Sensor-Based Assessment of Soil Salinity during the First Years of Transition from Flood to Sprinkler Irrigation
Abstract
1. Introduction
2. Materials and Methods
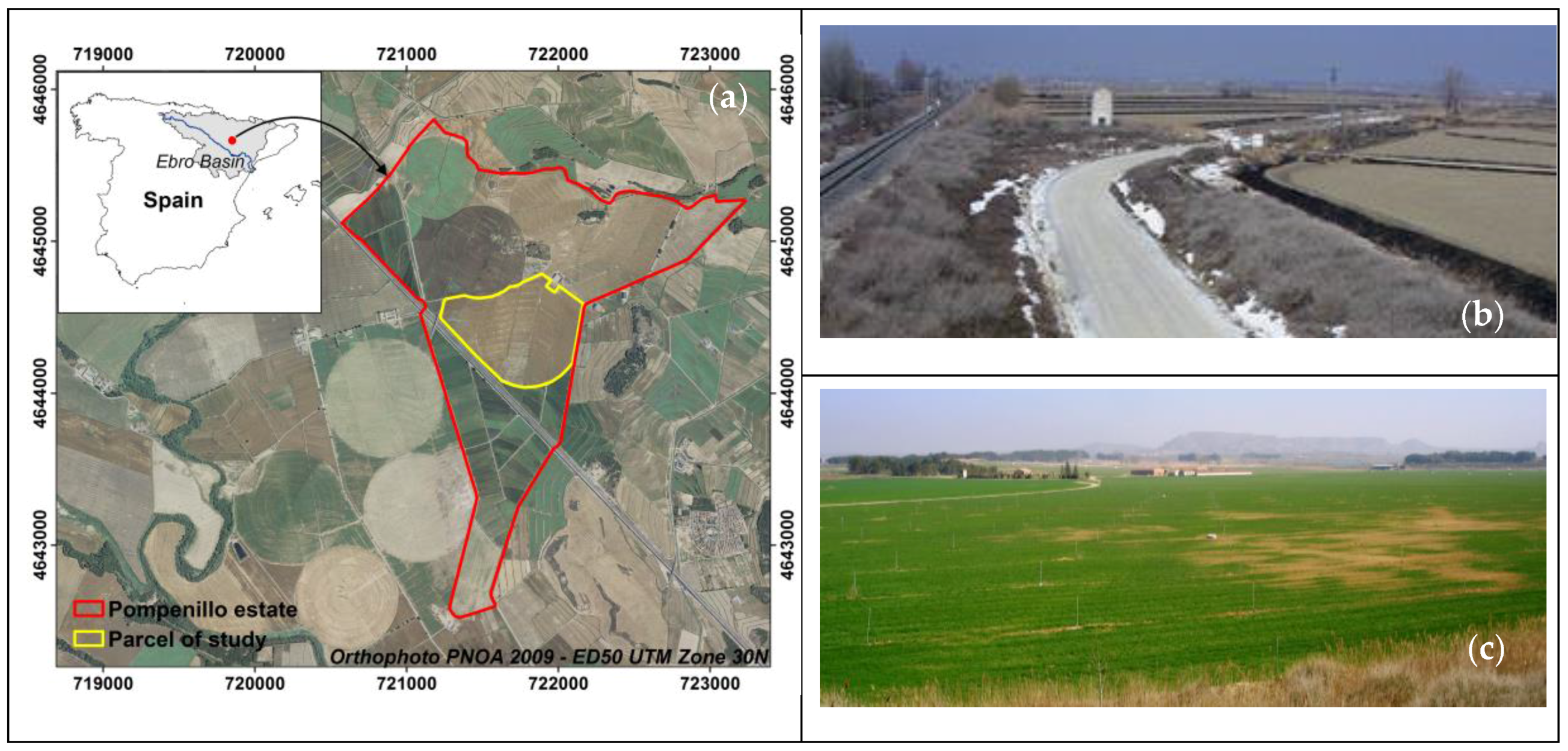
2.1. Study Area
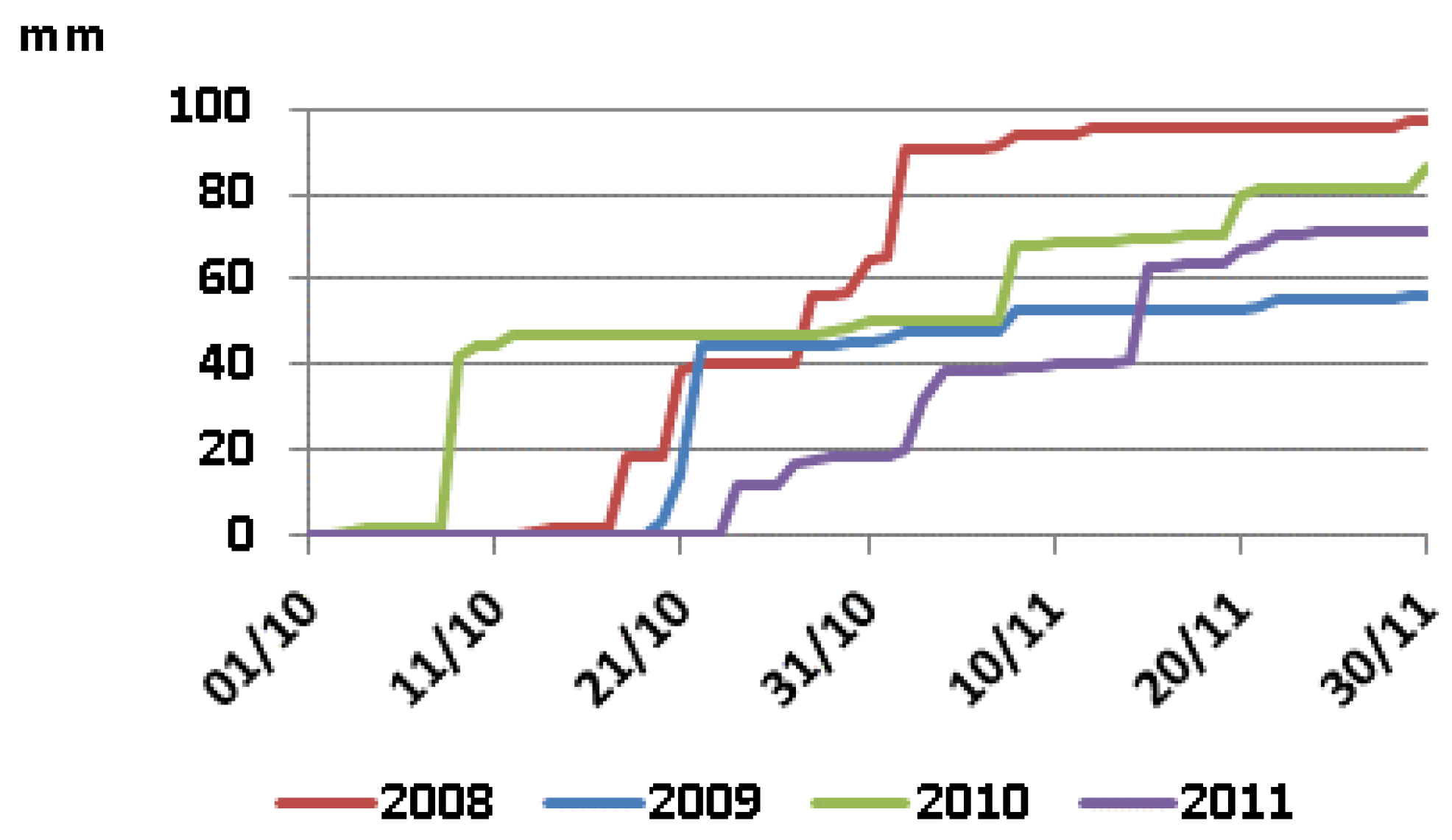
2.2. EMI Readings and Acquisitions of Soil Samples
2.3. EMI Readings Calibrations
2.4. Soil Salinity Mapping
2.5. Vegetative Activity
3. Results
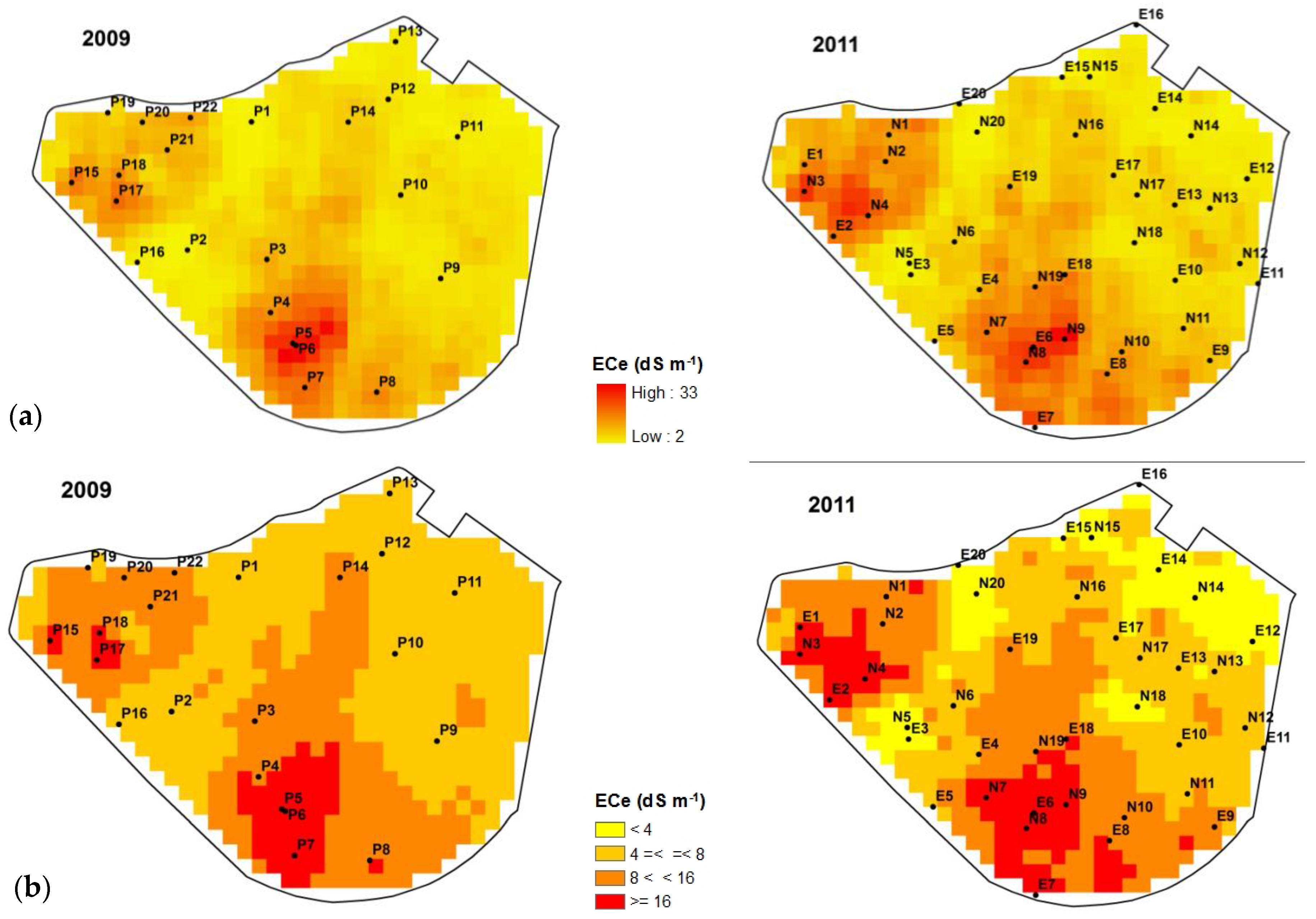
3.1. Soil Salinity
3.2. Vegetative Activity
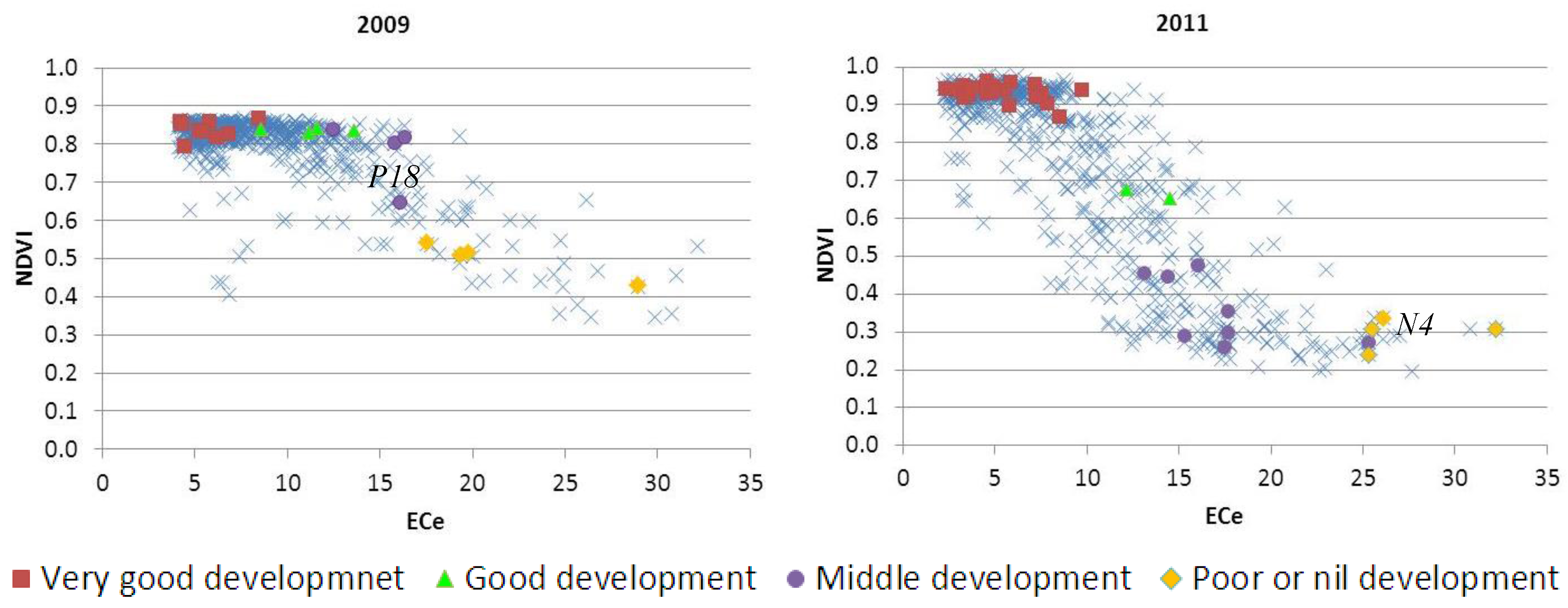
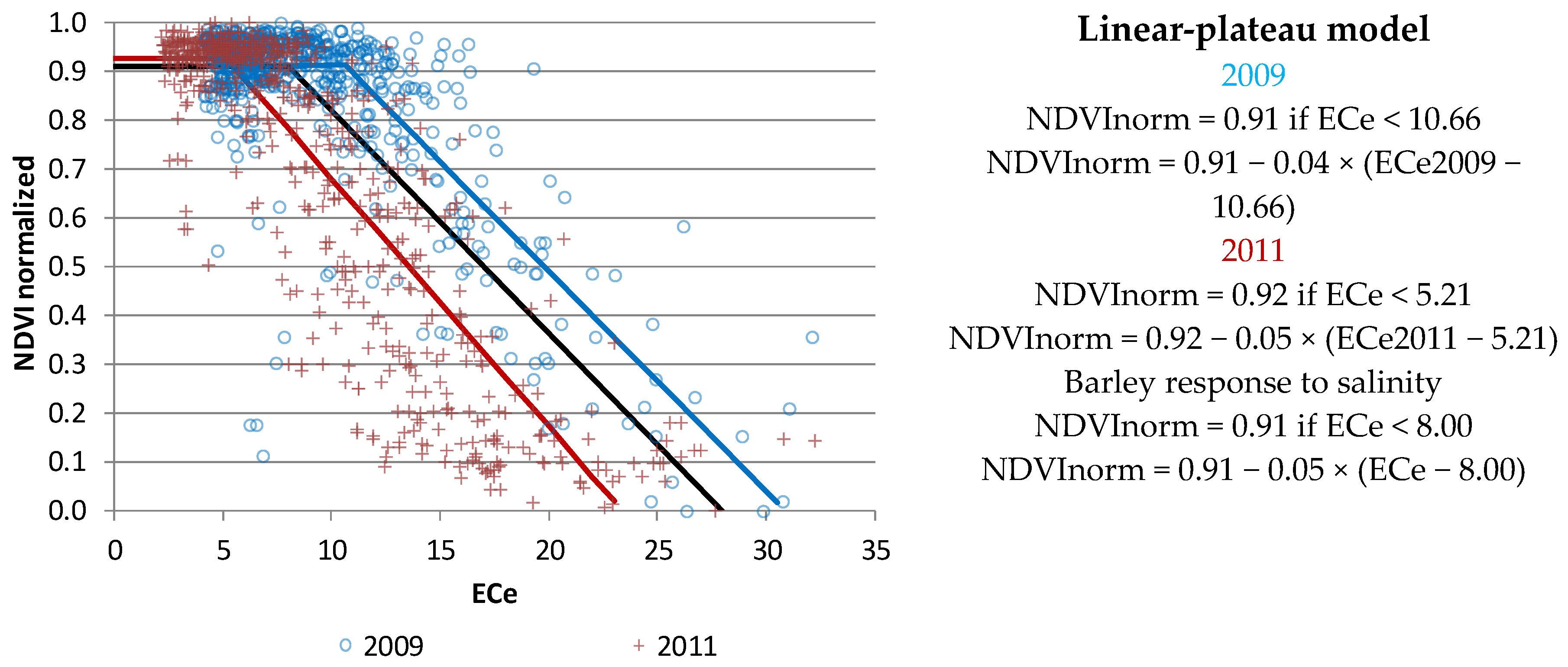
3.3. Relationship between Vegetative Activity and Soil Salinity
4. Discussion
4.1. Vegetative Activity and Its Relationship with Soil Salinity
4.2. Integration in Agricultural Management
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Cai, X.M.; McKinney, D.C.; Rosegrant, M.W. Sustainability analysis for irrigation water management in the Aral Sea region. Agric. Syst. 2003, 76, 1043–1066. [Google Scholar] [CrossRef]
- Marshall, G.R.; Jones, R.E. Significance of supply response for estimating agricultural costs of soil salinity. Agric. Syst. 1997, 53, 231–252. [Google Scholar] [CrossRef]
- Qadir, M.; Quillerou, E.; Nangia, V.; Murtaza, G.; Singh, M.; Thomas, R.J.; Drechsel, P.; Noble, A.D. Economics of salt-induced land degradation and restoration. Nat. Resour. Forum 2014, 38, 282–295. [Google Scholar] [CrossRef]
- Zekri, S.; Albisu, L.M. Economic-impact of soil-salinity in agriculture. A case study of Bardenas area, Spain. Agric. Syst. 1993, 41, 369–386. [Google Scholar] [CrossRef]
- Feijoo, M.L.; Calvo, E.; Albiac, J. Economic and environmental policy analysis of the Flumen-Monegros irrigation system in Huesca, Spain. Geogr. Anal. 2000, 32, 187–204. [Google Scholar] [CrossRef]
- Esteban, E.; Albiac, J. Salinity pollution control in the presence of farm heterogeneity: An empirical analysis. Water Econ. Policy 2016, 2, 20. [Google Scholar] [CrossRef]
- Grunwald, S.; Vasques, G.M.; Rivero, R.G. Fusion of soil and remote sensing data to model soil properties. Adv. Agron. 2015, 131, 1–109. [Google Scholar] [CrossRef]
- Corwin, D.L.; Lesch, S.M. Apparent soil electrical conductivity measurements in agriculture. Comput. Electron. Agric. 2005, 46, 11–43. [Google Scholar] [CrossRef]
