Characteristics and Temperature Compensation of Non-Dispersive Infrared (NDIR) Alcohol Gas Sensors According to Incident Light Intensity
Abstract
1. Introduction
2. Theoretical Background
2.1. Basic Structure and Principle of Non-Dispersive Infrared Gas Sensor
2.1.1. Infrared Source
2.1.2. Optical Waveguide
2.1.3. Infrared Detector
2.1.4. Geometric Realization of an Elliptical Cell
3. Sensor Module Fabrication and Analysis
3.1. Sensor Module Design Considerations and 3D Modelling
3.2. Signal Conditioning Circuit
3.3. Simulation Result
4. Experimental Method
5. Result & Discussion
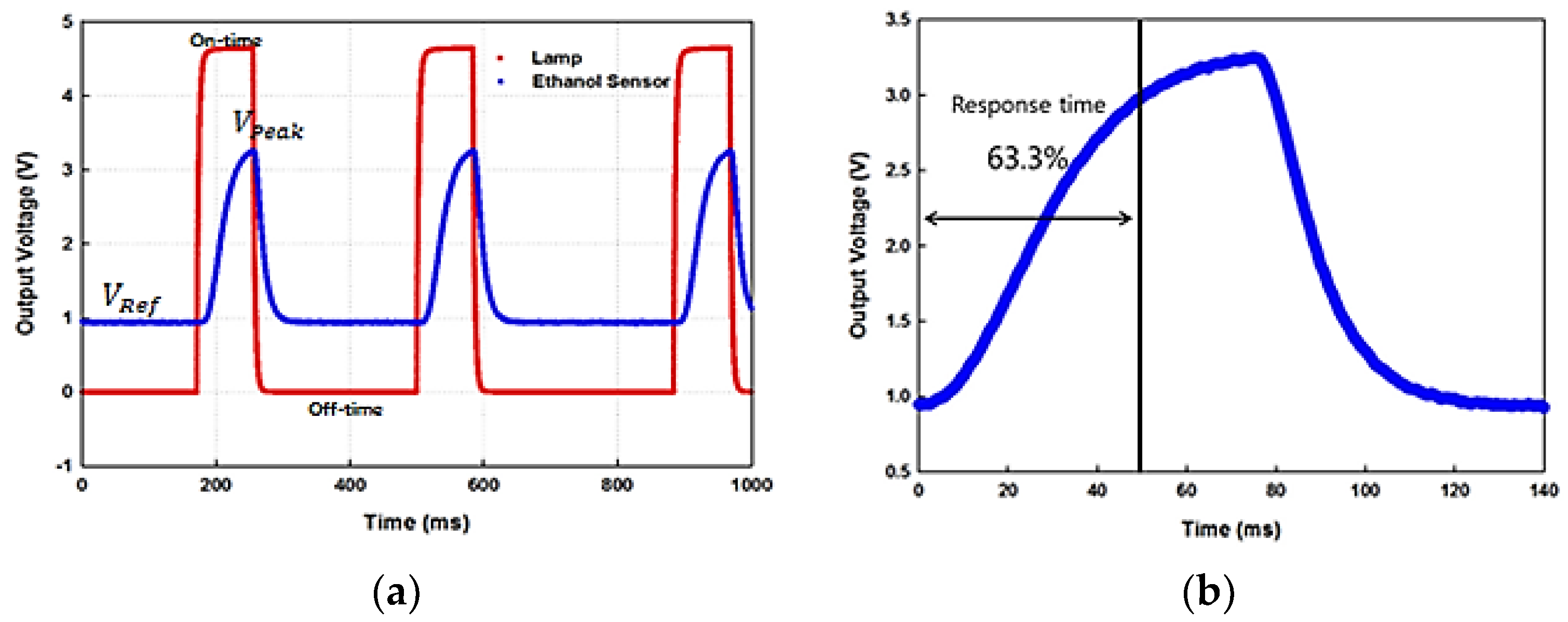
5.1. On-Off and Response Time
5.2. Output in Absence of Target Gas
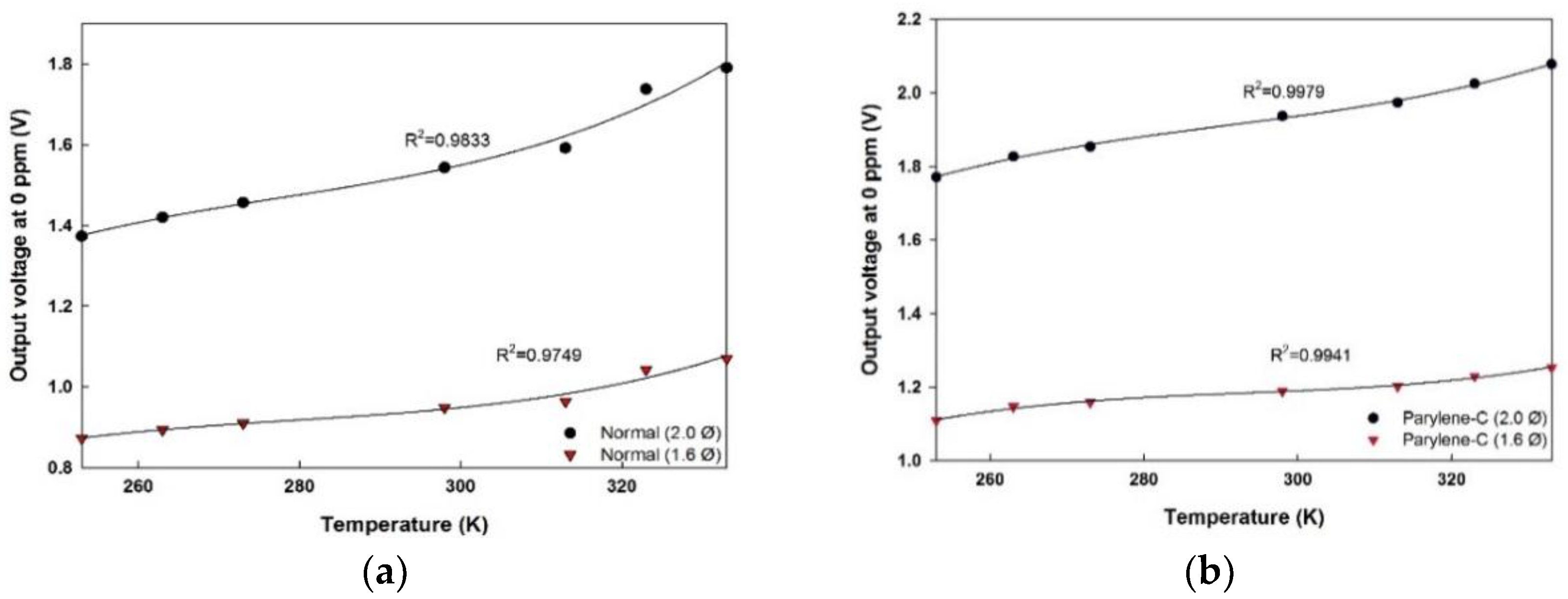
5.3. Temperature Dependency
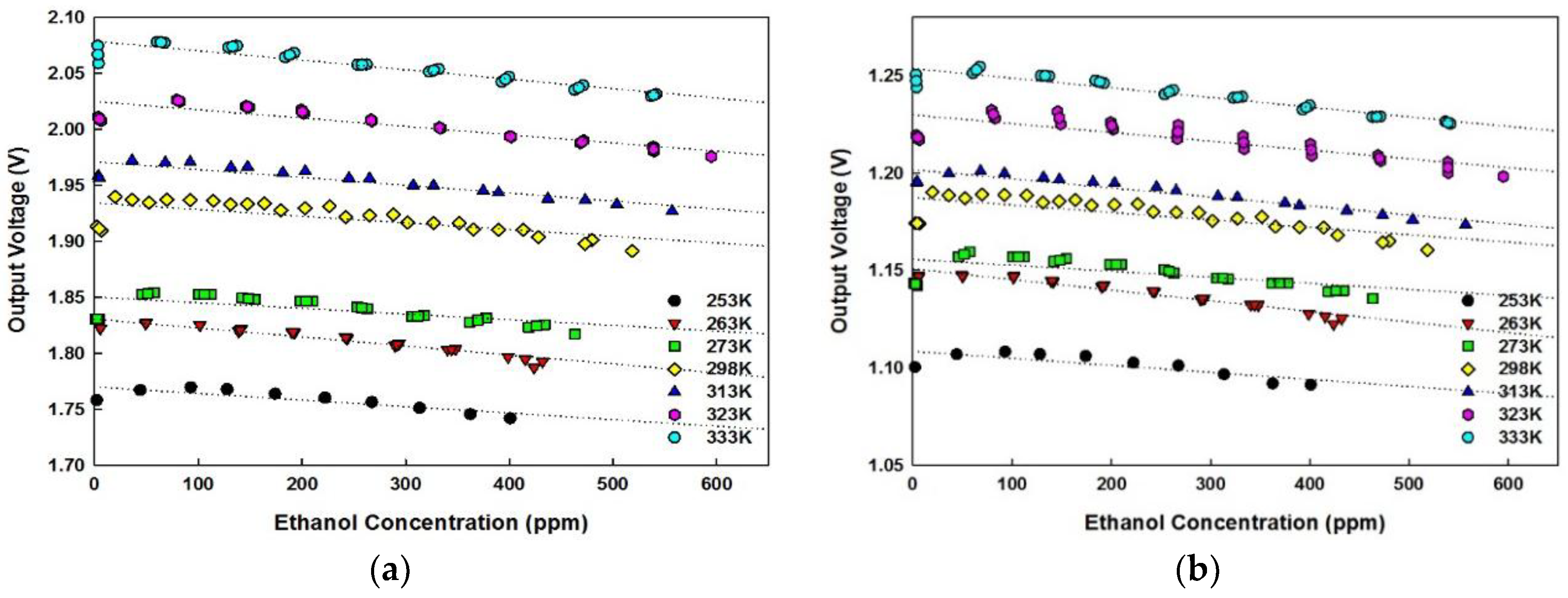
5.4. Temperature Compensation
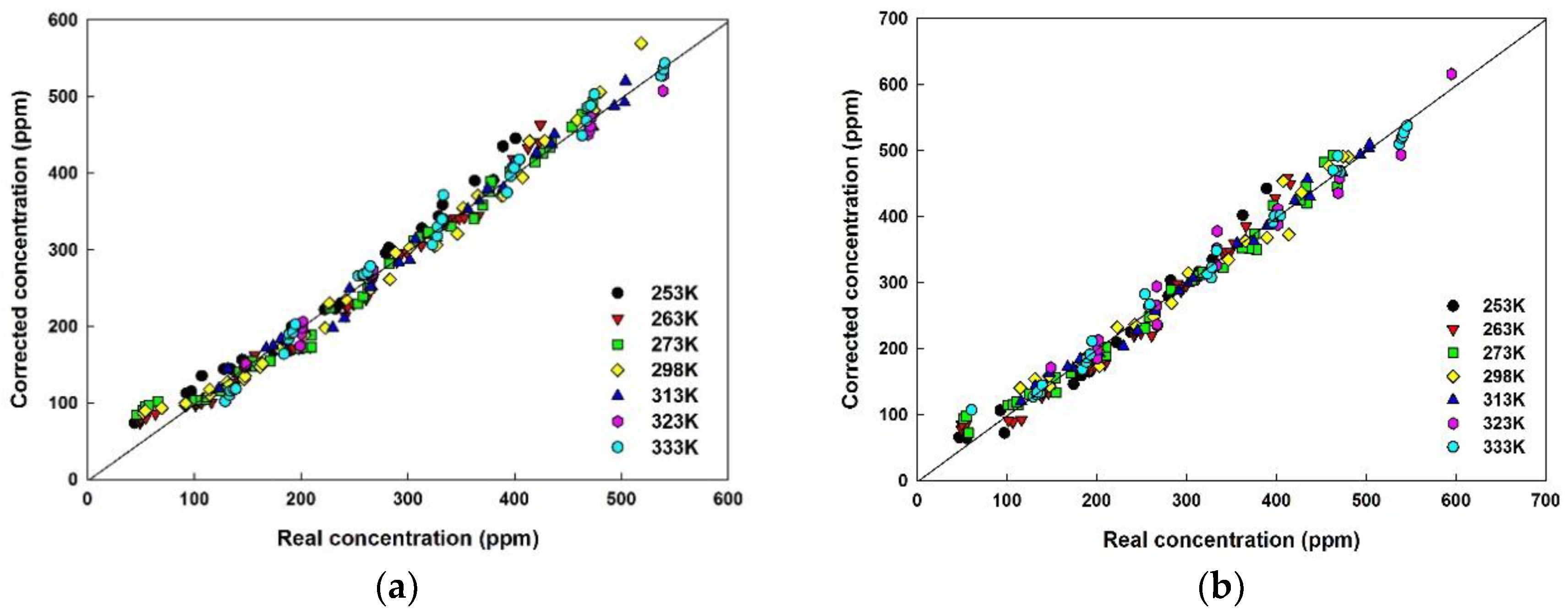
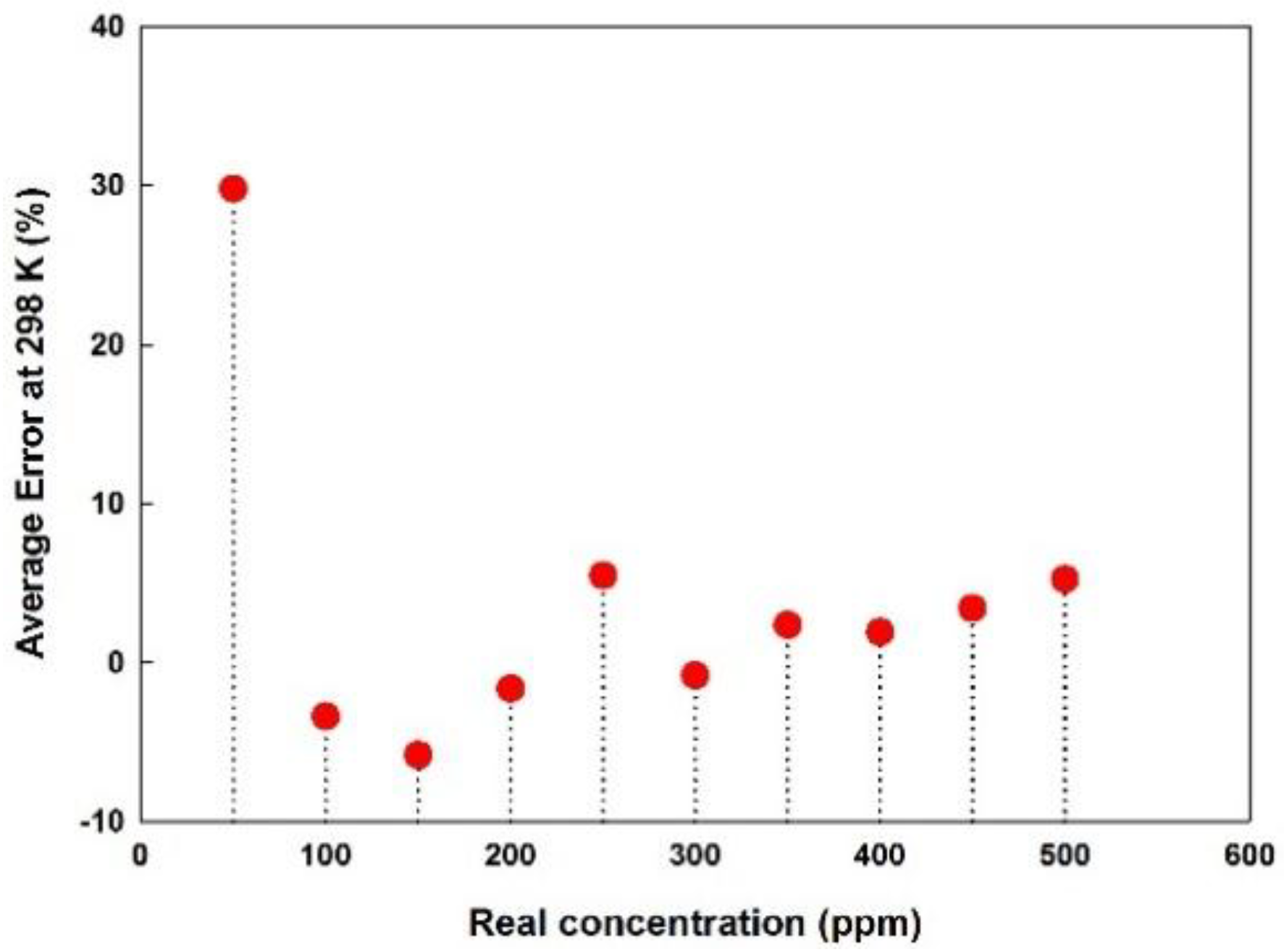
5.5. Error and Detection Limit
6. Comparison and Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Elder, R.W.; Shults, R.A.; Sleet, D.A.; Nichols, J.L.; Zaza, S.; Thompson, R.S. Effectiveness of Sobriety Checkpoints for Reducing Alcohol-Involved Crashes. Traffic Inj. Prev. 2002, 3, 266–274. [Google Scholar] [CrossRef]
- Blomberg, R.D.; Peck, R.C.; Moskowitz, H.; Burns, M.; Fiorentino, D. The Long Beach/Fort Lauderdale Relative Risk Study. J. Saf. Res. 2009, 40, 285–292. [Google Scholar] [CrossRef] [PubMed]
