Sensitivity Enhancement of a Surface Plasmon Resonance with Tin Selenide (SnSe) Allotropes
Abstract
:1. Introduction
2. Calculation Models and Methods
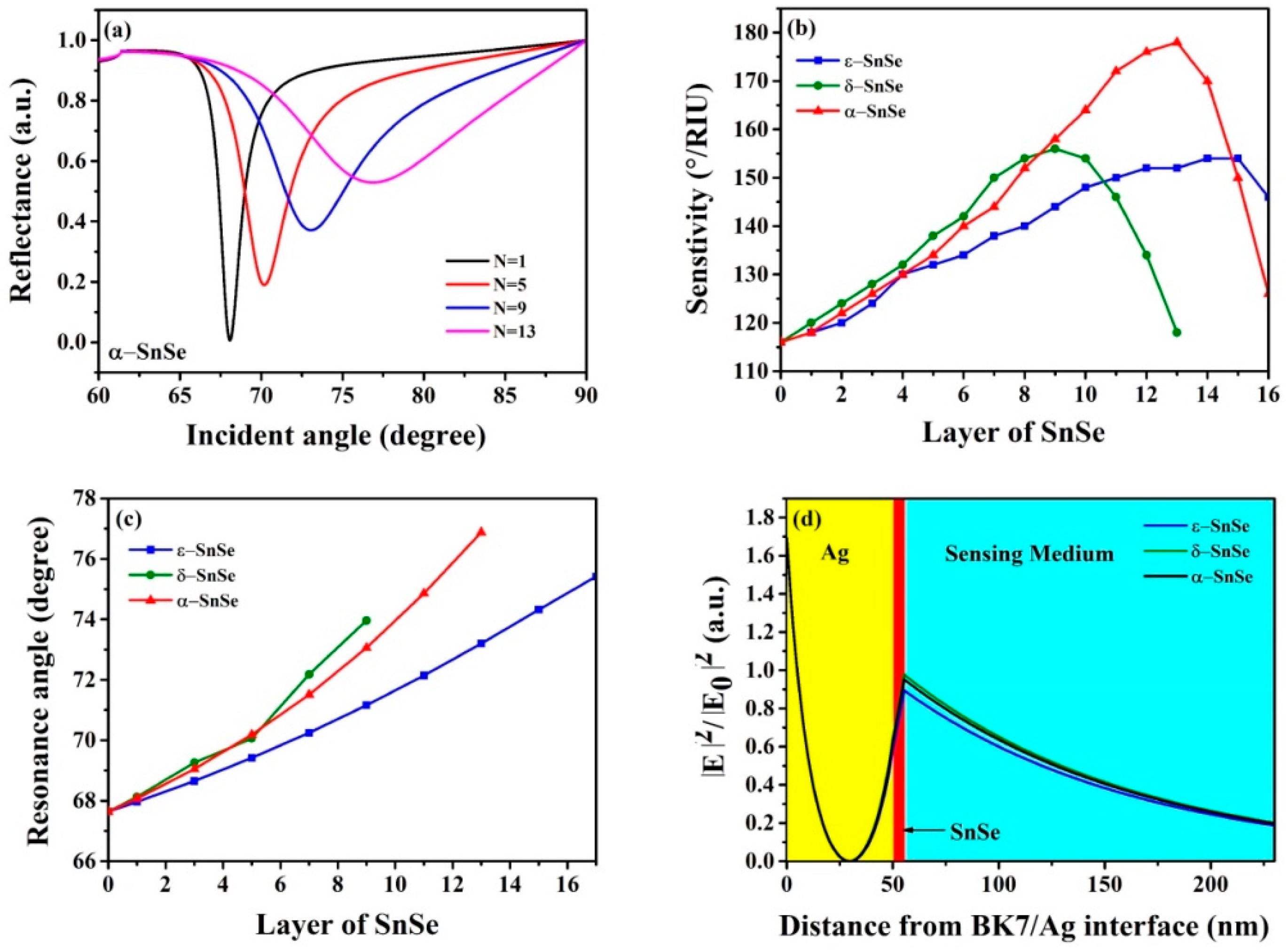
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Liedberg, B.; Nylander, C.; Lunström, I. Surface plasmon resonance for gas detection and biosensing. Sens. Actuators 1983, 4, 299–304. [Google Scholar] [CrossRef]
- Zeng, S.; Baillargeat, D.; Ho, H.-P.; Yong, K.-T. Nanomaterials enhanced surface plasmon resonance for biological and chemical sensing applications. Chem. Soc. Rev. 2014, 43, 3426–3452. [Google Scholar] [CrossRef] [PubMed]
- Shalabney, A.; Abdulhalim, I. Sensitivity-enhancement methods for surface plasmon sensors. Laser Photonics Rev. 2011, 5, 571–606. [Google Scholar] [CrossRef]
- Huang, Y.; Ho, H.P.; Kong, S.K.; Kabashin, A.V. Phase-sensitive surface plasmon resonance biosensors: Methodology, instrumentation and applications. Annalen Der Physik 2012, 524, 637–662. [Google Scholar] [CrossRef]
- Ouyang, Q.; Zeng, S.; Jiang, L.; Qu, J.; Dinh, X.-Q.; Qian, J.; He, S.; Coquet, P.; Yong, K.-T. Two-dimensional transition metal dichalcogenide enhanced phase-sensitive plasmonic biosensors: Theoretical insight. J. Phys. Chem. C 2017, 121, 6282–6289. [Google Scholar] [CrossRef]
