Hemodialysis Impact on Motor Function beyond Aging and Diabetes—Objectively Assessing Gait and Balance by Wearable Technology
Abstract
1. Introduction
2. Methods
2.1. Study Population
2.2. Demographic and Clinical Information
2.3. Gait Test
2.4. Balance Test
2.5. Statistical Analysis
3. Results
4. Discussion
Limitations
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Obembe, A.O.; Olaogun, M.O.; Adedoyin, R. Gait and balance performance of stroke survivors in South-Western Nigeria—A cross-sectional study. Pan Afr. Med. J. 2014, 17, 6. [Google Scholar] [CrossRef] [PubMed]
- Ellis, T.; Cavanaugh, J.T.; Earhart, G.M.; Ford, M.P.; Foreman, K.B.; Dibble, L.E. Which measures of physical function and motor impairment best predict quality of life in Parkinson’s disease? Parkinsonism Relat. Disord. 2011, 17, 693–697. [Google Scholar] [CrossRef] [PubMed]
- Steffen, T.M.; Hacker, T.A.; Mollinger, L. Age-and gender-related test performance in community-dwelling elderly people: Six-Minute Walk Test, Berg Balance Scale, Timed Up & Go Test, and gait speeds. Phys. Ther. 2002, 82, 128–137. [Google Scholar] [PubMed]
- Benjuya, N.; Melzer, I.; Kaplanski, J. Aging-induced shifts from a reliance on sensory input to muscle cocontraction during balanced standing. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2004, 59, M166–M171. [Google Scholar] [CrossRef]
- Wolfson, L.I.; Whipple, R.; Amerman, P.; Kaplan, J.; Kleinberg, A. Gait and balance in the elderly: Two functional capacities that link sensory and motor ability to falls. Clin. Geriatr. Med. 1985, 1, 649–659. [Google Scholar] [CrossRef]
- Laughton, C.A.; Slavin, M.; Katdare, K.; Nolan, L.; Bean, J.F.; Kerrigan, D.C.; Phillips, E.; Lipsitz, L.A.; Collins, J.J. Aging, muscle activity, and balance control: Physiologic changes associated with balance impairment. Gait Posture 2003, 18, 101–108. [Google Scholar] [CrossRef]
- Johansen, K.L.; Chertow, G.M.; Da Silva, M.; Carey, S.; Painter, P. Determinants of physical performance in ambulatory patients on hemodialysis. Kidney Int. 2001, 60, 1586–1591. [Google Scholar] [CrossRef] [PubMed]
- Erken, E.; Ozelsancak, R.; Sahin, S.; Yılmaz, E.E.; Torun, D.; Leblebici, B.; Kuyucu, Y.E.; Sezer, S. The effect of hemodialysis on balance measurements and risk of fall. Int. Urol. Nephrol. 2016, 48, 1705–1711. [Google Scholar] [CrossRef] [PubMed]
- Shin, S.; Chung, H.R.; Fitschen, P.J.; Kistler, B.M.; Park, H.W.; Wilund, K.R.; Sosnoff, J.J. Postural control in hemodialysis patients. Gait Posture 2014, 39, 723–727. [Google Scholar] [CrossRef] [PubMed]
- Foundation, N.K. Diabetes and Chronic Kidney Disease. Available online: https://www.kidney.org/news/newsroom/factsheets/Diabetes-And-CKD (accessed on 15 June 2018).
