Design of Highly Selective Gas Sensors via Physicochemical Modification of Oxide Nanowires: Overview
Abstract
:1. Introduction
2. Metal Oxide Nanowires
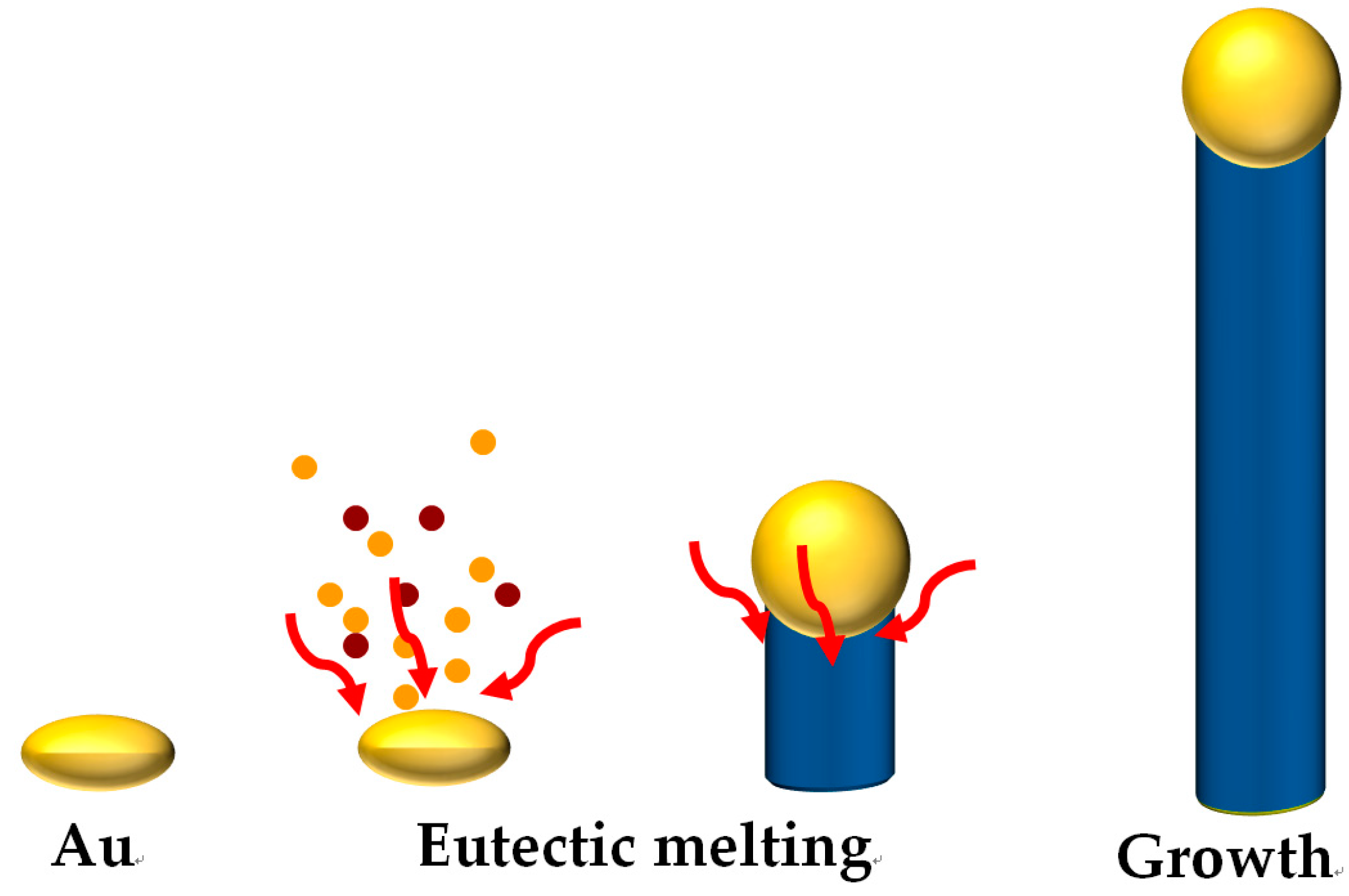
2.1. Growth of Single Crystalline Metal Oxide Nanowires
2.2. Gas-Sensing Mechanism of Metal Oxide Nanowire Networks
2.3. Metal Oxide Nanowire Network Gas Sensors: Strengths and Limitations
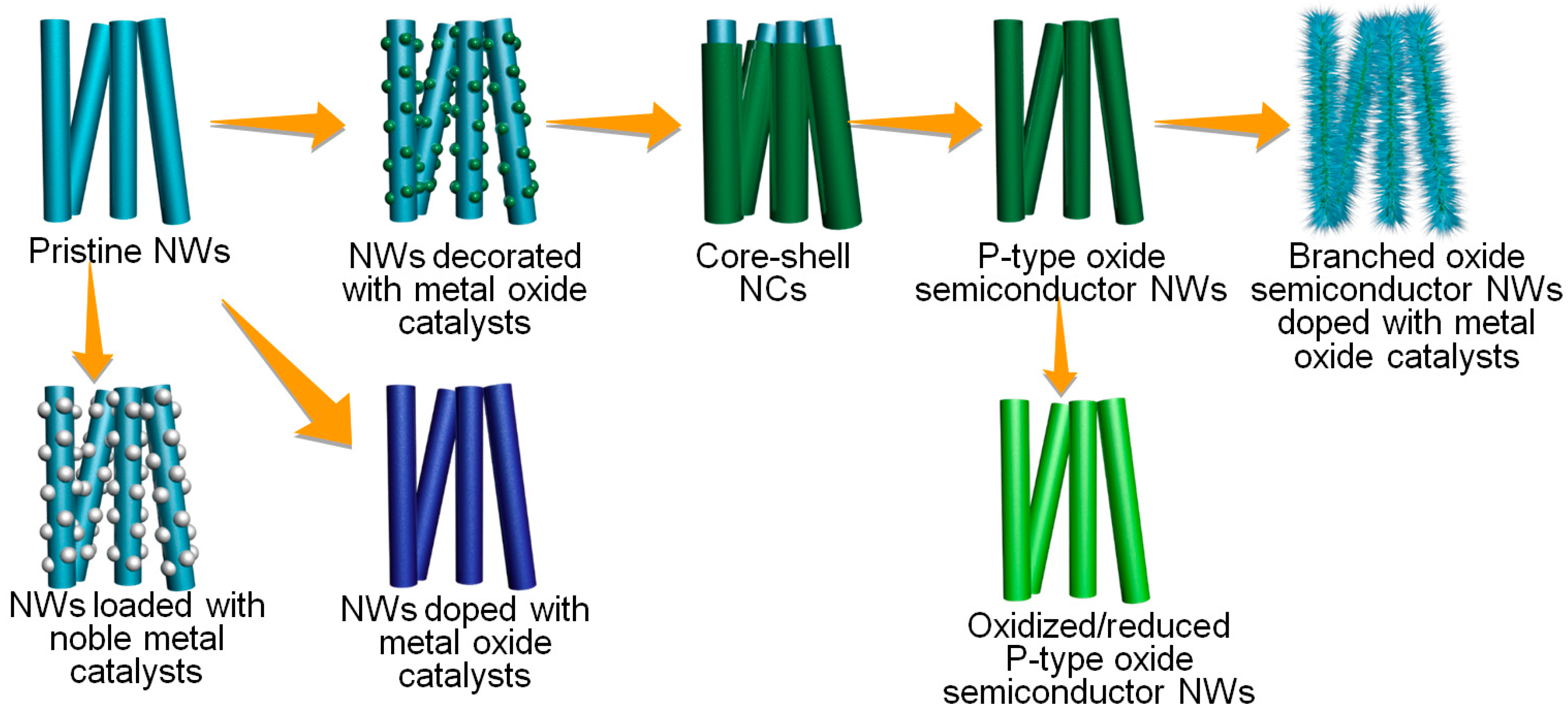
3. Physicochemical Modifications for the Enhancement of Selectivity
3.1. Noble Metal Doping/Loading
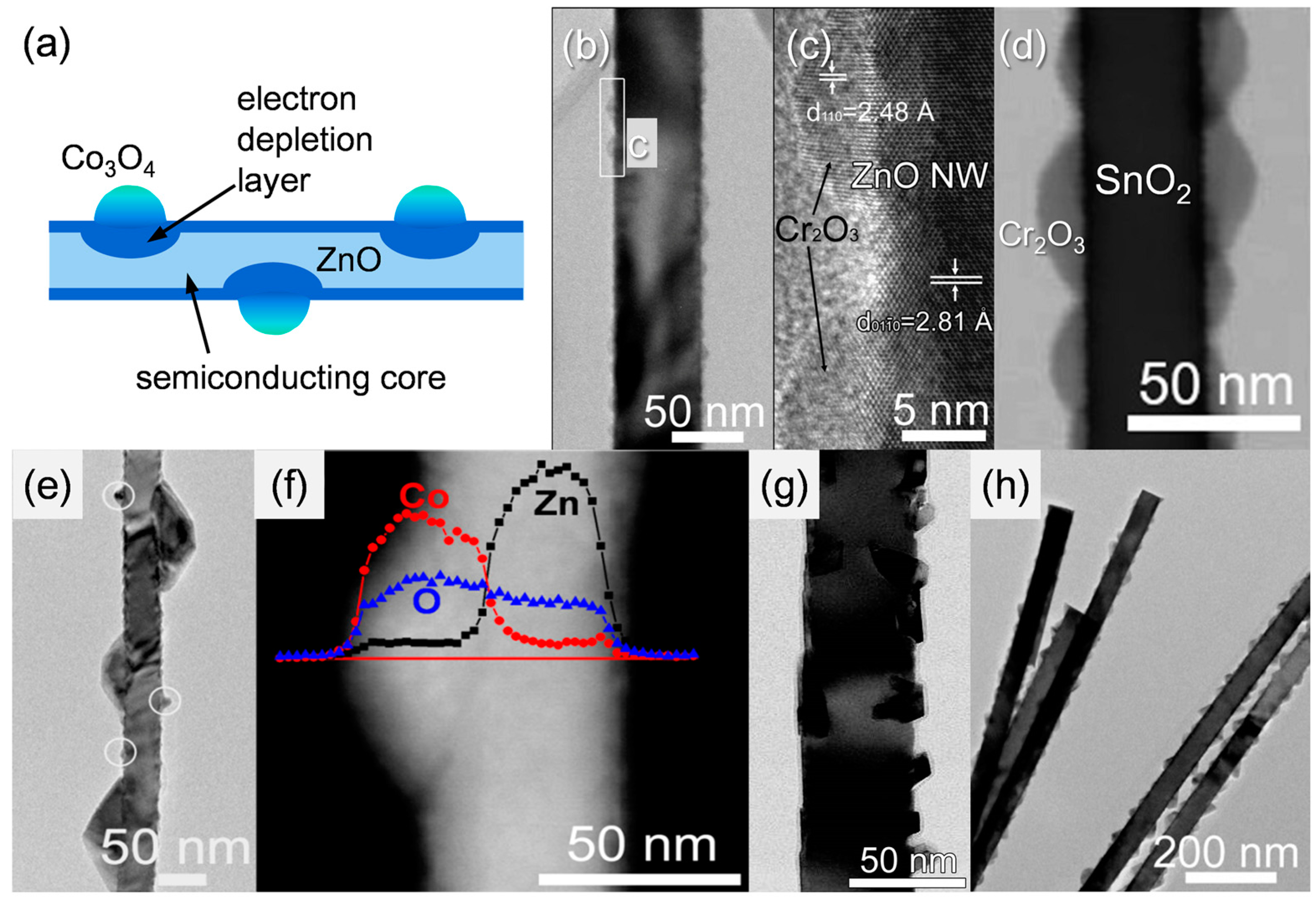
3.2. Transition Metal Oxide Doping/Decoration
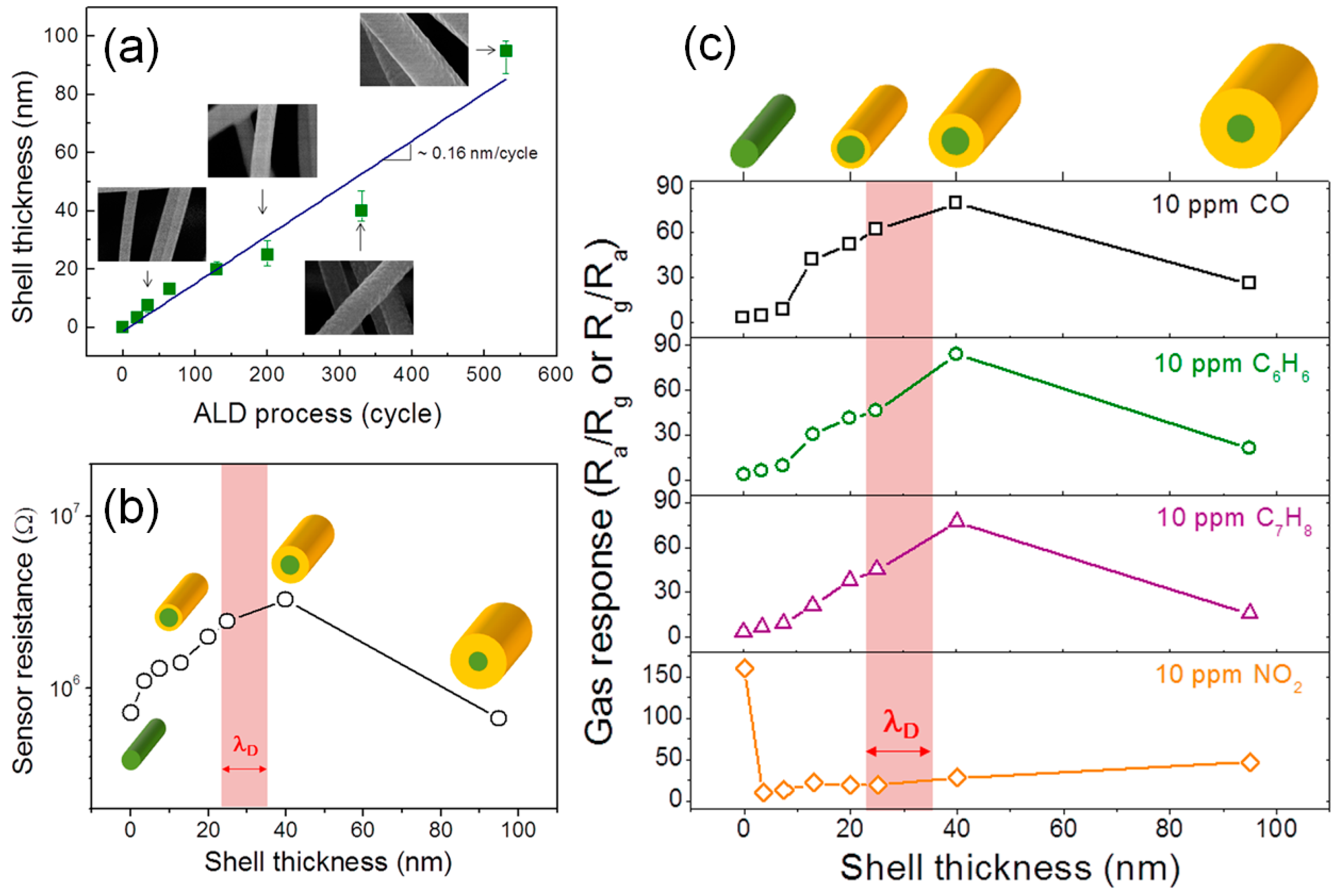
3.3. Core-Shell Structures
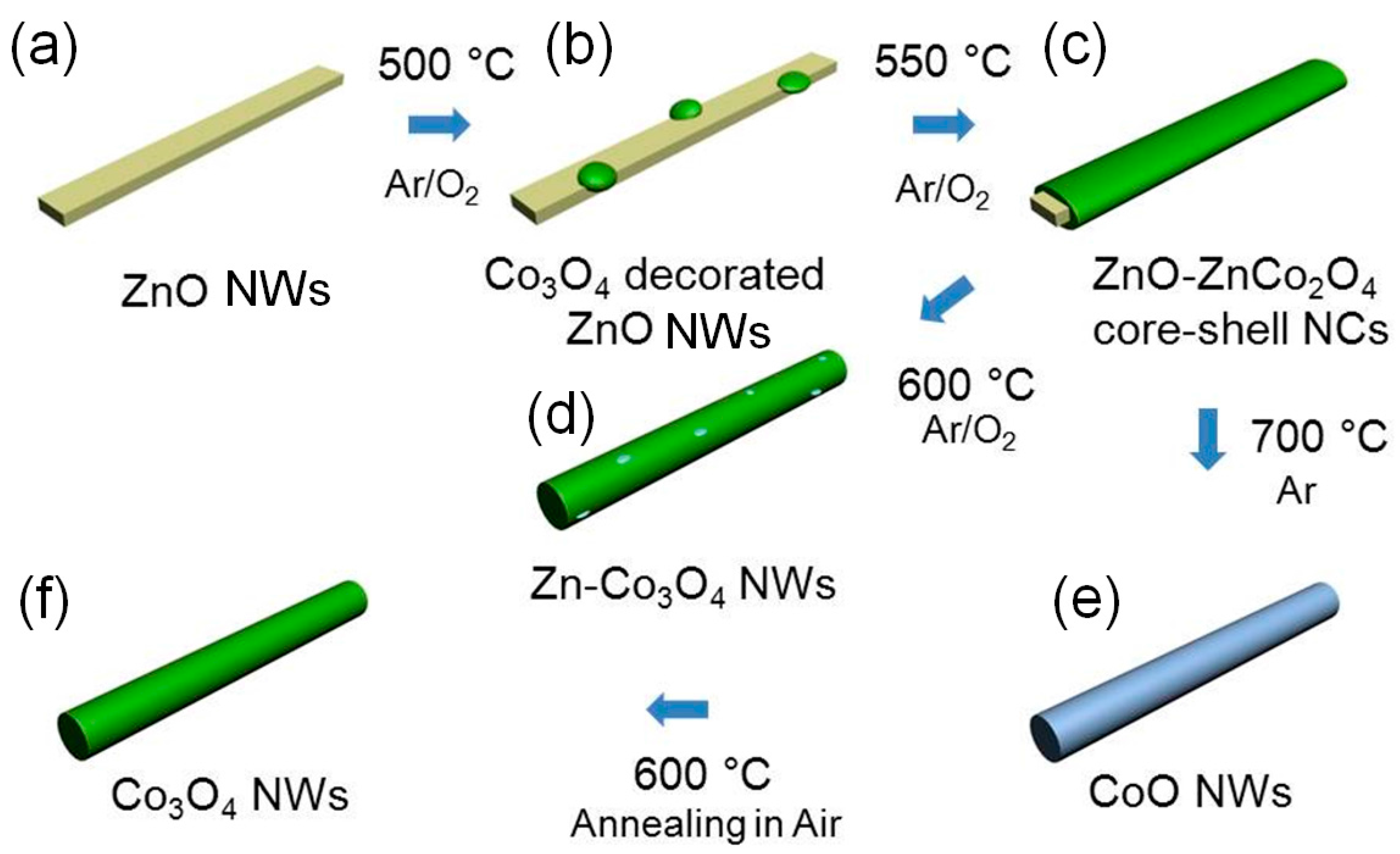
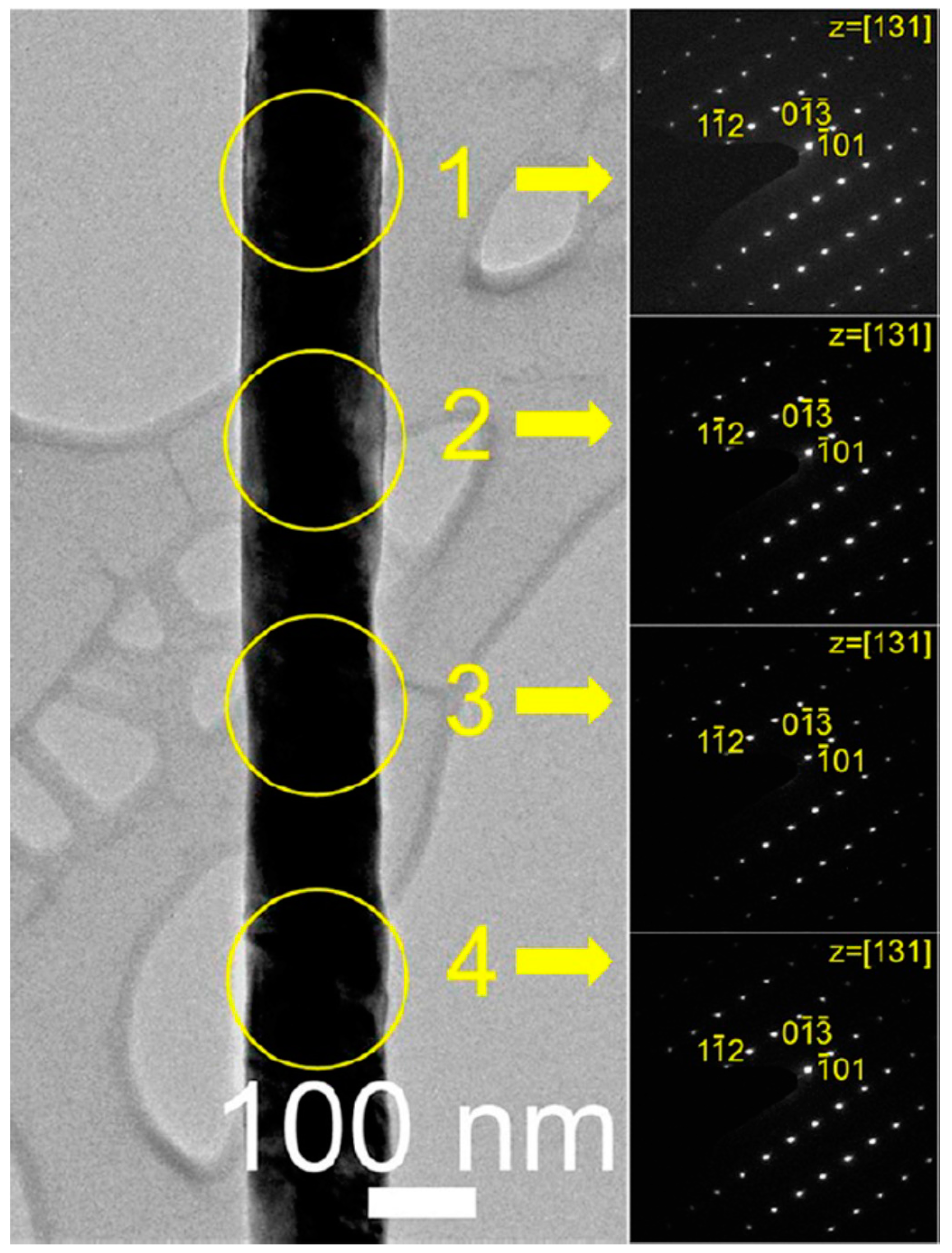
3.4. New Physicochemical Route for the Preparation of p-Type Metal Oxide Nanowires
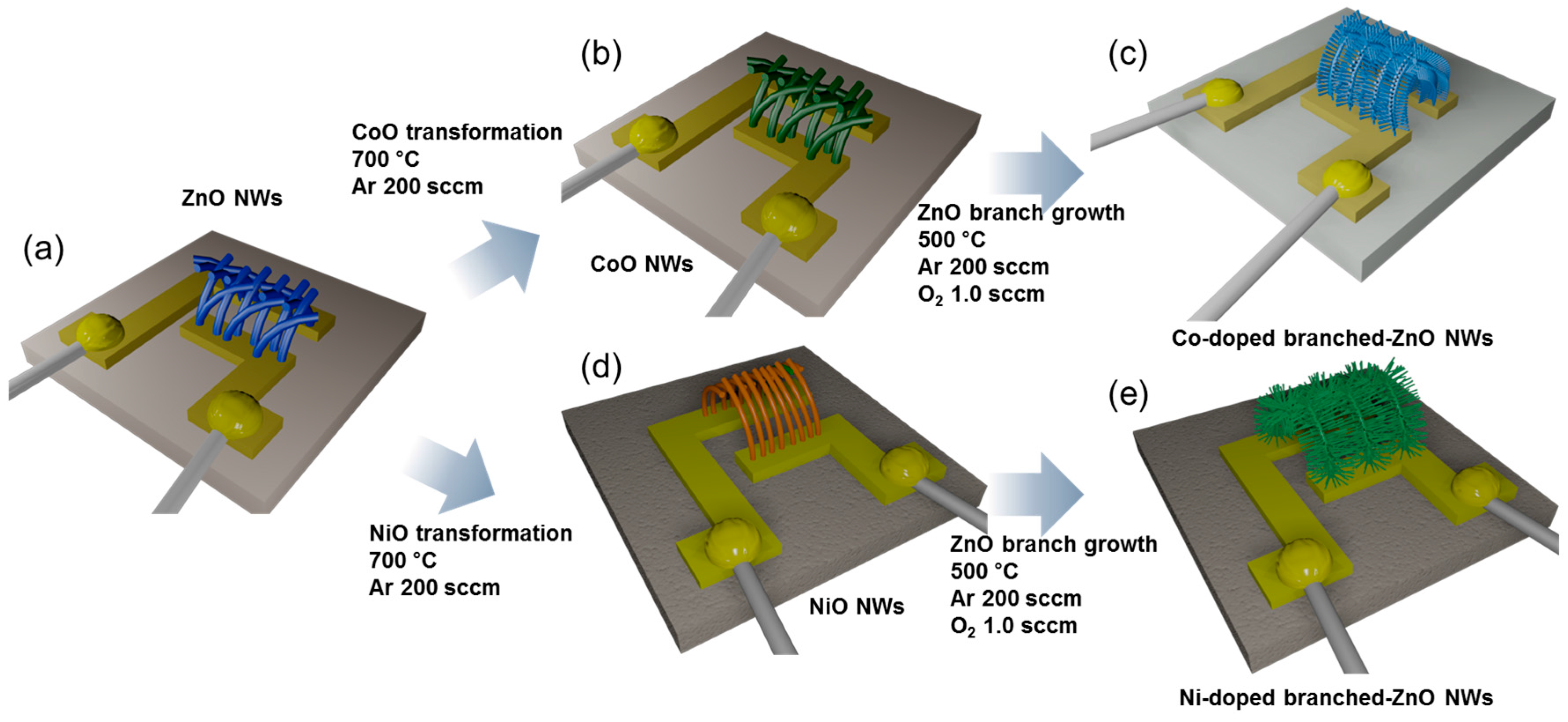
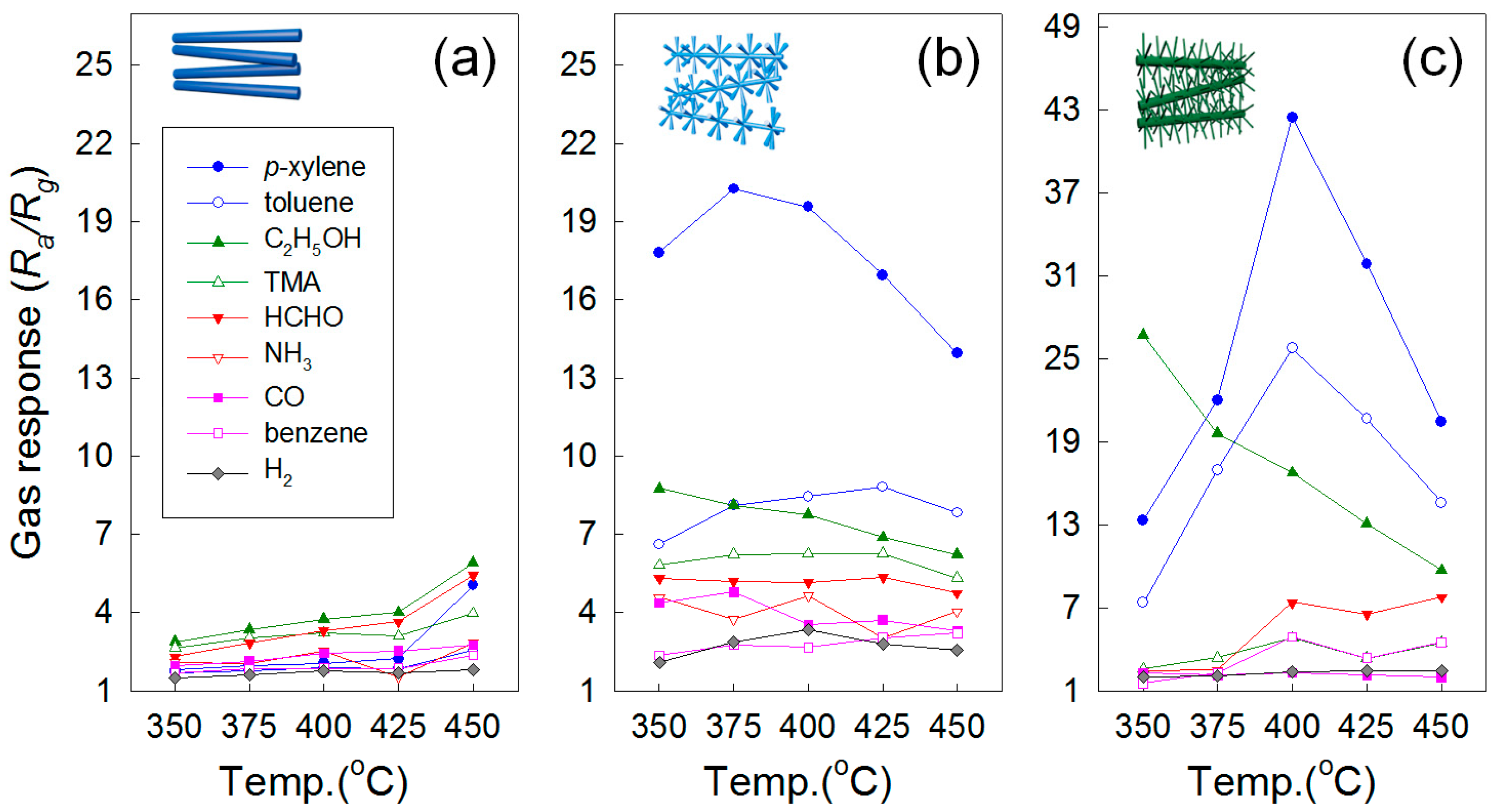
3.5. Hierarchical Structures
3.6. Oxide Nanowires for Selective Gas Detection
4. Other Approaches to Enhance Gas Selectivity
5. Conclusions
Acknowledgments
Conflicts of Interest
References
- Xia, Y.; Yang, P.; Sun, Y.; Wu, Y.; Mayers, B.; Gates, B.; Yin, Y.; Kim, F.; Yan, H. One-dimensional nanostructures: Synthesis, characterization, and applications. Adv. Mater. 2003, 15, 353–389. [Google Scholar] [CrossRef]
- Cheng, Y.; Xiong, P.; Fields, L.; Zheng, J.P.; Yang, R.S.; Wang, Z.L. Intrinsic characteristics of semiconducting oxide nanobelt field-effect transistors. Appl. Phys. Lett. 2006, 89, 093114–093116. [Google Scholar] [CrossRef]
- Kolmakov, A.; Moskovits, M. Chemical sensing and catalysis by one-dimensional metal-oxide nanostructures. Annu. Rev. Mater. Res. 2004, 34, 151–180. [Google Scholar] [CrossRef]
- Comini, E. Metal oxide nano-crystals for gas sensing. Anal. Chim. Acta 2006, 568, 28–40. [Google Scholar] [CrossRef] [PubMed]
- Wan, Q.; Li, Q.H.; Chen, Y.J.; Wang, T.H.; He, X.L. Fabrication and ethanol sensing characteristics of ZnO nanowire gas sensors. Appl. Phys. Lett. 2004, 84, 3654–3656. [Google Scholar] [CrossRef]
- Youn, S.K.; Ramgir, N.; Wang, C.; Subannajui, K.; Cimalla, V.; Zacharias, M. Catalyst-free growth of ZnO nanowires based on topographical confinement and preferential chemisorption and their use for room temperature CO detection. J. Phys. Chem. C 2010, 114, 10092–10100. [Google Scholar] [CrossRef]
- Hwang, I.-S.; Lee, J.-H. Gas sensors using oxide nanowire networks: An overview. J. Nanoeng. Nanomanuf. 2011, 1, 4–17. [Google Scholar] [CrossRef]
- Hwang, I.-S.; Lee, E.-B.; Kim, S.-J.; Choi, J.-K.; Cha, J.-H.; Lee, H.-J.; Ju, B.-K.; Lee, J.-H. Gas sensing properties of SnO2 nanowires on micro-heater. Sens. Actuators B Chem. 2009, 154, 295–300. [Google Scholar] [CrossRef]
- Law, M.; Kind, H.; Messer, B.; Kim, F.; Yang, P. Photochemical sensing of NO2 with SnO2 nanoribbon nanosensors at room temperature. Angew. Chem. Int. Ed. 2002, 114, 2511–2514. [Google Scholar] [CrossRef]
- Fields, L.L.; Zheng, J.P.; Cheng, Y.; Xiong, P. Room-temperature low-power hydrogen sensor based on a single tin dioxide nanobelt. Appl. Phys. Lett. 2006, 88, 263102–263104. [Google Scholar] [CrossRef]
- Sysoev, V.V.; Schneider, T.; Goschnick, J.; Kiselev, I.; Habicht, W.; Strelcov, E.; Kolmakov, A. Percolating SnO2 nanowire network as a stable gas sensor: Direct comparison of long-term performance versus SnO2 nanoparticle films. Sens. Actuators B Chem. 2009, 139, 699–703. [Google Scholar] [CrossRef]
- Sysoev, V.V.; Goschnik, J.; Schneider, T.; Strelcov, E.; Kolmakov, A. A gradient microarray electronic nose based on percolating SnO2 nanowires sensing elements. Nano Lett. 2007, 7, 3182–3188. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.-J.; Hwang, I.-S.; Park, J.-G.; Choi, K.J.; Park, J.-H.; Lee, J.-H. Novel fabrication of an SnO2 nanowire gas sensor with high sensitivity. Nanotechnology 2008, 19, 095508. [Google Scholar] [CrossRef] [PubMed]
- Hsueh, T.-J.; Hsu, C.-L.; Chang, S.-J.; Chen, I.-C. Laterally grown ZnO nanowire ethanol gas sensors. Sens. Actuators B Chem. 2007, 126, 473–477. [Google Scholar] [CrossRef]
