Power Approaches for Implantable Medical Devices
Abstract
:1. Introduction
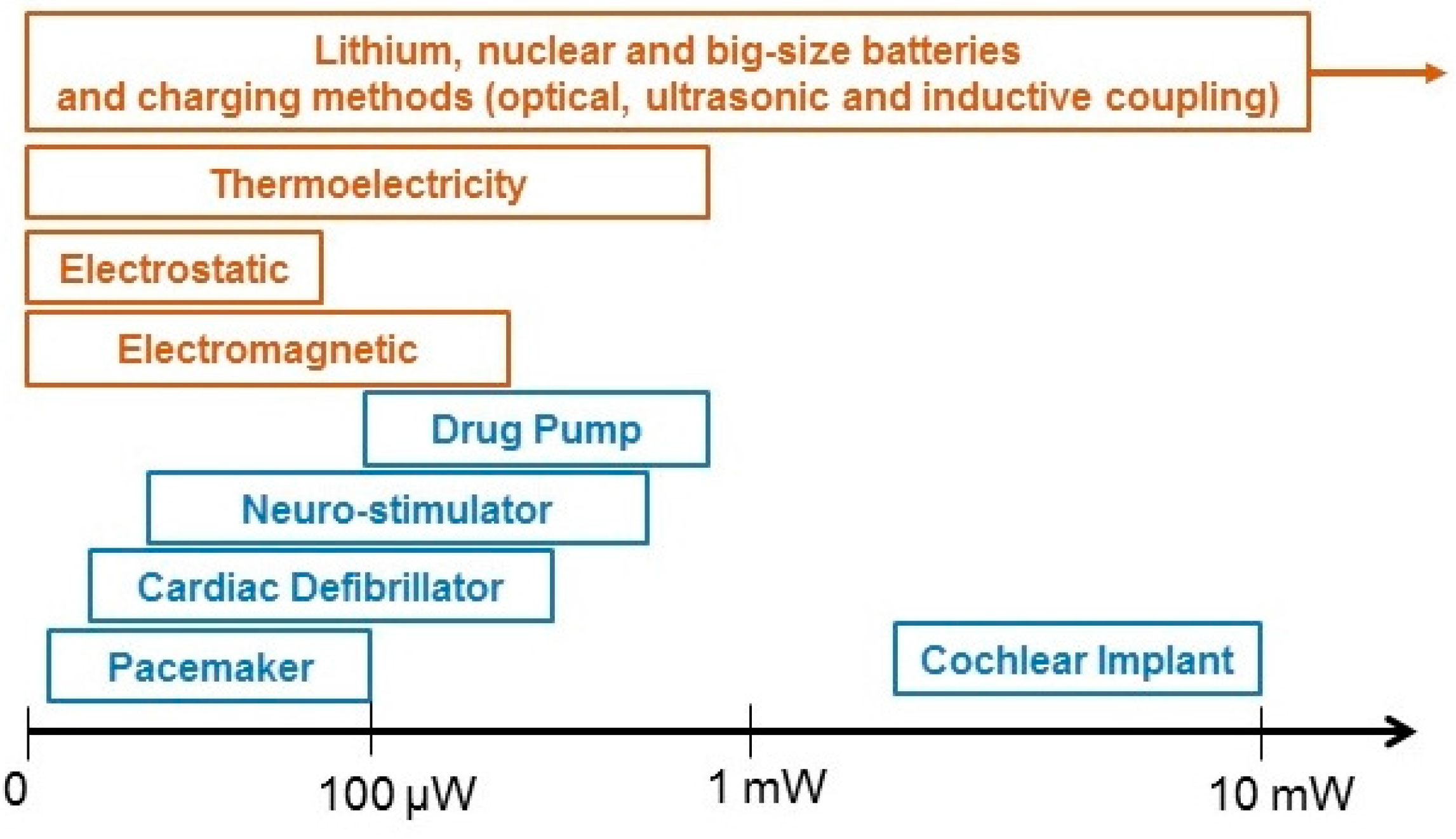

2. Methods to Power IMDs
2.1. Independent Systems
2.1.1. Lithium Batteries
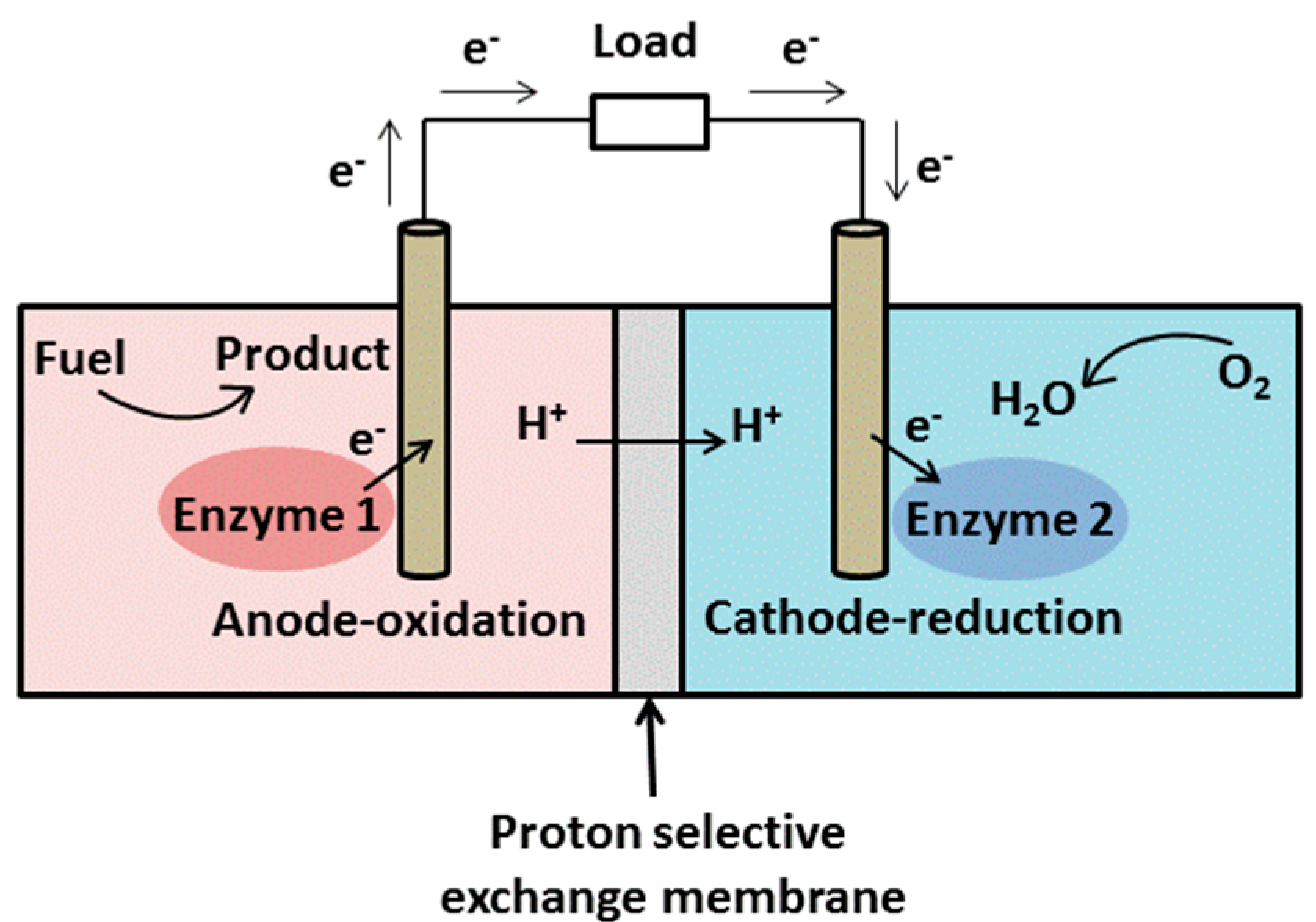
2.1.2. Bio-Fuel Cells
2.1.3. Nuclear Batteries
2.1.4. Thermoelectricity
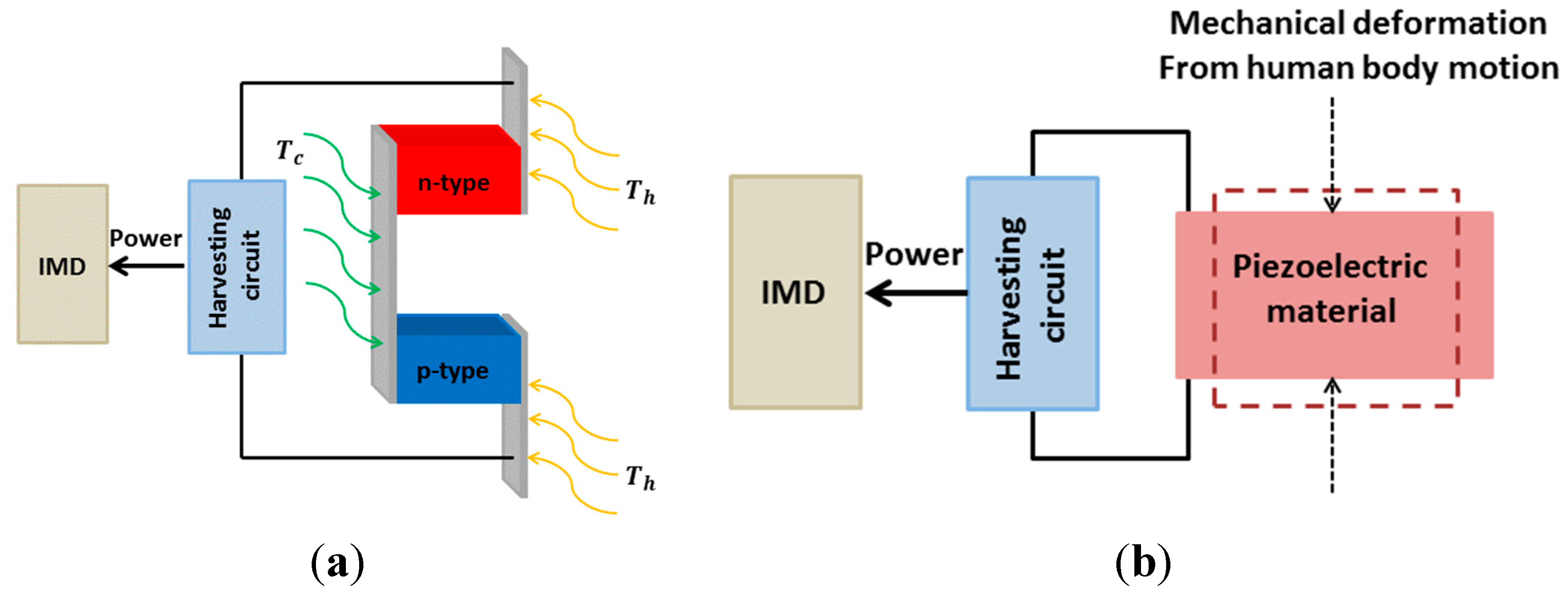
2.1.5. Piezoelectricity
2.1.6. Electrostatic Generators
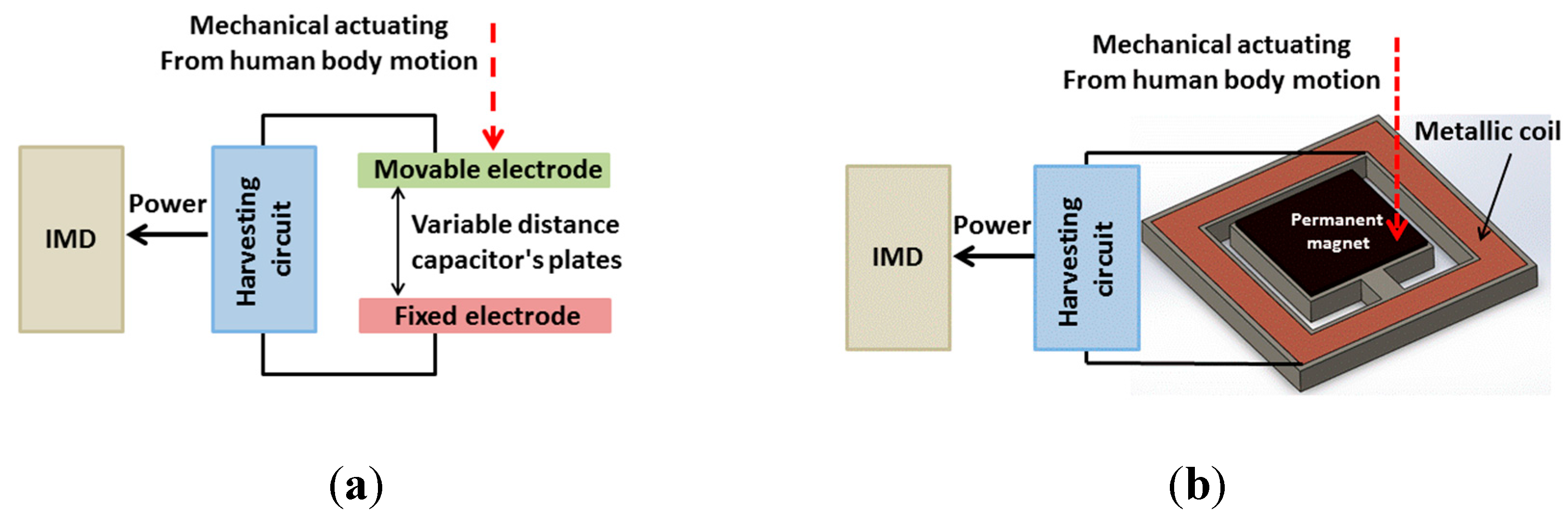
2.1.7. Electromagnetic Generators
2.2. Systems with an External Unit
2.2.1. Optical Charging
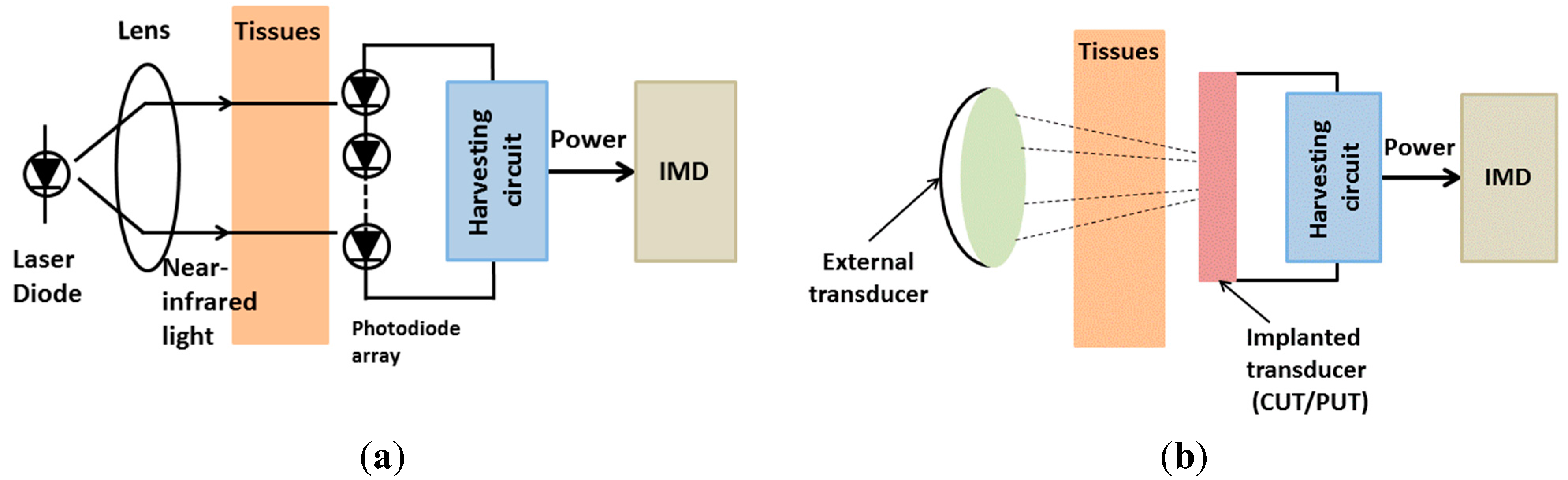
2.2.2. Ultrasonic Transducer
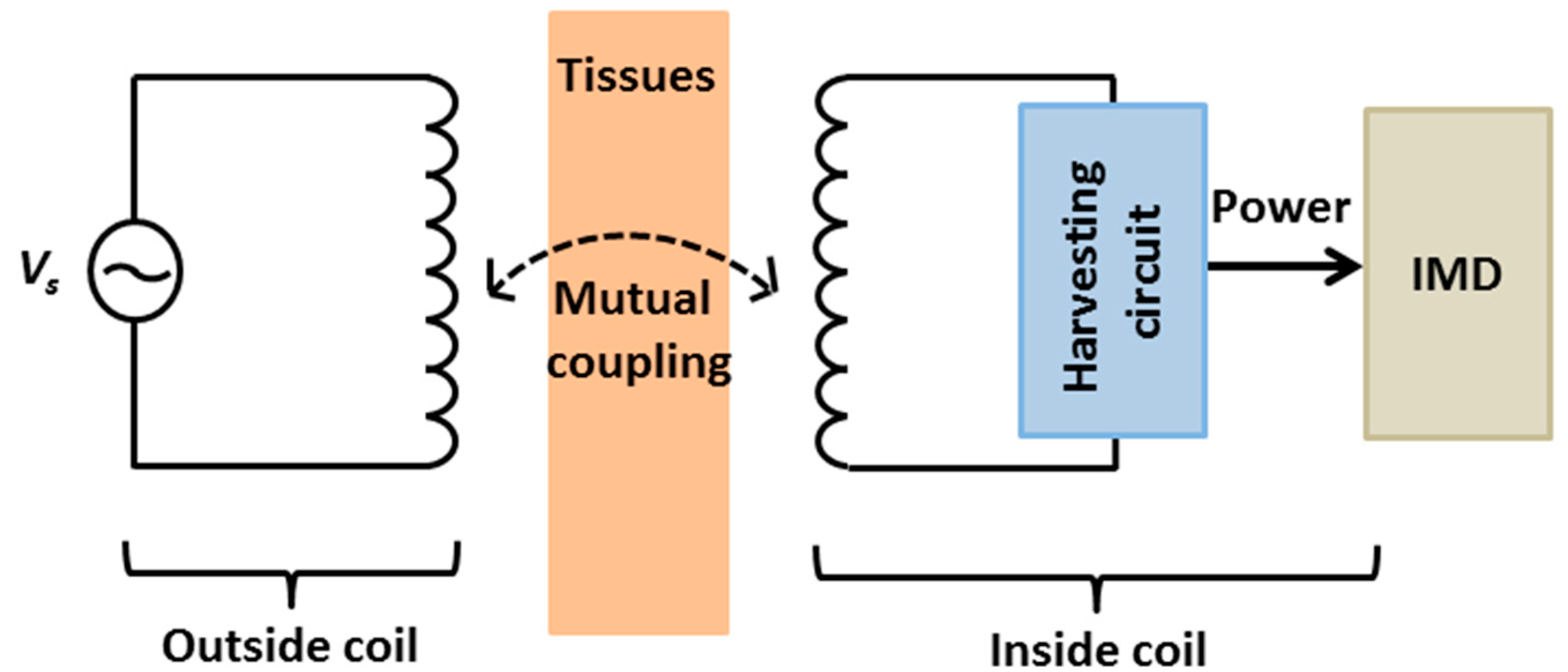
2.2.3. Inductive Coupling
3. Discussion
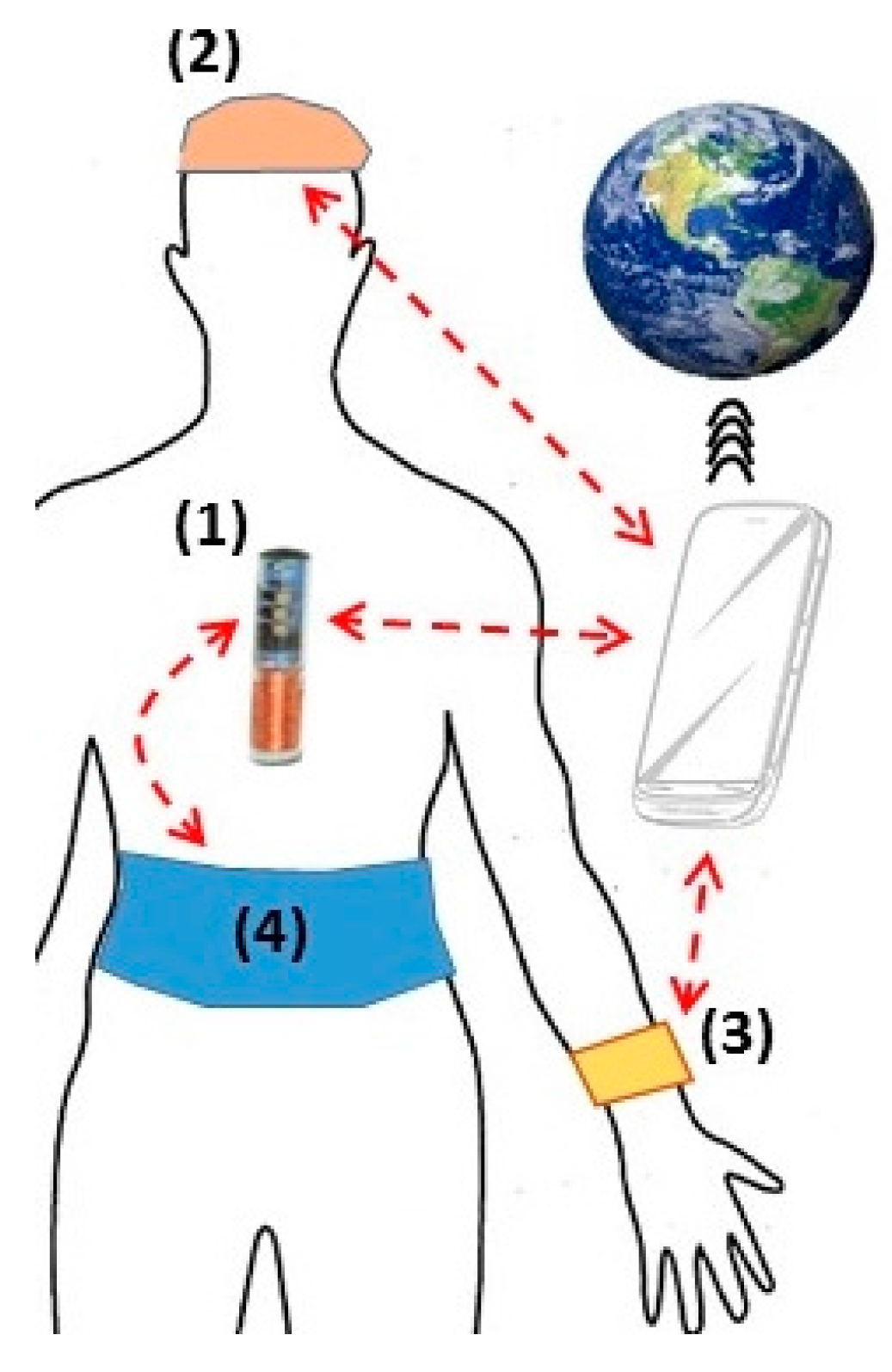
3.1. Implantable Medical Devices: Roles and Future Expectations
3.2. IMDs Power Source Approaches: Advantages and Limitations
| Energy Harvesting Method | Approaches | Generated Power | References | Advantages | Disadvantages |
|---|---|---|---|---|---|
| Independent system | Lithium batteries | 210 W·h/kg | [27] | Compatibility with flexible electronic | Size |
| 300 W·h/kg | [76] | Toxicity | |||
| Bio-fuel cells | 2.4 μW | [83] | Recycle materials | Lifetime | |
| Biocompatibility with human body | Low output power | ||||
| Nuclear batteries | 50 μW | [27] | Longer service life (>15 years) | Radioactive danger | |
| Stable output energy | Expensive | ||||
| Thermoelectricity | 5.8 µW | [91] | Unlimited lifetime | Low output power | |
| 1 µW | [92] | ||||
| 180 μW/cm2 | [93] | ||||
| Piezoelectricity | 2.1–69.8 W | [98] | High output power | Limited implantable locations | |
| 0.33 μW | [104] | No additional voltage source | Biocompatibility issues | ||
| 1 W | [46] | ||||
| Electrostatic | 36 μW | [54,109] | High output power | Additional voltage source | |
| 58 μW | [54,109] | High output impedance | |||
| 80 µW | [108] | ||||
| Electromagnetic | 40–200 μW | [112,113] | Unlimited implantable locations | Complexity in fabrication technologies | |
| 1.1 mW | [115] | ||||
| 400 μW | [116] | ||||
| Systems with external unit | Optical charging | 22 mW/cm | [119] | High output power | Large dimension |
| Ultrasonic transducer | 1.5 mW/cm2 | [124,126] | Data transfer | Low output power | |
| May be used for different depths | Side effects | ||||
| Inductive coupling | 19 mW | [134] | High data rate and power transmission | Limited carrier frequency due to tissue absorptions | |
