1. Introduction
Metal oxides have been extensively studied for chemoresistive gas sensors because of their high reactivity with various gas molecules as well as their adoptability to modern microfabrication technologies [
1]. It is well known that the molecular adsorption of gas phases followed by the chemical reaction with the oxide occurs by the charge exchange between the gas molecule and oxide, and the consequent change of conductance change in the oxide is translated to the sensing signal through the metal electrodes underneath [
2,
3,
4]. In such oxide gas sensors, the surface plays the role of the sensory receptor by the supply of gas molecular ionosorption sites while the bulk structure donates charges for the ionosorption reaction on the surface. At the same time, the bulk structure also works as a transducer, which reveals the change of conductance (or resistance) as an electric signal. A typical geometry for the operation of metal oxide gas sensors is configured by the receptor and transducer integrated into the single metal oxide body.
Since the sensor operating principle is strongly affected by the structure of the sensor, both ‘nanosize effect’ and ‘nanosize limit effect’ have been reported in conduction-type gas sensors. The nanosize effect is a well-known observation in chemoresistive gas sensors, which describes the increase in the sensing response as the dimension of the sensor structure decreases to a nanosize [
4,
5,
6,
7,
8,
9,
10]. However, when the size decreases smaller than the depletion depth scale, the total depletion of carriers leads to the insulating nature in the sensor structure followed by the decrease of the sensor signal. Such declining response at the dimension below the depletion depth scale has been introduced as the nanosize limit [
10,
11]. Therefore, the highest response level can be achieved at the size comparable with that of the depletion depth for a given doping concentration of the oxide.
CNTs have been repeatedly reported to show the sensing capability toward NH
3 at room temperature (RT, ~25 °C) [
12,
13,
14,
15,
16,
17]. The interaction of NH
3 with the Fe and Fe
2O
3 at RT also was reported to occur via the dissociative molecular adsorption of NH
3 on the Fe and Fe
2O
3 clusters [
18,
19,
20,
21,
22,
23,
24,
25,
26,
27], respectively. Hematite exhibited higher ammonia sensing performance at the elevated temperature (150 °C) than that at RT [
26]. Meanwhile, the composite sensors, tin oxide-based carbon, showed a sensing capability toward NH
3 [
28,
29,
30] and NO
2 [
31]. Hieu et al. [
28] showed that the SnO
2/multi-wall CNT composite detected NH
3 at RT. Lee et al. [
29] reported about the SnO
2/carbon nanoflake composite for the detection of NH
3 at 400 °C. The different operating temperatures were possibly originated from the different mixing ratios between the carbon (a p-type conducting sensor operating at RT) and SnO
2 (an n-type conducting sensor operating at high temperature). Rigoni et al. [
30] also detected NH
3 using the indium tin oxide (ITO) and SWCNT composite at RT. Interestingly, the conducting type of the composite sensor changed from the p-type semiconductor to the n-type with high NH
3 adsorption due to the injection of many electrons into the structure.
