1. Background
Crown-of-thorns starfish (CoTS;
Acanthaster spp., excluding
A. brevispinus) are renowned for their ability to devastate coral reef ecosystems [
1]. This is primarily because local densities of CoTS can increase from normally very low densities (<1 starfish per hectare) to extremely high densities (>1000 starfish per hectare) during periodic population outbreaks (e.g., [
2]). Moreover, CoTS are one of the largest and most efficient predators on scleractinian corals [
3]. Whereas most other individual coral-feeding organisms (e.g.,
Chaetodon butterflyfishes, and
Drupella snails) cause only localized injuries or tissue-loss [
4,
5], adult CoTS can kill entire corals, including relatively large colonies. High densities of CoTS will, therefore, cause rapid and extensive short- to long-term coral depletion. In French Polynesia, for example, high densities of CoTS caused systematic coral loss around the entire circumference of the island of Moorea, killing > 96% of coral between 2005 and 2010 [
6]. More broadly, outbreaks of
Acanthaster spp. are a major contributor to sustained declines in coral cover and degradation of coral reefs at many locations throughout the Indo-West Pacific [
7,
8,
9].
While there has been considerable research, and a large number of scientific articles (>940) focused on
Acanthaster spp., extending back to the 1960s [
10,
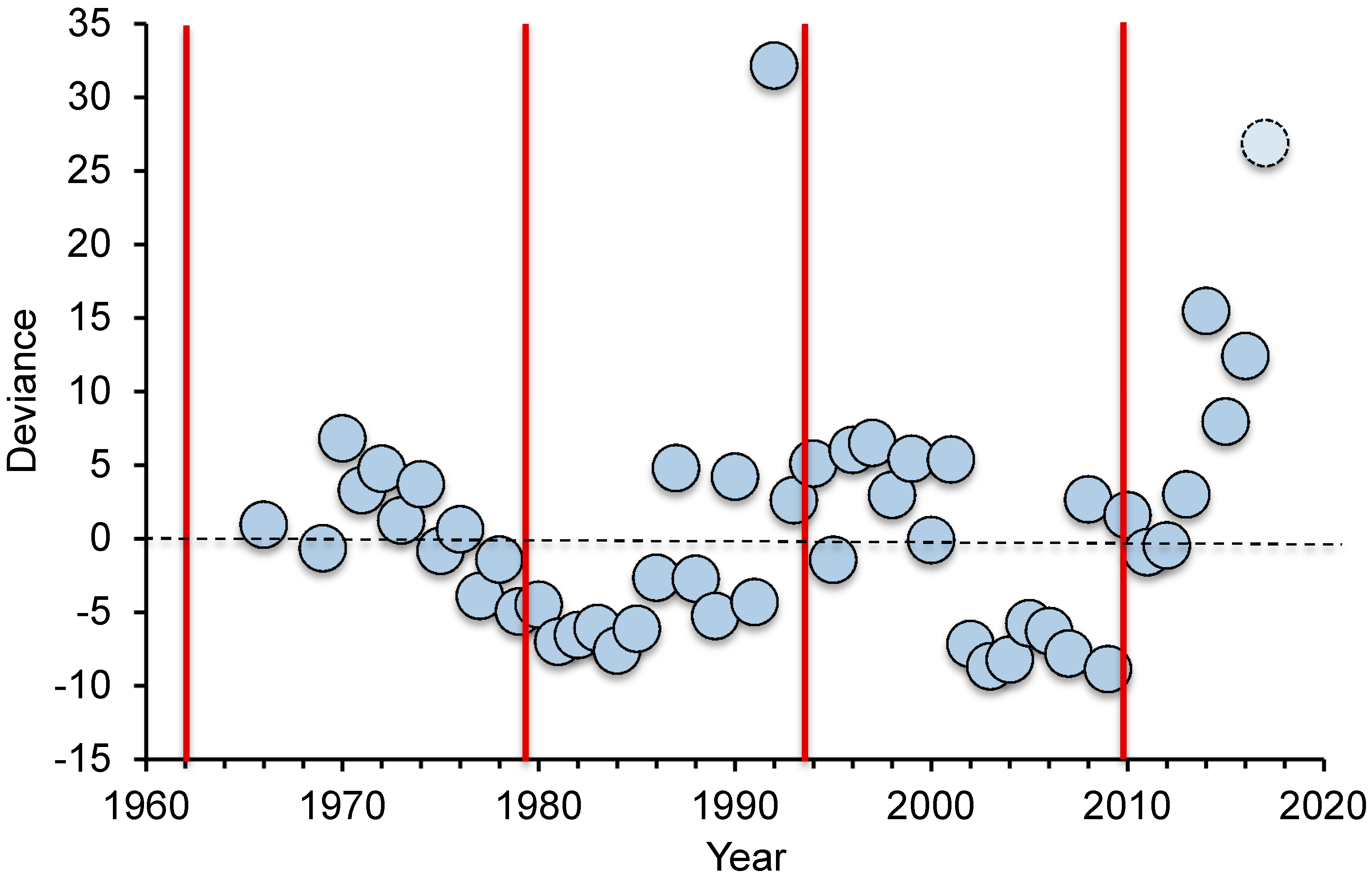
11], research interest and funding has waxed and waned through this period (
Figure 1). In Australia, a disproportionate number of research papers on CoTS have followed the initiation of each new wave of outbreaks (in 1962, 1979, 1993 and 2009), with apparent declines in publications as outbreaks subside. The main exception to this pattern was in 1979 to 1992, where the number of papers published on CoTS was lower than expected even after the initiation of the outbreak in 1979, whereas publication output was highest in 1992, immediately prior to the start of the third documented wave of outbreaks. The high number of publications in 1992 (46 publications) was partly due to two separate special issues on
Acanthaster spp. [
12,
13], as well as an explicit recognition of the need to study CoTS during non-outbreak periods [
14,
15]. Scientific, management and political interest in CoTS did decline towards the end of the third documented wave of outbreaks in the early 2000s (
Figure 1), with increasing concern about climate change and coral bleaching deflecting some attention from CoTS outbreaks. There has however, been a sustained increase in the number of scientific studies and publications on CoTS from 2010–2017 (
Figure 1).
There is greatly renewed interest in
Acanthaster spp. for two reasons. Firstly, renewed outbreaks of CoTS are occurring on the Great Barrier Reef (GBR), Australia, combined with unprecedented outbreaks at many locations throughout the Indo-Pacific [
1]. Secondly, outbreaks of CoTS remain a major contributor to sustained decline in coral cover at many reefs throughout the Indo-Pacific (e.g., [
16,
17]) and scientists and managers alike recognize the critical need to halt and reverse this decline. Preventing and/or containing CoTS outbreaks is generally considered to be one of the most feasible management actions to reduce rates of coral mortality (e.g., [
7]), thereby improving the capacity of reef systems to cope with threats due to climate change and other anthropogenic disturbances [
18]. Despite persistent knowledge gaps regarding the ultimate cause(s) of outbreaks, and the considerable time and cost required for effective management of established outbreaks, outbreaks of CoTS are one of the principal causes of major coral loss (along with severe tropical storms and mass coral bleaching) that are amenable to direct and immediate intervention [
7]. De’ath et al. [
7] suggested that preventing outbreaks of
Acanthaster spp. on the GBR could in itself reverse sustained declines in coral cover. The same is probably true for other reef regions, where outbreaks of CoTS are among the major causes of acute coral loss [
9,
17,
19,
20].
The purpose of this review is not to provide a comprehensive overview of CoTS biology or research, which are already available elsewhere [
1,
21,
22], but to consider major advances (and apparent failings) in CoTS research over the past few decades. Our assessment of research progress is based on a critical judgment of the extent to which research (including original research presented within this special issue) has addressed the knowledge gaps highlighted by a seminal review in 1986 [
21]. Ongoing research interest in
Acanthaster spp. is largely driven by persistent controversy around the cause(s) of outbreaks and corresponding management actions [
1]. This special issue (
Biology,
Ecology and Management of Crown-of-Thorns Starfish) reflects the latest resurgence in scientific interest and research on
Acanthaster spp., unequivocally focused on better understanding the initiation and spread of outbreaks, as well as refining the capability and capacity for effective management. The papers presented in this special issue address: (i) environmental drivers of fertilization and early development [
23,
24]; (ii) larval nutrition, larval development and implications for the ‘nutrient enrichment hypothesis’ [
25,
26,
27,
28,
29]; (iii) predation and implications for the ‘predator removal hypothesis’ [
30,
31,
32]; (iv) factors influencing settlement [
27,
30]; (v) dispersal and genetics [
33]; (vi) longevity, growth, and size-and-age relationship [
34,
35]; (vii) movement [
36]; and (viii) control and management [
16,
37].
Aside from research to address current issues related to the effective management of CoTS outbreaks, there have also been some fundamentally new directions for CoTS research in the last decade, enabled by advances in research methods and analytical capabilities. Therefore, this review also considers some of the foremost new directions for CoTS research, related to (i) systematics and biogeography, (ii) genetic and genomic sampling, (iii) ecological modelling, and (iv) projected effects of environmental change. Notably, genetic sequencing of CoTS populations from throughout their geographic range (Red Sea to the eastern Pacific) has resolved that there are several distinct species [
38,
39];
Acanthaster planci, which is restricted to the northern Indian Ocean, is readily distinguishable from the Pacific species (
A. cf.
solaris) based on color, as well as a general lack of spines along the aboral distal portion of the arms (
Figure 2). Overall, there are at least four distinct and geographically separated species [
38,
39], though most of the research and knowledge of CoTS biology and ecology comes from research in the western Pacific on
A. cf.
solaris. However, species-specific differences in behavior and biology may account for geographic variation in the occurrence of outbreaks, and their impacts on reef ecosystems [
1]. Thus, there is a definite need for comparative studies across multiple species of
Acanthaster, testing for differences in key demographic rates (e.g., growth and reproductive output) as well as feeding rates and dietary preferences.
2. Advances in CoTS Research
It has been 30 years since the publication of a comprehensive review titled, “The
Acanthaster Phenomenon” by Moran [
21]. Moran [
21] reported that crown-of-thorns starfish are probably the single most studied species (or species complex) on coral reefs, and yet there were many knowledge gaps that directly hampered effective management of population outbreaks. Accordingly, Moran [
21] presented a list of 41 questions considered fundamental to understanding the causes and consequences of outbreaks. These questions were intended to guide research through the subsequent years and decades to improve both understanding and management of outbreaks of
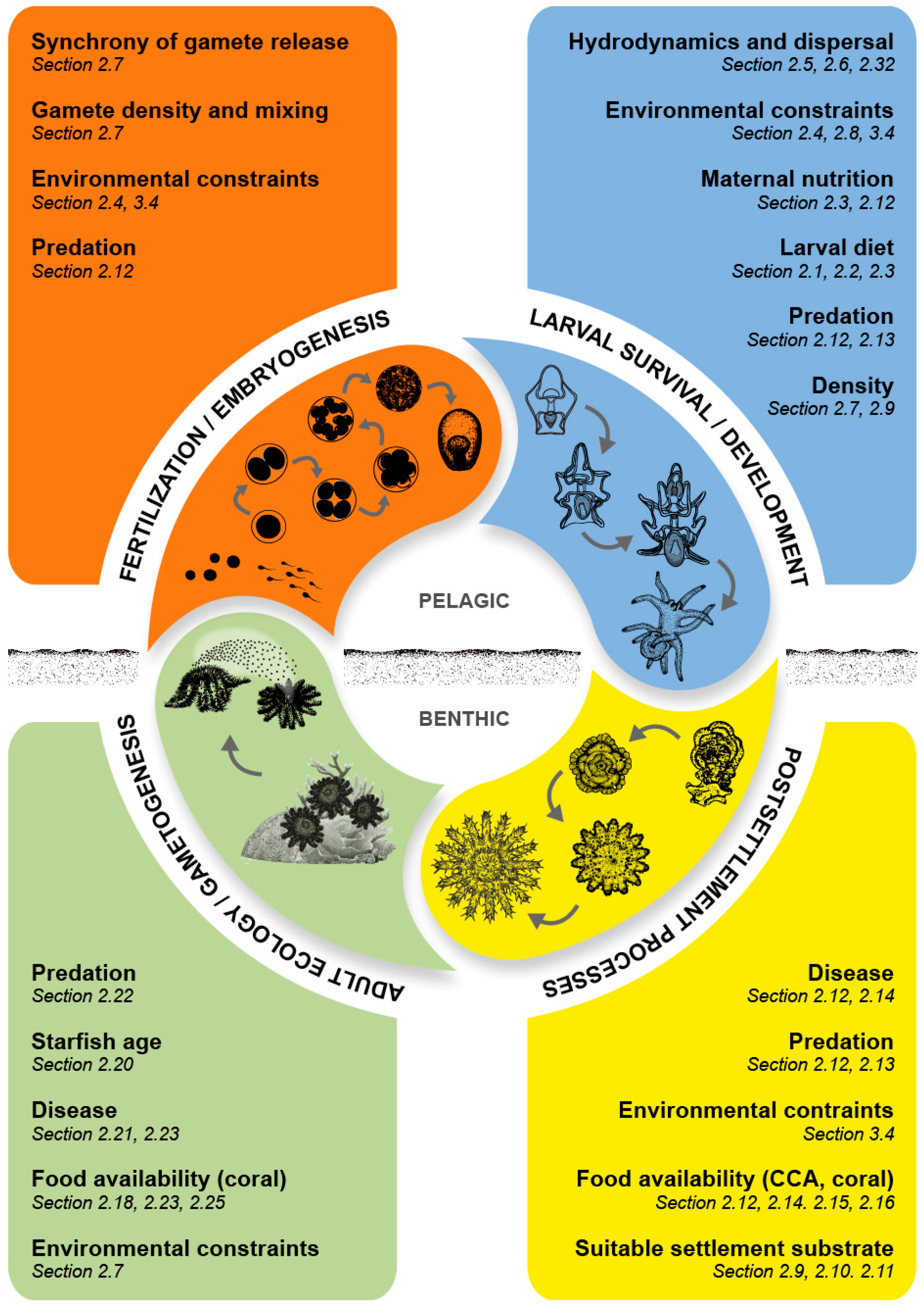
Acanthaster spp. They were presented within three broad categories (Larvae and Juveniles—21 questions, Adults—11 questions, and Effects on Communities and Processes—3 questions) together with a further six overarching questions. To assess the advances (or lack thereof) in our understanding of the biology of CoTS over the last three decades, as well as causes and consequences of outbreaks, we review progress against each of these 41 research questions, specifically highlighting the contributions of research articles presented in this issue. Where relevant, questions have been grouped together under a single section heading to minimize repetition.
2.1. Questions 1 and 3 (Larvae and Juveniles)—Are high nutrient conditions needed for the enhanced survival of larvae in the field? Can larvae develop and settle under ‘non-bloom’ nutrient conditions in the field? If so, can high densities of larvae be sustained under these conditions?
While receiving considerable attention, these questions are LARGELY UNRESOLVED.
One of the foremost hypotheses proposed to account for outbreaks of CoTS, the
larval starvation hypothesis, is predicated on a link between rates of effective larval development and availability of suitable prey (mostly unicellular phytoplankton; e.g., [
40,
41,
42,
43]). Early studies by Lucas [
40] suggested that rates of development and survivorship increased with increasing algal concentrations. Moreover, Okaji [
41] demonstrated that CoTS larvae grew faster and had a higher survivorship with increasing chlorophyll-
a (chl-
a) levels (>2 μg chl-
a L
−1), suggesting that enrichment of nearshore waters and phytoplankton assemblages would lead to increased densities of
A. cf.
solaris larvae [
44,
45]. Conversely, field-based experiments conducted by Olson [
46], using an apparatus designed to rear larvae in situ, suggested that larvae grew well under low chl-
a conditions. However, these results were potentially confounded by contamination and retention of phytoplankton within the experimental apparatus, resulting in higher than expected chl-
a concentrations [
41]. Nevertheless, in situ studies of larval development and survival, with simultaneous sampling of environmental parameters (e.g., cell-counts, chl-
a, organic carbon content), are still needed.