| 150 mW | [20] | ||||
| 50 mW | [135,136] | No batteries needed | Side effects | ||
| 6.15 mW | [141] |
3.3. Inductive Coupling: Possibilities and Challenges
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Price, C.P.; Kricka, L.J. Improving healthcare accessibility through point-of-care technologies. Clin. Chem. 2007, 53, 1665–1675. [Google Scholar] [PubMed]
- Kim, D.-H.; Lu, N.; Ma, R.; Kim, Y.-S.; Kim, R.-H.; Wang, S.; Wu, J.; Won, S.M.; Tao, H.; Islam, A. Epidermal electronics. Science 2011, 333, 838–843. [Google Scholar] [CrossRef] [PubMed]
- Cramer, J.A. Microelectronic systems for monitoring and enhancing patient compliance with medication regimens. Drugs 1995, 49, 321–327. [Google Scholar] [CrossRef] [PubMed]
- Sia, S.K.; Kricka, L.J. Microfluidics and point-of-care testing. Lab Chip 2008, 8, 1982–1983. [Google Scholar] [CrossRef] [PubMed]
- Cao, H.; Landge, V.; Tata, U.; Seo, Y.-S.; Rao, S.; Tang, S.-J.; Tibbals, H.; Spechler, S.; Chiao, J. An implantable, batteryless, and wireless capsule with integrated impedance and PH sensors for gastroesophageal reflux monitoring. IEEE Trans. Biomed. Eng. 2012, 59, 3131–3139. [Google Scholar] [PubMed]
- Cao, H.; Rao, S.; Tang, S.-J.; Tibbals, H.F.; Spechler, S.; Chiao, J.-C. Batteryless implantable dual-sensor capsule for esophageal reflux monitoring. Gastrointest. Endos. 2013, 77, 649–653. [Google Scholar] [CrossRef] [PubMed]
- Cao, H.; Yu, F.; Zhao, Y.; Scianmarello, N.; Lee, J.; Dai, W.; Jen, N.; Beebe, T.; Li, R.; Ebrahimi, R. Stretchable electrochemical impedance sensors for intravascular detection of lipid-rich lesions in new zealand white rabbits. Biosens. Bioelectron. 2014, 54, 610–616. [Google Scholar] [CrossRef] [PubMed]
- Webster, J.G. Design of Cardiac Pacemakers; IEEE Press: Piscataway, NJ, USA, 1995. [Google Scholar]
- Wilson, B.S.; Finley, C.C.; Lawson, D.T.; Wolford, R.D.; Eddington, D.K.; Rabinowitz, W.M. Better speech recognition with cochlear implants. Nature 1991, 352, 236–238. [Google Scholar] [CrossRef] [PubMed]
- Breit, S.; Schulz, J.B.; Benabid, A.-L. Deep brain stimulation. Cell Tissue Res. 2004, 318, 275–288. [Google Scholar] [CrossRef] [PubMed]
- Zeng, F.-G. Trends in cochlear implants. Trends Amplif. 2004, 8, 1–34. [Google Scholar] [CrossRef] [PubMed]