- Zhang, D.; Liu, Z.; Li, C.; Tang, T.; Liu, X.; Han, S.; Lei, B.; Zhou, C. Detection of NO2 down to ppb levels using individual and multiple In2O3 nanowire devices. Nano Lett. 2004, 4, 1919–1924. [Google Scholar] [CrossRef]
- Cao, B.; Chen, J.; Tang, X.; Zhou, W. Growth of monoclinic WO3 nanowire array for highly sensitive NO2 detection. J. Mater. Chem. 2009, 19, 2323–2327. [Google Scholar] [CrossRef]
- Chen, X.; Wong, C.K.Y.; Yan, C.A.; Zhang, G. Nanowire-based gas sensors. Sens. Actuators B Chem. 2013, 177, 178–195. [Google Scholar] [CrossRef]
- Wagner, R.S.; Ellis, W.C. Vapor-Liquid-Solid mechanism of single crystal growth. Appl. Phys. Lett. 1964, 4, 89–90. [Google Scholar] [CrossRef]
- Liu, F.; Li, L.; Mo, F.; Chen, J.; Deng, S.; Xu, N. A catalyzed-growth route to directly form micropatterned WO2 and WO3 nanowire arrays with excellent field emission behaviors at low temperature. Cryst. Growth Des. 2010, 10, 5193–5199. [Google Scholar] [CrossRef]
- Lei, M.; Sheng, Y.; Wan, L.; Bi, K.; Huang, K.; Jia, R.; Wang, Y. A novel self-catalytic route to zinc stannate nanowires and cathodoluminescence and electrical transport properties of a single nanowire. J. Alloy Compd. 2016, 657, 394–399. [Google Scholar] [CrossRef]
- Shen, Y.; Turner, S.; Yang, P.; van Tendeloo, G.; Lebedev, O.I.; Wu, T. Epitaxy-enabled vapor-liquid-solid growth of tin-doped indium oxide nanowires with controlled orientations. Nano Lett. 2014, 14, 4342–4351. [Google Scholar] [CrossRef] [PubMed]
- Fu, Y.Y.; Chen, J.; Zhang, J. Synthesis of Fe2O3 nanowires by oxidation of iron. Chem. Phys. Lett. 2001, 350, 491–494. [Google Scholar] [CrossRef]
- Xu, C.H.; Woo, C.H.; Shi, S.Q. Formation of CuO nanowires on foil. Chem. Phys. Lett. 2004, 399, 62–66. [Google Scholar] [CrossRef]
- Lim, J.H.; Choi, J. Formation of niobium oxide nanowires by thermal oxidation. J. Ind. Eng. Chem. 2009, 15, 860–864. [Google Scholar] [CrossRef]
- Gu, Z.; Paranthaman, M.P.; Xu, J.; Pan, J.W. Aligned ZnO nanorod arrays grown directly on zinc foils and zinc spheres by a low-temperature oxidization method. ACS Nano 2009, 3, 273–278. [Google Scholar]
- Rackauskas, S.; Nasibulin, A.G.; Jiang, H.; Tian, Y.; Kleshch, V.I.; Sainio, J.; Obraztsova, E.D.; Bokova, S.N.; Obraztsov, A.N.; Kauppinen, E.I. A novel method for metal oxide nanowire synthesis. Nanotechnology 2009, 20, 165603. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.-H.; Katoch, A.; Choi, S.-W.; Kim, S.S. Growth and sensing properties of networked p-CuO nanowires. Sens. Actuators B Chem. 2015, 212, 190–195. [Google Scholar] [CrossRef]
- Steinhauer, S.; Chapelle, A.; Menini, P.; Sowwan, M. Local CuO nanowire growth on microhotplates: In situ electrical measurement and gas sensing application. ACS Sens. 2016, 1, 503–507. [Google Scholar] [CrossRef]
- Law, J.B.K.; Thong, J.T.L. Improving the NH3 gas sensitivity of ZnO nanowire sensors by reducing the carrier concentration. Nanotechnology 2008, 19, 205502. [Google Scholar] [CrossRef] [PubMed]
- Yamazoe, N.; Fuchigami, J.; Kishikawa, M.; Seiyama, T. Interactions of tin oxide surface with O2, H2O and H2. Surf. Sci. 1979, 86, 335–344. [Google Scholar] [CrossRef]
- Chang, S.-C. Oxygen chemisorption on tin oxide: Correlation between electrical conductivity and EPR measurements. J. Vac. Sci. Technol. 1980, 17, 366–369. [Google Scholar] [CrossRef]
- Cretu, V.; Postica, V.; Mishra, A.K.; Hoppe, M.; Tiginyanu, I.; Mishra, Y.K.; Chow, L.; Leeuw, N.H.; Adelung, R.; Lupan, O. Synthesis, characterization and DFT studies of zinc-doped copper oxide nanocrystals for gas sensing applications. J. Mater. Chem. A 2016, 4, 6527–6539. [Google Scholar] [CrossRef]
- Yamazoe, N.; Suematsu, K.; Shimanoe, K. Surface chemistry of neat tin oxide sensor for response to hydrogen. Sens. Actuators B Chem. 2016, 227, 403–410. [Google Scholar] [CrossRef]
- Xu, C.N.; Tamaki, J.; Miura, N.; Yamazoe, N. Grain size effects on gas sensitivity of porous SnO2-based elements. Sens. Actuators B Chem. 1991, 3, 147–155. [Google Scholar] [CrossRef]
- Fan, Z.; Wang, D.; Chang, P.C.; Tseng, W.Y.; Lu, J.G. ZnO nanowire field-effect transistor and oxygen sensing property. Appl. Phys. Lett. 2004, 85, 5923–5925. [Google Scholar] [CrossRef]
