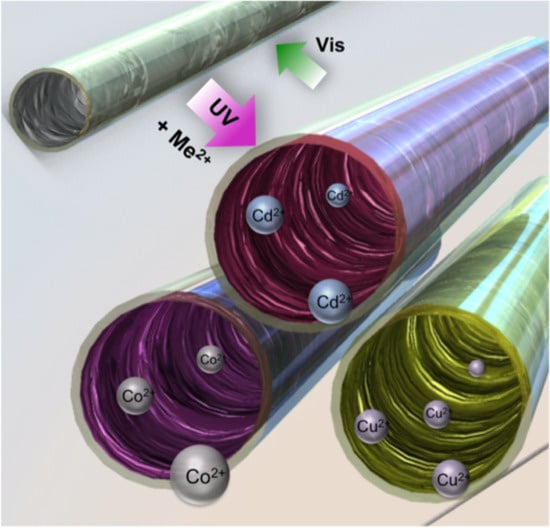
Micro-Capillary Coatings Based on Spiropyran Polymeric Brushes for Metal Ion Binding, Detection, and Release in Continuous Flow
Abstract
1. Introduction
2. Experimental
2.1. Materials
2.2. Synthesis of Spiropyran Norbornene Monomer (SP)
2.3. Synthesis of Spiropyran Polymeric Brushes (PolySP)
2.4. Light Source