- Cao, H.; Li, A.-L.; Nguyen, C.M.; Peng, Y.-B.; Chiao, J.-C. An integrated flexible implantable micro-probe for sensing neurotransmitters. IEEE Sens. J. 2012, 12, 1618–1624. [Google Scholar] [CrossRef]
- Kringelbach, M.L.; Owen, S.L.; Aziz, T.Z. Deep-brain stimulation. 2007, 8, 623–635. [Google Scholar] [CrossRef] [PubMed]
- Kern, D.S.; Kumar, R. Deep brain stimulation. Neurologist 2007, 13, 237–252. [Google Scholar] [CrossRef] [PubMed]
- Sousa, J.E.; Costa, M.A.; Abizaid, A.; Abizaid, A.S.; Feres, F.; Pinto, I.M.; Seixas, A.C.; Staico, R.; Mattos, L.A.; Sousa, A.G. Lack of neointimal proliferation after implantation of sirolimus-coated stents in human coronary arteries a quantitative coronary angiography and three-dimensional intravascular ultrasound study. Circulation 2001, 103, 192–195. [Google Scholar] [CrossRef] [PubMed]
- Bloomfield, D.M.; Steinman, R.C.; Namerow, P.B.; Parides, M.; Davidenko, J.; Kaufman, E.S.; Shinn, T.; Curtis, A.; Fontaine, J.; Holmes, D. Microvolt t-wave alternans distinguishes between patients likely and patients not likely to benefit from implanted cardiac defibrillator therapy a solution to the multicenter automatic defibrillator implantation trial (MADIT) II conundrum. Circulation 2004, 110, 1885–1889. [Google Scholar] [CrossRef] [PubMed]
- Wood, M.A.; Ellenbogen, K.A. Cardiac pacemakers from the patient’s perspective. Circulation 2002, 105, 2136–2138. [Google Scholar] [CrossRef] [PubMed]
- Mond, H.G.; Proclemer, A. The 11th world survey of cardiac pacing and implantable cardioverter—Defibrillators: Calendar year 2009—A world society of arrhythmia’s project. Pacing Clin. Electrophysiol. 2011, 34, 1013–1027. [Google Scholar] [CrossRef] [PubMed]
- Antonioli, F.B.; Consiglio, F.; Grassi, G.; Lebrun, R.; Zanardi, F. Stimulatore cardiac impiantabile con nuova a stato solido al litio. Minerva 1973, 64, 2298–2305. [Google Scholar]
- Lenaerts, B.; Puers, R. An inductive power link for a wireless endoscope. Biosens. Bioelectron. 2007, 22, 1390–1395. [Google Scholar] [CrossRef] [PubMed]
- Silay, K.M.; Dondi, D.; Larcher, L.; Declercq, M.; Benini, L.; Leblebici, Y.; Dehollain, C. Load Optimization of an Inductive Power Link for Remote Powering of Biomedical Implants. In Proceedings of the IEEE International Symposium on Circuits and Systems, Taipei, Taiwan, 24–27 May 2009; pp. 533–536.
- De Donaldson, N.N.; Perkins, T. Analysis of resonant coupled coils in the design of radio frequency transcutaneous links. Med. Biol. Eng. Comput. 1983, 21, 612–627. [Google Scholar] [CrossRef]
