Daily Life Benefits and Usage Characteristics of Dynamic Arm Supports in Subjects with Neuromuscular Disorders
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
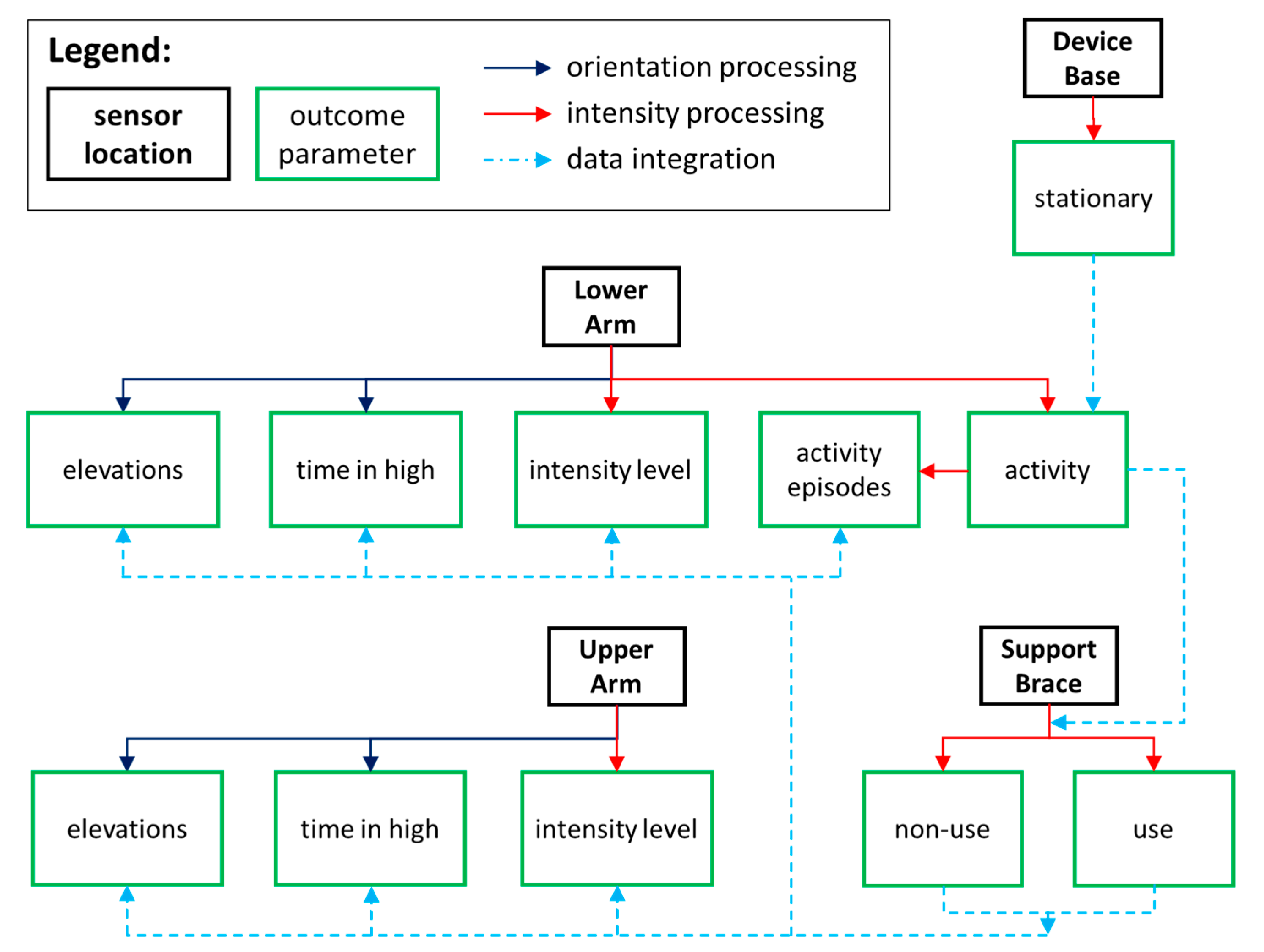
2.2. Activity Sensors
2.3. Data Processing
2.4. Self-Reports
2.5. Data Synthesis
2.6. Statistical Analysis
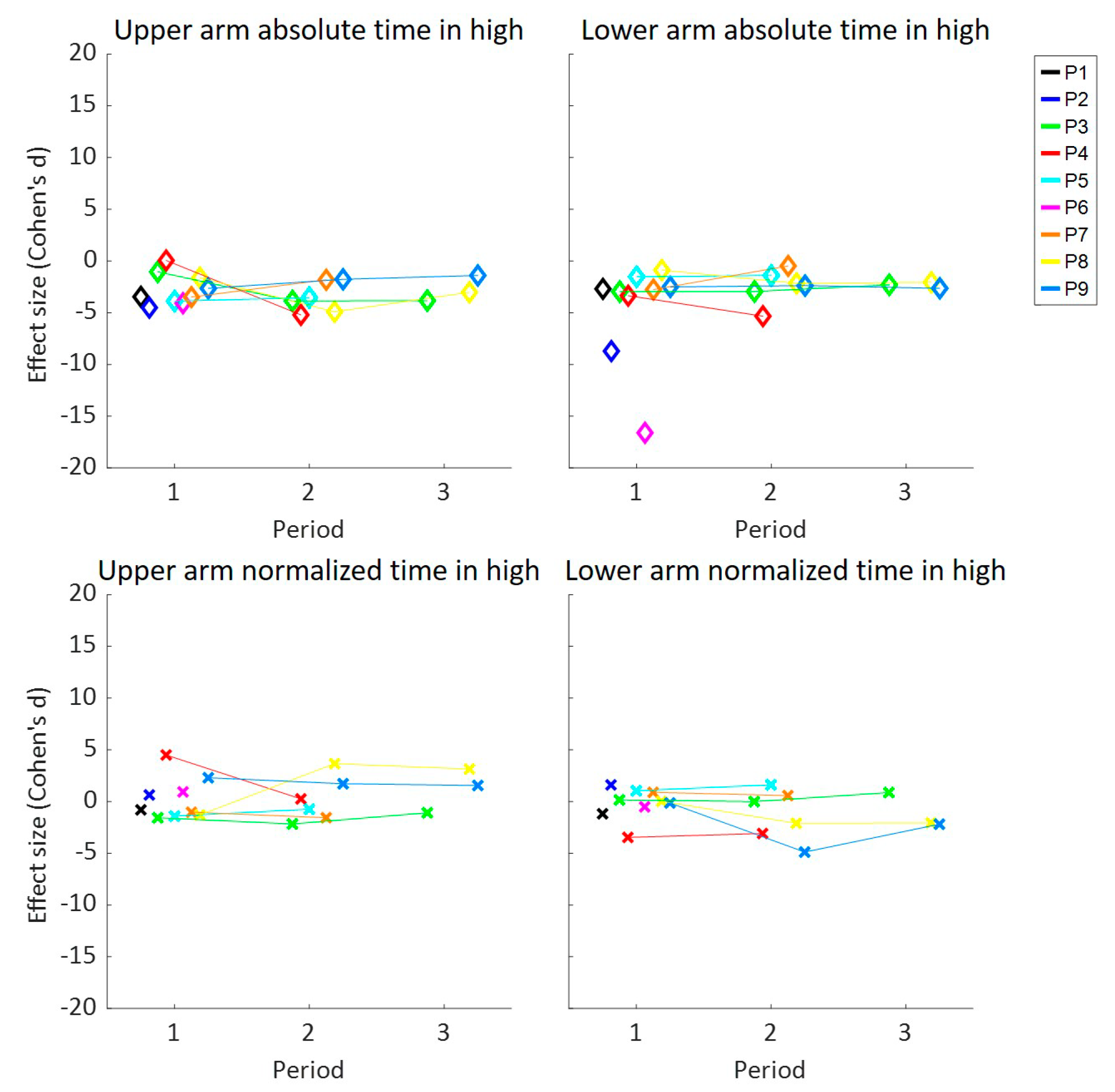
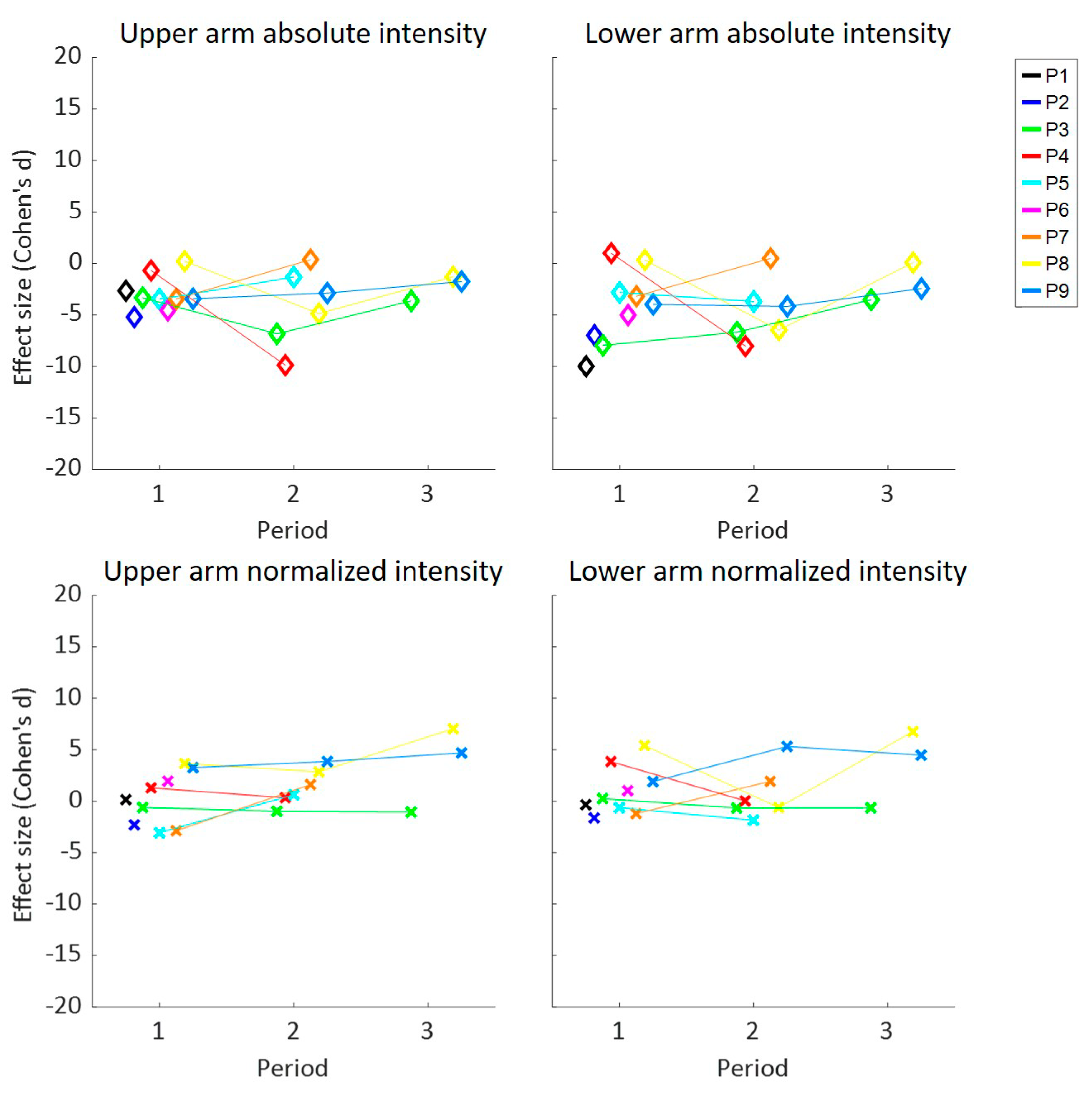
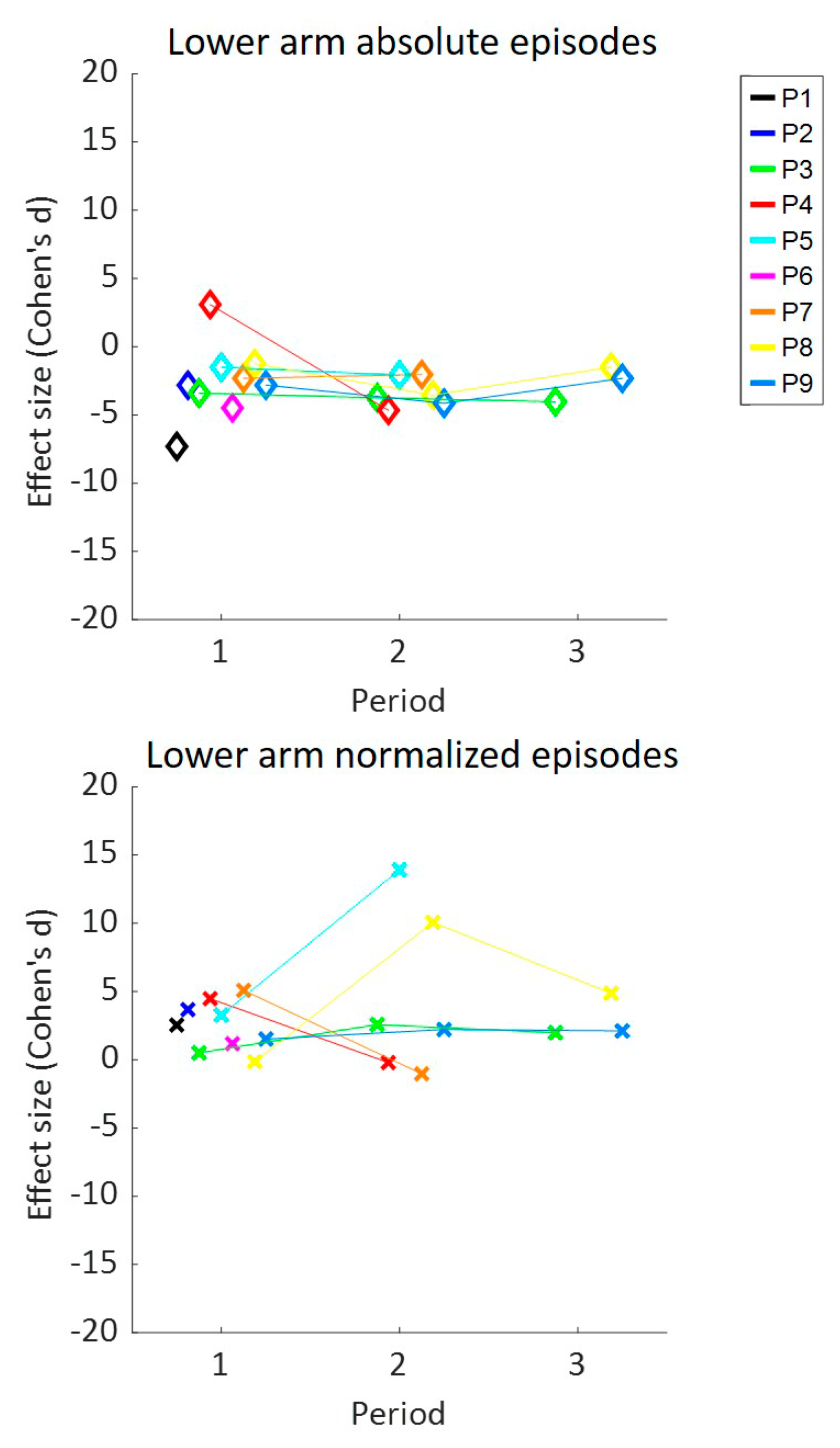
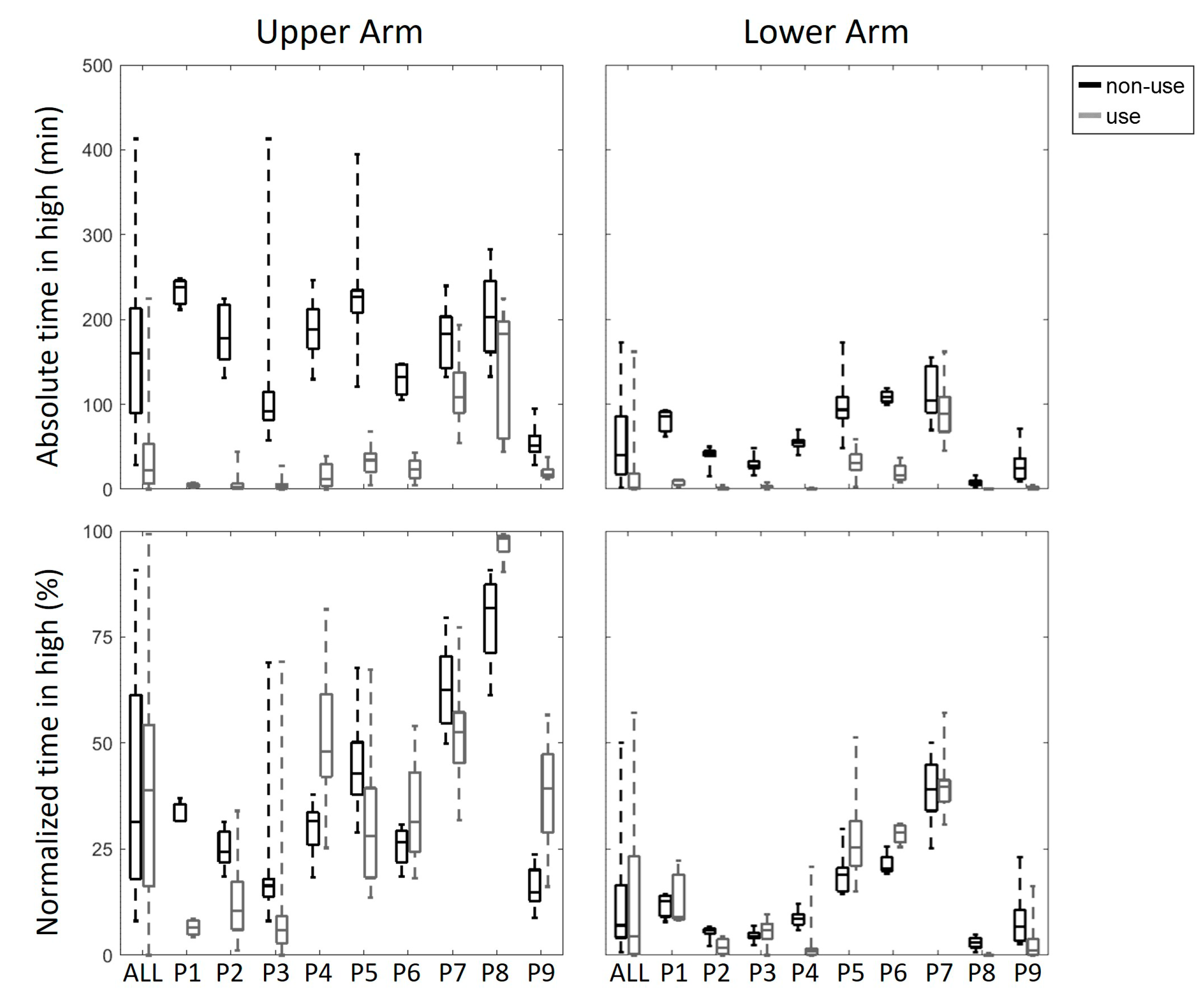
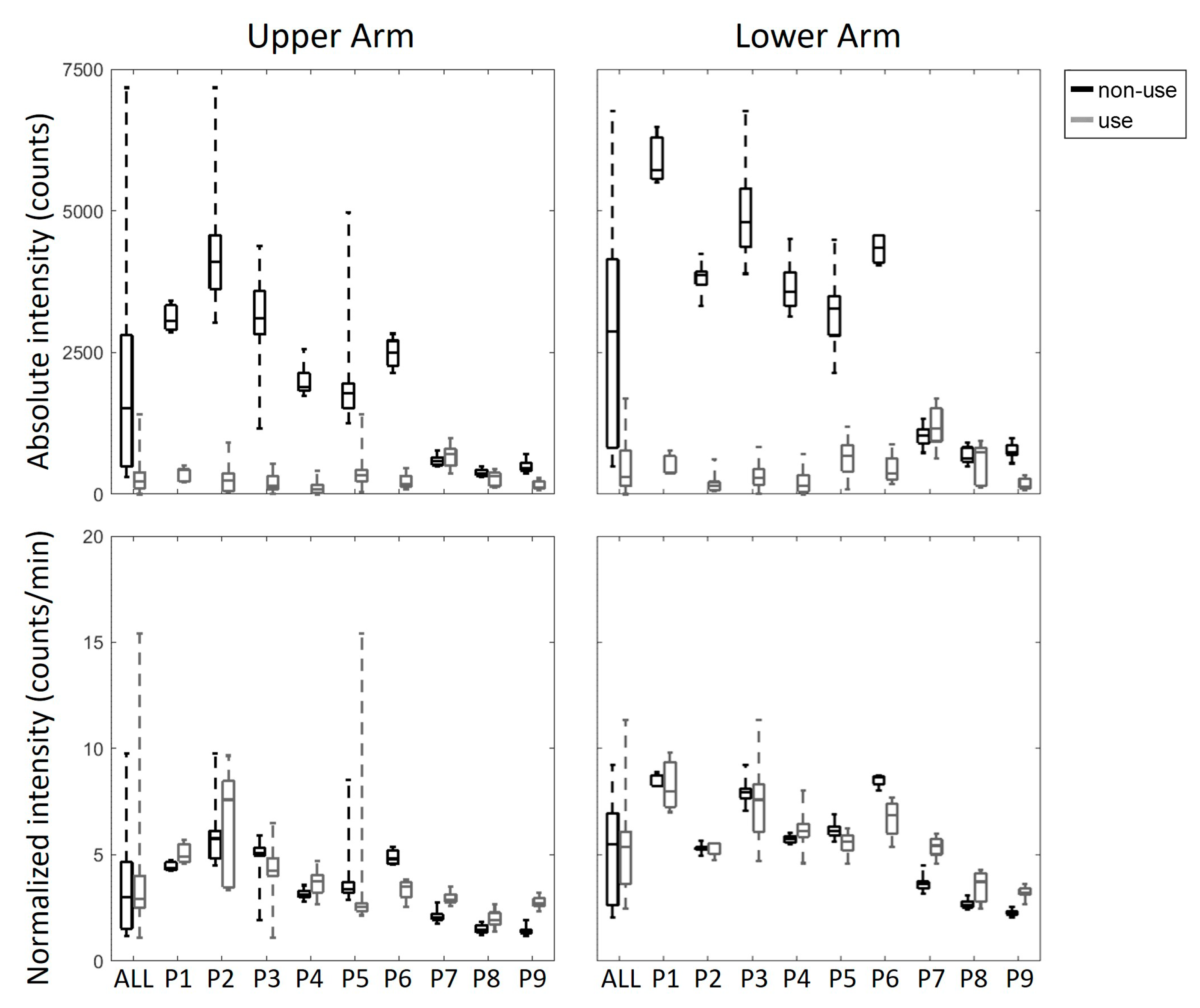
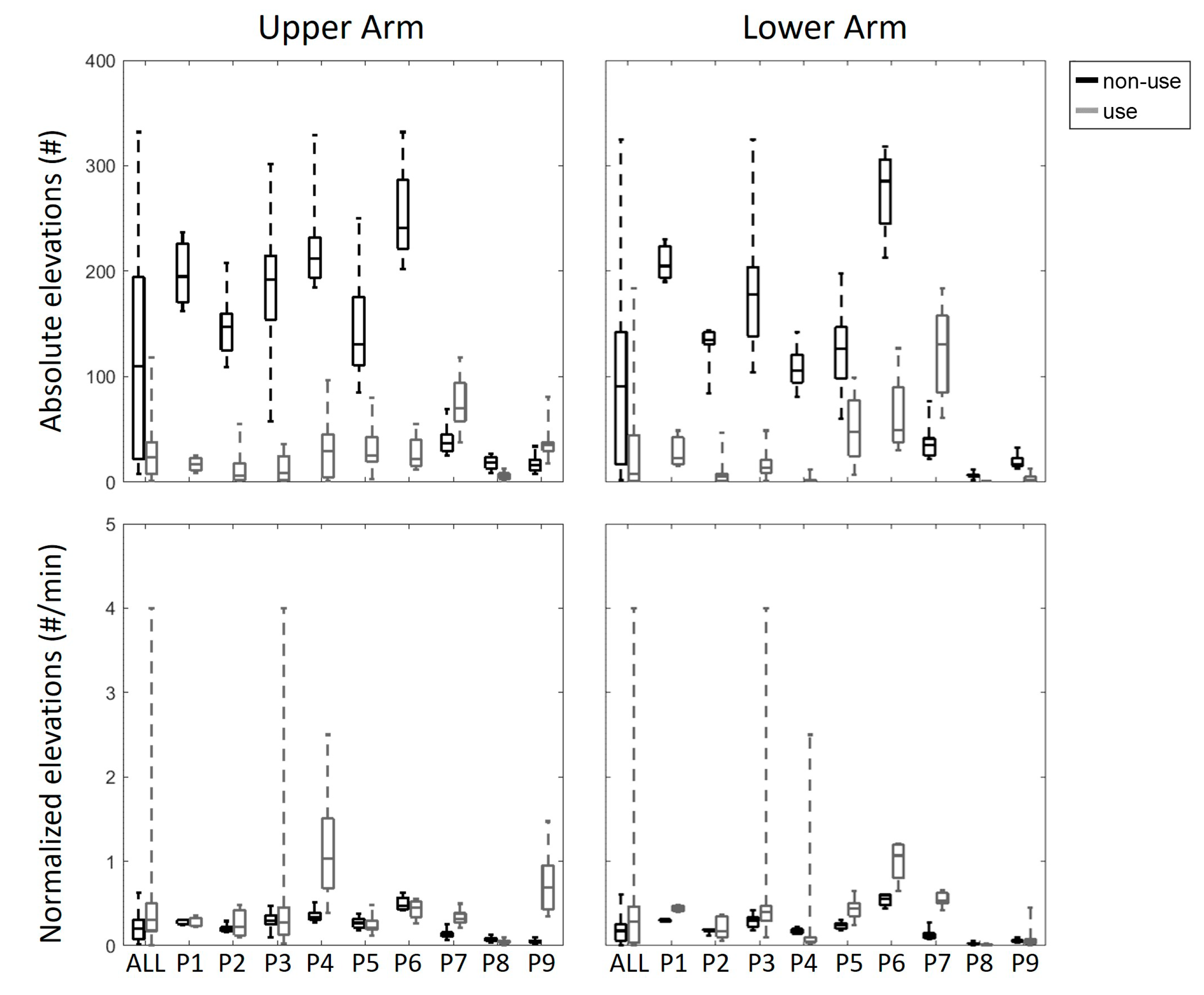
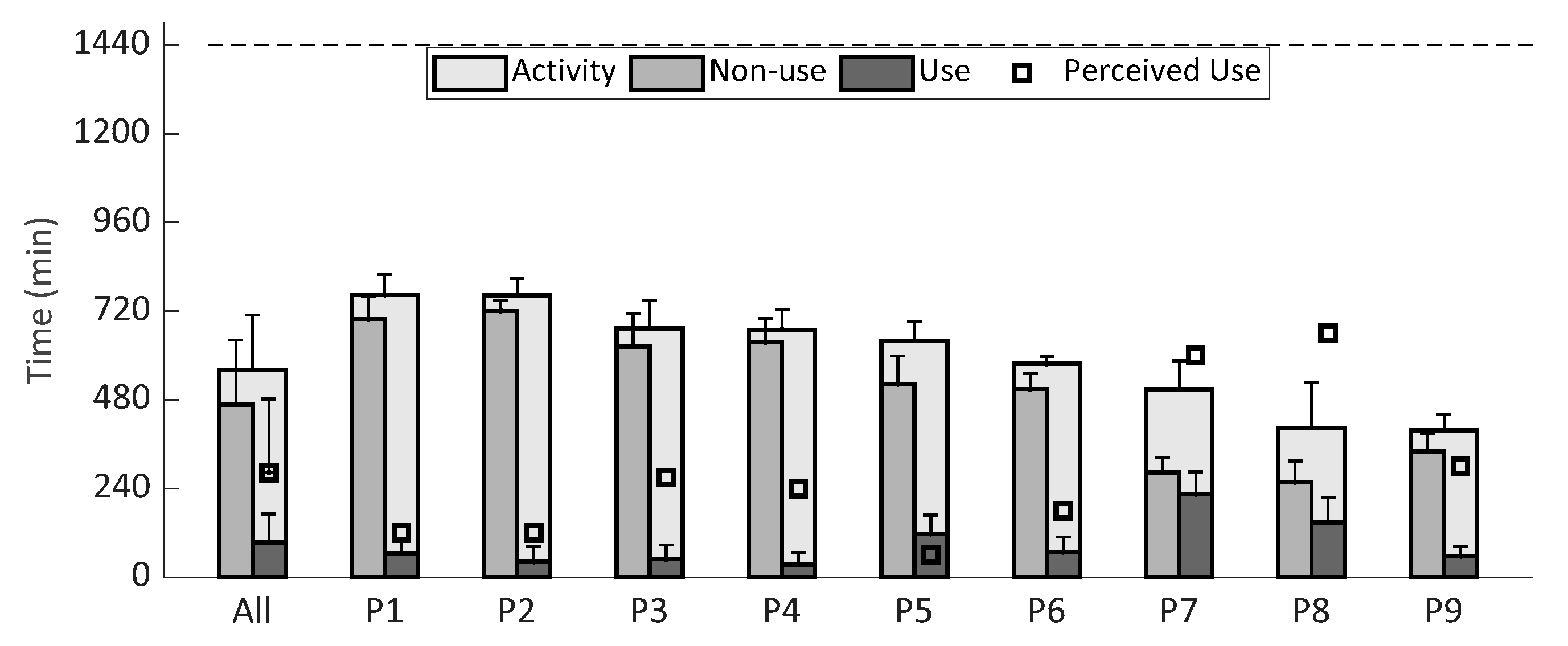
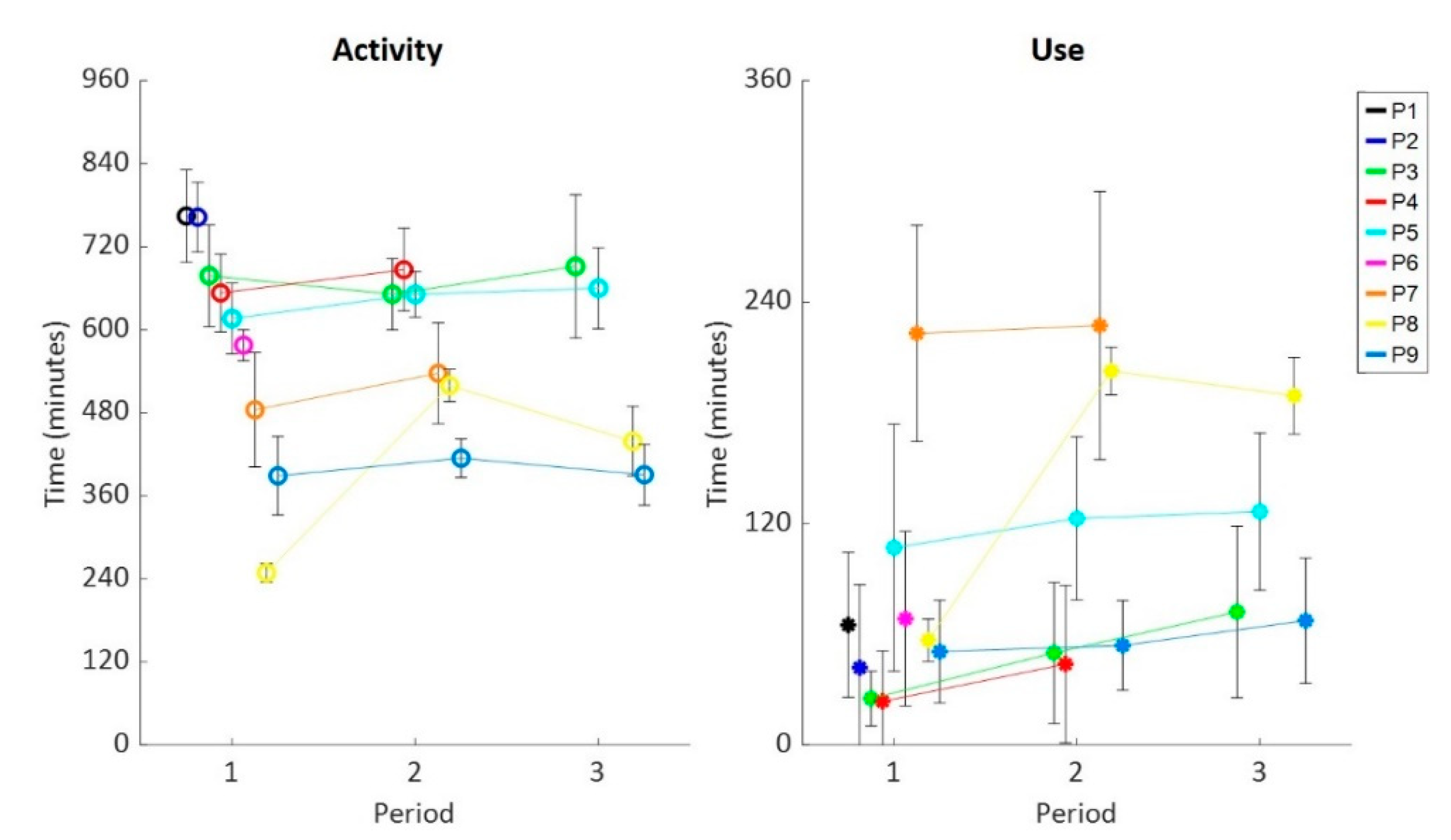
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A


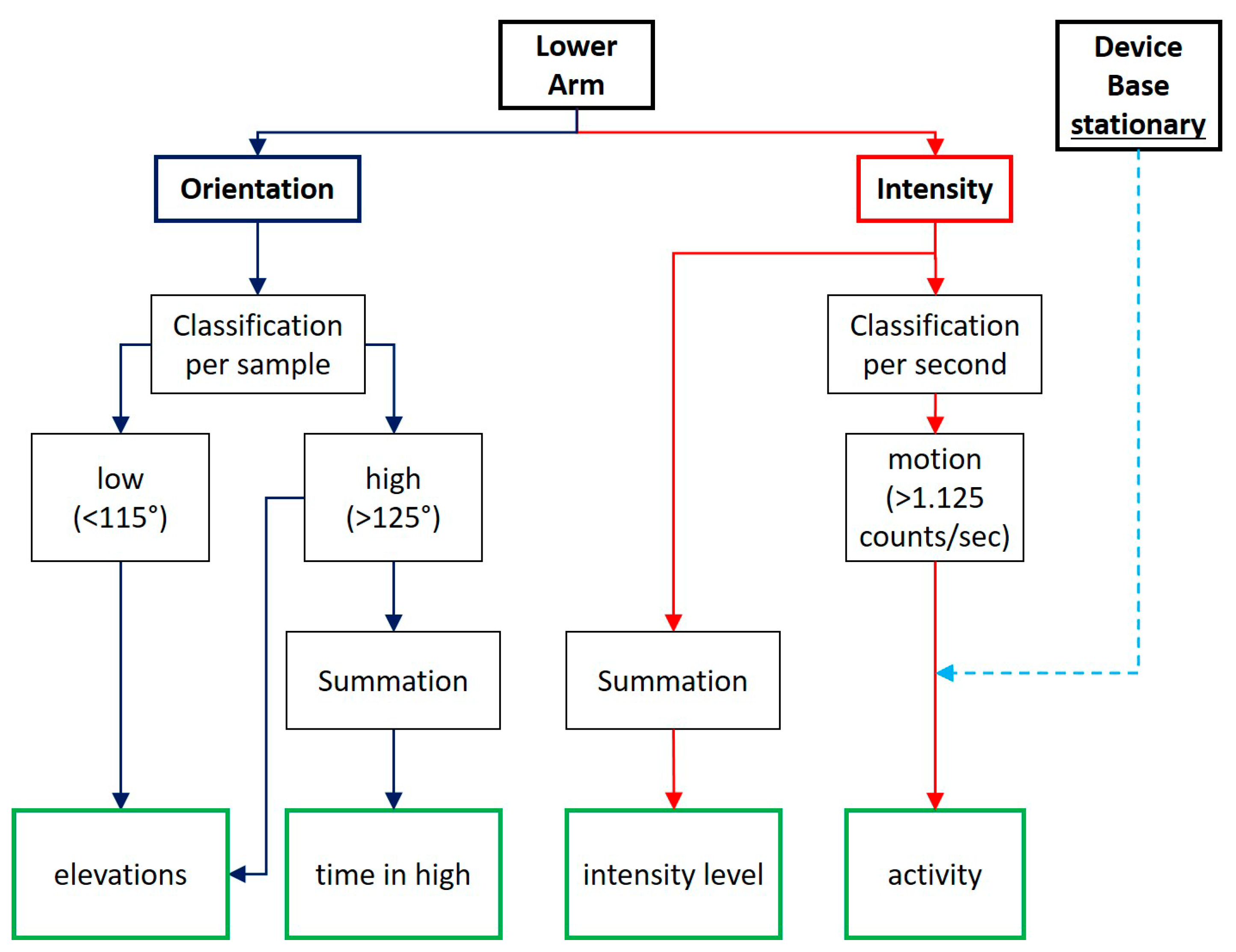
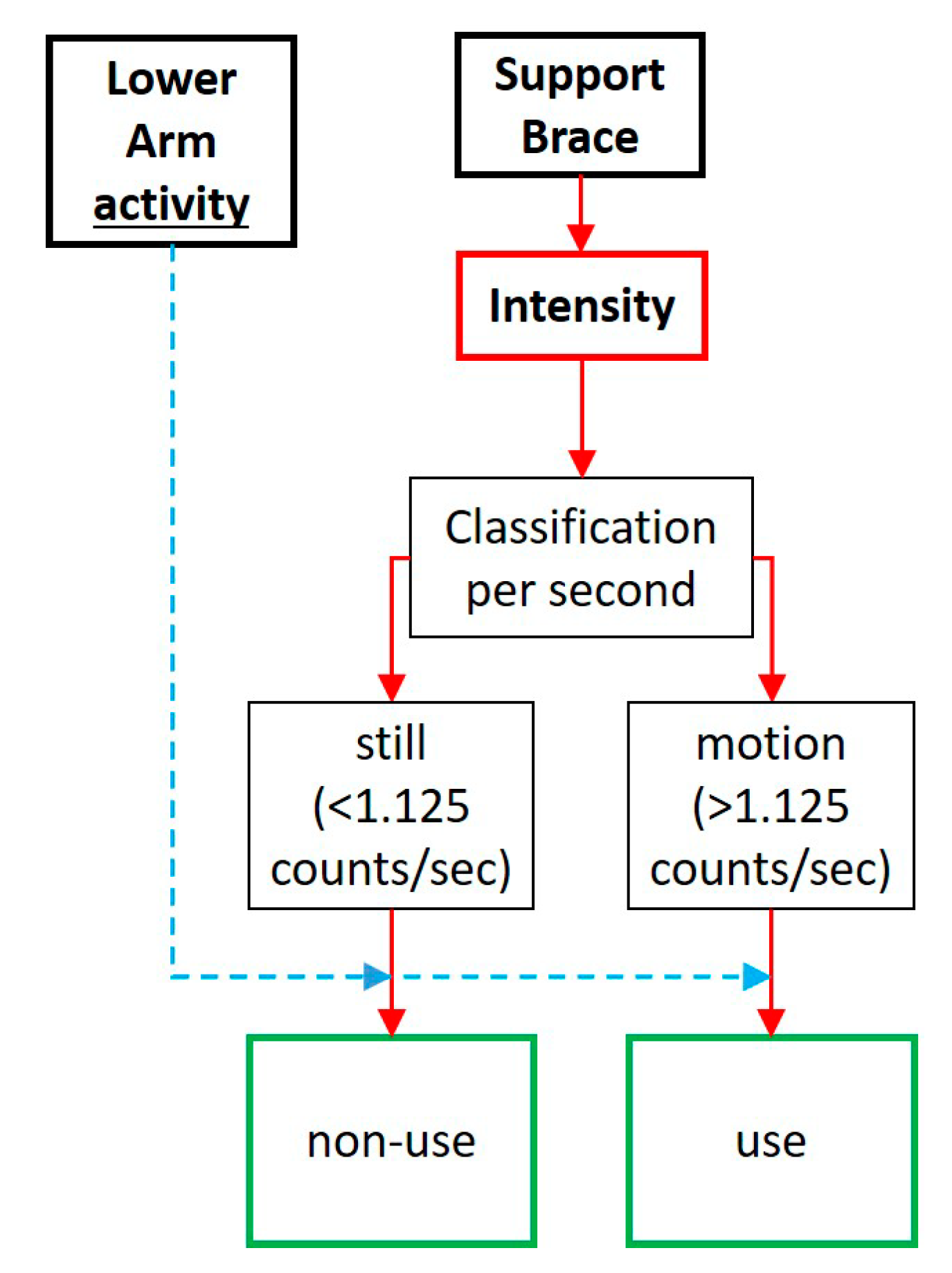
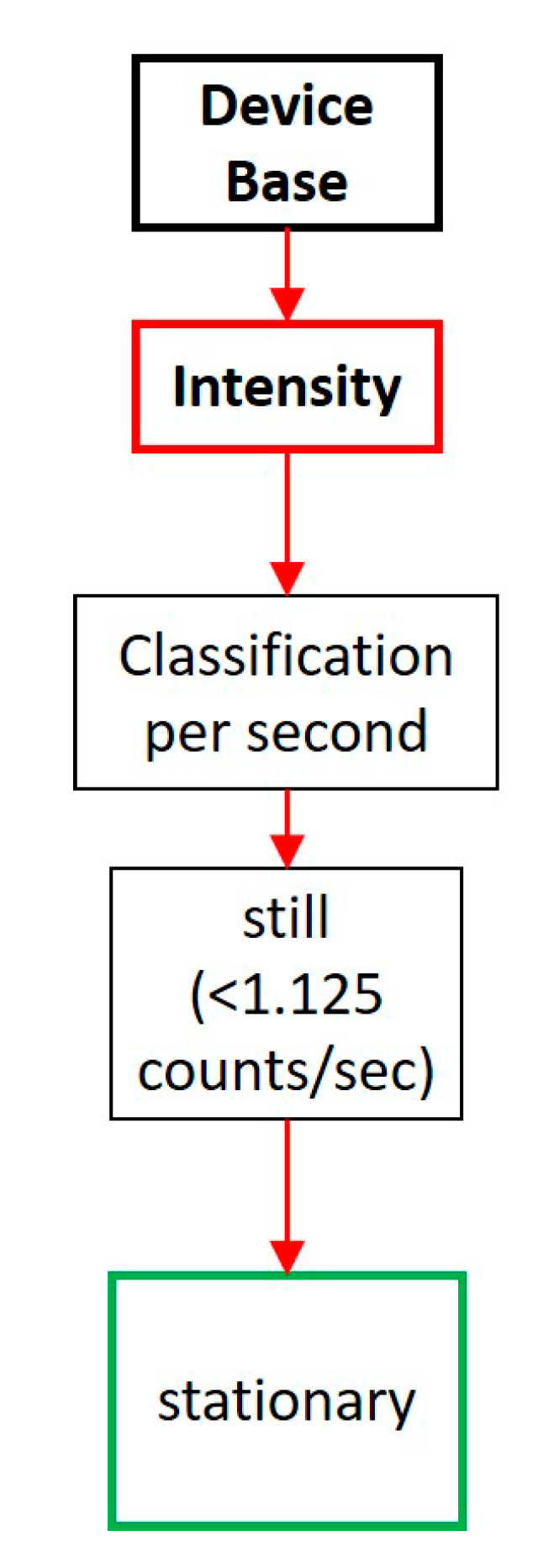
| Time in High UA | Time in High LA | Intensity UA | Intensity LA | Elevations UA | Elevations LA | Episodes LA | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| abs | norm | abs | norm | abs | norm | abs | norm | abs | norm | abs | norm | abs | norm | |
| All | −1.9 | −0.1 | −1.6 | 0.0 | −1.2 | 0.2 | −1.3 | 0.1 | −1.1 | 0.6 | −0.9 | 1.0 | −1.9 | 1.7 |
| P1 | −10.5 | −16.3 | −5.9 | 0.2 | −6.5 | 1.0 | −7.8 | −0.1 | −4.0 | −0.1 | −4.8 | 4.0 | −5.7 | 4.1 |
| P2 | −3.5 | −0.8 | −2.7 | −1.2 | −2.7 | 0.1 | −10.0 | −0.3 | −3.6 | 0.4 | −2.9 | 0.2 | −7.3 | 2.5 |
