Diversity of Pyrrolizidine Alkaloids in the Boraginaceae Structures, Distribution, and Biological Properties
Abstract
:1. Introduction
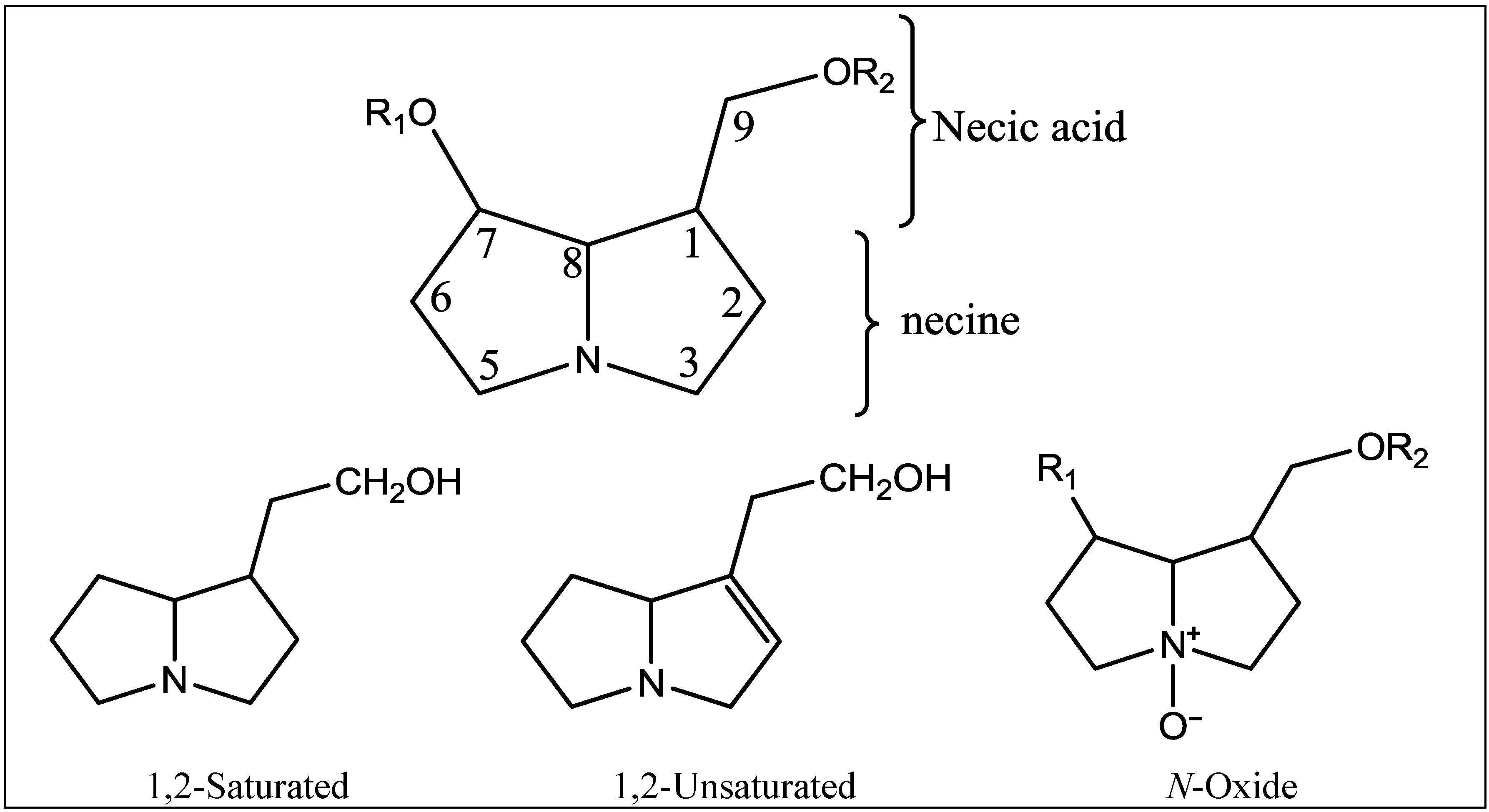
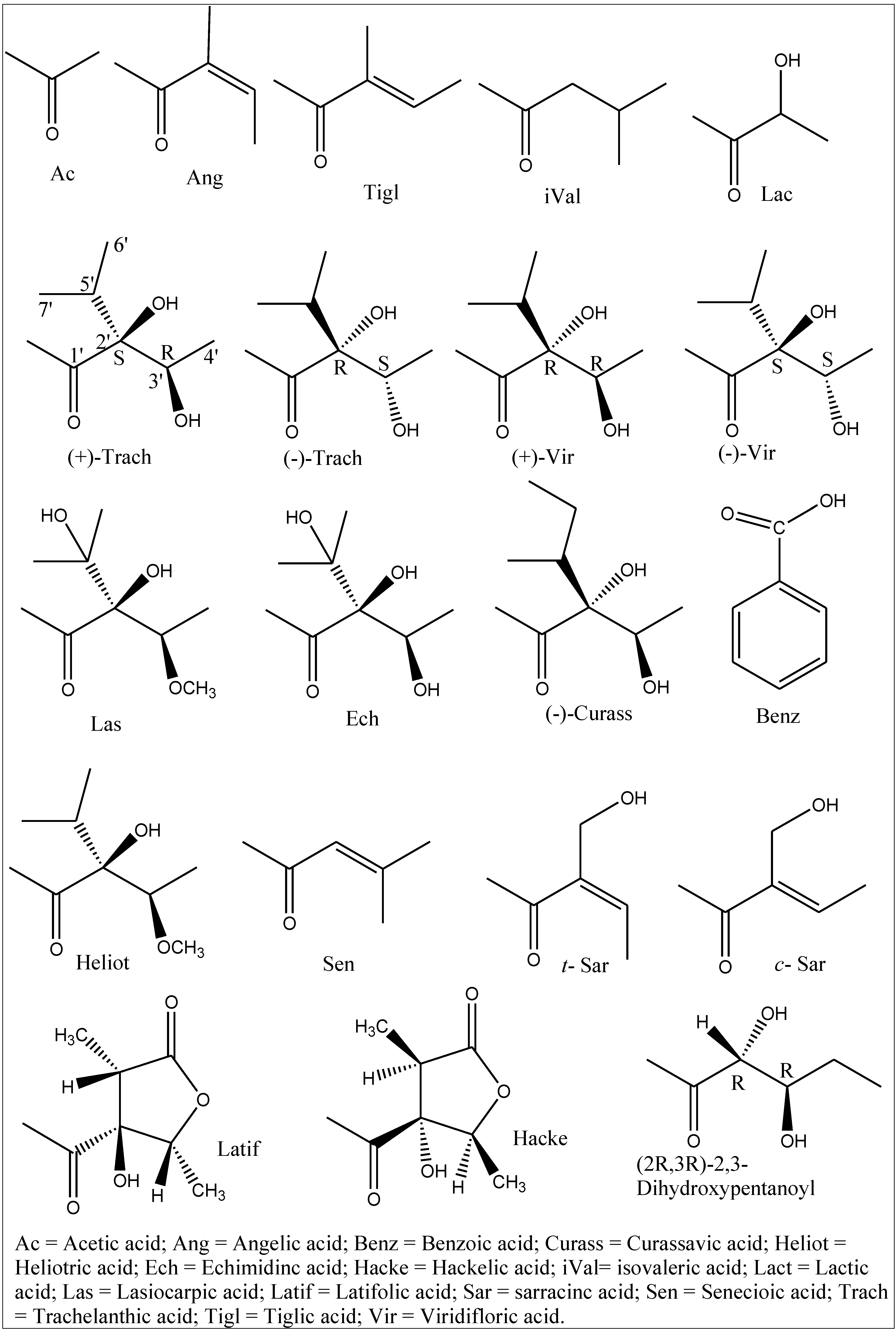
| Species | Pyrrolizidine alkaloids | References |
|---|---|---|
| Alkanna orientalis | 7-Angeloylretronecine, 9-angeloylretronecine, 7-tigloylretronecine, 9-tigloylretronecine, 7-seneioylretronecine, 9-senecioylretronecine, triangularine, dihydroxytriangularine, triangularicine, dihydroxytriangularicine, 7-angeloyl-9-(hydroxypropenoyl) retronecine, 7-tigloyl-9-(hydroxypropenoyl) retronecine, 7-angeloyl-9-(2,3-dihydroxypropanoyl) retronecine, 7-tigloyl-9-(2,3-dihydroxypropanoyl) retronecine. | [40,41] |
| A. tinctoria (A. tuberculata) | 7-Angeloylretronecine, 7-tigloylretronecine, 9-tigloylretronecine, triangularine, dihydroxytriangularine, triangularicine, dihydroxytriangularicine, 7-acetyl-9-sarracinoylretronecine, 7-angeloyl-9-(hydroxypropenoyl) retronecine, 7-tigloyl-9-(hydroxypropenoyl) retronecine, 7-angeloyl-9-(2,3-dihydroxypropanoyl) retronecine, 7-tigloyl-9-(2,3-dihydroxypropanoyl) retronecine. | [40,41] |
| Amsinckia carinata | Echiumine, furcatine, 3'-acetylfurcatine, intermedine, 7-acetylintermedine, lycopsamine, myoscorpine, supinine. | [42] |
| A. douglasiana | Amabiline, cynaustrline, intermedine, lycopsamine, tessellatine, 9-(3'-acetylviridifloryl)turniforcidine. | [42,43] |
| A. eastwoodiae | Amabiline, intermedine, 3'-acetylintermedine, echiumine, lindelofine, lycopsamine, supinine, tessellatine. | [42] |
| A. furacata | Furcatine, intermedine, 3'-acetylfurcatine, lycopsamine, supinine, tessellatine. | [42] |
| A. grandiflora | Amabiline, intermedine, lycopsamine, 3'-aetyllycopsamine, 7-acetyllycopsamine, tessellatine, 9-acetyltessellatine. | [42] |
| A. hispida | Echiumine, intermedine, lycopsamine. | [44] |
| A. intermedia | Echiumine, intermedine, lycopsamine, sincamidine. | [44] |
| A. lunaris | Lycopsamine, 3'-acetyllycopamine, intermedine, supinine, tesslatine, 9'-acetyltessellatine. | [42] |
| A. lycopsoides | Amabiline, echiumine, intermedine, 3'-acetylintermedine, lycopsamine, supinine, tessellatine. | [42,44] |
| A. lycopsoides × menziesii var. intermedia | Amabiline, intermedine, 3'-acetylintermedine, lycopsamine, supinine, tessellatine. | [42] |
| A. menziesii | Intermedine, lycopsamine, 3'-acetyllycopsamine, 7-acetyllycopsamine, 3',7-diacetyllycopsamine. | [45] |
| A. menziesii var. intermedia | Amabiline, cynaustraine (or steroisomer), echiumine, 3'-acetylechiumine, intermedine, 3'-acetylintermedine, 7-acetylintermedine, 3',7-diacetylintermedine, lindelfoline, lycopsamine, 3'-acetyllycopsamine, 7-acetyllycopsamine, myscorpine, 3'-acetylmyscorpine, supinine, symlandine, tessellatine. | [42,43] |
| A. retrosa | Amabiline, intermedine, 3'-acetylintermedine, lycopsamine, myscorpine, 3'-acetylmyscorpine, supinine, tessellatine. | [42] |
| A. spectabilis var. microcarpa | Intermedine, 3'-acetylintermedine, lindelofine, lycopsamine, myscorpine, supinine, tessellatine. | [42] |
| A. specabtilis var. spectabilis | Intermedine, 3'-actylintermedine, lindelofine, lycopsamine, supinine. | [42] |
| A. spectabilis var. nicolai | Intermedine, lindelofine, lycopsamine, tessellatine, trachelanthamine, supinine. | [42] |
| A. tessellata | Intermedine, lycopsamine, 3'-acetylintermedine, 3'-acetyllycopsamine, 7-acetylintermedine, 7-acetyllycopsamine, 3',7-diacetylintermedine, 3'-7-diacetyllycopsamine. | [46] |
| A. tessellata var. gloriosa | Amabiline, intermedine, lycopsamine, supinine, tessellatine, tracelanthamine, viridiflorine, 3'-acetylviridiflorine. | [42,43] |
| A. tessellata var. tessellata | Amabiline, intermedine, lycopsamine, 3'-acetyllycopsamine, tessellatine, 3'-acetyltessellatine, 9-acetyltessellatine, 9,3'-acetyltessellatine, 9-(3'-acetylviridifloryl)-turniforcidine. | [42] |
| A. vernicosa | Furcatine, intermedine, lycopsamine, supinine, 7-trachelanthyl retronecine. | [42] |
| Anchusa arvensis (=Lycopsis arvensis) | Echinatine, intermedine, 7-acetylintermedine, lycopsamine, 7-acetyllycopsamine, 3',7-diacetylintermedine (or its isomer 3',7-diaetyllycopsamine), supinine, 9-acetyltrachelanthamidine, 9-angeloyltrachelanthamidine. | [40,47] |
| A. hispida (=Gastrocotyle hispida) | 7-Angeloylheliotridine, intermedine, lyopsamine, 7-acetyllycopsamine, retronecine, trachelanthamine and its isomer. | [40] |
| A. milleri | Heliotridine, 7-angeloylheliotridine, rinderine, retronecine, supinine, viridiflorine, 9-curassavoyleliotridine, 7-acetyl-9-curassavoylheliotridine. | [40] |
| A. officinalis | Intermedine, curassavine, lycopsamine, 7-aceyllycopsamine. | [47,48,49] |
| A. strigosa | Heliotridine 2S-hydroxy-2S(1S-hydroxyethyl)-4-methyl-pentanoyl ester, platyneine N-oxide 2S-hydroxy-2S(1S-hydroxyethyl)-4-methyl-pentanoyl ester, retronecine 2S-hydroxy-2S(1S-hydroxyethyl)-4-methyl-pentanoyl ester and its N-oxide, retronecine 2S-hydroxy-2S(1R-hydroxyethyl)-4-methyl-pentanoyl ester and its N-oxide, retronecine 2S-hydroxy-2S(1S-hydroxyethyl)-[1'S-hydroxyethyl)-4-methylpentanoyl]-4-methyl-pentanoyl ester, supinidine N-oxide 2S-hydroxy-2S(1S-hydroxyethyl)-4-methyl-pentanoyl ester, trachelanthamidine 2S-hydroxy-2S(1S-hydroxyethyl)-4-methyl-pentanoyl ester. | [50,51] |
| Arnebia decumbens | 7-angeloylrtronecine, 9-angeloylretronecine, 7-tigloylrtronecine, 9-tigloylretronecine, europine, heliotrine, lycopsamine, rinderine, supinine. | [52] |
| Arnebia euchroma | 7-angeloylrtronecine, 9-angeloylretronecine | [53] |
| A. hispidissima | Echimidine, monocrotaline. | [54] |
| Asperugo procumbens | Amabiline (or supinine), echinatine. | [47] |
| Borago officinalis | Amabiline, intermedine, 7-acetylintermedine, lycopsamine, 7-acetyllycopsamine, supinine, thesinine, thesinine-4'- O-β-D-glucoside. | [55,56] |
| Caccinia crassifolia | Supinine, Heliotridine or retronecine trachelanthate. | [57] |
| C. glauca | Retronecine 7:9 -dibenzoate. | [58] |
| Cerinthe glabra | Lycopsamine, 3'-acetyllycopsamine, 7-acetyllycopsamine, 3',7-diacetyllycopsamine, supinine, 7-hydroxymethyl butyryl-9-viridifloryl-retronecine (or isomer). | [59] |
| Cerinthe minor | Intermedine, 7-angeloylretronecine, 9-angeloyl-7-viridiflorylretronecine, lycopsamine. | [60,61] |
| Cordia myxa | macrophylline | [62] |
| C. sinensis | floridanine | [62] |
| Cryptantha cana | Intermdine, 3'-acetylintermedine, 7-acetylintemedine, lycopsamine, 3'-acetyllycopsamine, 7-acetyllycopsamine. | [63] |
| C. clevelandii | Intermdine, 3'-acetylintermedine, echiumine, 2'',3''-epoxyechiumine, thero-2',3' dihydroxyechiumine, erytho-3''-chloro-2''-hydroxyechiumine. | [63] |
| C. confertiflora | Amabiline, intermedine, lycopsamine, tessellatine, 9-acetyltessellatine. | [63] |
| C. crassipes | Lycopsamine, intermedine and their 3'-acetyllycopsamine, 3'-acetylintermedine, 7-acetyllycopsamine, 7-acetylintermedine, amabiline, echiumine, dihydroechumine, echimiplatine, lepanthine. | [64] |
| C. fendleri | 7-Angeloylretronecine, 9-angeloylretronecine, latifoline, neolatifoline. | [63] |
| C. flava | Intermedine, 3'-acetylintermedine, 7-acetylintemedine, lycopsamine, 3'-acetyllycopsamine, 7-acetyllycopsamine. | [63] |
| C. inequata | Echimidine, acetylechimidine, echiuplatine, methylechiuplatine, lycopsamine, intermedine, dihydroxyechiumine. | [65] |
| C. jamesii | Intermedine, lycopsamine, 3'- acetyllycopsamine, 7-aetyllyopsamine. | [66] |
| C. leiocarpa | 7-Angeloylretronecine, 9-angeloylretronecine, echiumine, intermedine, 3'-acetylintermedine, 2'',3''-epoxyechiumine, thero-2',3'-dihydroxyechiumine, erytho-3''-chloro-2''-hydroxyechiumine | [63] |
| C. thyrsiflora | Intermedine, 3'-acetylintermedine, 7-acetylintemedine, lycopsamine, 3'-acetyllycopsamine, 7-acetyllycopsamine | [63] |
| C. utahensis | Cryptanthine | [65] |
| C. virgata | Intermedine, 3'-acetylintermedine, 7-acetylintemedine, lycopsamine, 3'-acetyllycopsamine, 7-acetyllycopsamine | [63] |
| C. virginiensis | Amabiline, intermedine, lycopsamine, tessellatine. | [63] |
| Cynoglossum amabile | Amabiline, echinatine, 7-acetylechinatine, lycopsamine, rinderine, supinine. 9-angeloylretronecine, 9-angeloyl-7-viridiflorylretronecine, | [60,67,68] |
| C. australe | Cynausine, cynaustraline. | [68] |
| C. clandestinum | 9-Angeloyl-7-viridiflorylretronecine, trachelanhamine | [60] |
| C. columnae | N-oxides of echintine, rinderine, 3’-acetylrinderine, 7-tigloyl-9-(2-deoxy-2-methyl)echimidinyl heliotridine. | [69] |
| C. creticum | Cynoglossamine, echinatine, 3'-acetylechinatine, heliosupine, 3'-acetylheliosupine, 7-angeloylheliotridine, 7-angeloyl-9-(methylbutyryl) heliotridine, 7α-angeloyl-1-chloromethyl-1,2-dehydropyrrolizidine, 7-senecioylheliotridine, rinderine, 3'-acetylrenderine, supinine, trachelanthamine (or isomer). | [70,71,72] |
| C. furcatum (C. zeylanicum) | Echinatine, isoechinatine, neocoromandaline, cynaustraline, lactodine, viridinatine | [73,74,75,76] |
| C. germanicum | Echinatine, viridiflorine. | [77] |
| C. glochidiatum | Amabiline | [78] |
| C. lanceolatum | Cynaustine, cynaustraline. | [78] |
| C. latifolium | 7-Angeloylheliotridine, latifoline. | [79] |
| C. macrostylum | Echinatine, heliosupine. | [80] |
| C. montanum | Cynaustine, cynaustraline, echinatine, heliosupine. | [81] |
| C. nervosum | Heliotrine, echinatine, rinderine and their N-oxides. | [82] |
| C. officinale | Echinatine, 3'-acetylechinatine, 7-angeloylechinatine, heliosupine, 3'-acetylheliosupine, 7-angeloylheliotridine, 7-angeloyl-1-formyl-6,7-dihydro-5H-pyrrolizidine, 7-angeloyl-9-(2-methylbutyryl)heliotridine, 7-angeloyl-9-(2,3-dihydroxybutyryl) heliotridine, 7-tigloylheliotridine, rinderine, 7-angeloylrinderine, trachelanthamine, viridiflorine. | [67,83,84] |
| C. pictum | Echinatine, heliosupine, pictumine. | [85] |
| C. viridiforum | Heliosupine, viridiflorine. | [86] |
| Echium amoenum | Echimidine, echimidine isomer (tigloyl), 7-angeloylretronecine, 7-tigloylretronecine. | [87] |
| E. angustifolium | Echimidine | [88] |
| E. diffusum | Heliotridine or retronecine esters. | [28] |
| E. glomeratum | 7-Angeloylretronecine, 9-angeloylretronecine, (7S,8R)petranine, (7S,8S)petranine. | [89] |
| E. horridum | Echimidine, echimidine isomer (tigloyl), lycopsamine, 7-acetyllycopsamine, 7-angeloyllycopsamine, 7-tigloyllycopsamine, 7-angeloylretronecine, 7-tigloylretronecine, 7-angeloyl-9-(2-methylbutyryl)retronecine, 7-tigloyl-9-(2-methylbutyryl) retronecine, 7-angeloyl-9-(2, 3-dihydroxybutyryl)retronecine, 7-tigloyl-9-(2, 3-dihydroxybutyryl)retronecine, uplandicine. | [90] |
| E. humile | Echimidine, echihumiline, lycopsamine, 7-acetyllycopsamine, 7-senecioyllycopsamine, pycnanthine, 7-seneioylretronecine, 9-seneioylretronecine, 7-(2-methylutyryl)retronecine, 7-(methybutyryl)-9-(2,3-dihydroxybutyryl)retronecine, 7-(2-methybutyryl)-9-echimidinylretronecine. | [91] |
| E. hypertropicum | Echimidine, echihumiline, 7-(2-methylbutyryl)-9-echimidinylretronecine, 7-senecioylretronecine, 9-angeloylretronecine, lycopsamine, 7-acetyl-lycopsamine | [92] |
| E. italicum | Echimidine | [28] |
| E. lycopsis (=E. plantagineum) | Echimidine, echiumine, uplandicine, lycopsamine, intermedine, echimplatine, echiuplatine, 3'-acetylintermedine, 3'-acetyllycopsamine, 3'-acetylechiumine, 9-angeloylretronecine, leptanthine. | [4,93,94] |
| E. pininana | Echimidine, ehiupinine, 3'-aetylintermedine, hydroxymyoscorpine, myoscorpine. | [95] |
| E. rauwolfii | Echimidine, echimidine isomer (tigloyl), 7-acetyllycopsamine, 7-angeloyllycopsamine, 7-tigloyllycopsamine, 7-angeloylretronecine, 7-tigloylretronecine, 7-angeloyl-9-(2-methylbutyryl)retronecine, 7-tigloyl-9-(2-methylbutyryl)retronecine, 7-angeloyl-9-(2, 3-dihydroxybutyryl)retronecine, 7-tigloyl-9-(2, 3-dihydroxybutyryl)retronecine, uplandicine. | [90] |
| E. sericeum | Echimidine, symlandine (or symphytine). | [54] |
| E. setosum | Echimidine, echimidine isomer (tigloyl), 7-angeloylretronecine, 7-tigloylretronecine, 9-angeloylretronecine, 9-tigloylretronecine, 7-angeloyl-9-(2-methylbutryl) retronecine, 7-tigloyl-9-(2-methylbutryl) retronecine, 7-angeloyl-9-(2,3-dimethylbutryl) retronecine, 7-angeloyl-9-(2,3-dihydroxybutryl) retronecine, uplandicine. | [96] |
| E. simplex | 7-Angeloylretronecine, 9-angeloylretronecine. | [61] |
| E. stenosiphon Webb subsp. stenosiphon | Echimidine, 7-(2-methylbutyryl)-9-echimidinylretronecine | [92] |
| E. tuberculatum | Echimidine,7-angeloyl-9-viridiflorylretronecine, 7-viridiflorylretronecine. | [60] |
| E. vulgare | Asperumine, heliosupine, 3'-acetylheliosupine, Echinatine, echiuplantine, leptanthine, echimiplantine, echivulgarine, vulgarine, 7-O-acetylvulgarine, echimidine, echimidine isomer (tigloyl ?), 3'-acetylechimidine, 5'-acetylechimidine, echihumiline, retronecine, 7-angeloylretronecine, 9-angeloylretronecine, 7-tigloylretronecine, 9-tigloylretronecine, 7-(2-methylbutyryl) retronecine, 9-(2-methylbutyryl) retronecine, 7-angeloyl-9-(2-methylbutryl) retronecine, 7-tigloyl-9-(2-methylbutryl) retronecine, 7-angeloyl-9-(2, 3-di methylbutryl) retronecine, 7-tigloyl-9-(2, 3-dihydroxybutryl)retronecine, uplandicine | [11,96,97,98] |
| E. wildpretti | Echimidine and its N-oxide. | [99] |
| Ehretia aspera | Ehretinine | [100] |
| Eritrichium rupestre | 7-Angeloylretronecine, 7-angeloyl-9-viridiflorylretronecine, 7-viridiflorylretronecine | [60] |
| Hackelia californica | Hackelidine, longitubine, 7-acetylhackelidine, 9-latifolylretronecine, 7-acetyl-9-latifolylretronecine | [101,102] |
| H. floribunda | Latifoline and its N-oxide. | [103] |
| H. longituba | Latifoline, neolatifoline, longitubine, 7-angeloylretronecie, 9-angeloylretronecine. | [104] |
| Heliotropium acutifolium | Heliotrine | [105] |
| H. amplexicaule | Indicine | [29] |
| H. angiospermum | Subulacine, lindelofidine, retronecine, supinidine, trachelanthamidine. | [106] |
| H. arbainense | Europine, heliotrine, lasiocarpine. | [71] |
| H. arborescens (=H. peruvianum) | Indicine, 3'-acetylindicine, lasiocarpine. | [107] |
| H. arguzioides | Heliotrine, trichodesmine. | [28] |
| H. bacciferum | Europine, heliotrine, heleurine and their N-oxides, supinine. | [108,109] |
| H. bovei | Europine, 7-acetyleuropine, lasiocarpine, 5'-acetyllasiocarpine, lasiocarpine N-oxide, 5'-acetyllasiocarpine N-oxide. | [110] |
| H. bracteatum | Helibractinecine, retronecine, helibracteatinine, helibracteatine | [111,112] |
| H. bursiferum | 7-Angeloylretronecine. | [113] |
| H. circinatum | 7-angeloylheliotrine, echinatine, europine, heleurine, heliotrine, lasiocarpine. | [114] |
| H. confertifolium | lindelofidine, retronecine, supinidine, trachelanthamidine. | [106] |
| H. crassifolium | Ilamine, europine and their N-oxides. | [115] |
| H. curassavicum | Coromandaline, coromandalinine, curassavine, curassavinine, curassanecine, heliocurassavine, heliocurassavinine, heliocurassavicine, heliocoromandaline, heliovicine, 7-angeloylheliotridine, trachelanhamidine, retronecine, supinidine. | [106,116,117,118] |
| H. curassavicum var. argentium | 9-(3'-isovaleryl) viridifloryl retronecine, 9-(3'-acetyl) viridifloryl retronecine. | [119] |
| H. curassavicum var. curassavicum | 9-(3'-isovaleryl) viridifloryl retronecine, 9-(3'-acetyl) viridifloryl retronecine. | [119] |
| H. dasycarpum | heliotrine | [120] |
| H. digynum (H. luteum) | Europine, heliotrine, 7-angeloylheliotrine, lasiocarpine. | [121] |
| H. disciforme | Heliotrine, 2'-actylheliotrine, heliotrine N-oxide, heleurine, heliorine N-oxide. | [122] |
| H. dissitiflorum | Heliotrine, heliotrine N-oxide, europine, 5'-deoxylasiocarpine. | [123] |
| H. eichwaldii | Heliotrine, 7-angeloylheliotrine, lasiocarpine. | [124] |
| H. esfandiarii | Europine, europine N-oxide. | [125] |
| H. europaeum | Europine, acetyleuropine, heleurine, heliotrine, 7-angeloylheliotrine, lasiocarpine, 6-acetyllasiocarpine, heliotrine N-oxide, dehydroheliotrine, 5'-acetyllasiocarpine N-oxide, N-(dihydropyrrolizinomethyl)-heliotrine, supinine. | [126,127] |
| H. floridum | Floridine, floridinine, floridimine, heliovicine, 3'-acetyltrachelanthamine. | [128] |
| H. foliosisimum | lindelofidine, retronecine, supinidine, trachelanthamidine. | [106] |
| H. fruticosum | lindelofidine, retronecine, supinidine, trachelanthamidine. | [106] |
| H. hirsutissimum | Europine, heliotrine, heleurine, lasiocarpine, 3'-acetyllasiocarpine, 5'-acetyllasiocarpine, supinine, N-oxides of acetylasiocarpine, 3'-acetyleheliosupine. | [29,129] |
| H. indicum | Echinatine, helindicine, heliotrine, heleurine, indicine, acetylindicine, indicinine, lasiocarpine, lycopsamine, rinderine, supinine, lindelofidine, retronecine, supinidine, trachelanthamine. | [106,116,130,131,132] |
| H. keralense | Intermedine, isolycopsamine, retronesine. | [133] |
| H. lasiocarpum | Heliotrine, lasiocarpine. | [29] |
| H. marifolium | Europine, heliotrine, indicine, lasiocarpine. | [29] |
| H. maris mortui | Europine, lasiocarpine. | [29,71] |
| H. megalanthum | Lycopsamine, megalanthonine. | [134] |
| H. molle | subulacine | [29] |
| H. olgae | Heliotrine, incanine. | [135] |
| H. ovalifolium | Heliofoline, retronecine. | [136] |
| H. peruvianum | Rinderine | [29] |
| H. popovii subsp. gillianum | Heliotrine | [28] |
| H. procumbens | Lindelofidine, retronecine, supinidine, trachelanthamidine. | [106] |
| H. queretaroanum | Lindelofidine, retronecine, supinidine, trachelanthamidine. | [106] |
| H. racemosum | Lindelofidine, retronecine, supinidine, trachelanthamidine. | [106] |
| H. ramosissimum | Heliotrine | [28] |
| H. rotundifolium | Europine, 5'-acetyleuropine, heliotrine, lasiocarpine. | [137,138] |
| H. scabrum | Heliscabine, retronecine. | [139] |
| H. sessei | Lindelofidine, retronecine, supinidine, trachelanthamidine. | [106] |
| H. spathulatum | Amabiline, coromandaline, coromandalinine, heliovicine, curassavinine, curassavine, heliospathine, heliospathuline, lindelofidine, retronecine, supinidine, trachelanthamidine. | [116,140] |
| H. steudneri | Lycopsamine | [27] |
| H. strigosum | Strigosine, trachelanthamidine | [25,141] |
| H. suaveolens | Echinatine, europine, heliotrine, lasiocarpine. | [29] |
| H. subulatum | Subulacine; retronecine, heliotrine, 7-angeloylheliotridine | [142] |
| H. supinum | Echinatine, heliosupine, heliotrine, 7-angeloylheliotridine (and its trachelanthic and viridifloric esters), lasiocarpine, supinine. | [27,143] |
| H. ternatum | subulacine | [27] |
| H. transalpinum | Intermedine, indicine, lycopsamine, rinderine, 3'-acetylrinderine, supinine. | [144] |
| H. transalpinum var. transalpinum | Transalpinecine, subulacine. | [145] |
| H. transoxanum | Heliotrine | [105] |
| H. wigginsii | Lindelofidine, retronecine, supinidine, trachelanthamidine. | [106] |
| Lappula glochidiata | Echinatine | [146] |
| L. intermedia (Echinospermum intermedium) | Lasiocarpine | [147] |
| L. myosotis | Intermedine, lycopsamine , 7-acetylintermedine, 7-acetyllycopsamine. | [35,148] |
| L. spinocarpos | Amabiline, intermedine, 7-angeloylheiotridine, 9- heliotrinoylretronecine, lycopsamine, 7-acetyllycopsamine, retronecine, trachelanthamine, supinine, viridiflorine. | [40] |
| Lindelofia anchusoides (L. macrostyla) | Lindelofamine, lindelofine. | [28] |
| L. angustifolia | Amabiline, echinatine | [78] |
| L. longiflora | Echinatine and its N-oxide. | [149] |
| L. olgae | Viridiflorine | [150] |
| L. pterocarpa | Viridiflorine | [151] |
| L. spectabilis | Echinatine, 3'-acetylechinatine, monocorotaline. | [124] |
| L. stylosa | Echinatine, lindelofine, viridiflorine. | [135] |
| L. tschimganica | Echinatine, carategine, viridiflorine. | [152] |
| Lithospermum canesens | Canesine, canescenine, 3'-acetycanesine, 3'-acetylcanescenine, lycopsamine, 7-acetyllycopsamine, 7-acetylintermedine. | [153,154] |
| L. erythrorhizon | Intermedine, myoscorpine, hydroxymyoscorpine. | [155] |
| L. officinale | Lithosenine, acetyllithosenine. | [156] |
| L. purpureocoeruleum | Lycopsamine | [60] |
| Macrotomia echioides | Macrotomine | [28] |
| Mertensia bakeri | Lycopsamine | [157] |
| M. ciliata | Intermedine, lycopsamine. | [157] |
| Messerschmidia argentea | Indicine, 3'-acetylindicine, and their N-oxide. | [158] |
| M. sibirica | Lycopsamine, 9-angeloylretronecine. | [28] |
| Moltikiopsis ciliata (Lithospermum callosum) | Echinatine, heliotrine. | [108] |
| Myosotis scorpioides (=M. palustris) | Myoscorpine, symphytine, scorpioidine, 7-acetylscorpioidine. | [159] |
| M. sylvatica | Heliosupine, 3'-acetylheliosupine, 9-angeloylretronecine, trachelanthamine. | [25] |
| Neatostema apulum | Amabiline, lycopsamine and their N-oxides. | [160] |