2.5. Methods
3. Results and Discussion
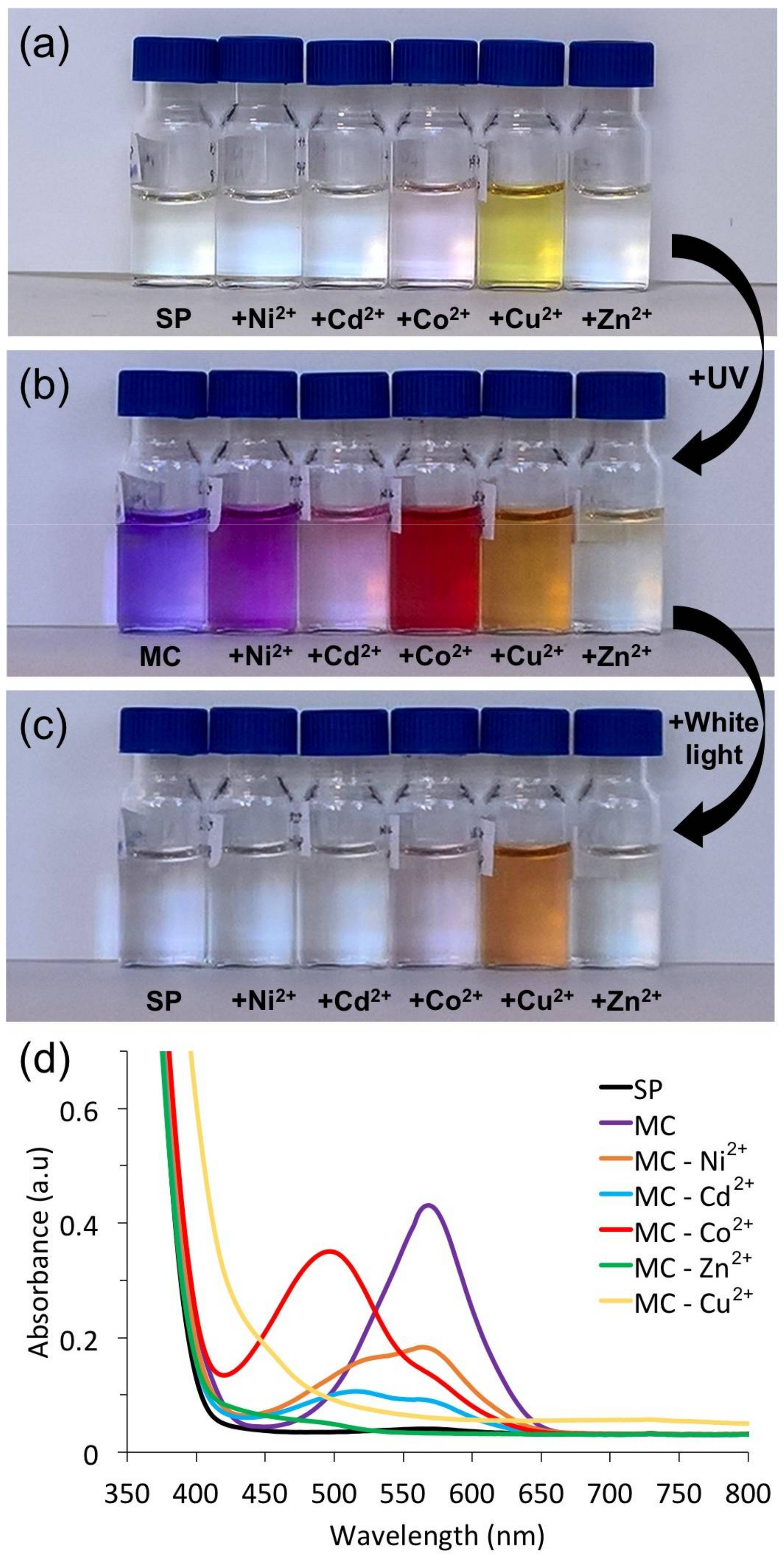
3.1. Photochromism of Spiropyrans
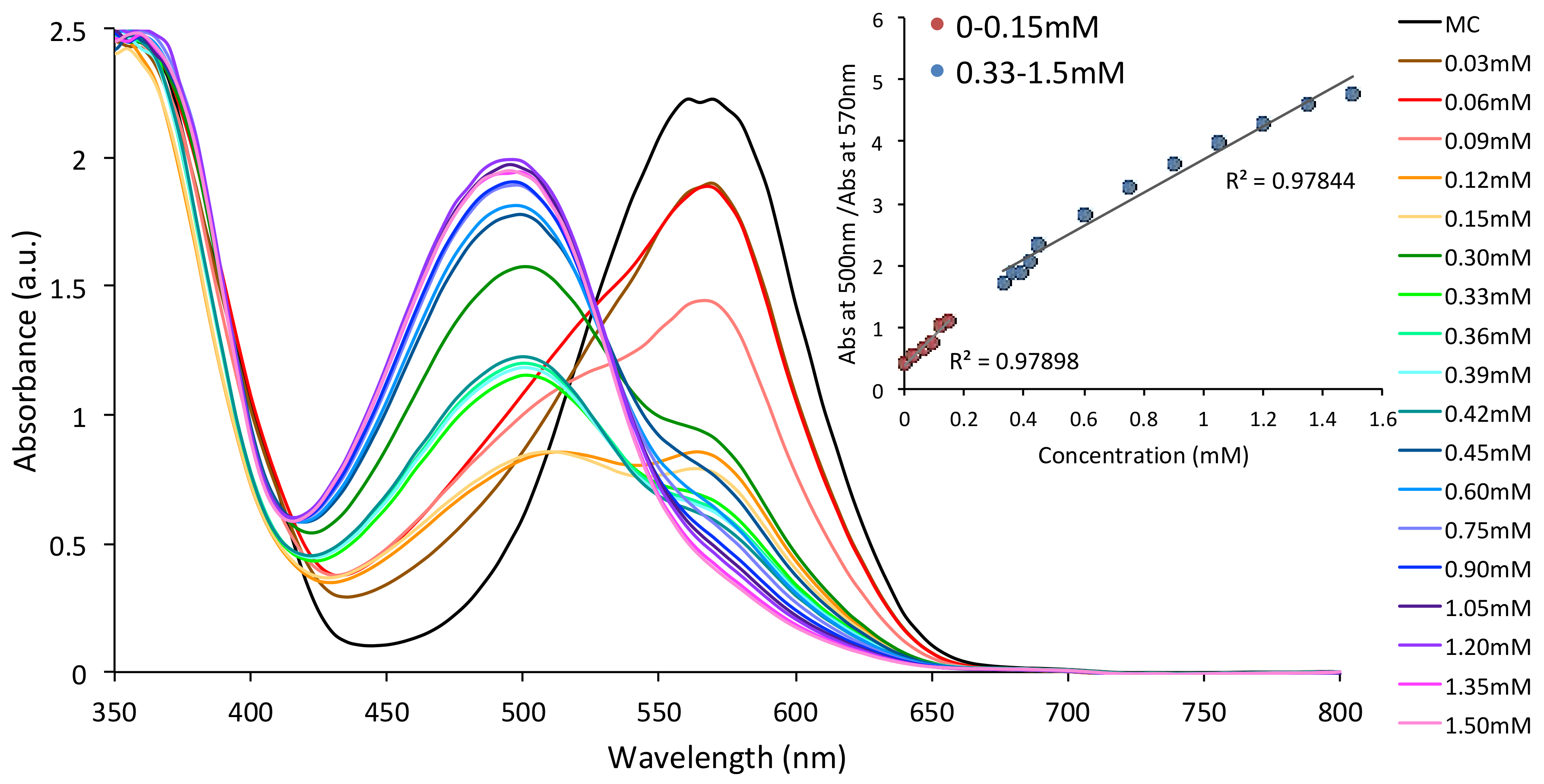
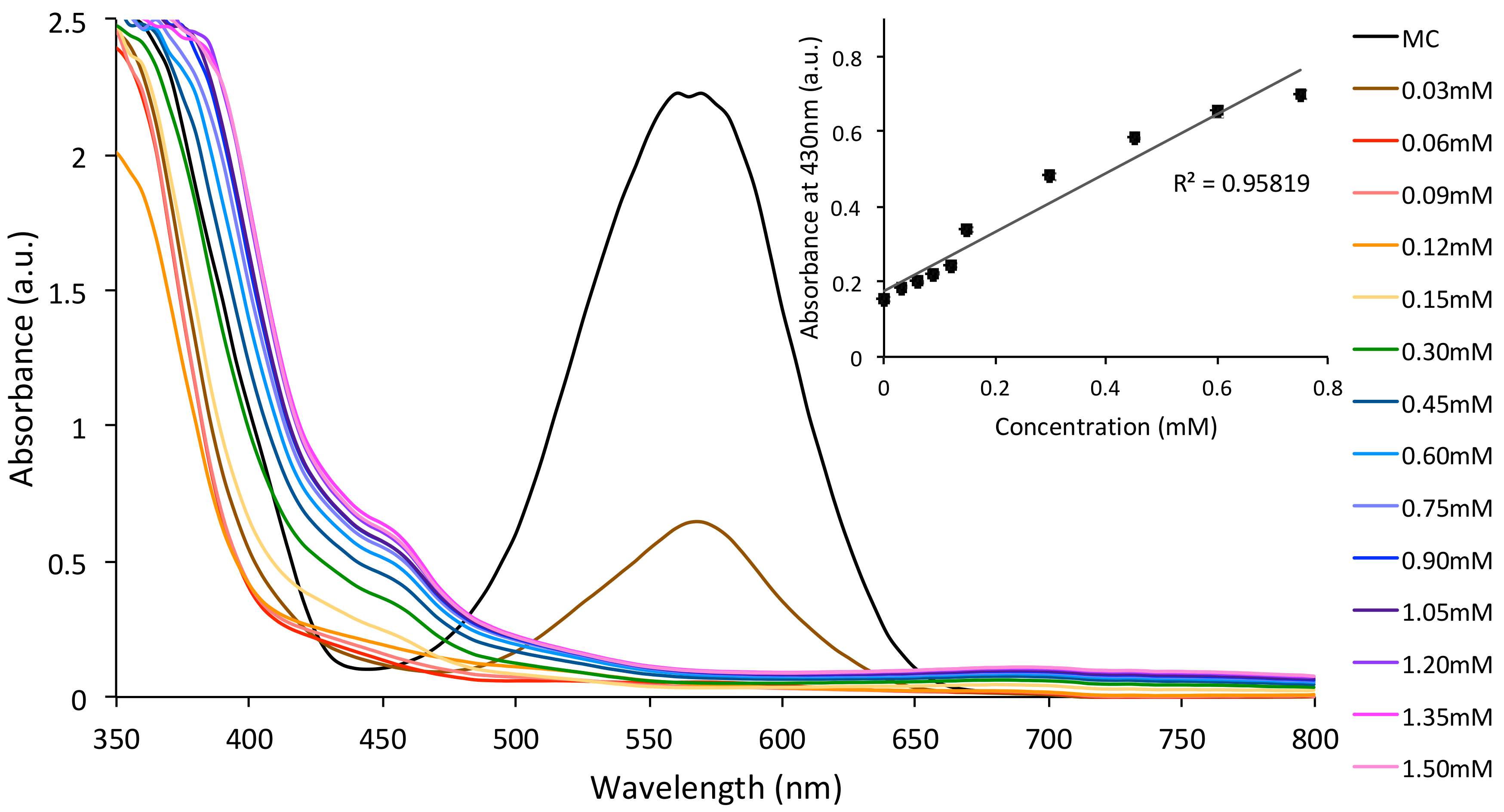