- Lee, J.-H. Gas sensors using hierarchical and hollow oxide nanostructures: Overview. Sens. Actuators B Chem. 2009, 140, 319–336. [Google Scholar] [CrossRef]
- Kim, B.-K.; Choi, S.-D. SnO2 thin film gas sensor fabrication by ion beam deposition. Sens. Actuators B Chem. 2004, 98, 239–246. [Google Scholar]
- Shoyama, M.; Hashimoto, N. Effect of poly ethylene glycol addition on the microstructure and sensor characteristics of SnO2 thin films prepared by sol-gel method. Sens. Actuators B Chem. 2003, 93, 585–589. [Google Scholar] [CrossRef]
- Korotchenkov, G. Gas response control through structural and chemical modification of metal oxide films: State of the art and approaches. Sens. Actuators B Chem. 2005, 107, 209. [Google Scholar] [CrossRef]
- Hwang, I.-S.; Kim, Y.-S.; Kim, S.-J.; Ju, B.-K.; Lee, J.-H. A facile fabrication of semiconductor nanowires gas sensor using PDMS patterning and solution deposition. Sens. Actuators B Chem. 2009, 136, 224–229. [Google Scholar] [CrossRef]
- Van den Broek, A.M.W.T.; Feenstra, L.; de Baat, C. A review of the current literature on aetiology and measurement methods of halitosis. J. Dent. 2007, 35, 627–635. [Google Scholar] [CrossRef] [PubMed]
- Loesche, W.J.; Kazor, C. Microbiology and treatment of halitosis. Periodontology 2000 2002, 28, 256–279. [Google Scholar] [CrossRef] [PubMed]
- Deng, C.; Zhang, J.; Yu, X.; Zhang, W.; Zhang, X. Determination of acetone in human breath by gas chromatography-mass spectrometry and solid-phase microextraction with on-fiber derivation. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2004, 810, 269–275. [Google Scholar] [CrossRef]
- Tseliou, E.; Bessa, V.; Hillas, G.; Delimpoura, V.; Papadaki, G.; Roussos, C.; Papiris, S.; Bakakos, P.; Loukides, S. Exhaled nitric oxide and exhaled breath condensate pH in severe refractory asthma. Chest 2015, 138, 107–113. [Google Scholar] [CrossRef] [PubMed]
- Moon, H.G.; Choi, Y.R.; Shim, Y.-S.; Choi, K.-I.; Lee, J.-H.; Kim, J.-S.; Yoon, S.-J.; Park, H.-H.; Kang, C.-Y.; Jang, H.W. Extremely sensitive and selective NO probe based on villi-like WO3 nanostructures for application to exhaled breath analyzers. ACS Appl. Mater. Interfaces 2013, 5, 10591–10596. [Google Scholar] [CrossRef] [PubMed]
- Davis, S.; Spanel, P.; Smith, D. Quantitative analysis of ammonia on the breath of patients in end-stage renal failure. Kidney Int. 1997, 52, 223–228. [Google Scholar] [CrossRef]
- Horvath, I.; Loukides, S.; Wodehouse, T.; Kharitonov, S.; Cole, P.; Barnes, P. Increased levels of exhaled carbon monoxide in bronchiectasis: A new marker of oxidative stress. Thorax 1998, 53, 867–870. [Google Scholar] [CrossRef] [PubMed]
- Yamazoe, N.; Sakai, G.; Shimanoe, K. Oxide semiconductor gas sensors. Catal. Surv. Asia 2003, 7, 63–75. [Google Scholar] [CrossRef]
- Huang, X.-J.; Choi, Y.-K. Chemical sensors based on nanostructured materials. Sens. Actuators B Chem. 2007, 122, 659–671. [Google Scholar] [CrossRef]
- Haridas, D.; Sreenivas, K.; Gupta, V. Improved response characteristics of SnO2 thin film loaded with nanoscale catalyst for LPG detection. Sens. Actuators B Chem. 2008, 133, 270–275. [Google Scholar] [CrossRef]
- Xue, X.-Y.; Chen, Z.-H.; Xing, L.-L.; Ma, C.-H.; Chen, Y.-J.; Wang, T.-H. Enhanced optical and sensing properties of one-step synthesized Pt-ZnO nanoflowers. J. Phys. Chem. C 2010, 114, 18607–15509. [Google Scholar] [CrossRef]
- Yuasa, M.; Masaki, T.; Kida, T.; Shimanoe, K.; Yamazoe, N. Nano-sized PdO loaded SnO2 nanoparticles by reverse micelle method for highly sensitive CO gas sensor. Sens. Actuators B Chem. 2009, 136, 99–104. [Google Scholar] [CrossRef]
- Gaspera, E.D.; Guglielmi, M.; Martucci, A.; Giancaterini, L.; Cantalini, C. Enhanced optical and electrical gas sensing response of sol-gel based NiO-Au and ZnO-Au nanostructures thin films. Sens. Actuators B Chem. 2012, 164, 54–63. [Google Scholar] [CrossRef]
- Sun, G.-J.; Choi, S.-W.; Jung, S.-H.; Katoch, A.; Kim, S.S. V-groove SnO2 nanowire sensors: Fabrication and Pt-nanoparticle decoration. Nanotechnology 2013, 24, 025504. [Google Scholar] [CrossRef] [PubMed]
- Shen, Y.; Yamazaki, T.; Liu, Z.; Meng, D.; Kikuta, T. Hydrogen sensors made of undoped and Pt-doped SnO2 nanowires. J. Alloys Compd. 2009, 488, L21–L25. [Google Scholar] [CrossRef]
