1. Introduction
With the recent advancement in micro- and nanofabrication technologies, cantilever-based sensors have increasingly become versatile tools for various sensing applications due to their fascinating intrinsic flexibility and robustness. They have widely been embraced and applied in many scientific fields ranging from physical and chemical sensing, material characterization, magnetometry, explosive detection, to biological disease diagnosis [
1,
2,
3,
4]. For instance, they have immensely and significantly contributed to the biopharmaceutical development, manufacture and quality assurance, in which the titer of virus is related to the viral dosage and resultant performance of the vaccine [
5]. Moreover, these sensors play a critical role in protein aggregation studies, differentiation and concentration measurement of sub-visible protein aggregates and silicone oil droplets in pre-filled syringe aggregation studies. In drug delivery [
6,
7], cantilever-based sensors facilitate the measurement of the concentration and particle size of the delivery vector, which often influence the biological response to the drug. Similarly, in disease diagnostics, they have been deployed in determining the concentration of specific exosomes and microvesicles [
8], which may be indicative of the onset of a disease. They are also widely used in nanoparticle toxicology studies [
2] to determine the concentration of particles within a biological or ecological environment, which essentially influences the biological response to the nanoparticles from a toxicity perspective, etc.
In the measurement of particle concentration, for instance, some various measurement technologies such as dynamic light scattering, nanoparticle tracking analysis, and resonant mass sensing can be deployed. These technologies provide particle concentration measurement based on particle fluorescence, particle size analysis and ensemble particle zeta potential measurement, and particle mass measurements, respectively. For the latter, differential stress at the surface to the beam of the cantilever causes bending or deflection of the cantilever. Most equipment for determining small masses, e.g., mass spectrometers [
9] and suspended microchannel resonators [
10], often utilize complicated technology and are quite expensive. On the other hand, the scope of application of Quartz Crystal Microbalances (QCMs) has been extended beyond gaseous to liquid media (e.g., in the study of the environment, nanoparticles, etc.). However, the interaction of the QCM surface with the liquid often results in the formation of soft or viscoelastic films. Besides, spurious factors (e.g., stress, pressure, etc.) exist [
11], which potentially dampen the vibrations. This has subsequently necessitated the recent deployment of QCM with dissipation monitoring [
12] to measure both the resonant-frequency change (Δ
f) and energy loss (measured as dissipation change).
In providing a low-cost measurement approach, dipping a cantilever sensor into a magnetic particle solution has also recently been devised and implemented [
13]. However, this particle sampling and measurement approach results in low quality factor (
Q ~ 180) due to severe damping and often yields randomized adsorption of particles on the whole cantilever surface. Furthermore, uniformity in particle adsorption or distribution over the sensing region is never guaranteed. In our study, we explored and presented an alternative droplet-based approach that can be utilized to precisely deposit particles on a specified sensing area on a cantilever for particle concentration or mass sensing applications. We successfully demonstrated localized deposition (utilizing liquid dispensing) of monodispersed magnetic polystyrene (MPS) particles onto a piezoresistive silicon cantilever sensors. Furthermore, we discussed some uncertainty parameters that influenced our measurements.
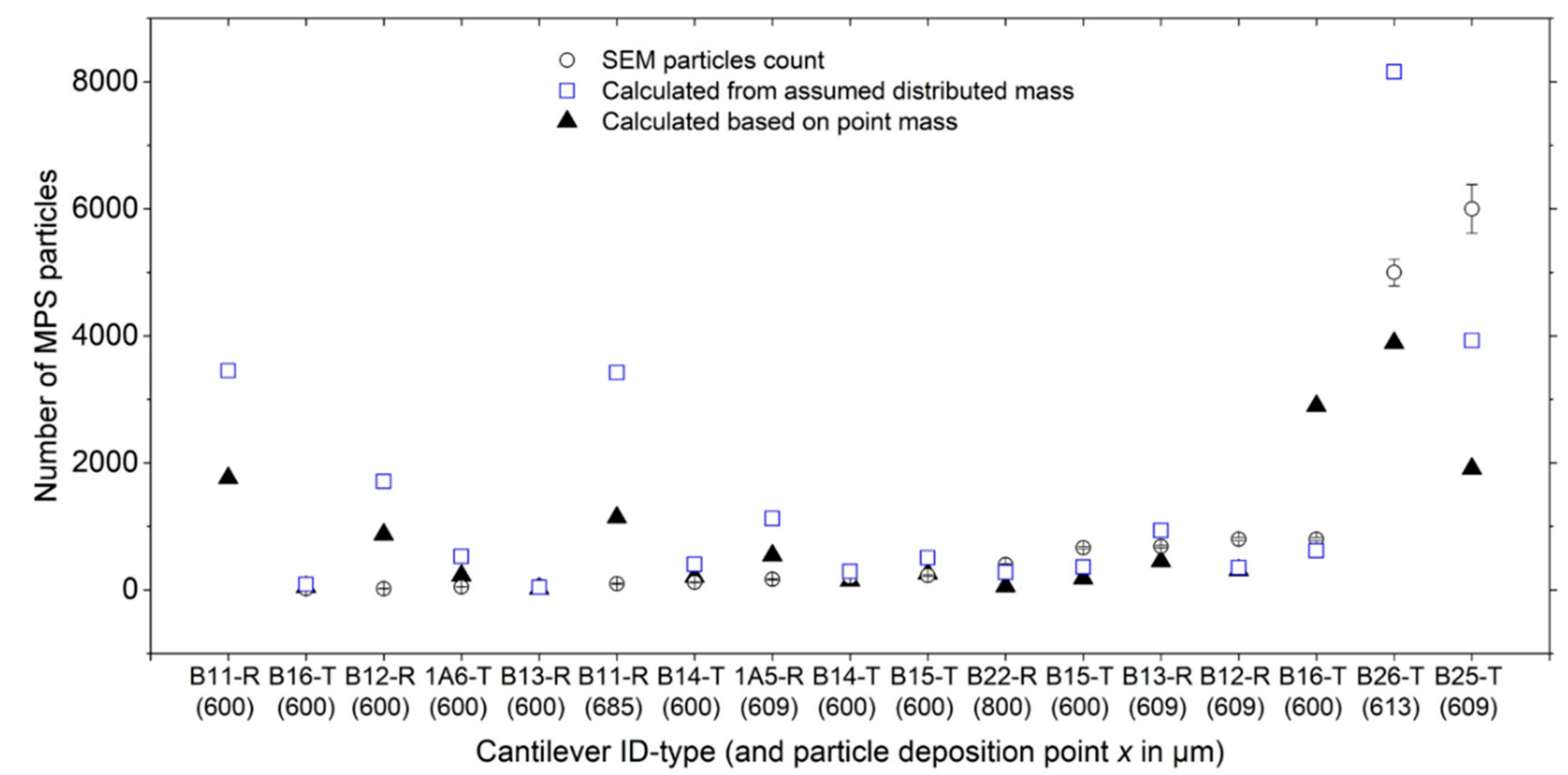
Through an optimized liquid-dispensing mechanism, we sub-sampled and loaded small water droplets with and without particles onto in-house fabricated cantilever sensors. Additionally, as a self-validating process, particle-laden droplets were similarly deposited on surface-treated silicon substrates and upon solvent evaporation, particle count estimations were performed using scanning electron microscopy (SEM). We also monitored and determined the droplet evaporation time (tev) and trends thereof for water droplets of known volume (Vd). The particles used herein included magnetic polystyrene (MPS) from Micromod GmbH, and polymethylmethacrylate (PMMA) from Sigma-Aldrich Inc. It is worth noting that the two test particles have comparable (nominal) diameters and densities, but contrasting magnetic properties, with MPS and PMMA being magnetic and non-magnetic, respectively. However, similar to silicon, PMMA has magnetic susceptibility of less than 1 and is thus diamagnetic in nature. The essence of using these two particle types was to compare the results and assess the performance of our measurement approach.