- RamRakhyani, A.K.; Mirabbasi, S.; Chiao, M. Design and optimization of resonance-based efficient wireless power delivery systems for biomedical implants. IEEE Trans.Biomed. Circuits Syst. 2011, 5, 48–63. [Google Scholar] [CrossRef] [PubMed]
- Holmes, C.F. The role of lithium batteries in modern health care. J. Power Sour. 2001, 97, 739–741. [Google Scholar] [CrossRef]
- Holmes, C.F. The bourner lecture: Electrochemical power sources—An important contributor to modern health care. J. Power Sour. 1997, 65. [Google Scholar] [CrossRef]
- Nathan, M. Microbattery technologies for miniaturized implantable medical devices. Curr. Pharm. Biotechnol. 2010, 11, 404–410. [Google Scholar] [CrossRef] [PubMed]
- Wei, X.; Liu, J. Power sources and electrical recharging strategies for implantable medical devices. Front. Energy Power Eng. China 2008, 2, 1–13. [Google Scholar] [CrossRef]
- Schmidt, C.L.; Skarstad, P.M. The future of lithium and lithium-ion batteries in implantable medical devices. J. Power Sour. 2001, 97, 742–746. [Google Scholar] [CrossRef]
- Bock, D.C.; Marschilok, A.C.; Takeuchi, K.J.; Takeuchi, E.S. Batteries used to power implantable biomedical devices. Electrochim. Acta 2012, 84, 155–164. [Google Scholar] [CrossRef] [PubMed]
- Bullen, R.A.; Arnot, T.; Lakeman, J.; Walsh, F. Biofuel cells and their development. Biosens. Bioelectron. 2006, 21, 2015–2045. [Google Scholar] [CrossRef] [PubMed]
- Dong, K.; Jia, B.; Yu, C.; Dong, W.; Du, F.; Liu, H. Microbial fuel cell as power supply for implantable medical devices: A novel configuration design for simulating colonic environment. Biosens. Bioelectron. 2013, 41, 916–919. [Google Scholar] [CrossRef] [PubMed]
- Drews, J.; Fehrmann, G.; Staub, R.; Wolf, R. Primary batteries for implantable pacemakers and defibrillators. J. Power Sour. 2001, 97, 747–749. [Google Scholar] [CrossRef]
- Mallela, V.S.; Ilankumaran, V.; Rao, N.S. Trends in cardiac pacemaker batteries. Indian Pacing Electrophysiol. J. 2004, 4, 201. [Google Scholar] [PubMed]
- Fernandez, C.; Garcia, O.; Prieto, R.; Cobos, J.; Gabriels, S.; van der Borght, G. Design Issues of a Core-Less Transformer for a Contact-Less Application. In Proceedings of the Seventeenth Annual IEEE Applied Power Electronics Conference and Exposition, Dallas, TX, USA, 10–14 March 2002; pp. 339–345.