- Srivastava, S.K.; Verma, R.; Gupta, B.D. Theoretical modeling of a self-referenced dual mode SPR sensor utilizing indium tin oxide film. Opt. Commun. 2016, 369, 131–137. [Google Scholar] [CrossRef]
- Wang, Q.H.; Kalantar-Zadeh, K.; Kis, A.; Coleman, J.N.; Strano, M.S. Electronics and optoelectronics of two-dimensional transition metal dichalcogenides. Nat. Nanotechnol. 2012, 7, 699. [Google Scholar] [CrossRef]
- Huang, X.; Zeng, Z.; Zhang, H. Metal dichalcogenide nanosheets: Preparation, properties and applications. Chem. Soc. Rev. 2013, 42, 1934–1946. [Google Scholar] [CrossRef]
- Lu, X.; Utama, M.I.B.; Lin, J.; Gong, X.; Zhang, J.; Zhao, Y.; Pantelides, S.T.; Wang, J.; Dong, Z.; Liu, Z. Large-area synthesis of monolayer and few-layer MoSe2 films on SiO2 substrates. Nano Lett. 2014, 14, 2419–2425. [Google Scholar] [CrossRef]
- Ouyang, Q.; Zeng, S.; Jiang, L.; Hong, L.; Xu, G.; Dinh, X.-Q.; Qian, J.; He, S.; Qu, J.; Coquet, P. Sensitivity enhancement of transition metal dichalcogenides/silicon nanostructure-based surface plasmon resonance biosensor. Sci. Rep. 2016, 6, 28190. [Google Scholar] [CrossRef]
