A Review of Wearable Solutions for Physiological and Emotional Monitoring for Use by People with Autism Spectrum Disorder and Their Caregivers
Abstract
1. Introduction
2. Physiological Changes in ASD Population, Their Relevance in the Context of Anxiety, Stress, and Challenging Behavior Detection, and the Potential for Wearable Assistive Technology
2.1. Autism, and Prevelance of Anxiety in ASD Population
2.2. Physiological Signals for Emotional Monitoring
2.2.1. Heart Rate, Heart Rate Variability, and Heart Rate Reactivity
2.2.2. Respiration Rate
2.2.3. Electrodermal Activity
2.2.4. Skin and Body Temperature
2.2.5. Cortisol Level
2.2.6. Blood Pressure
2.2.7. Blood Volume Pulse
2.2.8. Blood Oxygen Saturation
2.2.9. Electromyography
2.2.10. Electroencephalogram
2.3. Physiological Changes Associated with Anxiety, Stress in ASD Population
2.4. Emotional Changes Assosciated with Challenging Behavior in Population with ASD
2.5. The Potential for Wearable Technology for Stress Detection in ASD in Daily Life
3. Related Work
4. Review of Physiological and Emotional Wearable Monitoring Solutions
4.1. Research Methodology
- Commercially available devices that can monitor physiological signals or emotional states.
- Research prototypes that can monitor physiological signals or emotional states.
- Forthcoming solutions that are still under development which can claim to be able to monitor physiological signals or emotional states.
4.2. Commercial Wearable Devices
4.3. Research Prototypes
4.4. Emerging Solutions
5. Discussion
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| BLE | Bluetooth Low-Energy |
| HR | Heart Rate |
| HRV | Heart Rate Variability |
| RR | Respiration Rate |
| RSA | Respiratory Sinus Arrhythmia |
| ST | Skin Temperature |
| EDA | Electrodermal Activity |
| PPG | Photoplethysmogramy |
| BVP | Blood Volume Pulse |
| ECG | Electrocardiogramy |
| EEG | Electroencephalogramy |
| EMG | Electromyography |
| FDA | Drug and Food Administration |
| TENS | Transcutaneous electrical nerve stimulation |
| tDCS | Transcranial direct current stimulation |
References
- Salazar, F.; Baird, G.; Chandler, S.; Tseng, E.; O’sullivan, T.; Howlin, P.; Pickles, A.; Simonoff, E. Co-occurring psychiatric disorders in preschool and elementary school-aged children with autism spectrum disorder. J. Autism Dev. Disord. 2015, 45, 2283–2294. [Google Scholar] [CrossRef] [PubMed]
- White, S.W.; Oswald, D.; Ollendick, T.; Scahill, L. Anxiety in children andadolescents with autism spectrum disorders. Clin. Psychol. Rev. 2009, 29, 216–229. [Google Scholar] [CrossRef] [PubMed]
- White, S.W.; Oswald, D.; Ollendick, T.; Scahill, L.; Strang, J.F.; Kenworthy, L.; Daniolos, P.; Case, L.; Wills, M.C.; Martin, A.; et al. Depression and anxiety symptoms in children and adolescents with autism spectrum disorders without intellectual disability. Res. Autism Spectr. Disord. 2012, 6, 406–412. [Google Scholar] [CrossRef]
- Tantam, D. The challenge of adolescents and adults with asperger syndrome. Child Adolesc. Psychiatr. Clin. N. Am. 2003, 12, 143–163. [Google Scholar] [CrossRef]
- Farrugia, S.; Hudson, J. Anxiety in adolescents with Asperger syndrome: Negative thoughts, behavioural problems, and life interference. Focus Autism Dev. Disabil. 2006, 21, 25–35. [Google Scholar] [CrossRef]
- Bitsika, V.; Sharpley, C.F. Which Aspects of Challenging Behaviour Are Associated with Anxiety across two Age Groups of Young Males with an Autism Spectrum Disorder? J. Dev. Phys. Disabil. 2016, 28, 685–701. [Google Scholar] [CrossRef]
- Fitzpatrick, S.E.; Srivorakiat, L.; Wink, L.K.; Pedapati, E.V.; Erickson, C.A. Aggression in autism spectrum disorder: Presentation and treatment options. Neuropsychiatr. Dis. Treat. 2016, 12, 1525–1538. [Google Scholar] [CrossRef] [PubMed]
- Bird, G.; Cook, R. Mixed emotions: The contribution of alexithymia to the emotional symptoms of autism. Transl. Psychiatry. 2013, 3, e285. [Google Scholar] [CrossRef]
- Malik, M.; Bigger, J.T.; Camm, A.J.; Kleiger, R.E.; Malliani, A.; Moss, A.J.; Schwartz, P.J. Heart rate variability: Standards of measurement, physiological interpretation, and clinical use. Eur. Heart J. 1996, 14, 354–381. [Google Scholar] [CrossRef]
- Jeyhani, V.; Mahdiani, S.; Peltokangas, M.; Vehkaoja, A. Comparison of HRV parameters derived from photoplethysmography and electrocardiography signals. In Proceedings of the 37th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Milan, Italy, 25–29 August 2015; pp. 5952–5955. [Google Scholar]
- Moraes, J.L.; Rocha, M.X.; Vasconcelos, G.G.; Vasconcelos Filho, J.E.; de Albuquerque, V.H.C.; Alexandria, A.R. Advances in photopletysmography signal analysis for biomedical applications. Sensors 2018, 18, 1894. [Google Scholar] [CrossRef]
- Yasuma, F.; Hayano, J. Respiratory sinus arrhythmia: Why does the heartbeat synchronize with respiratory rhythm? Chest 2004, 125, 683–690. [Google Scholar] [CrossRef]
- Tonhajzerova, I.; Mestanik, M.; Mestanikova, A.; Jurko, A. Respiratory sinus arrhythmia as a non-invasive index of ‘brain-heart’ interaction in stress. Indian J. Med. Res. 2016, 144, 815–822. [Google Scholar] [CrossRef] [PubMed]
- Vinkers, C.H.; Penning, R.; Hellhammer, J.; Verster, J.C.; Klaessens, J.H.; Olivier, B.; Kalkman, C.J. The effect of stress on core and peripheral body temperature in humans. Stress 2013, 16, 520–530. [Google Scholar] [CrossRef] [PubMed]
- Villarejo, M.V.; Zapirain, B.G.; Zorrilla, A.M. A Stress Sensor Based on Galvanic Skin Response (GSR) Controlled by ZigBee. Sensors 2012, 12, 6075–6101. [Google Scholar] [CrossRef] [PubMed]
- Parlak, O.; Keene, S.T.; Marais, A.; Curto, V.F.; Salleo, A. Molecularly selective nanoporous membrane-based wearable organic electrochemical device for noninvasive cortisol sensing. Sci. Adv. 2018, 4, eaar2904. [Google Scholar] [CrossRef]
- Marvar, P.J.; Vinh, A.; Salim, T.; Lob, H.E.; Geem, D.; Ressler, K.J.; Harrison, D.G. T Lymphocytes and Vascular Inflammation Contribute to Stress-Dependent Hypertension. Biol. Psychiatry 2012, 71, 774–782. [Google Scholar] [CrossRef]
- Trapp, M.; Trapp, E.M.; Egger, J.W.; Domej, W.; Schillaci, G.; Avian, A.; Rohrer, P.M.; Hörlesberger, N.; Magometschnigg, D.; Cervar-Zivkovic, M.; et al. Impact of Mental and Physical Stress on Blood Pressure and Pulse Pressure under Normobaric versus Hypoxic Conditions. PLoS ONE 2014, 9, e89005. [Google Scholar] [CrossRef]
- Xie, J.; Wen, W.; Liu, G.; Chen, C.; Zhang, J.; Liu, H. Identifying strong stress and weak stress through blood volume pulse. In Proceedings of the International Conference on Progress in Informatics and Computing (PIC), Shanghai, China, 23–25 December 2016. [Google Scholar]
- Leader, D. Understanding Oxygen Saturation. VeryWellHealth. Available online: https://www.verywellhealth.com/oxygen-saturation-914796 (accessed on 5 October 2018).