- Nguyen, M.Q.; Hughes, Z.; Woods, P.; Seo, Y.-S.; Rao, S.; Chiao, J.-C. Field distribution models of spiral coil for misalignment analysis in wireless power transfer systems. IEEE Trans. Microw. Theory Tech. 2014, 62, 920–930. [Google Scholar] [CrossRef]
- Parsonnet, V.; Cheema, A. The nature and frequency of postimplant surgical interventions. Pacing Clin. Electrophysiol. 2003, 26, 2308–2312. [Google Scholar] [CrossRef] [PubMed]
- Ko, W.H.; Hynecek, J. Implant evaluation of a nuclear power source-betacel battery. IEEE Trans. Biomed. Eng. 1974, 3, 238–241. [Google Scholar] [CrossRef] [PubMed]
- Parsonnet, V.; Villanueva, A.; Driller, J.; Bernstein, A.D. Corrosion of pacemaker electrodes. Pacing Clin. Electrophysiol. 1981, 4, 289–295. [Google Scholar] [CrossRef] [PubMed]
- Halliwell, C.M.; Simon, E.; Toh, C.-S.; Cass, A.E.; Bartlett, P.N. The design of dehydrogenase enzymes for use in a biofuel cell: The role of genetically introduced peptide tags in enzyme immobilization on electrodes. Bioelectrochemistry 2002, 55, 21–23. [Google Scholar] [CrossRef]
- Simon, E.; Halliwell, C.M.; Toh, C.S.; Cass, A.E.; Bartlett, P.N. Immobilisation of enzymes on poly (aniline)–poly (anion) composite films. Preparation of bioanodes for biofuel cell applications. Bioelectrochemistry 2002, 55, 13–15. [Google Scholar] [CrossRef]
- Meral, I.; Mert, H.; Mert, N.; Deger, Y.; Yoruk, I.; Yetkin, A.; Keskin, S. Effects of 900-MHz electromagnetic field emitted from cellular phone on brain oxidative stress and some vitamin levels of guinea pigs. Brain Res. 2007, 1169, 120–124. [Google Scholar] [CrossRef] [PubMed]
- Priya, S.; Inman, D.J. Energy Harvesting Technologies; Springer: Berlin, Germany, 2009. [Google Scholar]
- Vullers, R.; van Schaijk, R.; Doms, I.; van Hoof, C.; Mertens, R. Micropower energy harvesting. Solid State Electron. 2009, 53, 684–693. [Google Scholar] [CrossRef]
- Harb, A. Energy harvesting: State-of-the-art. Renew. Energy 2011, 36, 2641–2654. [Google Scholar] [CrossRef]
- Glynne-Jones, P.; Beeby, S.; White, N. Towards a piezoelectric vibration-powered microgenerator. IEE Proc. Sci. Meas. Technol. 2001, 148, 68–72. [Google Scholar] [CrossRef]
- Kymissis, J.; Kendall, C.; Paradiso, J.; Gershenfeld, N. Parasitic Power Parvesting in Phoes, Wearable Computers. In Proceedings of the IEEE Second International Symposium on Digest of Papers, Pittsburg, PA, USA, 19–20 October 1998; pp. 132–139.
- Kim, H.S.; Kim, J.-H.; Kim, J. A review of piezoelectric energy harvesting based on vibration. Int. J. Precis. Eng. Manuf. 2011, 12, 1129–1141. [Google Scholar] [CrossRef]
- Almouahed, S.; Gouriou, M.; Hamitouche, C.; Stindel, E.; Roux, C. Self-Powered Instrumented Knee Implant for Early Detection of Postoperative Complications. In Proceedings of the 2010 Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Piscataway, NJ, USA, 31 September 2010; pp. 5121–5124.
- Lahuec, C.; Almouahed, S.; Arzel, M.; Gupta, D.; Hamitouche, C.; Jezequel, M.; Stindel, E.; Roux, C. A self-powered telemetry system to estimate the postoperative instability of a knee implant. IEEE Trans. Biomed. Eng. 2011, 58, 822–825. [Google Scholar] [CrossRef] [PubMed]
- Almouahed, S.; Gouriou, M.; Hamitouche, C.; Stindel, E.; Roux, C. The use of piezoceramics as electrical energy harvesters within instrumented knee implant during walking. IEEE ASME Trans. Mechatron. 2011, 16, 799–807. [Google Scholar] [CrossRef]
- Beeby, S.P.; Tudor, M.J.; White, N. Energy harvesting vibration sources for microsystems applications. Meas. Sci. Technol. 2006, 17, 175–195. [Google Scholar] [CrossRef]
- Miao, P.; Holmes, A.; Yeatman, E.; Green, T.; Mitcheson, P. Micro-Machined Variable Capacitors for Power Generation. In Proceedings of the Conference Series-Institute of Physics, Edinburgh, Scotland, 23–27 March 2003; pp. 53–58.
- Miyazaki, M.; Tanaka, H.; Nagano, T.; Ohkubo, N.; Kawahara, T. Electric-energy generation through variable-capacitive resonator for power-free LSI. IEICE Trans. Electron. 2004, 87, 549–555. [Google Scholar]
- Tashiro, R.; Kabei, N.; Katayama, K.; Ishizuka, Y.; Tsuboi, F.; Tsuchiya, K. Development of an electrostatic generator that harnesses the motion of a living body. Use of a resonant phenomenon. JSME Int. J. Ser. C 2000, 43, 916–922. [Google Scholar] [CrossRef]
- Murakawa, K.; Kobayashi, M.; Nakamura, O.; Kawata, S. A wireless near-infrared energy system for medical implants. IEEE Eng. Med. Biol. Mag. 1999, 18, 70–72. [Google Scholar] [CrossRef] [PubMed]
- Penner, A. Acoustically Powered Implantable Stimulating Device. U.S. Patent US8577460 B2, 5 November 2013. [Google Scholar]
- Willis, N.P.; Brisken, A.F.; Cowan, M.W.; Pare, M.; Fowler, R.; Brennan, J. Optimizing Energy Transmission in a Leadless Tissue Stimulation System. U.S. Patent US8718773 B2, 6 May 2014. [Google Scholar]
- Tran, B.C.; Mi, B.; Harguth, R.S. Systems and Methods for Controlling Wireless Signal Transfers between Ultrasound-Enabled Medical Devices. U.S. Patent WO2009102640 A1, 5 February 2013. [Google Scholar]
- Ozeri, S.; Shmilovitz, D.; Singer, S.; Wang, C.-C. Ultrasonic transcutaneous energy transfer using a continuous wave 650 kHz gaussian shaded transmitter. Ultrasonics 2010, 50, 666–674. [Google Scholar] [CrossRef] [PubMed]
- Ozeri, S.; Shmilovitz, D. Ultrasonic transcutaneous energy transfer for powering implanted devices. Ultrasonics 2010, 50, 556–566. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.; Yang, Z.; Hoang, L.; Weiland, J.; Humayun, M.; Liu, W. An integrated 256-channel epiretinal prosthesis. IEEE J. Solid State Circuits 2010, 45, 1946–1956. [Google Scholar] [CrossRef]
- Scrosati, B. History of lithium batteries. J. Solid State Electrochem. 2011, 15, 1623–1630. [Google Scholar] [CrossRef]
- Holmes, C. The lithium/iodine-polyvinylpyridine pacemaker battery-35 years of successful clinical use. ECS Trans. 2007, 6, 1–7. [Google Scholar]
- Linden, D.; Reddy, T.B. Handbook of Batteries, 3rd ed.; McGraw-Hill: New York, NY, USA, 2002. [Google Scholar]
- Nagao, M.; Pitteloud, C.; Kamiyama, T.; Otomo, T.; Itoh, K.; Fukunaga, T.; Kanno, R. Further understanding of reaction processes in electrolytic manganese dioxide electrodes for lithium cells. J. Electrochem. Soc. 2005, 152, E230–E237. [Google Scholar] [CrossRef]