| Nonnea lutea | 7-viridiflorylretronecine | [60] |
| N. setosa | 7-viridiflorylretronecine | [60] |
| Omphalodes verna | Isoretronocanol or its isomer. | [27] |
| Onosma alborosea | Intermedine, lycopsamine, 7-acetylintermedine, 7-acetylycopsamine. | [161] |
| O. alboroseum × sanguinolentum | 9-Angeloylretronecine, echimidine, lycopsamine, intermedine, 7-acetylintermedine, 7-acetylycopsamine. | [161,162] |
| O. arenaria | 7-Acetyllycopsamine, 5,6-diydro-7,9-dimethoxy-7H-pyrrolizine, 7-acetylretronecine, 7-acetyl-9-(2-methylbutryl) retronecine, 7-acetyl-9-(2,3-dimethylbutryl) retronecine, 7-acetyl-9-(2-hydroxy-3-methylbutryl) retronecine, 7-acetyl-9-(2,3-dihydroxybutryl) retronecine, 9-(butyryl-2-ene) supinidine, 3'-acetylsupinine, uplandicine. | [163] |
| O. arenaria subsp. pennina | Intermedine, lycopsamine, 7-acetylintermedine, 7-acetylycopsamine. | [161] |
| O. erecta | N-oxides of 7-O-acetylechinatine, viridinatine, stereoisomer, 7-epi-echimiplatine, onosmerectine. | [164] |
| O. hetrophyllum | Helioridine, 1-methylene-8α-pyrrolizidine. | [165] |
| O. leptantha | Echihumiline, 3’-acetylechihumiline, leptanthine and their N-oxides. | [166] |
| O. stellulatum | Echimidine, 7-viridiflorylretronecine, heliospathuline, leptanthine, lyopsamine and heir N-oxides, 7-acetylintermedine, dihydroechinatine, trahelanthamine, uplandicine. | [60,167] |
| Paracaryum himalayense | Viridiflorine | [151] |
| P. intermedium | 7-Angeloylheliotridine, 7-senecioylheliotridine, rinderine, 7-angeloylrindrine, 7-senecioylrinderine, viridiflorine. | [40] |
| P. regulosum | Echinatine, heliosupine, 7-angeloylheliotridine, rinderine, viridiflorine. | [40] |
| Paracynoglossum imeretinum | Echinatine, heliosupine. | [29] |
| Pulmonaria obscura | Intermedine, lycopsamine, 7-acetylintermedine, 7-acetylycopsamine. | [168] |
| Rindera austroechinata | Echinatine, rinderine, 7-angeloylheliotridine | [57,152] |
| R. baldschuanica | Echinatine, rinderine, trachelanthamine, turkestanine | [152] |
| R. cyclodonta | Echinatine | [120] |
| R. echinata | Echinatine, trachelanthamine | [120] |
| R. oblongifolia | Cerategine, echinatine, turkestanine. | [152] |
| R. umbellata | 7-Angeloyl-9-(+)-trachelanthyl heliotridine, lindelofine, punctanecine, 7-angeloyl heliotridane, 7-angeloyl heliotridine, heliosupine, 9-(+)-trachelanthyl-laburnine, echinatine. | [169] |
| Solenanthus circinnatus | Echinatine | [151] |
| S. coronatus | Echinatine | [135] |
| S. karateginus | Cerategine, ehinatine. | [151] |
| S. turkestanicus | Rinderine, turkestanine. | [135,150] |
| Symphytum aintabicum | Echimidine | [170] |
| S. asperum | Echimidine, symphytine, asperumine, ehinatine, heliosupine, acetylechimidine (or its isomer), aetyllyopsamine (or its isomer), symviridine | [171,172] |
| S. bohemium | Echimidine, lycopsamine, 7-acetyllycopsamine, symphytine. | [173] |
| S. caucasium | Asperumine, echimidine, echinatine, heliotrine, lasiocarpine. | [174] |
| S. consolidum | Echimidine, symphytine. | [175] |
| S. grandiflorum | Echimidine, lycopsamine, symphytine. | [176] |
| S. ibericum | Echimidine, lycopsamine, symphytine. | [176] |
| S. officinale | Lycopsamine, 7-acetyllycopsamine, symphytine, echimidine, echinatine, heliosupine, intermedine, 7-acetylintermedine, viridiflorine, symviridine. | [17,162,171,172,177,178,179] |
| S. orientale | Anadoline, echimidine, symphytine. | [28] |
| S. peregrinum | Intermedine, 7-acetylintermedine, lycopsamine, 7-acetyllycopsamine, symphytine. | [27] |
| S. sylvaticum subsp. sepulcrale var. sepulcrale | Echimidine N-oxide | [170] |
| S. tanaiense | Echimidine, lycopsamine, 7-acetyllycopsamine, symphytine. | [173] |
| S. tuberosum | 7-Angeloylretronecine; anadoline, echimidine, lycopsamine, 7-acetyllycopsamine, symphytine. | [28,162,176] |
| S × uplandicum | Echimidine, intermedine, 7-acetylintermedine lycopsamine, 7-acetyllycopsamine, symphytine, symlandine, symviridine, uplandicine. | [172,180] |
| Tournefortia sarmentosa | Supinine | [28] |
| T. sibirica | Turneforcine | [29] |
| T. sogdiana | Echinatine | [57] |
| Trahlenthus hissaricus | Trachelanthine, trachelanthamine, viridiforine. | [29] |
| T. korolkovii | Trachelanthine, trachelanthamidine, trachelanthamine. | [57,150] |
| Trichodesma africanum | Europine, intermedine, lycopsamine, trichodesmine, retronecine, viridiflorine | [40,71,181] |
| T. ehrenbergii | Senkirkine, supinine. | [54] |
| T. incanum | Inanine, trihodesmine. | [46] |
| T. zeylanicum | Supinine | [182] |
| Ulugbekia tschimganica | Uluganine | [183] |
| Compounds | Sources | References |
|---|---|---|
| 3'-Acetylcanesine | Lithospermum canesens | [153,154] |
| 3'-Acetylcanescenine | Lithospermum canesens | [153,154] |
| 7-Acetyl-9-curassavoylheliotridine | Anchusa milleri | [40] |
| 7-Acetyl-9-(2,3-dihydroxybutryl) retronecine | Onosma arenaria | [163] |
| 7-Acetyl-9-(2-dimethylbutryl) retronecine | Onosma arenaria | [163] |
| 3'-Acetylechinatine | Cynoglossum creticum | [70] |
| Cynoglossum officinale | [67] | |
| Lindelofia spectabilis | [124] | |
| Messerschmidia argentea | [158] | |
| 7-Acetylechinatine | Cynoglossum amabile | [67] |
| Cynoglossum officinale | [67,84] | |
| Onosma erecta | [69] | |
| 3'- Acetylechihumiline | Onosma leptantha | [166] |
| 3'-Acetylechiumine | Amsinckia menziessi var. intermedia | [42] |
| Cryptantha clevelandii | [63] | |
| 3'-Acetylechimidine | Echium vulgare | [96] |
| 5'-Acetylechimidine | Echium vulgare | [11] |
| 5'-Acetyleuropine | Heliotropium disciforme | [122] |
| Heliotropium rotundifloium | [138] | |
| 7-Acetyleuropine | Heliotropium bovi | [110] |
| 3'-Acetylfurcatine | Amsinckia carinata | [42] |
| Amsinckia furacata | [42] | |
| 3'-Acetylheliosupine | Cynoglossum creticum | [70] |
| Cynoglossum officinale | [67,83] | |
| Heliotropium hirsutissimum | [129] | |
| 7-Acetyl-9-(2-hydroxy-3-methylbutryl) retronecine | Onosma arenaria | [163] |
| 3'-Acetylindicine | Heliotropium arborescens (H. peruvianum) | [28] |
| Messerschmidia argentea | [158] | |
| 3'-Acetylintermedine | Amsinckia eastwoodiae | [42] |
| Amsinckia lycopsoides | [42] | |
| Amsinckia lycopsoides × menziesii var. intermedia | [42] | |
| Amsinckia menziesii var. intermedia | [43] | |
| Amsinckia retrosa | [42] | |
| Amsinckia spectabilis var. microcarpa | [42] | |
| Amsinckia spectabilis var. spectabilis | [42] | |
| Amsinckia tessellata | [46] | |
| Cryptantha cana | [63] | |
| Cryptantha crassipes | [64] | |
| Cryptantha clevelandii | [63] | |
| Cryptantha flava | [63] | |
| Cryptantha leiocarpa | [63] | |
| Cryptantha thyrsiflora | [63] | |
| Cryptantha virgata | [63] | |
| Echium pininana | [95] | |
| 7-Acetylintermedine | Amsinckia carinata | [42] |
| Amsinckia menziesii var. intermedia | [42] | |
| Amsinckia tessellata | [46] | |
| Anchusa arvensis | [40] | |
| Borago officinalis | [55] | |
| Cryptantha cana | [63] | |
| Cryptantha crassipes | [64] | |
| Cryptantha flava | [63] | |
| Cryptantha thyrsiflora | [63] | |
| Cryptantha virgata | [63] | |
| Lappula myostis | [35] | |
| Lithospermum canescens | [153,154] | |
| Onosma alborosea | [161] | |
| Onosma arenaria pennina | [161] | |
| Onosma stellulatum | [167] | |
| Pulmonaria obscura | [168] | |
| Symphytum officinale | [171,172,179] | |
| Symphytum peregrinum | [27] | |
| Symphytum × uplandicum | [180] | |
| 5'-Acetyllasiocarpine | Heliotropium hirsutissimum | [129] |
| 7-Acetyl-9-latifolylretronecine | Hackelia californica | [101] |
| 3'-Acetyllithosenine | Lithospermum officinale | [156] |
| 3'-Acetyllycopsamine | Amsinckia grandiflora | [42] |
| Amsinckia lunaris | [42] | |
| Amsinckia menziesii | [45] | |
| Amsinckia menziesii var. intermedia | [42] | |
| Amsinckia tessellata | [46] | |
| Amsinckia tessellata var. tessellata | [42] | |
| Cerinthe glabra | [40] | |
| Cryptantha cana | [63] | |
| Cryptantha crassipes | [64] | |
| Cryptantha flava | [63] | |
| Cryptantha jamesii | [66] | |
| Cryptantha thyrsiflora | [63] | |
| Echium lycopsis (E. plantagineum) | [93] | |
| 7-Acetyllycopsamine | Amsinckia grandiflora | [42] |
| Amsinckia menziesii | [45] | |
| Amsinckia menziesii var. intermedia | [42] | |
| Amsinckia tessellata | [46] | |
| Amsinckia tessellate var. tessellata | [42] | |
| Anchusa arvensis | [40] | |
| Anchusa hispida | [40] | |
| Anchusa officinalis | [48] | |
| Borago officinalis | [55] | |
| Cerinthe glabra | [40] | |
| Cryptantha cana | [63] | |
| Cryptantha crassipes | [64] | |
| Cryptantha flava | [63] | |
| Cryptantha jamesii | [66] | |
| Cryptantha thyrsiflora | [63] | |
| Cryptantha virgata | [63] | |
| Echium horridum | [90] | |
| Echium hypertropicum | [92] | |
| Echium humile | [91] | |
| Echium rauwolfi | [90] | |
| Lappula myostis | [35] | |
| Lappula spinocarpos | [40] | |
| Lithospermum canesens | [153,154] | |
| Onosma alborosea | [161] | |
| Onosma arenaria | [163] | |
| Onosma arenaria pennina | [161] | |
| Pulmonaria obscura | [168] | |
| Symphytum bohemium | [173] | |
| Symphytum officinale | [171,172,179] | |
| Symphytum peregrinum | [27] | |
| Symphytum tanaiense | [173] | |
| Symphytum tubertosum | [28] | |
| Symphytum × uplandicum | [180] | |
| 7-Acetyl-9-(2-methylbutyryl) retronecine | Onosma arenaria | [163] |
| 3'-Acetylmyscorpine | Amsinckia menziesii var. intermedia | [42] |
| Amsinckia retrosa | [42] | |
| 7-Acetylretronecine | Onosma arenaria | [163] |
| 3'-Acetylrinderine | Cynoglossum columnae | [69] |
| Heliotropium transplinum | [144] | |
| 7-Acetyl-9-sarracinoyl retronecine | Alkanna tinctoria | [40] |
| 7-Acetylscorpioidine | Myosotis scorpioides | [159] |
| 3'-Acetylsupinine | Onosma arenaria | [163] |
| 3'-Acetyltessellatine | Amsinckia tessellata var. tessellata | [42] |
| 9-Acetytessellatine | Amsinckia grandifkora | [42] |
| Amsinckia lunaris | [42] | |
| Amsinckia tessellata var. tessellata | [42] | |
| 3'-Acetyltrachelanthamine | Heliotropium floridum | [128] |
| 9-Acetyltrachelanthamine | Anchusa arvensis | [40] |
| 3'-Acetylviridiflorine | Amsinckia tessellata var. gloriosa | [42] |
| 9-(3'-Acetyl)viridiflory retronecine | Heliotropium curassavicum var. argentinum | [119] |
| Heliotropium curassavicum var. curassavicum | [119] | |
| 9-(3'-Acetylviridifloryl) turniforcidine | A. douglasiana | [42] |
| A. tessellata var. tessellata | [42] | |
| 7-Acetylvulgarine | Echium vulgare | [11] |
| Amabiline | Amsinckia douglasina | [42] |
| Amsinckia eastwoodiae | [42] | |
| Amsinckia grandiflora | [42] | |
| Amsinckia lycopsoides | [42] | |
| Amsinckia lycopsodes menziesii var. intermedium | [42] | |
| Amsinckia menziesii var intermedium | [42] | |
| Amsinckia retrosa | [42] | |
| Amsinckia tessellata var. gloriosa | [42] | |
| Amsinckia tessellata var. tessellata. | [42] | |
| Asperugo procumbens | [47] | |
| Borago officinalis | [55] | |
| Cryptantha confertiflora | [63] | |
| Cryptantha crassipes | [64] | |
| Cryptantha virginensis | [63] | |
| Cynoglossum amabile | [67,68] | |
| Cynoglossum glochidiatum | [78] | |
| Heliotropium spathulatum | [140] | |
| Lappula spinocarpos | [40] | |
| Lindelofia angustiflora | [78] | |
| Neatostema apulum | [160] | |
| Anadoline | Symphytum orientale | [28] |
| Symphytum tuberosum | [28] | |
| 7α-Angeloyl-1-chloromethy-1,2-dihydropyrrolizidine | Cynoglossum creticum | [70] |
| 7-Angeloyl-9-(2,3-dihydroxybutyryl)heliotridine | Cynoglossum officinale | [67] |
| 7-Angeloyl-9-(2,3-dihydroxybutyryl)retronecine | Echium horridum | [90] |
| Echium rauwolfii | [90] | |
| Echium setosum | [96] | |
| Onosma arenaria | [163] | |
| 7-Angeloyl-9-(2,3-dihydroxypropanoyl)retronecine | Alkanna orientalis | [40] |
| Alkanna tinctoria | [40] | |
| 7-Angeloyl-1-formyl-6,7-dihydro-5H-pyrrolizidine | Cynoglossum officinale | [67] |
| 7-Angeloyl-9-(hydroxypropenoyl)retronecine | Alkanna orientalis | [40] |
| Alkanna tinctoria | [40] | |
| 7-Angeloylechinatine | Cynoglossum officinale | [67] |
| 7-Angeloylheliotridine | Anchusa hispida (Gastrocotyle hispida) | [40] |
| Anchusa milleri | [40] | |
| Cynoglossum creticum | [70,72] | |
| Cynoglossum latifolium | [79] | |
| Cynoglossum officinale | [67,84] | |
| Heliotropium curassavicum | [117] | |
| Heliotropium supinum | [27,143] | |
| Lappula spinocarpos | [40] | |
| 7-Angeloylheliotridine | Paracaryum intermedium | [40] |
| Paracaryum regulosum | [40] | |
| Rindera austroechinata | [57] | |
| 7-Angeloylheliotrine | Heliotropium circinatum | [114] |
| Heliotropium digynum (H. luteum) | [121] | |
| Heliotropium eichwaldii | [124] | |
| Heliotropium europaeum | [127] | |
| Rindera umbellata | [169] | |
| 7-Angeloyllycopsamine | Echium horridum | [90] |
| Echium rauwolfii | [90] | |
| 7-Angeloyl-9-(2-methylbutyryl)heliotridine | Cynoglossum creticum | [70] |
| Cynoglossum offocinale | [67] | |
| 7-Angeloyl-9-(2-methylbutyryl)retronecine | Echium horridum | [90] |
| Echium rauwolfii | [90] | |
| 7-Angeloylretronecine | Alkanna orientalis | [40,41] |
| Alkanna tinctoria (A. tuberculata) | [40,184] | |
| Arnebia decumbens | [52] | |
| Arnebia euchroma | [53] | |
| Cerinthe minor | [60] | |
| Cryptantha fendleri | [63] | |
| Cryptantha leiocarpa | [63] | |
| Echium amoenum | [87] | |
| Echium glomeratum | [89] | |
| Echium horridum | [90] | |
| Echium rauwolfii | [90] | |
| Echium setosum | [96] | |
| Echium simplex | [162] | |
| Echium vulgare | [96] | |
| Eritrichium rupestre | [60] | |
| Hackelia longituba | [104] | |
| Heliotropium bursiferum | [113] | |
| Symphytum tuberosum | [162] | |
| 9-Angeloylretronecine | Alkanna orientalis | [40,41] |
| Arnebia decumbens | [52] | |
| Cryptantha fendleri | [63] | |
| Cryptantha leiocarpa | [63] | |
| Cynoglossum amabilie | [67] | |
| Echium glomaratum | [89] | |
| Echium hypertropicum | [92] | |
| Echium setosum | [96] | |
| Echium simplex | [162] | |
| Echium vulgare | [96] | |
| Hackelia longituba | [104] | |
| Messerchimidia sibrica | [28] | |
| Myosotis sylvatica | [25] | |
| 7-Angeloylrinderine | Cynoglossun officinale | [67] |
| 9-Angeloyltrachelanthamidine | Anchusa arvensis | [40] |
| 7-Angeloyl-9-(+)-trachelanthylheliotridine | Rindera umbellata | [169] |
| 9-Angeloyl-7-viridiflorylretronecine | Cerinthe minor | [60] |
| Cynoglossum amabile | [60] | |
| Cynoglossum clandestinum | [60] | |
| Echium tuberculatum | [60] | |
| Eritrichium rupestre | [60] | |
| Asperumine | Echium vulgare | [98] |
| Symphytum asperum | [172] | |
| Symphytum caucasium | [174] | |
| 9-(Butyryl-2-ene) supinidine | Onosma arenaria | [163] |
| Canescine | Lithospermum canescens | [153,154] |
| Canescenine | Lithospermum canescens | [153,154] |
| Carategine | Lindelofia tschimganica | [27] |
| Rindera oblongifolia | [152] | |
| Solanthus karateginus | [151] | |
| Coromandaline | Heliotropium curassavicum | [117,118] |
| Heliotropium spathulatum | [140] | |
| Coromandalinine | Heliotropium curassavicum | [117] |
| Heliotropium spathulatum | [140] | |
| Cryptanthine | Cryptantha utahensis | [65] |
| Curassanecine | Heliotropium curassavicum | [117] |
| Curassavine | Anchusa officinalis | [49] |
| Heliotropium curassavicum | [117] | |
| Heliotropium spathulatum | [140] | |
| Curassavinine | Heliotropium curassavicum | [117] |
| Heliotropium spathulatum | [140] | |
| 9-Curassavorylheliotridine | Anchusa milleri | [40] |
| Cynaustrine | Cynoglossum australe | [68] |
| Cynoglossum lanceolatum | [78] | |
| Cynoglossum montanum | [81] | |
| Cynaustraline | Amsinckia douglasiana | [42] |
| Cynoglossum australe | [68] | |
| Cynoglossum furcatum | [73] | |
| Cynoglossum lanceolatum | [78] | |
| Cynoglossum montanum | [81] | |
| Cynoglossamine | Cynoglossum creticum | [72] |
| Dehydroheliotrine | Heliotropium europaeum | [185] |
| 5-Deoxylasiocarpine | Heliotropium dissitiflorum | [123] |
| 3',7-Diacetylintermedine | Amsinckia menziesii var. intermedia | [42] |
| Amsinckia tessellata | [46] | |
| Anchusa arvensis | [40] | |
| 3',7-Diacetyllycopsamine | Amsinckia menziesii | [45] |
| Amsinckia tessellata | [46] | |
| 3',7-Diacetyllycopsamine | Anchusa arvensis | [40] |
| Cerinthe glabra | [59] | |
| 3',9-Diacetyltessellatine | Amsinckia tessellata var. tessellata | [42] |
| 5,6-Dihydro-7,9-dimethoxy-7H-pyrrolizine | Onosma arenaria | [163] |
| Dihydroechinatine | Onosma stellulatum | [167] |
| thero-2'',3''-Dihydroxyechiumine | Cryptantha clevelandiiCryptantha inequata | [63,65] |
| Cryptantha leiocarpa | [63] | |
| Dihydroxytriangularine | Alkanna orientalis | [40,41] |
| Alkanna tinctoria | [40,41] | |
| Dihydroxytriangularicine | Alkanna orientalis | [40] |
| Alkanna tinctoria | [40] | |
| Echihumiline | Echium hypertropicum | [92] |
| Echium humile | [91] | |
| Echium vulgare | [96] | |
| Onosma leptantha | [166] | |
| Echimidine | Arnebia hispidissima | [54] |
| Cryptantha inequata | [65] | |
| Echium amoenum | [87] | |
| Echium angustifolium | [88] | |
| Echium horridum | [90] | |
| Echium humile | [91] | |
| Echium hypertropicum | [92] | |
| Echium italicum | [28] | |
| Echium lycopsis (E. plantagineum) | [94] | |
| Echium pininana | [95] | |
| Echium rauwolfii | [90] | |
| Echium sericeum | [54] | |
| Echium. setosum | [96] | |
| Echium stenosiphon subsp. stenosiphon | [92] | |
| Echium tuberculatum | [60] | |
| Echium vulgare | [11,96] | |
| Echium wildpretti | [99] | |
| Onosma stellulatum | [60,167] | |
| Symphytum aintabicum | [170] | |
| Symphytum asperum | [171] | |
| Symphytum bohemium | [173] | |
| Symphytum caucasium | [174] | |
| Symphytum consolidum | [175] | |
| Symphytum sylvaticum | [170] | |
| Symphytum tuberosum | [162] | |
| Echimidine isomer (tigloyl) | Echium amoenum | [87] |
| Echium horridum | [90] | |
| Echimidine isomer (tigloyl) | Echium rauwolfii | [90] |
| Echium setosum | [96] | |
| Echium vulgare | [96] | |
| Echimiplatine | Cryptantha crassipes | [64] |
| Echium plantagineum | [93] | |
| Echium vulgare | [11] | |
| Echinatine | Asperugo procumbens | [47] |
| Cynoglossum amabile | [67,68] | |
| Cynoglossum columnae | [69] | |
| Cynoglossum creticum | [70,71,72] | |
| Cynoglossum furcatum (C. zeylanicum) | [74,75] | |
| Cynoglossum germanicum | [77] | |
| Cynoglossum macrostylum | [80] | |
| Cynoglossum montanum | [81] | |
| Cynoglossum nervosum | [82] | |
| Cynoglossum officinale | [67,84] | |
| Cynoglossum pictum | [85] | |
| Heliotropium circinatum | [114] | |
| Heliotropium indicum | [130] | |
| Heliotropium suaveolens | [29] | |
| Heliotropium supinum | [27,143] | |
| Lappula glochidiata | [146] | |
| Lindelofia longiflora | [149] | |
| Lindelofia spectabilis | [124] | |
| Lindelofia stylosa | [135] | |
| Moltikiopsis ciliata | [108] | |
| Paracaryum regulosum | [40] | |
| Paracynoglossum imeretium | [29] | |
| Rindera austroechinata | [57,152] | |
| Rindera baldschuanica | [152] | |
| Rindera cyclodonata | [120] | |
| Rindera echinata | [120] | |
| Rindera oblogifolia | [152] | |
| Rindera umbellata | [169] | |
| Solenanthus circinnatus | [151] | |
| Solenanthus coronatus | [135] | |
| Solenanthus karateginus | [151] | |
| Symphytum asperum | [172] | |
| Symphytum caucasium | [174] | |
| Symphytum officinale | [179] | |
| Tournefortia sogdiana | [57] | |
| Echiumine | Amsinckia carinata | [42] |
| Amsinckia eastwoodiae | [42] | |
| Amsinckia hispida | [44] | |
| Amsinckia intermedia | [44] | |
| Amsinckia lycopsoides | [44] | |
| Amsinckia menziesii var. intermedia | [42] | |
| Cryptantha clevelandii | [63] | |
| Cryptantha crassipes | [64] | |
| Cryptantha leiocarpa | [63] | |
| Echium lycopsis | [94] | |
| Echiupine | Echium pininana | [95] |
| Echiuplatine | Cryptantha inequata | [65] |
| Echium plantagineum | [93] | |
| Echium vulgare | [11] | |
| Echivulgarine | Echium vulgare | [11] |
| Ehretinine | Ehretia aspera | [100] |
| 2'',3''-Epoxyechiumine | Cryptantha clevelandii | [63] |
| Cryptantha leiocarpa | [63] | |
| Erythro-2'',3''-chloro-2''-hydroxyechiumine | Cryptantha clevelandii | [63] |
| Cryptantha leiocarpa | [63] | |
| 7-Epi-echimiplatine | Onosma erecta | [69] |
| 1α-2α-Epoxy-1β-hydroxymethyl-8α-pyrrolizidine | Heliotropium transalpinum var. transalpinum | [145] |
| Europine | Heliotropium arbainense | [71] |
| Heliotropium bacciferum | [108,109] | |
| Heliotropium bovi | [110] | |
| Heliotropium circinatum | [114] | |
| Heliotropium crassifolium | [115] | |
| Heliotropium digynum (H.luteum) | [121] | |
| Heliotropium dissitiflorum | [123] | |
| Heliotropium esfandiarii | [125] | |
| Heliotropium europaeum | [126,127,186] | |
| Heliotropium hirsutissinum | [129] | |
| Heliotropium marifolium | [29] | |
| Heliotropium maris mortui | [71] | |
| Heliotropium rotundifolium | [137,138] | |
| Floridanine | Cordia sinensis | [62] |