- Mishra, A.K.; Mishra, S.K.; Verma, R.K. Graphene and beyond graphene MoS2: A new window in surface-plasmon-resonance-based fiber optic sensing. J. Phys. Chem. C 2016, 120, 2893–2900. [Google Scholar] [CrossRef]
- Wei, P.-C.; Bhattacharya, S.; He, J.; Neeleshwar, S.; Podila, R.; Chen, Y.; Rao, A. The intrinsic thermal conductivity of SnSe. Nature 2016, 539, E1. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.-D.; Tan, G.; Hao, S.; He, J.; Pei, Y.; Chi, H.; Wang, H.; Gong, S.; Xu, H.; Dravid, V.P. Ultrahigh power factor and thermoelectric performance in hole-doped single-crystal SnSe. Science 2016, 351, 141–144. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.-D.; Lo, S.-H.; Zhang, Y.; Sun, H.; Tan, G.; Uher, C.; Wolverton, C.; Dravid, V.P.; Kanatzidis, M.G. Ultralow thermal conductivity and high thermoelectric figure of merit in SnSe crystals. Nature 2014, 508, 373. [Google Scholar] [CrossRef] [PubMed]
- Tan, G.; Zhao, L.-D.; Kanatzidis, M.G. Rationally designing high-performance bulk thermoelectric materials. Chem. Rev. 2016, 116, 12123–12149. [Google Scholar] [CrossRef] [PubMed]
- Hu, Z.-Y.; Li, K.-Y.; Lu, Y.; Huang, Y.; Shao, X.-H. High thermoelectric performances of monolayer SnSe allotropes. Nanoscale 2017, 9, 16093–16100. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Z.; Wang, Z.; Gillespie, D.E.; Lausted, C.; Zheng, Z.; Yang, M.; Zhu, J. Plain Silver Surface Plasmon Resonance for Microarray Application. Anal. Chem. 2015, 87, 1466–1469. [Google Scholar] [CrossRef]