Magnetic polymer particles (such as MPS) are usually synthesized by embedding magnetic nanoparticles (e.g., from pure metals such as Fe, Co and Ni or their oxides) into a non-magnetic spherical (polymer) matrix [
14]. Magnetic beads come in different compositions, sizes and shapes. These parameters determine their chemical and physical properties. These particles can be coupled with ligands that target specific molecules. This consequently makes them highly versatile and allows their widespread use in industrial and biomedical applications. For instance, in biomedicine, they are often useful in diagnosis and therapy, e.g., sorting and purification of cells, proteins and nucleic acids, as contrast reagents in magnetic resonance imaging (MRI), targeted drug delivery, diagnostics and targeted destruction of tumors by intercellular hyperthermia [
15,
16,
17]. In the industry, these particles similarly play a critical role, e.g., as inorganic solar cell constituents, chemically inert additives, cosmetic UV protection and color filters for LCDs [
18], optimization of bioengineering processes especially in the oil industry [
14], high-density data storage [
19,
20], etc. In environmental monitoring, magnetic nanoparticles can also be used directly for pollutant removal [
21], e.g., from contaminated water resources. They for instance facilitate the removal of mercury ions in aqueous solutions by sensing, sorting and quantifying them using a nanoparticle-based system with optical and electrochemical detection regimes [
22].
2. Materials and Methods
2.1. Magnetic Polystyrene Particles
In this study, we used magnetic polystyrene (MPS) beads of nominal diameter ~2 μm and density 1.1 gcm
−3, from micromod Partikeltechnologie GmbH (D-18119 Rostock, Germany). They are basically monodispersed particles consisting of magnetite (Fe
3O
4) around an organic matrix of a styrene-maleic acid (SMA) copolymer, which are finally encapsulated with a polymer (polystyrene) layer [
14] Hence, MPS exhibits a superparamagnetic behavior. The surface of the particles, herein used, is not chemically functionalized.
Originally, monodispersed MPS particle solution was obtained in an aqueous suspension of 50 mg/mL concentration and thoroughly mixed (by sonication) prior to use or dilution. The solution was otherwise diluted with deionized water to tune the concentration levels, i.e., into approximately 20, 10, 1 and 0.1 mg/mL. This was the main variable, besides the droplet size, through which the mass Δm or the number Np of particles was varied for every particle solution dispensed on the target substrate or sensing surfaces.
2.2. Silicon Substrates Preparation and Surface Treatment
The silicon bulk substrates (~15 × 15 mm2) used in this study were cut from an n-type Si (100) wafer (Siegert Wafer GmbH, Aachen, Germany). These wafers had resistivity and thickness of 1–10 Ωcm and 275 ± 15 µm, respectively. After cleaving, the substrates were individually cleaned by: (a) boiling (5 min) in an oxidant solution containing a mixture of sulfuric acid (H2SO4, 96%) and hydrogen peroxide (H2O2, 30%) in a volume ratio of 1:1; (b) immersing in a water bath (for 5 min); (c) rinsing with deionized water; and (d) blow drying with nitrogen gas. Such a cleaning process was necessary in removing organic contaminants (e.g., dust particles, grease, silica gel, etc.) and ionic or metal contaminants from the silicon surface, prior to use or surface treatment.
Upon cleaning, some pre-selected bulk substrates were then dipped in HF solution (6–7%) for about 5 s (to etch the native oxide from silicon surface). Immediately thereafter, some set of samples were treated with O
2 plasma (100-E, Technics Plasma GmbH) for 2 min at RF power of 220 W, while others were treated with PSS/PDDA/PSS [
23]. This was meant to yield different wettabilities (i.e., surface properties). Consequently, water contact angle
θ was measured for each set of substrates: as-cleaned and after O
2 plasma treatment.