Recent experimental studies have shown that elevated chlorophyll concentrations may not be necessary for CoTS larvae to complete development (reviewed in [
47]). Laboratory experiments by Wolfe et al. [
43,
47] demonstrated high larval survival and settlement success across a broad range of nutrient levels, and most importantly, below the lower threshold levels previously suggested by Fabricius et al. [
45]. The robust nature of larvae, even in oligotrophic conditions, may be driven by increased investment in provisioning of eggs by well-fed adults [
25,
48] and the morphological plasticity of larvae to respond to changes in the availability of exogenous prey [
25,
49]. Furthermore, there appears to be an upper threshold for optimal larval survival and development, whereby very high levels of nutrients have been shown to be deleterious to larval development for CoTS, and larval growth and development are maximized at close to normal background concentrations of chl-
a [
27,
43]. Larval survival and size at set intervals (4, 7, and 10 days after fertilization), as well as settlement rate and size of newly settled juveniles after 18 days were significantly lower for treatments with the highest phytoplankton concentration (100,000 cells per mL; 10 μg chl-
a per L) compared to intermediate food levels (10,000 cells per mL; 1 μg chl-
a per L) [
43]. Mortality rates for larvae under high algal concentration (100,000 cells per mL; 10 μg chl-
a per L) were significantly higher from very early in larval development, compared to low (1000 cells per mL; 0.1 μg chl-
a per L) and intermediate (10,000 cells per mL; 1 μg chl-
a per L) food levels [
27]. Taken together, these results suggest that chl-
a levels between 0.4 and 1.0 μg per L may be the optimal range for larval survival and development.
The use of
Proteomonas sulcata in both Wolfe et al. [
43] and Pratchett et al. [
27] was criticized by Brodie et al. [
29] because of the very low chl-
a concentration of this phytoplankton species. In addition, the high cell numbers (~100,000 cells per mL) are unlikely to occur in the field; even under flood conditions, algal cell numbers rarely exceed 1000 cells per ml [
50]. Similarly, nutrient enrichment of natural seawater used in larval feeding experiments presented in Fabricius et al. [
45] had maximum cell numbers of 4400 cells per ml (equivalent to ~5.2 µg chl-
a per L in their study). As these numbers illustrate, it is unfortunate that the “nutrient hypothesis” (or “terrestrial runoff hypothesis”) is currently expressed in terms of chl-
a concentration. Although chl-
a concentration is easy to measure, chl-
a content varies widely among species and is not necessarily reflective of the nutritional value of the algae. Further studies need to focus on energy content (or at least organic carbon content) of food organisms (see [
26]). Debate continues as to whether terrestrial runoff promotes primary outbreaks on the GBR [
44,
45] and/or plays an important role in fuelling subsequent secondary outbreaks [
1,
29].
One of the foremost limitations in understanding purported links between nutrient enrichment, increases in phytoplankton abundance or changes in phytoplankton composition, and the increased survivorship and settlement of
Acanthaster spp. is the lack of systematic monitoring of relevant variables. On the GBR, for example, we lack the necessary information to explicitly compare nutrient availability and phytoplankton assemblages among reefs considered important for initiating primary outbreaks, versus those that support secondary outbreaks or are generally unaffected by CoTS outbreaks. It is also now understood that the algorithms used in the GBR to estimate chl-
a from satellite remote sensing, in particular the MODIS satellite with the Aqua sensor, can give inaccurate and biased results; and therefore, usage should be avoided for absolute measures of chl-
a [
51]. In situ monitoring of nutrients, phytoplankton assemblages, and CoTS larvae, to relate size, abundance, and condition of larvae to local biological and environmental parameters (e.g., [
49,
52]) in the lead up to the next outbreak of
A. cf.
solaris on the GBR, would clearly establish whether CoTS larvae can develop and settle even in the absence of nutrient plumes and phytoplankton blooms. This would go a long way to resolving the extent to which sustained but gradual improvements in catchment management and water quality are an effective mechanism for limiting future outbreaks. In addition, new genetic methods (eDNA, next generation sequencing) to identify larvae [
53,
54] and potentially characterize phytoplankton abundance and community structure in the same water parcel provide opportunities to investigate the connection between nutrient inputs and changes in the quantity and quality of food for the larvae.
2.2. Question 2 (Larvae and Juveniles)—Do these types of conditions (i.e., high nutrient conditions and associated phytoplankton blooms) occur frequently in the field? If so, do they coincide with observed spawning periods and how long do they occur?
These questions are Largely Unresolved.
Spatial and temporal coincidence between high nutrient conditions and enhanced survivorship of CoTS larvae in the field would lend significant support to the
larval starvation hypothesis [
45,
55]. However, establishing these links is complicated by aforementioned controversies about levels of food availability necessary to ensure development and survival of CoTS larvae (
Section 2.1), as well as the inability to directly assess larval densities and survivorship in the wild. Fabricius et al. [
45] argued that interannual variation in the cumulative annual discharge from the Burdekin river (one of five major rivers that discharge into the GBR) corresponds with the initiation of outbreaks of
A. cf.
solaris after allowing for inevitable lags in the timing of floods (and corresponding increases in larval densities) versus first reports of elevated densities of adult starfish in 1962, 1979, 1993 and 2009. However, the lag between major flood events and corresponding outbreaks of
A. cf.
solaris ranges from 2–5 years, possibly due to limitations in detecting the specific onset of outbreaks [
1]. Moreover, major flood events may or may not initiate outbreaks, depending on the availability of coral food resources [
25,
48], the timing of floods relative to specific spawning periods and developmental rates for
A. cf.
solaris [
45] and whether larval food supply is, in fact, a major limiting factor.
While there is a definite need for further research to resolve interannual variability in the specific pattern and occurrence of spawning by
A. solaris on the GBR relative to particular flood events [
52], the general onset of reproduction and larval development (December–March) broadly coincides with periods of heavy rainfall and increased likelihood of flooding [
29]. Moreover, flood plumes and phytoplankton blooms occur frequently, almost annually within certain areas of the GBR (almost annually) and can persist for weeks to months [
29]. These floodwaters provide increases in nutrient concentrations, especially from within heavily modified catchments with intensive agriculture [
45,
50]. However, it is the confluence of nutrient enrichment from flood plumes and relatively clean offshore waters that enable the proliferation of phytoplankton, potentially explaining why outbreaks of
A. cf.
solaris predominantly occur on mid-shelf reefs. If, however, phytoplankton blooms sufficient to sustain elevated densities of CoTS larvae occur almost annually, this cannot explain why initial outbreaks (primary outbreaks) occur relatively infrequently and at specific locations in the northern GBR [
1]. Rather, persistent nutrient enrichment may be important in sustaining the proliferation and spread of outbreaks (secondary outbreaks) once they have become established at discrete reef locations [
29]. If food is limiting, however, we might expect successive waves of outbreaks to peter out, especially in areas of the central GBR, which have lowest nutrient inputs (but see [
56]).
Wooldridge and Brodie [
57] explicitly acknowledge that high nutrient conditions and associated phytoplankton blooms occur quite commonly during summer in the northern GBR, but do not always initiate outbreaks of
A. cf.
solaris. At Green Island, for example chl-
a concentrations exceeded 0.80 μg chl-
a per L for prolonged periods at least six times between 1969 and 1998, though outbreaks developed only twice, in 1979 and 1993. Elevated nutrients may therefore, be a necessary precursor for outbreaks to become established, but there are other conditions that must also be met. Modelling studies by Wooldridge and Brodie [
57] suggested that it is interannual variation in levels of larval retention (see [
58]) that may explain when, and perhaps where, primary outbreaks become established. Importantly, hydrodynamic conditions would have promoted high levels of self-recruitment, rather than dispersal of larvae among widely separated reefs, in years prior to outbreaks arising in 1979 and 1993 [
57]. However, it is yet to be tested whether primary outbreaks are directly attributable to iterative increases in population size, due to high levels of self-recruitment [
59]. As stated previously, increased monitoring of relevant metrics for food availability is needed alongside fine-scale spatial and temporal sampling to document the initiation and spread of CoTS outbreaks.
2.3. Question 4 (Larvae and Juveniles)—How important is diet in influencing the survival of larvae? Is survival more dependent on the diversity rather than density of food species? What other factors influence the survival of larvae?
The first two components are MOSTLY RESOLVED, but larval survivorship is yet to be studied in field settings (
Section 2.12).
The abundance of specific phytoplankton is much more important than overall abundance of phytoplankton (and corresponding chl-
a concentrations) for promoting rapid development and survival of CoTS larvae. Larvae of
A. cf.
solaris feed predominantly on mid-sized phytoplankton (e.g., dinoflagellates and pennate diatoms >5 µm) [
40,
60,
61,
62], whereas tropical coral reef waters are generally dominated by picoplankton (e.g., pelagic cyanobacteria—
Synechococcus and
Prochlorococcus <2 µm), which typically make up >50% of total primary production in oligotrophic waters (e.g., [
63,
64,
65]). As such, total chl-
a concentrations may grossly overestimate food availability for CoTS larvae. Elevated nutrients due to terrestrial runoff and upwelling do promote increased dominance of phytoplankton species with larger cell sizes [
65,
66,
67], potentially benefitting CoTS larvae, but it is nonetheless necessary to sample phytoplankton assemblages directly (rather than relying on chl-
a concentrations) to assess the conditions that promote larval survival and the onset of outbreaks.
Very few studies have specifically considered the feeding selectivity of CoTS larvae, other than showing that there is strong size selectivity. Okaji et al. [
68] demonstrated that clearance rates of larval feeding were significantly lower for smaller phytoplankton (1–2 µm) compared to phytoplankton with larger cell sizes (4–5 µm), even when smaller phytoplankton were overwhelmingly dominant. Also, heterotrophic bacteria appear to have a negligible role in larval nutrition [
69]; though CoTS larvae are able to assimilate and utilize dissolved organic matter (DOM) in the water column [
60,
70], as well as coral-derived organic matter, such as mucus and associated microorganisms [
28]. Mellin et al. [
26] tested for selective feeding by CoTS larvae among phytoplankton species of similar size (>5 µm). CoTS larvae consistently preferred algal species with the highest energetic content (
Chaetoceros,
Dunaliella) over microalgae with lower energy content (
Pavlova lutheri,
Phaeodactylum tricornutum), which would presumably lead to higher growth rates and elevated survival of CoTS larvae, though this was not tested. Although algal species used in that study have been commonly used in aquaculture and are within the size-range preferred by CoTS, these species are not naturally present at high concentrations on the GBR (except for
Chaetoceros spp.). It is important therefore, to establish feeding selectivity for algal species that are particularly dominant during flood events (e.g.,
Skeletonema; [
50]) and DOM, as well as testing for variation in larval growth and survivorship with changes in the availability of specific prey types.
The extent to which development and survival rates of CoTS larvae are constrained by exogenous food availability is equivocal, and somewhat dependent on several other factors (e.g., maternal provisioning of larvae, and vulnerability to predation). Caballes et al. [
48] demonstrated that differences in the nutritional condition of female
A. cf.
solaris, based on contrasting diets (e.g.,
Acropora versus
Porites), have a major bearing on the growth and performance of their progeny. Well-fed females provision their offspring with increased levels of endogenous energetic reserves, which not only allows larvae to withstand prolonged periods of starvation, but also enables them to grow larger and feed more efficiently [
25,
49]. Larval survivorship in the wild will also be limited by competition, predation and environmental constraints on development (see
Section 2.13 and
Section 2.14). If higher food concentrations also benefit predators of CoTS, this has the potential to suppress larval survival and outbreaks [
71]. There are no empirical data on rates of predation for CoTS larvae in the wild, and such information will be very difficult to obtain, though CoTS larvae are certainly vulnerable to predation [
72].
2.4. Question 5 (Larvae and Juveniles)—Do certain physical conditions occur in the field that cause the increased survival of larvae? Do these conditions act in conjunction with any other factors?
These questions are LARGELY RESOLVED, though there is scope for more work on synergistic effects of climate-induced changes in physical conditions.
The specific physical conditions that promote larval development and survival for
Acanthaster spp. has received a great deal of attention (e.g., [
40,
73]) and there has been a recent resurgence in research on the environmental tolerances of CoTS larvae aimed at establishing the vulnerability of
Acanthaster spp. to ocean warming and acidification [
24,
74,
75,
76]. Temperature is widely regarded as the foremost abiotic factor influencing development rates and survivorship of planktonic larvae [
77], and
Acanthaster spp. are sensitive to extreme temperatures at all stages of their life-cycle [
24,
61]. Fitness of CoTS larvae is generally highest at 26–30 °C [
24,
76]. There is increasing evidence that CoTS larvae are adversely affected when exposed to temperatures ≥ 30 °C [
75], suggesting that ocean warming suppresses population outbreaks at low latitudes. Projected effects of ocean warming are further compounded by constraints on fertilization and larval development due to ocean acidification [
24,
74,
75], though it is possible that
Acanthaster spp. could acclimate or adapt to changing environmental conditions [
78].
Aside from temperature, salinity is considered to have an important influence on reproductive success for
Acanthaster spp. [
24,
79]. While echinoderms are generally very sensitive to changes in salinity, Lucas [
79] showed that larval survival was 3-fold higher at 30‰ salinity compared to ambient conditions. If so, temporary declines in salinity could further enhance the beneficial effects of nutrient inputs, during flooding [
44,
55]. However, Caballes et al. [
24] showed that rates of fertilization, gastrulation and cleavage were generally high between 30‰ and 34‰ and declined significantly at salinities <30‰.
2.5. Question 6 and 7 (Larvae and Juveniles)—How long do [CoTS] larvae spend in the plankton before settling? What is the maximum period of time they can spend in this phase and yet still be able to settle? How far can [CoTS] larvae be dispersed in the field?
These questions are LARGELY RESOLVED.
The time that larvae spend in the plankton, or planktonic larval duration (PLD), is constrained by the minimum pre-competency period, which is the necessary time for larvae to complete development before being capable of settling, and the maximum competency period, which is maximum time that larvae can spend in the plankton and still be capable of effective settlement [
27]. The minimum time taken for CoTS to develop into late-stage brachiolaria larvae, which are assumed to be competent to settle, is just 9 days [
80], though actual settlement has never been documented <14 days post-fertilization [
46]. At the other end of the spectrum, Pratchett et al. [
27] recorded settlement among larvae of
A. cf.
solaris up to 43 days post-fertilization. However, settlement rates peaked at 22 days for optimal (intermediate) food levels and declined through time due to limited survivorship >30 days post-fertilization and reduced settlement competency of surviving larvae [
27].
While CoTS larvae may settle in as little as 9–14 days after fertilization, their maximum competency period and capacity for long-distance dispersal is largely unknown. The maximum recorded longevity for CoTS larvae is 50 days [
27], and could be even longer under conditions of limited food availability and if deprived of opportunities to settle. However, CoTS larvae are planktotrophic and must maintain certain levels of energetic reserves to complete metamorphosis and settlement [
27]. Moreover, rates of larval mortality in the wild are likely to be even higher than have been documented in experimental studies (e.g., due to predation), such that few larvae are likely to persist beyond 30 days, let alone 50 days. Assuming average daily rates of natural mortality for echinoderm larvae (~0.16 per day; [
81]), CoTS larvae will have a survival probability of approximately 0.82% after 30 days, and 0.03% after 50 days. For the most part, CoTS larvae are expected to be dispersed only 10 s–100 s km between reefs [
82], if not entrained within the confines of their natal reef [
83,
84]. Genetic sampling of CoTS populations demonstrated that there is effective connectivity (reflective of ecological significant levels of larval dispersal) between reefs separated by <1000 km [
33,
85,
86]. However, there tends to be very strong genetic differentiation of CoTS populations among geographic provinces [
87,
88], not to mention distinct species in different ocean basins [
39], suggesting that there is extremely limited connectivity, and therefore, negligible larval dispersal, at distances of >1000 km.
