Electrochemical Screening and Evaluation of Lamiaceae Plant Species from South Africa with Potential Tyrosinase Activity
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Materials
2.2. Preparation of Plant Extracts
2.3. Chemicals and Reagents
2.4. Apparatus
2.5. Cyclic Voltammetry Measurement
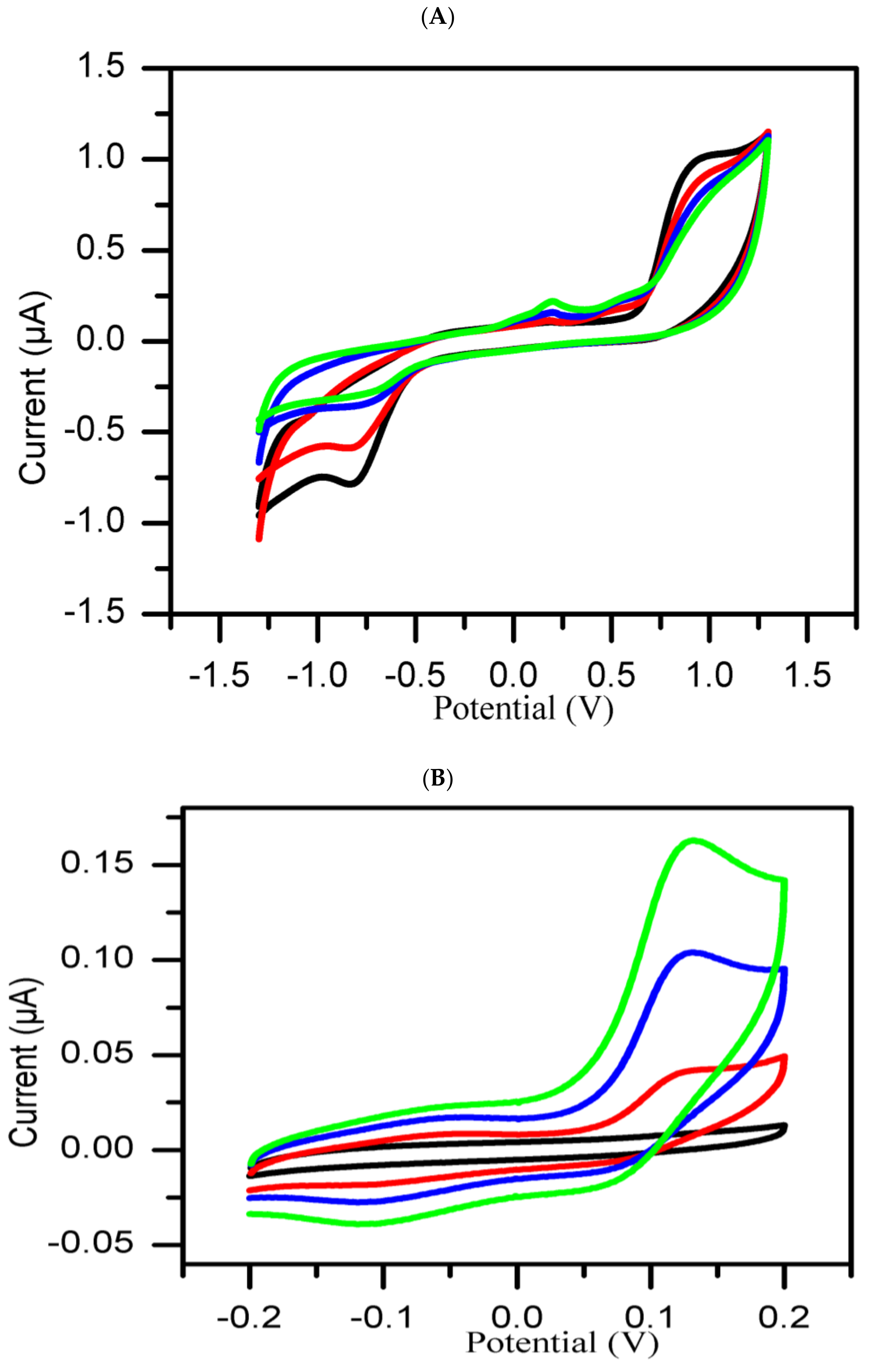
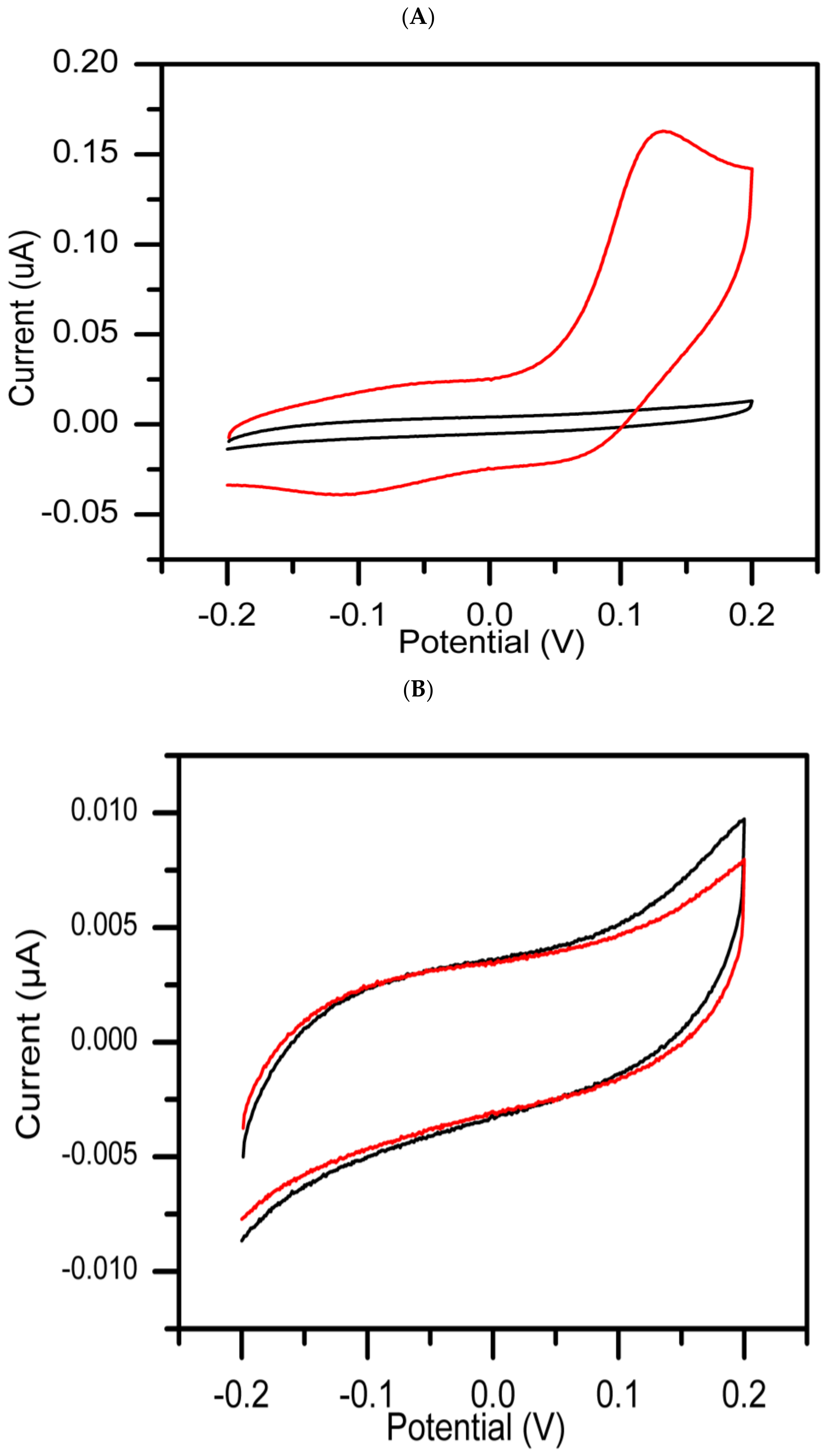
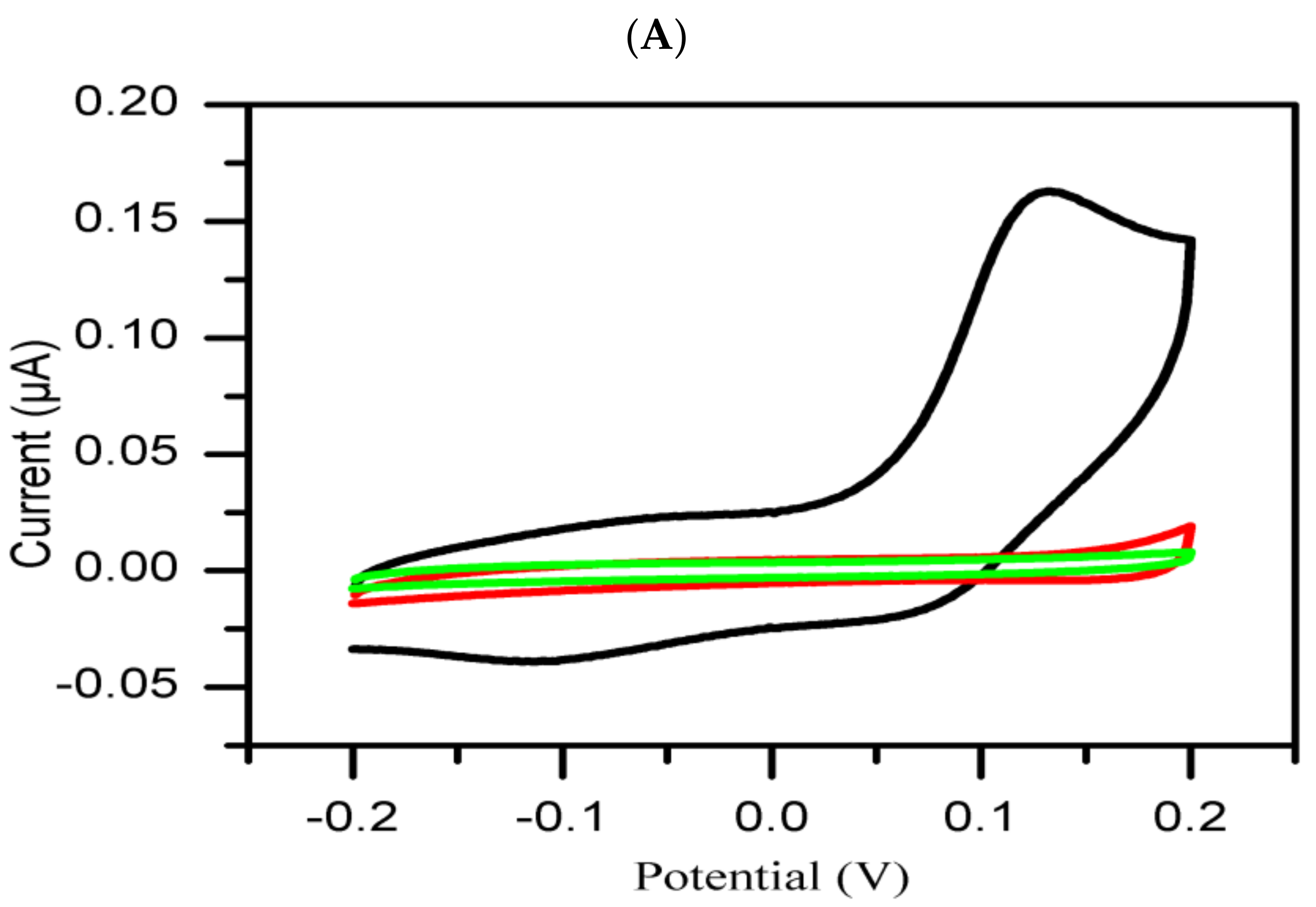
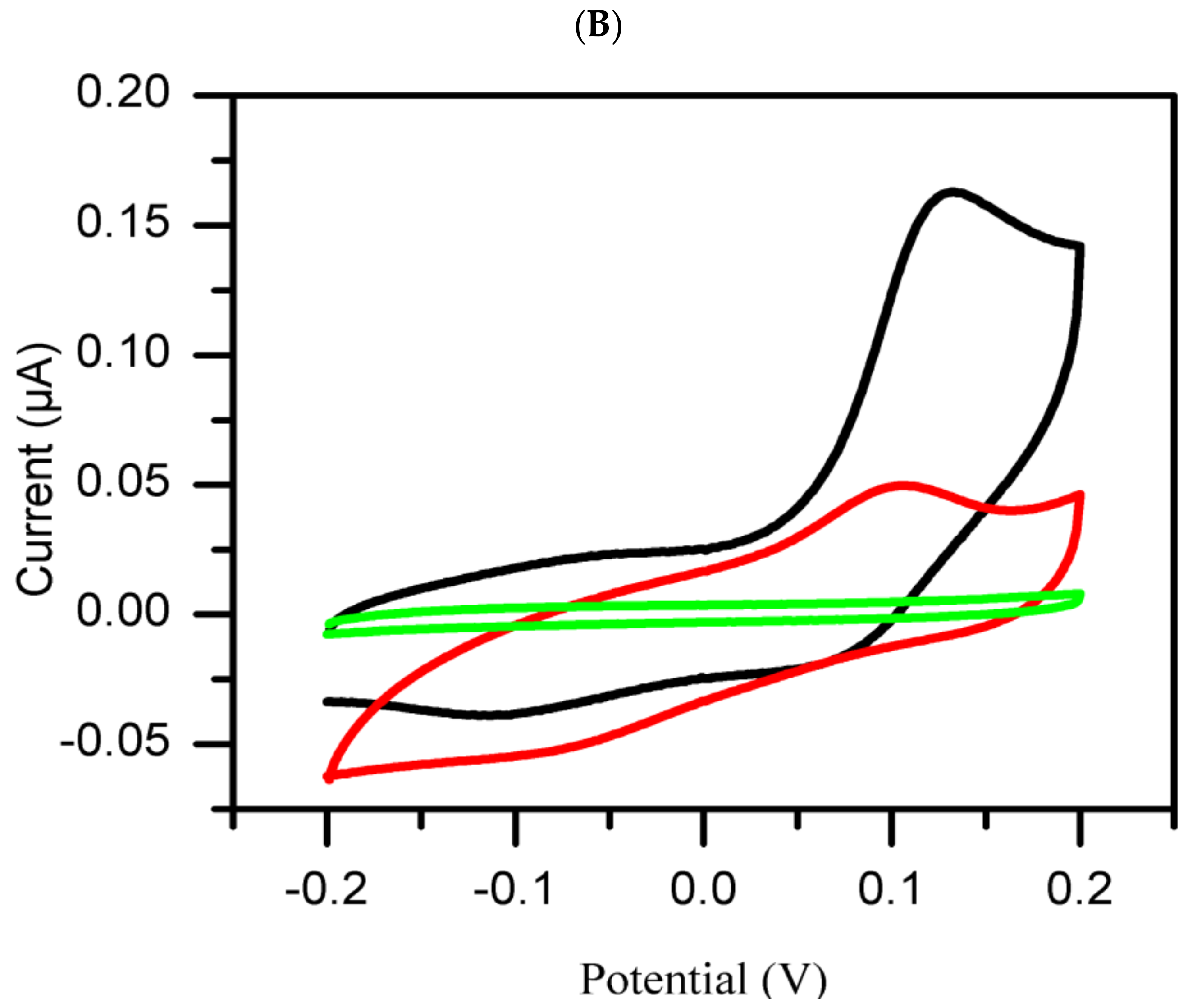
3. Results and Discussion
Cyclic Voltammetry Measurement
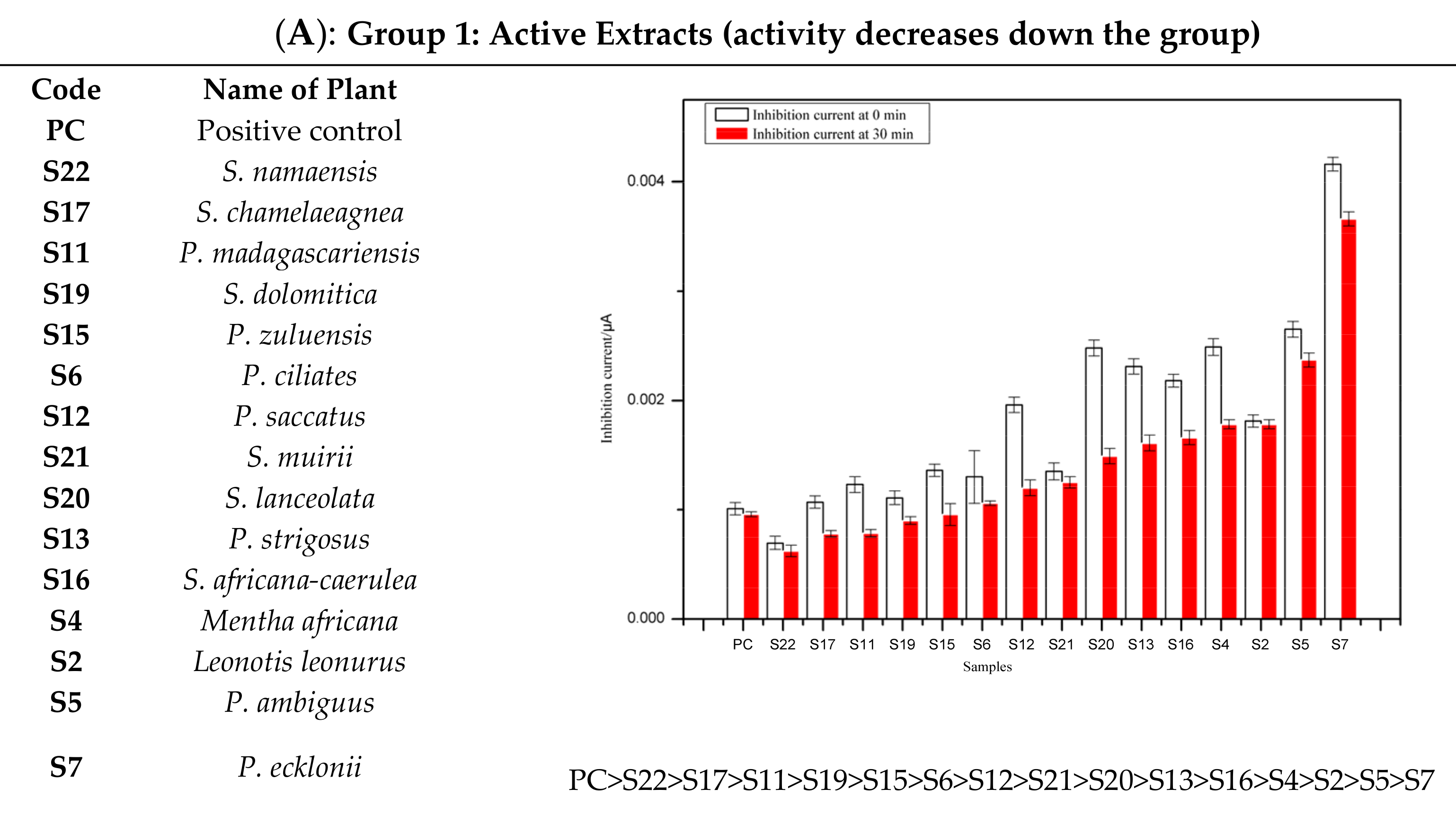
- Group 1:
- The extracts in this group referred to as active, showed inhibitory activity potential against tyrosinase enzyme compared to standard kojic acid used as positive control, which gave a significant decrease in inhibition current with time.
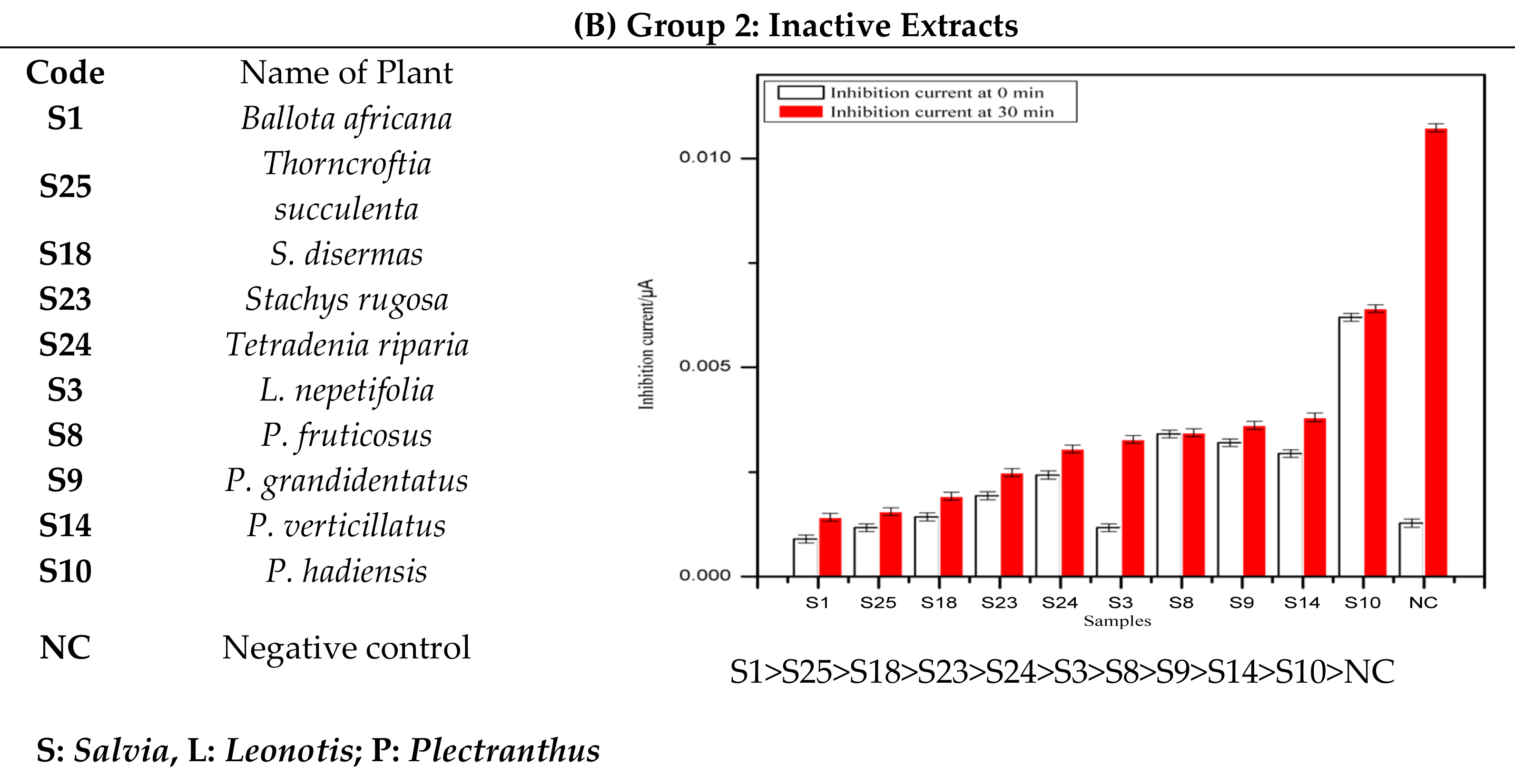
- Group 2:
- The extracts in this group are classified as inactive on the basis of their poor inhibitory activity against tyrosinase enzyme as compared to the standard negative control (DMSO). The inhibition current significantly increasesd with time.
