Label-Free and Redox Markers-Based Electrochemical Aptasensors for Aflatoxin M1 Detection
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents and Aptamers
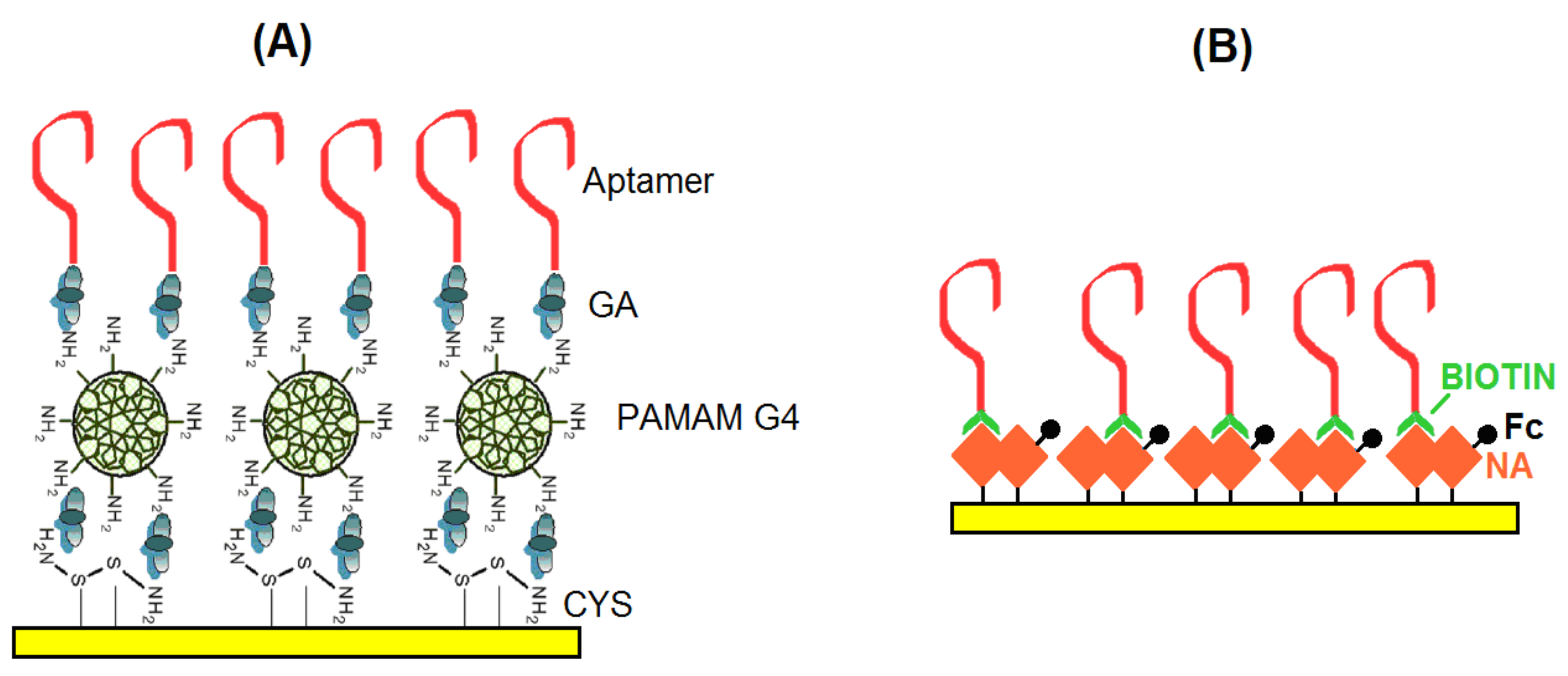
2.2. Preparation of Aptasensors
2.2.1. Aptasensors Based on Immobilization Aptamers at Dendrimer Modified Surface
2.2.2. Aptasensors Based on Biotinylated Aptamers Immobilized on Neutravidin Layers
2.3. Electrochemical Measurements
2.3.1. Instrumentation
2.3.2. Validation of the Aptasensors in Milk Samples
3. Results and Discussion
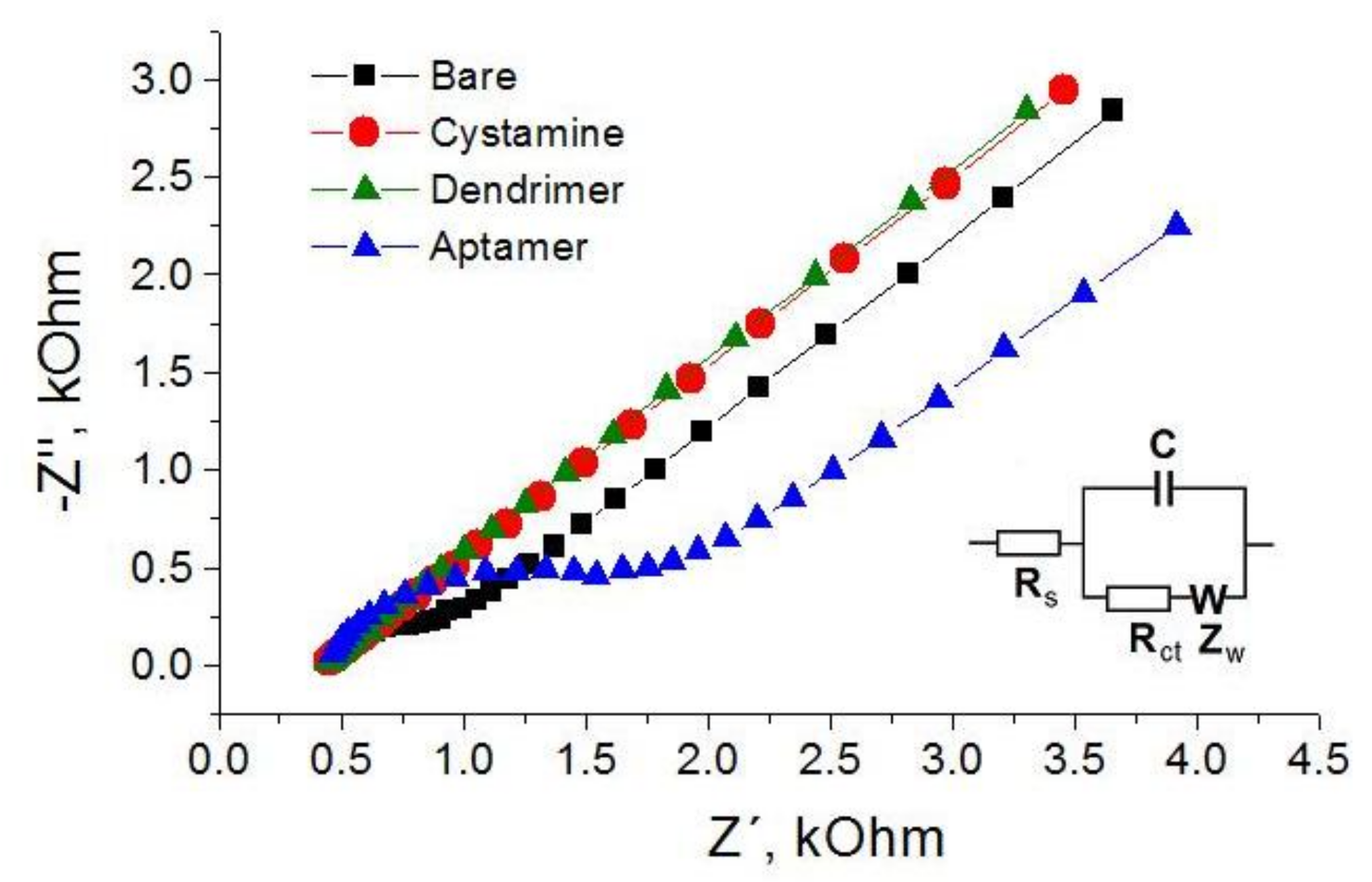
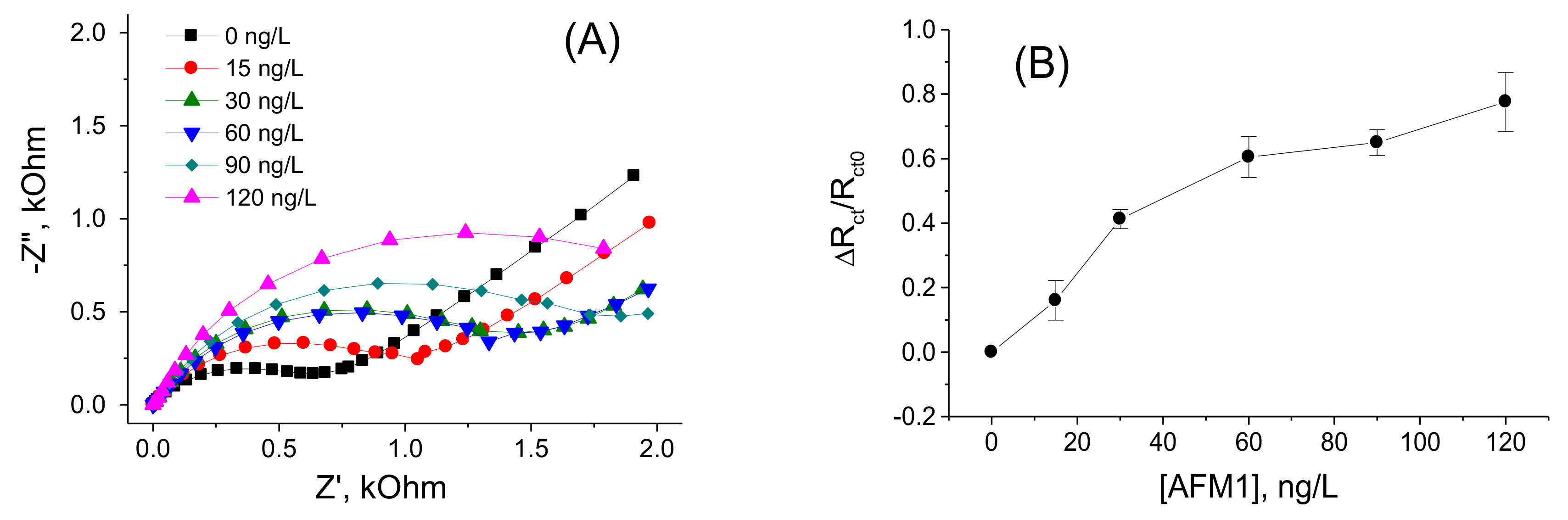
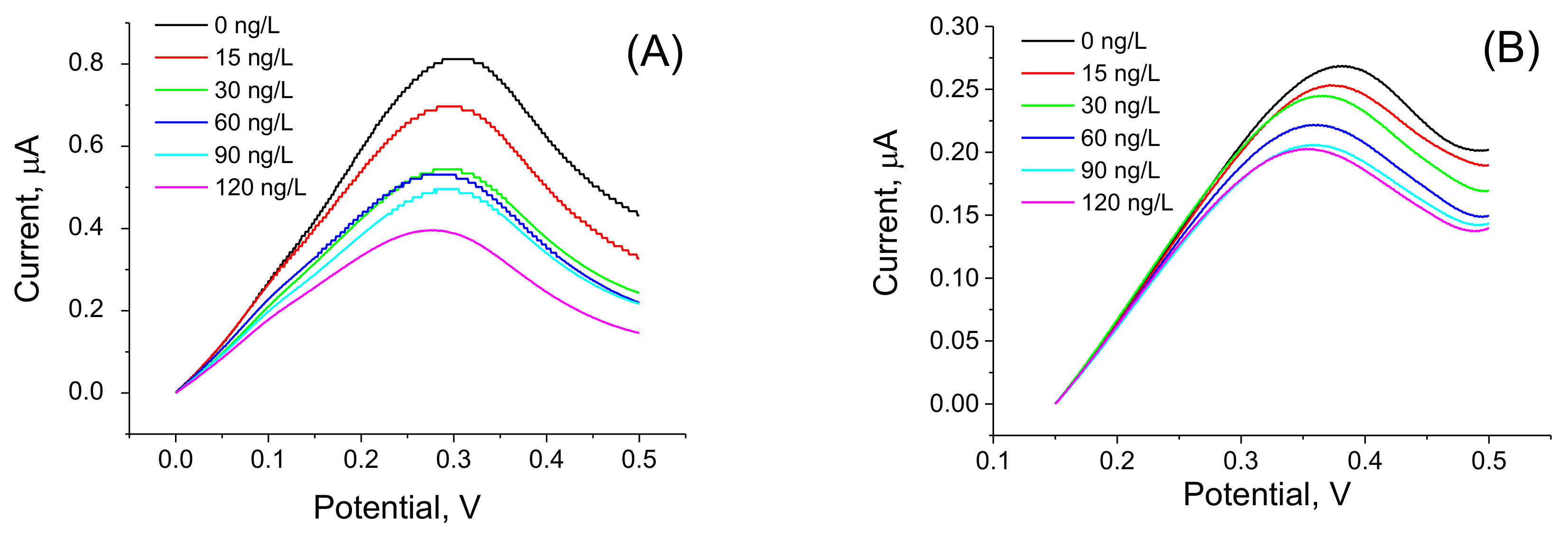
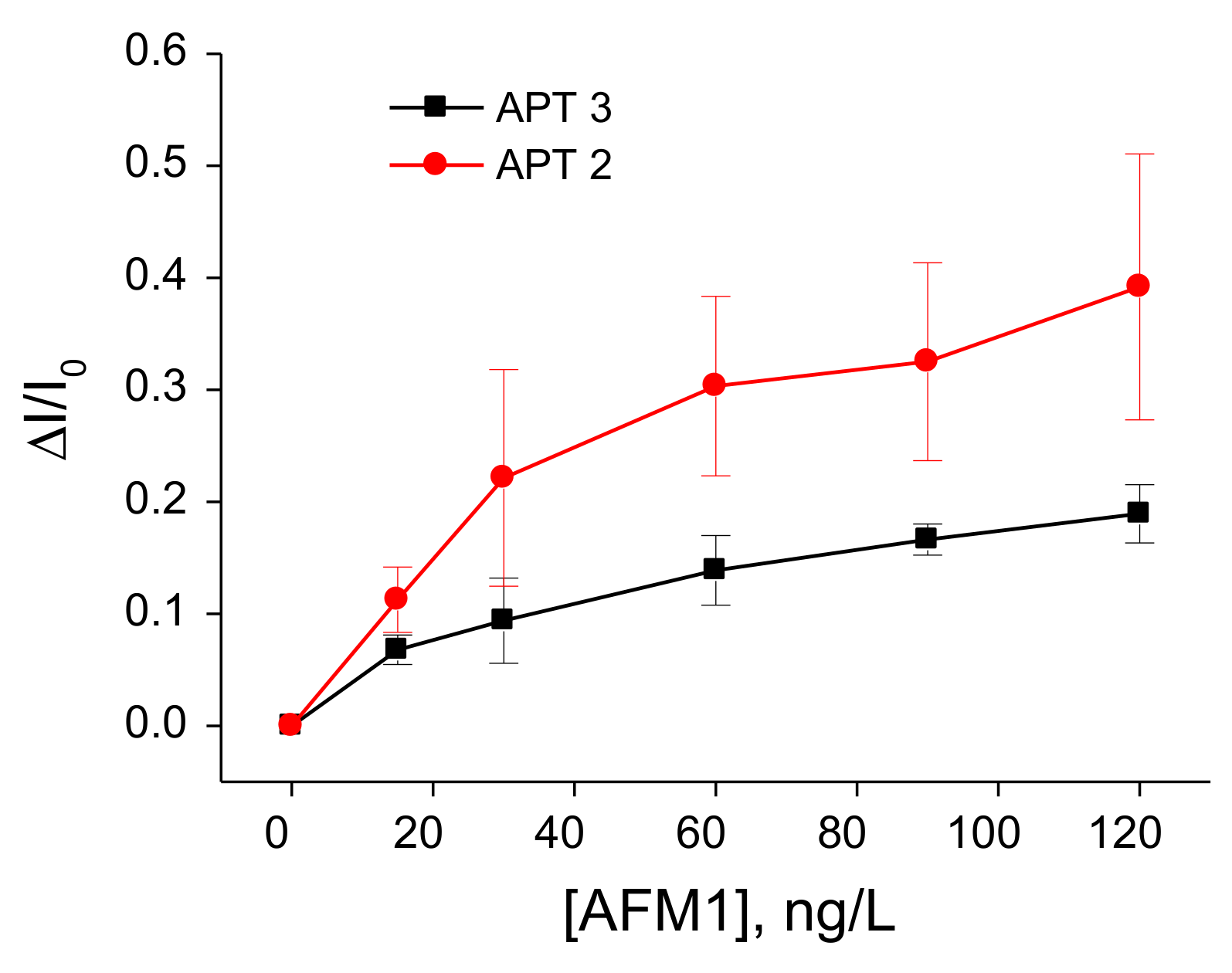
3.1. Determination of AFM1 by Aptasensors Depending on Aptamer Immobilization and Detection
3.2. Comparison of the Sensitivity of Immuno- and Aptasensors in AFM1 Detection
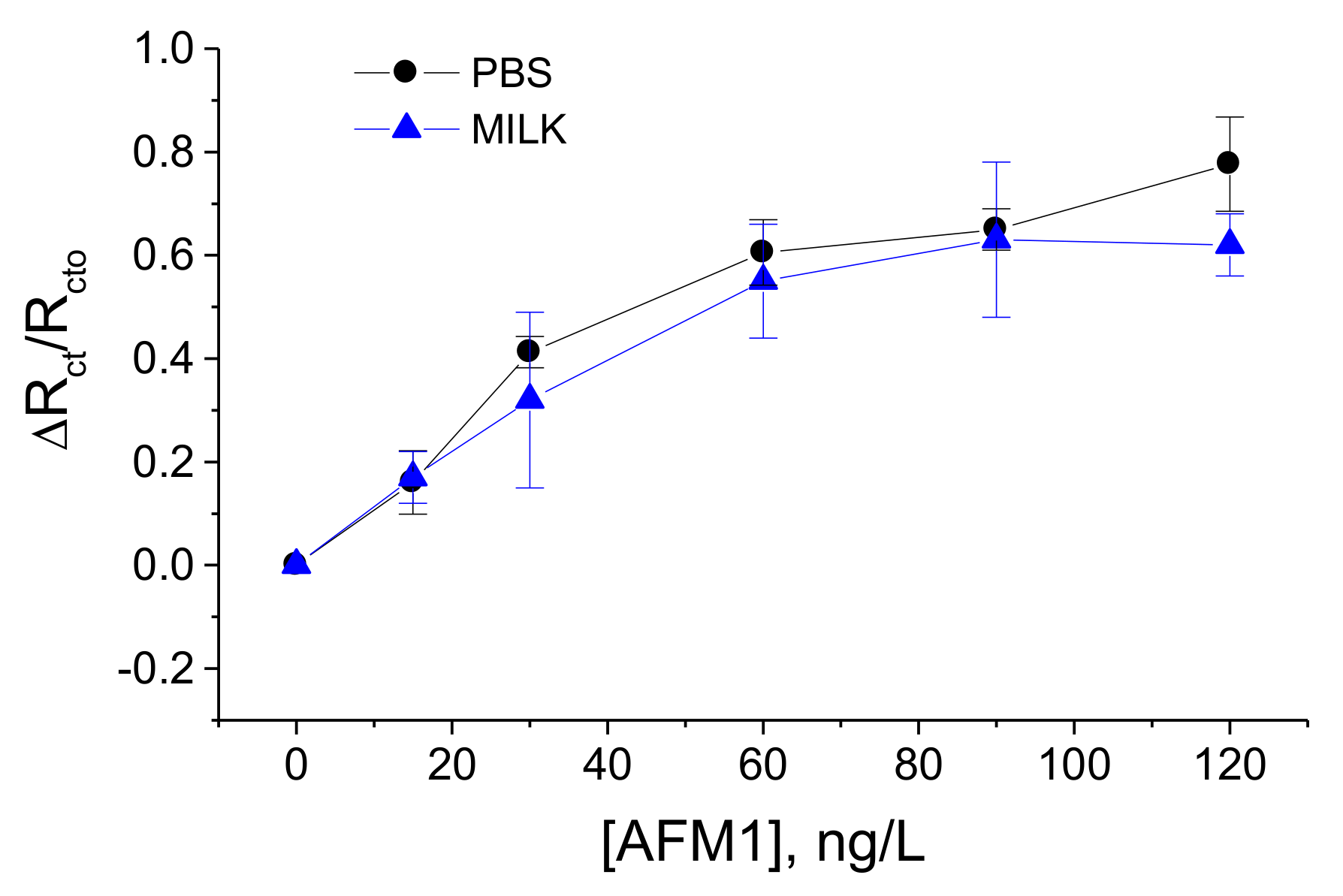
3.3. Validation of the Biosensors in Spiked Milk Samples
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Richard, J.L. Some major mycotoxins and their mycotoxicosis—An overview. Int. J. Food Microbiol. 2007, 119, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Bennett, J.W.; Klich, M. Mycotoxins. Microbiol. Rev. 2003, 16, 497–516. [Google Scholar] [CrossRef]
- Hussein, H.; Brasel, J. Toxicity, metabolism and impact of mycotoxins on human and animals. Toxicology 2001, 167, 101–134. [Google Scholar] [CrossRef]
- Fallah, A.A. Assessment of aflatoxin M1 contamination in pasteurized and UHT milk marketed in central part of Iran. Food Chem. Toxicol. 2010, 48, 988–991. [Google Scholar] [CrossRef] [PubMed]
- Prandini, A.; Tansini, G.; Sigolo, S.; Filippi, L.; Laporta, M.; Piva, G. On the occurrence of aflatoxin M1 in milk and dairy products. Food Chem. Toxicol. 2009, 47, 984–991. [Google Scholar] [CrossRef] [PubMed]