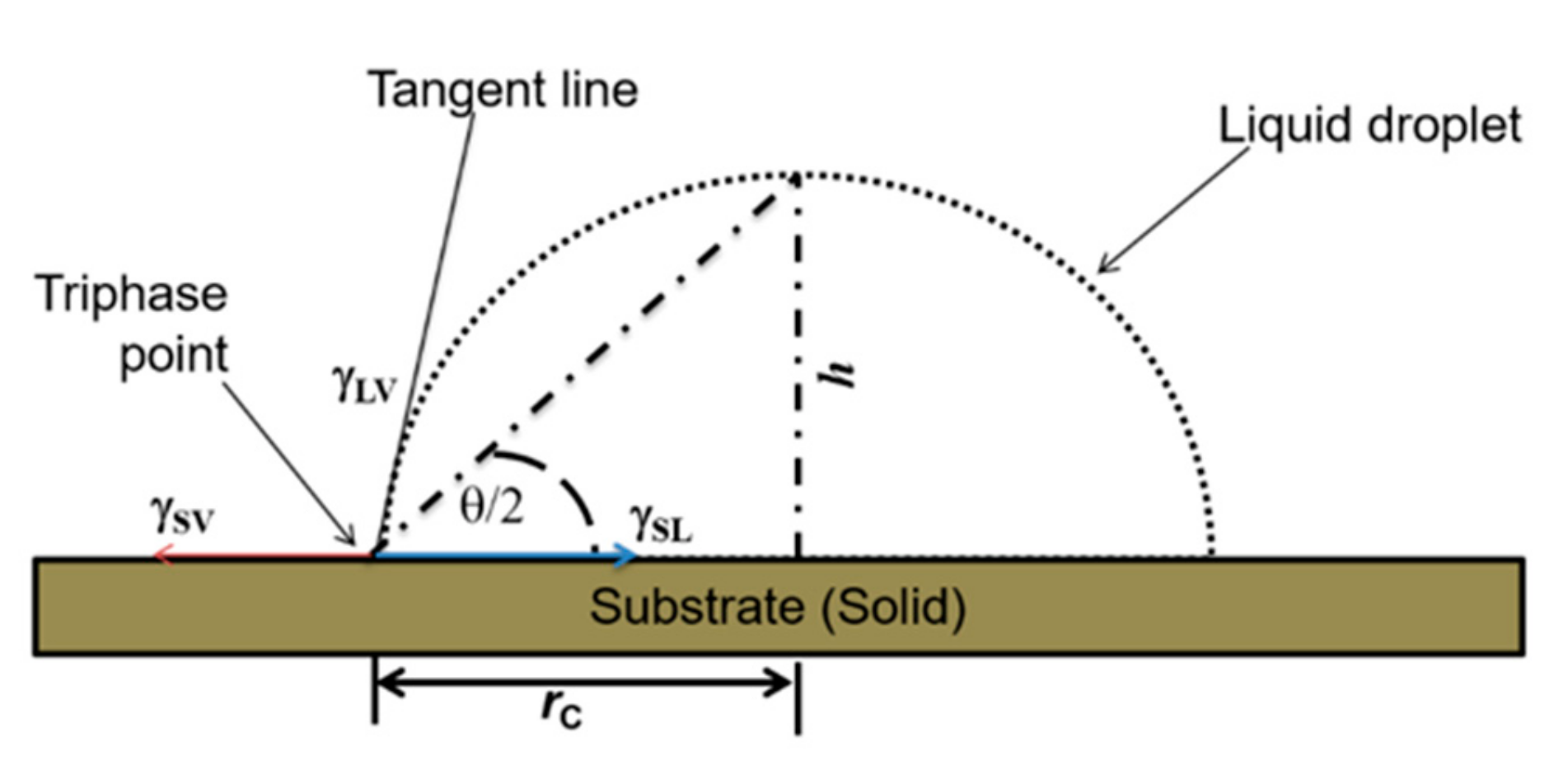
Measurement of contact angle was carried out by the sessile-drop method using a dispensing system integrated with an imaging system which comprises of a light source, a collimating mask, an adjustable stage, and a digital microscope camera (Mz-902, Oowl Tech Ltd., Hong Kong, China). The latter was helpful in visualizing and capturing the water droplet on each silicon substrate. Careful attention was taken to generate a homogeneous background behind the drop. For this reason, we positioned a diffuser between the lamp and the water droplet. The height of the camera was also adjusted so that the actual drop and its reflection on the silicon surface could clearly be observed. Consequently, the intersection point at the solid–liquid–air interfaces would precisely be determined. The internal nominal diameter of the dispensing tip (Nordson EFD Inc., East Providence, RI, USA) was ~0.10 mm, and a low pressure (~0.06 bar) was applied to dispense a droplet at room temperature (21.8 ± 0.5 °C).
In determining the contact angle, the profile of the drop image was extracted by an image-fitting in ImageJ [
24] using the Low Bond Axisymmetric Drop Shape Analysis (LBADSA) method [
25]. Primarily, by observing the shape of such a drop profile, the contact angle
θ can be measured, in which case
θ is the angle formed between the drop tangent and the solid surface at the tri-phase contact point of a simple spherical droplet, as depicted in
Figure 1. This approach allows for calculation of
θ from sessile drop images based on the Young–Laplacian equation (Equation (1)) for spherical approximation for half-angle method, in which
rc and
h denote the contact radius and the height of the drop above the solid surface, respectively.
