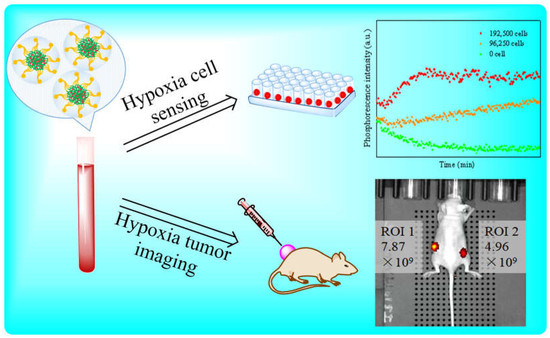
Amphiphilic Fluorine-Containing Block Copolymers as Carriers for Hydrophobic PtTFPP for Dissolved Oxygen Sensing, Cell Respiration Monitoring and In Vivo Hypoxia Imaging with High Quantum Efficiency and Long Lifetime
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials and Reagents
2.2. Instruments
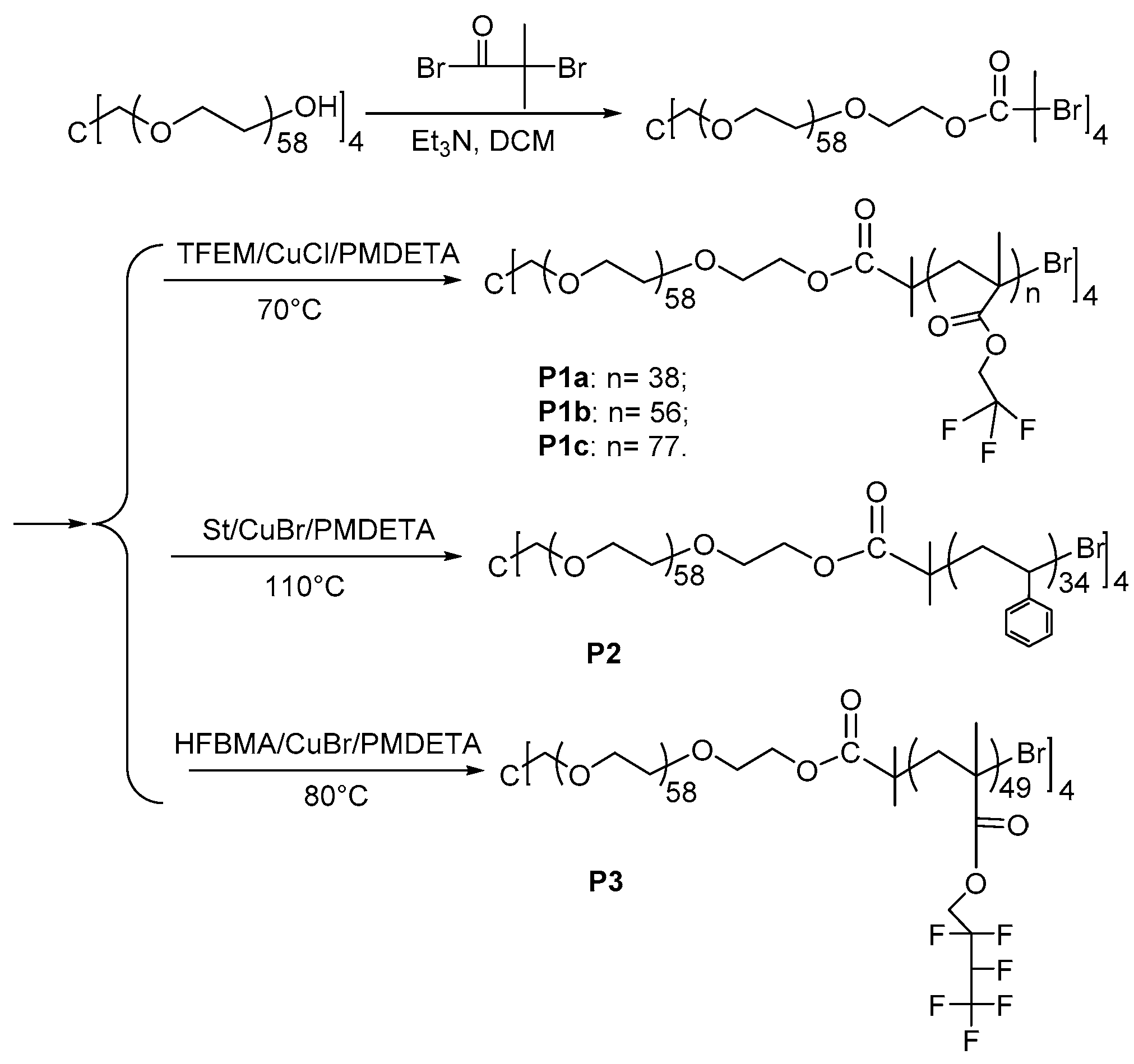
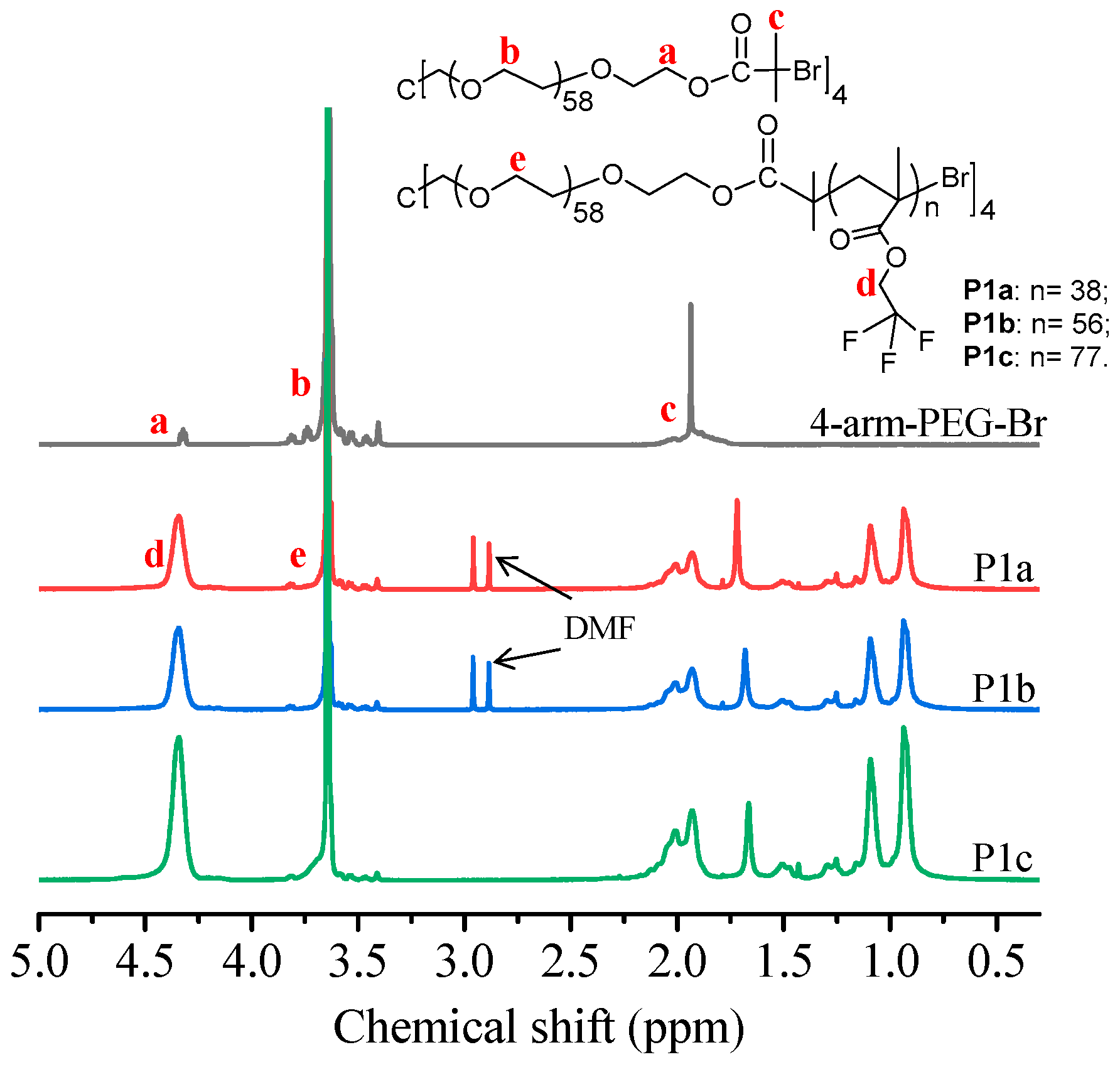
2.3. Synthesis of Macroinitiator 4-Arm-PEG-Br and Amphiphilic Star Polymers
2.4. Preparation of Micelles with PtTFPP
2.5. Critical Micelle Concentrations Determination of Block Polymers
2.6. Quantum Efficiency Determination
2.7. Response to Oxygen
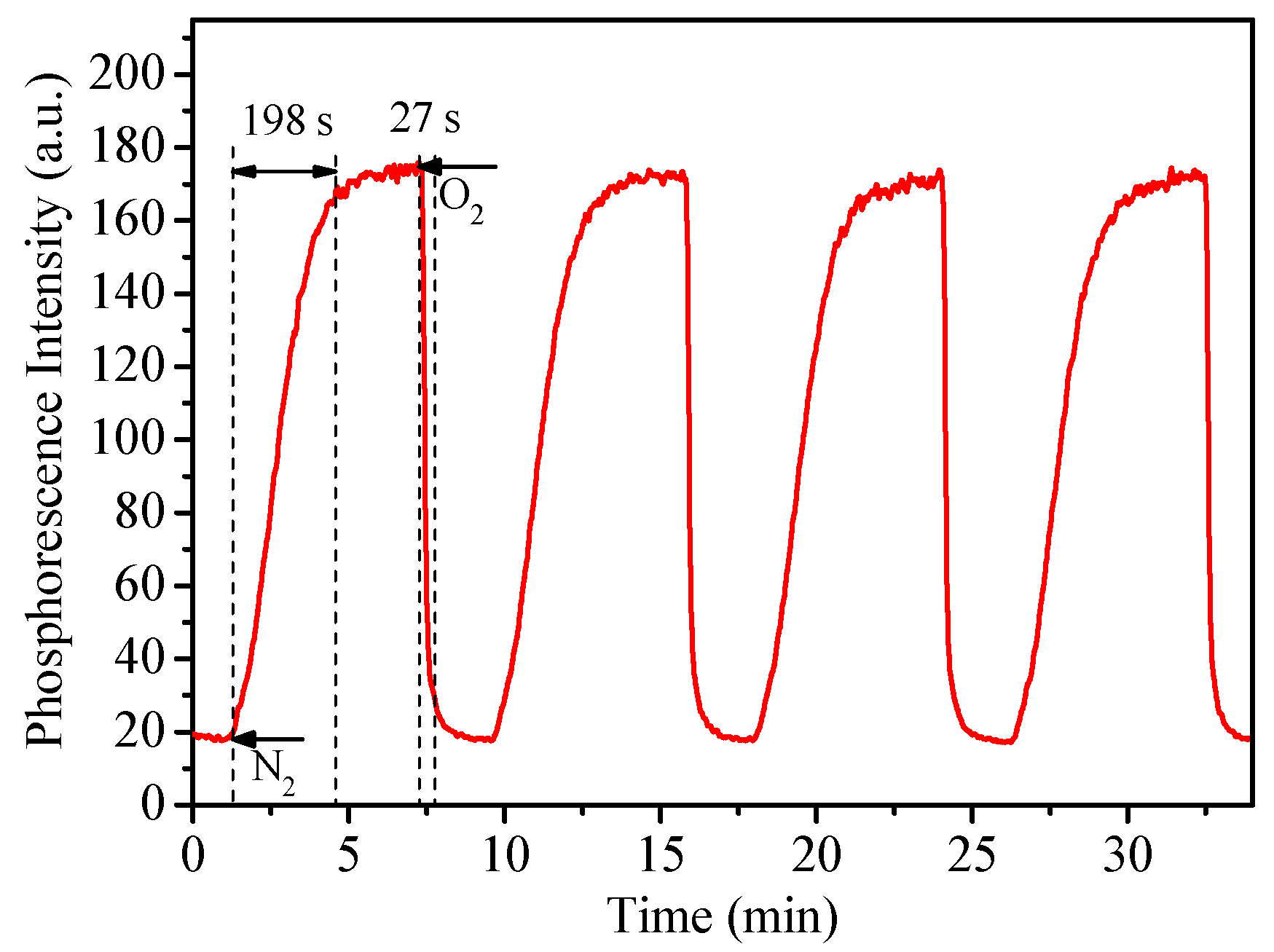
2.8. Response Time Measurement
2.9. Phosphorescence Lifetime Determination
2.10. E. coli Culture
2.11. Cell Culture
2.12. Mice Culture and In Vivo Imaging
3. Results and Discussion
3.1. Design and Synthesis of Block Copolymers
3.2. Critical Micelle Concentrations Determination
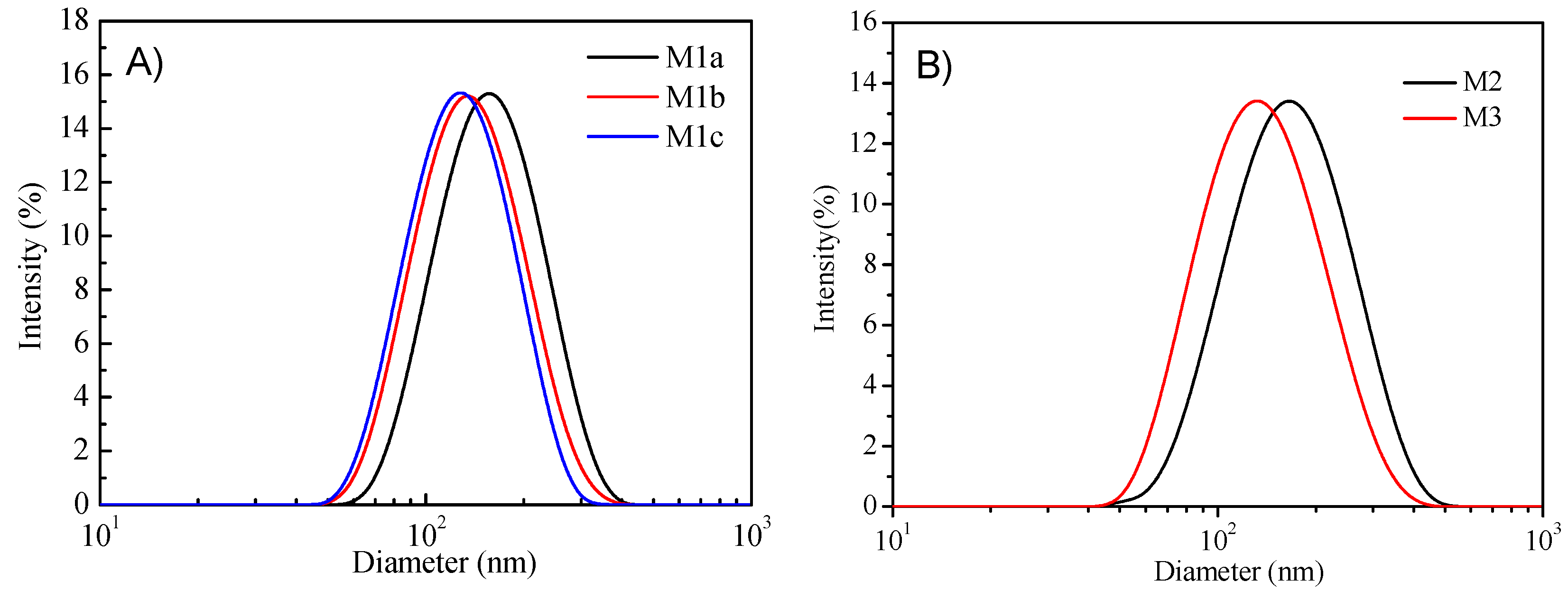
3.3. Micelle Preparation and Their Photophysical Properties
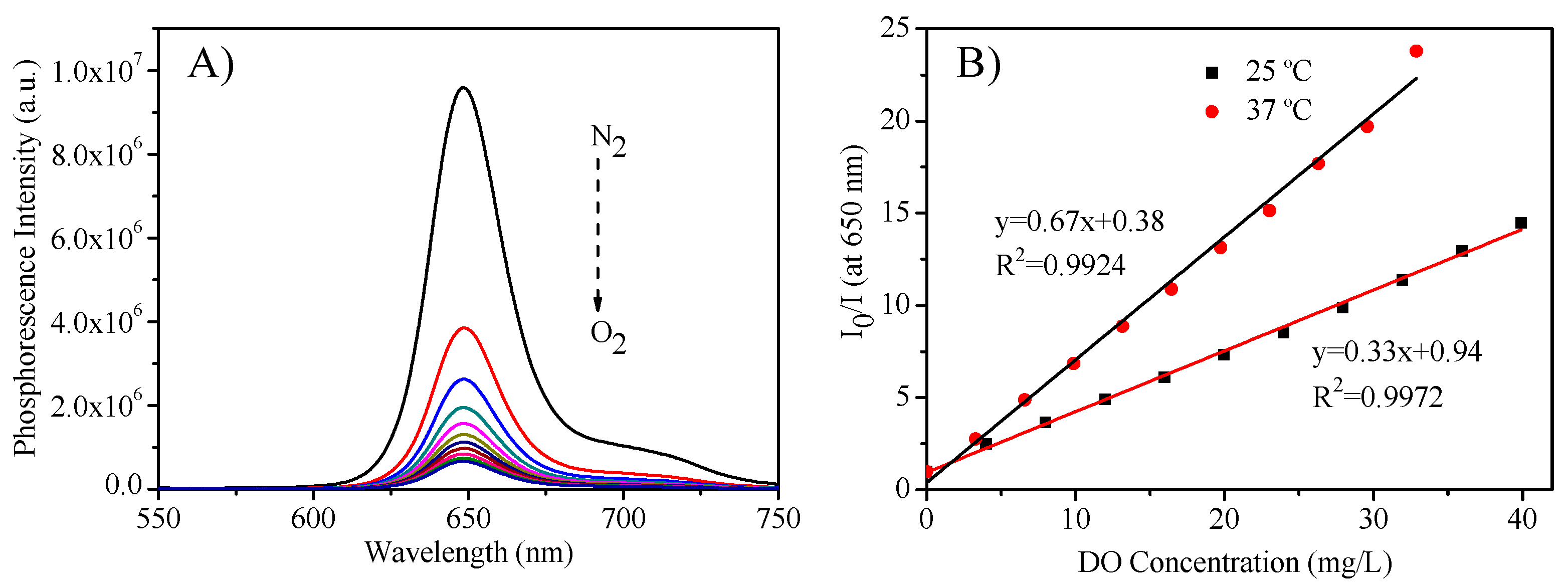
3.4. Oxygen Sensing Properties
3.5. The Use of the Sensors for Monitoring Cellular Respiration
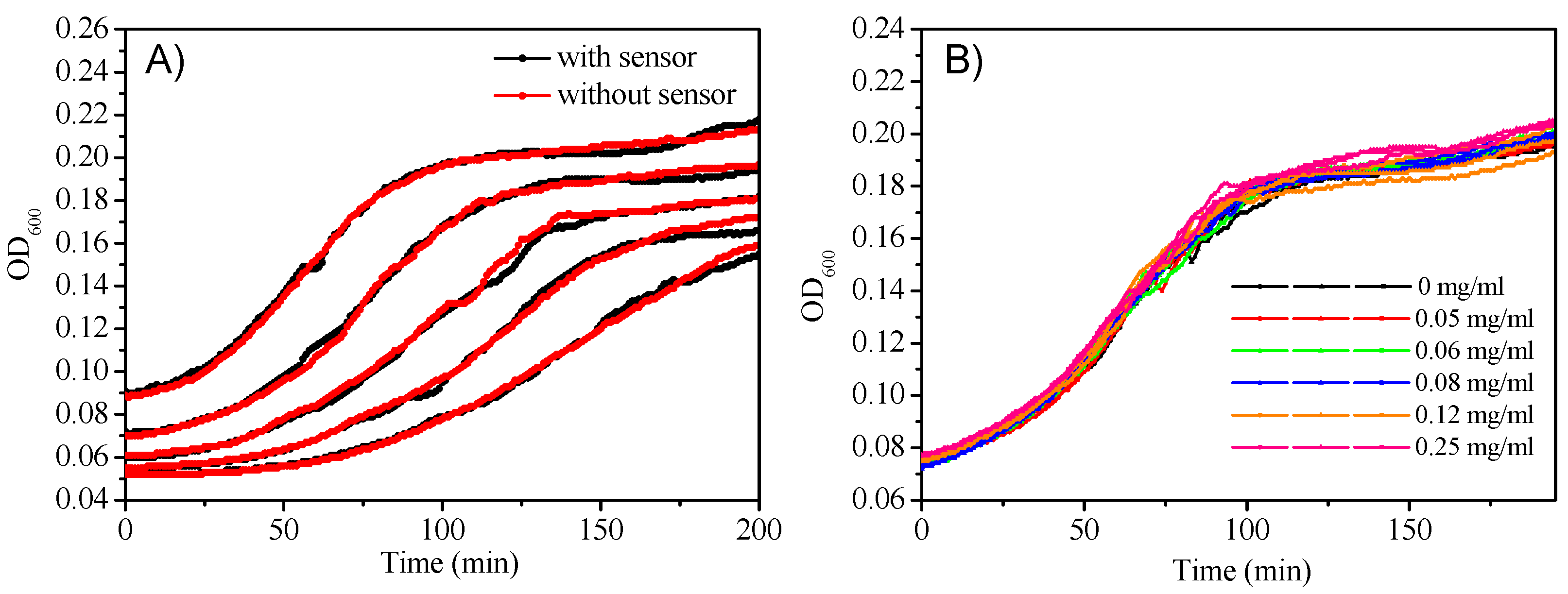
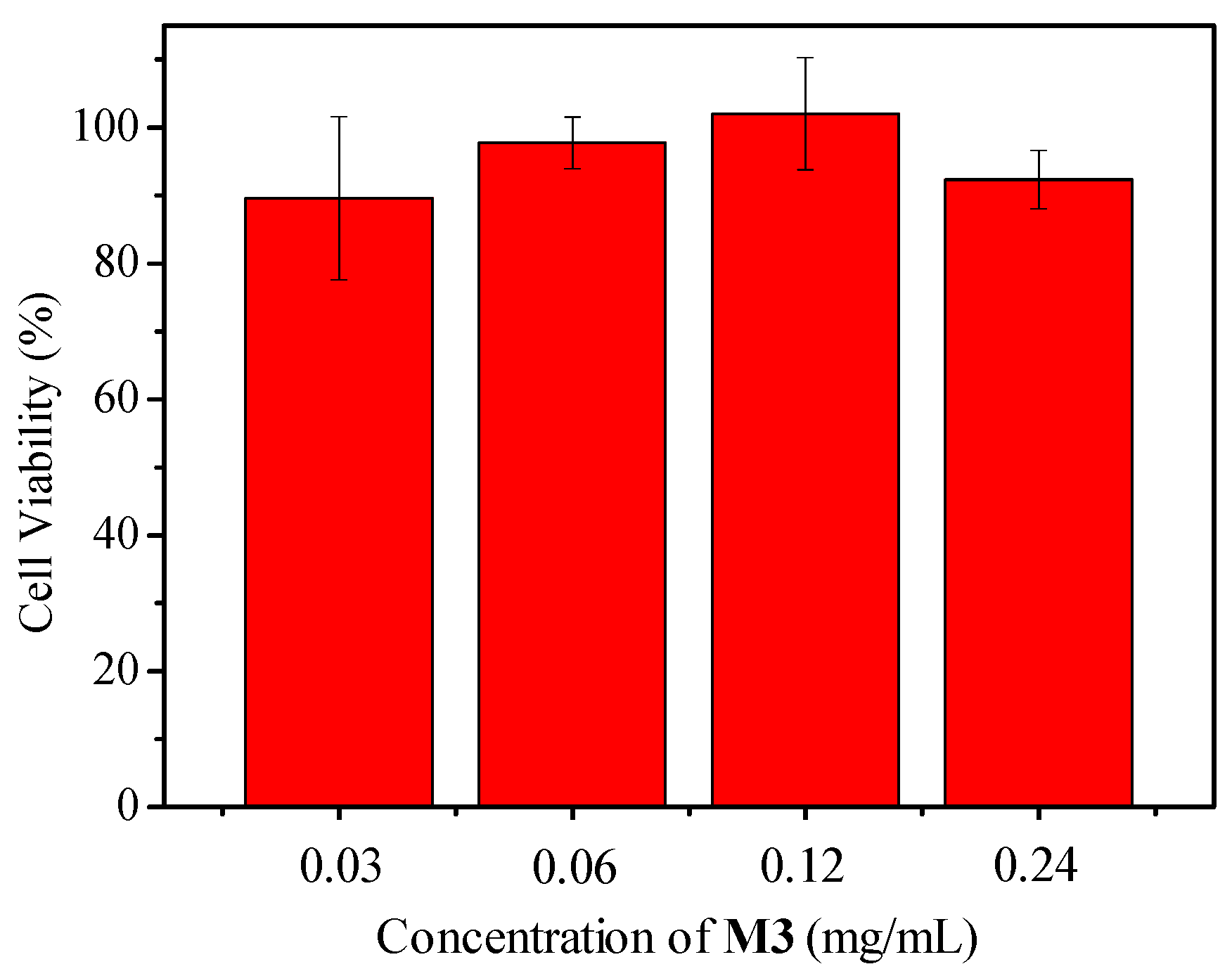
3.5.1. Monitoring the Oxygen Consumption during the Growth of E. coli and the Cytotoxicity Evaluation
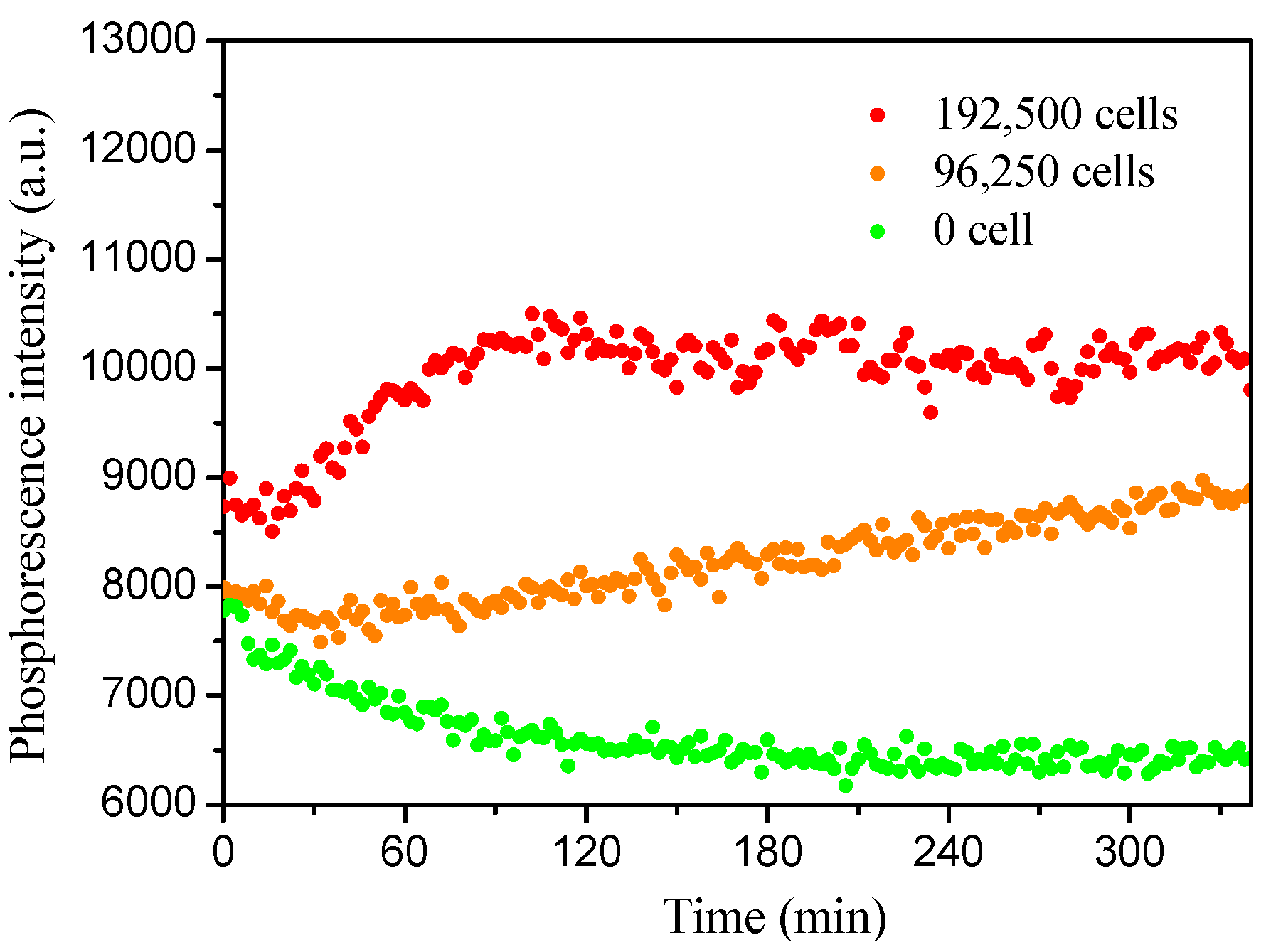
3.5.2. Monitoring Cell Respiration of J774A.1 Cells
3.6. In Vivo Hypoxia Imaging
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Vaupel, P.; Hoeckel, M.; Mayer, A. Detection and characterization of tumor hypoxia using PO2 histography. Antioxid. Redox Signal. 2007, 9, 1221–1235. [Google Scholar] [CrossRef] [PubMed]
- Iwai, T.; Obara, K.; Ito, C.; Furukawa, H.; Oka, J.-I. Hydroxyobtustyrene protects neuronal cells from chemical hypoxia-induced cell death. J. Nat. Med. 2018, 72, 915–921. [Google Scholar] [CrossRef] [PubMed]
