Enhanced Humidity Sensitivity with Silicon Nanopillar Array by UV Light
Abstract
:1. Introduction
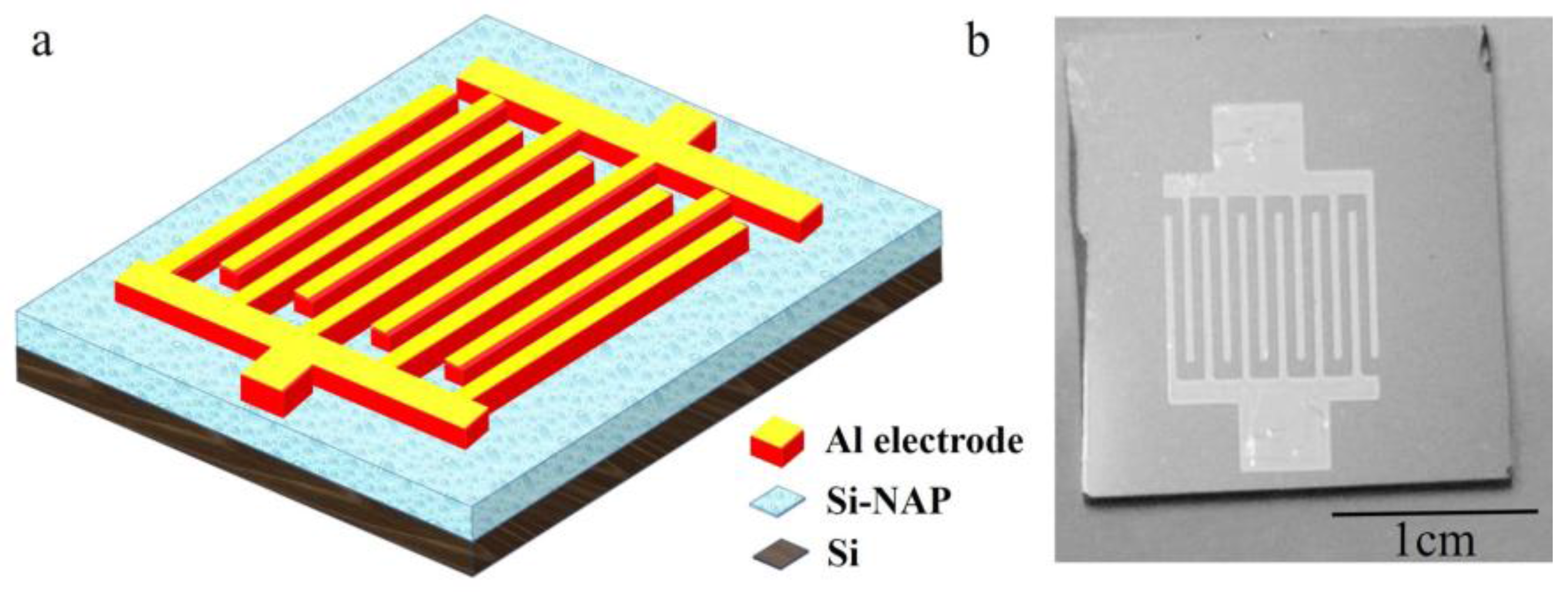
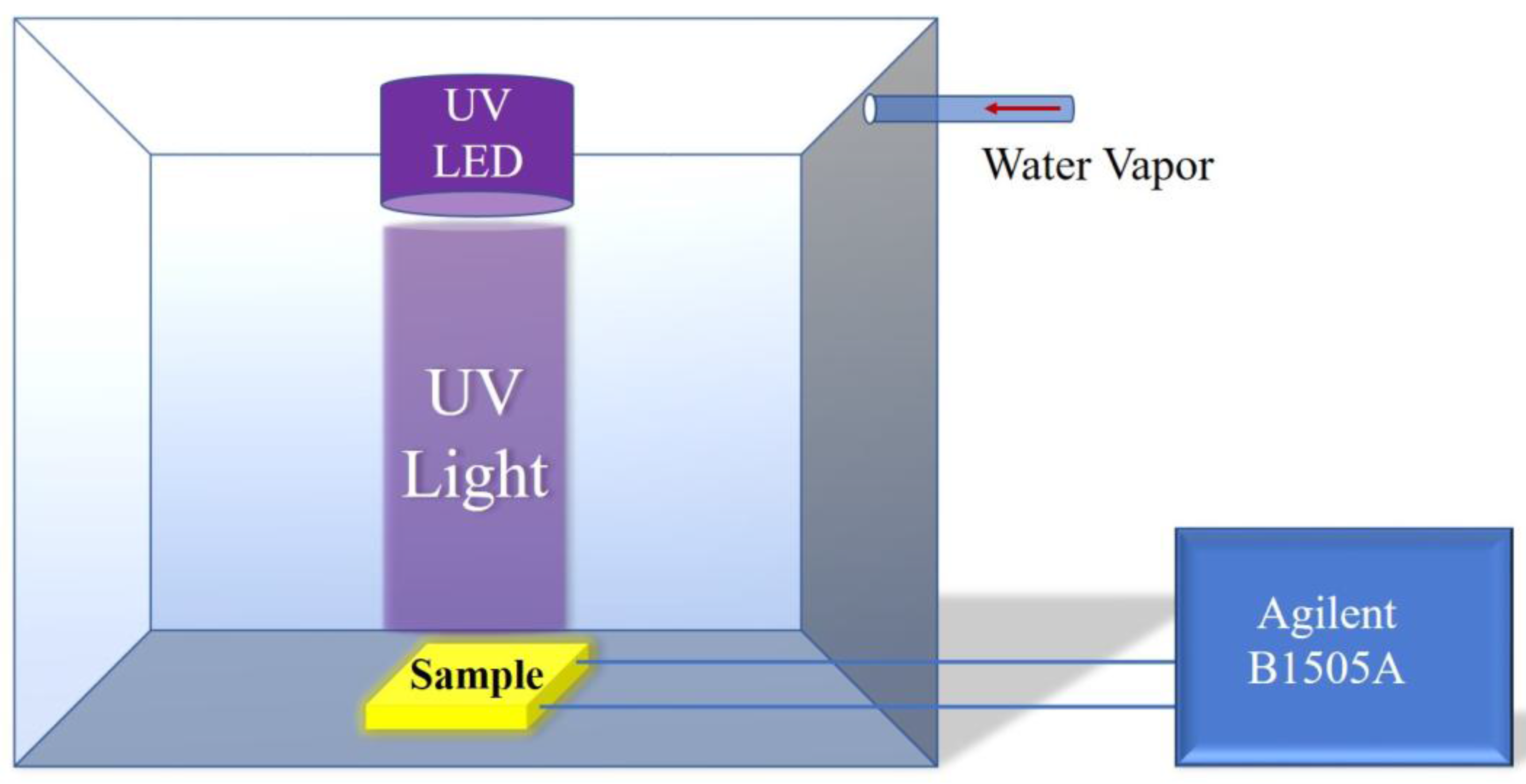
2. Experiment
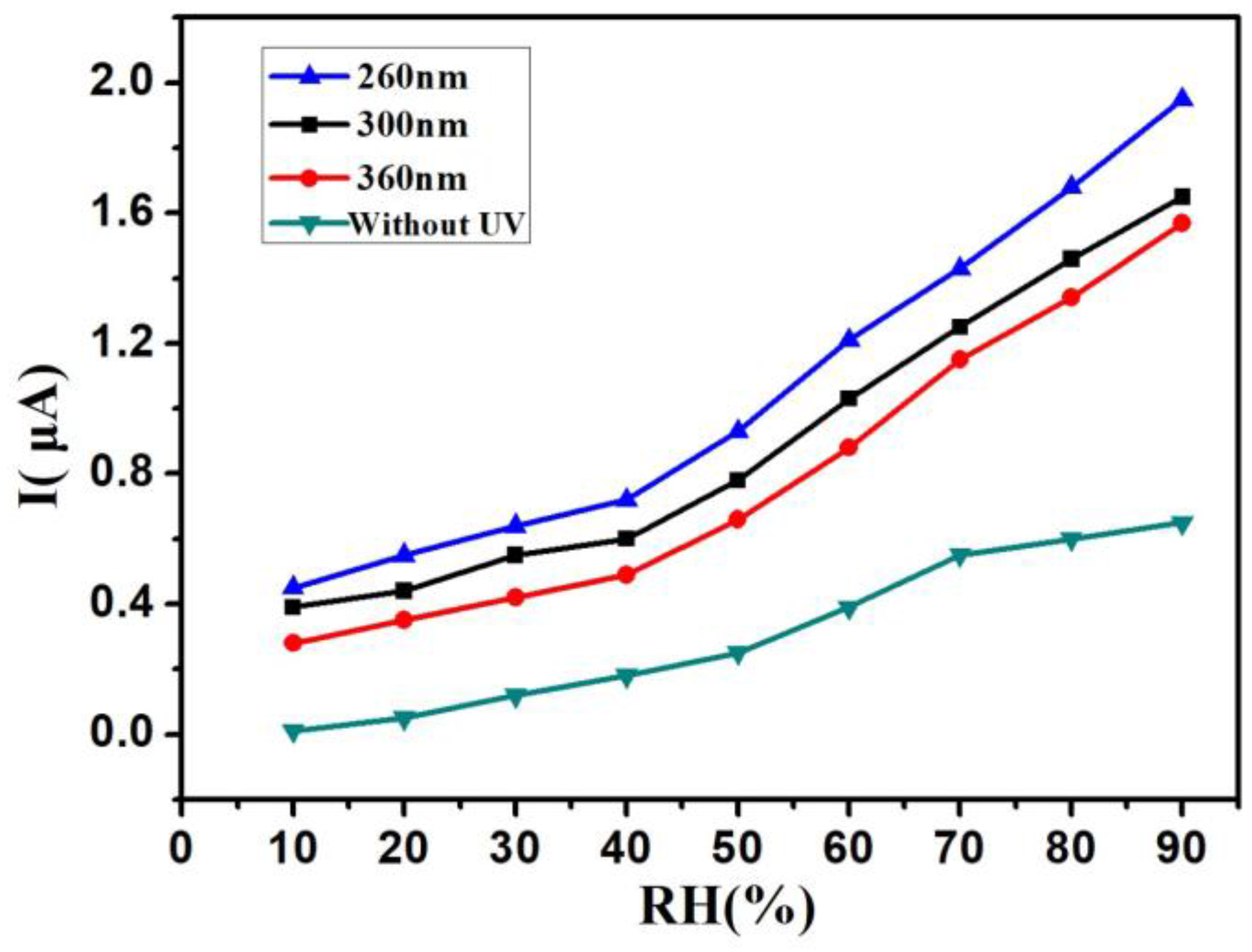
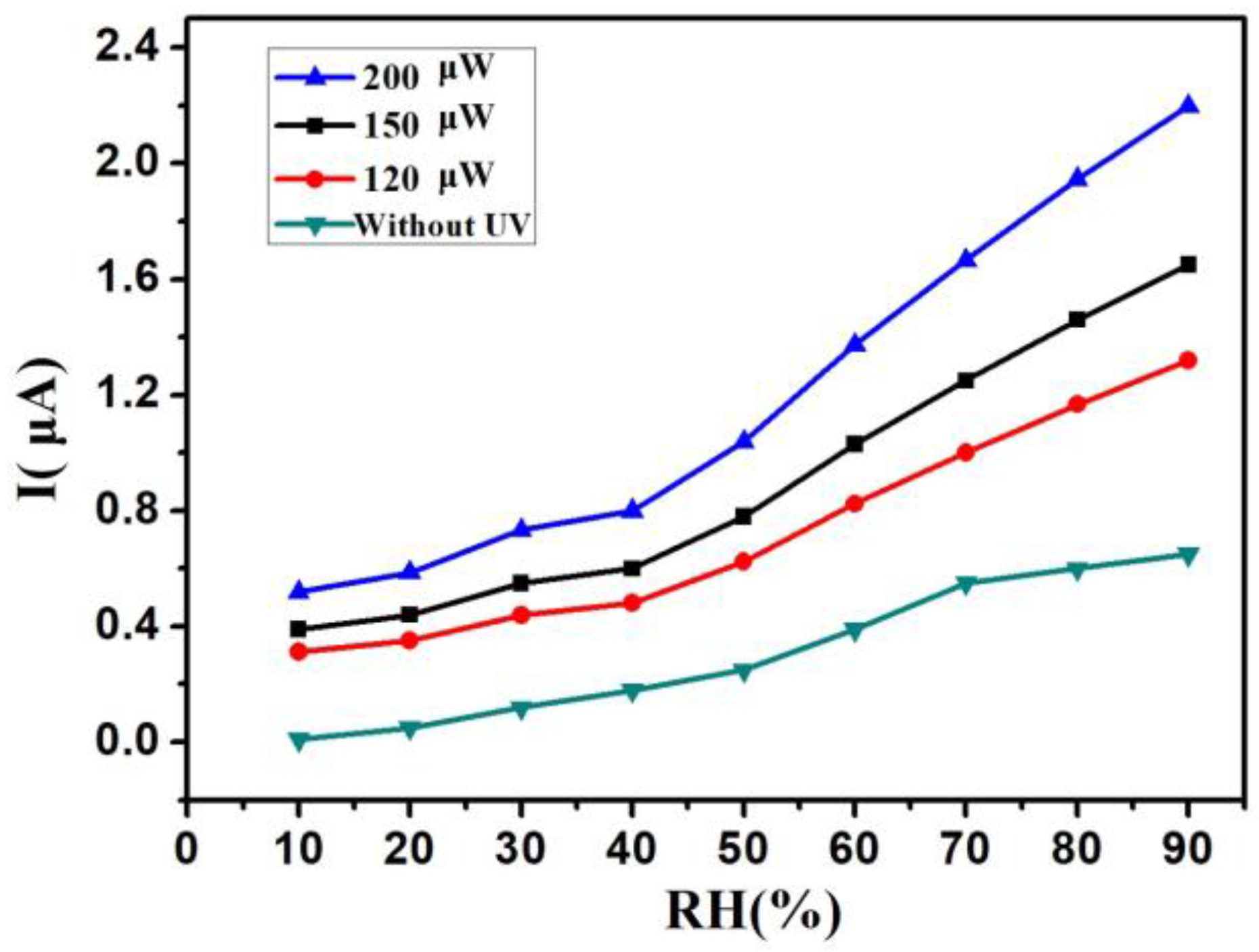
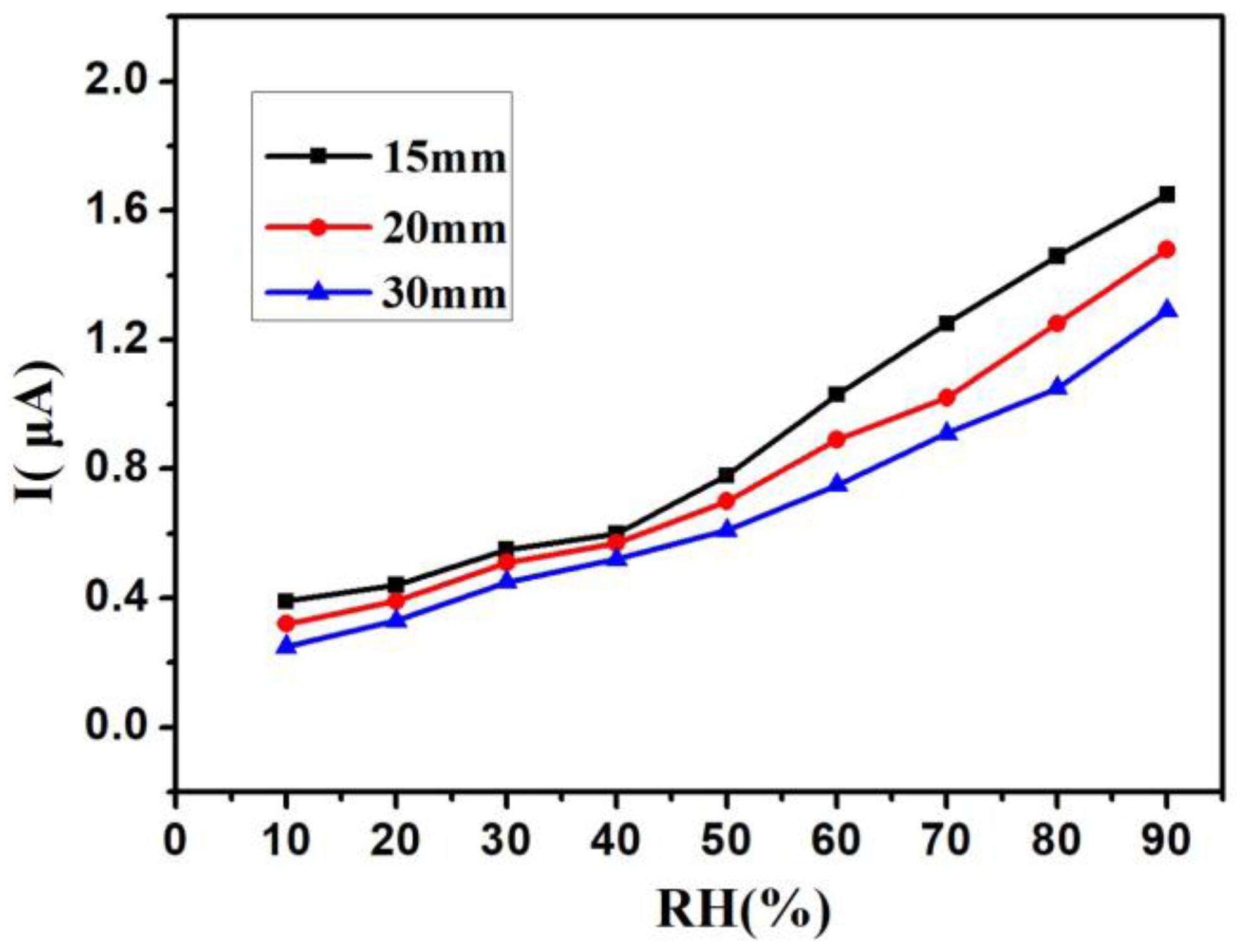
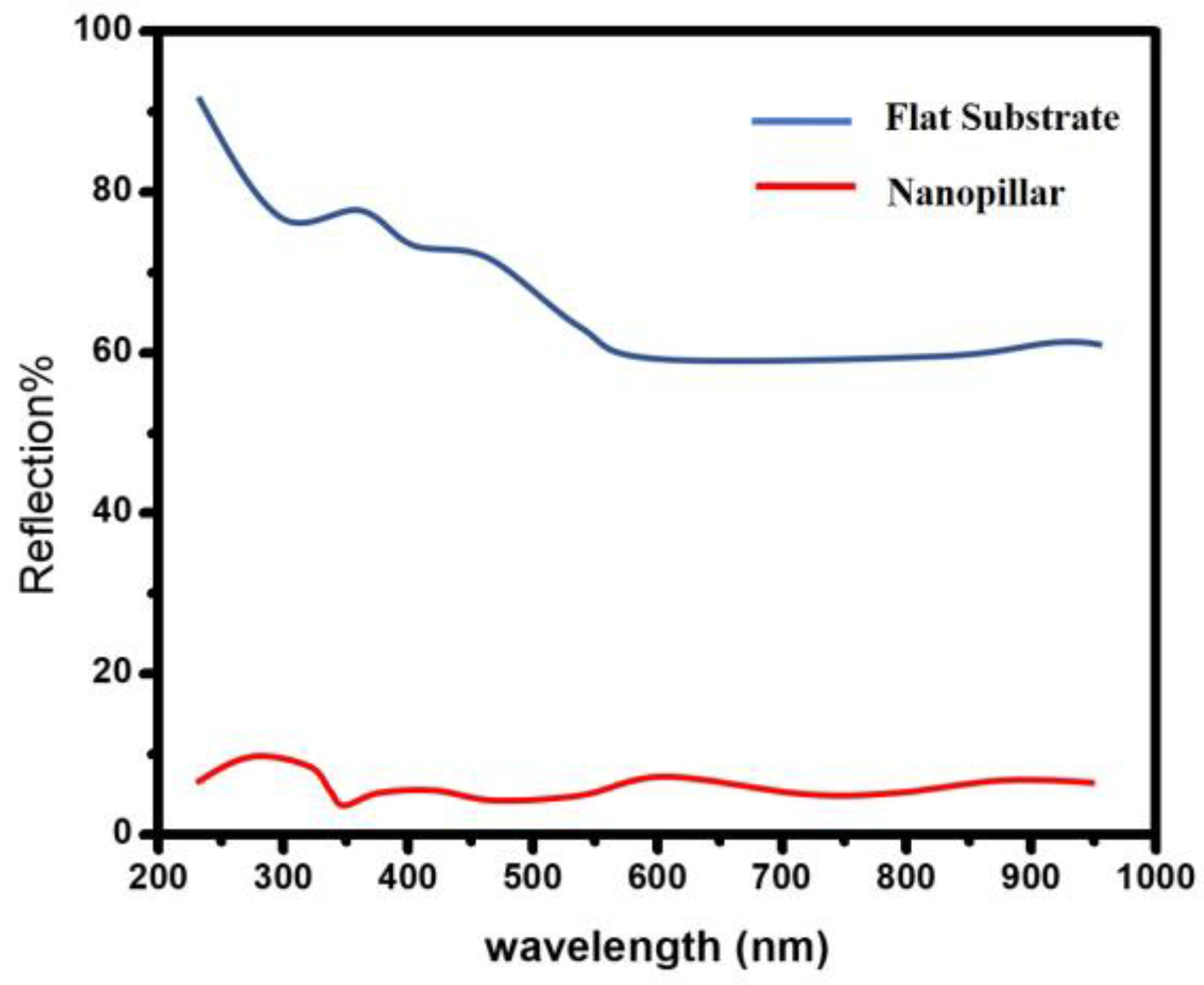
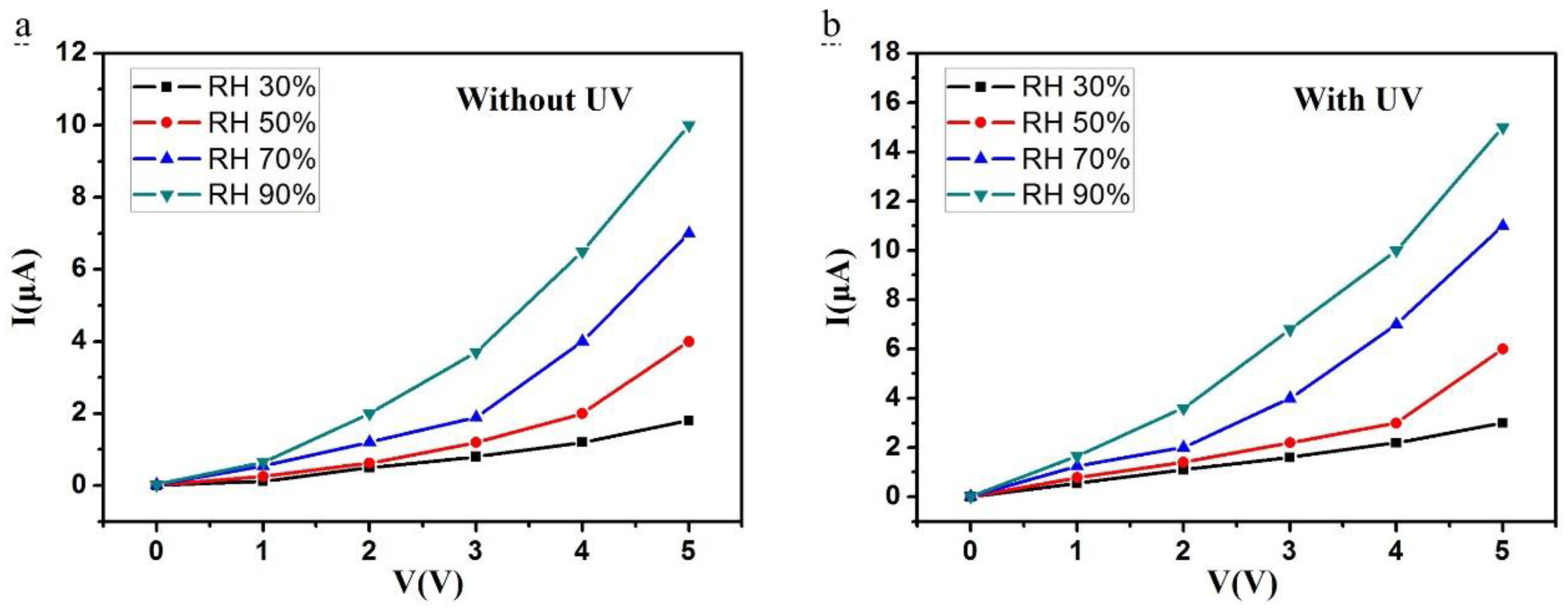
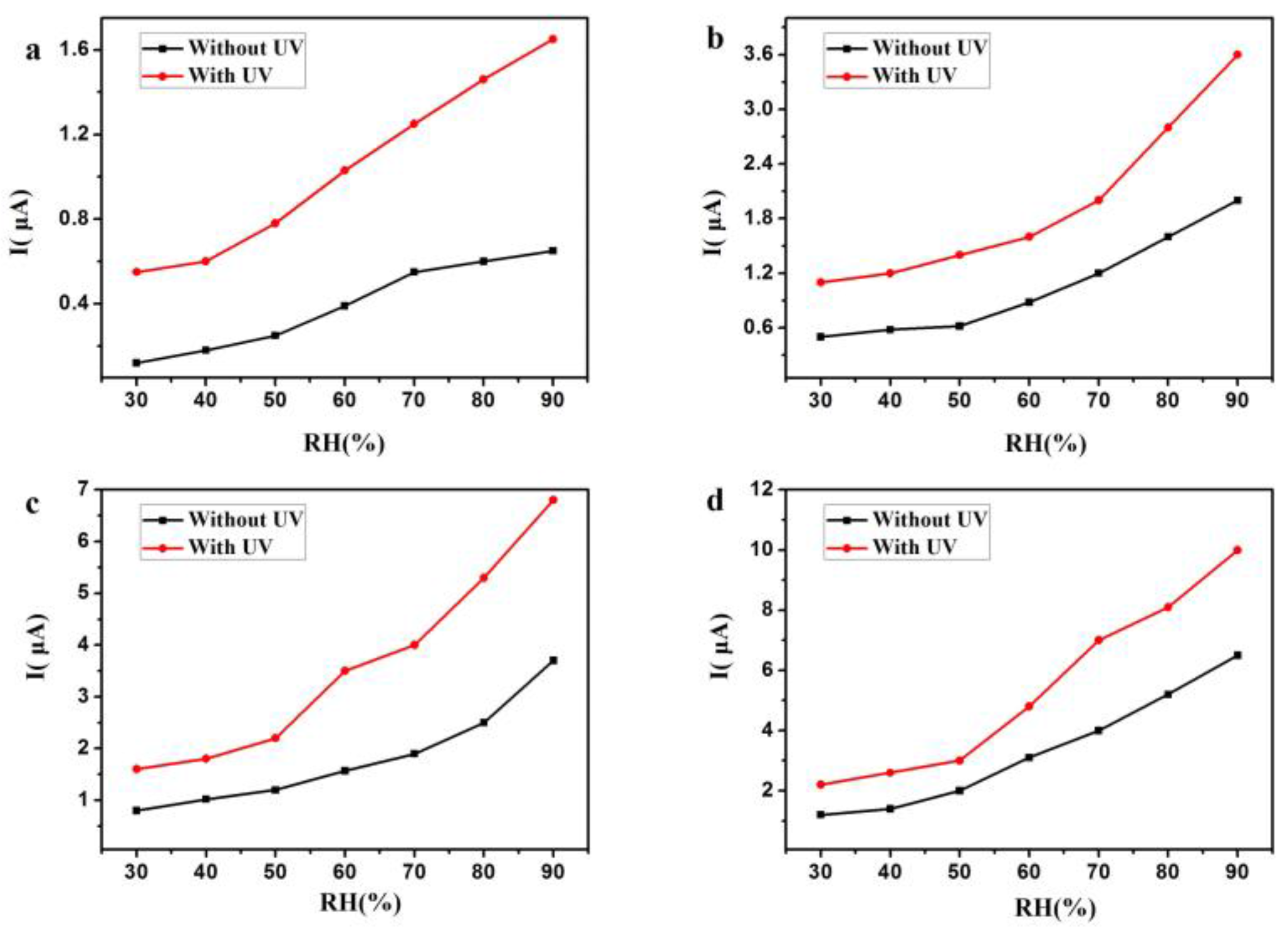
3. Results and Discussion
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Borini, S.; White, R.; Wei, D. Ultrafast Graphene Oxide Humidity Sensors. ACS Nano 2013, 7, 11166–11173. [Google Scholar] [CrossRef] [PubMed]
- Yeo, T.L.; Sun, T.; Grattan, K.T.V. Fibre-optic sensor technologies for humidity and moisture measurement. Sens. Actuators A Phys. 2008, 144, 280–295. [Google Scholar] [CrossRef]
