Spatial-Temporal Signals and Clinical Indices in Electrocardiographic Imaging (I): Preprocessing and Bipolar Potentials
Abstract
1. Introduction
2. Methods and Materials
2.1. Notation and ECGI Signal Preprocessing Stages
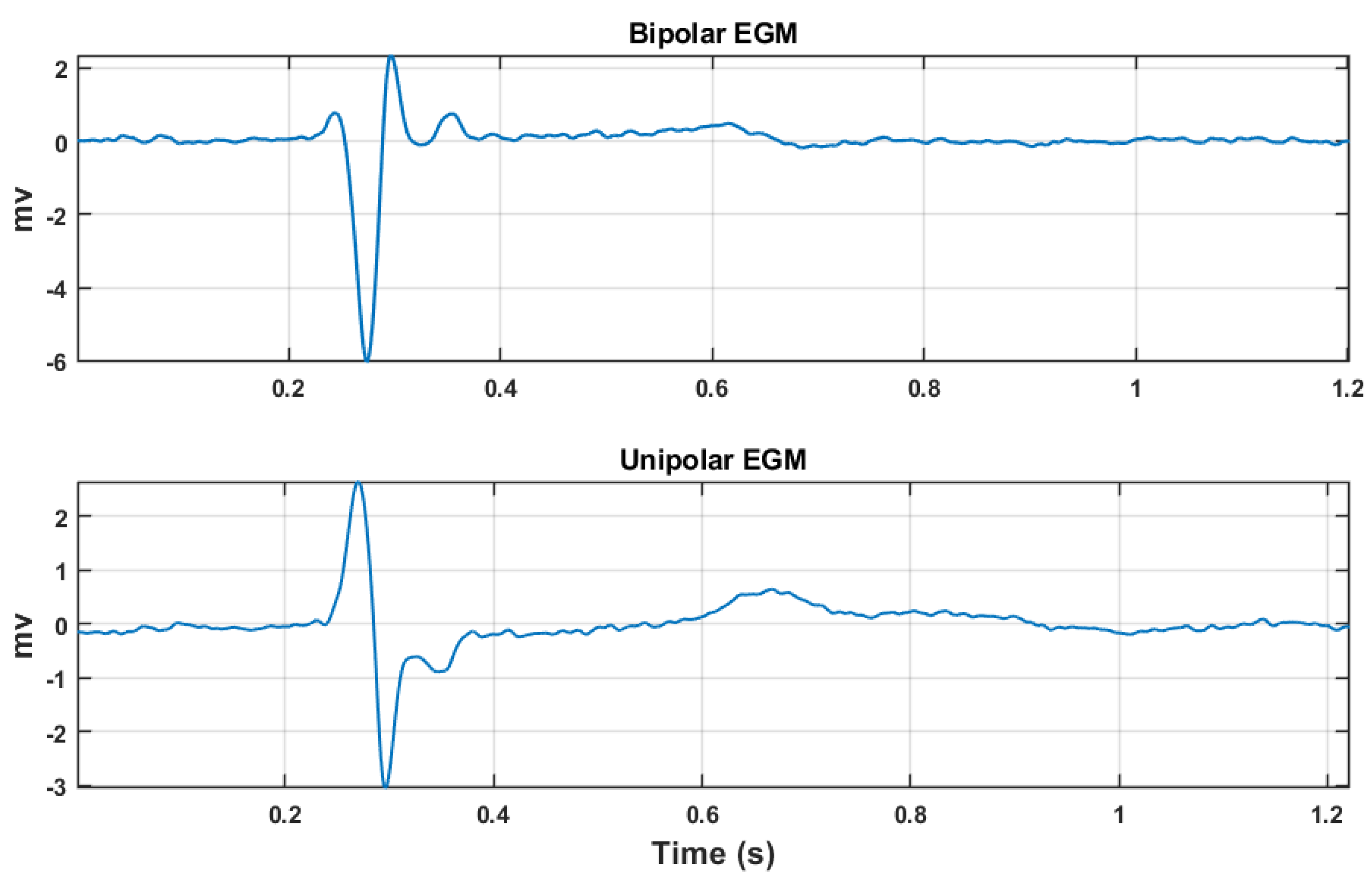
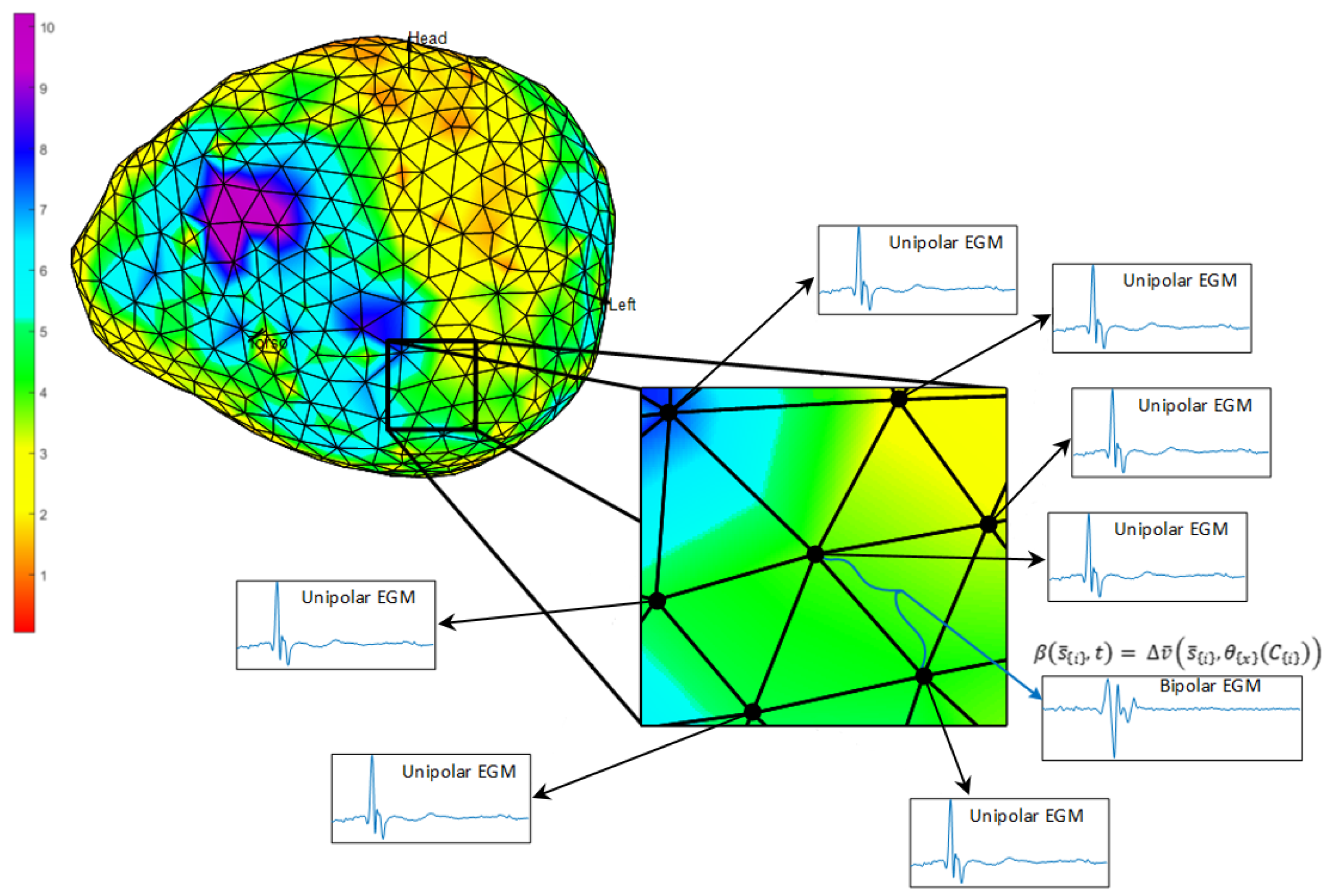
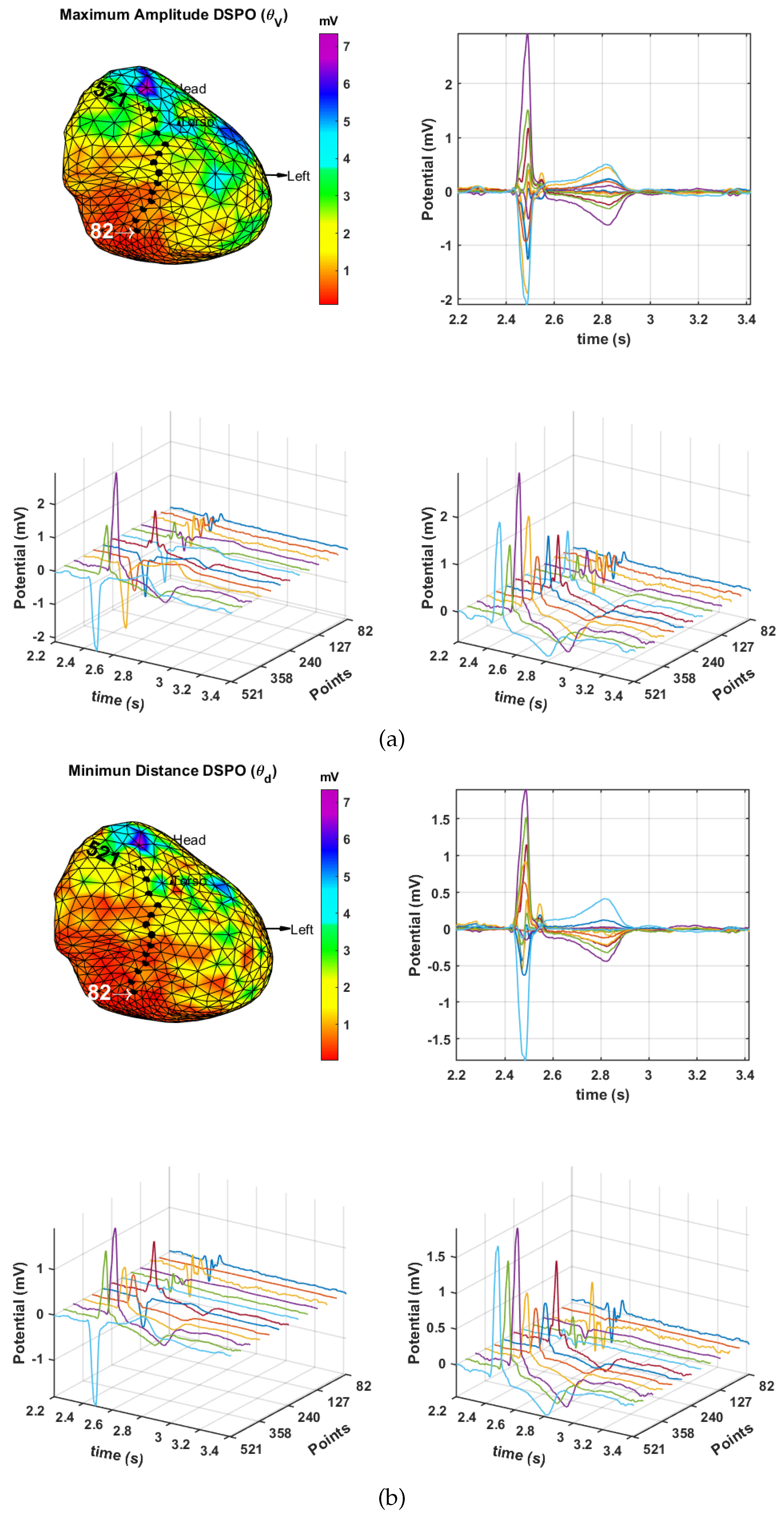
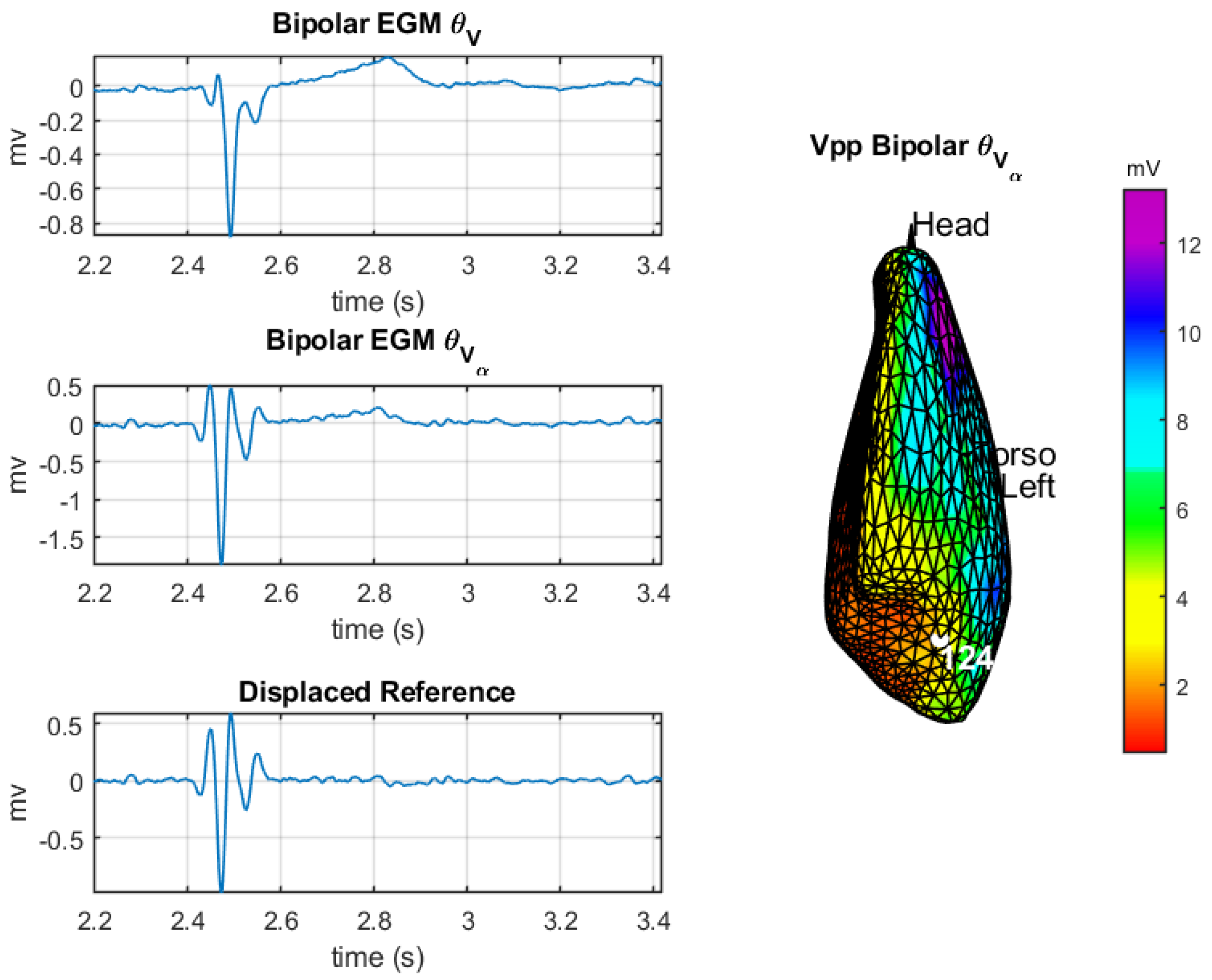
2.2. DSPO for Bipolar Potentials
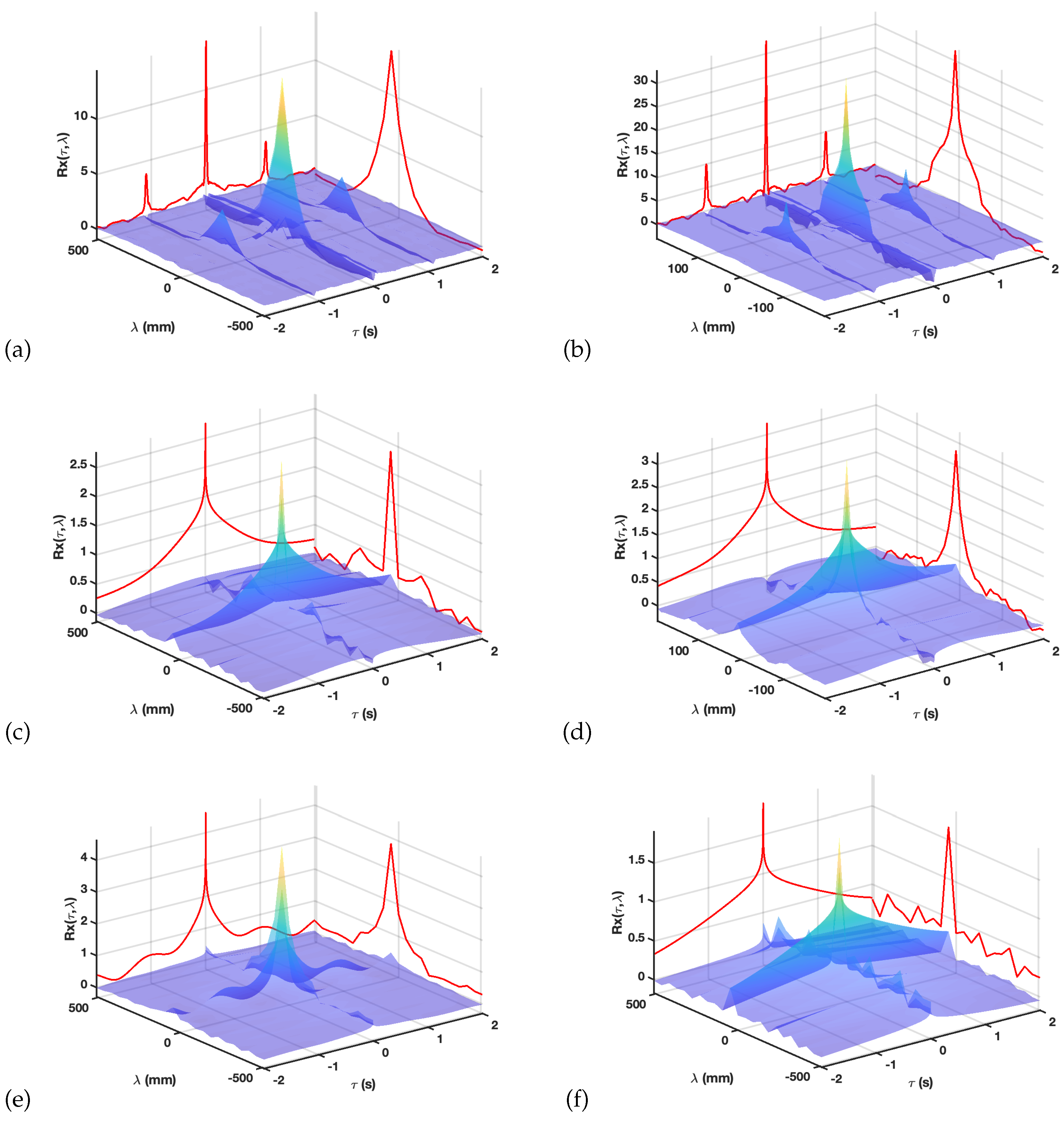
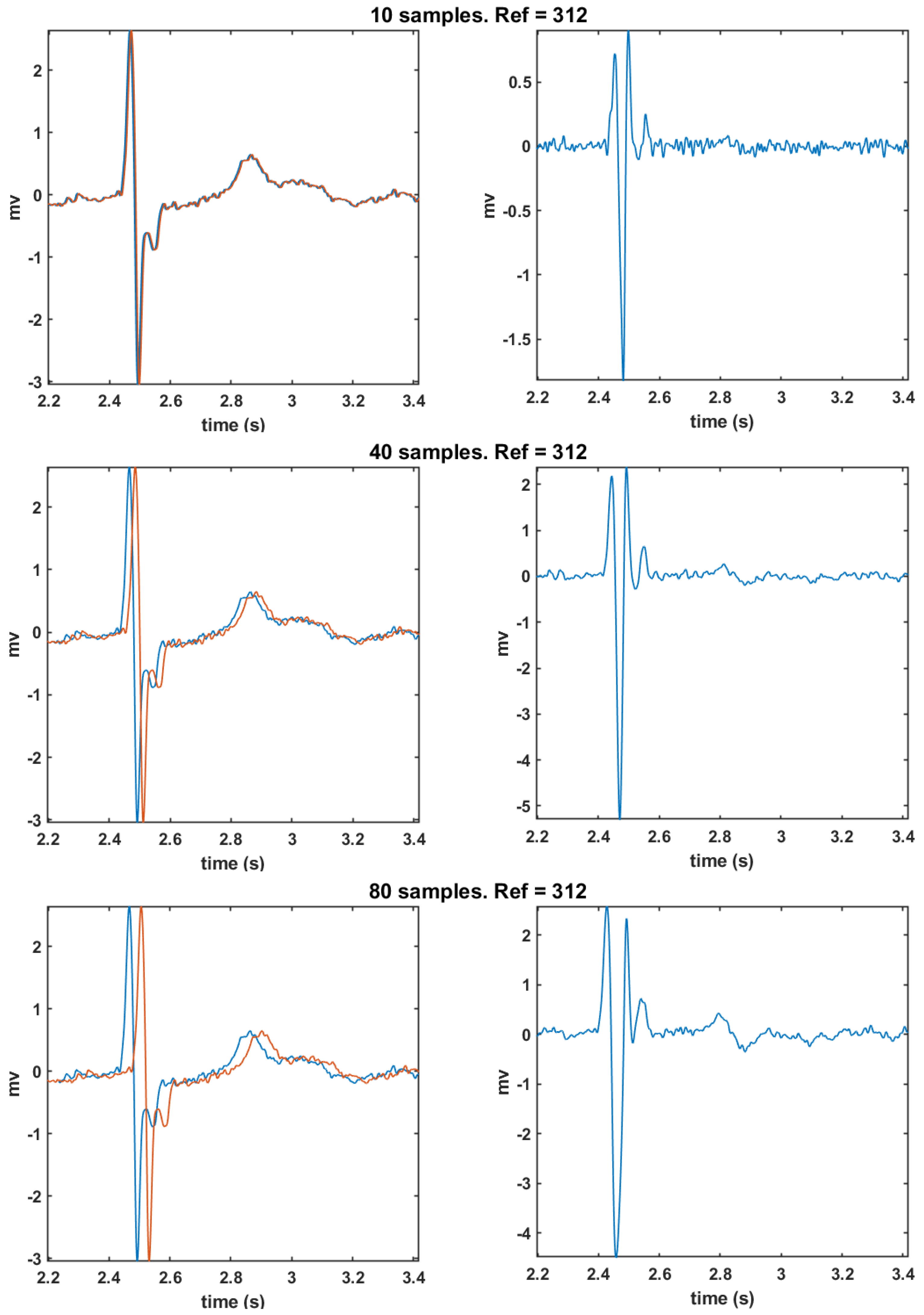
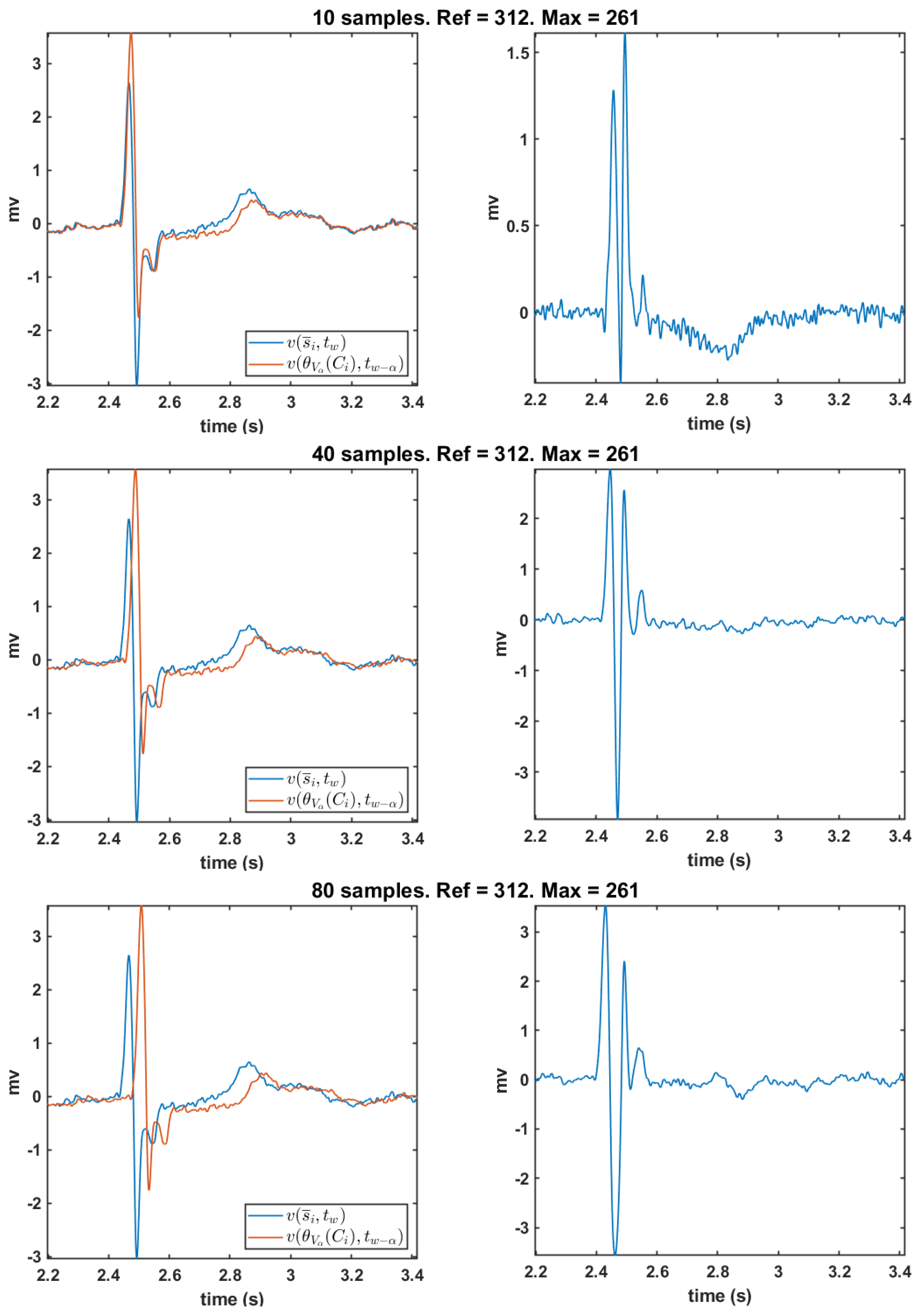
2.3. Time Derivative and Time Delay Analysis
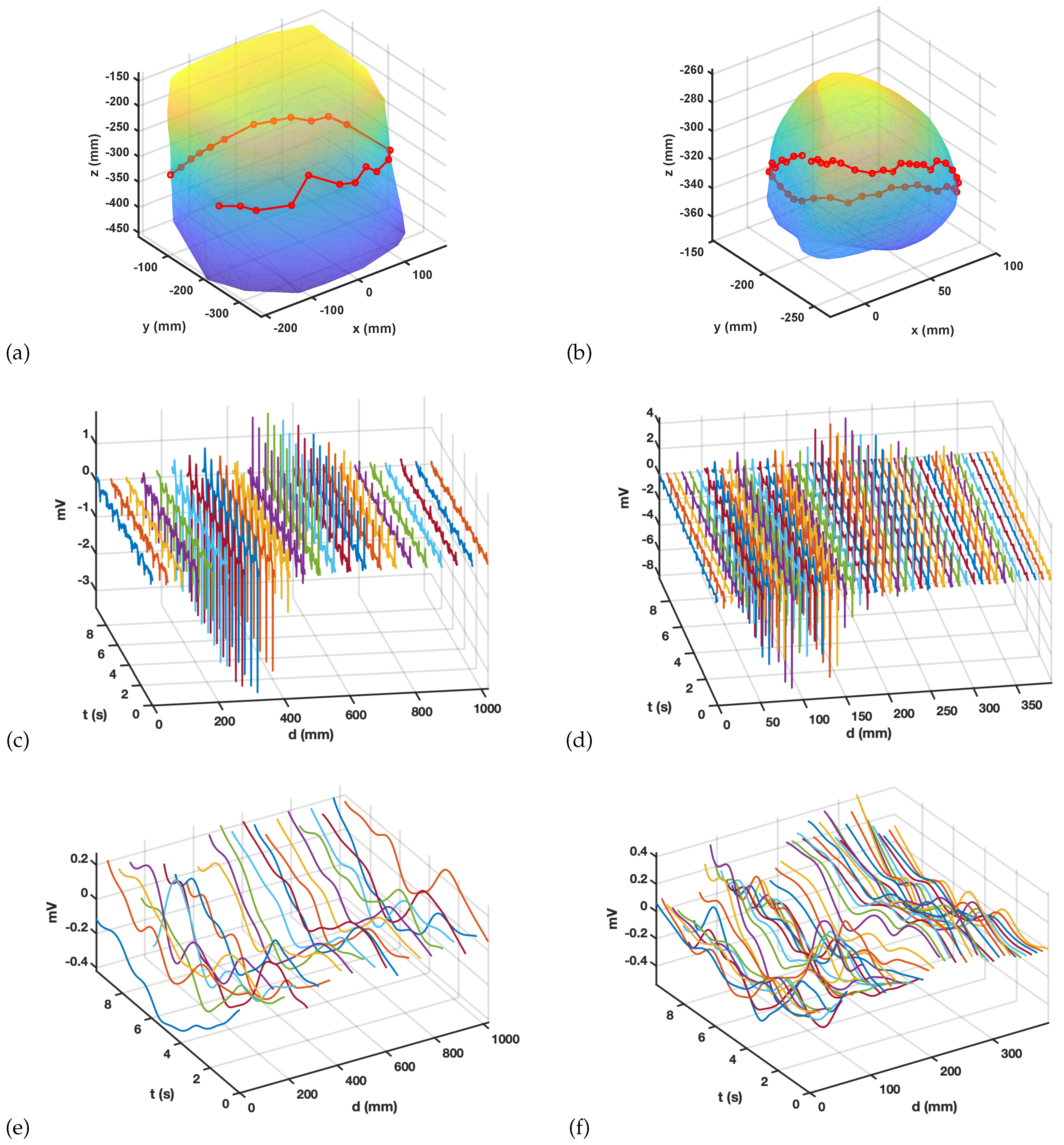
2.4. Data Sets
3. Experiments and Results
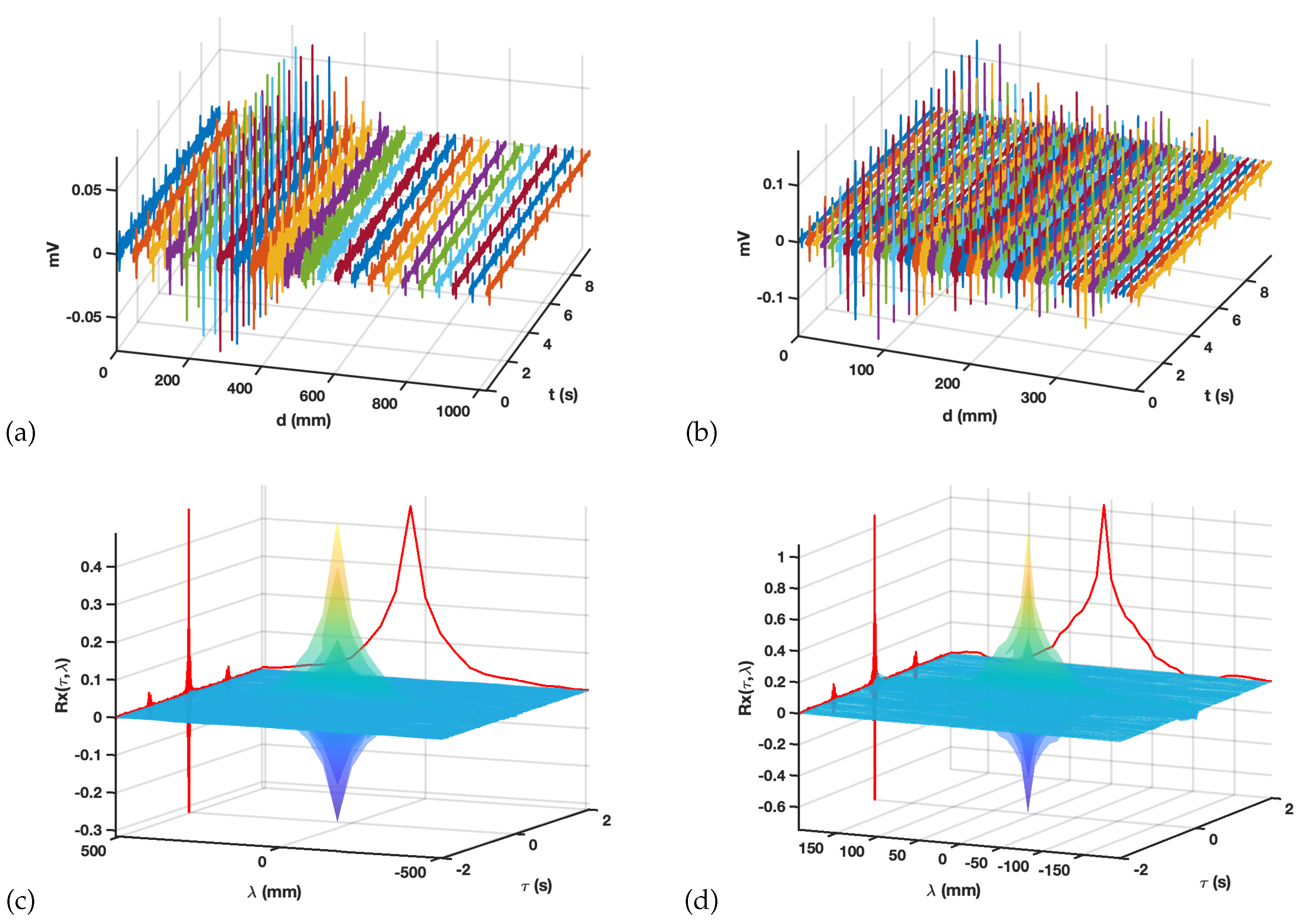
3.1. Preprocessing and Spatial-Temporal Correlations
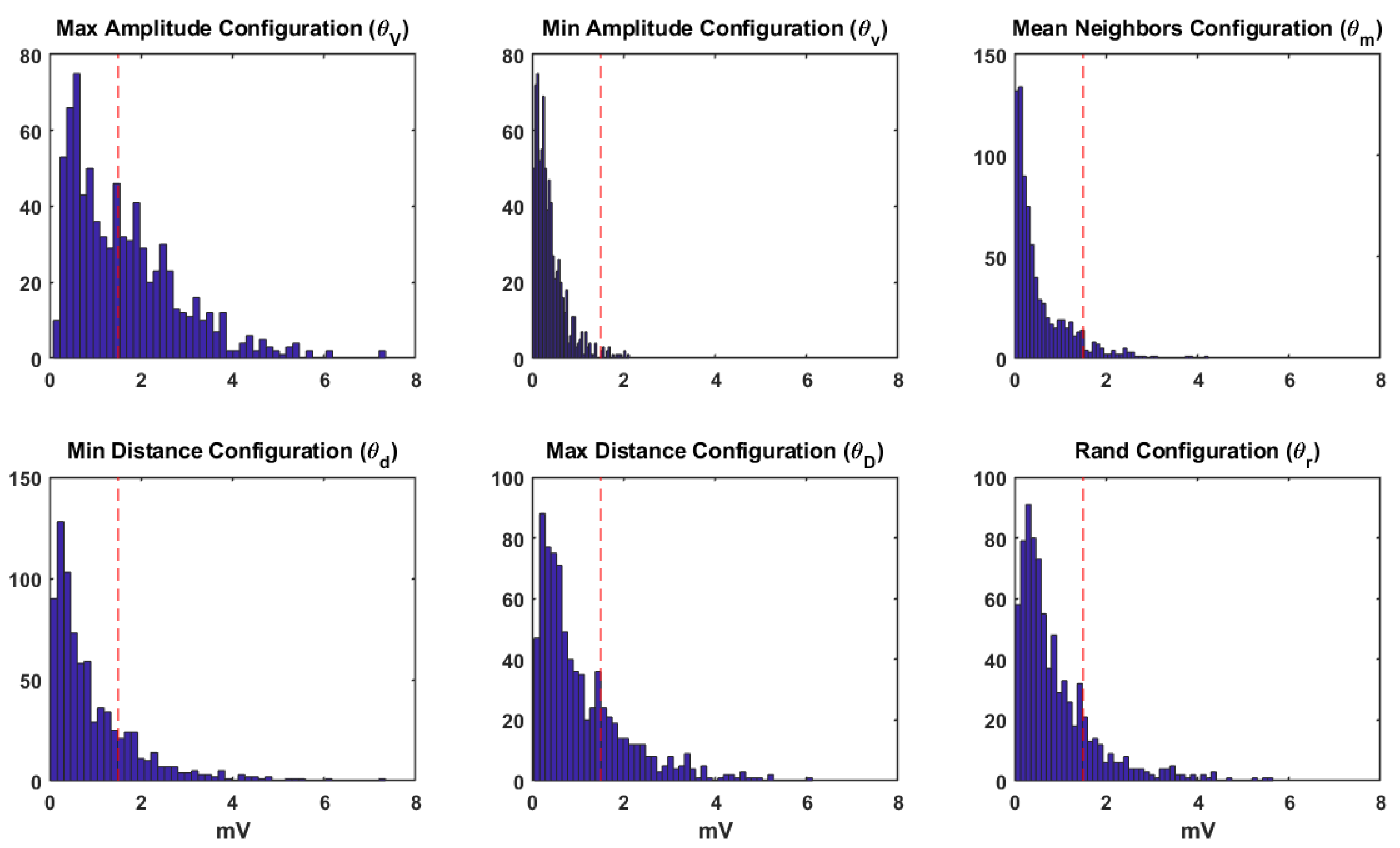
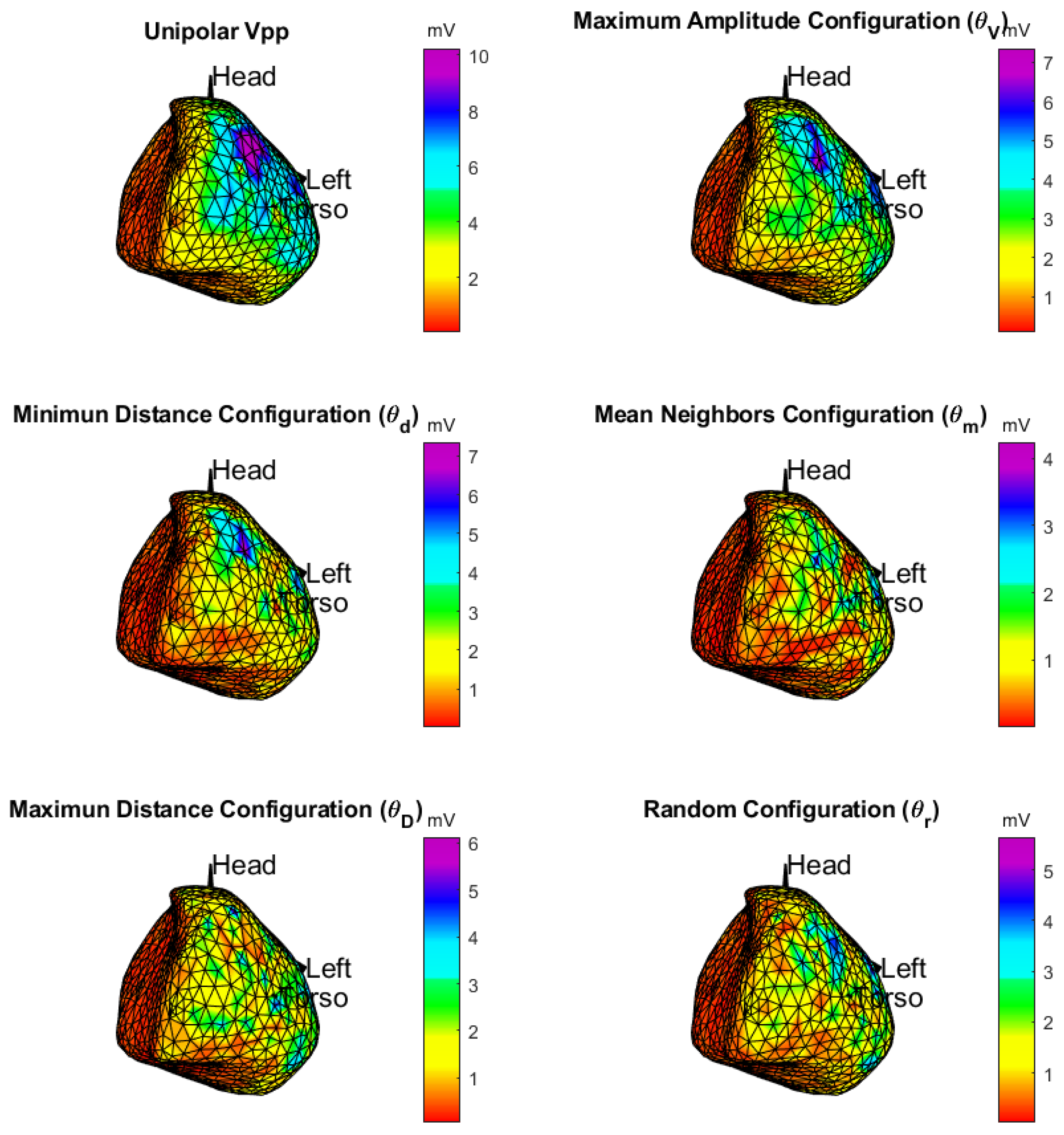
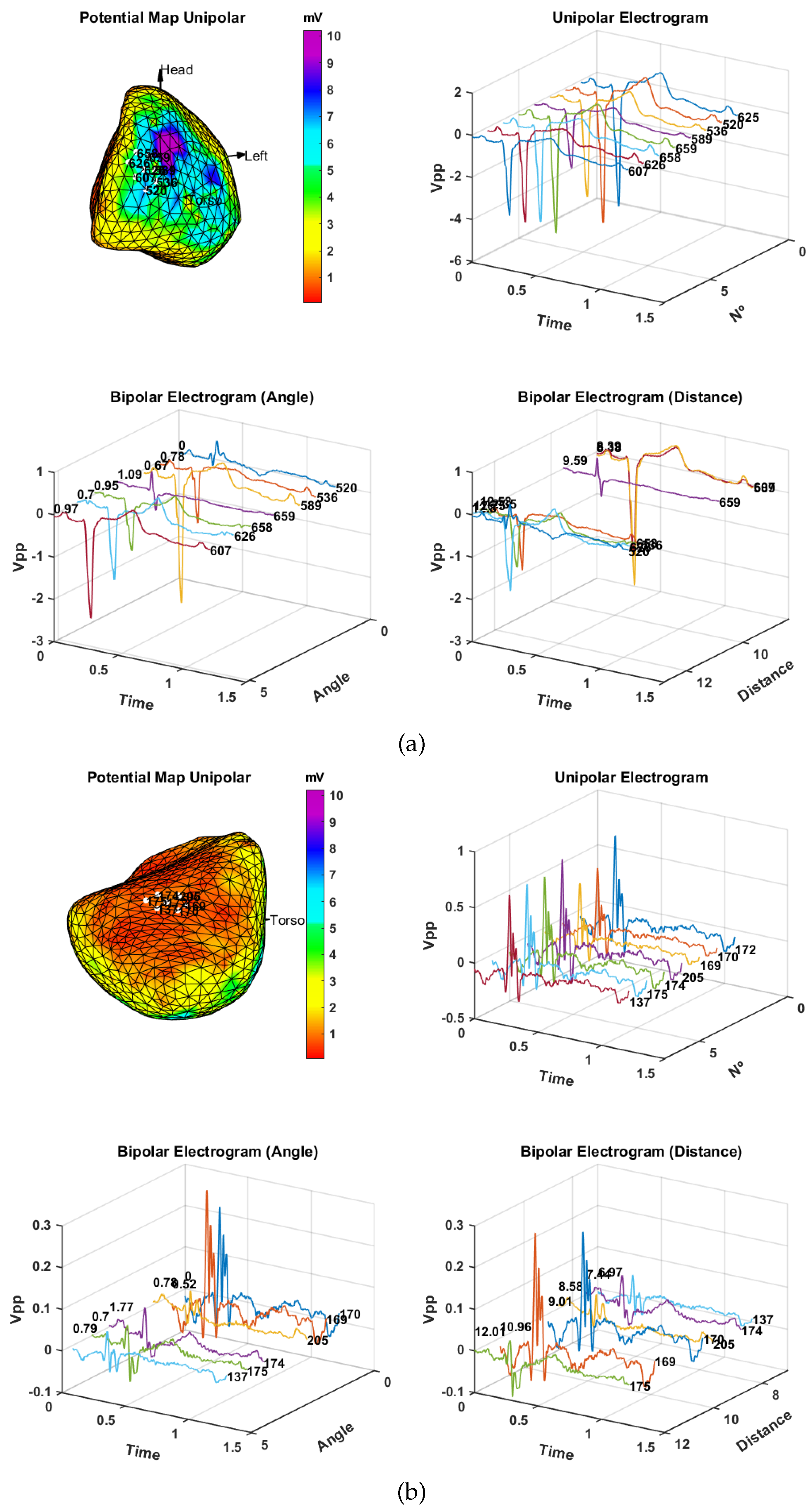
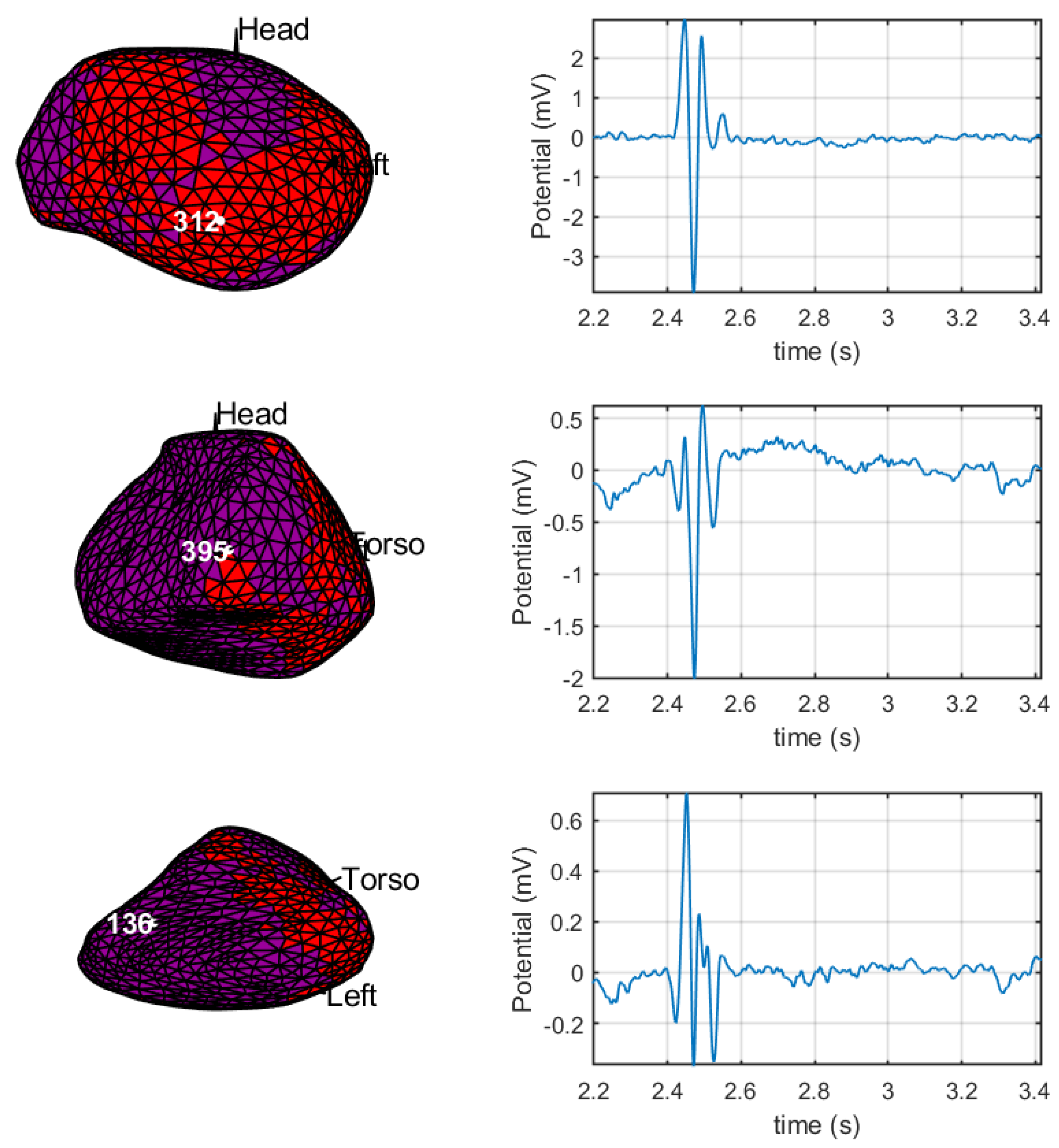
3.2. Spatial Consistency of Bipolar Potentials
3.3. Time Delay from Empirical Considerations
4. Discussion and Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Arunachalam, S.P. Novel Approaches for Quantitative Electrogram Analysis for Rotor Identification: Implications for Ablation in Patients with Atrial Fibrillation. Ph.D. Thesis, University of Minnesota, Minneapolis, MN, USA, 2017. [Google Scholar]
- Zeppenfeld, K.; Kiès, P.; Wijffels, M.C.; Bootsma, M.; van Erven, L.; Schalij, M.J. Identification of successful catheter ablation sites in patients with ventricular tachycardia based on electrogram characteristics during sinus rhythm. Heart Rhythm 2005, 2, 940–950. [Google Scholar] [CrossRef]
- Frontera, A.; Takigawa, M.; Martin, R.; Thompson, N.; Cheniti, G.; Massoullié, G.; Duchateau, J.; Wielandts, J.Y.; Teijeira, E.; Kitamura, T.; et al. Electrogram signature of specific activation patterns: Analysis of atrial tachycardias at high-density endocardial mapping. Heart Rhythm 2018, 15, 28–37. [Google Scholar] [CrossRef]
