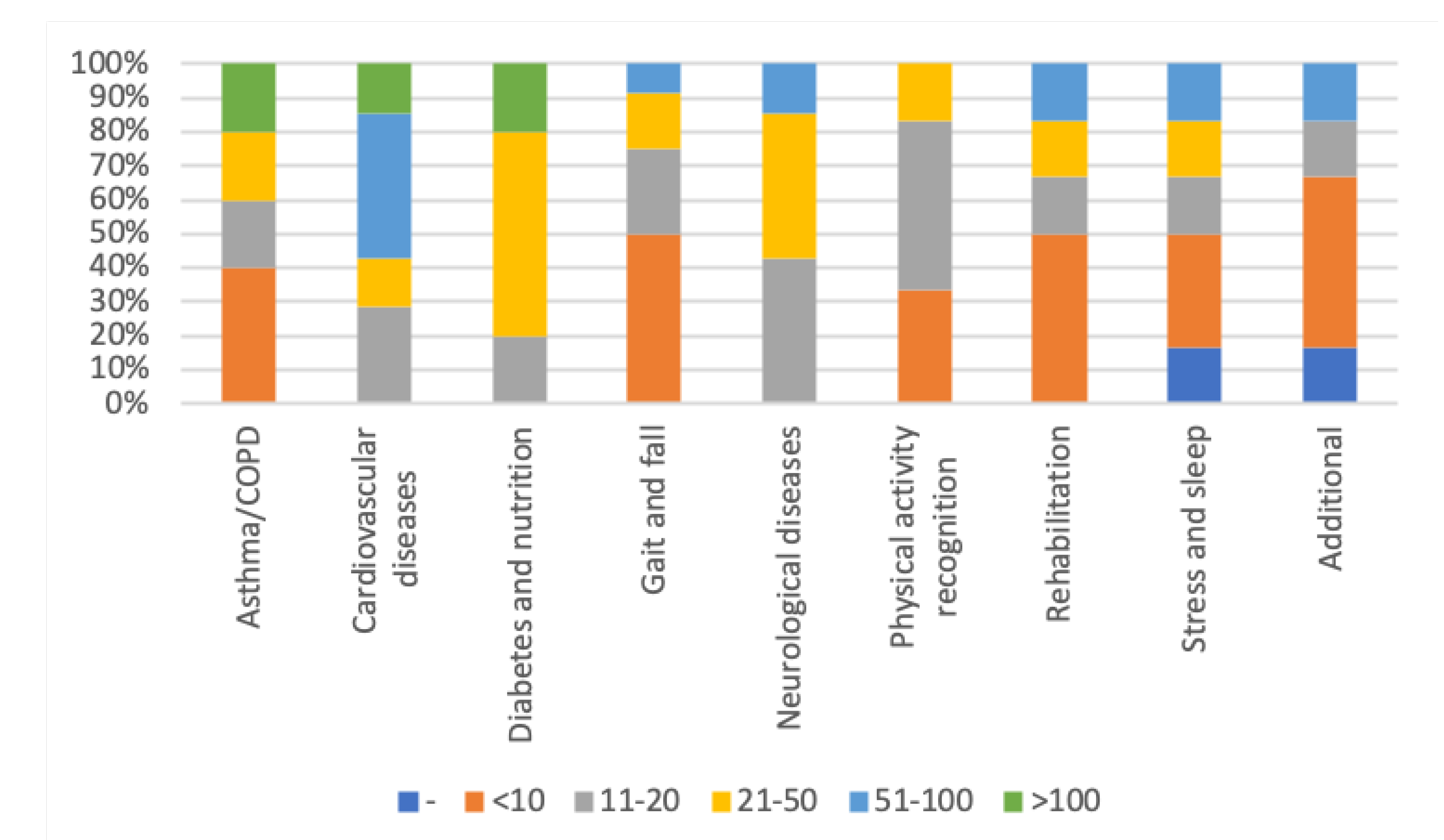
| Author, Year | Ref. | Article Category | Research Design | No. of Participants | Sensor Category |
|---|---|---|---|---|---|
| Bonnevie et al. 2019 | [13] | Asthma/COPD | Observational | 104 | Vital signs |
| 5 | |||||
| Caulfield et al. 2014 | [14] | Asthma/COPD | Observational | 10 | Physical activity |
| Estrada et al. 2016 | [15] | Asthma/COPD | Observational | 1 | Other |
| Katsaras et al. 2011 | [16] | Asthma/COPD | Randomized control | 48 | Other |
| Naranjo-Hernández et al. 2018 | [17] | Asthma/COPD | Observational | 2 | Vital signs |
| 9 | |||||
| Huang et al. 2014a | [18] | Cardiovascular diseases | - | 225 | ECG |
| Huang et al. 2014b | [19] | Cardiovascular diseases | Case-control | 84 | ECG |
| Javaid et al. 2018 | [20] | Cardiovascular diseases | Observational | 60 | Other |
| Li et al. 2019 | [3] | Cardiovascular diseases | Observational | 16 | Other |
| Raad et al. 2015 | [21] | Cardiovascular diseases | - | 30 | ECG |
| - | 2 | ||||
| Simjanoska et al. 2018 | [22] | Cardiovascular diseases | Observational | 16 | ECG |
| 3 | |||||
| 25 | |||||
| 7 | Dataset ECG | ||||
| Susič and Stanič 2016 | [23] | Cardiovascular diseases | - | 13 | ECG |
| Al-Taee et al. 2015 | [24] | Diabetes and nutrition | - | 22 | Other |
| Alshurafa et al. 2014 and Alshurafa et al. 2015 | [25,26] | Diabetes and nutrition | Observational | 10 | Other |
| 20 | |||||
| Dong and Biswas 2017 | [27] | Diabetes and nutrition | Observational | 14 | Other |
| Onoue et al. 2017 | [28] | Diabetes and nutrition | Randomized control | 101 | Physical activity |
| Atallah 2012 | [29] | Gait and fall | Observational | 34 | Physical activity |
| Godfrey et al. 2014 | [30] | Gait and fall | Observational | 24 | Physical activity |
| Lee et al. 2015 | [31] | Gait and fall | Observational | 11 | Physical activity |
| Liang et al. 2012 | [32] | Gait and fall | Observational | 8 | Physical activity |
| Liang et al. 2018 | [33] | Gait and fall | Observational | 18 | Physical activity |
| Paiman et al. 2016 | [34] | Gait and fall | Observational | 2 | Other |
| Tino et al. 2011 | [35] | Gait and fall | Observational | 3 | Other |
| Williams et al. 2015 | [36] | Gait and fall | Observational | 5–6 | Physical activity |
| Wu et al. 2013 | [4] | Gait and fall | Observational | 7 | Physical activity |
| Wu et al. 2019 | [37] | Gait and fall | Observational | 15 | Physical activity |
| Zhao et al. 2012 | [38] | Gait and fall | Observational | 8 | Physical activity |