2.6. Question 8 and 14 (Larvae and Juveniles)—What factors are important in causing dispersal [of CoTS larvae]? Do larvae tend to settle on those reefs from which they were propagated or do they generally recruit to reefs other than the parent reef?
The first component is WELL RESOLVED, but there is limited empirical information regarding rates of dispersal versus retention.
CoTS larvae are, for the most part, passively dispersed by ocean currents [
89]. Therefore, the primary factors that influence dispersal are (i) how long larvae can persist while still retaining the capacity to settle (see
Section 2.5) and (ii) oceanographic conditions, specifically, the strength and direction of water movement, during spawning and larval development [
85]. On the GBR, the extent to which larvae are retained and settle on their natal reefs (self-recruitment), rather than being dispersed, has been modelled for at least one small sub-region and may vary among years with changes in ocean current velocities driven by El Niño Southern Oscillation (ENSO) [
57]. However, it is not clear whether such effects exist in other regions at other phases of the ENSO cycle, particularly those that are putative seed areas for outbreaks. Weak or variable along-shore currents, which occur during neutral phases of the Southern Oscillation Index (SOI) may promote strong larval retention or very limited dispersal, which is fundamental to the progressive accumulation of CoTS within a given location and is likely to give rise to primary outbreaks [
57,
59]. In contrast, strong directional (southerly) along-shore currents associated with strong El Niño or La Niña conditions will increase the likelihood of inter-reef dispersal, which could lead to proliferation of outbreaks once they become established [
57]. These ideas are however, based on temporal autocorrelation in the initiation of outbreaks, rather than explicit empirical data on rates of self-recruitment versus larval dispersal.
High rates of self-recruitment by
Acanthaster spp. may be reinforced by conspecific chemo attraction of settling larvae towards feeding aggregations of adult CoTS [
2,
30]. In static choice chamber experiments, Cowan et al. [
30] showed that CoTS larvae were significantly attracted to adult conspecifics, which could lead to elevated rates of settlement on reefs already infested with high densities of CoTS. While settling in the presence of adult conspecifics may seem maladaptive due to ultimate competition for coral prey, this strategy may limit predation by sessile invertebrates [
61,
90] leading to overall increases in larval survivorship. However, the spread of population outbreaks, determined based on field surveys [
56,
91,
92] and modelling [
56,
58,
82,
83,
84,
93,
94] shows that at least some larvae must be dispersed and settle on non-natal reefs, regardless of the presence of adult CoTS.
Improvements in hydrodynamic models, combined with advances in computational power and new methods for analyzing patterns of particle dispersal, are providing increasingly resolved and tractable models to inform patterns of initiation and spread for CoTS outbreaks [
58,
82,
93,
95,
96]. However, these models are potentially very sensitive to the precise timing of spawning and the relevant speed and direction of currents, and predictions arising from these models need explicit testing based on extensive spatial and temporal sampling to resolve the occurrence and timing of outbreaks. Alternatively, genetic approaches may be used to explicitly resolve actual connections among discrete populations to validate dispersal patterns. However, using genetics to track the spread of outbreaks has proved difficult for CoTS due to the low levels of genetic differentiation apparent when using existing markers [
33,
97,
98]. On the GBR, for example, genetic sampling during outbreaks has failed to resolve any structure [
33,
97], indicating rapid expansion in population size from multiple, undifferentiated latent populations. Similarly, studies elsewhere in the Pacific have identified largely homogeneous populations within specific reef systems [
86,
99,
100], though CoTS generally exhibit substantial regional, archipelagic genetic structuring [
88], reflective of limited large-scale dispersal. Greater resolution in genetic structure among outbreak populations, enabling greater insight into the source and spread of outbreaks, may be possible using more extensive and comprehensive sampling (e.g., single-nucleotide polymorphisms or SNPs), but these are yet to be tested for any
Acanthaster sp.
2.7. Question 9 (Larvae And Juveniles)—Is there a positive correlation between larval density, recruitment density, and adult density?
Given previous limitations in sampling larvae and recruits, this question has not been addressed and is UNRESOLVED.
High densities of CoTS larvae and high recruitment will intuitively lead to increased densities of adult CoTS, and are a fundamental precursor to manifest rapid population outbreaks (mostly, secondary outbreaks). However, the more important question is whether it is the local densities of larvae (larval supply) or effective rates of recruitment that generally limit adult densities? There are also important, and as yet unresolved issues, about how far and how fast CoTS can move within and among reef habitats after they have settled (
Section 2.18), which will determine relevant scales of recruitment limitation. Most hypotheses that seek to explain the initiation of CoTS outbreaks assume that larval supply is generally limiting, such that outbreaks arise due to increased reproductive success and/or larval survivorship [
1]. However, the inability to quantify larval supply, settlement, and recruitment has so far prevented explicit testing of such assumptions. New methods aimed at measuring these processes are being developed and tested [
34,
53] and will not only provide new opportunities to test questions pertaining to recruitment limitation, but may also provide an early warning system for detecting new and renewed outbreaks.
The relationship between adult densities of
Acanthaster spp. and their corresponding reproductive output versus local densities of larvae or juveniles (stock-recruitment relationships) is important for understanding the role of adult biology and behavior in initiating outbreaks, as well as informing the effectiveness of population regulation based on culling of adult starfish. Given the high fecundity of individual CoTS [
101], larval production is likely to asymptote at relatively low adult densities [
102,
103] such that larval production may be largely insensitive to changes in adult abundance above a certain threshold. If so, this would mean that local densities would need to be reduced to very low levels before adult culling would have any meaningful impact on reproductive output and the progression of outbreaks. Moreover, the distribution and proximity of spawning starfish may be more important than adult densities in determining reproductive success (though it would be expected that there must be some relationship between these factors). Babcock and Mundy [
104] showed that
A. cf.
solaris achieve remarkably high rates of fertilization even when spawning male and female starfish were separated by >30 m. However, fertilization success is fundamentally dependent on spawning synchrony, which appears to be triggered via intrinsic cues (pheromones) and will therefore, be most effective when starfish are aggregated [
52]. Still, it is unclear whether
Acanthaster spp. actively aggregate to spawn, and what environmental cues induce spawning.
Recent modelling of fertilization success in CoTS suggests that both density and aggregation are important to reproductive success at low densities with a threshold density for enhanced reproductive success of 3 starfish per hectare when individuals are moderately aggregated [
105]. Reproductive success increased linearly above this density. At the highest levels of density and aggregation fertilization success for individuals did not increase due the increases in unsuccessful polyspermic fertilizations although population level zygote production did continue to increase [
105]. The reduced fertilization success at high levels of aggregation may in part explain the relative lack of aggregation by CoTS at the time of spawning [
106].
2.8. Question 10 (Larvae and Juveniles)—Where do larvae occur in the water column? Does their position vary throughout their planktonic period? What factors are responsible for determining their position?
These questions are LARGELY UNRESOLVED and important for understanding the environmental and nutritional conditions to which larvae are exposed during their development.
In laboratory cultures, hatched gastrulae of
A. cf.
solaris swim upward and remain close to the surface (negative geotaxis) throughout much of the formative period of their development [
61]. Larvae then become negatively buoyant at the late brachiolaria stage and actively orientate towards the bottom in search of potential settlement substrates [
41]. It is assumed therefore, that CoTS larvae are likely to be largely concentrated in surface waters in the wild. Accordingly, CoTS DNA have been detected in near-surface plankton tows along 320 km of coastline in the northern GBR [
53]. Similarly, plankton tows at 7 m below the surface along reefs in southern Japan have also yielded high concentrations of advanced-stage brachiolaria larvae [
107]. CoTS larvae are also capable of swimming, albeit at limited speeds (~0.4 mm per second), via ciliary movement that causes the body to rotate on its long axis [
61]. The horizontal transport of CoTS larvae has typically been considered a passive process that is mainly mediated by currents [
58,
87,
89]. However, current velocity and direction can vary with depth, so the vertical position of larvae and larval behavior in response to thermoclines, haloclines, or pycnoclines will have important implications for distance and direction of larval transport (e.g., [
108,
109,
110,
111]). Explicit field sampling is still required to ascertain the specific position of CoTS larvae at different stages throughout their development and under varying conditions; but this may now be possible with the development of new genetic quantification methods [
54].
2.9. Question 11 and 13 (Larvae and Juveniles)—Where do larvae settle in the field? Is it in shallow or deep water on reefs? Are there particular areas on reefs which are more suitable for settlement than others? Do they settle in high densities?
There have been significant insights on patterns and rates of settlement by specific, localized studies (e.g., [
112]), but these questions are MOSTLY UNRESOLVED.
One of the foremost controversies surrounding settlement patterns for
Acanthaster spp. is the extent to which larvae settle in deep-water (>20 m) versus shallow reef environments. Although earlier reports suggested that settlement occurred in shallow reef environments [
2,
113] and high densities of newly settled and juvenile
Acanthaster spp. have never been recorded in deep water (>20 m depth), the deep-water recruitment hypothesis [
114] has gained a lot of attention. CoTS larvae were suggested to settle mainly in deep water, at the base of reef slopes [
114] because (i) highest rates of settlement and metamorphosis occurred on coral rubble encrusted with the sciaphilic crustose coralline algae (CCA),
Lithothamnium pseudosorum, which was found predominantly in deep water habitats (but are also common in caves, crevices and overhangs in shallow water; S. Uthicke, personal observation); (ii) late-stage brachiolaria larvae are negatively buoyant and are expected to be concentrated along reef margins, such that larvae will ultimately fall out in deep water [
83]; (iii) few newly settled (0+ year class) starfish have historically been found in shallow reef environments; and (iv) on some reefs (e.g., Davies Reef in the central GBR), high densities of adult CoTS were initially detected moving up from deep water [
114]. To test whether
Acanthaster spp. preferentially settle in shallow (<5 m depth) or deep-water habitats (>20 m depth), standardized settlement collectors (e.g., [
115]) should be deployed across a range of depths, and this research is currently underway. It is possible however, that CoTS larvae settle across a broad range of depths, but settle preferentially in areas that provide best access to food and shelter. Alternatively, they may settle indiscriminately among habitats, but have vastly different rates of post-settlement survival depending on local abundance of prey, shelter, and/or predators (
Section 2.11).
Field-based studies on patterns of CoTS settlement are largely focused on the detection of newly settled CoTS, rather than explicitly measuring settlement rates in different habitats (but see [
115]). Even so, understanding the habitat preferences of newly settled CoTS has been greatly constrained by the small size and cryptic nature of newly settled individuals [
112,
114]. Until recently, few newly settled (0+ year class)
A. cf.
solaris had ever been detected on the GBR [
11,
114,
116]. At Suva Reef in Fiji, however, high densities of very small (10–32 mm in diameter)
A. cf.
solaris were detected in July 1984 [
112]. These individuals were presumed to have settled
en masse in January 1984, but were not detected until they were ≥6 months old. Even so, the newly settled (0+ year class) starfish were mainly found on the encrusting coralline algae,
Porolithon onkodes, on the underside of rubble and coral blocks [
112], suggesting that they had settled in the area and habitat in which they were found. Significant densities of newly settled
A. cf.
solaris were found immediately behind the exposed reef crest, in very shallow habitat dominated by rubble and intact skeletons of robust corals dislodged during cyclones and tsunamis [
112]. Newly settled CoTS were also sighted off the reef slope on the windward side of Suva Reef (6–8 m depth), indicating that their distribution extended subtidally [
112]. At Iriomote Island, Japan, Habe et al. [
117] detected highest densities (0.82 individuals per m
2) of newly settled CoTS on reef slopes (6.0–9.3 m depth), and lowest densities (0.06. individuals per m
2) on the reef flat (0.8–1.2 m).
During recent opportunistic sampling on the GBR, Wilmes et al. [
34] collected 3532 juvenile
A. cf.
solaris ranging in size from 3 to 64 mm in diameter. Sampling was conducted across 64 reefs throughout the course of 2015, with searching concentrated on visible patches of CCA on dead corals or coral rubble. Newly settled (0+ year class) starfish were collected from a range of depths (up to 15 m depth) and habitats. However, collections were mainly intended to inform growth models (see
Section 2.16) and so limited information was collected regarding the specific habitat conditions and exact densities. Despite limited success in the past [
116,
118], current work by Wilmes et al. [
34] demonstrates that 0+ year juveniles can be effectively sampled in the field, and much more sampling is required to establish variation in rates of settlement across different reef areas, depths and habitats. There are also methods available for measuring settlement rates of
Acanthaster spp. using settlement collectors constructed from high surface area plastic biospheres [
115], which will be important to differentiate where larvae settle versus where they survive and are actually recorded several months after settlement (
Section 2.9,
Section 2.10 and
Section 2.11). Previous constraints on the use of settlement collectors were the time and effort required to manual sort and visually detect newly settled CoTS, though modification of genetic sampling protocols used for larval detection [
53,
54] may overcome such constraints.
2.10. Question 12 (Larvae and Juveniles)—Do larvae tend to settle on a particular type of surface? What factors are important in determining the type of surface chosen by larvae for settlement?
These questions are LARGELY RESOLVED.
Settlement preferences of
Acanthaster spp. are dictated by both physical and biological habitat structure. Larvae settle preferentially in habitats with fine-scale topographic complexity, so that the larvae are completely hidden within the carbonate matrix, or among coral rubble, prior to metamorphosis [
80]. Ormond and Campbell [
119] demonstrated that skeletons of dead
Acropora hyacinthus were among the most preferred settlement substrates, probably owing to the fine-scale complexity provided by individual calices and branchlets. Conversely, CoTS will rarely settle on glass or ceramic tiles [
119,
120,
121]. However, CCA is a strong settlement inducer, and biological stimuli may override physical microhabitat preferences [
120]. Observations of newly settled
A. cf.
solaris in the field [
34,
112,
122], have revealed a strong and consistent association with CCA, which is expected given that newly settled starfish feed almost exclusively on coralline algae [
61,
112,
123].
Settlement experiments conducted under laboratory conditions [
27,
42,
43,
61,
121,
124] reaffirm that CCA is important for inducing CoTS settlement. When examined microscopically, the surface of CCA is roughly textured—this fine scale topographic complexity can provide a tactile stimulus for larvae to settle [
80]. Conversely, Johnson et al. [
121] argued that a tactile stimulus was unlikely given that settlement was high on live CCA as opposed to physically similar, but boiled, bleached, or autoclaved CCA fragments. They also observed high rates of settlement among larvae that were physically separated from CCA using a mesh, suggesting that settlement may be chemically mediated [
121]. However, bioassays with common marine invertebrate settlement inducers, γ-amino butyric acid (GABA) and potassium chloride (KCl) at different concentrations, did not induce settlement and metamorphosis in CoTS larvae [
121]. Antibiotic treatment of highly inductive shards of CCA significantly reduced settlement to low levels, indicating that settlement may be mediated by chemical cues produced by epiphytic bacteria [
121]. Settlement was inhibited in the absence of bacteria and larvae always settled on sections of CCA thalli that had high densities of bacteria, but not where epiphytic bacteria were sparse [
121,
125]. However, surface bacteria were not inductive when isolated from soluble algal compounds, suggesting that bacteria require the algal substrate to produce inductive compounds or that compounds from both the bacteria and CCA are required to induce settlement [
125]. It appears that both tactile and chemical stimuli may play a role in determining settlement preferences, though further field sampling is required to establish the extent to which these preferences determine settlement patterns in the wild (e.g., [
119]).
2.11. Question 15 (Larvae and Juveniles)—Do juveniles tend to be in shallow or deep water on reefs? Does this location vary depending on whether or not the reef has recently suffered an outbreak of adults?
Given limited effective sampling of recruits, these questions are LARGELY UNRESOLVED.