- Tokuda, M.; Tedrow, U.B.; Inada, K.; Reichlin, T.; Michaud, G.F.; John, R.M.; Epstein, L.M.; Stevenson, W.G. Direct comparison of adjacent endocardial and epicardial electrograms: Implications for substrate mapping. J. Am. Heart Assoc. 2013, 2, e000215. [Google Scholar] [CrossRef] [PubMed]
- Rudy, Y.; Messinger-Rapport, B. The inverse problem in electrocardiography: Solutions in terms of epicardial potentials. Crit. Rev. Biomed. Eng. 1988, 16, 215–268. [Google Scholar] [PubMed]
- Rudy, Y.; Taccardi, B. Noninvasive imaging and catheter imaging of potentials, electrograms, and isochrones on the ventricular surfaces. J. Electrocardiol. 1998, 30, 19–23. [Google Scholar] [CrossRef]
- Rudy, Y. Electrocardiographic imaging: A noninvasive imaging modality for characterization of intramural myocardial activation. J. Electrocardiol. 1999, 32, 1–6. [Google Scholar] [CrossRef]
- Ghanem, R.; Burnes, J.; Waldo, A.; Rudy, Y. Electrocardiographic Imaging: Noninvasive Reconstruction of Epicardial Measures of Dispersion of Repolarization. Biomed. Tech. Eng. 2001, 46, 201–203. [Google Scholar] [CrossRef]
- Burnes, J.E.; Taccardi, B.; Rudy, Y. A noninvasive imaging modality for cardiac arrhythmias. Circulation 2000, 102, 2152–2158. [Google Scholar] [CrossRef]
- Ramanathan, C.; Jia, P.; Ghanem, R.; Calvetti, D.; Rudy, Y. Noninvasive electrocardiographic imaging (ECGI): Application of the generalized minimal residual (GMRes) method. Ann. Biomed. Eng. 2003, 31, 981–994. [Google Scholar] [CrossRef]
- Ramanathan, C.; Ghanem, R.N.; Jia, P.; Ryu, K.; Rudy, Y. Noninvasive electrocardiographic imaging for cardiac electrophysiology and arrhythmia. Nat. Med. 2004, 10, 422–428. [Google Scholar] [CrossRef]
- Ramanathan, C.; Jia, P.; Ghanem, R.; Ryu, K.; Rudy, Y. Activation and repolarization of the normal human heart under complete physiological conditions. Proc. Natl. Acad. Sci. USA 2006, 103, 6309–6314. [Google Scholar] [CrossRef] [PubMed]
- Lim, H.S.; Zellerhoff, S.; Derval, N.; Denis, A.; Yamashita, S.; Berte, B.; Mahida, S.; Hooks, D.; Aljefairi, N.; Shah, A.J.; et al. Noninvasive mapping to guide atrial fibrillation ablation. Card. Electrophysiol. Clin. 2015, 7, 89–98. [Google Scholar] [CrossRef] [PubMed]