- Eknoyan, G.; Lameire, N.; Barsoum, R.; Eckardt, K.-U.; Levin, A.; Levin, N.; Locatelli, F.; Macleod, A.; Vanholder, R.; Walker, R. The burden of kidney disease: Improving global outcomes. Kidney Int. 2004, 66, 1310–1314. [Google Scholar] [CrossRef] [PubMed]
- Lockhart, T.E.; Barth, A.T.; Zhang, X.; Songra, R.; Abdel-Rahman, E.; Lach, J. Portable, non-invasive fall risk assessment in end stage renal disease patients on hemodialysis. In Wireless Health; ACM: New York, NY, USA, 2010; pp. 84–93. [Google Scholar]
- Soangra, R.; Lockhart, T.E.; Lach, J.; Abdel-Rahman, E.M. Effects of hemodialysis therapy on sit-to-walk characteristics in end stage renal disease patients. Ann. Biomed. Eng. 2013, 41, 795–805. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Sabbagh, M.; Wyman, R.; Liebsack, C.; Kunik, M.E.; Najafi, B. Instrumented trail-making task to differentiate persons with no cognitive impairment, amnestic mild cognitive impairment, and Alzheimer disease: A proof of concept study. Gerontology 2017, 63, 189–200. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Lee, H.; Lee, J.; Schwenk, M.; Najafi, B. Motor planning error: Toward measuring cognitive frailty in older adults using wearables. Sensors 2018, 18, 926. [Google Scholar] [CrossRef] [PubMed]
- Lebel, K.; Nguyen, H.; Duval, C.; Plamondon, R.; Boissy, P. Capturing the Cranio-Caudal Signature of a Turn with Inertial Measurement Systems: Methods, Parameters Robustness and Reliability. Front. Bioeng. Biotechnol. 2017, 5. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, H.; Lebel, K.; Bogard, S.; Goubault, E.; Boissy, P.; Duval, C. Using Inertial Sensors to Automatically Detect and Segment Activities of Daily Living in People with Parkinson‘s Disease. IEEE Trans. Neural Syst. Rehabil. Eng. 2018, 26, 197–204. [Google Scholar] [CrossRef] [PubMed]
- Razjouyan, J.; Naik, A.D.; Horstman, M.J.; Kunik, M.E.; Amirmazaheri, M.; Zhou, H.; Sharafkhaneh, A.; Najafi, B. Wearable Sensors and the Assessment of Frailty among Vulnerable Older Adults: An Observational Cohort Study. Sensors 2018, 18, 1336. [Google Scholar] [CrossRef] [PubMed]
- Piaggesi, A.; Låuchli, S.; Bassetto, F.; Biedermann, T.; Marques, A.; Najafi, B.; Palla, I.; Scarpa, C.; Seimetz, D.; Triulzi, I. Advanced therapies in wound management: Cell and tissue based therapies, physical and bio-physical therapies smart and IT based technologies. J. Wound Care 2018, 27, S1–S137. [Google Scholar] [CrossRef] [PubMed]
- Aminian, K.; Najafi, B. Capturing human motion using body-fixed sensors: Outdoor measurement and clinical applications. Comput. Anim. Virtual Worlds 2004, 15, 79–94. [Google Scholar] [CrossRef]
- Miller, J.D.; Najafi, B.; Armstrong, D.G. Current standards and advances in diabetic ulcer prevention and elderly fall prevention using wearable technology. Curr. Geriatr. Rep. 2015, 4, 249–256. [Google Scholar] [CrossRef]