The distribution of juvenile CoTS will be largely dictated by patterns of larval settlement (see
Section 2.9), though these patterns may be greatly altered and obscured by differential rates of post-settlement mortality and movement [
126]. Mortality rates for newly settled (post-metamorphosed) juvenile CoTS are unknown, but are expected to be very high due to the combination of predation, disease, and food limitation ([
126];
Section 2.12). In terms of moderating the distribution and abundance of juvenile CoTS, the key question is whether rates of mortality vary spatially (e.g., among habitat types or with depth). Keesing and Halford [
126] suggested that known predators on very small CoTS occur in particularly high abundance among dead coral rubble, though it is also possible that high complexity of these habitats moderates actual predation rates. Conversely, predators associated with coral-rich habitats may represent an even greater threat to survival of juvenile CoTS [
30]. There will also be an inherent tradeoff between the risk of predation and the necessary settlement cues and food resources that will determine the extent to which juvenile CoTS are associated with different habitat types [
30]. Another key factor that will potentially influence the survival and therefore relative abundance of juvenile CoTS in different habitats is the availability of suitable prey, and corresponding effects on the size and growth of individuals (
Section 2.15).
The locomotor capacity of
A. cf.
solaris is size-dependent [
127], such that very small CoTS move very slowly [
61] and are unlikely to venture far from where they settle [
126]. With sustained directional movement, which is unlikely given their generally cryptic behavior and limited energetic reserves, newly settled CoTS could travel only 5 m per week. As juvenile CoTS transition from feeding on CCA to coral, it is to be expected that their distribution and habitat-associations will also change. Notably, coral feeding juveniles are predominantly found in areas with moderate to high cover of scleractinian corals [
112,
116], whereas newly settled individuals targeting CCA will tend to be more abundant in habitats with low coral cover. In Fiji, for example, high densities of newly settled
A. cf.
solaris were first detected immediately behind the exposed reef crest in habitat with very limited coral cover [
112]. The following year, the same cohort of starfish was concentrated along the reef crest, feeding on abundant coral within this zone [
112,
128]. There has not however, been equivalent sampling in other areas to establish the generalities of these ontogenetic shifts in habitat use. It is also very likely that the distribution of juvenile CoTS will be affected by depletion of coral prey by high densities of adults. Moran [
21] suggested that juvenile starfish predominate in shallow-water habitats on reefs subject to outbreaks because large adults generally avoid habitats subject to high levels of wave action and surge. During the initiation of outbreaks, however, CoTS larvae may settle at the base of reef slopes and then gradually move to shallow reef environments as they grow and mature ([
21,
121]; see
Section 2.9).
2.12. Question 16 (Larvae and Juveniles)—What are the mortality rates of larvae and juveniles in the field?
Field-based rates of survivorship/mortality are LARGELY UNRESOLVED, and represent a major limitation in understanding the population dynamics of Acanthaster spp.
While there is some preliminary information regarding mortality rates of CoTS larvae and juveniles from laboratory and experimental studies, estimating natural mortality rates for CoTS throughout the formative stages of their life history (
Figure 3) is extremely challenging. In culture, concentrations of CoTS larvae steadily decline with time [
25,
27,
43,
48], indicative of significant rates of intrinsic larval mortality [
81], which depend on food availability (
Section 2.1) and food quality (
Section 2.3). Mortality rates recorded for larvae in laboratory cultures are substantial [
43]. However, there is increasing evidence that both larvae and juveniles are vulnerable to predation (
Section 2.13), which will further increase rates of mortality. Pratchett et al. [
27] showed that larval survivorship and settlement rates were highest for larvae maintained at intermediate food levels, while higher and lower levels of food availability resulted in higher mortality rates and delayed development. The recent feeding history and nutritional condition of maternal gamete sources of larvae may also interact with larval diet to affect larval survival, growth, and development [
25].
The factors affecting survival during the settlement and metamorphosis are still poorly understood. Rearing of larvae in laboratory conditions and settlement assays show high mortality rates during settlement and metamorphosis [
27,
43,
61,
126]. Yamaguchi [
61] suggested that ~50% of larvae are consumed by epibenthic fauna during settlement (see also [
30]). However, settlement assays on CCA that were carefully cleaned of epifauna still resulted in mortality rates as high as 84% during settlement [
126], which suggests that the physiological condition of competent larvae may influence intrinsic mortality rates. More recent laboratory assays have shown that larval nutritional history influences mortality rates during settlement [
27,
43]. The availability of suitable microhabitats for settlement (
Section 2.10) may also influence mortality rates [
126]; for example, mortality rates increased abruptly in larvae that have not settled after 30 days [
27]. Minor changes in mortality rates before and during settlement can potentially have a significant impact on the eventual adult population size, particularly over the course of several generations, especially given that a single female starfish can produce over 100 million eggs in a single year [
101]. Further investigations of factors influencing mortality rates during these critical phases, under natural conditions, are warranted.
Mortality rates of post-settlement juveniles are likely to be influenced by predation, food availability, and disease [
129]. Keesing and Halford [
130] recorded significant daily mortality rates (~6.5%) for small juveniles (~1-mm diameter) in the field. Mortality rates appear to decline with size, whereby mortality rates for ~3-mm and ~5-mm juveniles were 1.24% and 0.45% per day, respectively [
130]. Visual predators (e.g., reef fishes) have also been implicated as a source of mortality due to the cryptic and nocturnal behavior of juvenile CoTS. However, in a field experiment where laboratory-reared juvenile starfish were placed in an area with suspected fish predators present, Sweatman [
131] found that losses attributable to predation were low (0.13% per day)—much lower than the mortality rate due to predation (1.5% per day) predicted to have an impact on population regulation [
132]. These field studies highlight the importance of epibenthic predators (e.g., [
30]) in regulating population sizes during the post-settlement stage [
133]. The influence of food availability may be more pronounced once CoTS shift their diet from CCA to coral, since herbivorous juveniles are unlikely to be food limited in the field (
Section 2.14). Food availability and the conditions of juvenile CoTS may also influence susceptibility to predation [
126] and disease [
112,
128]. Using hypothetical rates of post-settlement mortality, Keesing and Halford [
126] argued that small changes in post-settlement mortality can have a disproportionate effect on the population size of adult starfish.
2.13. Question 17 (Larvae and Juveniles)—Is predation important in determining the density of larvae and juveniles? What are the main predators of each stage?
These questions are LARGELY UNRESOLVED.
Early field observations [
11] and laboratory experiments [
135] suggested that CoTS larvae are unpalatable to planktivorous fishes, such that predation was considered to exert limited influence on larval mortality. CoTS larvae contain steroidal saponins, which may have specific anti-predatory functions, as demonstrated by experimental assays showing planktivorous fishes discriminating against saponin-impregnated food pellets [
136]. However, more recent experiments have found that planktivorous fishes readily consumed CoTS larvae [
72]. Predation on larvae by scleractinian corals [
2], predaceous zooplankton [
137], and fishes [
11] may therefore be an important determinant of larval survivorship. There have not, however, been any studies aimed at estimating predation or mortality rates of CoTS larvae in the field, and the relative contributions of different groups of predators is unknown. Cowan et al. [
72] showed that planktivorous damselfish may be capable of consuming sufficient numbers of CoTS larvae (up to 158 larvae per hour) to effectively suppress larval settlement, especially when starfish are in low abundance. This level of predation may be sufficient to prevent the onset of outbreaks. However, reef-based predators are likely to be overwhelmed by extremely high densities of larvae [
72], accounting for the propagation of outbreaks once they become established.
Unlike larvae, newly settled CoTS have long been considered to be extremely vulnerable to predation [
21,
138]. Indeed, the highly cryptic and generally nocturnal habits of newly settled
Acanthaster spp. are considered to be adaptations to moderate natural predation rates. Keesing and Halford [
126] demonstrated that epibenthic predators were the major source of mortality for captive reared
A. cf.
solaris that were deployed to field environments within boxes filled with freshly collected rubble. Predation rates were estimated to be 5.05% per day for 1-month old
A. cf.
solaris, which declined to 0.85% per day for 4-month old starfish [
130]. These results suggest that epibenthic predators may be a major factor in regulating local densities of
Acanthaster spp. However, natural predation rates may be moderated by selective settlement within microhabitats with relatively few benthic predators [
30]. Cowan et al. [
30] demonstrated that competent larvae of
A. cf.
solaris were able to detect some predators in the substrate and preferentially settled in microhabitats without predators, where possible. Variation in the abundance of benthic predators may therefore, influence settlement patterns of
A. cf.
solaris, even if they do not cause significant predation mortality in newly settled CoTS [
30].
2.14. Question 18 (Larvae and Juveniles)—Apart from predation what other factors are important in causing the mortality of juveniles (e.g., disease, lack of nutrients)?
This question is LARGELY UNRESOLVED.
Food limitation and constraints on the physiological condition of juvenile CoTS will have consequences for survivorship, though the ultimate factors responsible for mortality will be predation and/or disease. Disease was shown to contribute to mass-mortality of juvenile CoTS in Fiji, which was attributed to an undescribed sporozoan pathogen (intracellular parasite), which ultimately resulted in the extirpation of the entire cohort [
128]. The general susceptibility of juvenile CoTS to disease is largely unknown, but likely depends on the conspecific densities and individual condition, which in turn may be influenced by availability of specific prey. While certain coralline algae may promote higher growth or survivorship (
Section 2.15), crustose coralline algae are considered to be ubiquitous within potential settlement habitats [
139,
140,
141]. Therefore, it is expected to be coral prey that ultimately constrains the growth and survivorship of juvenile CoTS. In the absence of suitable coral prey, juvenile CoTS may continue to feed on a CCA for >2 years [
123,
126], though timely transition to coral prey leads to marked increases in growth. In the wild, it is expected that CoTS that have limited access to coral prey will experience high rates of mortality [
126], though this is yet to be explicitly tested (
Section 2.12). If newly settled CoTS can withstand temporary or localized depletion of coral prey by continuing to feed on coralline algae, these latent populations may proliferate following the recovery of coral populations and assemblages, potentially accounting for the sudden onset of population outbreaks.
2.15. Question 19 (Larvae and Juveniles)—What type of food do juveniles eat in the field? do they show any feeding preferences?
Feeding preferences of juvenile CoTS are POORLY RESOLVED, but the question also needs to be REPHRASED to focus on the fitness consequences associated with differential access to preferred versus non-preferred prey (see
Section 2.16).
Newly-settled
Acanthaster spp. have been reported to feed on a wide variety (at least 12 different species) of coralline algae [
112,
117,
122], including
Lithothamnium pseudosorum and
Porolithon onkodes [
125,
142], though the fitness consequences of settling and feeding on differential species of coralline algae have not been considered. Johnson et al. [
121] suggested that the most preferred coralline algae is
L. pseudosorum but this was based on settlement (rather than feeding) preferences. Higher growth and survival rates as a result of feeding on the preferred species of coralline algae may be fundamental in understanding settlement preferences, as well as accounting for spatial and temporal variation in population dynamics.
Coral-feeding juvenile
Acanthaster spp. do exhibit pronounced feeding preferences as shown by a recent laboratory study [
143]. In this study, juvenile
A. cf.
solaris that were given the choice between eight species of coral (
Acropora formosa, A. millepora, A. tenuis, Pavona cactus, Echinopora lamellosa, Pocillopora damicornis, Stylophora pistillata and Porites lutea) and preferred
Acropora tenuis while avoiding
S. pistillata, E. lamellosa and P. lutea [
143]. While these laboratory studies show that juvenile CoTS do have distinct feeding preferences, field surveys of feeding preferences of juveniles (based on feeding scars on corals upon collection of juveniles;
sensu [
144]), are needed to evaluate the ecological impact of feeding by juvenile CoTS in the field, where prey choices are likely to be influenced by a wide variety of different factors [
21]. For example, juvenile CoTS (1+ year old; mean size ~ 35 mm) sampled from Green Island and Fitzroy Island (Great Barrier Reef) were consistently observed feeding on bushy scleractinian corals such as
Acropora echinata and
Stylophora mordax [
11], which may reflect preference for complex habitats to evade predators rather than inherent feeding preferences.
2.16. Question 20 (Larvae and Juveniles)—Wow fast do juveniles grow in the field? Is it similar to that recorded in the laboratory? How important is diet in determining the growth rate of juveniles?
General patterns of juvenile growth are WELL RESOLVED, but it is unknown whether different species of coralline algae significantly influence growth of algal-feeding juveniles.
Growth rates of
Acanthaster spp. vary with ontogeny, but can also vary in response to environmental conditions (e.g., temperature), food availability and conspecific densities [
123]. On the GBR, growth rates of newly-settled (0+ year)
A. cf.
solaris increase exponentially from 0.03–0.04 mm per day at 1-month to 0.11–0.22 mm per day at 12-months [
34], which is comparable to laboratory-based growth estimates for
A. cf.
solaris [
123]. Growth rates further accelerate after 12-months or as soon as individuals switch from feeding on CCA to scleractinian corals and peak at 20–30 mm per month when
A. cf.
solaris are ~100–200 mm diameter for both wild and captive individuals [
61,
123].
Broad changes in diet (from coralline algae to coral) and overall prey availability have a major impact on growth rates for juvenile CoTS [
61,
123]. Most notably, growth is relatively slow during the algal-feeding phase, but accelerates after switching to coral prey [
41,
61,
112,
117,
123,
145]. Accordingly, Lucas [
123] showed that
A. cf.
solaris constrained to feeding on CCA for 2 years were up to 20 times smaller than counterparts from the same cohort provided with access to coral prey. Given that growth and survival of newly settled juveniles may represent a critical bottleneck to recruitment, more work is required to understand the relative importance of different types of coralline algae. After switching to coral prey, growth rates certainly vary according to availability of different coral prey; laboratory-reared juveniles maintained on an exclusive diet of
Acropora formosa grew at 12.0 mm per month compared 0.1 mm per month for those maintained on a diet of
Porites lichen [
126]. Difference in growth rates may be due to variable nutritional content of specific coral diets. Laboratory-reared juveniles maintained under
Acropora or
Pocillopora feeding treatments grew at the same rate despite consumption of
Acropora being twice as much compared to
Pocillopora [
145].
2.17. Question 21 (Larvae and Juveniles)—How far do juveniles move in the field?
Locomotor capacities of newly settled CoTS are WELL RESOLVED, but there has been limited consideration of rates and patterns of movement by larger juveniles (up to 10–15 cm total diameter).
Research on the movement of newly settled
Acanthaster spp. is limited to short-term experimental studies during which individuals were deprived of access to prey and placed on petri dishes or bare sand. Two-week old juveniles (<2 mm total diameter) placed in a clean dish without food moved at 1 cm per minute [
61], while larger juveniles (19–70 mm total diameter) moved over bare sand at rates of 2.34 to 6.67 cm per minute [
11]. However, CoTS generally settle on or near their preferred prey (CCA) and spend most of their time feeding and hiding, rather than moving [
61,
126]. Keesing and Halford [
130] deployed pervious boxes to reef habitats to measure predation rates on captive reared
A. cf.
solaris. Through the course of these experiments and explicit escape controls, Keesing and Halford [
130] demonstrated that 1-month old starfish move very little (<26 cm) on time frames of 1–2 weeks, though CoTS certainly become more mobile as they grow. Larger juveniles (up to 10–15 cm total diameter), which have increased capacity for movement, but are still very vulnerable to predation, may be expected to frequently move between feeding sites and predator refuges, and may also move over larger distances in search of more optimal habitats. If there are significant ontogenetic shifts in habitat use, it is likely that these occur once starfish attain sufficient size to maximize food intake and energetic reserves, but have not yet invested in reproduction.
2.18. Question 1 (Adults)—Are adults capable of moving between reefs?
Adult CoTS are certainly capable of moving between reefs under certain conditions, but the question needs to be REPHRASED to explicitly consider the maximum distances over which adult CoTS can and do move between reefs.
Rates of movement for CoTS have been extensively studied, mostly to inform the extent of their impacts on coral assemblages and reef ecosystems [
127]. Over small distances (meters), CoTS are capable of moving at 33 to 51 cm per minute [
11,
127,
146], with maximum rates of movement (which probably reflect escape responses) moderated by the size of the starfish and the complexity of the substrate [
127,
147]. If maximum rates of movement recorded on sand [
127] can be sustained, large
A. cf.
solaris could travel up to 520 m per day. Even so, it would take weeks to months for
A. cf.
solaris to move several kilometers between reefs, and longer-term and larger-scale movement will be greatly constrained by habitat heterogeneity, resource acquisition, and diurnally restricted periods of activity. Ultimately, the likelihood of adult CoTS moving between reefs will depend on the distance separating adjacent reefs as well as the nature of the intervening habitat.
