Investigation of Polyurethane Matrix Membranes for Salivary Nitrate ISFETs to Prevent the Drift
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of NO3-ISFETs without Purification
2.3. Determination of Calibration Curves and Selectivity Coefficients in NO3-ISFETs
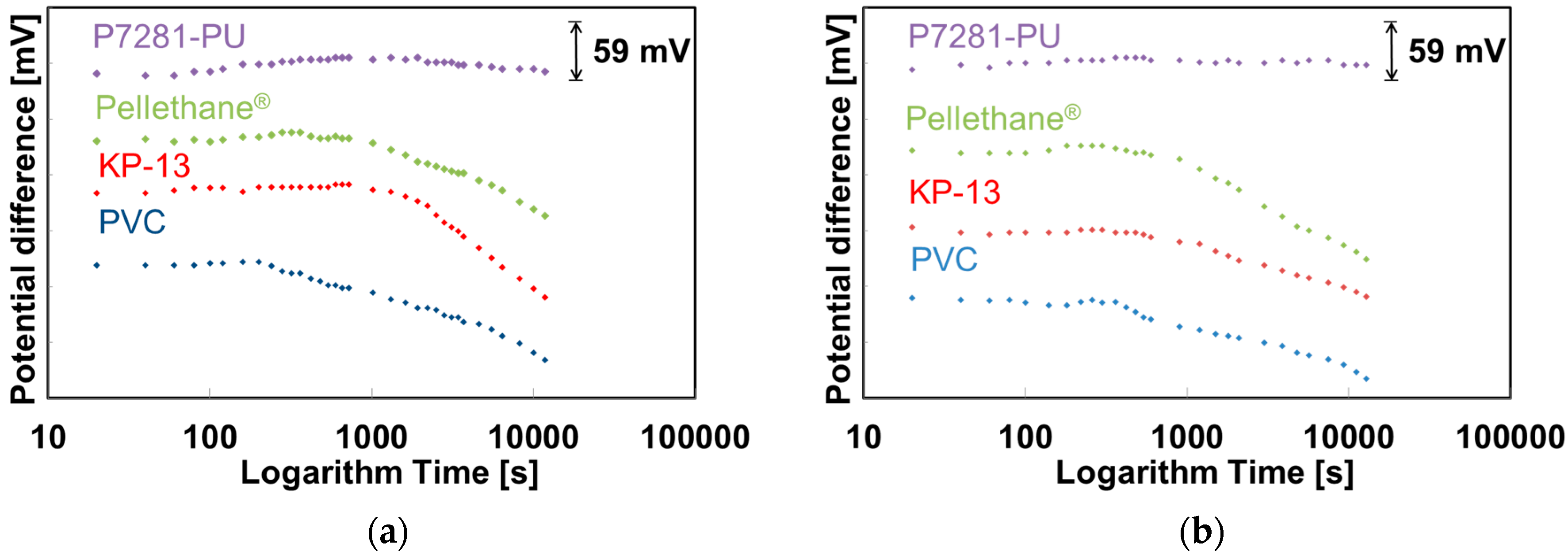
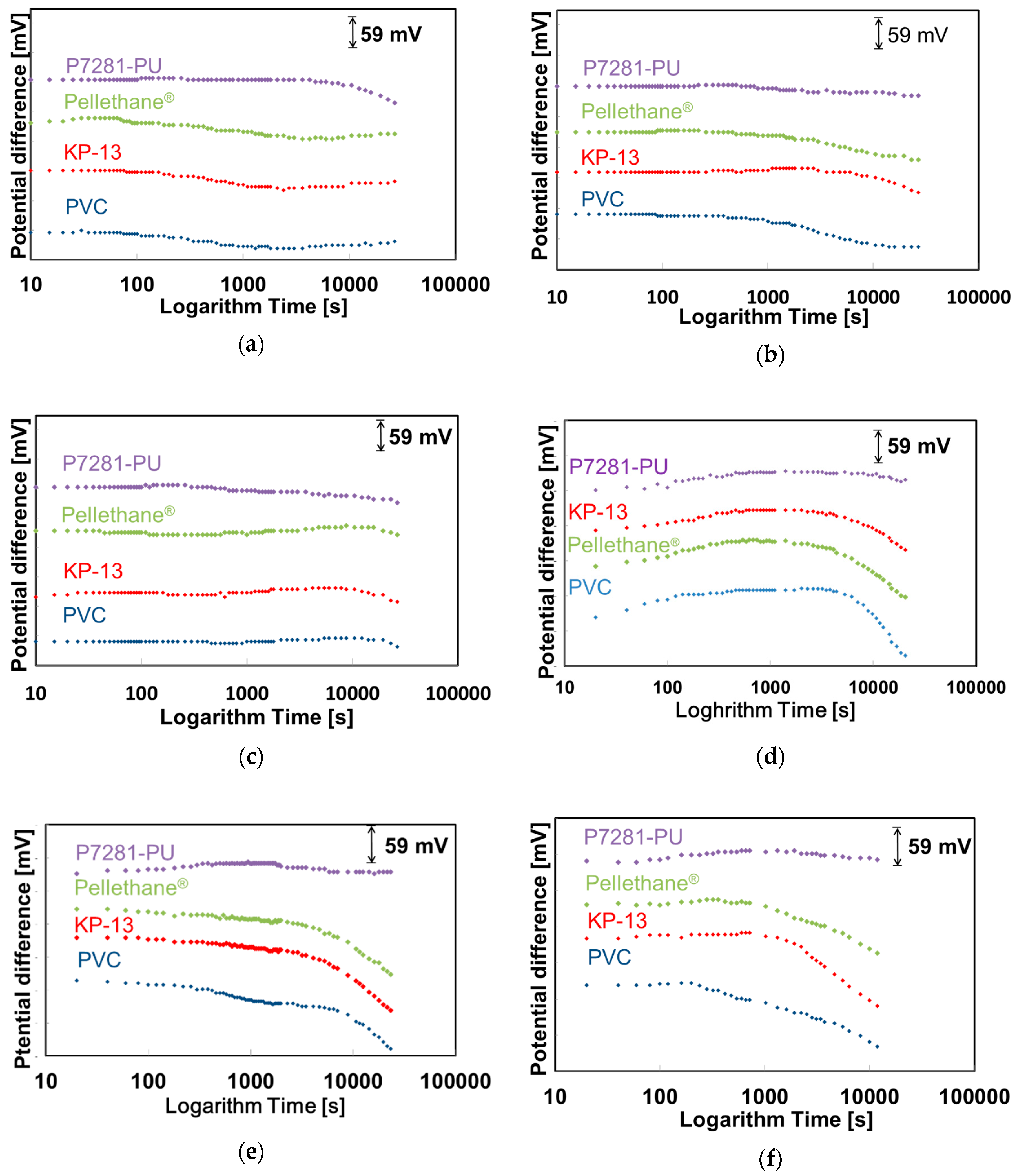
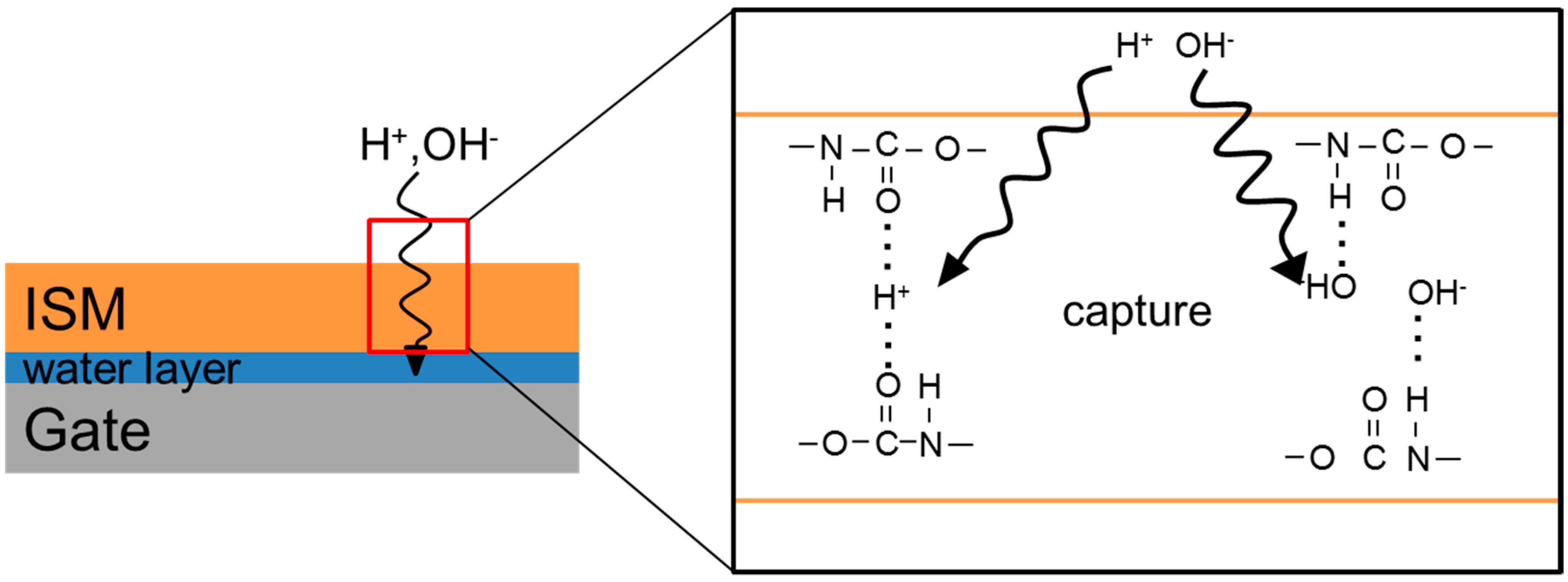
2.4. Evaluation of Initial Drift of the NO3-ISFETs
3. Results and Discussion
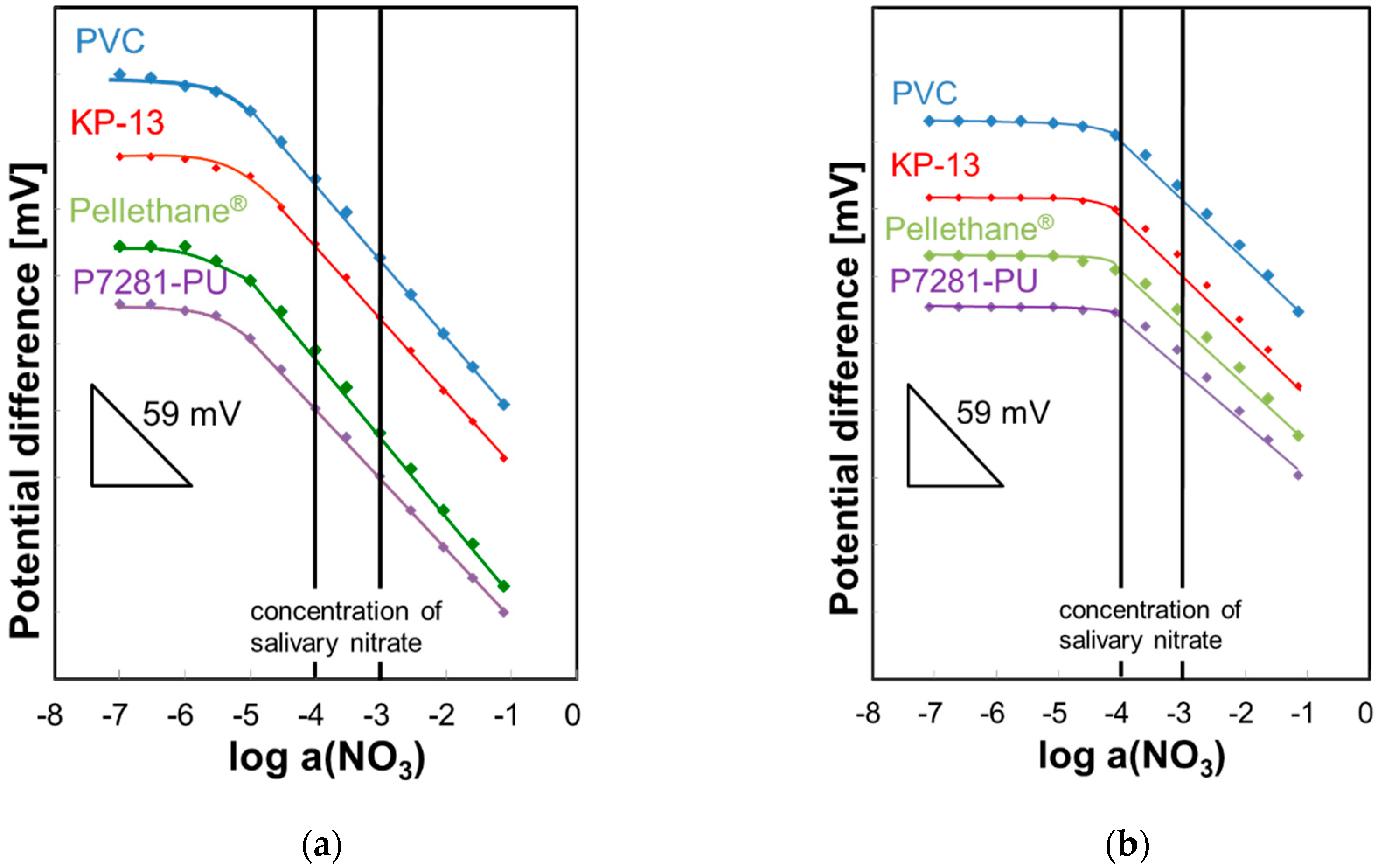
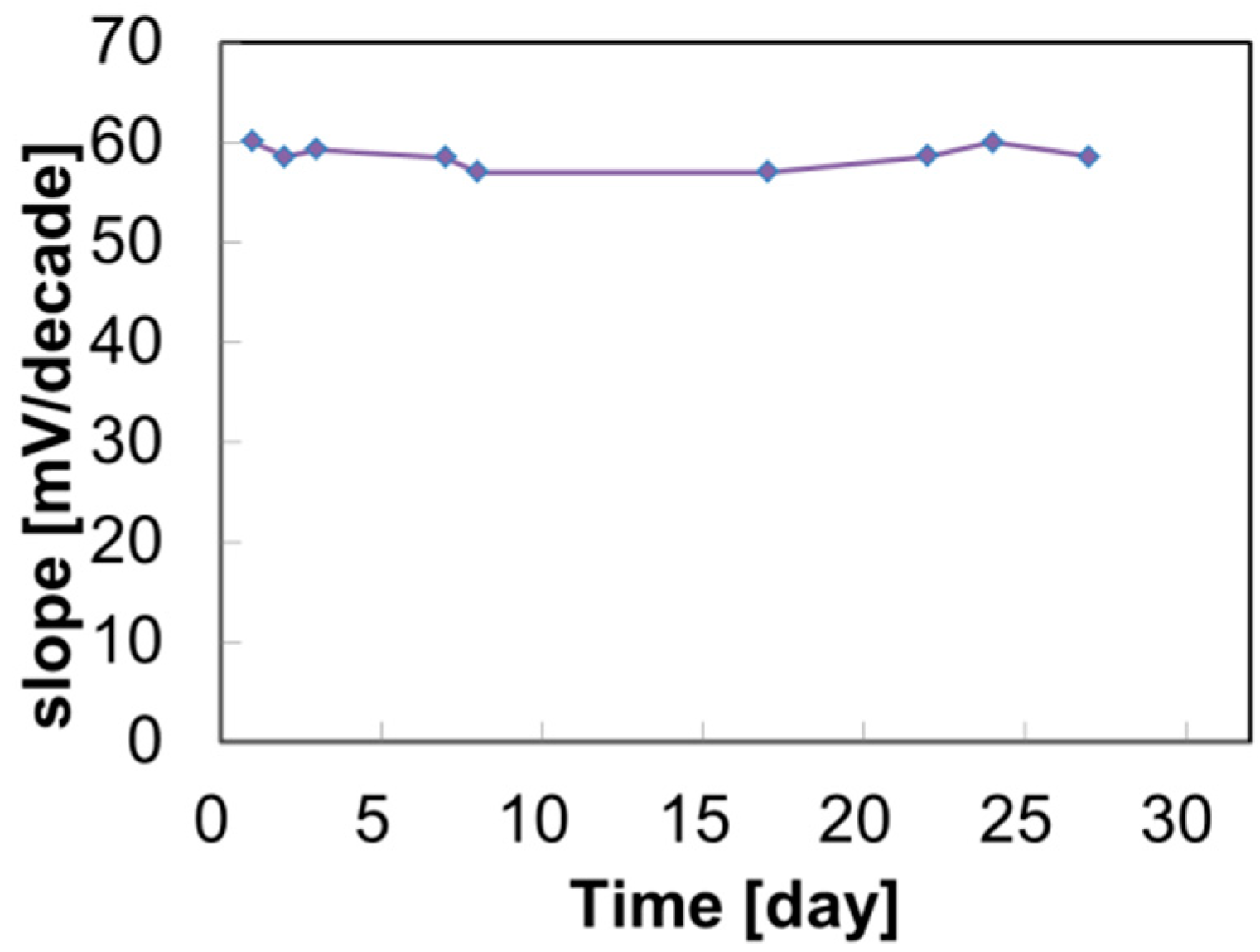
3.1. Calibration Curves and Selectivity Coefficients
3.2. Evaluation of Initial Drift in Saliva and Various Solutions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
References
- Hammen, C. Stress and depression. In Annual Review of Clinical Psychology; Annual Reviews: Palo Alto, CA, USA, 2005; Volume 1, pp. 293–319. [Google Scholar]
- Kendler, K.S.; Gardner, C.O. Depressive vulnerability, stressful life events and episode onset of major depression: A longitudinal model. Psychol. Med. 2016, 46, 1865–1874. [Google Scholar] [CrossRef] [PubMed]