- Razjouyan, J.; Lee, H.; Parthasarathy, S.; Mohler, J.; Sharafkhaneh, A.; Najafi, B. Improving Sleep Quality Assessment Using Wearable Sensors by Including Information From Postural/Sleep Position Changes and Body Acceleration: A Comparison of Chest-Worn Sensors, Wrist Actigraphy, and Polysomnography. J. Clin. Sleep Med. 2017, 13, 1301–1310. [Google Scholar] [CrossRef] [PubMed]
- Najafi, B.; Ron, E.; Enriquez, A.; Marin, I.; Razjouyan, J.; Armstrong, D.G. Smarter Sole Survival: Will Neuropathic Patients at High Risk for Ulceration Use a Smart Insole-Based Foot Protection System? J. Diabetes Sci. Technol. 2017, 11, 702–713. [Google Scholar] [CrossRef] [PubMed]
- Razjouyan, J.; Grewal, G.S.; Talal, T.K.; Armstrong, D.G.; Mills, J.L.; Najafi, B. Does Physiological Stress Slow Down Wound Healing in Patients With Diabetes? J. Diabetes Sci. Technol. 2017, 11, 685–692. [Google Scholar] [CrossRef] [PubMed]
- Najafi, B.; Armstrong, D.G.; Mohler, J. Novel wearable technology for assessing spontaneous daily physical activity and risk of falling in older adults with diabetes. J. Diabetes Sci. Technol. 2013, 7, 1147–1160. [Google Scholar] [CrossRef] [PubMed]
- Yardley, L.; Beyer, N.; Hauer, K.; Kempen, G.; Piot-Ziegler, C.; Todd, C. Development and initial validation of the Falls Efficacy Scale-International (FES-I). Age Ageing 2005, 34, 614–619. [Google Scholar] [CrossRef] [PubMed]
- Orme, J.G.; Reis, J.; Herz, E.J. Factorial and discriminant validity of the Center for Epidemiological Studies Depression (CES-D) scale. J. Clin. Psychol. 1986, 42, 28–33. [Google Scholar] [CrossRef]
- Fried, L.P.; Tangen, C.M.; Walston, J.; Newman, A.B.; Hirsch, C.; Gottdiener, J.; Seeman, T.; Tracy, R.; Kop, W.J.; Burke, G. Frailty in older adults: Evidence for a phenotype. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2001, 56, M146–M157. [Google Scholar] [CrossRef]
- Young, M.J.; Breddy, J.L.; Veves, A.; Boulton, A.J. The prediction of diabetic neuropathic foot ulceration using vibration perception thresholds: A prospective study. Diabetes Care 1994, 17, 557–560. [Google Scholar] [CrossRef] [PubMed]
- Xu, D.; Li, J.; Zou, L.; Xu, Y.; Hu, D.; Pagoto, S.L.; Ma, Y. Sensitivity and specificity of the ankle—Brachial index to diagnose peripheral artery disease: A structured review. Vasc. Med. 2010, 15, 361–369. [Google Scholar]
- Organization, W.H. Use of Glycated Haemoglobin (HbA1c) in the Diagnosis of Diabetes Mellitus; The Organization Google Scholar: Geneva, Switzerland, 2011. [Google Scholar]
- Delbaere, K.; Close, J.C.; Mikolaizak, A.S.; Sachdev, P.S.; Brodaty, H.; Lord, S.R. The falls efficacy scale international (FES-I). A comprehensive longitudinal validation study. Age Ageing 2010, 39, 210–216. [Google Scholar] [CrossRef] [PubMed]