When tracked for periods of days to weeks (by relocating uniquely tagged individuals at regular intervals), CoTS move <35 m per day and mostly move only after they have depleted coral prey within the immediate area [
2]. Adult CoTS also tend to avoid crossing open expanses of sand (e.g., [
148]). Sigl and Laforsch [
36] demonstrated that well-fed
A. cf.
solaris remain within shelter, whereas starved individuals more readily leave shelter and travel over sand, presumably in search of food. Suzuki et al. [
149] reported large numbers of adult CoTS moving across shallow sand flats in Ishigaki Island, southern Japan, which were in very poor condition and ultimately became stranded at low tide. For the most part, adult CoTS in good condition and with reasonable access to coral prey will have limited impetus to move, whereas individuals that are starving are probably unlikely to succeed in traversing large distances between reefs, despite demonstrated capacity for detection of reef structures and selective migration toward coral-rich areas via “vision” or chemoreception [
150,
151,
152,
153]. It is very likely that CoTS can and do move between close positioned reefs, especially where there is contiguous reef habitat connecting reefs, but the limited temporal and spatial scales of previous movement studies (as well as the predominant focus on movement within coral habitats) do not really inform the capacity of CoTS to travel large distances between reefs. Acoustic tagging may provide new opportunities to assess the scale and occurrence of inter-reef movement and migrations by adult CoTS, assuming that small acoustic tags can be permanently affixed to the body of these starfish.
2.19. Question 2 and 3 (Adults)—How rapidly do [adult CoTS] grow in the field? Is their rate of growth similar to that recorded in the laboratory? Can the age of a starfish be determined from its size?
These questions are LARGELY RESOLVED, though there are some persistent controversies surrounding the ability to reliably age adult CoTS using biological proxies.
Growth rates of
Acanthaster spp. have long been considered to be extremely plastic [
123,
154]. In the extreme, Lucas [
123] reported a 20-fold difference in the size of
A. cf.
solaris at 2 years of age, depending on whether they did or did not transition from feeding on coralline algae to coral prey. Substantial differences in the size of CoTS within any given population [
59] may, therefore, reflect individual differences in growth, rather than differences in age, whereby the latter would reflect the multiple cohorts within the same population. That said, direct comparisons of size-at-age data from previous studies, including laboratory-based measurements of Lucas [
123], have revealed remarkable consistency in age-specific growth rates [
1]. Growth rates of
A. cf.
solaris are maximized (100–150 mm per year) among 1+ and 2+ annual age classes, and thereafter, follow a von Bertalanffy growth function. There are however, apparent differences in the extent to which CoTS exhibit finite versus indeterminate growth [
154], as well as variation in asymptotic size [
35]. These differences are largely manifest when comparing between outbreaking and non-outbreak populations [
155], whereby growth is increasingly constrained (and potentially finite) during severe population outbreaks due to strong intraspecific competition for food and scarcity of prey resources [
156]. MacNeil et al. [
35] compared the size-structure of
A. cf.
solaris populations across 17 reefs on the GBR and showed that the asymptotic size varied among reefs (from <300 mm to >400 mm total diameter, with smaller asymptotic sizes recorded on reefs with higher CoTS densities.
The sigmoidal growth exhibited by
A. cf.
solaris, combined with variation in their asymptotic size, may obscure the general relationship between size and age, especially for larger and older individuals. For this reason, the capacity to distinguish individual cohorts based on population size-structure and retrospectively establish interannual variation in rates of settlement based on size has been contentious (but see [
157]). Therefore, various size-independent proxies of age have been explored for
Acanthaster spp. including spine length and pigment bands on spines [
158]. Care is needed to consistently sample the longest spines from the upper portion of non-adjacent arms, specifically avoiding regenerating spines and arms [
159]. Stump [
160] used mark-recapture to confirm that spine-banding couplets are deposited annually for
A. cf.
solaris at Davies Reef in the central GBR. While absolute age-estimates based on spine banding still need to be validated, this method enables coarse estimates and comparisons of age-structure (and therefore growth) among discrete populations [
35]. Validation of age estimates is critically dependent upon determining the specific timing of initial band formation [
158], which may vary spatially and especially among distinct species (
Section 3.1).
2.20. Question 4 and 8 (Adults)—How long do adults survive in the field? Do adult starfish enter a senile phase in the field where their growth declines greatly and they become infertile?
Maximum longevity is still unclear, but predominant patterns of growth and longevity are WELL RESOLVED.
Demographics of CoTS populations are strongly dependent on food availability (e.g., [
123,
154]), and may also vary with environmental conditions (especially temperature). In captivity, Lucas [
123] demonstrated that
A. cf.
solaris grew to >300 mm total diameter within 3 years, but then largely stopped growing and reproducing, and mostly died within 4–5 years. The limited size of these starfish and the early onset of apparent senescence were suggested to be experimental artefacts, and at least partly attributable to food limitation. In the wild,
Acanthaster spp. can grow to >750 mm total diameter and live >8 years [
157,
161]. On the GBR, large individuals of
A. cf.
solaris (>600 mm total diameter) have been recorded before the onset of active outbreaks, potentially representing individuals that have lived throughout an entire outbreak cycle [
157]. If so, these individuals may be >14 years old, though it is also possible that these starfish simply recruited during non-outbreak periods and grew quickly or survived longer due to abundant coral prey and limited food competition. For outbreak populations, the maximum size of
A. cf.
solaris is generally <400 mm (e.g., [
35]), which probably reflects constraints on growth and longevity due to local depletion of prey resources and density-dependent mortality [
156,
162]. Even so, there is no evidence of size- or age-specific onset of senility among wild populations [
101,
129]. Babcock et al. [
101] demonstrated that there was an exponential increase in egg and sperm production with increasing size, and no apparent reduction in reproductive investment for individuals up to 500 mm total diameter.
2.21. Question 5 and 7 (Adults)—What are the rates of mortality for adults in the field? Are there any other factors which are important in causing the mortality of adult starfish (e.g., disease)?
These questions are LARGELY UNRESOLVED though there is considerable evidence that CoTS are highly susceptible to disease.
Aside from documented declines in the local abundance of
Acanthaster spp. at the end of outbreaks [
59], there is very limited information on rates (or causes) of natural mortality for CoTS. Moran [
21] proposed a comprehensive field study to explicitly quantify mortality and longevity for
Acanthaster spp., but such studies are still hampered by the limited capacity to tag and/or identify individual starfish over necessary periods of several years, especially during major outbreaks. If or when conducted, such studies should attempt to discern density-dependent effects on population dynamics and individual demography, or at the very least, test for differences in key demographic rates between outbreak and low-density populations. Information relating to the biology and ecology of CoTS in low-density populations is particularly lacking, largely owing to the logistic constraints on gathering sufficient data when starfish are few and far between (e.g., [
14]). Conversely, recurrent sampling of outbreak populations [
112,
128] has revealed marked temporal and spatial variability in the abundance and size structure of CoTS, indicating high rates of mortality among smaller CoTS. Zann et al. [
112,
128] estimated that 99% of CoTS that recruited to the barrier reef off Suva, Fiji, died within 2 years. However, this high rate of mortality may have been anomalous due to the high incidence (10%) of pathogenesis among small and juvenile CoTS.
Echinoderms are particularly susceptible to disease [
163], and disease has been implicated in mass mortalities of numerous species of urchins and starfish (e.g., [
164,
165]). Accordingly,
A. cf.
solaris has been seen to exhibit symptoms of disease, both in captivity [
123,
166] and in the wild [
112,
167]. Background levels of pathogenesis among populations of
Acanthaster spp. appear to be generally very low [
168], though the probability of infections arising, as well as rates of transmission among individuals are likely to increase with population density [
112,
128]. Susceptibility to disease is also likely to increase following prey depletion and declines in the condition of adult starfish, leading to further increases in the incidence and importance of disease after outbreaks are well-established (
Section 2.23). Further research is clearly needed to better understand the ultimate fate of individual CoTS, though it seems likely that most succumb to either predation and/or pathogenesis, even if their vulnerability to such processes varies with size, age, prey availability, energetic condition and behavior.
2.22. Question 6 (Adults)—What is the rate of predation on adults on reefs? What are the main predators of adult starfish? Are these predators sufficient to limit adult population levels? Do the densities of these predators fluctuate markedly through time?
There is increasing evidence that adult CoTS are susceptible to predation, but these specific questions are LARGELY UNRESOLVED.
Adult
Acanthaster spp. were initially thought to be relatively immune to predation due to their elaborate physical and chemical defenses [
136]. However, an ever-increasing array of coral reef organisms have been reported to feed on adult CoTS [
31,
32,
169]. For the most part, these nominal predators have been observed feeding on the remains of dead or moribund adult CoTS [
31] and it is unknown to what extent these predators actually kill adult CoTS. The main predators that are known to kill and individually consume adult CoTS are the giant triton (
Charonia tritonis) and the stellar pufferfish (
Arothron stellatus). The abundance of
C. tritonis is purported to have been much higher prior to extensive harvesting in the 1950s to 1960s, which coincided with the first reported mass outbreak of
A. cf.
solaris on the GBR and prompted concerns that the removal of predators may have caused or contributed to outbreaks (the
predator removal hypothesis; [
170]).
Charonia tritonis is now universally rare on the GBR and on all other reef systems, potentially contributing to increased instabilities in the abundance of
Acanthaster spp. Similarly,
A. stellatus and other large predatory pufferfishes are widespread, but uncommon [
31]. Moreover, pufferfishes are not subject to fishing and are unlikely to vary in abundance among reefs that are open versus closed to fishing. On the other hand, lethrinids, such as
Lethrinus nebulosus or
L. miniatus, are targeted by many fisheries and may also be important predators of
Acanthaster spp. [
31]. Large polyps of the corallimorpharian
Paracorynactis hoplites have also been observed fully ingesting and killing adult CoTS [
171,
172,
173], but the distribution, abundance and rates of CoTS consumption by this highly cryptic predator are unknown. Overall, there is still considerable research needed to identify important predators, let alone establishing their relative preference for CoTS, and respective predation rates at different CoTS densities.
Although there are established methods for measuring relative rates of predation and mortality for echinoderms in the field [
174,
175], there are no empirical data on rates of predator-induced mortality for
Acanthaster spp. For now, relative rates of predation are inferred based on the proportion of CoTS with conspicuous injuries [
32,
169], which are attributed to partial predation. In the Philippines, the incidence of injuries was higher for CoTS populations sampled from inside of marine protected areas (MPAs) where fishing was prohibited, which would be consistent with higher abundance of potential predators. On the GBR, Messmer et al. [
32] found marked inter-reef differences in the proportion of CoTS with injuries, but these differences did not correspond with inter-reef differences in fisheries regulations. It is possible that the incidence and severity of injuries has no relation whatsoever to rates of predator-induced mortality, because (i) rates of regeneration and therefore, persistence of injuries vary depending on the physiological condition and energetic reserves of starfish [
156], and/or (ii) it is an altogether different suite of predators that cause injuries versus outright mortality of CoTS [
32]. High incidence of sub-lethal predation may nonetheless have important effects on the individual fitness and population dynamics of
Acanthaster spp., diverting energy to tissue repair that would otherwise contribute to increased fecundity.
Predation may contribute to population regulation in several ways, including (i) direct reductions in the local densities of juvenile or adult CoTS; (ii) disrupting spawning aggregations [
176]; (iii) reducing individual fecundity through partial predation (V. Messmer, unpublished data); and/or (iv) modifying settlement rates and behavior (
Section 2.13). However, the initiation and spread of CoTS outbreaks cannot be definitively attributed to declines in the abundance of reef-based-predators, caused by sustained and on-going fishing (
Section 2.22). If local densities of
Acanthaster spp. are normally regulated by predation then overall declines in the abundance of predators might be expected to result in gradual and sustained increases in baseline abundance of CoTS [
55], rather than periodic or recurrent outbreaks. This is why Moran [
21] asked whether there are any putative predators that fluctuate markedly in time and potentially account for oscillations in the abundance of
Acanthaster spp. In reality, reduced predation pressure may contribute to increased instability in the population dynamics of
Acanthaster spp., thereby contributing to increased incidence or severity of outbreaks on individual reefs subject to increased fishing pressure [
133,
177]. On this basis, it would be prudent to limit or prohibit fishing in areas known to be important for the initiation of reef-wide outbreaks, though more work is still required to reconcile the specific mechanistic links between fishing and CoTS outbreaks [
101].
2.23. Question 9 (Adults)—What causes the rapid disappearance of adult starfish which has been observed in the field? Is it related to density dependent factors (e.g., crowding causing loss of condition)? What happens to the majority of starfish? Do they die (e.g., from disease) or do they move to another reef?
These questions are POORLY RESOLVED and potentially very important for informing management of population outbreaks.
Rapid and pronounced declines in the abundance of CoTS following major outbreaks [
2,
11,
59] are suggested to occur because starfish either die following extensive prey depletion and subsequent starvation, or move en masse to find alternate sources of prey [
170]. However, precipitous declines in the local abundance of CoTS may [
170] or may not [
178] coincide with comprehensive depletion of scleractinian corals. At Lizard Island (northern GBR) in 1999, localized outbreaks ended even though mean coral cover was still >22% [
178]. Moreover, CoTS can survive without food for many months [
21], and so it seems unlikely that it is a lack of coral per se, that causes elevated mortality or initiates mass exodus from reefs. It is possible, however, that limited access to preferred coral prey (e.g.,
Acropora) leads to compromised health and condition of CoTS [
48,
123], despite relatively high overall coral cover on reefs subject to moderate CoTS outbreaks.
The extent to which CoTS actually move between reefs is largely unresolved (
Section 2.18). Even though starvation is a potential trigger for movement by CoTS [
36] and there will be strong motivation to move away from reefs following extensive depletion of coral prey, by the time this happens, the starfish are likely to have already depleted much of their energy reserves thereby constraining the capacity for long-distance migration. Given the high densities and compromised condition of CoTS towards the end of outbreaks, it seems most likely that that rapid transmission of highly virulent and opportunistic pathogens is responsible for localized population collapse [
22,
112]. Moran [
21] argued that mass-mortalities of CoTS are unlikely to have gone completely unnoticed, though it is possible that diseased starfish seek refuge within the reef matrix and are highly cryptic, or that sick individuals are targeted by opportunistic reef predators (e.g., [
179]).
There is a potentially important link between food availability, the nutritional condition of CoTS, and their vulnerability to opportunistic pathogens [
22], which are highly prevalent on and within the tissues of these starfish [
168]. Mills [
162] showed that CoTS increasingly invest in prophylaxis at high densities of conspecific, where there is an increased risk of infection. While this would be expected to confer greater resistance to pathogenesis, and therefore, reduced relative rates of mortality, it would be maladaptive to increase investment in prophylaxis unless risks were significantly increased. In addition, there are likely to be significant constraints on energy investment for CoTS towards the end of outbreaks following selective depletion of preferred coral prey (e.g.,
Acropora) if not comprehensive coral loss [
178]. If crown-of-thorns starfish continue to invest disproportionately into immune defense even when prey are scarce, energy reserves will be depleted even more rapidly, thereby making individuals even more prone to disease [
180,
181].
2.24. Question 10 (Adults)—Do the skeletal components of starfish accumulate in the sediments after times of outbreaks? Do more spines tend to accumulate during outbreaks than during times when starfish densities are low?
The specific questions posed here are WELL RESOLVED, but skeletal elements cannot be used to resolve the specific incidence and timing of past outbreaks.