- Koehler, U.; Hildebrandt, O.; Kroenig, J.; Grimm, W.; Otto, J.; Hildebrandt, W.; Kinscherf, R. Chronic hypoxia and cardiovascular risk. Clinical significance of different forms of hypoxia. Herz 2018, 43, 291–297. [Google Scholar] [CrossRef] [PubMed]
- Schramm, A.; Grunewald, S.; Lorenz, R.; Classen, J.; Naumann, M. Parkinsonism due to bilateral basal ganglia lesions following mastocytosis-induced hypoxia. J. Neurol. 2004, 251, 1270–1272. [Google Scholar] [CrossRef] [PubMed]
- Salminen, A.; Kauppinen, M.; Kaamiranta, K. Hypoxia/ischemia activate processing of amyloid precursor protein: Impact of vascular dysfunction in the pathogenesis of Alzheimer’s disease. J. Neurochem. 2017, 140, 536–549. [Google Scholar] [CrossRef] [PubMed]
- Thienpont, B.; Steinbacher, J.; Zhao, H.; D’Anna, F.; Kuchnio, A.; Ploumakis, A.; Ghesquiere, B.; Van Dyck, L.; Boeckx, B.; Schoonjans, L.; et al. Tumour hypoxia causes DNA hyper-methylation by reducing TET activity. Nature 2016, 537, 63–68. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; He, G.; Qing, H.; Zhou, W.; Dobie, F.; Cai, F.; Staufenbiel, M.; Huang, L.E.; Song, W. Hypoxia facilitates Alzheimer’s disease pathogenesis by up-regulating BACE1 gene expression. Proc. Natl. Acad. Sci. USA 2006, 103, 18727–18732. [Google Scholar] [CrossRef] [PubMed]
- Semenza, G.L. Oxygen sensing, hypoxia-inducible factors, and disease pathophysiology. Annu. Rev. Pathol. Mech. Dis. 2014, 9, 47–71. [Google Scholar] [CrossRef] [PubMed]
- Giaccia, A.J.; Simon, M.C.; Johnson, R. The biology of hypoxia: The role of oxygen sensing in development, normal function, and disease. Genes Dev. 2004, 18, 2183–2194. [Google Scholar] [CrossRef] [PubMed]
- Kinoshita, K. Electrochemical Oxygen Technology; Wiley: New York, NY, USA, 1992. [Google Scholar]
- Winkler, L.W. Die bestimmung des im wasser gelösten sauerstoffes. Eur. J. Inorg. Chem. 1888, 21, 2843–2854. [Google Scholar] [CrossRef]
- Han, B.H.; Manners, I.; Winnik, M.A. Phosphorescence quenching of dyes adsorbed to silica thin-layer chromatography plates. Anal. Chem. 2005, 77, 8075–8085. [Google Scholar] [CrossRef] [PubMed]
