A Hybrid Approach to Detect Driver Drowsiness Utilizing Physiological Signals to Improve System Performance and Wearability
Abstract
:1. Introduction
2. Related Work
2.1. Vehicle-Based Measures
2.2. Subjective Measures
2.3. Driver Behavioral Measures
2.4. Driver Physiological Measures
- to detect drowsiness using EEG and ECG alone, and combining both modalities to improve the performance of the system;
- to propose a channel reduction paradigm to reduce the number of electrodes and to increase the practicality of the proposed technique in real driving environments.
3. Materials and Methods for Inducing and Detecting Driver Drowsiness
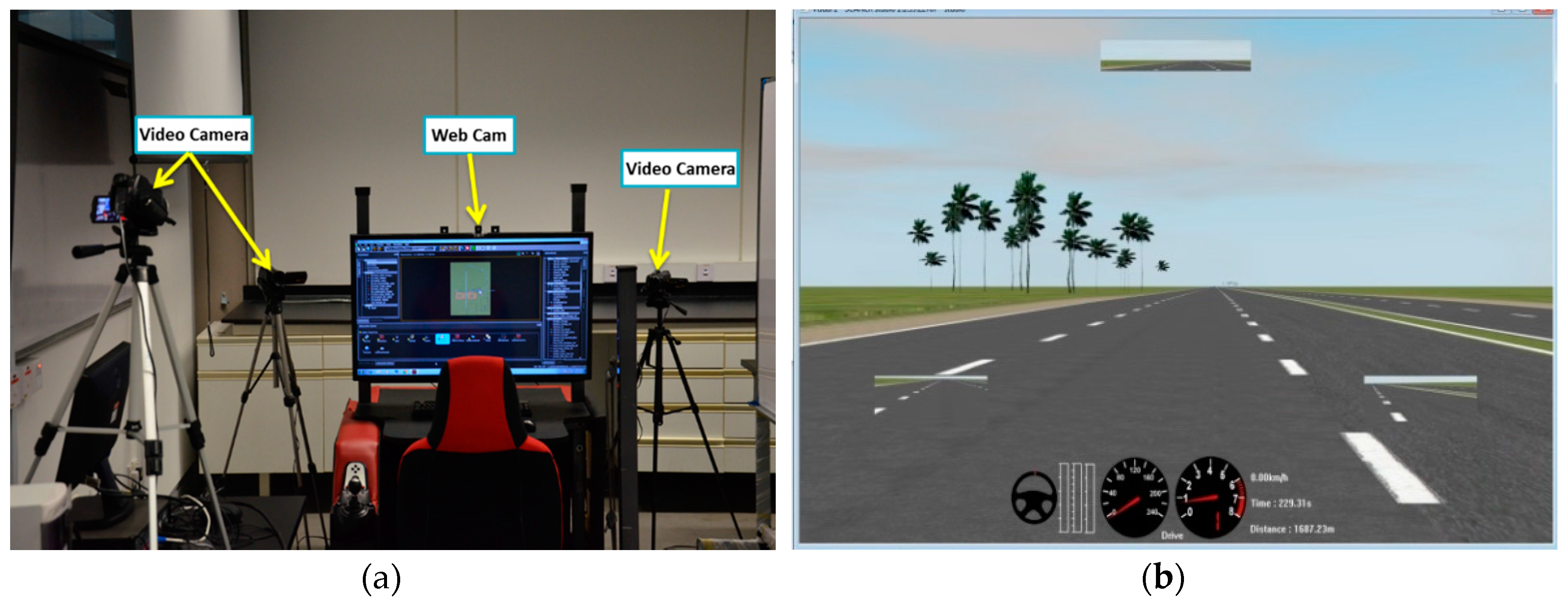
3.1. Simulator-Based Driving Environment
3.2. Sample Population
3.3. Materials