Adult CoTS have about 2000 calcareous skeletal elements; these persist in reef sediments and are readily recognizable. It is expected therefore, that fluctuations in the prevalence of CoTS skeletal elements within distinct layers of reef sediments could be used to test whether outbreaks occurred prior to the first documented outbreaks in the 1950s–1960s, and test whether the frequency of outbreaks has changed through time [
182,
183,
184]. However, the use of ossicles to reconstruct the history of outbreaks has been controversial. Initial studies sampled the sediments in the lagoons of 44 reefs spread widely across the GBR region and found higher numbers of ossicles in surface sediments at reefs with active outbreaks of CoTS [
182]. Sub-surface sampling using an airlift found remains of starfish in sediments that dated from more than 3000 years before present, with some suggestion of higher concentrations at 250–300 year intervals [
182]. These results were reassessed by Moran et al. [
185] who concluded that, while the occurrence of skeletal elements confirms that CoTS have been present on the GBR for a long time, these data cannot confirm or refute the occurrence of outbreaks prior to the 1960s.
Walbran et al. [
183] sampled sediments in multiple sites at two reefs in the north central GBR using a vibro-corer which retained the structure of the sediment cores better than the airlift used by Frankel [
182]. Walbran et al. [
183] concluded that CoTS have been present on the GBR for at least 8000 years and that the general density of ossicles was noticeably higher in sediments that were 1000–2000 years old than in the more recent deposits. However, individual outbreaks lasting only a few years could not be resolved due to reworking of sediments. A series of subsequent papers identified potential weaknesses in the study which could undermine these broad conclusions: possible taphonomic changes were not considered [
186], a number of assumptions that are the basis for the link between recent starfish populations and the incidence of ossicles in surface sediments were untested [
187], and differential rates of sedimentation and compaction, both natural and those resulting from the coring process, were not accounted for [
188]. While Henderson and Walbran [
184] point out that many of these potential problems are unsubstantiated, it is clear that the interpretation of the sediment record is not straightforward. Ultimately, bioturbation and differential compaction mean that the position of ossicles in reef sediments is an unreliable indicator of their relative age. A robust estimate of the timing of past outbreaks awaits the development of a method of aging individual ossicles of CoTS at a cost low enough to allow large sample sizes. The high magnesium calcite content of ossicles makes them unsuitable for established techniques such as U-series dating.
2.25. Question 11 (Adults)—Do adults show a distinct preference for certain types of coral?
This question has been WELL RESOLVED.
Numerous studies have reported that
Acanthaster spp. feed predominantly on certain coral taxa, mainly
Acropora and
Montipora, while rarely feeding on other taxa, such as
Porites and
Turbinaria (reviewed by [
1,
21,
22]). Strong selective feeding is expected to cause differential mortality and directional shifts in the structure of coral assemblages, potentially contributing to increased diversity through selective removal of dominant coral taxa [
189]. However, differential consumption of coral taxa may not necessarily reflect inherent feeding preferences [
190]. Potts [
190] argued that field-based patterns of feeding by
Acanthaster spp. are moderated by accessibility to different prey and that they become conditioned to feed disproportionately on locally abundant corals. Moreover, Moran [
21] pointed out that few of the early studies on the feeding habits of CoTS employed methods necessary to document explicit feeding preferences, largely failing to account for the differential abundance or availability of different coral prey. Since that time, there have been several quantitative field studies demonstrating that
Acanthaster spp. consume and deplete different corals disproportionately to their availability [
19,
20,
144,
191,
192], reflective of distinct feeding preferences. Compilation of data (forage ratios) from these distinct studies reveal that
Acropora and
Montipora are consistently consumed more than expected based on their availability [
1]. Conversely, several coral genera (including
Porites,
Pectinia,
Galaxea, and
Echinopora) were generally consumed less than expected based on their availability, though actual forage ratios and inherent feeding preferences vary with concentration and condition of CoTS, as well as the size, abundance and distribution of prey.
While
Acanthaster spp. do exhibit distinct feeding preferences, CoTS outbreaks do not necessarily lead to directional shifts in the structure of coral assemblages [
6], nor to persistent changes in coral diversity [
178]. During major outbreaks, feeding selectivity may be apparent in the sequential depletion of different coral taxa, but even the least-preferred corals (e.g.,
Porites) are consumed and often locally depleted (e.g., [
2,
6,
11]). Selective effects of CoTS feeding on coral assemblages will be most apparent during relatively moderate outbreaks (e.g., [
178]). At Lizard Island in the 1990s, outbreaks of
A. cf.
solaris caused overall declines in coral cover of <30% [
178]. Despite moderate declines in live coral cover, there were marked shifts in the structure of coral assemblages, with disproportionate declines (50–80% depending on species and location) in abundance of formerly dominant
Acropora corals. Nonetheless, coral diversity declined because CoTS were not sufficiently averse to less common taxa.
2.26. Question 1 (Effects On Communities and Processes)—Do coral communities recover from outbreaks of starfish? How long does this take? Is the pattern of recovery similar for most types of reefs and for different scales of disturbance?
Recovery of coral assemblages (both in terms of total coral cover and community composition) is well studied and LARGELY RESOLVED.
Estimates of the time required for coral assemblages to recover from outbreaks of
Acanthaster spp. (and other major disturbances) range from <5 years to >100 years [
193,
194,
195,
196], depending on the spatial extent and magnitude of coral loss, as well as the specific types of corals that are affected. In extreme cases, coral assemblages may never regain their initial structure, even where overall coral cover returns to pre-disturbance levels [
197], owing to fundamental shifts in community dynamics. Completely denuded reefs also recover much more slowly than reefs in which at least some corals survive to grow and reproduce [
198,
199]. Moreover, it is increasingly apparent that major disturbances are occurring too frequently to allow for recovery of coral assemblages in the intervening period [
9].
The recovery of coral assemblages following outbreaks of
A. cf.
solaris was explicitly studied on the GBR based on temporal dissimilarity in taxonomic composition of benthic assemblages on reefs monitored annually from 1993 to 2005 [
200]. While coral assemblages did often exhibit effective recovery from CoTS outbreaks throughout this period, the time taken for community reassembly after outbreaks of
A. cf.
solaris was longer than for other major disturbances, such as severe tropical storms and bleaching [
200]. Notably, recovery also took longer in reef areas where fishing was permitted, where 8–10 years were necessary for coral communities to return to their pre-outbreak composition, compared to an average of 6–7 years inside no-take areas [
200]. This difference was attributed to lower severity of outbreaks and corresponding reductions in the magnitude of coral loss inside no-take areas, potentially linked to greater predation on juvenile or adult starfish where fisheries exploitation was restricted [
133].
2.27. Question 2 (Effects on Communities and Processes)—What effect do outbreaks have on other communities (e.g., fish, soft corals)? Is this effect permanent or do these communities recover from such a disturbance?
These questions are WELL RESOLVED, though the effects of coral loss on reef-associated organisms are informed by studies considering a diverse array of different disturbances.
Coral depletion, regardless of whether it is caused by CoTS outbreaks or by other large-scale disturbances (e.g., climate-induced coral bleaching), has adverse effects across a wide range of marine organisms that depend on corals for food, shelter or recruitment [
201,
202,
203,
204]. Extensive coral depletion also provides increased space for colonisation by soft corals [
178,
205] or algae [
206] which can, in turn, lead to increases in abundance of habitat generalists and herbivores. The specific effects of outbreaks of
Acanthaster spp. on coral reef fishes will depend on the magnitude (extent and severity) and selectivity of coral loss. However, the corals that are most critical in supporting both coral-dwelling (e.g.,
Acropora,
Stylophora and
Pocillopora; [
204]) and coral-feeding fishes (
Acropora and
Pocillopora; [
4]) are the same corals that are preferred by
Acanthaster spp. (
Section 2.25) and are often rapidly and comprehensively depleted during CoTS outbreaks [
6]. Not surprisingly, therefore, declines in the abundance of coral-dependent fishes during outbreaks of
Acanthaster spp., are often disproportionate to overall levels of coral loss [
6,
207,
208].
Approximately 10% of coral reef fishes are directly dependent upon scleractinian coral for food and shelter [
202] and mostly small-bodied fishes with limited fisheries importance [
204]. However, the effects of extensive coral depletion caused by outbreaks of
Acanthaster spp. extend well beyond the few fishes that are directly dependent on live corals for food and shelter [
209], especially where the effects of coral depletion are compounded by structural collapse of three-dimensional habitats and/or increased dominance of macroalgae. In southern Japan, Sano et al. [
209] recorded 65% declines in the abundance and diversity of reef fishes on reefs that were devastated by outbreaks of
A. cf.
solaris. Sheltered reef environments that supported extensive growth of tall staghorn
Acropora corals, providing complex 3-dimensional habitats, were rapidly reduced to flat, homogeneous rubble fields, following the comprehensive consumption of corals by CoTS [
209]. Importantly, fundamental shifts in the structure of coral reef habitats, initiated by severe coral loss, can impact on the abundance of many large-bodied and carnivorous fishes [
208], directly undermining fisheries productivity [
210].
Given their strong reliance on corals and coral-dominated habitats, the abundance and diversity of reef fishes are expected to recover in direct accordance with recovery and reassembly of coral assemblages [
211,
212]. Inherent lags in recovery may sometimes occur due to vagaries in larval supply and recruitment by fishes, and persistent shifts in community structure may occur due to differential rates of coral recovery [
197]. Of greater concern, however, is that the time required for effective recovery relative to projected increases in the incidence of major disturbances [
213], whereby the effects of CoTS outbreaks on coral and non-coral communities will be increasingly compounded by other major disturbances, such as mass coral bleaching. Coral recovery and community reassembly may also be constrained by the occupation of reef substrates by non-coral sessile organisms (e.g., macroalgae) that prevent or hamper growth and recruitment of corals [
214], highlighting the importance of fisheries management to promote ecosystem resilience on coral reefs. Accordingly, Mellin et al. [
200] showed that recovery of both fish and coral communities was faster (<6 years) within no-take areas, relative to reefs that were open to fishing (>9 years).
2.28. Question 3 (Effects on Communities and Processes)—What effect do outbreaks have on reef processes such as calcification, primary production, and reef growth? Question 2 (Overarching)—Whether [CoTS] outbreaks play an important part in reefal processes and the development of reef structure?
The specific effects of widespread coral mortality and corresponding fluctuations in abundance of reef-building corals (regardless of actual cause) on reef processes are WELL RESOLVED.
Net calcification, which is important for rates of reef growth (or accretion) of contemporary reef systems [
215,
216], is highly sensitive to ecological perturbations and changes in the overall abundance of reef-building corals [
217]. Importantly, significant declines in the abundance of corals, and especially fast growing coral species (such as
Acropora) will significantly affect reef carbonate budgets and net framework production [
215,
216]. In the Caribbean, for example, sustained and selective coral loss (specifically, functional loss of
Acropora species) has resulted in >40% declines in rates of carbonate production, such that net carbonate production is below the threshold necessary to sustain positive reef accretion [
215]. While the causes of reef degradation in the Caribbean do not include outbreaks of
Acanthaster spp. (which does not occur in the Atlantic), recurrent and ongoing outbreaks of
Acanthaster spp. may be undermining critical ecosystem process throughout the Indo-Pacific. If coral assemblages can recover and reassemble reasonably quickly following disturbances (
Section 2.26), temporary declines in coral cover and calcification will have negligible effects on carbonate budgets. Periodic mortality and structural collapse of
Acropora corals may actually contribute to reef accretion through increased production of carbonate sediment [
218]. However, outbreaks of
Acanthaster spp. are a major contributor to sustained declines in the abundance of reef-building corals recorded at key locations throughout the Indo-Pacific (e.g., GBR [
7,
8]; Japan [
16]; Indonesia [
20]; Guam [
219]; Maldives [
17]; French Polynesia [
6,
9]), and certainly have disproportionate impacts on faster-growing corals (
Section 2.25). Functional replacement of reef-building corals by other habitat-forming sessile fauna and flora, may partially offset declines in primary productivity and carbonate production due to widespread coral loss. However, it is now unequivocal that reef ecosystems with very limited (<10%) cover of reef-building corals support a fraction of the species found in coral-rich habitats [
220], which has significant consequences for ecosystem function and productivity.
2.29. Question 1 (Overarching)—Why [CoTS] outbreaks occur and whether they are natural or unnatural phenomena?
This question has preoccupied much of the discussion around Acanthaster spp., but remains LARGELY UNRESOLVED.
The extent to which outbreaks of
Acanthaster spp. are caused or exacerbated by anthropogenic activities (e.g., inputs of nutrients and pollutants, and overfishing of key predators) is widely disputed [
1,
71]. Fundamentally, the unique and extreme life-history characteristics of
Acanthaster spp., such as exceptional fecundity recorded for
A. cf.
solaris [
101] predispose them to major population fluctuations [
103]. However, when outbreaks of
Acanthaster spp. were documented in the late 1960s [
2,
11], it was immediately assumed by many scientists that these were new and unprecedented phenomena, such that scientists were compelled to link the sudden occurrence of outbreaks to sustained and ongoing degradation of coastal environments, due to coastal development [
2], land-based run-off of nutrients and pollutants [
221], and/or exploitation of marine species [
222]. Several scientists (e.g., [
137,
223,
224,
225]) did argue that outbreaks of
Acanthaster spp. were probably a natural phenomenon that had occurred across the Indo-Pacific well before the 1960s.
While there are no rigorous quantitative estimates of CoTS densities prior to the 1960s, historical accounts, mostly from anecdotal information, support the view that outbreaks have occurred in the past [
223,
224,
226]. In particular, there is information suggesting that CoTS were unusually common at certain locations in the 1940s [
113], though some of these reports may refer to normal spawning aggregations of CoTS [
227], rather than actual outbreaks. Birkeland [
228] and Flanigan and Lamberts [
229] also proposed that the significance of CoTS in Micronesian and Samoan cultures could be indicative of previous instances of high abundance. However, Moran [
21] argued that the cultural importance of
Acanthaster spp. could be a result of its sinister appearance and toxicity, rather than periodic abundance. On the GBR, it appears likely that outbreaks of
A. cf.
solaris were underway in 1913, given the relative ease with which Clark [
230] collected CoTS in the Torres Strait. It is unknown however, whether there were progressive waves of outbreaks that propagated the length of the GBR, and there is insufficient information to establish the timing and spread of outbreaks that occurred prior to the 1960s.
Even if outbreaks did occur on the GBR prior to the 1960s, it has been suggested that the frequency and/or intensity of outbreaks is now much higher than it was in the past [
45,
113,
231]. Fabricius et al. [
45] refer to a model of coupled oscillations between coral cover and CoTS densities, suggesting that the incidence of outbreaks has increased from one outbreak in 50–80 years to one in 15 years over the last 200 years, attributing this increased incidence to higher nutrient loads from river discharge. The principal basis of these assertions is that long-term development of the GBR would not have been possible given the current frequency and severity of CoTS outbreaks (see also [
221]). However, the increased susceptibility of reef ecosystems to outbreaks of
Acanthaster spp. and protracted periods required for subsequent recovery and reassembly of contemporary coral assemblages is not in itself evidence that outbreaks are unnatural [
22]. Rather, other factors, such as chronic threats posed by increasing anthropogenic activities (e.g., fishing and harvesting, sedimentation, eutrophication, and pollutants) may have undermined the capacity of reef ecosystems to withstand natural disturbances [
232], eroding ecosystem resilience and altering population and community responses to persistent and ongoing disturbances. The best (albeit inferential) evidence that there have been temporal shifts in the incidence and/or intensity of CoTS outbreaks comes from cores of massive
Porites and
Diploastrea colonies, which purportedly record the incidence of CoTS feeding scars throughout their lifespan [
231]. DeVantier and Done [
231] concede that the ability to discern previous outbreaks declines as you extend further back, even when comparing among known outbreaks events in the 1960s and 1970s. Nonetheless, there is evidence of outbreaks occurring prior to the 1960s, and mainly in the 1930s. Devantier and Done [
231] conclude that outbreaks of CoTS have been a persistent feature of the GBR for centuries, but may have gone from isolated and sporadic events in the 1930s to frequent and widespread events since the 1960s.