- Qi, Q.; Zhang, T. Properties of humidity sensing ZnO nanorods-base sensor fabricated by screen-printing. Sens. Actuators B Chem. 2008, 133, 638–643. [Google Scholar] [CrossRef]
- Farahani, H.; Wagiran, R.; Hamidon, M. Humidity Sensors Principle, Mechanism, and Fabrication Technologies: A Comprehensive Review. Sensors 2014, 14, 7881–7939. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Park, S.; Yeow, J.T.W. A capacitive humidity sensor based on ordered macroporous silicon with thin film surface coating. Sens. Actuators B Chem. 2010, 149, 136–142. [Google Scholar] [CrossRef]
- Duy, T.P.; Chung, G.S. Effects of rapid thermal annealing on humidity sensor based on graphene oxide thin films. Sens. Actuators B Chem. 2015, 220, 1050–1055. [Google Scholar]
- Burman, D.; Ghosh, R.; Santra, S. Highly proton conducting MoS2/graphene oxide nanocomposite based chemoresistive humidity sensor. RSC Adv. 2016, 6, 57424. [Google Scholar] [CrossRef]
- Jha, R.K.; Guha, P.K. Liquid exfoliated pristine WS2 nanosheets for ultrasensitive and highly stable chemiresistive humidity sensors. Nanotechnology 2016, 27, 475503. [Google Scholar] [CrossRef] [PubMed]
- Georgieva, B.; Podolesheva, I.; Spasov, G. Nanosized Thin SnO2 Layers Doped with Te and TeO2 as Room Temperature Humidity Sensors. Sensors 2014, 14, 8950–8960. [Google Scholar] [CrossRef] [PubMed]
- Song, X.; Qi, Q.; Zhang, T. A humidity sensor based on KCl-doped SnO2 nanofibers. Sens. Actuators B Chem. 2009, 138, 368–373. [Google Scholar] [CrossRef]
- Tomer, V.K.; Duhan, S. Highly sensitive and stable relative humidity sensors based on WO3 modified mesoporous silica. Appl. Phys. Lett. 2015, 106, 063105. [Google Scholar] [CrossRef]
