Surface Acoustic Waves (SAW)-Based Biosensing for Quantification of Cell Growth in 2D and 3D Cultures
Abstract
:1. Introduction
2. Working Principle
3. Design and Fabrication of Bio-Sensor
3.1. Device Design
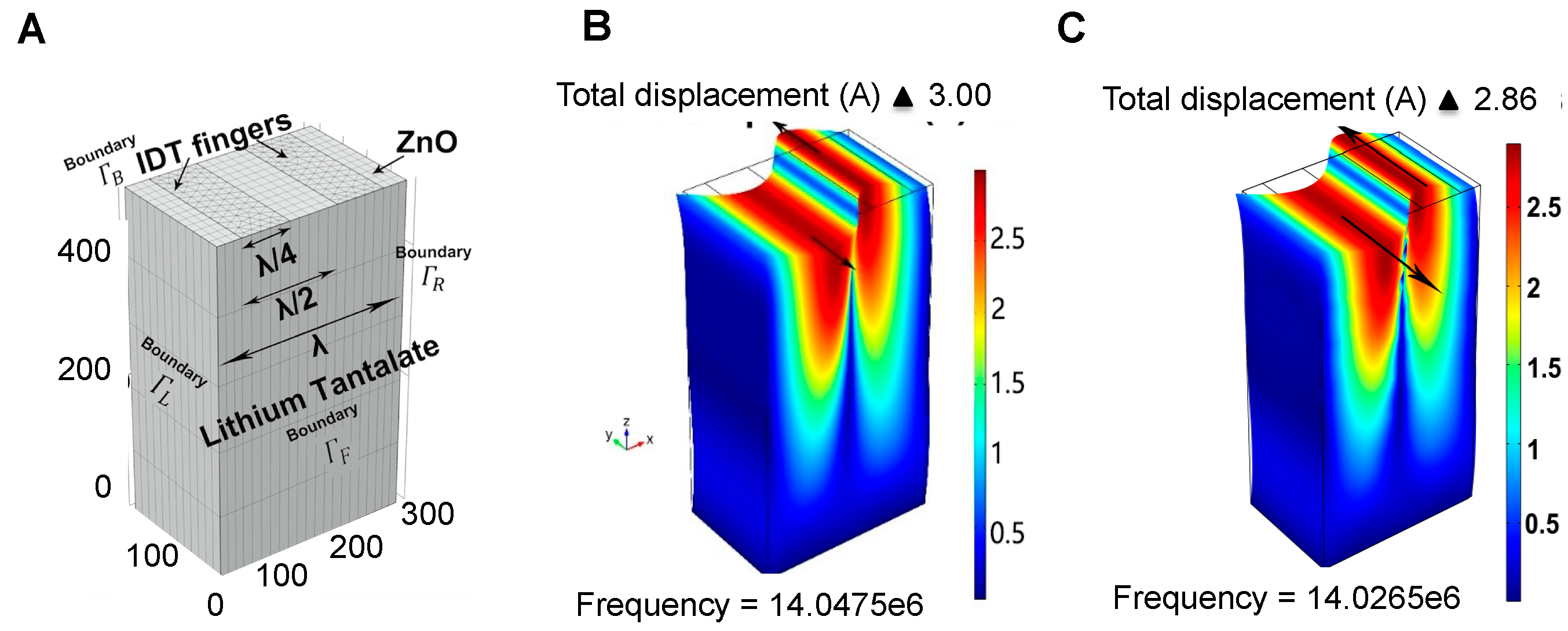
| PARAMETERS | SETTINGS |
|---|---|
| Wavelength (λ) | 297 μm |
| Number of reflecting fingers | 30 pairs |
| Finger width | 74.25 μm |
| Wavelength of reflecting fingers | 297 μm |
| Number of fingers | 30 pairs |
| Well diameter | 6.5 mm |
| SAW velocity | 4160 m/s |
| ZnO layer thickness | 200 nm |
| Finger height | 100 nm |
| Operation frequency | 14.05 MHz |
3.2. Device Fabrication

4. Experimental Setup
4.1. Experimental Protocol for SAW Measurement
4.2. Experimental Protocol for Measuring Cell Viability
4.3. Experimental Protocol for Measuring Cell Proliferation
4.4. Experimental Protocol for Culturing 3D Tumoroids
5. Results and Discussion