- Beltrán, E.; Ibáñez, M.; Sancho, J.V.; Cortés, M.Á.; Yus, V.; Hernández, F. UHPLC-MS/MS highly sensitive determination of aflatoxins, the aflatoxin metabolite M1 and ochratoxin A in baby food and milk. Food Chem. 2011, 126, 737–744. [Google Scholar] [CrossRef]
- Bognanno, M.; La Fauci, L.; Ritieni, A.; Tafuri, A.; De Lorenzo, A.; Micari, P.; Di Renzo, L.; Ciappellano, S.; Sarullo, V.; Galvano, F. Survey of the occurrence of Aflatoxin M1 in ovine milk by HPLC and its confirmation by MS. Mol. Nutr. Food Res. 2006, 50, 300–305. [Google Scholar] [CrossRef] [PubMed]
- Rastogi, S.; Dwivedi, P.D.; Khanna, S.K.; Das, M. Detection of aflatoxin M1 contamination in milk and infant milk products from Indian markets by ELISA. Food Control 2004, 15, 287–290. [Google Scholar] [CrossRef]
- Tuerk, C.; Gold, L. Systematic evolution of ligands by exponential enrichment: RNA ligands to bacteriophage T4 DNA polymerase. Science 1990, 249, 505–510. [Google Scholar] [CrossRef]
- Ellington, A.D.; Szostak, J.W. In vitro selection of RNA molecules that bind specific ligands. Nature 1990, 346, 818–822. [Google Scholar] [CrossRef]
- Hianik, T. Affinity biosensors for detection immunoglobulin E and cellular prions. Antibodies vs. DNA aptamers. Electroanalysis 2016, 28, 1764–1776. [Google Scholar] [CrossRef]
- Micheli, L.; Grecco, R.; Badea, M.; Moscone, D.; Palleschi, G. An electrochemical immunosensor for aflatoxin M1 determination in milk using screen-printed electrodes. Biosens. Bioelectron. 2005, 21, 588–596. [Google Scholar] [CrossRef] [PubMed]
