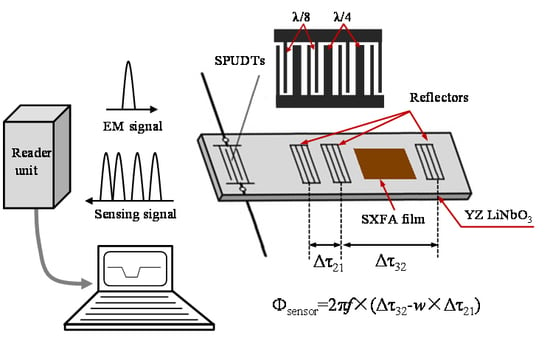
Development of a Wireless and Passive SAW-Based Chemical Sensor for Organophosphorous Compound Detection
Abstract
:1. Introduction
- The sensor chip is maintenance-free because it is passive, and does not require any power supply to operate;
- The working environment can be very hazardous such as contaminated and high voltage areas because of the wireless measurement;

2. Design Considerations
2.1. Reader Unit
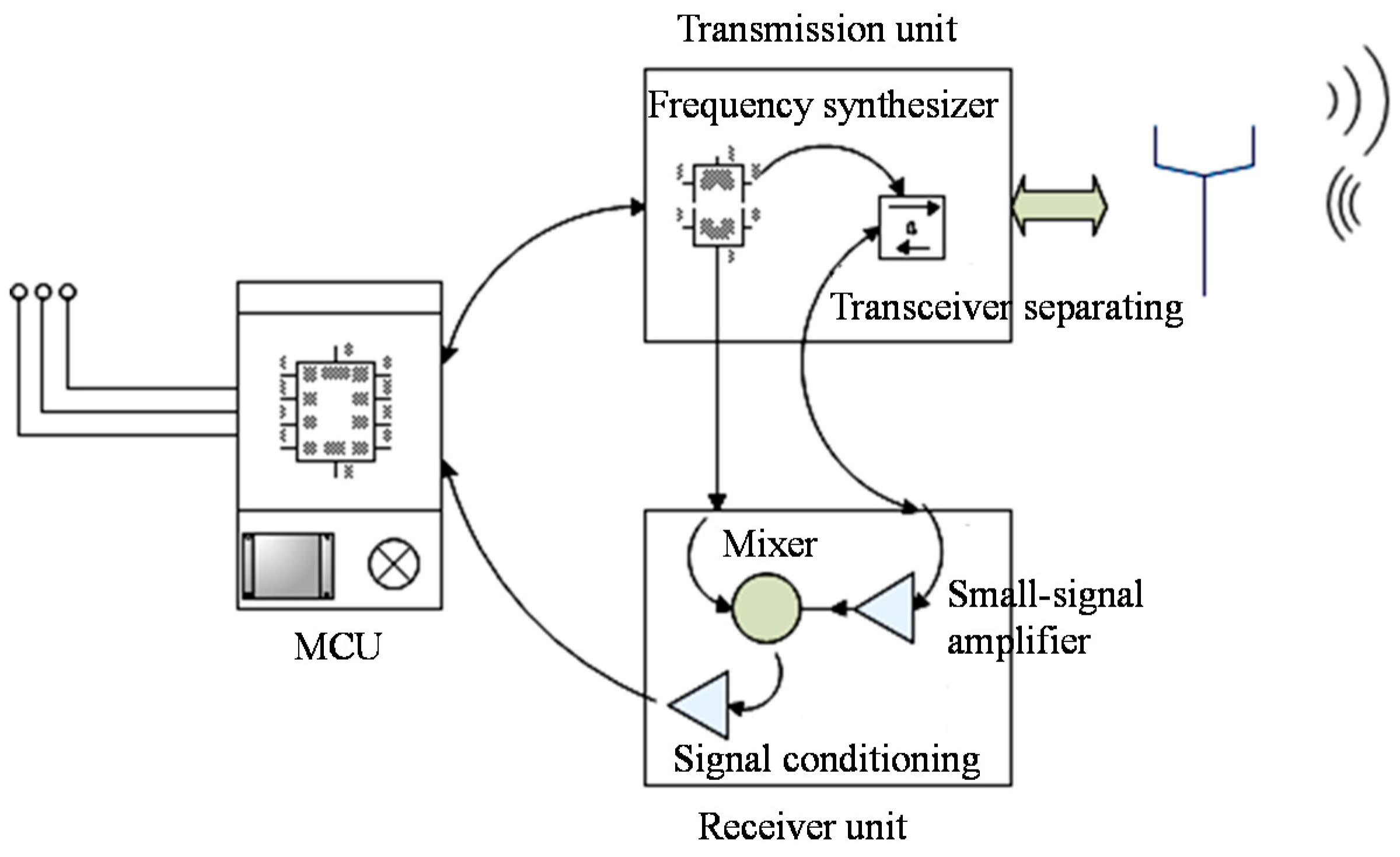
2.2. Design on the SAW Sensor Chip
| Design Parameters | Value |