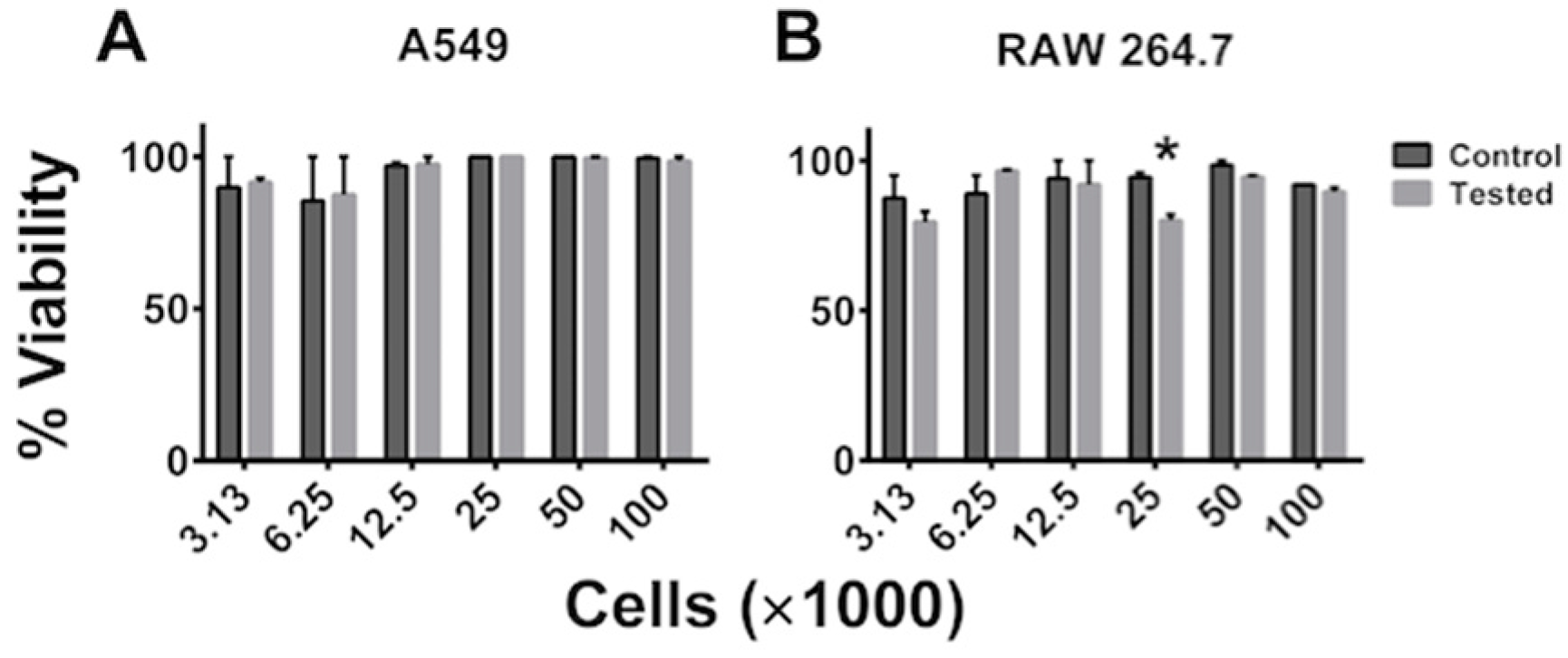
5.1. Cell Viability and Cell Proliferation Is Not Affected after SAW Measurements
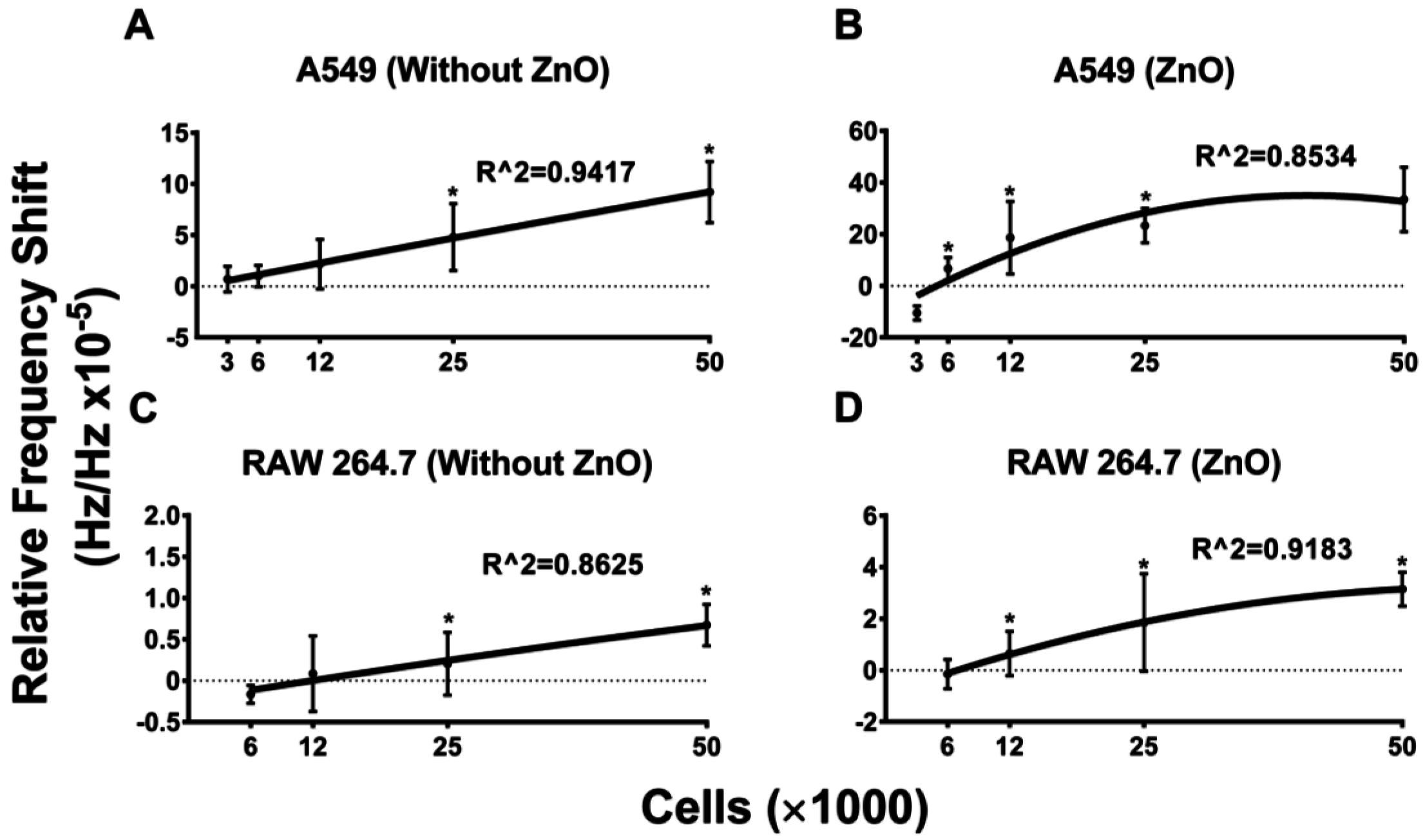
5.2. Frequency Shift Increases with Increasing Cell Concentration and Sensitivity Is Further Aided by the Use of ZnO
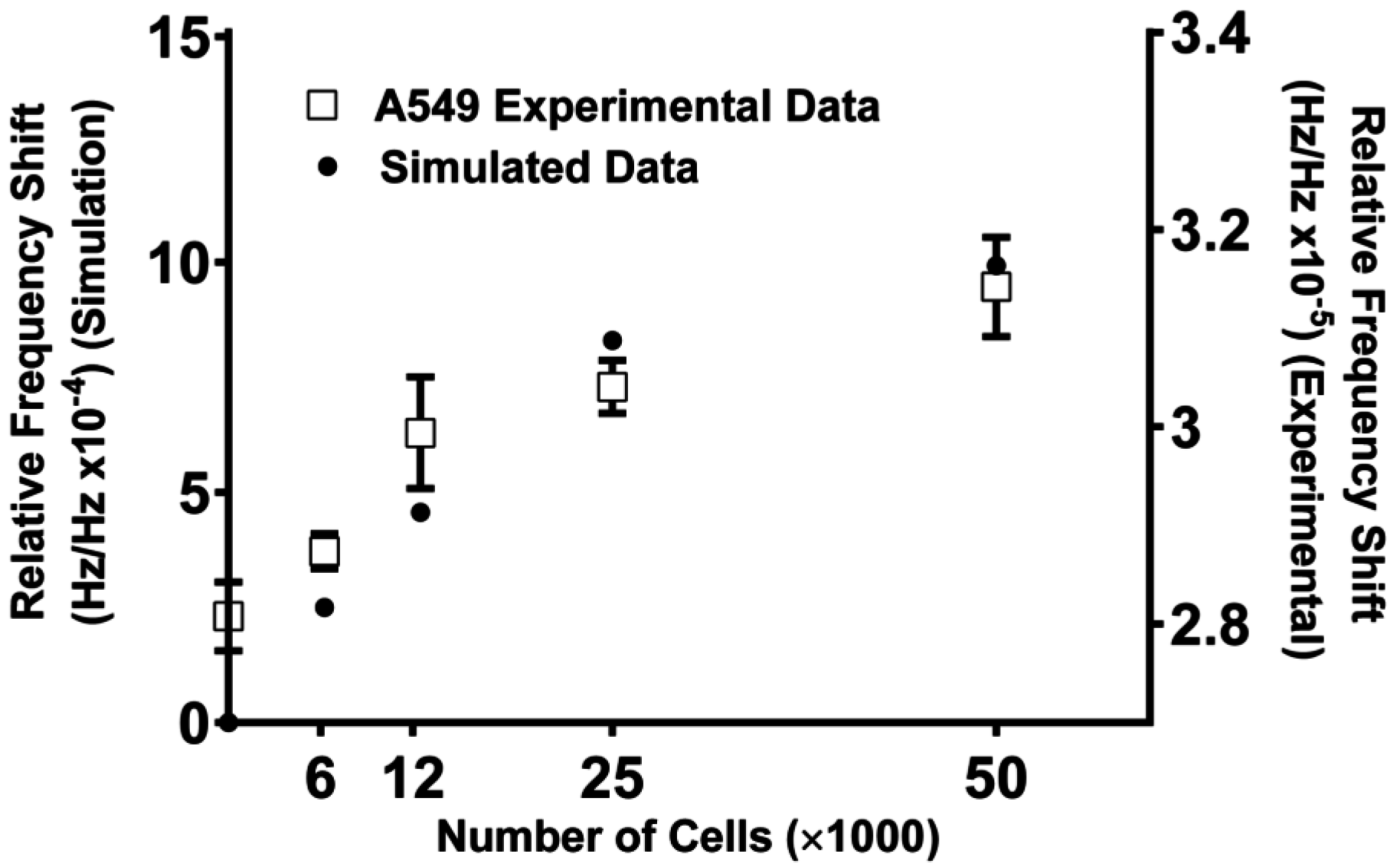
5.3. SAW Measurements of Cell Density Match Simulation Results
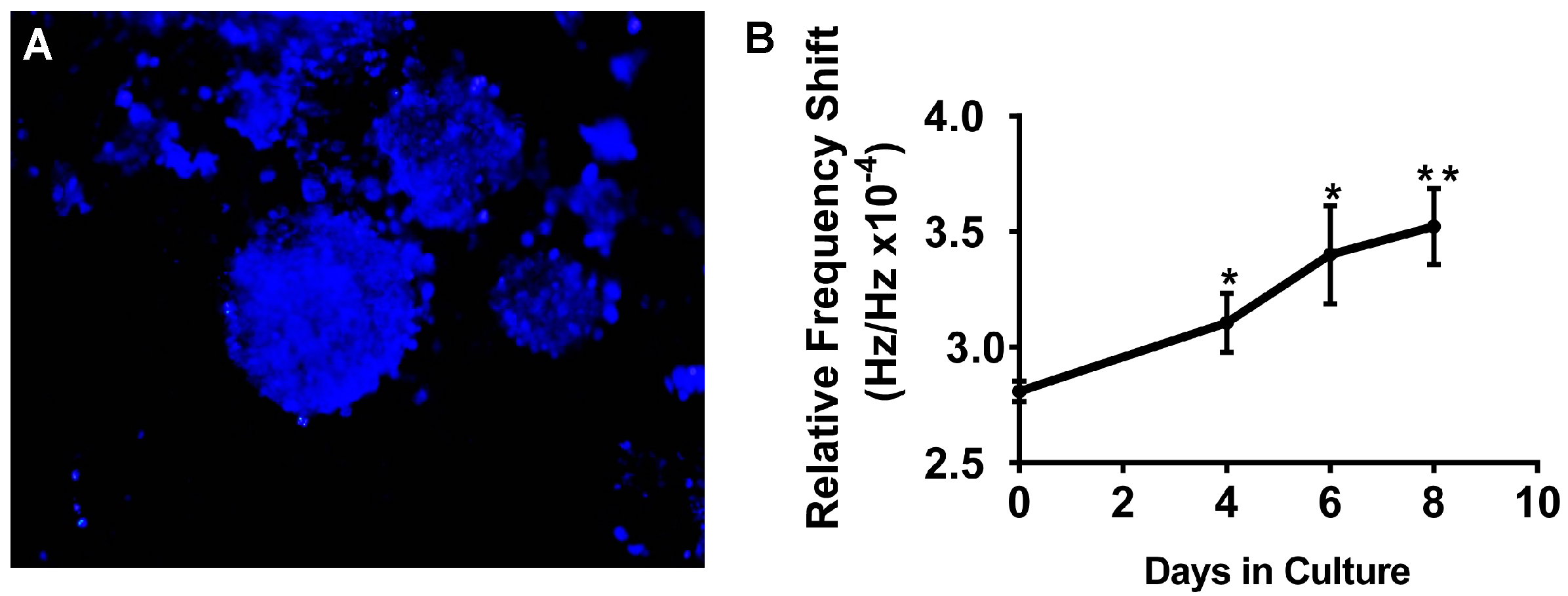
5.4. SAW Measurements Aid in Monitoring Growth of A549 3D Spheroid Cultures
6. Conclusions
Supplementary Files
Supplementary File 1Acknowledgments
Author Contributions
Conflicts of Interest
References
- Shen, C.; Liou, S. Surface acoustic wave gas monitor for ppm ammonia detection. Sens. Actuators B Chem. 2008, 131, 673–679. [Google Scholar] [CrossRef]
- Onen, O.; Ahmad, A.A.; Guldiken, R.; Gallant, N.D. Surface Modification on Acoustic Wave Biosensors for Enhanced Specificity. Sensors 2012, 12, 12317–12328. [Google Scholar] [CrossRef] [PubMed]