In this study, we prepared composite sensors composed of iron or hematite (Fe2O3) and single-wall carbon nanotubes (SWCNTs), Fe:SWCNTs, and Fe2O3:SWCNTs, respectively, for the NH3 detection at RT, and scrutinized their sensing behaviours with pure hematite and SWCNT sensors. Fe:SWCNTs and Fe2O3:SWCNTs nanocomposite structures showed the enhanced NH3 sensing response at RT owing to the synergistic effect. In addition, based on the comparative study with single-body gas sensors, hematite or SWCNTs sensors, we explored the underlying sensing mechanism of the composite structure. The semiconductor depletion model for the nanosize effect and the nanosize limit effect was thoroughly revisited, thus we could overcome the nanosize limit via the new concept of separated receptors and transducers for the high-response chemoresistive gas sensors. While the large surface area of hematite nanoparticles with a thin carbon layer provides adsorption sites for ammonia gas molecules, the charges necessary for the ionosorption are supplied from the SWCNTs. The net result is the appearance of far more extended depletion in SWCNTs, enabling an enhanced sensor signal. We elaborate the origin of the synergy effect and the principle of the new sensor scheme. Furthermore, we showed that such a sensor scheme can be a general route to overcome the nanosize limit regardless of the junction types by comparison with the p-p junction composite of Ni2O3:SWCNT.
3. Results and Discussion
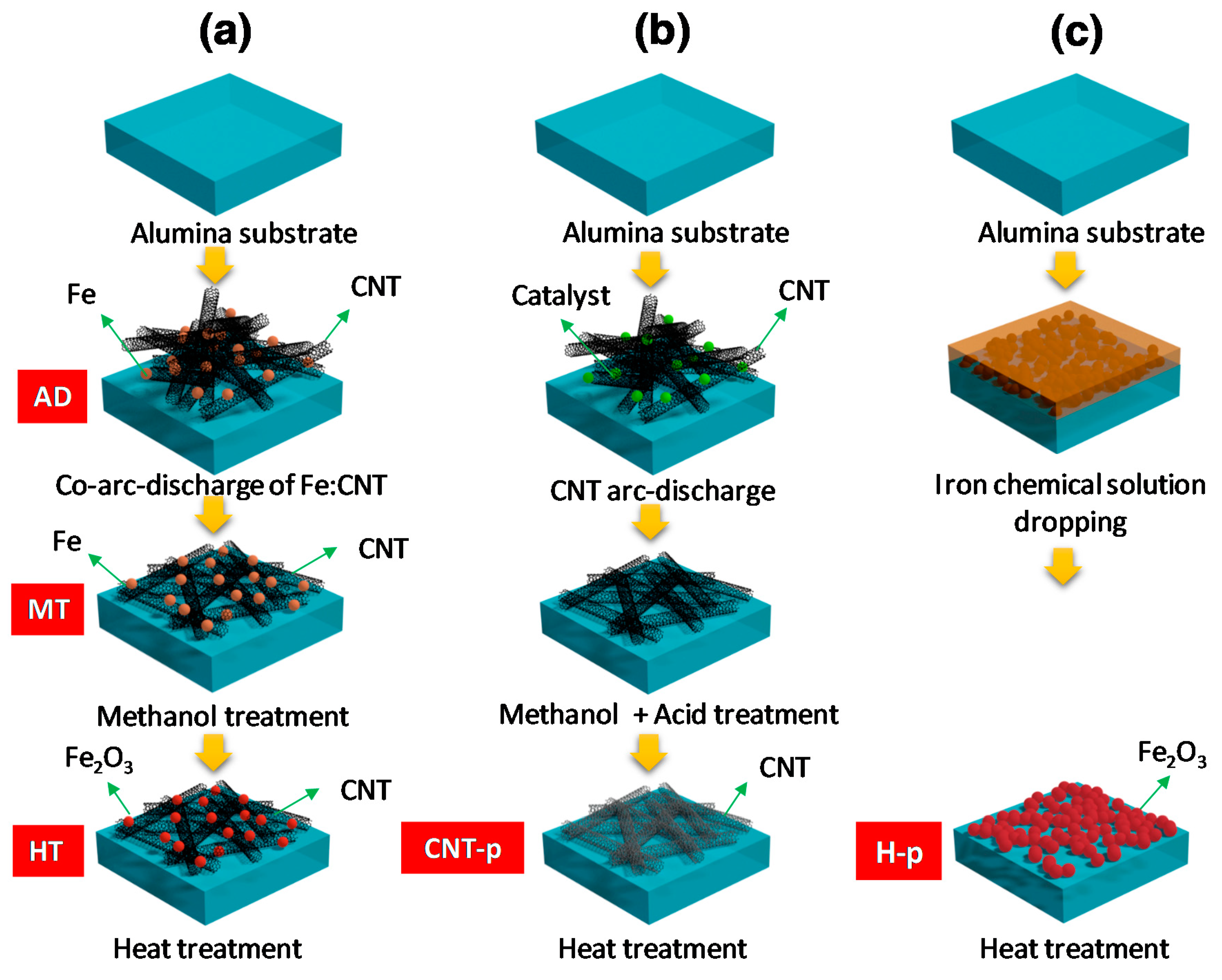
The sample AD (the Fe:CNT composite) showed a highly porous morphology with large volume among the SWCNTs as shown in the SEM image (
Figure 2a). The methanol treatment led the porous composite structure to collapse and form a compact mat-like structure (sample MT) due to the hydrophobicity of the SWCNTs (
Figure 2b) [
38,
39]. The sample HT (the Fe
2O
3:CNT composite) was obtained by thermal oxidation of the MT sample, in which Fe nanoparticles were oxidized to Fe
2O
3 nanoparticles while preserving the porosity of the MT sample as well as the thin carbon layer on the surface (
Figure 2c). Transmission electron microscopy (TEM) images clearly presents the dispersed hematite particles on the SWCNT network in the HT sample (
Figure 2d). The entangled SWCNT bundles with the diameter of ~10 nm supported Fe
2O
3 nanoparticles, of which the size range is large from several to ~30 nm. The high-resolution TEM image with the electron diffraction pattern of the inset (
Figure 2e) confirms the crystalline Fe
2O
3 nanoparticles in the Fe
2O
3:CNT composite structure. The hematite particles were encapsulated by amorphous or graphitic carbon layers as shown by
Figure 2e,f. The carbon layers encapsulating the Fe and Fe
2O
3 nanoparticles were preserved during the oxidation process at 400 °C.
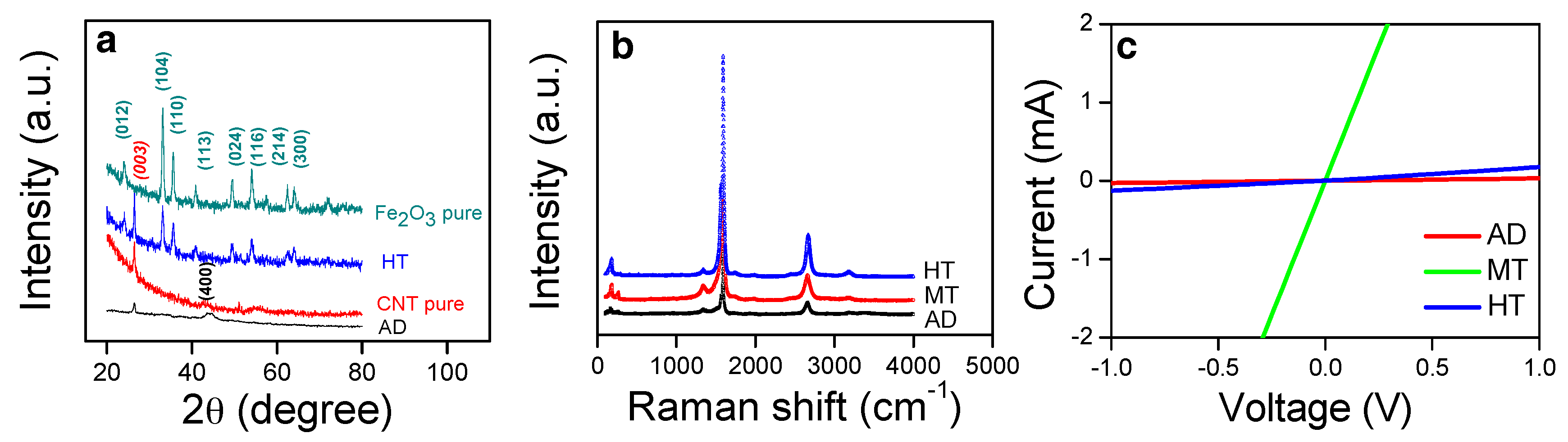
The XRD patterns of the AD and HT samples are shown in
Figure 3a. Those of pure Fe
2O
3 (H-p) and SWCNT (CNT-p) are displayed together for the comparison. The diffraction peak at ~44° matches (400) of Fe in the AD sample, indicating the distribution of Fe nanoparticles on SWCNTs. Meanwhile, diffraction peaks corresponding to the polycrystalline hematite (Fe
2O
3; JCPDS 00-024-0072) and graphite (JCPDS 98-002-0123) were identified in the HT sample. The Raman spectra of the composites are shown in
Figure 3b. The G- and D-mode peaks at ~1600 cm
−1 and ~1400 cm
−1 represent the graphitic lattice and the defective graphite structure, respectively, therefore the intensity ratio of G to D peaks can be considered a measure of the crystalline characteristic of the single-wall CNT [
40,
41]. All composites revealed the similar intensity ratio of G to D peaks indicating the preserved graphitic character in SWCNT during the process. This indicates that the crystallinity of the CNT in the composites was not noticeably degraded after the methanol or heat treatment, as exhibited by the sharp G-mode peak with respect to the D-mode peak.