- Aeschliman, S.D.; Blue, M.S.; Williams, K.B.; Cobb, C.M.; MacNeill, S.R. A preliminary study on oxygen saturation levels of patients during periodontal surgery with and without oral conscious sedation using diazepam. J. Periodontol. 2003, 74, 1056–1059. [Google Scholar] [CrossRef]
- Health Library—Electromyography (EMG). Available online: https://www.hopkinsmedicine.org/healthlibrary/test_procedures/neurological/electromyography_92,p07656 (accessed on 10 October 2018).
- Luijcks, R.; Hermens, H.J.; Bodar, L.; Vossen, C.J.; van Os, J.; Lousberg, R. Experimentally Induced Stress Validated by EMG Activity. PLoS ONE 2014, 9, e95215. [Google Scholar] [CrossRef]
- Taelman, J. Analysis of Electrophysiological Measurements during Stress Monitoring. Ph.D. Thesis, Katholieke Universiteit Leuven, Leuven, Belgium, 2011. Available online: ftp://ftp.esat.kuleuven.be/sista/jtaelman/reports/phd_Joachim.pdf (accessed on 10 September 2018).
- Michel, C.M.; Murray, M.M.; Lantz, G.; Gonzalez, S.; Spinelli, L.; Petralta, R.G.D. EEG source imaging. Clin. Neurophysiol. 2004, 115, 2195–2222. [Google Scholar] [CrossRef]
- Ryu, K.; Myung, R. Evaluation of mental workload with a combined measure based on physiological indices during a dual task of tracking and mental arithmetic. Inter. J. Ind. Ergon. 2005, 35, 991–1009. [Google Scholar] [CrossRef]
- Harmony, T.; Fernández, T.; Silva, J.; Bernal, J.; Díaz-Comas, L.; Reyes, A.; Marosi, E.; Rodríguez, M. EEG delta activity: An indicator of attention to internal processing during performance of mental tasks. Int. J. Psychophysiol. 1996, 24, 161–171. [Google Scholar] [CrossRef]
- Steensel, F.J.A.; Bögels, S.M.; Perrin, S. Anxiety disorders in children and adolescents with autistic spectrum disorders: A meta-analysis. Clin. Child. Fam. Psychol. Rev. 2011, 14, 302–317. [Google Scholar] [CrossRef] [PubMed]
- Simonoff, E.; Pickles, A.; Charman, T.; Chandler, S.; Loucas, T.; Baird, G. Psychiatric disorders in children with autism spectrum disorders: Prevalence, comorbidity, and associated factors in a population-derived sample. J. Am. Acad. Child Adolesc. Psychiatry 2008, 47, 921–929. [Google Scholar] [CrossRef] [PubMed]
- Merikangas, K.R.; He, J.P.; Burstein, M.; Swanson, S.A.; Avenevoli, S.; Cui, L.; Benjet, C.; Georgiades, K.; Swendsen, J. Lifetime Prevalence of Mental Disorders in US Adolescents: Results from the National Comorbidity Study-Adolescent Supplement (NCS-A). J. Am. Acad. Child Adolesc. Psychiatry 2010, 49, 980–989. [Google Scholar] [CrossRef] [PubMed]
- Jansen, L.M.; Gispen-de Wied, C.C.; Wiegant, V.M.; Westenberg, H.G.; Lahuis, B.E.; van Engeland, H. Autonomic and Neuroendocrine Responses to a Psychosocial Stressor in Adults with Autistic Spectrum Disorder. J. Autism Dev. Disord. 2006, 36, 891–899. [Google Scholar] [CrossRef] [PubMed]
- Levine, T.P.; Stephen, J.S.; Pescosolido, M.; Rodino, A.; Elia, G.; Lester, B. Physiologic arousal to social stress in children with Autism. Res. Autism Spectr. Disord. 2012, 6, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Goodwin, M.S.; Groden, J.; Velicer, W.F.; Lipsitt, L.P.; Grace, B.M.; Hofmann, S.G.; Groden, G. Cardiovascular arousal in individuals with autism. Focus Autism Dev. Disabil. 2006, 21, 100–123. [Google Scholar] [CrossRef]
- Berntson, G.G.; Bigger, J.T.; Eckberg, D.L.; Grossman, P.; Kaufmann, P.G.; Malik, M.; Nagaraja, H.N.; Porges, S.W.; Saul, J.P.; Stone, P.H.; et al. Heart Rate Variability: Origins, Methods, and Interpretive Caveats. Psychophysiology 1997, 34, 623–648. [Google Scholar] [CrossRef]
- Groden, J.; Goodwin, M.S.; Baron, M.G.; Groden, G.; Velicer, W.F.; Lipsitt, L.P.; Hofmann, S.G.; Plummer, B. Assessing Cardiovascular Responses to Stressors in Individuals With Autism Spectrum Disorders. Focus Autism Dev. Disabil. 2005, 20, 244–252. [Google Scholar] [CrossRef]
- Kushki, A.; Brian, J.; Dupuis, A.; Anagnostou, E. Functional autonomic nervous system profile in children with autism spectrum disorder. Mol. Autism 2014, 5, 39. [Google Scholar] [CrossRef] [PubMed]
- Goodwin, M.S.; Özdenizci, O.; Cumpanasoiu, C.; Tian, P.; Guo, Y.; Stedman, A.; Peura, C.; Mazefsky, C.; Siegel, M.; Erdoğmuş, D.; et al. Predicting imminent aggression onset in minimally-verbal youth with autism spectrum disorder using preceding physiological signals. In Proceedings of the 12th EAI International Conference on Pervasive Computing Technologies for Healthcare, New York, NY, USA, 21–24 May 2018; pp. 201–207. [Google Scholar]
- Sappok, T.; Budczies, J.; Dziobek, I.; Bölte, S.; Dosen, A.; Diefenbacher, A. The missing link: Delayed emotional development predicts challenging behavior in adults with intellectual disability. J. Autism Dev. Dis. 2014, 44, 786–800. [Google Scholar] [CrossRef] [PubMed]
- Rojahn, J.; Schroeder, S.R.; Hoch, T.A. Self-Injurious Behavior in Intellectual Disabilities; Elsevier: New York, NY, USA, 2007; Volume 2. [Google Scholar]
- Carr, E.G.; Durand, V.M. Reducing behavior problems through functional communication training. J. Appl. Behav. Anal. 1985, 18, 111–126. [Google Scholar] [CrossRef]
- Heslop, P.; Macaulay, F. Hidden Pain?: Self-Injury and People with Learning Disabilities; Bristol Crisis Service for Women: Bristol, UK, 2009. [Google Scholar]
- Leibenluft, E.; Gardner, D.L.; Cowdry, R.W. Special feature the inner experience of the borderline self-mutilator. J. Personal. Disord. 1987, 1, 317–324. [Google Scholar] [CrossRef]
- Suyemoto, K.L. The functions of self-mutilation. Clin. Psychol. Rev. 1998, 18, 531–554. [Google Scholar] [CrossRef]
- Emerson, E. Challenging Behaviour: Analysis and Intervention in People with Severe Intellectual Disabilities; Cambridge University Press: New York, NY, USA, 2001. [Google Scholar]
