Origin and Domestication of Native Amazonian Crops
Abstract
:1. Introduction
2. The Crops
2.1. Manioc
2.2. Cacao
2.3. Peach Palm
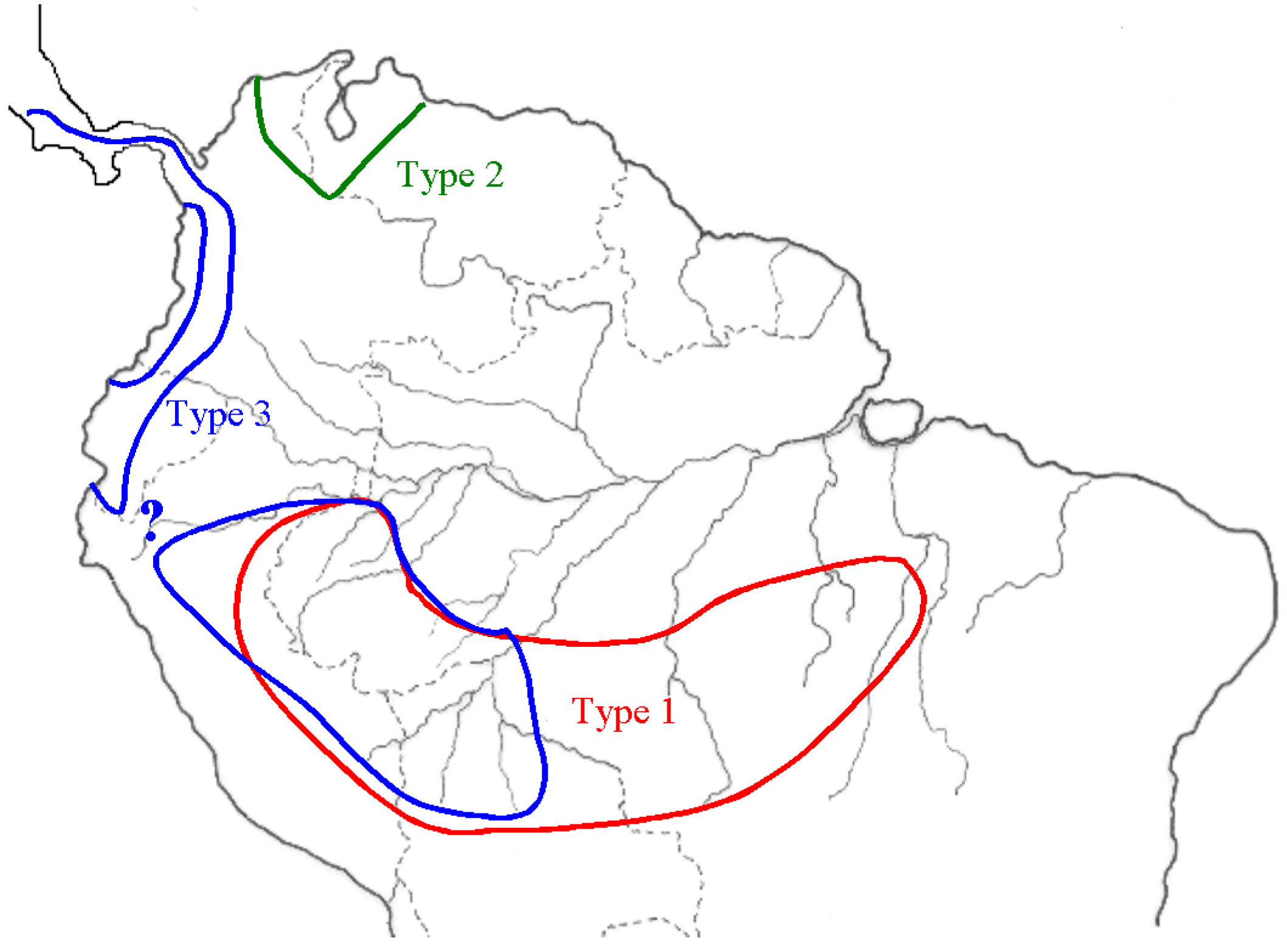
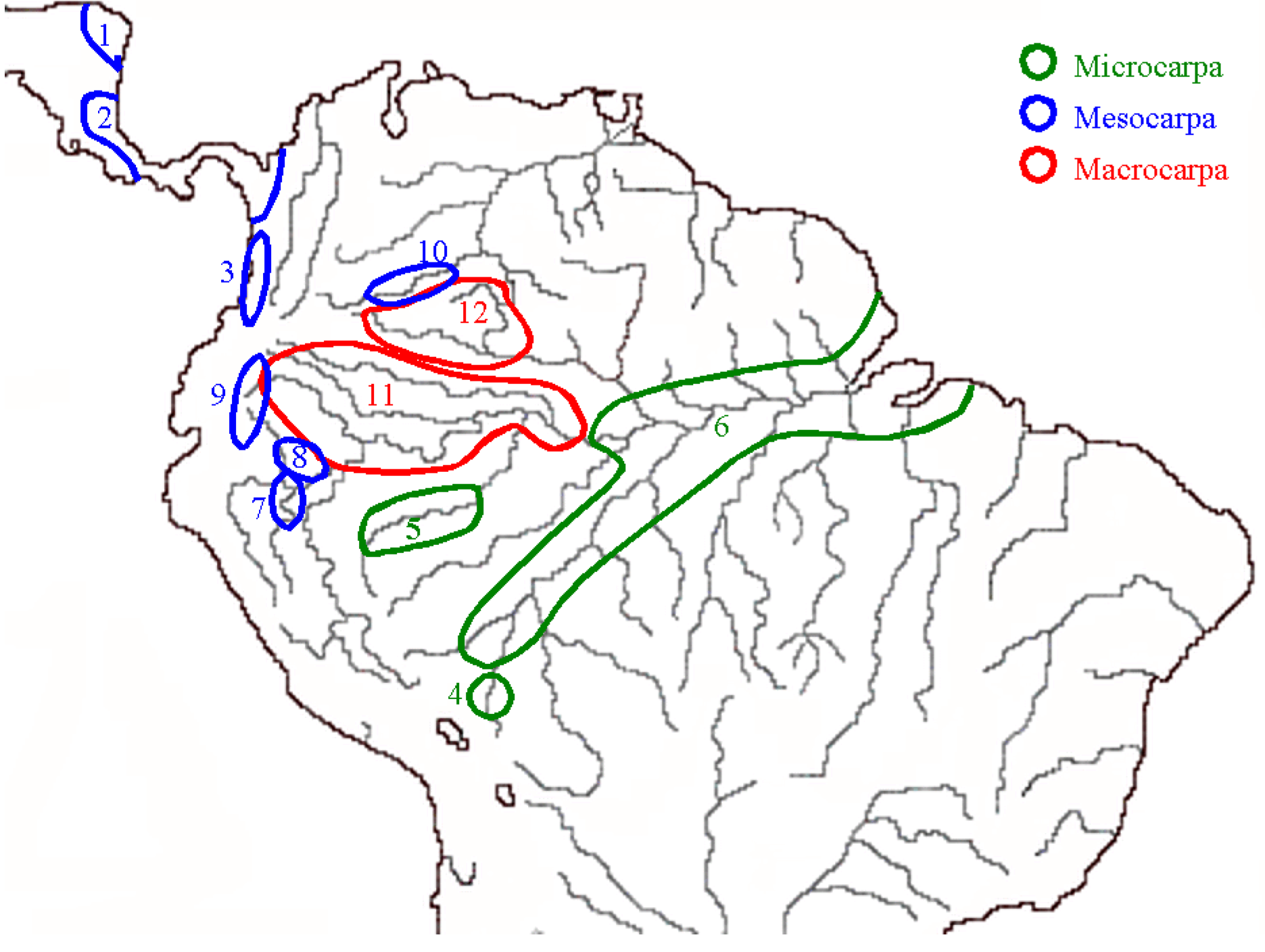
2.4. Capsicum Peppers
2.5. Pineapple
2.6. Inga
2.7. Guaraná
2.8. Brazil Nut
2.9. Cupuassu
3. Patterns of Diversity
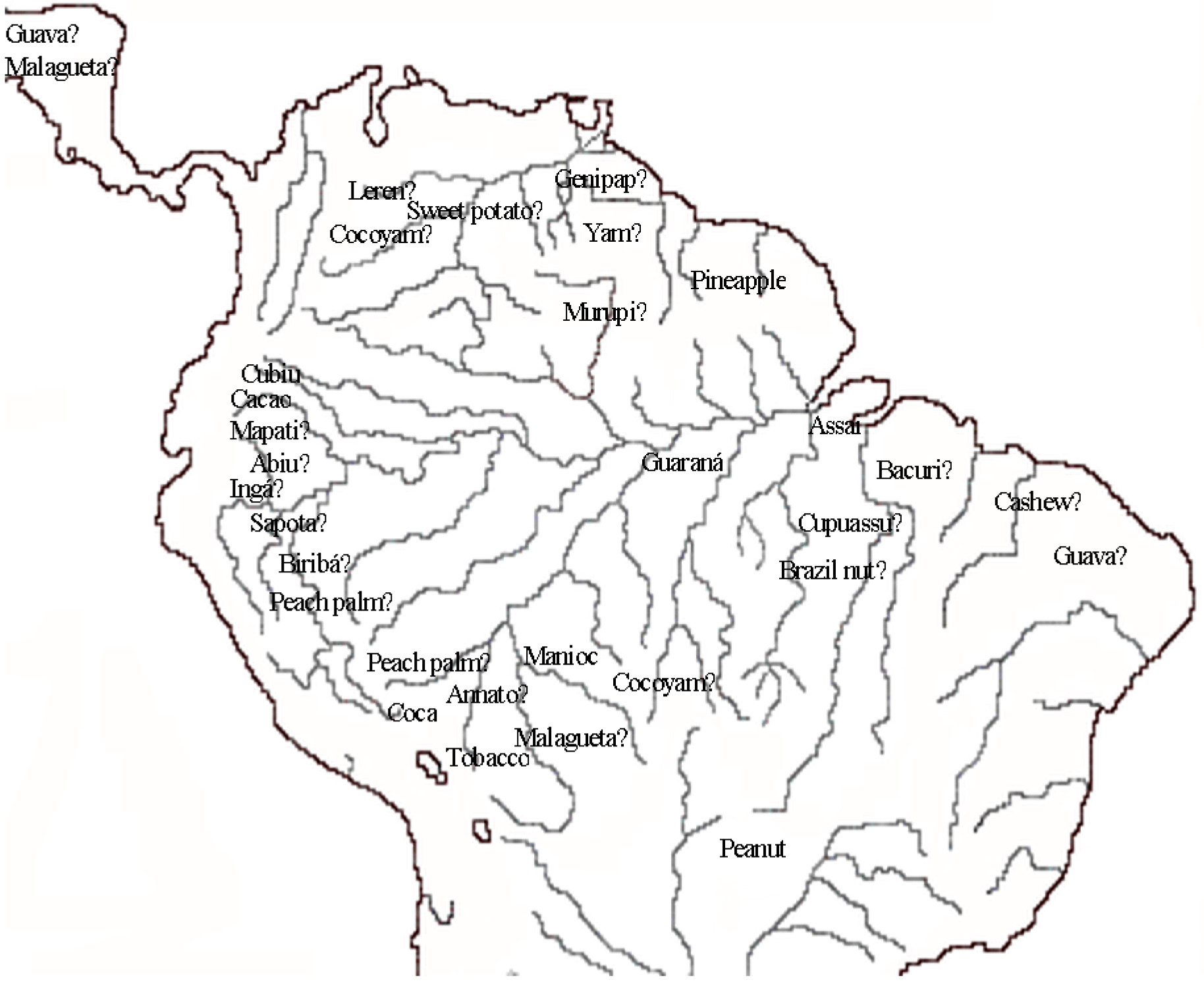
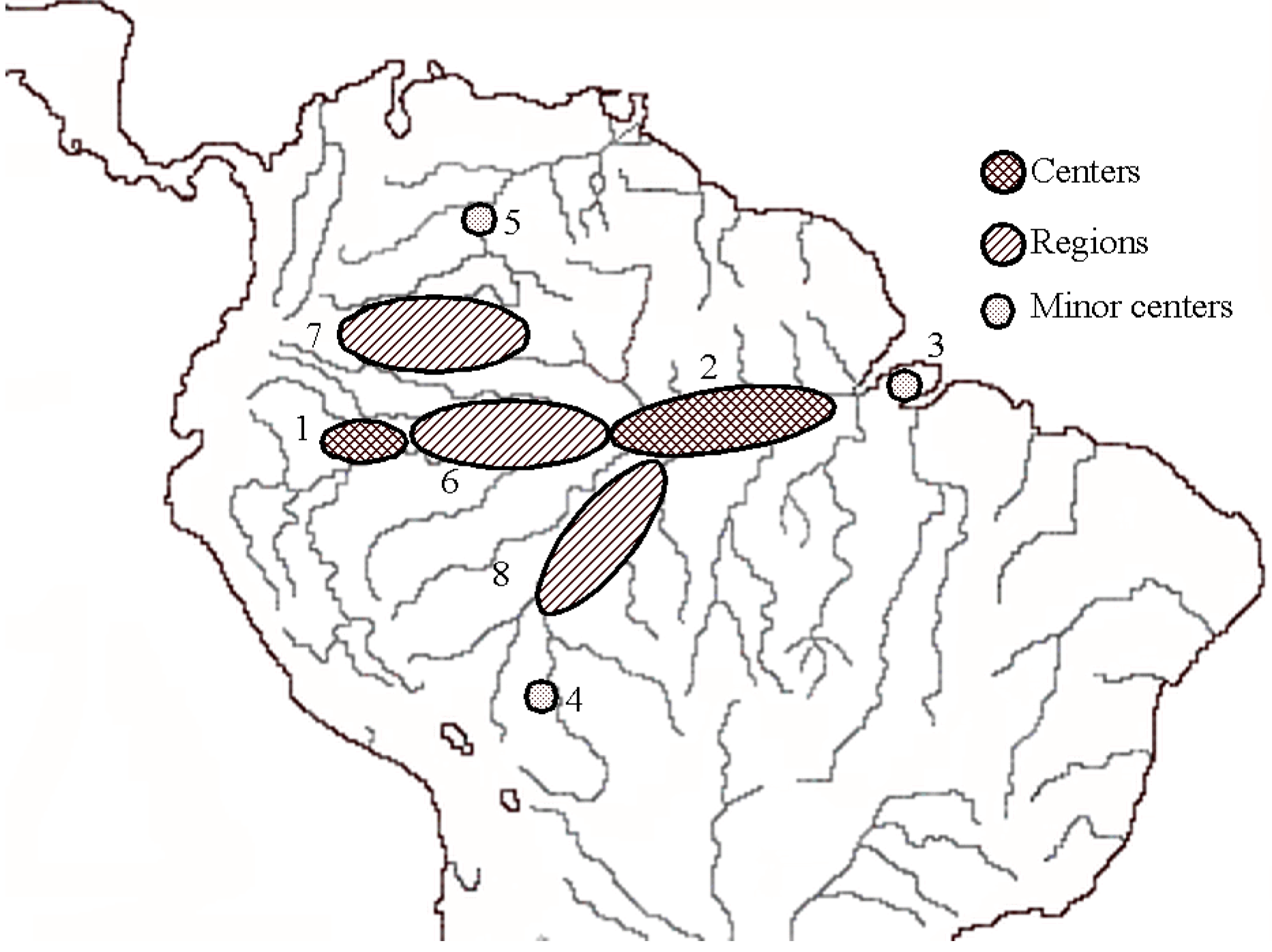
4. Conclusions
Acknowledgements
References and Notes
- Clement, C.R. 1492 and the loss of Amazonian crop genetic resources. I. The relation between domestication and human population decline. Econ. Bot. 1999, 53, 188–202. [Google Scholar] [CrossRef]
- Schaal, B.A.; Olsen, K.M.; Carvalho, L.J.C.B. Evolution, domestication, and agrobiodiversity in the tropical crop cassava. In Darwin’s Harvest: New Approaches to the Origins, Evolution, and Conservation of Crops; Motley, T.J., Zerega, N., Cross, H., Eds.; Columbia University Press: New York, NY, USA, 2006; pp. 269–284. [Google Scholar]
- Hancock, J.F. Plant Evolution and the Origin of Crop Species, 2nd ed.; CABI Publishing: Wallingford, Oxon, UK, 2004; p. 313. [Google Scholar]
- Clement, C.R. Domestication of the pejibaye palm (Bactris gasipaes): past and present. In The Palm—Tree of Life. Biology, Utilization and Conservation; Balick, M.J., Ed.; The New York Botanical Garden: Bronx, NY, USA, 1988; Advances in Economic Botany; Volume 6, pp. 155–174. [Google Scholar]
- Clement, C.R. A center of crop genetic diversity in western Amazonia. BioScience 1989, 39, 624–631. [Google Scholar] [CrossRef]
- Pearsall, D.M. The origins of plant cultivation in South America. In The Origins of Agriculture: An International Perspective; Cowan, C.W., Watson, P.J., Eds.; Smithsonian Institution Press: Washington, DC, USA, 1992; pp. 173–206. [Google Scholar]
- Clement, C.R. 1492 and the loss of Amazonian crop genetic resources. II. Crop biogeography at contact. Econ. Bot. 1999, 53, 203–216. [Google Scholar] [CrossRef]
- Balée, W. The culture of Amazonian forests. In Resource Management in Amazonia: Indigenous and Folk Strategies; Posey, D.A., Balée, W., Eds.; The New York Botanical Garden: Bronx, NY, USA, 1989; Advances in Economic Botany; Volume 7, pp. 1–21. [Google Scholar]
- Avise, J.C. Phylogeography: The History and Formation of Species; Harvard University Press: Cambridge, MA, USA, 2000; p. 447. [Google Scholar]
- Pickersgill, B. Domestication of plants in the Americas: insights from Mendelian and molecular genetics. Ann. Bot. 2007, 100, 925–940. [Google Scholar] [CrossRef]