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kim, Y.J.; Uyama, H. Tyrosinase inhibitors from natural and synthetic sources: Structure, inhibition mechanism and perspective for the future. Cell. Mol. Life Sci. 2005, 62, 1707–1723. [Google Scholar] [CrossRef] [PubMed]
- Summers, B. A lightening tour of skin-brightening options. Pharm. Cosmet. Rev. 2006, 33, 29–30. [Google Scholar]
- Mapunya, M.B.; Nikolova, R.V.; Lall, N. Melanogenesis and antityrosinase activity of selected south african plants. Evid. Based Complement. Altern. Med. 2012, 2012, 374017. [Google Scholar] [CrossRef] [PubMed]
- Costin, G.E.; Hearing, V.J. Human skin pigmentation: Melanocytes modulate skin color in response to stress. FASEB J. 2007, 21, 976–994. [Google Scholar] [CrossRef] [PubMed]
- Brenner, M.; Hearing, V.J. The Protective Role of Melanin Against UV Damage in Human Skin. Photochem Photobiol. 2008, 84, 539–549. [Google Scholar] [CrossRef] [PubMed]
- Chompo, J.; Upadhyay, A.; Fukuta, M.; Tawata, S. Effect of Alpinia zerumber components on antioxidant and skin disease-related enzymes. BMC Complement. Altern. Med. 2012, 12, 106–114. [Google Scholar] [CrossRef] [PubMed]