- Kessler, R.C. The effects of stressful life events on depression. Annu. Rev. Psychol. 1997, 48, 191–214. [Google Scholar] [CrossRef] [PubMed]
- Charmandari, E.; Tsigos, C.; Chrousos, G. Endocrinology of the stress response. In Annual Review of Physiology; Annual Reviews: Palo Alto, CA, USA, 2005; Volume 67, pp. 259–284. [Google Scholar]
- Schwab, K.O.; Heubel, G.; Bartels, H. Free epinephrine, norepinephrine and dopamine in saliva and plasma of healthy-adults. Eur. J. Clin. Chem. Clin. Biochem. 1992, 30, 541–544. [Google Scholar] [PubMed]
- Zetterquist, W.; Pedroletti, C.; Lundberg, J.O.N.; Alving, K. Salivary Contribution to Exhaled Nitric Oxide. Eur. Respir. J. 1999, 13, 327–333. [Google Scholar] [CrossRef] [PubMed]
- Sato, E.F.; Choudhury, T.; Nishikawa, T.; Inoue, M. Dynamic aspect of reactive oxygen and nitric oxide in oral cavity. J. Clin. Biochem. Nutr. 2008, 42, 8–13. [Google Scholar] [CrossRef] [PubMed]
- Fleming, I.; Busse, R. NO: The primary EDRF. J. Mol. Cell. Cardiol. 1999, 31, 5–14. [Google Scholar] [CrossRef] [PubMed]