| Floridimine | Heliotropium floridum | [128] |
| Floridine | Heliotropium floridum | [128] |
| Floridinine | Heliotropium floridum | [128] |
| Furcatine | Amsinckia carinata | [42] |
| Amsinckia furacata | [42] | |
| Amsinckia vernicosa | [42] | |
| Hackelidine | Hackelia californica | [102] |
| Heleurine | Heliotropium bacciferum | [109] |
| Heliotropium circinatum | [114] | |
| Heliotropium disciforme | [122] | |
| Heliotropium europaeum | [126,127,186] | |
| Heliotropium hirsutissimum | [129] | |
| Heliotropium indicum | [131] | |
| Helibracteatine | Heliotropium bracteatum | [112] |
| Helibractinecine | Heliotropium bracteatum | [111] |
| Helibracteatinecine | Heliotropium bracteatum | [112] |
| Helibracteatinine | Heliotropium bracteatum | [112] |
| Heliofoline | Heliotropium ovalifolium | [136] |
| Helindicine | Heliotropium indicum | [130] |
| Heliocoromandaline | Heliotropium curassavicum | [117] |
| Heliocurassavine | Heliotropium curassavicum | [117] |
| Heliocurassavicine | Heliotropium curassavicum | [117] |
| Heliocurassavinine | Heliotropium curassavicum | [117] |
| Heliospathine | Heliotropium spathulatum | [140] |
| Heliospathuline | Heliotropium spathulatum | [140] |
| Onosma stellulatum | [167] | |
| Heliosupine | Cynoglossium creticum | [70,71,72] |
| Cynoglossium macrostylum | [80] | |
| Cynoglossium montatum | [81] | |
| Cynoglossium officinale | [67,83,84] | |
| Cynoglossium pictum | [85] | |
| Cynoglossium viridiforum | [86] | |
| Heliotropium supinum | [27,143] | |
| Myosotis sylvatica | [25] | |
| Paracaryum regulosum | [90] | |
| Paracynoglossum imeretium | [29] | |
| Rindera umbellata | [169] | |
| Symphytum asperum | [171] | |
| Symphytum officinale | [179] | |
| Heliotridine | Anchusa milleri | [90] |
| Onosma heterophyllum | [165] | |
| Heliotridine 2S-hydroxy-2S(1S-hydroxyethyl0-4-methyl-pentanoyl ester | Anchusa strigosa | [50] |
| Heliotrine | Arenbia decumbens | [52] |
| Cynoglossum nervosum | [82] | |
| Heliotropium acutifolium | [105] | |
| H. arbainense | [71] | |
| H. bacciferum | [108,109] | |
| Heliotropium circinatum | [114] | |
| Heliotropium dasycarpum | [120] | |
| Heliotropium digynum | [121] | |
| Heliotropium disciforme | [122] | |
| Heliotropium dissitiflorum | [123] | |
| Heliotropium eichwaldii | [124] | |
| Heliotropium europaeum | [127,185,186] | |
| Heliotropium hirsutissimum | [129] | |
| Heliotropium indicum | [130,131] | |
| Heliotropium lasiocarpum | [29] | |
| Heliotrine | Heliotropium marifolium | [29] |
| Heliotropium olgae | [135] | |
| Heliotropium popovii subsp. gillianum | [28] | |
| Heliotropium rotundifolum | [137,138] | |
| Heliotropium suaveolens | [29] | |
| Heliotropium supinum | [27,143] | |
| Heliotropium transoxanum | [27] | |
| Molyikiopsis ciliate (Lithospermum callosum) | [108] | |
| Symphytum caucasium | [174] | |
| Heliovicine | Heliotropium spathulatum | [140] |
| Heliscabine | Heliotropium scabrum | [139] |
| Hydroxymyoscorpine | Echium pininana | [95] |
| Ilamine | Heliotropium crassifolium | [115] |
| Incanine | Heliotropium olgae | [29] |
| Trichodesma incanum | [46] | |
| Indicine | Heliotropium amplexicaule | [29] |
| Heliotropium arborescens (H. pruvianum) | [107] | |
| Heliotropium indicum | [130,131] | |
| Heliotropium marifolium | [29] | |
| Heliotropium transalpinum | [144] | |
| Messerschmidia argentea | [158] | |
| Indicinine | Heliotropium indicum | [130,131] |
| Isoechinatine | Cynoglossum furcatum (C. zeylanicum) | [75] |
| Isolycopsamine | Heliotropium keralense | [190] |
| Isoretronocanol (or its isomer) | Omphalodes verna | [27] |
| 9-(3'-Isovaleryl)viridiflory retronecine | Heliotropium curassavicum var. argentinum | [119] |
| Heliotropium curassavicum var. curassavicum | [119] | |
| Intermedine | Amsinckia carinata | [42] |
| Amsinckia douglasiana | [42] | |
| Amsinckia eastwoodiae | [42] | |
| Amsinckia furacata | [42] | |
| Amsinckia. grandiflora | [42] | |
| Amsinckia hispida | [44] | |
| Amsinckia intermedia | [44] | |
| Amsinckia lunaris | [42] | |
| Amsinckia lycopsoides | [42,44] | |
| Amsinckia lycopsoides × menziesii var. intermedia | [42] | |
| Amsinckia menziesii | [45] | |
| Amsinckia menziesii var. intermedia | [42] | |
| Amsinckia retrosa | [42] | |
| Intermedine | Amsinckia spectabilis var. microcarpa | [42] |
| Amsinckia spectabilis var. spectabilis | [42] | |
| Amsinckia spectabilis var. nicolai | [42] | |
| Amsinckia tessellata | [46] | |
| Amsinckia tessellata var. gloriosa | [42] | |
| Amsinckia tessellata var. tessellata | [42] | |
| Amsinckia vernicosa | [42] | |
| Anchusa arvensis (Lycopsis arvensis) | [40] | |
| Anchusa hispidia (Gastrocotyle hispidia) | [40] | |
| Anchusa officinalis | [48] | |
| Borago officinalis | [55] | |
| Cerinthe minor | [61] | |
| Cryptantha cana | [63] | |
| Cryptantha clevelandii | [63] | |
| Cryptantha confertiflora | [63] | |
| Cryptantha flava | [63] | |
| Cryptantha inequata | [65] | |
| Cryptantha jamesii | [66] | |
| Cryptantha leiocarpa | [63] | |
| Cryptantha thyrsiflora | [63] | |
| Cryptantha virgata | [63] | |
| Cryptantha virginiensis | [63] | |
| Heliotropium keralense | [133] | |
| Heliotropium transalpinum | [144] | |
| Lappula myostis | [35] | |
| Lappula spinocarpas | [40] | |
| Lithospermum erythrorhizon | [155] | |
| Mertensia ciliate | [157] | |
| Onosma alborosea | [161] | |
| Onosma arenaria pennina | [161] | |
| Pulmonaria obscura | [168] | |
| Symphytum peregrinum | [27] | |
| Symphytum × uplandicum | [180] | |
| Trichodesma africanum | [71] | |
| Lactodine | Cynoglossum furcatum | [76,187] |
| Lasiocarpine | Heliotropium arbainense | [71] |
| Heliotropium bovei | [110] | |
| Heliotropium circinatum | [114] | |
| Heliotropium digynum | [121] | |
| Heliotropium eichwaldii | [124] | |
| Heliotropium europaeum | [126,127,186] | |
| Heliotropium hirsutissimum | [129] | |
| Heliotropium indicum | [130,131] | |
| Heliotropium lasiocarpum | [29] | |
| Heliotropium marifolium | [29] | |
| Heliotropium maris mortui | [137] | |
| Heliotropium rotundifolium | [137,138] | |
| Heliotropium suaveolens | [29] | |
| Lappula intrmedia | [27] | |
| Symphytum caucasium | [174] | |
| Symphytum officinale | [177] | |
| Latifoline | Cryptantha fendleri | [63] |
| Cynoglossum latifolium | [79] | |
| Hackelia californica | [101] | |
| Hackelia floribunda | [103] | |
| Hackelia longituba | [104] | |
| Lindelofia stylosa | [27] | |
| 9-Latifolylretronecine | Hackelia californica | [101] |
| Leptanthine | Cryptantha crassipes | [64] |
| Echium plantagineum | [93] | |
| Echium vulgare | [11] | |
| Onosma leptantha | [166] | |
| Onosma stellulatum | [167] | |
| Lindelofine | Amsickia menzesii var. intermedia | [42] |
| Amsickia spectabilis var. microcarpa | [42] | |
| Amsickia spectabilis var. spectabilis | [42] | |
| Amsickia spectabilis var. nicola | [42] | |
| Lindelofia anchusoides (L. macrostyle) | [28] | |
| Rindera umbellata | [169] | |
| Lindelofamine | Lindelofia anchusoides (L. macrostyle) | [28] |
| Lindelofidine | Heliotropium angiospermum | [106] |
| Heliotropium confertifolum | [106] | |
| Heliotropium curassavicum | [106] | |
| Heliotropium foliosissimum | [106] | |
| Heliotropium fruticosum | [106] | |
| Heliotropium gregii | [106] | |
| Heliotropium indicum | [106,116] | |
| Heliotropium molle | [106] | |
| Heliotropium procumbens | [106] | |
| Heliotropium queretaroanum | [106] | |
| Heliotropium spathulatum | [116] | |
| Heliotropium sessei | [106] | |
| Heliotropium racemosum | [106] | |
| Heliotropium ternatum | [106] | |
| Heliotropium wigginsii | [106] | |
| Lithosenine | Lithospermum officinale | [156] |
| Longitubine | Hackelia californica | [101] |
| Hackelia logituba | [104] | |
| Lycopsamine | Amsinckia carinata | [42] |
| Amsinckia douglasiana | [42] | |
| Amsinckia eastwoodiae | [42] | |
| Amsinckia furacata | [42] | |
| Amsinckia grandiflora | [42] | |
| Amsinckia hispida | [44] | |
| Amsinckia intermedia | [44] | |
| Amsinckia lunaris | [42] | |
| Amsinckia lycopsoides | [42,44] | |
| Amsinckia lycopsoides × menziesii var. intermedia | [42] | |
| Amsinckia menziesii | [45] | |
| Amsinckia menziesii var. intermedia | [42] | |
| Amsinckia retrosa | [42] | |
| Amsinckia spectabilis var. microcarpa | [42] | |
| Amsinckia spectabilis var. spectabilis | [42] | |
| Amsinckia spectabilis var. nicolai | [42] | |
| Amsinckia tessellata | [46] | |
| Amsinckia tessellata var. gloriosa | [42] | |
| Amsinckia tessellata var. tessellata | [42] | |
| Amsinckia vernicosa | [42] | |
| Anchusa arvensis (Lycopsis arvensis) | [40] | |
| Anchusa hispidia (Gastrocotyle hispidia) | [40] | |
| Anchusa officinalis | [48] | |
| Arnebia decumbens | [52] | |
| Borago officinalis | [55] | |
| Cerinthe glabra | [59] | |
| Cerinthe minor | [61] | |
| Cryptantha cana | [63] | |
| Cryptantha confertiflora | [63] | |
| Cryptantha flava | [63] | |
| Cryptantha inequata | [65] | |
| Cryptantha jamesii | [66] | |
| Cryptantha thyrsiflora | [63] | |
| Cryptantha virgata | [63] | |
| Cryptantha virginiensis | [63] | |
| Cynoglossum amabile | [67] | |
| Echium hypertropicum | [92] | |
| Heliotropium transalpinum | [144] | |
| Heliotropium megalanthum | [134] | |
| Heliotropium steudneri | [27] | |
| Lappula myostis | [35] | |
| Lappula spinocarpas | [40] | |
| Lithospermum purpureocoeruleum | [60] | |
| Mertensia bakeri | [157] | |
| Mertensia ciliate | [157] | |
| Mertensia sibirica | [28] | |
| Neatostema apulum | [60] | |
| Onosma alborosea | [161] | |
| Onosma arenaria pennina | [161] | |
| Onosma stellulatum | [167] | |
| Pulmonaria obscura | [168] | |
| Symphytum bohemium | [173] | |
| Symphytum grandiflorum | [176] | |
| Symphytum ibericum | [176] | |
| Symphytum peregrinum | [27] | |
| Symphytum officinale | [171,177] | |
| Symphytum tanaiense | [173] | |
| Symphytum tuberosum | [162,176] | |
| Symphytum × uplandicum | [180] | |
| Trichodesma africanum | [71] | |
| Macrophylline | Cordia myxa | [62] |
| Macrotamine | Macrotomia echioides | [28] |
| Megalanthonine | Heliotropium megalanthum | [134] |
| Methyechiuplatine | Cryptantha inequata | [65] |
| 1-Methylene-8α-pyrrolizidine | Onosma heterophyllum | [165] |
| 7-(2-Methylbutyryl)retronecine | Echium humile | [91] |
| Echium vulgare | [96] | |
| 9-(2-Methylbutyryl)retronecine | Echium vulgare | [96] |
| 7-(2-Methylbutyryl)-9-(2,3-dihydroxybutyryl)retronecine | Echium humile | [91] |
| Echium vulgare | [96] | |
| 7-(2-Methylbutyryl)-9-echimidinyl retronecine | Echium humile | [91] |
| Echium hypertropicum | [92] | |
| Echium stenosiphon subsp. stenosiphon | [92] | |
| Echium vulgare | [96] | |
| Monocrotaline | Arnebia hispidissima | [54] |
| Lindelofia spectabilis | [124] | |
| Myoscorpine | Amsinckia carinata | [42] |
| Amsinckia menziesii var. intermedia | [42] | |
| Amsinckia retrosa | [42] | |
| Amsinckia spectabilis var. microcarpa | [42] | |
| Echium pininana | [95] | |
| Linelfolia erythrorhizon | [155] | |
| Myosotis scorpioides | [159] | |
| Neocoromandaline | Cynoglossum furcatum | [74] |
| Neolatifoline | Cryptantha fendleri | [63] |
| Hackelia logituba | [104] | |
| Onosmerectine | Onosma erecta | [69] |
| (7S,8R)Petranine | Echium glomeratum | [89] |
| (7S,8S)Petranine | Echium glomeratum | [89] |
| Pictumine | Cynoglossum pictum | [28] |
| Platynecine | Cryptantha leiocarpa | [63] |
| Platynecine N-oxide 2S-hydroxy-2S(1S-hydroxyethyl)-4-methyl-pentanosyl ester | Anchusa strigosa | [50,51] |
| Punctanecine | Rindera umbellata | [169] |
| Pycnanthine | Echium humile | [91] |
| Retronecine | Anchusa hispida (Gastrocotyle hispida) | [40] |
| Echium vulgare | [96] | |
| Heliotropium angiospermum | [106] | |
| Heliotropium confertifolum | [106] | |
| Heliotropium curassavicum | [106,116] | |
| Heliotropium foliosissimum | [106] | |
| Heliotropium fruticosum | [106] | |
| Heliotropium gregii | [106] | |
| Heliotropium indicum | [106,116] | |
| Heliotropium keralense | [133] | |
| Heliotropium molle | [106] | |
| Heliotropium ovalifolium | [136] | |
| Heliotropium procumbens | [106] | |
| Heliotropium queretaroanum | [106] | |
| Heliotropium racemosum | [106] | |
| Heliotropium scabrum | [139] | |
| Heliotropium spathulatum | [106,116] | |
| Heliotropium sessei | [106] | |
| Heliotropium ternatum | [106] | |
| Heliotropium wigginsii | [106] | |
| Lappula spinocarpos | [40] | |
| Trichodesma africanum | [40] | |
| Retronecine-7:9- dibenzoate | Caccinea glauca | [58] |
| Retronecine 2S-hydroxy-2S(1S-hydroxyethyl)-4-methyl-pentanosyl ester | Anchusa strigosa | [50,51] |
| Retronecine 2S-hydroxy-2S(1R-hydroxyethyl)-4-methyl-pentanosyl ester | Anchusa strigosa | [50,51] |
| Rinderine | Anchusa milleri | [40] |
| Arnebia decumbens | [52] | |
| Cynoglossum columnae | [69] | |
| Cynoglossum creticum | [70,72] | |
| Cynoglossum officinale | [67,84] | |
| Heliotropium indicum | [130,131] | |
| Heliotropium peruvianum | [29] | |
| Heliotropium transalpinum | [144] | |
| Paracaryum intermedium | [40] | |
| Paracaryum regulosum | [40] | |
| Rindera austroechinata | [57] | |
| Rindera baldschuanica | [152] | |
| Solanthus turkestanicus | [150] | |
| Scorpioidine | Myosotis scorpioides | [159] |
| 7-Senecioylhelotridine | Cynoglossum creticum | [70] |
| Paracarum intermedium | [40] | |
| 7-Senecioylretronecine | Alkanna orientalis | [40] |
| Echium hypertropicum | [92] | |
| Echium humile | [91] | |
| 9-Senecioylretronecine | Alkanna orientalis | [40] |
| Echium humile | [91] | |
| 7-Senecioylrinderine | Paracaryum intermedium | [40] |
| 7-Senecioyllycopsamine | Echium humile | [91] |
| Sincamidine | Amsinckia intermedia | [44] |
| Senkirkine | Trichodesma ehrenbergii | [54] |
| Strigosine | Heliotopium strigosum | [25,141] |
| Subulacine | Heliotopium angiospermum | [29] |
| Heliotopium molle | [29] | |
| Heliotopium subulacatum | [29] | |
| Heliotopium ternatum | [27] | |
| Heliotopium transalpinum var. transalpinum | [145] | |
| Supinine | Amsinckia carinata | [42] |
| Amsinckia eastwoodiae | [42] | |
| Amsinckia furacata | [42] | |
| Amsinckia lunaris | [42] | |
| Amsinckia lycopsoides | [42,44] | |
| Amsinckia lycopsoides × menziesii var. intermedia | [42] | |
| Amsinckia menziesii var. intermedia | [42] | |
| Amsinckia retrosa | [42] | |
| Amsinckia spectabilis var. microcarpa | [42] | |
| Amsinckia spectabilis var. spectabilis | [42] | |
| Amsinckia spectabilis var. nicolai | [42] | |
| Amsinckia tessellate var. gloriosa | [42] | |
| Amsinckia tessellate var. tessellate | [42] | |
| Anchusa arvensis (Lycopsis arvensis) | [40] | |
| Anchusa melleri | [40] | |
| Arnebia decumbens | [52] | |
| Borago officinalis | [55] | |
| Caccina crassifolia | [57] | |
| Cerinthe glabra | [59] | |
| Cynoglossum amabile | [67] | |
| Cynoglossum creticum | [70] | |
| Heliotropium bacciferum | [109] | |
| Heliotropium europaeum | [127] | |
| Heliotropium hirsutissimum | [129] | |
| Heliotropium indicum | [130,131] | |
| Heliotropium supinum | [27,143] | |
| Heliotropium transalpinum | [144] | |
| Lappula spinocarpos | [40] | |
| Tournefortia samentosa | [28] | |
| Trichodesma ehrenbergii | [54] | |
| Trichodesma zeylanicum | [182] | |
| Supinidine | Heliotropium angiospermum | [106] |
| Heliotropium confertifolum | [106] | |
| Heliotropium curassavicum | [106,116] | |
| Heliotropium foliosissimum | [106] | |
| Heliotropium fruticosum | [106] | |
| Heliotropium gregii | [106] | |
| Heliotropium indicum | [106,116] | |
| Heliotropium molle | [106] | |
| Heliotropium procumbens | [106] | |
| Heliotropium queretaroanum | [106] | |
| Heliotropium racemosum | [106] | |
| Heliotropium spathulatum | [106,116] | |
| Heliotropium sessei | [106] | |
| Heliotropium wigginsii | [106] | |
| Supinidine N-oxide 2S-hydroxy-2S(1S-hydroxyethyl)-4-methyl-pentanoyl ester | Anchusa strigosa | [50,51] |
| Symlandine | Amsinckia menziesii var. inermedia | [42] |
| Echium sericeum | [54] | |
| Symphytum × uplandicum | [180] | |
| Symphytine | Myosotis scorpioides | [159] |
| Symphytum asperum | [171] | |
| Symphytum bohemium | [173] | |
| Symphytum consolidum | [175] | |
| Symphytum grandiflorum | [176] | |
| Symphytum ibericum | [176] | |
| Symphytum officinale | [171,177,178,179] | |
| Symphytum orientale | [28] | |
| Symphytum peregrinum | [27] | |
| Symphytum tanaiense | [173] | |
| Symphytum tuberosum | [28,176] | |
| Symphytum × uplandicum | [180] | |
| Symviridine | Symphytum asperum | [172] |
| Symphytum officinale | [172,177] | |
| Symphytum × uplandicum | [172] | |
| Tessellatine | Amsinckia douglasiana | [42] |
| Amsinckia eastwoodiae | [42] | |
| Amsinckia furacata | [42] | |
| Amsinckia grandiflora | [42] | |
| Amsinckia lunaris | [42] | |
| Amsinckia lycopsoides | [42,44] | |
| Amsinckia lycopsoides × menziesii var. intermedia | [42] | |
| Amsinckia menziesii var. intermedia | [42] | |
| Amsinckia retrosa | [42] | |
| Amsinckia spectabilis var. microcarpa | [42] | |
| Amsinckia spectabilis var. nicolai | [42] | |
| Amsinckia tessellate var. gloriosa | [42] | |
| Amsinckia tessellate var. tessellate | [42] | |
| Cryptantha confertiflora | [63] | |
| Cryptantha virginiensis | [63] | |
| Thesinine | Borago officinalis | [56] |
| Thesinine-4'-O-β-D-glucoside | Borago officinalis | [56] |
| 7-Tigloyl-9-(2-deoxy-2-methyl)echimidinylheliotridinEchium | Cynoglossum columnae | [69] |
| 7-Tigloyl-9-(2,3-dihydroxybutyryl)retronecine | Echium horridum | [90] |
| Echium rauwolfii | [90] | |
| Echium setosum | [96] | |
| Echium vulgare | [96] | |
| 7-Tigloyl-9-(2,3-dihydroxypropanoyl)retronecine | Alkanna orientalis | [40] |
| Alkanna tincotoria | [40] | |
| 7-Tigloylheliotridine | Cynoglossum officinale | [67] |
| 7-Tigloyllycopsamine | Echium horridum | [90] |
| Echium rauwolfii | [90] | |
| 7-Tigloyl-9-(2-methybutyryl)retronecine | Echium horridum | [90] |
| Echium rauwolfii | [90] | |
| Echium setosum | [96] | |
| Echium vulgare | [96] | |
| 7-Tigloylretronecine | Alkanna tinctoria | [40] |
| Arnebia decumbens | [40] | |
| Echium amoenum | [87] | |
| Echium setosum | [96] | |
| Echium vulgare | [96] | |
| 9-Tigloylretronecine | Alkanna orientalis | [40] |
| Alkanna tincotoria | [40] | |
| Arnebia decumbens | [52] | |
| Echium horridum | [90] | |
| Echium rauwolfii | [90] | |
| Echium setosum | [96] | |
| Echium vulgare | [96] | |
| Trachelanthamidine | Heliotropium angiospermum | [106] |
| Heliotropium confertifolum | [106] | |
| Heliotropium curassavicum | [106,116,118] | |
| Heliotropium foliosissimum | [106] | |
| Heliotropium fruticosum | [106] | |
| Heliotropium gregii | [106] | |
| Heliotropium indicum | [106,116] | |
| Heliotropium molle | [106] | |
| Heliotropium procumbens | [106] | |
| Heliotropium queretaroanum | [106] | |
| Heliotropium racemosum | [106] | |
| Heliotropium sessei | [106] | |
| Heliotropium spathulatum | [106,116] | |
| Heliotropium strigosa | [25] | |
| Heliotropium wigginsii | [106] | |
| Trachelanthamine | Anchusa hispida | [40] |
| Cynoglossum clandestinum | [60] | |
| Cynoglossum creticum | [70] | |
| Cynoglossum officinale | [67] | |
| Lappula spinocarpos | [40] | |
| Myosotis sylvatica | [25] | |
| Onosma stellulatum | [167] | |
| Rindera balaschuanica | [152] | |
| R. echinata | [120] | |
| Trachelanthus hissaricus | [29] | |
| Trachelanthus korolkovii | [57,150] | |
| Trachelanthine | Trachelanthus hissaricus | [29] |
| Trachelanthus korolkovii | [150] | |
| 7-Trachelanthyl-laburnine | Rindera umbellata | [169] |
| 7-Trachelanthylretronecine | Amsinckia vernicosa | [42] |
| Transalpinecine | Heliotropium transalpinum var. transalpinum | [145] |
| Triangularine | Alkanna orientalis | [40] |
| Alkanna tinctoria | [40] | |
| Triangularicine | Alkanna orientalis | [40] |
| Alkanna tinctoria | [40] | |
| Trichodesmine | Heliotropium arguzioides | [28] |
| Trichodesma africanum | [40,181] | |
| T. incanum | [46] | |
| Turkestanine | Rindera baldschuanica | [152] |
| Rindera oblongifolia | [152] | |
| Solenanthus turkestanicus | [135,150] | |
| Uplandicine | Echium rauwolfii | [90] |
| Echium setosum | [96] | |
| Echium vulgare | [96] | |
| Onosma arenaria | [163] | |
| Onosma stellulatum | [167] | |
| Symphytum × uplandicum | [180] | |
| Uluganine | Ulugbekia tshimganica | [183] |
| Viridantine | Cynoglossum furcatum | [76,187] |
| Onosma erecta | [69] | |
| Viridiflorine | Anchusa milleri | [40] |
| Cynoglossum germanicum | [77] | |
| Cynoglossum officinale | [40,84] | |
| Cynoglossum viridiforum | [97] | |
| Lappula spinocarpus | [40] | |
| Lindelofia olgae | [150] | |
| Lindelofia pterocarpa | [151] | |
| Lindelofia stylosa | [135] | |
| Lindelofia tschimganic | [152] | |
| Paracaryum intermedium | [40] | |