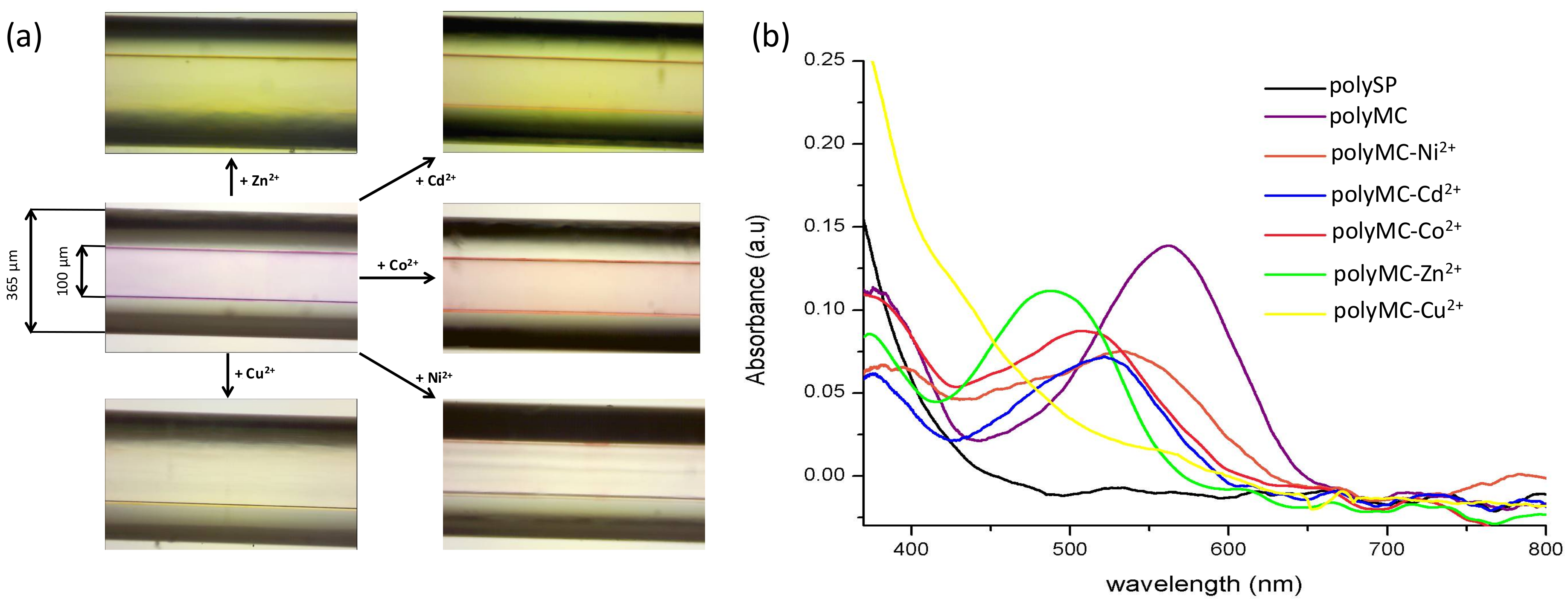
3.2. Metal Ion Binding—Solution Studies
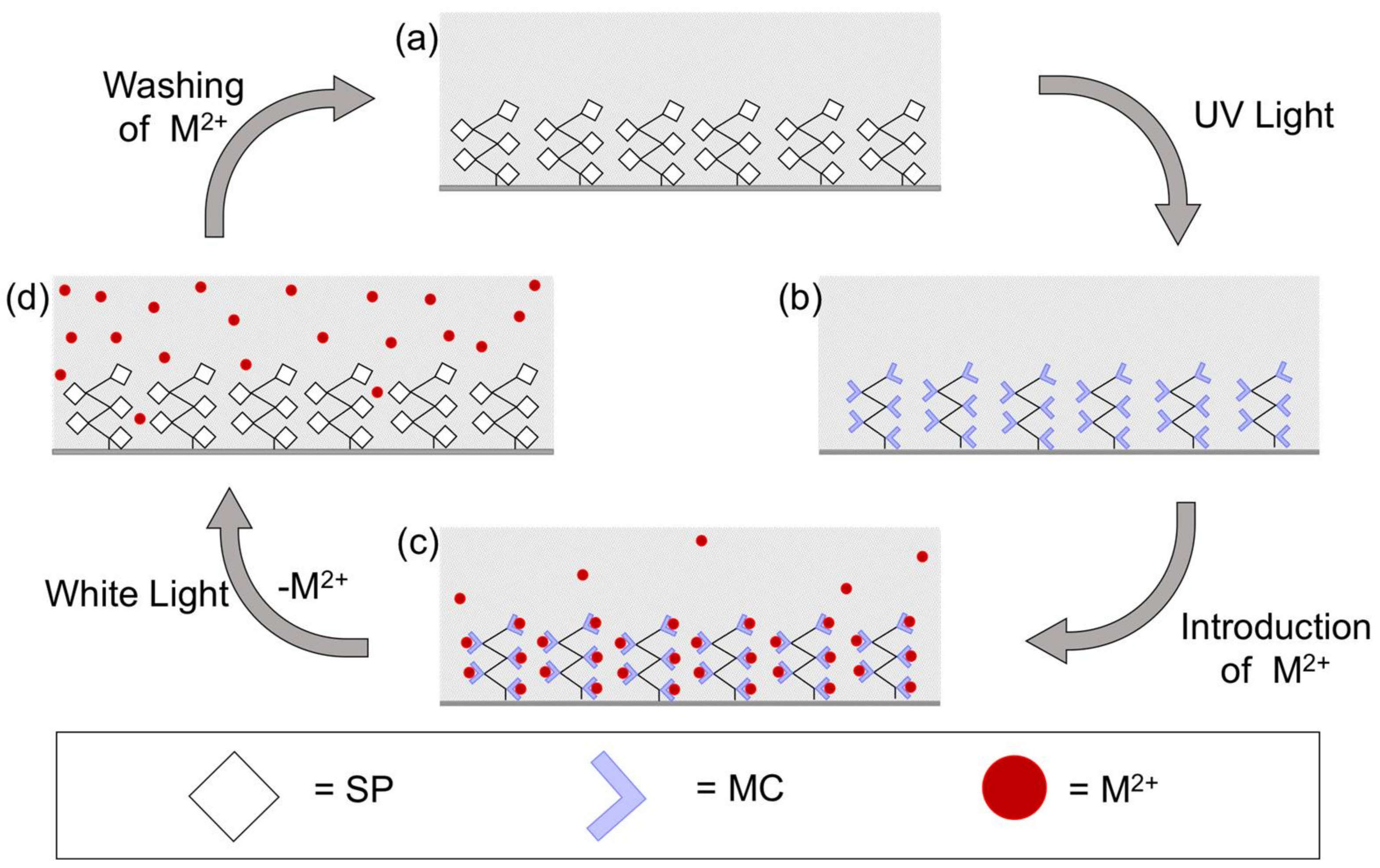
3.3. Metal Ion Binding—Micro-Capillary Studies