The electrical properties of the composite structures are measured as shown in
Figure 3c. All composite samples exhibited excellent ohmic contacts with the Au electrodes at RT in air as indicative by the linearity in the current versus voltage curves. The high resistance of ~3.1 × 10
5 Ohm of the as-deposited Fe:CNT composite (AD) originates from the high porosity of the structure. The high porosity in the entangled nanowires comprises relatively a small number of contacts among the CNTs, which leads to the high resistance by a small cross-section area for the current path. The other Fe:CNT composite after the methanol treatment, the MT sample, shows two orders of magnitude lowered resistance of ~6.8 × 10
3 Ohm. The methanol treatment led to the collapse of the porous structure followed by formation of the compact mat-like structure [
38,
39], in which the cross-section area for the current path is large due to the great increase in the number of contacts among the CNTs. HT reveals the same compact structure with MT, however, Fe nanoparticles were oxidized to semiconducting oxide particles, Fe
2O
3. Despite similar compact structures between MT and HT, the much higher resistance of Fe
2O
3 than Fe caused a far increase in the resistance, ~1.3 × 10
4 Ohm. A partial burn-out of the SWCNTs could contribute to the increase of the resistance in the HT structure [
38].
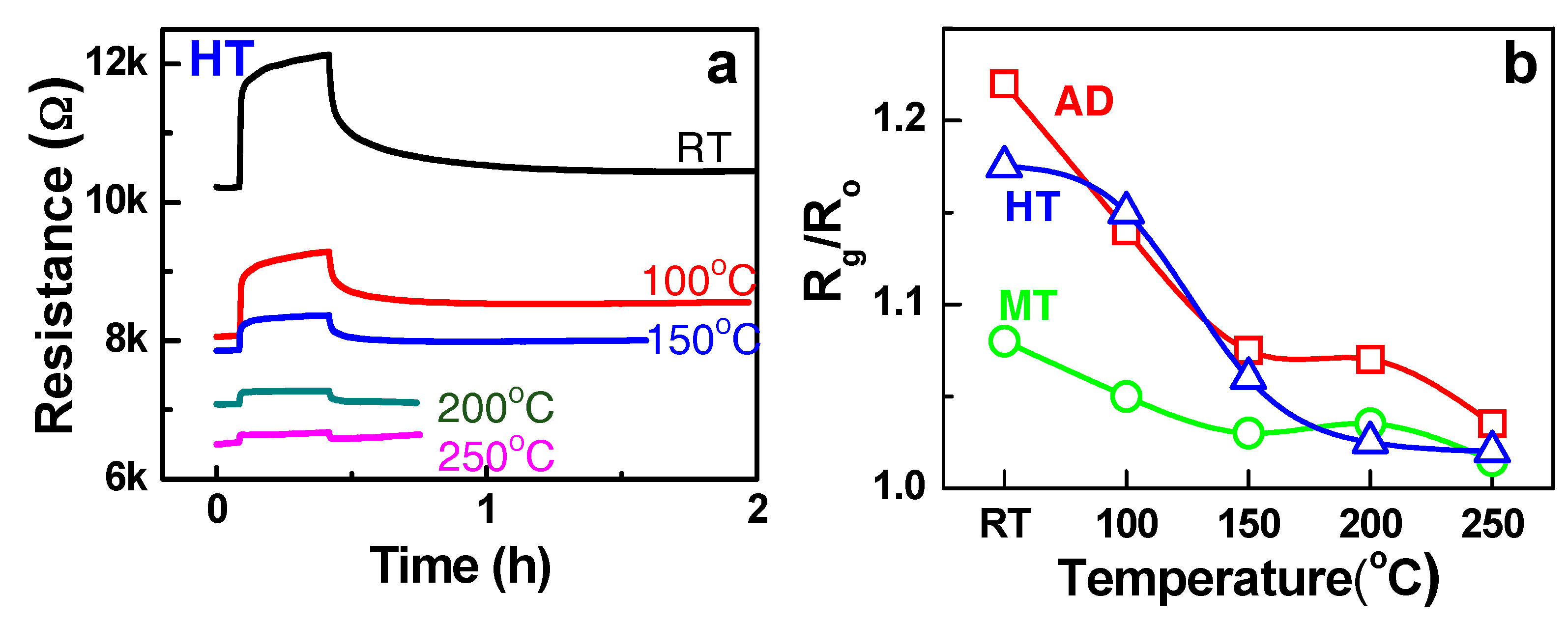
The resistance changes of the HT composite toward 500 ppm NH
3 gas diluted in dry air were measured at different temperatures, as shown in
Figure 4a. The increase of the resistance upon exposure to the reducing gas like NH
3 indicates that the HT composite is a p-type sensor, in which the positive charge carrier hole (h
+) is supplied to the adsorbed NH
3 molecules, thereby enabling ionosorption (e.g.,
). The change of the resistance was highest at RT, however it decreased by the increase of the temperature. The similar p-type semiconducting sensing behaviours and the temperature dependence were observed in the Fe:SWCNT composite (AD and MT samples) as shown in
Figure 4b. The RT sensing response was attributed to the molecular adsorption of NH
3 on the sensor (not combustion of NH
3). Furthermore, the p-type behaviours in the sensing mechanism indicates that p-type SWCNTs are the main contributor for the charge exchange during NH
3 adsorption.