| Zhong et al. 2019 | [39] | Gait and fall | Observational | 56 | Physical activity |
| Giuberti et al. 2015 | [40] | Neurological diseases | Observational | 24 | Physical activity |
| Gong et al. 2015, Gong et al. 2016 | [41,42] | Neurological diseases | Case-control | 41 | Physical activity |
| Kuusik et al. 2018 | [43] | Neurological diseases | Observational | 51 | Physical activity |
| Sok et al. 2018 | [44] | Neurological diseases | Observational | 13 | Physical activity |
| Stamate et al. 2017 and Stamate et al. 2018 | [45,46] | Neurological diseases | Observational | 12 | Other |
| Castro et al. 2017 and Rodriguez et al. 2017 | [5,6] | Physical activity recognition | Observational | 3 | Other |
| Doron et al. 2013 | [7] | Physical activity recognition | Observational | 65 | Other |
| 20 | |||||
| Rednic et al. 2012 | [47] | Physical activity recognition | Observational | 17 | Physical activity |
| Xu et al. 2014 | [8] | Physical activity recognition | Observational | 14 | Other |
| Xu et al. 2016 | [9] | Physical activity recognition | Observational | 4 | Other |
| 3 | Physical activity | ||||
| 5 | |||||
| 6 | |||||
| Argent et al. 2019 | [48] | Rehabilitation | Observational | 15 | Physical activity |
| Banos et al. 2015 | [49] | Rehabilitation | Observational | 10 | Other |
| Lee et al. 2018 | [50] | Rehabilitation | Case-control | 30 | Physical activity |
| Timmermans et al. 2010 | [51] | Rehabilitation | Observational | 9 | Physical activity |
| Whelan et al. 2017 | [52] | Rehabilitation | Observational | 55 | Physical activity |
| Xu et al. 2017 | [53] | Rehabilitation | Observational | 6 | Other |
| Lin et al. 2012 | [54] | Stress and sleep | Case-control | 18 (6/12) | Physical activity |
| Nakamura et al. 2017 | [55] | Stress and sleep | Observational | 4 | Other |
| Parnandi and Gutierrez-Osuna 2017 | [56] | Stress and sleep | Randomized control | 25 | Other |
| Uday et al. 2018 | [57] | Stress and sleep | Observational | 10 | Other |
| Umemura et al. 2017 | [58] | Stress and sleep | Case-control | 54 | Other |
| Velicu et al. 2016 | [10] | Stress and sleep | Observational | - | - |
| Ayzenberg and Picard 2014 | [59] | Additional | Crossover | 10 | Other |
| Pagán et al. 2016 | [60] | Additional | Observational | 2 | Other |
| Rawasdeh et al. 2017 | [61] | Additional | Observational | 55 | ECG |
| Seeger et al. 2012 | [11] | Additional | - | - | Other |
| Wannenburg and Malekian 2015 | [12] | Additional | Observational | 4–8 | Vital signs |
| Wu et al. 2018 | [62] | Additional | Observational | 20 | ECG |