|---|---|
| Operation frequency (MHz) | 434 |
| Piezoelectric substrate | YZ LiNbO3 |
| Thickness of the Al electrodes (nm) | 300 |
| Distance between SPUDT and the first reflector (λ: wavelength) | 250.25 |
| Distance between the second and the third reflector (λ) | 399.75 |
| The length of the second reflector(λ) | 2.25 |
| Acoustic velocity (m/s) | 3492 |
| Aperture (λ) | 80 |
| Number of SPUDT finger pairs | 15 |
| Distance between the first and the second reflector (λ) | 30.75 |
| Length of the first reflector (λ) | 2.25 |
| Length of the third reflector (λ) | 4.25 |
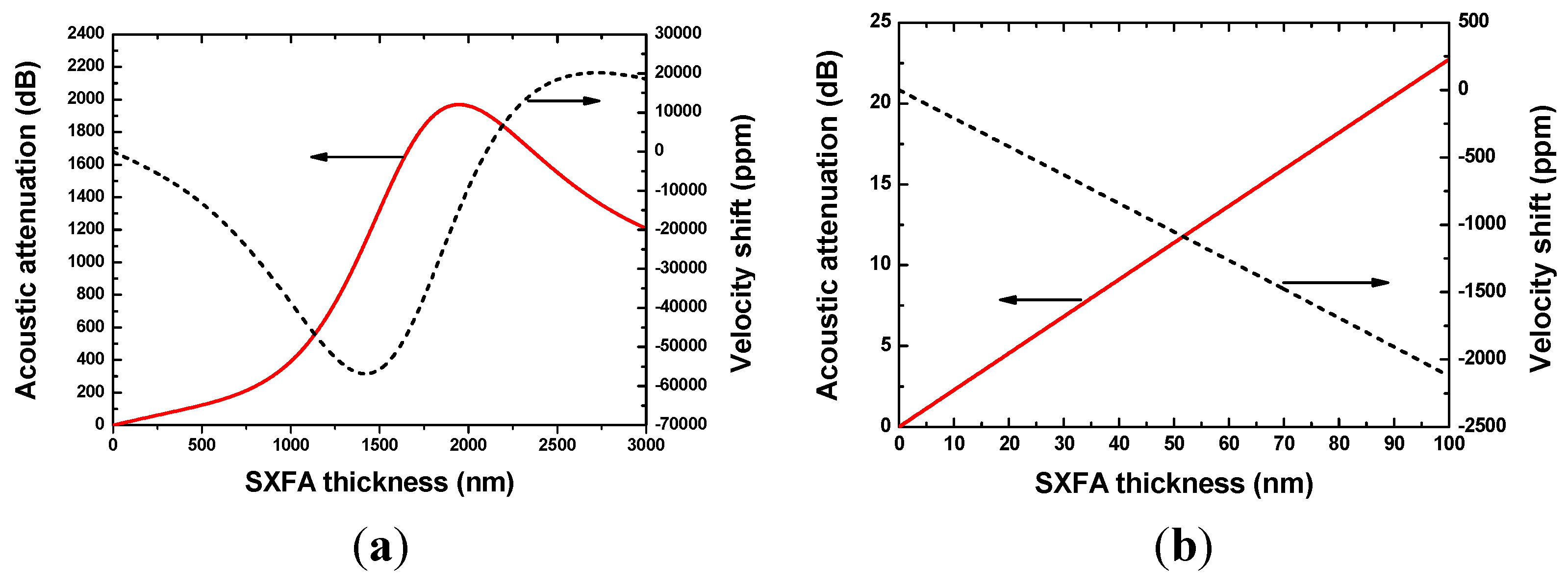
2.3. Theoretical Analysis of the Sensor Response
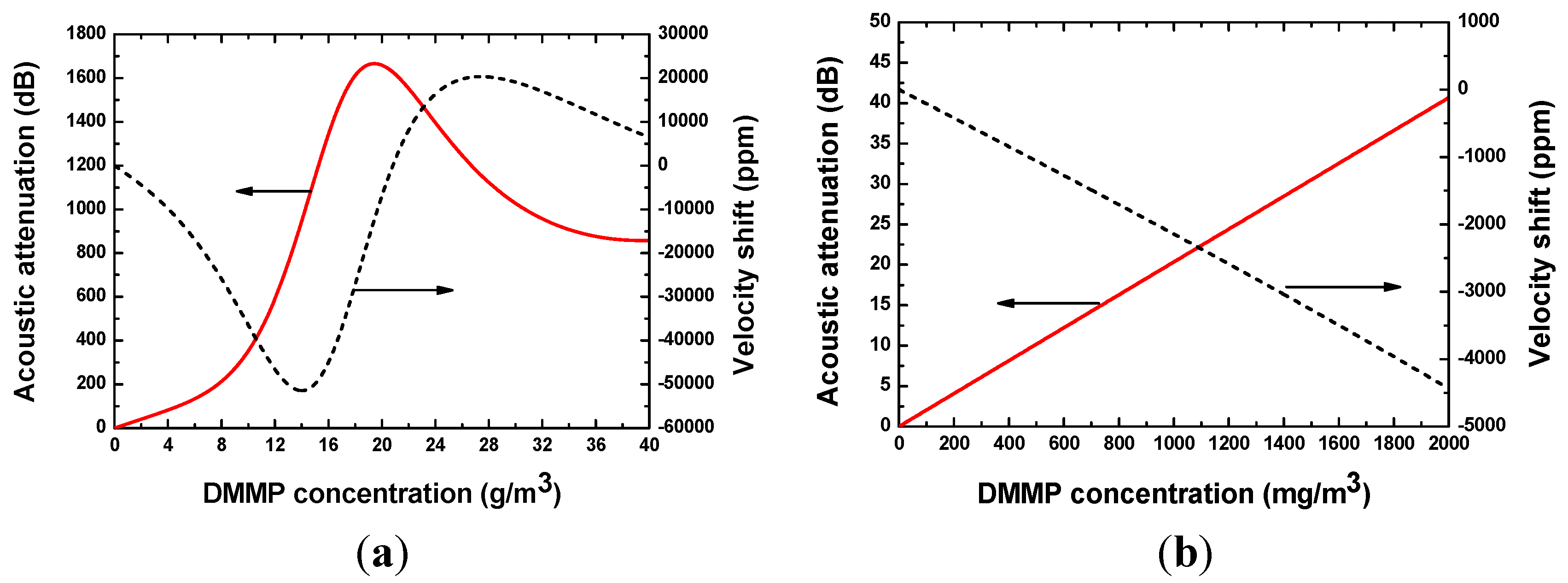
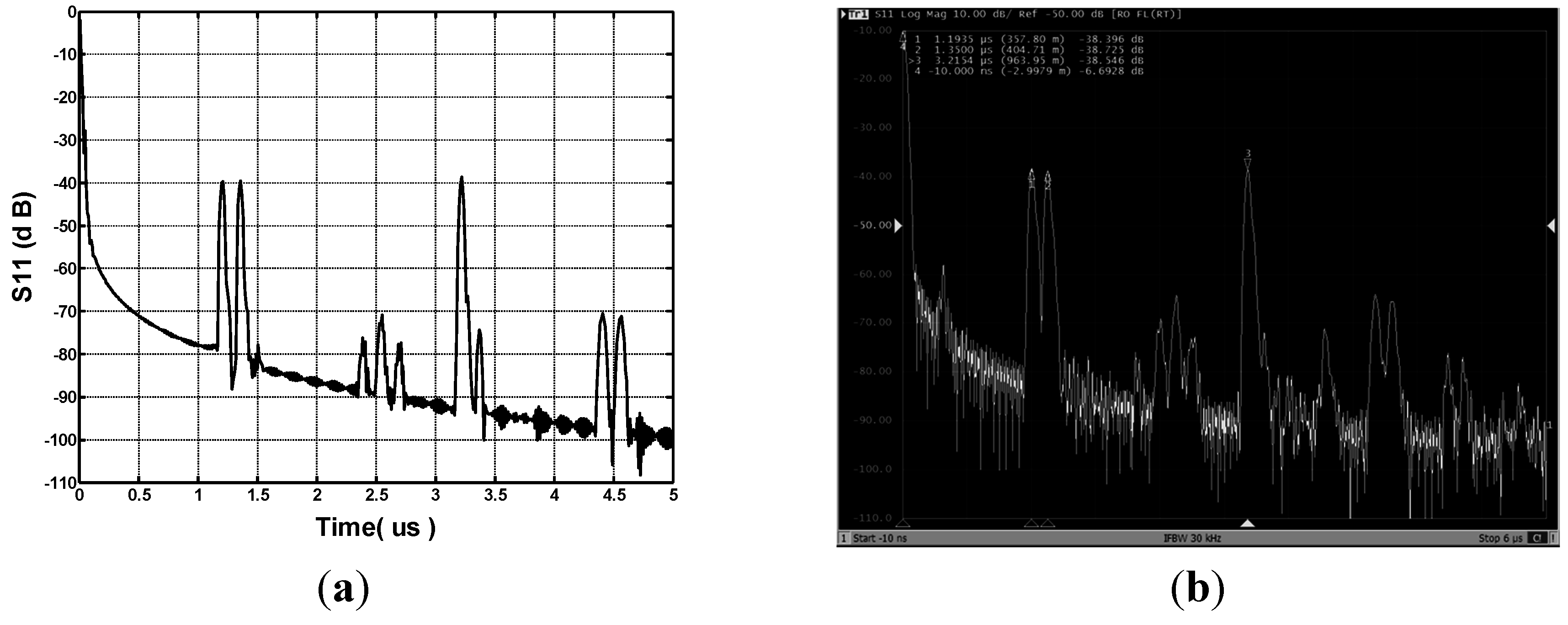
3. Technique Realization
3.1. SAW Sensor
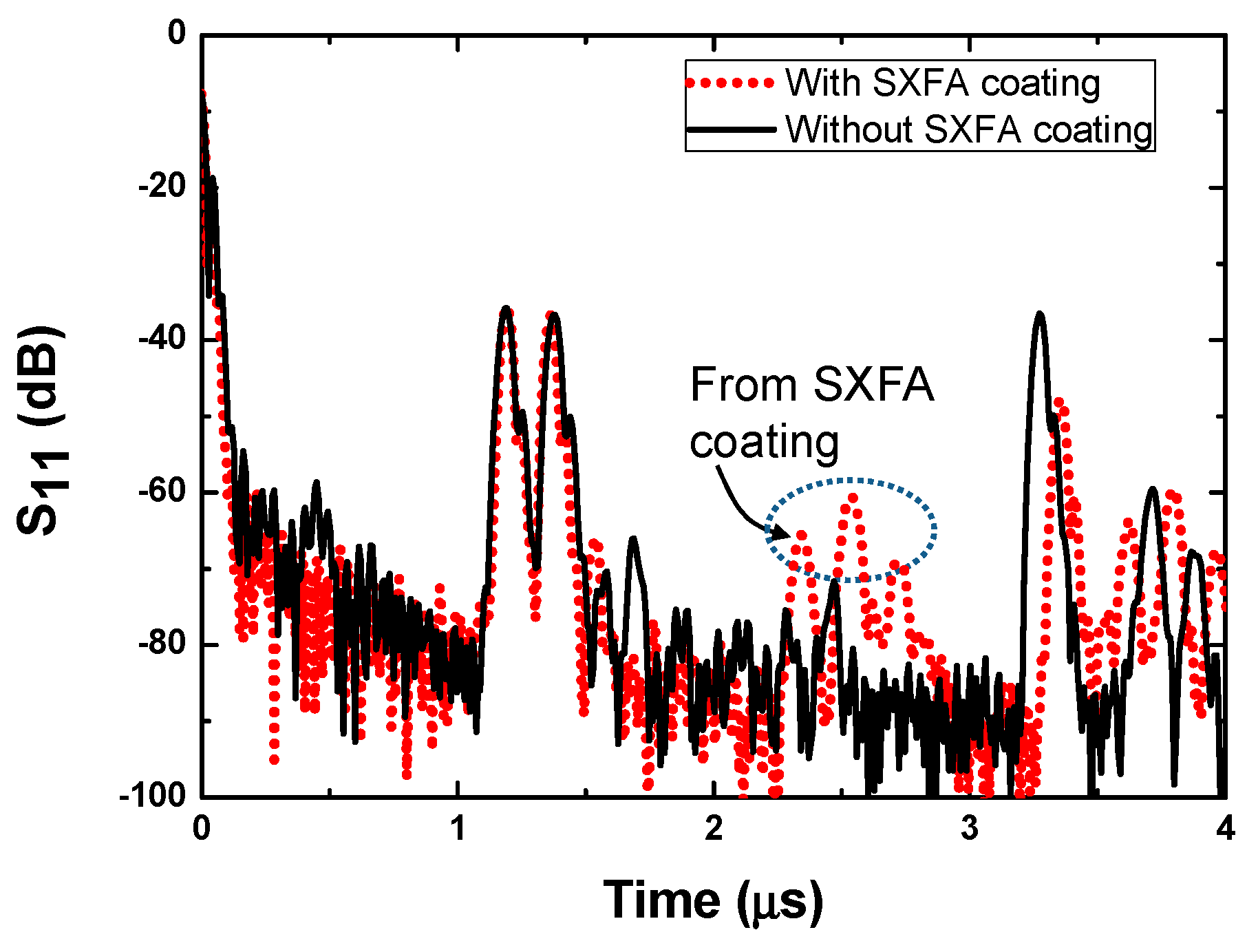
3.2. Sensor Experimental Setup
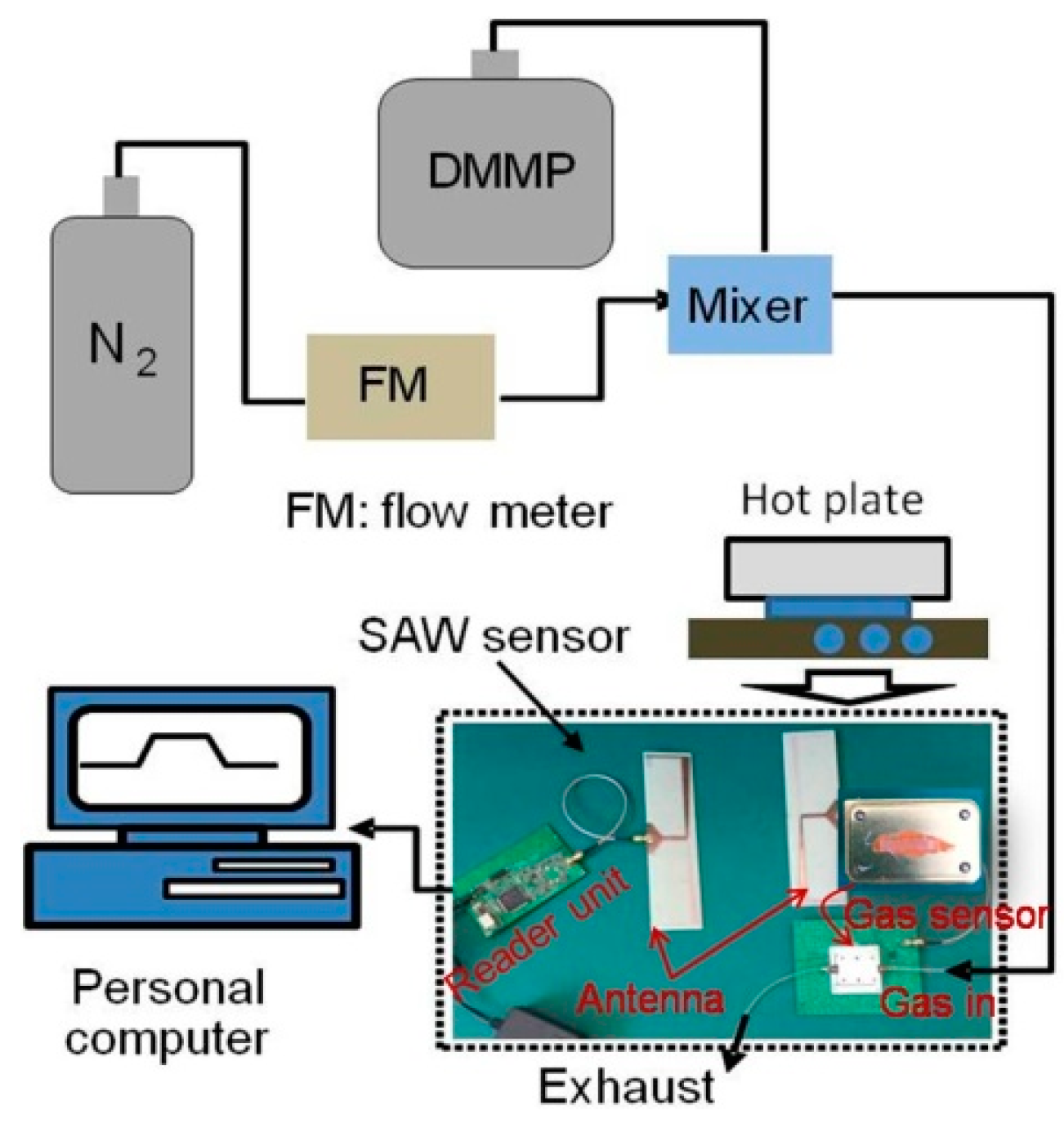
4. Wireless Measurement of the SAW Sensor
4.1. DMMP Adsorption
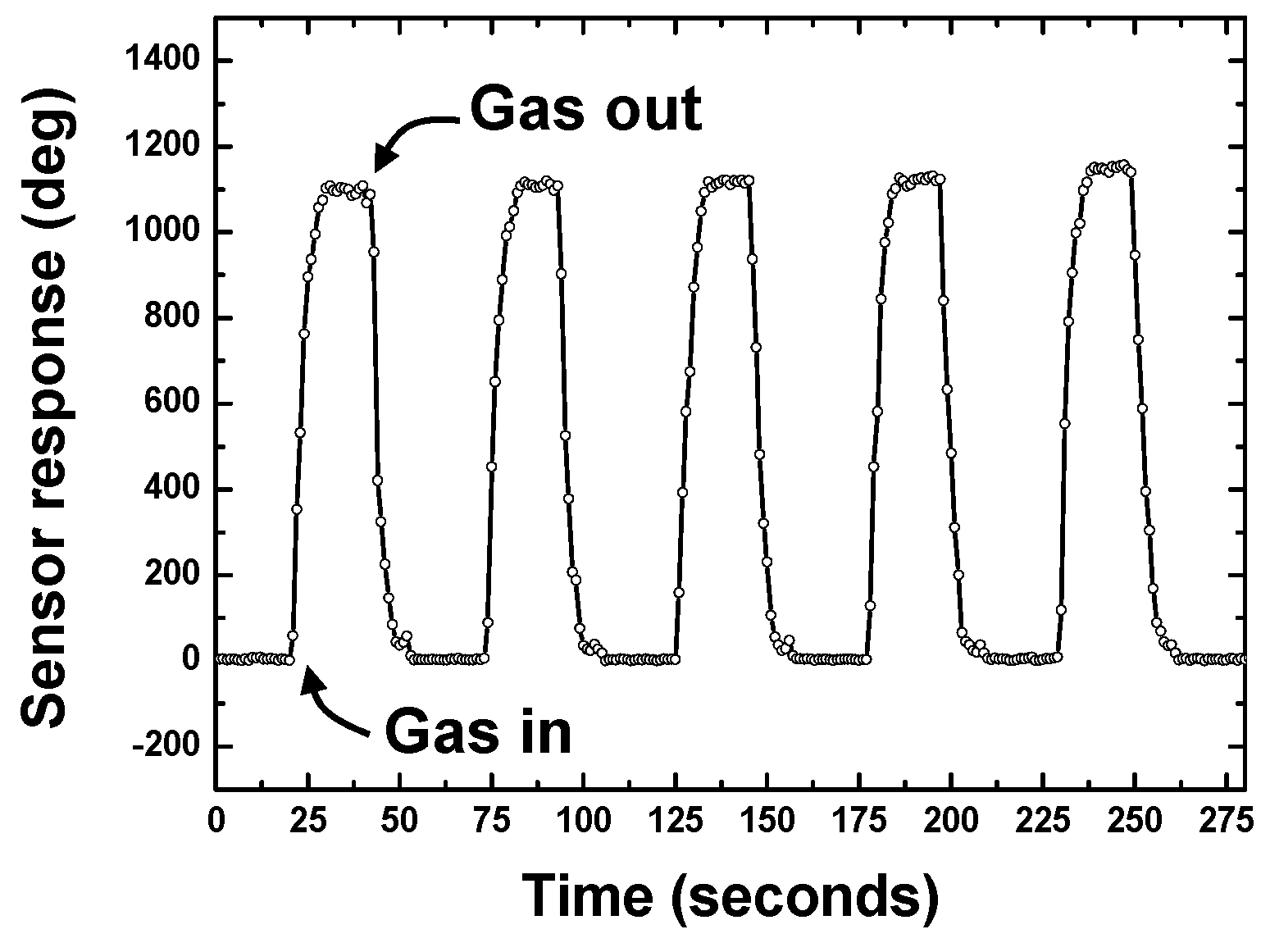
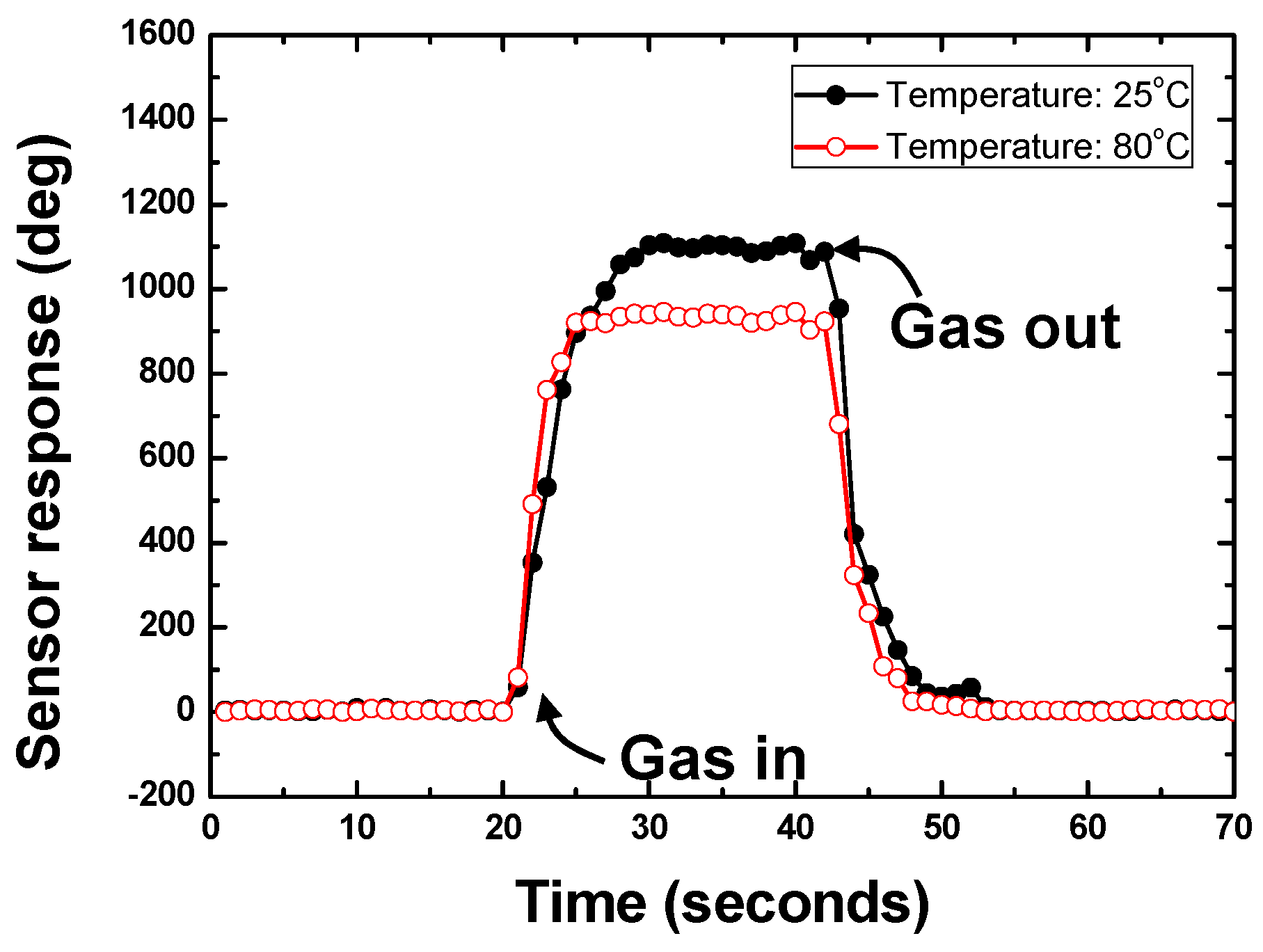
4.2. Temperature Effect on Sensor Response
