Combined Dielectrophoresis and Impedance Systems for Bacteria Analysis in Microfluidic On-Chip Platforms
Abstract
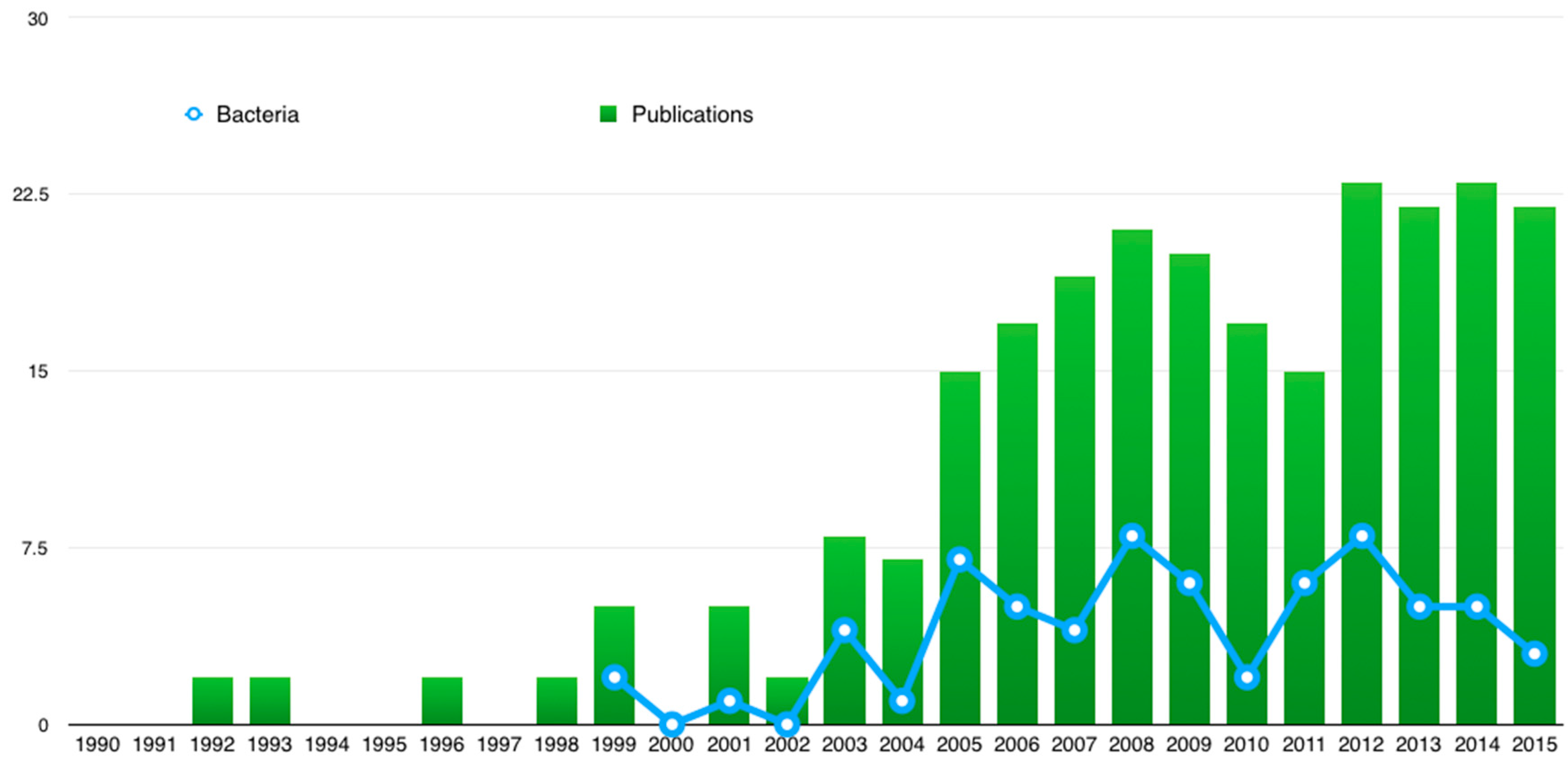
:1. Introduction
2. Theoretical Background
2.1. Dielectrophoresis (DEP)
2.2. Impedance (IA)
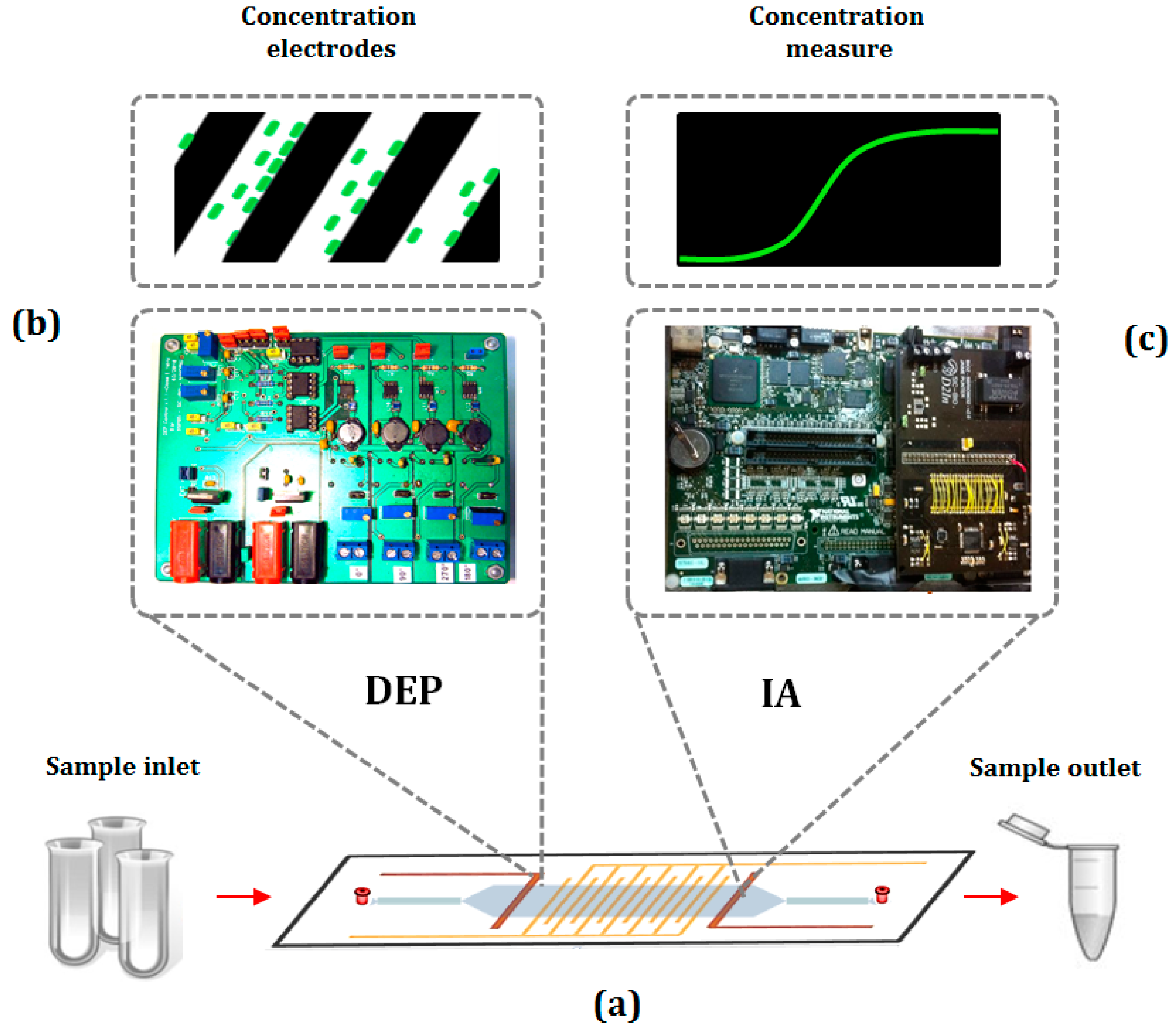
2.3. The Combined Approach for Bacteria Concentration and Detection
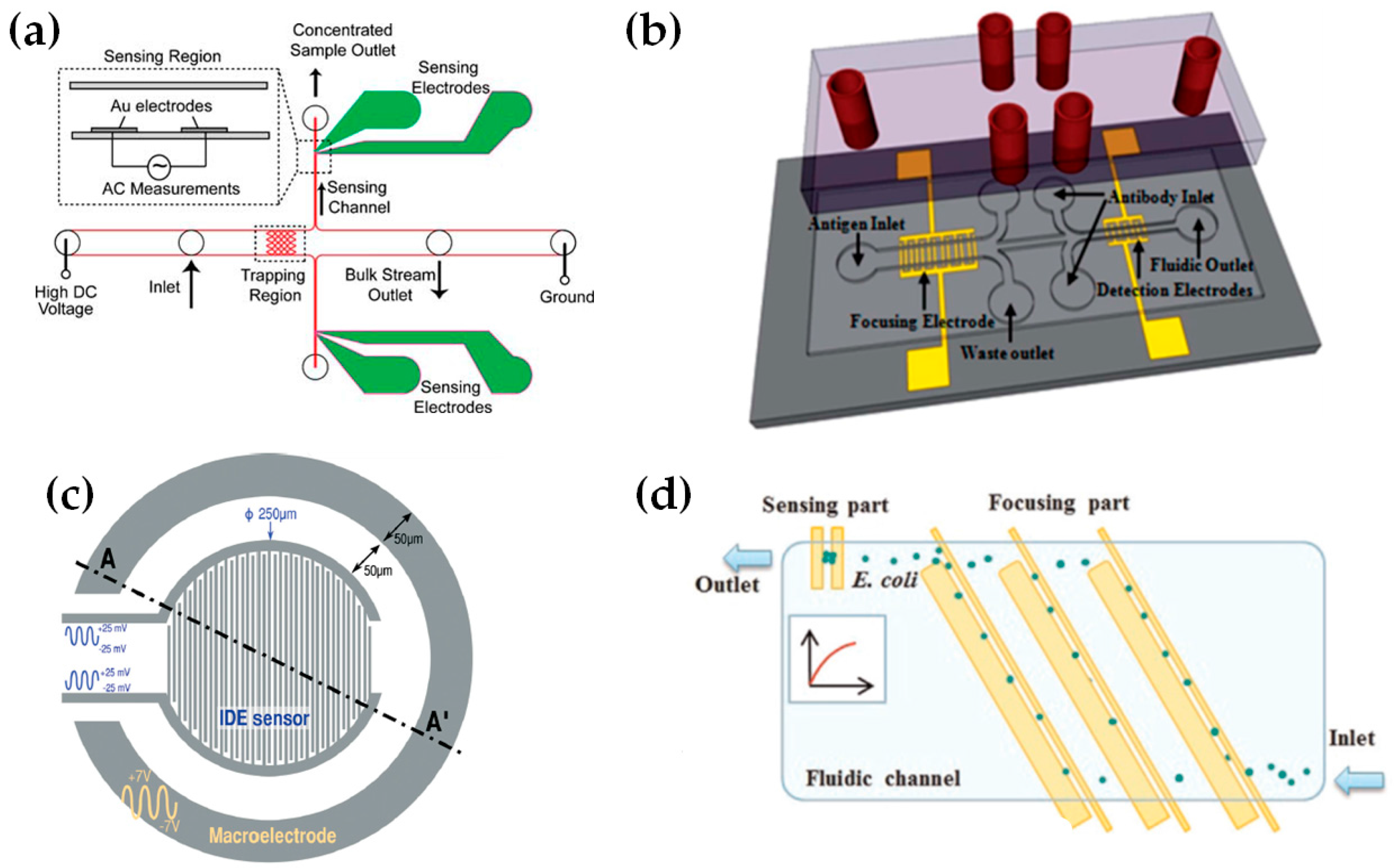
2.4. Recent Approaches
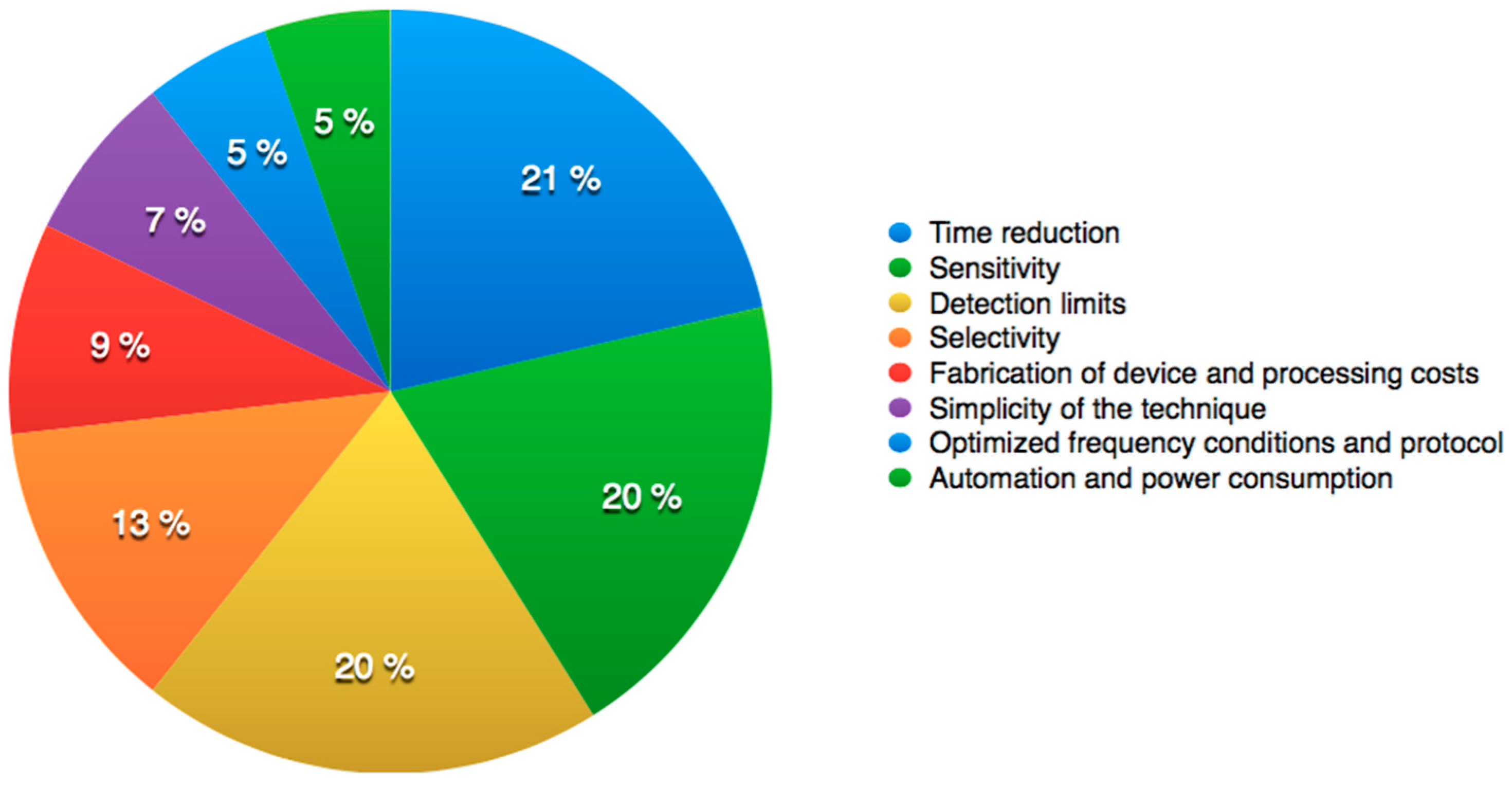
3. Operational Improvements of Combined DEP and IA Targeting Bacteria
3.1. Selectivity and Sensitivity
3.2. Fouling
3.3. Buffer Conductivity Variations
4. Future Perspectives of DEP and IA On-Chip Platforms
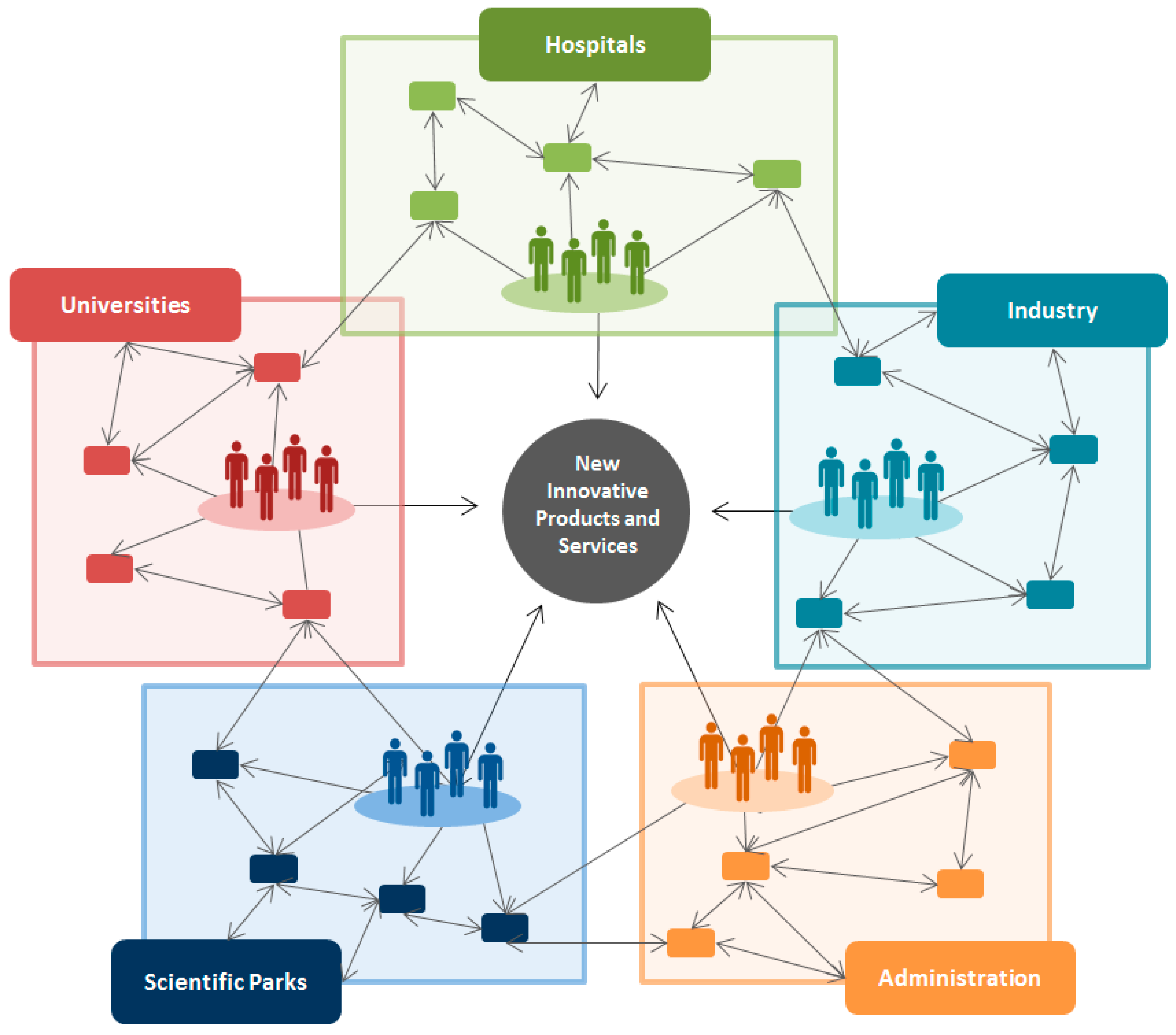
5. Technology Transfer and Social Return Challenges in Microelectronics
6. Concluding Comments
Acknowledgments
Conflicts of Interest
Appendix
| Method | Type | Principle | Advantage | Limitation | Ref. |
|---|---|---|---|---|---|
| Capillary Electrophoresis (CE) | Electro-dynamic | Separation method based in sublimities capillaries and micro/nano fluidic changes | Technique that brings speed, quantifiability, reproducibility and automation | Long separation times, poor specificity, sensitivity of the analyte to the surrounding analytical environment, requirements for sample purity, and microbe aggregation. high salt buffers | [13] |
| Mass Spectrometry (MS) | Chemical Method | Identification of cells by breaking them into ionized molecular fragments and measuring mass/charge ratio of the products | Fast technique with high sensitivity, quantitative and qualitative analysis, differentiates isotopes | Lack of sample purity, chemical differences in cell species, variations between stages of cell development | [12] |
| Centrifugation | Physical Method | Separation technique based on the centrifugal force that separate particles in solution according to their size, shape, density, and viscosity | Rapid, inexpensive, simple, non-specific; amenable to large sample sizes | Bacteria adhere to and sediment with matrix components | [6] |
| Filtration | Physical Method | Mechanic force used to separate solids from fluids, liquids or gases by interposing a medium through which only the fluid can pass | Rapid, inexpensive, simple, non-specific; amenable to large sample sizes | Limited to low particulate foods that will not clog the filter and by the volume of sample that can be passed through the filter (i.e., sample filterability). Sample pre-treatment with enzymes and detergents can increase sample filterability but may adversely affect cell viability | [6] |
| Immunoseparation | Biological Method | Separation technique based the use of immunoglobulins (antibodies) reactive with the particles to be separated | rapid, simple, standards methods available | high-non-specific binding | [6] |
| Raman microprobe spectroscopy (RMS) | Microscopy | Spectroscopic fingerprint from the microbial sample. Provides quantitative and qualitative information that can be used to characterize, discriminate and identify micro-organisms at the single-cell level | High sensitivity and unique molecular specificity | The signal in direct aqueous solution detection is often weak because of the small polarizability of most biological molecules compared with dye probe molecules | [15,16] |
| ELISA | Immunologic | Use of antibodies to which enzymes have been covalently bound. The antigen is rapped so that it may be the target micro-organism or target toxin | Useful for detection of infectious and toxigenic bacteria (ex. C. perfringens a toxin in the intestinal contents of animals). Able to differentiate the e and b toxins | Is time-consuming, not very sensitive, and involves laborious multiple steps | [162] |
| Polymerase Chain Reaction (PCR) | Nucleic acid probe-based method | Is an in vitro technique, which allows the amplification of a specific DNA region that lies between two regions of a known DNA sequence | Rapidly detects a wide range of micro-organisms in foods, the environment and in biological material. Cheaper and robust technique | A major disadvantage is that the amount of DNA sequence known for a given organism may be limited | [18] |