- Wang, Z.; Cheng, Z.; Singh, V.; Zheng, Z.; Wang, Y.; Li, S.; Song, L.; Zhu, J. Stable and Sensitive Silver Surface Plasmon Resonance Imaging Sensor Using Trilayered Metallic Structures. Anal. Chem. 2014, 86, 1430–1436. [Google Scholar] [CrossRef]
- Zhao, S.; Wang, H.; Zhou, Y.; Liao, L.; Jiang, Y.; Yang, X.; Chen, G.; Lin, M.; Wang, Y.; Peng, H. Controlled synthesis of single-crystal SnSe nanoplates. Nano Res. 2015, 8, 288–295. [Google Scholar] [CrossRef]
- Zeng, S.; Hu, S.; Xia, J.; Anderson, T.; Dinh, X.-Q.; Meng, X.-M.; Coquet, P.; Yong, K.-T. Graphene–MoS2 hybrid nanostructures enhanced surface plasmon resonance biosensors. Sens. Actuators B-Chem. 2015, 207, 801–810. [Google Scholar] [CrossRef]
- Gupta, B.; Sharma, A.K. Sensitivity evaluation of a multi-layered surface plasmon resonance-based fiber optic sensor: A theoretical study. Sens. Actuators B-Chem. 2005, 107, 40–46. [Google Scholar] [CrossRef]
- Cui, Z.; Wang, X.; Ding, Y.; Li, M. Exploration work function and optical properties of monolayer SnSe allotropes. Superlattices Microstruct. 2018, 114, 251–258. [Google Scholar] [CrossRef]
