Physiological Responses During Hybrid BNCI Control of an Upper-Limb Exoskeleton
Abstract
:1. Introduction
2. Materials and Methods
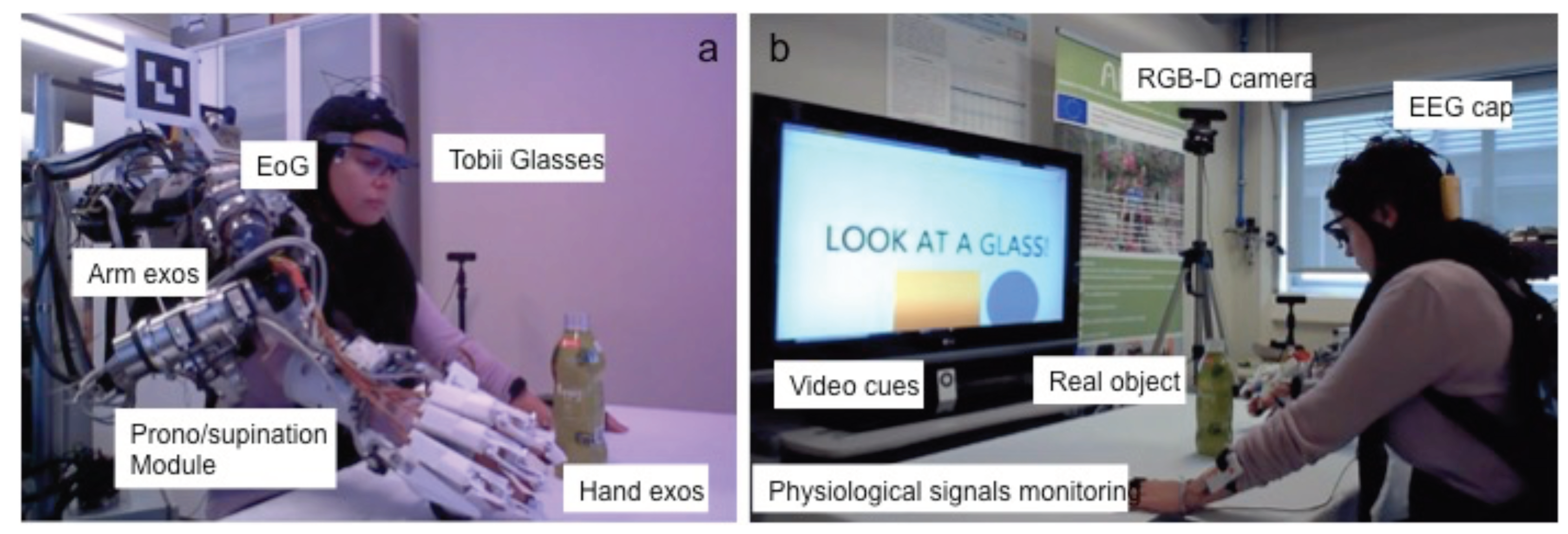
2.1. Multimodal Sensory System
2.1.1. Hybrid Bnci
2.1.2. Physiological Monitoring System
2.2. Arm Exoskeleton
2.2.1. Shoulder-Elbow Exoskeleton
2.2.2. Hand-Wrist Exoskeleton
2.3. Participants
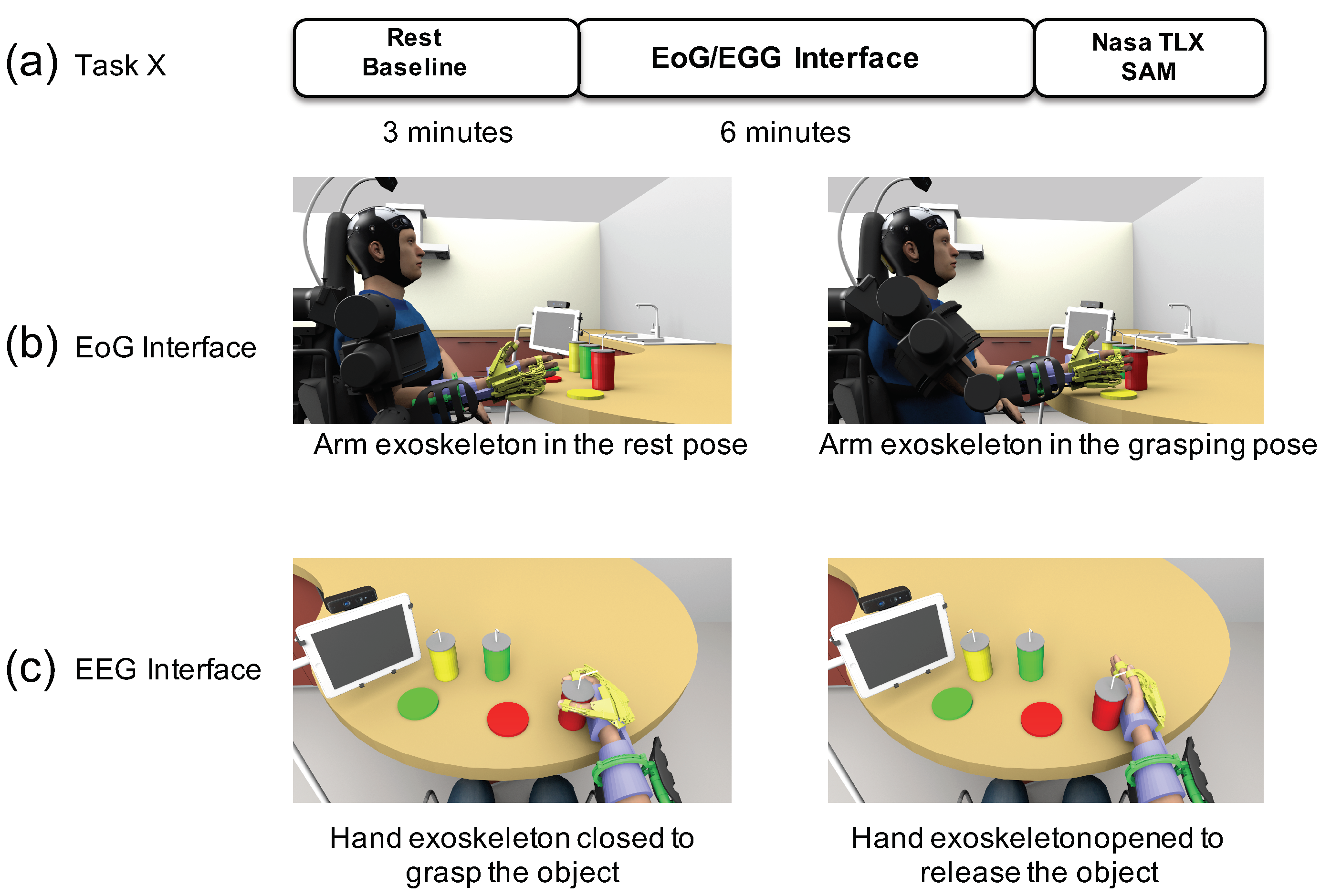
2.4. Experimental Protocol
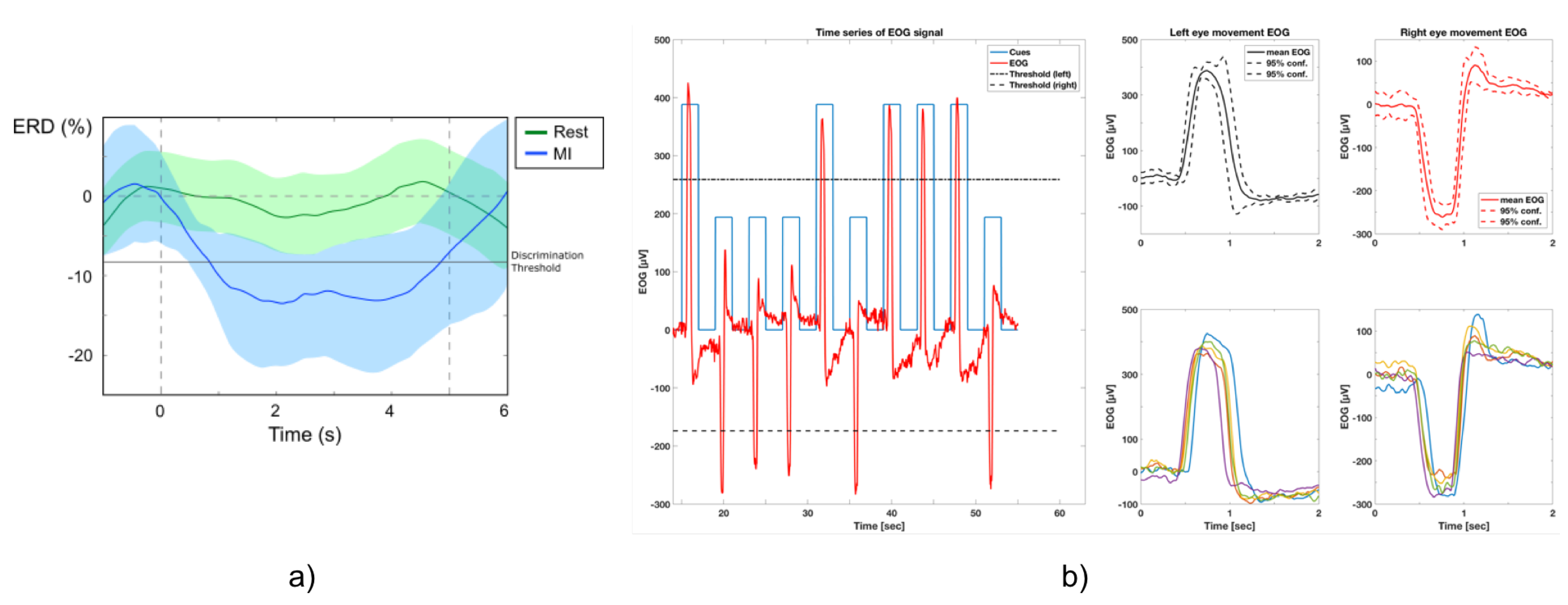
- Set-up, calibration and BNCI training: Calibration of the BNCI system comprises two parts: in the first part, participants are instructed to either relax or imagine hand-grasping motions following a visual cue displayed on a computer screen. To identify the optimal frequency for detection of motor-imagery related desynchronization of sensorimotor rhythms (SMR, 8–15Hz) of the subject, a power spectrum estimation is performed, selecting the frequency that shows largest even-related desynchronization (ERD) during motor-imagery and event-related synchronization (ERS) during relax. Based on the maximum values for ERD and ERS, an optimal discrimination threshold is computed and used for later online BCI control. EoG is recorded in accordance to the standard EoG placements at the left and right outer canthus (LOC/ROC). In the EoG-related part of the calibration, subjects are instructed to move their eyes to the left or to the right following randomized visual cues (arrow to the left, arrow to the right). A detection threshold for full left and right eye movements is set at 80% of the average of maximal EoG signal recorded during presentation of the visual cues.
