Monitoring Plant Status and Fertilization Strategy through Multispectral Images
Abstract
1. Introduction
2. Materials and Methods
2.1. Location and Growing Conditions
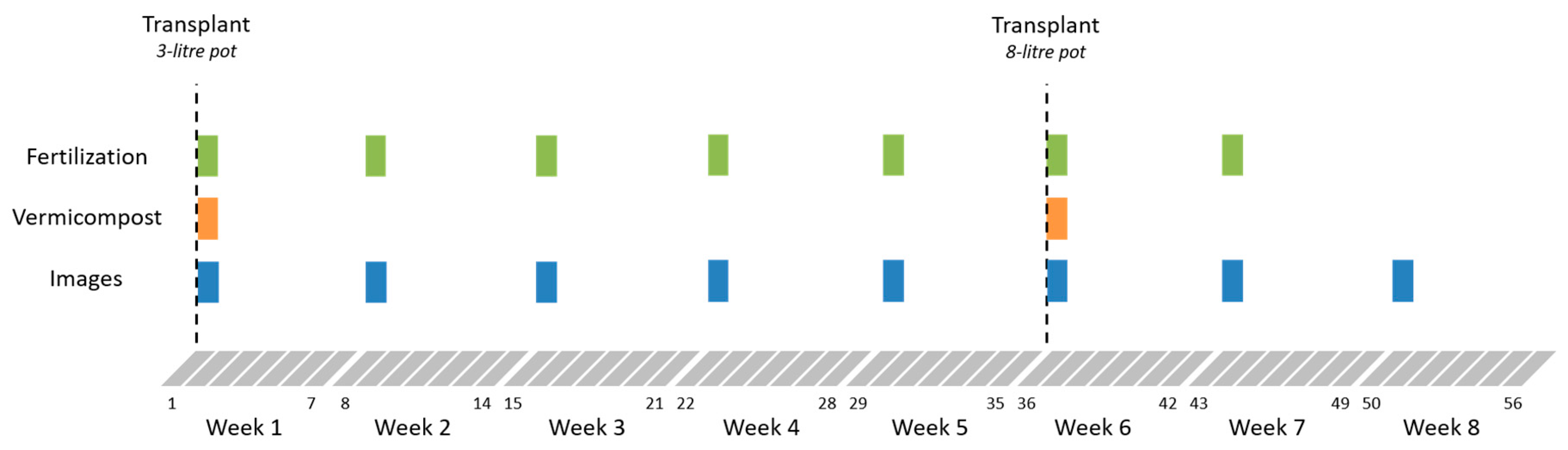
2.2. Trial Design and Fertilization
2.3. System Overview
2.4. Computational System for Image Analysis
2.4.1. Image Pre-Processing
2.4.2. Vegetation Indices
2.4.3. Plant Extraction (Segmentation)
2.4.4. Morphological Analysis
2.4.5. Statistical Analysis
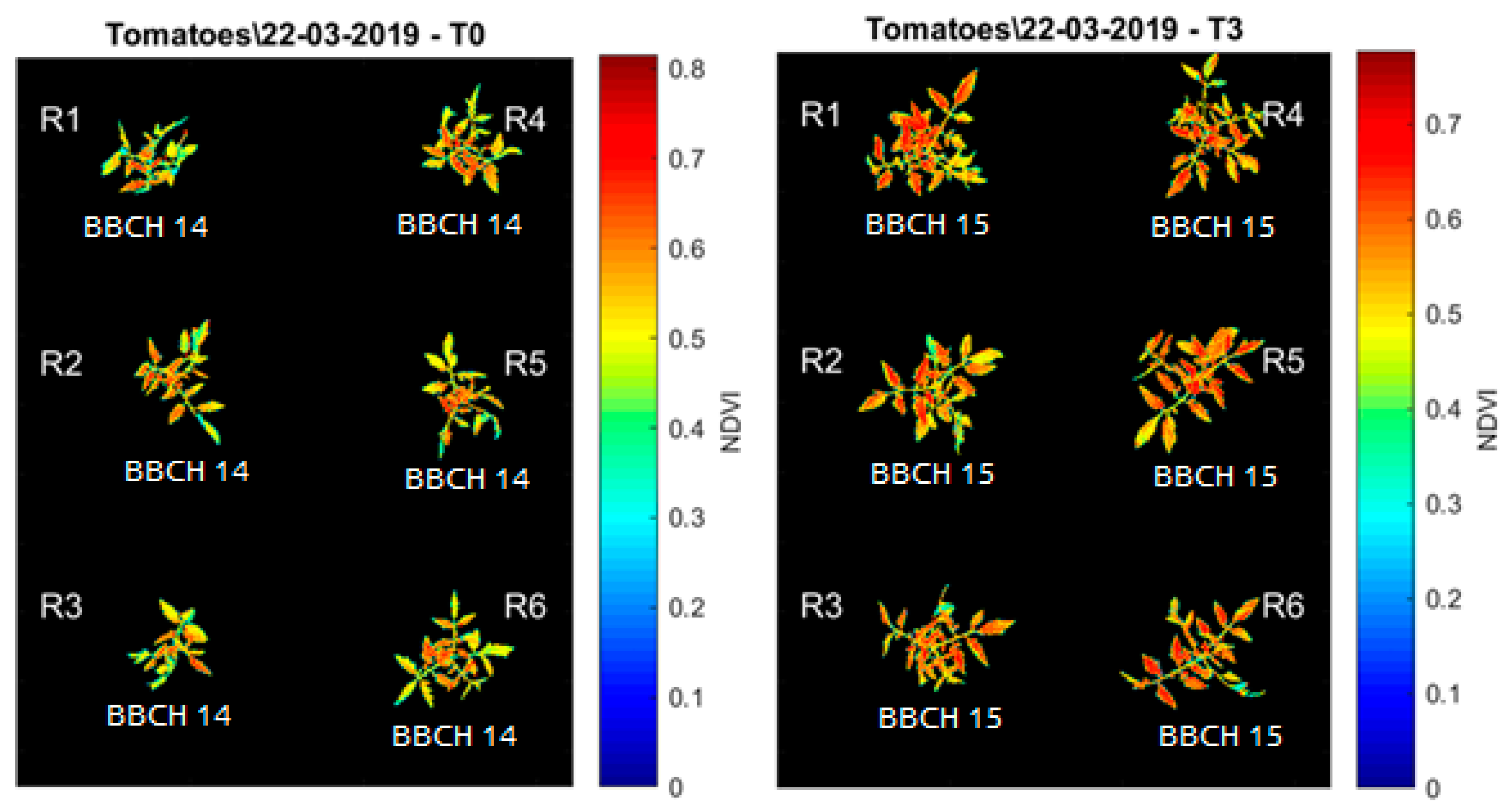
3. Results and Discussion
Discussion
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hidalgo-Baz, M.; Martos-Partal, M.; González-Benito, O. Attitudes vs. purchase behaviors as experienced dissonance: The roles of knowledge and consumer orientations in organic market. Front. Psychol. 2017, 8, 248. [Google Scholar] [CrossRef]
- Pino, G.; Peluso, A.M.; Guido, G. Determinants of regular and occasional consumers’ intentions to buy organic food. J. Consum. Aff. 2012, 46, 157–169. [Google Scholar] [CrossRef]
- Ribeiro, A.C.; Guimarães, P.T.G.; Alvarez, V.V.H. Recomendações Para o Uso de Corretivos e Fertilizantes em Minas Gerais—5° Aproximação, 1st ed.; Universidade Federal de Viçosa: Viçosa, Brazil, 1999; p. 359. [Google Scholar]
- Agrios, G.N. Introduction to Plant Pathology; Elsevier Academic Press Publication: Amsterdam, The Netherlands, 2005; pp. 257–262. [Google Scholar]
- Brown, J.F.; Ogle, H.J. Plant Pathogens and Plant Diseases; Rockvale Publications for the Division of Botany: Armidale, Australia, 1997; pp. 156–157. [Google Scholar]
- Wang, Y.; Ying, H.; Yin, Y.; Zheng, H.; Cui, Z. Estimating soil nitrate leaching of nitrogen fertilizer from global meta-analysis. Sci. Total Environ. 2019, 657, 96–102. [Google Scholar] [CrossRef]
- Du, Y.D.; Niu, W.Q.; Gu, X.B.; Zhang, Q.; Cui, B.J. Water-and nitrogen-saving potentials in tomato production: A meta-analysis. Agric. Water Manag. 2018, 210, 296–303. [Google Scholar] [CrossRef]
- Pedersen, S.M.; Lind, K.M. Precision Agriculture–From Mapping to Site-Specific Application. In Precision Agriculture: Technology and Economic Perspectives; Springer: Cham, Switzerland, 2017; p. 25. [Google Scholar]
- Chen, C.; Pan, J.; Lam, S.K. A review of precision fertilization research. Environ. Earth Sci. 2014, 71, 4073–4080. [Google Scholar] [CrossRef]
- Candiago, S.; Remondino, F.; De Giglio, M.; Dubbini, M.; Gatteli, M. Evaluating multispectral images and vegetation indices for precision farming applications from UAV images. Remote Sens. 2015, 7, 4026–4047. [Google Scholar] [CrossRef]
