Benthic Predators Influence Microhabitat Preferences and Settlement Success of Crown-of-Thorns Starfish (Acanthaster cf. solaris)
Abstract
:1. Introduction
2. Materials and Methods
2.1. Collection and Maintenance of Study Species
2.2. Preparation of Cues
2.3. Predation Rates by Benthic Predators
2.4. Static Choice Chambers
2.5. Settlement Assays
3. Results
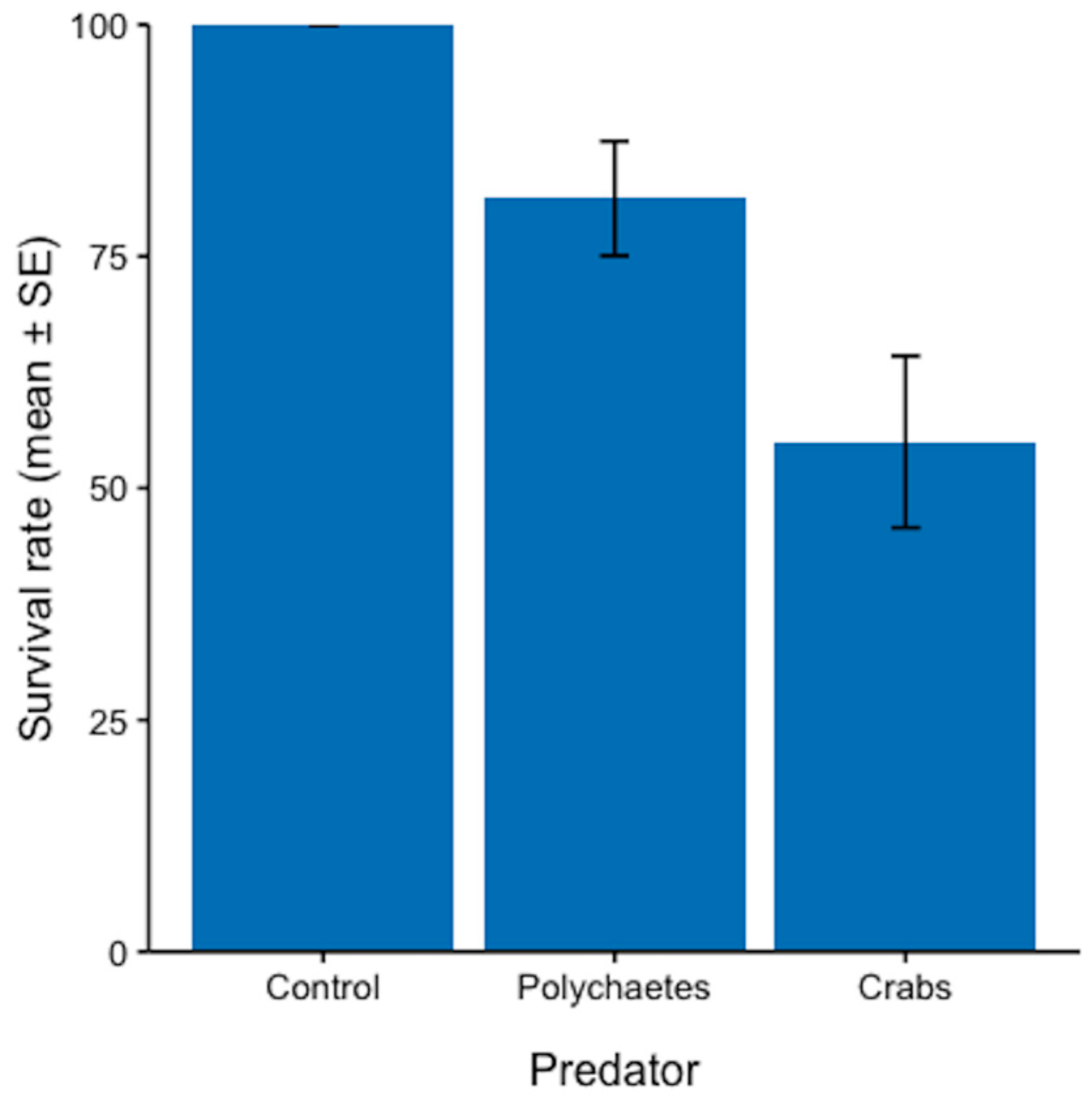
3.1. Potential Predators
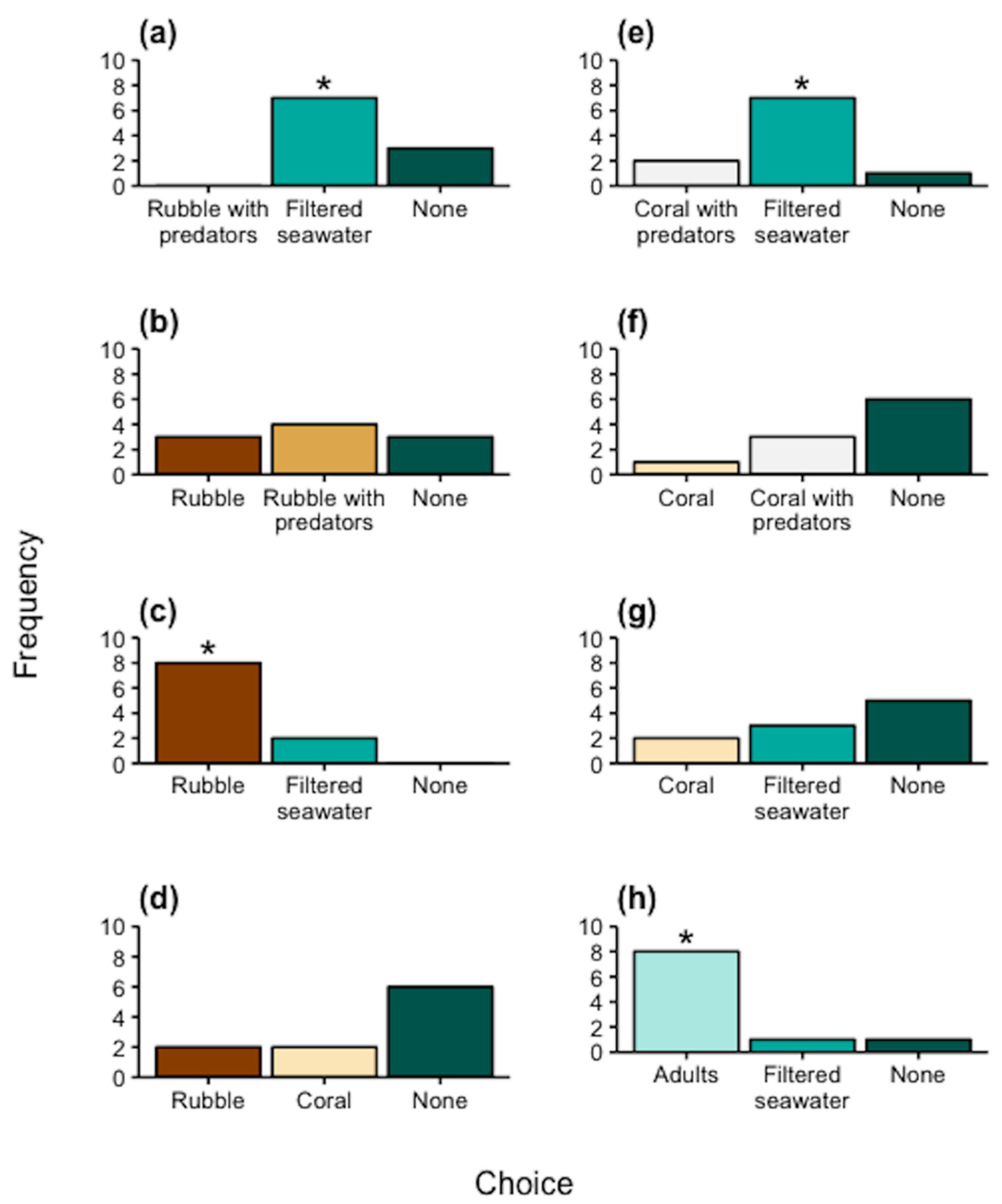
3.2. Static Choice Chambers
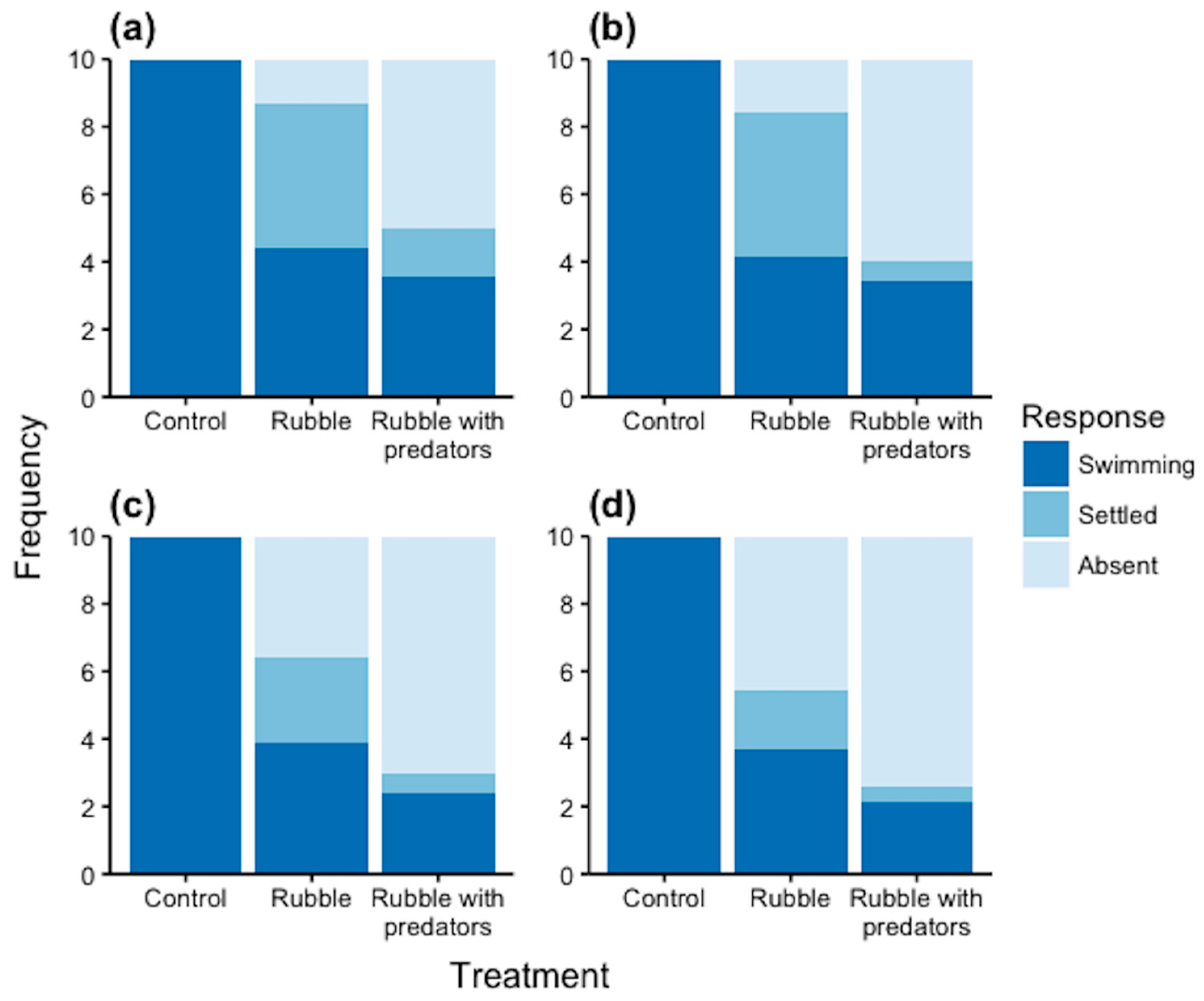
3.3. Settlement Assays
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Almany, G.R.; Webster, M.S. The predation gauntlet: Early post-settlement mortality in reef fishes. Coral Reefs 2006, 25, 19–22. [Google Scholar] [CrossRef]
- Cowan, Z.-L.; Dworjanyn, S.A.; Caballes, C.F.; Pratchett, M.S. Predation on crown-of-thorns starfish larvae by damselfishes. Coral Reefs 2016, 35, 1253–1262. [Google Scholar] [CrossRef]
- Yamaguchi, M. Early life histories of coral reef asteroids, with special reference to Acanthaster planci (L.). In Biology and Geology of Coral Reefs; Jones, O., Endean, R., Eds.; Academic Press: New York, NY, USA, 1973; pp. 369–387. [Google Scholar]
- Keesing, J.; Halford, A. Field measurement of survival rates of juvenile Acanthaster planci, techniques and preliminary results. Mar. Ecol. Prog. Ser. 1992, 85, 107–114. [Google Scholar] [CrossRef]
- Morgan, S.G. The larval ecology of marine communities. In Marine Community Ecology; Bertness, M.D., Gaines, S.D., Hay, M.E., Eds.; Sinauer Associates: Sunderland, MA, USA, 2001; pp. 159–181. [Google Scholar]
- Gosselin, L.A.; Qian, P.-Y. Juvenile mortality in benthic marine invertebrates. Mar. Ecol. Prog. Ser. 1997, 146, 265–282. [Google Scholar] [CrossRef]