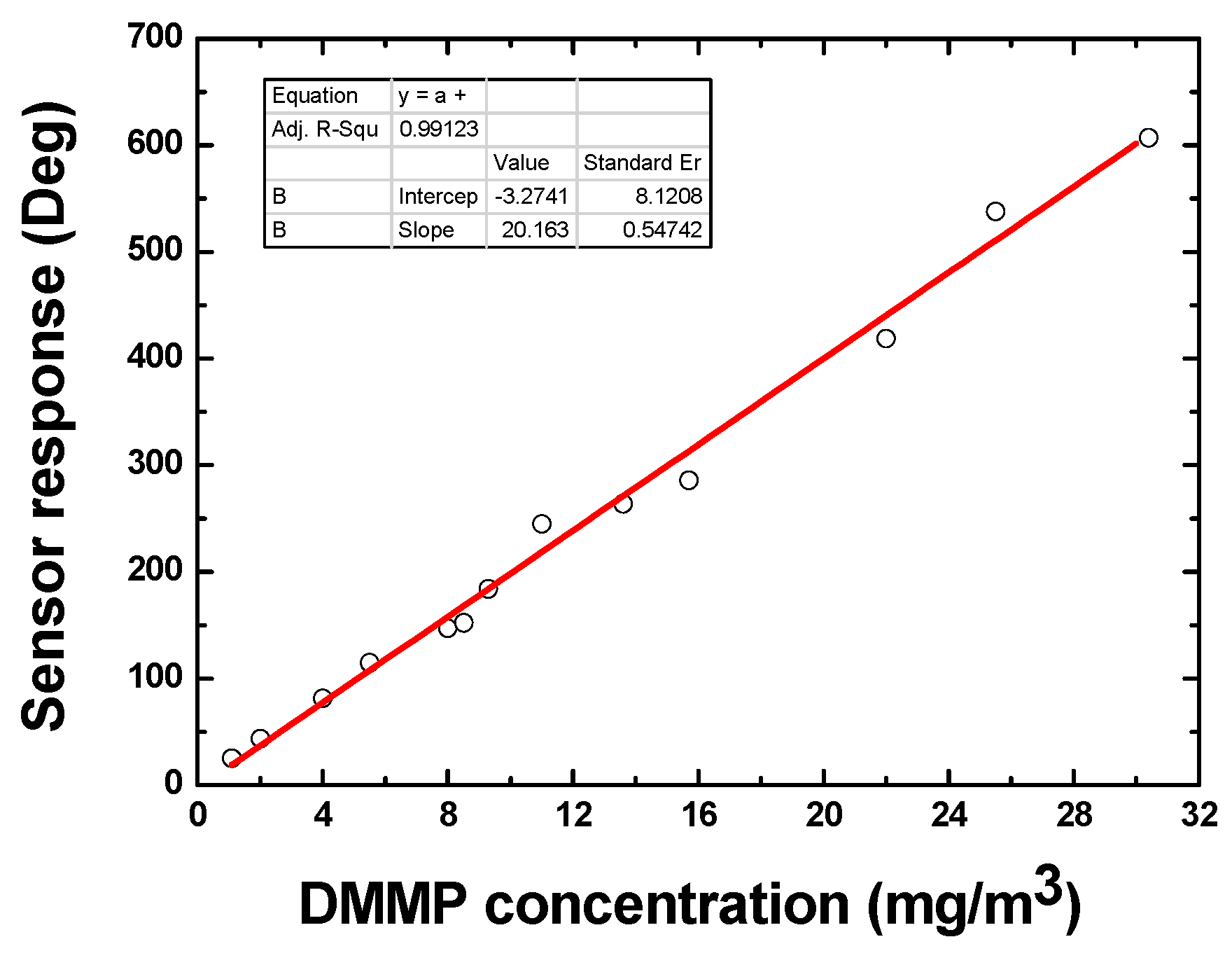
4.3. Sensitivity Evaluation
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Ricco, A.J.; Crooks, R.M.; Osbourn, G.C. Surface acoustic wave chemical sensor arrays: New chemically sensitive interfaces combined with novel cluster analysis to detect volatile organic compounds and mixtures. Acc. Chem. Res. 1998, 31, 289–296. [Google Scholar] [CrossRef]
- Ballantine, D.S., Jr.; White, R.M.; Martin, S.J. Acoustic Wave Sensors: Theory, Design &Physico-Chemical Applications; Academic Press: San Diego, CA, USA, 1996. [Google Scholar]