| P3 | −1.6 | −1.6 | −2.8 | 0.3 | −4.2 | −0.9 | −5.0 | −0.3 | −3.0 | 0.2 | −2.5 | 0.3 | −3.8 | 0.8 |
| P4 | −4.7 | 0.5 | −7.5 | −1.0 | −6.0 | 1.0 | −6.3 | 0.6 | −3.3 | 0.8 | −6.0 | 0.1 | −4.8 | 0.5 |
| P5 | −3.4 | −1.2 | −1.9 | 1.0 | −2.4 | −0.2 | −3.2 | −1.0 | −2.2 | −0.2 | −1.2 | 1.5 | −1.9 | 3.6 |
| P6 | −4.5 | 0.6 | −8.7 | 1.6 | −5.2 | −2.3 | −7.0 | −1.6 | −3.5 | −0.5 | −3.4 | 2.1 | −2.8 | 3.7 |
| P7 | −1.6 | −1.5 | −0.7 | 0.2 | 0.3 | 2.1 | 0.4 | 2.6 | 1.2 | 2.3 | 1.8 | 4.3 | −1.5 | −0.5 |
| P8 | −1.3 | 2.3 | −1.8 | −2.2 | −1.2 | 2.1 | −0.6 | 0.4 | −2.1 | −1.3 | −2.3 | −1.5 | −0.6 | 1.6 |
| P9 | −1.8 | 1.8 | −1.6 | −0.8 | −2.5 | 3.8 | −3.3 | 3.1 | 1.5 | 2.0 | −2.6 | 0.2 | −2.7 | 1.9 |
| Perceived Activity (min) | Perceived Use (min) | Perceived DAS Use (%) | Perceived DAS Benefit Scale (%) | Ratio Left-Right Arm Activity | |
|---|---|---|---|---|---|
| P1 | 120 | 120 | 100 | 80 | 50/50 |
| P2 | 300 | 120 | 40 | 80 | 33/67 |
| P3 | 270 | 270 | 100 | 90 | 70/30 |
| P4 | 720 | 240 | 33 | 60 | 33/67 |
| P5 | 60 | 60 | 100 | 67 | 40/60 |
| P6 | 300 | 180 | 60 | 70 | 30/70 |
| P7 | 600 | 600 | 100 | 100 | 40/60 |
| P8 | 660 | 660 | 100 | 100 | 40/60 |
| P9 | 840 | 300 | 36 | 100 | 40/60 |


References
- Deenen, J.C.; Horlings, C.G.; Verschuuren, J.J.; Verbeek, A.L.; van Engelen, B.G. The epidemiology of neuromuscular disorders: A comprehensive overview of the literature. J. Neuromuscul. Dis. 2015, 2, 73–85. [Google Scholar] [CrossRef] [PubMed]