- Connell, J.H. The consequences of variation in initial settlement vs. post-settlement mortality in rocky intertidal communities. J. Exp. Mar. Bio. Ecol. 1985, 93, 11–45. [Google Scholar] [CrossRef]
- Rowley, R.J. Settlement and recruitment of sea urchins (Strongylocentrotus spp.) in a sea-urchin barren ground and a kelp bed: Are populations regulated by settlement or post-settlement processes? Mar. Biol. 1989, 100, 485–494. [Google Scholar] [CrossRef]
- Lucas, J.S. Environmental influences on the early development of Acanthaster planci (L.). In Crown-Thorns Starfish Seminar Proceedings; Australian Government Publishing Service: Canberra, Australia, 1975; pp. 109–121. [Google Scholar]
- Lucas, J.; Hart, R.; Howden, M.; Salathe, R. Saponins in eggs and larvae of Acanthaster planci (L.) (Asteroidea) as chemical defences against planktivorous fish. J. Exp. Mar. Bio. Ecol. 1979, 40, 155–165. [Google Scholar] [CrossRef]
- Barnett, D.; Dean, P.W.; Hart, R.J.; Lucas, J.S.; Salathe, R.; Howden, M.E.H. Determination of contents of steroidal saponins in starfish tissues and study of their biosynthesis. Comp. Biochem. Physiol. Part B Comp. Biochem. 1988, 90, 141–145. [Google Scholar] [CrossRef]
- Johnson, C.R.; Sutton, D.C.; Olson, R.R.; Giddins, R. Settlement of crown-of-thorns starfish: Role of bacteria on surfaces of coralline algae and a hypothesis for deepwater recruitment. Mar. Ecol. Prog. Ser. 1991, 71, 143–162. [Google Scholar] [CrossRef]
- Pawlik, J.R. Chemical ecology of the settlement of benthic marine invertebrates. Oceanogr. Mar. Biol. 1992, 30, 273–335. [Google Scholar]
- Dixson, D.L.; Munday, P.L.; Pratchett, M.; Jones, G.P. Ontogenetic changes in responses to settlement cues by anemonefish. Coral Reefs 2011, 30, 903–910. [Google Scholar] [CrossRef]