- Onen, O.; Sisman, A.; Gallant, N.D.; Kruk, P.; Guldiken, R. A urinary Bcl-2 surface acoustic wave biosensor for early ovarian cancer detection. Sensors 2012, 12, 7423–7437. [Google Scholar] [CrossRef] [PubMed]
- Vivancos, J.-L.; Rácz, Z.; Cole, M.; Gardner, J.W. Surface acoustic wave based analytical system for the detection of liquid detergents. Sens. Actuators B Chem. 2012, 171–172, 469–477. [Google Scholar] [CrossRef]
- Pomowski, A.; Baricham, C.; Rapp, B.E.; Matern, A.; Länge, K. Acoustic Biosensors Coated With Phosphorylcholine Groups for Label-Free Detection of Human C-Reactive Protein in Serum. IEEE Sens. J. 2015, 15, 4388–4392. [Google Scholar] [CrossRef]
- Nomura, T.; Saitoh, A.; Horikoshi, Y. Measurement of acoustic properties of liquid using liquid flow SH-SAW sensor system. Sens. Actuators B Chem. 2001, 76, 69–73. [Google Scholar] [CrossRef]
- Deobagkar, D.D.; Limaye, V.; Sinha, S.; Yadava, R.D.S. Acoustic wave immunosensing of Escherichia coli in water. Sens. Actuators B. Chem. 2005, 104, 85–89. [Google Scholar] [CrossRef]
- Roederer, J.E.; Bastiaans, G.J. Microgravimetric Immunoassay with Piezoelectric Crystals. Anal. Chem. 1983, 55, 2333–2336. [Google Scholar] [CrossRef]
- Kondoh, J.; Tabushi, S.; Matsui, Y.; Shiokawa, S. Development of methanol sensor using a shear horizontal surface acoustic wave device for a direct methanol fuel cell. Sens. Actuators B Chem. 2008, 129, 575–580. [Google Scholar] [CrossRef]
- Nomura, T.; Saitoh, A.; Miyazaki, T. Liquid sensor probe using reflecting SH-SAW delay line. Sens. Actuators B Chem. 2003, 91, 298–302. [Google Scholar] [CrossRef]
- Litao, Y.; Fu, Y.; Li, Z.; Li, Y.; Ma, J.; Placido, F.; Walton, A.J.; Zu, X. Love mode surface acoustic wave ultraviolet sensor using ZnO films deposited on. Sens. Actuators A. Phys. 2013, 193, 87–94. [Google Scholar]