- Deenen, J.C.; van Doorn, P.A.; Faber, C.G.; van der Kooi, A.J.; Kuks, J.B.; Notermans, N.C.; Visser, L.H.; Horlings, C.G.; Verschuuren, J.J.; Verbeek, A.L.; et al. The epidemiology of neuromuscular disorders: Age at onset and gender in the Netherlands. Neuromuscul. Disord. NMD 2016, 26, 447–452. [Google Scholar] [CrossRef] [PubMed]
- Bergsma, A.; Janssen, M.M.H.P.; Geurts, A.C.H.; Cup, E.H.; de Groot, I.J.M. Different profiles of upper limb function in four types of neuromuscular disorders. Neuromuscul. Disord. NMD 2017, 27, 1115–1122. [Google Scholar] [CrossRef] [PubMed]
- Janssen, M.M.H.P.; Bergsma, A.; Geurts, A.C.H.; de Groot, I.J.M. Patterns of decline in upper limb function of boys and men with DMD: An international survey. J. Neurol. 2014, 261, 1269–1288. [Google Scholar] [CrossRef] [PubMed]
- van der Heide, L.; van Ninhuijs, B.; Bergsma, A.; Gelderblom, G.; van der Pijl, D.; de Witte, L. An overview and categorization of dynamic arm supports for people with decreased arm function. Prosthet. Orthot. Int. 2014, 38, 287–302. [Google Scholar] [CrossRef] [PubMed]
- van der Heide, L.; de Witte, L. The perceived functional benefit of dynamic arm supports in daily life. J. Rehabil. Res. Dev. 2016, 533, 1139–1150. [Google Scholar] [CrossRef]