- Dubois, R.; Shah, A.J.; Hocini, M.; Denis, A.; Derval, N.; Cochet, H.; Sacher, F.; Bear, L.; Duchateau, J.; Jais, P.; et al. Non-invasive cardiac mapping in clinical practice: Application to the ablation of cardiac arrhythmias. J. Electrocardiol. 2015, 48, 966–974. [Google Scholar] [CrossRef] [PubMed]
- Rudy, Y. Noninvasive ECG imaging (ECGI): Mapping the arrhythmic substrate of the human heart. Int. J. Cardiol. 2017, 237, 13–14. [Google Scholar] [CrossRef] [PubMed]
- Rudy, Y. Role for electrocardiographic imaging in cardiac resynchronization therapy? Heart Rhythm 2018, 15, 1070–1071. [Google Scholar] [CrossRef] [PubMed]
- Duchateau, J.; Sacher, F.; Pambrun, T.; Derval, N.; Chamorro-Servent, J.; Denis, A.; Ploux, S.; Hocini, M.; Jaïs, P.; Bernus, O.; et al. Performance and limitations of noninvasive cardiac activation mapping. Heart Rhythm 2019, 16, 435–442. [Google Scholar] [CrossRef]
- Wang, D.; Kirby, R.; Johnson, C. Resolution Strategies for the Finite-Element-Based Solution of the ECG Inverse Problem. IEEE Trans. Biomed. Eng. 2010, 57, 220–237. [Google Scholar] [CrossRef]
- Cluitmans, M.; Brooks, D.H.; MacLeod, R.; Dössel, O.; Guillem, M.S.; van Dam, P.M.; Svehlikova, J.; He, B.; Sapp, J.; Wang, L.; et al. Validation and Opportunities of Electrocardiographic Imaging: From Technical Achievements to Clinical Applications. Front. Physiol. 2018, 9, 1305. [Google Scholar] [CrossRef]
- Duchateau, J.; Coudière, Y.; Haïssaguerre, M.; Dubois, R. Virtual Bipolar and Laplacian Electrodes for Activation Map Construction in ECGi. In Proceedings of the 2015 Computing in Cardiology Conference (CinC), Nice, France, 6–9 September 2015; pp. 949–952. [Google Scholar]
- Shenasa, M.; Hindricks, G.; Callans, D.J.; Miller, J.M.; Josephson, M.E. Cardiac Mapping; John Wiley & Sons: Oxford, UK, 2019. [Google Scholar]
- Caulier-Cisterna, R.; Blanco-Velasco, M.; Goya-Esteban, R.; Muñoz-Romero, S.; Sanromán-Junquera, M.; García-Alberola, A.; Rojo-Álvarez, J. Spatial-temporal Signals and Clinical Indices in Electrocardiographic Imaging (II): Electrogram Clustering, Fragmentation, and T-wave Alternans. Sensors 2020, 20, 3070. [Google Scholar]