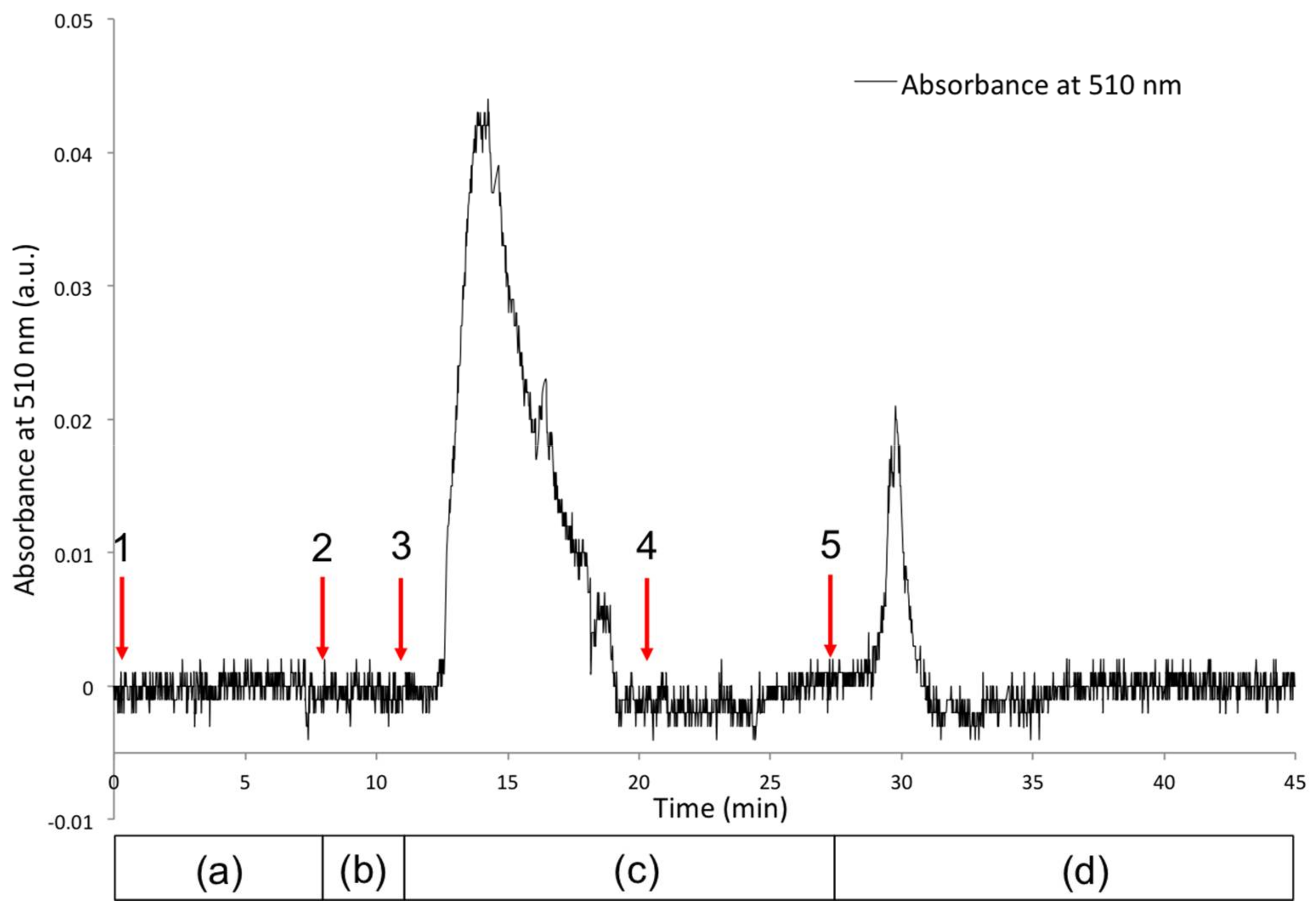
- Passive form (polySP) in the presence of solvent; capillary is colourless, no Co2+ present; low baseline signal at 510 nm (Figure 6a).