Furthermore, the angle
θ depends on the surface tensions for solid–liquid (
γsl), solid–air (
γsv), and liquid–air (
γlv) interfaces. For small drops (of liquid density
ρ), surface tension dominates over gravity,
g ≈ 9.81 ms
−2. Consequently, a drop tends to form a spherical-cap shape to minimize total free surface energy thereby resulting into a capillary length, or capillary constant
Lc, given as:
The parameter Δ
ρ represents the difference in density of the involved phases (i.e., liquid and air). For water (
ρ =
ρw = 1 gcm
−3,
γlv = 0.0728 Nm
−1) at room temperature and standard pressure conditions,
Lc ≈ 1.35 × 10
−5 m
−2. The LBADSA method further considers the pressure gradient across a surface to the drop curvature, and the contact angle is usually approximated based on a perturbation solution of the contour of an axisymmetric sessile drop and assumes gravity as the only external force, and follows Equations (2) and (3). Equation (3) relates the principal radii of curvature (
R1 and
R2) at any point on the drop, the height
h of the drop, the capillary constant
Lc and the radius of curvature
b at the apex.
In our study, as-cleaned samples yielded
θ = 47.3° ± 2.7°, whereas contact angle after O
2 plasma treatment (2 min) was about 9°. Obviously, our surface treatment with O
2 plasma yielded enhanced hydrophilicity compared to the 24.5–46.5° (for 1–20 min O
2-RIE plasma treatment duration) reported by Alam et al. [
26]. It is worth noting that a short-time lapse between surface treatment and contact angle measurements was observed in our study. Such short time gap does not compromise the results, which can otherwise be a consequence of buildup of static charges. Moreover, for our evaluation of the experimental contact angle, we assumed a negligible effect of gravity and a constant droplet volume. This can therefore be attributed to the measurement of contact angles immediately after O
2 plasma treatment.
2.3. Fabrication of Silicon-Based Piezoresistive Cantilever Mass Sensors
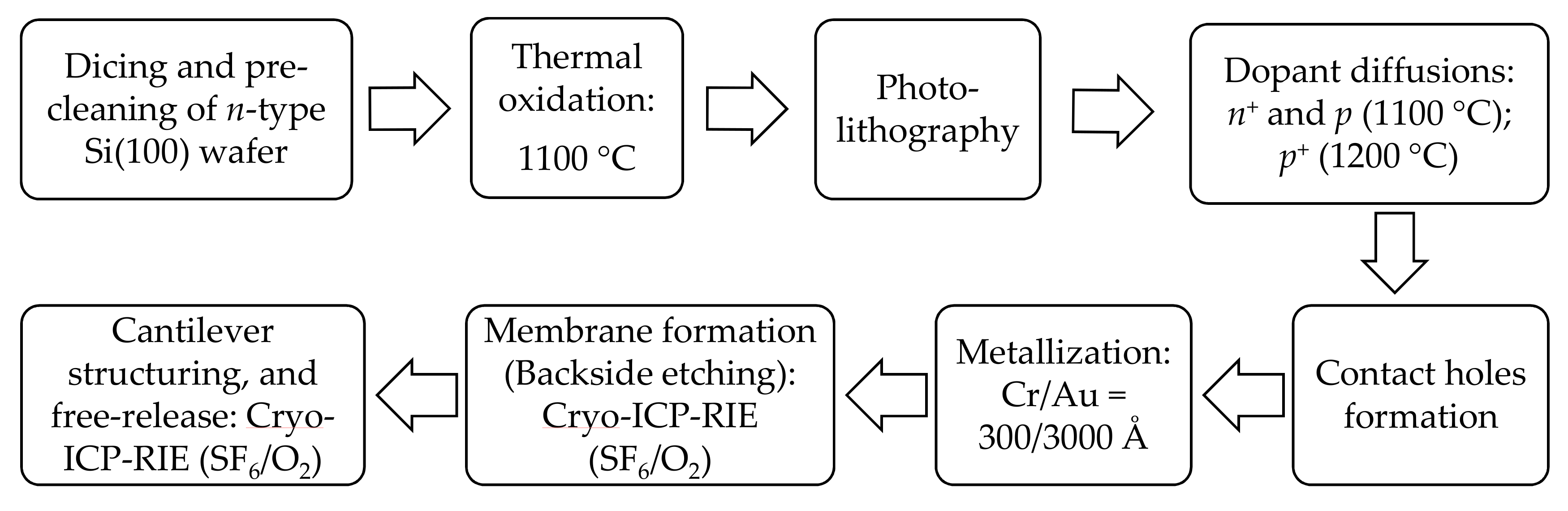
Fabrication process of our cantilever-based sensors was exclusively done in-house, using
n-type (100) silicon (as a base material) and conventional bulk micromachining technologies as described by Wasisto et al. [
27]. The process mainly entails substrate preparation (i.e., dicing the bulk Si wafer into 30 × 30 mm
2 pieces and cleaning them accordingly; cf.
Section 2.2), thermal oxidation, photolithography, dopant diffusions (phosphorous and boron), etching processes (i.e., HF dip and cryogenic ICP-RIE: SF
6/O
2), contact holes formation, and metallization of electrical contacts (Cr/Au = 300/3000 Å) coupled with a lift-off process. The general cantilever fabrication process utilizing bulk micromachining is schematically illustrated in
Figure 2.
Fabrication process basically began with the deposition of an isolation layer (SiO2) on top of the pre-cleaned silicon substrate at 1100 °C using thermal oxidation process. Hereafter, an intermediary substrate-cleaning process was performed by sonication in acetone and water rinses followed by nitrogen blow-dry. The silica layer (~100 nm) provided an excellent protection during dopant diffusion processes, in which case heating furnace temperature was set and maintained at 1100 °C (n+- and p-diffusions) and at 1200 °C (p+-diffusion).
