The Effect of Pain Relief on Daily Physical Activity: In-Home Objective Physical Activity Assessment in Chronic Low Back Pain Patients after Paravertebral Spinal Block
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Paravertebral Facet Injection
2.3. Patient-Reported Outcome Measures (PROMs)
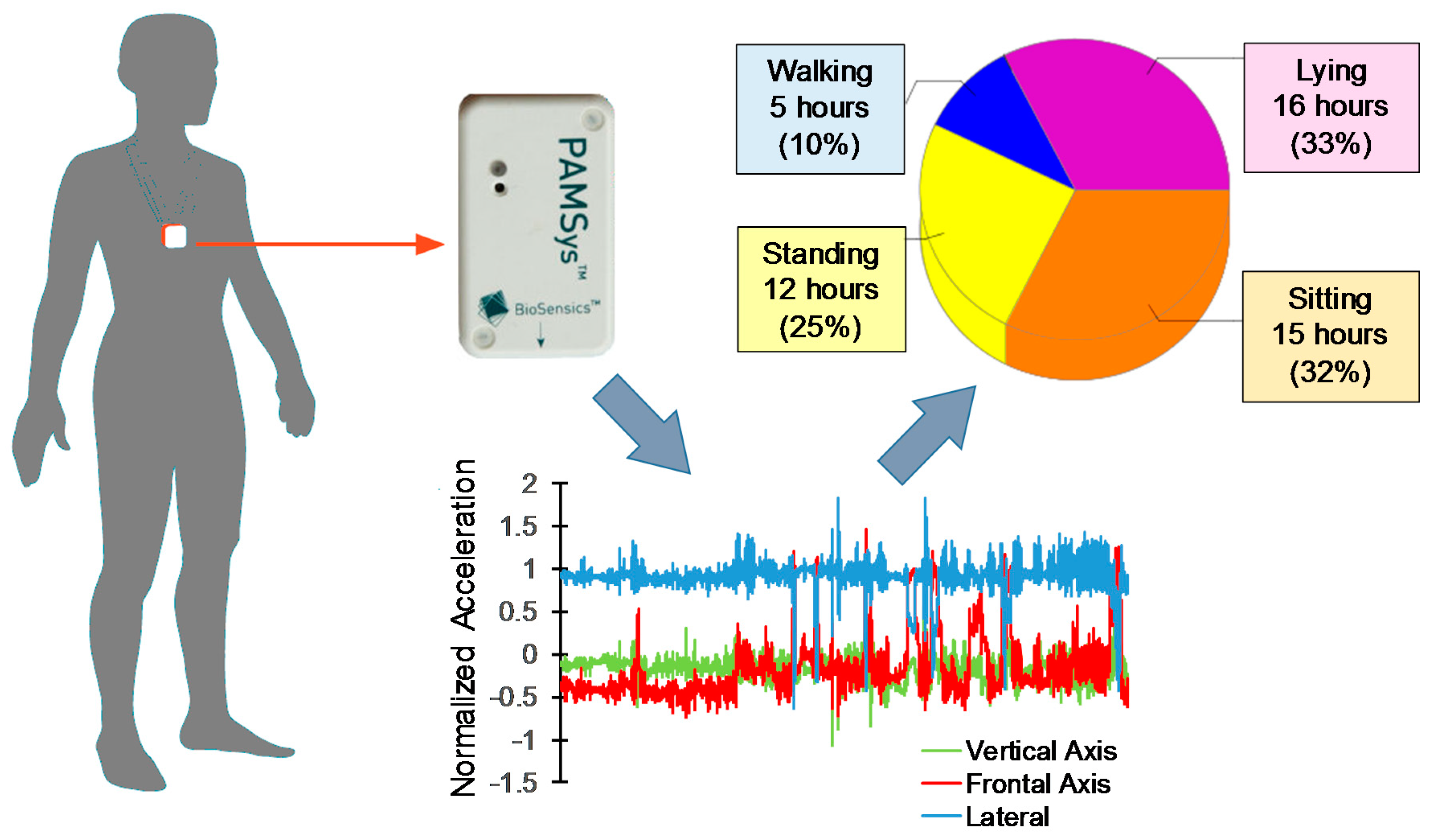
2.4. Objective Motor Performance Measurements
2.5. Data Analysis
3. Results
3.1. Participants
3.2. Changes in PROMs Following Spinal Injection
3.3. Changes in DPA (PAMSys) Data
3.4. Changes in Motor Performance Following Spinal Injection
4. Discussion
4.1. Differences between Subjective and Objective In-Clinic and In-Home LBP Improvements
4.2. Limitations and Future Directions
4.3. Clinical Implications
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Suri, P.; Miyakoshi, A.; Hunter, D.J.; Jarvik, J.G.; Rainville, J.; Guermazi, A.; Li, L.; Katz, J.N. Does lumbar spinal degeneration begin with the anterior structures? A study of the observed epidemiology in a community-based population. BMC Musculoskelet. Disord. 2011, 12, 202. [Google Scholar] [CrossRef] [PubMed]
- Toosizadeh, N.; Nussbaum, M.A.; Bazrgari, B.; Madigan, M.L. Load-relaxation properties of the human trunk in response to prolonged flexion: Measuring and modeling the effect of flexion angle. PLoS ONE 2012, 7, e48625. [Google Scholar] [CrossRef] [PubMed]
- Freburger, J.K.; Holmes, G.M.; Agans, R.P.; Jackman, A.M.; Darter, J.D.; Wallace, A.S.; Castel, L.D.; Kalsbeek, W.D.; Carey, T.S. The rising prevalence of chronic low back pain. Arch. Internal Med. 2009, 169, 251–258. [Google Scholar] [CrossRef] [PubMed]
- Chou, R.; Qaseem, A.; Snow, V.; Casey, D.; Cross, J.T.; Shekelle, P.; Owens, D.K. Diagnosis and treatment of low back pain: A joint clinical practice guideline from the American College of Physicians and the American Pain Society. Ann. Internal Med. 2007, 147, 478–491. [Google Scholar] [CrossRef]
- Koes, B.; Van Tulder, M.; Thomas, S. Diagnosis and treatment of low back pain. Br. Med. J. 2006, 332, 1430–1434. [Google Scholar] [CrossRef] [PubMed]
- Kalichman, L.; Hunter, D.J. Lumbar Facet Joint Osteoarthritis: A Review. Semin. Arthritis Rheum. 2007, 37, 69–80. [Google Scholar] [CrossRef] [PubMed]
- Manchikanti, L.; Buenaventura, R.M.; Manchikanti, K.N.; Ruan, X.; Gupta, S.; Smith, H.S.; Christo, P.J.; Ward, S.P. Effectiveness of therapeutic lumbar transforaminal epidural steroid injections in managing lumbar spinal pain. Pain Phys. 2012, 15, E199–E245. [Google Scholar]
- Weinstein, S.M.; Herring, S.A. Lumbar epidural steroid injections. Spine J. 2003, 3, 37–44. [Google Scholar] [CrossRef]