- Pan, Y.; Yang, L.; Mu, N.; Shao, S.; Wang, W.; Xie, X.; He, S. Selective surface acoustic wave-based organophosphorus sensor employing a host-guest self-assembly monolayer of β-cyclodextrin derivative. Sensors 2015, 15, 17916–17925. [Google Scholar] [CrossRef] [PubMed]
- Afzala, A.; Iqbala, N. Advanced vapor recognition materials for selective and fast responsive surface acoustic wave sensors: A review. Anal. Chem. Acta 2013, 787, 36–49. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Hu, H.; Chen, G.; Xie, X.; He, S. Optimization of a BSP3-coated surface acoustic wave chemical sensor. IEEE Sens. J. 2015, 15, 6730–6737. [Google Scholar] [CrossRef]
- Joo, B.S.; Huh, J.; Lee, D. Fabrication of polymer SAW sensor array to classify chemical warfare agents. Sens. Actuators B Chem. 2007, 121, 47–53. [Google Scholar] [CrossRef]
- Barie, N.; Bucking, M.; Rapp, M. A novel electronic nose based on miniaturized SAW sensor arrays coupled with SPME enhanced headspace-analysis and its use for rapid determination of volatile organic compounds in food quality monitoring. Sens. Actuators B Chem. 2006, 114, 482–488. [Google Scholar] [CrossRef]
- Binder, A.; Bruckner, G.; Schobernig, N.; Schmitt, D. Wireless surface acoustic wave pressure and temperature sensor with unique identification based on LiNbO3. IEEE Sens. J. 2013, 13, 1801–1805. [Google Scholar] [CrossRef]
- Reindl, L. Wireless passive SAW identification marks and sensors. In Proceedings of the 2nd International Symposium on Acoustic Wave Devices for Future Mobile Communication Systems, Chiba, Japan, 3–5 March 2004; pp. 1–15.