- Pallottino, F.; Antonucci, F.; Costa, C.; Bisaglia, C.; Figorilli, S.; Menesatti, P. Optoelectronic proximal sensing vehicle-mounted technologies in precision agriculture: A review. Comput. Electron. Agric. 2019, 162, 859–873. [Google Scholar] [CrossRef]
- Prey, L.; Von Bloh, M.; Scgumidhalter, U. Evaluating RGB imaging and multispectral active and hyperspectral passive sensing for assessing early plant vigor in winter wheat. Sensors 2018, 18, 2931. [Google Scholar] [CrossRef]
- Kitic, G.; Tagarakis, A.; Cselyuszka, N.; Panic, M.; Birgermajer, S.; Sakulski, D.; Matovik, J. A new low-cost portable multispectral optical device for precise plant status assessment. Comput. Electron. Agric. 2019, 162, 300–308. [Google Scholar] [CrossRef]
- Freidenreich, A.; Barraza, G.; Jayachandran, K.; Khoddamzadeg, A.A. Precision Agriculture Application for Sustainable Nitrogen Management of Justicia brandegeana Using Optical Sensor Technology. Agriculture 2019, 9, 98. [Google Scholar] [CrossRef]
- Liu, L.; Song, B.; Zhang, S.; Liu, X. A Novel Principal Component Analysis Method for the Reconstruction of Leaf Reflectance Spectra and Retrieval of Leaf Biochemical Contents. Remote Sens. 2017, 9, 1–23. [Google Scholar] [CrossRef]
- Osco, L.P.; de Arruda, M.D.S.; Junior, J.M.; da Silva, N.B.; Ramos, A.P.M.; Moryia, É.A.S.; NobuhiroImai, N.; RobertoPereira, D.; EduardoCreste, J.; TakashiMatsubara, E.; et al. A convolutional neural network approach for counting and geolocating citrus-trees in UAV multispectral imagery. ISPRS J. Photogramm. Remote Sens. 2020, 160, 97–106. [Google Scholar] [CrossRef]
- Wallis, C.I.; Homeier, J.; Peña, J.; Brandl, R.; Farwig, N.; Bendix, J. Modeling tropical montane forest biomass; productivity and canopy traits with multispectral remote sensing data. Remote Sens. Environ. 2019, 225, 77–92. [Google Scholar] [CrossRef]
- Zeng, L.; Wardlow, B.D.; Xiang, D.; Hu, S.; Li, D.A. Review of vegetation phenological metrics extraction using time-series, multispectral satellite data. Remote Sens. Environ. 2020, 237, 1–20. [Google Scholar] [CrossRef]
- Meddens, A.J.; Hicke, J.A.; Vierling, L.A. Evaluating the potential of multispectral imagery to map multiple stages of tree mortality. Remote Sens. Environ. 2011, 115, 1632–1642. [Google Scholar] [CrossRef]
- Huang, Y.; Lan, Y.; Hoffmann, W.C. Use of airborne multi-spectral imagery in pest management systems. Agricultural Engineering International: CIGR J. 2008, 10, 1–14. [Google Scholar]