| Ligase chain reaction (LCR) | Nucleic acid probe-based method | An in vitro nucleic acid amplification technique that exponentially amplifies targeted DNA sequences | Possesses unique advantages for sensitive and specific miRNA detection. LCR exhibits better specificity than primer extension-based amplification, such as PCR, RCA, LAMP | Limited by gel electrophoresis separation or heterogeneous analysis process, which brought about multiplex steps, high cost, and long analysis time | [17] |
| Microarrays | Nucleic acid method | Analysis of large numbers of genes at a high resolution by the hybridization of labelled DNA to a substrate containing thousands of surface-immobilised DNA’s or oligonucleotides | Micro-arrays allow thousands of specific DNA or RNA sequences to be detected simultaneously on a small glass or silica slide only 1–2 cm2 in size | Micro-array instruments are expensive, of limited availability and require much skill in extracting useful information from the plethora of available data. However, this is an exciting area that appears headed for a very bright future | [18] |
References
- Kirk, M.D.; Pires, S.M.; Black, R.E.; Caipo, M.; Crump, J.A.; Devleesschauwer, B.; Döpfer, D.; Fazil, A.; Fischer-Walker, C.L.; Hald, T.; et al. World Health Organization Estimates of the Global and Regional Disease Burden of 22 Foodborne Bacterial, Protozoal, and Viral Diseases, 2010: A Data Synthesis. PLOS Med. 2015, 12, e1001921. [Google Scholar] [CrossRef] [PubMed]
- Prieto, M.; Colin, P.; Fernández-Escámez, P.; Alvarez-Ordóñez, A. Epidemiology, Detection, and Control of Foodborne Microbial Pathogens. Biomed Res. Int. 2015. [Google Scholar] [CrossRef] [PubMed]
- Salter, S.J. The food-borne identity. Nat. Rev. Microbiol. 2014, 12, 533. [Google Scholar] [CrossRef] [PubMed]
- Cabral, J.P.S. Water Microbiology. Bacterial Pathogens and Water. Int. J. Environ. Res. Public Health 2010, 7, 3657–3703. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Ye, Z.; Ying, Y. New trends in impedimetric biosensors for the detection of foodborne pathogenic bacteria. Sensors 2012, 12, 3449–3471. [Google Scholar] [CrossRef] [PubMed]
- Stevens, K.A.; Jaykus, L.-A. Bacterial separation and concentration from complex sample matrices: A review. Crit. Rev. Microbiol. 2004, 30, 7–24. [Google Scholar] [CrossRef] [PubMed]
- Lapizco-Encinas, B.H.; Davalos, R.V.; Simmons, B.A.; Cummings, E.B.; Fintschenko, Y. An insulator-based (electrodeless) dielectrophoretic concentrator for microbes in water. J. Microbiol. Methods 2005, 62, 317–326. [Google Scholar] [CrossRef] [PubMed]
- Yager, P.; Edwards, T.; Fu, E.; Helton, K.; Nelson, K.; Tam, M.R.; Weigl, B.H. Microfluidic diagnostic technologies for global public health. Nature 2006, 442, 412–418. [Google Scholar] [CrossRef] [PubMed]
- Urdea, M.; Penny, L.A.; Olmsted, S.S.; Giovanni, M.Y.; Kaspar, P.; Shepherd, A.; Wilson, P.; Dahl, C.A.; Buchsbaum, S.; Moeller, G.; et al. Requirements for high impact diagnostics in the developing world. Nature 2006, 444, 73–79. [Google Scholar] [CrossRef] [PubMed]
- Lapizco-Encinas, B.H.; Simmons, B.A.; Cummings, E.B.; Fintschenko, Y. Dielectrophoretic concentration and separation of live and dead bacteria in an array of insulators. Anal. Chem. 2004, 76, 1571–1579. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Yan, X.; Feng, X.; Wang, J.; Du, W.; Wang, Y.; Chen, P.; Xiong, L.; Liu, B.-F. Agarose-Based Microfluidic Device for Point-of-Care Concentration and Detection of Pathogen. Anal. Chem. 2014, 86, 10653–10659. [Google Scholar] [CrossRef] [PubMed]
- Jones, P.; DeMichele, A.; Kemp, L.; Hayes, M. Differentiation of Escherichia coli serotypes using DC gradient insulator dielectrophoresis. Anal. Bioanal. Chem. 2014, 406, 183–192. [Google Scholar] [CrossRef] [PubMed]
- Petr, J.; Maier, V. Analysis of microorganisms by capillary electrophoresis. TrAC Trends Anal. Chem. 2012, 31, 9–22. [Google Scholar] [CrossRef]
- Dastider, S.G.; Barizuddin, S.; Dweik, M.; Almasri, M. A micromachined impedance biosensor for accurate and rapid detection of E. coli O157:H7. RSC Adv. 2013, 3, 26297–26306. [Google Scholar] [CrossRef]
- Yang, X.; Gu, C.; Qian, F.; Li, Y.; Zhang, J.Z. Highly sensitive detection of proteins and bacteria in aqueous solution using surface-enhanced Raman scattering and optical fibers. Anal. Chem. 2011, 83, 5888–5894. [Google Scholar] [CrossRef] [PubMed]