- Nogués, J.; Robinson, D.A.; Herrero, J. Incorporating electromagnetic induction methods into regional soil salinity survey of irrigation districts. Soil Sci. Soc. Am. J. 2006, 70, 2075–2085. [Google Scholar] [CrossRef]
- Guo, Y.; Huang, J.; Shi, Z.; Li, H. Mapping spatial variability of soil salinity in a coastal paddy field based on electromagnetic sensors. PLoS ONE 2015, 10, e0127996. [Google Scholar] [CrossRef] [PubMed]
- Triantafilis, J.; Ahmed, M.F.; Odeh, I.O.A. Application of a mobile electromagnetic sensing system (MESS) to assess cause and management of soil salinization in an irrigated cotton-growing field. Soil Use Manag. 2002, 18, 330–339. [Google Scholar] [CrossRef]
- Urdanoz, V.; Amezketa, E.; Clavería, I.; Ochoa, V.; Aragüés, R. Mobile and georeferenced electromagnetic sensors and applications for salinity assessment. Span. J. Agric. Res. 2008, 6, 469–478. [Google Scholar] [CrossRef]
- Metternicht, G.I.; Zinck, J.A. Remote sensing of soil salinity: Potentials and constraints. Remote Sens. Environ. 2003, 85, 1–20. [Google Scholar] [CrossRef]
- Muller, S.J.; van Niekerk, A. An evaluation of supervised classifiers for indirectly detecting salt-affected areas at irrigation scheme level. Int. J. Appl. Earth Obs. Geoinf. 2016, 49, 138–150. [Google Scholar] [CrossRef]
- Dominguez-Beisiegel, M.; Castañeda, C.; Mougenot, B.; Herrero, J. Analysis and mapping of the spectral characteristics of fractional green cover in salinewetlands (NE Spain) using field and remote sensing data. Remote Sens. 2016, 8, 590. [Google Scholar] [CrossRef]
- Gorji, T.; Sertel, E.; Tanik, A. Recent satellite technologies for soil salinity assessment with special focus on mediterranean countries. Fresenius Environ. Bull. 2017, 26, 196–203. [Google Scholar]
- Eldeiry, A.A.; Garcia, L.A.; Reich, R.M. Soil salinity sampling strategy using spatial modeling techniques, remote sensing, and field data. J. Irrig. Drain. Eng. ASCE 2008, 134, 768–777. [Google Scholar] [CrossRef]
- Lobell, D.B.; Ortíz-Monasterio, J.I.; Gurrola, F.C.; Valenzuela, L. Identification of saline soils with multiyear remote sensing of crop yields. Soil Sci. Soc. Am. J. 2007, 71, 777–783. [Google Scholar] [CrossRef]