3.4. Data Collection
3.5. Subjective Measure
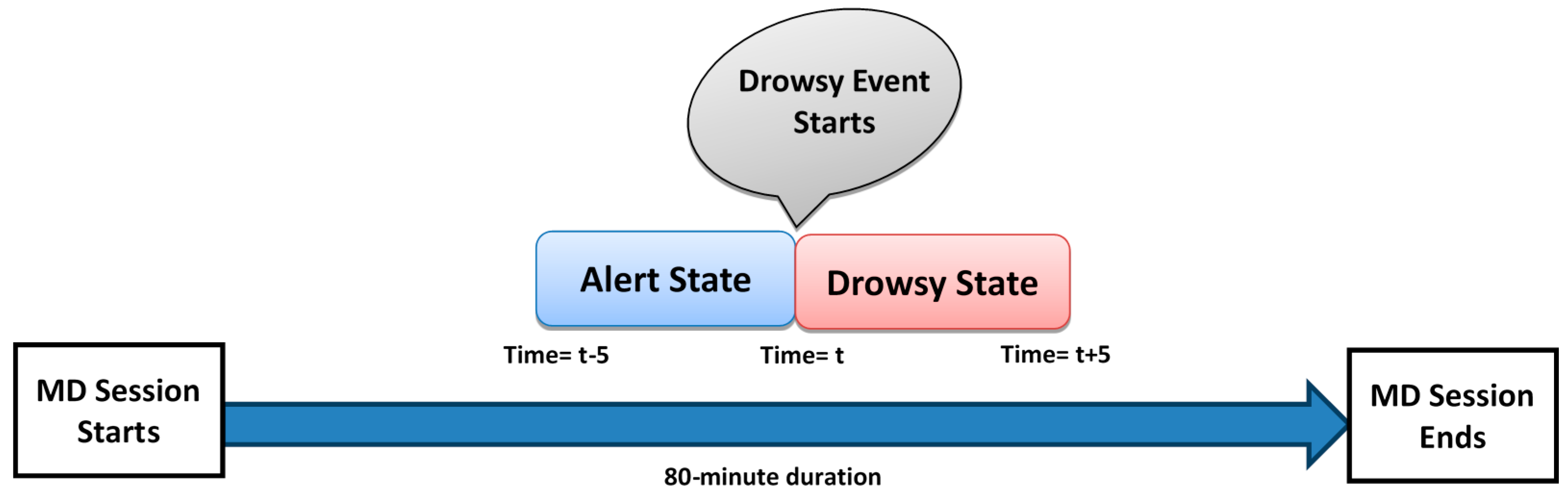
3.6. Ground Truth Management Using Observational Analysis
3.7. Physiological Data Pre-Processing
3.8. Feature Extraction of Physiological Data
3.8.1. EEG Time Domain Feature Extraction
3.8.2. EEG Frequency Domain Feature Extraction
3.8.3. ECG Data Feature Extraction
3.9. Feature Selection
3.10. Classifying Drowsiness and Channel Reduction
4. Results and Discussion
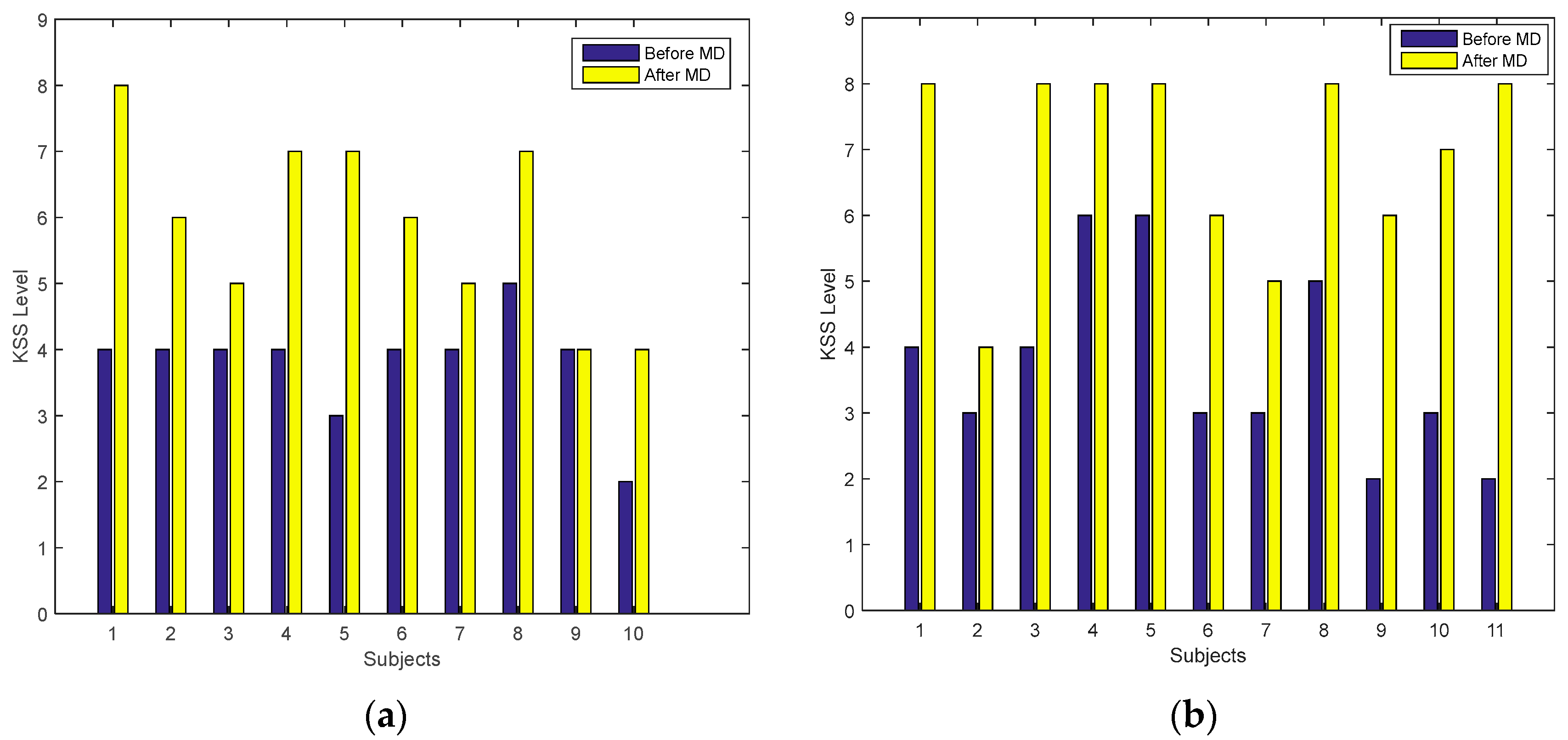
4.1. Subjective Measures
4.2. Physiological Measures
4.2.1. EEG Time Domain Analysis
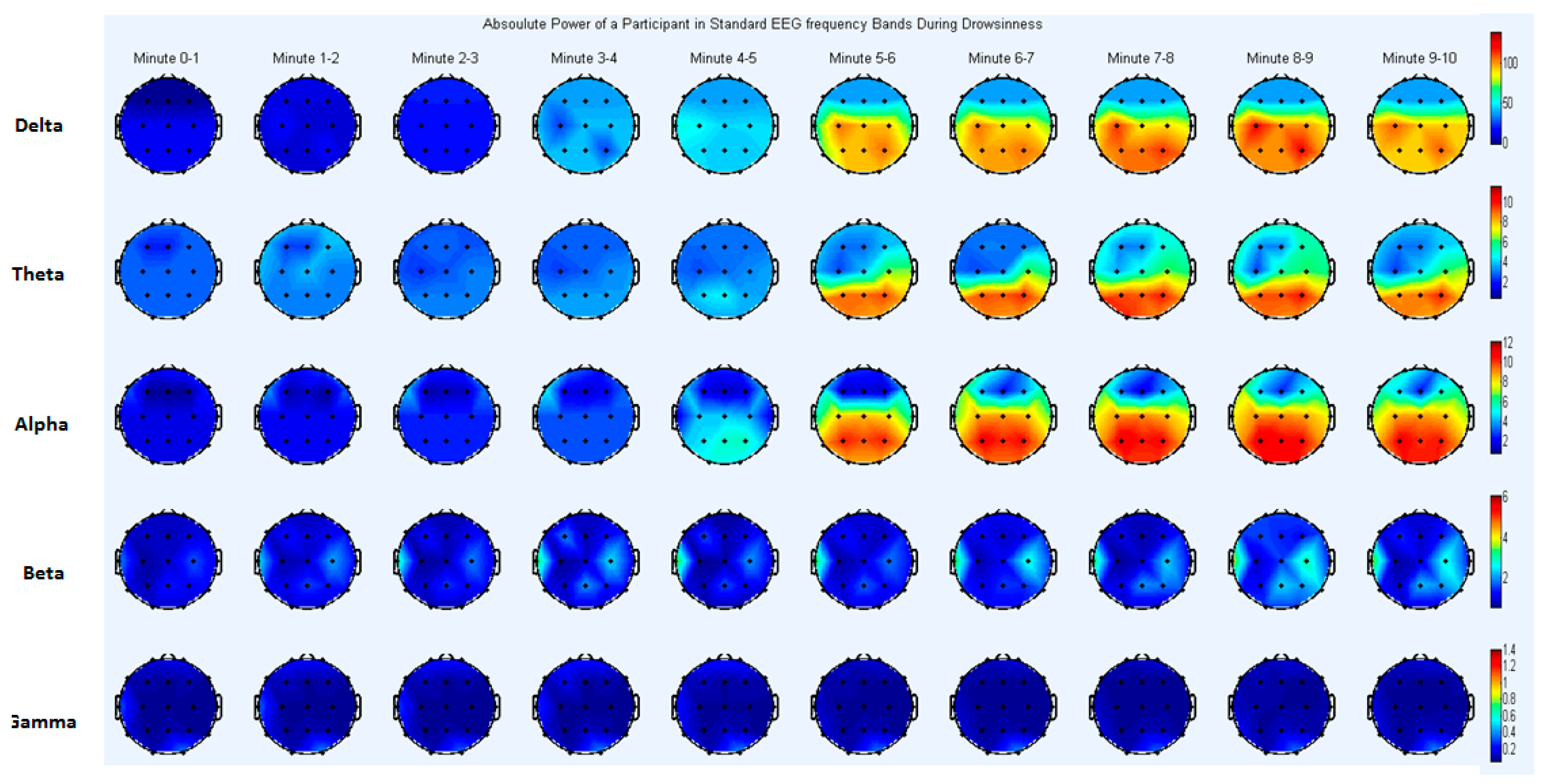
4.2.2. EEG Frequency Domain Analysis
Absolute Power Analysis
Relative Power Analysis
4.2.3. ECG Results
4.3. Performance Analysis by Combining EEG and ECG Channels Compared with Using Them Alone
4.4. Channel Reduction to Improve Wearability
5. Conclusions and Future Work
- The study was conducted in a simulator-based environment in which the room temperature, lighting conditions and other environmental variables were constant. However, in real driving conditions these variables constantly change, introducing more artifacts into the physiological signals due to diverse environmental conditions and driving behavior. Therefore, future studies should examine the performance of this system under real driving conditions with variable environmental conditions.
- To develop a real time drowsiness monitoring device, the computational complexity of the proposed system should be decreased. This includes the optimization of the drowsiness detection algorithm and the removal of various artifacts in real time.
- The effects of drowsiness should be further explored using other physiological signals such as electrooculogram (EOG) and electromyogram (EMG). EMG sensors are less limited in terms of wearability compared with EEG sensors, which require drivers to wear a large cap while driving.
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Traffic Safety Facts. Available online: https://crashstats.nhtsa.dot.gov/Api/Public/Publication/811449 (accessed on 15 April 2017).
- State of the Road Fatigue Fact Sheet. Available online: https://www.fatiguemanagementtrainingonline.com.au/blog/state-of-the-road-fatigue-fact-sheet/ (accessed on 30 March 2017).