- Choi, S.-W.; Katoch, A.; Sun, G.-J.; Kim, S.S. Bimetallic Pd/Pt nanoparticle-functionalized SnO2 nanowires for fast response and recovery to NO2. Sens. Actuators B Chem. 2013, 181, 446–453. [Google Scholar] [CrossRef]
- Zhang, Y.; Xu, J.; Xu, P.; Zhu, Y.; Chen, X.; Yu, W. Decoration of ZnO nanowires with Pt nanoparticles and their improved gas sensing and photocatalytic performance. Nanotechnology 2010, 21, 285501. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.S.; Park, J.Y.; Choi, S.-W.; Kim, H.S.; Na, H.G.; Yang, J.C.; Kim, H.W. Significant enhancement of the sensing characteristics of In2O3 nanowires by functionalization with Pt nanoparticles. Nanotechnology 2010, 21, 415502. [Google Scholar] [CrossRef] [PubMed]
- Zou, X.; Wang, J.; Liu, X.; Wang, C.; Jiang, Y.; Wang, Y.; Xiao, X.; Ho, J.C.; Li, J.; Jiang, C.; et al. Rational design of sub-parts per million specific gas sensors array based on metal nanoparticles decorated nanowire enhancement-mode transistors. Nano Lett. 2013, 13, 3287–3292. [Google Scholar] [CrossRef] [PubMed]
- Kolmakov, A.; Klenov, D.; Lilach, Y.; Stemmer, S.; Moskovits, M. Enhanced gas sensing by individual SnO2 nanowires and nanobelts functionalized with Pd catalyst particles. Nano Lett. 2005, 5, 667–673. [Google Scholar] [CrossRef] [PubMed]
- Baik, J.M.; Zielke, M.; Kim, M.H.; Turner, K.L.; Wodtke, A.M.; Moskovits, M. Tin-oxide-nanowire-based electronic nose using heterogeneous catalysis as a functionalization strategy. ACS Nano 2010, 4, 3117–3122. [Google Scholar] [CrossRef] [PubMed]
- Baik, J.M.; Kim, M.H.; Larson, C.; Yavuz, C.T.; Stucky, G.D.; Wodtke, A.M.; Moskovits, M. Pd-sensitized single vanadium oxide nanowires: Highly responsive hydrogen sensing based on the metal-insulator transition. Nano Lett. 2009, 9, 3980–3984. [Google Scholar] [CrossRef] [PubMed]
- Joshi, R.K.; Hu, Q.; Alvi, F.; Joshi, N.; Kumar, A. Au decorated zinc oxide nanowires for CO sensing. J. Phys. Chem. C 2009, 113, 16199–16202. [Google Scholar] [CrossRef]
- Hongsith, N.; Viriyaworasakul, C.; Mangkorntong, P.; Magkorntong, N.; Choopun, S. Ethanol sensor based on ZnO and Au-doped ZnO nanowires. Ceram. Int. 2008, 34, 823–826. [Google Scholar] [CrossRef]
- Xiang, Q.; Meng, G.; Zhang, Y.; Xu, J.; Xu, P.; Pan, Q.; Yu, W. Ag nanoparticle embedded-ZnO nanorods synthesized via a photochemical method and its gas-sensing properties. Sens. Actuators B Chem. 2010, 143, 635–640. [Google Scholar] [CrossRef]
- Hwang, I.-S.; Choi, J.-K.; Woo, H.-S.; Kim, S.-J.; Jung, S.-Y.; Seong, T.-Y.; Kim, I.-D.; Lee, J.-H. Facile control of C2H5OH sensing characteristics by decorating discrete Ag nanoclusters on SnO2 nanowire networks. ACS Appl. Mater. Interfaces 2011, 3, 3140–3145. [Google Scholar] [CrossRef] [PubMed]
- Joshi, R.K.; Kruis, F.E. Influence of Ag particle size on ethanol sensing of SnO1.8:Ag nanoparticle films: A method to develop parts per billion level gas sensors. Appl. Phys. Lett. 2006, 89, 153116. [Google Scholar] [CrossRef]
- Chen, X.H.; Moskovits, M. Observing catalysis through the agency of the participating electrons: Surface-chemistry-induced current changes in a tin oxide nanowire decorated with silver. Nano Lett. 2007, 7, 807–812. [Google Scholar] [CrossRef] [PubMed]
- Yamazoe, N. New approaches for improving semiconductor gas sensors. Sens. Actuators B Chem. 1991, 5, 7–19. [Google Scholar] [CrossRef]
- Kandoi, S.; Gokhale, A.A.; Grabow, L.C.; Dumesic, J.A.; Mavrikakis, M. Why Au and Cu are more selective than Pt for preferential oxidation of CO at low temperature. Catal. Lett. 2004, 93, 93–100. [Google Scholar] [CrossRef]
- Haruta, M. Gold as a novel catalyst in the 21st century: Preparation, working mechanism and applications. Gold Bull. 2004, 37, 27–36. [Google Scholar] [CrossRef]
- Schubert, M.M.; Hackenberg, S.; van Veen, A.C.; Muhler, M.; Plzak, V.; Behm, R.J. CO oxidation over supported gold catalyst-“inter” and “active” support materials and their role for the oxygen supply during reaction. J. Catal. 2001, 197, 113–122. [Google Scholar] [CrossRef]
- Tien, L.C.; Sadik, P.W.; Norton, D.P.; Voss, L.F.; Pearton, S.J.; Wang, H.T.; Kang, B.S.; Ren, F.; Jun, J.; Lin, J. Hydrogen sensing at room temperature with Pt-coated ZnO thin films and nanorods. Appl. Phys. Lett. 2005, 87, 222106. [Google Scholar] [CrossRef]