- Odeh, I.O.A.; Onus, A. Spatial analysis of soil salinity and soil structural stability in a semiarid region of New South Wales, Australia. Environ. Manag. 2008, 42, 265–278. [Google Scholar] [CrossRef] [PubMed]
- Guo, W.; Maas, S.J.; Bronson, K.F. Relationship between cotton yield and soil electrical conductivity, topography, and landsat imagery. Precis. Agric. 2012, 13, 678–692. [Google Scholar] [CrossRef]
- Alexakis, D.D.; Daliakopoulos, I.N.; Panagea, I.S.; Tsanis, I.K. Assessing soil salinity using WorldView-2 multispectral images in Timpaki, Crete, Greece. Geocarto Int. 2018, 33, 321–338. [Google Scholar] [CrossRef]
- Muller, S.J.; van Niekerk, A. Identification of WorldView-2 spectral and spatial factors in detecting salt accumulation in cultivated fields. Geoderma 2016, 273, 1–11. [Google Scholar] [CrossRef]
- Vermeulen, D.; Van Niekerk, A. Evaluation of a WorldView-2 image for soil salinity monitoring in a moderately affected irrigated area. J. Appl. Remote Sens. 2016, 10, 026025. [Google Scholar] [CrossRef]
- Wu, J.; Vincent, B.; Yang, J.; Bouarfa, S.; Vidal, A. Remote sensing monitoring of changes in soil salinity: A case study in inner Mongolia, China. Sensors 2008, 8, 7035–7049. [Google Scholar] [CrossRef] [PubMed]
- Singh, G.; Bundela, D.S.; Sethi, M.; Lal, K.; Kamra, S.K. Remote sensing and geographic information system for appraisal of salt-affected soils in India. J. Environ. Qual. 2010, 39, 5–15. [Google Scholar] [CrossRef] [PubMed]
- Bouaziz, M.; Matschullat, J.; Gloaguen, R. Improved remote sensing detection of soil salinity from a semi-arid climate in northeast Brazil. C. R. Geosci. 2011, 343, 795–803. [Google Scholar] [CrossRef]
- Jin, X.M.; Vekerdy, Z.; Zhang, Y.K.; Liu, J.T. Soil salt content and its relationship with crops and groundwater depth in the Yinchuan plain (China) using remote sensing. Arid Land Res. Manag. 2012, 26, 227–235. [Google Scholar] [CrossRef]
- Pakparvar, M.; Gabriels, D.; Aarabi, K.; Edraki, M.; Raes, D.; Cornelis, W. Incorporating legacy soil data to minimize errors in salinity change detection: A case study of Darab plain, Iran. Int. J. Remote Sens. 2012, 33, 6215–6238. [Google Scholar] [CrossRef]