- Shan, C.; Yang, H.; Han, D.; Zhang, Q.; Ivaska, A.; Niu, L. Electrochemical Determination of NADH and Ethanol Based on Ionic Liquid-Functionalized Graphene. Biosens. Bioelectron. 2010, 25, 1504–1508. [Google Scholar] [CrossRef] [PubMed]
- Inyawilert, K.; Wisitsoraat, A.; Sriprachaubwong, C.; Tuantranont, A.; Phanichphant, S.; Liewhiran, C. Rapid Ethanol Sensor Based on Electrolytically-Exfoliated Graphene-Loaded Flame-Made In-Doped SnO2 Composite Film. Sens. Actuators B Chem. 2015, 209, 40–55. [Google Scholar] [CrossRef]
- Sudha, M.; Radha, S.; Kirubaveni, S.; Kiruthika, R.; Govindaraj, R.; Santhosh, N. Experimental Study on Structural, Optoelectronic and Room Temperature Sensing Performance of Nickel Doped ZnO Based Ethanol Sensors. Solid State Sci. 2018, 78, 30–39. [Google Scholar] [CrossRef]
- Modjtahedi, A.; Amirfazli, A.; Farhad, S. Low Catalyst Loaded Ethanol Gas Fuel Cell Sensor. Sens. Actuators B Chem. 2016, 234, 70–79. [Google Scholar] [CrossRef]
- Svanberg, S. Atomic and Molecular Spectroscopy: Basic Aspects and Practical Applications; Springer: Berlin, Germany, 2012; pp. 1–186. [Google Scholar]
- Hummelgard, C.; Bryntse, I.; Bryzgalov, M.; Henning, J.A.; Martin, H.; Norén, M.; Rödjegard, H. Low-Cost NDIR Based Sensor Platform for Sub-Ppm Gas Detection. Urban Clim. 2015, 14, 342–350. [Google Scholar] [CrossRef]