- Kryliouk, O.; Park, H.J.; Wang, H.T.; Kang, B.S.; Anderson, T.J.; Ren, F.; Pearton, S.J. Pt-coated InN nanorods for selective detection of hydrogen at room temperature. J. Vac. Sci. Technol. B 2005, 23, 1891–1894. [Google Scholar] [CrossRef]
- Hu, P.; Du, G.; Zhou, W.; Cui, J.; Lin, J.; Liu, H.; Liu, D.; Wang, J.; Chen, S. Enhancement of ethanol vapor sensing of TiO2 nanobelts by surface engineering. ACS Appl. Mater. Interfaces 2010, 2, 3263–3269. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zheng, Z.; Yang, F. Highly sensitive and selective alcohol sensors based on Ag-doped In2O3 coating. Ind. Eng. Chem. Res. 2010, 49, 3539–3543. [Google Scholar] [CrossRef]
- Kim, H.-J.; Lee, J.-H. Highly sensitive and selective gas sensors using p-type oxide semiconductors: Overview. Sens. Actuators B Chem. 2014, 195, 189–196. [Google Scholar] [CrossRef]
- Xue, X.; Xing, L.; Chen, Y.; Shi, S.; Wang, Y.; Wang, T. Synthesis and H2S sensing properties of CuO-SnO2 core/shell PN-junction nanorods. J. Phys. Chem. C 2008, 112, 12157–12160. [Google Scholar] [CrossRef]
- Giebelhaus, I.; Varechkina, E.; Fischer, T.; Rumyantseva, M.; Ivanov, V.; Gaskov, A.; Morante, J.R.; Arbiol, J.; Tyrra, W.; Mathur, S. One-dimensional CuO-SnO2 p-n heterojunctions for enhanced detection of H2S. J. Mater. Chem. A 2013, 1, 11261–11268. [Google Scholar] [CrossRef]
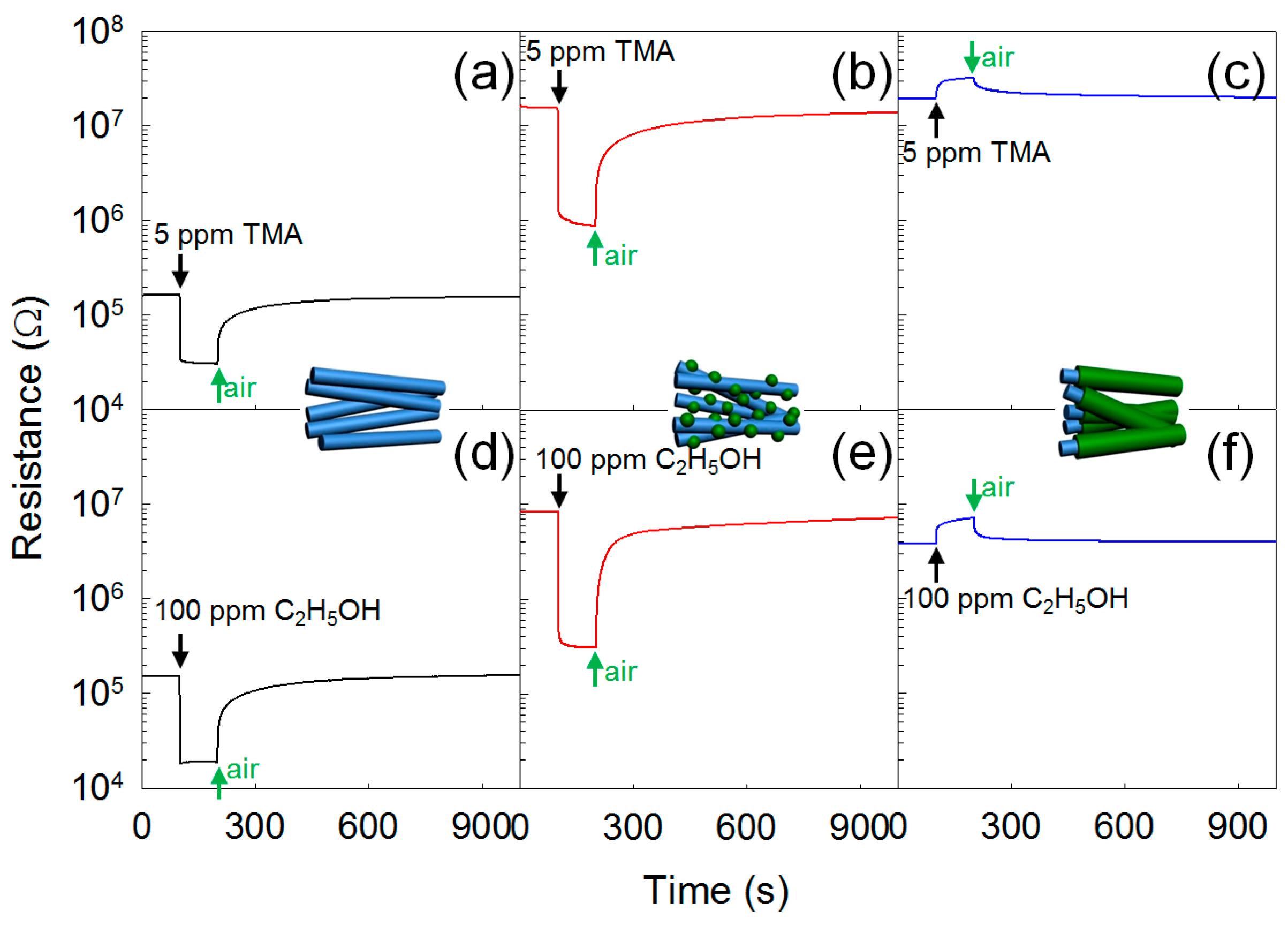
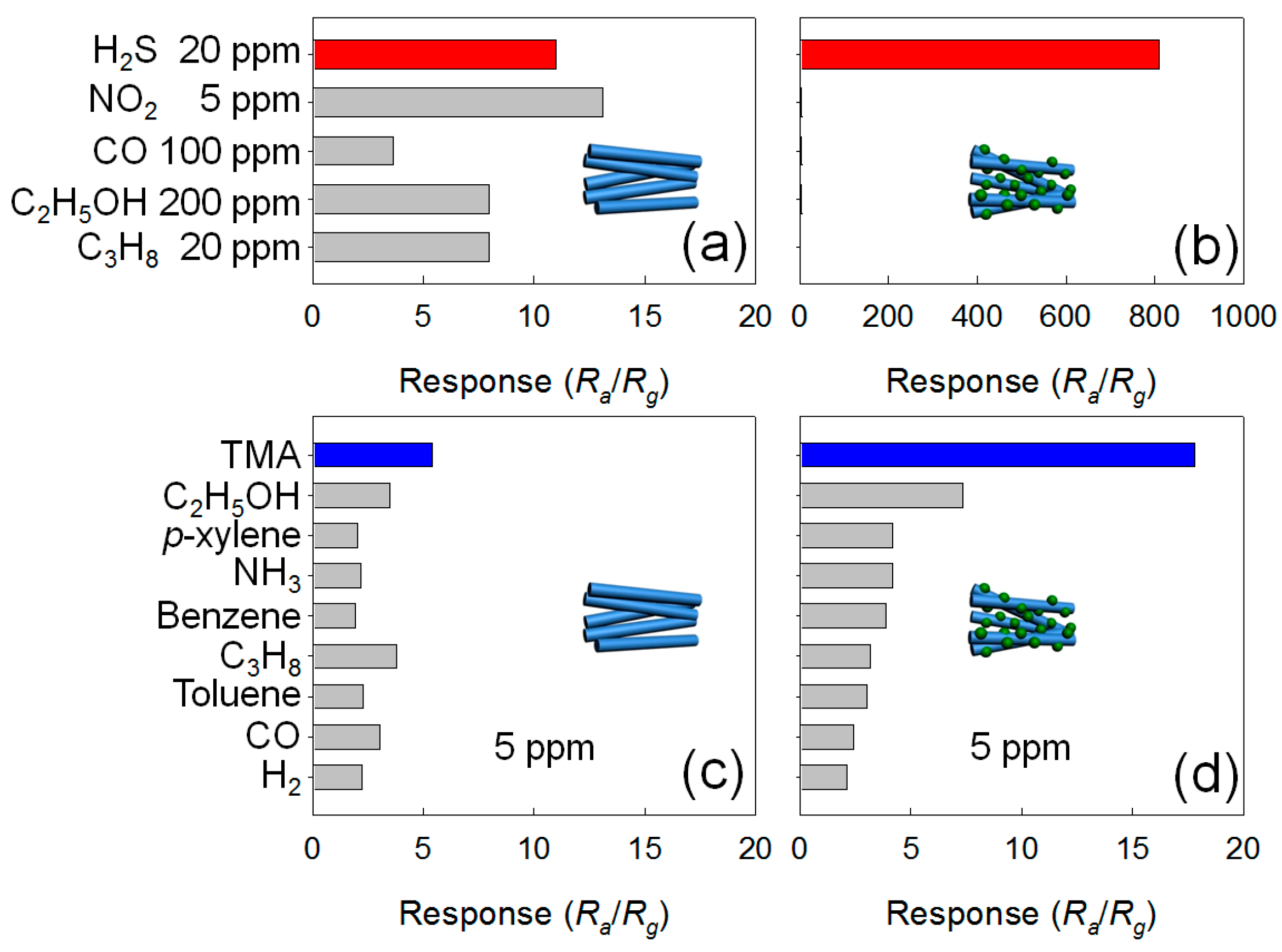
- Woo, H.-S.; Na, C.W.; Kim, I.-D.; Lee, J.-H. Highly sensitive and selective trimethylamine sensor using one-dimensional ZnO-Cr2O3 hetero-nanostructures. Nanotechnology 2012, 23, 245501. [Google Scholar] [CrossRef] [PubMed]
- Kwak, C.-H.; Woo, H.-S.; Lee, J.-H. Selective trimethylamine sensors using Cr2O3-decorated SnO2 nanowires. Sens. Actuators B Chem. 2014, 204, 231–238. [Google Scholar] [CrossRef]
- Na, C.W.; Woo, H.-S.; Kim, I.-D.; Lee, J.-H. Selective detection of NO2 and C2H5OH using a Co3O4-decorated ZnO nanowire network sensor. Chem. Commun. 2011, 47, 5148–5150. [Google Scholar] [CrossRef] [PubMed]
- Na, C.W.; Woo, H.-S.; Lee, J.-H. Design of highly sensitive volatile organic compound sensors by controlling NiO loading on ZnO nanowire networks. RSC Adv. 2012, 2, 414–417. [Google Scholar] [CrossRef]
- Na, C.W.; Park, S.-Y.; Chung, J.-H.; Lee, J.-H. Transformation of ZnO nanobelts into single-crystalline Mn3O4 nanowires. ACS Appl. Mater. Interfaces 2012, 4, 6565–6572. [Google Scholar] [CrossRef] [PubMed]
- Hieu, N.V.; Kim, H.-R.; Ju, B.-K.; Lee, J.-H. Enhanced performance of SnO2 nanowires ethanol sensor by functionalizing with La2O3. Sens. Actuators B Chem. 2008, 133, 228–234. [Google Scholar] [CrossRef]
- Khanna, A.; Kumar, R.; Bhatti, S.S. CuO doped SnO2 thin films as hydrogen sulfide gas sensor. Appl. Phys. Lett. 2003, 82, 4388–4390. [Google Scholar] [CrossRef]
- Kumar, V.; Sen, S.; Muthe, K.P.; Gaur, N.K.; Gupta, S.K.; Yakhmi, J.V. Copper doped SnO2 nanowires as highly sensitive H2S gas sensor. Sens. Actuators B Chem. 2009, 138, 587–590. [Google Scholar] [CrossRef]
- Woo, H.-S.; Kwak, C.-H.; Kim, I.-D.; Lee, J.-H. Selective, sensitive, and reversible detection of H2S using Mo-doped ZnO nanowire network sensors. J. Mater. Chem. A 2014, 2, 6412–6418. [Google Scholar] [CrossRef]
- Kwak, C.-H.; Woo, H.-S.; Abdel-Hady, F.; Wazzan, A.A.; Lee, J.-H. Vapor-phase growth of urchin-like Mg-doped ZnO nanowire networks and their application to highly sensitive and selective detection of ethanol. Sens. Actuators B Chem. 2016, 223, 527–534. [Google Scholar] [CrossRef]
- Wan, Q.; Wang, T.H. Single-crystalline Sb-doped SnO2 nanowires: Synthesis and gas sensor application. Chem. Commun. 2005, 30, 3481–3843. [Google Scholar] [CrossRef] [PubMed]
- Hwang, I.-S.; Choi, J.-K.; Kim, S.-J.; Dong, K.Y.; Kwon, J.-H.; Ju, B.-K.; Lee, J.-H. Enhanced H2S sensing characteristics of SnO2 nanowire functionalized with CuO. Sens. Actuators B Chem. 2009, 142, 105–110. [Google Scholar] [CrossRef]
- Borck, Ø.; Hyldegaard, P.; Schroder, E. Adsorption of methylamine on α-Al2O3 (0001) and α-Cr2O3 (0001): Density functional theory. Phys. Rev. B 2007, 75, 035403. [Google Scholar] [CrossRef]