- Staerz, A.; Berthold, C.; Russ, T. The oxidizing effect of humidity on WO3 based sensors. Sens. Actuators B Chem. 2016, 237, 54–58. [Google Scholar] [CrossRef]
- Hsu, C.L.; Su, I.L.; Hsueh, T.J. Tunable Schottky contact humidity sensor based on S-doped ZnO nanowires on flexible PET substrate with piezotronic effect. J. Alloys Compd. 2017, 705, 722–733. [Google Scholar] [CrossRef]
- Mahjoub, M.A.; Monier, G.; Robert-Goumet, C. Synthesis and Study of Stable and Size-Controlled ZnO-SiO2 Quantum Dots: Application as a Humidity Sensor. J. Phys. Chem. C 2016, 120, 11652–11662. [Google Scholar] [CrossRef]
- Buvailo, A.I.; Xing, Y.; Hines, J. TiO2/LiCl-Based Nanostructured Thin Film for Humidity Sensor Applications. ACS Appl. Mater. Interfaces 2011, 3, 528–533. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Pan, Y.Z.; Huang, S.S. Resistive and capacitive response of nitrogen-doped TiO2 nanotubes film humidity sensor. Nanotechnology 2011, 22, 025501. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Kolmakov, A. Electronic Control of Chemistry and Catalysis at the Surface of an Individual Tin Oxide Nanowire. J. Phys. Chem. B 2005, 109, 1923–1929. [Google Scholar] [CrossRef] [PubMed]
- Kolmakov, A.; Chen, X.H.; Moskovits, M. Functionalizing Nanowires with Catalytic Nanoparticles for Gas Sensing Application. J. Nanosci. Nanotechnol. 2008, 8, 111–121. [Google Scholar] [CrossRef] [PubMed]
- da Silva, L.F.; M’Peko, J.C.; Catto, A.C.; Bernardini, S.; Mastelaro, V.R.; Aguir, K.; Ribeiro, C.; Longo, E. UV-enhanced ozone gas sensing response of ZnO-SnO2 heterojunctions at room temperature. Sens. Actuators B Chem. 2017, 240, 573–579. [Google Scholar] [CrossRef]