- Hodgkinson, J.; Smith, R.; Ho, W.O.; Saffell, J.R.; Tatam, R.P. Non-Dispersive Infra-Red (NDIR) Measurement of Carbon Dioxide at 4.2 μm in a Compact and Optically Efficient Sensor. Sens. Actuators B Chem. 2013, 186, 580–588. [Google Scholar] [CrossRef]
- Yi, S. Temperature Compensation Methods of Nondispersive Infrared CO2 Gas Sensor with Dual Ellipsoidal Optical Waveguide. Sens. Mater. 2017, 29, 243–252. [Google Scholar]
- Tan, Q.; Pei, X.; Zhu, S.; Sun, D.; Liu, J.; Xue, C.; Liang, T.; Zhang, W.; Xiong, J. Development of an Optical Gas Leak Sensor for Detecting Ethylene, Dimethyl Ether and Methane. Sensors 2013, 13, 4157–4169. [Google Scholar] [CrossRef] [PubMed]
- Xing, X.; Lei, Y.; Shang, W.; Du, L.; Guo, P. Theoretical Research on Ellipsoidal Structure Methane Gas Detection Based on Near Infrared Light Sources of PbSe Quantum Dots. J. Nano 2017, 2017, 1–7. [Google Scholar] [CrossRef]
- Park, J.; Yi, S. Nondispersive Infrared Ray CH4 Gas Sensor Using Focused Infrared Beam Structures. Sens. Mater. 2011, 23, 147–158. [Google Scholar]
- Sklorz, A.; Janßen, S.; Lang, W. Detection Limit Improvement for NDIR Ethylene Gas Detectors Using Passive Approaches. Sens. Actuators B Chem. 2012, 175, 246–254. [Google Scholar] [CrossRef]
- Hök, B.; Pettersson, H.; Andersson, A.K.; Haasl, S.; Åkerlund, P. Breath Analyzer for Alcolocks and Screening Devices. IEEE Sens. J. 2010, 10, 10–15. [Google Scholar] [CrossRef]