- Switch back to solvent without Co2+; signal returns to baseline indicating unbound Co2+ has been removed. Capillary still has a reddish colour suggesting bound Co2+ is present in the coating (Figure 6c).
- White light source is turned ON; coating reverts to colourless (polySP) passive form; bound Co2+ is simultaneously released (Figure 6d) and detected as the peak at ca. 30 min.
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Fischer, E.; Hirshberg, Y. Formation of Coloured Forms of Spirans by Low-Temperature Irradiation; Royal Society of Chemistry: Cambridge, UK, 1952; pp. 4522–4524. [Google Scholar]
- Triolo, C.; Patanè, S.; Mazzeo, M.; Gambino, S.; Gigli, G.; Allegrini, M. Pure optical nano-writing on light-switchable spiropyrans/merocyanine thin film. Opt. Express 2014, 22, 283–288. [Google Scholar] [CrossRef] [PubMed]
- Berkovic, G.; Krongauz, V.; Weiss, V. Spiropyrans and spirooxazines for memories and switches. Chem. Rev. 2000, 100, 1741–1754. [Google Scholar] [CrossRef] [PubMed]
- Kadowaki, S. Photochromic Lens for Eye Glasses. U.S. Patent 9,335,566, 10 May 2016. [Google Scholar]
- Johnson, N.M.; Smolin, Y.Y.; Shindler, C.; Hagaman, D.; Soroush, M.; Lau, K.K.; Ji, H.-F. Photochromic dye-sensitized solar cells. AIMS Mater. Sci. 2015, 2, 503–509. [Google Scholar] [CrossRef]
- Xie, X.; Crespo, G.A.; Mistlberger, G.; Bakker, E. Photocurrent generation based on a light-driven proton pump in an artificial liquid membrane. Nat. Chem. 2014, 6, 202–207. [Google Scholar] [CrossRef] [PubMed]
- Xie, X.; Bakker, E. Creating electrochemical gradients by light: From bio-inspired concepts to photoelectric conversion. Phys. Chem. Chem. Phys. 2014, 16, 19781–19789. [Google Scholar] [CrossRef] [PubMed]
- Xie, X.; Mistlberger, G.N.; Bakker, E. Reversible photodynamic chloride-selective sensor based on photochromic spiropyran. J. Am. Chem. Soc. 2012, 134, 16929–16932. [Google Scholar] [CrossRef] [PubMed]
- Florea, L.; Hennart, A.; Diamond, D.; Benito-Lopez, F. Synthesis and characterisation of spiropyran-polymer brushes in micro-capillaries: Towards an integrated optical sensor for continuous flow analysis. Sens. Actuators B Chem. 2012, 175, 92–99. [Google Scholar] [CrossRef]
- Florea, L.; McKeon, A.; Diamond, D.; Benito-Lopez, F. Spiropyran polymeric microcapillary coatings for photodetection of solvent polarity. Langmuir 2013, 29, 2790–2797. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Stumpel, J.E.; Ziółkowski, B.; Florea, L.; Diamond, D.; Broer, D.J.; Schenning, A.P. Photoswitchable ratchet surface topographies based on self-protonating spiropyran-NIPAAM hydrogels. ACS Appl. Mater. Interfaces 2014, 6, 7268–7274. [Google Scholar] [CrossRef] [PubMed]
- Williamson, A.; Ferro, M.; Leleux, P.; Ismailova, E.; Kaszas, A.; Doublet, T.; Quilichini, P.; Rivnay, J.; Rózsa, B.; Katona, G. Localized neuron stimulation with organic electrochemical transistors on delaminating depth probes. Adv. Mater. 2015, 27, 4405–4410. [Google Scholar] [CrossRef] [PubMed]
- Francis, W.; Dunne, A.; Delaney, C.; Florea, L.; Diamond, D. Spiropyran based hydrogels actuators—Walking in the light. Sens. Actuators B Chem. 2017, 250, 608–616. [Google Scholar] [CrossRef]
- Dunne, A.; Delaney, C.; Florea, L.; Diamond, D. Solvato-morphologically controlled, reversible NIPAAM hydrogel photoactuators. RSC Adv. 2016, 6, 83296–83302. [Google Scholar] [CrossRef]