Establishing whether outbreaks of CoTS are caused (or exacerbated) by anthropogenic activities was considered fundamental in addressing the dilemma of whether to regulate population densities of
Acanthaster spp. and prevent future outbreaks [
233]. In reality, the decision to aggressively defend coral reef ecosystems against the devastation caused by CoTS outbreaks has already been made, as evidenced by intensive and extensive culling programs in operation throughout the Indo-Pacific, wherein over 17 million CoTS have so far been killed and/or removed from reefs across the Indo-Pacific [
1]. The more important questions that must be addressed are whether increased actions to improve water quality (specifically, addressing land-use practices to reduce nutrient inputs) within reef environments and/or increased fisheries restrictions can reduce the frequency or intensity of future CoTS outbreaks. We also need to consider whether these indirect management actions are ultimately going to be more effective than direct intervention (e.g., culling programs) in minimizing the incidence of outbreaks and reducing coral loss at various timeframes. Given persistent uncertainties surrounding the proximal cause(s) of outbreaks and the likelihood that multiple factors will be involved in the initiation and subsequent spread of outbreaks [
57,
71], it seems prudent to maintain a multipronged approach to managing outbreaks of
Acanthaster spp. ([
18];
Section 2.33). Nevertheless, intensifying efforts to improve water quality, as well as redressing over-fishing on coral reefs, are important regardless of whether they ameliorate the threat posed by outbreaks of
Acanthaster spp. Meanwhile, evaluating the relative roles of these factors in initiating outbreaks of CoTS will require more integrated and intensive monitoring to explicitly resolve spatial and temporal gradients in biological communities and environmental conditions (
Section 2.2) relative to the larval abundance, settlement rates, and post-settlement survival of CoTS [
71].
2.30. Question 3 (Overarching)—Why some reefs are more susceptible to [CoTS] outbreaks than others?
This question refers to differences in the occurrence and severity of outbreaks among reefs within the same reef system (e.g., the GBR), which is LARGELY UNRESOLVED
On the GBR, outbreaks of
A. cf.
solaris are only ever recorded on a specific subset of reefs [
21]. Outbreaks are not, for example, recorded on most nearshore reefs and are only rarely recorded on outermost reefs [
21]. The reasons for these patterns are often discussed, but generally unknown. The specific factors that influence susceptibility of reefs to population outbreaks of
Acanthaster spp. will vary depending on whether outbreaks arise independently (primary outbreaks) or result from extensive delivery of larvae spawned by high-density populations on nearby or upstream reefs (secondary outbreaks). The independent initiation of outbreaks on individual reefs is likely to result from the progressive accumulation of starfish over multiple cohorts [
59], which will be conditional upon entrainment of larvae and sustained levels of self-recruitment [
57]. The initiation of primary outbreaks may also be promoted by local depletion of putative predators (through fisheries exploitation and trophic cascades [
133,
177]) leading to increased survival of larvae, settlers, juveniles or adult CoTS within reef environments, and/or localized nutrient enrichment, due to river discharge [
45] or upwelling of nutrient rich waters [
234], which overcomes normal food-limitation and facilitates increased development and survivorship of CoTS larvae [
55]. Outbreaks of
A. cf.
solaris on the GBR are initiated on mid-shelf reefs between Cooktown and Cairns, and mainly in the northern portion of this area (commonly referred to as the “initiation box”). This region is characterized by high densities of individual reefs and highly restricted water movement [
58], which will promote steady and sustained increases in local densities of
A. cf.
solaris during successive years of spawning. However, limited spatial and temporal resolution in monitoring means that it is unclear where exactly outbreaks originate. This prohibits meaningful comparisons of putative predator densities or nutrient concentrations among reefs that do and do not sustain primary outbreaks. There is also very limited monitoring of biological communities and environmental conditions at spatial and temporal scales relevant to explain the initiation of CoTS outbreaks (
Section 2.1 and
Section 2.2).
For secondary outbreaks, the predominant factor that will influence (or at least fundamentally constrain) when and where outbreaks arise is the extent of larval delivery via hydrodynamic connectivity [
58,
83]. That said, the delivery of high densities of CoTS larvae to individual reefs would not necessarily result in the establishment of population outbreaks if there were local constraints on larval survival and development or high rates post-settlement mortality (
Figure 3). Previous discussions on the role of river discharge and nutrient pulses on the GBR have focused largely on the initiation of primary outbreaks [
45]. If, however, larval development and survival are severely constrained by very low nutrient concentrations and limited food availability (
Section 2.1), it seems logical that this would limit both the initiation and spread of CoTS outbreaks [
1,
29]. Pratchett et al. [
1] suggested that primary outbreaks may propagate over extended periods independent of any major flood events, whereas it is the subsequent spread of outbreaks that might benefit from major flood events, due to enhanced food availability and elevated larval survival across large areas of the reef [
29]. Even if there are large numbers of well-fed and competent larvae arriving at a reef, settlement and post-settlement survival might be constrained by habitat structure, availability of coralline algae or coral prey, as well as the local abundance putative predators (
Figure 3). Specific habitat requirements and settlement preferences are yet to be established for
Acanthaster spp. (
Section 2.9), though it is likely that there will be inter-reef variation in settlement and survival rates corresponding with differences in reef topography, habitat availability, and also the recent disturbance history. Importantly, there are expected to by interactions between CoTS outbreaks and other disturbances that cause coral loss and habitat degradation [
71]. In general, outbreaks might be expected to be less likely to arise on reefs where coral cover has been supressed by recent distrubances, but these disturbances might also increase local availability of critical settlment habitat (dead, but intact coral skeletons, and/or coral rubble). Outbreaks of
A. cf.
solaris have certainly occurred on reefs with low coral cover in the aftermath of major disturbances (e.g., GBR [
59], Guam [
219], Okinawa [
235]), and contribute to further coral loss.
2.31. Question 4 (Overarching)—Why some [CoTS] outbreaks cause extensive coral mortality while others do not?
This question is LARGELY RESOLVED, though more research is needed to explore species–specific differences in biology and behavior (
Section 3.1).
Although outbreaks of
Acanthaster spp. are capable of causing extensive coral depletion over vast areas (e.g., [
2,
11,
236,
237]), outbreaks vary greatly, not only in the size and density of starfish, but also in their effects [
21]. Most notably, high densities of
A. cf.
solaris occurred for more than 18 months (1969–1970) at Molokai Island, Hawaii, but had negligible effect on local coral assemblages [
238]. Within the Pacific, devastating effects of CoTS outbreaks on coral assemblages are mainly restricted to the central and western Pacific, including French Polynesia [
6,
9], Australia’s GBR [
7,
11], Micronesia [
2,
237], and southern Japan [
16,
239,
240]. In contrast, CoTS outbreaks cause minimal coral loss in the eastern Pacific [
138,
241]. These patterns might be explained by the relative dominance of
Acropora spp. [
22];
Acropora spp. tend to dominate coral assemblages in the central and western Pacific, and are consistently among the corals that are first and worst affected during outbreaks (e.g., [
19,
178]). In the north and east Pacific (e.g., the main Hawaiian islands and Panama)
Acropora is relatively scarce and coral assemblages tend to be dominated by
Pocillopora, which is much less susceptible to CoTS attack [
138,
241,
242], owing to the defensive behavior of infauna, especially
Trapezia crabs.
Geographic variation in the effects of
Acanthaster spp. may also result from differences in the population dynamics and behavior among the four nominal sister species distributed in different parts of the Indo-Pacific [
38,
39].
Acanthaster spp. from throughout the Indo-Pacific ostensibly look and behave the same way, but devastating impacts of crown-of-thorns starfish appear to be confined to the Pacific, which is the geographical range of
A. cf.
solaris [
39,
243]. This warrants explicit comparisons of reproductive and larval ecology, demographic rates, feeding rates and feeding preferences among
Acanthaster spp. from each of the four distinct sub-populations identified by Vogler et al. [
38], extending the studies in the Pacific and the Red Sea to both southern and northern Indian Ocean regions.
2.32. Question 5 (Overarching)—How [CoTS] outbreaks are propagated over large distances?
This question is ambiguous, but is assumed to refer to the apparent coincidence of outbreaks and population connections across widely separated locations, which has Been Largely Resolved.
The propagation of CoTS outbreaks among adjacent reefs and within reef systems is variously ascribed to larval dispersal (
Section 2.5 and
Section 2.6) versus inter-reef movement by adult starfish (
Section 2.18), depending on relevant inter-reef distances. At smaller spatial scales (100 s to 1000 s of meters) it is conceivable that connectivity among reefs is achieved through movement of adults [
113], whereas large-scale dispersal is largely, if not exclusively, achieved through dispersal of planktonic larvae. Planktonic larvae may also be dispersed on oceanic currents to provide connections among widely dispersed locations. Indeed, the pan-Pacific range of
A. cf.
solaris is a potential indicator of its broad dispersal capability [
88]. In the 1960s, and again in the late 1970s, outbreaks of
Acanthaster spp. occurred more or less synchronously across multiple locations throughout the Indo-Pacific [
21] suggesting that these outbreaks were inter-connected. The only viable mechanism that would enable connections among widely separated locations, leading to simultaneous or successive outbreaks in discrete locations, is large-scale dispersal of very large number of larvae. Indeed, outbreaks of
A. cf.
solaris that occurred in Central Province, Papua New Guinea in the early 2000s likely represented the ultimate and inevitable conclusion of the northerly progression of outbreaks on the GBR throughout the 1990s [
19]. However, Timmers et al. [
88] explicitly tested for larval dispersal among distinct geographic regions in the Pacific by examining genetic structure of the highly variable mitochondrial control region (mtDNA). While there was evidence of very occasional larval exchange among geographic regions, there was no possibility that outbreaks were propagated from one region to another through mass larval dispersal. Strong genetic structure, indicative of limited larval exchange, was particularly apparent at scales of >1000 km [
88], suggesting that the simultaneous occurrence of outbreaks across a broad range of locations are triggered by large-scale climatic features, such as ENSO [
244] or increased temperature [
113]. Moreover, the increasing incidence of ENSO anomalies since the 1960s may account for increased incidence of CoTS outbreaks since that time [
231]. However, searches for environmental triggers of CoTS outbreaks, based on spatiotemporal correlation have been hampered by imprecise accounts of when outbreaks actually started across different locations [
231]. The apparent coincidence in the occurrence of outbreaks may simply result from increased research and reporting following reports of renewed outbreaks at key locations. Even Moran [
21] conceded it is unlikely that discrete and disparate outbreaks of
Acanthaster spp. originate as a result of the same single process.
2.33. Question 6 (Overarching)—Whether special management policies need to be formulated in order to prepare for the occurrence of future [CoTS] outbreaks?
There is an unequivocal need for increased proactive (cf. reactive) management of CoTS outbreaks, but the specific management policies and strategies are LARGELY UNRESOLVED and yet to be implemented.
Contemporary management of CoTS outbreaks is largely focused on culling or removing adult starfish, with the intention of minimizing ongoing coral loss through reductions in the number and size of adult starfish and/or containing the spread of outbreaks. To address the current outbreak on the GBR, for example, a targeted control program is in place that kills upwards of 100,000
A. cf.
solaris each year. Currently, the most efficient and accepted method for culling is to individually inject starfish with specific chemicals [
37,
179,
245] that cause immediate and comprehensive mortality. Manual controls are very labor intensive, though the recent development of a single-injection method [
246] has increased the efficiency of in-water culling programs by at least 250%. Single-injection methods also eliminate the need to manually handle adult CoTS, which is purported to result in spontaneous spawning, though this seems unlikely given the delayed responses of CoTS to specific spawning inducers during experimental studies [
52]. Single-injection methods are now so efficient that major constraints on effective population control relate to detectability of outbreak populations [
247], which is contingent upon both timely surveillance and improved understanding of the spatial dynamics (within and among reefs) of CoTS populations. Even with the current suite of tools, there are significant opportunities to further improve the efficiency and therefore, effectiveness of CoTS control programs (e.g., “CoTSBot” [
248]). The concern however, is that effective control of CoTS outbreaks across the entire extent of the GBR will be prohibitively costly. Moreover, ineffective control (e.g., incomplete eradication at specific locations) may simply prolong the outbreak and fail to actually protect local coral assemblages. In reality, outbreaks do not affect all reefs (
Section 2.30) and never occur simultaneously across the entire expanse of the GBR [
56]. Moreover, outbreaks on most reefs can be traced back to initial outbreaks on a few discrete reefs within the initiation box [
56], such that timely investment and focused management activity in these areas could contain the spread of outbreaks and minimize reef-wide coral loss.
Despite the consistent and recurring patterns of CoTS outbreaks on the GBR [
1,
56] there have been very few management policies that have been specifically formulated to prepare for the occurrence of future outbreaks. This lack of planning, combined with inevitable diversion of research and management focus during non-outbreak periods [
249] and limited capacity to detect the early onset of outbreaks, results in inevitable delays in responding to new outbreaks [
250]. The issue is even more complex and more pronounced outside of the GBR where there is no apparent pattern to the timing and location of outbreaks. A proactive management policy with dedicated funding that can be immediately accessed when initiation of future outbreaks are imminent or actually detected is sorely needed and currently lacking [
249]. Ongoing control programs across the Indo-Pacific are estimated to have cost up to US
$44 million [
1], and have been largely ineffective in protecting reef systems from outbreaks of
Acanthaster spp. once they become established [
247]. Timely intervention and the containment of outbreaks before they can spread may, therefore, save greatly on management costs and increase management effectiveness (e.g., [
148,
251]). Established disaster funds have proven successful in the Australian agriculture sector to support pro-active monitoring, early detection and early prevention of locust plagues [
252]. Under this scenario, farmers locally monitor and pre-emptively control locusts at the initiation of an outbreak (and are reimbursed by the fund) and the government targets high-risk locations. A similar commission should be established for
Acanthaster spp. to coordinate early detection and rapid response for future outbreaks. Such a fund needs to have well established trigger levels for action and to be well resourced given the significant cost and efficiency benefits associated with rapid response.
Timely intervention to manage new and renewed outbreaks of
Acanthaster spp. is partly constrained by the reliance on detection of elevated densities of relatively large starfish to signify the onset of outbreaks [
56,
59,
253,
254]. The development and refinement of early-warning systems, focused on measuring larval densities, settlement rates, and/or local abundance of newly settled juveniles is, therefore, a priority not only for improved understanding of population dynamics (
Section 2.7), but also for management. Moreover, it is important to maintain research and management traction throughout non-outbreak periods (
Figure 4), both to address significant knowledge gaps pertaining to the dynamics of non-outbreak populations and to consider additional preventative actions (e.g., sustained culling of low density populations) that may further increase management effectiveness.
3. New Directions
While many of the questions posed by Moran [
21] regarding the biology of CoTS and management of outbreaks remain pertinent, there are several emerging issues and research methods that have deflected much of the recent research attention. For example, when Moran’s [
21] review was published, taxonomists concurred that crown-of-thorns starfish found on coral reefs throughout the Indo-Pacific (including the Red Sea) were just one species,
Acanthaster planci (Linnaeus 1758). However, molecular sampling throughout the last 30 years has suggested that there are multiple species of
Acanthaster that inhabit coral reefs [
38,
87,
255,
256]. Most notably, Vogler et al. [
38] sampled CoTS from the Red Sea to the eastern Pacific, and revealed at least four strongly differentiated clades, which has stimulated necessary research into the systematics and nomenclature [
39] and raises many important questions about the biogeography of CoTS. Moreover, genomic data are providing unprecedented insights into the intrinsic mechanistic basis of CoTS behavior [
176], providing new options for population control and management. It is these fields, along with advances in population modelling that are expected to advance understanding and management of CoTS outbreaks in coming years.