- Toda, M.; Den, R.; Hasegawa-Ohira, M.; Morimoto, K. Effects of woodland walking on salivary stress markers cortisol and chromogranin A. Complement. Ther. Med. 2013, 21, 29–34. [Google Scholar] [CrossRef] [PubMed]
- Cozma, S.; Martu, C.; Ghiciuc, C.M.; Patacchioli, F.R.; Cozma, L.C.D. Salivary alpha-amylase activity—A novel non-invasive biomarker in clinical pathology. Revista De Chimie 2018, 69, 728–730. [Google Scholar]
- Fogt, E.J.; Untereker, D.F.; Norenberg, M.S.; Meyerhoff, M.E. Response of ion-selective field effect transistors to carbon-dioxide and organic-acids. Anal. Chem. 1985, 57, 1995–1998. [Google Scholar] [CrossRef] [PubMed]
- Janata, J. 20 Years of ion-selective field-effect transistors. Analyst 1994, 119, 2275–2278. [Google Scholar] [CrossRef]
- Li, X.H.; Verpoorte, E.M.J.; Harrison, D.J. Elimination of neutral species interference at the ion-sensitive membrane semiconductor-device interface. Anal. Chem. 1988, 60, 493–498. [Google Scholar] [CrossRef]
- Reichmuth, P.; van der Wal, P.D.; Puntener, M.; Schoning-Hammer, A.; Morf, W.E.; de Rooij, N.F.; Pretsch, E. Reducing the interference from CO2 or organic acids in ion-selective polymer membrane sensors having a field-effect transistor as internal reference element. Anal. Chim. Acta 2002, 464, 79–88. [Google Scholar] [CrossRef]
- Abramova, N.; Borisov, Y.; Bratov, A.; Gavrilenko, P.; Dominguez, C.; Spiridonov, V.; Suglobova, E. Application of an ion-selective field effect transistor with a photocured polymer membrane in nephrology for determination of potassium ions in dialysis solutions and in blood plasma. Talanta 2000, 52, 533–538. [Google Scholar] [CrossRef]
- Espadastorre, C.; Meyerhoff, M.E. Thrombogenic Properties of untreated and poly(ethylene oxide)-modified polymeric matrices useful for preparing intraarterial ion-selective electrodes. Anal. Chem. 1995, 67, 3108–3114. [Google Scholar] [CrossRef]
- Wakida, S. Polymeric matrix membrane field-effect transistors—Sodium-ion sensors for medical applications. In ACS Symposium Series; ACS Publications: Washington, DC, USA, 1992; Volume 487, pp. 246–251. [Google Scholar]