- Johnson, C.S. Development and utility of manganese oxides as cathodes in lithium batteries. J. Power Sour. 2007, 165, 559–565. [Google Scholar] [CrossRef]
- Greatbatch, W.; Holmes, C.; Takeuchi, E.; Ebel, S. Lithium/carbon monofluoride (Li/CFx): A new pacemaker battery. Pacing Clin. Electrophysiol. 1996, 19, 1836–1840. [Google Scholar] [CrossRef] [PubMed]
- Amatucci, G.G.; Pereira, N. Fluoride based electrode materials for advanced energy storage devices. J. Fluor. Chem. 2007, 128, 243–262. [Google Scholar] [CrossRef]
- Davis, S.; Takeuchi, E.S.; Tiedemann, W.; Newman, J. Simulation of pulse discharge of the Li-CFx x system. J. Electrochem. Soc. 2008, 155, A24–A28. [Google Scholar] [CrossRef]
- Zhang, S.S.; Foster, D.; Read, J. Carbothermal treatment for the improved discharge performance of primary Li/CFx battery. J. Power Sour. 2009, 191, 648–652. [Google Scholar] [CrossRef]
- Takeuchi, E.S.; Piliero, P. Lithium/silver vanadium oxide batteries with various silver to vanadium ratios. J. Power Sour. 1987, 21, 133–141. [Google Scholar] [CrossRef]
- Kim, Y.J.; Lee, C.-Y.; Marschilok, A.C.; Takeuchi, K.J.; Takeuchi, E.S. AgxVOPO4: A demonstration of the dependence of battery-related electrochemical properties of silver vanadium phosphorous oxides on Ag/V ratios. J. Power Sour. 2011, 196, 3325–3330. [Google Scholar] [CrossRef] [PubMed]
- Crespi, A.; Schmidt, C.; Norton, J.; Chen, K.; Skarstad, P. Modeling and characterization of the resistance of lithium/SVO batteries for implantable cardioverter defibrillators. J. Electrochem. Soc. 2001, 148, A30–A37. [Google Scholar] [CrossRef]
- Chen, K.; Merritt, D.R.; Howard, W.G.; Schmidt, C.L.; Skarstad, P.M. Hybrid cathode lithium batteries for implantable medical applications. J. Power Sour. 2006, 162, 837–840. [Google Scholar] [CrossRef]
- Gomadam, P.M.; Merritt, D.R.; Scott, E.R.; Schmidt, C.L.; Skarstad, P.M.; Weidner, J.W. Modeling Li/CFx-SVO Hybrid-Cathode Batteries. J. Electrochem. Soc. 2007, 154, A1058–A1064. [Google Scholar] [CrossRef]
- Whittingham, M.S. Lithium batteries and cathode materials. Chem. Rev. 2004, 104, 4271–4302. [Google Scholar] [CrossRef] [PubMed]
- Rahimi-Eichi, H.; Baronti, F.; Chow, M.-Y. Online adaptive parameter identification and state-of-charge coestimation for lithium-polymer battery cells. IEEE Trans. Ind. Electron. 2014, 61, 2053–2061. [Google Scholar] [CrossRef]
- Gwon, H.; Hong, J.; Kim, H.; Seo, D.-H.; Jeon, S.; Kang, K. Recent progress on flexible lithium rechargeable batteries. Energy Environ. Sci. 2014, 7, 538–551. [Google Scholar] [CrossRef]
- Grove, W.R. Xxiv. On voltaic series and the combination of gases by platinum. Lond. Edinb. Philos. Mag. J. Sci. 1839, 14, 127–130. [Google Scholar]
- Potter, M.C. Electrical Effects Accompanying the Decomposition of Organic Compounds. In Proceedings of the Royal Society of London. Series B, Containing Papers of a Biological Character, London, UK, 14 September 1911; pp. 260–276.
- Cohen, B. The bacterial culture as an electrical half-cell. J. Bacteriol. 1931, 21, 18–19. [Google Scholar]
- Yahiro, A.; Lee, S.; Kimble, D. Bioelectrochemistry: I. Enzyme Utilizing Bio-Fuel Cell Studies. Biochim. Biophys. Acta Spec. Sect. Biophys. Subj. 1964, 88, 375–383. [Google Scholar] [CrossRef]
- Mano, N.; Mao, F.; Heller, A. Characteristics of a miniature compartment-less glucose-O2 biofuel cell and its operation in a living plant. J. Am. Chem. Soc. 2003, 125, 6588–6594. [Google Scholar] [CrossRef] [PubMed]
- Cadei, A.; Dionisi, A.; Sardini, E.; Serpelloni, M. Kinetic and thermal energy harvesters for implantable medical devices and biomedical autonomous sensors. Meas. Sci. Technol. 2014, 25. [Google Scholar] [CrossRef]
- Huesgen, T.; Woias, P.; Kockmann, N. Design and fabrication of mems thermoelectric generators with high temperature efficiency. Sens. Actuators A Phys. 2008, 145, 423–429. [Google Scholar] [CrossRef]
- Snyder, G.J. Thermoelectric energy harvesting. In Energy Harvesting Technologies; Springer: Berlin, Germany, 2009; pp. 325–336. [Google Scholar]
- Torfs, T.; Leonov, V.; Vullers, R. Pulse oximeter fully powered by human body heat. Sens. Transducers J. 2007, 80, 1230–1238. [Google Scholar]
- Yadav, A.; Pipe, K.; Shtein, M. Fiber-based flexible thermoelectric power generator. J. Power Sour. 2008, 175, 909–913. [Google Scholar] [CrossRef]
- Rowe, D.M. Crc Handbook of Thermoelectrics; CRC Press: Boca Raton, FL, USA, 1995. [Google Scholar]
- Xie, J.; Lee, C.; Feng, H. Design, fabrication, and characterization of cmos mems-based thermoelectric power generators. Microelectromech. Syst. J. 2010, 19, 317–324. [Google Scholar] [CrossRef]
- Stark, I.; Stordeur, M. New Micro Thermoelectric Devices Based on Bismuth Telluride-Type Thin Solid Films. In Proceedings of the Eighteenth International Conference on Thermoelectrics, Piscataway, NJ, USA, 29 August–2 September 1999; pp. 465–472.
- Strasser, M.; Aigner, R.; Lauterbach, C.; Sturm, T.; Franosch, M.; Wachutka, G. Micromachined cmos thermoelectric generators as on-chip power supply. Sens. Actuators A Phys. 2004, 114, 362–370. [Google Scholar] [CrossRef]
- Settaluri, K.T.; Lo, H.; Ram, R.J. Thin thermoelectric generator system for body energy harvesting. J. Electron. Mater. 2012, 41, 984–988. [Google Scholar] [CrossRef]
- Manbachi, A.; Cobbold, R.S. Development and application of piezoelectric materials for ultrasound generation and detection. Ultrasound 2011, 19, 187–196. [Google Scholar] [CrossRef]
- Anton, S.R.; Sodano, H.A. A review of power harvesting using piezoelectric materials (2003–2006). Smart Mater. Struct. 2007, 16, R1–R21. [Google Scholar] [CrossRef]
- Sodano, H.A.; Inman, D.J.; Park, G. A review of power harvesting from vibration using piezoelectric materials. Shock Vib. Dig. 2004, 36, 197–206. [Google Scholar] [CrossRef]
- González, J.L.; Rubio, A.; Moll, F. Human powered piezoelectric batteries to supply power to wearable electronic devices. Int. J. Soc. Mater. Eng. Resour. 2002, 10, 34–40. [Google Scholar] [CrossRef]
- Niu, P.; Chapman, P.; Riemer, R.; Zhang, X. Evaluation of Motions and Actuation Methods for Biomechanical Energy Harvesting. In Proceedings of the IEEE 35th Annual Power Electronics Specialists Conference, Aachen, Germany, 20–25 June 2004; pp. 2100–2106.