- Familiarization: The familiarization phase only consisted of showing the user the functioning of the finite-state machine and the visual interface. No additional training was required, since the user was already familiarized with the EEG/EoG interface.
- Experimental phase: Each subject had to perform two different tasks during 6 min each one: (i) a task triggered by the EoG interface to reach an object controlling the arm exoskeleton; (ii) a task triggered by the EEG interface to grasp an object controlling the hand exoskeleton. To increase the number of data points for the analysis, subjects were asked to perform two times the EoG task and four times the EEG task. After completing each task with one interface, the NASA-TLX questionnaire and self-assessment manikin (SAM) were submitted to the user to evaluate the subjective workload required to perform the task.
3. Results
3.1. Interfaces and Performance
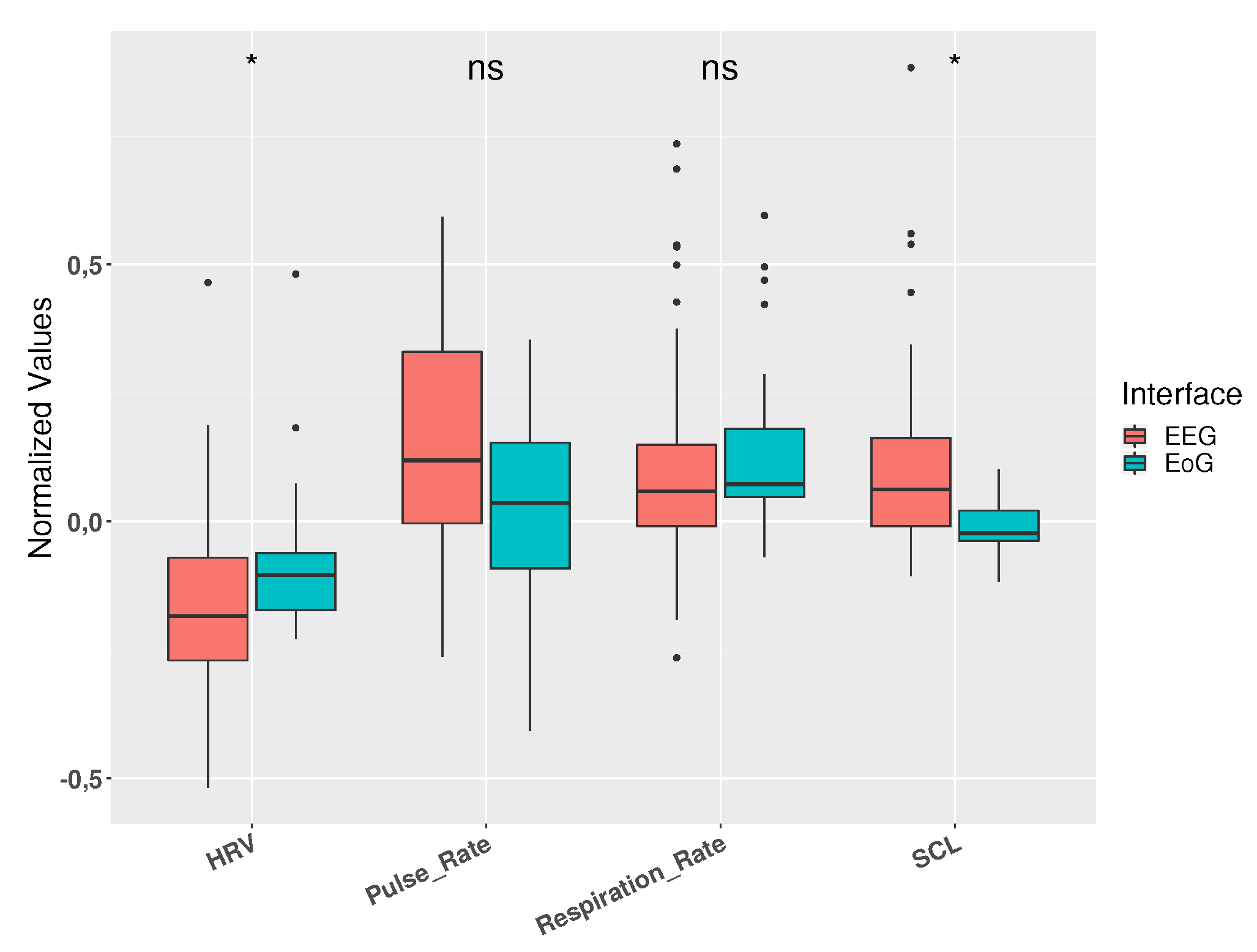
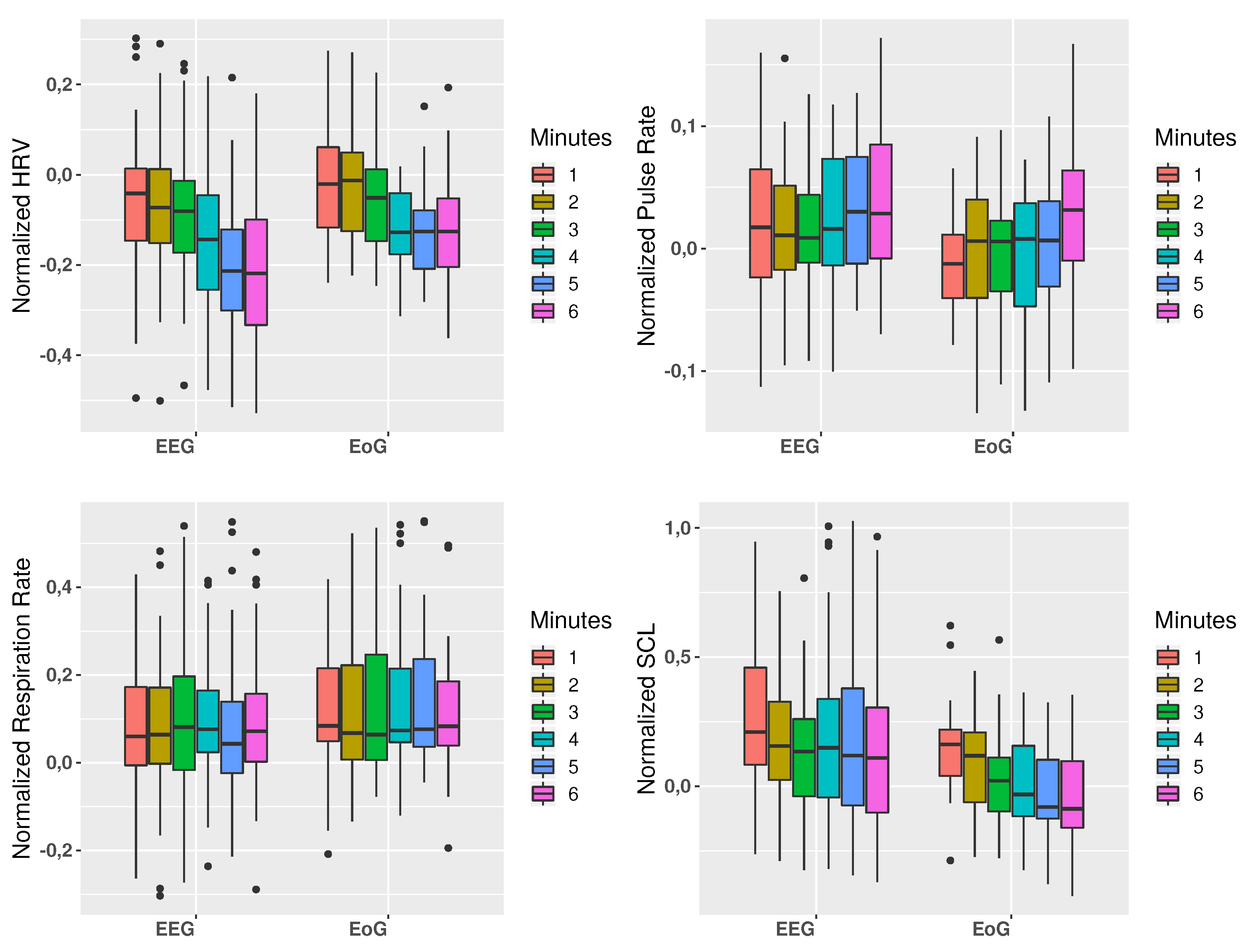
3.2. Physiological Measurements
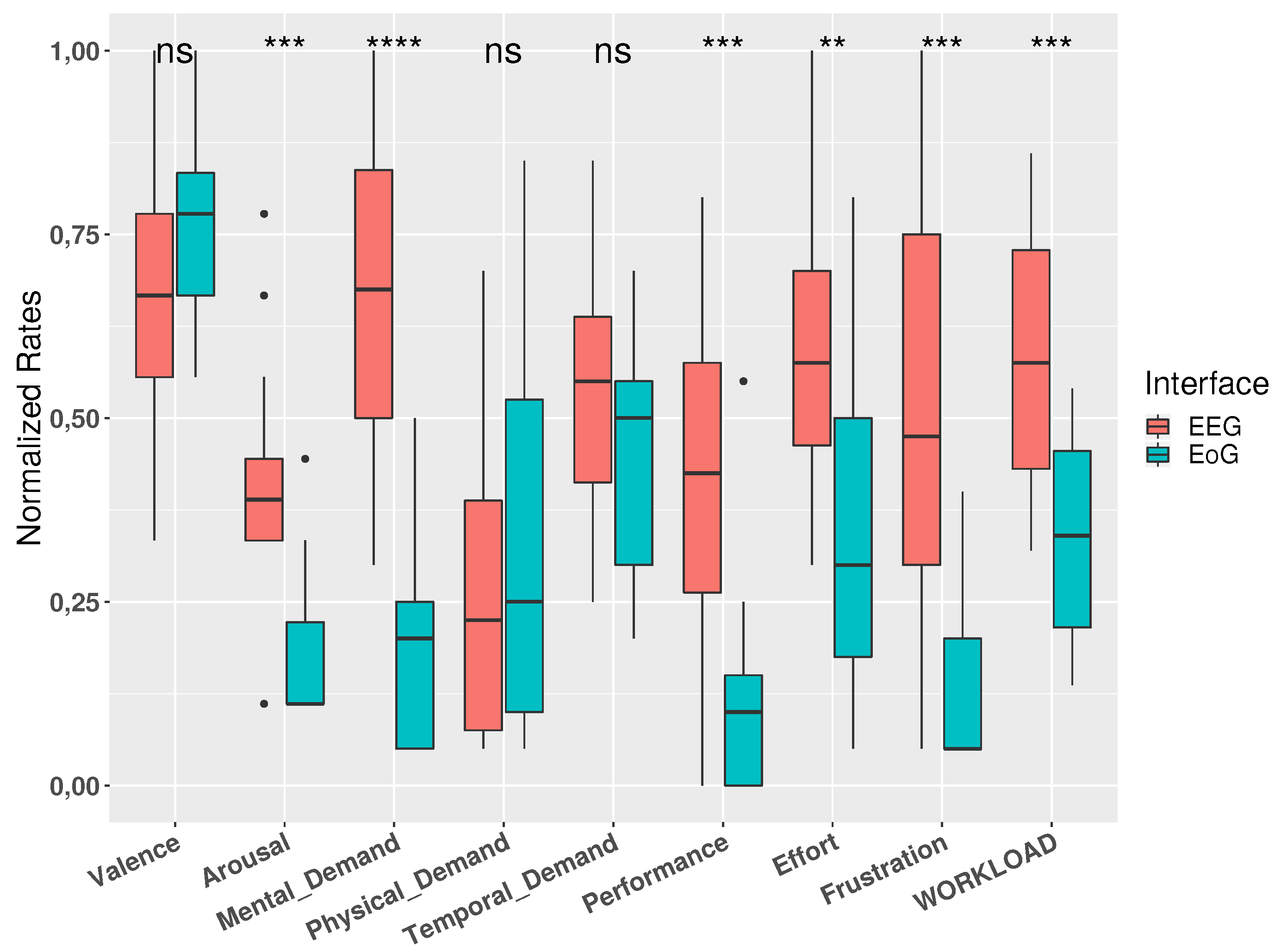
3.3. Subjective Ratings
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- European Commision. People with disabilities have equal rights. In The European Disability Strategy 2010–2020; European Commision: Brussels, Belgium, 2010; ISBN 978-92-79-16836-9. [Google Scholar]
- Müller-Putz, G.R.; Breitwieser, C.; Cincotti, F.; Leeb, R.; Schreuder, M.; Leotta, F.; Tavella, M.; Bianchi, L.; Kreilinger, A.; Ramsay, A.; et al. Tools for brain–computer interaction: A general concept for a hybrid BCI. Front. Neuroinform. 2011, 5, 30. [Google Scholar] [CrossRef] [PubMed]
- Choi, I.; Rhiu, I.; Lee, Y.S.; Yun, M.W.; Nam, C.S. A Systematic Review of Hybrid Brain-Computer Interfaces: Taxonomy and Usability Perspectives. PLoS ONE 2017, 12, e0176674. [Google Scholar] [CrossRef] [PubMed]
- Su, Y.; Qi, Y.; Luo, J.X.; Wu, B.; Yang, F.; Li, Y.; Zhuang, Y.T.; Zheng, X.X.; Chen, W.D. A hybrid brain-computer interface control strategy in a virtual environment. J. Zhejiang Univ. Sci. C 2011, 12, 351–361. [Google Scholar] [CrossRef]
- Pan, J.; Xie, Q.; He, Y.; Wang, F.; Di, H.; Laureys, S.; Yu, R.; Li, Y. Detecting awareness in patients with disorders of consciousness using a hybrid brain-computer interface. J. Neural Eng. 2014, 11, 56007. [Google Scholar] [CrossRef] [PubMed]