- Moriuchi, T. Development of stable ISFETs for salivary nitrate to acute stress monitoring. In Proceedings of the 17th International Meeting on Chemical Sensors, Vienna, Austria, 15–19 July 2018. [Google Scholar] [CrossRef]
- Wakida, S. Stable ISFET sensors for stress monitoring. In Proceedings of the Advanced Environmental, Chemical, and Biological Sensing Technologies XV, Baltimore, MD, USA, 14–19 April 2019. [Google Scholar]
- Wakida, S.I.; Okumura, T.; Shibutani, Y.; Liu, H. Highly sensitive nitrate-sensing materials for ion-selective field-effect transistors for single-drop rain analysis. Sens. Mater. 2007, 19, 235–247. [Google Scholar]
- Tanaka, Y.; Naruishi, N.; Fukuya, H.; Sakata, J.; Saito, K.; Wakida, S. Simultaneous determination of nitrite, nitrate, thiocyanate and uric acid in human saliva by capillary zone electrophoresis and its application to the study of daily variations. J. Chromatogr. A 2004, 1051, 193–197. [Google Scholar] [CrossRef]

| Polymer Matrix | |||||
|---|---|---|---|---|---|
| I | SCN | NO2 | Cl | SO4 | |
| PVC | 0.82 | 0.95 | −1.53 | −2.55 | −4.10 |
| KP-13 | 0.56 | 0.95 | −1.53 | −2.59 | −4.03 |
| Pellethane® | 0.78 | 1.09 | −1.50 | −2.71 | −4.11 |