- Moos, R. Solid State Gas Sensors–Industrial Application; Fleischer, M., Lehmann, M., Eds.; Springer: Berlin/Heidelberg, Germany, 2011; pp. 173–188. [Google Scholar]
- Wang, X.; Wolfbeis, O.S. Optical methods for sensing and imaging oxygen: Materials, spectroscopies and applications. Chem. Soc. Rev. 2014, 43, 3666–3761. [Google Scholar] [CrossRef] [PubMed]
- Yoshihara, T.; Hirakawa, Y.; Hosaka, M.; Nangaku, M.; Tobita, S. Oxygen imaging of living cells and tissues using luminescent molecular probes. J. Photochem. Photobiol. C 2017, 30, 71–95. [Google Scholar] [CrossRef]
- Pereira, N.A.M.; Laranjo, M.; Casalta-Lopes, J.; Serra, A.C.; Pineiro, M.; Pina, J.; Sergio Seixas de Melo, J.; Senge, M.O.; Botelho, F.M.; Martelo, L.; et al. Platinum (II) ring-fused chlorins as near-infrared emitting oxygen sensors and photodynamic agents. ACS Med. Chem. Lett. 2017, 8, 310–315. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Y.; Liu, Y.; Shang, J.; Ma, J.; Wang, R.; Deng, L.; Guo, Y.; Zhong, F.; Bai, M.; Zhang, S.; et al. Phosphorescence monitoring of hypoxic microenvironment in solid-tumors to evaluate chemotherapeutic effects using the hypoxia-sensitive Iridium (III) coordination compound. PLoS ONE 2015, 10, e0121293. [Google Scholar] [CrossRef] [PubMed]
- Eastwood, D.; Gouterma, M. Porphyrins XVIII luminescence of (Co), (Ni), Pd, Pt complexes. J. Mol. Spectrosc. 1970, 35, 359–375. [Google Scholar] [CrossRef]
- Sun, K.; Tang, Y.; Li, Q.; Yin, S.; Qin, W.; Yu, J.; Chiu, D.T.; Liu, Y.; Yuan, Z.; Zhang, X.; et al. In vivo dynamic monitoring of small molecules with implantable polymer-dot transducer. ACS Nano 2016, 10, 6769–6781. [Google Scholar] [CrossRef] [PubMed]
- Sakadzic, S.; Roussakis, E.; Yaseen, M.A.; Mandeville, E.T.; Srinivasan, V.J.; Arai, K.; Ruvinskaya, S.; Devor, A.; Lo, E.H.; Vinogradov, S.A.; et al. Two-photon high-resolution measurement of partial pressure of oxygen in cerebral vasculature and tissue. Nat. Methods 2010, 7, 755–759. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; Zhou, X.; Cao, T.; Zhang, K.Y.; Yang, L.; Liu, S.; Liang, H.; Yang, H.; Li, F.; Huang, W. Fluorescent/phosphorescent dual-emissive conjugated polymer dots for hypoxia bioimaging. Chem. Sci. 2015, 6, 1825–1831. [Google Scholar] [CrossRef] [PubMed]