- Weissman, M.M.; Sholomskas, D.; Pottenger, M.; Prusoff, B.A.; Locke, B.Z. Assessing depressive symptoms in five psychiatric populations: A validation study. Am. J. Epidemiol. 1977, 106, 203–214. [Google Scholar] [CrossRef] [PubMed]
- Bracewell, N.; Game, F.; Jeffcoate, W.; Scammell, B. Clinical evaluation of a new device in the assessment of peripheral sensory neuropathy in diabetes. Diabetic Med. 2012, 29, 1553–1555. [Google Scholar] [CrossRef] [PubMed]
- Wyatt, M.F.; Stickrath, C.; Shah, A.; Smart, A.; Hunt, J.; Casserly, I.P. Ankle—brachial index performance among internal medicine residents. Vasc. Med. 2010, 15, 99–105. [Google Scholar] [CrossRef] [PubMed]
- Aminian, K.; Najafi, B.; Büla, C.; Leyvraz, P.-F.; Robert, P. Spatio-temporal parameters of gait measured by an ambulatory system using miniature gyroscopes. J. Biomech. 2002, 35, 689–699. [Google Scholar] [CrossRef]
- Aminian, K.; Trevisan, C.; Najafi, B.; Dejnabadi, H.; Frigo, C.; Pavan, E.; Telonio, A.; Cerati, F.; Marinoni, E.; Robert, P. Evaluation of an ambulatory system for gait analysis in hip osteoarthritis and after total hip replacement. Gait Posture 2004, 20, 102–107. [Google Scholar] [CrossRef]
- Najafi, B.; Helbostad, J.L.; Moe-Nilssen, R.; Zijlstra, W.; Aminian, K. Does walking strategy in older people change as a function of walking distance? Gait Posture 2009, 29, 261–266. [Google Scholar] [CrossRef] [PubMed]
- Lindemann, U.; Najafi, B.; Zijlstra, W.; Hauer, K.; Muche, R.; Becker, C.; Aminian, K. Distance to achieve steady state walking speed in frail elderly persons. Gait Posture 2008, 27, 91–96. [Google Scholar] [CrossRef] [PubMed]
- Grewal, G.; Sayeed, R.; Yeschek, S.; Menzies, R.A.; Talal, T.K.; Lavery, L.A.; Armstrong, D.G.; Najafi, B. Virtualizing the assessment: A novel pragmatic paradigm to evaluate lower extremity joint perception in diabetes. Gerontology 2012, 58, 463–471. [Google Scholar] [CrossRef] [PubMed]
- Najafi, B.; Miller, D.; Jarrett, B.D.; Wrobel, J.S. Does footwear type impact the number of steps required to reach gait steady state?: An innovative look at the impact of foot orthoses on gait initiation. Gait Posture 2010, 32, 29–33. [Google Scholar] [CrossRef] [PubMed]
- Najafi, B.; Horn, D.; Marclay, S.; Crews, R.T.; Wu, S.; Wrobel, J.S. Assessing Postural Control and Postural Control Strategy in Diabetes Patients Using Innovative and Wearable Technology. J. Diabetes Sci. Technol. 2010, 4, 780–791. [Google Scholar] [CrossRef] [PubMed]
- Najafi, B.; Bharara, M.; Talal, T.K.; Armstrong, D.G. Advances in balance assessment and balance training for diabetes. Diabetes Manag. 2012, 2, 293. [Google Scholar] [CrossRef]