- Dixson, D.L.; Abrego, D.; Hay, M.E. Chemically mediated behavior of recruiting corals and fishes: A tipping point that may limit reef recovery. Science 2014, 345, 892–897. [Google Scholar] [CrossRef] [PubMed]
- Sweatman, H. Field evidence that settling coral reef fish larvae detect resident fishes using dissolved chemical cues. J. Exp. Mar. Bio. Ecol. 1988, 124, 163–174. [Google Scholar] [CrossRef]
- Johnson, C.R.; Muir, D.G.; Reysenbach, A.L. Characteristic bacteria associated with surfaces of coralline algae: A hypothesis for bacterial induction of marine invertebrate larvae. Mar. Ecol. Prog. Ser. 1991, 74, 281–294. [Google Scholar] [CrossRef]
- Johnson, C.R.; Sutton, D.C. Bacteria on the surface of crustose coralline algae induce metamorphosis of the crown-of-thorns starfish Acanthaster planci. Mar. Biol. 1994, 120, 305–310. [Google Scholar] [CrossRef]
- Brauer, R.W.; Jordan, M.J. Triggering of the stomach eversion reflex of Acanthaster planci by coral extracts. Nature 1970, 228, 344–346. [Google Scholar] [CrossRef] [PubMed]
- Ormond, R.; Campbell, A.; Head, S.; Moore, R.; Rainbow, P.; Saunders, A. Formation and breakdown of aggregations of the crown-of-thorns starfish, Acanthaster planci (L.). Nature 1973, 246, 167–169. [Google Scholar] [CrossRef]
- Haszprunar, G.; Spies, M. An integrative approach to the taxonomy of the crown-of-thorns starfish species group (Asteroidea: Acanthaster): A review of names and comparison to recent molecular data. Zootaxa 2014, 3841, 271–284. [Google Scholar] [CrossRef] [PubMed]
- Glynn, P.W. Acanthaster population regulation by a shrimp and a worm. In Proceedings of the 4th International Coral Reef Symposium, Manila, Philippines, 18–22 May 1981; Gomez, E.D., Birkeland, C.E., Buddemeier, R.W., Johannes, R.E., Marsh, J.A., Jr., Tsuda, R.T., Eds.; 1982; Volume 2, pp. 607–612. [Google Scholar]