- Haumont, T.; Rahman, T.; Sample, W.; King, M.M.; Church, C.; Henley, J.; Jayakumar, S. Wilmington Robotic Exoskeleton: A novel device to maintain arm improvement in muscular disease. J. Pediatr. Orthop. 2011, 31, e44–e49. [Google Scholar] [CrossRef]
- Herder, J.L.; Vrijlandt, N.; Antonides, T.; Cloosterman, M.; Mastenbroek, P.L. Principle and design of a mobile arm support for people with muscular weakness. J. Rehabil. Res. Dev. 2006, 43, 591–604. [Google Scholar] [CrossRef]
- Kumar, A.; Phillips, M.F. Use of powered mobile arm supports by people with neuromuscular conditions. JRRD 2013, 50, 61–70. [Google Scholar] [CrossRef]
- Rahman, T.; Sample, W.; Seliktar, R.; Alexander, M.; Scavina, M. A body-powered functional upper limb orthosis. JRRD 2000, 37, 675–680. [Google Scholar]
- Estilow, T.; Glanzman, A.M.; Powers, K.; Moll, A.; Flickinger, J.; Medne, L.; Tennekoon, G.; Yum, S.W. Use of the wilmington robotic exoskeleton to improve upper extremity function in patients with duchenne muscular dystrophy. Am. J. Occup. Ther. 2018, 72, 7202345010p1–7202345010p5. [Google Scholar] [CrossRef] [PubMed]
- Coscia, M.; Cheung, V.C.K.; Tropea, P.; Koenig, A.; Monaco, V.; Bennis, C.; Micera, S.; Bonato, P. The effect of arm weight support on upper limb muscle synergies during reaching movements. J. Neuroeng. Rehabil. 2014, 11, 22. [Google Scholar] [CrossRef] [PubMed]