- Abbas, A.; Khan, S.; Hussain, N.; Hanjra, M.A.; Akbar, S. Characterizing soil salinity in irrigated agriculture using a remote sensing approach. Phys. Chem. Earth 2013, 55–57, 43–52. [Google Scholar] [CrossRef]
- Ivits, E.; Cherlet, M.; Tóth, T.; Lewińska, K.E.; Tóth, G. Characterisation of productivity limitation of salt-affected lands in different climatic regions of Europe using remote sensing derived productivity indicators. Land Degrad. Dev. 2013, 24, 438–452. [Google Scholar] [CrossRef]
- Wu, W.; Al-Shafie, W.M.; Mhaimeed, A.S.; Ziadat, F.; Nangia, V.; Payne, W.B. Soil salinity mapping by multiscale remote sensing in Mesopotamia, Iraq. IEEE J. Sel. Top. Appl. Earth Obs. Remote Sens. 2014, 7, 4442–4452. [Google Scholar] [CrossRef]
- Scudiero, E.; Skaggs, T.H.; Corwin, D.L. Regional-scale soil salinity assessment using Landsat ETM plus canopy reflectance. Remote Sens. Environ. 2015, 169, 335–343. [Google Scholar] [CrossRef]
- López-Lozano, R.; Casterad, M.A.; Herrero, J. Site-specific management units in a commercial maize plot delineated using very high resolution remote sensing and soil properties mapping. Comput. Electron. Agric. 2010, 73, 219–229. [Google Scholar] [CrossRef]
- Luz, P.B.; Heermann, D. A statistical approach to estimating runoff in center pivot irrigation with crust conditions. Agric. Water Manag. 2005, 72, 33–46. [Google Scholar] [CrossRef]
- Silva, L.L. Fitting infiltration equations to centre-pivot irrigation data in a mediterranean soil. Agric. Water Manag. 2007, 94, 83–92. [Google Scholar] [CrossRef]
- Salvador, R.; Martínez-Cob, A.; Cavero, J.; Playán, E. Seasonal on-farm irrigation performance in the Ebro basin (Spain): Crops and irrigation systems. Agric. Water Manag. 2011, 98, 577–587. [Google Scholar] [CrossRef]
- Rengasamy, P.; Olsson, K.A. Sodicity and soil structure. Aust. J. Soil Res. 1991, 29, 935–952. [Google Scholar] [CrossRef]
- Rodríguez, R.; Herrero, J.; Porta, J. Micromorphological assessment of drain siltation risk indexes in a saline sodic soil in Monegros irrigation district (Spain). In Soil Micromorphology: A Basic and Applied Science; Developments in Soil Science; Douglas, L.A., Ed.; Elsevier: Amsterdam, The Netherlands, 1990; Volume 19, pp. 41–52. ISBN 0-444-88302-9. [Google Scholar]