- West, H.; Quinn, N.; Horswell, M. Remote sensing for drought monitoring & impact assessment: Progress, past challenges and future opportunities. Remote Sens. Environ. 2019, 232, 1–14. [Google Scholar]
- Lu, R. Multispectral imaging for predicting firmness and soluble solids content of apple fruit. Postharvest Biol. Technol. 2004, 31, 47–157. [Google Scholar] [CrossRef]
- Lleó, L.; Barreiro, P.; Ruiz-Altisent, M.; Herrero, A. Multispectral images of peach related to firmness and maturity at harvest. J. Food Eng. 2009, 93, 229–235. [Google Scholar] [CrossRef]
- Gerhards, R.; Oebel, H. Practical experiences with a system for site-specific weed control in arable crops using real-time image analysis and GPS-controlled patch spraying. Weed Res. 2006, 46, 185–193. [Google Scholar] [CrossRef]
- Christensen, S.; Søgaard, H.T.; Kudsk, P.; Nørrenmark, M.; Lund, I.; Nadimi, E.S.; Jørgensen, R. Site-specific weed control technologies. Weed Res. 2009, 49, 233–241. [Google Scholar] [CrossRef]
- Ávila-Navarro, J.; Franco, C.A.; Rasmussen, J.; Nielsen, J. Color classification methods for perennial weed detection in cereal crops. In Progress in Pattern Recognition, Image Analysis, Computer Vision, and Applications, Proceedings of the 23rd Iberoamerican Congress, CIARP 2018, Madrid, Spain, 19–22 November 2018; Springer: Cham, Switzerland, 2019; pp. 117–123. [Google Scholar]
- Partel, V.; Kakarla, S.C.; Ampatzidis, Y. Development and evaluation of a low-cost and smart technology for precision weed management utilizing artificial intelligence. Comput. Electron. Agric. 2019, 157, 339–350. [Google Scholar] [CrossRef]
- Ampatzidis, Y.; De Bellis, L.; Luvisi, A. iPathology: Robotic applications and management of plants and plant diseases. Sustainability 2017, 9, 1010–1024. [Google Scholar] [CrossRef]
- Cavender-Bares, K.; Bares, C. Robotic Platform and Method for Performing Multiple Functions in Agricultural Systems. U.S. Patent Application n. 16/188,422, 14 March 2019. [Google Scholar]
- Veys, C.; Chatziavgerinos, F.; AlSuwaidi, A.; Hibbert, J.; Hansen, M.; Bernotas, G.; Smith, M.; Yin, H.; Rolfe, S.; Grieve, B. Multispectral imaging for presymptomatic analysis of light leaf spot in oilseed rape. Plant Methods 2019, 15, 4. [Google Scholar] [CrossRef]
- Grimstad, L.; From, P. The Thorvald II agricultural robotic system. Robotics 2017, 6, 24–38. [Google Scholar] [CrossRef]
- Shamshiri, R.R.; Weltzien, C.; Hameed, I.A.; Yule, I.J.; Grift, T.E.; Balasundram, S.K.; Pitonakova, L.; Ahmad, D.; Chowdhary, G. Research and development in agricultural robotics: A perspective of digital farming. Int. J. Agric. Biol. Eng. 2018, 11, 1–14. [Google Scholar] [CrossRef]