- Rojo-Álvarez, J.L.; Goya-Esteban, R.; Muñoz-Romero, S.; García-Alberola, A.; Melgarejo-Meseguer, F.M.; Blanco-Velasco, M. T-Wave Alternans Analysis with Electrocardiographic Imaging. In Proceedings of the 2018 Computing in Cardiology Conference (CinC), Maastricht, The Netherlands, 23–26 September 2018; pp. 1–4. [Google Scholar]
- Villabriga, B.; Romaniega, T. Manual de Arritmias y Electrofisiología Cardíaca; Pulso Ediciones: Madrid, Spain, 2010. [Google Scholar]
- Josephson, M.E. Josephson’s Clinical Cardiac Electrophysiology; Wolters Kluwer: Baltimore, MD, USA, 2016. [Google Scholar]
- Mont, L. Manual de Electrofisiología clínica y Ablación; Marge Médica Books: Barcelona, Spain, 2011. [Google Scholar]
- Rudy, Y.; Ramanathan, C.; Ghosh, S.; Zipes, D. Noninvasive electrocardiographic imaging (ECGI): Methodology and excitation of the normal human heart. In Cardiac Electrophysiology: From Cell to Bedside, 5th ed.; Saunders Elsevier: Philadelphia, PA, USA, 2009; pp. 467–472. [Google Scholar]
- Oster, H.S.; Taccardi, B.; Lux, R.L.; Ershler, P.R.; Rudy, Y. Electrocardiographic imaging: Noninvasive characterization of intramural myocardial activation from inverse-reconstructed epicardial potentials and electrograms. Circulation 1998, 97, 1496–1507. [Google Scholar] [CrossRef]
- Burnes, J.E.; Taccardi, B.; MacLeod, R.S.; Rudy, Y. Noninvasive ECG imaging of electrophysiologically abnormal substrates in infarcted hearts: A model study. Circulation 2000, 101, 533–540. [Google Scholar] [CrossRef] [PubMed]
- Ghanem, R.N.; Jia, P.; Ramanathan, C.; Ryu, K.; Markowitz, A.; Rudy, Y. Noninvasive electrocardiographic imaging (ECGI): Comparison to intraoperative mapping in patients. Heart Rhythm 2005, 2, 339–354. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Cuculich, P.S.; Zhang, J.; Desouza, K.A.; Vijayakumar, R.; Chen, J.; Faddis, M.N.; Lindsay, B.D.; Smith, T.W.; Rudy, Y. Noninvasive electroanatomic mapping of human ventricular arrhythmias with electrocardiographic imaging. Sci. Transl. Med. 2011, 3, 98ra84. [Google Scholar] [CrossRef] [PubMed]