- Vizcayno, C.; García-González, M.T.; Gutiérrez, M.; Rodríguez, R. Mineralogical, chemical and morphological features of salt accumulations in the Flumen-Monegros district, NE Spain. Geoderma 1995, 68, 193–210. [Google Scholar] [CrossRef]
- Nogués, J.; Herrero, J.; Rodríguez-Ochoa, R.; Boixadera, J. Land evaluation in a salt-affected irrigated district using an index of productive potential. Environ. Manag. 2000, 25, 143–152. [Google Scholar] [CrossRef] [PubMed]
- Herrero, J.; Castañeda, C. Temporal changes in soil salinity at four saline wetlands in NE Spain. Catena 2015, 133, 145–156. [Google Scholar] [CrossRef]
- García-González, M.T. Fine-grained laminated quaternary sediments in the Ebro Valley (Spain): Characteristics and formation. Clay Miner. 1996, 31, 173–181. [Google Scholar] [CrossRef]
- Herrero, J.; Castañeda, C. A paddy on sodic varved sediment and plant life constraints, NE Spain. Catena 2018, 164, 56–61. [Google Scholar] [CrossRef]
- Playán, E.; Pérez-Coveta, O.; Martínez-Cob, A.; Herrero, J.; García-Navarro, P.; Latorre, B.; Brufau, P.; Garcés, J. Overland water and salt flows in a set of rice paddies. Agric. Water Manag. 2008, 95, 645–658. [Google Scholar] [CrossRef]
- Betrán, J.A. Mejora de los suelos de la finca Pompenillo. In Proyecto Fin de Carrera; Escuela Técnica Superior de Ingenieros Agrónomos: Lérida, Spain, 1986. [Google Scholar]
- Abdu, H.; Robinson, D.A.; Jones, S.B. Comparing bulk soil electrical conductivity determination using the Dualem-1S and EM38-DD electromagnetic induction instruments. Soil Sci. Soc. Am. J. 2007, 71, 189–196. [Google Scholar] [CrossRef]
- United States Salinity Laboratory Staff. Diagnosis and improvement of saline and alkali soils. In Agriculture Handbook; United States Department of Agriculture: Washington, DC, USA, 1954; Volume 60, p. 160. [Google Scholar]
- Ayers, R.S.; Westcot, D.W. Water Quality for Agriculture; Irrigation and Drainage Paper 29, Rev. 1; FAO: Rome, Italy, 1985; p. 97. [Google Scholar]
- Díaz, L.; Herrero, J. Salinity estimates in irrigated soils using electromagnetic induction. Soil Sci. 1992, 154, 151–157. [Google Scholar] [CrossRef]