- Allison, B.Z.; Brunner, C.; Kaiser, V.; Muller-Putz, G.R.; Neuper, C.; Pfurtscheller, G. Toward a hybrid brain computer interface based on imagined movement and visual attention. J. Neural Eng. 2010, 7, 026007. [Google Scholar] [CrossRef] [PubMed]
- Úbeda, A.; Iáñez, E.; Badesa, J.; Morales, R.; Azorín, J.M.; García, N. Control strategies of an assistive robot using a Brain-Machine Interface. In Proceedings of the 2012 IEEE/RSJ International Conference on Intelligent Robots and Systems, Vilamoura, Portugal, 7–12 October 2012; pp. 3553–3558. [Google Scholar]
- Yu, T.; Xiao, J.; Wang, F.; Zhang, R.; Gu, Z.; Cichocki, A.; Li, Y. Enhanced motor imagery training using a hybrid BCI with feedback. IEEE Trans. Biomed. Eng. 2015, 62, 1706–1717. [Google Scholar] [CrossRef] [PubMed]
- Brunner, C.; Allison, B.Z.; Krusienski, D.J.; Mullerputz, G.R.; Pfurtscheller, G.; Neuper, C. Improved signal processing approaches in an offline simulation of a hybrid brain-computer interface. J. Neurosci. Methods 2010, 188, 165–173. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Samuel, O.W.; Zhang, X.; Wang, H.; Fang, P.; Li, G. A motion-classification strategy based on sEMG-EEG signal combination for upper-limb amputees. J. Neuroeng. Rehabil. 2017, 14, 2. [Google Scholar] [CrossRef] [PubMed]
- Kawase, T.; Sakurada, T.; Koike, Y.; Kansaku, K. A hybrid BMI- based exoskeleton for paresis: EMG control for assisting arm movements. J. Neural Eng. 2017, 14, 016015. [Google Scholar] [CrossRef] [PubMed]
- Scherer, R.; Mller-Putz, G.R.; Pfurtscheller, G. Self-initiation of eeg-based brain-computer communication using the heart rate response. J. Neural Eng. 2007, 4, L23–L29. [Google Scholar] [CrossRef] [PubMed]
- Pfurtscheller, G.; Allison, B.Z.; Brunner, C.; Bauernfeind, G.; Escalante, T.S.; Scherer, R.; Zander, T.O.; Mueller-Putz, G.; Neuper, C.; Birbaumer, N. The hybrid bci. Front. Neurosci. 2010, 4, 30. [Google Scholar] [CrossRef] [PubMed]
- Witkowski, M.; Cortese, M.; Cempini, M.; Mellinger, J.; Vitiello, N.; Soekadar, S.R. Enhancing brain–machine interface (BMI) control of a hand exoskeleton using electrooculography (EoG). J. Neuroeng. Rehabil. 2014, 11, 165. [Google Scholar] [CrossRef] [PubMed]
- Surjo, R. Soekadar, Matthias Witkowski, Nicola Vitiello, Niels Birbaumer An EEG/EoG-based hybrid brain-neural computer interaction (BNCI) system to control an exoskeleton for the paralyzed hand. Biomed. Tech. 2015, 60, 199–205. [Google Scholar] [CrossRef]