- Hufnagel, C.; Chambres, P.; Bertrand, P.R.; Dutheil, F. The need for objective measures of stress in autism. Front. Psychol. 2017, 8, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Majumder, S.; Mondal, T.; Deen, M.J. Wearable Sensors for Remote Health Monitoring. Sensors 2017, 17, 130–175. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.; Park, H.; Bonato, P.; Chan, L.; Rodgers, M. A review of wearable sensors and systems with application in rehabilitation. J. Neuroeng. Rehabil. 2012, 9, 21. [Google Scholar] [CrossRef] [PubMed]
- Kamišalic, A.; Fister, I., Jr.; Turkanovic, M.; Karakatic, S. Sensors and functionalities of non-invasive wrist-wearable devices: A review. Sensors 2018, 18, 1714. [Google Scholar] [CrossRef] [PubMed]
- Gao, W.; Brooks, G.A.; Klonoff, D.C. Wearable physiological systems and technologies for metabolic monitoring. J. Appl. Physiol. 2017, 124, 548–556. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Zhang, L.; Tao, X.; Ding, X. Review of flexible temperature sensing networks for wearable physiological monitoring. Adv. Healthcare Mater. 2017, 6, 1601371. [Google Scholar] [CrossRef] [PubMed]
- Nag, A.; Mukhopadhyay, S.C.; Kosel, J. Wearable Flexible Sensors: A Review. IEEE Sensors J. 2017, 17, 3949–3960. [Google Scholar] [CrossRef]
- Stacey, K.M.; Hill, N.; Woods, D. Physiological monitoring for healthy military personnel. J. R. Army Med. Corps 2018, 164, 290–292. [Google Scholar] [CrossRef] [PubMed]
- Baig, M.M.; GholamHosseini, H.; Moqeem, A.A.; Mirza, F.; Lindén, M. A Systematic Review of Wearable Patient Monitoring Systems—Current Challenges and Opportunities for Clinical Adoption. J. Med. Syst. 2017, 41, 115. [Google Scholar] [CrossRef] [PubMed]
- Reinertsen, E.; Clifford, G.D. A review of physiological and behavioral monitoring with digital sensors for neuropsychiatric illnesses. Physiol. Meas. 2018, 39, 05TR01. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, M.H.; Aydin, A.; Brunckhorst, O.; Dasgupta, P.; Ahmed, K. A review of wearable technology in medicine. J. R. Soc. Med. 2016, 109, 372–380. [Google Scholar] [CrossRef] [PubMed]
- Alberdia, A.; Aztiria, A.; Basarab, A. Towards an automatic early stress recognition system for office environments based on multimodal measurements: A review. J. Biomed. Inform. 2016, 59, 49–75. [Google Scholar] [CrossRef] [PubMed]
- Peake, J.M.; Kerr, G.; Sullivan, J.P. A Critical Review of Consumer Wearables, Mobile Applications, and Equipment for Providing Biofeedback, Monitoring Stress, and Sleep in Physically Active Populations. Front. Physiol. 2018, 9, 743. [Google Scholar] [CrossRef]
- Koo, S.H.; Gaul, K.; Rivera, S.; Pan, T.; Fong, D. Wearable Technology Design for Autism Spectrum Disorders. Arch. Des. Res. 2018, 31, 37–55. [Google Scholar] [CrossRef]
- Cabibihan, J.; Javed, H.; Aldosari, M.; Frazier, T.W.; Elbashir, H. Sensing Technologies for Autism Spectrum Disorder Screening and Intervention. Sensors 2017, 17, 46–71. [Google Scholar] [CrossRef]
- Taj-Eldin, M.; O’Flynn, B.; Galvin, P.; Ryan, C. A Review of Wearable Tracking and Emotional Monitoring Solutions for Individuals with Autism and Intellectual Disability. In Proceedings of the Fourth International Conference on Smart Portable, Wearable, Implantable and Disability-oriented Devices and Systems, Barcelona, Spain, 22–26 July 2018. [Google Scholar]
- Fitbit. Available online: https://www.fitbit.com/shop/charge3 (accessed on 5 October 2018).
- Hexoskin, Inc. Available online: https://www.hexoskin.com/pages/health-research (accessed on 6 October 2018).
- Benedetto, S.; Caldato, C.; Bazzan, E.; Greenwood, D.C.; Pensabene, V.; Actis, P. Actis Assessment of the Fitbit Charge 2 for monitoring heart rate. PLoS ONE 2018, 13, e192691. [Google Scholar] [CrossRef] [PubMed]
- Garmin. Available online: https://buy.garmin.com/en-US/US/p/605739 (accessed on 10 October 2018).
- Scosche. Available online: https://www.scosche.com/rhythm24-waterproof-armband-heart-rate-monit (accessed on 10 October 2018).
- McCarthy, C.; Pradhan, N.; Redpath, C.; Adler, A. Validation of the Empatica E4 wristband. In Proceedings of the IEEE EMBS International Student Conference (ISC), Ottawa, ON, Canada, 29–31 May 2016. [Google Scholar] [CrossRef]
- Empatica, Inc. Available online: https://store.empatica.com/products/e4-wristband?variant=39588207747 (accessed on 30 October 2018).
- Empatica Inc. Available online: https://www.empatica.com/embrace/ (accessed on 23 October 2018).
- Empatica Srl. Available online: https://blog.empatica.com/embrace-is-now-an-fda-approved-medical-device-bf506d991b1c (accessed on 23 October 2018).
- PulseOn. Available online: http://pulseon.com/ohr/ohr-tracker (accessed on 5 October 2018).
- Pieti, J.; Mehrang, S.; Tolonen, J.; Helander, E.; Jimison, H.; Pavel, M.; Korhonen, I. Evaluation of the accuracy and reliability for photoplethysmography based heart rate and beat-to-beat detection during daily activities. In EMBEC & NBC 2017; Springer: Singapore, 2017; Volume 65, pp. 145–148. [Google Scholar]
- The ViSi Mobile. Available online: http://www.visimobile.com/visi-mobile/ (accessed on 10 October 2018).
- Weenk, M.; van Goor, H.; Frietman, B.; Engelen, L.J.; van Laarhoven, C.J.; Smit, J.; Bredie, S.J.H.; van de belt, T.H. Continuous monitoring of vital signs using wearable devices on the general ward: Pilot study. JMIR Mhealth Uhealth 2017, 5, e91. [Google Scholar] [CrossRef] [PubMed]
- Zypher Technology. Available online: https://www.zephyranywhere.com/media/download/bioharness3-user-manual.pdf (accessed on 9 October 2018).
- Nepi, D.; Sbrollini, A.; Agostinelli, A. Validation of the heart-rate signal provided by the Zephyr bioharness 3.0. In Proceedings of the Computing in Cardiology Conference (CinC), Vancouver, BC, Canada, 11–14 September 2016; Volume 43, pp. 361–364. [Google Scholar]
- Equivital, Inc. Available online: http://www.equivital.com/assets/common/SEM_Data_Sheet_General_HIDA3330-DSG-02.2_.2_.pdf (accessed on 10 October 2018).