- Marotta, N.E.; Beavers, K.R.; Bottomley, L.A. Limitations of surface enhanced Raman scattering in sensing DNA hybridization demonstrated by label-free DNA oligos as molecular rulers of distance-dependent enhancement. Anal. Chem. 2013, 85, 1440–1446. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Z.; Zhou, Y.; Gao, S.; Cheng, Y.; Li, Z. Homogeneous and sensitive detection of microRNA with ligase chain reaction and lambda exonuclease-assisted cationic conjugated polymer biosensing. ACS Appl. Mater. Interfaces 2014, 6, 6181–6185. [Google Scholar] [CrossRef] [PubMed]
- Deisingh, A.K.; Thompson, M. Detection of infectious and toxigenic bacteria. Analyst 2002, 127, 567–581. [Google Scholar] [CrossRef] [PubMed]
- Whitesides, G. The origins and the future of microfluidics. Nature 2006, 442, 368–373. [Google Scholar] [CrossRef] [PubMed]
- Zordan, M.D.; Grafton, M.M.G.; Acharya, G.; Reece, L.M.; Cooper, C.L.; Aronson, A.I.; Park, K.; Leary, J.F. Detection of pathogenic E. coli O157:H7 by a hybrid microfluidic SPR and molecular imaging cytometry device. Cytometry. A 2009, 75, 155–162. [Google Scholar] [CrossRef] [PubMed]
- Hamada, R.; Takayama, H. Improvement of dielectrophoretic impedance measurement method by bacterial concentration utilizing negative dielectrophoresis. J. PhysicsConference Ser. 2011, 307, 1–6. [Google Scholar] [CrossRef]
- Chin, C.D.; Linder, V.; Sia, S.K. Commercialization of microfluidic point-of-care diagnostic devices. Lab Chip 2012, 12, 2118–2134. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Jiang, H.; Zhang, L.; Zhang, C.; Wang, Z.; Chen, X. An Energy-Efficient ASIC for Wireless Body Sensor Networks in Medical Applications. IEEE Trans. Biomed. Circuits Syst. 2010, 4, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Figeys, D.; Pinto, D. Lab-on-a-chip: A revolution in biological and medical sciences. Anal. Chem. 2000, 72, 330A–335A. [Google Scholar] [CrossRef] [PubMed]
- Heo, J.; Hua, S.Z. An overview of recent strategies in pathogen sensing. Sensors 2009, 9, 4483–4502. [Google Scholar] [CrossRef] [PubMed]
- Ríos, A.; Zougagh, M.; Avila, M. Miniaturization through lab-on-a-chip: Utopia or reality for routine laboratories? A review. Anal. Chim. Acta 2012, 740, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Mackenzie, A.; Orrbine, E.; Hyde, L.; Benoit, M.; Chan, F.; Park, C.; Alverson, J.; Lembke, A.; Hoban, D.; Kennedy, W. Performance of the ImmunoCard STAT! E. coli O157:H7 test for detection of Escherichia coli O157:H7 in stools. J. Clin. Microbiol. 2000, 38, 1866–1868. [Google Scholar] [PubMed]
- Acheson, D.; McEntire, J.; Thorpe, C.M. Foodborne Illness: Latest Threats and Emerging Issues, an Issue of Infectious Disease Clinics; Elsevier Health Sciences: Philadelphia, PA, USA, 2013. [Google Scholar]
- Cole, K. Permeability and impermeability of cell membranes for ions. Cold Spring Harb. Symp. Quant. Biol. 1940, 8, 110–122. [Google Scholar] [CrossRef]
- Kim, M.; Jung, T.; Kim, Y.; Lee, C.; Woo, K.; Seol, J.H.; Yang, S. A microfluidic device for label-free detection of Escherichia coli in drinking water using positive dielectrophoretic focusing, capturing, and impedance measurement. Biosens. Bioelectron. 2015, 74, 1011–1015. [Google Scholar] [CrossRef] [PubMed]
- Del Moral Zamora, B.; Álvarez Azpeitia, J.M.; Oliva Brañas, A.M.; Colomer-Farrarons, J.; Castellarnau, M.; Miribel-Català, P.L.; Homs-Corbera, A.; Juárez, A.; Samitier, J. Dielectrophoretic concentrator enhancement based on dielectric poles for continuously flowing samples. Electrophoresis 2015, 36, 1405–1413. [Google Scholar] [CrossRef] [PubMed]
- Pohl, H.A.; Hawk, I. Separation of living and dead cells by dielectrophoresis. Science 1966, 152, 647–649. [Google Scholar] [CrossRef] [PubMed]
- Pethig, R.; Markx, G.H. Applications of dielectrophoresis in biotechnology. Trends Biotechnol. 1997, 15, 426–432. [Google Scholar] [CrossRef]
- Lapizco-Encinas, B.H. Editorial: Dielectrophoresis 2015. Electrophoresis 2015, 36, 1385. [Google Scholar] [CrossRef] [PubMed]
- Hamada, R.; Takayama, H.; Shonishi, Y.; Mao, L.; Nakano, M.; Suehiro, J. A rapid bacteria detection technique utilizing impedance measurement combined with positive and negative dielectrophoresis. Sens. Actuators B Chem. 2013, 181, 439–445. [Google Scholar] [CrossRef]
- Del Moral-Zamora, B.; Punter-Villagrassa, J.; Oliva-Brañas, A.M.; Álvarez-Azpeitia, J.M.; Colomer-Farrarons, J.; Samitier, J.; Homs-Corbera, A.; Miribel-Català, P.L. Combined dielectrophoretic and impedance system for on-chip controlled bacteria concentration: Application to Escherichia coli. Electrophoresis 2015, 36, 1130–1141. [Google Scholar] [CrossRef] [PubMed]