| Paracaryum regulosum | [40] | |
| Symphytum officinale | [179] | |
| Trachelanthus hissricus | [29] | |
| Trichodesma africanum | [40] | |
| 7-Viridiflorylretronecine | Echium tuberculatum | [60] |
| Eritrichium rupestre | [60] | |
| Nonnea lutea | [60] | |
| Nonnea setosa | [60] | |
| Onosma stellulatum | [60] | |
| Vulgarine | Echium vulgare | [11] |
2. Phytochemical Analysis of PAs
2.1. Mass Spectrometry
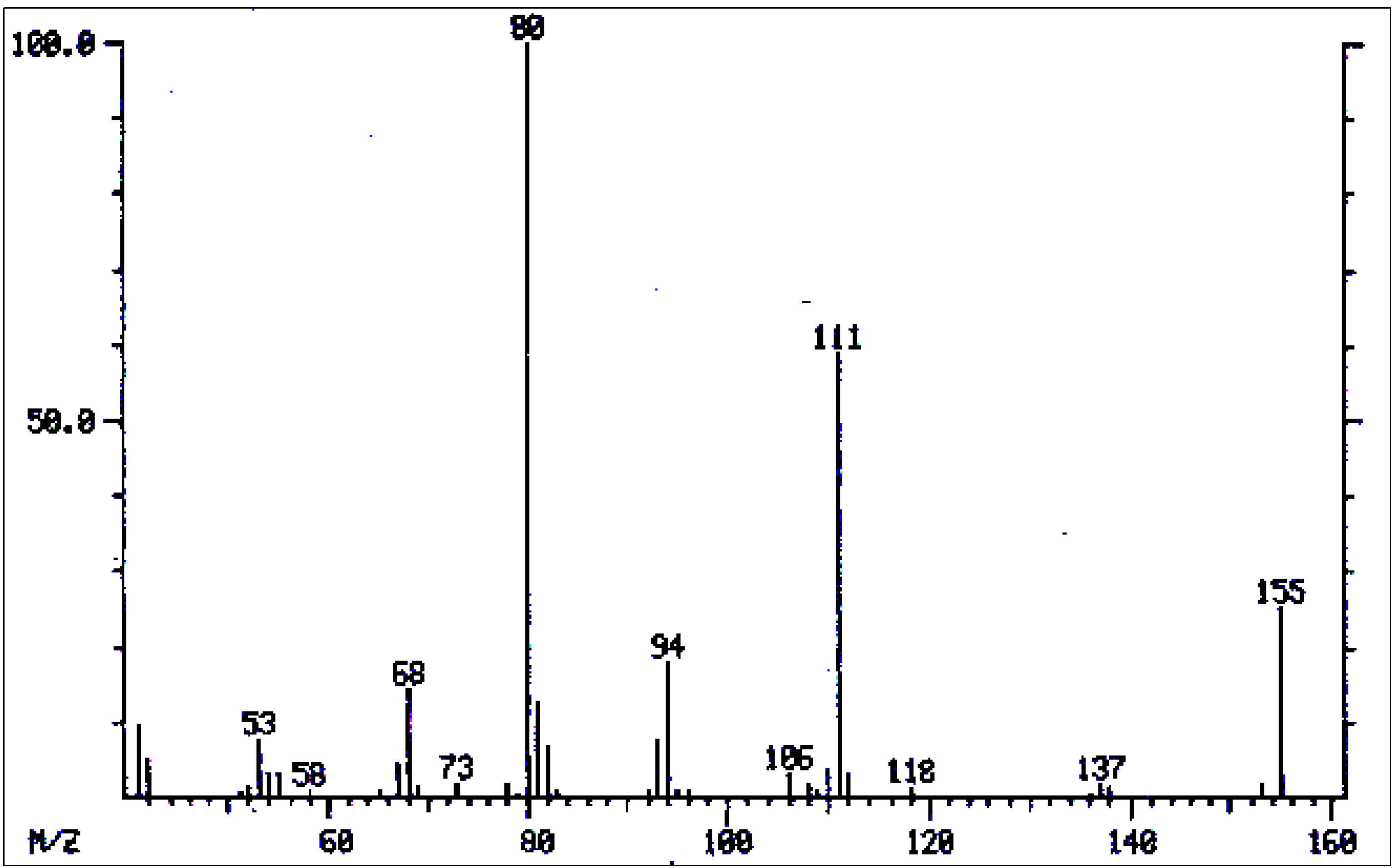
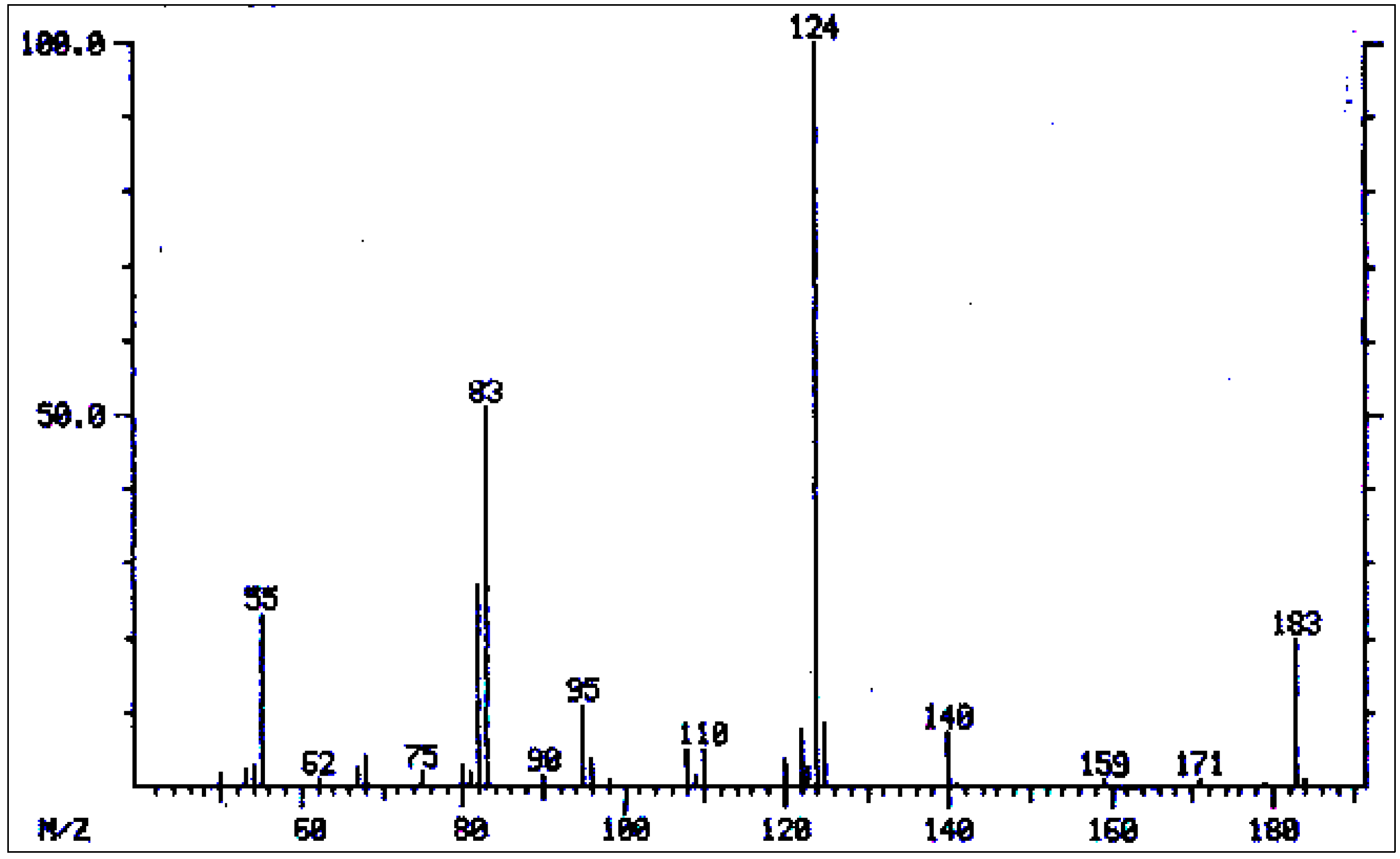
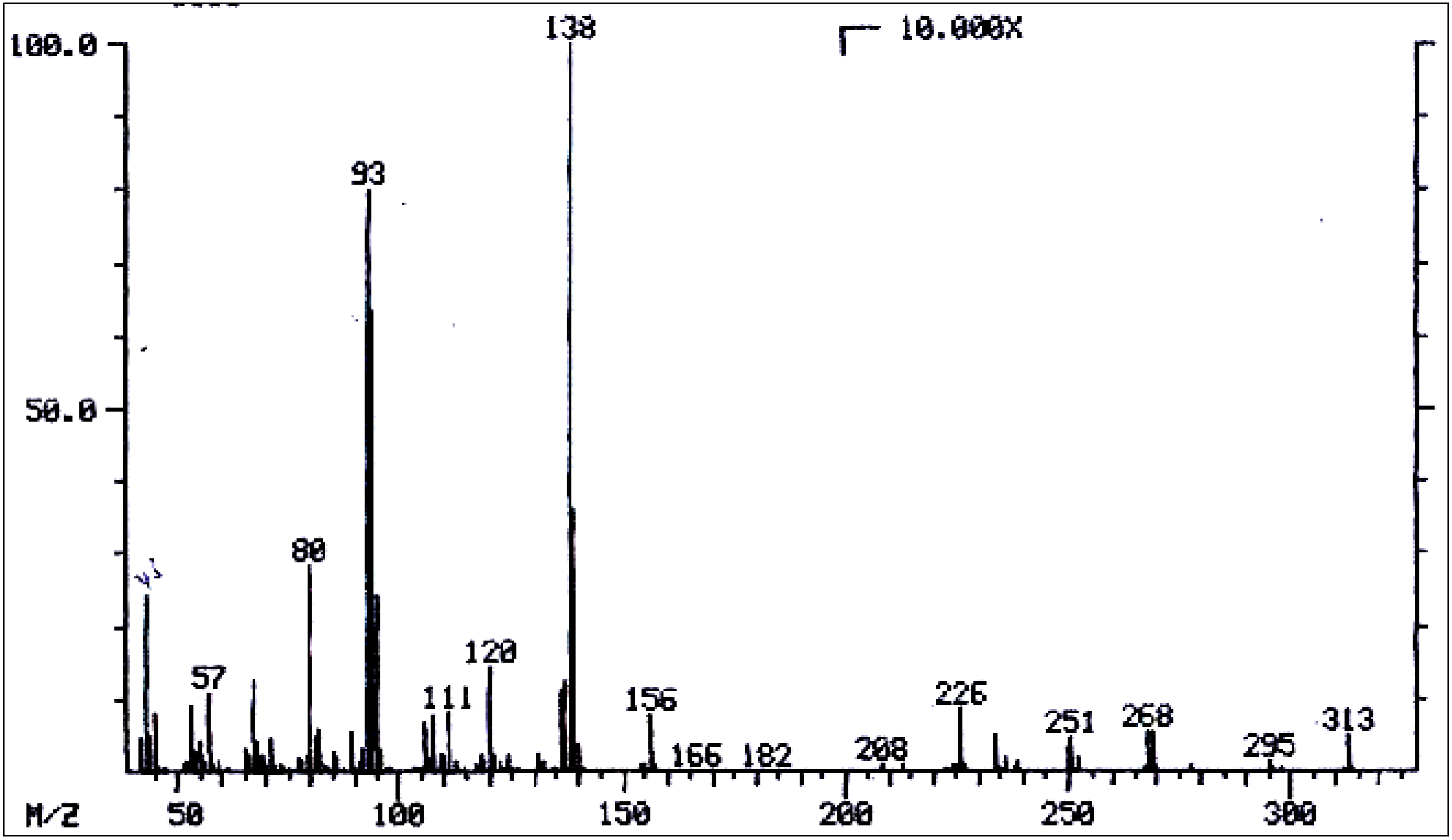
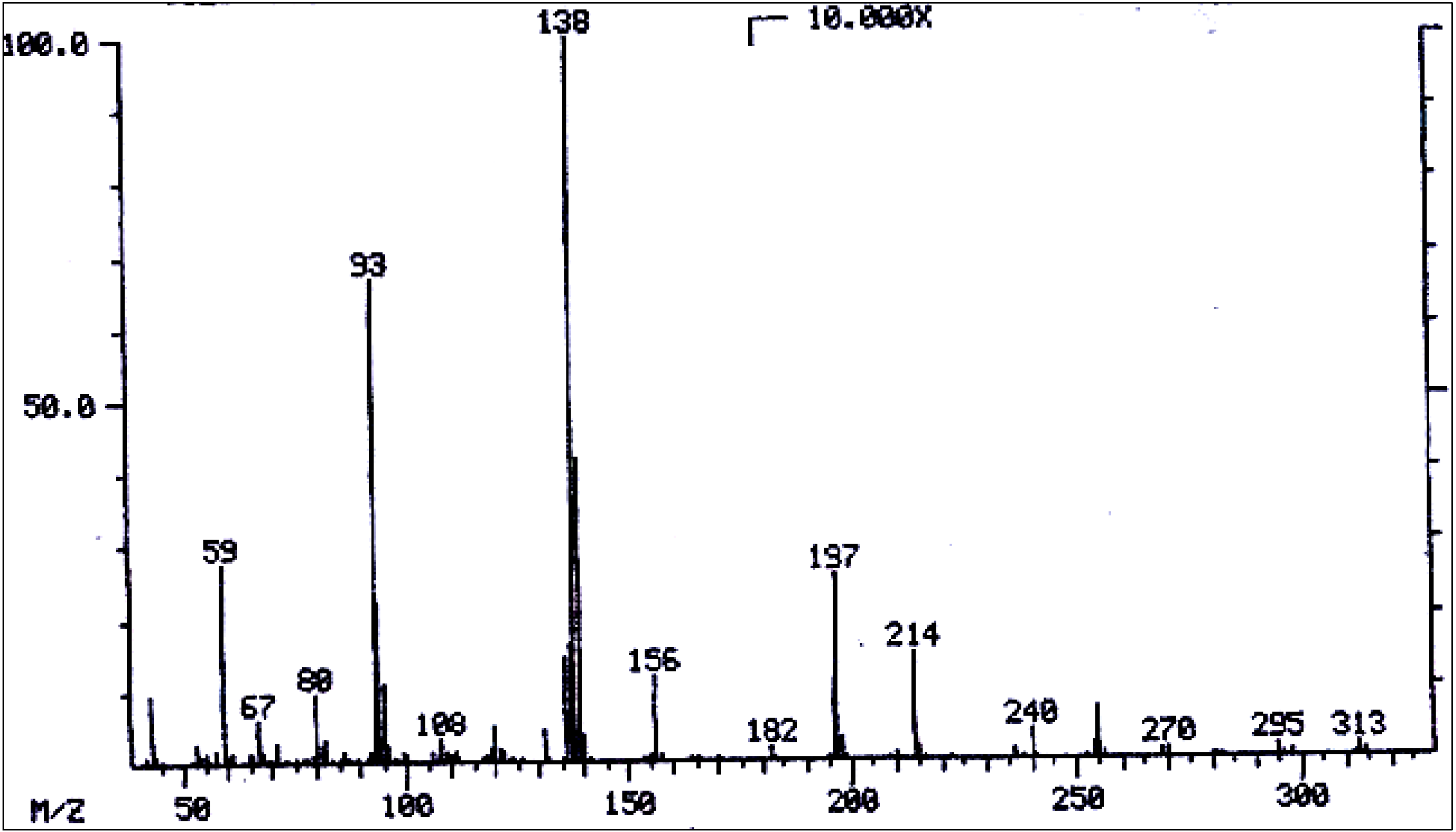

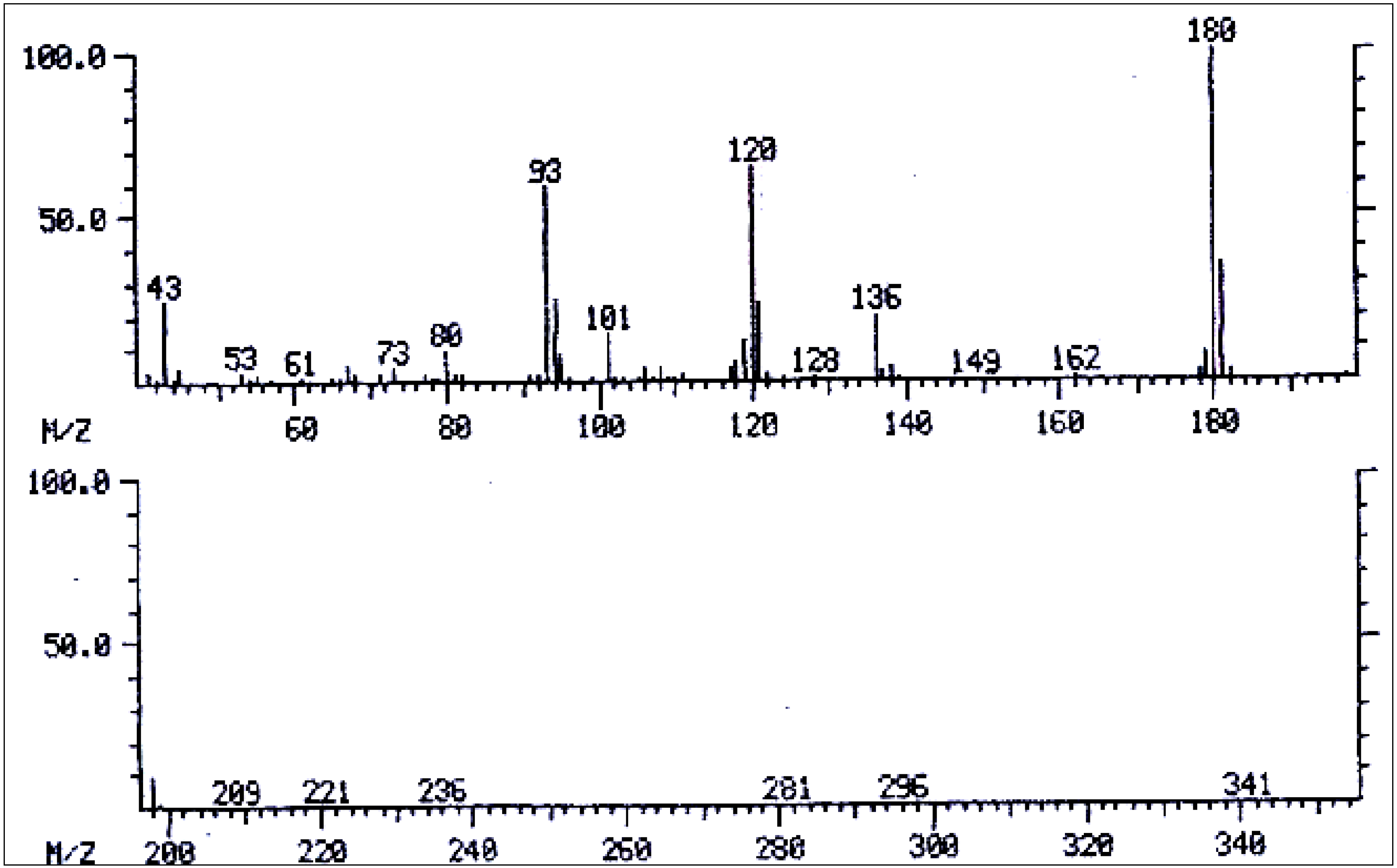
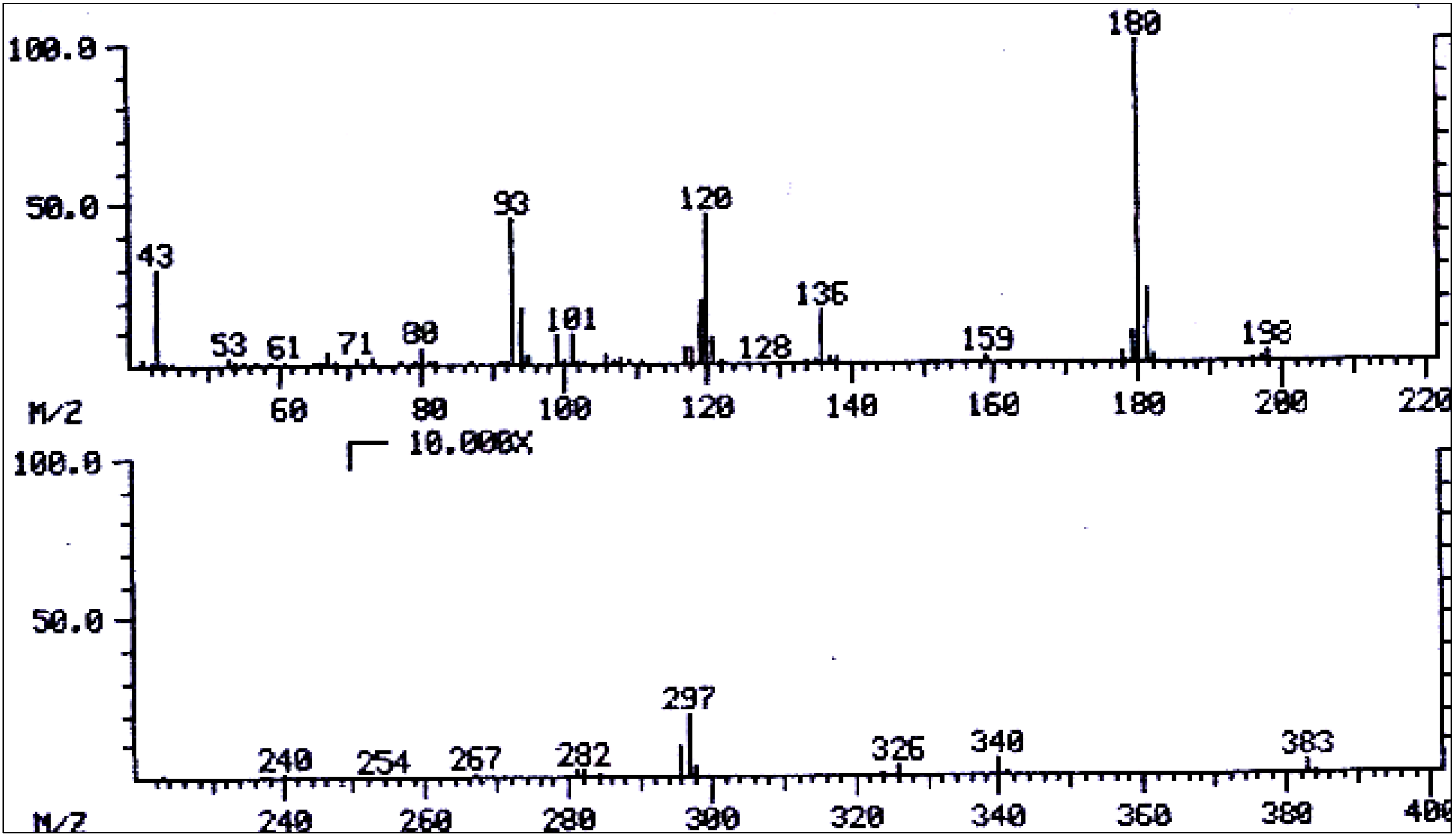
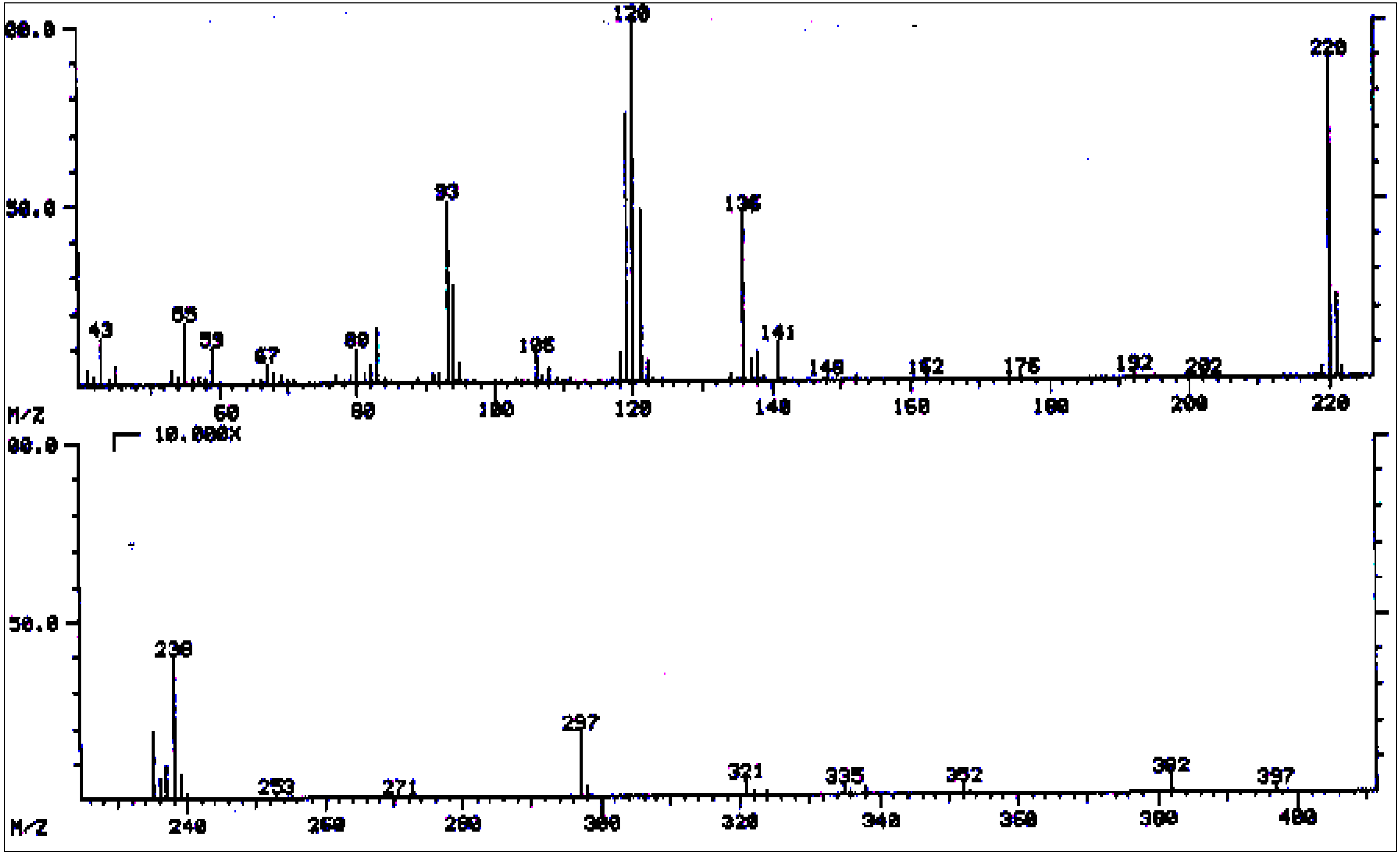
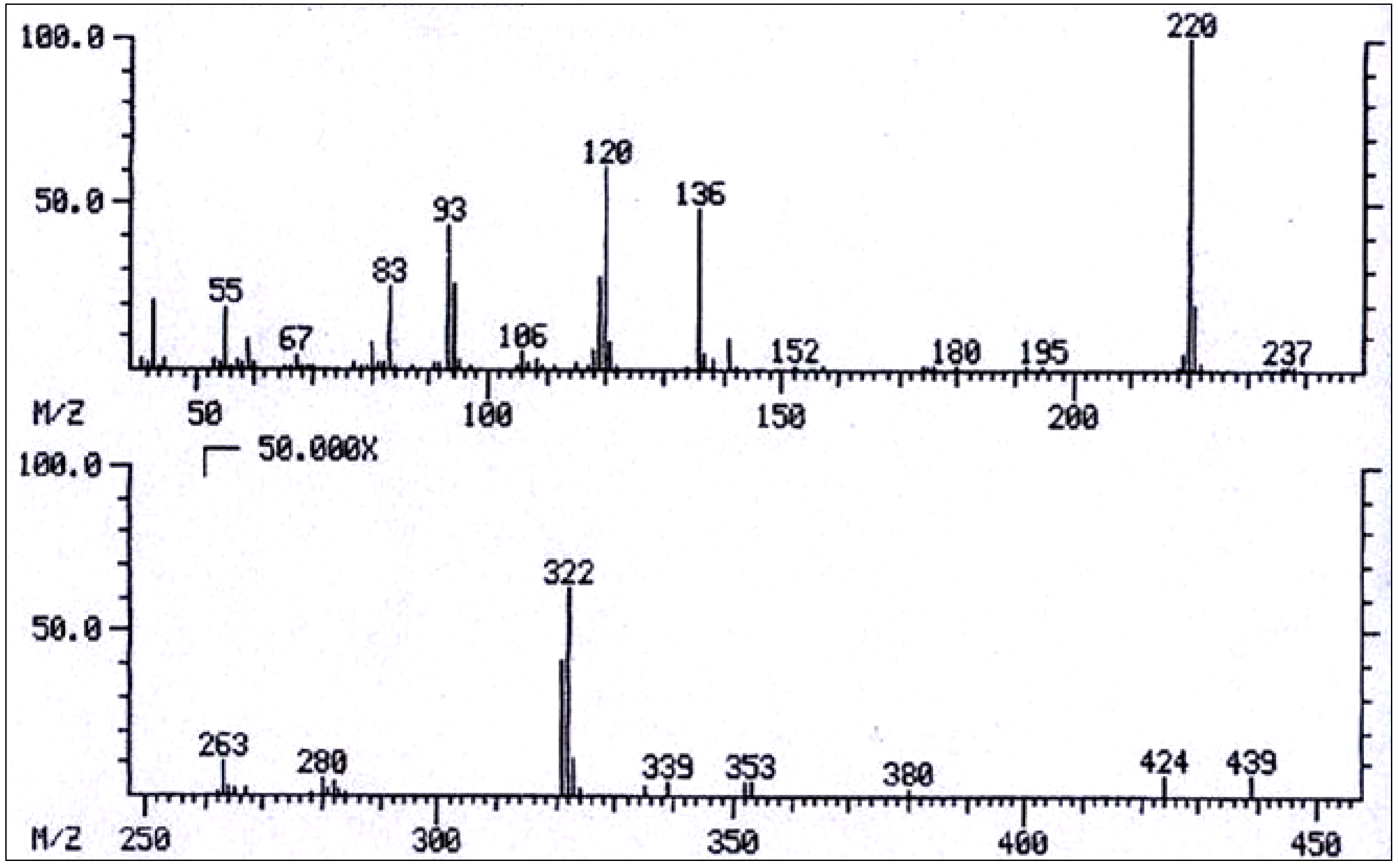
| No. | Alkaloid | RI | [M]+ | Characteristic ions m/z (relative abundance) | References |
|---|---|---|---|---|---|
| 1 | 3'-Acetylcanescine | - | 441 | 441(0.7), 426(2.6), 355(2.4), 255(6.3), 238(62.4), 220(18.3), 180(39), 136(47), 120(100), 93(74), 80(20). | [154] |
| 2 | 3'- Acetylcanescenine | - | 441 | 441(1.2), 426(2.4), 398(1.6), 355(1.2), 255(5.7), 238(58.2), 220(20.3), 180(44.3), 136(43.4), 120(100), 93(80.3), 80(22.1). | [154] |
| 3 | 7-Acetyl-9-curassavoylheliotridine | 2275 | 355 | 295(0.1), 268(0.2), 198(8), 181(36), 180(100), 136(22), 121(26), 120(84), 119(12), 95(15), 93(35), 80(17), 67(10), 57(12), 45(9), 43(48). | [40] |
| 4 | 7-Acetyl-9-(2,3-dihydroxybutryl) retronecine | 2092 | 299 | 239(3), 181(20), 180(100), 136(20), 120(59), 101(20), 94(32), 93(73), 80(12), 67(5), 55(5), 43(15). | [163] |
| 5 | 7-Acetyl-9-(2,3-dimethylbutryl) retronecine | 1947** | 295 | 235(8), 180(100), 136(20), 120(36), 101(30), 94(30), 93(65), 80(8), 43(42). | [163] |
| 6 | 3'-Acetylechinatine | 2220 | 341 | 326(0.1), 298(1), 255(2), 254(2), 181(2), 156(4), 139(21), 138(100), 137(10), 136(10), 120(6), 99(6), 94(20), 93(71), 80(8), 67(5), 43(22). | [84] |
| 7 | 7-Acetylechinatine | 2235 | 341 | 341(0.1), 281(2), 198(6), 181(39), 180(100), 136(18), 121(35), 120(70), 119(28), 101(9), 94(18), 93(55), 80(7), 43(33). | [67] |
| 8 | 3'-Acetylechiumine | 2245* | 423 | 423(2), 380(3), 338(3), 337(6), 336(3), 323(2), 280(3), 263(3), 256(2), 238(5), 237(6), 221(34), 220(100), 219(15), 159(5), 141(19), 138(12), 136(54), 121(20), 120(76), 119(34), 106(12), 94(42), 93(56), 83(27), 80(15), 59(3), 55(34), 53(17). | [42] |
| 9 | 3'-Acetylechimidine | 2640 | 439 | 439(0.1), 424(0.1), 322(1), 238(2), 221(25), 220(100), 219(3), 138(5), 137(6), 136(49), 121(8), 120(61), 119(28), 106(6(, 94(24), 93(46), 83(14), 59(11), 55(13). | [91] |
| 10 | 5'-Acetyleuropine | - | 371 | 156(18), 138(100), 94(36), 93(75), 59(74). | [138] |
| 11 | 7-Acetyleuropine | - | 371 | 371(2.7), 356(1), 311(3), 282(5), 180(100), 120(72), 93(65), 80(17), 59(95). | [110] |
| 12 | 3'-Acetylfurcatine | 3188* | 425 | 223(28), 222(100), 143(10), 136(36), 121(15), 120(82), 94(27), 93(61), 85(21), 80(7), 57(63). | [42] |
| 13 | 3'-Acetylheliosupine | 2640 | 439 | 424 (0.1), 321(1), 221 (28), 220(100), 141 (10), 138(4), 137(5), 136 (45), 121(14), 120 (89), 119(82), 106(7), 94 (21), 93(40), 83(11), 80(5), 59(8), 55(11), 43(22). | [67] |
| 14 | 7-Acetyl-9-(2-hydroxy-3-methylbutryl) retronecine | 2024** | 297 | 237(5), 180(100), 136(25), 120(52), 119(20), 101(25), 94(45), 93(95), 80(15), 43(35). | [163] |
| 15 | 3'-Acetylindicine | 2195 | 341 | 341(2), 255(2), 181(5), 138(100), 93(48), 43(10). | [158] |
| 16 | 3'-Acetylintermedine | 2255 | 341 | 341(5), 298 (4), 255 (16), 139(20),138(100), 137 (12),136 (12), 94 (30), 93 (71), 80 (10), 43 (21). | [40] |
| 17 | 7-Acetylintermedine | 2220 | 341 | 296(4), 281(3), 181(40), 180(100), 136(25), 121(30), 120(78), 119(15), 101(19), 95(10), 94(30), 93(73), 80(11), 43(40). | [40] |
| 18 | 5'-Acetyllasiocarpine | - | 453 | 363, 335, 321, 263, 220(100), 141, 136, 121, 120, 119, 106, 94, 93, 83, 80. | [186] |
| 19 | 3'-Acetyllithosenine | - | 457 | 457(0.78), 442(2), 339(1), 255(2), 238(100), 237(5), 222(10), 220(25), 138(30), 137(8), 136(49), 121(36), 120(75), 119(36), 95(9), 94(34), 93(57), 80(12). | [156] |
| 20 | 3'-Acetyllycopsamine | 2255 | 341 | 341(5), 298(4), 255(16), 139(20), 138(100), 137(12), 136(12), 94 (30), 93(71), 80 (10), 43(21). | [40] |
| 21 | 7-Acetyllycopsamine | 2230 | 341 | 341(1), 296(4), 281(3), 181(32), 180(100), 136(20), 121(35), 120(70), 119(11), 101 (19), 95(10), 94 (28), 93(61), 80(10), 43(36). | [91] |
| 22 | 7-Acetyl-9-(2-methylbutyryl) retronecine | 1914** | 281 | 221(12), 195(5), 180(100), 136(25), 120(42), 119(15), 101(35), 94(45), 93(80), 80(13), 67(100), 57(18), 53(5), 43(23). | [163] |
| 23 | 3'-Acetylmyscorpine | 3290* | 423 | 221(30), 220(100), 136(47), 120(67), 119(35), 94(39), 93(51), 83(32), 80(19), 55(39). | [42] |
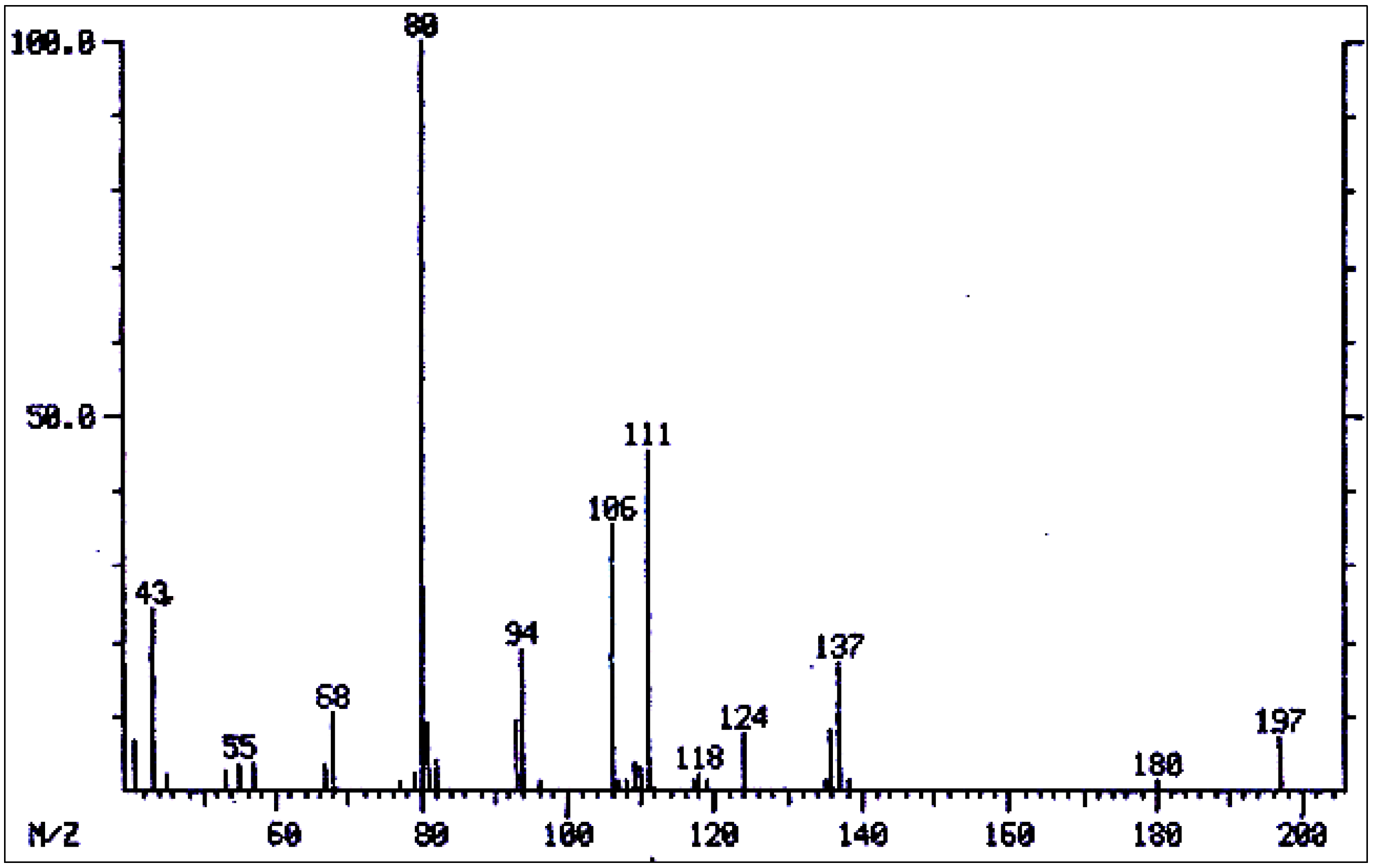
| 24 | 7-Acetylretronecine | 1532** | 197 | 180(3), 137(12), 111(40), 106(30), 94(20), 80(100), 68(3), 43(30). | [163] |
| 25 | 3'-Acetylrinderine | 2222 | 341 | 326(0.1), 298(1), 255(1), 254(0.5), 181(5), 156(5), 139(30), 138(100), 137(17), 136(17), 120(10), 99(10), 95(9), 94(35), 93(95), 80(13), 67(9), 43(36). | [70] |
| 26 | 7-Acetyl-9-(sarracinoyl) retronecine | 2125 | 295 | 235(1), 197(32), 196(10), 181(8), 180(55), 179(15), 136(34), 121(12), 120(42), 119 (20), 101(19), 94(40), 93100), 80(15), 67(6), 53(9), 43(20). | [40] |
| 27 | 7-Acetylscorpioidine | - | 423 | 423(0.8), 181(23), 180(92), 179(17), 136(23), 121(10), 120(57), 119(45), 95(6), 94(35), 93(57), 83(100), 80(15), 55(39), 43(30). | [159] |
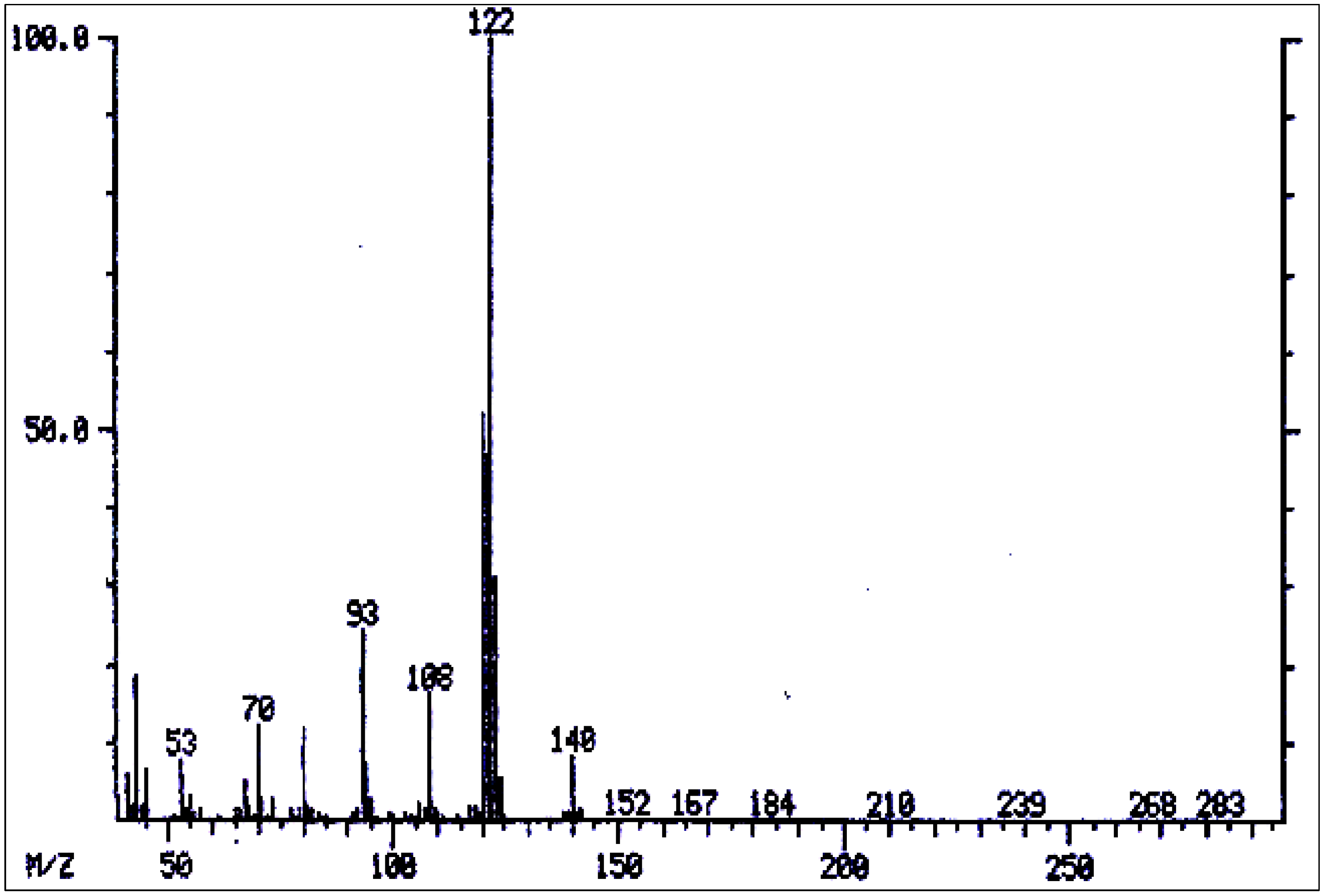
| 28 | 3'-Acetylsupinine | 2080** | 325 | 284(.02), 299(10), 136(10), 122(100), 120(48), 108(13), 101(13), 93(50), 80(13), 70(9), 53(6), 43(15). | [163] |
| 29 | 3'-Acetyltessellatine | 3064* | 341 | 341(41), 324(10), 323(10), 299(2), 280(8), 255(2), 254(4), 248(2), 238(2), 237(5), 236(17), 198(8), 181(4), 180(2), 156(7), 154(12), 138(22), 137(28), 136(27), 124(23), 120(63), 111(66), 108(30), 106(61), 99(17), 94(35), 93(14), 80(100), 55(10), 53(10). | [42] |
| 30 | 9-Acetytessellatine | 2962* | 341 | 341(7), 299(6), 282(11), 281(15), 248(3), 238(15), 237(16), 236(14), 198(18), 181(12), 180(10), 179(9), 153(13), 138(49), 136(45), 121(26), 120(100), 119(32), 108(11), 106(12), 94(52), 93(89), 80(28), 53(11). | [42] |
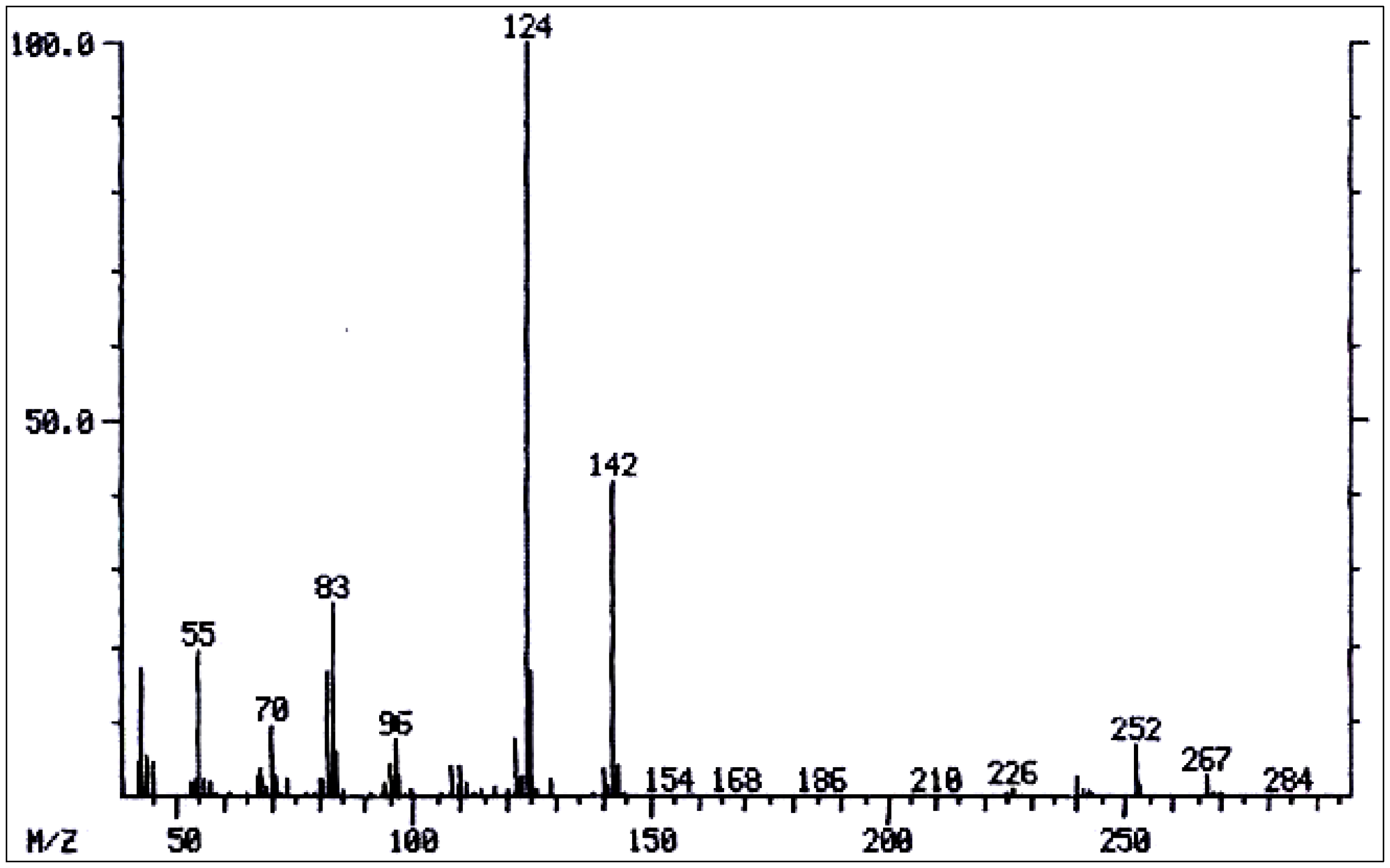
| 31 | 3'-Acetyltrachelanthamine | - | 327 | 327(1), 284(2), 240(6), 184(6), 142(27), 125(38), 124(100), 84(14), 83(59), 82(54). | [128] |
| 32 | 9-Acetyltrachelanthamidine | 1395 | 183 | 140(8), 125(8), 124(100), 110(5), 95(10), 83(50), 82 (27), 55(23). | [40] |
| 33 | 3'-Acetylviridiflorine | 2767* | 327 | 327(3), 284(6), 268(2), 267(2), 241(5), 240(9), 225(4), 184(4), 159(2), 142(21), 140(4), 125(17), 124(100), 83(14), 82(12), 55(19). | [42] |
| 34 | 9-(3'-Acetylviridifloryl) retronecine | - | 341 | 341(4.7), 255(3), 182(5), 157(4), 138(100), 120(10), 93(92), 85(4), 83(20), 80(16), 43(38). | [119] |
| 35 | 9-(3'-Acetyl)viridifloryl turneforcidine (or isomer) | 3050* | 343 | 343(5), 325(4), 300(12), 299(5), 257(14), 256(50), 212(13), 200(15), 197(14), 159(10), 158(36), 141(19), 140(30), 138(16), 122(21), 120(33), 117(10), 106(12), 96(44), 95(90), 83(11), 82(100), 69(17), 55(32). | [42] |
| 36 | 7- Acetylvulgarine | - | 439 | 440(M++ 1), 422(40), 380(15), 358(5), 340(65), 296(42), 282(85), 180(100). | [11] |
| 37 | Amabiline | 1985, 2652* | 283 | 383(1), 140(8), 123 (30), 122(100), 121(46), 120(51), 108(17), 93(25), 80(13), 70 (17), 53(7), 45(6), 43(19). | [67] |
| 38 | Anadoline | - | 381 | 381(6), 204(6), 167(6), 149(23), 139(22), 138(93), 137(45), 136(33), 120(17), 119(17), 118(13), 117(18), 111(12), 109(15), 100915), 97(17), 95(27), 94(55), 93(88), 85(25), 83(100), 82(17), 81(27), 80(27), 73(14), 71(37), 70(17), 67(27), 57(73), 56(17), 55(83), 54(13), 53(20). | [202] |
| 39 | 7α-Angeloyl-1-chloromethy-1,2-dihydropyrrolizidine | 1815 | 255 | 220(40), 172(15), 155(45), 136(23), 130(24), 129(32), 128(63), 121(11), 120(94), 119 (24), 106(30), 94 (100), 93(20), 83(20), 80(17), 67(8), 55(35). | [70] |
| 40 | 7-Angeloyl-9-(2,3-dihydroxybutyryl)heliotridine | 2333 | 339 | 339(1), 324(1), 294(1), 239(6), 222(25), 221(25), 220(65), 219(8), 138 (20), 137(10), 136(81), 121(24), 120(100), 119(85), 106(15), 94(50), 93(85), 83(24), 80(18), 75(2), 57(10), 55(25), 45(10). | [67] |
| 41 | 7-Angeloyl-9-(2,3-dihydroxybutyryl)retronecine | 2315 | 339 | 339(1), 239(5), 238(5), 237(5), 221(25), 220(99), 219(15), 141(20), 138(10), 137(11), 136 (100), 121(15), 120(83), 119(34), 106(10), 94(55), 93(95), 83(41), 80(20), 75(2), 57(10), 55(40), 45(10). | [96] |
| 42 | 7-Angeloyl-9-(2,3-dihydroxypropanoyl)retronecine | 2300 | 325 | 294(0.5), 237(5), 255(9), 221(10), 220(67), 219(16), 141(22), 138(5), 137(11), 136(100), 121(11), 120(57), 119(40), 106(10), 94(62), 93(95), 83(37), 80(17), 55(32). | [40] |
| 43 | 7-Angeloyl-1-formyl-6,7-dihydro-5H-pyrrolizidine | 1920 | 233 | 215(3), 150(100), 134(92), 133(35), 122(4), 106(15), 105(38), 104(15), 83(10), 79(16), 55(20). 46 | [67] |
| 44 | 7-Angeloyl-9-(hydroxypropenoyl)retronecine | 2053 | 307 | 221(13), 220(95), 207(24), 181(8), 141(30), 137(10), 136(100), 120(53), 119 (24), 106(10), 94(63), 93(77), 83(43), 80(21), 67(9), 55(41), 43 (26). | [40] |
| 45 | 7-Angeloylechinatine | 2467 | 381 | 336(2), 281(1), 238(11), 221(42), 220(86), 141(11), 138(11), 137(9), 136(55), 121(78), 120(100), 119(60), 117(4), 106(9), 99(3), 94 (30), 93(52), 83(17), 80(10), 55(17), 45(8), 43(21). | [67] |
| 46 | 7-Angeloylheliotridine | 1820 | 237 | 219(1), 154(2), 137(42), 136(20), 124(25), 111(35), 106(86), 94(25), 83(10), 80(100), 68(10), 55(20). | [70] |
| 47 | 7-Angeloylheliotine | - | 395 | 395(5), 295(7), 220(100), 136(57), 120(80), 119(70), 93(43), 83(34), 59(55), 43(18). | [127] |
| 48 | 7-Angeloyllycopsamine | 2460* | 381 | 381(0.1), 336(0.9), 281(0.5), 238(0.5), 220(100), 136(65), 121(40), 120(90), 94(40), 93(85), 83(50), 80(15), 55(48). | [90] |
| 49 | 7-Angeloyl-9-(2-methylbutyryl)heliotridine | 2180 | 321 | 321(0.5), 221(43), 220 (70), 195(5), 141(26), 138(4), 137(9), 136(85), 121(13), 120(100), 119(65), 106(13), 94(59), 93(71), 83 (23), 80(15), 67(4), 57(27), 55(30). | [67] |
| 50 | 7-Angeloyl-9-(2-methylbutyryl)retronecine | 2155 | 321 | 221(35), 220(100), 195(5), 141 (25), 138(3), 137(9), 136(90), 121(6), 120(53), 119(20), 106(8), 94(50), 93(70), 83(35), 80(15), 57(20), 55(35). | [96] |
| 51 | 7-Angeloylretronecine | 1787 | 237 | 237(2), 219 (3), 204(0.5), 191(1), 154(2), 138(5), 137(23), 136(18), 124(23), 111(38), 106(40), 94(20), 93(6), 83(11), 80(100), 55(22). | [96] |
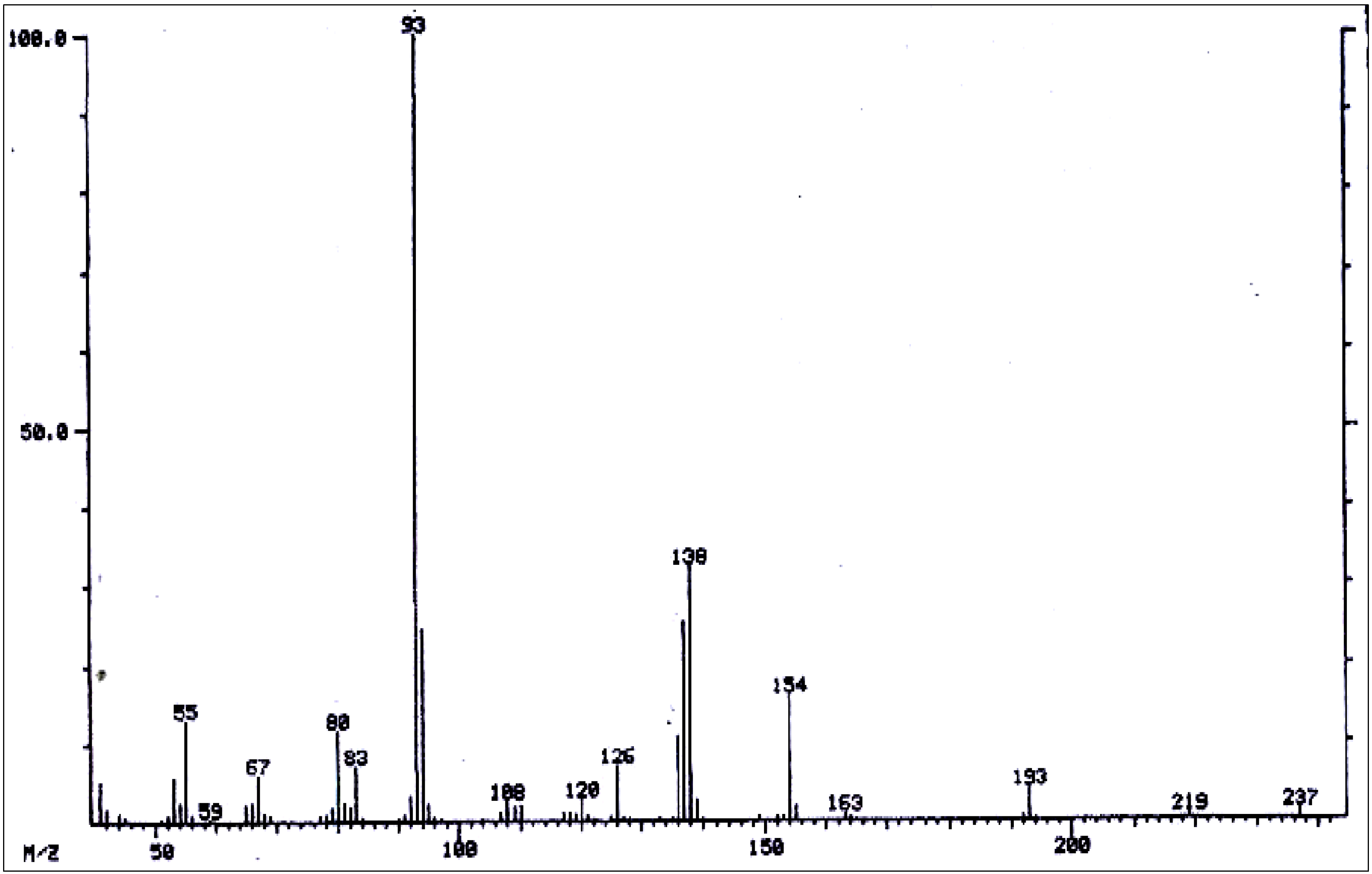
| 52 | 9-Angeloylretronecine | 1797 | 237 | 237(1), 219(0.5), 193(3), 154(16), 138(32), 137(25), 136(10), 126(7), 120(2), 108(2), 94(25), 93(100), 83(8), 80(10), 55(13). | [96] |
| 53 | 7-Angeloylrinderine | 2465 | 381 | 381(0.1), 336(2), 281(1), 238(10), 221(34), 220(80), 141(11), 138(10), 137(9), 136(53), 121(70), 120(100), 119(59), 117(4), 106(10), 94(28), 93(50), 83(15), 80(10), 55(17), 45(8), 43(25). | [67] |
| 54 | 9-Angeloyltrachelamthamidine | 1700 | 223 | 140(1), 125 (30), 124(100), 123(20), 122(15), 110(4), 95(13), 83(45), 82(16), 70(6), 55(27). | [40] |
| 55 | 9-Angeloyl-7-viridiflorylretronecine | - | 381 | 220(8.3), 138(7.3), 137(9.8), 136(9.9), 120(28.6), 117(50), 106(29), 94(34), 93(23.1), 91(34.6), 79(40.8), 67(39.6), 64(81.8), 60(31), 58(57), 57(85), 55(100). | [60] |
| 56 | Asperumine | - | 397 | 380, 336, 220, 138, 120. | [203] |
| 57 | 9-(Butyryl-2-ene) supinidine | 1674** | 207 | 180(5), 159(7), 157(7), 122(100), 121(6), 120(23), 108(6), 93(15), 80(17), 71(25), 57(7), 53(11), 45(11), 43(14). | [163] |
| 58 | Canescine | - | 399 | 399(0.4), 384(4), 355(1), 338(0.3), 256(10), 238(66), 220(21), 180(11.6), 136(42), 120(100), 93(65), 80(20). | [154] |
| 59 | Canescenine | - | 399 | 399(0.1), 384(1.6), 355(0.4), 338(0.3), 256(9.6), 238(67.5), 220(20.6), 180(14.7), 136(45.1), 120(100), 93(61.6), 80(20.2). | [154] |
| 60 | Coromandaline | - | 285 | 285(2), 267(7), 241(3), 240(9), 142(65), 125(23), 124(100), 83(28), 82(18). | [140] |
| 61 | Coromandalinine | - | 283 | 283(0.8), 239(0.4), 238(0.3), 140(7), 123(27), 122(100), 121(35), 120(40), 108(13), 94(8), 93(20.5), | [140] |
| 62 | Cryptanthine | 353 | 354[M++1](1), 272(2), 254(3), 238(2), 138(2), 120(100), 118(2). | [65] | |
| 63 | Curassanecine | - | 157 | 126(14), 98(10), 83(100) 82(23). | [117] |
| 64 | Curassavine | - | 299 | 299(0.7), 281(1.4), 255(2.6), 254(5), 142(80), 125(27), 124(100). | [140] |
| 65 | Curassavinine | - | 297 | 297(0.3), 253(0.1), 252(0.1), 241(0.1), 239(0.1), 140(8), 123(31), 122(100), 121(44), 120(41), 108(13), 94(6.4), 93(20). | [140] |
| 66 | 9-Curassavoylheliotridine | 2190 | 313 | 295(2), 269(0.5), 268(0.5), 251(0.5), 226(0.9), 156(8), 139(35), 138(100), 120(15), 111(9), 106(8), 95(25), 94(64), 93(80), 80(28), 67(13), 57(11), 45(8), 43(25). | [40] |
| 67 | Cynaustine | - | 283 | C15H25NO4, 122(100). | [68] |
| 68 | Cynaustraline (or stereoisomer) | 2682* | 285 | 285(2), 267(4), 252(5), 242(1), 241(1), 240(3), 226(1), 142(34), 125(15), 124(100), 83(24), 82(14), 55(25). | [42] |
| 69 | Cynoglossamine | - | 445 | 147(100), 138(27), 119(17), 93(23). | [138] |
| 70 | 5-Deoxylasiocarpine | - | 395 | 396[M + H] + (2), 295(5.5), 363(1), 220(80), 120(83), 93(68), 83(32). | [123] |
| 71/72 | 3',7-Diacetylintermedine/3',7-Diacetyllycopsamine | 2340 | 383 | 340(5), 297(10), 296(10), 181(22), 180(100), 136(18), 121(8), 120(47), 119(20), 101(10), 99(10), 94(18), 93(47), 80(5), 43(30). | [40] |
| 73 | 3',9-Diacetyltessellatine | 3090* | 383 | 383(2), 342, 341(37)(7), 324(21), 323(10), 297(8), 296(6), 280(10), 238(2), 237(6), 236(18), 198(10), 181(7), 180(4), 179(8), 154(14), 153(22), 138(20), 136(49), 121(18), 120(100), 119(55), 108(12), 106(19), 99(13), 94(37), 93(82), 80(25), 53(11). | [42] |
| 74 | 5,6-Dihydro-7,9-dimethoxy-7H-pyrrolizine | 1415** | 181 | 150(90), 134(5), 120(100), 119(35), 106(12), 91(3), 79(5). | [163] |
| 75 | Dihydroechinatine | - | 301 | MS2, 284, 258, 240, 140, 122, 96. | [167] |
| 76 | thero-2'',3''-Dihydroxyechiumine | - | 415 | 415(0.5), 371(2), 254(56), 210(26), 166(5), 138(30), 136(37), 120(100), 93(70). | [63] |
| 77 | Dihydroxytriangularine | 2525 | 369 | 338(1), 324 (2), 269(30), 252(30), 237(40), 221(25), 220(100), 219(10), 141(20), 138(8), 137(10), 136(80), 121(20), 120(80), 119(30), 106 (8), 95 (9), 94 (45), 93(81), 83(39), 80(10), 55(28). | [40] |
| 78 | Dihydroxytriangularicine | 2525 | 369 | 338(0.1), 324 (0.1), 269(4), 252(4), 237(5), 221(24), 220(100), 219(3), 141(28), 138(6), 137(8), 136(67), 121(24), 120(75), 119(35), 106(8), 95(8), 94(47), 93(90), 83(41), 80(11), 55(24). | [40] |
| 79 | Echihumiline | 2578 | 397 | 397(0.1), 382 (0.2), 352 (0.1), 338(0.1), 321 (0.1), 297 (2), 238 (2), 221 (20), 220 (100), 219 (4), 138 (6), 137 (6), 136 (40), 121 (15), 120 (39), 119 (16), 106 (4), 94 (20), 93 (30), 83 (36), 80 (6), 59 (5), 55 (6). | [91] |
| 80 | Echimidine | 2560 | 397 | 397(0.1), 382 (0.1), 352 (0.1), 297(2), 221(21), 220(100), 219(5), 138(5), 137(6), 136(48), 121(26), 120(75), 119(30), 106(5), 94(30), 93 (61), 83(39), 80(10), 59(10), 55(25), 43(18). | [96] |
| 81 | Echimidine isomer (tigloyl) | 2580 | 397 | 397(0.1), 382 (0.1), 297(2), 238(3), 221(21), 220(100), 219(3), 138(5), 137(6), 136(48), 121(28), 120(62), 119(23), 106(5), 94(28), 93 (46), 83(30), 80(10), 59(10), 55(20), 43(18). | [96] |
| 82 | Echinatine | 2175 | 299 | 284(0.1), 254(1), 156(10), 139(30), 138 (100), 137(7), 136(4), 95(9), 94(25), 93 (46), 80(7), 67(5), 53(3), 43(12). | [70] |
| 83 | Echiumine | 3178* | 381 | 381(0.4), 338(1), 337(1), 336(1), 281(2), 255(1), 238(9), 237(2), 221(35), 220(100), 141(16), 138(8), 136(47), 121(32), 120(69), 119(20), 106(7), 94(39), 93(50), 83(28), 80(17), 59(1), 55(37), 53(9). | [42] |
| 84 | Echiuplatine | 381 | 383[M++1](3), 382(68), 365(1), 364(1), 322(2), 320(24), 300(5), 282(7), 280(1), 238(4), 220(51), 138(2), 120(100), 118(2). | [65] | |
| 85 | Echiupinine | - | 381 | 381(0.3), 337(1), 336(1), 282(0.2), 281(2), 212(38), 220(100), 136(43), 121(33), 120(55), 119(19), 118(18), 117(14), 103(16), 94(24), 93(47), | [95] |
| 86 | Echivulgarine | - | 479 | 480(M++ 1), 462(35), 418(10), 398(10), 380(90), 336(40), 322(100), 220(70). | [11] |
| 87 | Ehretinine | - | 275 | 275, 140(100), 123, 97. | [100] |
| 88 | 2'',3''-Epoxyechiumine | - | 397 | 254(9), 237(33), 236(100), 164(17), 157(4), 138(4), 136(19), 121(24), 120(25), 94(27), 93(44), 80(12), 71(4), 43(30). | [63] |
| 89 | Erythro-2'',3''-chloro-2''-hydroxyechiumine | - | 435 | 435(0.5), 274(37), 273(32), 272(92), 254(10), 236(13), 208(9), 138(10), 136(25), 121(26), 120(100), 94(32), 93(45), 80919), 71(19). | [63] |
| 90 | 1α-2α-Epoxy-1β-hydroxymethyl-8α-pyrrolizidine | - | 155 | 126(4), 124(13), 96(10), 80(4), 71(50, 70(100), 68(10), 67(5), 56(6), 55(65). | [106] |
| 91 | Europine | 2217 | 329 | 314(1), 256(5), 240(25), 239(20), 156(10), 139(20), 138(100), 120(5), 94(20), 93(50), 80(10), 67(4), 59(20), 43(5). | [52] |
| 92 | Floridanine | - | 441 | 426, 397, 382, 168, 152, 151(100), 150, 149, 123, 122, 110, 96, 94. | [62,204] |
| 93 | Floridimine | - | 301 | 301(1), 239(8), 226(7), 142(36), 124(100), 95(12), 94(10), 83(55), 55(23). | [128] |
| 94 | Floridine | - | 343 | 343(1), 328(2), 284(1), 240(2), 239(3), 225(10), 142(8), 124(100), 83(27). | [128] |
| 95 | Floridinine | - | 301 | 301(1), 242(5), 239(7), 226(5), 167(1), 142(46), 125(19), 124(100), 122(8), 110(6), 96(8), 95(7), 83(36), 82(24), 70(12), 59(12). | [128] |
| 96 | Furcatine | 3138* | 383 | 383(0.2), 339(1), 281(1), 240(6), 223(35), 222(100), 143(18), 136(33), 121(27), 120(75), 119(21), 94(44), 93(65), 85(13), 80(19), 74(8), 73(5), 60(1), 57(46). | [42] |
| 97 | Heleurine | 1970 | 297 | 239(1), 198(3), 181(6), 140(15), 131(6), 123(30), 122(100), 120(60), 119(70), 108(16), 93(17), 80(10), 70(11), 59(26). | [109] |
| 98 | Helibracteatine | - | 255 | 255(0.5), 238(6), 237(1), 224(9), 156(43.4), 155(45.5), 138(16.3), 122(2.3), 111(99.7), 99(31), 98(100), 82(35). | [112] |
| 99 | Helibractinecine | - | 173 | 173(22), 156(4), 155(11), 142(7), 129(10), 124(1), 112(5), 99(83), 98(83), 95(6), 83(7), 82(100). | [111] |
| 100 | Helibracteatinecine | - | 173 | 173(20), 156(4.5), 155(14), 142(3), 129(12), 124(1), 112(5), 99(85), 98(100), 82(83.5). | [112] |
| 101 | Helibracteatinine | - | 255 | 255(2), 238(3), 237(3.5), 224(9), 198(7), 181(60), 156(16), 155(93), 154(10), 124(12), 122(21), 100(23), 98(20), 83(23), 82(100). | [112] |
| 102 | Helifoline | - | 255 | 237(3), 156(5), 138(11), 137(3), 112(27), 111(100), 99(9), 98(42), 94(41), 83(9), 82(20), 80(24). | [136] |
| 103 | Helifolinecine | - | 173 | 155(9.5), 129(67), 124(3), 116(6), 112(8), 98(100), 82(59), 80(10). | [136] |
| 104 | Helindicine | - | 281 | 281(35), 207(100), 191(15), 149(37), 135(33), 109(34), 97(73), 95(55), 83(57), 81(46). | [130] |
| 105 | Heliocoromandaline | - | 285 | 285(1), 267(4), 252(4), 240(6), 142(41), 125(17), 124(100), 84(63), 83(20), 82(15). | [117] |
| 106 | Heliocurassavine | - | 299 | 299(1), 284(1), 281(1), 255(3), 254(10), 252(8), 243(7), 226(8), 142(48), 124(100), 83(56), 82(40). | [117] |
| 107 | Heliocurassavicine | - | 285 | 285(1), 267(3), 252(3), 240(3), 142(49), 125(22), 124(100), 84(63), 83(58), 82(41). | [117] |
| 108 | Heliocurassavinine | - | 285 | 285(1), 267(4), 252(4), 240(3), 142(55), 125(24), 124(100), 84(22), 83(48), 82(48), 81(36). | [117] |
| 109 | Heliospathine | - | 313 | 313(1.3), 269(2), 268(1), 156(12), 139(43), 138(100), 137(15), 120(11), 95(20), 94(57), 93(75), 80(10). | [140] |
| 110 | Heliospathuline | - | 299 | 299(14.7), 282(6), 281(16.5), 238(18), 237(19), 236(20), 156(38), 139(30), 137(36), 136(20), 124(19), 120(60), 111(100), 108(51), 106(63), 94(41), 93(13), 80(78), 45(12). | [140] |
| 111 | Heliosupine | 2553 | 397 | 382(0.1), 352(0.1), 297(2), 238(4), 221(29), 220(100), 141(11), 138(10), 137(8), 136(50), 121(40), 120(95), 119(70), 106(10), 94(26), 93(52), 83 (12), 80(10), 59(10), 55(15), 43(15). | [70] |
| 112 | Heliotridine | 1447 | 155 | 138(2), 111(55), 94(18), 80(100), 68(15). | [40] |
| 113 | Heliotridine 2S-hydroxy-2S(1S-hydroxyethyl-4-methyl-pentanoyl ester | - | 313 | MS2: 269, 270, 224, 156, 138, 120, 94. | [50] |
| 114 | Heliotrine | 2100 | 313 | 255(0.3), 214(0.5), 197(1), 156(5), 139(22), 138(100), 136(13), 120(7), 94(41), 93(83), 80(40), 59(85). | [52] |
| 115 | Heliovicine | - | 285 | 267(5), 252(5), 240(4), 226(3), 175(1), 142(35), 124(100), 96(11), 95(9), 83(25), 82(20), 55(33). | [128] |