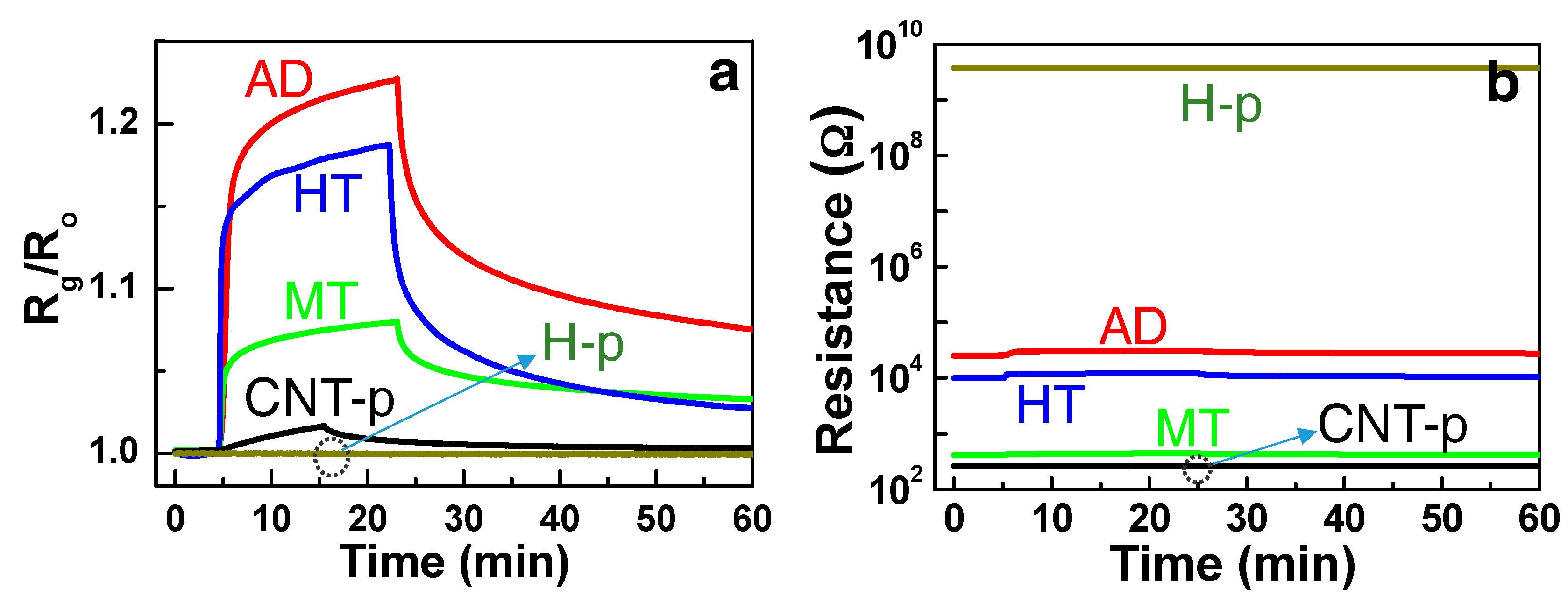
The RT response-recovery curves of the composite structures (AD, MT, and HT) are presented in
Figure 5a. The response and recovery times of the HT structure were ~1.2 and ~1.8 min, respectively, as estimated in previous studies [
8,
9]. The measurements from pure SWCNT (CNT-p) and pure hematite (H-p) structures with 500 ppm NH
3 are also plotted for comparison. The corresponding resistance changes of the sensor structures are presented in
Figure 5b. It can be immediately noted that the H-p sensor with very high resistance showed negligible response and the CNT-p sensor with very low resistance also showed a small response to NH
3 at RT. However, the composite-type sensors revealed the enhanced chemoresistive response level. The observed sensing response with the composites is elucidated by the synergistic interplay between the hole carrier concentration in the transducer and the adsorption sites density of the receptor. Holes are supplied by p-type SWCNTs, of which concentration is directly translated to the sensor resistance. The density of adsorption sites depends on the physical and chemical nature of the nanoparticles distributed on SWCNTs. We thoroughly investigate the synergetic effect in the sensing response by examination of the electronic and morphological properties of the structures.
Ionosorption is chemical adsorption (chemisorption) of the gas molecule on the material surface as an ionic state, which can be completed by either capture or release of electrons by or from the material, respectively. Therefore, a molecule impinging on the receptor can end up with adsorption followed by the charge exchange with the transducer. The charge supplied to or extracted from the transducer leads to a resistance change of the transducer. The ratio of resistances before and after gas adsorption (R
o and R
g, respectively) is taken as the sensor response signal, which is defined by Equation (1),
for sensing of oxidizing gases on n-type doped materials or reducing gases on p-type materials. The reverse (
) can be defined for sensing oxidizing gases on p-type doped materials or reducing gases on n-type materials.