- Hansen, W.N. Electric fields produced by the propagation of plane coherent electromagnetic radiation in a stratified medium. JOSA 1968, 58, 380–390. [Google Scholar] [CrossRef]
- Maharana, P.K.; Jha, R. Chalcogenide prism and graphene multilayer based surface plasmon resonance affinity biosensor for high performance. Sens. Actuators B-Chem. 2012, 169, 161–166. [Google Scholar] [CrossRef]
- Homola, J. Surface plasmon resonance sensors for detection of chemical and biological species. Chem. Rev. 2008, 108, 462–493. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Chu, H.S.; Koh, W.S.; Li, E.P. Highly sensitive graphene biosensors based on surface plasmon resonance. Opt. Express 2010, 18, 14395–14400. [Google Scholar] [CrossRef] [PubMed]
- Homola, J. Present and future of surface plasmon resonance biosensors. Anal. Bioanal. Chem. 2003, 377, 528–539. [Google Scholar] [CrossRef]
- Piliarik, M.; Homola, J. Surface plasmon resonance (SPR) sensors: Approaching their limits? Opt. Express 2009, 17, 16505–16517. [Google Scholar] [CrossRef]
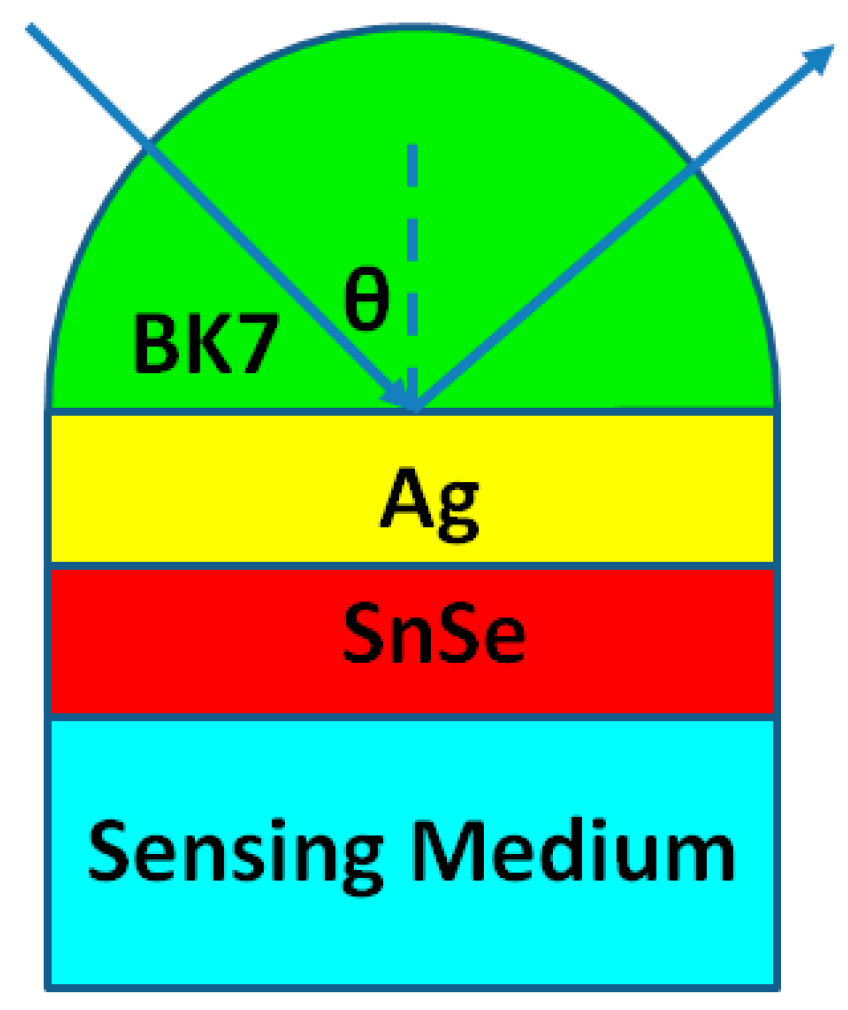
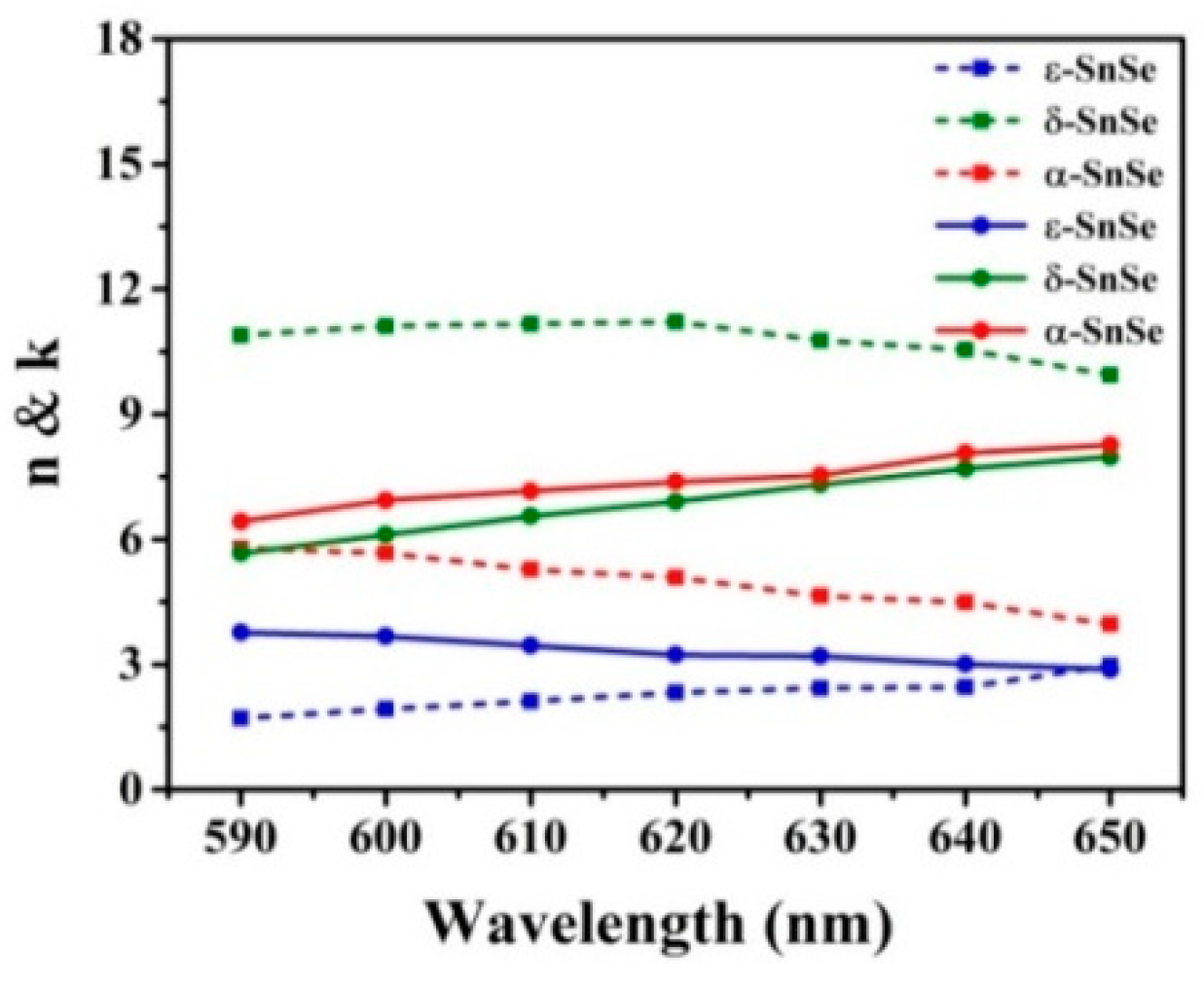
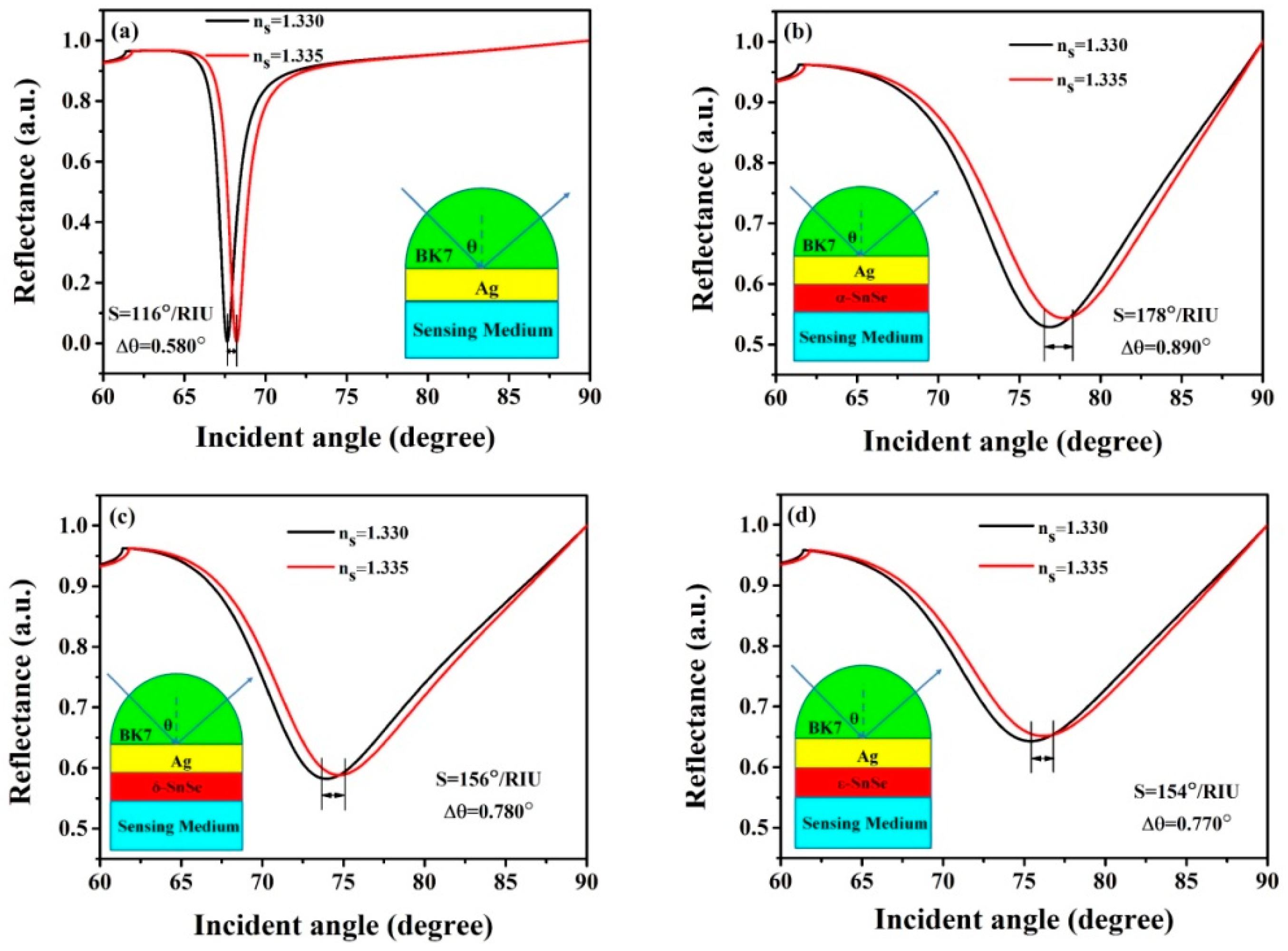
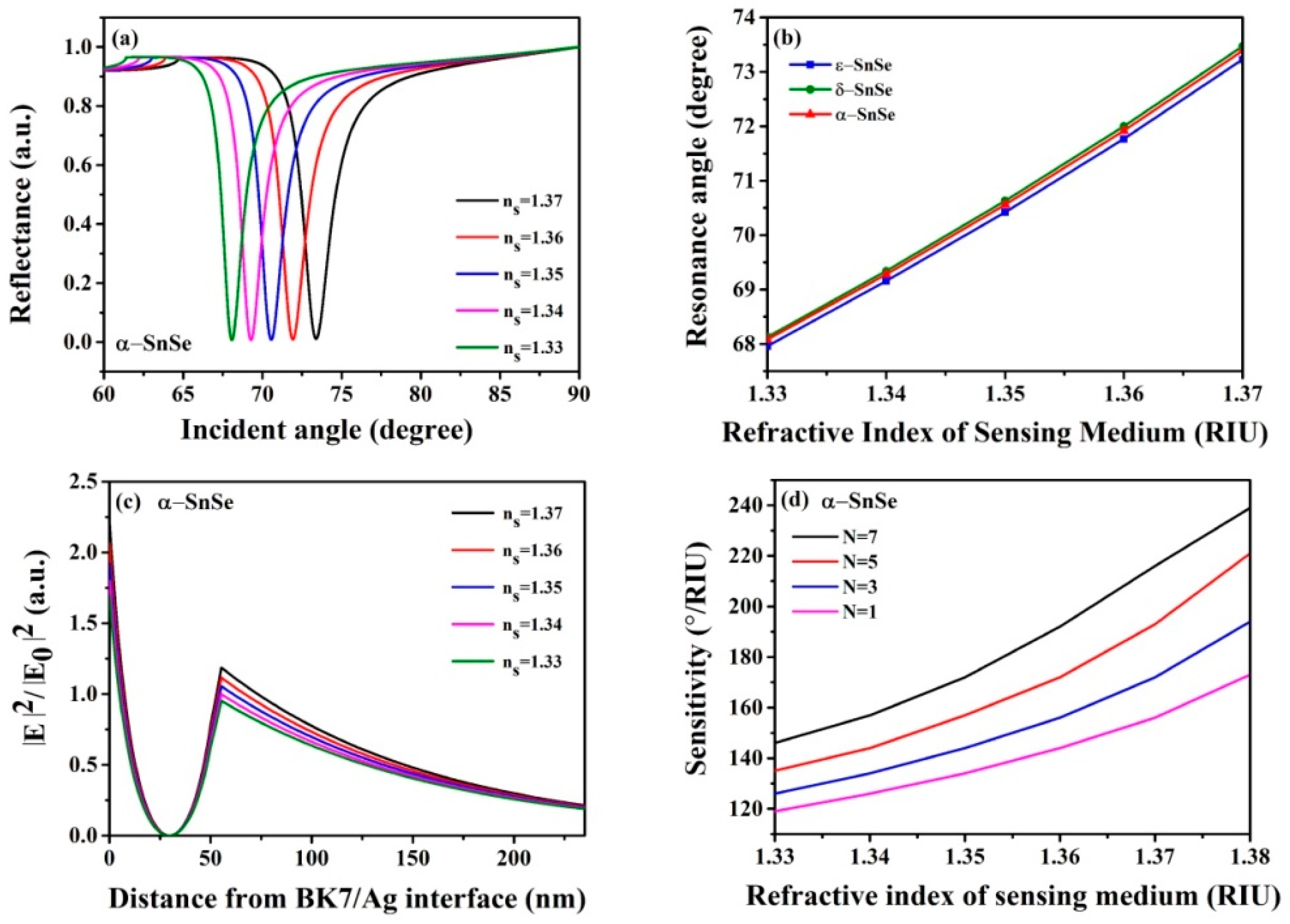