- Jinkawa, T.; Sakai, G.; Tamaki, J.; Miura, N.; Yamazoe, N. Relationship between ethanol gas sensitivity and surface catalytic property of tin oxide sensors modified with acidic or basic oxides. J. Mol. Catal. A Chem. 2000, 155, 193–200. [Google Scholar] [CrossRef]
- Kim, S.-J.; Na, C.W.; Hwang, I.-S.; Lee, J.-H. One-pot hydrothermal synthesis of CuO-ZnO composite hollow spheres for selective H2S detection. Sens. Actuators B Chem. 2012, 168, 83–89. [Google Scholar] [CrossRef]
- Liang, X.; Kim, T.-H.; Yoon, J.-W.; Kwak, C.-H.; Lee, J.-H. Ultrasensitive and ultraselective detection of H2S using electrospun CuO-loaded In2O3 nanofiber sensors assisted by pulse heating. Sens. Actuators B Chem. 2015, 209, 934–942. [Google Scholar] [CrossRef]
- Lee, S.C.; Kim, S.Y.; Hwang, B.W.; Jung, S.Y.; Ragupathy, D.; Son, I.S.; Lee, D.D.; Kim, J.C. Improvement of H2S sensing properties of SnO2-based thick film gas sensors promoted with MoO3 and NiO. Sensors 2013, 13, 3889–3901. [Google Scholar] [CrossRef] [PubMed]
- Matsuda, S.; Kamo, T.; Imahashi, J.; Nakajima, F. Adsorption and oxidative desorption of hydrogen sulfide by molybdenum trioxide-titanium dioxide. Ind. Eng. Chem. Res. 1982, 21, 18–22. [Google Scholar] [CrossRef]
- Ohtomo, A.; Shiroki, R.; Ohkubo, I.; Koinuma, H.; Kwasaki, M. Thermal stability of supersaturated MgxZn1−xO alloy films and MgxZn1−xO/ZnO heterointerface. Appl. Phys. Lett. 1999, 75, 4088–4090. [Google Scholar] [CrossRef]
- Minemoto, T.; Negami, T.; Nishiwaki, S.; Takahara, H.; Hamakawa, Y. Preparation of Zn1-xMgxO films by radio frequency magnetron sputtering. Thin Solid Films 2000, 372, 173–176. [Google Scholar] [CrossRef]
- Kim, T.H.; Park, J.J.; Nam, S.H.; Park, H.S.; Cheong, N.R.; Song, J.K.; Park, S.M. Fabrication of Mg-doped thin films by laser ablation of Zn:Mg target. Appl. Surf. Sci. 2009, 255, 5264–5266. [Google Scholar] [CrossRef]
- Park, S.; An, S.; Mun, Y.; Lee, C. UV-Enhanced NO2 gas sensing properties of SnO2-core/ZnO-shell nanowires at room temperature. ACS Appl. Mater. Interfaces 2013, 5, 4285–4292. [Google Scholar] [CrossRef] [PubMed]
- Le, D.T.T.; Trung, D.D.; Chinh, N.D.; Binh, D.T.T.; Hong, H.S.; Duy, N.V.; Hoa, N.D.; Hieu, N.V. Facile synthesis of SnO2-ZnO core-shell nanowires for enhanced ethanol-sensing performance. Curr. Appl. Phys. 2013, 13, 1637–1642. [Google Scholar]
- Hwang, I.-S.; Kim, S.-J.; Choi, J.-K.; Choi, J.; Ji, H.; Kim, G.-T.; Cao, G.; Lee, J.-H. Synthesis and gas sensing characteristics of highly crystalline ZnO-SnO2 core-shell nanowires. Sens. Actuators B Chem. 2010, 148, 595–600. [Google Scholar] [CrossRef]
- Choi, S.-W.; Katoch, A.; Sun, G.-J.; Kim, J.-H.; Kim, S.-H.; Kim, S.-S. Dual functional sensing mechanism in SnO2-ZnO core-shell nanowires. ACS Appl. Mater. Interfaces 2014, 6, 8281–8287. [Google Scholar] [CrossRef] [PubMed]
- Na, C.W.; Woo, H.-S.; Kim, H.-J.; Jeong, U.; Chung, J.-H.; Lee, J.-H. Controlled transformation of ZnO nanobelts into CoO/Co3O4 nanowires. CrystEngComm 2012, 14, 3737–3741. [Google Scholar] [CrossRef]
- Schwebel, T.; Fleischer, M.; Meixner, H. A selective, temperature compensated O2 sensor based on Ga2O3 thin films. Sens. Actuators B Chem. 2000, 65, 176–180. [Google Scholar] [CrossRef]
- Ogita, M.; Hiro, K.; Nakanishi, Y.; Hatanaka, Y. Ga2O3 thin film for oxygen sensor at high temperature. Appl. Surf. Sci. 2001, 175–176, 721–725. [Google Scholar] [CrossRef]
- Fleischer, M.; Meixner, H. Sensing reducing gases at high temperatures using long-term stable Ga2O3 thin films. Sens. Actuators B Chem. 1992, 6, 257–261. [Google Scholar] [CrossRef]
- Fleischer, M.; Giber, J.; Meixner, H. H2-induced changes in electrical conductance of β-Ga2O3 thin film system. Appl. Phys. A 1992, 54, 560–566. [Google Scholar] [CrossRef]
- Schwebel, T.; Fleischer, M.; Meixner, H.; Kohl, C.D. CO-sensor for domestic use based on high temperature stable Ga2O3 thin films. Sens. Actuators B Chem. 1998, 49, 46–51. [Google Scholar] [CrossRef]
- Jin, C.; Park, S.; Kim, H.; Lee, C. Ultrasensitive multiple networked Ga2O3-core/ZnO-shell nanorod gas sensors. Sens. Actuators B Chem. 2012, 161, 223–228. [Google Scholar] [CrossRef]