- Kim, J.; Lee, K.; Yi, S. NDIR Ethanol Gas Sensor with Two Elliptical Optical Structures. In Proceedings of the Eurosensors 2016, Budapest, Hungary, 4–7 September 2016; Volume 168, pp. 359–362. [Google Scholar]
- Kim, J.; Shin, S.; Yi, S. Effects of Infrared Energy on Dual Elliptical NDIR Ethanol Gas Sensors. In Proceedings of the Eurosensors 2017, Paris, France, 3–6 September 2017; Volume 1, pp. 1–4. [Google Scholar]
- Fonseca, L.; Rubio, R.; Santander, J.; Calaza, C.; Sabaté, N.; Ivanov, P.; Figueras, E.; Gràcia, I.; Cané, C.; Udina, S.; et al. Qualitative and Quantitative Substance Discrimination Using a CMOS Compatible Non-Specific NDIR Microarray. Sens. Actuators B Chem. 2009, 141, 396–403. [Google Scholar] [CrossRef]
- Dinh, T.V.; Choi, I.Y.; Son, Y.S.; Kim, J.C. A Review on Non-Dispersive Infrared Gas Sensors: Improvement of Sensor Detection Limit and Interference Correction. Sens. Actuators B Chem. 2016, 231, 529–538. [Google Scholar] [CrossRef]
- Kahouli, A.; Sylvestre, A.; Ortega, L.; Jomni, F.; Yangui, B.; Kahouli, A.; Sylvestre, A.; Ortega, L.; Jomni, F.; Yangui, B.; et al. Structural and Dielectric Study of Parylene C Thin Films. Appl. Phys. Lett. 2013, 152901, 1–3. [Google Scholar] [CrossRef]
- Parylene Properties & Characteristics. Available online: http://www.vp-scientific.com/parylene_properties.html (accessed on 4 June 2018).