- Frisoli, A.; Procopio, C.; Chisari, C.; Creatini, I.; Bonfiglio, L.; Bergamasco, M.; Rossi, B.; Carboncini, M. Positive effects of robotic exoskeleton training of upper limb reaching movements after stroke. J. NeuroEng. Rehabil. 2012, 9, 36. [Google Scholar] [CrossRef] [PubMed]
- Barsotti, M.; Leonardis, D.; Loconsole, C.; Solazzi, M.; Sotgiu, E.; Procopio, C.; Chisari, C.; Bergamasco, M.; Frisoli, A. A full upper limb robotic exoskeleton for reaching and grasping rehabilitation triggered by MI-BCI. In Proceedings of the 2015 IEEE International Conference on Rehabilitation Robotics (ICORR), Singapore, 11–14 August 2015; pp. 49–54. [Google Scholar]
- Pedrocchi, A.; Ferrante, S.; Ambrosini, E.G.; Olla, M.; Casellato, C.; Schauer, T.; Klauer, C.; Pascual, J.; Vidaurre, C.; Gfohler, M.; et al. Mundus project: MUltimodal neuroprosthesis for daily upper limb support. J. Neuroeng. Rehabil. 2013, 10, 6. [Google Scholar] [CrossRef] [PubMed]
- Kaufmann, T.; Vögele, C.; Sütterlin, S.; Lukito, S.; Kübler, A. Effects of resting heart rate variability on performance in the P300 brain-computer interface. Int. J. Psychophysiol. 2012, 83, 336–341. [Google Scholar] [CrossRef] [PubMed]
- Myrden, A.; Chau, T. Effects of user mental state on EEG-BCI performance. Front. Hum. Neurosci. 2015, 9, 308. [Google Scholar] [CrossRef] [PubMed]
- Myrden, A.; Chau, T. A Passive EEG-BCI for Single-Trial Detection of Changes in Mental State. IEEE Trans. Neural Syst. Rehabil. Eng. 2017, 25, 345–356. [Google Scholar] [CrossRef] [PubMed]
- Mohammadpour, M.; Mozaffari, S. Classification of EEG-based attention for brain computer interface. In Proceedings of the 2017 3rd Iranian Conference on Intelligent Systems and Signal Processing (ICSPIS), Shahrood, Iran, 20–21 December 2017; pp. 34–37. [Google Scholar]
- Li, Y.; Li, X.; Ratcliffe, M.; Liu, L.; Qi, Y.; Liu, Q. A real-time EEG-based BCI system for attention recognition in ubiquitous environment. In Proceedings of the 2011 International Workshop on Ubiquitous Affective Awareness and Intelligent Interaction (UAAII ’11), Beijing, China, 18 September 2011; pp. 33–40. [Google Scholar]
- Ushiba, J.; Soekadar, S.R. Brain-machine interfaces for rehabilitation of poststroke hemiplegia. Prog. Brain Res. 2016, 228, 163–183. [Google Scholar] [PubMed]