Photolithography was then undertaken to pattern and transfer various geometric shapes/structures (e.g., U-shaped Wheatstone bridge piezo resistors, connection lines and contact pads) on a mask to the surface of the silicon substrates. A preliminary surface preparation process involving bubbling of hexamethyldisilazane (HMDS) within an enclosed chamber was executed (at ~115 °C for ~5 min) to enhance hydrophobicity of the substrate. Hereafter, pattern transfer ensued. Firstly, the prepared substrate was held onto a vacuum chunk and a photoresist (AZ 5214) was dispensed onto the silicon surface and spin-coated (e.g., 3500 rpm for 35 s). The sample was then partially evaporated in a soft baking process (110 °C for ~50 s) to improve adhesion and uniformity, and optimize light absorbance characteristics of the photoresist. To transfer the mask image to the resist-coated wafer, both the mask and substrate were mounted onto respective stages/holders on the mask aligner equipment (MBJ4, SÜSS MicroTec AG, Garching, Germany) followed by alignment and UV exposure (for specified time period, e.g., 35 s). To dissolve the soluble areas of the exposed photoresist and realize visible patterns on the wafer, the sample was immersed and gently agitated in a developer chemical solution (AZ 726) for ~60 s and thoroughly rinsed with deionized water. The resulting pattern after resist development would then be inspected (under a light microscope) followed by a hard-baking process (at 100 °C for ~30 min). Subsequently, the hardened-patterned sample was immersed in a buffered hydrofluoric acid for about 10 min to remove the entire SiO2 layer or ~5 s to clear the native oxide as desirable.
After forming piezo resistors, contact holes, electrical connections and contact pads, the sample was cryogenically etched (ICP-RIE, with SF
6/O
2) from the backside to create a membrane. Lastly, the patterning process (by lithography) was undertaken to form the desired free-ends (i.e., rectangular or triangular beam configurations [
28]) of our cantilever sensors. This was then followed by the free-releasing of the cantilever sensors from the sample through a cryogenic dry etching process. The length and width of rectangular cantilevers were 1000 µm and 170 µm, respectively, while the size of the equilateral-shaped free-end triangular cantilever was ~700 µm. The thickness of our sensors was approximately 37.9 ± 0.2 µm, and the mass
m0 of cantilever was 15.01 ± 0.11 µg and 24.65 ± 0.18 µg for rectangular and triangular cantilevers, respectively.
2.4. Droplet Dispensing Apparatus
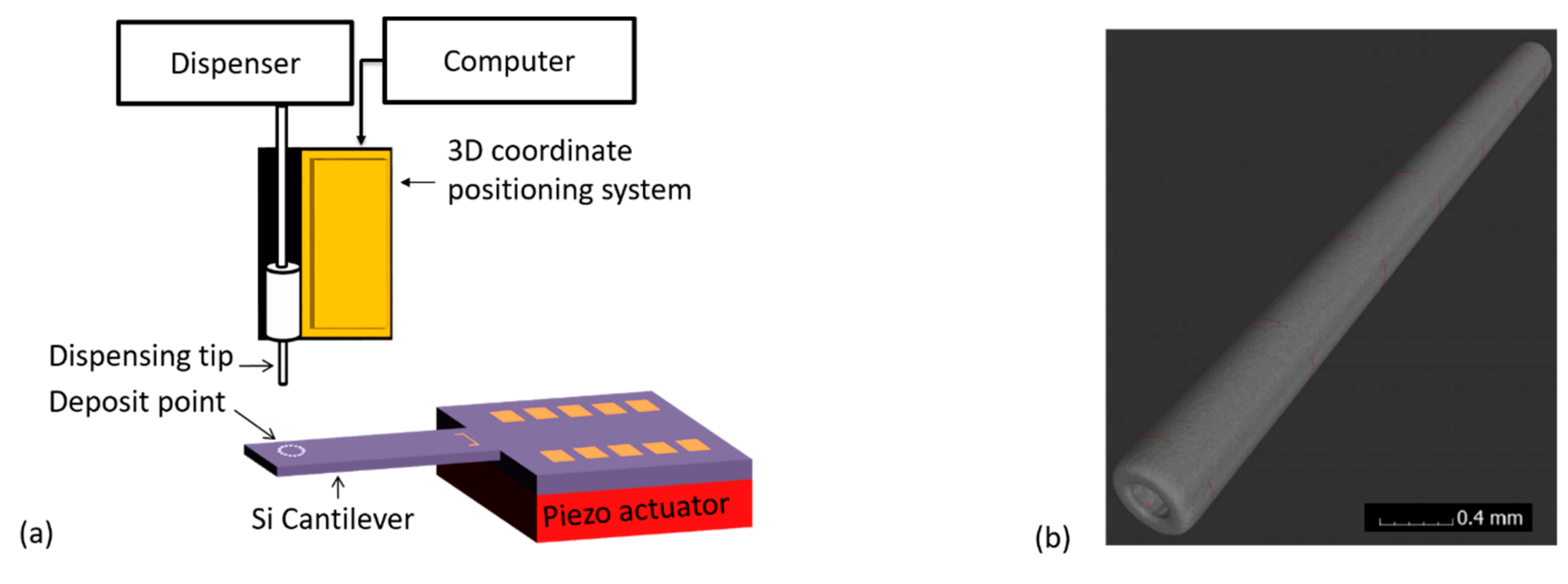
Water droplet with/without MPS particles was generated from a needle tip (of specified size) by applying specified dispensing pressure over a defined pulse duration. By keeping the size of the needle tip constant, the drop size could reliably be tuned mainly by varying the magnitude of the dispensing pressure and/or time. Through X-ray computer tomography (xCT) measurements (undertaken at PTB), we precisely calibrated and determined the internal and external diameters of our dispensing stainless needle tip (see
Figure 3) to be 0.1169 ± 0.004 mm and 0.2357 ± 0.0005 mm, respectively. The latter was also necessary and helpful in calibrating our USB digital camera (Mz-902, Oowltech).