- Prange, G.B.; Jannink, M.J.A.; Stienen, A.H.A.; van der Kooij, H.; IJzerman, M.J.; Hermens, H.J. Influence of gravity compensation on muscle activation patterns during different temporal phases of arm movements of stroke patients. Neurorehabil. Neural Repair 2009, 23, 478–485. [Google Scholar] [CrossRef] [PubMed]
- Ellis, M.D.; Sukal, T.; DeMott, T.; Dewald, J.P.A. Augmenting clinical evaluation of hemiparetic arm movement with a laboratory-based quantitative measurement of kinematics as a function of limb loading. Neurorehabil. Neural Repair 2008, 22, 321–329. [Google Scholar] [CrossRef]
- Cruz, A.; Callaway, L.; Randall, M.; Ryan, M. Mobile arm supports in Duchenne muscular dystrophy: A pilot study of user experience and outcomes. Disabil. Rehabil. Assist. Technol. 2020, 1–10. [Google Scholar] [CrossRef]
- van der Heide, L.; Ramakers, I.; Essers, J.M.N.; Gelderblom, G.; De Witte, L.; Meijer, K. Is it possible to assess the effects of dynamic arm supports on upper extremity range of motion during activities of daily living in the domestic setting using a portable motion capturing device?—A pilot study. Technol. Disabil. 2017, 29, 91–99. [Google Scholar] [CrossRef]
- van der Heide, L.; Gelderblom, G.; de Witte, L. Effects and effectiveness of dynamic arm supports. Am. J. Phys. Med. Rehabil. 2015, 94, 44–62. [Google Scholar] [CrossRef]