- Emshwiller, E. Genetic data and plant domestication. In Documenting Domestication: New Genetic and Archaeological Paradigms; Zeder, M.A., Bradley, D.G., Emshwiller, E., Smith, B.D., Eds.; University of California Press: Berkeley, CA, USA, 2006; pp. 99–122. [Google Scholar]
- Zeder, M.A. Central questions in the domestication of plants and animals. Evol. Anthropol. 2006, 15, 105–117. [Google Scholar] [CrossRef]
- Lebot, V. Tropical Root and Tuber Crops: Cassava, Sweet Potato, Yams and Aroids; CAB International: Oxford, UK, 2009; Crop Production Science in Horticulture Series; Volume 17, p. 413. [Google Scholar]
- Rogers, D.J.; Appan, S.G. Manihot and Manihotoides (Euphorbiaceae): A Computer-Assisted Study; Hafner Press: New York, NY, USA, 1973. [Google Scholar]
- Reichel-Dolmatoff, G. Arqueologia de Colombia: Un Texto Introductorio; Fundación Segunda Expedición Botánica: Bogotá, Colombia, 1986. [Google Scholar]
- Allem, A.C. The origin and taxonomy of cassava. In Cassava: Biology, Production and Utilization; Hillocks, R.J., Thresh, J.M., Bellotti, A.C., Eds.; CAB International: Oxford, UK, 2001; pp. 1–16. [Google Scholar]
- Fregene, M.A.; Vargas, J.; Ikea, J.; Angel, F.; Tohme, J.; Asiedu, R.A.; Akoroda, M.O.; Roca, W.M. Variability of chloroplast DNA and nuclear ribosomal DNA in cassava (Manihot esculenta Crantz) and its wild relatives. Theor. Appl. Genet. 1994, 89, 719–727. [Google Scholar]
- Roa, A.C.; Maya, M.M.; Duque, M.C.; Tohme, A.C.; Allem, A.C.; Bonierbale, M.W. AFLP analysis of relationships among cassava and other Manihot species. Theor. Appl. Genet. 1997, 95, 741–750. [Google Scholar] [CrossRef]
- Allem, A.C. The origin of Manihot esculenta Crantz (Euphorbiaceae). Genet. Resour. Crop Evol. 1994, 41, 133–150. [Google Scholar] [CrossRef]
- Olsen, K.M.; Schaal, B.A. Evidence on the origin of cassava: phylogeography of Manihot esculenta. Proc. Natl. Acad. Sci. USA 1999, 96, 5586–5591. [Google Scholar] [CrossRef]
- Olsen, K.M. SNPs, SSRs and inferences on cassava’s origin. Plant Mol. Biol. 2004, 56, 517–526. [Google Scholar] [CrossRef]
- Léotard, G.; Duputié, A.; Kjellberg, F.; Douzery, E.J.P.; Debain, C.; Granville, J.J.; McKey, D. Phylogeography and the origin of cassava: new insights from the northern rim of the Amazonian basin. Mol. Phylogenet. Evol. 2009, 53, 329–334. [Google Scholar] [CrossRef]
- Mühlen, G.S.; Martins, P.S.; Ando, A. Variabilidade genética de etnovariedades de mandioca, avaliada por marcadores de DNA. Sci. Agric. 2000, 57, 319–328. [Google Scholar]