- Schubert, J.; Krabbe, A.; Birk, M.; Wagner, G. Parylene Anti-Reflection Coating of a Quasi-Optical Hot-Electron- Bolometric Mixer at Terahertz Frequencies. Infrared Phys. Technol. 2001, 42, 41–47. [Google Scholar]
- Yasuda, H.; Chun, B.H.; Cho, D.L.; Lin, T.J.; Yang, D.J.; Antonelli, J.A. Interface-Engineered Parylene C Coating for Corrosion Protection of Cold-Rolled Steel. Corrosion 1996, 52, 169–176. [Google Scholar] [CrossRef]
- Li, Y.; Su, L.; Shou, C.; Yu, C.; Deng, J.; Fang, Y. Surface-Enhanced Molecular Spectroscopy (SEMS) Based on Perfect-Absorber Metamaterials in the Mid-Infrared. Sci. Rep. 2013, 3, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Lee, J.; Lee, K.; Yi, S. Enhanced Characteristics of Nondispersive Infrared CO2 Gas Sensor by Deposition of Hydrophobic Thin Film. In Proceedings of the Eurosensors 2017, Paris, France, 3–6 September 2017; Volume 1, pp. 1–5. [Google Scholar]
- Mendes, L.B.; Ogink, N.W.M.; Van Dooren, H.J.C.; Mosquera, J.; Gerais, M. NDIR Gas Sensor for Spatial Monitoring of Carbon Dioxide Concentrations in Naturally Ventilated Livestock Buildings. Sensors 2015, 15, 11239–11257. [Google Scholar] [CrossRef] [PubMed]
- Gordon, I.E.; Rothman, L.S.; Hill, C.; Kochanov, R.V.; Tan, Y.; Bernath, P.F.; Birk, M.; Boudon, V.; Campargue, A.; Chance, K.V.; et al. The HITRAN2016 Molecular Spectroscopic Database. J. Quant. Spectrosc. Radiat. Transf. 2017, 203, 3–69. [Google Scholar] [CrossRef]
- NIST Chemistry WebBook. 2018. Available online: https://webbook.nist.gov/chemistry/ (accessed on 28 June 2018).