| 116 | Heliscabine | - | 255 | 255(4), 156(42), 138(12), 111(100), 99(33), 98(92), 82(77), 80(10). | [139] |
| 117 | Hydroxymyoscorpine | - | 397 | 397(0.2), 382(0.5), 338(0.1), 297(3.6), 221(21.3), 220(100), 136(40), 121(16), 120(41), 119(16.6), 94(18.4), 93(36). | [95] |
| 118 | 7-(Hydroxy-methylbutyryl)-9-viridifloryl retronecine | 2560** | 399 | 384(2), 354(0.4), 296(0.3), 282(2), 256(6), 239(21), 238(55), 138(15), 136(21), 121(35), 120(100), 108(2), 101(5), 95(10), 94(20), 93(42), 83(5), 80(6), 73(5), 67(3), 59(12), 55(0.5), 43(25). | [59] |
| 119 | Ilamine | - | 313 | 314(M+ +1, 18), 224(6), 139(3), 122(52), 120(25), 93(20), 80(8), 59(100). | [115] |
| 120 | Incanine | - | 337 | 294(2), 250(4), 222(10), 206(8), 155(5), 136(100), 120(77), 119(85), 94(42), 93(56), 80(26), 53(17), 43(69). | [46] |
| 121 | Indicine | 2126 | 299 | 255(4), 156(11), 138(100), 93(95). | [158] |
| 122 | Isoechinatine (9-(+)-viridiflorylheliotridine) | - | 299 | 299(11), 156(10), 139(33), 138(100), 137(14), 94(32), 93(68), 80(18). | [75,138] |
| 123 | Isolycopsamine | - | 299 | 281(12.3), 236(10.3), 138(67), 137(42), 136(34), 124(21), 121(15), 120(70), 119(15), 118(15), 117(14), 111(68), 110(21), 109(11), 108(49), 106(81), 95(17), 94(43), 93(28), 80(100). | [133] |
| 124 | Isoretronocanol enantiomer | 1883* | 141 | 141(20), 140(9), 124(13), 110(12), 108(11), 83(100), 82(56), 70(10), 68(11), 55(48). | [42] |
| 125 | 9-(3'-Isovaleryl)viridifloryl retronecine | - | 383 | 383(0.2), 255(3), 240(3), 223(5), 20194), 138(100), 120(10), 93(66), 85(28), 80(10), 57(19), 43(10), | [119] |
| 126 | Intermedine | 2133 | 299 | 156(9), 139(35), 138(100), 137(13), 136(13), 120(10), 95(15), 94(50), 93(80), 80(14), 67(9), 45(7), 43(18). | [40] |
| 127 | Lactodine | - | 227 | 227(9), 138(32), 137(41), 124(15), 111(60), 106(21), 94(44), 80(100). | [76,187] |
| 128 | Lasiocarpine | - | 411 | 396(2), 311(4), 279(5), 221(43), 220(11), 137(20), 136(49), 124(22), 120(74), 119(42), 106(15), 95(49), 93(23), 93(33), 83(39). | [71] |
| 129 | Latifoline | - | 393 | 393(6), 293(9), 221(10), 220(74), 219(35), 137(11), 136(100), 120(49), 119(42), 118(8), 94(33), 93(75), 83(29), 80(17), 67(9). | [103,104] |
| 130 | 9-Latifolylretronecine | - | 311 | 312(M+ +1, 40), 195(40), 161(20), 119(100). | [101] |
| 131 | Lepanthine | - | 315 | MS2: 298, 156, 138, 120, 94. | [166,167] |
| 132 | Lindelofine (or steroisomer) | 2678* | 285 | 285(5), 267(5), 252(6), 242(1), 241(1), 240(1), 226(1), 142(30), 125(21), 124(100), 83(26), 82(15), 55(30). | [42] |
| 133 | Lithosenine | - | 415 | 415(0.2), 400(10), 297(8), 256(3), 238(100), 237(15), 222(16), 220(24), 138(30), 137(8), 136(49), 121(36), 120(75), 119(36), 95(9), 94(34), 93(57), 80(12) | [156] |
| 134 | Longitubine | - | 353 | 353(29), 293(12), 181(29), 180(78), 136(75), 120(69), 119(62), 118(15), 106(10), 101(22), 94(58), 93(100), 80(32), 67(17). | [104] |
| 135 | Lycopsamine | 2145 | 299 | 299(0.5), 254(1), 156(8), 139(31), 138 (100), 137(12), 136(12), 120(10), 108(4), 95(15), 94 (55), 93(84), 80(14), 67(10), 45(8), 43(20). | [91] |
| 136 | Macrophylline | - | 239 | 98, 83(100), 55. | [62] |
| 137 | Megalanthonine | - | 301 | 301(2), 283(2), 256(11), 240(8), 212(19), 158(86), 141(15), 140(43), 138(14), 124(10), 122(18), 114(13), 97(18), 96(44), 95(62), 83(15), 82(100), 55(32). | [134] |
| 138 | 1-Methylene-8α-pyrrolizidine | 1274 | 139 | 139(15), 95(100). | [205] |
| 139 | 7-(2-Methylbutyryl)retronecine | 1760 | 239 | 239(20), 222(2), 154(20), 138(12), 137(12), 136(28), 124(37), 120(20), 111(28), 108(15), 106(28), 94(30), 93(20), 80 (100), 68(10), 57(19). | [96] |
| 140 | 9-(2-Methylbutyryl)retronecine | 1795 | 239 | 239(2), 195(5), 154(3), 138(98), 137(8), 136(5), 120(6), 108(5), 94(60), 93(100), 80 (27), 67(15), 57(30). | [96] |
| 141 | 7-(2-Methylbutyryl)-9-(2,3-dihydroxybutyryl)retronecine | 2285 | 341 | 341(1), 239(10), 223(20), 222(100), 143(18), 138(11), 137(7), 136(50), 121(17), 120(80), 119(30), 106(8), 94(40), 93(83), 85(15), 80(15), 57(30), 45(9). | [91] |
| 142 | 7-(2-Methylbutyryl)-9-echimidinyl retronecine | 2512 | 399 | 399(0.1), 384(0.5), 354(0.5), 297(1), 223(20), 222(100), 221(5), 143(15), 138(8), 137(8), 136(40), 121(30), 120(95), 119(30), 106(9), 94(31), 93 (70), 85(15), 80(15), 67(8), 59(15), 57(28), 45(8), 43(24). | [91] |
| 143 | Monocrotaline | 2268 | 325 | 236(35), 136(46), 120(100), 119(64), 93(36), 80(12), 43(69). | [195] |
| 144 | Myoscorpine | 3212* | 381 | 381(0.2), 338(1), 337(2), 336(2), 281(2), 255(1), 238(9), 237(3), 221(33), 220(100), 141(22), 138(8), 136(46), 121(34), 120(71), 119(22), 106(8), 94(36), 93(55), 83(39), 80(19), 59(4), 55(42), 53(11). | [42] |
| 145 | Neolatifoline | - | 393 | 393(7), 293(11), 221(16), 220(85), 219(31), 137(13), 136(100), 120(43), 119(48), 118(15), 94(39), 93(79), 83(32), 80(16), 67(8). | [104] |
| 146 | (7S,8R)Petranine | - | 285 | ESIMS (positive mode) m/z 286.11966 | [89] |
| 147 | (7S,8S)Petranine | - | 285 | ESIMS (positive mode) m/z 286.12162 | [89] |
| 148 | Platynecine | - | 157 | 113(18), 82(100), 68(18), 67(4), 55(27). | [106] |
| 149 | Platynecine N-Oxide 2S-hydroxy-2S(1S-hydroxyethyl)-4-methyl-pentanosyl ester | - | 331 | 314, 288, 174, 156, 112. | [50] |
| 150 | Pycnanthine | 2793 | 397 | 397(0.8), 382(0.4), 354(2), 352(2), 281(3), 254(12), 237(32), 236(100), 235(7), 138(26), 137(14), 136(74), 121(46), 120(77), 119(20), 99(20), 94(50), 93(58), 80(20), 71(20), 58(15), 45(20), 44(22), 43(20). | [91] |
| 151 | Punctanecine | - | 383 | 383(0.2), 368(0.5), 338(3), 296(2), 283(3), 266(4), 240(25), 222(19), 155(13), 140(45), 139(40), 137(73), 123(25), 122(48), 120(14), 96(23), 95(26), 83(27), 82(100). | [169,206] |
| 152 | Retronecine | 1430 2190* | 155 | 155(26), 138(2), 111(55), 94(15), 80(100), 68(15). | [40] |
| 153 | Retronecine dibenzoate | 2785 | 363 | 363(0), 258(10), 241(20), 136(43), 119(55), 105(100), 94(60), 93(78), 77(35). | [207] |
| 154 | Retronecine 2S-hydroxy-2S(1S-hydroxyethyl)-4-methyl-pentanosyl ester | - | 313 | MS2: 396, 270, 224, 156, 138. | [50] |
| 155 | Retronecine 2S-hydroxy-2S(1S-hydroxyethyl)-[1'S-hydroxyethyl)-4-methylpentanoyl]-4-methyl-pentanoyl ester, | - | 471 | ESIMS: m/z 472 [M + H]+ , for C24H41NO8 | [51] |
| 156 | Rinderine | 2155 | 299 | 284(0.1), 254(0.6), 156(9), 139(35), 138(100), 137(10), 136(10), 120(5), 95 (12), 94(23), 93(70), 80(12), 67(7), 53(5), 43(16). | [70] |
| 157 | Scorpioidine | - | 381 | 381(7), 199(7), 139(11), 138(69), 137(28), 136(13), 120(9), 101(8), 95(4), 94(21), 93(41), 83(100), 80(9), 67(6), 57(6), 55(34), 53(6), 43(10). | [159] |
| 158 | 7-Senecioylhelotridine | 1870 | 237 | 137(35), 136(18), 124(20), 111(35), 106(90), 94(21), 83(15), 80(100), 68(8), 55(18). | [70] |
| 159 | 7-Senecioylretronecine | 1816 | 237 | 237(4), 154(4), 137(30), 136(16), 124(25), 111(37), 106(40), 94(21), 83(23), 80(100), 68 (8), 55(15). | [91] |
| 160 | 9-Senecioylretronecine | 1833 | 237 | 237(1), 193(2), 155(11), 154(10), 138(23), 137(28), 136(15), 126(9), 109(5), 94(25), 93(100), 83(20), 80(16), 67(6), 55(11). | [91] |
| 161 | 7-Senecioylrinderine | 2515 | 381 | 336(0.1), 221(28), 220(61), 138(10), 137(15), 136(50), 121(75), 120(100), 119(85), 106(23), 95(15), 94(48), 93(72), 83(27), 80(30), 55(40), 43(40). | [40] |
| 162 | 7-Senecioyllycopsamine | 2497 | 381 | 336(0.5), 281(1), 238(5), 221(28), 220(100), 219(4), 141(15), 138(14), 137(12), 136(80), 121(38), 120(70), 119(20), 94(38), 93(61), 83 (76), 80(11), 55(15), 45(8), 43(28). | [91] |
| 163 | Sincamidine | - | 313 | C16H27NO5(similar to heliotrine) | [44] |
| 164 | Senkirkine | 2460 | 365 | 365(1), 266(10), 250(6), 222(9), 168(24), 153(41), 80(38), 43(100). | [54,195] |
| 165 | Subulacine | - | 155 | 155(17), 126(4), 124(13), 96(10), 80(4), 71(5), 70(100), 68(10), 56(6), 55(65). | [106,208] |
| 166 | Supinine | 1978 | 283 | 283(0.2), 140(6), 123(25), 122(100), 121(40), 120(49), 108(11), 93(20), 80(8), 70(7), 53(5), 45(4), 43(12). | [70] |
| 167 | Supinidine | 1887* | 139 | 139(40), 138(12), 122(32), 120(14), 111(15), 110(18), 108(30), 94(13), 80(100), 70(4), 68(17), 55(14), 53(17). | [42] |
| 168 | Supinidine N-oxide 2S-hydroxy-2S(1S-hydroxyethyl)-4-methyl-pentanoyl ester | - | 313 | MS2: 296, 270, 224, 156, 138. | [50] |
| 169 | Symlandine | 3194* | 381 | 381(0.3), 338(1), 337(2), 336(2), 281(2), 255(1), 238(9), 237(3), 221(30), 220(100), 141(19), 138(10), 136(47), 121(35), 120(72), 119(21), 106(9), 94(36), 93(52), 83(31), 80(17), 59(3), 55(40), 53(12). | [42] |
| 170 | Symphytine | 3240* | 381 | 381(0.4), 338(1), 337(1), 336(3), 281(2), 255(1), 238(9), 237(2), 221(33), 220(100), 141(21), 138(7), 136(44), 121(33), 120(70), 119(20), 106(7), 94(36), 93(52), 83(33), 80(18), 59(3), 55(38), 53(12). | [42] |
| 171 | Symviridine | - | 381 | 381(0.3), 337(0.1), 336(0.1), 281(0.2), 221(34.5), 220(100), 136(44.2), 121(22), 120(50), 119(11), 118(15), 117(7.6), 103(10), 95(23.5), 93(44.5). | [172] |
| 172 | Tessellatine | 2930* | 299 | 299(7), 281(7), 256(5), 255(3), 248(4), 238(11), 237(9), 236(11), 156(19), 139(25), 138(64), 137(23), 124(16), 120(40), 111(45), 108(29), 106(39), 94(54), 93(40), 80(100), 53(17). | [42] |
| 173 | 7-Tigloyl-9-(2,3-dihydroxybutyryl)retronecine | 2325 | 339 | 239(5), 238(5), 237(8), 221(22), 220(90), 219 (20), 141(20), 138(10), 137(11), 136(100), 121(15), 120(80), 119(35), 106(10), 94(58), 93(90), 83(46), 80(20), 75(2), 57(9), 55(40), 45(10) . | [96] |
| 174 | 7-Tigloyl-9-(2,3-dihydroxypropanoyl)retronecine | 2320 | 325 | 237(8), 225(12), 221(12), 220(70), 141(27), 138(5), 137(12), 136(93), 121(8), 120(55), 119(48), 106(10), 94(59), 93(100), 83(55), 80(18), 55(24). | [40] |
| 175 | 7-Tigloylheliotridine | 1873 | 237 | 237(0.5), 137(55), 136(15), 124(19), 120(5), 111(41), 107(10), 106(100), 94(20), 86(6), 83(12), 80(95), 68(5), 55(19). | [67] |
| 176 | 7-Tigloyl-9-( hydroxypropenoyl) retronecine | 2073 | 307 | 221(14), 220(100), 207(27), 181(10), 141(42), 137(10), 136(95), 120(53), 119(28), 106(10), 94(63), 93(84), 83(50), 80(20), 67(9), 55(37), 43(24). | [40] |
| 177 | 7-Tigloyllycopsamine | 2473* | 381 | 336(1), 281(1.5), 238(1), 221(20), 220(100), 136(60), 121(30), 120(80), 94(40), 93(70), 83(50), 80(18), 55(40). | [90] |
| 178 | 7-Tigloyl-9-(2-methybutyryl)retronecine | 2170 | 321 | 221(36), 220(100), 195(5), 141(32), 138(2), 137(10), 136(82), 121(6), 120(53), 119(25), 106(10), 94(53), 93(80), 83(40), 80(15), 57(20), 55(33). | [96] |
| 179 | 7-Tigloylretronecine | 1816 | 237 | 237(3), 219 (0.5), 154(2), 138(3), 137(29), 136 (15), 124(25), 120(5), 111(44), 106(50), 94(20), 93(6), 83(15), 80 (100), 55(22). | [96] |
| 180 | 9-Tigloylretronecine | 1843 | 237 | 193(5), 154(15), 138(20), 137(26), 136(13), 126(7), 119(5), 109(4), 94(23), 93(100), 83(10), 80(12), 55(18). | [96] |
| 181 | Trachelanthamidine | 1853* | 141 | 141(24), 140(10), 124(15), 110(9), 108(7), 83(100), 82(62), 70(8), 68(8), 55(45). | [42] |
| 182 | Trachelanthamine | 1970 | 285 | 267(5), 252(4), 240(4), 142(50), 125(18), 124(100), 110(4), 96(6), 83(20), 82(11), 70(6), 55(14), 43(12). | [70] |
| 183 | Trachelanthamidine 2S-hydroxy-2S(1S-hydroxyethyl)-4-methyl-pentanoyl ester | - | 299 | ESIMS: m/z 300 [M + H]+, 142, 89, 83, 82, 55. | [51] |
| 184 | 7-Trachelanthyl retronecine | 2899* | 299 | 299(12), 281(6), 236(11), 156(25), 138(50), 120(47), 111(45), 108(29), 106(40), 94(40), 93(320, 80(100). | [42] |
| 185 | Transalpinecine | - | 173 | 98, 83, 70. | [145] |
| 186 | Triangularine | 2375 | 335 | 237(23), 236(6), 235(4), 221(5), 220(30), 219(18), 141(12), 137(12), 136(100), 121(28), 120(64), 119(35), 95(12), 94(60), 93(98), 83(58), 80(15), 55(30). | [40] |
| 187 | Triangularicine | 2394 | 335 | 237(30), 236 (6), 235(3), 221(5), 220(31), 219(10), 141(18), 137(10), 136(83), 121(27), 120(51), 119(40), 95(10), 94(55), 93(100), 83(60), 80(15), 55(25). | [40] |
| 188 | Trichodesmine | 2388 | 353 | 353(1), 264(56), 222(5), 136(36), 120(100), 93(23), 81(16), 43(40). | [195] |
| 189 | Turneforcidine | - | 157 | 113(20), 82(100), 68(21), 67(6), 55(32). | [106] |
| 190 | Uluganine | 399 | 399(2), 384(8), 356(1), 355(3), 354(2). | [183] | |
| 191 | Uplandicine | 2337 | 357 | 357(4), 342(5), 297(23), 281(4), 256(4), 207 (7), 206(52), 181(80), 180 (100), 179 (43), 136(75), 121(49), 120(85), 119 (50), 101(23), 94(55), 93(74), 80(28), 73(74), 59(23), 45(20), 44(69), 43(48). | [163] |
| 192 | Viridinatine | - | 443 | 282(17), 138(41), 137(86), 124(32), 111(70), 106(42), 94(44), 80(100). | [76,187] |
| 193 | Viridiflorine | 1983 | 285 | 284(0.3), 267(5), 252(5), 240(5), 142 (70), 125 (23), 124 (100), 117(3), 110 (5), 83 (35), 82 (22), 70 (15), 55(25), 43(21). | [67] |
| 194 | 9-Viridifloryl turneforcidine (or isomer) | 2394* | 301 | 301(0.3), 283(8), 257(8), 256(14), 212(18), 159(21), 158(68), 141(17), 140(50), 138(32), 122(24), 121(12), 120(31), 117(10), 106(13), 96(43), 95(81), 83(20), 82(100), 69(22), 55(40). | [42] |
| 195 | Vulgarine | - | 397 | 398[M+ 1] +, 380(18), 336(5), 316(5), 298(40), 254(15), 240(52), 138(100), 120(14). | [11] |

2.1.1. Gas Liquid Chromatography-Mass Spectrometry (GLC-MS)
2.1.2. Liquid Chromatography-Mass Spectrometry (LC-MS)
2.2. Nuclear Magnetic Resonance (NMR)
2.3. Enzyme-Linked Immunoassay
3. Tissue Culture
4. Variation of PAs between Plant Organs and Developmental Stages
| Alkaloid | C-1 | C-2 | C-3 | C-5 | C-6 | C-7 | C-8 | C-9 | C-1' | C-2' | C-3' | C-4' | C-5' | C-6' | C-7' | C-1'' | C-2'' | C-3'' | C-4'' | C-5'' | C-6'' | solvent | References |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Necine Base | |||||||||||||||||||||||
| Helibractinecine | 82.8 | 38.3 | 53.4 | 52.5 | 36.2 | 73.7 | 71.2 | 68.6 | CDCL3-CD3OD | [111] | |||||||||||||
| Helibracteatinecine | 82.6 | 34.1 | 53.0 | 52.9 | 33.3 | 79.7 | 73.0 | 67.2 | CDCL3-CD3OD | [112] | |||||||||||||
| Turneforcidine | 40.8 | 32.6 | 56.2 | 52.8 | 37.5 | 72.0 | 73.7 | 65.6 | CD3OD-ND3 | [222] | |||||||||||||
| Transalpinecine | 82.3 | 84.4 | 59.2 | 55.9 | 28.0 | 24.6 | 72.3 | 63.9 | CD3OD | [145] | |||||||||||||
| Subulacine | 70.9 | 63.4 | 54.0 | 58.4 | 27.1 | 26.2 | 67.9 | 60.2 | CD3OD | [145] | |||||||||||||
| 1α-2α-epoxy-1β-hydroxymethyl-8α-pyrrolizidine | 83.7 | 65.2 | 62.3 | 56.2 | 27.9 | 25.0 | 72.1 | 64.0 | CD3OD | [145] | |||||||||||||
| Tertiary base and N-oxide alkaloids | |||||||||||||||||||||||
| 3'-Acetylcanescine | 133.0 | 127.5 | 62.6 | 53.6 | 34.4 | 74.4 | 75.4 | 62.3 | 175.1 | 84.3 | 72.4-(CO:170.4), (Me: 21.2) | 17.2 | 33.1 | 17.2 | 17.1 | 171.7 | 46.9 | 69.1 | 29.4 | 29.2 | CDCL3/D6-DMSO | [154] | |
| 3'-Acetylcanescenine | 133.0 | 127.5 | 62.6 | 53.6 | 34.4 | 74.4 | 75.4 | 62.3 | 175.1 | 84.3 | 73.7-(CO:170.4), (Me: 21.3) | 16.0 | 33.1 | 17.8 | 14.2 | 171.7 | 46.9 | 69.1 | 29.4 | 29.2 | CDCL3/D6-DMSO | [154] | |
| 3’-Acetylechihumiline N-oxide | 133.0 | 124.9 | 79.4 | 70.5 | 33.9 | 73.3 | 95.6 | 63.0 | 174.7 | 85.6 | 74.8-(Me: 21.6) | 61.1 | 74.0 | 27.3 | 26.4 | 166.8 | 116.2 | 161.9 | 28.0 | 21.0 | CD3OD | [166] | |
| 7-Acetylechinatine N-oxide | 133.0 | 124.3 | 79.0 | 70.1 | 33.4 | 73.8 | 95.0 | 62.1 | 175.0 | 85.2 | 72.4 | 18.2 | 34.0 | 18.5 | 16.7 | 171.5 | 21.1 | CD3OD | [69] | ||||
| 7-Acetyleuropine | 135.0 | 128.3 | 62.3 | 54.4 | 30.6 | 76.9 | 78.8 | 62.6 | 174.2 | 84.0 | 79.2- (OMe:56.8) | 13.3 | 73.4 | 26.7 | 24.9 | 171.3 | 21.5 | CDCL3 | [110] | ||||
| 3'-Acetylindicine | 132.3 | 130.7 | 62.8 | 53.0 | 36.1 | 71.1 | 78.5 | 63.3 | 174.5 | 81.6 | 71.8-(CO:170.0), (Me 21.1) | 14.3 | 32.3 | 17.2 | 16.3 | CDCL3 | [158] | ||||||
| 3'-Acetylindicine N-Oxide | 132.4 | 121.9 | 78.0 | 69.6 | 34.7 | 69.6 | 95.8 | 62.1 | 173.8 | 81.9 | 72.4-(CO:170.3), (Me: 21.2) | 14.0 | 33.0 | 17.3 | 16.8 | CDCL3 | [158] | ||||||
| 3'-Acetyllithonine | 132.5 | 128.2 | 62.2 | 53.0 | 33.8 | 73.5 | 74.8 | 61.5 | 172.3 | 82.9 | 72.1-(CO:169.0), (Me: 20.5) | 14.7 | 72.4 | 24.8 | 25.9 | 170.5 | 47.3 | 68.1 | 28.8 | 28.9 | CDCL3 | [156] | |
| 3'-Acetylrinderine | 135.0 | 128.4 | 61.9 | 54.2 | 34.0 | 75.2 | 80.4 | 62.4 | 174.3 | 81.8 | 72.5-(CO:110.2), (Me 13.9) | 21.1 | 33.1 | 16.8 | 17.2 | CDCL3 | [70] | ||||||
| 3'-Acetyltrachelanthamine | 44.7 | 30.4 | 54.1 | 54.9 | 25.6 | 31.5 | 67.8 | 67.9 | 174.3 | 81.6 | 72.2 -(CO: 169.7), (Me: 21.1) | 13.9 | 32.8 | 16.7 | 17.2 | CDCL3 | [128] | ||||||
| Canescine | 133.0 | 127.5 | 62.6 | 53.6 | 34.4 | 74.4 | 75.4 | 62.3 | 175.1 | 84.3 | 69.5 | 17.2 | 33.1 | 17.2 | 17.1 | 171.7 | 46.9 | 69.1 | 29.4 | 29.2 | CDCL3/D6-DMSO | [154] | |
| Canescenine | 133.0 | 127.5 | 62.6 | 53.6 | 34.4 | 74.4 | 75.4 | 62.3 | 175.1 | 84.3 | 71.1 | 16.0 | 33.1 | 17.8 | 14.2 | 171.7 | 46.9 | 69.1 | 29.4 | 29.2 | CDCL3/D6-DMSO | [154] | |
| Cryptanthine | 133.9 | 125.8 | 62.9 | 53.9 | 34.7 | 75.4 | 75.6 | 60.7 | 167.7 | 127.4 | 139.3 | 16.0 | 20.6 | 175.4 | 77.4 | 71.4 | 16.7 | 22.6 | [65] | ||||
| Echihumiline | 132.8 | 127.6 | 62.0 | 53.7 | 34.2 | 73.7 | 76.1 | 62.0 | 174.3 | 83.1 | 69.8 | 18.5 | 72.4 | 26.0 | 24.9 | 165.5 | 115.3 | 159.0 | 27.7 | 20.4 | CDCL3 | [91] | |
| Echimidine isomer | 133.3 | 129 | 63.2 | 54.1 | 34.8 | 74.2 | 76 | 62.9 | 174.7 | 83.7 | 70.0 | 18.9 | 73.8 | 26.5 | 25.2 | 167.5 | 128.9 | 138.2 | 14.8 | 12.3 | CDCL3 | [87] | |
| Echiupinine | 133.0 | 128.1 | 62.3 | 53.7 | 34.3 | 72.8 | 75.6 | 62.2 | 175.1 | 82.9 | 68.7 | 17.2 | 32.9 | 17.1 | 16.9 | 167.1 | 115.6 | 158.1 | 27.5 | 20.3 | CDCL3 | [95] | |
| Echiupinine N-oxide | 131.8 | 128.1 | 78.2 | 69.1 | 34.3 | 71.4 | 93.7 | 60.2 | 174.9 | 84.0 | 69.4 | 16.9 | 33.2 | 17.5 | 17.2 | 166.2 | 114.5 | 160.3 | 27.6 | 20.6 | CDCL3 | [95] | |
| 7-Epi-echimiplatine N-oxide | 132.4 | 125.9 | 79.7 | 70.2 | 35.8 | 71.4 | 96.7 | 63.6 | 176.1 | 86.9 | 71.1 | 19.0 | 75.6 | 26.3 | 27.0 | D2O | [164] | ||||||
| 2'',3''-Epoxyechiumine | noise | 128.5 | 62.7 | 53.6 | 34.4 | 75.3 | 75.4 | 62.5 | 175.1 | 82.9 | 69.4 | 17.1 | 33.0 | 16.9 | 17.3 | noise | 77.2 | 60.0 | 13.6 | 19.3 | CDCL3 | [63] | |
| Erythro-3''-chloro-2''-hydroxyechiumine | 132.6 | 127.8 | 62.4 | 53.7 | 34.5 | 75.4 | 75.8 | 62.3 | 173.6 | 83.0 | 69.4 | 16.9 | 33.0 | 17.1 | 17.3 | 175.1 | 77.2 | 62.8 | 17.9 | 23.0 | CDCL3 | [63] | |
| Floridine | 44.2 | 31.2 | 54.0 | 54.0 | 25.4 | 29.5 | 67.6 | 66.2 | 173.4 | 83.4 | 72.7 -(CO: 169.6), (Me: 21.2) | 15.1 | 73.5 | 26.2 | 24.9 | CDCL3 | [128] | ||||||
| Floridinine | 43.3 | 28.6 | 53.8 | 54.0 | 24.6 | 30.6 | 68.4 | 66.2 | 174.6 | 82.5 | 69.8 | 18.6 | 73.9 | 25.0 | 26.0 | CDCL3 | [128] | ||||||
| Floridimine | 44.3 | 28.7 | 54.2 | 54.3 | 25.3 | 30.6 | 67.6 | 63.2 | 174.7 | 84.1 | 69.9 | 18.6 | 73.8 | 26.0 | 24.9 | CDCL3 | [128] | ||||||
| Helindicine | 134.2 | 125.3 | 62.5 | 55.5 | 36.7 | 70.5 | 80.5 | 61.7 | 175.6 | 84.5 | 70.5 | 17.3 | 34.2 | 17.2 | 17.6 | CD3OD | [130] | ||||||
| Heliotridine 2S-hydroxy-2S-(1S-hydroxyethyl)-4-methyl-pentanoyl ester | 134.0 | 123.5 | 62.0 | 55.9 | 35.0 | 70.8 | 79.4 | 62.2 | ? | ? | 45.0 | 25.2 | 23.1 | 24.4 | 73.9-(8':17.4) | CD3OD | [50] | ||||||
| Heliscabine | 81.1 | 38.4 | 52.8 | 52.0 | 36.0 | 72.8 | 69.8 | 70.0 | 168.1 | 127.2 | 139.7 | 16.0 | 20.6 | CDCL3 | [139] | ||||||||
| Heliospathine | 133.1 | 128.9 | 62.4 | 53.9 | 36.1 | 70.5 | 78.2 | 62.2 | 174.5 | 84.4 | 71.2 | 17.4 | 38.9 | 24.7 | 12.1-(8':11.9) | CDCL3 | [140] | ||||||
| Heliospathuline | 139.1 | 124.2 | 62.4 | 53.9 | 34.8 | 76.5 | 76.2 | 59.3 | 174.3 | 83.8 | 72.4 | 16.5 | 31.9 | 15.6 (7'':17.2) | CDCL3 | [140] | |||||||
| Hydroxymyoscorpine | 132.8 | 128.5 | 62.8 | 53.7 | 34.3 | 73.8 | 75.7 | 62.5 | 174.2 | 83.0 | 69.6 | 18.4 | 73.5 | 25.9 | 24.8 | 167.1 | 128.6 | 137.8 | 14.4 | 11.9 | CDCL3 | [95] | |
| Isoechinatine (9-(+)-viridiflorylheliotridine) | 136.1 | 126.3 | 61.7 | 54.3 | 33.8 | 74.7 | 79.8 | 61.9 | 174.3 | 83.9 | 71.6 | 17.5 | 32.2 | 17.8 | 15.9 | CDCL3 | [72,75] | ||||||
| Iso-lycopsamine | 139.1 | 123.2 | 63.0 | 53.4 | 34.9 | 76.4 | 76.5 | 59.5 | 174.1 | 82.8 | 69.4 | 16.5 | 33.38 | 17.2 (7'':12.27) | CDCL3 | [133] | |||||||
| Lactodine | 138.1 | 123.2 | 63.8 | 54.1 | 35.1 | 69.4 | 76.5 | 58.9 | 173.3 | 68.4 | 16.3 | CDCL3 | [187] | ||||||||||
| Leptanthine | 133.7 | 124.6 | 61.7 | 54.9 | 36.3 | 70.0 | 79.9 | 61.6 | 174.8 | 85.1 | 70.2 | 18.0 | 74.2 | 26.0 | 25.4 | CD3OD | [166] | ||||||
| Lithosenine N-oxide | 133.7 | 124.0 | 78.0 | 69.7 | 36.3 | 71.2 | 96.8 | 62.5 | 175.5 | 86.1 | 71.2 | 19.1 | 75.2 | 27.0 | 26.3 | CD3OD | [166] | ||||||
| Lithosenine | 133.0 | 126.9 | 62.2 | 53.1 | 33.8 | 73.4 | 74.8 | 61.4 | 173.5 | 83.4 | 68.9 | 18.0 | 72.5 | 24.7 | 25.9 | 170.3 | 47.5 | 68.1 | 28.9 | 29.0 | CDCL3 | [156] | |
| Megalanthonine | 36.0 | 31.2 | 51.1 | 54.6 | 36.8 | 69.5 | 72.8 | 66.2 | 174.5 | 84.1 | 72.0 | 17.0 | 32.0 | 15.6 | 17.8 | CDCL3 | [134] | ||||||