- Shi, H.; Ma, X.; Zhao, Q.; Liu, B.; Qu, Q.; An, Z.; Zhao, Y.; Huang, W. Ultrasmall phosphorescent polymer dots for ratiometric oxygen sensing and photodynamic cancer therapy. Adv. Funct. Mater. 2014, 24, 4823–4830. [Google Scholar] [CrossRef]
- Dmitriev, R.I.; Borisov, S.M.; Duessmann, H.; Sun, S.; Mueller, B.J.; Prehn, J.; Baklaushev, V.P.; Klimant, I.; Papkovsky, D.B. Versatile conjugated polymer nanoparticles for high-resolution O2 imaging in cells and 3D tissue models. ACS Nano 2015, 9, 5275–5288. [Google Scholar] [CrossRef] [PubMed]
- Lemon, C.M.; Karnas, E.; Han, X.; Bruns, O.T.; Kempa, T.J.; Fukumura, D.; Bawendi, M.G.; Jain, R.K.; Duda, D.G.; Nocera, D.G. Micelle-encapsulated quantum dot-porphyrin assemblies as in vivo two-photon oxygen sensors. J. Am. Chem. Soc. 2015, 137, 9832–9842. [Google Scholar] [CrossRef] [PubMed]
- Su, F.; Alam, R.; Mei, Q.; Tian, Y.; Youngbull, C.; Johnson, R.H.; Meldrum, D.R. Nanostructured oxygen sensor–using micelles to incorporate a hydrophobic platinum porphyrin. PLoS ONE 2012, 7, e33390. [Google Scholar] [CrossRef] [PubMed]
- Zou, X.; Pan, T.; Jiang, J.; Li, G.; Song, C.; Sun, R.; Yang, Z.; Sun, D.; Hou, C.; Chen, M.; et al. Poly(epsilon-caprolactone)-containing graft copolymers for ratiometric extracellular oxygen sensing. Sens. Actuator B Chem. 2017, 248, 108–118. [Google Scholar] [CrossRef]
- Zhao, Q.; Pan, T.; Xiang, G.; Mei, Z.; Jiang, J.; Li, G.; Zou, X.; Chen, M.; Sun, D.; Jiang, S.; et al. Highly efficient ratiometric extracellular oxygen sensors through physical incorporation of a conjugated polymer and PtTFPP in graft copolymers. Sens. Actuator B Chem. 2018, 273, 242–252. [Google Scholar] [CrossRef]