- Akintola, A.A.; van de Pol, V.; Bimmel, D.; Maan, A.C.; van Heemst, D. comparative analysis of the equivital eq02 lifemonitor with holter ambulatory ecg device for continuous measurement of ecg, heart rate, and heart rate variability: A validation study for precision and accuracy. Front. Physiol. 2016, 7, 391. [Google Scholar] [CrossRef] [PubMed]
- Zephyr Performance Systems. Available online: https://www.zephyranywhere.com/benefits/physiological-biomechanical (accessed on 6 September 2018).
- Cherif, N.H.; Mezghani, N.; Gaudreault, N.; Ouakrim, Y.; Mouzoune, I.; Boulay, P. Physiological data validation of the hexoskin smart textile. In Proceedings of the 11th International Joint Conference on Biomedical Engineering System and Technologies (BIOSTEC 2018), Funchal, Portugal, 19–21 January 2018. [Google Scholar] [CrossRef]
- Polar Electro. Available online: https://www.polar.com/us-en/products/accessories/h10_heart_rate_sensor (accessed on 10 October 2018).
- Vogelaere, P.; De Meyer, F.; Duquet, W.; Vandevelde, P. Sports tester PE 3000 vs Holter ECG for the measurement of heart frequency. Sci. Sports 1986, 1, 321–329. [Google Scholar] [CrossRef]
- Plews, D.J.; Scott, B.; Altini, M.; Wood, M.; Kilding, A.E.; Laursen, P.B. Comparison of heart rate variability recording with smart phone photoplethysmographic, Polar H7 chest strap and electrocardiogram methods. Int. J. Sports Physiol. Perform. 2017, 12, 1324–1328. [Google Scholar] [CrossRef] [PubMed]
- Polar Electro Oy. Available online: https://www.polar.com/en/products/pro/V800 (accessed on 10 October 2018).
- Giles, D.; Draper, N.; Neil, W. Validity of the Polar V800 heart rate monitor to measure RR intervals at rest. Eur. J. Appl. Physiol. 2016, 116, 563–571. [Google Scholar] [CrossRef] [PubMed]
- Polar Electro Oy. Available online: https://www.polar.com/en/products/compare?product1=91059 (accessed on 12 October 2018).
- Qardio, Inc. Available online: https://www.getqardio.com/qardiocore-wearable-ecg-ekg-monitor-iphone/ (accessed on 10 October 2018).
- Is QardioCore Clinically Validated? Available online: https://support.getqardio.com/hc/en-us/articles/115000256725-Is-QardioCore-clinically-validated- (accessed on 10 October 2018).
- Under Armour, Inc. Available online: https://www.underarmour.com/en-us/pid1247081 (accessed on 10 October 2018).
- Heart Rate Variability Logger. Available online: https://www.marcoaltini.com/blog/heart-rate-variability-logger-app-details (accessed on 10 October 2018).
- Wahoo Fitness. Available online: https://eu.wahoofitness.com/devices/heart-rate-monitors/wahoo-tickr-x-heart-rate-strap (accessed on 10 October 2018).
- Elite HRV. Available online: https://elitehrv.com/compatible-devices (accessed on 10 October 2018).
- Suunto. Available online: https://www.suunto.com/Products/sports-watches/suunto-9/suunto-9-baro-black/ (accessed on 9 October 2018).
- Suunto, Inc. Available online: https://www.suunto.com/Products/Heart-Rate-Belts/SuuntoSmartSensor/ (accessed on 9 October 2018).
- Bouillod, A.; Cassirame, J.; Bousson, J.M.; Sagawa, Y., Jr.; Tordi, N. Accuracy of the Suunto system for heart rate variability analysis during a tilt-test. Rev. Bras. Cineantropometria Desempenho Hum. 2015, 17, 409–417. [Google Scholar] [CrossRef]
- Apple. Available online: https://www.apple.com/ie/apple-watch-series-4/ (accessed on 5 October 2018).
- Hernando, D.; Roca, S.; Sancho, J.; Alesanco, Á.; Bailón, R. Validation of the apple watch for heart rate variability measurements during relax and mental stress in healthy subjects. Sensors 2018, 18, 2619. [Google Scholar] [CrossRef] [PubMed]
- Healbe, Inc. Available online: https://healbe.com/us/gobe2/ (accessed on 15 October 2018).
- MC10 Inc. Available online: https://www.mc10inc.com/our-products#biostamp-npoint (accessed on 8 September 2018).
- MC10 Inc. Available online: https://www.mc10inc.com/press-media/fda-510k-clearance-for-the-biostamp-npoint-system (accessed on 10 October 2018).
- VialConnect. Available online: https://vitalconnect.com/solutions/vitalpatch/ (accessed on 10 September 2018).
- Lief Therapeutics, Inc. Available online: https://store.getlief.com/products/lief-device (accessed on 15 October 2018).
- Komodo Technologies. Available online: http://komodotec.com/product/aio-sleeve/ (accessed on 10 September 2018).
- Brain Power, LLC. Available online: http://www.brain-power.com/ (accessed on 23 October 2018).
- Using Technology to Recognise Emotions in Autistic People, Graduation Report, University of Twente. Available online: https://essay.utwente.nl/73095/1/Graduation_report_Notenboom.pdf (accessed on 10 October 2018).
- Imani, S.; Bandodkar, A.J.; Mohan, A.M.V.; Kumar, R.; Yu, S.; Wang, J.; Mercier, P.P. A wearable chemical–electrophysiological hybrid biosensing system for real-time health and fitness monitoring. Nat. Commun. 2016, 7, 11650. [Google Scholar] [CrossRef] [PubMed]
- Yoon, S.; Sim, J.K.; Cho, Y. A flexible and wearable human stress monitoring patch. Sci. Rep. 2016, 6, 23468. [Google Scholar] [CrossRef]
- Williams, M.A.; Roseway, A.; O’Dowd, C.; Czerwinski, M.; Moriss, M.M. Swarm: An actuated wearable for mediating affect. In Proceedings of the Ninth International Conference on Tangible, Embedded, and Embodied Interaction, Stanford, CA, USA, 16–19 January 2015. [Google Scholar]
- Guo, C.; Chen, Y.V.; Qian, Z.C.; Ma, Y.; Dinh, H.; Anasingaraju, S. Designing a Smart Scarf to Influence Group Members’ Emotions in Ambience: Design Process and User Experience. In Proceedings of the International Conference on Universal Access in Human-Computer Interaction, Toronto, ON, Canada, 17–22 July 2016; pp. 392–402. [Google Scholar]
- Hui, T.K.L.; Sherratt, R.S. Coverage of Emotion Recognition for Common Wearable Biosensors. Biosensors 2018, 8, 30. [Google Scholar] [CrossRef]
- Tang, T.Y.; Zhejiang, W. Helping Neuro-typical Individuals to “Read” the Emotion of Children with Autism Spectrum Disorder: An Internetof-Things Approach. In Proceedings of the 15th International Conference on Interaction Design and Children, Manchester, UK, 21–24 June 2016. [Google Scholar]
- Torrado, J.C.; Gomez, J.; Montoro, G. Emotional Self-Regulation of Individuals with Autism Spectrum Disorders: Smartwatches for Monitoring and Interaction. Sensor 2017, 17, 1359. [Google Scholar] [CrossRef] [PubMed]
- AlHanai, T.W.; Ghassemi, M.M. Predicting Latent Narrative Mood using Audio and Physiologic Data. In Proceedings of the Association for the Advancement of Artificial Intelligence (AAAI) Conference, San Francisco, CA, USA, 4–9 February 2017. [Google Scholar]
- Northrup, C.M.; Lantz, J.; Hamlin, T. Wearable stress sensors for children with autism spectrum disorder with in situ alerts to caregivers via a mobile phone. Iproceedings 2016, 2. [Google Scholar] [CrossRef]
- Simm, W.; Ferrario, M.A.; Gradinar, A.; Smith, M.T.; Forshaw, S.; Smith, I.; Whittl, J. Anxiety and Autism: Towards Personalized Digital Health. In Proceedings of the 2016 CHI Conference on Human Factors in Computing Systems, San Jose, CA, USA, 7–12 May 2016. [Google Scholar]
- Sentio Solutions Inc. Available online: https://www.myfeel.co/science (accessed on 10 October 2018).