- King, A. The future of agriculture. Nature 2017, 544, S21–S23. [Google Scholar] [CrossRef]
- Xue, J.; Fan, Y.; Su, B.; Fuentes, S. Assessment of canopy vigor ınformation from kiwifruit plants based on a digital surface model from unmanned aerial vehicle ımagery. Int. J. Agric. Biol. Eng. 2019, 12, 165–171. [Google Scholar] [CrossRef]
- Xue, J.; Su, B. Significant Remote Sensing Vegetation Indices: A Review of Developments and Applications. J. Sens. 2017, 2017, 1–17. [Google Scholar] [CrossRef]
- Moriarty, C.; Cowley, D.C.; Wade, T.; Nichol, C.J. Deploying multispectral remote sensing for multi-temporal analysis of archaeological crop stress at Ravenshall, Fife, Scotland. Archaeol. Prospect. 2019, 26, 33–46. [Google Scholar] [CrossRef]
- Fahlgren, N.; Gehan, M.A.; Baxter, I. Lights, camera, action: High-throughput plant phenotyping is ready for a close-up. Curr. Opin. Plant Biol. 2015, 24, 93–99. [Google Scholar] [CrossRef]
- Berry, J.C.; Fahlgren, N.; Pokorny, A.A.; Bart, R.S.; Veey, K.M. An automated, high-throughput method for standardizing image color profiles to improve image-based plant phenotyping. PeerJ 2018, 6, e5727. [Google Scholar] [CrossRef]
- Yogamalagan, R.; Karthikeyan, B. Segmentation techniques comparison in image processing. Int. J. Eng. Technol. 2013, 5, 307–313. [Google Scholar]
- Soille, P. Morphological image analysis applied to crop field mapping. Image Vis. Comput. 2010, 18, 1025–1032. [Google Scholar] [CrossRef]
- Van Eysinga, J.R.; Smilde, K.W. Nutritional Disorders in Glasshouse Tomatoes, Cucumbers and Lettuce; Centre for Agricultural Publishing and Documentation: Wageningen, The Netherlands, 1981. [Google Scholar]
- Veley, K.; Berry, J.; Fentress, S.; Schachtman, D.; Baxter, I.; Bart, R. High-throughput profiling and analysis of plant responses over time to abiotic stress. Plant Direct 2017, 1, e00023. [Google Scholar] [CrossRef]
- Liang, Z.; Pandey, P.; Stoerger, V.; Xu, Y.; Qiu, Y.; Ge, Y.; Schnable, C. Conventional and hyperspectral time-series imaging of maize lines widely used in field trials. Gigascience 2018, 7, 1–11. [Google Scholar] [CrossRef]
- Tariq, M.; Siddiqi, A.; Narejo, G.B.; Andleeb, S. A Cross Sectional Study of Tumors Using Bio-medical Imaging Modalities. Curr. Med. Imaging Rev. 2019, 15, 66–73. [Google Scholar] [CrossRef]
- Jiang, J. Design of Meshing Assembly Algorithms for Industrial Gears Based on Image Recognition. In International Conference on Advanced Information Systems Engineering; Springer: Cham, Switzerland, 2019; pp. 64–72. [Google Scholar]