- Vig, A.; Radoi, A.; Muñoz-Berbel, X.; Gyemant, G.; Marty, J.-L. Impedimetric aflatoxin M1 immunosensor based on colloidal gold and silver electrodeposition. Sens. Actuators B Chem. 2009, 138, 214–220. [Google Scholar] [CrossRef]
- Parker, C.O.; Tothill, I.E. Development of an electrochemical immunosensor for aflatoxin M1 in milk with focus on matrix interference. Biosens. Bioelectron. 2009, 24, 2452–2457. [Google Scholar] [CrossRef] [PubMed]
- Neagu, D.; Perrino, S.; Micheli, L.; Palleschi, G.; Moscone, D. Aflatoxin M1 determination and stability study in milk samples using a screen-printed 96-well electrochemical microplate. Int. Dairy J. 2009, 19, 753–758. [Google Scholar] [CrossRef]
- Paniel, N.; Radoi, A.; Marty, J.L. Development of an electrochemical biosensor for the detection of aflatoxin M1 in milk. Sensors 2010, 10, 9439–9448. [Google Scholar] [CrossRef] [PubMed]
- Bacher, G.; Pal, S.; Kanungo, L.; Bhand, S. A label-free silver wire based impedimetric immunosensor for detection of aflatoxin M1 in milk. Sens. Actuators B Chem. 2012, 168, 223–230. [Google Scholar] [CrossRef]
- Zhang, X.; Liu, L.; Chen, X.; Kuang, H.; Song, S.; Xu, C. Immunochromatographic strip development for ultrasensitive analysis of aflatoxin M1. Anal. Methods 2013, 5, 6567–6571. [Google Scholar] [CrossRef]
- Karczmarczyk, A.; Baeumner, A.J.; Feller, K.H. Rapid and sensitive inhibition-based assay for the electrochemical detection of ochratoxin A and aflatoxin M1 in red wine and milk. Electrochim. Acta 2017, 243, 82–89. [Google Scholar] [CrossRef]
- Chalyan, T.; Pasquardini, L.; Gandolfi, D.; Guider, R.; Samusenko, A.; Zanetti, M.; Pucker, G.; Pederzolli, C.; Pavesi, L. Aptamer-and Fab’-functionalized microring resonators for aflatoxin M1 detection. IEEE J. Sel. Top. Quantum Electron. 2017, 23, 350–357. [Google Scholar] [CrossRef]