- Jang, Y.-G.; Kim, W.-S.; Kim, D.-H.; Hong, S.-H. Fabrication of Ga2O3/SnO2 core-shell nanowires and their ethanol gas sensing properties. J. Mater. Res. 2011, 26, 2322–2327. [Google Scholar] [CrossRef]
- Yamazoe, N.; Shimanoe, K. New perspective of gas sensor technology. Sens. Actuators B Chem. 2015, 138, 100–107. [Google Scholar] [CrossRef]
- Hübner, M.; Simion, C.E.; Tomescu-Stănoiu, A.; Pokhrel, S.; Barsan, N.; Weimer, U. Influence of humidity on CO sensing with p-type CuO think film gas sensors. Sens. Actuators B Chem. 2011, 153, 347–353. [Google Scholar] [CrossRef]
- Iwamoto, M.; Yoda, Y.; Yamazoe, N.; Seiyama, T. Study of metal oxide catalysts by temperature programmed desorption. 4. Oxygen adsorption on various metal oxides. J. Phys. Chem. 1978, 82, 2564–2570. [Google Scholar] [CrossRef]
- Kim, K.-M.; Jeong, H.-M.; Kim, H.-R.; Choi, K.-I.; Kim, H.-J.; Lee, J.-H. Selective Detection of NO2 Using Cr-Doped CuO Nanorods. Sensors 2012, 12, 8013–8025. [Google Scholar] [CrossRef] [PubMed]
- Deng, J.; Zhang, R.; Wang, L.; Lou, Z.; Zhang, T. Enhanced sensing performance of the Co3O4 hierarchical nanorods to NH3 gas. Sens. Actuators B Chem. 2015, 209, 449–455. [Google Scholar] [CrossRef]
- Jeong, H.-M.; Kim, H.-J.; Rai, P.; Yoon, J.-W.; Lee, J.-H. Cr-doped Co3O4 nanorods as chemiresistor for ultraselective monitoring of methyl benzene. Sens. Actuators B Chem. 2014, 201, 482–489. [Google Scholar] [CrossRef]
- Park, J.; Shen, X.; Wang, G. Solvothermal synthesis and gas-sensing performance of Co3O4 hollow nanospheres. Sens. Actuators B Chem. 2009, 136, 494–498. [Google Scholar] [CrossRef]
- Kim, H.-J.; Jeong, H.-M.; Kim, T.-H.; Chung, J.-H.; Kang, Y.C.; Lee, J.-H. Enhanced ethanol sensing characteristics of In2O3-decorated NiO hollow nanostructures via modulation of hole accumulation layer. ACS Appl. Mater. Interfaces 2014, 6, 18197–18204. [Google Scholar] [CrossRef] [PubMed]
- Cao, A.-M.; Hu, J.-S.; Liang, H.-P.; Song, W.-G.; Wan, L.-J.; He, X.-L.; Gao, X.-G.; Xia, S.-H. Hierarchically structured cobalt oxide (Co3O4): The morphology control and its potential in sensors. J. Phys. Chem. B 2006, 110, 15858–15863. [Google Scholar] [CrossRef] [PubMed]
- Hwang, S.-J.; Choi, K.-I.; Yoon, J.-W.; Kang, Y.C.; Lee, J.-H. Pure and palladium-loaded Co3O4 hollow hierarchical nanostructures with giant and ultraselective Chemiresistivity to xylene and toluene. Chem. Eur. J. 2015, 21, 5872–5878. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.-J.; Yoon, J.-W.; Choi, K.-I.; Jang, H.W.; Umar, A.; Lee, J.-H. Ultraselective and sensitive detection of xylene and toluene for monitoring indoor air pollution using Cr-doped NiO hierarchical nanostructures. Nanoscale 2013, 5, 7066–7073. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.-S.; Dai, Z.; Jeong, S.-Y.; Kwak, C.-H.; Kim, B.-Y.; Kim, D.-H.; Jang, H.W.; Park, J.-S.; Lee, J.-H. Monolayer Co3O4 inverse opals a multifunctional sensors for volatile organic compounds. Chem. Eur. J. 2016, 22, 7012–7017. [Google Scholar]
- Wu, Y.; Yan, H.; Yang, P. Semiconductor nanowire array: Potential substrates for photocatalysis and photovoltaics. Top. Catal. 2002, 19, 197–202. [Google Scholar] [CrossRef]
- Lee, J.Y.; Kim, D.S.; Park, J. Chemical conversion reaction between CdS nanobelts and ZnS nanobelts by vapor transport. Chem. Mater. 2007, 19, 4663–4669. [Google Scholar] [CrossRef]
- Kim, H.S.; Sung, T.K.; Jang, S.Y.; Myung, Y.; Cho, Y.J.; Lee, C.-W.; Park, J.; Ahn, J.-P.; Kim, J.-G.; Kim, Y.-J. Gas phase substitution synthesis of Cu1.8S and Cu2S superlattice nanowires from CdS nanowires. CrystEngComm 2011, 13, 2091–2095. [Google Scholar] [CrossRef]
- Lan, Z.H.; Liang, C.H.; Hsu, C.W.; Wu, C.T.; Lin, H.M.; Dhara, S.; Chen, K.H.; Chen, L.C.; Chen, C.C. Nanohomojunction (GaN) and nanoheterojunction (InN) nanorods on one-dimensional GaN nanowire substrates. Adv. Funct. Mater. 2004, 14, 233–237. [Google Scholar] [CrossRef]
- Wang, X.; Gao, P.; Li, J.; Summers, C.J.; Wang, Z.L. Rectangular porous ZnO-ZnS nanocables and ZnS nanotubes. Adv. Mater. 2002, 14, 1732–1735. [Google Scholar] [CrossRef]