- Ziółkowski, B.; Florea, L.; Theobald, J.; Benito-Lopez, F.; Diamond, D. Self-protonating spiropyran-co-NIPAM-co-acrylic acid hydrogel photoactuators. Soft Matter 2013, 9, 8754–8760. [Google Scholar] [CrossRef]
- Klajn, R. Spiropyran-based dynamic materials. Chem. Soc. Rev. 2014, 43, 148–184. [Google Scholar] [CrossRef] [PubMed]
- Benito-Lopez, F.; Scarmagnani, S.; Walsh, Z.; Paull, B.; Macka, M.; Diamond, D. Spiropyran modified micro-fluidic chip channels as photonically controlled self-indicating system for metal ion accumulation and release. Sens. Actuators B Chem. 2009, 140, 295–303. [Google Scholar] [CrossRef]
- Fries, K.H.; Driskell, J.D.; Samanta, S.; Locklin, J. Spectroscopic analysis of metal ion binding in spiropyran containing copolymer thin films. Anal. Chem. 2010, 82, 3306–3314. [Google Scholar] [CrossRef] [PubMed]
- Florea, L.; Diamond, D.; Benito-Lopez, F. Photo-responsive polymeric structures based on spiropyran. Macromol. Mater. Eng. 2012, 297, 1148–1159. [Google Scholar] [CrossRef]
- Florea, L.; Diamond, D.; Benito-Lopez, F. Opto-smart systems in microfluidics. In Research Perspectives on Functional Micro- and Nanoscale Coatings; IGI Global: Hershey, PA, USA, 2016; p. 265. [Google Scholar]
- Francis, W.; Fay, C.; Florea, L.; Diamond, D. Self-propelled chemotactic ionic liquid droplets. Chem. Commun. 2015, 51, 2342–2344. [Google Scholar] [CrossRef] [PubMed]
- Sun, T.; Qing, G.; Su, B.; Jiang, L. Functional biointerface materials inspired from nature. Chem. Soc. Rev. 2011, 40, 2909–2921. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, C.; Arribart, H.; Giraud Guille, M.M. Biomimetism and bioinspiration as tools for the design of innovative materials and systems. Nat. Mater. 2005, 4, 277. [Google Scholar] [CrossRef] [PubMed]
- Kobatake, S.; Yamada, T.; Uchida, K.; Kato, N.; Irie, M. Photochromism of 1, 2-bis (2,5-dimethyl-3-thienyl) perfluoro-cyclopentene in a single crystalline phase. J. Am. Chem. Soc. 1999, 121, 2380–2386. [Google Scholar] [CrossRef]
- Durr, H.; Bouas-Laurent, H. Photochromism: Molecules and Systems; Elsevier: Amsterdam, The Netherlands, 2003; pp. 314–467. [Google Scholar]
- Bénard, S.; Yu, P. New spiropyrans showing crystalline-state photochromism. Adv. Mater. 2000, 12, 48–50. [Google Scholar] [CrossRef]
- Harada, J.; Kawazoe, Y.; Ogawa, K. Photochromism of spiropyrans and spirooxazines in the solid state: Low temperature enhances photocoloration. Chem. Commun. 2010, 46, 2593–2595. [Google Scholar] [CrossRef] [PubMed]
- Chernyshev, A.V.; Voloshin, N.A.; Metelitsa, A.V.; Tkachev, V.V.; Aldoshin, S.M.; Solov’eva, E.; Rostovtseva, I.A.; Minkin, V.I. Metal complexes of new photochromic chelator: Structure, stability and photodissociation. J. Photochem. Photobiol. A Chem. 2013, 265, 1–9. [Google Scholar] [CrossRef]
- Phillips, J.P.; Mueller, A.; Przytal, F. Photochromic chelating agents. J. Am. Chem. Soc. 1965, 87, 4020–4021. [Google Scholar] [CrossRef]
- Taylor, L.D.; Nichoslon, J.; Davis, R.B. Photochromic chelating agents. Tetrahedron Lett. 1967, 8, 1585–1588. [Google Scholar] [CrossRef]