- Glynn, P.W. An amphinoid worm predator of the crown-of-thorns sea star and general predation on asteroids in eastern and western pacific coral reefs. Bull. Mar. Sci. 1984, 35, 54–71. [Google Scholar]
- Anderson, M.J.; Gorley, R.N.; Clarke, K.R. PERMANOVA+ for PRIMER: Guide to Software and Statistical Methods; PRIMER-E Ltd.: Plymouth, UK, 2008. [Google Scholar]
- Mileikovsky, S.A. On predation of pelagic larvae and early juveniles of marine bottom invertebrates by adult benthic invertebrates and their passing alive through their predators. Mar. Biol. 1974, 26, 303–311. [Google Scholar] [CrossRef]
- Zann, L.; Brodie, J.; Berryman, C.; Naqasima, M. Recruitment, ecology, growth and behavior of juvenile Acanthaster planci (L.) (Echinodermata: Asteroidea). Bull. Mar. Sci. 1987, 41, 561–575. [Google Scholar]
- McCallum, H.I. Effects of predation on organisms with pelagic larval stages: Models of metapopulations. In Proceedings of the 6th International Coral Reef Symposium, Townsville, Australia, 8–12 August 1988; Choat, J.H., Barnes, D., Borowitzka, M.A., Coll, J.C., Davies, P.J., Flood, P., Hatcher, B.G., Hopley, D., Hutchings, P.A., Kinsey, D., et al., Eds.; 1988; Volume 2, pp. 101–106. [Google Scholar]
- McCallum, H.I. Effects of predation on Acanthaster: Age-structured metapopulation models. In Acanthaster and the Coral Reef: A Theoretical Perspective; Bradbury, R., Ed.; Springer-Verlag: Berlin/Heidelberg, Germany, 1990; pp. 208–219. [Google Scholar]
- Grassle, J.F. Variety in coral reef communities. Biol. Geol. Coral Reefs 1973, 2, 247–270. [Google Scholar]
- Dean, H. The use of polychaetes (Annelida) as indicator species of marine pollution: A review. Rev. Biol. Trop. 2008, 56, 11–38. [Google Scholar]