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences, 2nd ed.; Erlbaum: Hillsdale, NJ, USA, 1988. [Google Scholar]
- Pop-Busui, R.; Roberts, L.; Pennathur, S.; Kretzler, M.; Brosius, F.C.; Feldman, E.L. The management of diabetic neuropathy in CKD. Am. J. Kidney Dis. 2010, 55, 365–385. [Google Scholar] [CrossRef] [PubMed]
- Timar, B.; Timar, R.; Gaiță, L.; Oancea, C.; Levai, C.; Lungeanu, D. The impact of diabetic neuropathy on balance and on the risk of falls in patients with type 2 diabetes mellitus: A cross-sectional study. PLoS ONE 2016, 11, e0154654. [Google Scholar] [CrossRef] [PubMed]
- Arnold, R.; Issar, T.; Krishnan, A.V.; Pussell, B.A. Neurological complications in chronic kidney disease. JRSM Cardiovasc. Dis. 2016, 5, 2048004016677687. [Google Scholar] [CrossRef] [PubMed]
- Petrofsky, J.; Lee, S.; Macnider, M.; Navarro, E. Autonomic, endothelial function and the analysis of gait in patients with type 1 and type 2 diabetes. Acta Diabetol. 2005, 42, 7–15. [Google Scholar] [CrossRef] [PubMed]
- Jhamb, M.; Weisbord, S.D.; Steel, J.L.; Unruh, M. Fatigue in patients receiving maintenance dialysis: A review of definitions, measures, and contributing factors. Am. J. Kidney Dis. 2008, 52, 353–365. [Google Scholar] [CrossRef] [PubMed]
- Floege, J.; Ehlerding, G. Beta-2-microglobulin-associated amyloidosis. Nephron 1996, 72, 9–26. [Google Scholar] [CrossRef] [PubMed]
- Lattanzio, F.; Corsonello, A.; Abbatecola, A.M.; Volpato, S.; Pedone, C.; Pranno, L.; Laino, I.; Garasto, S.; Corica, F.; Passarino, G. Relationship between renal function and physical performance in elderly hospitalized patients. Rejuv. Res. 2012, 15, 545–552. [Google Scholar] [CrossRef] [PubMed]
- Menz, H.B.; Lord, S.R.; St George, R.; Fitzpatrick, R.C. Walking stability and sensorimotor function in older people with diabetic peripheral neuropathy. Arch. Phys. Med. Rehabi. 2004, 85, 245–252. [Google Scholar] [CrossRef]
- Richardson, J.K. Factors associated with falls in older patients with diffuse polyneuropathy. J. Am. Geriatr. Soc. 2002, 50, 1767–1773. [Google Scholar] [CrossRef] [PubMed]
- Conner-Kerr, T.; Templeton, M.S. Chronic fall risk among aged individuals with type 2 diabetes. Ostomy 2002, 48, 28–34, 35. [Google Scholar]
- Johansen, K.L.; Chertow, G.M.; Ng, A.V.; Mulligan, K.; Carey, S.; Schoenfeld, P.Y.; Kent-Braun, J.A. Physical activity levels in patients on hemodialysis and healthy sedentary controls. Kidney Int. 2000, 57, 2564–2570. [Google Scholar] [CrossRef] [PubMed]
- Rhee, C.M.; Leung, A.M.; Kovesdy, C.P.; Lynch, K.E.; Brent, G.A.; Kalantar-Zadeh, K. Updates on the management of diabetes in dialysis patients. Semin. Dialysis 2014, 27, 135–145. [Google Scholar] [CrossRef] [PubMed]