- Singh, K.; Kumar, S.; Kaur, P. Automatic detection of rust disease of Lentil by machine learning system using microscopic images. Int. J. Electr. Comput. Eng. (IJECE) 2019, 9, 660–666. [Google Scholar] [CrossRef]
- Hsieh, H.; Chung, C.; Huang, C. Using the NDVI and mean shift segmentation to extract landslide areas in the Lioukuei Experimental Forest region with multi-temporal FORMOSAT-2 images. Taiwan J. For. Sci. 2017, 32, 203–222. [Google Scholar]
- Bhandari, A.K.; Kumar, A.; Singh, K. Feature Extraction using Normalized Difference Vegetation Index (NDVI): A Case Study of Jabalpur City. Procedia Technol. 2012, 6, 612–621. [Google Scholar] [CrossRef]
- Manakos, I.; Braun, M. Land Use and Land Cover Mapping in Europe: Practices & Trends, Remote Sensing and Digital Image Processing 18; Springer Science + Business Media: Berlin/Heidelberg, Germany, 2014; pp. 363–381. [Google Scholar]
- Padilla, F.M.; Peña-Fleitas, M.T.; Gallardo, M.; Thompson, R.B. Threshold values of canopy reflectance indices and chlorophyll meter readings for optimal nitrogen nutrition of tomato. Ann. Appl. Biol. 2015, 166, 271–285. [Google Scholar] [CrossRef]
- Fortes, R.; Prieto, M.H.; García-Martin, A.; Córdoba, A.; Martínez, L.; Campillo, C. Using NDVI and guided sampling to develop yield prediction maps of processing tomato crop. Span. J. Agric. Res. 2015, 16, 1–9. [Google Scholar] [CrossRef]
- Oliveira, T.F.; Pinto, F.A.C.; Silva, D.J.H. Spectral Vegetation Indices applied to Nitrogen Sufficiency Index: A Strategy with Potential to Increase Nitrogen Use Efficiency in Tomato Crop. Eng. Agrícola 2019, 39, 118–126. [Google Scholar] [CrossRef]
- Gianquinto, G.; Orsini, F.; Fecondini, M.; Mezzetti, M.; Sambo, P.; Bona, S. A methodological approach for defining spectral indices for assessing tomato nitrogen status and yield. Eur. J. Agron. 2011, 35, 135–143. [Google Scholar] [CrossRef]
- Kipp, S.; Mistele, B.; Schimidhalter, U. The performance of active spectral reflectance sensor as influenced by measuring distance, device temperature and light intensity. Comput. Electron. Agric. 2014, 100, 24–33. [Google Scholar] [CrossRef]
- Mooy, L.M.; Hasan, A.; Onsili, R. Growth and yield of Tomato (Lycopersicum esculentum Mill.) as influenced by the combination of liquid organic fertilizer concentration and branch pruning. IOP Conf. Ser. Earth Environ. Sci. 2019, 260, 1–8. [Google Scholar] [CrossRef]