- Berger, T.; Fischer, G.; Pfeifer, B.; Modre, R.; Hanser, F.; Trieb, T.; Roithinger, F.X.; Stuehlinger, M.; Pachinger, O.; Tilg, B.; et al. Single-beat noninvasive imaging of cardiac electrophysiology of ventricular pre-excitation. J. Am. Coll. Cardiol. 2006, 48, 2045–2052. [Google Scholar] [CrossRef] [PubMed]
- Berger, T.; Pfeifer, B.; Hanser, F.F.; Hintringer, F.; Fischer, G.; Netzer, M.; Trieb, T.; Stuehlinger, M.; Dichtl, W.; Baumgartner, C.; et al. Single-beat noninvasive imaging of ventricular endocardial and epicardial activation in patients undergoing CRT. PLoS ONE 2011, 6, e16255. [Google Scholar] [CrossRef] [PubMed]
- Andrews, C.M.; Srinivasan, N.T.; Rosmini, S.; Bulluck, H.; Orini, M.; Jenkins, S.; Pantazis, A.; McKenna, W.J.; Moon, J.; Lambiase, P.D.; et al. Electrical and Structural Substrate of Arrhythmogenic Right Ventricular Cardiomyopathy Determined Using Noninvasive Electrocardiographic Imaging and Late Gadolinium Magnetic Resonance Imaging. Circ. Arrhythm. Electrophysiol. 2017, 10, e005105. [Google Scholar] [CrossRef]
- Cuculich, P.; Schill, M.; Kashani, R.; Mutic, S.; Lang, A.; Cooper, D.; Faddis, M.; Gleva, M.; Noheria, A.; Smith, T.; et al. Noninvasive Cardiac Radiation for Ablation of Ventricular Tachycardia. N. Engl. J. Med. 2017, 377, 2325–2336. [Google Scholar] [CrossRef]
- Bear, L.R.; LeGrice, I.J.; Sands, G.B.; Lever, N.A.; Loiselle, D.S.; Paterson, D.J.; Cheng, L.K.; Smaill, B.H. How Accurate Is Inverse Electrocardiographic Mapping?: A Systematic In Vivo Evaluation. Circ. Arrhythm. Electrophysiol. 2018, 11, e006108. [Google Scholar] [CrossRef]
- Andrews, C.; Cupps, B.P.; Pasque, M.K.; Rudy, Y. Electromechanics of the Normal Human Heart In Situ. Circ. Arrhythmia Electrophysiol. 2019, 12, e007484. [Google Scholar] [CrossRef]
- Vijayakumar, R.; Silva, J.N.; Desouza, K.A.; Abraham, R.L.; Strom, M.; Sacher, F.; Van Hare, G.F.; Haïssaguerre, M.; Roden, D.M.; Rudy, Y. Electrophysiologic substrate in congenital long QT syndrome: Noninvasive mapping with electrocardiographic imaging (ECGI). Circulation 2014, 130, 1936–1943. [Google Scholar] [CrossRef]