- Delport, E.G.; Cucuzzella, A.R.; Marley, J.K.; Pruitt, C.M.; Fisher, J.R. Treatment of lumbar spinal stenosis with epidural steroid injections: A retrospective outcome study. Arch. Phys. Med. Rehabil. 2004, 85, 479–484. [Google Scholar] [CrossRef]
- Ackerman, W.E., III; Ahmad, M. The efficacy of lumbar epidural steroid injections in patients with lumbar disc herniations. Anesth. Analg. 2007, 104, 1217–1222. [Google Scholar] [CrossRef] [PubMed]
- Friedly, J.L.; Comstock, B.A.; Turner, J.A.; Heagerty, P.J.; Deyo, R.A.; Sullivan, S.D.; Bauer, Z.; Bresnahan, B.W.; Avins, A.L.; Nedeljkovic, S.S. A randomized trial of epidural glucocorticoid injections for spinal stenosis. N. Engl. J. Med. 2014, 371, 11–21. [Google Scholar] [CrossRef] [PubMed]
- Tomkins-Lane, C.C.; Conway, J.; Hepler, C.; Haig, A.J. Changes in objectively measured physical activity (performance) after epidural steroid injection for lumbar spinal stenosis. Arch. Phys. Med. Rehabil. 2012, 93, 2008–2014. [Google Scholar] [CrossRef] [PubMed]
- Toosizadeh, N.; Harati, H.; Yen, T.-C.; Fastje, C.; Mohler, J.; Najafi, B.; Dohm, M. Paravertebral spinal injection for the treatment of patients with degenerative facet osteoarthropathy: Evidence of motor performance improvements based on objective assessments. Clin. Biomech. 2016, 39, 100–108. [Google Scholar] [CrossRef] [PubMed]
- General Assembly of the World Medical Association. World Medical Association Declaration of Helsinki: Ethical principles for medical research involving human subjects. J. Am. Coll. Dent. 2014, 81, 14. [Google Scholar]
- Manchikanti, L.; Boswell, M.V.; Singh, V.; Benyamin, R.M.; Fellows, B.; Abdi, S.; Buenaventura, R.M.; Conn, A.; Datta, S.; Derby, R.; et al. Comprehensive evidence-based guidelines for interventional techniques in the management of chronic spinal pain. Pain Phys. 2009, 12, 699–802. [Google Scholar]
- Langley, G.; Sheppeard, H. The visual analogue scale: Its use in pain measurement. Rheumatol. Int. 1985, 5, 145–148. [Google Scholar] [CrossRef] [PubMed]
- Fairbank, J.C.; Pynsent, P.B. The Oswestry disability index. Spine 2000, 25, 2940–2953. [Google Scholar] [CrossRef] [PubMed]
- Kempen, G.I.; Yardley, L.; Van Haastregt, J.C.; Zijlstra, G.R.; Beyer, N.; Hauer, K.; Todd, C. The Short FES-I: A shortened version of the falls efficacy scale-international to assess fear of falling. Age Ageing 2007, 37, 45–50. [Google Scholar] [CrossRef] [PubMed]
- Ware Jr, J.E.; Kosinski, M.; Keller, S.D. A 12-Item Short-Form Health Survey: Construction of scales and preliminary tests of reliability and validity. Med. Care 1996, 34, 220–233. [Google Scholar] [CrossRef]