- Couniot, N.; Francis, L.A.; Flandre, D. Resonant dielectrophoresis and electrohydrodynamics for high-sensitivity impedance detection of whole-cell bacteria. Lab Chip 2015, 15, 3183–3191. [Google Scholar] [CrossRef] [PubMed]
- Suehiro, J.; Hamada, R.; Noutomi, D.; Shutou, M.; Hara, M. Selective detection of viable bacteria using dielectrophoretic impedance measurement method. J. Electrostat. 2003, 57, 157–168. [Google Scholar] [CrossRef]
- Chen, L.; Zheng, X.-L.; Hu, N.; Yang, J.; Luo, H.-Y.; Jiang, F.; Liao, Y.-J. Research Progress on Microfluidic Chip of Cell Separation Based on Dielectrophoresis. Chinese J. Anal. Chem. 2015, 43, 300–309. [Google Scholar] [CrossRef]
- Dwivedi, H.P.; Jaykus, L.-A. Detection of pathogens in foods: The current state-of-the-art and future directions. Crit. Rev. Microbiol. 2011, 37, 40–63. [Google Scholar] [CrossRef] [PubMed]
- Demircan, Y.; Özgür, E.; Külah, H. Dielectrophoresis: Applications and future outlook in point of care. Electrophoresis 2013, 34, 1008–1027. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, I.; Monjushiro, H.; Watarai, H. Measurement of dielectrophoretic mobility of single micro-particles in a flow channel. Analyst 2005, 130, 1340–1342. [Google Scholar] [CrossRef] [PubMed]
- Crane, J.; Pohl, H. A study of living and dead yeast cells using dielectrophoresis. J. Electrochem. Soc. 1968, 115, 584–586. [Google Scholar] [CrossRef]
- Allsopp, D.W.E.; Milner, K.R.; Brown, A.P.; Betts, W.B. Impedance technique for measuring dielectrophoretic collection of microbiological particles. J. Phys. D Appl. Phys. 1999, 32, 1066–1074. [Google Scholar] [CrossRef]
- Medoro, G.; Manaresi, N. A lab-on-a-chip for cell detection and manipulation. Sens. J. 2003, 3, 317–325. [Google Scholar] [CrossRef]
- Sabounchi, P.; Morales, A.M.; Ponce, P.; Lee, L.P.; Simmons, B.A.; Davalos, R.V. Sample concentration and impedance detection on a microfluidic polymer chip. Biomed. Microdevices 2008, 10, 661–670. [Google Scholar] [CrossRef] [PubMed]
- Jones, T.B. Electromechanics of Particles; Cambridge University Press: New York, NY, USA, 2005. [Google Scholar]
- Van den Driesche, S.; Rao, V.; Puchberger-Enengl, D.; Witarski, W.; Vellekoop, M.J. Continuous cell from cell separation by traveling wave dielectrophoresis. Sens. Actuators B Chem. 2012, 170, 207–214. [Google Scholar] [CrossRef]
- Ino, K.; Ishida, A.; Inoue, K.Y.; Suzuki, M.; Koide, M.; Yasukawa, T.; Shiku, H.; Matsue, T. Electrorotation chip consisting of three-dimensional interdigitated array electrodes. Sens. Actuators B Chem. 2011, 153, 468–473. [Google Scholar] [CrossRef]
- Issadore, D.; Franke, T.; Brown, K.A.; Westervelt, R.M. A microfluidic microprocessor: Controlling biomimetic containers and cells using hybrid integrated circuit/microfluidic chips. Lab Chip 2010, 10, 2937–2943. [Google Scholar] [CrossRef] [PubMed]
- Cheng, I.-F.; Chung, C.-C.; Chang, H.-C. High-throughput electrokinetic bioparticle focusing based on a travelling-wave dielectrophoretic field. Microfluid. Nanofluid. 2010, 10, 649–660. [Google Scholar] [CrossRef]
- Miled, M.; Sawan, M. Dielectrophoresis-based integrated lab-on-chip for nano and micro-particles manipulation and capacitive detection. IEEE Trans. Biomed. Circuits Syst. 2012, 6, 120–132. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.Y.; Do, J.; Premasiri, W.R.; Ziegler, L.D.; Klapperich, C.M. Rapid point-of-care concentration of bacteria in a disposable microfluidic device using meniscus dragging effect. Lab Chip 2010, 10, 3265–3270. [Google Scholar] [CrossRef] [PubMed]
- Yoo, J.; Cha, M.; Lee, J. Bacterial concentration detection with dielectrophoresis and capacitive measurement. In Proceedings of the NSTI Nanotechnology Conference and Trade Show, Boston, MA, USA, 1–5 June 2008; pp. 611–614.
- Yang, L.; Bashir, R. Electrical/electrochemical impedance for rapid detection of foodborne pathogenic bacteria. Biotechnol. Adv. 2008, 26, 135–150. [Google Scholar] [CrossRef] [PubMed]
- Everwand, M.; Anselmetti, D.; Regtmeier, J. On-Chip Continuous Flow Interaction Studies of DNA and Protein Complexed DNA. In Proceedings of the 14th International Conference on Miniaturized Systems for Chemistry and Life Sciences, Groningen, The Netherlands, 3–7 October 2010; pp. 19–21.