- Awake Labs, Inc. Available online: http://awakelabs.com/home/ (accessed on 6 September 2018).
- Kushki, A.; Anagnostou, E. Anxiety Meter. U.S. Patent 20160000365A1, 7 January 2016. [Google Scholar]
- Gaia LLC. Available online: https://www.gaiawearables.com/ (accessed on 23 September 2018).
- Hedman, E.B. In-Situ Measurement of Electrodermal activity during Occupational Therapy. Master’s Thesis, University of Colorado, Denver, CO, USA, 2010. Available online: https://static1.squarespace.com/static/540e15d2e4b0271ac53fa946/t/54821edae4b0090d4b367956/1417813722496/hedman-masters.pdf (accessed on 23 October 2018).
- TouchPoint, Inc. Available online: https://www.touchpointeurope.com/products/touchpoint-kit-single-person (accessed on 6 June 2018).
- Serin, A.; Hageman, N.S.; Kade, E. The Therapeutic Effect of Bilateral Alternating Stimulation Tactile Form Technology on the Stress Response. J. Biotechnol. Biomed. Sci. 2018, 1, 42–47. [Google Scholar] [CrossRef]
- Bio Technologies. Available online: http://www.biohug.com/Vest (accessed on 23 October 2018).
- Muaremi, A.; Arnrich, B.; Tröster, G. Towards Measuring Stress with Smartphones and Wearable Devices During Workday and Sleep. Bionanoscience 2013, 3, 172–183. [Google Scholar] [CrossRef] [PubMed]
- Gjoreski, M.; Luštrek, M.; Gams, M.; Gjoreski, H. Monitoring stress with a wrist device using context. J. Biomed. Inform. 2017, 73, 159–170. [Google Scholar] [CrossRef] [PubMed]
- Padmaja, B.; Prasad, V.R.; Sunitha, K.V. A Machine Learning Approach for Stress Detection using a Wireless Physical Activity Tracker. Int. J. Mach. Learn. Comput. 2018, 8, 33–38. [Google Scholar] [CrossRef]
- Thync Global Inc. Available online: https://www.thync.com/ (accessed on 16 October 2018).
- Yoshimoto, S.; Babygirija, R.; Dobner, A.; Ludwig, K.; Takahashi, T. Anti-stress effects of transcutaneous electrical nerve stimulation (TENS) on colonic motility in rats. Dig. Dis. Sci. 2012, 57, 1213–1221. [Google Scholar] [CrossRef]
- Fregni, F.; Macedo, I.C.; Spezia-Adachi, L.N.; Scarabelot, V.L.; Laste, G.; Souza, A.; Sanches, P.R.S.; Caumo, W.; Torres, I.L.S. Transcranial direct current stimulation (tDCS) prevents chronic stress-induced hyperalgesia in rats. Brain Stimul. 2018, 11, 299–301. [Google Scholar] [CrossRef]
- ChooseMuse, Inc. Available online: http://www.choosemuse.com/ (accessed on 16 October 2018).
- Healthcare Inc. Available online: https://www.cnet.com/videos/medical-grade-blood-pressure-in-a-fitness-watch-omron-heartguide-measures-your-hypertension/ (accessed on 7 October 2018).
- Sentio Solutions Inc. Available online: https://www.myfeel.co/reserve (accessed on 15 October 2018).
- Awake Labs. Available online: https://awakelabs.com/news-clinical-trial-community-pilot/ (accessed on 17 October 2018).
- Dymaxia Inc. Available online: https://dymaxiainc.com/ (accessed on 23 October 2018).
- mPath. Available online: http://www.buildempathy.com/the-moxo-sensor/ (accessed on 23 October 2018).
- iBreve, Ltd. Available online: https://www.ibreve.com/ (accessed on 15 October 2018).
- Europa. Available online: https://ec.europa.eu/eipp/desktop/en/projects/project-9143.html (accessed on 17 October 2018).
- KiddoWear, Inc. Available online: https://www.kiddowear.com/ (accessed on 15 October 2018).
- Spire, Inc. Available online: https://spire.io/pages/spire-health-tag-main (accessed on 15 October 2018).
- Federal Drug Adminstraton. Available online: https://clinicaltrials.gov/ct2/show/results/NCT02786771 (accessed on 16 October 2018).
- SKIIN, Inc. Available online: https://skiin.com/technology / (accessed on 15 October 2018).
- Bedell Innovations. Available online: https://betapage.co/product/u-check-it (accessed on 15 October 2018).
- Moodmetric. Available online: http://www.moodmetric.com/ (accessed on 15 October 2018).
- Galvanic, Inc. Available online: https://thepip.com/en-eu/ (accessed on 16 October 2018).
- Bhayee, S.; Tomaszewski, P.; Lee, D.H.; Moffat, G.; Pino, L.; Moreno, S.; Farb, N.A.S. Attentional and affective consequences of technology supported mindfulness training: A randomised, active control, efficacy trial. BMC Psychol. 2016, 4, 60. [Google Scholar] [CrossRef]
- Urban. Available online: https://webshop.bellabeat.com/products/leaf-urban (accessed on 15 October 2018).