- Wangthong, S.; Tonsiripakdee, I.; Monhaphol, T.; Nonthabenjawan, R.; Wanichwecharungruang, P.S. TLC developing technique for tyrosinase inhibitor detection. Biomed. Chromatogr. 2007, 21, 94–100. [Google Scholar] [CrossRef] [PubMed]
- Bartosz, M.; Kedziora, J.; Bartosz, G. The copper complex of captopril is not a superoxide dismutase mimic. Artefacts in DMPO spin trapping. Free Radic. Res. 1996, 24, 391–396. [Google Scholar] [CrossRef] [PubMed]
- Espín, J.C.; Wichers, H.J. Effect of captopril on mushroom tyrosinase activity in vitro. Biochim. Biophys. Acta 2001, 1544, 289–300. [Google Scholar] [CrossRef]
- Kubo, I.; Kinst-Hori, I.; Ishiguro, K.; Chaudhuri, S.K.; Sanchez, Y.; Oruga, T. Tyrosinase inhibitory flavonoids from Heterotheca inuloides and their structural functions. Bioorg. Med. Chem. Lett. 1994, 4, 1443–1446. [Google Scholar] [CrossRef]
- Kermasha, S.; Bao, H.; Bisakowski, B. Biocatalysis of tyrosinase using catechin as substrate in selected organic solvent media. J. Mol. Catal. B Enzym. 2001, 11, 929–938. [Google Scholar] [CrossRef]