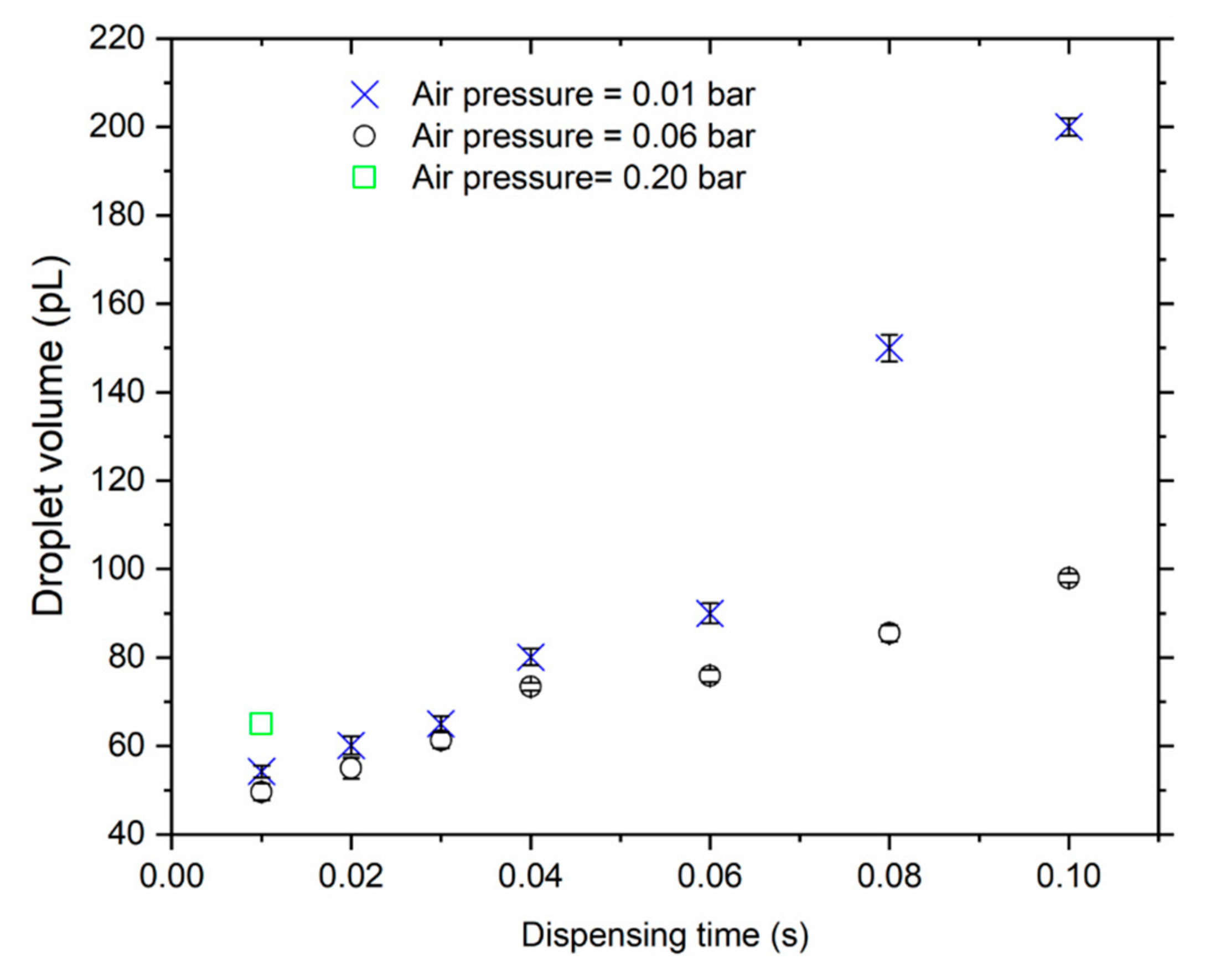
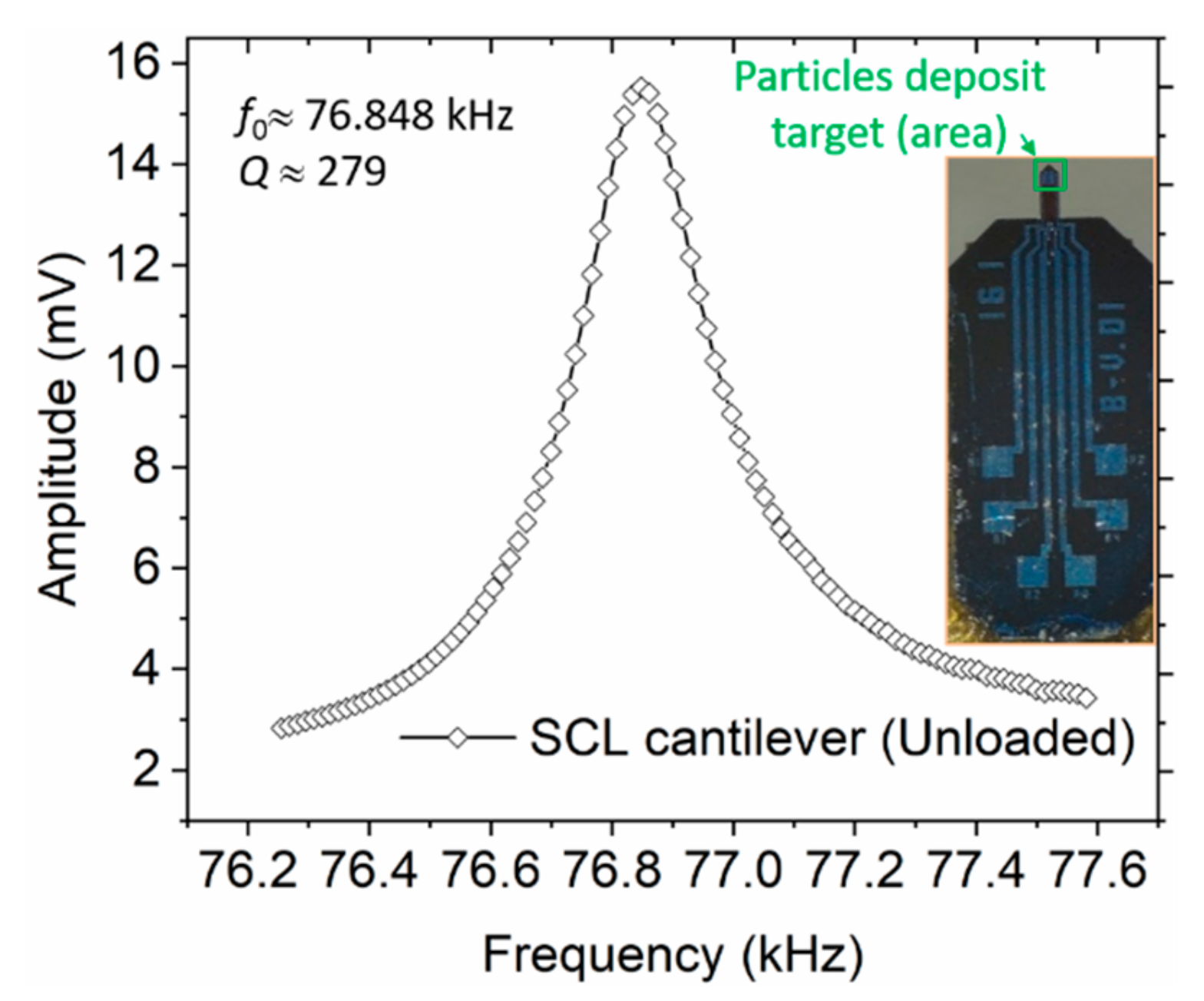
Firstly, each cantilever mass sensor was mounted on top of a piezo actuator within the sampler and carefully aligned in the horizontal plane. At this point, the particles collection target point would be defined (X,Y), i.e., by specifying a coordinate along the axisymmetric and the vibrating axis of the cantilever. The needle tip would then be mounted into a barrel (containing a thoroughly mixed solution of MPS particles) and aligned directly on top of deposit target point. During the alignment process, a 3D micro-positioning system (with a resolution of 10 μm) was used to carefully move the needle tip to the target point; and with an aid of a camera, the height from the sensor and the tip would carefully be adjusted (Z-coordinate). To further enhance the accuracy of positioning the tip on the sensing surface and minimize sensor breakages, a camera was also incorporated in the alignment process. The tip was aligned and adjusted very closely above the sensor target deposit point, in orders of few microns. Finally, the droplet was generated (1500XL, Nordson EFD Inc., Rhode Island,USA) and deposited on the sensor by applying defined dispensing parameters (i.e., air pressure and pulse duration). Typically, minimum dispensing pressure (~60 mbar) was applied and different pulse durations were used. This was desirable in generating a small droplet as much as possible.