- López-Bruna, D.; Herrero, J. El comportamiento del sensor electromagnético y su calíbracíón frente a la salinidad edáfica. Agronomie 1996, 16, 95–105. [Google Scholar] [CrossRef]
- Herrero, J.; Ba, A.A.; Aragüés, R. Soil salinity and its distribution determined by soil sampling and electromagnetic techniques. Soil Use Manag. 2003, 19, 119–126. [Google Scholar] [CrossRef]
- Herrero, J.; Hudnall, W.H. Measurement of soil salinity using electromagnetic induction in a paddy with a densic pan and shallow water table. Paddy Water Environ. 2014, 12, 263–274. [Google Scholar] [CrossRef]
- Herrero, J.; Netthisinghe, A.; Hudnall, W.H.; Pérez-Coveta, O. Electromagnetic induction as a basis for soil salinity monitoring within a mediterranean irrigation district. J. Hydrol. 2011, 405, 427–438. [Google Scholar] [CrossRef]
- Bower, C.A.; Huss, R.B. Rapid conductometric method for estimating gypsum in soils. Soil Sci. 1948, 66, 199–204. [Google Scholar] [CrossRef]
- Kunze, G.W. Pretreatment for mineralogical analysis. In Methods of Soil Analysis. Part 1. Physical and Mineralogical Properties, Including Statistics of Measurement and Sampling. Agronomy Monograph 9.1; Black, C.A., Ed.; ASA, SSSA: Madison, WI, USA, 1965; pp. 568–577. [Google Scholar] [CrossRef]
- Artieda, O.; Herrero, J.; Drohan, P.J. Refinement of the differential water loss method for gypsum determination in soils. Soil Sci. Soc. Am. J. 2006, 70, 1932–1935. [Google Scholar] [CrossRef]
- Soil Survey Division Staff. Soil Survey Manual; Handbook 18; Natural Resources Conservation Service, USDA: Washington, DC, USA, 1993.
- Amezketa, E.; Urdanoz, V.; Barinagarrementeria, I.; Albizua, L.; Berkane, Y.; Portero, C.; Casterad, M.A. Validación de índices espectrales para detectar salinidad edáfica en cebada mediante sensores electromagnéticos terrestres. In Teledetección: Bosques y Cambio Climático; DL:AS-3588-2011; Recondo, C., Pendás, E., Eds.; XIV Congreso de la Asociación Española de Teledetección: Asturias, Spain, 2011; pp. 277–280. [Google Scholar]
- Grieve, C.; Grattan, S.; Maas, E. Plant Salt Tolerance. In Agricultural Salinity Assessment and Management, 2nd ed.; Wallender, W.W., Tanji, K.K., Eds.; ASCE: Ritton, VA, USA, 2011; pp. 405–459. ISBN 978-0-7844-1169-8. [Google Scholar]