- Sharma, A.; Goud, K.; Hayat, A.; Bhand, S.; Marty, J. Recent advances in electrochemical-based sensing platforms for aflatoxins detection. Chemosensors 2016, 5, 1. [Google Scholar] [CrossRef]
- Rhouati, A.; Catanante, G.; Nunes, G.; Hayat, A.; Marty, J.L. Label-free aptasensors for the detection of mycotoxins. Sensors 2016, 16, 2178. [Google Scholar] [CrossRef] [PubMed]
- Rapini, R.; Marrazza, G. Electrochemical aptasensors for contaminants detection in food and environment: Recent advances. Bioelectrochemistry 2017, 118, 47–61. [Google Scholar] [CrossRef] [PubMed]
- Gurban, A.M.; Epure, P.; Oancea, F.; Doni, M. Achievements and prospects in electrochemical-based biosensing platforms for aflatoxin M1 detection in milk and dairy products. Sensors 2017, 17, 2951. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, B.H.; Tran, L.D.; Do, Q.P.; Le Nguyen, H.; Tran, N.H.; Nguyen, P.X. Label-free detection of aflatoxin M1 with electrochemical Fe3O4/polyaniline-based aptasensor. Mater. Sci. Eng. C 2013, 33, 2229–2234. [Google Scholar] [CrossRef] [PubMed]
- Pandey, A.K.; Rajput, Y.S.; Sharma, R.; Singh, D. Immobilized aptamer on gold electrode senses trace amount of aflatoxin M1. Appl. Nanosci. 2017, 7, 893–903. [Google Scholar] [CrossRef] [PubMed]
- Istamboulié, G.; Paniel, N.; Zara, L.; Granados, L.R.; Barthelmebs, L.; Noguer, T. Development of an impedimetric aptasensor for the determination of aflatoxin M1 in milk. Talanta 2016, 146, 464–469. [Google Scholar] [CrossRef]
- Smolko, V.; Shurpik, D.; Porfireva, A.; Evtugyn, G.; Stoikov, I.; Hianik, T. Electrochemical aptasensor based on poly (Neutral Red) and carboxylated pillar[5]arene for sensitive determination of aflatoxin M1. Electroanalysis 2018, 30, 486–496. [Google Scholar] [CrossRef]