- Stoney, R.; Donohoe, B.; Geraghty, D.; O’Donnell, G.E. The development of surface acoustic wave sensors (SAWs) for process monitoring. Procedia CIRP 2012, 1, 569–574. [Google Scholar] [CrossRef]
- Li, B.; Yassinem, O.; Kosel, J. A surface acoustic wave passive and wireless sensor for magnetic fields, temperature, and humidity. IEEE Sens. J. 2015, 15, 453–462. [Google Scholar] [CrossRef]
- Springer, A.; Weigel, R.; Pohl, A.; Seifert, F. Wireless identification and sensing using surface acoustic wave devices. Mechatronics 1999, 9, 745–756. [Google Scholar] [CrossRef]
- Wang, W.; Lee, K.; Kim, T.; Yang, S.; Woo, I.; Park, I. A novel wireless, passive CO2 sensor incorporating a surface acoustic wave reflective delay line. Smart Mater. Struct. 2007, 16, 1382–1389. [Google Scholar] [CrossRef]
- Lim, C.-B.; Wang, W.; Yang, S.S.; Lee, K.K. Development of a wireless, battery-free SAW volatile organic compounds sensor integrated with temperature sensor. Sens. Lett. 2011, 9, 82–86. [Google Scholar] [CrossRef]
- Wang, W.; Lim, C.; Lee, K.; Yang, S. Wireless surface acoustic wave chemical sensor for simultaneous measurement of CO2 and humidity. J. Micro/Nanolith. MEMS MOEMS 2009, 8. [Google Scholar] [CrossRef]
- Lim, C.; Wang, W.; Yang, S.S.; Lee, K.K. Development of SAW-based multi-gas sensor for simultaneous detection of CO2 and NO2. Sens. Actuators B Chem. 2011, 154, 9–16. [Google Scholar] [CrossRef]
- Lieberzeit, P.A.; Christian, P.; Dickert, F.L.; Gerhard, F. SAW RFID-tags for mass-sensitive detection of humidity and vapors. Sensors 2009, 9, 9805–9815. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.S.; Chen, Y.Y.; Wu, T.T. A passive wireless hydrogen surface acoustic wave sensor based on Pt-coated ZnO nanorods. Nanotechnology 2010, 21. [Google Scholar] [CrossRef] [PubMed]
- Westafer, R.S.; Levitin, G.; Hess, D.W.; Michael, H.B.; William, D.H. Detection of ppb ozone using a dispersive surface acoustic wave reflective delay line with integrated reference signal. Sens. Actuators B Chem. 2014, 192, 406–413. [Google Scholar] [CrossRef]
- Wang, W.; He, S.T.; Liu, M.H.; Pan, Y. Advances in SXFA-coated SAW chemical sensor for organophosphorous compound detection. Sensors 2011, 11, 1526–1541. [Google Scholar] [CrossRef] [PubMed]
- Grate, J.W. Siloxanes with Strongly Hydrogen Bond Donating Functionalities. U.S. Patent No. 5,756,631, 26 May 1998. [Google Scholar]
- Hauser, R.; Carinthian, T.R.; Villach, A.; Fachberger, R.; Bruckner, G.; Smetana, W. A wireless SAW-based temperature sensor for harsh environment. In Proceedings of the 2004 IEEE Sensors, Vienna, Austria, 24–27 October 2004; pp. 860–863.
- Reindl, L.; Shrena, I.M. Wireless measurement of temperature using surface acoustic wave sensors. IEEE Trans. Ultrason. Ferroelectr. Freq. Control 2004, 51, 1457–1463. [Google Scholar] [CrossRef] [PubMed]
- Peter, R.; Harmann, C.S. Passive long range and high temperature ID systems based on SAW technology. In Proceedings of the Sensor Conference, Nuremberg, Germany, 13–15 May 2003; pp. 335–340.
- Wang, W.; Lee, K.K.; Yang, S.S. Optimal design on SAW sensor for wireless pressure measurement based on reflective delay line. Sens. Actuators A Phys. 2007, 139, 2–6. [Google Scholar] [CrossRef]
- Martin, S.J.; Frye, G.C.; Senturla, S.D. Dynamics and response of polymer-coated surface acoustic wave devices: Effect of viscoelastic properties and film resonance. Anal. Chem. 1994, 66, 2201–2219. [Google Scholar] [CrossRef]
- Grate, J.W. Hydrogen-bond acidic polymers for chemical vapor sensing. Chem. Rev. 2008, 108, 726–745. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xu, F.-Q.; Wang, W.; Xue, X.-F.; Hu, H.-L.; Liu, X.-L.; Pan, Y. Development of a Wireless and Passive SAW-Based Chemical Sensor for Organophosphorous Compound Detection. Sensors 2015, 15, 30187-30198. https://doi.org/10.3390/s151229793
Xu F-Q, Wang W, Xue X-F, Hu H-L, Liu X-L, Pan Y. Development of a Wireless and Passive SAW-Based Chemical Sensor for Organophosphorous Compound Detection. Sensors. 2015; 15(12):30187-30198. https://doi.org/10.3390/s151229793
Chicago/Turabian StyleXu, Fang-Qian, Wen Wang, Xu-Feng Xue, Hao-Liang Hu, Xin-Lu Liu, and Yong Pan. 2015. "Development of a Wireless and Passive SAW-Based Chemical Sensor for Organophosphorous Compound Detection" Sensors 15, no. 12: 30187-30198. https://doi.org/10.3390/s151229793
APA StyleXu, F.-Q., Wang, W., Xue, X.-F., Hu, H.-L., Liu, X.-L., & Pan, Y. (2015). Development of a Wireless and Passive SAW-Based Chemical Sensor for Organophosphorous Compound Detection. Sensors, 15(12), 30187-30198. https://doi.org/10.3390/s151229793