- Shao, N.; Jin, J.Y.; Wang, H.; Zhang, Y.; Yang, R.H.; Chan, W.H. Tunable photochromism of spirobenzopyran via selective metal ion coordination: An efficient visual and ratioing fluorescent probe for divalent copper ion. Anal. Chem. 2008, 80, 3466–3475. [Google Scholar] [CrossRef] [PubMed]
- Wojtyk, J.C.; Kazmaier, P. Effects of metal ion complexation on the spiropyran–merocyanine interconversion: Development of a thermally stable photo-switch. Chem. Commun. 1998, 16, 1703–1704. [Google Scholar] [CrossRef]
- Natali, M.; Giordani, S. Interation studies between photochromic spiropyrans and transition metal cations: The curious case of copper. Org. Biomol. Chem. 2012, 10, 1162–1171. [Google Scholar] [CrossRef] [PubMed]
- Görner, H.; Chibisov, A.K. Complexes of spiropyran-derived merocyannies with metal ions themally activated and light-induced processes. J. Chem. Soc. Faraday Trans. 1998, 94, 2557–2564. [Google Scholar] [CrossRef]
- Fries, K.; Samanta, S.; Orski, S.; Locklin, J. Reversible colorimetric ion sensors based on surface initiated polymerization of photochromic polymers. Chem. Commun. 2008, 47, 6288–6290. [Google Scholar] [CrossRef] [PubMed]
- Natali, M.; Aakeröy, C.; Desper, J.; Giordani, S. The role of metal ions and counterions in the switching behavior of a carboxylic acid functionalized spiropyran. Dalton Trans. 2010, 39, 8269–8277. [Google Scholar] [CrossRef] [PubMed]
- Fries, K.H.; Sheppard, G.R.; Bilbrey, J.A.; Locklin, J. Tuning chelating groups and comonomers in spiropyran-containing copolymer thin films for color-specific metal ion binding. Polym. Chem. 2014, 5, 2094–2102. [Google Scholar] [CrossRef]
- Ghasemi, J.; Niazi, A.; Maeder, M. Spectrophotometric studies on the protonation and nickel complexation equilibria of 4-(2-pyridylazo) resorcinol using global analysis in aqueous solution. J. Braz. Chem. Soc. 2007, 18, 267–272. [Google Scholar] [CrossRef]
- Ghasemi, J.; Peyman, H.; Meloun, M. Study of complex formation between 4-(2-pyridylazo) resorcinol and Al3+, Fe3+, Zn2+, and Cd2+ ions in an aqueous solution at 0.1 M ionic strength. J. Chem. Eng. Data 2007, 52, 1171–1178. [Google Scholar] [CrossRef]

© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dunne, A.; Delaney, C.; McKeon, A.; Nesterenko, P.; Paull, B.; Benito-Lopez, F.; Diamond, D.; Florea, L. Micro-Capillary Coatings Based on Spiropyran Polymeric Brushes for Metal Ion Binding, Detection, and Release in Continuous Flow. Sensors 2018, 18, 1083. https://doi.org/10.3390/s18041083
Dunne A, Delaney C, McKeon A, Nesterenko P, Paull B, Benito-Lopez F, Diamond D, Florea L. Micro-Capillary Coatings Based on Spiropyran Polymeric Brushes for Metal Ion Binding, Detection, and Release in Continuous Flow. Sensors. 2018; 18(4):1083. https://doi.org/10.3390/s18041083
Chicago/Turabian StyleDunne, Aishling, Colm Delaney, Aoife McKeon, Pavel Nesterenko, Brett Paull, Fernando Benito-Lopez, Dermot Diamond, and Larisa Florea. 2018. "Micro-Capillary Coatings Based on Spiropyran Polymeric Brushes for Metal Ion Binding, Detection, and Release in Continuous Flow" Sensors 18, no. 4: 1083. https://doi.org/10.3390/s18041083
APA StyleDunne, A., Delaney, C., McKeon, A., Nesterenko, P., Paull, B., Benito-Lopez, F., Diamond, D., & Florea, L. (2018). Micro-Capillary Coatings Based on Spiropyran Polymeric Brushes for Metal Ion Binding, Detection, and Release in Continuous Flow. Sensors, 18(4), 1083. https://doi.org/10.3390/s18041083