- Lam, C.S. A Review of the Recent Development of Temperature Stable Cuts of Quartz for SAW Applications. In Proceedings of the Fourth International Symposium on Acoustic Wave Devices for Future Mobile Communication Systems, Chiba, Japan, 3–5 March 2010.
- Shilton, R.J.; Travagliati, M.; Beltram, F.; Cecchini, M. Microfluidic pumping through miniaturized channels driven by ultra-high frequency surface acoustic waves. Appl. Phys. Lett. 2014, 105. [Google Scholar] [CrossRef]
- Li, F. The Acoustic Wave Sensor and Soft Lithography Technologies for Cell Biological Studies; ProQuest, UMI Dissertation Publishing: Ann Arbor, MI, USA, 2011. [Google Scholar]
- Jo, M.C.; Guldiken, R. Effects of polydimethylsiloxane (PDMS) microchannels on surface acoustic wave-based microfluidic devices. Microelectron. Eng. 2014, 113, 98–104. [Google Scholar] [CrossRef]
- Oh, H.; Lee, K.J.; Baek, J.; Yang, S.S.; Lee, K. Development of a high sensitive pH sensor based on shear horizontal surface acoustic wave with ZnO nanoparticles. Microelectron. Eng. 2013, 111, 154–159. [Google Scholar] [CrossRef]
- Chivukula, V.; Ciplys, D.; Shur, M.; Dutta, P. ZnO nanoparticle surface acoustic wave UV sensor. Appl. Phys. Lett. 2010, 96, 3–6. [Google Scholar] [CrossRef]
- Powell, D.A.; Kalantar-zadeh, K.; Wlodarski, W. Numerical calculation of SAW sensitivity: Application to ZnO/LiTaO3 transducers. Sens. Actuators A Phys. 2004, 115, 456–461. [Google Scholar] [CrossRef]
- Powell, D.A.; Kalantar-zadeh, K.; Ippolito, S.; Wlodarski, W. A layered SAW device based on ZnO/LiTaO3 for liquid media sensing applications. In Proceedings of the 2002 IEEE International Ultrasonics Symposium, Munich, Germany, 8–11 October 2002; Volume 1, pp. 493–496.