| Methylechiuplatine | 133.5 | 128.0 | 62.9 | 54.0 | 34.7 | 73.6 | 76.1 | 61.3 | 171.3 | 44.9 | 69.7 | 44.8 | 172.2 | 51.8:OMe | 27.3 | 166.9 | 127.7 | 139.2 | 15.9 | 20.6 | CDCL3 | [65] | |
| Myoscorpine | 133.1 | 128.1 | 62.7 | 53.8 | 34.4 | 73.7 | 75.8 | 62.2 | 175.1 | 82.9 | 69.3 | 17.2 | 32.9 | 17.1 | 16.9 | 167.0 | 128.5 | 137.8 | 14.4 | 11.9 | CDCL3 | [95] | |
| Myoscorpine N-oxide | 131.8 | 124.0 | 7 8.4 | 69.3 | 34.3 | 72.4 | 93.8 | 60.1 | 14.9 | 84.0 | 69.4 | 16.9 | 33.1 | 17.5 | 17.2 | 166.2 | 127.9 | 139.0 | 14.6 | 12.0 | CDCL3 | [95] | |
| Onosmerectine N-oxide | 133.5 | 123.8 | 78.6 | 69.7 | 35.7 | 70.7 | 96.7 | 62.5 | 175.3 | 85.7 | 74.6 | 70.8 | 18.7 | 26.6 | 25.9 | CD3OD | [164] | ||||||
| (7S, 8R)-Petranine | 134.4 | 121.1 | 70.9 | 63.4 | 34.7 | 70.2 | 87.7 | 59.8 | 167.5 | 126.9 | 140.3 | 16.1 | 20.6 | 68.8 | CDCL3 | [89] | |||||||
| (7S, 8S)-Petranine | 134.6 | 121.3 | 71.2 | 63.6 | 34.8 | 70.6 | 88.1 | 59.9 | 167.0 | 127.0 | 149.9 | 16.3 | 20.8 | 69.1 | CDCL3 | [89] | |||||||
| Platynecine N-oxide 2S-hydroxy-2S-(1S-hydroxyethyl)-4-methyl-pentanoyl ester | 37.3 | 30.6 | 73.0 | 70.6 | 35.8 | 70.1 | 91.5 | 67.3 | ? | ? | 44.5 | 25.3 | 23.3 | 24.8 | 73.9-(8':17.4) | CD3OD | [50] | ||||||
| Pycnanthine | 132.8 | 126.0 | 62.2 | 53.9 | 34.2 | 72.4 | 75.8 | 61.7 | 174.4 | 83.6 | 71.3 | 15.8 | 32.0 | 17.7 | 17.0 | 165.5 | 112.4 | 160.1 | 66.8 | 30.3 | CDCL3 | [91] | |
| Retronecine 2S-hydroxy-2S-(1S-hydroxyethyl)-4-methyl-pentanoyl ester | 133.2 | 123.8 | 62.0 | 54.0 | 36.7 | 70.0 | 80.0 | 61.0 | 175.4 | 82.3 | 45.2 | 25.3 | 23.3 | 24.6 | 73.9-(8':17.6) | CD3OD | [51] | ||||||
| Retronecine N-oxide 2S-hydroxy-2S-(1S-hydroxyethyl)-4-methyl-pentanoyl ester | 133.9 | 123.4 | 78.8 | 70.0 | 35.7 | 70.6 | 97.2 | 62.4 | 175.9 | 82.0 | 45.0 | 25.2 | 23.3 | 24.6 | 73.6-(8':17.7) | CD3OD | [51] | ||||||
| Retronecine N-oxide 2S-hydroxy-2S-(1R-hydroxyethyl)-4-methyl-pentanoyl ester | 134.0 | 123.4 | 78.8 | 70.0 | 35.8 | 70.6 | 97.0 | 62.0 | 175.5 | 81.0 | 44.0 | 25.8 | 23.9 | 24.3 | 74.0-(8':16.5) | CD3OD | [51] | ||||||
| Retronecine 2S-hydroxy-2S-(1S-hydroxyethyl)-2S-[(1'S-hydroxyethyl)-4-methylpentanoyl]-4-methyl-pentanoyl ester | 135.4 | 124.0 | 62.0 | 54.9 | 36.6 | 70.4 | 79.8 | 61.7 | 175.7 | 82.2 | 43.6 | 25.2 | 24.0 | 24.7 | 73.4-(8': 7.4) | 180.0 | 81.3 | 45.0 | 25.2 | 23.1 | 24.7 (7'':73.6) (8'':17.0) | CD3OD | [51] |
| Symviridine | 133 | 127.8 | 62.7 | 53.8 | 34.3 | 72.7 | 75.8 | 62.3 | 174.4 | 83.4 | 70.7 | 17.1 | 32.9 | 17.7 | 16.0 | 166.9 | 115.5 | 158.2 | 27.5 | 20.3 | CDCL3 | [172] | |
| Supinidine N-oxide 2S-hydroxy-2S-(1S-hydroxyethyl)-4-methyl-pentanoyl ester | 137.7 | 123.0 | 76.9 | 71.1 | 25.2 | 28.3 | 90.2 | 61.2 | 176.2 | 82.0 | 45.2 | 25.2 | 23.5 | 24.6 | 73.3-(8':17.6) | CD3OD | [51] | ||||||
| Tessellatine | 138.6 | 124.5 | 63.0 | 53.4 | 34.7 | 75.7 | 76.0 | 59.6 | 173.1 | 83.5 | 71.4 | 16.9 | 31.9 | 17.6 (7'':15.7) | CDCL3 | [42] | |||||||
| Threo-2'',3''-dihydroxyechiumine | 132.9 | 125.0 | 63.3 | 54.0 | 34.8 | 74.9 | 75.8 | 63.0 | 174.9 | 83.2 | 69.8 | 16.9 | 33.2 | 17.2 | 17.2 | 175.4 | 78.7 | 71.5 | 16.5 | 22.1 | CDCL3 | [63] | |
| Trachelanthamidine 2S-hydroxy-2S-(1S-hydroxyethyl)-4-methyl-pentanoyl ester | 48.0 | 29.7 | 55.9 | 55.8 | 25.6 | 31.3 | 71.9 | 63.4 | 181.6 | 81.8 | 44.1 | 25.0 | 24.0 | 25.0 | 73.9-(8':17.4) | CD3OD | [51] | ||||||
| Uplandicine | 132.7 | 127.7 | 62.1 | 53.4 | 34.0 | 73.6 | 75.5 | 62.2 | 174.2 | 83.2 | 69.6 | 18.5 | 73.6 | 24.7 | 25.9 | 170.2 | 21.1 | - | - | - | CDCL3 | [163] | |
| Viridiflorine | 44.5 | 31.5 | 54.5 | 54.2 | 25.5 | 30.0 | 67.8 | 67.2 | 174.4 | 83.6 | 71.0 | 17.2 | 32.1 | 17.9 | 15.9 | [42] | |||||||
| Viridinatine | 139.3 | 124.2 | 64.2 | 54.2 | 36.7 | 77.3 | 77.3 | 59.4 | 174.1 | 82.8 | 68.4 | 16.2 | 33.5 | 17.2 | 17.3 | 176.2 | 83.2 | 69.1 | 16.8 | 34.12 | 18.1 (7'':18.9) | CDCL3 | [187] |
| Viridinatine N-oxide stereoisomer | 132.5 | 125.4 | 79.5 | 70.1 | 35.8 | 71.3 | 96.6 | 63.3 | 176.6 | 85.9 | 70.9 | 17.6 | 34.6 | 18.9 | 18.4 | 176.1 | 86.2 | 72.3 | 18.6 | 34.2 | 18, 17.2 | ||
| Alkaloid | H-1 | H-2 | H-3 | H-5 | H-6 | H-7 | H-8 | H-9 | H-3' | H-4' | H-5' | H-6' | C-7' | H-2'' | H-3'' | H-4'' | H-5'' | solvent | References |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Necine Base | |||||||||||||||||||
| Helibractinecine [C8H15NO3] | 1.94 | α 3.13 β 2.93 | α 3.07 β 2.91 | 2.03 | 4.50 | 3.34 | α 3.60 β 3.66 | CDCL3-CD3OD | [111] | ||||||||||
| Helibracteatinecine [C8H15NO3] | 1.63 | α 3.26 β 2.55 | α 3.26 β 2.52 | α 1.84 β 2.11 | 3.94 | 3.12 | α 3.79 β 3.84 | CDCL3-CD3OD | [112] | ||||||||||
| Turneforcidine [C8H15NO2] | 2.53 | α 1.68 β 2.10 | α 3.08 β 2.55 | α 3.03 β 2.70 | α 2.00 β 1.99 | 4.18 | 3.16 | α 3.57 β 3.53 | CD3OD-ND3 | [222] | |||||||||
| Transalpinecine [C8H15NO3] | 4.18 | α 3.86 β 3.38 | α 3.06 β 3.36 | α 1.95 β 2.16 | α 1.88 β 2.66 | 3.99 | α 3.90 β 3.61 | CD3OD | [145] | ||||||||||
| Subulacine [C8H15NO2] | 3.95 | α 3.79 β 3.38 | α 2.97 3.62 | α 1.96 2.24 | α 2.19 2.26 | 4.45 | α 3.97 β 3.79 | CD3OD | [145] | ||||||||||
| 1α-2α-epoxy-1β-hydroxymethyl-8α-pyrrolizidine [C8H15NO2] | 4.59 | α 3.81 β 3.89 | α 3.23 β 3.50 | α 2.08 β 2.23 | α 2.00 β 2.30 | 4.31 | α 3.93 β 3.69 | CD3OD | [145] | ||||||||||
| Tertiary base and N-oxide alkaloids | |||||||||||||||||||
| 3'-Acetylcanescine [C22H35NO8] | 5.83 | α 3.95 β 3.38 | α 3.33 β 2.65 | 2.09 | 5.38 | 4.35 | α 4.79 β 4.67 | 5.21-(COMe: 2.00) | 1.19 | 2.00 | 0.94 | 0.91 | 2.45 | 1.26 | 1.26 | CDCL3/D6-DMSO | [154] | ||
| 3'-Acetylcanescenine [C22H35NO8] | 5.83 | α 3.95 β 3.38 | α 3.33 β 2.65 | 2.09 | 5.38 | 4.35 | α 4.74 β 4.73 | 5.17 -(COMe: 2.03) | 1.19 | 2.00 | 0.94 | 0.91 | 2.45 | 1.26 | 1.26 | CDCL3/D6-DMSO | [154] | ||
| 3’-Acetylechihumiline N-oxide [C22H33NO9] | 6.03 | α 4.37 β 4.69 | 3.90 – 3.75 (m) | α 2.20 β 2.78 | 5.76 | 4.83 | 4.72 | 5.46 -(COMe: 1.98) | 1.34 | 1.25 | 1.35 | 5.67 | - | 1.92 | 2.18 | CD3OD | [166] | ||
| 3'-Acetylechinatine [C17H27NO6] | 5.76 | α 3.37 β 3.92 | α 2.68 β 3.30 | α 1.91 β 1.98 | 4.17 | 3.96 | α 4.78 β 4.96 | 5.21-(COMe: 2.04) | 1.29 | 2.01 | 0.88 | 0.96 | CDCL3 | [70] | |||||
| 7-Acetylechinatine N-oxide | 6.00 | α 4.70 β 4.38 | α 3.83 β 3.79 | α 2.74 β 2.23 | 5.69 | 4.88 | 4.78 | 3.96 | 1.42 | 2.17 | 0.90 | 0.93 | 2.06 | CD3OD | [164] | ||||
| 7-Acetyleuropine [C18H29NO7] | 5.81 | α 4.02 β 3.38 | α 3.28 β 2.86 | 1.93 | 5.09 | 4.16 | α 4.93 β 4.87 | 3.81-(OMe: 3.27) | 1.27 | 1.19 | 1.31 | 2.06 | CDCL3 | [110] | |||||
| 3'-Acetylindicine [C17H27NO6] | 5.92 | α 3.96 β 3.45 | α 3.29 β 2.76 | α 1.98 β 2.02 | 4.30 | 4.21 | 4.80 | 5.20 -(COMe: 2.04) | 1.23 | 2.09 | 0.95 | 0.92 | CDCL3 | [158] | |||||
| 3'-Acetylindicine N-Oxide [C17H27NO7] | 5.79 | 4.45 | 3.73-3.80 | α 2.01 β 2.62 | 4.70 | 4.66 | 4.83 | 5.23 -(COMe: 2.03) | 1.24 | 2.05 | 0.97 | 0.91 | CDCL3 | [158] | |||||
| 3'-Acetyllithosenine [C22H35NO9] | 5.75 | α 3.77 β 3.29 | α 3.17 β 2.54 | 1.96 | 5.23 | 4.16 | α 4.68 β 4.59 | 5.28 -(COMe: 1.87) | 1.13 | 1.25 | 1.11 | 2.33 | 1.17 | 1.17 | CDCL3 | [156] | |||
| 3'-Acetylrinderine [C17H27NO6] | 5.76 | α 3.37 β 3.92 | α 2.68 β 3.30 | α 1.91 β 1.98 | 4.21 | 3.96 | α 4.66 β 4.93 | 5.28 -(COMe: 2.02) | 1.25 | 2.01 | 0.92 | 0.98 | CDCL3 | [70] | |||||
| 3'-Acetyltrachelanthamine [C17H29NO5] | 2.09 | α 1.70 β 2.02 | α 3.28 β 2.60 | α 3.07 β 2.65 | α 1.89 β 1.84 | α 2.05 β 1.63 | 3.38 | 4.16 | 5.22 -(COMe: 1.97) | 1.23 | 2.02 | 0.90 | 0.96 | CDCL3 | [128] | ||||
| Canescine [C20H33NO7] | 5.83 | α 3.95 β 3.38 | α 3.33 β 2.65 | 2.09 | 5.38 | 4.35 | α 4.79 β 4.67 | 4.07 | 1.19 | 2.00 | 0.94 | 0.91 | 2.45 | 1.26 | 1.26 | CDCL3/ D6-DMSO | [154] | ||
| Canescenine [C20H33NO7] | 5.83 | α 3.95 β 3.38 | α 3.33 β 2.65 | 2.09 | 5.38 | 4.35 | α 4.74 β 4.73 | 3.89 | 1.19 | 2.00 | 0.94 | 0.91 | 2.45 | 1.26 | 1.26 | CDCL3/ D6-DMSO | [154] | ||
| Cryptanthine | 5.74 | α 4.0 β 3.37 | α 3.40 β 2.61 | 2.12 | 5.3 | 4.39 | α 4.84 β 4.64 | - | 6.1 | 1.98 | 1.89 | 3.8 | 1.17 | 1.24 | CDCL3 | [65] | |||
| 5'-Deoxylasiocarpine [C21H33NO6] | 5.8 | α 3.50 β 4.18 | α 3.00 β 3.40 | 1.8 | 5.12 | 4.37 | 4.88 | 3.7 -(OMe: 3.22) | 1.21 | 2.20 | 0.95 | 0.94 | 6.13 | 1.95 | 1.82 | CDCL3 | [123] | ||
| Echihumiline [C20H31NO7] | 5.87 | α 3.35 β 3.72 | α 2.83 β 2.83 | 2.09 | 5.48 | 4.13 | α 4.65 β 4.96 | 4.21 | 1.26 | - | 1.24 | 1.31 | 5.59 | - | 1.90 | 2.15 | CDCL3 | [91] | |
| Echihumiline N-oxide [C20H31NO8] | 5.97 | α 4.54 β 4.77 | α 3.73 β 4.13 | α 2.24 β 2.89 | 5.77 | 5.45 | α 4.71 β 4.94 | 4.22 | 1.27 | - | 1.25 | 1.30 | 5.57 | - | 1.92 | 2.17 | CDCL3 | [91] | |
| Echimidine isomer (tigloyl) [C20H31NO7] | 5.81 | α 3.34 β 3.83 | α 2.60 β 3.27 | 2.05 | 5.37 | 4.32 | α 4.60 β 4.86 | 4.12 | 1.21 | 1.17 | 1.26 | - | 6.05 | 1.91 | 1.76 | CDCL3 | [87] | ||
| 7-Epi-echimiplatine N-oxide [C15H25NO7] | 5.89 | α 4.42 β 4.21 | α 3.74 β 3.59 | α 2.47 β 2.01 | 4.67 | 4.55 | 4.78 | 4.18 | 1.14 | 1.16 | 1.21 | D2O | [164] | ||||||
| Floridine [C17H29NO6] | 2.23 | α 2.15 β 2.15 | α 3.75 β 2.77 | α 3.48 β 2.65 | 2.04 | α 2.20 β 1.70 | 4.01 | α 4.16 β 4.27 | 5.45 -(COMe: 1.98) | 1.36 | - | 1.25 | 1.40 | CDCL3 | [128] | ||||
| Floridinine [C15H27NO5] | 2.36 | α 2.08 β 2.20 | α 3.51 β 2.66 | α 3.24 β 2.85 | α 2.0 β 1.94 | α 2.15 β 1.63 | 3.70 | α 4.20 β 4.53 | 4.19 | 1.27 | - | 1.28 | 0.96 | CDCL3 | [128] | ||||
| Floridimine [C15H27NO5] | 2.30 | α 2.17 β 2.28 | α 3.89 β 2.83 | α 3.59 β 2.89 | 2.11 | α 2.26 β 1.80 | 4.30 | α 4.35 β 4.45 | 4.26 | 1.32 | - | 1.28 | 1.33 | CDCL3 | [128] | ||||
| Helibracteatine [C13H21NO4] | 1.85 | α 3.22 β 2.54 | α 3.22 β 2.54 | α 1.85 β 2.05 | 4.08 | 3.30 | α 4.35 β 4.46 | 6.10 | 1.99 | 1.91 | CDCL3 + D2O | [112] | |||||||
| Helibracteatinine [C13H21NO4] | α 1.80 β 2.10 | α 3.35 β 2.78 | α 3.20 β 2.60 | 1.80 | 5.09 | 3.30 | α 3.64 β 3.84 | 6.12 | 1.98 | 1.88 | CDCL3 + D2O | [112] | |||||||
| Helindicine [C15H23NO4] | 5.94 | α 4.40 β 3.92 | α 3.97 β 3.34 | 2.15 | 4.63 | 4.90 | α 4.98 β 4.80 | 4.05 | 1.17 | 2.03 | 0.95 | 0.93 | CD3OD | [130] | |||||
| Heliotridine 2S-hydroxy-2S-(1S-hydroxyethyl)-4-methyl-pentanoyl ester [C16H27NO5] | 5.95 | 3.70 4.18 | 3.10 2.64 | 2.10 | 4.52 | 4.60 | 4.85 | 1.74 1.79 | 1.82 | 0.87 | 0.99 | 3.78-(8':1.15) | CD3OD | [50] | |||||
| Heliscabine [C13H21NO4] | 1.86 | α 3.42 β 3.05 | α 3.30 β 3.05 | 2.16 | 4.66 | 3.64 | α 4.25 β 4.35 | 6.10 | 1.99 | 1.89 | CDCL3 | [139] | |||||||
| Ilamine [C16H27NO5] | 5.81 | 3.45 4.10 | 2.62 3.35 | 1.86 | 1.66 2.10 | 4.43 | 4.78 | 3.81 | 1.28 | 1.31 | 1.20 (8-OMe: 3.30) | CDCL3 | [115] | ||||||
| Ilamine N-oxide [C16H27NO6] | 5.81 | α 3.43 β 4.55 | α 3.48 β 3.79 | α 1.99 β 1.89 | 2.50 | 4.81 | α 4.85 β 4.69 | 3.80 | 1.26 | 1.29 | 1.22-(8-OMe:3.30) | CDCL3 | [115] | ||||||
| Isoechinatine (9-(+)-Viridiflorylheliotridine) [C15H25NO5] | 5.66 | 3.30 3.83 | 2.55 3.22 | 1.80 1.92 | 4.13 | 3.97 | 4.70 5.00 | 3.92 | 1.24 | 2.13 | 0.89 | 0.85 | CDCL3 | [72,75] | |||||
| Leptanthine [C15H25NO6] | 5.90 | α 3.90 β 4.40 | 3.90 | 2.20 | 4.65 | 4.95 | 4.90 | 4.20 – Me 1.98 | 1.29 | 1.28 | 1.32 | [166] | |||||||
| Leptanthine N-oxide [C15H25NO7] | 6.05 | α 4.65 β 4.85 | 4.1 – 4.0 (m) | α 2.23 β 2.63 | 4.83 | 5.20 | α 4.92 β 4.98 | 4.22 – Me 1.98 | 1.28 | 1.25 | 1.31 | [166] | |||||||
| Lactodine [C11H17NO4] | 5.68 | α 3.90 β 3.34 | α 3.26 β 2.59 | α 1.93 β 1.87 | 4.10 | 4.02 | α 5.01 β 4.89 | 4.20 | 1.39 | CDCL3 + D2O | [187] | ||||||||
| Lithosenine [C20H33NO8] | 5.78 | α 3.77 β 3.29 | α 3.16 β 2.57 | 1.96 | 5.21 | 4.20 | α 4.66 β 4.52 | 4.03 | 1.13 | 1.17 | 1.11 | 2.33 | 1.16 | 1.16 | CDCL3 | [156] | |||
| Megalanthonine [C15H27NO5] | 2.78 | α 1.89 β 2.15 | α 3.26 β 2.61 | α 3.21 β 2.73 | α 2.03 β 2.14 | 4.29 | 3.48 | α 4.09 β 4.46 | 2.64 | - | 2.64 | 3.70 | 1.34 | 6.80 | 1.96 | 1.80 | CDCL3 | [134] | |
| Methylechiuplatine [C20H29NO7] | 5.77 | α 3.97 β 3.37 | α 3.32 β 2.64 | 2.09 | 5.40 | 4.33 | α 4.68 β 4.62 | [65] | |||||||||||
| Neo coramandaline [C15H27NO4] | 1.9 | α 1.85 β 1.55 | α 3.20 β 2.50 | α 3.05 β 2.64 | α 1.85 β 1.55 | α 1.90 β 1.35 | 3.56 | α 4.30 β 4.20 | 3.98 | 1.23 | 2.14 | 0.88 | 0.92 | CDCL3 | [74] | ||||
| Onosmerectine N-oxide [C15H25NO7] | 5.96 | α 4.66 β 4.45 | 3.87 | α 2.58 β 2.12 | 4.75 | 4.85 | α 4.92 β 4.88 | 4.22 | 1.26 | 1.31 | 1.27 | CD3O | [164] | ||||||
| (7S, 8R)-Petranine [C14H20CINO3] | 5.82 | α 4.55 β 4.77 | α 4.09 β 4.35 | α 2.48 β 2.70 | 4.92 | 5.36 | 4.78 | 6.18 | 2.00 | 1.90 | α5.42 β 5.75 | CDCL3 | [89] | ||||||
| (7S, 8S)-Petranine [C14H20CINO3] | 5.83 | α 4.54 β 4.78 | α 4.09 β 4.35 | α 2.51 β 2.69 | 4.94 | 5.38 | 4.88 | 6.20 | 2.02 | 1.90 | α5.71 β 5.39 | CDCL3 | [89] | ||||||
| Platynecine N-oxide 2S-hydroxy-2S-(1S-hydroxyethyl)-4-methyl-pentanoyl ester [C16H29NO6] | 2.22 | α 1.90 β 2.33 | α 3.30 β 3.89 | α 3.61 β 3.87 | α 2.07 β 2.14 | 4.52 | 3.84 | α 4.29 β 3.85 | α 1.76 β 1.70 | 1.82 | 0.87 | 0.98 | 3.78-(8':1.15) | CD3OD | [50] | ||||
| Pycnanthine [C20H31NO7] | 5.83 | α 3.55 β 4.06 | α 2.80 β 3.50 | 2.15 | 5.47 | 4.12 | α 4.65 β 4.75 | 3.99 | 1.26 | 2.15 | 0.93 | 0.85 | 5.92 | - | 3.65 | 2.06 | CDCL3 | [91] | |
| Retronecine 2S-hydroxy-2S-(1S-hydroxyethyl)-4-methyl-pentanoyl ester [C16H27NO5] | 5.96 | α 3.98 β 4.46 | α 3.49 β 3.87 | 2.19 | 4.64 | 4.90 | 4.95 | 1.75 | 1.83 | 0.88 | 0.99 | 3.788'-1.17 | CD3OD | [51] | |||||
| Retronecine N-oxide 2S-hydroxy-2S-(1S-hydroxyethyl)-4-methyl-pentanoyl ester [C16H27NO6] | 5.93 | α 4.36 β 4.65 | α 3.70 β 3.82 | α 2.63 β 2.08 | 4.72 | 4.68 | 4.85 | α 1.73 β 1.80 | 1.82 | 0.88 | 0.98 | 3.76-(8':1.16) | CD3OD | [51] | |||||
| Retronecine N-oxide 2S-hydroxy-2S-(1R-hydroxyethyl)-4-methyl-pentanoyl ester [C16H27NO6] | 5.95 | α 4.36 β 4.60 | α 3.70 β 3.85 | α 2.60 β 2.08 | 4.72 | 4.69 | 4.82 | α 1.66 β 1.47 | 1.74 | 0.88 | 1.00 | 3.91-(8':1.22) | CD3OD | [51] | |||||
| Retronecine 2S-hydroxy-2S-(1S-hydroxyethyl)-2S-[(1'S-hydroxyethyl)-4-methylpentanoyl]-4-methyl-pentanoyl ester [C24H41NO8] | 5.95 | α 3.96 β 4.39 | α 3.29 β 3.89 | 2.18 | 4.61 | 4.91 | 4.94 | α 1.79 β 1.82 | 1.80 | 0.95 | 0.99 | 3.70-(8':1.16) | 1.79 | 1.85 | α 1.81 β 0.87 | 3.77-(6'':1.14) | CD3OD | [51] | |
| Symviridine [C20H31NO6] | 5.84 | α4.02 β3.43 | α 3.40 β 2.68 | 2.10 | 5.41 | 4.48 | α 4.78 β 4.66 | 3.97 | 1.22 | 2.13 | 0.86 | 0.92 | 5.57 | - | 1.88 | 2.12 | CDCL3 | [172] | |
| Supinidine N-oxide 2S-hydroxy-2S-(1S-hydroxyethyl)-4-methyl-pentanoyl ester [C16H27NO4] | 5.90 | α 4.36 β 4.56 | α 3.61 β 3.67 | α 2.08 β 2.38 | 2.50 2.03 | 4.66 | 4.77 | α 1.73 β 1.80 | 1.83 | 0.88 | 0.98 | CD3OD | [51] | ||||||
| Trachelanthamidine 2S-hydroxy-2S-(1S-hydroxyethyl)-4-methyl-pentanoyl ester [C16H29NO4] | 2.28 | 1.90 2.27 | α 3.22 β 3.48 | α 3.74 β 3.12 | 2.08 2.19 | 2.25 1.97 | 3.99 | 3.69 3.63 | 1.77 1.69 | 1.83 | 0.94 | 1.00 | 3.77-(8':1.15) | CD3OD | [51] | ||||
| Uplandicine [C17H27NO7] | 5.83 | α 3.38 β 3.93 | α 2.70 β 3.35 | 2.06 | 5.34 | 4.43 | α 4.62 β 4.87 | 4.14 | 1.22 | - | 1.26 | 1.19 | 1.98 | CDCL3 | [163] | ||||
| Viridinatine [C22H37NO8] | 5.71 | α 3.80 β 3.30 | α 3.16 β 2.19 | α 1.92 β 1.85 | 5.48 | 4.10 | α 4.98 β 4.76 | 3.90 | 1.28 | 2.15 | 0.86 | 0.93 | - | 3.95 | 1.32 | 2.14 (6'':0.90) (7'':1.00) | CDCL3 + D2O | [187] | |
| Viridinatine N-oxide stereoisomer [C22H37NO9] | 5.86 | α 4.51 β 4.22 | α 3.73 β 3.61 | α 2.45 β 2.00 | 4.67 | 5.57 | 4.75 | 4.04 | 1.04 | 1.89 | 0.75–0.78 | - | 3.93 | 1.10 | 2.05, 0.81 | D2O | [164] | ||
5. Biological Activity of PAs
5.1. Antimicrobial Activity
5.2. Sequestration in Insects and Antifeedant Properties
5.3. Biological Importance
6. Chemotaxonomic Information of PAs in Boraginaceae
6.1. Genus Amsinckia
6.2. Genus Cynoglossum
6.3. Genus Echium
6.4. Genus Heliotropium
6.5. Genus Paracaryum and Paracynoglossum
6.6. Genus Rindera
6.7. Genus Symphytum
6.8. Other Boraginaceae Species
7. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Wink, M. Plant breeding: Importance of plant secondary metabolites for protection against pathogens and herbivores. Theor. Appl. Genet. 1988, 75, 225–233. [Google Scholar] [CrossRef]
- Wink, M. Evolution of secondary metabolites from an ecological and molecular phylogenetic perspective. Phytochemistry 2003, 64, 3–19. [Google Scholar] [CrossRef]
- Detzel, A.; Wink, M. Attraction, deterrence or intoxication of bees (Apis mellifera) by plant allelochemicals. Chemoecology 1993, 4, 8–18. [Google Scholar] [CrossRef]
- Culvenor, C.C.J.; Edgar, J.A.; Smith, L.W. Pyrrolizidine alkaloids in Honey from Echium plantagineum L. J. Agric. Food Chem. 1981, 29, 958–960. [Google Scholar] [CrossRef]
- Hartmann, T. Chemical ecology of pyrrolizidine alkaloids. Planta 1999, 207, 483–495. [Google Scholar] [CrossRef]
- Prakash, A.S.; Pereira, T.N.; Reilly, P.E.A.; Seawright, A.A. Pyrrolizidine alkaloids in human diet. Mutation Res. 1999, 443, 53–67. [Google Scholar] [CrossRef]
- Edgar, J.A.; Smith, L.W. Transfer of Pyrrolizidine Alkaloids into Eggs: Food Safety Implications. ACS Symp. Ser. 1999, 754, 118–128. [Google Scholar] [CrossRef]
- Edgar, J.A.; Roeder, E.; Molyneux, R.J. Honey from plants containing pyrrolizidine alkaloids: A potential threat to health. J. Agric. Food Chem. 2002, 50, 2719–2730. [Google Scholar] [CrossRef]
- Fu, P.P.; Yang, Y.; Xia, Q.; Chou, M.W.; Cui, Y.Y.; Lin, A.G. Pyrrolizidine alkaloids-tumorigenic components in Chinese herbal medicines and dietary supplements. J. Food Drug Anal. 2002, 10, 198–211. [Google Scholar]
- Azadbakht, M.; Talavaki, M. Qualitative and quantitative determination of pyrrolizidine alkaloids of wheat and flour contaminated with Senecio in Mazandaran province farm. Int. J. Pharm. Res. 2003, 2, 179–183. [Google Scholar]
- Boppré, M.; Colegate, S.M.; Edgar, J.A. Pyrrolizidine alkaloids of Echium vulgare honey found in pure pollen. J. Agric. Food Chem. 2005, 53, 594–600. [Google Scholar] [CrossRef]
- Boppré, M.; Colegate, S.M.; Edgar, J.A.; Fischer, W.O. Hepatotoxic pyrrolizidine alkaloids in pollen and drying-related implication for commercial processing of bee pollen. J. Agric. Food Chem. 2008, 56, 5662–5672. [Google Scholar] [CrossRef]
- Sharma, R.A.; Singh, B.; Singh, D.; Chandrawat, P. Ethnomedicinal, pharmacological properties and chemistry of some medicinal plants of Borginaceae in India. J. Med. Plant Res. 2009, 3, 1153–1175. [Google Scholar]
- European Food Safety Authority. Opinion of the scientific panel on contamination in the food chain on a request from the European Commission related to pyrrolizidine alkaloids as undesirable substances in animal feed. EFSA J. 2007, 447, 1–51. [Google Scholar]
- Kakar, F.; Akbarian, Z.; Leslie, T.; Mustafa, M.L.; Watson, J.; van Egmond, H.P.; Omar, M.F.; Mofleh, J. An outbreak of hepatic veno-occlusive disease in western Afghanistan associated with exposure to wheat flour contaminated with pyrrolizidine alkaloids. J. Toxicol. 2010. [Google Scholar] [CrossRef]
- Kempf, M.; Heil, S.; Haβlauer, I.; Schmidt, L.; von der Ohe, K.; Theuring, C.; Reinhard, A.; Schreier, P.; Beuerle, T. Pyrrolizidine alkaloids in pollen and pollen products. Mol. Nutr. Food Res. 2010, 54, 292–300. [Google Scholar] [CrossRef]
- Staiger, C. Comfrey: A Clinical Overview. Phytother. Res. 2012, 26, 1441–1448. [Google Scholar]
- Cooper, R.A.; Huxtable, R.J. The relationship between reactivity of metabolites of pyrrolizidine alkaloids and extrahepatic toxicity. Proc. West Pharmacol. Soc. 1999, 42, 13–16. [Google Scholar]
- Kim, H.Y.; Stermitz, F.R.; Li, J.K.; Coulombe, R.A. Comparative DNA cross-linkage by activated pyrrolizidine alkaloids. Food Chem. Toxicol. 1999, 37, 619–625. [Google Scholar] [CrossRef]
- Schmeller, T.; El-Shazly, A.; Wink, M. Allelochemical activities of pyrrolizidine alkaloids: Interaction with neuroreceptors and acetylcholine related enzymes. J. Chem. Ecol. 1997, 23, 399–416. [Google Scholar] [CrossRef]
- Wink, M.; Roberts, M.F. Compartmentation of alkaloid synthesis, transport and storage. In Alkaloids, Biochemistry, Ecology and Medicinal Application; Roberts, M.F., Wink, M., Eds.; Plenum Press: New York, NY, USA and London, UK, 1998; Chapter 10; pp. 239–269. [Google Scholar]
- Wink, M.; Schmeller, T.; Latz-Brüning, B. Modes of action of allelochemical alkaloids: Interaction with neuroreceptors, DNA and other molecular targets. J. Chem. Ecol. 1998, 24, 1881–1937. [Google Scholar] [CrossRef]
- Bull, L.B.; Culvenor, C.C.J.; Dick, A.T. The Pyrrolizidine Alkaloids; North Holland: Amsterdam, The Netherlands, 1968. [Google Scholar]
- McLean, E.K. The toxic activities of pyrrolizidine (Senecio) alkaloids. Pharmacol. Rev. 1970, 22, 429–476. [Google Scholar]
- Smith, L.W.; Culvenor, C.C.J. Plant sources of hepatotoxic pyrrolizidine alkaloids. J. Nat. Prod. 1981, 44, 129–152. [Google Scholar]
- Robins, D.J. The pyrrolizidine alkaloids. Fortschr. Chem. Org. Naturst. 1982, 41, 115–203. [Google Scholar]
- Mattocks, A.R. Chemistry and Toxicology of Pyrrolizidine Alkaloids; Academic Press: London, UK and New York, NY, USA, 1986. [Google Scholar]