- Aminian, K.; Najafi, B. Capturing human motion using body—Fixed sensors: Outdoor measurement and clinical applications. Comput. Anim. Virtual Worlds 2004, 15, 79–94. [Google Scholar] [CrossRef]
- Mohler, M.J.; Wendel, C.S.; Taylor-Piliae, R.E.; Toosizadeh, N.; Najafi, B. Motor performance and physical activity as predictors of prospective falls in community-dwelling older adults by frailty level: Application of wearable technology. Gerontology 2016, 62, 654–664. [Google Scholar] [CrossRef] [PubMed]
- Najafi, B.; Armstrong, D.G.; Mohler, J. Novel Wearable Technology for Assessing Spontaneous Daily Physical Activity and Risk of Falling in Older Adults with Diabetes; SAGE Publications: Los Angeles, CA, USA, 2013. [Google Scholar]
- Najafi, B.; Crews, R.T.; Wrobel, J.S. Importance of time spent standing for those at risk of diabetic foot ulceration. Diabetes Care 2010, 33, 2448–2450. [Google Scholar] [CrossRef] [PubMed]
- Paraschiv-Ionescu, A.; Buchser, E.; Rutschmann, B.; Najafi, B.; Aminian, K. Ambulatory system for the quantitative and qualitative analysis of gait and posture in chronic pain patients treated with spinal cord stimulation. Gait Posture 2004, 20, 113–125. [Google Scholar] [CrossRef] [PubMed]
- Toosizadeh, N.; Mohler, J.; Lei, H.; Parvaneh, S.; Sherman, S.; Najafi, B. Motor performance assessment in Parkinson’s disease: Association between objective in-clinic, objective in-home, and subjective/semi-objective measures. PLoS ONE 2015, 10, e0124763. [Google Scholar] [CrossRef] [PubMed]
- De Bruin, E.D.; Najafi, B.; Murer, K.; Uebelhart, D.; Aminian, K. Quantification of everyday motor function in a geriatric population. J. Rehabil. Res. Dev. 2007, 44, 417–428. [Google Scholar] [CrossRef] [PubMed]
- Aminian, K.; Najafi, B.; Büla, C.; Leyvraz, P.-F.; Robert, P. Spatio-temporal parameters of gait measured by an ambulatory system using miniature gyroscopes. J. Biomech. 2002, 35, 689–699. [Google Scholar] [CrossRef]
- Thiede, R.; Toosizadeh, N.; Mills, J.L.; Zaky, M.; Mohler, J.; Najafi, B. Gait and balance assessments as early indicators of frailty in patients with known peripheral artery disease. Clin. Biomech. 2016, 32, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Zampieri, C.; Salarian, A.; Carlson-Kuhta, P.; Aminian, K.; Nutt, J.G.; Horak, F.B. The instrumented timed up and go test: Potential outcome measure for disease modifying therapies in Parkinson’s disease. J. Neurol. Neurosurg. Psychiatry 2010, 81, 171–176. [Google Scholar] [CrossRef] [PubMed]
- Niv, D.; Kreitler, S. Pain and quality of life. Pain Pract. 2001, 1, 150–161. [Google Scholar] [CrossRef] [PubMed]