- Renaud, M.; Sterken, T.; Fiorini, P.; Puers, R.; Baert, K.; van Hoof, C. Scavenging Energy from Human Body: Design of a Piezoelectric Transducer. In Proceedings of the 13th International Conference on Solid-State Sensors, Actuators and Microsystems, Digest of Technical Papers, Seoul, Korea, 5–9 June 2005; pp. 784–787.
- Chen, H.; Liu, M.; Jia, C.; Wang, Z. Power harvesting using PZT ceramics embedded in orthopedic implants. IEEE Trans. Ultrason. Ferroelectr. Freq. Control 2009, 56, 2010–2014. [Google Scholar] [CrossRef] [PubMed]
- Platt, S.R.; Farritor, S.; Garvin, K.; Haider, H. The use of piezoelectric ceramics for electric power generation within orthopedic implants. IEEE ASME Trans. Mechatron. 2005, 10, 455–461. [Google Scholar] [CrossRef]
- Mateu, L.; Fonellosa, F.; Moll, F. Electrical Characterization of a Piezoelectric Film-Based Power Generator for Autonomous Wearable Devices. In Proceedings of the Conference on Design of Circuits and Integrated Systems, Albacete, Spain, 19–21 November 2013; pp. 677–682.
- Mateu, L.; Moll, F. Optimum piezoelectric bending beam structures for energy harvesting using shoe inserts. J. Intell. Mater. Syst. Struct. 2005, 16, 835–845. [Google Scholar] [CrossRef]
- Sohn, J.; Choi, S.B.; Lee, D. An investigation on piezoelectric energy harvesting for mems power sources. Proc. Inst. Mech. Eng. Part C J.Mech. Eng. Sci. 2005, 219, 429–436. [Google Scholar] [CrossRef]
- Cheng, D.K. Field and Wave Electromagnetics; Addison-Wesley: New York, NY, USA, 1989. [Google Scholar]
- Tsai, N.-C.; Sue, C.-Y. Review of mems-based drug delivery and dosing systems. Sens. Actuators A Phys. 2007, 134, 555–564. [Google Scholar] [CrossRef]
- Roundy, S.J. Energy Scavenging for Wireless Sensor Nodes with a Focus on Vibration to Electricity Conversion; University of California: Berkeley, CA, USA, 2003. [Google Scholar]
- Miao, P.; Mitcheson, P.; Holmes, A.; Yeatman, E.; Green, T.; Stark, B. Mems inertial power generators for biomedical applications. Microsyst. Technol. 2006, 12, 1079–1083. [Google Scholar] [CrossRef]
- Tashiro, R.; Kabei, N.; Katayama, K.; Tsuboi, E.; Tsuchiya, K. Development of an electrostatic generator for a cardiac pacemaker that harnesses the ventricular wall motion. J. Artif. Org. 2002, 5, 0239–0245. [Google Scholar] [CrossRef]
- Paulo, J.; Gaspar, P. Review and Future Trend of Energy Harvesting Methods for Portable Medical Devices. In Proceedings of the World Congress on Engineering, London, UK, 30 June–2 July 2010; pp. 168–196.
- Williams, C.; Yates, R.B. Analysis of a micro-electric generator for microsystems. Sens. Actuators A Phys. 1996, 52, 8–11. [Google Scholar] [CrossRef]
- Irani, A.; Bianco, M.; Tran, D.; Deyoung, P.D.; Wyld, M.L.R.; Li, T.H. Energy Generating Systems for Implanted Medical Devices. U.S. Patent US20090171404 A1, 2 July 2009. [Google Scholar]
- Goto, H.; Sugiura, T.; Harada, Y.; Kazui, T. Feasibility of using the automatic generating system for quartz watches as a leadless pacemaker power source. Med. Biol. Eng. Comput. 1999, 37, 377–380. [Google Scholar] [CrossRef] [PubMed]
- Roberts, P.; Stanley, G.; Morgan, J.M. Harvesting the energy of cardiac motion to power a pacemaker. Circulation 2008, 118, 679–680. [Google Scholar] [CrossRef] [Green Version]
- Nasiri, A.; Zabalawi, S.A.; Jeutter, D.C. A linear permanent magnet generator for powering implanted electronic devices. IEEE Trans. Power Electron. 2011, 26, 192–199. [Google Scholar] [CrossRef]
- Amirtharajah, R.; Chandrakasan, A.P. Self-powered signal processing using vibration-based power generation. IEEE J. Solid State Circuits 1998, 33, 687–695. [Google Scholar] [CrossRef]
- Parkhouse, L. Photovoltaic Powered Charging Apparatus for Implanted Rechargeable Batteries. U.S. Patent US7003353 B1, 21 February 2006. [Google Scholar]
- Lee, J.B.; Chen, Z.; Allen, M.G.; Rohatgi, A.; Arya, R. A miniaturized high-voltage solar cell array as an electrostatic mems power supply. Microelectromech. Syst. J. 1995, 4, 102–108. [Google Scholar] [CrossRef]
- Goto, K.; Nakagawa, T.; Nakamura, O.; Kawata, S. An implantable power supply with an optically rechargeable lithium battery. IEEE Trans. Biomed. Eng. 2001, 48, 830–833. [Google Scholar] [CrossRef] [PubMed]
- Jr Ackermann, D.M.; Smith, B.; Kilgore, K.L.; Peckham, P.H. Design of a High Speed Transcutaneous Optical Telemetry Link. In Proceedings of the 28th Annual International Conference of the IEEE Engineering in Medicine and Biology Society, New York, NY, USA, 30 August–3 September 2006; pp. 2932–2935.
- Olivo, J.; Carrara, S.; de Micheli, G. Energy harvesting and remote powering for implantable biosensors. IEEE Sens. J. 2011, 11, 1573–1586. [Google Scholar] [CrossRef]
- Banerji, S.; Goh, W.L.; Cheong, J.H.; Je, M. CMUT Ultrasonic Power Link Front-End for Wireless Power Transfer Deep in Body. In Proceedings of the IEEE MTT-S International Microwave Workshop Series on RF and Wireless Technologies for Biomedical and Healthcare Applications (IMWS-BIO), Singapore, 9–11 December 2013; pp. 1–3.
- Christensen, D.B.; Roundy, S. Ultrasonically powered piezoelectric generators for bio-implantable sensors: Plate versus diaphragm. J. Intell. Mater. Syst. Struct. 2015. [Google Scholar] [CrossRef]
- Cochran, G.V.; Kadaba, M.P.; Palmieri, V.R. External ultrasound can generate microampere direct currents in vivo from implanted piezoelectric materials. J. Orthop. Res. 1988, 6, 145–147. [Google Scholar] [CrossRef] [PubMed]
- Seo, D.; Carmena, J.M.; Rabaey, J.M.; Maharbiz, M.M.; Alon, E. Model validation of untethered, ultrasonic neural dust motes for cortical recording. J. Neurosci. Methods 2015, 244, 114–122. [Google Scholar] [CrossRef] [PubMed]
- Phillips, W.; Towe, B.; Larson, P. An Ultrasonically-Driven Piezoelectric Neural Stimulator. In Proceedings of the 25th Annual International Conference of the IEEE Engineering in Medicine and Biology Society, Cancun, Mexico, 17–21 September 2003; pp. 1983–1986.
- Kennedy, J.; Ter Haar, G.; Cranston, D. High intensity focused ultrasound: Surgery of the future? British J. Radiol. 2003, 76, 590–609. [Google Scholar] [CrossRef] [PubMed]
- Tesla, N. Apparatus for Transmitting Electrical Energy. U.S. Patent US1119732 A, 1 December 1914. [Google Scholar]
- Nishimura, T.H.; Eguchi, T.; Hirachi, K.; Maejima, Y.; Kuwana, K.; Saito, M. A Large Air Gap Flat Transformer for a Transcutaneous Energy Transmission System. In Proceedings of the 25th Annual IEEE Power Electronics Specialists Conference, PESC’94 Record, Taipei, Taiwan, 20–25 June 1994; pp. 1323–1329.