- Rizk, A.M. Naturally Occurring Pyrrolizidine Alkaloids; CRC Press: Boca Raton, FL, USA, 1991. [Google Scholar]
- Hartmann, T.; Witte, L. Chemistry, biology and chemoecology of pyrrolizidine alkaloids. In Alkaloids: Chemical and Biological Perspectives; Pelletier, S.W., Ed.; Elsevier Science Ltd.: Kidlington, UK, 1995; Vol. 9, Charpter 4; pp. 155–233. [Google Scholar]
- Roeder, E. Medicinal plants in Europe containing pyrrolizidine alkaloids. Pharmazie 1995, 50, 83–98. [Google Scholar]
- Roeder, E. Analysis of pyrrolizidine alkaloids. Curr. Org. Chem. 1999, 3, 557–576. [Google Scholar] [CrossRef]
- Roeder, E. Medicinal plants in China containing pyrrolizidine alkaloids. Pharmazie 2000, 55, 711–725. [Google Scholar]
- Wink, M. Plant secondary metabolism: Diversity, function and its evolution. Nat Prod. Commun. 2008, 3, 1205–1216. [Google Scholar]
- Wink, M.; van Wyk, B.E. Mind-altering and Poisonous Plants of the World; Briza Publications: Pretoria, South Africa, 2008. [Google Scholar]
- Roeder, E.; Wiedenfeld, H. Pyrrolizidine alkaloids in medicinal plants of Mongolia, Nepal and Tibet. Pharmazie 2009, 64, 699–716. [Google Scholar]
- Wiedenfeld, H. Plants containing pyrrolizidine alkaloids: Toxicity and problems. Food Addit. Contam. 2011, 28, 282–292. [Google Scholar] [CrossRef]
- Jiang, Y.; Fu, P.P.; Lin, G. Hepatotoxicity of naturally occurring pyrrolizidine alkaloids. Asian J. Pharmacodyn. Pharmacokinet. 2006, 6, 187–192. [Google Scholar]
- Dreger, M.; Stanisławska, M.; Krajewska-Patan, A.; Mielcarek, S.; Mikołajczak, L.P.; Buchwald, W. Pyrrolizidine alkaloids–chemistry, biosynthesis, pathway, toxicity, safety and perspectives of medicinal usage. Herba Polonica 2009, 55, 127–147. [Google Scholar]
- Roeder, E.; Wiedenfeld, H. Plants containing pyrrolizidine alkaloids used in the Traditional Indian Medicine—including Ayurveda. Pharmazie 2013, 68, 83–92. [Google Scholar]
- El-Shazly, A.; El-Domiaty, M.; Witte, L.; Wink, M. Pyrrolizidine alkaloids in members of the Boraginaceae from Sinia (Egypt). Biochem. Syst. Ecol. 1998, 26, 619–639. [Google Scholar] [CrossRef]
- Roeder, E.; Sarg, T.; El-Dahmy, S.; Abdel-Ghani, A. Pyrrolizidine alkaloids from Alkanna orientalis. Fitoterapia 1992, 63, 405–408. [Google Scholar]
- Kelley, R.B.; Seiber, J.N. Pyrrolizidine alkaloid chemosystematics in Amsinckia. Phytochemistry 1992, 31, 2369–2387. [Google Scholar] [CrossRef]
- Kelley, R.B.; Seiber, J.N. Pyrrolizidine alkaloid from Amsinckia. Phytochemistry 1992, 31, 2513–2518. [Google Scholar] [CrossRef]
- Culvenor, C.C.J.; Smith, L.W. Alkaloids of Amsinckia species: A. intermedia, A. hispida and A. lycoposoides. Aust. J. Chem. 1966, 19, 1955–1964. [Google Scholar] [CrossRef]
- Roitman, J.N. Pyrrolizidine alkaloids of Amsinckia menziesii. Aust. J. Chem. 1983, 36, 769–778. [Google Scholar] [CrossRef]
- Cooper, R.A.; Bowers, R.J.; Beckham, C.J.; Huxtable, R.J. Preparative separation of pyrrolizidine alkaloids by high-speed counter-current chromatography. J. Chromatogr. A 1996, 732, 43–50. [Google Scholar] [CrossRef]
- Pedersen, E. Pyrrolizidine alkaloids in Danish species of the family Boraginaceae. Arch. Pharm. Chem. Sci. Ed. 1975, 3, 55–64. [Google Scholar]
- Broch-Due, A.I.; Aasen, A.J. Alkaloids of Anchusa officinalis L. Identification of pyrrolizidine alkaloid lycopsamine. Acta Chem. Scand. Ser. B. 1980, 34, 75–77. [Google Scholar] [CrossRef]
- Hendriks, H.; Bruins, A.P.; Huizing, H.J. Detection of curassavine and some related pyrrolizidine alkaloids in an Anchusa officinalis strain with means of positive ion and negative ion chemical ionization GC-MS. Biomed. Environ. Mass Spctrom. 1988, 17, 129–132. [Google Scholar] [CrossRef]
- Siciliano, T.; de Leo, M.; Bader, A.; de Tommasi, N.; Vrieling, K.; Braca, A.; Morelli, I. Pyrrolizidine alkaloids from Anchusa strigosa and their antifeedant activity. Phytochemistry 2005, 66, 1593–1600. [Google Scholar] [CrossRef]
- Braca, A.; Bader, A.; Siciliano, T.; Morelli, I.; Tommasi, N. New pyrrolizidine alkaloid and glycosides from Anchusa strigosa. Planta Med. 2003, 69, 835–841. [Google Scholar] [CrossRef]
- El-Dahmy, S.; Adel Ghani, A. Alkaloids of Arnebia decumbens Vent. Az. J. Pharm. Sci. 1995, 15, 24–34. [Google Scholar]
- Roeder, E.; Rengel-Mayer, B. Pyrrolizidine alkaloids from Arnebia euchroma. Planta Med. 1993, 59, 192. [Google Scholar]
- Wassel, G.; El-Menshawi, B.; Saeed, A.; Mahran, G. Toxic pyrrolizidine alkaloids of certain Boraginaceae. Acta Pharm. Suec. 1987, 24, 199–204. [Google Scholar]
- Langer, T.; Franz, C. Determination of pyrrolizidine alkaloids in commercial samples of borage seed oil products by GC-MS. Sci. Pharm. 1997, 65, 321–328. [Google Scholar]
- Herrmann, M.; Joppe, H.; Schmaus, G. Thesinine-4'-O-β-D-glucoside the first glycosylated plant pyrrolizidine alkaloid from Borago officinalis. Phytochemistry 2002, 60, 399–402. [Google Scholar] [CrossRef]
- Telezhenetskaya, M.V.; Matkarmov, A.D.; Khadzhibekov, S.N.; Yunusov, S.Y. Alkaloids of some Central Asian plants. Khim. Prir. Soedin 1987, 3, 663–664. [Google Scholar]
- Siddiqi, M.A.; Suri, K.A.; Suri, O.P.; Atal, C.K. A new pyrrolizidine alkaloid from Caccinia glauca. Pytochemistry 1978, 17, 2049–2050. [Google Scholar] [CrossRef]
- El-Shazly, A. Pyrrolizidine alkaloids of Cerinthe glabra Miller (Boraginaceae). Zagazig. J. Pharm. Sci. 2004, 13, 1–5. [Google Scholar]
- Mroczek, T.; Baj, S.; Chrobok, A.; Glowniak, K. Screening for pyrrolizidine alkaloids in plant materials by electron ionization RP-HPLC-MS with thermabeam interface. Bio. Med. Chromatogr. 2004, 18, 745–751. [Google Scholar] [CrossRef]
- Roeder, E.; Wiedenfeld, H.; Kaus, K.J. Das Pyrrolizidinalkaloid Intermedin aus Cerinthe minor L. Sci. Pharm. 1990, 58, 9–13. (In German) [Google Scholar]
- Wassel, G.; El-Menshawi, B.; Saeed, A.; Mahran, G.; Reisch, J. New sources of pyrrolizidine alkaloids: Genus Cordia (Ehretiaceae) and Schismus (Gramineae). Sci. Pharm. 1987, 55, 163–166. [Google Scholar]
- Stermiz, F.; Pass, M.A.; Kelley, R.B.; Liddell, J.R. Pyrrolizidine alkaloids from Cryptantha species. Phytochemistry 1993, 33, 383–387. [Google Scholar] [CrossRef]
- Williams, M.T.; Warnock, B.J.; Betz, J.M.; Beck, J.J.; Gardner, D.R.; Lee, S.T.; Molyneux, R.J.; Colegate, S.M. Detection of high levels of pyrrolizidine-N-oxides in the endangered plant Cryptantha crassipes (Terlingua creek cat’s-eye) using HPLC-ESI-MS. Phytochem. Anal. 2011, 22, 532–540. [Google Scholar] [CrossRef]
- Colegate, S.M.; Gardner, D.R.; Davis, T.Z.; Betz, J.M.; Panter, K.E. Dehydropyrrolizidine alkaloids in two Cryptantha species: Including two new open chain diesters one of which is amphoteric. Phytochem. Anal. 2013, 24, 201–212. [Google Scholar] [CrossRef]
- Beck, J.J.; Stermitz, F.R. Pyrrolizidine alkaloids from Brickellia grandiflora and Cryptantha jamesii. Biochem. Syst. Ecol. 2002, 30, 1079–1081. [Google Scholar] [CrossRef]
- El-Shazly, A.; Sarg, T.; Ateya, A.; Abdel-Aziz, E.; Witte, L.; Wink, M. Pyrrolizidine alkaloids of Cynoglossum officinale and Cynoglossum amabile (Family Boraginaceae). Biochem. Syst. Ecol. 1996, 24, 415–421. [Google Scholar] [CrossRef]
- Culvenor, C.C.J.; Smith, L.W. Alkaloids of Cynoglossum australe and C. amabile. Aust. J. Chem. 1967, 20, 2499–2503. [Google Scholar] [CrossRef]
- Damianakos, H.; Jeziorek, M.; Pietrosiuk, A.; Sykłowska-Baranek, K.; Chinou, I. Isolation of pyrrolizidine alkaloids from Cynoglossum columnae Ten. (Boraginaceae). Planta Med. 2013. [Google Scholar] [CrossRef]
- El-Shazly, A.; Sarg, T.; Witte, L.; Wink, M. Pyrrolizidine alkaloids from Cynoglossum creticum. Phytochemistry 1996, 42, 1217–1221. [Google Scholar] [CrossRef]
- Zalkow, L.H.; Bonetii, S.; Gelbaum, L.T.; Gordon, M.M.; Patil, B.B.; Shani, A.; van Derveer, I.D. Pyrrolizidine alkaloid from middle eastern plans. J. Nat. Prod. 1979, 42, 603–614. [Google Scholar] [CrossRef]
- Asibal, C.F.; Glinski, J.A.; Gelbaum, L.T.; Zalkow, L.H. Pyrrolizidine alkaloids from Cynoglossum creticum. Synthesis of pyrrolizidine alkaloids echinatine, rinderine and analogues. J. Nat. Prod. 1989, 52, 109–118. [Google Scholar] [CrossRef]
- Chen, Z.W.; Tang, J.; Li, J.; Zhang, Q.; Zhao, X.; Hu, J. Constituents of Cynoglossum zeylanicum. Zhongguo Yaoke Daxue Xuebao (J. Chin. Pharm.) 1987, 18, 51–53. (In Chinese) [Google Scholar]
- Ravi, S.; Lakshmanan, A.J. Coromandaline, a pyrrolizidine alkaloid from Cynoglossum furcatum. Ind. J. Chem. 2000, 39B, 80–82. [Google Scholar]
- Ravicumar, R.; Lakshmanan, A.J. Isoechinatine, a pyrrolizidine alkaloid from Cynoglossum furcatum. Ind. J. Chem. 2004, 43B, 406–409. [Google Scholar]
- Ravi, S.; Ravikumar, R.; Lakshmanan, A.J. Pyrrolizidine alkaloids from Cynoglossum furcatum. J. Asian Nat. Prod. Res. 2008, 10, 307–310. [Google Scholar] [CrossRef]
- Mattoks, A.R.; Pigott, C.D. Pyrrolizidine alkaloids from Cynoglossum germanicum. Phytochemistry 1990, 29, 2871–2872. [Google Scholar] [CrossRef]
- Suri, K.A.; Sawhney, R.S.; Atal, C.K. Pyrrolizidine alkaloids from Cynoglossum lanceolatum, C. glochidiatum and Lindelofia angustifolia. Indian J. Pharm. 1975, 37, 69–70. [Google Scholar]
- Crowely, H.C.; Culvenor, C.C.J. Alkaloids of Cynoglossum latifolium, latifoline and 7-angeloylretronecine. Aust. J. Chem. 1962, 15, 139–144. [Google Scholar] [CrossRef]
- Kelly, H.A.; Robins, D.J. Pyrrolizidine alkaloids from Cynoglossum macrostylum. Fitoterapia 1992, 63, 91. [Google Scholar]
- Guner, N. Pyrrolizidine alkaloids from Cynoglossum montanum. Marmara Univ. Eczacilik Derg. 1987, 3, 57–60. [Google Scholar]
- Hagan, D.; Robins, D.J. Pyrrolizidine alkaloids from Cynoglossum nervosum. Fitoterapia 1991, 62, 186–187. [Google Scholar]
- Resch, J.F.; Meinwald, J. A revised structure for acetylheliosupine. Pytochemistry 1982, 21, 2430–2431. [Google Scholar] [CrossRef]
- Van Dam, N.M.; Witte, L.; Theuring, C.; Hartmann, T. Distribution, biosynthesis and turnover of pyrrolizidine alkaloids in Cynoglossum officinale. Phyochemistry 1995, 39, 287–292. [Google Scholar] [CrossRef]
- Man’ko, I.V.; Marchenko, L.G. Pictumine, a new alkaloid from Cynoglossum pictum. Khim. Prir. Soedin. 1972, 5, 655–656. [Google Scholar]
- Man’ko, I.V. Alkaloids of Cynoglossum amabile and C. viridiflorum. Rastit. Resur. 1972, 8, 243–246. [Google Scholar]
- Mehrabani, M.; Ghannadi, A.; Sajjadi, E.; Ghassemi, N.; Shams-Ardakani, M. Toxic pyrrolizidine alkaloids of Echium amoenum Fisch. & Mey. DARU J. Pharm. Sci. 2006, 14, 122–127. [Google Scholar]
- Sarg, T.; El-Dahmy, S.; Abdel-Aziz, E.; Abdel-Ghani, A.; Roeder, E. Pyrrolizidine alkaloids from Echium angustifolium. Fitoterapia 1992, 63, 466–468. [Google Scholar]
- Alali, F.Q.; Tahboub, Y.R.; Ibrahim, E.S.; Qandil, A.; Tawaha, K.; Burgess, J.P; Sy, A.; Nakanishi, Y.; Kroll, D.J.; Oberlies, N.H. Pyrrolizidine alkaloids from Echium glomeratum (Boraginaceae). Phytochemistry 2008, 69, 2341–2346. [Google Scholar] [CrossRef]
- El-Shazly, A.; Abdel-All, M.; Tei, A.; Wink, M. Pyrrolizidine alkaloids from Echium rauwolfii and Echium horridum (Boraginaceae). Z. Naturforsch. 1999, 54c, 295–300. [Google Scholar]
- El-Shazly, A.; Sarg, T.; Ateya, A.; Abdel-Aziz, E.; El-Dahmy, S.; Witte, L.; Wink, M. Pyrrolizidine and tetrahydroisoquinoline alkaloids from Echium humile. Phytochemistry 1996, 42, 225–230. [Google Scholar] [CrossRef]
- Carvalho, J.C.B.; Almeida, H.D.S.; Lobo, J.F.R.; Ferreira, J.L.P.; Oliveira, A.P.; Leandro Rocha, L. Pyrrolizidine alkaloids in two endemic capeverdian Echium species. Biochem. Syst. Ecol. 2013, 50, 1–6. [Google Scholar]
- Colegate, S.M.; Edgar, J.A.; Knill, A.M.; Lee, S.T. Solid-phase extraction and HPLC-MS profiling of pyrrolizidine alkaloids and their N-oxides: A case study of Echium plantagineum. Phytochem. Anal. 2005, 16, 108–119. [Google Scholar] [CrossRef]
- Culvenor, C.C.J. The alkaloids of Echium plantagineum L. Aust. J. Chem. 1956, 9, 512–520. [Google Scholar] [CrossRef]
- Roeder, E.; Liu, K.; Bourauel, T. Pyrrolizidine alkaloids from Echium pininana. Phytochemistry 1991, 30, 3107–3110. [Google Scholar] [CrossRef]
- El-Shazly, A.; Sarg, T.; Ateya, A.; Abdel-Aziz, E.; El-Dahmy, S.; Witte, L.; Wink, M. Pyrrolizidine alkaloids from Echium setosum and Echium vulgare. J. Nat. Prod. 1996, 59, 310–313. [Google Scholar] [CrossRef]
- Man’ko, I.V. Alkaloids of Cynoglossum officinale and Echium vulgare and standard preparation from Cynoglossum officinale. Farm. Zh. (Kiev) 1964, 19, 22–26. [Google Scholar]
- Karimov, A.; Telezhenetskaya, M.V.; Lutfullin, K.; Yunusov, S.Y. Alkaloids of Echium vulgaris and Berberis oblonga. Khim. Prir. Soedin. 1975, 11, 433–434. [Google Scholar]
- Dominguez, D.M.; Reina, M.; Santos-Guerra, A.; Santana, O.; Agullo, T.; Lopez-Balbo, C.; Gonzalez-Coloma, A. Pyrrolizidine alkaloids from Canarian endemic plants and their biological effects. Biochem. Syst. Ecol. 2008, 36, 153–166. [Google Scholar] [CrossRef]
- Suri, O.P.; Jamwal, R.S.; Suri, K.A.; Atal, C.K. Ehretinine, a noval pyrrolizidine alkaloid from Ehretia aspera. Phytochemistry 1980, 19, 1273–1274. [Google Scholar] [CrossRef]
- L’Empereur, K.M.; Li, Y.; Stermitz, F. Pyrrolizidine alkloids from Hackelia californica and Gnophaela latipennis, and H. californica-hasted arctiid moth. J. Nat. Prod. 1989, 52, 360–366. [Google Scholar] [CrossRef]
- Li, Y. Two new pyrrolizidine alkaloids from Hackelia californica (Gray) Johnston (Boraginaceae). Huaxue Xuebao 1990, 48, 415–418. [Google Scholar]
- Hagglund, K.M.; L’Empereur, K.M.; Roby, M.K.; Stermitz, F.R. Latifoline and latifoline N-oxide from Hackelia floribunda. J. Nat. Prod. 1985, 48, 638–639. [Google Scholar] [CrossRef]
- Roitman, J.N. Longitubine and neolatifoline, new pyrrolizidine alkaloids from Hackelia longituba. Aust. J. Chem. 1988, 41, 1827–1833. [Google Scholar]
- Akramov, S.T.; Shadmanov, Z.; Samatov, A.; Yunusov, S.Y. Alkaloids of Senecio jacobaea, Heliotropium acutiflorum and H. transoxanum. Khim. Prir. Soedin. 1966, 4, 258. [Google Scholar]
- Birecka, H.; Frohlich, M.W.; Glickman, M. Free and esterified necines in Heliotropium species from Mexico and Texas. Phytochemistry 1983, 22, 1167–1171. [Google Scholar] [CrossRef]
- Marquez, V.C. Chromatographic separation of Bulensia retamo, Heliotropium arborescens and Cestrum auriculatum. Bol. Soc. Quim. Peru. 1961, 27, 161–172. [Google Scholar]
- Rizk, A.M.; Hammouda, F.M.; Roeder, E.; Wiedenfeld, H.; Ismail, S.I.; Hassan, N.M. Occurrence of pyrrolizidine alkaloids in Heliotropium bacciferum Forssk. Sci. Pharm. 1988, 56, 105–110. [Google Scholar]
- Farrag, N.M.; Adel-Aziz, E.M.; El-Shafae, A.M.; Ateya, A.M.; El-Domiaty, M.M. Pyrrolizidine alkaloids of Heliotropium bacciferum Frossk from Egypt. Int. J. Pharmacogn. 1996, 34, 374–377. [Google Scholar] [CrossRef]
- Reina, M.; Mericli, A.H.; Cabbera, R.; Gonzalez-Coloma, A. Pyrrolizidine alkaloid from Heliotropium bovei. Phytochemistry 1995, 38, 355–358. [Google Scholar] [CrossRef]
- Lakshmanan, A.J.; Shanmugasundaram, S. Helibractinecine, a pyrrolizidine alkaloid from Heliotropium bracteatum. Phytochemistry 1994, 36, 245–248. [Google Scholar] [CrossRef]
- Lakshmanan, A.J.; Shanmugasundaram, S. Ester alkaloid from Heliotropium bracteatum. Phytochemistry 1995, 40, 291–294. [Google Scholar] [CrossRef]
- Marquina, G.; Laguna, A.; Velez, H.; Ripperger, H. 9-Angeloylretronecine N-oxide from Heliotropium bursiferum. Pharmazie 1988, 34, 55–56. [Google Scholar]
- Eröksüz, H.; Eröksüz, Y.; Ozer, H.; Ceribasi, A.O.; Tosun, F.; Tamer, U.; Kizilay, C.A. Toxicity of dietery Heliotropium circinatum to rats. Vet. Hum. Toxicol. 2003, 45, 198–201. [Google Scholar]
- Farsam, H.N.; Yassa, N.; Sarkhail, P.; Shafiee, A. New pyrrolizidine alkaloids from Heliotropium crassifolium. Planta Med. 2000, 66, 389–391. [Google Scholar] [CrossRef]
- Catalfamo, J.L.; Martin, W.B.; Birecka, H. Accumulation of alkaloids and their necines in Heliotropium curassavicum, H. spathulatum and H. indicum. Phytochemistry 1982, 21, 2669–2675. [Google Scholar] [CrossRef]
- Mohanraj, S.; Subramanian, P.S. Curassavine, an alkaloid from Heliotropium curassavicum Linn. with a C8 necic acid skeleton. J. Chem. Soc. Chem. Comm. 1978. [Google Scholar] [CrossRef]
- Subramanian, P.S.; Mohanraj, S.; Cockrum, P.A.; Culvenor, C.C.J.; Edgar, J.A.; Farhn, J.L.; Smith, L.W. The alkaloids of Heliotropium curassavicum. Aust. J. Chem. 1980, 33, 1357–1363. [Google Scholar] [CrossRef]
- Davicino, J.G.; Pestchanker, M.J.; Giodano, O.S. Pyrrolizidine alkaloids from Heliotropium curassavicum. Phytochemistry 1988, 27, 960–962. [Google Scholar] [CrossRef]
- Akramov, S.T.; Kiyamitdinova, F.; Yunusov, S.Y. Alkaloids of Rindera cyclodonata, Rinderia echinata and Heliotropium dasycarpum. Khim. Prir. Soedin. 1967, 3, 288–289. [Google Scholar]
- Hammouda, F.M.; Rizk, A.M.; Ismail, S.I.; Atteya, S.Z.; Ghaleb, H.A.; Madkour, M.K.; Pohland, A.E.; Wood, G. Poisonous plants containing edible ones and toxic substances in plant foods. Part 3, Pyrrolizidine alkaloids from Heliotropium digynum Forssk (H. luteum Poir.). Pharmazie 1984, 39, 703–705. [Google Scholar]
- Farsam, H.; Yassa, N.; Shafiee, A.; Amanlou, M. Pyrrolizidine alkaloids from Heliotropium disciforme. Pharm. Pharmacol. Lett. 1998, 8, 79–80. [Google Scholar]
- Shafiee, A.; Salimi, M.; Farsam, H.; Yassa, N. Pyrrolizidine alkaloids from Heliotropium dissitiflorum Boiss. DARU J. Pharm. Sci. 2002, 10, 168–170. [Google Scholar]
- Suri, O.P.; Sawhney, R.S.; Atal, C.K. Pyrrolizidine alkaloids from Heliotropium eichwaldii and Lindelofia spectabilis. Indian J. Chem. 1975, 13, 505–506. [Google Scholar]
- Yassa, N.H.; Farsam, H.; Shafiee, A.; Rustaiyan, A. Pyrrolizidine alkaloids from Heliotropium esfandiarii. Planta Med. 1996, 62, 583–584. [Google Scholar] [CrossRef]
- Yassa, N.H.; Farsam, H.; Rustaiyan, A.; Shafiee, A. Alkaloids of Boraginaceae II [1], pyrrolizidine alkaloids of Heliotropium europaeum L. population Garmsar. J. Sci. Islamic Repub. Iran 1999, 10, 39–42. [Google Scholar]
- Tosun, F.; Tamer, U. Determination of pyrrolizidine alkaloids in the seeds of Heliotropium europaeum by GC-MS. J. Fac. Pharm. Ankara 2004, 33, 7–9. [Google Scholar]
- Reina, M.; Gonzalez-Coloma, A.; Gutierrez, C.; Cabbera, R.; Henriquez, J.; Villarroel, L. Bioactive saturated pyrrolizidine alkaloid from Heliotropium floridum. Phytochemistry 1997, 46, 845–853. [Google Scholar]
- Constantinidis, T.; Harvala, C.; Skaltsounis, A.L. Pyrrolizidine N-oxide alkaloids of Heliotropium hirsutissimum. Phytochemistry 1993, 32, 1335–1337. [Google Scholar] [CrossRef]
- Souza, J.S.N.; Machado, L.L.; Pessoa, O.D.L.; Braz-Filho, R.; Overk, C.R.; Yao, P.; Cordell, G.A.; Lemos, T.L.G. Pyrrolizidine alkaloids from Heliotropium indicum. J. Braz. Chem. Soc. 2005, 16, 1410–1414. [Google Scholar] [CrossRef]
- Singh, J.P.; Pandey, D.P.; Singh, A.; Singh, R. Alkaloids of Heliotropium indicum. J. Ind. Chem. Soc. 2005, 82, 175–176. [Google Scholar]
- Dash, G.K.; Abdullah, M.S. A review on Heliotropium indicum L. (Boraginaceae). Int. J. Pharm. Sci. Res. 2013, 4, 1253–1258. [Google Scholar]
- Ravi, S.; Lakshmanan, A.J.; Herz, W. Isolycopsamine, a pyrrolizidine alkaloid from Heliotropium keralense. Phytochemistry 1990, 29, 361–364. [Google Scholar] [CrossRef]
- Reina, M.; Gonzalez-Coloma, A.; Gutierrez, C.; Cabbera, R.; Henriquez, J.; Villarroel, L. Pyrrolizidine alkaloids from Heliotropium megalanthum. J. Nat. Prod. 1998, 61, 1418–1420. [Google Scholar] [CrossRef]
- Kiyamitdinova, F.; Akramov, S.T.; Yunusov, S.Y. Alkaloids from the family Boraginaceae. Khim. Prir. Soedin. 1967, 3, 411–412. [Google Scholar]
- Mohanraj, S.; Kulanthaivel, P.; Subramanian, P.S.; Herz, W. Helifoline, a pyrrolizidine alkaloid from Heliotropium ovalifolium. Phytochemistry 1981, 20, 1991–1995. [Google Scholar] [CrossRef]
- Zalkow, L.H.; Gelbaum, L.; Keinan, E. Isolation of the pyrrolizidine alkaloid europine N-oxide from Heliotropium maris-mortui and H. rotundifolium. Phytochemistry 1978, 17, 172. [Google Scholar] [CrossRef]
- Asibal, C.F.; Gelaum, L.T.; Zalkow, L.H. Pyrrolizidine alkaloids from Heliotropium rotundifolium. J. Nat. Prod. 1989, 52, 726–731. [Google Scholar]
- Lakshmanan, A.J.; Shanmugasundaram, S. Heliscabine, a pyrrolizidine ester alkaloid from Heliotropium scabrum. Phytochemistry 1995, 39, 473–475. [Google Scholar] [CrossRef]
- Roeder, E.; Breitmaier, E.; Birecka, H.; Frohlich, M.W.; Badzies-Crombach, A. Pyrrolizidine alkaloids of Heliotropium spathulatum. Phytochemistry 1991, 30, 1703–1706. [Google Scholar] [CrossRef]
- Mattocks, A.R. Strigosine, the major alkaloid of Heliotropium strigosum. J. Chem. Soc. 1964, 1974–1977. [Google Scholar] [CrossRef]
