Flame-Oxidized Stainless-Steel Anode as a Probe in Bioelectrochemical System-Based Biosensors to Monitor the Biochemical Oxygen Demand of Wastewater
Abstract
:1. Introduction
2. Materials and Methods
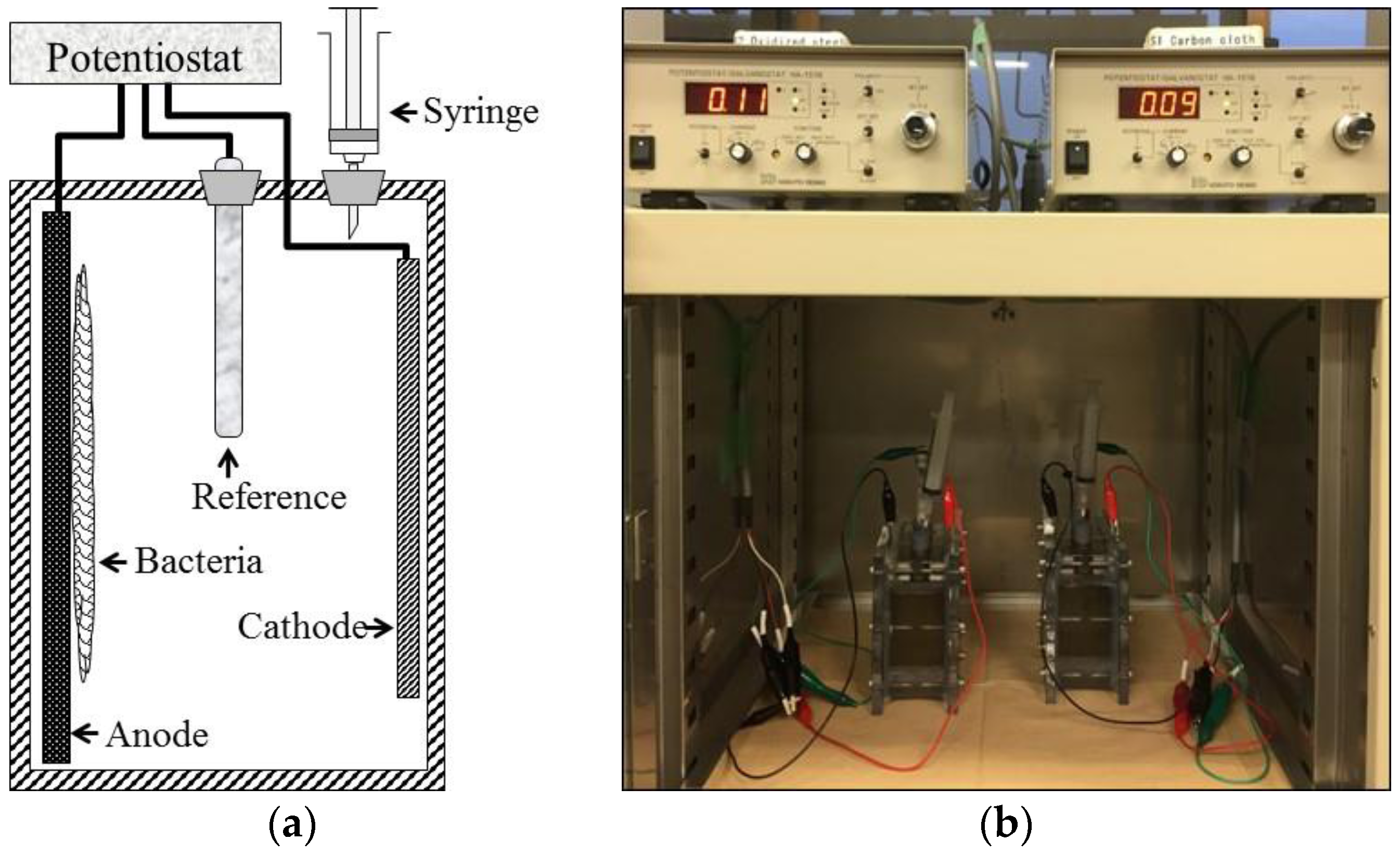
2.1. Biosensor Construction
2.2. Biosensor Operation and Analysis
3. Results and Discussion
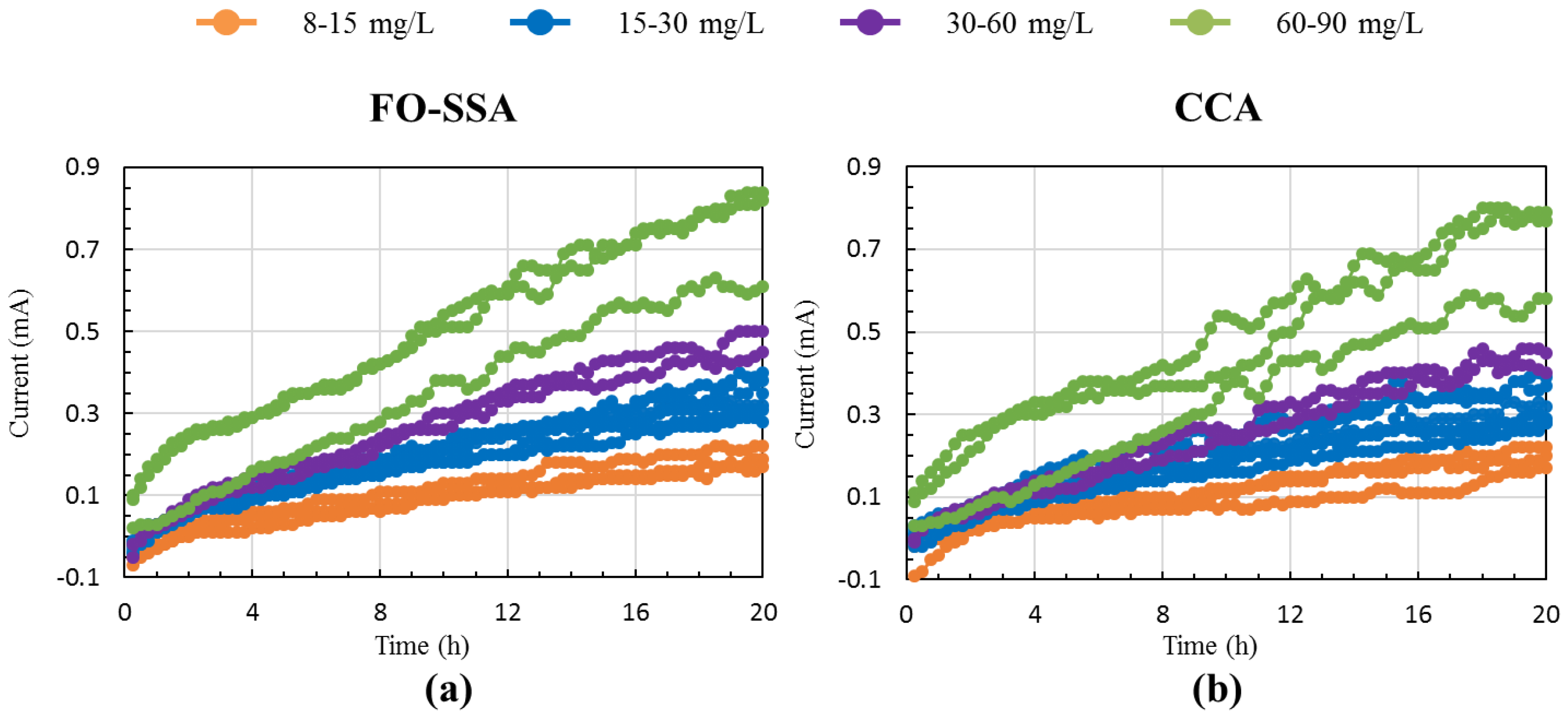
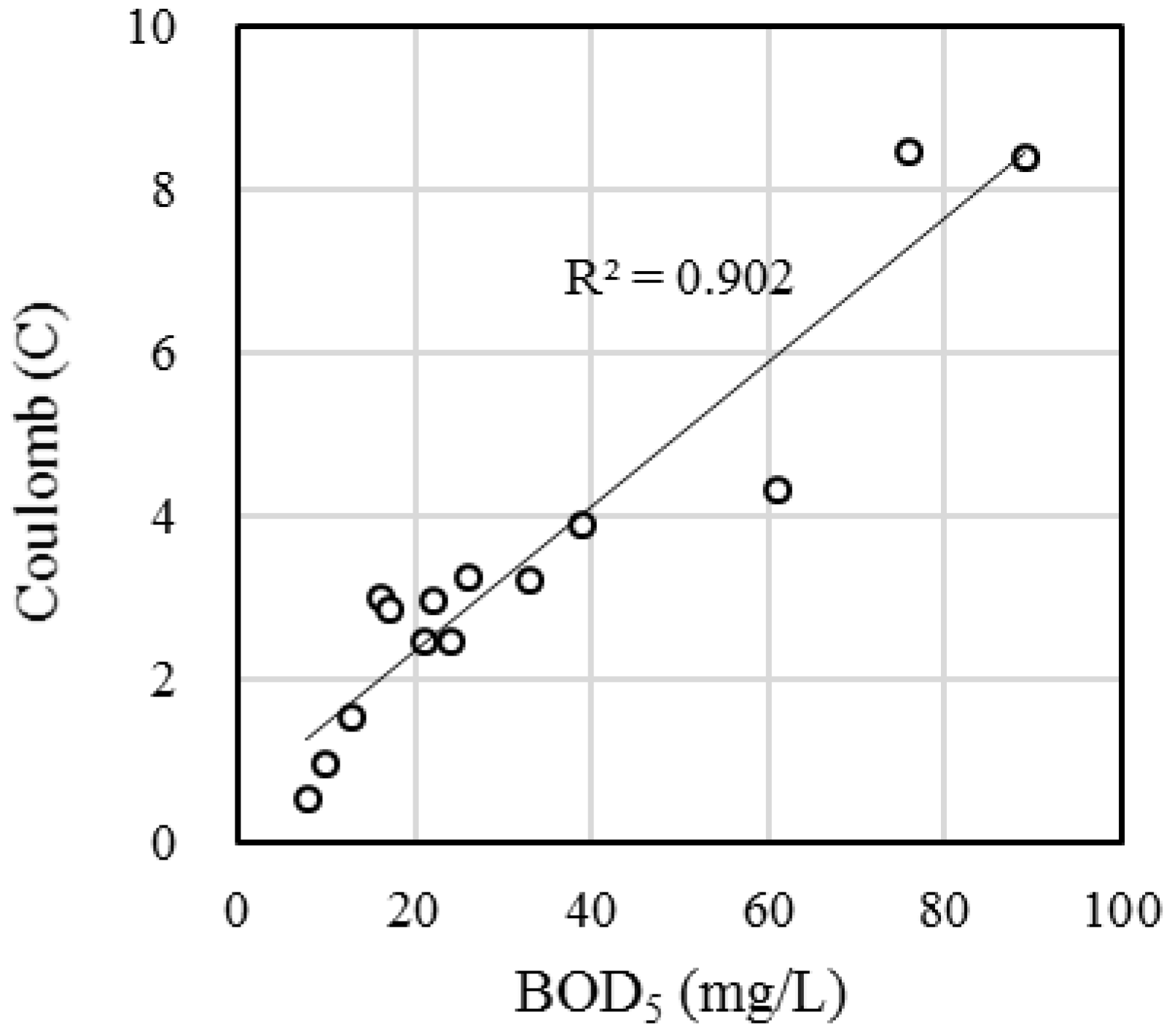
3.1. BOD Monitoring
3.2. Efficacy of the FO-SSA as a BES-Based BOD Biosensor Probe
3.3. Potential Applications of FO-SSA-Equipped BOD Biosensors
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Sun, J.Z.; Kingori, G.P.; Si, R.W.; Zhai, D.D.; Liao, Z.H.; Sun, D.Z.; Zheng, T.; Yong, Y.C. Microbial fuel cell-based biosensors for environmental monitoring: A review. Water Sci. Technol. 2015, 71, 801–809. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.H.; Chang, I.S.; Gil, G.C.; Park, H.S.; Kim, H.J. Novel BOD (biological oxygen demand) sensor using mediator-less microbial fuel cell. Biotechnol. Lett. 2003, 25, 541–545. [Google Scholar] [CrossRef] [PubMed]