| SnSe Allotropes | dAg (nm) | L | S (°/RIU) | θmin (Degree) | Δθ (Degree) | Rmin (a. u.) |
|---|---|---|---|---|---|---|
| without SnSe | 52 | 0 | 116 | 67.64 | 0.58 | 0.02690 |
| α-SnSe | 52 | 1 | 118 | 68.08 | 0.59 | 0.00003 |
| 52 | 8 | 152 | 72.27 | 0.76 | 0.3954 | |
| 52 | 13 | 178 | 76.91 | 0.89 | 0.5854 | |
| δ-SnSe | 52 | 17 | 118 | 68.14 | 0.59 | 0.01282 |
| 52 | 5 | 138 | 70.60 | 0.69 | 0.4446 | |
| 52 | 9 | 156 | 73.98 | 0.78 | 0.6327 | |
| ε-SnSe | 52 | 13 | 118 | 67.96 | 0.59 | 0.00100 |
| 52 | 8 | 138 | 70.70 | 0.69 | 0.4513 | |
| 52 | 15 | 156 | 74.33 | 0.78 | 0.6476 | |
| without SnSe | 50 | 0 | 116 | 67.64 | 0.58 | 0.00590 |
| α-SnSe | 50 | 1 | 118 | 68.08 | 0.59 | 0.00680 |
| 50 | 8 | 152 | 72.25 | 0.76 | 0.3295 | |
| 50 | 13 | 178 | 76.86 | 0.89 | 0.5288 | |
| δ-SnSe | 50 | 17 | 120 | 68.13 | 0.60 | 0.03950 |
| 50 | 5 | 138 | 70.59 | 0.69 | 0.3805 | |
| 50 | 9 | 156 | 73.95 | 0.78 | 0.5821 | |
| ε-SnSe | 50 | 13 | 118 | 67.96 | 0.59 | 0.01420 |
| 50 | 8 | 138 | 70.69 | 0.69 | 0.3875 | |
| 50 | 15 | 154 | 74.31 | 0.77 | 0.5987 | |
| without SnSe | 48 | 0 | 116 | 67.64 | 0.58 | 0.02691 |
| α-SnSe | 48 | 1 | 118 | 68.08 | 0.59 | 0.00003 |
| 48 | 8 | 150 | 72.23 | 0.75 | 0.2632 | |