- Elias, M.; Mühlen, G.S.; McKey, D.; Roa, A.C.; Tohme, J. Genetic diversity of traditional South American landraces of cassava (Manihot esculenta Crantz): an analysis using microsatellites. Econ. Bot. 2004, 58, 242–256. [Google Scholar] [CrossRef]
- McKey, D.; Beckerman, S. Chemical ecology, plant evolution and traditional manioc cultivation systems. In Tropical Forests, People and Food: Biocultural Interactions and Applications to Development; Hladik, C.M., Hladick, A., Linares, O.F., Pagezy, H., Semple, A., Hadley, M., Eds.; Parthenon: Carnforth, UK, and UNESCO: Paris, France; 1993; pp. 83–112. [Google Scholar]
- Wilson, W.M.; Dufour, D.L. Why “bitter” cassava? Productivity of “bitter” and “sweet” cassava in a Tukanoan Indian settlement in the Northwest Amazon. Econ. Bot. 2002, 56, 49–57. [Google Scholar] [CrossRef]
- Wilson, W.M. Cassava (Manihot esculenta Crantz), cyanogenic potential, and predation in Northwestern Amazonian: the Tukanoan perspective. Hum. Ecol. 2003, 31, 403–417. [Google Scholar] [CrossRef]
- Wilson, W.M.; Dufour, D.L. Ethnobotanical evidence for cultivar selection among the Tukanoans: Manioc (Manihot esculenta Crantz) in the Northwest Amazon. Cult. Agric. 2006, 28, 122–130. [Google Scholar] [CrossRef]
- Martins, P.S. Dinâmica evolutiva em roças de caboclos amazônicos. In Diversidade Biológica e Cultural da Amazônia; Vieira, I.C.G., Silva, J.M.C., Oren, D.C., D'Incao, M.A., Eds.; Museu Paraense Emílio Goeldi: Belém, Brazil, 2001; pp. 369–384. [Google Scholar]
- Pujol, B.; Renoux, F.; Elias, M.; Rival, L.; McKey, D. The unappreciated ecology of landrace populations: conservation consequences of soil seedbanks in cassava. Biol. Conserv. 2007, 136, 541–551. [Google Scholar] [CrossRef]
- Duputié, A.; Massol, F.; David, P.; Haxaire, C.; McKey, D. Traditional Amerindian cultivators combine directional and ideotypic selection for sustainable management of cassava genetic diversity. J. Evol. Biol. 2009, 22, 1317–1325. [Google Scholar]
- Rival, L.; McKey, D. Domestication and diversity in manioc (Manihot esculenta Crantz ssp. esculenta, Euphorbiaceae). Curr. Anthropol. 2008, 49, 1119–1128. [Google Scholar] [CrossRef]
- Fraser, J.A.; Clement, C.R. Dark Earths and manioc cultivation in Central Amazonia: a window on pre-Columbian agricultural systems? Bol. Mus. Paraense Emílio Goeldi Ciênc. Hum. 2008, 3, 175–194. [Google Scholar] [CrossRef]