- Sahayadhas, A.; Sundaraj, K.; Murugappan, M. Detecting driver drowsiness based on sensors: A review. Sensors 2012, 12, 16937–16953. [Google Scholar] [CrossRef] [PubMed]
- Richman, J.S.; Moorman, J.R. Physiological time-series analysis using approximate entropy and sample entropy. Am. J. Physiol. Heart Circ. Physiol. 2000, 278, H2039–H2049. [Google Scholar] [PubMed]
- Slater, J.D. A definition of drowsiness: One purpose for sleep? Med. Hypotheses 2008, 71, 641–644. [Google Scholar] [CrossRef] [PubMed]
- Mardi, Z.; Ashtiani, S.N.M.; Mikaili, M. EEG-based drowsiness detection for safe driving using chaotic features and statistical tests. J. Med. Signals Sens. 2011, 1, 130. [Google Scholar] [PubMed]
- Tunçer, O.; Güvenç, L.; Coşkun, F. Vision based lane keeping assistance control triggered by a driver inattention monitor. In Proceedings of the International Conference on Systems Man and Cybernetics (SMC), Istanbul, Turkey, 10–13 October 2010. [Google Scholar]
- Sayed, R.; Eskandarian, A. Unobtrusive drowsiness detection by neural network learning of driver steering. J. Automob. Eng. 2001, 215, 969–975. [Google Scholar] [CrossRef]
- Pomerleau, D. RALPH: Rapidly adapting lateral position handler. In Proceedings of the Intelligent Vehicles’ Symposium, Detroit, MI, USA, 25–26 September 1995. [Google Scholar]
- Thiffault, P.; Bergeron, J. Monotony of road environment and driver fatigue: A simulator study. Accid. Anal. Prev. 2003, 35, 381–391. [Google Scholar] [CrossRef]
- Vural, E. Video-based detection of driver fatigue. Ph.D. Thesis, Sabanci University, Istanbul, Turkey, 2009. [Google Scholar]
- Khushaba, R.N.; Kodagoda, S.; Lal, S.; Dissanayake, G. Intelligent driver drowsiness detection system using Uncorrelated Fuzzy Locality Preserving Analysis. In Proceedings of the Intelligent Robots and Systems (IROS) Conference, San Francisco, CA, USA, 25–30 September 2011. [Google Scholar]
- Shahid, A.; Wilkinson, K.; Marcu, S. Stanford Sleepiness Scale (SSS), STOP, THAT and One Hundred Other Sleep Scales, 1st ed.; Springer: New York, NY, USA, 2012; pp. 369–370. [Google Scholar]
- Stanford Sleepiness Scale. Available online: http://www.stanford.edu/~dement/sss.html (accessed on 2 February 2017).
- Shahid, A.; Wilkinson, K.; Marcu, S.; Colin, M.; Shapiro, M. Karolinska Sleepiness Scale (KSS) in STOP, THAT and One Hundred Other Sleep Scales, 1st ed.; Springer: New York, NY, USA, 2012; pp. 209–210. [Google Scholar]
- Simons, R.; Martens, M.; Ramaekers, J.; Krul, A.; Klöpping-Ketelaars, I.; Skopp, G. Effects of dexamphetamine with and without alcohol on simulated driving. Psychopharmacology 2012, 222, 391–399. [Google Scholar]
- Tremaine, R.; Dorrian, J.; Lack, L.; Lovato, N.; Ferguson, S.; Zhou, X.; Roach, G. The relationship between subjective and objective sleepiness and performance during a simulated night-shift with a nap countermeasure. Appl. Ergon. 2010, 42, 52–61. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, K.A.; Itoi, A.; Dement, W.C. Awareness of sleepiness and ability to predict sleep onset: Can drivers avoid falling asleep at the wheel? Sleep Med. 2007, 9, 71–79. [Google Scholar] [CrossRef] [PubMed]
- Azim, T.; Jaffar, M.A.; Mirza, A.M. Automatic Fatigue Detection of Drivers through Pupil Detection and Yawning Analysis. In Proceedings of the 4th Innovative Computing, Information and Control (ICICIC), Kaohsiung, Taiwan, 7–9 December 2009. [Google Scholar]
- Danisman, T.; Danisman, T.; Bilasco, I.M.; Djeraba, C.; Ihaddadene, N. Drowsy driver detection system using eye blink patterns. In Proceedings of the International Conference on Machine and Web Intelligence (ICMWI), Algiers, Algeria, 3–5 October 2010. [Google Scholar]