- Zhang, J.; Cooper, D.H.; Desouza, K.A.; Cuculich, P.S.; Woodard, P.K.; Smith, T.W.; Rudy, Y. Electrophysiologic Scar Substrate in Relation to VT: Noninvasive High-Resolution Mapping and Risk Assessment with ECGI. PACE 2016, 39, 781–791. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Hocini, M.; Strom, M.; Cuculich, P.S.; Cooper, D.H.; Sacher, F.; Haïssaguerre, M.; Rudy, Y. The Electrophysiological Substrate of Early Repolarization Syndrome: Noninvasive Mapping in Patients. JACC. Clin. Electrophysiol. 2017, 3, 894–904. [Google Scholar] [CrossRef] [PubMed]
- Everss-Villalba, E.; Melgarejo-Meseguer, F.M.; Blanco-Velasco, M.; Gimeno-Blanes, F.J.; Sala-Pla, S.; Rojo-Álvarez, J.L.; García-Alberola, A. Noise Maps for Quantitative and Clinical Severity Towards Long-Term ECG Monitoring. Sensors 2017, 17, 2448. [Google Scholar] [CrossRef] [PubMed]
- Desjardins, B.; Crawford, T.; Good, E.; Oral, H.; Chugh, A.; Pelosi, F.; Morady, F.; Bogun, F. Infarct architecture and characteristics on delayed enhanced magnetic resonance imaging and electroanatomic mapping in patients with postinfarction ventricular arrhythmia. Heart Rhythm 2009, 6, 644–651. [Google Scholar] [CrossRef] [PubMed]
- Josephson, M.E. Clinical Cardiac Electrophysiology: Techniques and Interpretations; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2008. [Google Scholar]
- Ellenbogen, K.A.; Tung, R.; Frankel, D. Essential Concepts of Electrophysiology through Case Studies: Intracardiac EGMs; Kenneth, A., Ellenbogen, M.D., Eds.; CardioText: Minneapolis, MN, USA, 2015. [Google Scholar] [CrossRef]
- Maheshwari, S.; Acharyya, A.; Puddu, P.E.; Mazomenos, E.B.; Leekha, G.; Maharatna, K.; Schiariti, M. An automated algorithm for online detection of fragmented QRS and identification of its various morphologies. J. R. Soc. Interface 2013, 10, 20130761. [Google Scholar] [CrossRef]
- Rudy, Y. The electrocardiogram and its relationship to excitation of the heart. Dev. Cardiovasc. Med. 1995, 151, 201. [Google Scholar]