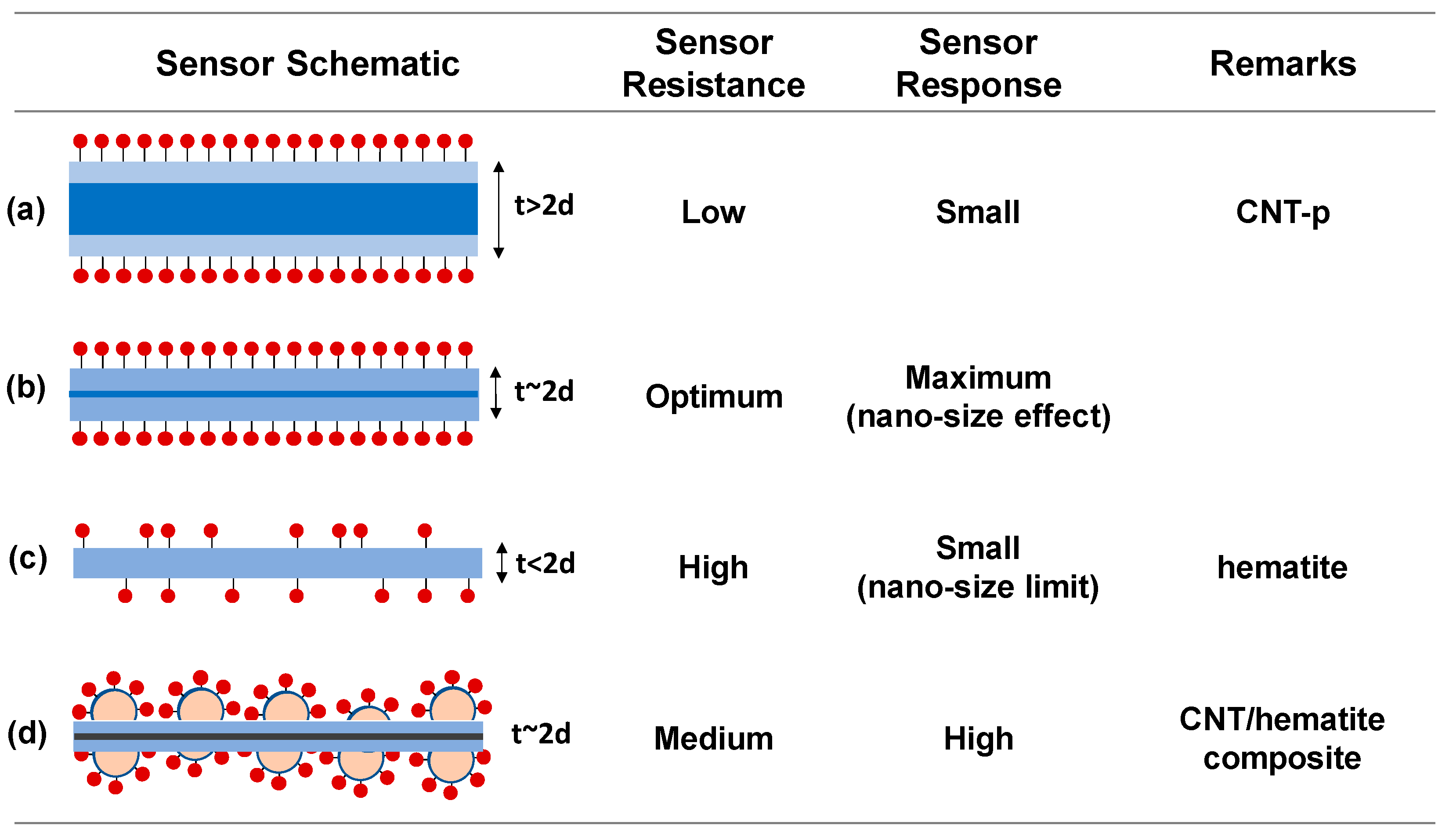
The simple condition, the uniform thin film oxide sensor with a thickness (t), is illustrated in
Figure 6a. The resistance is inversely proportional to the thickness,
(A is arbitrary). If a reducing gas adsorbs by the hole supply from the p-type film (such as the NH
3 adsorption on SWCNT), the surface region becomes depleted of holes with the depletion depth (d) from the each side, leading to the change of the film resistance,
[
36]. Therefore, the response of the thin film sensor is given by Equation (2),
S approaches the unity (= 1) or negligible response in the condition of t >> d (
Figure 6a) [
10]. The nano-size effect in gas sensing is observed by the enhancement of response as thickness (t) approaches twice of the depletion depth, i.e., t = ~2d, of which the condition is illustrated in
Figure 6b. The nano-size effect is highest when t = ~2d or the film thickness is comparable with the depletion depth in the given doping concentration. Since the depletion in the transducer occurs by the molecular adsorption of the gas phase on the receptor surface and consequent charge extraction, the total depletion condition is given by
, where n
d and N
ad are the carrier concentration of the transducer (cm
−3) and the areal density of molecular adsorption on the receptor (cm
−2), respectively, in the steady state for a given gas concentration. Considering the resistance and the adsorption site intensity in the structure, the difference in the sensing response among AD, MT, and HT (see room temperature data in
Figure 4b) can be elucidated by the sensor structural conditions between
Figure 6a,b.
However, in the case of t < 2d, the total charges in the transducer structure are smaller than the steady-state adsorption molecules (n
dt < N
ad). Only part of the steady-state adsorption sites can be occupied. The completely depleted state in the transducer before and after the gas adsorption is depicted in
Figure 6c, where the absence of the sensing response, i.e., S = ~1, is detected. It should be noted that Equation (2) cannot be applied to this condition. The maximum response as proposed in
Figure 6b begins to decline as the thickness decreases like the schematic in
Figure 6c. We already called this the declining response with the decrease in the sensor dimension below the depletion depth scale as ‘nanosize effect limit’. We had repeatedly observed the nanosize effect limit in several oxide gas sensors [
9,
10,
11,
42]. We have to note that both of the nanosize effect (increase in the sensing response with decreasing the sensor size towards the depletion depth scale) and the nanosize limit (decrease in the sensing response with further decreasing the sensor size below the critical limit of the depletion depth) are conceived from the independence between the receptor and transducer action in the single-body scheme of the gas sensor. Therefore, simultaneous control of the dimensions of the oxide element, carrier concentration, and adsorption site density is required to achieve the maximum response signal in the conventional single-body oxide sensors.
To discuss the synergetic effect of the composite sensors, we need to understand the origin of zero and negligible response levels from the H-p and CNT-p sensors, respectively, as shown in
Figure 5a. Detection of NH
3 in CNT-based sensors at RT has been reported [
12,
13,
14,
15,
16,
17], and the relatively good recovery at RT was available due to the weak adsorption binding between CNT and NH
3 [
43]. However, the CNT-p exhibited a small response level (S = ~1.02 or R
g = ~R
o in Equation (1)), indicating that the resistance modulation in the SWCNT by the NH
3 adsorption is very small. The small response is attributed to the relatively high hole carrier concentration (i.e., high conductance in SWCNTs) compared with the small adsorption site density of the SWCNTs (n
dt >> N
ad). This case corresponds to the condition schemed in
Figure 6a, where the adsorption sites are occupied at the steady state, however, the consequent surface deletion depth is relatively small.