- Fan, X.; Yin, B.C.; Sun, Y.F. Yawning detection for monitoring driver fatigue. In Proceedings of the International Conference on Machine Learning Cybernetics (ICMLC), Hong Kong, China, 19–22 August 2007. [Google Scholar]
- Lee, D.; Oh, S.; Heo, S.; Hahn, M. Drowsy driving detection based on the driver’s head movement using infrared sensors. In Proceedings of the 2nd International Symposium on Universal Communication, Osaka, Japan, 15–16 December 2008. [Google Scholar]
- Popieul, J.C.; Simon, P.; Loslever, P. Using driver’s head movements evolution as a drowsiness indicator. In Proceedings of the IEEE Intelligent Vehicles Symposium, Columbus, OH, USA, 9–11 June 2003. [Google Scholar]
- Ying, Y.; Jing, S.; Wei, Z. The monitoring method of driver’s fatigue based on neural network. In Proceedings of the International Conference on Mechatronics and Automation, Harbin, China, 5–8 August 2007. [Google Scholar]
- Yu, X. Real-Time Nonintrusive Detection of Driver Drowsiness; CTS 09-15 Technical Report; University of Minnesota: Minneapolis, MN, USA, May 2009. [Google Scholar]
- Lin, C.T.; Chang, C.J.; Lin, B.S.; Hung, S.H.; Chao, C.F.; Wang, I.J. A real-time wireless brain–computer interface system for drowsiness detection. IEEE Trans. Biomed. Circuit Syst. 2010, 4, 214–222. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.T.; Wu, R.C.; Liang, S.F.; Chao, W.H.; Chen, Y.J.; Jung, T.P. EEG-based drowsiness estimation for safety driving using independent component analysis. IEEE Trans. Biomed. Circuit Syst. 2005, 52, 2726–2738. [Google Scholar]
- Papadelis, C.; Chen, Z.; Kourtidou-Papadeli, C.; Bamidis, P.D.; Chouvarda, I.; Bekiaris, E.; Maglaveras, N. Monitoring sleepiness with on-board electrophysiological recordings for preventing sleep-deprived traffic accidents. Clinic. Neurophysiol. 2007, 118, 1906–1922. [Google Scholar] [CrossRef] [PubMed]
- Michail, E.; Kokonozi, A.; Chouvarda, I.; Maglaveras, N. EEG and HRV markers of sleepiness and loss of control during car driving. In Proceedings of the Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Vancouver, BC, Canada, 25–28 August 2008. [Google Scholar]
- Yang, G.; Lin, Y.; Bhattacharya, P. A driver fatigue recognition model based on information fusion and dynamic Bayesian network. Inf. Sci. 2010, 180, 1942–1954. [Google Scholar] [CrossRef]
- Picot, A.; Charbonnier, S.; Caplier, A. On-line detection of drowsiness using brain and visual information, Systems, Man and Cybernetics, Part A: Systems and Humans. IEEE Trans. Syst. Hum. 2012, 42, 764–775. [Google Scholar]
- Elsenbruch, S.; Harnish, M.J.; Orr, W.C. Heart rate variability during waking and sleep in healthy males and females. Sleep 1999, 22, 1067–1071. [Google Scholar] [CrossRef] [PubMed]
- Tsunoda, M.; Endo, T.; Hashimoto, S.; Honma, S.; Honma, K.I. Effects of light and sleep stages on heart rate variability in humans. Psychiatry Clin. Neurosci. 2001, 55, 285–286. [Google Scholar] [CrossRef] [PubMed]
- Hu, S.; Bowlds, R.L.; Ye, G.; Yu, X. Pulse wave sensor for non-intrusive driver’s drowsiness detection. In Proceedings of the Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Minneapolis, MN, USA, 3–6 September 2009. [Google Scholar]
- Furman, G.D.; Baharav, A. Investigation of drowsiness while driving utilizing analysis of heart rate fluctuations. In Proceedings of the IEEE Computing in Cardiology, Belfast, UK, 26−29 September 2010. [Google Scholar]
- Sahayadhas, A.; Sundaraj, K.; Murugappan, M. Drowsiness detection during different times of day using multiple features. Australas. Phys. Eng. Sci. Med. 2013, 36, 243–250. [Google Scholar] [CrossRef] [PubMed]
- Enobio. Neuroelectrics. Available online: http://www.neuroelectrics.com/products/enobio (accessed on 17 April 2017).