- Hui, S.; Zhong, W.; Lee, C. A critical review of recent progress in mid-range wireless power transfer. IEEE Trans.Power Electron. 2013, 29, 4500–4511. [Google Scholar] [CrossRef]
- Lee, H.-M.; Ghovanloo, M. A power-efficient wireless capacitor charging system through an inductive link. IEEE Trans. Circuits Syst. Express Br. 2013, 60, 707–711. [Google Scholar] [CrossRef] [PubMed]
- Neagu, C.; Jansen, H.; Smith, A.; Gardeniers, J.; Elwenspoek, M. Characterization of a planar microcoil for implantable microsystems. Sens. Actuators A Phys. 1997, 62, 599–611. [Google Scholar] [CrossRef]
- Cao, H.; Thakar, S.K.; Fu, T.; Sheth, M.; Oseng, M.L.; Landge, V.; Seo, Y.-S.; Chiao, J.-C. A Wireless Strain Sensor System for Bladder Volume Monitoring. In Proceedings of the IEEE MTT-S International Microwave Symposium Digest (MTT), Baltimore, MD, USA, 5–10 June 2011; pp. 1–4.
- Parramon, J.; Doguet, P.; Marin, D.; Verleyssen, M.; Munoz, R.; Leija, L.; Valderrama, E. Asic-Based Batteryless Implantable Telemetry Microsystem for Recording Purposes. Engineering in Medicine and Biology Society. In Proceedings of the 19th IEEE Annual International Conference, Chicago, IL, USA, 30 October–2 November 1997; pp. 2225–2228.
- Catrysse, M.; Hermans, B.; Puers, R. An inductive power system with integrated bi-directional data-transmission. Sens. Actuators A Phys. 2004, 115, 221–229. [Google Scholar] [CrossRef]
- Ghovanloo, M.; Najafi, K. A wireless implantable multichannel microstimulating system-on-a-chip with modular architecture. IEEE Trans. Neural Syst. Rehabil. Eng. 2007, 15, 449–457. [Google Scholar] [CrossRef] [PubMed]
- Bui, A.L.; Fonarow, G.C. Home monitoring for heart failure management. J. Am. Coll. Cardiol. 2012, 59, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Cao, H.; Tata, U.; Landge, V.; Li, A.-L.; Peng, Y.-B.; Chiao, J.-C. A Wireless Bladder Volume Monitoring System Using a Flexible Capacitance-Based Sensor. In Proceedings of the Topical Conference on Biomedical Wireless Technologies, Networks, and Sensing Systems, Austin, TX, USA, 20–23 January 2013; pp. 34–36.
- Tran, R.T.; Choy, W.M.; Cao, H.; Qattan, I.; Chiao, J.C.; Ip, W.Y.; Yeung, K.W.K.; Yang, J. Fabrication and characterization of biomimetic multichanneled crosslinked-urethane-doped polyester tissue engineered nerve guides. J. Biomed. Mater. Rese. Part A 2014, 102, 2793–2804. [Google Scholar] [CrossRef] [PubMed]
- Kirkland, N.; Birbilis, N.; Staiger, M. Assessing the corrosion of biodegradable magnesium implants: A critical review of current methodologies and their limitations. Acta Biomater. 2012, 8, 925–936. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Bashirullah, R. A wireless power interface for rechargeable battery operated medical implants. IEEE Trans. Circuits Syst. II Express Br. 2007, 54, 912–916. [Google Scholar] [CrossRef]
- Galán, T.; Prieto-Simón, B.; Alvira, M.; Eritja, R.; Götz, G.; Bäuerle, P.; Samitier, J. Label-free electrochemical DNA sensor using “click”-functionalized pedot electrodes. Biosens. Bioelectron. 2015, 74, 751–756. [Google Scholar] [CrossRef] [PubMed]
- Weber, J.E.; Pillai, S.; Ram, M.K.; Kumar, A.; Singh, S.R. Electrochemical impedance-based DNA sensor using a modified single walled carbon nanotube electrode. Mater. Sci. Eng. C 2011, 31, 821–825. [Google Scholar] [CrossRef]
- Thipmanee, O.; Samanman, S.; Sankoh, S.; Numnuam, A.; Limbut, W.; Kanatharana, P.; Vilaivan, T.; Thavarungkul, P. Label-free capacitive DNA sensor using immobilized pyrrolidinyl pna probe: Effect of the length and terminating head group of the blocking thiols. Biosens. Bioelectron. 2012, 38, 430–435. [Google Scholar] [CrossRef] [PubMed]
- Cao, H.; Zhao, Y.; Kouki, A.B.; Tai, Y.-C.; Hsiai, T.K. A wireless ecg recording system for small animal models of heart regeneration. In Proceedings of the IEEE MTT-S International Microwave Symposium (IMS), Phoenix, AZ, USA, 17–22 May 2015; pp. 1–3.
- Cao, H.; Yu, F.; Zhao, Y.; Zhang, X.; Tai, J.; Lee, J.; Darehzereshki, A.; Bersohn, M.; Lien, C.-L.; Chi, N.C. Wearable multi-channel microelectrode membranes for elucidating electrophysiological phenotypes of injured myocardium. Integr. Biol. 2014, 6, 789–795. [Google Scholar] [CrossRef] [PubMed]
- Zierhofer, C.M.; Hochmair, E.S. Geometric approach for coupling enhancement of magnetically coupled coils. IEEE Trans. Biomed. Eng. 1996, 43, 708–714. [Google Scholar] [CrossRef] [PubMed]
- Zierhofer, C.M.; Hochmair, E.S. High-efficiency coupling-insensitive transcutaneous power and data transmission via an inductive link. IEEE Trans. Biomed. Eng. 1990, 37, 716–722. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Liu, W.; Sivaprakasam, M.; Kendir, G.A. Design and analysis of an adaptive transcutaneous power telemetry for biomedical implants. IEEE Trans. Circuits Syst. I Regul. Pap. 2005, 52, 2109–2117. [Google Scholar] [CrossRef]
- Baarman, D.W. Adaptive Inductive Power Supply. U.S. Patent US7522878 B2, 21 April 2009. [Google Scholar]
- Sample, A.P.; Meyer, D.; Smith, J.R. Analysis, experimental results, and range adaptation of magnetically coupled resonators for wireless power transfer. IEEE Trans. Ind. Electron. 2011, 58, 544–554. [Google Scholar] [CrossRef]
- Fotopoulou, K.; Flynn, B.W. Wireless power transfer in loosely coupled links: Coil misalignment model. IEEE Trans. Magn. 2011, 47, 416–430. [Google Scholar] [CrossRef]
- Wu, W.; Fang, Q. Design and Simulation of Printed Spiral Coil Used in Wireless Power Transmission Systems for Implant Medical Devices. In Proceedings of the Annual International Conference of the IEEE Engineering in Medicine and Biology Society, Boston, Massachusetts USA, 30 August–3 September 2011; pp. 4018–4021.
- Nguyen, M.Q.; Chou, Y.; Plesa, D.; Rao, S.; Chiao, J. Multiple inputs and multiple outputs wireless power combining and delivering systems. IEEE Trans. Power Electron. 2015, 30, 6254–6263. [Google Scholar] [CrossRef]
- Yoon, I.-J.; Ling, H. Investigation of near-field wireless power transfer under multiple transmitters. IEEE Antennas Wirel. Propag. Lett. 2011, 10, 662–665. [Google Scholar] [CrossRef]
- Lee, K.; Cho, D.-H. Diversity analysis of multiple transmitters in wireless power transfer system. IEEE Trans. Magn. 2013, 49, 2946–2952. [Google Scholar] [CrossRef]
- Nguyen, M.Q.; Plesa, D.; Rao, S.; Chiao, J.-C. A Multi-Input and Multi-Output Wireless Energy Transfer System. In Proceedings of the IEEE MTT-S International Microwave Symposium (IMS), Tampa Bay, FL, USA, 1–6 Junuary 2014; pp. 1–3.
- Walleczek, J. Electromagnetic field effects on cells of the immune system: The role of calcium signaling. FASEB J. 1992, 6, 3177–3185. [Google Scholar] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Amar, A.B.; Kouki, A.B.; Cao, H. Power Approaches for Implantable Medical Devices. Sensors 2015, 15, 28889-28914. https://doi.org/10.3390/s151128889
Amar AB, Kouki AB, Cao H. Power Approaches for Implantable Medical Devices. Sensors. 2015; 15(11):28889-28914. https://doi.org/10.3390/s151128889
Chicago/Turabian StyleAmar, Achraf Ben, Ammar B. Kouki, and Hung Cao. 2015. "Power Approaches for Implantable Medical Devices" Sensors 15, no. 11: 28889-28914. https://doi.org/10.3390/s151128889
APA StyleAmar, A. B., Kouki, A. B., & Cao, H. (2015). Power Approaches for Implantable Medical Devices. Sensors, 15(11), 28889-28914. https://doi.org/10.3390/s151128889