- Eisberg, R.; Resnick, R.; Brown, J. Quantum Physics of Atoms, Molecules, Solids, Nuclei, and Particles; Wiley John & Sons: Hoboken, NJ, USA, 1986; Volume 39. [Google Scholar]
- Kim, J.; Yi, S. Effects of Temperature and Humidity on NDIR CO2 Gas Sensor. J. Sens. Sci. Technol. 2017, 26, 179–185. [Google Scholar] [CrossRef]
- Ranganath, G.S. Black-Body Radiation. Resonance 2008, 13, 115–133. [Google Scholar] [CrossRef]
- Ma, L.; Yang, J.; Nie, J. Two Forms of Wien‘s Displacement Law. Education 2009, 3, 566–568. [Google Scholar]
- Maikala, R.V. Modified Beer’s Law—Historical Perspectives and Relevance in near-Infrared Monitoring of Optical Properties of Human Tissue. Int. J. Ind. Ergon. 2010, 40, 125–134. [Google Scholar] [CrossRef]
- Sieber, I.; Eggert, H.; Suphan, K.; Oliver, N. Optical Modeling of the Analytical Chamber of an IR Gas Sensor. Lamp 2001, 4408, 272–282. [Google Scholar]
- Viola, R. High-Luminosity Multipass Cell for Infrared Imaging Spectroscopy. Appl. Opt. 2006, 45, 2805–2809. [Google Scholar] [CrossRef] [PubMed]
- Martin, H. Gas Sensor. Swedish Patent SE 510549, 13 November 1995. [Google Scholar]
- Zhang, Y.; Gao, W.; Song, Z.; An, Y.; Li, L.; Song, Z.; Yu, W.W.; Wang, Y. Design of a Novel Gas Sensor Structure Based on Mid-Infrared Absorption Spectrum. Sens. Actuators B Chem. 2010, 147, 5–9. [Google Scholar] [CrossRef]
- Zheng, C.T.; Ye, W.L.; Li, G.L.; Yu, X.; Zhao, C.X.; Song, Z.W.; Wang, Y.D. Performance Enhancement of a Mid-Infrared CH4 detection Sensor by Optimizing an Asymmetric Ellipsoid Gas-Cell and Reducing Voltage-Fluctuation: Theory, Design and Experiment. Sens. Actuators B Chem. 2011, 160, 389–398. [Google Scholar] [CrossRef]