- Matyjaszewski, K.; Miller, P.J.; Pyun, J.; Kickelbick, G.; Diamanti, S. Synthesis and characterization of star polymers with varying arm number, length, and composition from organic and hybrid inorganic/organic multifunctional initiators. Macromolecules 1999, 32, 6526–6535. [Google Scholar] [CrossRef]
- Ren, J.M.; McKenzie, T.G.; Fu, Q.; Wong, E.H.H.; Xu, J.; An, Z.; Shanmugam, S.; Davis, T.P.; Boyer, C.; Qiao, G.G. Star Polymers. Chem. Rev. 2016, 116, 6743–6836. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; Fan, W.; Li, L.; Chu, P.K.; Yeung, K.W.K.; Wu, S.; Xu, Z. Novel anionic fluorine-containing amphiphilic self-assembly polymer micelles for potential application in protein drug carrier. J. Fluor. Chem. 2012, 141, 21–28. [Google Scholar] [CrossRef]
- Westbrook, A.W.; Ren, X.; Moo-Young, M.; Chou, C.P. Application of hydrocarbon and perfluorocarbon oxygen vectors to enhance heterologous production of hyaluronic acid in engineered bacillus subtilis. Biotechnol. Bioeng. 2018, 115, 1239–1252. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.W.S.; Wong, K.Y.; Li, X.M.; Leung, Y.B.; Chan, C.S.; Chan, K.S. Halogenated platinum porphyrins as sensing materials for luminescence-based oxygen sensors. J. Mater. Chem. 1993, 3, 1031–1035. [Google Scholar] [CrossRef]
- Rosen, M.J.; Mathias, J.H.; Davenport, L. Aberrant aggregation behavior in cationic gemini surfactants investigated by surface tension, interfacial tension, and fluorescence methods. Langmuir 1999, 15, 7340–7346. [Google Scholar] [CrossRef]
- Chen, C.-Y.; Kim, T.H.; Wu, W.-C.; Huang, C.-M.; Wei, H.; Mount, C.W.; Tian, Y.; Jang, S.-H.; Pun, S.H.; Jen, A.K.Y. pH-dependent, thermosensitive polymeric nanocarriers for drug delivery to solid tumors. Biomaterials 2013, 34, 4501–4509. [Google Scholar] [CrossRef] [PubMed]
- Lai, S.W.; Hou, Y.J.; Che, C.M.; Pang, H.L.; Wong, K.Y.; Chang, C.K.; Zhu, N.Y. Electronic spectroscopy, photophysical properties, and emission quenching studies of an oxidatively robust perfluorinated platinum porphyrin. Inorg. Chem. 2004, 43, 3724–3732. [Google Scholar] [CrossRef] [PubMed]