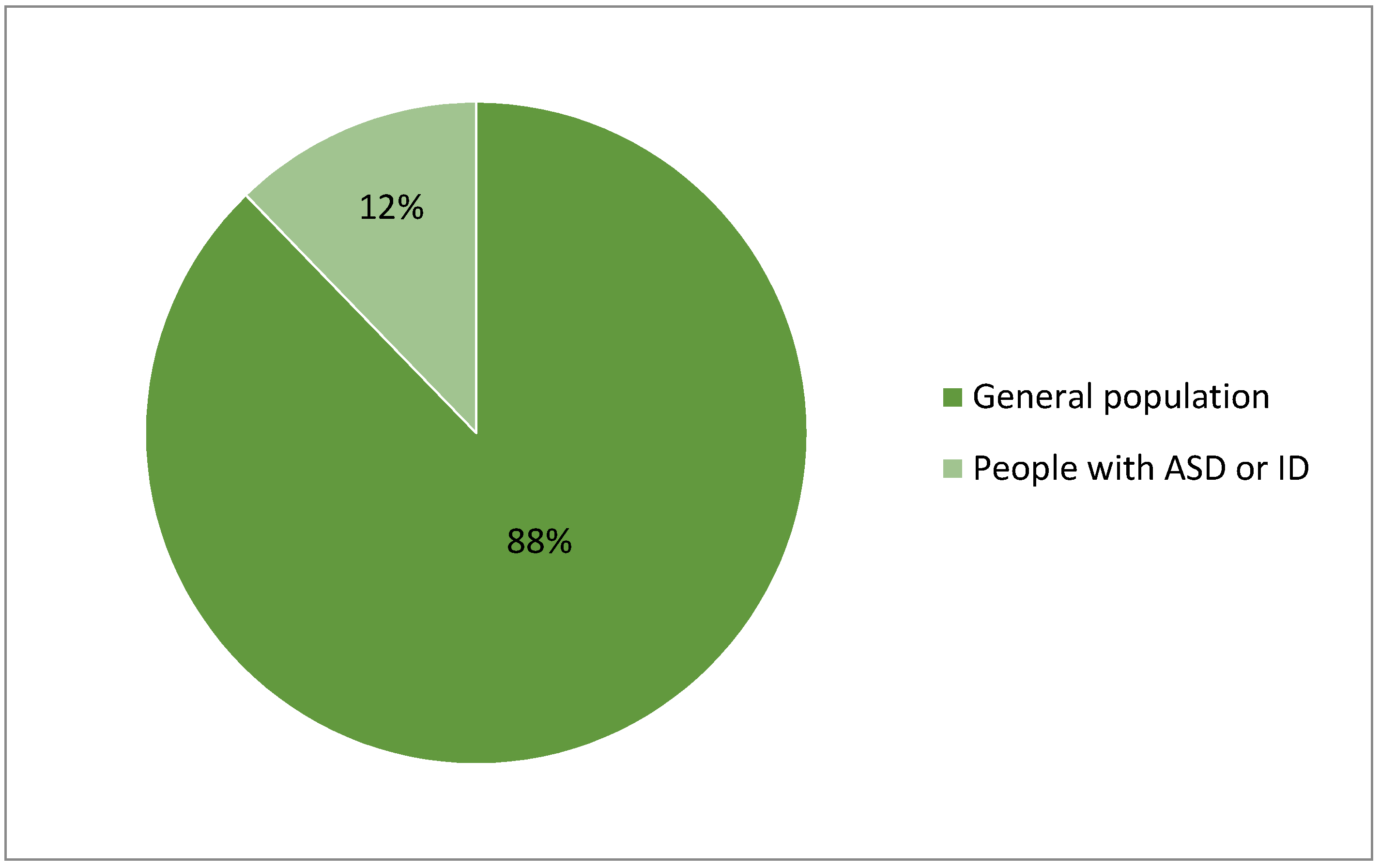
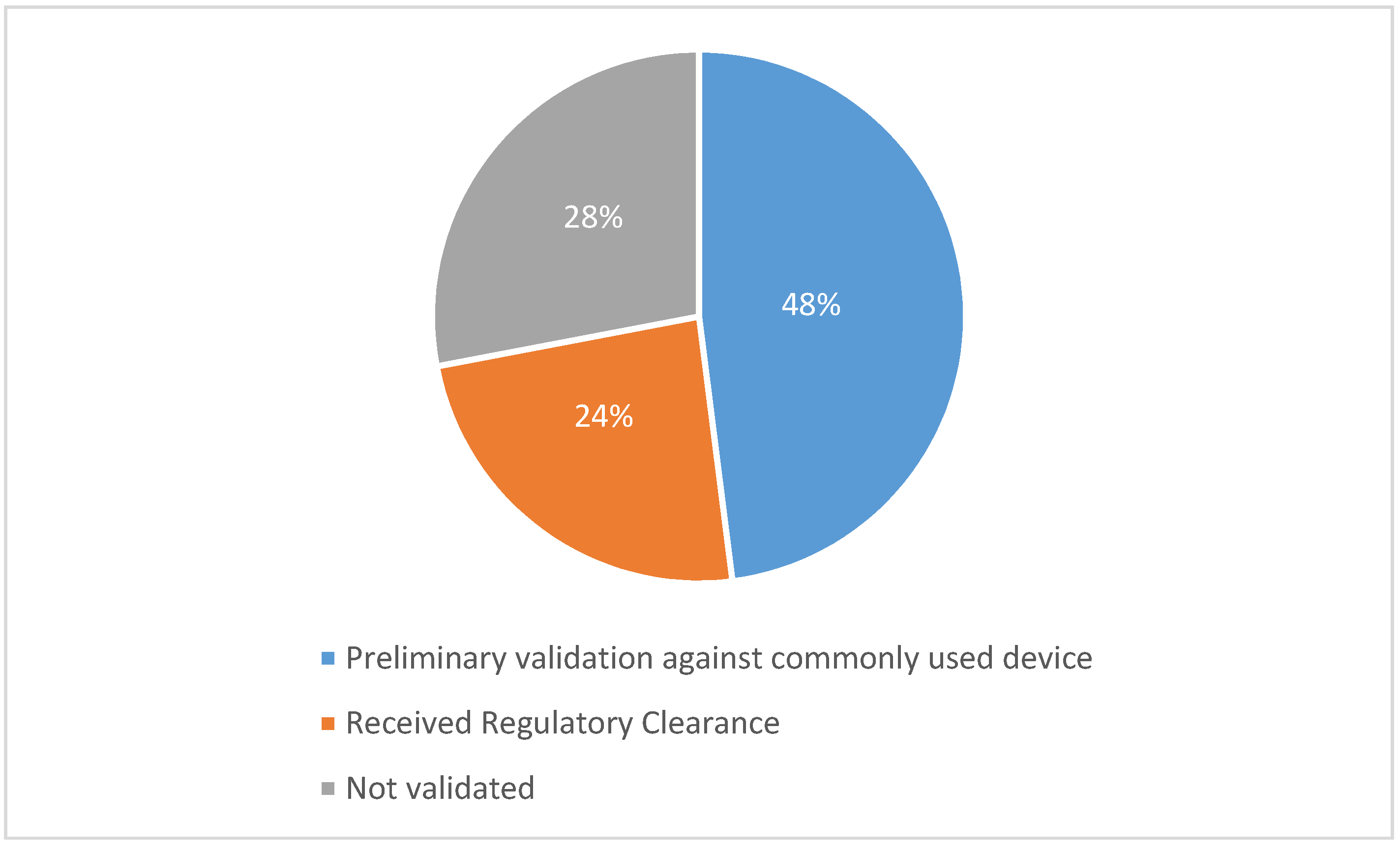
| Product, Producer | Purpose | Device Form Factor | Sensors/Parameters | Support for Wireless Technology | Clinical Validity | Suitability for Individual with ASD |
|---|---|---|---|---|---|---|
| Vivosmart 4, Garmin Inc., Olathe, KS, USA [64] | Monitoring stress score using HRV, sleep quality, and activity (step, distance, calories, floors) | Wristband | HR, HRV, accelerometer, oxygen saturation | Yes, Bluetooth Low Energy (BLE) and ANT+ | Not clinically validated | ☆☆☆ |
| Rhythm24, Scosche Inc., Oxnard, CA, USA [65] | Monitoring heart rate data | Armband | HR, HRV | Yes, BLE/ANT+ Connection | Investigation showed reliable readings [66] | ☆☆☆ |
| E4 wristband, Empatica Inc., Cambridge, MA, USA [67] | Collecting physiological and movement data only | Wristband | HR, HRV, EDA, ST, acceleration | Yes, BLE and data uploaded to cloud | Comparable readings to chest strap [66] | ☆☆☆☆ |
| Embrace, Empatica Inc., Cambridge, MA, USA [68] | Alerts epileptics (or caregivers) when an attack occurs in ASD population | Watch | HR using PPG, EDA, ST, and activity | Yes, using BLE | Clinically validated and FDA approved [69] | ☆☆☆☆☆ |
| PulseOn OHR, PulseOn, Espoo, Finland [70] | Monitoring physiological and movement data | Wristband | HR, HRV, acceleration | Yes, real-time transfer to application using BLE | HR/HRV are reliable only during resting condition [71] | ☆☆☆ |
| ViSi Mobile, Sotera Wireless Inc., San Diego, CA [72] | Monitoring physiological data | Wristband | ECG, HR, pulse rate, BP, RR, SpO2, and ST | Yes, using WiFi | FDA approved for blood pressure monitoring [73] | ☆☆☆ |
| BioHarness 3.0, Zephyr Technology, Annapolis, MD, USA [74] | Physiological and activity data collection | Chest strap | HR, HRV, EDA, body temperature, RR, activity, location | Yes, using BLE and LP–PAN [74] | Only reliable HR but HRV is not suitable for clinical evaluations [75] | ☆☆ |
| EQ02 LifeMonitor, Equivital Inc., Cambridge, UK [76] | Physiological and activity data collection | Chest Belt | ECG; HR, HRV, RR, EDA, ST, acceleration | Yes, using Bluetooth 2.1 | EQ02 can accurately measure ECG and HRV [77] | ☆ |
| Zephyr belt, Medtronic, Inc., Minneapolis, MN, USA [78] | Sports health monitoring | Belt | HR, HRV, RR | Yes, using BLE | Not clinically validated | ☆ |
| Hexoskin Smart Shirt, Hexoskin, Inc., Montreal, QC, Canada [62] | Physiological and activity data collection and monitoring quality of sleep | Shirt | HR, HRV, Heart rate recovery, RR and volume, acceleration and power | Yes, using BLE and uploaded to cloud | Clinically validated to obtain reliable HR and RR [79] | ☆☆☆ |
| Polar H7 & H10, Polar Electro Oy, Kempele, Finland [80] | Monitoring HR activity | Chest Strap | HR, HRV using ECG | Yes, BLE protocol | Clinically comparable data [81,82] | ☆ |
| Polar V800, Polar Electro Oy, Kempele, Finland [83] | Monitoring HR activity | Watch | HR, HRV using light source sensor | Yes, BLE protocol | Clinically validated during rest [84] | ☆☆☆☆ |
| Polar Vantage V, Polar Electro Oy, Kempele, Finland [85] | Monitoring HR and physical activity | Watch | HR, HRV, accelerometer | Yes, BLE protocol | Not clinically validated | ☆☆☆☆ |
| QardioCore, Qardio, Inc., San Francisco, CA, USA [86] | Monitoring HR, ST, activity tracking | Can be mounted on chest | HR, HRV using ECG sensor, RR, ST, acceleration | Yes, BLE protocol | FDA (510) cleared [87] | ☆ |
| Armour39® Module & Chest Strap, Under Armour, Inc., Baltimore, MD, USA [88] | Monitoring heart rate, calorie | Chest Strap | HR, HRV using ECG | Yes, BLE protocol | HR/HRV are reported to be accurate [89] | ☆ |
| Wahoo TICKR X, Wahoo Fitness, Atlanta, GA, USA [90] | Monitoring HR, calorie tracking, insights of physical activity | Chest Strap | HR, HRV using ECG acceleration | Yes, using BLE and ANT+ | Bluetooth is HR/HRV accurate, ANT+ is just HR accurate [91] | ☆ |
| Suunto 9, Suunto, Inc., Vantaa, Finland [92] | Monitoring HR activity | Watch | HR, HRV | Yes, using BLE | Not clinically validated | ☆☆☆☆ |
| Suunto Smart Sensor, Suunto, Inc., Vantaa, Finland [93] | Monitoring HR activity | Chest Strap | HR, HRV | Yes, using BLE | Tests showed reliable HR and HRV [94] | ☆☆ |
| Apple Watch Series 4, Apple, Inc. [95] | HR and physical activity | Watch | HR, HRV | Yes, using BLE and to cloud | FDA cleared, initial tests showed good HR and HRV data [96] | ☆☆☆ |
| GoBe 2, Healbe, Corp., Moscow, Russia [97] | Emotional monitoring, calorie intake, sleep quality | Wristband | HR, EDA, impedance, BP, and acceleration sensor | Yes, using BLE | Not clinically validated | ☆☆☆ |