- Pinnegar, J.K.; Polunin, N.V.C.; Francour, P.; Badalamenti, F.; Chemello, R.; Harmelin-Vivien, M.-L.; Hereu, B.; Milazzo, M.; Zabala, M.; D’Anna, G.; et al. Trophic cascades in benthic marine ecosystems: Lessons for fisheries and protected-area management. Environ. Conserv. 2000, 27, 179–200. [Google Scholar] [CrossRef]
- Sukumaran, S.; Vijapure, T.; Kubal, P.; Mulik, J.; Rokade, M.A.; Salvi, S.; Thomas, J.; Naidu, V.S. Polychaete community of a marine protected area along the west coast of India-prior and post the tropical cyclone, Phyan. PLoS ONE 2016, 11, e0159368. [Google Scholar] [CrossRef] [PubMed]
- Chesher, R.H. Destruction of Pacific corals by the sea star Acanthaster planci. Science 1969, 165, 280–283. [Google Scholar] [CrossRef] [PubMed]
- Pratchett, M.S.; Caballes, C.F.; Rivera-Posada, J.A.; Sweatman, H.P.A. Limits to understanding and managing outbreaks of crown-of-thorns starfish (Acanthaster spp.). Oceanogr. Mar. Biol. Annu. Rev. 2014, 52, 133–200. [Google Scholar]
- Dworjanyn, S.A.; Pirozzi, I.; Liu, W. The effect of the addition of algae feeding stimulants to artificial diets for the sea urchin Tripneustes gratilla. Aquaculture 2007, 273, 624–633. [Google Scholar] [CrossRef]
- Keesing, J.K.; Wiedermeyer, W.L.; Okaji, K.; Halford, A.R.; Hall, K.C.; Cartwright, C.M. Mortality rates of juvenile starfish Acanthaster planci and Nardoa spp. measured on the Great Barrier Reef, Australia and in Okinawa, Japan. Oceanol. Acta 1996, 19, 441–448. [Google Scholar]
- Rivera-Posada, J.; Caballes, C.F.; Pratchett, M.S. Size-related variation in arm damage frequency in the crown-of-thorns sea star, Acanthaster planci. J. Coast. Life Med. 2014, 2, 187–195. [Google Scholar]