- Peroni, N. Taxonomia Folk e Diversidade Intra-específica de Mandioca (Manihot esculenta Crantz) em Roças de Agricultura Tradicional em Áreas de Mata Atlântica do Sul do Estado de São Paulo. Master’s Thesis, Escola Superior de Agricultura "Luiz de Queiroz", Universidade de São Paulo, Piracicaba, Brazil, 1998. [Google Scholar]
- Elias, M.; Rival, L.; McKey, D. Perception and management of cassava (Manihot esculenta Crantz) diversity among Makushi Amerindians of Guyana (South America). J. Ethnobiol. 2000, 20, 239–265. [Google Scholar]
- Elias, M.; Penet, L.; Vindry, P.; McKey, D.; Panaud, O.; Robert, T. Unmanaged sexual reproduction and the dynamics of genetic diversity of a vegetatively propagated crop plant, cassava (Manihot esculenta Crantz), in a traditional farming system. Mol. Ecol. 2001, 10, 1895–1907. [Google Scholar] [CrossRef]
- Sambatti, J.B.M.; Martins, P.S.; Ando, A. Folk taxonomy and evolutionary dynamics of cassava: A case study in Ubatuba, Brazil. Econ. Bot. 2001, 55, 93–105. [Google Scholar] [CrossRef]
- Peroni, N.; Kageyama, P.; Begossi, A. Molecular differentiation, diversity, and folk classification of "sweet"' and "bitter'' cassava (Manihot esculenta) in Caiçara and Caboclo management systems (Brazil). Genet. Resour. Crop Evol. 2007, 54, 1333–1349. [Google Scholar] [CrossRef]
- Peroni, N. Ecologia e Genética da Mandioca na Agricultura Itinerante do Litoral Sul Paulista: Uma Análise Espacial e Temporal. Doctoral Thesis, 2004; p. 246. [Google Scholar]
- Dias, L.A.S.; Resende, M.D.V. Domesticação e melhoramento de cacau. In Domesticação e Melhoramento de Plantas: Espécies Amazônicas; Borém, A., Lopes, M.T.G., Clement, C.R., Eds.; Editora da Univ. Fed. Viçosa: Viçosa, Minas Gerais, Brazil, 2009; pp. 251–274. [Google Scholar]
- Patiño, V.M. Historia y Dispersión de los Frutales Nativos del Neotrópico; Centro Internacional de Agricultura Tropical: Cali, Colombia, 2002; p. 655. [Google Scholar]
- Brown, C.H. Development of agriculture in prehistoric Mesoamerica: The linguistic evidence. In Pre-Columbian Foodways; Staller, J.E., Carrasco, M., Eds.; Springer: Berlin, Germany, 2009; pp. 71–107. [Google Scholar]
- Cuatrecasas, J. Cacao and its allies: A taxonomic revision of the genus Theobroma. Contrib. US Natl. Herb. 1964, 35, 379–614. [Google Scholar]
- Motamayor, J.C.; Lanaud, C. Molecular analysis of the origin and domestication of Theobroma cacao L. In Managing Plant Genetic Diversity; Engels, J.M.M., Ramanatha Rao, V., Brown, A.H.D., Jackson, M.T., Eds.; IPGRI: Rome, Italy, 2002; pp. 77–87. [Google Scholar]
- Cheesman, E. Notes on the nomenclature, classification and possible relationships of cocoa populations. Trop. Agric. 1944, 21, 144–159. [Google Scholar]