- Park, J.; Min, N.; Kweon, S.; Yi, S. Novel NDIR CO2 sensor with two concave mirrors. In Proceedings of the Asia-Pacific Conference of Transducers and Micro-Nano Technology (APCOT) 2006, Singapore, 25–28 June 2006; p. 137. [Google Scholar]
- Jang, S.; Chung, S.; Yi, S. Characteristics of an Optical Waveguide with Two Identical Elliptical Structures. J. Korean Inst. Gas. 2014, 18, 48–54. [Google Scholar] [CrossRef]
- Kim, J.; Yi, S. Properties of Non-Dispersive Infrared Ethanol Gas Sensors according to the Irradiation Energy. J. Sens. Sci. Technol. 2017, 26, 168–172. [Google Scholar] [CrossRef]
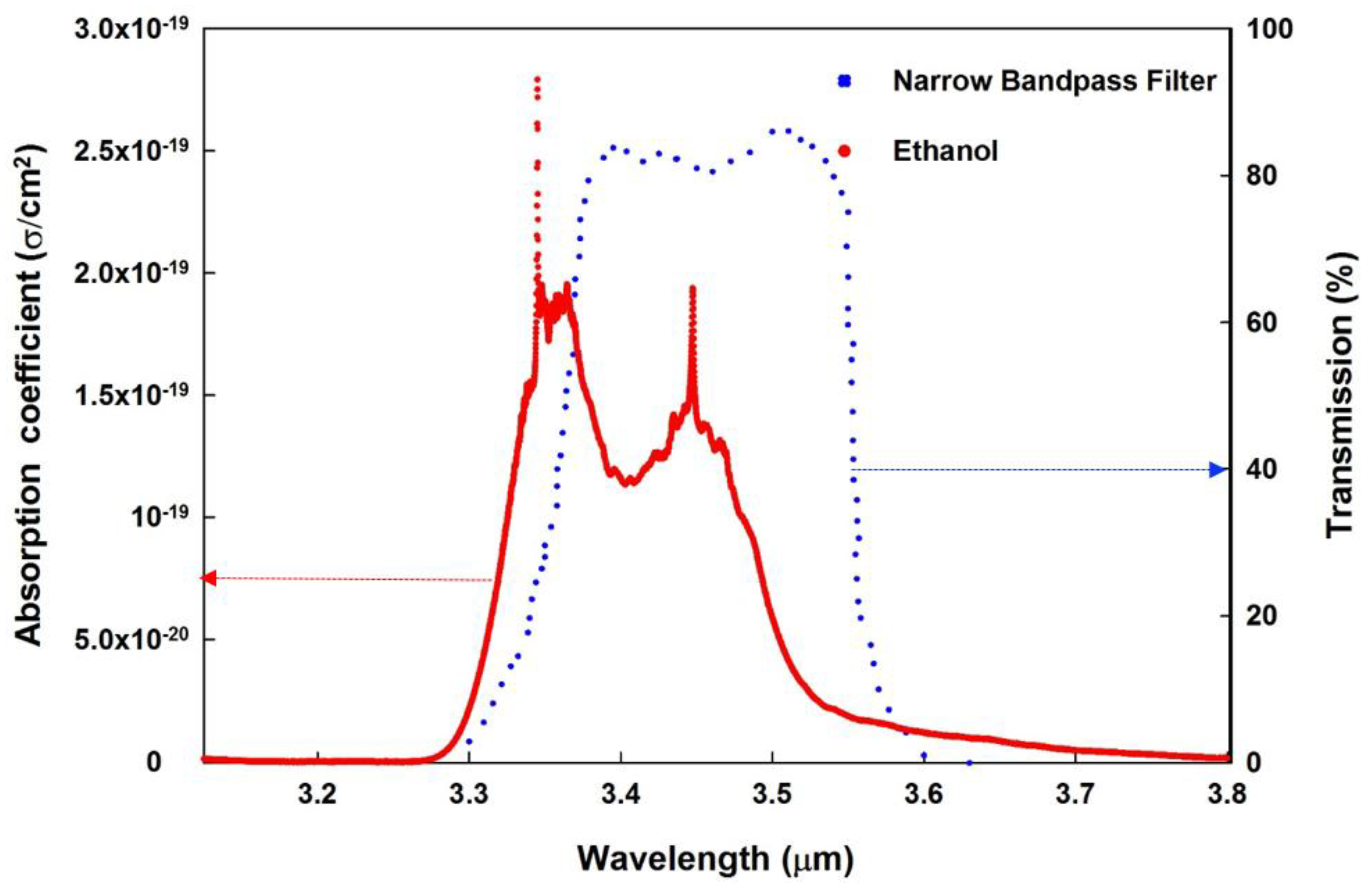
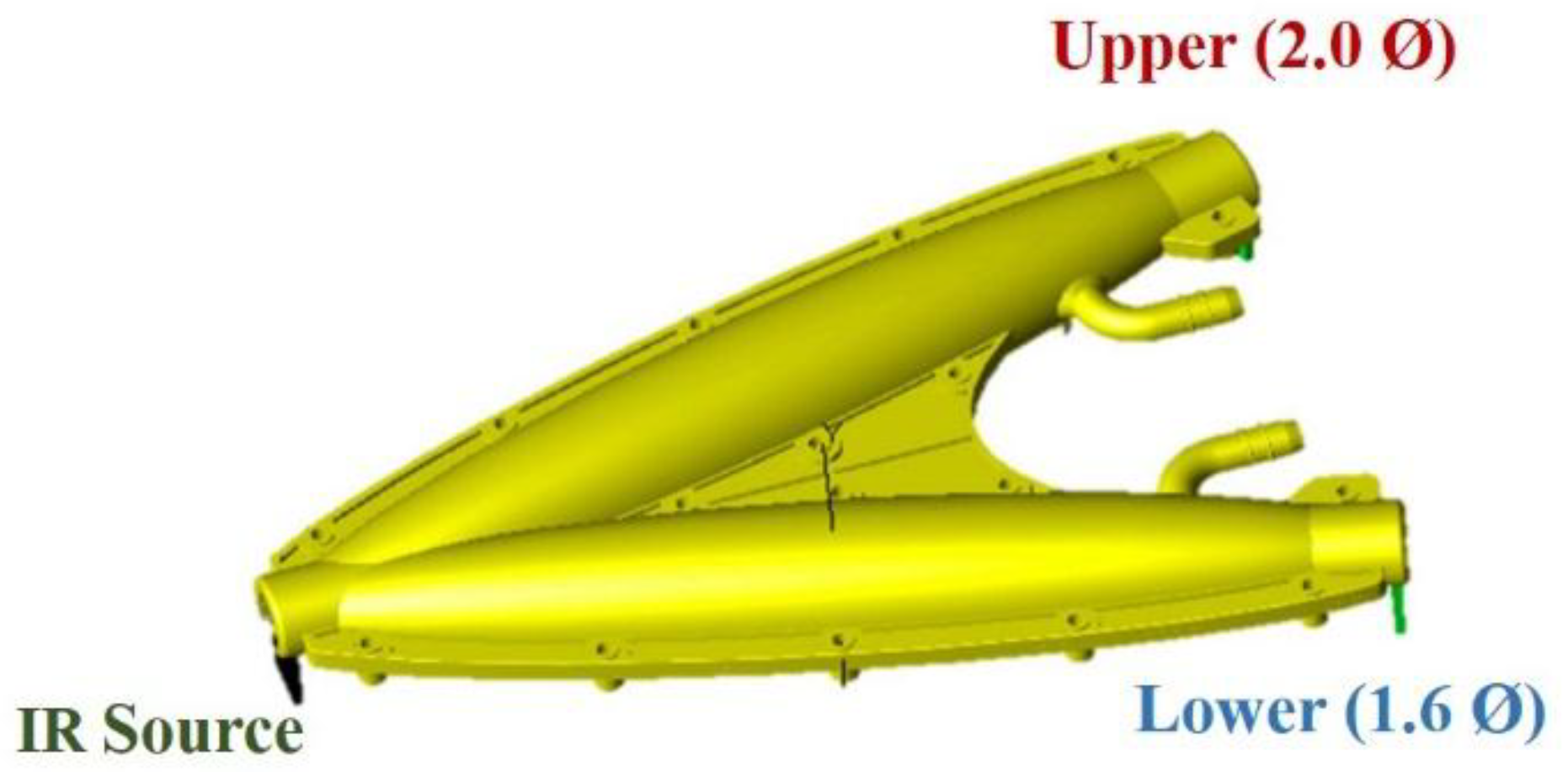

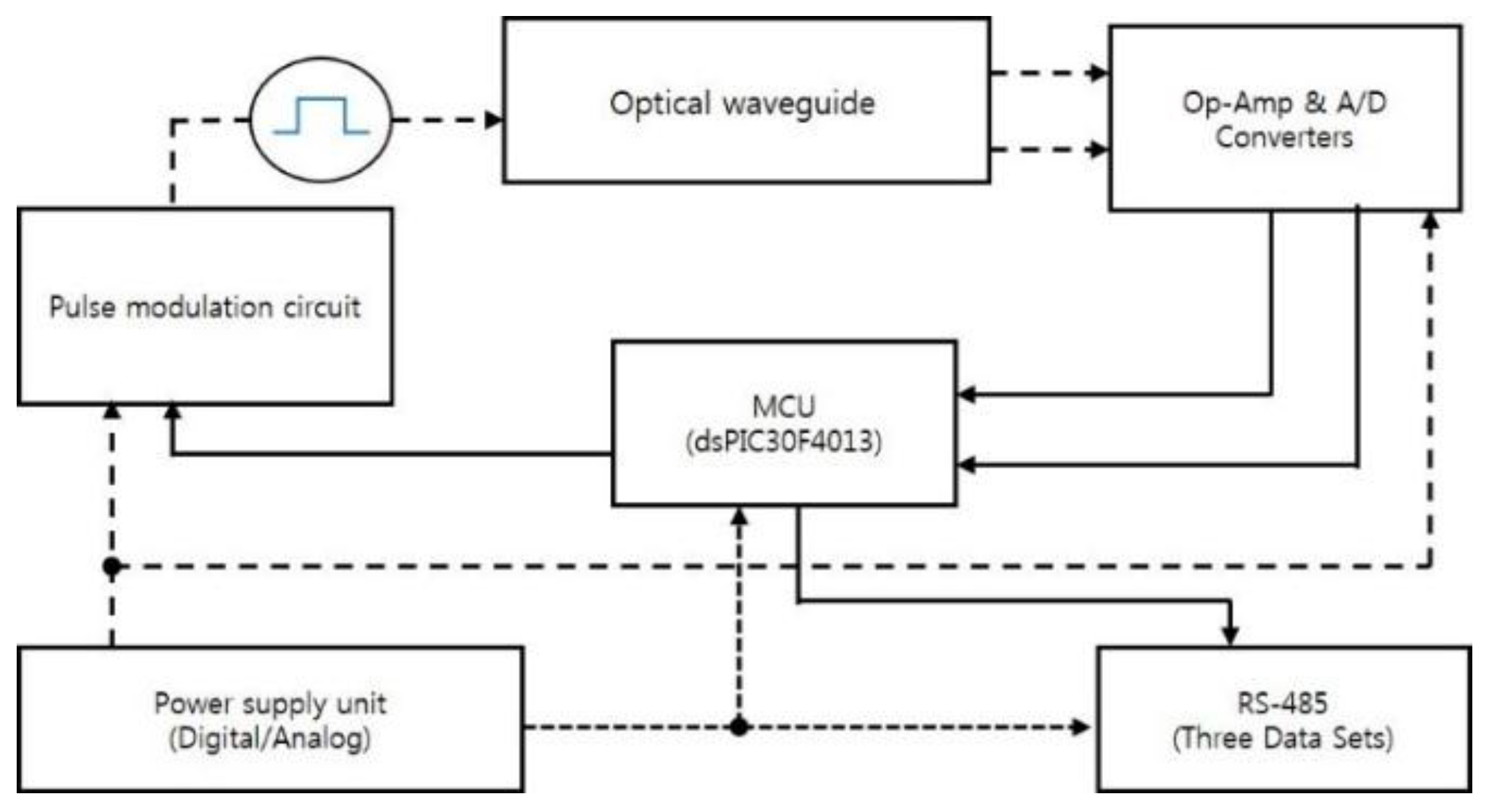

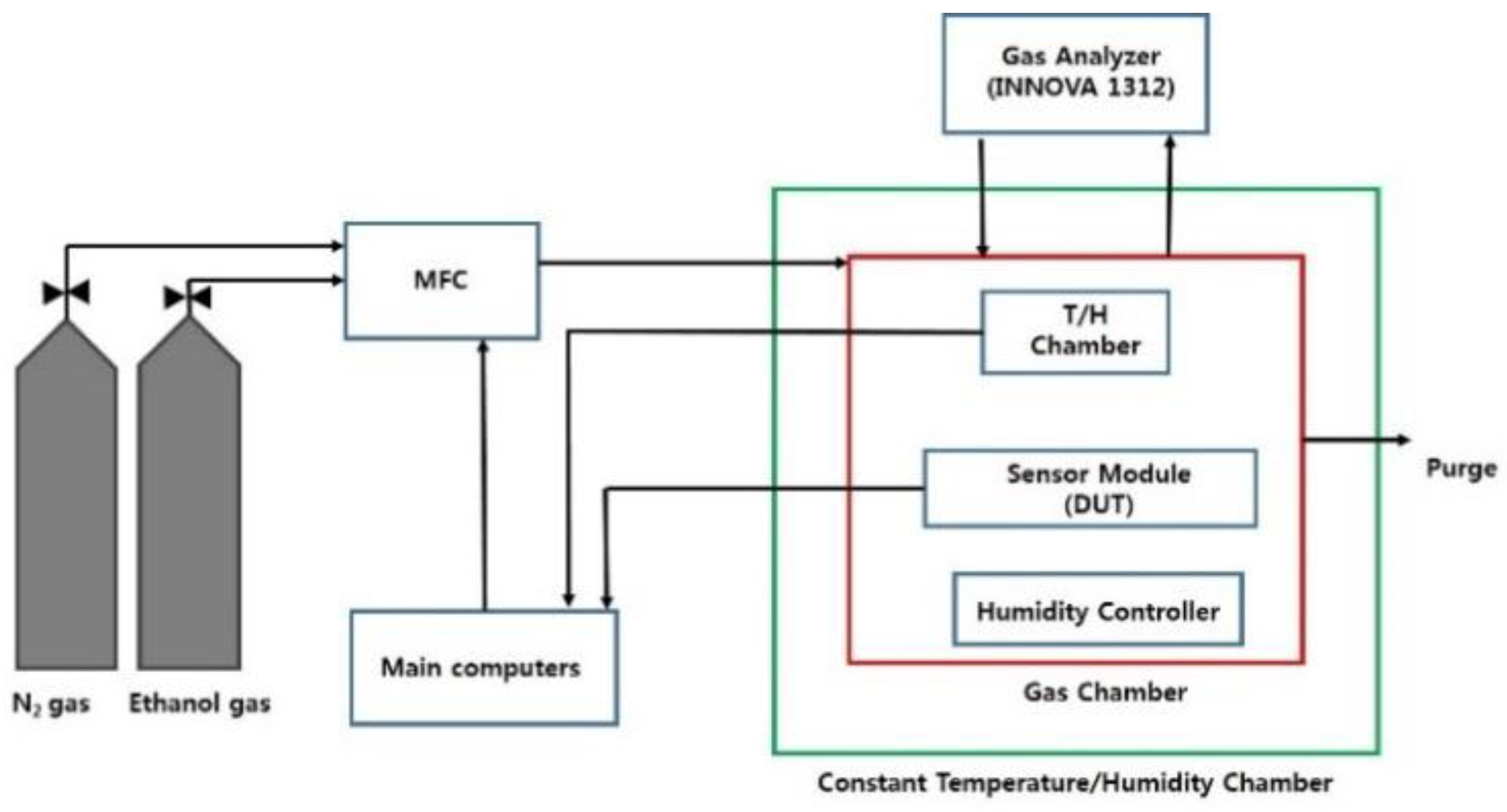
| Parameter | Typical Value |
|---|---|
| Spectral output range | 1–10 μm |
| Emitter Surface Area | 1.7 × 1.7 mm2 |
| Working temperature | 750 °C |
| Power consumption | 980 mW |
| Modulation frequency | 0–100 Hz (optimized at 2.5 Hz) |
| Warm up time | <30 ms |
| Lifetime | >5000 h at 750 °C |
| Temp. (K) | Normal (2.00 Ø) | Parylene-C (2.00 Ø) | ||
|---|---|---|---|---|
| VoF (T) | VoF (T) | |||
| 253 | 1.3736 | 5.9 × 10−5 | 1.7712 | 5.0 × 10−5 |
| 263 | 1.4204 | 5.6 × 10−5 | 1.8271 | 5.1 × 10−5 |
| 273 | 1.4572 | 5.1 × 10−5 | 1.8536 | 4.8 × 10−5 |
| 298 | 1.5435 | 5.4 × 10−5 | 1.9375 | 5.0 × 10−5 |
| 313 | 1.5920 | 5.3 × 10−5 | 1.9731 | 4.8 × 10−5 |