- Delorme, A.; Makeig, S. EEGLAB: An open source toolbox for analysis of single-trial EEG dynamics including independent component analysis. J. Neurosci. Methods 2004, 134, 9–21. [Google Scholar] [CrossRef] [PubMed]
- Camm, A.; Malik, M.; Bigger, J.T.; Breithardt, G.; Cerutti, S.; Cohen, R.J.; Coumel, P.; Fallen, E.L.; Kennedy, H.L.; Kleiger, R.E.; et al. Heart rate variability: standards of measurement, physiological interpretation and clinical use. Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology. Circulation 1996, 93, 1043–1065. [Google Scholar]
- Vicente, J; Laguna, P.; Bartra, A.; Bailón, R. Detection of driver’s drowsiness by means of HRV analysis. Comput. Cardiol. 2011, 38, 89–92. [Google Scholar]
- Massey, F.J. The Kolmogorov-Smirnov test for goodness of fit. J. Am. Stat. Assoc. 1951, 46, 68–78. [Google Scholar] [CrossRef]
- Hall, M.; Hall, M.; Frank, E.; Holmes, G.; Pfahringer, B.; Reutemann, P.; Witten, I.H. The WEKA data mining software: An update. ACM SIGKDD Explor. Newslett. 2009, 11, 10–18. [Google Scholar] [CrossRef]
- Chouvarda, I.; Papadelis, C.; Kourtidou-Papadeli, C.; Bamidis, P.D.; Koufogiannis, D.; Bekiaris, E.; Maglaveras, N. Non-linear analysis for the sleepy drivers problem. In Proceedings of the 12th World Congress on Health (Medical) Informatics and Building Sustainable Health Systems, 1st ed.; IOS Press: Amsterdam, The Netherlands, 15 September 2007. [Google Scholar]
- Santamaria, J.; Chiappa, K.H. The EEG of Drowsiness; Demos Publications: New York, NY, USA, 1987. [Google Scholar]
- Niedermeyer, E. The Normal EEG of the Waking Adult. Electroencephalogr. Basic Princ. Clin. Appl. Relat. Fields 2005, 167, 155–164. [Google Scholar]
- Picot, A.; Charbonnier, S.; Caplier, A. On-line automatic detection of driver drowsiness using a single electroencephalographic channel. In Proceedings of the 30th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Vancouver, BC, Canada, 20–25 August 2008. [Google Scholar]
- Lal, S.K.; Craig, A. Driver fatigue: Electroencephalography and psychological assessment. Psychophysiology 2002, 39, 313–321. [Google Scholar] [CrossRef] [PubMed]
- Awais, M.; Badruddin, N.; Drieberg, M. A non-invasive approach to detect drowsiness in a monotonous driving environment. In Proceedings of the IEEE TENCON Region 10 Conference, Bangkok, Thailand, 22–25 October 2014. [Google Scholar]
- Sörnmo, L.; Laguna, P. Bioelectrical Signal Processing in Cardiac and Neurological Applications; Elsevier Academic Press: Burlington, MA, USA, 2005. [Google Scholar]
- Tobaldini, E.; Nobili, L.; Strada, S.; Casali, K.R.; Braghiroli, A.; Montano, N. Heart rate variability in normal and pathological sleep. Front. Physiol. 2013, 4, 294. [Google Scholar] [CrossRef] [PubMed]
- Vicente, J.; Laguna, P.; Bartra, A.; Bailón, R. Drowsiness detection using heart rate variability. Med. Biol. Eng. Comput. 2016, 54, 927–937. [Google Scholar] [CrossRef] [PubMed]