- Nicola, S.; Basoccu, L. Nitrogen and N, P, K relation affect tomato seedling growth, yield and earliness. In III International Symposium on Protected Cultivation in Mild Winter Climates; ISHS Acta Horticulturae 357; ISHS: Buenos Aires, Argentina, 1994; Volume 357, pp. 95–102. [Google Scholar]
- Basoccu, L.; Nicola, S. Supplementary light and pretransplant nitrogen effects on tomato seedling growth and yield. Hydroponics Transpl. Prod. 1994, 396, 313–320. [Google Scholar] [CrossRef]
- Melton, R.R.; Dufault, R.J. Nitrogen, phosphorus, and potassium fertility regimes affect tomato transplant growth. HortScience 1991, 26, 141–142. [Google Scholar] [CrossRef]
- Scholberg, J.; Mcneal, B.L.; Boote, K.J.; Jones, J.W.; Locascio, S.J.; Olson, S.M. Nitrogen stress effects on growth and nitrogen accumulation by field-grown tomato. Agron. J. 2000, 92, 159–167. [Google Scholar] [CrossRef]
- Elia, A.; Giulia, C. Agronomic and physiological responses of a tomato crop to nitrogen input. Eur. J. Agron. 2012, 40, 64–74. [Google Scholar] [CrossRef]
- Hariyadi, B.W.; Nisak, F.; Nurmalasari, I.R.; Kogoya, Y. Effect of Dose and Time of Npk Fertilizer Application on the Growth and Yield of Tomato Plants (Lycopersicum esculentum Mill). Agric. Sci. 2019, 2, 101–111. [Google Scholar]
- Whitaker, R.T.; Mirzargar, M.; Kirby, R.M. Contour boxplots: A method for characterizing uncertainty in feature sets from simulation ensembles. IEEE Trans. Vis. Comput. Graph. 2013, 19, 2713–2722. [Google Scholar] [CrossRef]


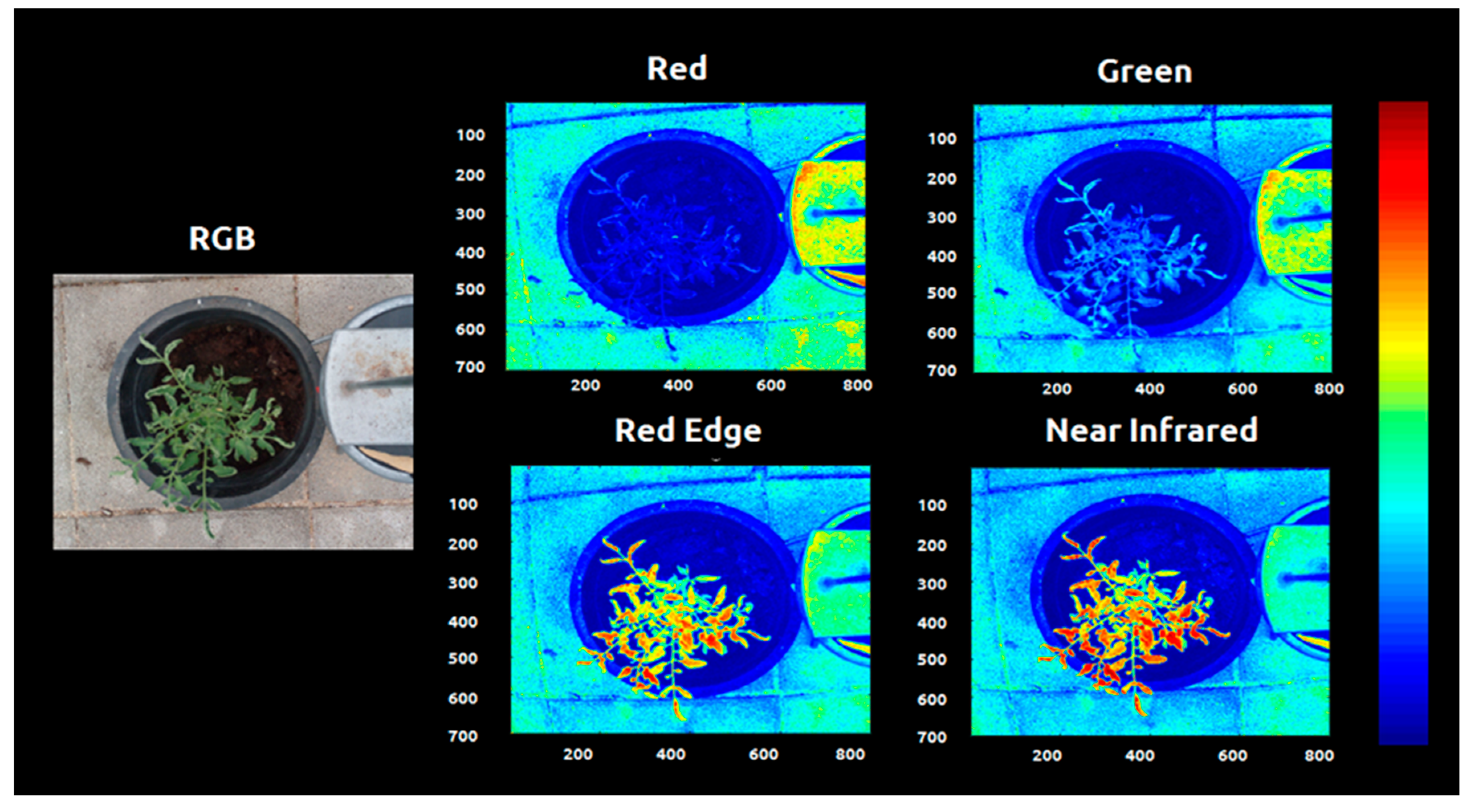
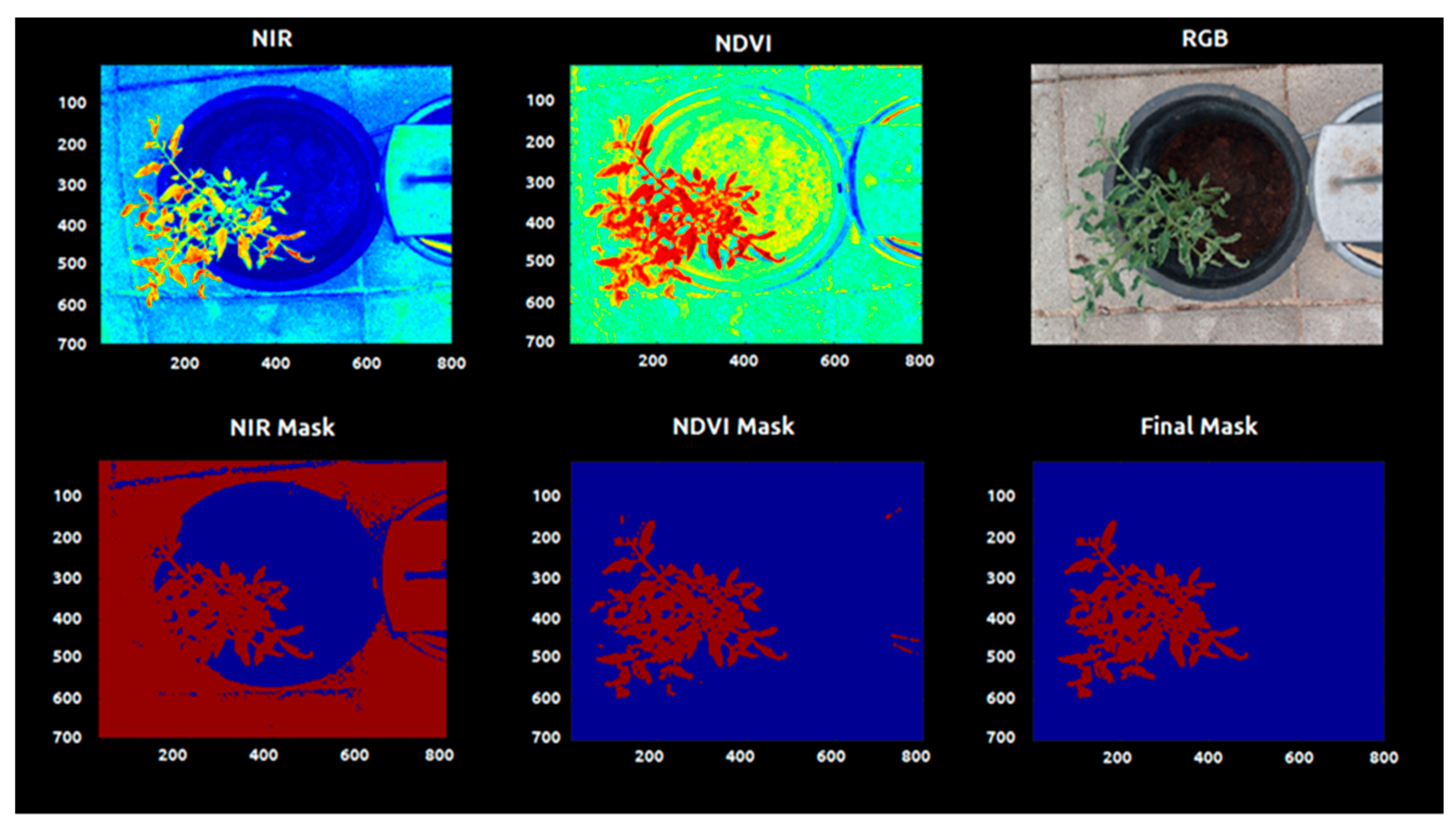
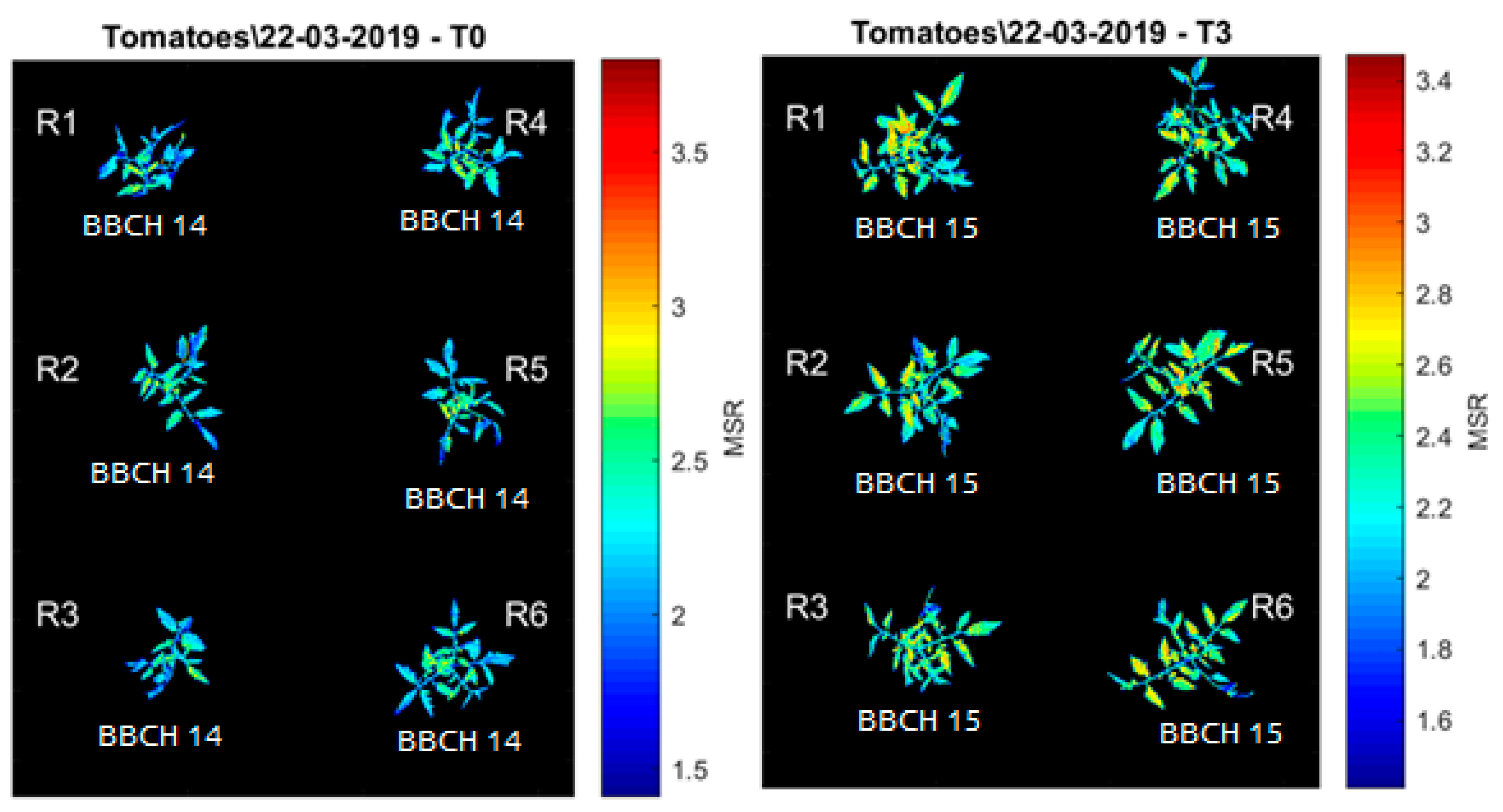
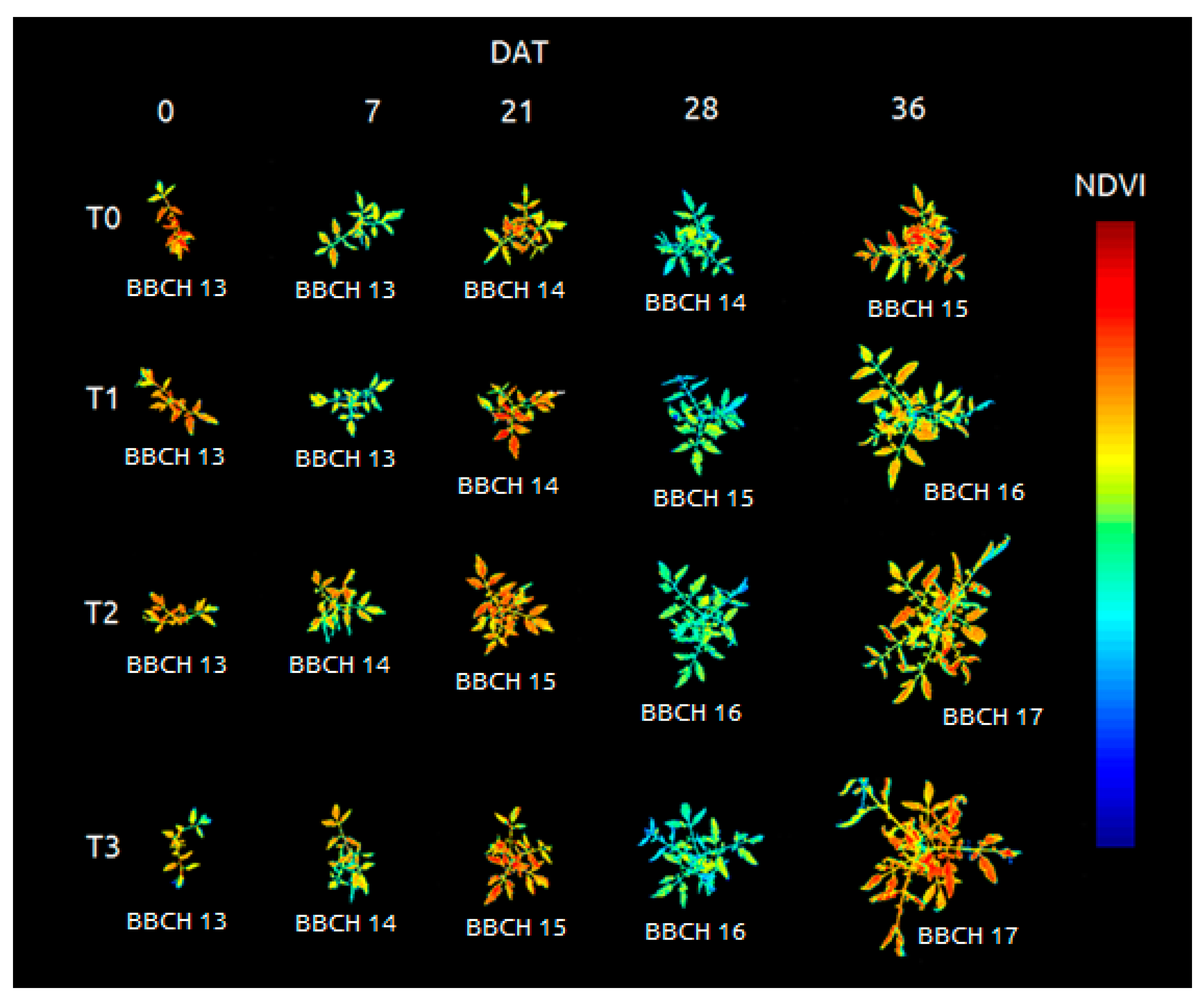


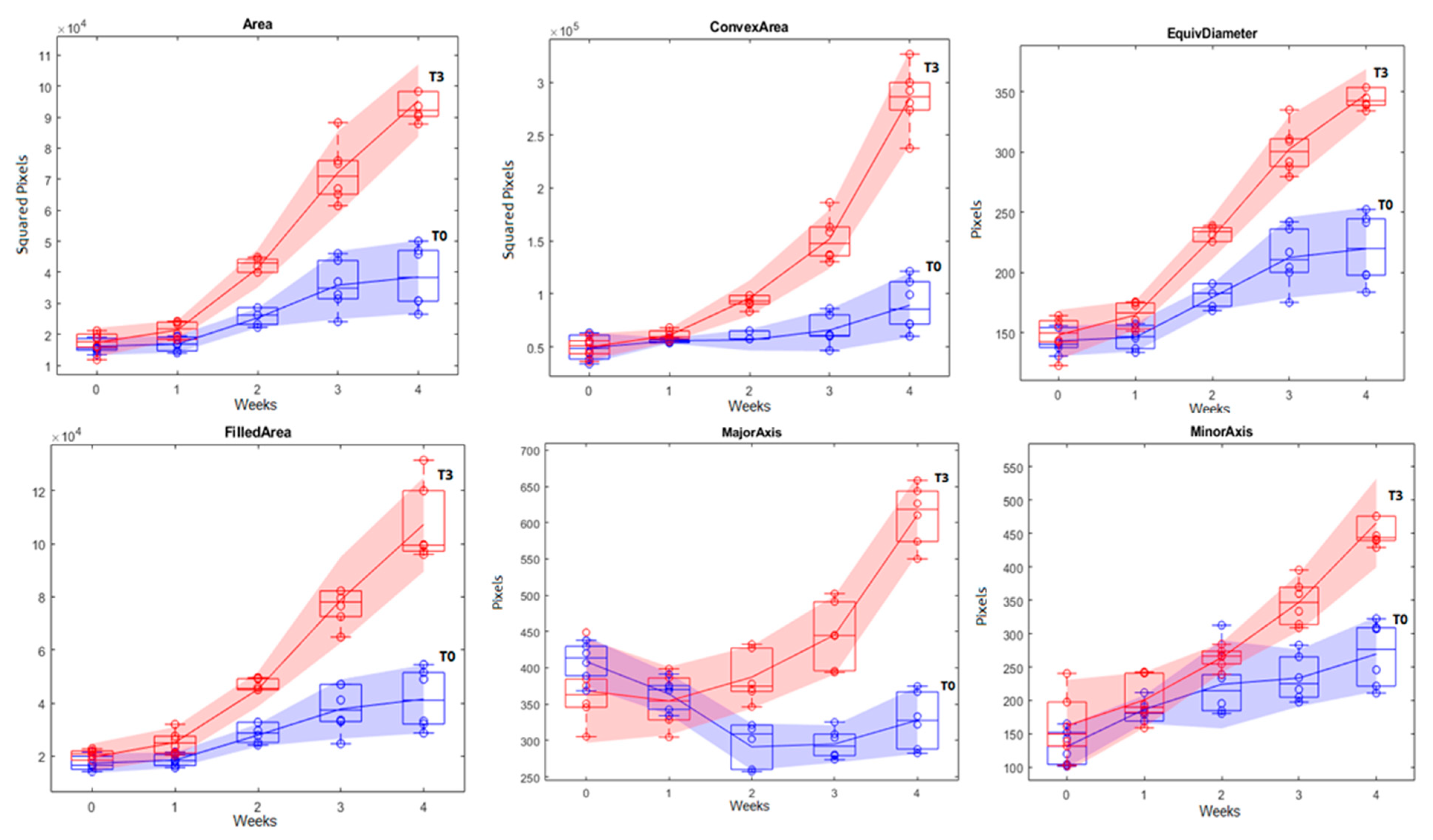
| Images | Shift Factor |
|---|---|
| Green | [50, 23] |
| Near Infrared | [41, −34] |
| Red Edge | [68, −11] |
| Index | Abbreviation | Formula | Application |
|---|---|---|---|
| Normalized Difference Vegetation Index | NDVI | Measuring green vegetation through normalized ration ranging from −1 to 1. | |
| Index | GNDVI | Modification of NDVI, more sensitive to chlorophyll content. | |
| Normalized Difference Vegetation Index | RENDVI | Modification of NDVI, using Red-Edge information related to plant health. | |
| Green Normalized Difference Vegetation Index | NLI | Modification of NDVI used to emphasize linear relations with vegetation parameters. | |
| Red-Edge Normalized Difference Vegetation Index | OSAVI | Variation of NDVI in order to reduce the soil effect | |
| Nonlinear Vegetation Index | GRVI | Related with leaf production and stress | |
| Optimized Soil Adjusted Vegetation Index | MSR | A combination of renormalized NDVI and SR to improve sensitivity to vegetable characteristics | |