- Oosterhoff, P.; Meijborg, V.M.; van Dam, P.M.; van Dessel, P.F.; Belterman, C.N.; Streekstra, G.J.; de Bakker, J.M.; Coronel, R.; Oostendorp, T.F. Experimental Validation of Noninvasive Epicardial and Endocardial Activation Imaging. Circ. Arrhythm. Electrophysiol. 2016, 9, e004104. [Google Scholar] [CrossRef][Green Version]
- Rudy, Y.; Plonsey, R. A comparison of volume conductor and source geometry effects on body surface and epicardial potentials. Circulation 1980, 46, 283–291. [Google Scholar] [CrossRef]
- Ghosh, S.; Rudy, Y. Application of L1-norm regularization to epicardial potential solution of the inverse electrocardiography problem. Ann. Biomed. Eng. 2009, 37, 902–912. [Google Scholar] [CrossRef]
- Oster, H.S.; Rudy, Y. The use of temporal information in the regularization of the inverse problem of electrocardiography. IEEE Trans. Biomed. Eng. 1992, 39, 65–75. [Google Scholar] [CrossRef]
- Cantwell, C.D.; Roney, C.H.; Ng, F.S.; Siggers, J.H.; Sherwin, S.; Peters, N.S. Techniques for automated local activation time annotation and conduction velocity estimation in cardiac mapping. Comput. Biol. Med. 2015, 65, 229–242. [Google Scholar] [CrossRef]
- Liang, J.J.; Callans, D.J. Electrophysiologic Testing and Cardiac Mapping. In Cardiac Arrhythmias, Pacing and Sudden Death; Springer: Cham, Switzerland, 2017; pp. 75–86. [Google Scholar]
- Takahashi, Y.; Iwai, S.; Yamashita, S.; Masumura, M.; Suzuki, M.; Yabe, K.; Sato, Y.; Hirao, K.; Isobe, M. Novel mapping technique for localization of focal and reentrant activation during atrial fibrillation. J. Cardiovasc. Electrophysiol. 2017, 28, 375–382. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.W.; Jia, P.; Ershler, P.R.; Taccardi, B.; Lux, R.L.; Khoury, D.S.; Rudy, Y. Noncontact endocardial mapping: Reconstruction of electrograms and isochrones from intracavitary probe potentials. J. Cardiovasc. Electrophysiol. 1997, 8, 415–431. [Google Scholar] [CrossRef] [PubMed]