- Motamayor, J.C.; Lachenaud, P.; Silva e Mota, J.W.; Loor, R.; Kuhn, D.N.; Brown, J.S.; Schnell, R.J. Geographic and genetic population differentiation of the Amazonian chocolate tree (Theobroma cacao L.). PLoS ONE 2008, 3(10), e3311. [Google Scholar] [CrossRef]
- Pritchard, J.K.; Stephens, M.; Donnelly, P. Inference of population structure using multilocus genotype data. Genetics 2000, 155, 945–959. [Google Scholar]
- Huber, J. Notas sobre a patria e distribuição geographica das arvores fructiferas do Pará. Bol. Mus. Paraense Emílio Goeldi 1904, 4, 375–406. [Google Scholar]
- Barrau, J. Sur l’origine du cacaoyer, Theobroma cacao Linné, Sterculiacées. J. Agric. Trad. Bot. Appl. 1979, 26, 171–180. [Google Scholar]
- Henderson, J.S.; Joyce, R.A.; Hall, G.R.; Hurst, W.J.; McGovern, P.E. Chemical and archaeological evidence for the earliest cacao beverages. Proc. Natl. Acad. Sci. USA 2007, 104, 18937–18940. [Google Scholar]
- Rindos, D. The Origins of Agriculture: An Evolutionary Perspective; Academic Press: San Diego, California, USA, 1984; p. 325. [Google Scholar]
- Anderson, E. Plants, Man and Life; Dover: Mineola, NY, USA, 2005; p. 251. [Google Scholar]
- Clement, C.R.; Rival, L.; Cole, D.M. Domestication of peach palm (Bactris gasipaes Kunth): The roles of human mobility and migration. In Shifting Spaces, Changing Times: Mobility, Migration and Displacement in Indigenous Lowland South America; Alexiades, M.N., Ed.; Berghahn Books: Oxford, UK, 2009; pp. 117–140. [Google Scholar]
- Mora Urpí, J.; Weber, J.C.; Clement, C.R. Peach palm. Bactris gasipaes Kunth; Institute of Plant Genetics and Crop Plant Research—IPK: Gatersleben, Germany, International Plant Genetic Resources Institute—IPGRI: Rome, Italy; 1997; Promoting the conservation and use of underutilized and neglected crops, Volume 20; p. 83. [Google Scholar]
- Henderson, A. Bactris (Palmae). Flora Neotropica 2000, 79, 1–181. [Google Scholar]
- Clement, C.R.; Santos, R.P.; Desmouliere, S.J.M.; Ferreira, E.J.L.; Farias Neto, J.T. 2009. Ecological adaptation of wild peach palm, its in situ conservation and deforestation-mediated extinction in southern Brazilian Amazonia. PLoS ONE 2009, 4, e4564. [Google Scholar] [CrossRef]
- Clement, C.R. Pejibaye (Bactris gasipaes). In Evolution of Crop Plants, 2nd ed.; Smartt, J., Simmonds, N.W., Eds.; Longman: London, UK, 1995; pp. 383–388. [Google Scholar]