2.5. Resonant Mass Sensing Instrumentation
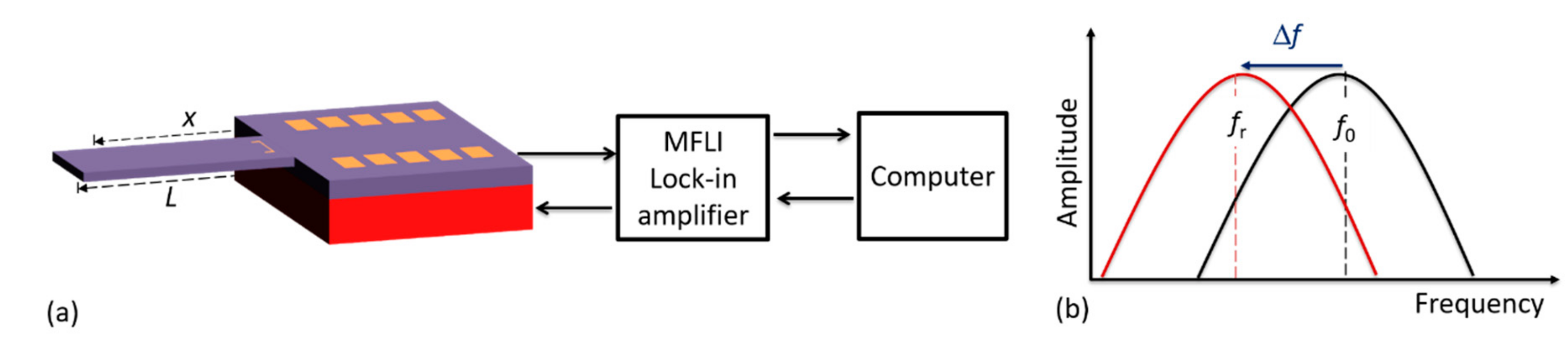
In detecting cantilever bending due to the mass of the droplet or adsorbed particles, different sensing or read-out methods can be deployed, such as piezoelectric, electrodynamic, tunneling, hard contact, optical reflection, interferometric, capacitive, and piezoresistive techniques [
29]. The latter mechanism was preferably integrated into our cantilevers during the fabrication process and was realized in the form of a U-shaped Wheatstone bridge by diffusing piezo resistors into the cantilever. To supply voltage (1 V
dc) and detect the voltage output from the bridge, a lock-in amplifier (MFLI, Zurich Instruments Ltd.) was connected to the cantilever contact pads (as shown in
Figure 4). Since our microcantilevers were externally excited with an in-plane piezoelectric stack actuator (P-121.05, from PI Ceramics GmbH, Lederhose, Germany), an excitation voltage (9.9 V
pp) was similarly supplied to the actuator by the MFLI instrument. The connections to and from the MFLI were accomplished through coaxial cables and SubMiniature version A (SMA) connectors. The required MFLI parameters (e.g., input voltage) and the signal read-out from sensor (e.g., frequency response) were managed using a computer via a control software.