- Essers, J.M.N.; Murgia, A.; Peters, A.A.; Janssen, M.M.H.P.; Meijer, K. Recommendations for Studies on Dynamic Arm Support Devices in People with Neuromuscular Disorders: A Scoping Review with Expert-Based Discussion. 2020. Available online: https://osf.io/ucpjq (accessed on 2 April 2020).
- Janssen, M.M.H.P.; Lobo-Prat, J.; Bergsma, A.; Vroom, E.; workshop participants. 2nd Workshop on Upper-Extremity Assistive Technology for People with Duchenne: Effectiveness and Usability of Arm Supports. Neuromuscul. Disord. NMD 2019, 29, 651–656. [Google Scholar] [CrossRef]
- World Health Organization. International Classification of Functioning, Disability and Health: ICF. World Health Organization. Available online: https://apps.who.int/iris/handle/10665/42407 (accessed on 2 April 2020).
- van der Heide, L.; Roentgen, U.; de Witte, L. Evaluation of dynamic arm supports in real life environments. Stud. Health Technol. Inform. 2017, 242, 517–522. [Google Scholar]
- Gandolla, M.; Antonietti, A.; Longatelli, V.; Pedrocchi, A. The effectiveness of wearable upper limb assistive devices in degenerative neuromuscular diseases: A systematic review and meta-analysis. Front. Bioeng. Biotechnol. 2020, 7, 450. [Google Scholar] [CrossRef]
- Bailey, R.R.; Lang, C.E. Upper extremity activity in adults: Referent values using accelerometry. J. Rehabil. Res. Dev. 2014, 50, 1213–1222. [Google Scholar] [CrossRef] [PubMed]