Meanwhile, the origin of the no response (S = ~0) in the H-p sensor (
Figure 5a) is different with that in the CNT-p sensor. It is reported that an n-type hematite sensor responded to ammonia at a high temperature like other oxide gas sensors [
26]. However, the hematite film exhibited an agglomeration among nanometric hematite particles, thereby revealing a very high resistance (>GΩ) due to fully depleted electron carriers as shown in
Figure 5b. The semi-insulating hematite film corresponds the condition of n
oxt << N
ad as presented in
Figure 6c. Ionosorption of 500 ppm NH
3 molecules followed by the release of electrons into the hematite could not lead to any detectable resistance change in the H-p sensor, as manifested by the nearly zero response.
Contrary to the poor sensing responses of the SWCNT and hematite, the enhanced response in their composite structures can be understood by investigation about their electronic and morphological structures. Namely, the resistance of the Fe:SWCNT composite is comprised by competition between the high-resistance Schottky contact between the Fe and SWCNT and low-resistance ohmic contact among the SWCNTs. Therefore, the resistances of the composites are located in between that of CNT-p and H-p (
Figure 5b), which is directly reflected to the operating condition. The sensor operating conditions of the composites lie between the conditions presented in
Figure 6a,c. Meanwhile, the proposed morphology of the composite sensors with the enhanced sensing response is depicted in
Figure 6d. The high-performance composite structure can be designed by consideration about two aspects. (1) The carbon-encapsulated Fe (or Fe
2O
3) nanoparticles with great surface area work as receptors for the NH
3 adsorption, and (2) the nanoparticle/SWCNT heterojunctions decrease the conductance of the SWCNT transducer. Both effects cooperatively push to the condition of
Figure 6b of higher response.
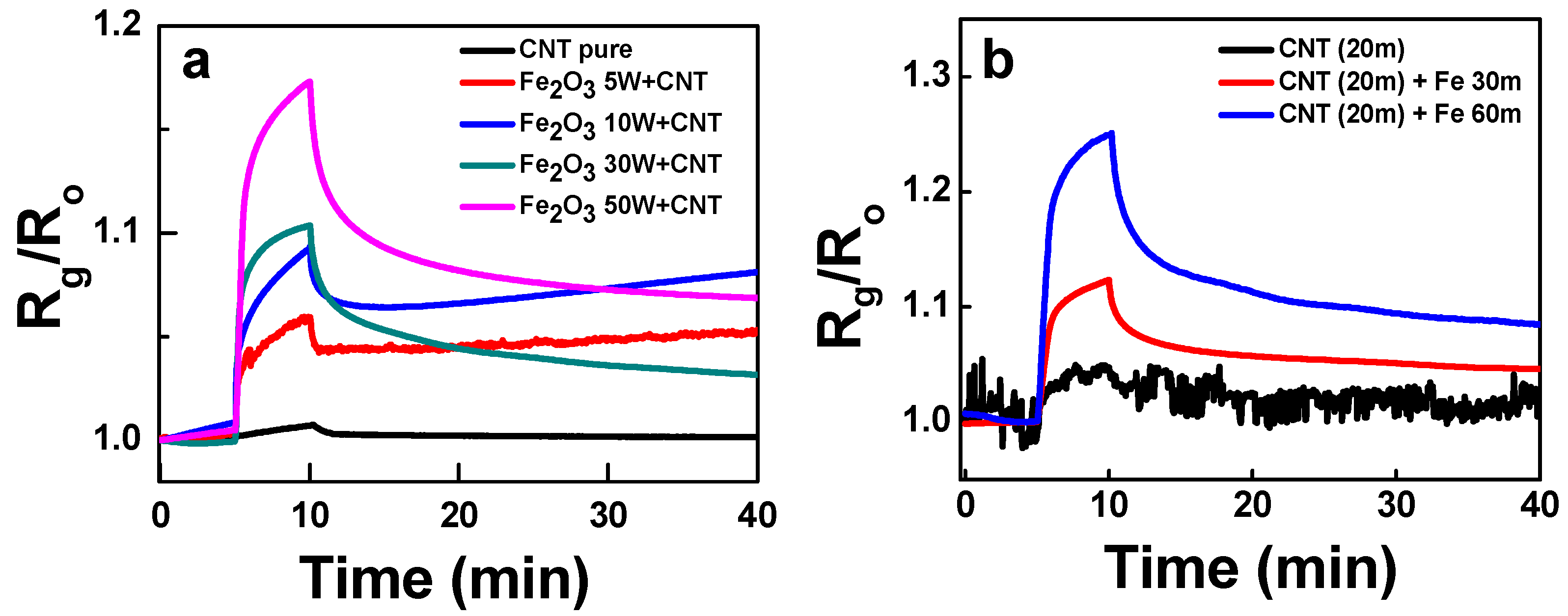
The effect of concentration and distribution of Fe
2O
3 nanoparticles on the sensor response of composites were examined as well. The Fe
2O
3 concentration in the Fe
2O
3:SWCNT composites could be increased by adding more Fe wires into the graphite rod during the co-arc-discharge process. The synthesis using the more Fe wires produced the composite with the higher carbon-encapsulated Fe
2O
3 concentration. The response to 500 ppm NH
3 of the structures are shown in
Figure 7a. The higher responses were obtained with the more Fe wires due to higher receptor concentration in the composites. Since the NH
3 adsorb on SWCNT as shown by the response of the CNT-p sensor, the high distribution of carbon-encapsulated hematite particles will increase the specific adsorption sites for NH
3.
Whereas, when Fe was sputter deposited on the mat-like SWCNTs thin film, a thin film of Fe seats on the SWCNT mat. The following oxidation process converts Fe to Fe
2O
3 nanoparticles agglomeration without the carbon shell layer. The sputter-based composite will show a morphology of thin film Fe
2O
3 nanoparticle receptor piled up on the mat-like SWCNT transducer. The cross-section views of the structures are shown in
Figure S3 that signifies increase in the thickness with the sputtering time. The sputter-based composite also revealed higher response with the Fe
2O
3 thickness (
Figure 7b). It was already repeatedly shown that the NH
3 molecules can adsorb on Fe or Fe
2O
3 nanoparticles at RT via the molecular adsorption reaction
or the dissociative adsorption reaction
, etc. [
19,
20,
21]. In this way, either the ammonia adsorption on carbon-encapsulated Fe
2O
3 or the direct adsorption on Fe
2O
3 observed in
Figure 7 can be supported.