- Connor, J.; Pak, C.C.; Schroit, A.J. Exposure of phosphatidylserine in the outer leaflet of human red blood cells. Relationship to cell density, cell age, and clearance by mononuclear cells. J. Biol. Chem. 1994, 269, 2399–2404. [Google Scholar] [PubMed]
- Ly, J.; Marticorena, R.; Donnelly, S. Red blood cell survival in chronic renal failure. Am. J. Kidney Dis. 2004, 44, 715–719. [Google Scholar] [CrossRef]
- Suki, W.N.; Zabaneh, R.; Cangiano, J.; Reed, J.; Fischer, D.; Garrett, L.; Ling, B.; Chasan-Taber, S.; Dillon, M.; Blair, A. Effects of sevelamer and calcium-based phosphate binders on mortality in hemodialysis patients. Kidney Int. 2007, 72, 1130–1137. [Google Scholar] [CrossRef] [PubMed]
- Vlassara, H.; Uribarri, J.; Cai, W.; Goodman, S.; Pyzik, R.; Post, J.; Grosjean, F.; Woodward, M.; Striker, G.E. Effects of sevelamer on HbA1c, inflammation, and advanced glycation end products in diabetic kidney disease. Clin. J. Am. Soc. Nephrol. 2012, 7, 934–942. [Google Scholar] [CrossRef] [PubMed]
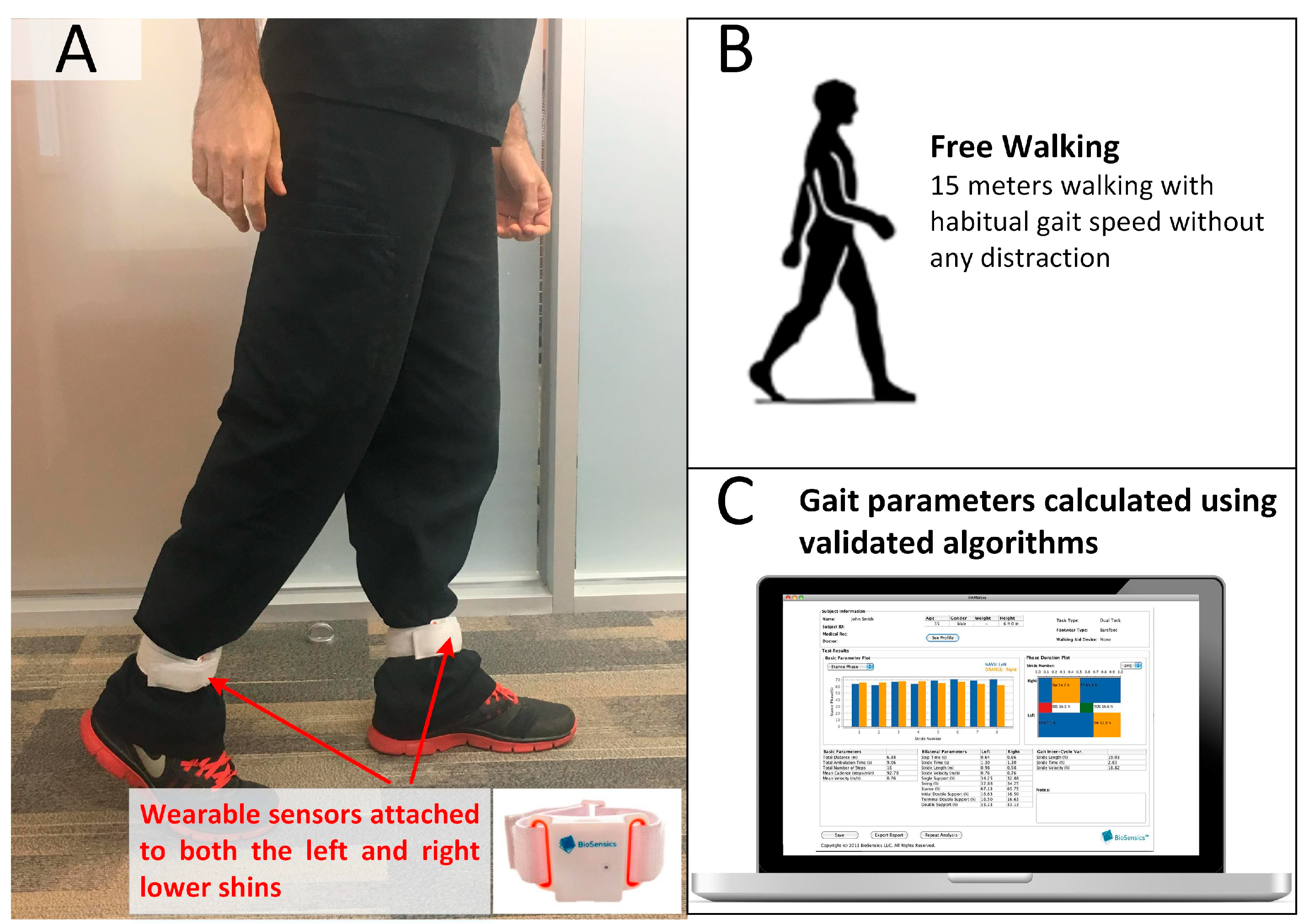
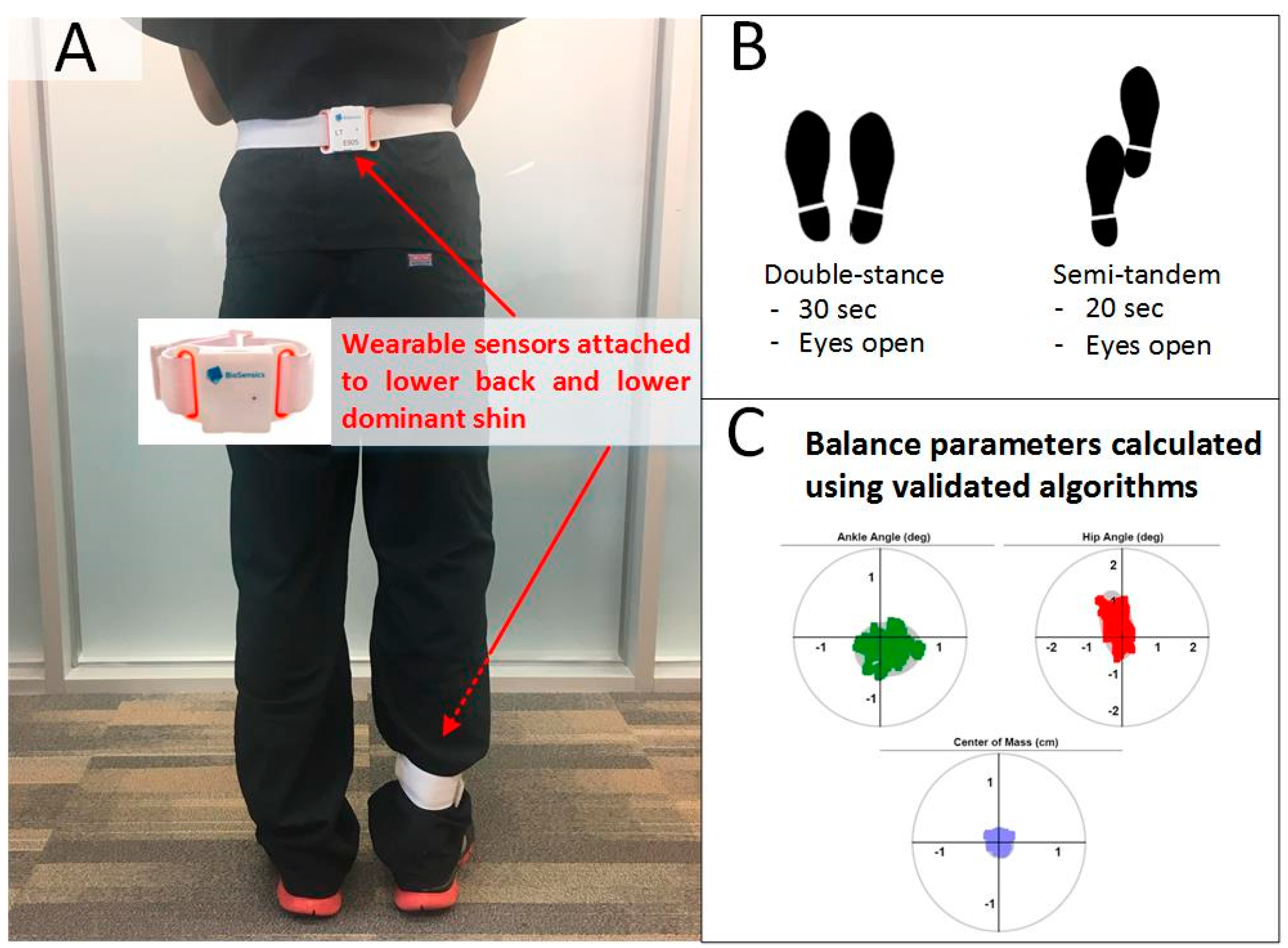
| Variable | Older Adults without Diabetes (DM−) | People with Diabetes (DM+) | p-Value | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Mid-Age Adults | Older Adults | Older DM− vs. Older DM+ | Mid-age DM+ vs. Older DM+ * | ||||||
| Total | HD− | HD+ | Total | HD− | HD+ | ||||
| Subject Number, n | 45 | 78 | 40 | 38 | 73 | 37 | 36 | - | - |
| Age, years (mean ± SD) | 73.4 ± 6.8 | 57.2 ± 4.2 | 56.5 ± 4.2 | 58.1 ± 4.1 | 71.4 ± 5.4 | 71.3 ± 4.6 | 71.5 ± 6.1 | 0.073 | <0.001 |
| Female, % | 71% | 51% | 55% | 47% | 52% | 49% | 56% | 0.041 | 0.924 |
| BMI, kg/m2 (mean ± SD) | 27.1 ± 5.0 | 31.1 ± 7.1 | 31.2 ± 6.1 | 31.1 ± 8.2 | 30.8 ± 5.9 | 29.9 ± 5.2 | 31.8 ± 6.5 | <0.001 | 0.780 |
| Fall History, % | 29% | 36% | 51% | 21% | 28% | 36% | 22% | 0.951 | 0.307 |
| High Concern about Falling, % | 36% | 65% | 80% | 50% | 74% | 78% | 69% | <0.001 | 0.230 |
| Depression, % | 13% | 28% | 27% | 29% | 36% | 29% | 42% | 0.005 | 0.246 |
| Frailty, % | 5% | 22% | 20% | 24% | 40% | 23% | 58% | <0.001 | 0.005 |
| Plantar Numbness, VPT (mean ± SD) | - | 32.0 ± 9.8 | 34.6 ± 8.9 | 29.4 ± 10.2 | 32.0 ± 10.1 | 35.0 ± 8.5 | 29.1 ± 10.7 | - | 0.982 |
| Diabetic Peripheral Neuropathy, % | - | 76% | 85% | 66% | 74% | 88% | 60% | - | 0.845 |
| Peripheral Artery Disease, % | - | 59% | 57% | 61% | 63% | 64% | 63% | - | 0.650 |
| HbA1c, % (mean ± SD) | - | 7.2 ± 2.2 | 7.9 ± 2.8 | 6.6 ± 1.5 | 7.0 ± 1.6 | 7.2 ± 2.0 | 6.8 ± 1.2 | - | 0.574 |
| Older DM− n = 45 | Mid-Age DM+ n = 78 | Older DM+ n = 73 | Mid-Age DM+ vs. Older DM− | Older DM+ vs. Older DM− | Older DM+ vs. Mid-Age DM+ | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Diff (%) | p-Value * | d * | Diff (%) | p-Value † | d† | Diff (%) | p-Value ‡ | d‡ | |||||