- Di Lorenzo, M.; Curtis, T.P.; Head, I.M.; Scott, K. A single-chamber microbial fuel cell as a biosensor for wastewaters. Water Res. 2009, 43, 3145–3154. [Google Scholar] [CrossRef] [PubMed]
- Pasternak, G.; Greenman, J.; Ieropoulos, I. Self-powered, autonomous Biological Oxygen Demand biosensor for online water quality monitoring. Sens. Actuator B-Chem. 2017, 244, 815–822. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, T.; Ookawa, N.; Ishida, M.; Kanamori, H.; Sasaki, H.; Katayose, Y.; Yokoyama, H. A novel open-type biosensor for the in-situ monitoring of biochemical oxygen demand in an aerobic environment. Sci. Rep. 2016, 6, 38552. [Google Scholar] [CrossRef] [PubMed]
- Guo, K.; Donose, B.C.; Soeriyadi, A.H.; Prevoteau, A.; Patil, S.A.; Freguia, S.; Gooding, J.J.; Rabaey, K. Flame oxidation of stainless steel felt enhances anodic biofilm formation and current output in bioelectrochemical systems. Environ. Sci. Technol. 2014, 48, 7151–7156. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, T.; Ishida, M.; Asakawa, S.; Kanamori, H.; Sasaki, H.; Ogino, A.; Katayose, Y.; Hatta, T.; Yokoyama, H. Enhanced electrical power generation using flame-oxidized stainless steel anode in microbial fuel cells and the anodic community structure. Biotechnol. Biofuels 2016, 9, 62. [Google Scholar] [CrossRef] [PubMed]
- Bond, D.R.; Lovley, D.R. Electricity production by Geobacter sulfurreducens attached to electrodes. Appl. Environ. Microbiol. 2003, 69, 1548–1555. [Google Scholar] [CrossRef] [PubMed]
- Yokoyama, H.; Ishida, M.; Yamashita, T. Comparison of Anodic Community in Microbial Fuel Cells with Iron Oxide-Reducing Community. J. Microbiol. Biotechnol. 2016, 26, 757–762. [Google Scholar] [CrossRef] [PubMed]

| Response Time (h) | FO-SSA | CCA |
|---|---|---|
| 1 | 0.805 | 0.696 |
| 2 | 0.823 | 0.752 |
| 3 | 0.845 | 0.789 |
| 4 | 0.860 | 0.803 |
| 5 | 0.874 | 0.820 |
| 6 | 0.884 | 0.836 |
| 7 | 0.894 | 0.851 |
| 8 | 0.902 | 0.867 |
| 10 | 0.918 | 0.895 |
| 12 | 0.924 | 0.912 |
| 16 | 0.937 | 0.930 |