- Ghanim Al-Rubaye, A.; Nabok, A.; Catanante, G.; Marty, J.-L.; Takács, E.; Székács, A.; Ghanim Al-Rubaye, A.; Nabok, A.; Catanante, G.; Marty, J.-L.; et al. Label-free optical detection of mycotoxins using specific aptamers immobilized on gold nanostructures. Toxins 2018, 10, 291. [Google Scholar] [CrossRef]
- Jalalian, S.H.; Ramezani, M.; Danesh, N.M.; Alibolandi, M.; Abnous, K.; Taghdisi, S.M. A novel electrochemical aptasensor for detection of aflatoxin M1 based on target-induced immobilization of gold nanoparticles on the surface of electrode. Biosens. Bioelectron. 2018, 117, 487–492. [Google Scholar] [CrossRef]
- Fojta, M.; Daňhel, A.; Havran, L.; Vyskočil, V. Recent progress in electrochemical sensors and assays for DNA damage and repair. Trends in Anal. Chem. 2016, 79, 160–167. [Google Scholar] [CrossRef]
- Siontorou, C.G.; Nikolelis, D.P.; Miernik, A.; Krull, U.J. Rapid methods for detection of Aflatoxin M1 based on electrochemical transduction by self-assembled metal-supported bilayer lipid membranes (s-BLMs) and on interferences with transduction of DNA hybridization. Electrochim. Acta 1998, 43, 3611–3617. [Google Scholar] [CrossRef]
- Hamula, C.L.A.; Guthrie, J.W.; Zhang, H.; Li, X.F.; Le, X.C. Selection and analytical applications of aptamers. TrAC Trends Anal. Chem. 2006, 25, 681–691. [Google Scholar] [CrossRef]
- Fischer, L.M.; Tenje, M.; Heiskanen, A.R.; Masuda, N.; Castillo, J.; Bentien, A.; Émneus, J.; Jakobsen, M.H.; Boisen, A. Gold cleaning methods for electrochemical detection applications. Microelectron. Eng. 2009, 86, 1282–1285. [Google Scholar] [CrossRef]
- Castillo, G.; Spinella, K.; Poturnayová, A.; Šnejdárková, M.; Mosiello, L.; Hianik, T. Detection of aflatoxin B1 by aptamer-based biosensor using PAMAM dendrimers as immobilization platform. Food Control 2015, 52, 9–18. [Google Scholar] [CrossRef]