| 323 | 1.7378 | 5.1 × 10−5 | 2.0255 | 4.8 × 10−5 |
| 333 | 1.7909 | 5.3 × 10−5 | 2.0784 | 5.1 × 10−5 |
| Design Principle | Error (%) | |
|---|---|---|
| One ethanol and one reference detector | ±5% | [16] |
| One ellipsoid partially covered with tape | 20–25% at <200 ppm −10 to 1% at >200 ppm | [41] |
| Hollow disk (1.6 Ø) inserted before one detector | ±15% at <100 ppm −1.2–3% at >100 ppm | [17] |
| Hollow disk (1.6 Ø) inserted before one sensor, detectors of one of the modules coated with Parylene-C | More than 10% for ≤50 ppm ±5% at >50 ppm | This |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hussain, H.; Kim, J.; Yi, S. Characteristics and Temperature Compensation of Non-Dispersive Infrared (NDIR) Alcohol Gas Sensors According to Incident Light Intensity. Sensors 2018, 18, 2911. https://doi.org/10.3390/s18092911
Hussain H, Kim J, Yi S. Characteristics and Temperature Compensation of Non-Dispersive Infrared (NDIR) Alcohol Gas Sensors According to Incident Light Intensity. Sensors. 2018; 18(9):2911. https://doi.org/10.3390/s18092911
Chicago/Turabian StyleHussain, Humaira, JinHo Kim, and SeungHwan Yi. 2018. "Characteristics and Temperature Compensation of Non-Dispersive Infrared (NDIR) Alcohol Gas Sensors According to Incident Light Intensity" Sensors 18, no. 9: 2911. https://doi.org/10.3390/s18092911
APA StyleHussain, H., Kim, J., & Yi, S. (2018). Characteristics and Temperature Compensation of Non-Dispersive Infrared (NDIR) Alcohol Gas Sensors According to Incident Light Intensity. Sensors, 18(9), 2911. https://doi.org/10.3390/s18092911