- Joshi, H.S.; Jamshidi, R.; Tor, Y. Conjugated 1,10-phenanthrolines as tunable fluorophores. Angew. Chem. Int. Ed. 1999, 38, 2722–2725. [Google Scholar] [CrossRef]
- Khanna, K.; Varshney, S.; Kakkar, A. Miktoarm star polymers: Advances in synthesis, self-assembly, and applications. Polym. Chem. 2010, 1, 1171–1185. [Google Scholar] [CrossRef]
- Ge, Z.; Liu, S. Supramolecular self-assembly of nonlinear amphiphilic and double hydrophilic block copolymers in aqueous solutions. Macromol. Rapid Commun. 2009, 30, 1523–1532. [Google Scholar] [CrossRef] [PubMed]
- Matyjaszewski, K. Atom transfer radical polymerization (ATRP): Current status and future perspectives. Macromolecules 2012, 45, 4015–4039. [Google Scholar] [CrossRef]
- Palmiero, U.C.; Sponchioni, M.; Manfredini, N.; Maraldi, M.; Moscatelli, D. Strategies to combine ROP with ATRP or RAFT polymerization for the synthesis of biodegradable polymeric nanoparticles for biomedical applications. Polym. Chem. 2018, 9, 4084–4099. [Google Scholar] [CrossRef]
- Boyer, C.; Corrigan, N.A.; Jung, K.; Diep, N.; Thuy-Khanh, N.; Adnan, N.N.M.; Oliver, S.; Shanmugam, S.; Yeow, J. Copper-mediated living radical polymerization (atom transfer radical polymerization and copper (0) mediated polymerization): From fundamentals to bioapplications. Chem. Rev. 2016, 116, 1803–1949. [Google Scholar] [CrossRef] [PubMed]
- Jones, M.C.; Leroux, J.C. Polymeric micelles—A new generation of colloidal drug carriers. Eur. J. Pharm. Biopharm. 1999, 48, 101–111. [Google Scholar] [CrossRef]
- Xu, R.; Wang, Y.; Duan, X.; Lu, K.; Micheroni, D.; Hu, A.; Lin, W. Nanoscale Metal-organic frameworks for ratiometric oxygen sensing in live cells. J. Am. Chem. Soc. 2016, 138, 2158–2161. [Google Scholar] [CrossRef] [PubMed]
- Fercher, A.; Borisov, S.M.; Zhdanov, A.V.; Klimant, I.; Papkovsky, D.B. Intracellular O2 sensing probe based on cell-penetrating phosphorescent nanoparticles. ACS Nano 2011, 5, 5499–5508. [Google Scholar] [CrossRef] [PubMed]