- Badria, F.A.; El-Gayyar, M.A. A new type of tyrosinase inhibitors from natural products as potential treat- ments for hyperpigmentation. Bollottino Chim. Farm. 2001, 140, 267–271. [Google Scholar]
- Sparavigna, A.; Tenconi, B.; Deponti, I.; Scarci, F.; Caserini, M. An innovative concept gel to prevent skin aging. J. Cosmet. Dermatol. Sci. Appl. 2013, 3, 271–280. [Google Scholar] [CrossRef]
- Tavares da Mota, G.S.; Arantes, A.B.; Sacchetti, G.; Spannoletti, A.; Ziosi, P.; Scalambra, E.; Vertuani, S.; Manfredini, S. Antioxidant activity of cosmetic formulations based on novel extracts from seeds of Brazilian Araucaria angustifolia (Bertoll) Kuntze. J. Cosmet. Dermatol. Sci. Appl. 2014, 4, 190–202. [Google Scholar]
- Sima, V.H.; Patris, S.; Aydogmus, Z.; Sarakbi, A.; Sandulescu, R.; Kauffmann, J.M. Tyrosinase immobilized magnetic nanobeads for the amperometric assay of enzyme inhibitors: Application to the skin whitening agents. Talanta 2011, 83, 980–987. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.C.; Tseng, T.S.; Hisiao, N.W.; Lin, Y.L.; Wen, Z.H.; Tsai, C.C.; Lee, Y.C.; Lin, H.H.; Tsai, K.C. Discovering of highly potent tyrosinase inhibitor, T1, with significant anti-melanogenesis ability by zebrafish in vivo assay and computational molecular modeling. Sci. Rep. 2015, 10, 1–8. [Google Scholar]