- Xu, C.; Wang, Y.; Cao, M.; Lu, Z. Dielectrophoresis of human red cells in microchips. Electrophoresis 1999, 20, 1829–1831. [Google Scholar] [CrossRef]
- Borgatti, M.; Bianchi, N.; Mancini, I.; Feriotto, G.; Gambari, R. New trends in non-invasive prenatal diagnosis: Applications of dielectrophoresis-based Lab-on-a-chip platforms to the identification and manipulation of rare cells. Int. J. Mol. Med. 2008, 21, 3–12. [Google Scholar] [CrossRef] [PubMed]
- Mernier, G.; Duqi, E.; Renaud, P. Characterization of a novel impedance cytometer design and its integration with lateral focusing by dielectrophoresis. Lab Chip 2012, 12, 4344–4349. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Canter, R.C.; Keten, G.; Vedantam, P.; Tzeng, T.-R.J.; Xuan, X. Continuous-flow particle and cell separations in a serpentine microchannel via curvature-induced dielectrophoresis. Microfluid. Nanofluidics 2011, 11, 743–752. [Google Scholar] [CrossRef]
- Patel, P.M.; Bhat, A.; Markx, G.H. A comparative study of cell death using electrical capacitance measurements and dielectrophoresis. Enzym. Microb. Technol. 2008, 43, 523–530. [Google Scholar] [CrossRef]
- Valero, A.; Braschler, T.; Rauch, A.; Demierre, N.; Barral, Y.; Renaud, P. Tracking and synchronization of the yeast cell cycle using dielectrophoretic opacity. Lab Chip 2011, 11, 1754–1760. [Google Scholar] [CrossRef] [PubMed]
- Cummings, E.B.; Singh, A.K. Dielectrophoresis in microchips containing arrays of insulating posts: Theoretical and experimental results. Anal. Chem. 2003, 75, 4724–4731. [Google Scholar] [CrossRef] [PubMed]
- Gallo-Villanueva, R.C.; Rodríguez-López, C.E.; Díaz-de-la-Garza, R.I.; Reyes-Betanzo, C.; Lapizco-Encinas, B.H. DNA manipulation by means of insulator-based dielectrophoresis employing direct current electric fields. Electrophoresis 2009, 30, 4195–4205. [Google Scholar] [CrossRef] [PubMed]
- Gupta, V.; Jafferji, I.; Garza, M.; Melnikova, V.O.; Hasegawa, D.K.; Pethig, R.; Davis, D.W. ApoStreamTM, a new dielectrophoretic device for antibody independent isolation and recovery of viable cancer cells from blood. Biomicrofluidics 2012, 6, 024133. [Google Scholar] [CrossRef] [PubMed]
- Wei, C.; Wei, T.-Y.; Liang, C.-H.; Tai, F.-C. The separation of different conducting multi-walled carbon nanotubes by AC dielectrophoresis. Diam. Relat. Mater. 2009, 18, 332–336. [Google Scholar] [CrossRef]
- Morgan, H.; Hughes, M.P.; Green, N.G. Separation of submicron bioparticles by dielectrophoresis. Biophys. J. 1999, 77, 516–525. [Google Scholar] [CrossRef]
- Gascoyne, P.R.C.; Vykoukal, J. Particle separation by dielectrophoresis. Electrophoresis 2002, 23, 1973–1983. [Google Scholar] [CrossRef]
- Gascoyne, P.R.C.; Vykoukal, J.V. Dielectrophoresis-Based Sample Handling in General-Purpose Programmable Diagnostic Instruments. Proc. IEEE. Inst. Electr. Electron. Eng. 2004, 92, 22–42. [Google Scholar] [CrossRef] [PubMed]
- Lui, C.; Cady, N.C.; Batt, C.A. Nucleic Acid-Based Detection of Bacterial Pathogens Using Integrated Microfluidic Platform Systems. Sensors 2009, 9, 3713–3144. [Google Scholar] [CrossRef] [PubMed]
- Zou, Z.; Lee, S.; Ahn, C.H. A Polymer Microfluidic Chip with Interdigitated Electrodes Arrays for Simultaneous Dielectrophoretic Manipulation and Impedimetric Detection of Microparticles. IEEE Sens. J. 2008, 8, 527–535. [Google Scholar] [CrossRef]
- Zou, Z.; Kai, J.; Rust, M.J.; Han, J.; Ahn, C.H. Functionalized nano interdigitated electrodes arrays on polymer with integrated microfluidics for direct bio-affinity sensing using impedimetric measurement. Sens. Actuators A Phys. 2007, 136, 518–526. [Google Scholar] [CrossRef]
- Gawad, S.; Schild, L.; Renaud, P.H. Micromachined impedance spectroscopy flow cytometer for cell analysis and particle sizing. Lab Chip 2001, 1, 76–82. [Google Scholar] [CrossRef] [PubMed]
- Gozzini, F.; Ferrari, G.; Sampietro, M. An instrument-on-chip for impedance measurements on nanobiosensors with attoFarad resoution. In Proceedings of the 2009 IEEE International Solid-State Circuits Conference, San Francisco, CA, USA, 8–12 February 2009; IEEE: San Francisco, CA, USA, 2009; pp. 346–347. [Google Scholar]
- Giaever, I.; Keese, C.R. A morphological biosensor for mammalian cells. Nature 1993, 366, 591–592. [Google Scholar] [CrossRef] [PubMed]
- Yang, M.; Lim, C.C.; Liao, R.; Zhang, X. A novel microfluidic impedance assay for monitoring endothelin-induced cardiomyocyte hypertrophy. Biosens. Bioelectron. 2007, 22, 1688–1693. [Google Scholar] [CrossRef] [PubMed]
- Varshney, M.; Li, Y. Interdigitated array microelectrodes based impedance biosensors for detection of bacterial cells. Biosens. Bioelectron. 2009, 24, 2951–2960. [Google Scholar] [CrossRef] [PubMed]
- Du, E.; Ha, S.; Diez-Silva, M.; Dao, M.; Suresh, S.; Chandrakasan, A.P. Electric impedance microflow cytometry for characterization of cell disease states. Lab Chip 2013, 13, 3903–3909. [Google Scholar] [CrossRef] [PubMed]
- Foudeh, A.M.; Fatanat Didar, T.; Veres, T.; Tabrizian, M. Microfluidic designs and techniques using lab-on-a-chip devices for pathogen detection for point-of-care diagnostics. Lab Chip 2012, 12, 3249–3266. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, M.C.; Kawde, A.-N.; Wang, J. Aptamer biosensor for label-free impedance spectroscopy detection of proteins based on recognition-induced switching of the surface charge. Chem. Commun. 2005, 4267–4269. [Google Scholar] [CrossRef] [PubMed]
- Tiruppathi, C.; Malik, A.B.; Del Vecchio, P.J.; Keese, C.R.; Giaever, I. Electrical method for detection of endothelial cell shape change in real time: assessment of endothelial barrier function. Proc. Natl. Acad. Sci. USA 1992, 89, 7919–7923. [Google Scholar] [CrossRef] [PubMed]
- Ramaswamy, B.; Yeh, Y.-T.T.; Zheng, S.-Y. Microfluidic device and system for point-of-care blood coagulation measurement based on electrical impedance sensing. Sens. Actuators B Chem. 2013, 180, 21–27. [Google Scholar] [CrossRef]
- Sun, T.; Morgan, H. Single-cell microfluidic impedance cytometry: A review. Microfluid. Nanofluidics 2010, 8, 423–443. [Google Scholar] [CrossRef]
- Xu, M.; Luo, X.; Davis, J.J. The label free picomolar detection of insulin in blood serum. Biosens. Bioelectron. 2013, 39, 21–25. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Bell, M.C.; Suni, I.I. Impedance biosensor for peanut protein Ara h 1. Anal. Chem. 2008, 80, 9157–9161. [Google Scholar] [CrossRef] [PubMed]
- Ramírez, N.; Regueiro, A.; Arias, O.; Contreras, R. Espectroscopía de impedancia electroquímica, herramienta eficaz para el diagnóstico rápido microbiológico. Biotecnol. Apl. 2009, 26, 72–78. [Google Scholar]