- Royo, A.; Aragüés, R. Validation of salinity crop production-functions obtained with the triple line source sprinkler system. Agron. J. 1993, 85, 795–800. [Google Scholar] [CrossRef]
- Martínez-Cob, A.; Aragüés, R.; Royo, A. Salt tolerance of barley (Hordeum vulgare L.) cultivars at the germination stage: Analysis of the response functions. Plant Soil 1987, 104, 53–56. [Google Scholar] [CrossRef]
- Maas, E.V.; Hoffman, G.J. Crop salt tolerance—Current assessment. J. Irrig. Drain. Eng. ASCE 1977, 103, 115–134. [Google Scholar]
- Van Genuchten, M.T.; Hoffman, G.J. Analysis of crop salt tolerance data. In Soil Salinity under Irrigation: Processes and Management; Ecology Studies; Shainberg, I., Shalhevet, J., Eds.; Springer-Verlag: New York, NY, USA, 1984; Volume 51, pp. 258–271. [Google Scholar]
- Cornacchione, M.V.; Suarez, D.L. Emergence, forage production, and ion relations of alfalfa in response to saline waters. Crop Sci. 2015, 55, 444–457. [Google Scholar] [CrossRef]
- Chu, L.; Kang, Y.; Wan, S. Influence of microsprinkler irrigation amount on water, soil, and pH profiles in a coastal saline soil. Sci. World J. 2014, 2014, 279895. [Google Scholar] [CrossRef] [PubMed]
- Dukes, M.D.; Perry, C. Uniformity testing of variable-rate center pivot irrigation control systems. Precis. Agric. 2006, 7, 205–218. [Google Scholar] [CrossRef]
- Yari, A.; Madramootoo, C.A.; Woods, S.A.; Adamchuk, V.I. Performance evaluation of constant versus variable rate irrigation. Irrig. Drain. 2017, 66, 501–509. [Google Scholar] [CrossRef]
- Hendriks, C.M.J.; Stoorvogel, J.J.; Claessens, L. Exploring the challenges with soil data in regional land use analysis. Agric. Syst. 2016, 144, 9–21. [Google Scholar] [CrossRef]



| Date of the Survey | Sites of EMI Reading | Soil Sampling | |||||
|---|---|---|---|---|---|---|---|
| Total | by ha | EMv < EMh, % | Number of Sites | Depth cm | Sampling Depth Interval, cm | Number of Samples | |
| 12 November 2009 | 3651 | 81 | 3.7 | 22 | 125 | 25 | 110 |
| 21 June 2011 | 2716 | 59 | 20.1 | 40 | 100 | 50 | 80 |
| Year | R2 % | S dS m−1 | Observations | |||
|---|---|---|---|---|---|---|
| Number | Kind | |||||
| 2009 | Equation (1) | EC1:5 = 0.46 + 0.50 × EMh | 87.7 | 0.35 | 22 | Sites |