- Schilling, R.J.; Peters, N.S.; Davies, D.W. Simultaneous endocardial mapping in the human left ventricle using a noncontact catheter: Comparison of contact and reconstructed electrograms during sinus rhythm. Circulation 1998, 98, 887–898. [Google Scholar] [CrossRef] [PubMed]
- Harlev, D.; Greenfield, P.; Amariglio, L. Cardiac mapping with catheter shape information. US Patent 9,526,434, 27 December 2016. [Google Scholar]
- Yeo, C.; Lemery, R.; Green, M. Contributions of Noncontact Mapping to the Understanding of Normal Atrial Activation. In Interatrial Block and Supraventricular Arrhythmias: Clinical Implications of Bayés’ Syndrome; Cardiotext: Minneapolis, MN, USA, 2017. [Google Scholar]
| DSPO | ||
|---|---|---|
| Amplitude | ||
| Random | ||
| Mean | ||
| Distance | ||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Caulier-Cisterna, R.; Sanromán-Junquera, M.; Muñoz-Romero, S.; Blanco-Velasco, M.; Goya-Esteban, R.; García-Alberola, A.; Rojo-Álvarez, J.L. Spatial-Temporal Signals and Clinical Indices in Electrocardiographic Imaging (I): Preprocessing and Bipolar Potentials. Sensors 2020, 20, 3131. https://doi.org/10.3390/s20113131
Caulier-Cisterna R, Sanromán-Junquera M, Muñoz-Romero S, Blanco-Velasco M, Goya-Esteban R, García-Alberola A, Rojo-Álvarez JL. Spatial-Temporal Signals and Clinical Indices in Electrocardiographic Imaging (I): Preprocessing and Bipolar Potentials. Sensors. 2020; 20(11):3131. https://doi.org/10.3390/s20113131
Chicago/Turabian StyleCaulier-Cisterna, Raúl, Margarita Sanromán-Junquera, Sergio Muñoz-Romero, Manuel Blanco-Velasco, Rebeca Goya-Esteban, Arcadi García-Alberola, and José Luis Rojo-Álvarez. 2020. "Spatial-Temporal Signals and Clinical Indices in Electrocardiographic Imaging (I): Preprocessing and Bipolar Potentials" Sensors 20, no. 11: 3131. https://doi.org/10.3390/s20113131
APA StyleCaulier-Cisterna, R., Sanromán-Junquera, M., Muñoz-Romero, S., Blanco-Velasco, M., Goya-Esteban, R., García-Alberola, A., & Rojo-Álvarez, J. L. (2020). Spatial-Temporal Signals and Clinical Indices in Electrocardiographic Imaging (I): Preprocessing and Bipolar Potentials. Sensors, 20(11), 3131. https://doi.org/10.3390/s20113131