- Xia, N.; Zhang, L.; Feng, Q.; Deng, D.; Sun, X.; Liu, L. Amplified voltammetric detection of tyrosinase and its activity with dopamine-gold nanoparticles as redox probes. Int. J. Electrochem. 2013, 8, 5487–5495. [Google Scholar]
- Rodríguez-Sevilla, E.; María-Teresa, R.S.; Romero-Romo, M.; Ibarra-Escutia, P.; Palomar-Pardavé, M. Electrochemical quantification of the antioxidant capacity of medicinal plants using biosensors. Sensors 2014, 14, 14423–14439. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Yang, X.; Han, Q.; Zheng, J. The investigation of electrochemistry behaviors of tyrosinase based on directly electrodeposited graphene on choline–gold nanoparticals. Preprints 2017, 22, 1047. [Google Scholar]
- Klopper, R.R.; Chatelain, C.; Banninger, V.; Habashi, C.; Steyn, H.M.; De Wet, B.C.; Arnold, T.H.; Gautier, L.; Smith, G.F.; Spichiger, R. Checklist of the Flowering Plants of Sub-Saharan Africa. An Index of Accepted Names and Synonyms; Southern African Botanical Diversity Network: Pretoria, South Africa, 2006. [Google Scholar]
- Vladimir-Knežević, S.; Blažeković, B.; Kindl, M.; Vladić, J.; Lower-Nedza, A.D.; Brantner, A.H. Acetylcholinesterase inhibitory, antioxidant and phytochemical properties of selected medicinal plants of the Lamiaceae family. Molecules 2014, 9, 767–782. [Google Scholar] [CrossRef] [PubMed]
- Aristov, A.; Habekost, A. Cyclic voltammetry—A versatile electrochemical method investigating electron transfer process. World J. Chem. Educ. 2015, 3, 115–119. [Google Scholar]
- Ozer, O.; Mutlu, B.; Kivcak, B. Antityrosinase activity of some plant extracts and formulations containing ellagic acid. Pharm. Biol. 2007, 45, 519–524. [Google Scholar] [CrossRef]
- Popoola, O.; Marnewich, J.L.; Rautenbach, F.; Ameer, F.; Iwuoha, E.I.; Hussein, A.A. Inhibition of oxidative stress and skin aging-related enzymes by prenylated chalcones and other flavonoids from Helichrysum teretifolium. Molecules 2015, 20, 7143–7155. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Kuang, D.; Feng, Y.; Zhang, F.; Xu, Z.; Liu, M.; Wang, D. Electrochemical tyrosine sensor based on a glassy carbon electrode modified with a nanohybrid made from graphene oxide and multiwalled carbon nanotubes. Microchim. Acta 2013, 180, 49–58. [Google Scholar] [CrossRef]