- Hianik, T.; Wang, J. Electrochemical aptasensors—Recent achievements and perspectives. Electroanalysis 2009, 21, 1223–1235. [Google Scholar] [CrossRef]
- Tassew, N.; Thompson, M. Kinetic characterization of TAR RNA-Tat peptide and neomycin interactions by acoustic wave biosensor. Biophys. Chem. 2003, 106, 241–252. [Google Scholar] [CrossRef]
- Radi, A.-E.; Acero Sánchez, J.L.; Baldrich, E.; O’Sullivan, C.K. Reagentless, reusable, ultrasensitive electrochemical molecular beacon aptasensor. J. Am. Chem. Soc. 2006, 128, 117–124. [Google Scholar] [CrossRef]
- Castillo, G.; Lamberti, I.; Mosiello, L.; Hianik, T. Impedimetric DNA aptasensor for sensitive detection of ochratoxin A in food. Electroanalysis 2012, 24, 512–520. [Google Scholar] [CrossRef]
- Miodek, A.; Castillo, G.; Hianik, T.; Korri-Youssoufi, H. Electrochemical aptasensor of human cellular prion based on multiwalled carbon nanotubes modified with dendrimers: A platform for connecting redox markers and aptamers. Anal. Chem. 2013, 85, 7704–7712. [Google Scholar] [CrossRef]
- Snejdarkova, M.; Svobodova, L.; Polohova, V.; Hianik, T. The study of surface properties of an IgE-sensitive aptasensor using an acoustic method. Anal. Bioanal. Chem. 2008, 390, 1087–1091. [Google Scholar] [CrossRef] [PubMed]
- Parker, C.O.; Lanyon, Y.H.; Manning, M.; Arrigan, D.W.M.; Tothill, I.E. Electrochemical immunochip sensor for aflatoxin M1 detection. Anal. Chem. 2009, 81, 5291–5298. [Google Scholar] [CrossRef] [PubMed]
| 1 | Immunosensors Immunosensors Microelectrode array immunosensor with antibodies immobilized by cross-linking with 1,4-phenylene diisothiocyanate. DNA Sensors Aptasensors 6–60 1.98 [25] |