| Ref. | Article Category | No. of Participants | Age Group | Age Statistics | Male | Female | Patient | Healthy |
|---|---|---|---|---|---|---|---|---|
| [13] | Asthma/COPD | 104 | 57–70 | 64 | 67 (64%) | 37 (36%) | 104 | |
| 5 | 50–66 | 62 | - | - | 5 | |||
| [14] | Asthma/COPD | 10 | 61.5 ± 5.7 | 5 | 5 | 10 | ||
| [15] | Asthma/COPD | 1 | - | - | 1 | 1 | ||
| [16] | Asthma/COPD | 48 | - | - | 48 | 48 | ||
| [17] | Asthma/COPD | 2 | 36 and 42 | 2 | 2 | |||
| 9 | 55–76 | 64 | 6 | 3 | 9 | |||
| [18] | Cardiovascular diseases | 225 | - | - | - | - | 225 | |
| [19] | Cardiovascular diseases | 84 | - | - | - | - | 1 group | 1 group |
| [20] | Cardiovascular diseases | 60 | - | 26.9 | 28 | 32 | 60 | |
| [3] | Cardiovascular diseases | 16 | - | - | - | - | - | - |
| [21] | Cardiovascular diseases | 30 | 20–23 | - | - | - | - | |
| 2 | - | - | - | - | 2 | |||
| [22] | Cardiovascular diseases | 16 | 16–72 | - | - | - | - | - |
| 3 | 25–27 | - | - | - | - | - | ||
| 25 | 20–73 | - | - | - | 14 | 11 | ||
| 7 | 20–74 | - | - | - | 7 | |||
| [23] | Cardiovascular diseases | 13 | - | 50.6 | 8 | 5 | 13 | |
| [24] | Diabetes and nutrition | 22 | - | - | - | - | 22 | |
| [25,26] | Diabetes and nutrition | 10 | 20–40 | 8 | 2 | - | - | |
| 20 | 20–40 | 12 | 8 | - | - | |||
| [27] | Diabetes and nutrition | 14 | - | - | 9 | 5 | 14 | |
| [28] | Diabetes and nutrition | 101 | - | 57.1 | 56 | 45 | 101 | |
| [29] | Gait and fall | 34 | - | 28.22 | 21 | 13 | 34 | |
| [30] | Gait and fall | 24 (12/12) | 20–40 | 32.5 | 7 | 5 | 12 | |
| 65.0 | 5 | 7 | 12 | |||||
| [31] | Gait and fall | 11 | - | 27.6 | 11 | 11 | ||
| [32] | Gait and fall | 8 | - | 23 | 8 | 8 | ||
| [33] | Gait and fall | 18 | - | 25 | 12 | 6 | 18 | |
| [34] | Gait and fall | 2 | 28 and 24 | - | 1 | 1 | 2 | |
| [35] | Gait and fall | 3 | 40–70 | - | - | - | - | - |
| [36] | Gait and fall | 5–6 (1/5) | 27 | - | 1 | - | - | |
| 21–36 | 27 | 4 | 1 | - | - | |||
| [4] | Gait and fall | 7 | - | - | - | - | - | - |
| [37] | Gait and fall | 15 | 20–27 | - | - | - | 15 | |
| [38] | Gait and fall | 8 | - | 28.5 | - | - | 8 | |
| [39] | Gait and fall | 56 (28/28) | - | 24.6 | 14 | 14 | 28 | |
| >55 | 66.1 | 18 | 10 | 28 | ||||
| [40] | Neurological diseases | 24 | 31–79 | 65.9 | 17 | 7 | 24 | |
| [41,42] | Neurological diseases | 41 (28/13) | - | 40.5 | 25% | 25% | 28 | 13 |
| - | 39.3 | 47% | 53% | |||||
| [43] | Neurological diseases | 51 | - | - | - | - | 51 | |
| [44] | Neurological diseases | 13 | 22–50 | - | 9 | 4 | 13 | |
| [45,46] | Neurological diseases | 12 | - | - | - | - | 12 | |