- Kovacs, F.M.; Abraira, V.; Zamora, J.; del Real, M.T.G.; Llobera, J.; Fernández, C. Correlation between pain, disability, and quality of life in patients with common low back pain. Spine 2004, 29, 206–210. [Google Scholar] [CrossRef] [PubMed]
- Keefe, F.J.; Hill, R.W. An objective approach to quantifying pain behavior and gait patterns in low back pain patients. Pain 1985, 21, 153–161. [Google Scholar] [CrossRef]
- Toosizadeh, N.; Yen, T.C.; Howe, C.; Dohm, M.; Mohler, J.; Najafi, B. Gait behaviors as an objective surgical outcome in low back disorders: A systematic review. Clin. Biomech. 2015, 30, 528–536. [Google Scholar] [CrossRef] [PubMed]
- Yen, T.C.; Toosizadeh, N.; Howe, C.; Dohm, M.; Mohler, J.; Najafi, B. Postural balance parameters as objective surgical assessments in low back disorders: A systematic review. J. Appl. Biomech. 2016, 32, 316–323. [Google Scholar] [CrossRef] [PubMed]
- Vlaeyen, J.W.; Linton, S.J. Fear-avoidance and its consequences in chronic musculoskeletal pain: A state of the art. Pain 2000, 85, 317–332. [Google Scholar] [CrossRef]
- Waddell, G.; Newton, M.; Henderson, I.; Somerville, D.; Main, C.J. A Fear-Avoidance Beliefs Questionnaire (FABQ) and the role of fear-avoidance beliefs in chronic low back pain and disability. Pain 1993, 52, 157–168. [Google Scholar] [CrossRef]
| Baseline | Immediate FU | One-Month FU | p-Value | CI (Upper) | CI (Lower) | Effect Size | |
|---|---|---|---|---|---|---|---|
| Pain at the moment, 0–10 (SD) | 5.63 (3.42) | 2.13 (2.01) | 2.92 (2.65) | <0.05 * | −0.42 | −4.74 | 0.81 |
| Pain in two weeks, 0–10 (SD) | 6.75 (2.76) | - | 3.50 (2.72) | 0.05 * | 0.07 | −5.73 | 1.01 |
| Oswestry, percentage (SD) | 41.8 (15.5) | - | 33.0 (17.6) | <0.05 * | −0.029 | −0.13 | 0.47 |
| SF-12, PCS (SD) | 25.2 (4.7) | - | 32.2 (13.0) | 0.08 | 15.03 | −1.30 | 0.65 |
| SF-12, MCS (SD) | 40.2 (17.1) | - | 49.8 (6.4) | 0.07 | 17.31 | −0.85 | 0.89 |
| Short FES-I, 7–28 (SD) | 18.3 (6.1) | - | 15.2 (3.9) | 0.29 | 2.96 | −7.96 | 0.43 |
| Postural Summary | Baseline | Immediate FU | One-Month FU | p-Value (t-Test) | p-Value (ANOVA) | Effect Size (ANOVA) |
|---|---|---|---|---|---|---|
| Sitting, min (SD) | 866.09 (421.10) | 974.66 (606.42) | 970.98 (462.76) | 0.46 | 0.54 | 0.10 |
| Standing, min (SD) | 465.43 (96.66) | 404.60 (156.06) | 595.07 (269.72) | 0.33 | 0.25 | 0.44 |
| Walking, min (SD) | 136.58 (66.35) | 141.44 (49.48) | 122.91 (92.87) | 0.90 | 0.79 | 0.11 |
| Maximum Walking Duration (s) | 290.38 (105.33) | 353.75 (222.35) | 251.67 (55.90) | 0.50 | 0.74 | 0.33 |
| Lying, min (SD) | 1411.27 (386.41) | 1348.46 (696.76) | 1190.72 (382.21) | 0.73 | 0.37 | 0.18 |
| % Sitting (SD) | 30.08 (14.62) | 33.94 (21.02) | 33.72 (16.07) | 0.44 | 0.54 | 0.10 |
| % Standing (SD) | 15.16 (3.36) | 14.14 (5.60) | 20.66 (9.36) | 0.35 | 0.25 | 0.43 |