- Ferreira, E. The phylogeny of pupunha (Bactris gasipaes Kunth, Palmae) and allied species. In Evolution, Variation, and Classification of palms; Henderson, A., Borchsenius, F., Eds.; The New York Botanical Garden: New York, NY, USA, 1999; Memoirs of the New York Botanical Garden; Volume 83, pp. 225–236. [Google Scholar]
- Rodrigues, D.P.; Astolfi Filho, S.; Clement, C.R. Molecular marker-mediated validation of morphologically defined landraces of pejibaye (Bactris gasipaes) and their phylogenetic relationships. Genet. Resour. Crop Evol. 2004, 51, 871–882. [Google Scholar]
- Morcote-Rios, G.; Bernal, R. Remains of palms (Palmae) at archaeological sites in the New World: a review. Bot. Rev. 2001, 67, 309–350. [Google Scholar] [CrossRef]
- Mora Urpí, J. Origen y domesticación. In Palmito de Pejibaye (Bactris gasipaes Kunth): Su Cultivo e Industrialización; Mora-Urpí, J., Gainza E., J., Eds.; Editorial de la Universidad de Costa Rica: San José, Costa Rica, 1999; pp. 17–24. [Google Scholar]
- Hernández-Ugalde, J.A.; Mora Urpí, J.; Rocha Nuñez, O. Diversidad genética y relaciones de parentesco de las poblaciones silvestres y cultivadas de pejibaye (Bactris gasipaes, Palmae), utilizando marcadores microsatelites. Rev. Biol. Trop. 2008, 56, 217–245. [Google Scholar]
- Couvreur, T.L.P.; Billotte, N.; Risterucci, A.M.; Lara, C.; Vigouroux, Y.; Ludeña, B.; Pham, J. L.; Pintaud, J.C. Close genetic proximity between cultivated and wild Bactris gasipaes Kunth revealed by microsatellite markers in Western Ecuador. Genet. Resour. Crop Evol. 2006, 53, 1361–1373. [Google Scholar] [CrossRef]
- Mora-Urpí, J.; Clement, C.R. Races and populations of peach palm found in the Amazon basin. In Final Report (revised): Peach Palm (Bactris gasipaes H.B.K.) Germplasm Bank; Clement, C.R., Coradin, L., Eds.; Instituto Nacional de Pesquisas da Amazônia/Centro Nacional de Recursos Genéticos: Manaus, Brazil, 1988; pp. 78–94. [Google Scholar]
- Rojas Vargas, S.; Ramírez, P.; Mora-Urpí, J. 1999. Polimorfismo isoenzimático en cuatro razas y un híbrido de Bactris gasipaes (Palmae). Rev. Biol. Trop. 1999, 47, 755–761. [Google Scholar]
- Nei, M. Estimation of average heterozygosity and genetic distance from small numbers of individuals. Genetics 1978, 89, 583–590. [Google Scholar]
- Silva, C.C. Análise molecular e validação de raças primitivas de pupunha (Bactris gasipaes) por meio de marcadores RAPD. Masters Thesis, Universidade Federal de São Carlos/Universidade Federal do Amazonas, Manaus, Brazil, 2004. [Google Scholar]
- Cristo-Araújo, M. Uma coleção nuclear de pupunha na Amazônia brasileira. Master’s Thesis, Universidade Federal do Amazonas, Manaus, Amazonas, Brazil, 2008; p. 91. [Google Scholar]