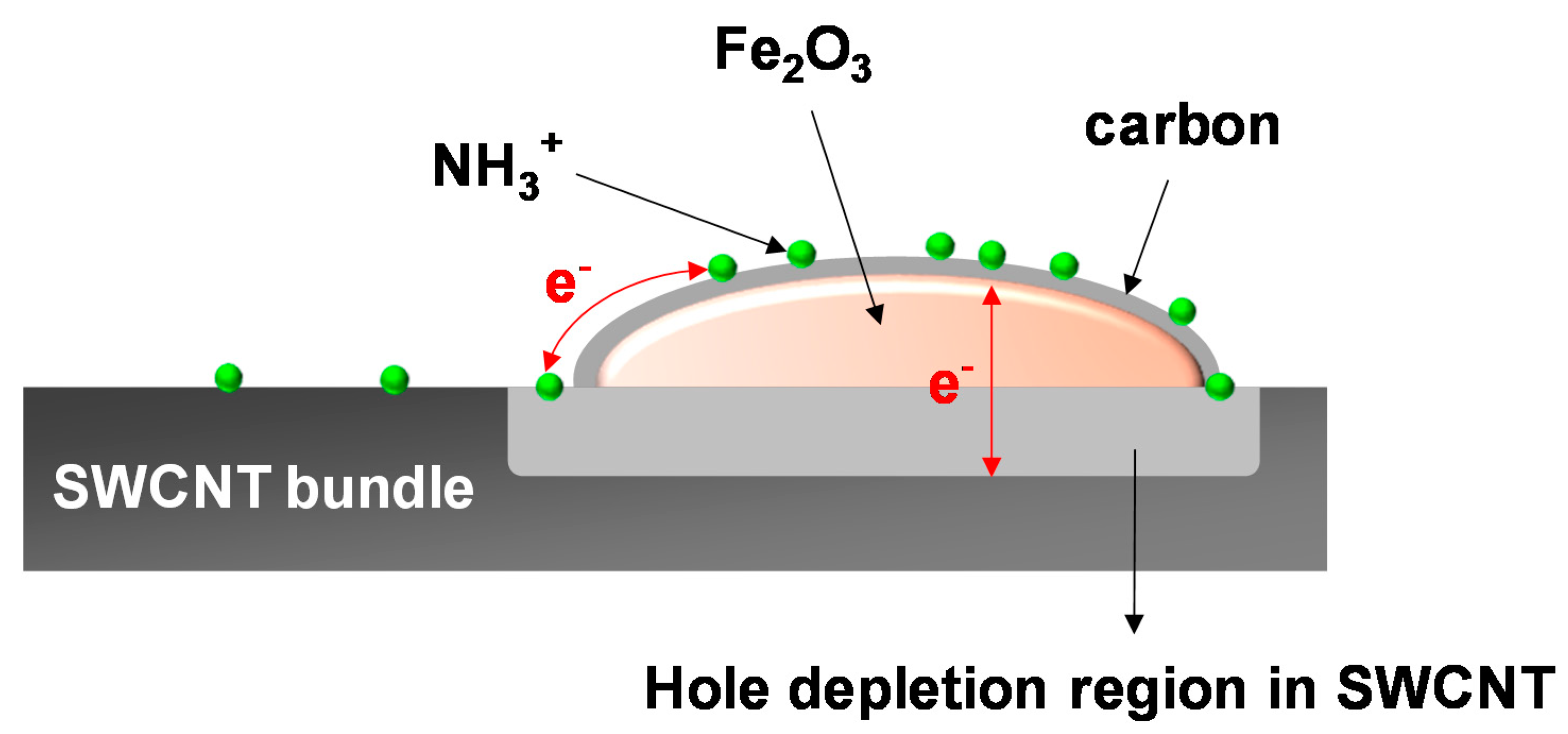
The microscopic view about the ammonia adsorption on the carbon-encapsulated hematite nanoparticle is presented in
Figure 8. The Fe
2O
3/SWCNT heterojunction results in the formation of the hole depletion region in the SWCNT. When the NH
3 molecules adsorb on the carbon-encapsulated Fe
2O
3 receptor at RT, electrons released via the adsorption reaction are transferred to the SWCNTs to recombine with holes. The electron transfer can be achieved by either surface conduction or tunneling through the hematite particles. The resultant increase in the hole depletion depth in the SWCNT transducer is translated to the sensor signal following Equation (2). From this perspective, an enhanced sensor response, i.e., far increased resistance modulation of the SWCNT transducer, is available by increase in the adsorption site density for the NH
3 molecules. The higher adsorption can be achieved by the higher concentration of nanoparticle receptors.
We also fabricated the Ni
2O
3:SWCNT as the p-type-oxide:SWCNT composite structure by the same method used for the preparation of the Fe
2O
3:SWCNT composite sensor. The temperature dependence of sensing responses to 100 ppm NH
3 is presented in
Figure S4. All results follow those from the Fe
2O
3:SWCNT composite. This indicates that the enhanced SWCNT sensor performance can be achieved by adding any type of receptor if it can provide adsorption sites for NH
3.