- Soekadar, S.R.; Birbaumer, N.; Slutzky, M.W.; Cohen, L.G. Brain-Machine Interfaces in Neurorehabilitation of Stroke. Neurobiol. Dis. 2015, 83, 172–179. [Google Scholar] [CrossRef] [PubMed]
- Soekadar, S.R.; Witkowski, M.; Gómez, C.; Opisso, E.; Medina, J.; Cortese, M.; Cempini, M.; Carrozza, M.C.; Cohen, L.G.; Birbaumer, N.; et al. Hybrid EEG/EoG-based brain/neural hand exoskeleton restores fully independent daily living activities after quadriplegia. Sci. Robot. 2016, 1, eaag3296. [Google Scholar] [CrossRef]
- Soekadar, S.R.; Nann, M.; Crea, S.; Trigili, E.; Gómez, C.; Opisso, E.; Cohen, L.G.; Birbaumer, N.; Vitiello, N. Restoration of Finger and Arm Movements Using Hybrid Brain/Neural Assistive Technology in Everyday Life Environments. In Brain-Computer Interface Research; Guger, C., Mrachacz-Kersting, N., Allison, B., Eds.; SpringerBriefs in Electrical and Computer Engineering; Springer: Cham, Switzerland, 2019. [Google Scholar]
- Crea, S.; Nann, M.; Trigili, E.; Cordella, F.; Baldoni, A.; Badesa, F.J.; Catalán, J.M.; Zollo, L.; Vitiello, N.; Aracil, N.G.; et al. Feasibility and safety of shared EEG/EOG and vision-guided autonomous whole-arm exoskeleton control to perform activities of daily living. Scienfic Rep. 2018, 8, 10823. [Google Scholar] [CrossRef] [PubMed]
- Pfurtscheller, G.; da Silva, L.F.H. Event-related EEG/MEG synchronization and desynchronization: Basic principles. Clin. Neurophysiol. 1999, 110, 1842–1857. [Google Scholar] [CrossRef]
- Toyama, S.; Takano, K.; Kansaku, K. A non-adhesive solid-gel electrode for a non-invasive brain-machine interface. Front. Neurol. 2012, 3, 114. [Google Scholar] [CrossRef] [PubMed]
- McFarl, D.J. The advantages of the surface Laplacian in brain-computer interface research. Int. J. Psychophysiol. 2015, 97, 271–276. [Google Scholar] [CrossRef] [PubMed]
- Crea, S.; Cempini, M.; Moisè, M.; Baldoni, A.; Trigili, E.; Marconi, D.; Cortese, M.; Giovacchini, F.; Posteraro, F.; Vitiello, N. A novel shoulder-elbow exoskeleton with series elastic actuators. In Proceedings of the 6th IEEE International Conference on Biomedical Robotics and Biomechatronics (BioRob), Singapore, 26–29 June 2016; pp. 1248–1253. [Google Scholar]
- Trigili, E.; Crea, S.; Moisè, M.; Baldoni, A.; Cempini, M.; Ercolini, G.; Marconi, D.; Posteraro, F.; Carrozza, M.C.; Vitiello, N. Design and Experimental Characterization of a Shoulder-Elbow Exoskeleton with Compliant Joints for Post-Stroke Rehabilitation. IEEE/ASME Trans. Mechatron. 2019, 24, 1485–1496. [Google Scholar] [CrossRef]