- Martínez, A.K.; Gaitán-Solis, E.; Duque, M.C.; Bernal, R.; Tohme, J. Primer Note: Microsatellite loci in Bactris gasipaes (Arecaceae): Their isolation and characterization. Mol. Ecol. Notes 2002, 2, 408–410. [Google Scholar] [CrossRef]
- Billotte, N.; Couvreur, T.; Marseillac, N.; Brottier, P.; Perthuis, B.; Vallejo, M.; Noyer, J.-L.; Jacquemoud-Collet, J.-P.; Risterucci, A.-M.; Pintaud, J.-C. A new set of microsatellite markers for the peach palm (Bactris gasipaes Kunth): Characterization and across-taxa utility within the tribe Cocoeae. Mol. Ecol. Notes 2004, 4, 580–582. [Google Scholar] [CrossRef]
- Rodrigues, D.P.; Vinson, C.; Ciampi, A.Y.; Farias, I.P.; Lemes, M.R.; Astolfi-Filho, S.; Clement, C.R. Novel microsatellite markers for Bactris gasipaes (Palmae). Mol. Ecol. Notes 2004, 4, 575–576. [Google Scholar] [CrossRef]
- Hernández-Ugalde, J.A. Descripción de la diversidad y estructura genética de las poblaciones silvestres y cultivadas de pejibaye (Bactris gasipaes Kunth), utilizando marcadores microsatélites. Master’s Thesis, Universidad de Costa Rica, San José, Costa Rica, 2005. [Google Scholar]
- Reis, V.M. Relações Genéticas entre Raças e Populações da Coleção Nuclear de Pupunha (Bactris gasipaes Kunth) Avaliadas com Microssatélites. Master’s Thesis, Universidade Federal do Amazonas, Manaus, Brazil, 2009; p. 83. [Google Scholar]
- Shaw, J.; Lickey, E.B.; Schilling, E.E.; Small, R.L. Comparison of whole chloroplast genome sequences to choose noncoding regions for phylogenetic studies in angiosperms: The tortoise and the hare. III. Am. J. Bot. 2007, 94, 275–288. [Google Scholar] [CrossRef]
- Barbieri, R.L.; Neitzke, R.S. Pimentas do gênero Capsicum. Cor, fogo e sabor. In Origem e Evolução de Plantas Cultivadas; Barbieri, R.L., Stumpf, E.R.T., Eds.; Embrapa: Brasília, Brasil, 2008; pp. 728–745. [Google Scholar]
- Reifschneider, F.J.B. Capsicum. Pimentas e Pimentões no Brasil; Embrapa Comunicação para Transferência de Tecnologia: Brasília, DF, Brasil, 2000; p. 113. [Google Scholar]
- Long-Solís, J. Capsicum y Cultura: La Historia del Chilli, 2nd ed.; Fondo de Cultura Económica: Ciudad de México, México, 1998; p. 203. [Google Scholar]
- Pickersgill, B.P.; Heiser, C.B. Origins and distribution of plants domesticated in the New World Tropics. In Origins of Agriculture; Reed, C.A., Ed.; Mouton: The Hague, The Netherlands, 1977; pp. 803–835. [Google Scholar]