| [5,6] | Physical activity recognition | 3 | - | - | - | - | - | - |
| [7] | Physical activity recognition | 65 | - | - | - | - | - | - |
| 20 | - | - | - | - | - | - | ||
| [47] | Physical activity recognition | 17 | - | - | 10 | 7 | - | - |
| [8] | Physical activity recognition | 14 | - | - | - | - | - | - |
| [9] | Physical activity recognition | 4 | - | - | - | - | - | - |
| 3 | - | - | - | - | 3 | |||
| 5 | - | - | - | - | 5 | |||
| 6 | - | - | 3 | 3 | - | - | ||
| [48] | Rehabilitation | 15 | - | 63 | 6 | 9 | 15 | |
| [49] | Rehabilitation | 10 | 21–37 | - | 8 | 2 | - | - |
| [50] | Rehabilitation | 20 | - | 54.4 | - | - | 20 | |
| 10 | 53.8 | - | - | 10 | ||||
| [51] | Rehabilitation | 9 | - | 60.7 | 5 | 4 | 9 | |
| [52] | Rehabilitation | 55 | - | 24.21 | 37 | 18 | 55 | |
| [53] | Rehabilitation | 6 | - | 72.5 | 3 | 3 | - | - |
| [54] | Stress and sleep | 18 (6/12) | 19–22 overall | - | 5 | 1 | - | - |
| 11 | 1 | - | - | |||||
| [55] | Stress and sleep | 4 | 25–36 | - | 4 | 4 | ||
| [56] | Stress and sleep | 25 | 19–33 | - | 15 | 10 | - | - |
| [57] | Stress and sleep | 10 | - | - | - | - | 10 | |
| [58] | Stress and sleep | 54 (26/28) | - | 22 | - | - | 54 | |
| - | 21 | - | - | - | - | |||
| [10] | Stress and sleep | - | - | - | - | - | - | - |
| [59] | Additional | 10 | 25–35 | 30.8 | 9 | 1 | 10 | |
| [60] | Additional | 2 | - | - | 2 | 2 | ||
| [61] | Additional | 55 | 18–22 | - | 50% | 50% | - | - |
| [11] | Additional | - | - | - | - | - | - | - |
| [12] | Additional | 4–8 (4/4) | - | - | - | - | - | - |
| - | - | - | - | - | - | |||
| [62] | Additional | 20 | - | - | - | - | 20 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kristoffersson, A.; Lindén, M. A Systematic Review on the Use of Wearable Body Sensors for Health Monitoring: A Qualitative Synthesis. Sensors 2020, 20, 1502. https://doi.org/10.3390/s20051502
Kristoffersson A, Lindén M. A Systematic Review on the Use of Wearable Body Sensors for Health Monitoring: A Qualitative Synthesis. Sensors. 2020; 20(5):1502. https://doi.org/10.3390/s20051502
Chicago/Turabian StyleKristoffersson, Annica, and Maria Lindén. 2020. "A Systematic Review on the Use of Wearable Body Sensors for Health Monitoring: A Qualitative Synthesis" Sensors 20, no. 5: 1502. https://doi.org/10.3390/s20051502
APA StyleKristoffersson, A., & Lindén, M. (2020). A Systematic Review on the Use of Wearable Body Sensors for Health Monitoring: A Qualitative Synthesis. Sensors, 20(5), 1502. https://doi.org/10.3390/s20051502