- Stewart, G.N. The charges produced by the growth of bacteria in the molecular concentration and electrical condustivity of culture media. J. Exp. Med. 1899, 4, 235–243. [Google Scholar] [CrossRef] [PubMed]
- Grossi, M.; Lanzoni, M.; Pompei, A.; Lazzarini, R.; Matteuzzi, D.; Riccò, B. An embedded portable biosensor system for bacterial concentration detection. Biosens. Bioelectron. 2010, 26, 983–990. [Google Scholar] [CrossRef] [PubMed]
- Yang, L. Electrical impedance spectroscopy for detection of bacterial cells in suspensions using interdigitated microelectrodes. Talanta 2008, 74, 1621–1629. [Google Scholar] [CrossRef] [PubMed]
- Suehiro, J.; Noutomi, D.; Hamada, R.; Hara, M. Selective detection of specific bacteria using dielectrophoretic impedance measurement method combined with an antigen–antibody reaction. J. Electrostat. 2003, 58, 229–246. [Google Scholar] [CrossRef]
- Easter, M.C.; Gibson, D.M. Rapid and automated detection of salmonella by electrical measurements. J. Hyg. 1985, 94, 245–262. [Google Scholar] [CrossRef] [PubMed]
- Smith, P.J.; Bolton, F.J.; Gayner, V.E.; Eccles, A. Improvements to a lysine medium for detection of salmonellas by electrical conductance. Lett. Appl. Microbiol. 2008, 11, 84–86. [Google Scholar] [CrossRef]
- Blivet, D.; Salvat, G.; Humbert, F.; Colin, P. Development of a new culture medium for the rapid detection of Salmonella by indirect conductance measurements. J. Appl. Microbiol. 1998, 84, 399–403. [Google Scholar] [CrossRef] [PubMed]
- Varshney, M.; Li, Y. Interdigitated array microelectrode based impedance biosensor coupled with magnetic nanoparticle-antibody conjugates for detection of Escherichia coli O157:H7 in food samples. Biosens. Bioelectron. 2007, 22, 2408–2414. [Google Scholar] [CrossRef] [PubMed]
- Timms, S.; Colquhoun, K.O.; Fricker, C.R. Detection of Escherichia coli in potable water using indirect impedance technology. J. Microbiol. Methods 1996, 26, 125–132. [Google Scholar] [CrossRef]
- Gomez-Sjoberg, R.; Morisette, D.T.; Bashir, R. Impedance microbiology-on-a-chip: Microfluidic bioprocessor for rapid detection of bacterial metabolism. J. Microelectromechan. Syst. 2005, 14, 829–838. [Google Scholar] [CrossRef]
- Glassmoyer, K.E.; Russell, S.M. Evaluation of a selective broth for detection of Staphylococcus aureus using impedance microbiology. J. Food Prot. 2001, 64, 44–50. [Google Scholar] [PubMed]
- Spiller, E.; Schöll, A.; Alexy, R.; Kümmerer, K.; Urban, G.A. A microsystem for growth inhibition test of Enterococcus faecalis based on impedance measurement. Sens. Actuators B Chem. 2006, 118, 182–191. [Google Scholar] [CrossRef]
- Richards, J.; Jason, A.; Hobbs, G.; Gibson, D.; Christie, R. Electronic measurement of bacterial growth. J. Phys. E 1978, 11, 560–568. [Google Scholar] [CrossRef] [PubMed]
- Nakano, M.; Ding, Z.; Kasahara, H.; Suehiro, J. Rapid microbead-based DNA detection using dielectrophoresis and impedance measurement. EPL 2014, 108, 28003. [Google Scholar] [CrossRef]
- Henning, A.; Bier, F.F.; Hölzel, R. Dielectrophoresis of DNA: Quantification by impedance measurements. Biomicrofluidics 2010, 4, 022803. [Google Scholar] [CrossRef] [PubMed]
- Krishnan, R.; Sullivan, B.D.; Mifflin, R.L.; Esener, S.C.; Heller, M.J. Alternating current electrokinetic separation and detection of DNA nanoparticles in high-conductance solutions. Electrophoresis 2008, 29, 1765–1774. [Google Scholar] [CrossRef] [PubMed]
- Suehiro, J.; Shutou, M.; Hatano, T.; Hara, M. High sensitive detection of biological cells using dielectrophoretic impedance measurement method combined with electropermeabilization. Sens. Actuators B Chem. 2003, 96, 144–151. [Google Scholar] [CrossRef]
- Nakano, M.; Ding, Z.; Suehiro, J. Dielectrophoresis and dielectrophoretic impedance detection of adenovirus and rotavirus. Jpn. J. Appl. Phys. 2016, 55, 017001. [Google Scholar] [CrossRef]
- Mohanty, S.K.; Ravula, S.K.; Engisch, K.L.; Frazier, A.B. A micro system using dielectrophoresis and electrical impedance spectroscopy for cell manipulation and analysis. In Proceedings of the 12th International Conference on TRANSDUCERS, Solid-State Sensors, Actuators and Microsystems, Boston, MA, USA, 8–12 June 2003; Volume 2, pp. 1055–1058.
- Ameri, S.K.; Singh, P.K.; Dokmeci, M.R.; Khademhosseini, A.; Xu, Q.; Sonkusale, S.R. All electronic approach for high-throughput cell trapping and lysis with electrical impedance monitoring. Biosens. Bioelectron. 2014, 54, 462–467. [Google Scholar] [CrossRef] [PubMed]
- Park, H.; Kim, D.; Yun, K.-S. Single-cell manipulation on microfluidic chip by dielectrophoretic actuation and impedance detection. Sens. Actuators B Chem. 2010, 150, 167–173. [Google Scholar] [CrossRef]
- Lan, K.-C.; Jang, L.-S. Integration of single-cell trapping and impedance measurement utilizing microwell electrodes. Biosens. Bioelectron. 2011, 26, 2025–2031. [Google Scholar] [CrossRef] [PubMed]
- Lennon, E.; Ostrovidov, S.; Senez, V.; Fujii, T. Dielectrophoresis, cell culture, and Electrical Impedance Spectroscopy Applied to Adherent Cells in a Single Biochip. In Proceedings of the 2006 International Conference on Microtechnologies in Medicine and Biology, Okinawa, Japan, 9–12 May 2006; pp. 165–168.
- Chuang, C.-H.; Huang, Y.-W.; Wu, Y.-T. System-level biochip for impedance sensing and programmable manipulation of bladder cancer cells. Sensors 2011, 11, 11021–11035. [Google Scholar] [CrossRef] [PubMed]
- Hirota, K.; Inagaki, S.; Hamada, R.; Ishihara, K.; Miyake, Y. Evaluation of a Rapid Oral Bacteria Quantification System Using Dielectrophoresis and the Impedance Measurement. Biocontrol Sci. 2014, 19, 45–49. [Google Scholar] [CrossRef] [PubMed]
- Ishii, Y.; Imamura, K.; Kikuchi, Y.; Miyagawa, S.; Hamada, R.; Sekino, J.; Sugito, H.; Ishihara, K.; Saito, A. Point-of-care detection of Tannerella forsythia using an antigen-antibody assisted dielectrophoretic impedance measurement method. Microb. Pathog. 2015, 82, 37–42. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, S.; Kuratani, M.; Tanaka, M.; Ito, T.; Kanemaki, N.; Shirai, M.; Nomura, R.; Nakano, K.; Asai, F. Measurement of oral bacterial counts in dogs by dielectrophoretic impedance. Fundam. Toxicol. Sci. 2015, 2, 83–87. [Google Scholar] [CrossRef]
- Radke, S.M.; Alocilja, E.C. A high density microelectrode array biosensor for detection of E. coli O157:H7. Biosens. Bioelectron. 2005, 20, 1662–1667. [Google Scholar] [CrossRef] [PubMed]
- Brown, A.P.; Betts, W.B.; Harrison, A.B.; O’Neill, J.G. Evaluation of a dielectrophoretic bacterial counting technique. Biosens. Bioelectron. 1999, 14, 341–351. [Google Scholar] [CrossRef]
- Suehiro, J.; Hatano, T.; Shutou, M.; Hara, M. Improvement of electric pulse shape for electropermeabilization-assisted dielectrophoretic impedance measurement for high sensitive bacteria detection. Sens. Actuators B Chem. 2005, 109, 209–215. [Google Scholar] [CrossRef]