- Nakamura, M.; Kumagai, N.H.; Sakai, K.; Okaji, K.; Ogasawara, K.; Mitarai, S. Spatial variability in recruitment of acroporid corals and predatory starfish along the Onna coast, Okinawa, Japan. Mar. Ecol. Prog. Ser. 2015, 540, 1–12. [Google Scholar] [CrossRef]
- Atema, J.; Kingsford, M.J.; Gerlach, G. Larval reef fish could use odour for detection, retention and orientation to reefs. Mar. Ecol. Prog. Ser. 2002, 241, 151–160. [Google Scholar] [CrossRef]
- Pineda, J.; Riebensahm, D.; Medeiros-Bergen, D. Semibalanus balanoides in winter and spring: Larval concentration, settlement, and substrate occupancy. Mar. Biol. 2002, 140, 789–800. [Google Scholar]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cowan, Z.-L.; Dworjanyn, S.A.; Caballes, C.F.; Pratchett, M. Benthic Predators Influence Microhabitat Preferences and Settlement Success of Crown-of-Thorns Starfish (Acanthaster cf. solaris). Diversity 2016, 8, 27. https://doi.org/10.3390/d8040027
Cowan Z-L, Dworjanyn SA, Caballes CF, Pratchett M. Benthic Predators Influence Microhabitat Preferences and Settlement Success of Crown-of-Thorns Starfish (Acanthaster cf. solaris). Diversity. 2016; 8(4):27. https://doi.org/10.3390/d8040027
Chicago/Turabian StyleCowan, Zara-Louise, Symon A. Dworjanyn, Ciemon F. Caballes, and Morgan Pratchett. 2016. "Benthic Predators Influence Microhabitat Preferences and Settlement Success of Crown-of-Thorns Starfish (Acanthaster cf. solaris)" Diversity 8, no. 4: 27. https://doi.org/10.3390/d8040027
APA StyleCowan, Z.-L., Dworjanyn, S. A., Caballes, C. F., & Pratchett, M. (2016). Benthic Predators Influence Microhabitat Preferences and Settlement Success of Crown-of-Thorns Starfish (Acanthaster cf. solaris). Diversity, 8(4), 27. https://doi.org/10.3390/d8040027