- Chang, R.-C.; Chu, S.-Y.; Hong, C.-S.; Chuang, Y.-T. A study of Love wave devices in ZnO/Quartz and ZnO/LiTaO3 structures. Thin Solid Films 2006, 498, 146–151. [Google Scholar] [CrossRef]
- Fu, Y.Q.; Luo, J.K.; Du, X.Y.; Flewitt, A.J.; Li, Y.; Markx, G.H.; Walton, A.J.; Milne, W.I. Recent developments on ZnO films for acoustic wave based bio-sensing and microfluidic applications: A review. Sens. Actuators B Chem. 2010, 143, 606–619. [Google Scholar] [CrossRef]
- Kondoh, J. A liquid-phase sensor using shear horizontal surface acoustic wave devices. Electron. Commun. Jpn. 2013, 96, 41–49. [Google Scholar] [CrossRef]
- Onen, O.; Guldiken, R. Investigation of guided surface acoustic wave sensors by analytical modeling and perturbation analysis. Sens. Actuators A Phys. 2014, 205, 38–46. [Google Scholar] [CrossRef]
- Guldiken, R.; Jo, M.C.; Gallant, N.D.; Demirci, U.; Zhe, J. Sheathless size-based acoustic particle separation. Sensors 2012, 12, 905–922. [Google Scholar] [CrossRef] [PubMed]
- Jo, M.C.; Guldiken, R. Active density-based separation using standing surface acoustic waves. Sens. Actuators A Phys. 2012, 187, 22–28. [Google Scholar] [CrossRef]
- Jo, M.C.; Guldiken, R. Dual surface acoustic wave-based active mixing in a microfluidic channel. Sens. Actuators A Phys. 2013, 196, 1–7. [Google Scholar] [CrossRef]
- Rocha-Gaso, M.-I.; March-Iborra, C.; Montoya-Baides, A.; Arnau-Vives, A. Surface generated acoustic wave biosensors for the detection of pathogens: A review. Sensors 2009, 9, 5740–5769. [Google Scholar] [CrossRef] [PubMed]
- Girard, Y.K.; Wang, C.; Ravi, S.; Howell, M.C.; Mallela, J.; Alibrahim, M.; Green, R.; Hellermann, G.; Mohapatra, S.S.; Mohapatra, S. A 3D Fibrous Scaffold Inducing Tumoroids: A Platform for Anticancer Drug Development. PLoS ONE 2013, 8. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, T.; Green, R.; Nair, R.R.; Howell, M.; Mohapatra, S.; Guldiken, R.; Mohapatra, S.S. Surface Acoustic Waves (SAW)-Based Biosensing for Quantification of Cell Growth in 2D and 3D Cultures. Sensors 2015, 15, 32045-32055. https://doi.org/10.3390/s151229909
Wang T, Green R, Nair RR, Howell M, Mohapatra S, Guldiken R, Mohapatra SS. Surface Acoustic Waves (SAW)-Based Biosensing for Quantification of Cell Growth in 2D and 3D Cultures. Sensors. 2015; 15(12):32045-32055. https://doi.org/10.3390/s151229909
Chicago/Turabian StyleWang, Tao, Ryan Green, Rajesh Ramakrishnan Nair, Mark Howell, Subhra Mohapatra, Rasim Guldiken, and Shyam Sundar Mohapatra. 2015. "Surface Acoustic Waves (SAW)-Based Biosensing for Quantification of Cell Growth in 2D and 3D Cultures" Sensors 15, no. 12: 32045-32055. https://doi.org/10.3390/s151229909
APA StyleWang, T., Green, R., Nair, R. R., Howell, M., Mohapatra, S., Guldiken, R., & Mohapatra, S. S. (2015). Surface Acoustic Waves (SAW)-Based Biosensing for Quantification of Cell Growth in 2D and 3D Cultures. Sensors, 15(12), 32045-32055. https://doi.org/10.3390/s151229909