| Green Ratio Vegetation Index | SR | Ratio of NIR scattering to chlorophyll and light absorption used for simple vegetation distinction | |
| Modified Simple Ratio | NDRER | Modification of NDVI, using Red-Edge instead of NIR. | |
| Simple ratio | SPI2 | Index used in areas with high variability in canopy structure | |
| Normalized Difference Red-Edge/Red | LCI | Index to assess chlorophyll content in areas of complete leaf coverage. |
| Morphological Property | Description |
|---|---|
| Area | Actual number of pixels in the region, returned as a scalar. |
| Convex Area | Number of pixels in the image that specifies the convex hull, with all pixels within the hull filled in (set to on), returned as a binary image (logical). The image is the size of the bounding box * of the region. |
| Eccentricity | Eccentricity of the ellipse that has the same second-moments as the region, returned as a scalar. The eccentricity is the ratio of the distance between the foci of the ellipse and its major axis length. The value is between 0 and 1. (0 and 1 are degenerate cases. An ellipse whose eccentricity is 0 is a circle, while an ellipse whose eccentricity is 1 is a line segment.) |
| Diameter Equivalent | Diameter of a circle with the same area as the region, returned as a scalar. Computed as . |
| Euler Number | Number of objects in the region minus the number of holes in those objects, returned as a scalar. |
| Extent | Ratio of pixels in the region to pixels in the total bounding box *, returned as a scalar. Computed as the area divided by the area of the bounding box *. |
| Filled Area | Number of on pixels in filled image, returned as a scalar. |
| Orientation | Angle between the x-axis and the major axis of the ellipse that has the same second-moments as the region, returned as a scalar. The value is in degrees, ranging from −90 degrees to 90 degrees. |
| Major Axis Length | Length (in pixels) of the major axis of the ellipse that has the same normalized second central moments as the region, returned as a scalar. |
| Minor Axis Length | Length (in pixels) of the minor axis of the ellipse that has the same normalized second central moments as the region, returned as a scalar |
| Perimeter | Distance around the boundary of the region returned as a scalar. This function computes the perimeter by calculating the distance between each adjoining pair of pixels around the border of the region |
| Solidity | Proportion of the pixels in the convex hull that are also in the region, returned as a scalar. Computed as area/convex area |