3.1. Systematics and Biogeography
It is now unequivocal that coral reef CoTS, which were formerly referred to as
A. planci throughout the Indo-Pacific (including the Red Sea) comprise at least four distinct species [
39]. Most notably,
A. planci occurs throughout the northeastern Indian Ocean, from the Sea of Oman to Christmas and Cocos Keeling Islands and is both genetically and phenotypically different from
A. cf.
solaris [
38,
256], which occurs throughout the Pacific and the Indo-Pacific archipelago. These two species co-occur and potentially hybridize at Christmas and Cocos (Keeling) Islands [
257]. The third major species (
A. mauritiensis) is generally restricted to the southwestern Indian Ocean, but does co-occur with
A. planci in the Oman Sea [
38]. There is also a fourth distinct species restricted to the Red Sea, which is yet to be formally described and named [
39]. The relatively high (8.8–10.6%) genetic distance among the four-aforementioned species, based on the COI marker used for ‘barcoding’, suggests that these species diverged 1.95 to 3.65 million years ago [
38]. Within the four nominal species, genetic divergence was greatest for the Pacific population [
39], and other studies have revealed conspicuous genetic structure when comparing among CoTS from different regions across the Pacific [
88,
99]. Moreover, distinctive phenotypes of
Acanthaster spp. have been recorded from the southern (Lord Howe Island) and northeastern Pacific (Hawai’i) suggesting that there may actually be more than one species in the Pacific [
39]. A recent report by Yuasa et al. [
258] also found
A. cf.
solaris and its short-spined sibling species,
A. brevispinus on the same reef in Kushimoto, Japan, which is in contrast with the previous assertion that natural interbreeding between these genetically compatible species is blocked by ecological isolation (different habitats) in regions of sympatry [
259]. This suggests that other possible blocks against interbreeding between these two distinct species may be present.
Confirmation that there are multiple CoTS species, which are geographically separated, raises important questions about generality of prior research, whereby much of the research on CoTS has been conducted in the western Pacific on
A. cf.
solaris (see
Section 2.1,
Section 2.2,
Section 2.3,
Section 2.5,
Section 2.10,
Section 2.11 and
Section 2.16). Interestingly, devastating outbreaks of
Acanthaster spp. are also reported mainly from the western Pacific [
21], suggesting that species-specific differences in feeding behavior and biology (e.g., reproductive potential) may account for geographic variation in the occurrence of outbreaks as well as their impacts on reef ecosystems [
1]. If there are significant interspecific differences in the biology of
Acanthaster spp., this may also provide significant insights into the intrinsic factors responsible for population outbreaks. Immediate priorities for comparative demographic studies among species would include: (i) estimates of size-specific fecundity, (ii) larval development and competency periods, and (iii) growth functions. Importantly, these demographic processes have already been quantified for
A. cf.
solaris [
27,
34,
35,
101,
129] and are key to understanding population dynamics.
3.2. Genetic and Genomic Sampling
High throughput sequencing technologies (HTS) are providing increasing opportunities to use genomic data to address ecological questions. As such, HTS has been used to describe molecular ecology and evolution, molecular mechanisms of development in animals, and how environmental factors such as those related to climate change influence animal life history. Methods such as RNA-Seq using Illumina technology have been widely used in many marine invertebrates, including corals [
260] and echinoderms to detect changes in gene expression [
261], or describe genetic variability among populations to assess future adaptation to global change [
262]. The era of genomics on CoTS using HTS technologies has just started to emerge. By sequencing the transcriptome of the male gonad of CoTS, using an RNA-Seq approach, key candidate genes involved in reproduction were identified [
263]. This study found that CoTS contain proteins, molecules, signalling pathways and key developmental genes that are known to have a role in sperm motility and signalling in other echinoderms [
263]. A recent study using genome sequencing and proteomics in combination with behavioral experiments, allowed the identification of key species-specific pheromones involved in their aggregation [
176]. Although this result provides a potential solution to control adult populations of CoTS in the GBR, little is known about the molecular basis underlying CoTS life history stages in response to ecological factors. Therefore, future studies using HTS approaches in combination with ecological experiments are necessary. A complete developmental transcriptome will be essential for the identification of (1) key genes and signalling pathways involved in CoTS developmental traits; (2) response to environmental factors addressed in this review; and (3) for understanding the molecular mechanism of calcification in CoTS. Moreover, genome sequencing of closely related species could provide a comparative genomic approach for population genomics and evolution within this group of animals. Other approaches such as eDNA could also serve as a tool to trace the distribution of early life stages of CoTS in the field [
53,
107,
264]. These data will not only contribute to a better understating of CoTS genomics, but will also be required for effective conservation and management.
3.3. Advances in Modelling
There have been significant recent advances in modelling CoTS populations (relevant to demographic and/or dispersal models), building on a long history of theoretical treatment of outbreak dynamics and the spread of outbreaks [
265]. Previous demographic models were mostly developed at a reef-scale and typically incorporated demographic processes and predator-prey relationships, but not dispersal [
266,
267]. For example, Morello et al. [
268] developed a model of intermediate complexity for ecosystem assessment (MICE) based on trophic interactions between CoTS and slow versus fast growing corals for Lizard Island. Although this model fit with observed population dynamics based on AIMS LTMP data [
133], it implicitly assumed low immigration and self-recruitment levels and failed to capture major peaks in CoTS abundance. The semi-individual based model developed by Chen et al. [
269] has further refined those predictions and was able to reconstruct historical abundances on a set of three reefs (including Lizard Island), however such a model remains too data demanding and computationally intensive to be transferred to the entire Great Barrier Reef. Furthermore, this model does not account for connectivity via larval dispersal, though it could be added.
At a regional scale, the development of large-scale hydrodynamic models for the GBR gave researchers the opportunity to recreate the initiation and spread of outbreaks [
82,
83,
84,
93]. However, Wolanski [
270] suggested that these predictions were built upon unrealistic assumptions and parameterization of the dispersal process and should therefore not be used for management. Despite this, these hydrodynamic models provided the foundation for Scandol’s [
271] interactive metapopulation models for CoTS management. Another hydrodynamic model was proposed by Bode and Mason [
272] and was used to define self-recruitment and connectivity for 321 reefs on the GBR [
273], and in turn model the effect of increasing nutrients on CoTS outbreaks and coral cover over a 150 year time period [
45]. This showed that moderate increases in nutrient availability can drastically increase larval survival and reduce coral cover. Importantly, these models were designed to predict population dynamics under a specific scenario (i.e., varying chl-
a levels) while keeping other parameters constant; however, such parameters were not calibrated against empirical data. Furthermore, such models did not account for uncertainty and how it propagated through the different steps, limiting their usefulness in terms of management and in developing decision-making strategies.
Recent developments in the hydrodynamic modelling for the GBR (e.g., eReefs) have reinvigorated attempts to understand CoTS population dynamics and provide management solutions. A connectivity network model for CoTS on the GBR based on hydrodynamic models generated from eReefs [
58] aimed to identify the most important areas for initiating and spreading CoTS outbreaks via their relative importance within the network. This approach has since been used as the basis of an adaptive management strategy, whereby reefs targeted for control are selected based upon their likelihood to spread future outbreaks [
274]. While it is a commendable and practical approach to inform management decisions, this model is solely based on dispersal and fails to incorporate demography or important biotic interactions [
275]. Furthermore, if models such as these are to be used for management, it is essential for them to be first validated against empirical data, and to account for uncertainty and its propagation throughout the model.
This recent progress in the field of CoTS modelling highlights important directions for future research: (i) despite the fact that there is no one-size-fits-all model, CoTS models should be better integrated across multiple scales so that those developed at reef scales can inform regional models (e.g., role of CoTS behavior, such as aggregation, on spatial distribution patterns). In turn, regional models provide a framework for defining conditions at the boundaries of reef-scale models (e.g., immigration rates from neighbouring reefs); (ii) demographic models should combine multiple and diverse sources of empirical data and specifically highlight where there is limited relevant information [
275]. This will provide an opportunity for better interactions between CoTS modellers and biologists to prioritize biological research; (iii) for CoTS models to be useful in terms of management, there needs to be a better appraisal of uncertainty (both data- and model-driven) and how it propagates though the different steps of the model. This implies the need to steer away from purely deterministic models and, instead, account for both environmental and demographic stochasticity. By documenting the uncertainty stemming from each step of the model and each scenario, more transparent CoTS models will allow managers to assess the impacts of different management strategies while accounting for the full range of their possible outcomes.
3.4. CoTS and Climate Change
Acanthaster spp., as well as their coral prey, are increasingly subject to environmental change caused by anthropogenic forcing in global climate systems. This issue was brought to the fore, during the recent mass bleaching on the GBR [
276], which is expected to have resulted in levels of coral mortality to rival the cumulative impact of entire cycles and reef-wide outbreaks of
A. cf.
solaris. Widespread and significant bleaching [
276] and subsequent coral mortality throughout the initiation box may ultimately constrain the initiation of future CoTS outbreaks, which are expected to occur in the early 2020s. However, projected changes in ocean temperatures and seawater chemistry are also expected to have direct effects on
Acanthaster spp. [
23,
24,
42,
74,
75,
76,
78] and juveniles [
74,
124,
277] especially during early life-history stages.
The optimal temperatures for embryonic and larval development of
A. cf.
solaris (28–29 °C), reflects ambient temperatures currently experienced during the reproductive season in the northern GBR [
24,
76]. Above these temperatures embryonic abnormality and mortality increase [
76,
78] and larval growth is impaired, as evident in the reduced size of the brachiolaria [
75]. Without acclimation to changing climate, even moderate ocean warming (1–2 °C) is therefore, likely to impose significant constraints on reproduction and settlement rates. When ocean acidification is also considered, warming (+2 °C) and reduced pH (pH 7.6–7.8) have additive negative effects, reducing larval size [
75]. As a single stressor acidification increases developmental abnormality in CoTS and reduces larval growth [
74]. Settlement by CoTS was also negatively affected when coralline algae were grown in low pH conditions [
74]. These negative effects on early development may be the bottleneck for population maintenance of CoTS in a changing ocean.
In contrast to larvae, juvenile CoTS are highly tolerant to increased temperature (+2 °C above ambient) and resilient to acidification (pH 7.6) [
124]. Growth and feeding rates of the algal-feeding juveniles were highest at 30 °C and pH 7.6. While growth increase at higher temperature is likely due to the direct effects of warming on physiology, faster growth in ocean acidification conditions was not expected. This was largely attributable to indirect effects of acidification on coralline algae [
277], which was more palatable (less calcified) and had a higher nutritional value (C:N) when grown at low pH. The cumulative effects of environmental change on individual fitness and population viability of
Acanthaster spp. still needs further consideration, along with explicit research into the vulnerability of coral feeding juveniles and adults to ocean warming and ocean acidification. However, any such effects may be largely irrelevant, given that climate-change poses a real and considerable threat to the availability of coral prey [
276], and has disproportionate impacts on
Acropora and other corals that are favored by
Acanthaster spp. Importantly, the loss of preferred coral prey will lead to declines in the quality and quantity of progeny [
25,
48], with significant consequences for population replenishment regardless of any direct effects of environmental change.
4. Discussion
Despite persistent controversy surrounding the specific cause(s) of outbreaks of
Acanthaster spp. [
1,
29], there has been substantial research (>950 publications) on
Acanthaster spp. over the past 30 years, leading to major advances in knowledge of their biology and ecology, as well as increased understanding of the effects of CoTS outbreaks within reef ecosystems (
Table 1). In all, we suggest that 59% (24 out of 41) of the questions posed by Moran [
21] have been effectively addressed and largely resolved in the last 30 years (
Table 1). Many of the questions that are still largely unresolved relate to ecological processes (e.g., food-limitation and predation) that pertain to population regulation. While these questions are critically important in establishing the fundamental cause(s) of outbreaks of
Acanthaster spp., a large part of the reason why these questions have not been effectively addressed is that the required research will be logistically very challenging. For example, measuring survivorship of CoTS larvae in the field is not really tractable, and it is clear that experimental studies conducted under laboratory conditions provide very limited insights into natural rates and causes of larval mortality (
Section 2.12). There are however, some unresolved questions that are not only tractable, but will contribute directly to increased management efficiency and effectiveness (
Table 1), and it is these research topics that should be given immediate priority. Notably, new technologies and opportunities to quantify the temporal and spatial patterns in the abundance of CoTS larvae [
53,
54], along with other established methods for measuring settlement rates [
115] should be incorporated into intensive and ongoing monitoring programs to provide an early warning system for new and renewed outbreaks of
Acanthaster spp. (
Section 2.33).
A large portion of recent studies on
Acanthaster spp. are confirmatory in nature, often refining or validating the exceptional insights of early researchers, such as Endean [
113] and Moran [
21]. For example, Pratchett et al. [
127] quantified short-term movement rates for adult
A. cf.
solaris over different substrate types. Despite using detailed video analyses to document movement under laboratory conditions, Pratchett et al. [
127] reported rates of movement that were broadly similar to those estimated based on field observations in the 1960s [
113]. Moreover, the more critical question to establish the capability of CoTS to move between reefs, which was also raised by Endean [
113], about how long adult CoTS can actually sustain near maximum rates of movement, has never been addressed. While specific and detailed experimental studies are critical in providing a stronger foundation for future research as well as the application of research findings, major advances in understanding and managing outbreaks of
Acanthaster spp. are reliant on (i) synthesis of prior research, both through qualitative reviews (e.g., [
1,
31,
129]; this review) and empirical-based models (
Section 3.3) to combine diverse data sources and identify critical knowledge gaps; (ii) coordinated and collaborative research efforts to effectively address logistically challenging questions at relevant scales of time and space, and in field settings; and (iii) improved integration of science and management, not only to expedite the update of new and important research findings, but to moderate research objectives to explicitly consider specific management tools and levers, and the relevant time-frames for considering different management actions.
Moran [
21] provided a thorough and comprehensive review of the state of knowledge for
Acanthaster spp. in the mid 1980s, during the height of the second documented outbreak on the GBR. At the time, most of the research and scientific discussion centered on the cause(s) of population outbreaks, and specifically, whether outbreaks are influenced by anthropogenic activities. Moran [
21] predicted that the staunch support for individual hypotheses and specific viewpoints would significantly impede scientific advances, such that research should not aim to confirm or refute individual hypotheses, especially not in isolation. Currently, in the midst of the fourth documented outbreak wave on the GBR, there is still ongoing debate and specifically targeted research to address individual causes of outbreaks. Some of this research is entirely pragmatic, focusing on the potential benefits of specific management actions [
133], rather than the specific mechanistic causes of outbreaks. However, focus needs to be given to specific, well-defined and tractable issues (
Table 2). Outbreaks of
Acanthaster spp. also need to be considered against the backdrop of other disturbances and threat to coral reef ecosystems. Worryingly for the health of the GBR is that the same areas that have so far been impacted by outbreaks of
A. cf.
solaris (northern and central sections; [
34,
35], have recently (2016 and 2017) been subject to the most widespread and most severe mass coral bleaching ever recorded [
276].
In conclusion, outbreaks of
Acanthaster spp. remain one of the most significant disturbances and major causes of coral loss across the Indo-Pacific [
6,
7,
8,
9,
17,
19,
20,
219,
235,
278]. While previous efforts to eradicate outbreaks have only been successful in relatively small and isolated areas [
22,
148], questioning whether it would ever be feasible or practical to prevent outbreaks. However, outbreaks are more amenable to direct intervention than most of the other major causes of widespread and significant coral loss (e.g., climate-induced coral bleaching), and therefore, sustained and ongoing research to improve understanding and management of outbreaks of
Acanthaster spp. is certainly warranted. Fundamentally, there remain considerable practical and logistical challenges to undertaking necessary research to better understand the population dynamics of
Acanthaster spp., though emerging technologies continually provide new opportunities and increased efficiencies to tackle research questions that were previously intractable or unfeasible (e.g., [
53,
54,
107]). The purpose of this review was to highlight research questions posed more than 30 years ago, that were considered fundamental in understanding and managing CoTS outbreaks [
21]. Despite significant research in the intervening period, a relatively large number (18 out of 41) of these questions remain largely unresolved (
Table 1). It was not that scientists completely neglected or disregarded these questions, and there has been some progress made to address many (21 out of 41) of the pertinent questions (
Section 2). There have also been new and emerging issues (
Section 3) that have partly deflected research attention. However, ongoing debate regarding the specific cause(s) of CoTS outbreaks is potentially detracting from effective research on this issue. Moving forward, the focus has to be on questions that will improve the effectiveness of management to reduce the frequency and likelihood of outbreaks arising, as well as detecting and containing outbreaks as soon as they start.