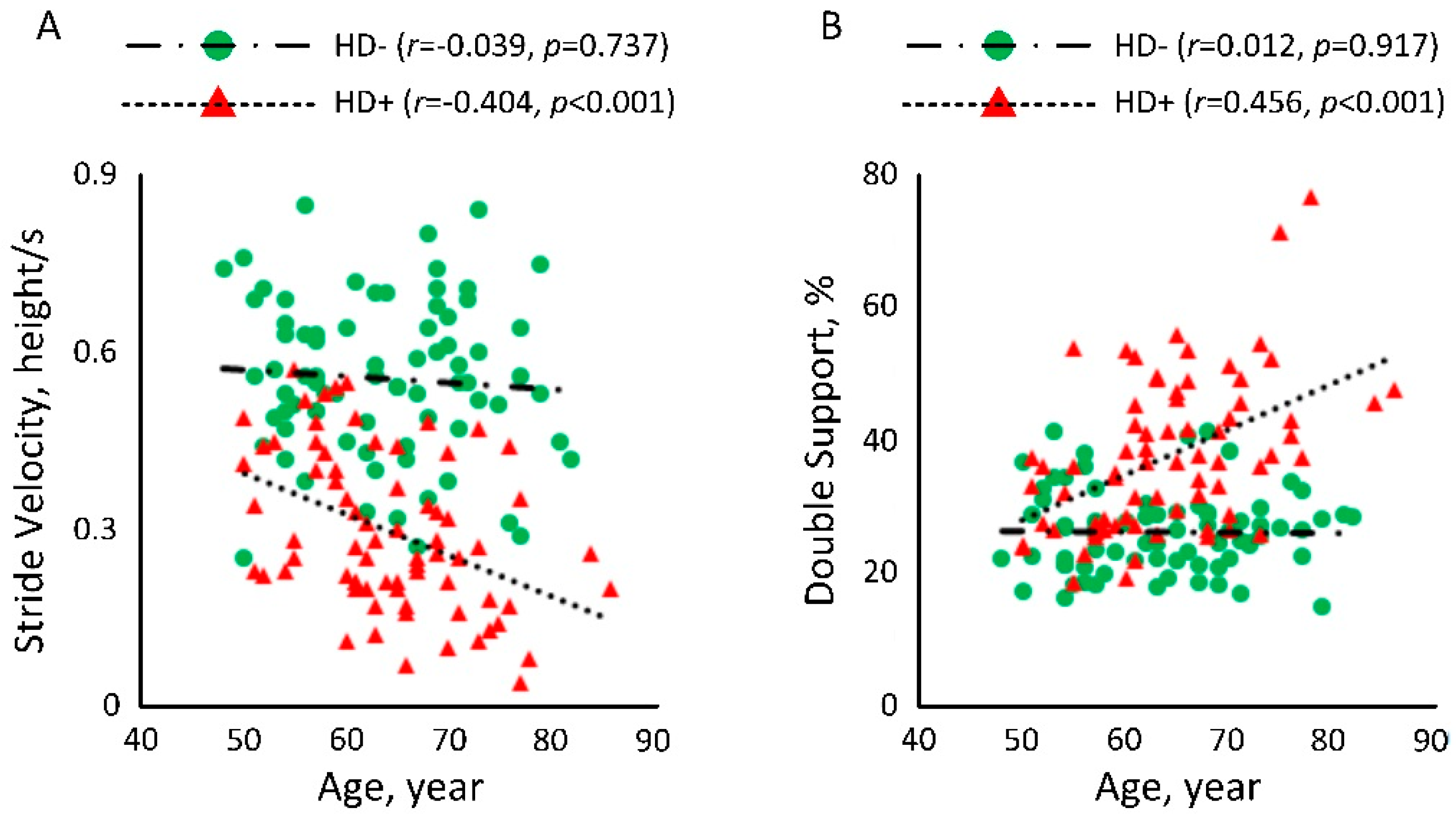
| Gait | Stride Velocity, m/s (mean ± SD) | 1.14 ± 0.17 | 0.75 ± 0.29 | 0.68 ± 0.36 | −34% | <0.001 | 1.55 | −40% | <0.001 | 1.61 | −10% | 0.171 | 0.22 |
| Normalized Stride Velocity, height/s (mean ± SD) | 0.70 ± 0.10 | 0.45 ± 0.17 | 0.40 ± 0.20 | −36% | <0.001 | 1.62 | −43% | <0.001 | 1.73 | −11% | 0.091 | 0.27 | |
| Stride Length, m (mean ± SD) | 1.23 ± 0.14 | 0.98 ± 0.31 | 0.89 ± 0.34 | −20% | <0.001 | 1.02 | −28% | <0.001 | 1.36 | −10% | 0.071 | 0.29 | |
| Normalized Stride Length, % height (mean ± SD) | 75.43 ± 7.14 | 59.42 ± 17.08 | 53.04 ± 18.42 | −21% | <0.001 | 1.11 | −30% | <0.001 | 1.53 | −11% | 0.029 | 0.36 | |
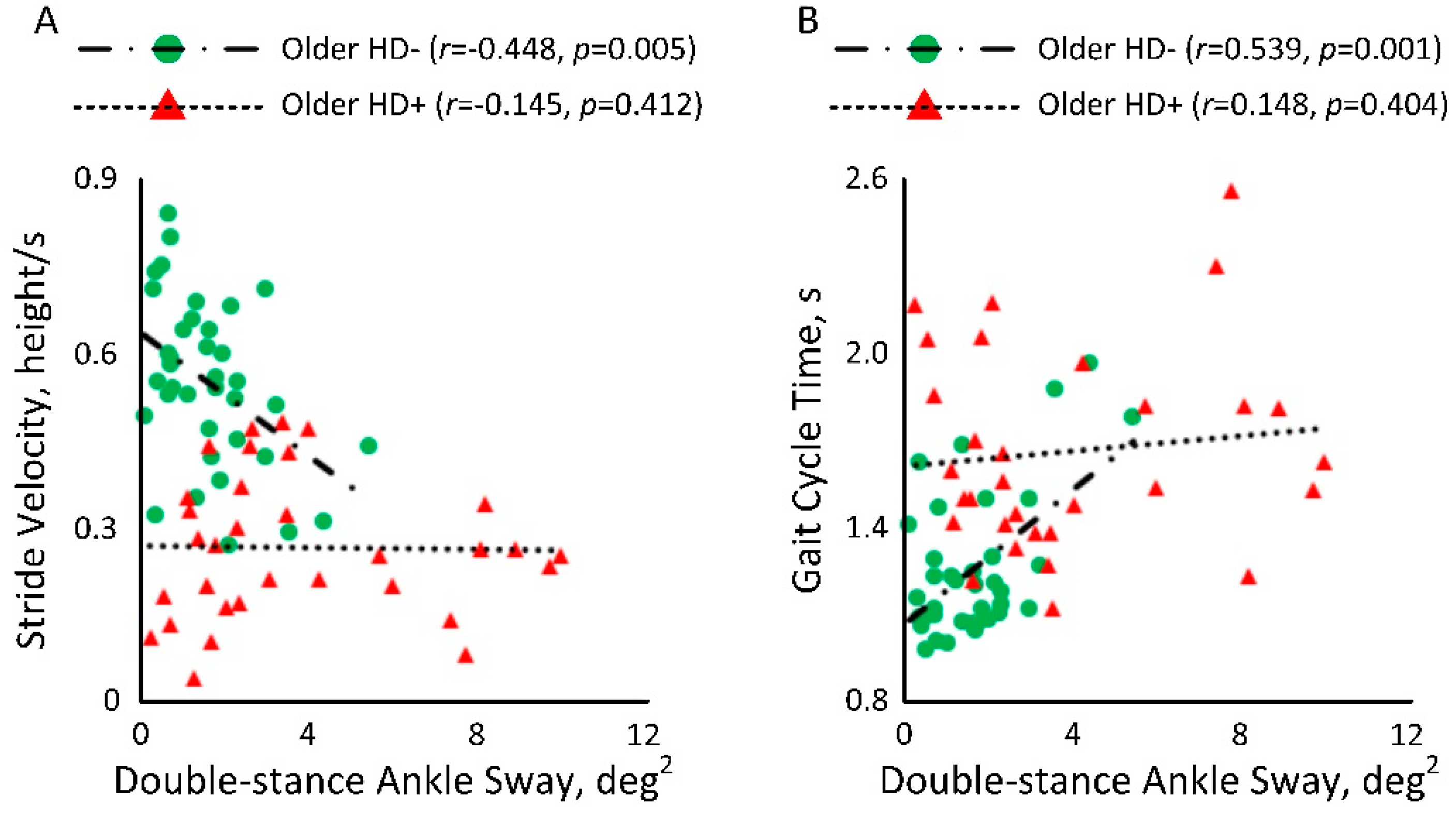
| Gait Cycle Time, s (mean ± SD) | 1.10 ± 0.11 | 1.39 ± 0.24 | 1.53 ± 0.52 | 26% | <0.001 | 1.34 | 38% | <0.001 | 1.06 | 10% | 0.036 | 0.34 | |
| Double Support, % (mean ± SD) | 22.66 ± 4.76 | 29.85 ± 8.94 | 34.92 ± 13.62 | 32% | <0.001 | 0.93 | 54% | <0.001 | 1.09 | 17% | 0.005 | 0.46 | |
| Balance Double-Stance | Ankle Sway, deg2 (mean ± SD) | 0.81 ± 0.75 | 2.24 ± 2.12 | 2.95 ± 3.09 | 177% | <0.001 | 0.86 | 264% | <0.001 | 0.79 | 32% | 0.105 | 0.27 |
| Hip Sway, deg2 (mean ± SD) | 0.94 ± 0.80 | 2.15 ± 2.43 | 3.15 ± 4.54 | 129% | 0.005 | 0.57 | 235% | 0.008 | 0.56 | 46% | 0.098 | 0.27 | |
| CoM Sway, cm2 (mean ± SD) | 0.16 ± 0.11 | 0.27 ± 0.24 | 0.36 ± 0.36 | 69% | 0.023 | 0.47 | 125% | 0.002 | 0.68 | 33% | 0.076 | 0.30 | |
| Normalized CoM Sway, cm2/height (mean ± SD SD) | 0.10 ± 0.07 | 0.16 ± 0.14 | 0.22 ± 0.22 | 60% | 0.022 | 0.47 | 120% | 0.002 | 0.67 | 38% | 0.065 | 0.31 | |
| Balance Semi-Tandem | Ankle Sway, deg2 (mean ± SD) | - | 2.44 ± 2.34 | 3.67 ± 4.14 | - | - | - | - | - | - | 51% | 0.044 | 0.38 |
| Hip Sway, deg2 (mean ± SD) | - | 2.32 ± 2.40 | 3.50 ± 3.73 | - | - | - | - | - | - | 51% | 0.034 | 0.40 | |
| CoM Sway, cm2 (mean ± SD) | - | 0.29 ± 0.29 | 0.74 ± 1.48 | - | - | - | - | - | - | 150% | 0.017 | 0.45 | |
| Normalized CoM Sway, cm2/height (mean ± SD) | - | 0.18 ± 0.17 | 0.23 ± 0.21 | - | - | - | - | - | - | 28% | 0.100 | 0.31 | |
| Mid-Age DM+ | Older DM+ | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| HD− n = 40 | HD+ n = 38 | Diff (%) | p-Value * | d * | HD− n = 37 | HD+ n = 36 | Diff (%) | p-Value * | d * | ||
| Gait | Stride Velocity, m/s (mean ± SD) | 0.93 ± 0.22 | 0.55 ± 0.22 | −41% | <0.001 | 1.68 | 0.96 ± 0.27 | 0.40 ± 0.20 | −58% | <0.001 | 2.31 |
| Normalized Stride Velocity, height/s (mean ± SD) | 0.56 ± 0.13 | 0.34 ± 0.13 | −39% | <0.001 | 1.68 | 0.55 ± 0.15 | 0.25 ± 0.12 | −55% | <0.001 | 2.14 | |
| Stride Length, m (mean ± SD) | 1.17 ± 0.20 | 0.78 ± 0.26 | −33% | <0.001 | 1.67 | 1.15 ± 0.22 | 0.62 ± 0.23 | −46% | <0.001 | 2.32 | |