- van der Geest, A.; Essers, J.M.N.; Bergsma, A.; Jansen, M.; de Groot, I.J.M. Monitoring daily physical activity of upper extremity in young and adolescent boys with Duchenne muscular dystrophy: A pilot study. Muscle Nerve 2019, 61, 293–300. [Google Scholar] [CrossRef] [PubMed]
- Braito, I.; Maselli, M.; Sgandurra, G.; Inguaggiato, E.; Beani, E.; Cecchi, F.; Cioni, G.; Boyd, R. Assessment of upper limb use in childrenwith typical development andneurodevelopmental disorders by inertialsensors: A systematic review. J. Neuroeng. Rehabil. 2018, 15, 94. [Google Scholar] [CrossRef] [PubMed]
- Michielsen, M.E.; Selles, R.W.; Stam, H.J.; Ribbers, G.M.; Bussmann, J.B. Quantifying nonuse in chronic stroke patients: A studyinto paretic, nonparetic, and bimanual upper-limbuse in daily life. Arch. Phys. Med. Rehabil. 2012, 93, 1975–1981. [Google Scholar] [CrossRef] [PubMed]
- Chadwell, A.; Kenney, L.; Granat, M.; Thies, S.; Galpin, A.; Head, J. Upper limb activity of twenty myoelectric prosthesis users and twenty healthy anatomically intact adults. Sci. Data 2019, 6, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Wattanaprakornkul, D.; Halaki, M.; Boettcher, C.; Cathers, I.; Ginn, K.A. A comprehensive analysis of muscle recruitment patterns during shoulder flexion: An electromyographic study. Clin. Anat. (N. Y.) 2011, 24, 619–626. [Google Scholar] [CrossRef]
- Focal Meditech, B.V. Dowing. Available online: https://focalmeditech.nl/nl/content/dowing (accessed on 2 April 2020).