- Lehner, P.; Larndorfer, C.; Garcia-Robledo, E.; Larsen, M.; Borisov, S.M.; Revsbech, N.-P.; Glud, R.N.; Canfield, D.E.; Klimant, I. LUMOS—A Sensitive and Reliable Optode System for Measuring Dissolved Oxygen in the Nanomolar Range. PLoS ONE 2015, 10, e0128125. [Google Scholar] [CrossRef] [PubMed]
- O’Riordan, T.C.; Fitzgerald, K.; Ponomarev, G.V.; Mackrill, J.; Hynes, J.; Taylor, C.; Papkovsky, D.B. Sensing intracellular oxygen using near-infrared phosphorescent probes and live-cell fluorescence imaging. Am. J. Physiol.-Reg. Integr. Comp. Physiol. 2007, 292, R1613–R1620. [Google Scholar] [CrossRef] [PubMed]
- Esipova, T.V.; Karagodov, A.; Miller, J.; Wilson, D.F.; Busch, T.M.; Vinogradov, S.A. Two new “protected” oxyphors for biological oximetry: Properties and application in tumor imaging. Anal. Chem. 2011, 83, 8756–8765. [Google Scholar] [CrossRef] [PubMed]
- Tian, Y.; Shumway, B.R.; Meldrum, D.R. A new cross-linkable oxygen sensor covalently bonded into poly(2-hydroxyethyl methacrylate)-co-polyacrylamide thin film for dissolved oxygen sensing. Chem. Mater. 2010, 22, 2069–2078. [Google Scholar] [CrossRef] [PubMed]
- Baggaley, E.; Botchway, S.W.; Haycock, J.W.; Morris, H.; Sazanovich, I.V.; Williams, J.A.G.; Weinstein, J.A. Long-lived metal complexes open up microsecond lifetime imaging microscopy under multiphoton excitation: From FLIM to PLIM and beyond. Chem. Sci. 2014, 5, 879–886. [Google Scholar] [CrossRef]
- Vaupel, P.; Harrison, L. Tumor hypoxia: Causative factors, compensatory mechanisms, and cellular response. Oncologist 2004, 9, 4–9. [Google Scholar] [CrossRef] [PubMed]



| Yield (%) | Conv. (%) | Mn(NMR) | Mn(GPC) | Mw/Mn | CMC (μg/mL) | |
|---|---|---|---|---|---|---|
| 4-arm-PEG-Br | 79 | - | 10,940 | 3960 | 1.1 | - |
| P1a | 50 | 76 | 33,790 | 5340 | 1.3 | 6.9 |
| P1b | 44 | 56 | 45,880 | 7650 | 1.2 | 8.8 |
| P1c | 40 | 51 | 62,680 | 10,710 | 1.3 | 10.5 |
| P2 | 26 | 34 | 24,780 | 6280 | 1.2 | 5.7 |
| P3 | 48 | 49 | 59,220 | 8370 | 1.5 | 6.0 |
| Micelles | Diameter (nm) | aPDI | Con (Polymer) (mg/mL) | Con (PtTFPP) (mg/mL) b | Encapsulation Efficiency (wt%) | Quantum Efficiency (%) | cI0/I100 | d Response Time (s) | e Lifetime (μs) | |||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 25 °C | 37 °C | t95-r | t95 | Air | N2 | |||||||