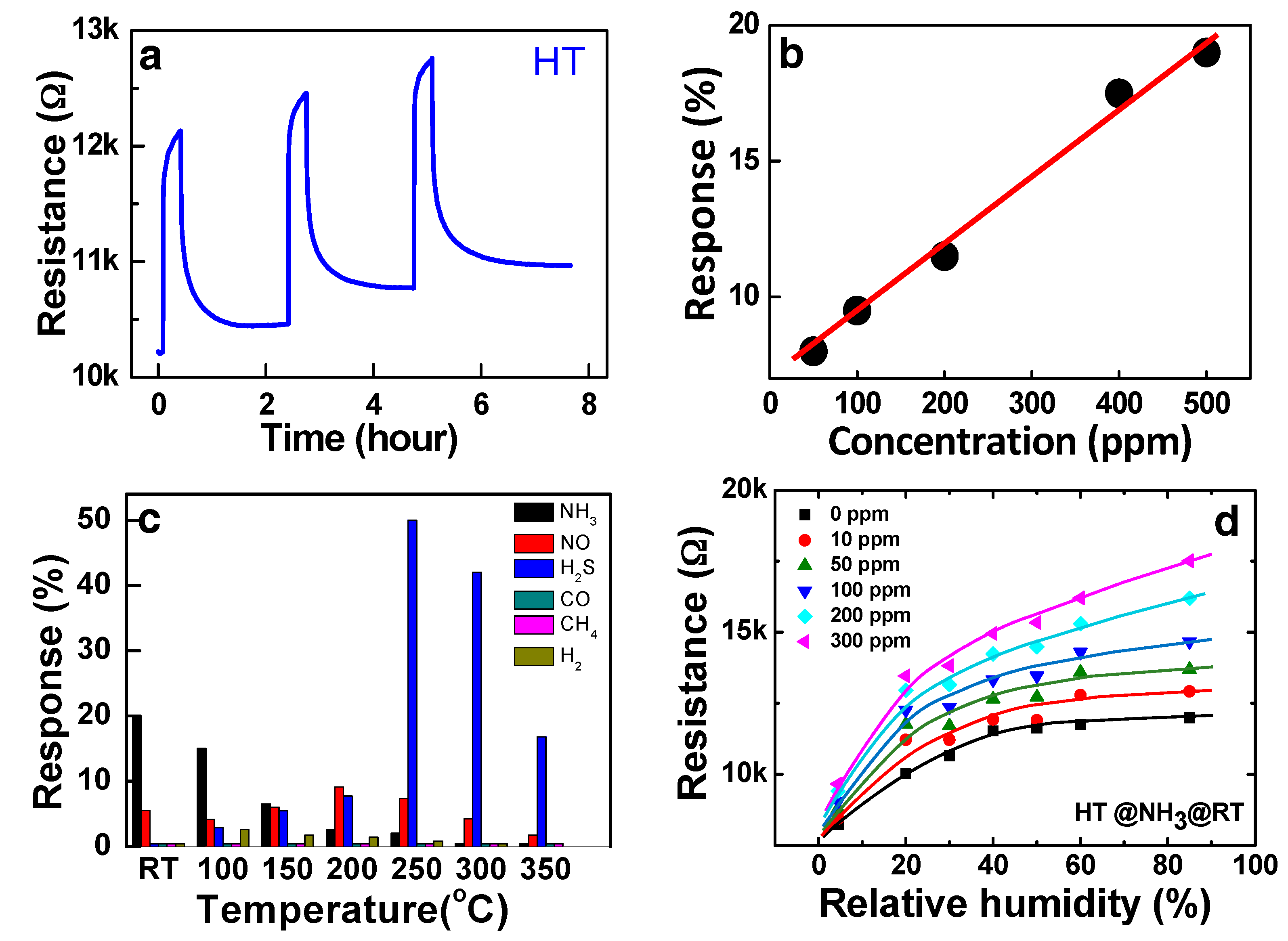
The various ammonia sensing properties of the HT sensor (Fe
2O
3:CNT composite) at RT are summarized in
Figure 9. In the test of the repeatability of sensing, the baseline resistance after recovery continued to increase by the repetition of the sensing cycles as shown in
Figure 9a. The chemical forms of ammonia adsorption can be variable in reality [
19,
20,
21], and a finite amount of
and/or
, which binds more strongly with the adsorption sites than
, remains adsorbed during the recovery cycle [
19]. The remnant adsorption can be accumulating as the response-recovery cycles are repeated. The linearity of the HT sensor at RT for ammonia detection was measured at the various concentrations in the range from 50 to 500 ppm as shown in
Figure 9b. The responses of the HT sample to other reducing and oxidizing gases were also examined, which shows high selectivity of the sensor to ammonia at RT (
Figure 9c). The preliminary examination shows that the composite structure can be further developed to commercial sensor structures. It is also interesting to note in
Figure 9c that the composite structure can be used as a H
2S sensor at high temperatures with good selectivity.
The humidity in the air is considered one of the most critical issues to operate the oxide gas sensor at RT. The H
2O molecules strongly affect the sensing signals via adsorption on the surface of the sensor. At low temperatures below 400 K, preferentially dissociated water molecules form the chemisorption layer of hydroxyl ions on the very surface, and further water molecular impingement forms multilayers of physisorbed water molecules [
44,
45,
46,
47]. The water molecular adsorption provides the ionic conduction path, thereby increasing the conductance of the oxide. In addition, electrons are released via the water molecular adsorption [
3], which also contributes to the further increase of the conductance in the case of n-type semiconducting oxides. We found that the n-type ZnO thin film can be used as a selective humidity sensor because it shows the conductance increase by relative humidity (RH) (
Figure S5). Rapid decrease of the sensing response above ~60% RH may account for an enhanced conduction due to several layers of the physisorbed water molecules. The conductivity change resulting from the physisorbed water molecules as well as the simultaneous electron injection into the oxide can manifest the humidity sensing of the ZnO.
Whereas, the behaviour of the p-type Fe
2O
3:CNT composite sensor is opposite with ZnO as shown in
Figure 9d, in which the conductance of the sensor was decreased by the humidity (see the curve with the zero NH
3 flow). Since SWCNTs also respond to water molecules [
48], the opposite humidity effect on the sensor conductance indicates that a part of the holes in the SWCNTs are compensated by the released electrons from the chemisorbed water molecule.
Figure 9d shows that the NH
3 gas in a ppm concentration range competitively adsorb on the surface in the humidity as indicative by sensing signals. Note that the ionic conduction by the adsorption of water molecules is not significant. Therefore, the ammonia sensing signal of HT in the humid condition still can result from the hole conduction mechanism in the SWCNT transducer. As observed, the ammonia gas concentration cannot be precisely determined in humidity unless the humidity is measured separately. The simplest solution is the monitoring of the humidity at the same location. Many semiconductor type humidity sensors have been developed to be mounted together with the chemoresistive sensor [
45]. In this case, a microprocessor to calibrate the signals based on the humidity is required as well.