- Nguyen, D.C.; Nguyen, D.Q. NO gas sensing kinetics at room temperature under UV light irradiation of In2O3 nanostructures. Sci. Rep. 2016, 6, 35066. [Google Scholar]
- Park, S.; An, S.; Mun, Y. UV-Enhanced NO2 Gas Sensing Properties of SnO2-Core/ZnO-Shell Nanowires at Room Temperature. ACS Appl. Mater. Interfaces 2013, 5, 4285–4292. [Google Scholar] [CrossRef] [PubMed]
- Gong, J.; Li, Y.; Chai, X. UV-Light-Activated ZnO Fibers for Organic Gas Sensing at Room Temperature. J. Phys. Chem. C 2010, 114, 1293–1298. [Google Scholar] [CrossRef]
- Li, W.; Hu, M.Y. Humidity sensing properties of morphology-controlled ordered silicon nanopillar. Appl. Surf. Sci. 2014, 317, 970–973. [Google Scholar] [CrossRef]
- Li, W.; Dai, E.; Bai, G. Depth-dependent humidity sensing properties of silicon nanopillar array. Sens. Actuators B Chem. 2016, 237, 526–533. [Google Scholar] [CrossRef]
- Li, W.; Feng, Z.; Dai, E. Organic vapour sensing properties of area-ordered and size-controlled silicon nanopillar. Sensors 2016, 16, 1880. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Liu, J. Fabrication of Ordered SnO2 Nanostructures with Enhanced Humidity Sensing Performance. Sensors 2017, 17, 2392. [Google Scholar] [CrossRef] [PubMed]

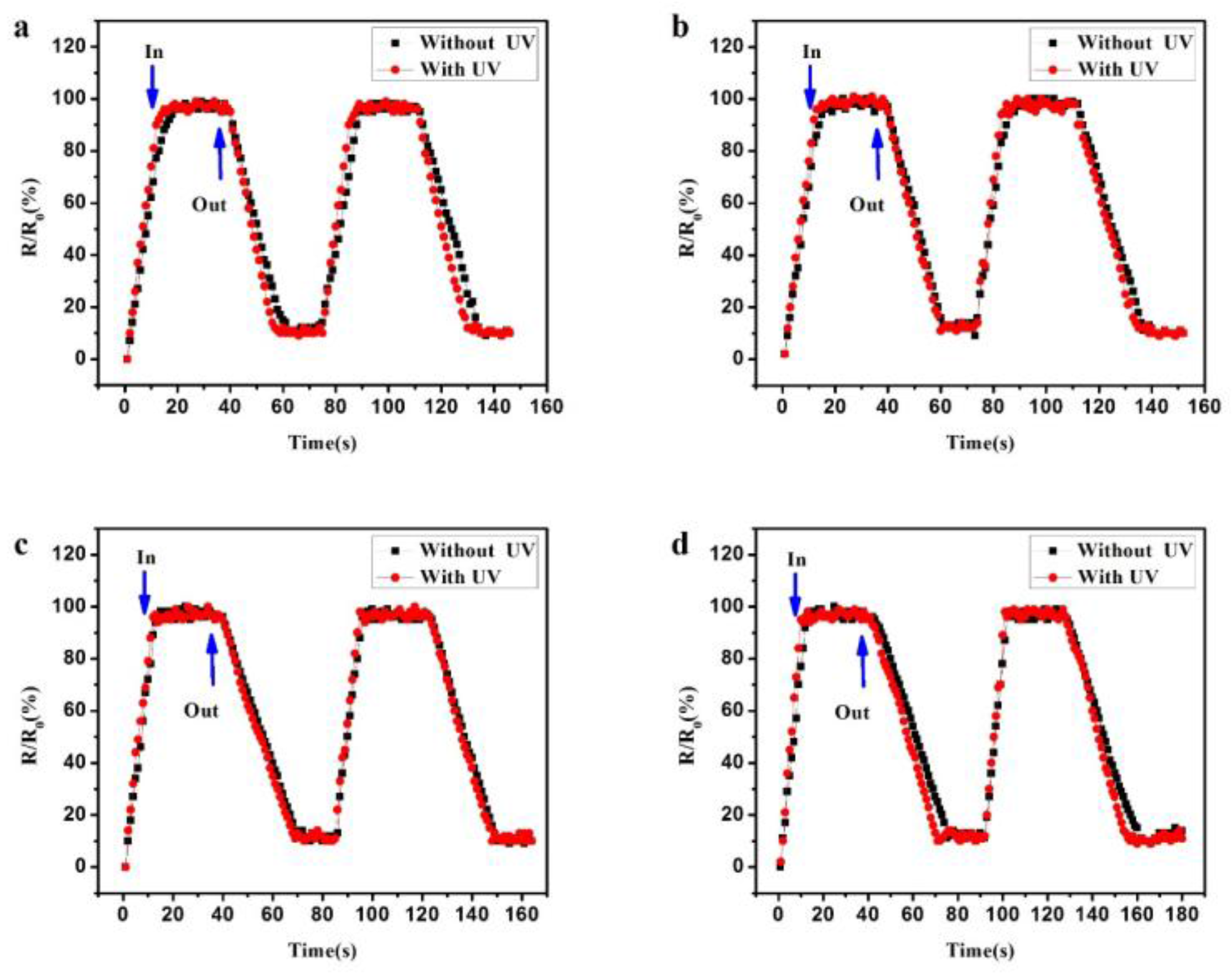
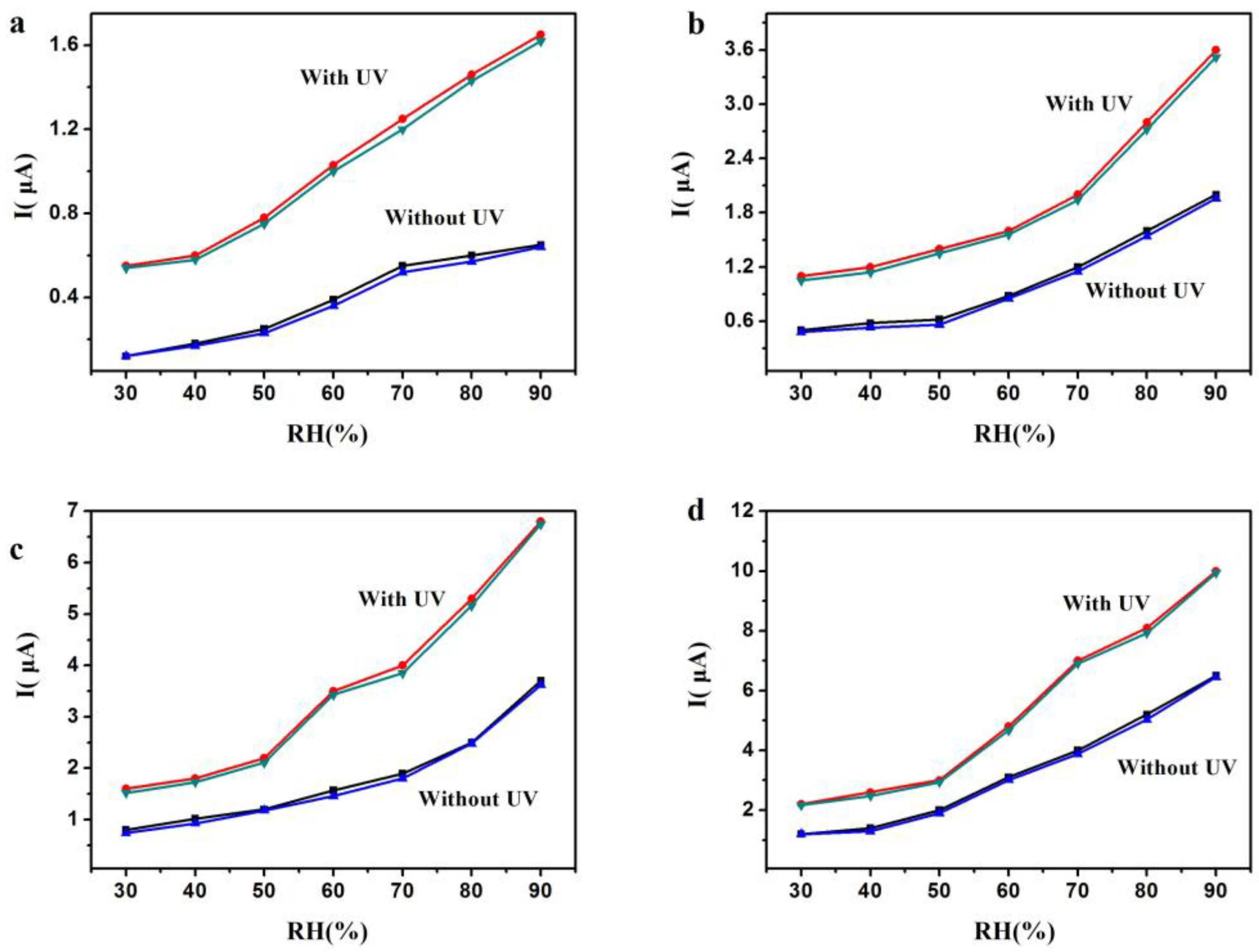
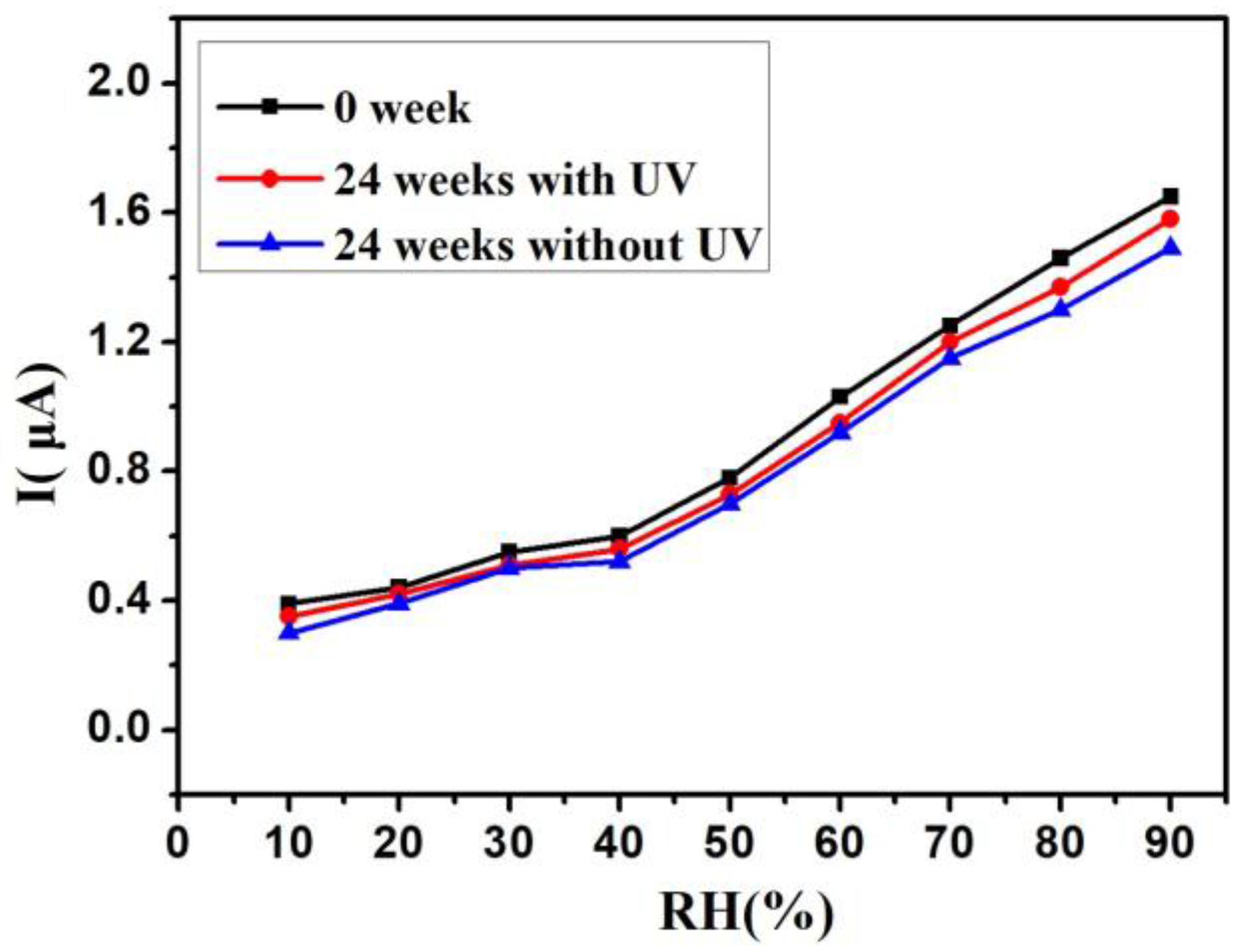
| RH | 30% | 50% | 70% | 90% | |
|---|---|---|---|---|---|
| Without UV | Response time | 17 s | 15 s | 14 s | 12 s |
| Recovery time | 23 s | 26 s | 30 s | 35 s | |
| With UV | Response time | 15 s | 13 s | 11 s | 10 s |
| Recovery time | 20 s | 23 s | 27 s | 31 s | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, W.; Ding, C.; Cai, Y.; Liu, J.; Wang, L.; Ren, Q.; Xu, J. Enhanced Humidity Sensitivity with Silicon Nanopillar Array by UV Light. Sensors 2018, 18, 660. https://doi.org/10.3390/s18020660
Li W, Ding C, Cai Y, Liu J, Wang L, Ren Q, Xu J. Enhanced Humidity Sensitivity with Silicon Nanopillar Array by UV Light. Sensors. 2018; 18(2):660. https://doi.org/10.3390/s18020660
Chicago/Turabian StyleLi, Wei, Chao Ding, Yun Cai, Juyan Liu, Linlin Wang, Qingying Ren, and Jie Xu. 2018. "Enhanced Humidity Sensitivity with Silicon Nanopillar Array by UV Light" Sensors 18, no. 2: 660. https://doi.org/10.3390/s18020660
APA StyleLi, W., Ding, C., Cai, Y., Liu, J., Wang, L., Ren, Q., & Xu, J. (2018). Enhanced Humidity Sensitivity with Silicon Nanopillar Array by UV Light. Sensors, 18(2), 660. https://doi.org/10.3390/s18020660