| M1a | 163 | 0.31 | 0.86 | 0.14 | 42 | 19.5 | 15 | 25 | 178 | 29 | 21 | 73 |
| M1b | 142 | 0.27 | 0.38 | 0.13 | 39 | 22.3 | 15 | 27 | 204 | 31 | 22 | 72 |
| M1c | 122 | 0.34 | 0.34 | 0.06 | 18 | 21.5 | 11 | 28 | 191 | 30 | 29 | 70 |
| M2 | 145 | 0.17 | 1.11 | 0.31 | 93 | 11.8 | 8 | 10 | 163 | 34 | 25 | 66 |
| M3 | 126 | 0.15 | 1.90 | 0.30 | 90 | 16.7 | 14 | 24 | 198 | 27 | 18 | 76 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, J.; Qiao, Y.; Pan, T.; Zhong, K.; Wen, J.; Wu, S.; Su, F.; Tian, Y. Amphiphilic Fluorine-Containing Block Copolymers as Carriers for Hydrophobic PtTFPP for Dissolved Oxygen Sensing, Cell Respiration Monitoring and In Vivo Hypoxia Imaging with High Quantum Efficiency and Long Lifetime. Sensors 2018, 18, 3752. https://doi.org/10.3390/s18113752
Li J, Qiao Y, Pan T, Zhong K, Wen J, Wu S, Su F, Tian Y. Amphiphilic Fluorine-Containing Block Copolymers as Carriers for Hydrophobic PtTFPP for Dissolved Oxygen Sensing, Cell Respiration Monitoring and In Vivo Hypoxia Imaging with High Quantum Efficiency and Long Lifetime. Sensors. 2018; 18(11):3752. https://doi.org/10.3390/s18113752
Chicago/Turabian StyleLi, Jiaze, Yuan Qiao, Tingting Pan, Ke Zhong, Jiaxing Wen, Shanshan Wu, Fengyu Su, and Yanqing Tian. 2018. "Amphiphilic Fluorine-Containing Block Copolymers as Carriers for Hydrophobic PtTFPP for Dissolved Oxygen Sensing, Cell Respiration Monitoring and In Vivo Hypoxia Imaging with High Quantum Efficiency and Long Lifetime" Sensors 18, no. 11: 3752. https://doi.org/10.3390/s18113752
APA StyleLi, J., Qiao, Y., Pan, T., Zhong, K., Wen, J., Wu, S., Su, F., & Tian, Y. (2018). Amphiphilic Fluorine-Containing Block Copolymers as Carriers for Hydrophobic PtTFPP for Dissolved Oxygen Sensing, Cell Respiration Monitoring and In Vivo Hypoxia Imaging with High Quantum Efficiency and Long Lifetime. Sensors, 18(11), 3752. https://doi.org/10.3390/s18113752