- Müller, T.; Gradl, G.; Howitz, S.; Shirley, S.; Schnelle, T.; Fuhr, G. A 3-D microelectrode system for handling and caging single cells and particles. Biosens. Bioelectron. 1999, 14, 247–256. [Google Scholar] [CrossRef]
- Fry, C.H.; Salvage, S.C.; Manazza, A.; Dupont, E.; Labeed, F.H.; Hughes, M.P.; Jabr, R.I. Cytoplasm resistivity of mammalian atrial myocardium determined by dielectrophoresis and impedance methods. Biophys. J. 2012, 103, 2287–2294. [Google Scholar] [CrossRef] [PubMed]
- Suehiro, J.; Yatsunami, R.; Hamada, R.; Hara, M. Quantitative estimation of biological cell concentration suspended in aqueous medium by using dielectrophoretic impedance measurement method. J. Phys. D. Appl. Phys. 1999, 32, 2814–2820. [Google Scholar] [CrossRef]
- Shafiee, H.; Caldwell, J.L.; Sano, M.B.; Davalos, R.V. Contactless dielectrophoresis: a new technique for cell manipulation. Biomed. Microdevices 2009, 11, 997–1006. [Google Scholar] [CrossRef] [PubMed]
- Sano, M.B.; Caldwell, J.L.; Davalos, R.V. Modeling and development of a low frequency contactless dielectrophoresis (cDEP) platform to sort cancer cells from dilute whole blood samples. Biosens. Bioelectron. 2011, 30, 13–20. [Google Scholar] [CrossRef] [PubMed]
- Suehiro, J.; Ohtsubo, A.; Hatano, T.; Hara, M. Selective detection of bacteria by a dielectrophoretic impedance measurement method using an antibody-immobilized electrode chip. Sens. Actuators B Chem. 2006, 119, 319–326. [Google Scholar] [CrossRef]
- Boehm, D.A.; Gottlieb, P.A.; Hua, S.Z. On-chip microfluidic biosensor for bacterial detection and identification. Sens. Actuators B Chem. 2007, 126, 508–514. [Google Scholar] [CrossRef]
- Dweik, M.; Stringer, R.C.; Dastider, S.G.; Wu, Y.; Almasri, M.; Barizuddin, S. Specific and targeted detection of viable Escherichia coli O157:H7 using a sensitive and reusable impedance biosensor with dose and time response studies. Talanta 2012, 94, 84–89. [Google Scholar] [CrossRef] [PubMed]
- Beck, J.D.; Shang, L.; Li, B.; Marcus, M.S.; Hamers, R.J. Discrimination between Bacillus species by impedance analysis of individual dielectrophoretically positioned spores. Anal. Chem. 2008, 80, 3757–3761. [Google Scholar] [CrossRef] [PubMed]
- Maas, S.L.N.; de Vrij, J.; van der Vlist, E.J.; Geragousian, B.; van Bloois, L.; Mastrobattista, E.; Schiffelers, R.M.; Wauben, M.H.M.; Broekman, M.L.D.; Nolte-’t Hoen, E.N.M. Possibilities and limitations of current technologies for quantification of biological extracellular vesicles and synthetic mimics. J. Control. Release 2015, 200, 87–96. [Google Scholar] [CrossRef] [PubMed]
- Filipe, V.; Poole, R.; Kutscher, M.; Forier, K.; Braeckmans, K.; Jiskoot, W. Fluorescence single particle tracking for the characterization of submicron protein aggregates in biological fluids and complex formulations. Pharm. Res. 2011, 28, 1112–1120. [Google Scholar] [CrossRef] [PubMed]
- Gwon, H.R.; Chang, S.T.; Choi, C.K.; Jung, J.-Y.; Kim, J.-M.; Lee, S.H. Development of a new contactless dielectrophoresis system for active particle manipulation using movable liquid electrodes. Electrophoresis 2014, 35, 2014–2021. [Google Scholar] [CrossRef] [PubMed]
- Demierre, N.; Braschler, T.; Muller, R.; Renaud, P. Focusing and continuous separation of cells in a microfluidic device using lateral dielectrophoresis. Sens. Actuators B Chem. 2008, 132, 388–396. [Google Scholar] [CrossRef]
- Demierre, N.; Braschler, T.; Linderholm, P.; Seger, U.; van Lintel, H.; Renaud, P. Characterization and optimization of liquid electrodes for lateral dielectrophoresis. Lab Chip 2007, 7, 355–365. [Google Scholar] [CrossRef] [PubMed]
- Hughes, M.P. Strategies for dielectrophoretic separation in laboratory-on-a-chip systems. Electrophoresis 2002, 23, 2569–2582. [Google Scholar] [CrossRef]
- Martinez-Duarte, R. Microfabrication technologies in dielectrophoresis applications—A review. Electrophoresis 2012, 33, 3110–3132. [Google Scholar] [CrossRef] [PubMed]
- Shaker, M.; Colella, L.; Caselli, F.; Bisegna, P.; Renaud, P. An impedance-based flow microcytometer for single cell morphology discrimination. Lab Chip 2014, 14, 2548–2555. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Li, S.; Cao, W.; Li, W.; Wen, W.; Alici, G. Improved concentration and separation of particles in a 3D dielectrophoretic chip integrating focusing, aligning and trapping. Microfluid. Nanofluid. 2012, 14, 527–539. [Google Scholar] [CrossRef]
- Volpatti, L.R.; Yetisen, A.K. Commercialization of microfluidic devices. Trends Biotechnol. 2014, 32, 347–350. [Google Scholar] [CrossRef] [PubMed]
- Regtmeier, J.; Eichhorn, R.; Viefhues, M.; Bogunovic, L.; Anselmetti, D. Electrodeless dielectrophoresis for bioanalysis: Theory, devices and applications. Electrophoresis 2011, 32, 2253–2273. [Google Scholar] [CrossRef] [PubMed]
- Kadri, N.A.; Fatoyinbo, H.O.; Hughes, M.P.; Labeed, F.H. Semi-automated Dielectrophoretic Cell Characterisation Module for Lab-on-Chip Applications. IFMBE Proc. 2011, 35, 582–586. [Google Scholar]
- Del Moral Zamora, B.; Alvarez Azpeitia, J.M.; Colomer-Farrarons, J.; Miribel-Catala, P.Ll.; Homs-Corbera, A.; Juarez, A.; Samitier, J. Towards Point-of-Use Dielectrophoretic Methods: A new Portable Multiphase Generator for Bacteria Concentration. IFMBE Proc. 2013, 41, 856–859. [Google Scholar]
- Mohammed, M.I.; Haswell, S.; Gibson, I. Lab-on-a-chip or Chip-in-a-lab: Challenges of Commercialization Lost in Translation. Proc. Technol. 2015, 20, 54–59. [Google Scholar] [CrossRef]
- Hogan, J. Lab on a chip: A little goes a long way. Nature 2006, 442, 351–352. [Google Scholar] [CrossRef] [PubMed]
- Islam, N.; Miyazaki, K. An empirical analysis of nanotechnology research domains. Technovation 2010, 30, 229–237. [Google Scholar] [CrossRef]
- Punter-Villagrasa, J.; Cid, J.; Páez-Avilés, C.; Rodríguez-Villarreal, I.; Juanola-Feliu, E.; Colomer-Farrarons, J.; Miribel-Català, P. An Instantaneous Low-Cost Point-of-Care Anemia Detection Device. Sensors 2015, 15, 4564–4577. [Google Scholar] [CrossRef] [PubMed]
- Butter, M.; Fischer, N.; Gjsberts, G.; Hartmann, C.; de Heide, M.; van der Zee, F. Horizon 2020: Key Enabling Technologies (KETs), Booster for European Leadership in the Manufacturing Sector; Study for the ITRE Committee: Brussels, Belgium, 2014. [Google Scholar]
- Páez-Avilés, C.; Juanola-Feliu, E.; Bogachan-Tahirbegi, I.; Mir, M.; Gonzales-Piñero, M.; Samitier, J. Innovation and Technology Transfer of Medical Devices Fostered by Cross-disciplinary Commmunities of Practitioners. Int. J. Innov. Manag. 2015, 19, 1–27. [Google Scholar] [CrossRef]