| 48 | 13 | 176 | 76.80 | 0.88 | 0.4673 | |
| δ-SnSe | 48 | 17 | 118 | 68.14 | 0.59 | 0.01281 |
| 48 | 5 | 138 | 70.58 | 0.69 | 0.3146 | |
| 48 | 9 | 154 | 73.92 | 0.77 | 0.5264 | |
| ε-SnSe | 48 | 13 | 118 | 67.96 | 0.59 | 0.00103 |
| 48 | 8 | 136 | 70.68 | 0.68 | 0.3218 | |
| 48 | 15 | 156 | 74.27 | 0.78 | 0.5447 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dai, X.; Liang, Y.; Zhao, Y.; Gan, S.; Jia, Y.; Xiang, Y. Sensitivity Enhancement of a Surface Plasmon Resonance with Tin Selenide (SnSe) Allotropes. Sensors 2019, 19, 173. https://doi.org/10.3390/s19010173
Dai X, Liang Y, Zhao Y, Gan S, Jia Y, Xiang Y. Sensitivity Enhancement of a Surface Plasmon Resonance with Tin Selenide (SnSe) Allotropes. Sensors. 2019; 19(1):173. https://doi.org/10.3390/s19010173
Chicago/Turabian StyleDai, Xiaoyu, Yanzhao Liang, Yuting Zhao, Shuaiwen Gan, Yue Jia, and Yuanjiang Xiang. 2019. "Sensitivity Enhancement of a Surface Plasmon Resonance with Tin Selenide (SnSe) Allotropes" Sensors 19, no. 1: 173. https://doi.org/10.3390/s19010173
APA StyleDai, X., Liang, Y., Zhao, Y., Gan, S., Jia, Y., & Xiang, Y. (2019). Sensitivity Enhancement of a Surface Plasmon Resonance with Tin Selenide (SnSe) Allotropes. Sensors, 19(1), 173. https://doi.org/10.3390/s19010173