| Normalized Stride Length, % height (mean ± SD) | 69.96 ± 10.01 | 48.11 ± 15.71 | −31% | <0.001 | 1.73 | 66.04 ± 11.47 | 39.50 ± 14.29 | −42% | <0.001 | 2.11 | |
| Gait Cycle Time, s (mean ± SD) | 1.29 ± 0.20 | 1.49 ± 0.24 | 15% | 0.001 | 0.83 | 1.26 ± 0.24 | 1.80 ± 0.60 | 43% | <0.001 | 1.15 | |
| Double Support, % (mean ± SD) | 26.30 ± 6.37 | 33.75 ± 9.67 | 28% | <0.001 | 0.87 | 26.34 ± 5.50 | 43.74 ± 14.22 | 66% | <0.001 | 1.42 | |
| Balance Double-Stance | Ankle Sway, deg2 (mean ± SD) | 1.48 ± 0.90 | 3.03 ± 2.68 | 105% | 0.002 | 0.76 | 1.54 ± 1.04 | 4.40 ± 3.82 | 187% | <0.001 | 1.19 |
| Hip Sway, deg2 (mean ± SD) | 1.29 ± 1.02 | 3.03 ± 3.09 | 134% | 0.003 | 0.72 | 1.55 ± 1.35 | 4.91 ± 5.97 | 217% | 0.001 | 0.90 | |
| CoM Sway, cm2 (mean ± SD) | 0.27 ± 0.21 | 0.28 ± 0.28 | 5% | 0.743 | 0.08 | 0.35 ± 0.26 | 0.37 ± 0.44 | 6% | 0.248 | 0.30 | |
| Normalized CoM Sway, cm2/height (mean ± SD) | 0.16 ± 0.12 | 0.17 ± 0.17 | 6% | 0.708 | 0.09 | 0.20 ± 0.15 | 0.23 ± 0.27 | 15% | 0.159 | 0.37 | |
| Balance Semi-Tandem | Ankle Sway, deg2 (mean ± SD) | 1.66 ± 1.13 | 2.99 ± 2.78 | 80% | 0.025 | 0.63 | 1.73 ± 1.78 | 4.85 ± 4.79 | 180% | 0.016 | 0.78 |
| Hip Sway, deg2 (mean ± SD) | 1.21 ± 0.71 | 3.10 ± 2.81 | 157% | 0.014 | 0.70 | 1.65 ± 1.63 | 4.63 ± 4.27 | 181% | 0.002 | 1.01 | |
| CoM Sway, cm2 (mean ± SD) | 0.31 ± 0.19 | 0.29 ± 0.35 | −6% | 0.709 | 0.11 | 0.37 ± 0.33 | 0.37 ± 0.37 | 0 | 0.483 | 0.23 | |
| Normalized CoM Sway, cm2/height (mean ± SD) | 0.18 ± 0.21 | 0.18 ± 0.35 | <1% | 0.639 | 0.13 | 0.21 ± 0.19 | 0.24 ± 0.23 | 14% | 0.339 | 0.31 | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhou, H.; Al-Ali, F.; Rahemi, H.; Kulkarni, N.; Hamad, A.; Ibrahim, R.; Talal, T.K.; Najafi, B. Hemodialysis Impact on Motor Function beyond Aging and Diabetes—Objectively Assessing Gait and Balance by Wearable Technology. Sensors 2018, 18, 3939. https://doi.org/10.3390/s18113939
Zhou H, Al-Ali F, Rahemi H, Kulkarni N, Hamad A, Ibrahim R, Talal TK, Najafi B. Hemodialysis Impact on Motor Function beyond Aging and Diabetes—Objectively Assessing Gait and Balance by Wearable Technology. Sensors. 2018; 18(11):3939. https://doi.org/10.3390/s18113939
Chicago/Turabian StyleZhou, He, Fadwa Al-Ali, Hadi Rahemi, Nishat Kulkarni, Abdullah Hamad, Rania Ibrahim, Talal K. Talal, and Bijan Najafi. 2018. "Hemodialysis Impact on Motor Function beyond Aging and Diabetes—Objectively Assessing Gait and Balance by Wearable Technology" Sensors 18, no. 11: 3939. https://doi.org/10.3390/s18113939
APA StyleZhou, H., Al-Ali, F., Rahemi, H., Kulkarni, N., Hamad, A., Ibrahim, R., Talal, T. K., & Najafi, B. (2018). Hemodialysis Impact on Motor Function beyond Aging and Diabetes—Objectively Assessing Gait and Balance by Wearable Technology. Sensors, 18(11), 3939. https://doi.org/10.3390/s18113939