- European Commision A European Strategy for Key Enabling Technologies—A Bridge to Growth and Jobs. A Comunication from the Commission to the European Parliament, the Council, the European Economic and Social Committee and the Committee of the Regions. 2012. Available online: http://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=COM:2012:0341:FIN:EN:PDF (accessed on 14 September 2016).
- Kawabata, T.; Washizu, M. Dielectrophoretic detection of molecular bindings. IEEE Trans. Ind. Appl. 2001, 37, 1625–1633. [Google Scholar] [CrossRef]
- Gomez, F. The future of microfluidic point-of-care diagnostic devices. Bioanalysis 2013, 5, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Tsai, C.; Cheng, K.; Chen, S. Low-Invasive Implantable Devices of Low-Power Consumption Using High-Efficiency Antennas for Cloud Health Care. IEEE J. Emerg. Sel. Top. Circuits Syst. 2012, 2, 14–23. [Google Scholar] [CrossRef]
- Jani, I.V.; Peter, T.F. How Point-of-Care Testing Could Drive Innovation in Global Health. N. Engl. J. Med. 2013, 368, 2319–2324. [Google Scholar] [CrossRef] [PubMed]
- Altomare, L.; Borgatti, M.; Medoro, G.; Manaresi, N.; Tartagni, M.; Guerrieri, R.; Gambari, R. Levitation and movement of human tumor cells using a printed circuit board device based on software-controlled dielectrophoresis. Biotechnol. Bioeng. 2003, 82, 474–479. [Google Scholar] [CrossRef] [PubMed]
- Andersen, P.D.; Jørgensen, B.H.; Lading, L.; Rasmussen, B. Sensor foresight—Technology and market. Technovation 2004, 24, 311–320. [Google Scholar] [CrossRef]
- Abel, G. Current status and future prospects of point-of-care testing around the globe. Expert Rev. Mol. Diagn. 2015, 15, 853–855. [Google Scholar] [CrossRef] [PubMed]
- Fu, B.E.; Yager, P.; Floriano, P.N.; Christodoulides, N.; Mcdevitt, J.T. Perspective on Diagnostics for Global Health. IEEE Pulse 2011, 2, 40–50. [Google Scholar] [CrossRef] [PubMed]
- Neužil, P.; Campos, C.D.M.; Wong, C.C.; Soon, J.B.W.; Reboud, J.; Manz, A. From chip-in-a-lab to lab-on-a-chip: Towards a single handheld electronic system for multiple application-specific lab-on-a-chip (ASLOC). Lab Chip 2014, 14, 2168–2176. [Google Scholar] [CrossRef] [PubMed]
- Maine, E.; Lubik, S.; Garnsey, E. Value creation strategies for science-based business: A study of advanced materials ventures. Innov. Manag. Policy Pract. 2013, 15, 35–51. [Google Scholar] [CrossRef]
- Hu, Y. Hyperlinked actors in the global knowledge communities and diffusion of innovation tools in nascent industrial field. Technovation 2013, 33, 38–49. [Google Scholar] [CrossRef]
- Van Looy, B.; Ranga, M.; Callaert, J.; Debackere, K.; Zimmermann, E. Combining entrepreneurial and scientific performance in academia: Towards a compounded and reciprocal Matthew-effect? Res. Policy 2004, 33, 425–441. [Google Scholar] [CrossRef]
- Kalisz, D.; Aluchna, M. Research and Innovation redefined. Perspectives on the European Union initiatives on Horizon 2020. Eur. Integr. Stud. 2012, 6, 140–149. [Google Scholar]
- Dogramatzis, D. Healthcare Biotechnology: A Practical Guide; CRC Press: Boca Raton, FL, USA, 2010. [Google Scholar]
- Juanola-Feliu, E.; Colomer-Farrarons, J.; Miribel-Català, P.; Samitier, J.; Valls-Pasola, J. Market challenges facing academic research in commercializing nano-enabled implantable devices for in-vivo biomedical analysis. Technovation 2012, 32, 193–204. [Google Scholar] [CrossRef]
- Leydesdorff, L. The Triple Helix, Quadruple Helix, …, and an N-Tuple of Helices: Explanatory Models for Analyzing the Knowledge-Based Economy? J. Knowl. Econ. 2011, 3, 25–35. [Google Scholar] [CrossRef]
- Cho, I.-H.; Irudayaraj, J. In-situ immuno-gold nanoparticle network ELISA biosensors for pathogen detection. Int. J. Food Microbiol. 2013, 164, 70–75. [Google Scholar] [CrossRef] [PubMed]
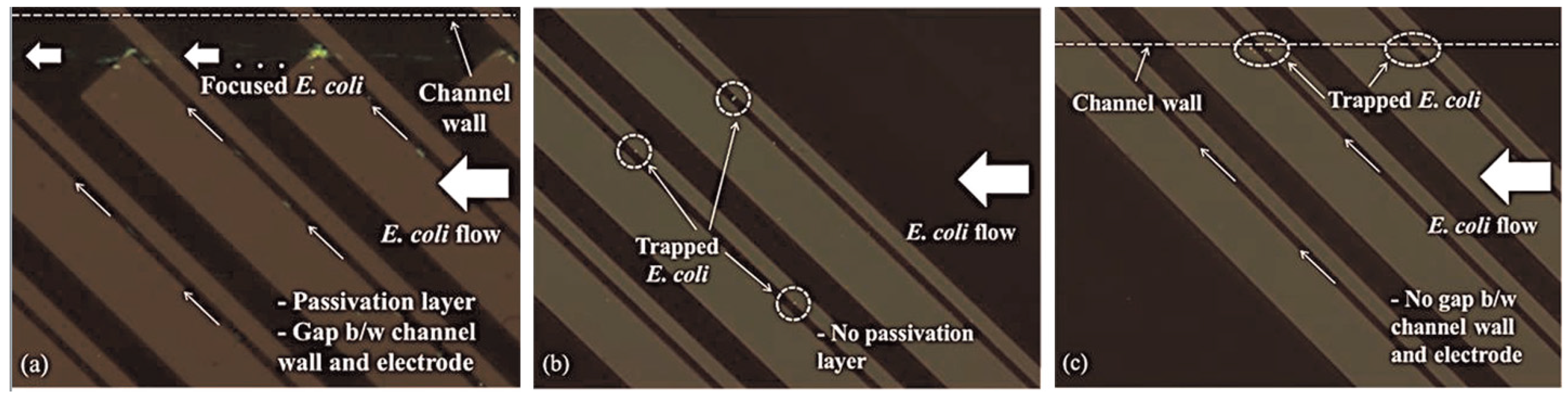
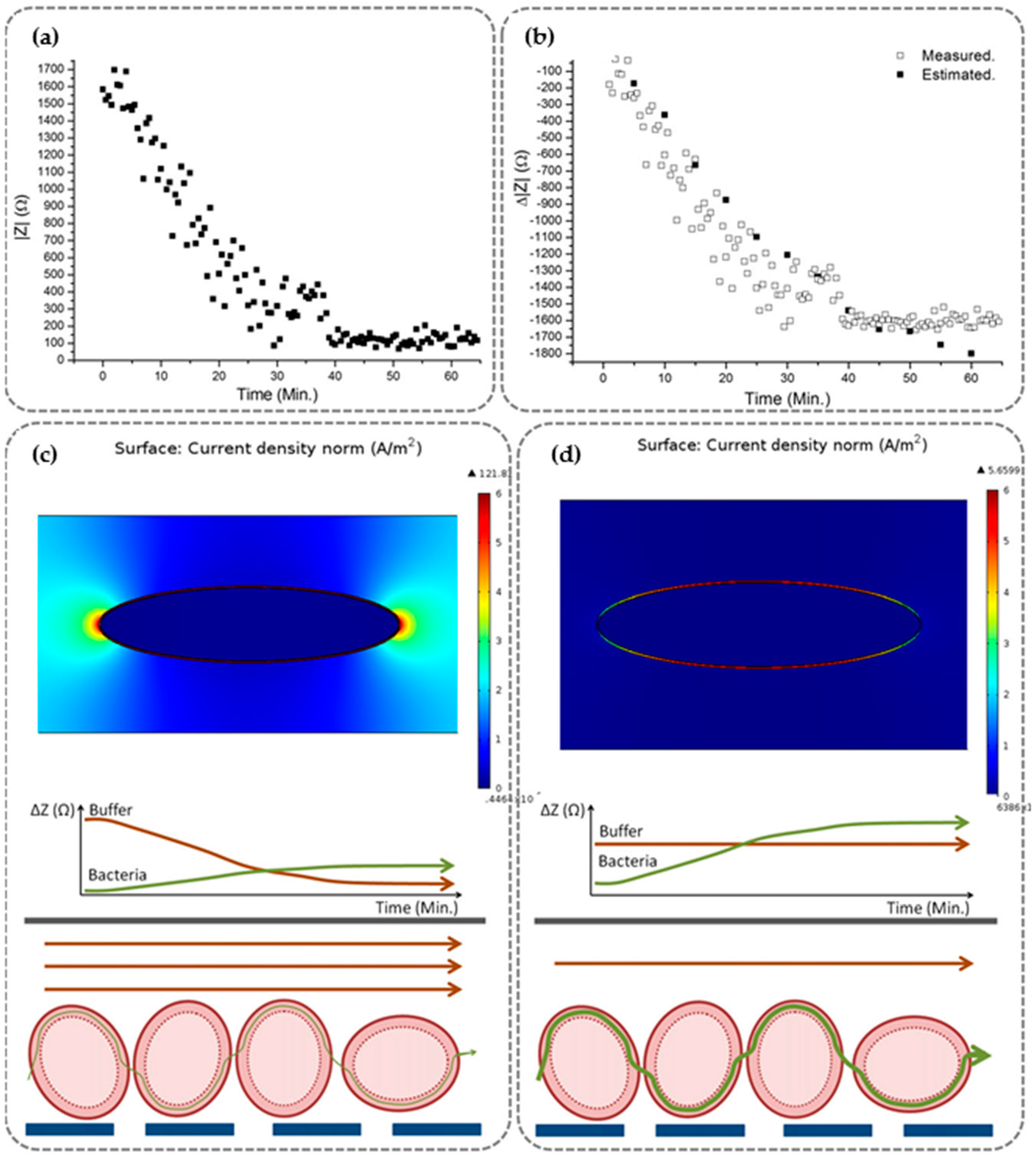
| Principle | Buffer | Conductivity | Bio-Affinity Element | Applied Frequency | Flow Rate Conditions | Bacteria | Sample Rate | Concentration | Signal Variation | Reference |
|---|---|---|---|---|---|---|---|---|---|---|
| DEP + IA | Manitol solution | 0.2 mS/m | polyclonal antibodies | 1 MHz | 9 × 102 μL/min | E. coli strain K12 | NA | 107 cells/mL | NA | [38] |
| EPA-DEP + IA | DI water | 0.2 mS/m | no element | 100 kHz | 5 × 102 μL/min | E. coli strain K12 | NA | 104 to 102 CFU/mL | NA | [116] |
| iDEP + IA | DI water | 1–2 μS/cm | fluorescent beads (2 μm) | 100 Hz | 40 μL/min | B. subtilis spores | 10 μL/min | 106 spores/Ml | NA | [46] |
| nDEPpDEP + IA | Manitol solution | 0.1 mS/m | no element | 1 kHz (nDEP) and 100 kHz (pDEP) | 0.27 m/s | E. coli strain K-12 (NBRC3301) | NA | NA | NA | [35] |
| pDEP + IA | PBS solution and DI water | low | polyclonal antibodies | 100 Hz–1 MHz | 2–4 μL/min | E. coli O157:H7 | 3 × 105 CFU/mL | 3 × 102 CFU/mL | NA | [14] |
| DEP + IA | Milli-Q water | 0.5 × 10−3 to 2.5 × 10−3 S/m | no element | 500 Hz to 5 kHz | 10 μL/min | E. coli 5K strains | NA | 2 × 107 cells/mL | 3.1% | [36] |
| DEP + IA + (AC-EO) | Phosphate buffered saline (PBS at pH 7.4) | 1.8 mS/m | no element | 10 kHz–63 MHz (AC-EO) | 5 μL/min | S. epidermidis ATCC 35984 | NA | 3.5 × 105 CFU/mL and 3.8 × 106 CFU/mL | NA | [37] |
| nDEP + IA | Drinking water | 0.0086 S/m (aprox) | no element | 1 kHz–10 MHz | 25 μL/min | E. coli ATTC 8739 | (150–1500 CFU/mL) | 300 CFU/mL | 1.13% ± 0.37% | [30] |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Páez-Avilés, C.; Juanola-Feliu, E.; Punter-Villagrasa, J.; Del Moral Zamora, B.; Homs-Corbera, A.; Colomer-Farrarons, J.; Miribel-Català, P.L.; Samitier, J. Combined Dielectrophoresis and Impedance Systems for Bacteria Analysis in Microfluidic On-Chip Platforms. Sensors 2016, 16, 1514. https://doi.org/10.3390/s16091514
Páez-Avilés C, Juanola-Feliu E, Punter-Villagrasa J, Del Moral Zamora B, Homs-Corbera A, Colomer-Farrarons J, Miribel-Català PL, Samitier J. Combined Dielectrophoresis and Impedance Systems for Bacteria Analysis in Microfluidic On-Chip Platforms. Sensors. 2016; 16(9):1514. https://doi.org/10.3390/s16091514
Chicago/Turabian StylePáez-Avilés, Cristina, Esteve Juanola-Feliu, Jaime Punter-Villagrasa, Beatriz Del Moral Zamora, Antoni Homs-Corbera, Jordi Colomer-Farrarons, Pere Lluís Miribel-Català, and Josep Samitier. 2016. "Combined Dielectrophoresis and Impedance Systems for Bacteria Analysis in Microfluidic On-Chip Platforms" Sensors 16, no. 9: 1514. https://doi.org/10.3390/s16091514
APA StylePáez-Avilés, C., Juanola-Feliu, E., Punter-Villagrasa, J., Del Moral Zamora, B., Homs-Corbera, A., Colomer-Farrarons, J., Miribel-Català, P. L., & Samitier, J. (2016). Combined Dielectrophoresis and Impedance Systems for Bacteria Analysis in Microfluidic On-Chip Platforms. Sensors, 16(9), 1514. https://doi.org/10.3390/s16091514