| Equation (2) | ECe = −0.41 + 9.20 × EC1:5 | 90.2 | 3.64 | 24 | Soil samples | |
| 2011 | Equation (3) | ECe = 1.35 + 7.75 × EMh | 94.1 | 2.42 | 40 | Sites |
| ECe, dS m−1 | Salinity Phases | Percent Surface | |
|---|---|---|---|
| 2009 | 2011 | ||
| ECe < 4 | Non-saline or Slightly saline | 0.0 | 17.4 |
| 4 ≤ ECe < 8 | Moderately saline | 55.1 | 35.0 |
| 8 ≤ ECe < 16 | Strongly saline | 36.5 | 33.9 |
| 16 ≤ ECe | Very strongly saline | 8.4 | 13.7 |
| Group A | Group B | Group C | Group D | |||||
|---|---|---|---|---|---|---|---|---|
| Very Good | Good | Middle | Poor or Nil | |||||
| 2009 | 2011 | 2009 | 2011 | 2009 | 2011 | 2009 | 2011 | |
| No. of samples | 32 | 32 | 6 | 6 | 12 | 12 | 9 | 9 |
| Max NDVI | 0.87 | 0.96 | 0.85 | 0.80 | 0.85 | 0.51 | 0.54 | 0.34 |
| Min NDVI | 0.79 | 0.85 | 0.83 | 0.65 | 0.64 | 0.26 | 0.36 | 0.20 |
| Range | 0.07 | 0.11 | 0.01 | 0.14 | 0.21 | 0.25 | 0.18 | 0.14 |
| Median | 0.84 | 0.93 | 0.84 | 0.68 | 0.80 | 0.33 | 0.46 | 0.29 |
| Mean | 0.84 | 0.93 | 0.84 | 0.70 | 0.77 | 0.37 | 0.47 | 0.28 |
| Standard deviation | 0.02 | 0.03 | 0.01 | 0.05 | 0.07 | 0.09 | 0.06 | 0.05 |
| Coefficient of variation | 2.46 | 2.93 | 0.65 | 7.42 | 9.70 | 25.05 | 13.32 | 17.93 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Casterad, M.A.; Herrero, J.; Betrán, J.A.; Ritchie, G. Sensor-Based Assessment of Soil Salinity during the First Years of Transition from Flood to Sprinkler Irrigation. Sensors 2018, 18, 616. https://doi.org/10.3390/s18020616
Casterad MA, Herrero J, Betrán JA, Ritchie G. Sensor-Based Assessment of Soil Salinity during the First Years of Transition from Flood to Sprinkler Irrigation. Sensors. 2018; 18(2):616. https://doi.org/10.3390/s18020616
Chicago/Turabian StyleCasterad, Mª Auxiliadora, Juan Herrero, Jesús A. Betrán, and Glen Ritchie. 2018. "Sensor-Based Assessment of Soil Salinity during the First Years of Transition from Flood to Sprinkler Irrigation" Sensors 18, no. 2: 616. https://doi.org/10.3390/s18020616
APA StyleCasterad, M. A., Herrero, J., Betrán, J. A., & Ritchie, G. (2018). Sensor-Based Assessment of Soil Salinity during the First Years of Transition from Flood to Sprinkler Irrigation. Sensors, 18(2), 616. https://doi.org/10.3390/s18020616