| 20 | 0.941 | 0.939 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liang, Q.; Yamashita, T.; Yamamoto-Ikemoto, R.; Yokoyama, H. Flame-Oxidized Stainless-Steel Anode as a Probe in Bioelectrochemical System-Based Biosensors to Monitor the Biochemical Oxygen Demand of Wastewater. Sensors 2018, 18, 607. https://doi.org/10.3390/s18020607
Liang Q, Yamashita T, Yamamoto-Ikemoto R, Yokoyama H. Flame-Oxidized Stainless-Steel Anode as a Probe in Bioelectrochemical System-Based Biosensors to Monitor the Biochemical Oxygen Demand of Wastewater. Sensors. 2018; 18(2):607. https://doi.org/10.3390/s18020607
Chicago/Turabian StyleLiang, Qiaochu, Takahiro Yamashita, Ryoko Yamamoto-Ikemoto, and Hiroshi Yokoyama. 2018. "Flame-Oxidized Stainless-Steel Anode as a Probe in Bioelectrochemical System-Based Biosensors to Monitor the Biochemical Oxygen Demand of Wastewater" Sensors 18, no. 2: 607. https://doi.org/10.3390/s18020607
APA StyleLiang, Q., Yamashita, T., Yamamoto-Ikemoto, R., & Yokoyama, H. (2018). Flame-Oxidized Stainless-Steel Anode as a Probe in Bioelectrochemical System-Based Biosensors to Monitor the Biochemical Oxygen Demand of Wastewater. Sensors, 18(2), 607. https://doi.org/10.3390/s18020607