- Fauziyah, R.S.; Gobikrishnan, S.; Indrawan, N.; Park, S.H.; Park, J.H.; Min, K.; Yoo, Y.J.; Park, D.H. A study on the electrochemical synthesis of L-DOPA using oxidoreductase enzymes: Optimization of an electrochemical process. J. Microbiol. Biotechnol. 2012, 22, 1446–1451. [Google Scholar]
- Chang, T.-S. An updated review of tyrosinase inhibitors. Int. J. Mol. Sci. 2009, 10, 2440–2475. [Google Scholar] [CrossRef] [PubMed]
- Ullah, S.; Son, S.; Yun, H.Y.; Kim, D.H.; Chun, P.; Moon, H.R. Tyrosinase inhibitors: A patent review (2011–2015). Expert Opin. Ther. Pat. 2016, 26, 347–362. [Google Scholar] [CrossRef] [PubMed]
- Suntar, I.; Akkol, E.K.; Senol, F.S.; Keles, H.; Orhan, I.E. Investigating wound healing, tyrosinase inhibitory and antioxidant activities of the ethanol extracts of Salvia cryptantha and Salvia cyanescens using in vivo and in vitro experimental models. J. Ethnopharmacol. 2011, 135, 71–77. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Etsassala, N.G.E.R.; Waryo, T.; Popoola, O.K.; Adeloye, A.O.; Iwuoha, E.I.; Hussein, A.A. Electrochemical Screening and Evaluation of Lamiaceae Plant Species from South Africa with Potential Tyrosinase Activity. Sensors 2019, 19, 1035. https://doi.org/10.3390/s19051035
Etsassala NGER, Waryo T, Popoola OK, Adeloye AO, Iwuoha EI, Hussein AA. Electrochemical Screening and Evaluation of Lamiaceae Plant Species from South Africa with Potential Tyrosinase Activity. Sensors. 2019; 19(5):1035. https://doi.org/10.3390/s19051035
Chicago/Turabian StyleEtsassala, Ninon G.E.R., Tesfaye Waryo, Olugbenga K. Popoola, Adewale O. Adeloye, Emmanuel I. Iwuoha, and Ahmed A. Hussein. 2019. "Electrochemical Screening and Evaluation of Lamiaceae Plant Species from South Africa with Potential Tyrosinase Activity" Sensors 19, no. 5: 1035. https://doi.org/10.3390/s19051035
APA StyleEtsassala, N. G. E. R., Waryo, T., Popoola, O. K., Adeloye, A. O., Iwuoha, E. I., & Hussein, A. A. (2019). Electrochemical Screening and Evaluation of Lamiaceae Plant Species from South Africa with Potential Tyrosinase Activity. Sensors, 19(5), 1035. https://doi.org/10.3390/s19051035