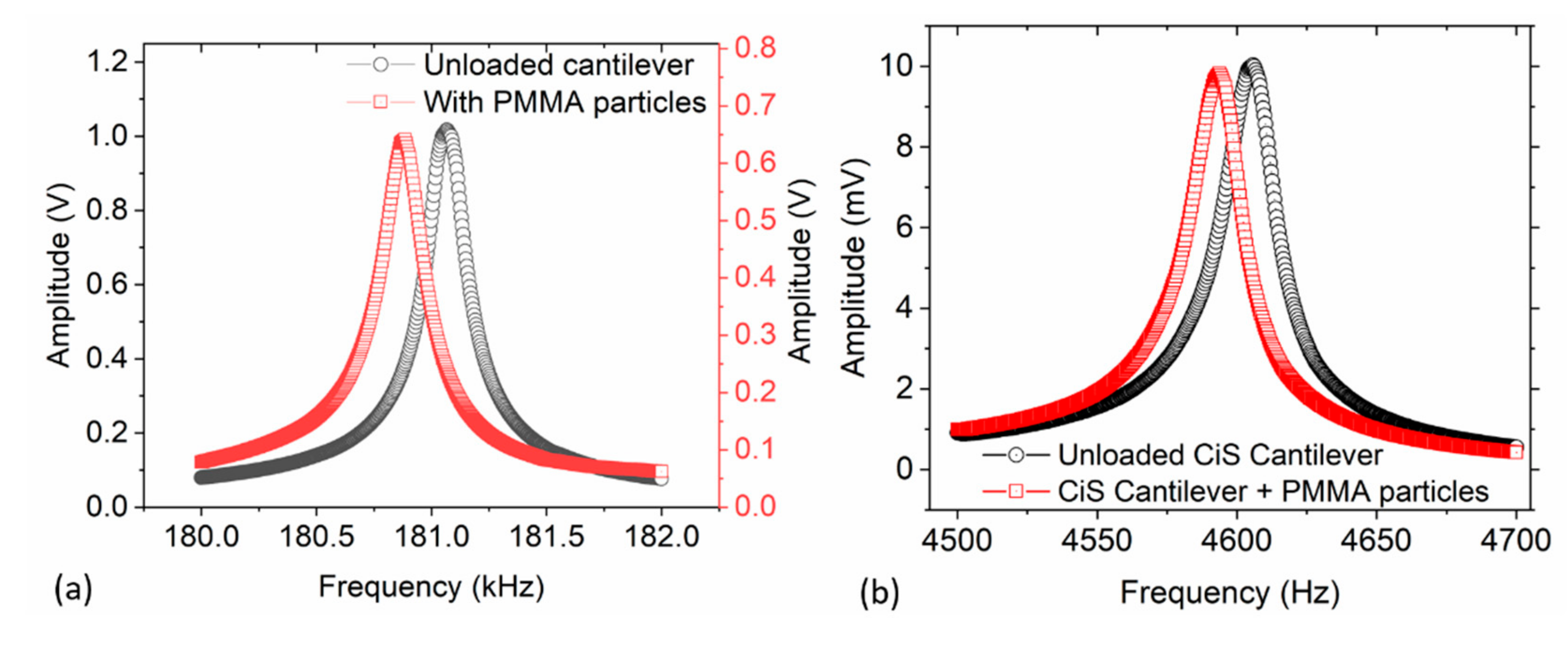
Initially, and for each particle adsorption cycle, the resonant frequencies of the cantilever before (
f0) and after (
fr) load deposition were measured. This was necessary to precisely determine the resonant frequency shift (Δ
f =
fr −
f0) due to the loaded mass Δ
m (i.e., mass of the droplet or adsorbed particles). Consequently, the particle and/or droplet mass was determined at a point
x from the fixed end of the cantilever beam of length
L. With the measured resonant frequency shift Δ
f, the mass of the cantilever
m0 and the mode-shape function
U(
xΔm), the drop or adsorbate mass Δ
m can be calculated according to [
30]:
where
The parameter
λ represents a modal constant, which is about 1.8751 for the first mode. To begin a new measurement cycle, the adsorbed particles were removed/cleaned from the cantilever surface and
f0 was measured again to average and compare with the initially obtained value.
2.6. Cleaning and Removal of Adsorbed Particles from the Cantilever
Whenever particles or analytes are collected on a silicon cantilever for mass detection or otherwise, they often adhere on the sensing surface. For instance, airborne particles such as cigarette smoke sticking on the surface of silicon cantilever can easily be removed by immersing and gentle agitating the cantilever in a glass containing acetone (for a few minutes) [
31]. However, after adsorbing liquid-dispersed MPS particles on the silicon cantilever, conventionally much more effort is required to remove and clear these particles from the sensing surface.
In this work, therefore, regeneration of particle-adsorbed cantilever sensors (
Figure 5a) was mainly performed by immersion in acetone and sonication for about 3–5 min. With this approach, the particles were detached and cleared from the surface (
Figure 5b). Since cleaning the fragile silicon cantilevers by sonication is a risky process, often bound to break and destroy the sensors, we clipped each particle-laden cantilever on the flat surface of a metallic mounting before immersing it in a glass containing acetone. This mechanism was successfully implemented and resulted in nearly 100% particle removal efficiency and alleviated the risk of damaging the cantilevers.