- Tawil, R.; McDermott, M.P.; Mendell, J.R.; Kissel, J.; Griggs, R.C. Facioscapulohumeral muscular dystrophy (FSHD): Design of natural history study and results of baseline testing. FSH-DY Group. Neurology 1994, 44, 442–446. [Google Scholar] [CrossRef]
- Bakhtiary, A.H.; Phoenix, J.; Edwards, R.H.; Frostick, S.P. The effect of motor learning in facioscapulohumeral muscular dystrophy patients. Eur. J. Appl. Physiol. 2000, 83, 551–558. [Google Scholar] [CrossRef]
- BV, M.I. MOX Accelerometry. Available online: https://www.accelerometry.eu (accessed on 2 April 2020).
- Bijnens, W.; Aarts, J.; Stevens, A.; Ummels, D.; Meijer, K. Optimization and Validation of an Adjustable Activity Classification Algorithm for Assessment of Physical Behavior in Elderly. Sensors 2019, 19, 5344. [Google Scholar] [CrossRef]
- IBM Corp. IBM SPSS Statistics for Windows, Version 25.0; IBM Corp: Armonk, NY, USA, 2017. [Google Scholar]
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences, 2nd ed.; Lawrence Erlbaum Associates: New York, NY, USA, 1988. [Google Scholar]
- Nam, H.S.; Seo, H.G.; Leigh, J.; Kim, Y.J.; Kim, S.; Bang, M.S. External Robotic Arm vs. Upper limb exoskeleton: What do potential users need? Appl. Sci. 2019, 9, 2471. [Google Scholar] [CrossRef]
- Pane, M.; Coratti, G.; Brogna, C.; Mazzone, E.S.; Mayhew, A.; Fanelli, L.; Messina, S.; D’Amico, A.; Catteruccia, M.; Scutifero, M.; et al. Upper limb function in Duchenne muscular dystrophy: 24 month longitudinal data. PLoS ONE 2018, 13, e0199223. [Google Scholar] [CrossRef] [PubMed]
- Lee, P.H.; Macfarlane, D.J.; Lam, T.H.; Stewart, S.M. Validity of the international physical activity questionnaire short form (IPAQ-SF): A systematic review. Int. J. Behav. Nutr. Phys. Act. 2011, 8, 115. [Google Scholar] [CrossRef] [PubMed]
- Prince, S.A.; Cardilli, L.; Reed, J.L.; Saunders, T.J.; Kite, C.; Douillette, K.; Fournier, K.; Buckley, J.P. A comparison of self-reported and device measured sedentary behaviour in adults: A systematic review and meta-analysis. Int. J. Behav. Nutr. Phys. Act. 2020, 17, 31. [Google Scholar] [CrossRef] [PubMed]
- Pitta, F.; Troosters, T.; Spruit, M.A.; Decramer, M.; Gosselink, R. Activity monitoring for assessment of physical activities in daily life in patients with chronic obstructive pulmonary disease. Arch. Phys. Med. Rehabil. 2005, 86, 1979–1985. [Google Scholar] [CrossRef]
- Bergsma, A.; Cup, E.H.C.; Geurts, A.C.H.; de Groot, I.J.M. Upper extremity function and activity in facioscapulohumeral dystrophy and limb-girdle muscular dystrophies: A systematic review. Disabil. Rehabil. 2014, 37, 1017–1032. [Google Scholar] [CrossRef]

| Gender (M/F) | Age (Y) | Diagnosis | Dynamic Arm Support | Dominant Side | Monitored Side | Total Days | |
|---|---|---|---|---|---|---|---|
| P1 | F | 61 | Congenital Myopathy | Gowing | Equal | Left | 3 |
| P2 | M | 44 | Amyotrophic Lateral Sclerosis | Gowing | Right | Right | 6 |
| P3 | M | 58 | Progressive Spinal Muscular Atrophy | Dowing (BL) | Right | Left | 21 |
| P4 | F | 30 | Limb Girdle Muscular Dystrophy | Armon Edero | Right | Right | 12 |
| P5 | F | 63 | Desminopathy | Sling (BL) | Right | Right | 16 |
| P6 | F | 60 | Limb Girdle Muscular Dystrophy | Dowing | Right | Right | 4 |
| P7 | M | 55 | Limb Girdle Muscular Dystrophy | Gowing | Right | Right | 13 |
| P8 | F | 60 | Spinal Muscular Atrophy type 3 | Gowing (BL) | Right | Left | 17 |
| P9 | M | 27 | Duchenne Muscular Dystrophy | Gowing (BL) | Right | Right | 21 |
| Primary | Secondary | ||
|---|---|---|---|
| Time absolute (min/day) | Time absolute (min/day) normalized (% activity) | Activity levels absolute (counts/day) normalized (counts/min) | Frequency absolute (#/day) normalized (#/min) |
| activity use non-use | UA use in high LA use in high UA non-use in high LA non-use in high | UA use intensity LA use intensity UA non-use intensity LA non-use intensity | UA use elevations LA use elevations UA non-use elevations LA non-use elevations LA use episodes LA non-use episodes |
| Reoccurring Activities | Only Possible with Device | Only Possible without Device |
|---|---|---|
| Eating/drinking (P1–5,7–9) Personal hygiene (P1–5,7–9) Computer activities (P1–5,7–9) House choirs and cooking (P1,4,5,7,8) Touch head (P7,8,9) | Eating/drinking (P4,5,8,9) Extended time computer work (P5,8) Touching head (P7,8) Personal hygiene (P8) | Driving a car (P4) Motions beyond the ambulant chair (P5) Typing (P7) |
| Unsuccessful with device | Unsuccessful without device | |
| Proper pronation/supination (P3) Maintaining arm within DAS (P3,4) Reaching below waist level (P3–5) Personal hygiene (P4) Folding laundry (P5) Support of wrist during hand to mouth (P5) Opening door handle (P7) Washing hand (P9) | Personal hygiene (P2) Reaching above shoulder (P3,4,5,7) House choirs/cooking (P4) Shake hands (P5) Flush toilet (P5) Opening door handle (P7) Eating/drinking (P7) Almost everything (P9) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Essers, J.; Murgia, A.; Peters, A.; Meijer, K. Daily Life Benefits and Usage Characteristics of Dynamic Arm Supports in Subjects with Neuromuscular Disorders. Sensors 2020, 20, 4864. https://doi.org/10.3390/s20174864
Essers J, Murgia A, Peters A, Meijer K. Daily Life Benefits and Usage Characteristics of Dynamic Arm Supports in Subjects with Neuromuscular Disorders. Sensors. 2020; 20(17):4864. https://doi.org/10.3390/s20174864
Chicago/Turabian StyleEssers, Johannes, Alessio Murgia, Anneliek Peters, and Kenneth Meijer. 2020. "Daily Life Benefits and Usage Characteristics of Dynamic Arm Supports in Subjects with Neuromuscular Disorders" Sensors 20, no. 17: 4864. https://doi.org/10.3390/s20174864
APA StyleEssers, J., Murgia, A., Peters, A., & Meijer, K. (2020). Daily Life Benefits and Usage Characteristics of Dynamic Arm Supports in Subjects with Neuromuscular Disorders. Sensors, 20(17), 4864. https://doi.org/10.3390/s20174864