- Zhang, B.; Jung, Y.; Chung, H.S.; Vugt, L.V.; Agarwal, R. Nanowire transformation by size-dependent cation exchange reactions. Nano Lett. 2010, 10, 149–155. [Google Scholar] [CrossRef] [PubMed]
- Woo, H.-S.; Kwak, C.-H.; Chung, J.-H.; Lee, J.-H. Highly selective and sensitive xylene sensors using Ni-doped branched ZnO nanowire networks. Sens. Actuators B Chem. 2015, 216, 358–366. [Google Scholar] [CrossRef]
- Zhang, Y.; Xu, J.; Xiang, Q.; Li, H.; Pan, Q.; Xu, P. Brush-like hierarchical ZnO nanostructures: Synthesis, photoluminescence and gas sensor properties. J. Phys. Chem. C 2009, 113, 3430–3435. [Google Scholar] [CrossRef]
- Thong, L.V.; Loan, L.T.N.; Hieu, N.V. Comparative study of gas sensor performance of SnO2 nanowires and their hierarchical nanostructures. Sens. Actuators B Chem. 2010, 150, 112–119. [Google Scholar]
- Khoang, N.D.; Trung, D.D.; Duy, N.D.; Hoa, N.D.; Hieu, N.V. Design of SnO2/ZnO hierarchical nanostructures for enhanced ethanol gas-sensing performance. Sens. Actuators B Chem. 2012, 174, 594–601. [Google Scholar] [CrossRef]
- Tharsika, T.; Haseeb, A.S.M.A.; Akbar, S.A.; Sabri, M.F.M.; Hoong, W.Y. Enhanced ethanol gas sensing properties of SnO2-core/ZnO-shell nanostructures. Sensors 2014, 14, 14586–14600. [Google Scholar] [CrossRef] [PubMed]
- Sen, S.; Kanitkar, P.; Sharma, A.; Muthe, K.P.; Rath, A.; Deshpande, S.K.; Kaur, M.; Aiyer, R.C.; Gupta, S.K.; Yakhmi, J.V. Growth of SnO2/W18O49 nanowire hierarchical heterostructure and their application as chemical sensor. Sens. Actuators B Chem. 2010, 147, 453–460. [Google Scholar] [CrossRef]
- Woo, H.-S.; Kwak, C.-H.; Chung, J.-H.; Lee, J.-H. Co-doped branched ZnO nanowires for ultraselective and sensitive detection of xylene. ACS Appl. Mater. Interfaces 2014, 6, 22553–22560. [Google Scholar] [CrossRef] [PubMed]
- Drobek, M.; Kim, J.-H.; Bechelany, M.; Vallicari, C.; Julbe, A.; Kim, S.S. MOF-based membrane encapsulated ZnO nanowires for enhanced gas sensor selectivity. ACS Appl. Mater. Interfaces 2016, 8, 8323–8328. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.-C.; Shen, G.; Zhou, C. Chemical sensors and electronic noses based on 1-D metal oxide nanostructures. IEEE Tans. Nanotechnol. 2008, 7, 668–682. [Google Scholar] [CrossRef]
- Park, W.J.; Kim, M.H.; Koo, B.H.; Choi, W.J.; Lee, J.-L.; Baik, J.M. Alternatively driven dual nanowire arrays by ZnO and CuO for selective sensing of gases. Sens. Actuators B Chem. 2013, 185, 10–16. [Google Scholar] [CrossRef]


| Catalyst | Structure | NW Material | Target Gas | References |
|---|---|---|---|---|
| Pt | Loading | SnO2 | NO2, Toluene, H2 | [54,55,56] |
| ZnO | C2H5OH | [57] | ||
| In2O3 | O2, H2 | [58,59] | ||
| Pd | Loading | SnO2, VO2 | H2 | [60,61,62] |
| Au | Loading | ZnO, In2O3 | CO, C2H5OH | [59,63,64,65] |
| ZnO | C2H5OH | |||
| Ag | Loading | SnO2 | C2H5OH | [66,67,68] |
| Catalyst | Structure | NW Material | Target Gas | References |
|---|---|---|---|---|
| CuO | Decoration | SnO2 | H2S | [78,79] |
| Cr2O3 | Decoration | ZnO, SnO2 | TMA | [80,81] |
| Co3O4 | Decoration | ZnO | NO2, C2H5OH | [82] |
| NiO | Decoration | ZnO | C2H5OH, HCHO | [83] |
| Mn3O4 | Decoration | ZnO | C2H5OH | [84] |
| La2O3 | Decoration | SnO2 | C2H5OH | [85] |
| Cu, CuO | Doping | SnO2 | H2S | [86,87] |
| Mo, Mo3O4 | Doping | ZnO | H2S | [88] |
| Mg, MgO | Doping | ZnO | C2H5OH | [89] |
| Sb | Doping | SnO2 | C2H5OH | [90] |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Woo, H.-S.; Na, C.W.; Lee, J.-H. Design of Highly Selective Gas Sensors via Physicochemical Modification of Oxide Nanowires: Overview. Sensors 2016, 16, 1531. https://doi.org/10.3390/s16091531
Woo H-S, Na CW, Lee J-H. Design of Highly Selective Gas Sensors via Physicochemical Modification of Oxide Nanowires: Overview. Sensors. 2016; 16(9):1531. https://doi.org/10.3390/s16091531
Chicago/Turabian StyleWoo, Hyung-Sik, Chan Woong Na, and Jong-Heun Lee. 2016. "Design of Highly Selective Gas Sensors via Physicochemical Modification of Oxide Nanowires: Overview" Sensors 16, no. 9: 1531. https://doi.org/10.3390/s16091531
APA StyleWoo, H.-S., Na, C. W., & Lee, J.-H. (2016). Design of Highly Selective Gas Sensors via Physicochemical Modification of Oxide Nanowires: Overview. Sensors, 16(9), 1531. https://doi.org/10.3390/s16091531