| BioStamp nPoint, MC10, Inc., Cambridge, CA, USA [98] | Monitoring heart rate, activity and posture data, sleep quality | Patch | HR, HRV, respiration rate, acceleration | Yes, using BLE and uploaded to cloud | FDA (510) cleared [99] | ☆ |
| VitalPatch, VitalConnect, San Jose, CA, USA [100] | HR, HRV, respiration rate, skin temperature, body posture, and activity | Patch | ECG for HR acceleration, thermistor for ST | Yes, data uploaded to the cloud | FDA (510) cleared, CE Marked, ISO 13485 certified. | ☆ |
| Lief Smart Patch, Lief Therapeutics, Inc., San Francisco, CA, USA [101] | Monitoring heart rate, emotional state, activity | Patch | HR, HRV using ECG sensor, RR, acceleration | Yes, using BLE | Not clinically validated | ☆☆☆ |
| AIO Sleeve, Komodo Technologies, Inc., Winnipeg, MB, Canada [102] | Physiological, activity data, monitoring stress level and sleep quality | Sleeve | HR and HRV using ECG, acceleration | Yes, using BLE protocol | Not clinically validated | ☆☆☆☆ |
| Empower Me, Brain Power, LLC., Cambridge, MA, USA [103] | Recognizing others’ emotions for ASD people | Glass | Acceleration | Yes, using BLE protocol | Clinically validated | ☆☆☆☆ |
| Reference, Institution, Year | Purpose | Device Form Factor | Sensors/Parameters | Support for Wireless Technology | Clinical Validity | Suitability for ASD |
|---|---|---|---|---|---|---|
| Imani et al. [105], University of California, San Diego, 2016 | Heart rate activity and sweat monitoring | Patch | HR from ECG sensor, concentrations of hydrogen and sodium ions, lactate in human sweat | Yes, using BLE protocol | Initial test on three subject and HR sensor comparable to commercial monitors | ☆☆ |
| Yoon et al. [106], Advanced Institute of Science and Technology, 2016 | Stress Monitoring | Patch | EDA, ST, and pulsewave | No | No | ☆☆☆ |
| Williams et al. [107], Microsoft, Inc., Redmond, WA, USA 2015 | Reflecting on user’ emotional state, recognizing other’s emotions and stress-relief system | Scarf | Heart rate (HR) | Yes, using BLE | Tested on small number of participants but not validated yet | ☆☆☆☆☆ |
| Guo et al. [108], Purdue University, 2016 | Recognizing emotions, enhancing positive emotions and mitigating negative ones. | Scarf | HR, Electrodermal activity (EDA), and use color-changing and odor emitting tool to promote positive emotions | Yes, using Infrared technology | Not validated yet | ☆☆☆☆☆ |
| Tang et al. [110], Wenzhou-Kean University, 2016 | Helping neuro-typical individuals recording emotional states of children with ASD | HR, perspiration, Pressure, and acceleration sensor | Yes, using WiFi to upload data to cloud | No | ☆☆ | |
| Parlak et al. [16], Stanford University, 2018 | Measuring stress noninvasively | Patch | Cortisol using novel electrochemical sensor | No | Not validated yet | ☆☆☆☆ |
| Notenboom [104], University of Twente, 2017 | Monitoring stress level for people with ASD | Watch | HR, EDA, ST, and oxygen sensor | No | Not validated yet | ☆☆☆☆☆ |
| Torrado et al. [111], Autonomous University of Madrid, 2017 | Inferring outburst patterns and self-regulation | Watch | HR monitor, accelerometer and gyroscope | Yes, BLE protocol | Not validated yet | ☆☆☆☆ |
| AlHanai et al. [112], Massachusetts Institute of Technology, 2017 | Predicting the mood using para-linguistic cues, linguistic content and the physiological state | Watch | ECG, PPG, accelerometer, gyroscope, bioimpedence, electric tissue impedance, EDA, ST, audio and text features | Yes, BLE protocol | Not validated yet | ☆☆☆☆☆ |
| Northrup et al., [113], The Center for Discovery, USA, 2016 | Monitoring stress to coordinate emotional arousal with contexts in ASD population | wristband | Electrodermal Activity (EDA) | Yes, BLE protocol | Preliminary tests showed good results [113] | ☆☆☆☆ |
| Simm et al. [114], Lancaster University, 2017 | Assessing anxiety | Wristband | HR, ST, and environmental sensing | No | Preliminary tests on adults with ASD [114] | ☆☆☆☆☆ |
| Hui et al. [109], University of Reading, 2018 | Emotional monitoring and assessment | Glove | Blood volume pulse, EDA, ST | Yes, using BLE to phone and data uploaded to cloud using WiFi/Cellular | Not validated | ☆☆☆☆ |
| Koo et al. [58], Konkuk University & University of California, Davis, 2018 | Monitoring Emotional states for people with ASD | Glove | EDA, HR and HRV using pulse oximeter | Yes, using BLE protocol | Not validated | ☆☆☆ |
| Solution Name, Company | Purpose | Device Form Factor | Sensors/Parameters | Support for Wireless Technology | Clinical Validity | Suitability for Individuals with ASD |
|---|---|---|---|---|---|---|