- Díez, J.A.; Blanco, A.; Catalán, J.M.; Badesa, F.J.; Lledó, L.D.; García-Aracil, N. Hand exoskeleton for rehabilitation therapies with integrated optical force sensor. Adv. Mech. Eng. 2018, 10, 2. [Google Scholar] [CrossRef]
- Novak, D.; Mihelj, M.; Munih, M. A survey of methods for data fusion and system adaptation using autonomic nervous system responses in physiological computing. Interact. Comput. 2012, 24, 154–172. [Google Scholar] [CrossRef]
- NASA. Nasa Task Load Index (TLX) v. 1.0 Manual; NASN: Washington, DC, USA, 1986. [Google Scholar]
- Bradley, M.M.; Lang, P.J. Measuring emotion: The self-assessment manikin and the semantic differential. J. Behav. Ther. Exp Psychiatry 1994, 25, 49–59. [Google Scholar] [CrossRef]
- Collet, C.; Averty, P.; Dittmar, A. Autonomic nervous system and subjective ratings of strain in air-traffic control. Appl. Ergon. 2009, 40, 23–32. [Google Scholar] [CrossRef] [PubMed]
- Egelund, N. Spectral analysis of heart rate variability as an indicator of driver fatigue. Ergonomics 1982, 25, 663–672. [Google Scholar] [CrossRef] [PubMed]
- Mascord, D.J.; Heath, R.A. Behavioral and physiological indices of fatigue in a visual tracking task. J. Saf. Res. 1992, 23, 19–25. [Google Scholar] [CrossRef]
- Borghini, G.; Astolfi, L.; Vecchiato, G.; Mattia, D.; Babiloni, F. Measuring neurophysiological signals in aircraft pilots and car drivers for the assessment of mental workload, fatigue and drowsiness. Neurosci. Biobehav. Rev. 2014, 44, 58–75. [Google Scholar] [CrossRef] [PubMed]
| Performance | Activation Time | |||
|---|---|---|---|---|
| EEG | EoG | EEG | EoG | |
| Min. | 21.43 | 96.77 | 1.55 | 0.85 |
| 1st Qu. | 51.01 | 100.00 | 1.92 | 0.94 |
| Median | 65.71 | 100.00 | 2.10 | 1.02 |
| Mean | 63.75 | 99.85 | 2.20 | 1.03 |
| 3rd Qu. | 79.09 | 100.00 | 2.46 | 1.11 |
| Max. | 93.75 | 100.00 | 3.20 | 1.25 |
| Skewness | −0.58 | −4.07 | 0.61 | 0.32 |
| Kurtosis | −0.74 | 15.27 | −0.44 | −0.71 |
| Mann-Whitney test | ||||
| p-value | <0.001 | <0.001 | ||
| Pearson Correlation | Wilcoxon Signed Rank Test | ||||
|---|---|---|---|---|---|
| (Signal vs. Time) | (Minute 1 vs. Minute 6) | ||||
| rho | p-Value | p-Value | |||
| HRV | −0.344 | 0.118 | <0.001 | <0.001 | |