- Singh, B.; Sahu, P.M.; Jain, S.C.; Singh, S. Antineoplastic and antiviral screening of pyrrolizidine alkaloids from Heliotropium subulatum. Pharm. Biol. 2002, 40, 581–586. [Google Scholar] [CrossRef]
- Crowley, H.C.; Culvenor, C.C.J. The alkaloids of Heliotropium supinum L., with observations on viridifloric acid. Aust. J. Chem. 1959, 12, 694–705. [Google Scholar] [CrossRef]
- Trigo, J.R.; Witte, L.; Brown, K.S.; Hartmann, T.; Barata, L. pyrrolizidine alkaloids in the acrtiid moth hyalurga syma. J. Chem. Ecol. 1993, 4, 669–679. [Google Scholar]
- Medina, J.C.M.; Gauze, G.F.; Vidott, G.J.; Sarragiotto, M.H.; Basso, E.A.; Peixoto, J.L.B. Structural characterization of saturated pyrrolizidine alkaloids from Heliotropium transalpinum var. transalpinum Vell by NMR spectroscopy and theoretical calculation. Tetrahedron Lett. 2009, 50, 2640–2642. [Google Scholar]
- Suri, K.A.; Suri, O.P.; Dhar, K L.; Atal, C.K. Chemical composition of Lappula glochidiata and Crotalaria anagyroides. Indian J. Chem. 1978, 16B, 78. [Google Scholar]
- Man’ko, I.V.; Vasil’kov, P.N. Lappula intermedia alkaloids. Tr. Leninger. Khim.-Farm. Inst. 1968, 26, 166. [Google Scholar]
- Wiedenfeld, H.; Amarsanaa, B.; Altanchimeg, D.; Narantuya, S. Pyrrolizidine alkaloid containing plants in Mongolian traditional medicine: Lappula myosotis Moench. Sci. Pharm. 2005, 73, 139–145. [Google Scholar]
- Kelly, H.A.; Robins, D.J. Pyrrolizidine alkaloids from Lindelofia longiflora. Fitoterapia 1990, 61, 89–90. [Google Scholar]
- Akramov, S.T.; Kiyamitdinova, F.; Yunusov, S.Y. Alkaloids of Solenanthus turkestanicus, Lindelofia olgae, and Trachelanthus korolkovi. Dokl. Akad. Nauk Uzb. SSR 1962, 19, 29. [Google Scholar]
- Akramov, S.T.; Kiyamitdinova, F.; Yunusov, S.Y. Alkaloids of Solenanthus circinatus, Paracaryum himalayense and Lindelofia pterocarpa. Dokl. Akad. Nauk Uzb. SSR 1964, 21, 28. [Google Scholar]
- Akramov, S.T.; Kiyamitdinova, F.; Yunusov, S.Y. Study of Rindera and Lindelofia. Dokl. Akad. Nauk Uzb. SSR 1965, 22, 35–38. [Google Scholar]
- Pietrosiuk, A.; Syklowska-Baranek, K.; Wiedenfeld, H.; Wolinowska, R.; Furmanowa, M.; Jaroszyk, E. The shikonin derivatives and pyrrolizidine alkaloids in hairy root cultures of Lithospermum canescens (Michx.) Lehm. Plant Cell Rep. 2006, 25, 1052–1058. [Google Scholar] [CrossRef]
- Wiedenfeld, H.; Pietrosiuk, A.; Furmanowa, M.; Roeder, E. Pyrrolizidine alkaloids from Lithospermum canescens Lehm. Z. Naturforsch. 2003, 58c, 173–176. [Google Scholar]
- Roeder, E.; Renge, B. Pyrrolizidine alkaloids from Lithospermum erythrorhizon. Phytochemistry 1990, 29, 690–693. [Google Scholar] [CrossRef]
- Krenn, L.; Wiedenfeld, H.; Roeder, E. Pyrrolizidine alkaloids from Lithospermum officinale. Phytochemistry 1994, 37, 275–277. [Google Scholar] [CrossRef]
- Li, Y.; Stermitz, F.R. Pyrrolizidine alkaloids from Mertensia species of Colorado. J. Nat. Prod. 1988, 51, 1289–1290. [Google Scholar] [CrossRef]
- Ogihara, K.; Miyagi, Y.; Higa, M.; Yogi, S. Pyrrolizidine alkaloids from Messerschmidia argentea. Phytochemistry 1997, 44, 545–547. [Google Scholar] [CrossRef]
- Resch, J.F.; Rosberger, D.F.; Meinwald, J.; Appling, J.W. Biologically active pyrrolizidine alkaloids from the true forget-me-not, Myosotis scorpioides. J. Nat. Prod. 1982, 45, 358–362. [Google Scholar] [CrossRef]
- Roeder, E.; Bourauel, T. Pyrrolizidine alkaloids from Neatostema apulum. Phytochemistry 1992, 31, 3613–3615. [Google Scholar] [CrossRef]
- Roeder, E.; Wiedenfeld, H.; Kroeger, R.; Teppner, H. Pyrrolizidine alkaloids of three taxa of Onosma (Boraginaceae-Lithospermeae). Phyton 1993, 33, 41–49. [Google Scholar]
- Roeder, E.; Wiedenfeld, H.; Kersten, R.; Kröger, R. Determination of open chain pyrrolizidine alkaloids by capillary gas chromatography. Planta Med. 1990, 56, 522. [Google Scholar]
- El-Shazly, A.; Adel-Ghani, A.; Wink, M. Pyrrolizidine Alkaloids from Onosma arenaria Waldst. and Kit (Boraginaceae). Biochem. Syst. Ecol. 2003, 31, 477–485. [Google Scholar] [CrossRef]
- Damianakos, H.; Sotiroudis, G.; Chinou, I. Pyrrolizidine alkaloids from Onosma erecta. J. Nat. Prod. 2013, 76, 1892–1835. [Google Scholar]
- Mellidis, A.S.; Papageorgiou, V.P. Pyrrolizidine alkaloids of the plant Onosma heterophylla. Chem. Chron. 1988, 17, 67–73. [Google Scholar]
- Kretsi, O.; Aligiannis, N.; Skaltsounis, A.L.; Chinou, I.B. Pyrrolizidine alkaloids from Onosma leptantha. Helv. Chim. Acta 2003, 86, 3136–3140. [Google Scholar] [CrossRef]
- Mroczek, T.; Ndjoko, K.; Glowniak, K.; Hostetmann, K. On-line structure characterization of pyrrolizidine alkaloids in Onosma stellulatum and Emilia coccinea by liquid chromatography-ion-trap mass spectrometry. J. Chromatogr. A. 2004, 1056, 91–97. [Google Scholar]
- Haberer, W.; Witte, L.; Hartmann, T.; Dobler, S. Pyrrolizidine alkaloids in Pulmonaria obscura. Planta Med. 2002, 68, 480–482. [Google Scholar] [CrossRef]
- Mandić, B.M.; Simić, M.R.; Vučković, I.M.; Vujisić, L.V.; Novaković, M.M.; Snežana, S.; Trifunović, S.S.; Snežana, D.; Nikolić-Mandić, S.D.; Vele, V.; Tešević, V.V.; Vajs, V.V.; Milosavljević, S.M. Pyrrolizidine alkaloids and fatty acids from the endemic plant species Rindera umbellata and the effect of lindelofine-N-oxide on tubulin polymerization. Molecules 2013, 18, 10694–10706. [Google Scholar] [CrossRef]
- Kurucu, S.; Kartal, M.; Choudary, M.I.; Topcu, G. Pyrrolizidine Alkaloids from Symphytum sylvaticum Boiss. subsp. sepulcrale. (Boiss. & Bal.) Greuter & Burdet var. sepulcrale and Symphytum aintabicum Hub.-Mor. & Wickens. Turk J Chem. 2002, 26, 195–199. [Google Scholar]
- Huizing, H.J.; Gadella, T.W.G.; Kliphuis, E. Chemotaxonomical investigation of the Symphytum officinale polyploidy complex and S. asperum (Boraginaceae): The pyrrolizidine alkaloids. Plant Syst. Evol. 1982, 140, 279–292. [Google Scholar] [CrossRef]
- Roeder, E.; Bourauel, T.; Neuberger, V. Symviridine, a new pyrrolizidine alkaloids from Symphytum species. Phytochemistry 1992, 31, 4041–4042. [Google Scholar] [CrossRef]
- Jaarsma, T.A.; Lohmanns, E.; Gadella, T.W.J.; Malingre, T.M. Chemotaxonomy of the Symphytum officinale agg. (Boraginaceae). Plant Syst. Evol. 1989, 167, 113–127. [Google Scholar] [CrossRef]
- Mel’kumova, Z.V.; Telzhenetskaya, M.V.; Yunusov, S.Y.; Man’ko, I.V. Refinement of the structure of asperumine. Khim. Prir. Soed. 1974, 4, 478–480. [Google Scholar]
- Huizing, H.J.; Malinerg, T.M. Purification and separation of pyrrolizidine alkaloids from Boraginaceae on a polystyrene-divinylbenzene resin. J. Chromatogr. 1979, 176, 274–279. [Google Scholar] [CrossRef]
- Jaarsma, T.A.; Lohmanns, E.; Hendriks, H.; Gadella, T.W.J.; Malingre, T.M. Chemo-and karyotaxonomic studies on some rhizomatous species of the genus Symphytum (Boraginaceae). Plant Syst. Evol. 1990, 169, 31–39. [Google Scholar] [CrossRef]
- Liu, F.; Wan, S.Y.; Jiang, Z.; Li, S.F.; Ong, E.S.; Osorio, J.C. Determination of pyrrolizidine alkaloids in comfrey by liquid chromatography-electrospray ionisation mass spectrometry. Talanta 2009, 15, 916–923. [Google Scholar]
- Furuya, T.; Araki, K. Studies on constituents of crude drugs. 1. Alkaloids of Symphytum officinale Linn. Chem. Pharm. Bull. 1968, 16, 2512–2516. [Google Scholar] [CrossRef]
- Roeder, E.; Wiedenfeld, H.; Stengl, P. 13C-NMR Daten der stereoisomeren Alkaloide aus Symphytum officinale L. Arc. Pharm. (Weinheim) 1982, 315, 87–88. [Google Scholar] [CrossRef]
- Culvenor, C.C.J.; Edgar, J.A.; Frahn, J.L.; Smith, L.W. The alkaloids of Symphytum × uplandicum (Russian comfrey). Aust. J. Chem. 1980, 33, 1105–1113. [Google Scholar] [CrossRef]
- Omar, M.; Defeo, J.; Youngken, H.W. Chemical and toxicity studies of Trichodesma africanum L. J. Nat. Prod. 1983, 46, 153–156. [Google Scholar] [CrossRef]
- O’Kelly, J.; Sargeant, K. Supinine from the seeds of Trichodesma zeylanicum R. Br. J. Chem. Soc. 1961, 484. [Google Scholar]
- Khassanova, M.A.; Abdullaev, U.A.; Telezhenetskaya, M.V.; Yunusov, S.Yu. Structure of uluganine. Khim. Prir. Soedin. 1974, 6, 809–810. [Google Scholar]
- Roeder, E.; Wiedenfeld, H.; Schraut, R. Pyrrolizidine alkaloids from Alkanna tinctoria. Phytochemistry 1984, 23, 2125–2126. [Google Scholar] [CrossRef]
- Culvenor, C.C.J.; Smith, L.W. Aquaternary N-dihydropyrrolizinomethyl derivative of heliotrine from Heliotropium Europaeum. Tetrahedron Lett. 1969, 41, 3603–3606. [Google Scholar] [CrossRef]
- Culvenor, C.C.J.; Johns, S.R.; Smith, L.W. Acetyllasiocarpine, an alkaloids of Heliotropium europaeum. Aust. J. Chem. 1975, 28, 2319–2322. [Google Scholar] [CrossRef]
- Ravi, S.; Ravikumar, R.; Lakshmanan, A.J. Pyrrolizidine alkaloids from Cynoglossum furcatum. Nig. J. Nat. Prod. Med. 2007, 11, 87–89. [Google Scholar]
- Crews, C.; Berthiller, F.; Krska, R. Update on analytical methods for toxic pyrrolizidine alkaloids. Anal. Bioanal. Chem. 2010, 396, 327–338. [Google Scholar] [CrossRef]
- Crews, C. Methods for Analysis of Pyrrolizidine Alkaloids in Natural Products; Ramawat, K.G., Merillon, J.M., Eds.; Springer-Verlag: Berlin & Heidelberg, Germany, 2013; pp. 1049–1068. [Google Scholar]
- Zalkow, L.H.; Asibal, C.F.; Glinski, J.A.; Bonetti, S.J.; Gelbaum, L.T.; van Derveer, D.; Powis, G. Macrocyclic pyrrolizidine alkaloids from Senecio anonymus. Separation of a complex alkaloid extract using droplet counter-current chromatography. J. Nat. Prod. 1988, 51, 690–702. [Google Scholar]
- Bicchi, C.; Caniato, R.; Tabacchi, R.; Tsoupras, G. Capillary gas chromatography/positive and negative ion chemical ionization mass spectrometry on pyrrolizidine alkaloids of Senecio inaequidens using ammonia and hydroxyl ions as the reagent species. J. Nat. Prod. 1989, 52, 32–41. [Google Scholar]
- Stelljes, M.E.; Kelley, R.B.; Molyneux, R.J.; Seiber, J.N. GC-MS determination of pyrrolizidine alkaloids in four Senecio species. J. Nat. Prod. 1991, 54, 759–773. [Google Scholar]
- Witte, L.; Rubiolo, P.; Bicchi, C.; Hartmann, T. Comparative analysis of pyrrolizidine alkaloids from natural sources by gas chromatography-mass spectrometry. Phytochemistry 1993, 32, 187–196. [Google Scholar]
- El-Shazly, A. Pyrrolizidine alkaloid profiles of some Senecio species from Egypt. Z. Naturforsch. 2002, 57c, 429–433. [Google Scholar]
- Asres, K.; Sporer, F.; Wink, M. Patterns of pyrrolizidine alkaloids in 12 Ethiopian Crotalaria species. Biochem. Syst. Ecol. 2004, 32, 915–930. [Google Scholar] [CrossRef]
- Crews, C.; Startin, J.R.; Clarke, P.A. Determination of pyrrolizidine alkaloids in honey from selected sites by solid phase extraction and HPLC-MS. Food Addit. Contam. 1997, 14, 419–428. [Google Scholar] [CrossRef]
- Lin, G.; Zhou, K.Y.; Zhao, X.G.; Wang, Z.T.; But, P.P. Determination of hepatotoxic pyrrolizidine alkaloids by on-line high performance liquid chromatography mass spectrometry with an electrospray interface. Rapid Commun. Mass Spectrom. 1998, 12, 1445–1456. [Google Scholar]
- Zhang, F.; Wang, C.H.; Wang, W.; Chen, L.X.; Ma, H.Y.; Zhang, C.F.; Zhang, M.; Bligh, S.W.; Wang, Z.T. Quantitative analysis by HPLC-MS2 of the pyrrolizidine alkaloid adonifoline in Senecio scandens. Phytochem. Anal. 2008, 19, 25–31. [Google Scholar] [CrossRef]
- Ruan, J.; Li, N.; Xia, Q.; Fu, P.P.; Peng, S.; Ye, Y.; Lin, G. Characteristic ion clusters as determinants for the identification of pyrrolizidine alkaloid N-oxides in pyrrolizidine alkaloid–containing natural products using HPLC-MS analysis. J. Mass Spectrom. 2012, 47, 331–337. [Google Scholar] [CrossRef]
- Neuner-Jehle, N.; Nesvadba, H.; Spiteller, G. Anwendung der Massenspektrometric zur Strukturaufklärung von Alkaloiden, 6. Mitt. Mh. Chem. 1965, 96, 321–338. (In German) [Google Scholar]
- Pedersen, E.; Larsen, E. Mass spectrometry of some pyrrolizidine alkaloids. Org. Mass Spectrom 1970, 4, 249–256. [Google Scholar] [CrossRef]
- Culvenor, C.C.J.; Edgar, J.A.; Frahn, J.L.; Smith, L.W.; Ulubelen, A.; Doganca, S. The structure of anadaloine. Aust. J. Chem. 1975, 28, 173–178. [Google Scholar] [CrossRef]
- Wuilloud, J.C.A.; Gratz, S.R.; Gamble, B.M.; Wolnik, K.A. Simultaneous analysis of hepatotoxic pyrrolizidine alkaloids and N-oxides in comfrey root by LC-ion trap mass spectrometry. Analyst 2004, 129, 150–156. [Google Scholar] [CrossRef]
- Roeder, E.; Wiedenfeld, H.; Hoeing, A. Pyrrolizidinalkaloide aus Senecio aureus. Planta Med. 1983, 49, 57–59. [Google Scholar]
- Flores, A.S.; de Azevedo Tozzi, A.M.G.; Trigo, J.R. Pyrrolizidine alkaloid profiles in Crotalaria species from Brazil: Chemotaxonomic significance. Biochem. Syst. Ecol. 2009, 37, 459–469. [Google Scholar]
- Mead, E.W.; Looker, M.; Gardner, D.R.; Stermitz, F.R. Pyrrolizidine alkaloids of Liatris punctata and its root parasite, Castilleja integra. Phytochemistry 1992, 31, 3255–3257. [Google Scholar] [CrossRef]
- Tei, A. Identifizierung und Strukturaufklärung von Alkaloiden und anderen Sekundärstoffen mit Hilfe von GC-MS und Kernresonanzspektroskopie. Ph.D. Thesis, Ruprecht-Karls-Universität Heidelberg, Heidelberg, Germany, 2000. [Google Scholar]
- Stermiz, F.R.; L’Empereur, K.M. Identity of “subulasine N-oxide” with 1β,2β-epoxy-1α-hydroxymethyl-8α-pyrrolizidine. Tetrahedron Lett. 1988, 29, 4943–4944. [Google Scholar] [CrossRef]
- Griffin, C.T.; Danaher, M.; Elliott, C.T.; Kennedy, D.G.; Furey, A. Detection of pyrrolizidine alkaloids in commercial honey using liquid chromatography–ion trap mass spectrometry. Food Chem. 2013, 136, 1577–1583. [Google Scholar] [CrossRef]
- Martinello, M.; Cristofoli, C.; Gallina, A.; Mutinelli, F. Easy and rapid method for the quantitative determination of pyrrolizidine alkaloids in honey by ultra performance liquid chromatography-mass spectrometry: An evaluation in commercial honey. Food Control 2014, 37, 146–152. [Google Scholar] [CrossRef]
- Logie, C.G.; Grue, M.R.; Liddell, J.R. Proton NMR spectroscopy of pyrrolizidine alkaloids. Phytochemistry 1994, 37, 43–109. [Google Scholar] [CrossRef]
- Roeder, E. Carbon-13 NMR spectroscopy of pyrrolizidine alkaloids. Phytochemistry 1990, 29, 11–29. [Google Scholar] [CrossRef]
- Bober, M.A.; Milco, L.A.; Miller, R.B.; Mount, M.; Wicks, B.; Kurth, M.J. A competitive enzyme-linked immunosorbent assay (ELISA), to detect retronecine and monocrotaline in vitro. Toxicon. 2004, 27, 1059–1064. [Google Scholar]
- Roeder, E.; Pflueger, T. Analysis of pyrrolizidine alkaloids: a competitive enzyme-linked immunoassay (ELISA) for the quantitative determination of some toxic pyrrolizidine alkaloids. Nat. Toxins. 1995, 3, 305–309. [Google Scholar] [CrossRef]
- Lee, S.T.; Schoch, T.K.; Stegelmeier, B.L.; Gardner, D.R.; Than, K.A.; Molyneus, R.J. Development of enzyme-linked immunosorbent assays for the hepatotoxic riddelliine and reddelliine N-oxide. J. Agric. Food Chem. 2001, 49, 4144–4151. [Google Scholar] [CrossRef]
- Cavallaro, V.; Than, K.A.; Colegate, S.M.; Edgar, J.A. An indirect competitive ELISA for pyrrolizidine alkaloids of Heliotropium europaeum. In Poisonous Plants and Related Toxins; Acamovic, T., Stewart, C.S., Pennycott, T.W., Eds.; CABI publishing: Wallingford, UK, 2004; pp. 114–119. [Google Scholar]
- Lee, S.T.; Knill, A.; Michalewicz, A.; Stevens, V.; Colegate, S.M. Heliotropium europaeum alkaloids: A quaternary pyrrolizidine alkaloids approach to elisa development. In Poisonous Plant Global Research and Solution; United States Department of Agriculture: Washington, DC, USA, 2007; Chapter 81; pp. 476–480. [Google Scholar]
- Von Borstel, K.; Hartmann, T. Selective uptake of pyrrolizidine N-oxides by cell suspension cultures from pyrrolizidine alkaloid producing plants. Plant Cell Rep. 1986, 5, 39–42. [Google Scholar] [CrossRef]
- Van Dam, N.M.; Verpoorte, R.; Meijden, E.V.D. Extreme differences in pyrrolizidine alkaloid levels between leaves of Cynoglossum officinale. Phytochemistry 1994, 37, 1013–1016. [Google Scholar] [CrossRef]
- Frölich, C.; Ober, D.; Hartmann, T. Tissue distribution, core biosynthesis and diversification of pyrrolizidine alkaloids of the lycopsamine type in three Boraginaceae species. Phytochemistry 2007, 68, 1026–1037. [Google Scholar] [CrossRef]
- Abd El-Mawla, A.M. Effect of cretin elicitors on production of pyrrolizidine alkaloids in hairy root cultures of Echium rauwolfii. Pharmazie 2010, 65, 224–226. [Google Scholar]
- Marin-Loaiza, J.C.; Ernst, L.; Beuerle, T.; THeuring, C.; Cespedes, C.L.; Hartmann, T. Pyrrolizidine alkaloids of the endemic Mexican genus Pittocaulon and assignment of stereoisomeric 1,2-saturated necine bases. Phytochemistry 2008, 69, 154–167. [Google Scholar] [CrossRef]
- Okusa, P.N.; Penge, O.; Devleeschouwer, M.; Duez, P. Direct and indirect antimicrobial and antioxidant activity of Cordia dilletii De Wild (Boraginaceae). J. Ethanopharmacol. 2007, 112, 476–481. [Google Scholar] [CrossRef]
- Joosten, L.; van Veen, J.A. Defensive properties of pyrrolizidine alkaloids against microorganisms. Phytochem. Rev. 2011, 10, 127–136. [Google Scholar] [CrossRef]
- Wink, M. Allelochemical properties and raison d’être of alkaloids. In The Alkaloids; Cordell, G., Ed.; Academic Press: New York, NY, USA, 1993; vol. 43, pp. 1–118. [Google Scholar]
- Boppre, M. Insects pharmacophagously utilizing defensive plant chemicals (pyrrolizidine alkaloids). Naturwissenschaften 1986, 73, 17–26. [Google Scholar] [CrossRef]
- Boppre, M. Lepidoptera and pyrrolizidine alkaloids. Exemplification of complexity in chemical ecology. J. Chem. Ecol. 1990, 16, 165–185. [Google Scholar] [CrossRef]
- Von Nickisch-Rosenegk, E.; Wink, M. Sequestration of pyrrolizidine alkaloids in several arctiid moths (Lepidoptera: Arctiidae). J. Chem. Ecol. 1993, 19, 1889–1903. [Google Scholar] [CrossRef]
- Dobler, S.; Haberer, W.; Witte, L.; Hartmann, T. Selective sequestration of pyrrolizidine alkaloids from diverse host plants by Longitarsus flea beetles. J. Chem. Ecol. 2000, 5, 1281–1298. [Google Scholar]
- Honda, Y.; Honda, K.; Omura, H. Major components in the hairpencil secretion of a butterfly, Euploea mulciber (Lepidoptera, Danaidae): Their origin and male behavioral responses to pyrrolizidine alkaloids. J. Insect Physiol. 2006, 52, 1043–1053. [Google Scholar] [CrossRef]
- Beuerle, T.; Theuring, C.; Klewer, N.; Schulz, S.; Hartmann, T. Absolute configuration of the creatonotines and callimorphines, two classes of arctiid-specific pyrrolizidine alkaloids. Insect Biochem. Mol. Biol. 2007, 37, 80–89. [Google Scholar] [CrossRef]
- Macel, M. Attract and deter: A dual role for pyrrolizidine alkaloids in plant-insect interaction. Phytochem. Rev. 2011, 10, 75–82. [Google Scholar] [CrossRef]
- Denmark, S.E.; Hurd, A.R. Synthesis of (+)-casuarine. J. Org. Chem. 2000, 65, 2875–2886. [Google Scholar] [CrossRef]
- Asano, N.; Ikeda, K.; Kasahara, M.; Arai, Y.; Kizu, H. Glycosidase-inhibiting pyrrolidines and pyrrolizidines with long side chain in Scilla peruviana. J. Nat. Prod. 2004, 67, 846–850. [Google Scholar] [CrossRef]
- Garcia-Moreno, M.I.; Rodriguez-Lucena, D.; Mellet, C.O.; Garcia Fernandez, J.M. Pseudoamide-type pyrrolidine and pyrrolizidine glycomimetics and their inhibitory activities against glycosidases. J. Org. Chem. 2004, 69, 3578–3581. [Google Scholar] [CrossRef]
- Nibret, E.; Sporer, F.; Asres, K.; Wink, M. Antitrypanosomal and cytotoxic activity of pyrrolizidine alkaloid-producing plants of Ethiopia. J. Pharm. Pharmacol. 2009, 61, 801–808. [Google Scholar] [CrossRef]
- Singh, B.; Sahu, P.M.; Singh, S. Antimicrobial activity of alkaloids from Heliotropium subulatum. Fitoterapia 2002, 73, 153–155. [Google Scholar] [CrossRef]
- Wink, M. Evolution of secondary metabolites in legumes (Fabaceae). S. Afr. J. Bot. 2013, 89, 164–175. [Google Scholar] [CrossRef]
- Pfister, J.A.; Molyneux, R.J.; Baker, D.C. Pyrrolizidine alkaloid content of hounds tongue (Cynoglossum officinale L.). J. Range Managem. 1992, 45, 254–256. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
El-Shazly, A.; Wink, M. Diversity of Pyrrolizidine Alkaloids in the Boraginaceae Structures, Distribution, and Biological Properties. Diversity 2014, 6, 188-282. https://doi.org/10.3390/d6020188
El-Shazly A, Wink M. Diversity of Pyrrolizidine Alkaloids in the Boraginaceae Structures, Distribution, and Biological Properties. Diversity. 2014; 6(2):188-282. https://doi.org/10.3390/d6020188
Chicago/Turabian StyleEl-Shazly, Assem, and Michael Wink. 2014. "Diversity of Pyrrolizidine Alkaloids in the Boraginaceae Structures, Distribution, and Biological Properties" Diversity 6, no. 2: 188-282. https://doi.org/10.3390/d6020188
APA StyleEl-Shazly, A., & Wink, M. (2014). Diversity of Pyrrolizidine Alkaloids in the Boraginaceae Structures, Distribution, and Biological Properties. Diversity, 6(2), 188-282. https://doi.org/10.3390/d6020188