| Time After Transplant (Days) | ||||||||
|---|---|---|---|---|---|---|---|---|
| 08-mar | 15-mar | 22-mar | 29-mar | 05-abr | 12-abr | 19-abr | 26-abr | |
| 0 | 7 | 14 | 21 | 28 | 36 | 43 | 50 | |
| NDVI | n.s | n.s | n.s | n.s | n.s | n.s | 0.02 ** | 0.05 ** |
| MSR | n.s | n.s | n.s | n.s | n.s | n.s | 0.074 ** | 0.03 ** |
| GRVI | n.s | n.s | n.s | n.s | n.s | n.s | 0.013 ** | 0.103 ** |
| Area | n.s | n.s | n.s | 0.002 ** | <0.0001 ** | <0.0001 ** | <0.0001 ** | <0.0001 ** |
| MajorAxisLength | n.s | n.s | n.s | <0.0001 | <0.0001 ** | 0.001 ** | 0.0008 ** | <0.0001 ** |
| MinorAxisLength | n.s | n.s | n.s | 0.0007 ** | <0.0001 ** | <0.0001 ** | <0.0001 ** | <0.0001 ** |
| ConvexArea | n.s | n.s | n.s | <0.0001 ** | <0.0001 ** | <0.0001 ** | <0.0001 ** | <0.0001 ** |
| FilledArea | n.s | n.s | n.s | 0.02 ** | <0.0001 ** | <0.0001 ** | <0.0001 ** | <0.0001 ** |
| Perimeter | n.s | n.s | n.s | 0.0016 ** | <0.0001 ** | <0.0001 ** | <0.0001 ** | 0.0002 ** |
| EquivDiameter | n.s | n.s | n.s | <0.0001 ** | <0.0001 ** | <0.0001 ** | <0.0001 ** | <0.0001 ** |
| Parameter | Tr. | Regression | R2 |
|---|---|---|---|
| Area | T0 | 0.937 | |
| T3 | 0.983 | ||
| Filled Area | T0 | 0.953 | |
| T3 | 0.977 | ||
| Perimeter | T0 | 0.559 | |
| T3 | 0.941 | ||
| Eq. Diameter | T0 | 0.938 | |
| T3 | 0.971 | ||
| Convex Area | T0 | 0.957 | |
| T3 | 0.992 | ||
| Major Axes | T0 | 0.933 | |
| T3 | 0.988 | ||
| Minor Axes | T0 | 0.976 | |
| T3 | 0.964 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cardim Ferreira Lima, M.; Krus, A.; Valero, C.; Barrientos, A.; del Cerro, J.; Roldán-Gómez, J.J. Monitoring Plant Status and Fertilization Strategy through Multispectral Images. Sensors 2020, 20, 435. https://doi.org/10.3390/s20020435
Cardim Ferreira Lima M, Krus A, Valero C, Barrientos A, del Cerro J, Roldán-Gómez JJ. Monitoring Plant Status and Fertilization Strategy through Multispectral Images. Sensors. 2020; 20(2):435. https://doi.org/10.3390/s20020435
Chicago/Turabian StyleCardim Ferreira Lima, Matheus, Anne Krus, Constantino Valero, Antonio Barrientos, Jaime del Cerro, and Juan Jesús Roldán-Gómez. 2020. "Monitoring Plant Status and Fertilization Strategy through Multispectral Images" Sensors 20, no. 2: 435. https://doi.org/10.3390/s20020435
APA StyleCardim Ferreira Lima, M., Krus, A., Valero, C., Barrientos, A., del Cerro, J., & Roldán-Gómez, J. J. (2020). Monitoring Plant Status and Fertilization Strategy through Multispectral Images. Sensors, 20(2), 435. https://doi.org/10.3390/s20020435