| Sensor Preparation | Method of Detection | Dynamic Range, ng/L | LOD, ng/L | Reference |
|---|---|---|---|---|
| Immunosensors | ||||
| Immobilization of the specific antibodies on the screen-printed electrode | ELISA | 30–160 | 25 | [12] |
| An amperometric immunosensor based on the gold-labeled antibodies immobilized at screen-printed electrodes | ELISA | 15–1000 | 15 | [13] |
| A screen-printed electrode array adapted with a standard 96-well microplate | ELISA | 5–250 | 1 | [15] |
| A sensor based on a competitive immunoassay using horseradish peroxidase (HRP) | Chronoamperometry | 10–500 | 10 | [16] |
| A label-free impedimetric immunosensor based on silver wire electrode | ELISA | 1–100 | 1 | [17] |
| Immunochromatographic strip with immobilized AFB1–bovine serum albumin as the immobilized antigen and anti-AFM1 antibody labeled with gold nanoparticles as tracers | ELISA | - | 200 | [18] |
| An electrochemical immunosensor with capture antibodies immobilized on the gold screen-printed electrode. Competitive assay | DPV | - | 37 | [19] |
| Microelectrode array immunosensor with antibodies immobilized by cross-linking with 1,4-phenylene diisothiocyanate. | ELISA | 1–100 | 8 | [42] |
| Antigen-binding fragments (Fab’) immobilized on silicon oxynitride micro ring resonators | MRR | - | 1641 | [20] |
| DNA Sensors | ||||
| Metal-supported bilayer lipid membranes (s-BLMs) | Amperometry | 0.5–6572 | 157 | [31] |
| Aptasensors | ||||
| Aptasensor with electrochemical Fe3O4/PANi interface | DPV | 6–60 | 1.98 | [25] |
| Aptasensor based on biotin-modified aptamer at streptavidin layer on a screen-printed electrode | CV, SWV | 1–105 | 1 | [26] |