| P7281-PU | 0.69 | 1.07 | −1.40 | −2.30 | −4.13 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Osaki, S.; Kintoki, T.; Moriuchi-Kawakami, T.; Kitamura, K.; Wakida, S.-i. Investigation of Polyurethane Matrix Membranes for Salivary Nitrate ISFETs to Prevent the Drift. Sensors 2019, 19, 2713. https://doi.org/10.3390/s19122713
Osaki S, Kintoki T, Moriuchi-Kawakami T, Kitamura K, Wakida S-i. Investigation of Polyurethane Matrix Membranes for Salivary Nitrate ISFETs to Prevent the Drift. Sensors. 2019; 19(12):2713. https://doi.org/10.3390/s19122713
Chicago/Turabian StyleOsaki, Shuto, Takuya Kintoki, Takayo Moriuchi-Kawakami, Kenichi Kitamura, and Shin-ichi Wakida. 2019. "Investigation of Polyurethane Matrix Membranes for Salivary Nitrate ISFETs to Prevent the Drift" Sensors 19, no. 12: 2713. https://doi.org/10.3390/s19122713
APA StyleOsaki, S., Kintoki, T., Moriuchi-Kawakami, T., Kitamura, K., & Wakida, S.-i. (2019). Investigation of Polyurethane Matrix Membranes for Salivary Nitrate ISFETs to Prevent the Drift. Sensors, 19(12), 2713. https://doi.org/10.3390/s19122713