| MyFeel wristband, Sentio Solutions Inc., Palo Alto, CA, USA [131] | Recognizing emotions | Wristband | HR, EDA, ST | Yes, using BLE | Preliminary study showed usefulness on 150 subjects | ☆☆☆☆ |
| Reveal, Awake Labs, Toronto, ON, Canada [34] | Monitoring stress and anxiety in ASD | Wristband | HR, EDA, ST | Yes, using BLE | Undergoing clinical validation and pilot study [132] | ☆☆☆☆☆ |
| Gaia Wearable, Gaia LLC., Las Vegas, NV, USA [118] | Monitoring stress, notifying individuals with ASD of meltdowns | Shirt | HR, EDA | Yes, using BLE | Not validated | ☆☆☆☆☆ |
| Dymaxia Wearable, Dymaxia, Inc., Toronto, ON, Canada [133] | Analyzing anxiety trends in ASD | Wristband | HR | Yes, using BLE | Not validated | ☆☆☆ |
| BioHug Vest, Bio Hug Technologies Ltd., [122] | Calming people with extreme levels of stress in people with ADHD, post-traumatic stress disorders (PTSD), ASD | Vest | Air pressure using Deep Pressure patent technology | Yes, using BLE | Not validated | ☆☆☆☆☆ |
| MOXO Wearable, mPath, Broomfield, CO, USA [134] | Monitoring stress levels | Watch | Electrodermal activity | No known | Preliminary studies showed good results [119] | ☆ |
| iBreve, iBreve, Ltd., Dublin, Ireland [135] | Monitoring breathing, stress, and activity for women | Clip | RR, HRV, and acceleration | Yes, using BLE | Initial tests to distinguish between different breathing [136] | ☆☆☆ |
| KiddoWear band, Good Parents Inc., San Francisco, CA, USA [137] | Monitoring health, stress, activity, and sleep levels | Wristband for kids | HR, ST, EDA, and acceleration | Yes, using BLE | Not validated yet | ☆☆☆☆ |
| Spire Health Tag, Spire, St. Louis, MO, USA [138] | Monitoring respiration and HR, sleep quality | Tag | HR, RR, and acceleration | Yes, using BLE | Undergoing clinical trial [139] | ☆☆☆☆☆ |
| SKIIN Smart Clothing, Myant Inc., Toronto, ON, Canada [140] | Monitoring respiration heart rate, and sleep | Textile clothing | HR, ST, RR and acceleration | Yes, using BLE | Not validated yet | ☆☆☆☆☆ |
| U-Check-It, Bedell Innovations, LLC., PA, USA [141] | Monitoring stress level, stress relief | Wristband | EDA, body temperature, acceleration | Yes, using BLE | Not validated yet | ☆☆☆☆ |
| Moodmetric Smart Ring, Vigofere Oy, Tampere, Finland [142] | Monitoring stress level | Ring | Electrodermal activity | Yes, using BLE | Not validated yet | ☆☆☆☆ |
| TouchPoints wristband, Touchpoint Solution™, [120] | Stress and anxiety relief | Wristband | Bi-lateral alternating stimulation –tactile (BLAST) | No | Patent-pending neuro-scientific technology | ☆☆☆☆ |
| The Pip, Galvanic, Inc., Calgary, AB, Canada [143] | Monitoring and reducing stress level | Teardrop-shape | Electrodermal activity | Yes, using BLE | Not validated yet | ☆ |
| Thync, Thync Global Inc., USA [126] | Improving mood (reduce stress, relax, and help in sleep) | Wearable on forehead | Neurostimulation (TENS and tDCS) | Yes, using BLE | Not validated yet | ☆ |
| Muse, InteraXon [129] | Monitoring brain activity and stress-relief | Headband | EEG sensor | Yes, using BLE | Preliminary tests [144] and undergoing clinical trial [139] | ☆ |
| HeartGuide, Omron Healthcare, Milton Keynes, UK [130] | Monitoring blood pressure, heart rate, sleep quality | Wristband | Optical blood pressure sensor | Yes, using BLE | Undergoing clinical validation but not validated yet. | ☆☆☆☆ |
| Leaf Urban, Bellabeat, San Francisco, CA, USA [145] | Stress prediction, sleep monitoring | Bracelet, necklace | Monitor activity, sleep, and RR | Yes, using BLE | Not validated yet | ☆☆☆ |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Taj-Eldin, M.; Ryan, C.; O’Flynn, B.; Galvin, P. A Review of Wearable Solutions for Physiological and Emotional Monitoring for Use by People with Autism Spectrum Disorder and Their Caregivers. Sensors 2018, 18, 4271. https://doi.org/10.3390/s18124271
Taj-Eldin M, Ryan C, O’Flynn B, Galvin P. A Review of Wearable Solutions for Physiological and Emotional Monitoring for Use by People with Autism Spectrum Disorder and Their Caregivers. Sensors. 2018; 18(12):4271. https://doi.org/10.3390/s18124271
Chicago/Turabian StyleTaj-Eldin, Mohammed, Christian Ryan, Brendan O’Flynn, and Paul Galvin. 2018. "A Review of Wearable Solutions for Physiological and Emotional Monitoring for Use by People with Autism Spectrum Disorder and Their Caregivers" Sensors 18, no. 12: 4271. https://doi.org/10.3390/s18124271
APA StyleTaj-Eldin, M., Ryan, C., O’Flynn, B., & Galvin, P. (2018). A Review of Wearable Solutions for Physiological and Emotional Monitoring for Use by People with Autism Spectrum Disorder and Their Caregivers. Sensors, 18(12), 4271. https://doi.org/10.3390/s18124271