| Hexaethyleneglycol-modified aptamers immobilized on a carbon screen-printed electrode | CV, EIS | 2-150 | 1.15 | [27] |
| Neutral Red electropolymerized film modified by pillar[5]arene | EIS | 5–120 | 0.5 | [28] |
| Optical label-free. Aptamers immobilized on nanostructured Au films | TIRE with LSRP | 10–105 | 10 | [29] |
| Hairpin-shaped aptamer immobilized on gold nanoparticles. Methylene blue as a redox probe | DPV | 2–600 | 0.9 | [30] |
| Aptamers immobilized at PAMAM dendrimers | EIS | 15–120 | 8.47 | This work |
| Biotinylated aptamers immobilized at neutravidin layer modified by ferrocene | DPV | 15–120 | 8.52 | This work |
| Concentration of AFM1, ng/L | Buffer ΔRct/Rcto | Milk ΔRct/Rcto | Recovery, % |
|---|---|---|---|
| 15 | 0.16 | 0.17 | 106.25% |
| 30 | 0.41 | 0.32 | 78.04% |
| 60 | 0.61 | 0.55 | 90.16% |
| 90 | 0.65 | 0.63 | 97.00% |
| 120 | 0.78 | 0.62 | 79.5% |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Karapetis, S.; Nikolelis, D.; Hianik, T. Label-Free and Redox Markers-Based Electrochemical Aptasensors for Aflatoxin M1 Detection. Sensors 2018, 18, 4218. https://doi.org/10.3390/s18124218
Karapetis S, Nikolelis D, Hianik T. Label-Free and Redox Markers-Based Electrochemical Aptasensors for Aflatoxin M1 Detection. Sensors. 2018; 18(12):4218. https://doi.org/10.3390/s18124218
Chicago/Turabian StyleKarapetis, Stefanos, Dimitrios Nikolelis, and Tibor Hianik. 2018. "Label-Free and Redox Markers-Based Electrochemical Aptasensors for Aflatoxin M1 Detection" Sensors 18, no. 12: 4218. https://doi.org/10.3390/s18124218
APA StyleKarapetis, S., Nikolelis, D., & Hianik, T. (2018). Label-Free and Redox Markers-Based Electrochemical Aptasensors for Aflatoxin M1 Detection. Sensors, 18(12), 4218. https://doi.org/10.3390/s18124218