- Patel, M.; Lal, S.K.L.; Kavanagh, D.; Rossiter, P. Applying neural network analysis on heart rate variability data to assess driver fatigue. Expert Syst. Appl. 2011, 38, 7235–7242. [Google Scholar] [CrossRef]
- Soomro, M.H.; Badruddin, N.; Yusoff, M.Z.; Jatoi, M.A. Automatic eye-blink artifact removal method based on EMD-CCA. In Proceedings of the International Conference on Complex Medical Engineering, Beijing, China, 25–28 May 2013. [Google Scholar]
- Javed, E.; Faye, I.; Malik, A.S.; Abdullah, J.M. A Hybrid Method to Improve the Reduction of Ballistocardiogram Artifact from EEG Data. In Proceedings of the International Conference on Neural Information Processing, Kuching, Sarawak, Malaysia, 3–6 November 2014. [Google Scholar]
| Channel | Significant Feature (p-Value) |
|---|---|
| P4 | , , (0.022), (0.013) |
| P7 | (0.018), Energy (0.005) |
| C3 | |
| O1 | (0.039), , Energy (0.009), |
| O2 |
| Channel | Significant Feature (p-Value) |
|---|---|
| P3 | |
| P4 | , , (0.014), |
| P7 | (0.044) |
| P8 | (0.023) |
| C3 | (0.006), |
| Cz | (0.032) |
| O1 | (0.048), |
| O2 | (0.039), (0.047) |
| State → | Alert State | Drowsy State | ||||||
|---|---|---|---|---|---|---|---|---|
| Features → | b | a | b | a | b | a | b | a |
| Mean | 0.54 | 0.32 | 2.01 | 859.82 | 0.46 | 0.37 | 1.39 | 1338.47 |
| STD | 0.10 | 0.08 | 0.98 | 114.12 | 0.08 | 0.06 | 0.59 | 121.61 |
| Accuracy | (a) Only ECG | ||
| 70.00% | Predicted Class | ||
| Actual Class | Alert | Drowsy | ← Classified as |
| 39 | 16 | Alert | |
| 17 | 38 | Drowsy | |
| Accuracy | (b) Only EEG | ||
| 76.36% | Predicted Class | ||
| Actual Class | Alert | Drowsy | ← Classified as |
| 43 | 12 | Alert | |
| 14 | 41 | Drowsy | |
| Accuracy | (c) Combining ECG and EEG | ||
| 80.90% | Predicted Class | ||
| Actual Class | Alert | Drowsy | ← Classified as |
| 46 | 9 | Alert | |
| 12 | 43 | Drowsy | |
| Channel Combination | Performance |
|---|---|
| O1, ECG | 79.82% |
| O2, ECG | 80.90% |
| P7, ECG | 80.00% |
| P4, ECG | 79.09% |
| C3, ECG | 76.36% |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Awais, M.; Badruddin, N.; Drieberg, M. A Hybrid Approach to Detect Driver Drowsiness Utilizing Physiological Signals to Improve System Performance and Wearability. Sensors 2017, 17, 1991. https://doi.org/10.3390/s17091991
Awais M, Badruddin N, Drieberg M. A Hybrid Approach to Detect Driver Drowsiness Utilizing Physiological Signals to Improve System Performance and Wearability. Sensors. 2017; 17(9):1991. https://doi.org/10.3390/s17091991
Chicago/Turabian StyleAwais, Muhammad, Nasreen Badruddin, and Micheal Drieberg. 2017. "A Hybrid Approach to Detect Driver Drowsiness Utilizing Physiological Signals to Improve System Performance and Wearability" Sensors 17, no. 9: 1991. https://doi.org/10.3390/s17091991
APA StyleAwais, M., Badruddin, N., & Drieberg, M. (2017). A Hybrid Approach to Detect Driver Drowsiness Utilizing Physiological Signals to Improve System Performance and Wearability. Sensors, 17(9), 1991. https://doi.org/10.3390/s17091991