| EEG | Pulse Rate | 0.134 | 0.018 | 0.030 | 0.064 |
| Resp. Rate | 0.011 | 0.0001 | 0.865 | 0.544 | |
| SCL | −0.086 | 0.007 | 0.234 | <0.001 | |
| HRV | −0.339 | 0.115 | 0.001 | 0.011 | |
| EoG | Pulse Rate | 0.156 | 0.024 | 0.087 | 0.007 |
| Resp. Rate | 0.016 | 0.0001 | 0.868 | 0.920 | |
| SCL | −0.306 | 0.094 | 0.001 | 0.001 | |
| Parameter | EEG | EoG | Mann-Whitney U Test | ||
|---|---|---|---|---|---|
| Ratings | Scales | Ratings | Scales | p-Value | |
| Mental Demand | 13.23 ± 4.06 | 3.82 ± 2.86 | 4.05 ± 1.03 | 3.00 ± 1.15 | <0.0001 |
| Physical Demand | 4.95 ± 3.56 | 6.55 ± 5.32 | 0.64 ± 0.89 | 3.36 ± 1.26 | 0.54 |
| Temporal Demand | 10.68 ± 2.99 | 8.82 ± 3.40 | 2.55 ± 1.35 | 3.10 ± 1.34 | 0.19 |
| Performance | 8.68 ± 4.10 | 2.36 ± 3.24 | 2.10 ± 0.86 | 0.45 ± 0.80 | <0.001 |
| Effort | 12.00 ± 3.54 | 7.18 ± 4.55 | 2.91 ± 1.05 | 3.73 ± 1.08 | 0.007 |
| Frustration | 10.45 ± 5.34 | 2.73 ± 2.27 | 2.77 ± 1.70 | 1.36 ± 0.79 | <0.001 |
| Workload | 58.38 ± 16.52 | 33.82 ± 14.78 | <0.001 | ||
| Valence | 5.95 ± 1.56 | 6.82 ± 1.40 | 0.14 | ||
| Arousal | 3.95 ± 1.56 | 1.73 ± 1.01 | <0.001 | ||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Badesa, F.J.; Diez, J.A.; Catalan, J.M.; Trigili, E.; Cordella, F.; Nann, M.; Crea, S.; Soekadar, S.R.; Zollo, L.; Vitiello, N.; et al. Physiological Responses During Hybrid BNCI Control of an Upper-Limb Exoskeleton. Sensors 2019, 19, 4931. https://doi.org/10.3390/s19224931
Badesa FJ, Diez JA, Catalan JM, Trigili E, Cordella F, Nann M, Crea S, Soekadar SR, Zollo L, Vitiello N, et al. Physiological Responses During Hybrid BNCI Control of an Upper-Limb Exoskeleton. Sensors. 2019; 19(22):4931. https://doi.org/10.3390/s19224931
Chicago/Turabian StyleBadesa, Francisco J., Jorge A. Diez, Jose Maria Catalan, Emilio Trigili, Francesca Cordella, Marius Nann, Simona Crea, Surjo R. Soekadar, Loredana Zollo, Nicola Vitiello, and et al. 2019. "Physiological Responses During Hybrid BNCI Control of an Upper-Limb Exoskeleton" Sensors 19, no. 22: 4931. https://doi.org/10.3390/s19224931
APA StyleBadesa, F. J., Diez, J. A., Catalan, J. M., Trigili, E., Cordella, F., Nann, M., Crea, S., Soekadar, S. R., Zollo, L., Vitiello, N., & Garcia-Aracil, N. (2019). Physiological Responses During Hybrid BNCI Control of an Upper-Limb Exoskeleton. Sensors, 19(22), 4931. https://doi.org/10.3390/s19224931