| % Walking (SD) | 4.74 (2.31) | 4.94 (1.76) | 4.27 (3.22) | 0.88 | 0.79 | 0.11 |
| % Lying (SD) | 49.01 (13.41) | 46.99 (24.17) | 41.35 (13.28) | 0.74 | 0.37 | 0.18 |
| Walking Characterizing | ||||||
| Total Steps (SD) | 7060.50 (3650.414) | 7068.38 (2902.22) | 5429.83 (4116.34) | 0.76 | 0.43 | 0.20 |
| Walking Duration Variability (%) | 106.99 (12.78) | 114.77 (30.73) | 103.17 (10.71) | 0.58 | 0.80 | 0.26 |
| Episode Cadence Average, steps/min (SD) | 67.16 (2.96) | 67.08 (7.42) | 66.45 (4.17) | 0.98 | 0.87 | 0.06 |
| Baseline | Immediate FU | One-Month FU | p-Value | CI (Upper) | CI (Lower) | Effect Size | |
|---|---|---|---|---|---|---|---|
| Normal Gait | |||||||
| Gait speed, m/s (SD) | 0.96 (0.24) | 1.02 (0.22) | 1.19 (0.14) | <0.05 * | 0.19 | 0.03 | 0.44 |
| Stride length, m (SD) | 1.18 (0.19) | 1.20 (0.20) | 1.35 (0.14) | 0.06 | 0.15 | −0.002 | 0.39 |
| Gait cycle time, s (SD) | 1.27 (0.11) | 1.21 (0.07) | 1.15 (0.09) | <0.01 * | −0.02 | −0.11 | 0.49 |
| Speed variability, % (SD) | 3.78 (3.17) | 2.59 (1.47) | 1.18 (0.57) | 0.07 | 0.11 | −2.60 | 0.58 |
| Cadence (steps/min) | 94.98 (4.27) | 99.88 (3.00) | 104.54 (4.29) | 0.03 * | 0.38 | 5.40 | 0.48 |
| TUG Test | |||||||
| Total TUG duration, s (SD) | 14.25 (3.90) | 12.17 (2.67) | 11.27 (1.42) | <0.05 * | −20.34 | −269.68 | 0.46 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chuan Yen, T.; Mohler, J.; Dohm, M.; Laksari, K.; Najafi, B.; Toosizadeh, N. The Effect of Pain Relief on Daily Physical Activity: In-Home Objective Physical Activity Assessment in Chronic Low Back Pain Patients after Paravertebral Spinal Block. Sensors 2018, 18, 3048. https://doi.org/10.3390/s18093048
Chuan Yen T, Mohler J, Dohm M, Laksari K, Najafi B, Toosizadeh N. The Effect of Pain Relief on Daily Physical Activity: In-Home Objective Physical Activity Assessment in Chronic Low Back Pain Patients after Paravertebral Spinal Block. Sensors. 2018; 18(9):3048. https://doi.org/10.3390/s18093048
Chicago/Turabian StyleChuan Yen, Tzu, Jane Mohler, Michael Dohm, Kaveh Laksari, Bijan Najafi, and Nima Toosizadeh. 2018. "The Effect of Pain Relief on Daily Physical Activity: In-Home Objective Physical Activity Assessment in Chronic Low Back Pain Patients after Paravertebral Spinal Block" Sensors 18, no. 9: 3048. https://doi.org/10.3390/s18093048
APA StyleChuan Yen, T., Mohler, J., Dohm, M., Laksari, K., Najafi, B., & Toosizadeh, N. (2018). The Effect of Pain Relief on Daily Physical Activity: In-Home Objective Physical Activity Assessment in Chronic Low Back Pain Patients after Paravertebral Spinal Block. Sensors, 18(9), 3048. https://doi.org/10.3390/s18093048