- Perry, L.; Dickau, R.; Zarrillo, S; Holst, I.; Pearsall, D.M.; Piperno, D.R.; Berman, M.J.; Cooke, R.G.; Rademaker, R.; Ranere, A.J.; Raymond, J.S.; Sandweiss, D.H.; Scaramelli, F.; Tarble, K.; Zeidler, J.A. Starch fossils and the domestication and dispersal of chili peppers (Capsicum spp. L.) in the Americas. Science 2007, 315, 986–988. [Google Scholar] [CrossRef]
- Pickersgill, B. Migrations of chili peppers, Capsicum spp., in the Americas. In Pre-Columbian Plant Migration; Stone, D., Ed.; Harvard University Press: Cambridge, MA, USA, 1984; Volume 76; pp. 105–123. [Google Scholar]
- Eshbaugh, W.H. Peppers: History and exploitation of a serendipitous new crop discovery. In New Crops; Janick, J., Simon, J.E., Eds.; John Wiley & Sons: NY, New York, 1993; pp. 132–139. [Google Scholar]
- Andrews, J. The peripatetic chili pepper: Diffusion of the domesticated Capsicums since Columbus. In Chilies to Chocolate: Food the Americas Gave the World; Foster, N., Cordell, L.S., Eds.; The University of Arizona Press: Tucson, AZ, USA, 1992; pp. 81–93. [Google Scholar]
- Heiser, C.B., Jr. Peppers. Capsicum (Solanaceae). In Evolution of Crop Plants, 2nd ed.; Smartt, J., Simmonds, N.W., Eds.; Longman Scientific & Technical: London, UK, 1995; pp. 449–451. [Google Scholar]
- Carvalho, S.I.C.; Bianchetti, L.B.; Ribeiro, C.S.C.; Lopes, C.A. Pimentas do gênero Capsicum no Brasil; Embrapa Hortaliças: Brasília, DF, Brasil, 2006; p. 27. [Google Scholar]
- Pickersgill, B. The genus Capsicum: A multidisciplinary approach to the taxonomy of cultivated and wild plants. Biol. Zentralbl. 1988, 107, 381–389. [Google Scholar]
- Walsh, B.M.; Hoot, S.B. Phylogenetic relationships of Capsicum (Solanaceae) using DNA sequences from two noncoding regions: The chloroplast atpb-rbcl spacer region and nuclear waxy introns. Int. J. Plant Sci. 2001, 162, 1409–1418. [Google Scholar] [CrossRef]
- Loaiza-Figueroa, F.; Ritland, K.; Laborde Cancino, J.A.; Tanksley, S.D. Patterns of genetic variation of the genus Capsicum (Solanaceae) in Mexico. Plant Syst. Evol. 1989, 165, 159–188. [Google Scholar] [CrossRef]
- Luciano de Bem Bianchetti. Embrapa Recursos Genéticos e Biotecnologia. personal communication to Charles R.
- Organization for Economic Co-operation and Development (OECD). Consensus Document on the Biology of the Capsicum annuum Complex (chili peppers, hot peppers and sweet peppers; OECD: Paris, France, 2006. Series on Harmonisation of Regulatory Oversight in Biotechnology, Number 36. Available online: http://www.oecd.org/ehs/ (accessed 2 November 2009).
- Coppens d'Eeckenbrugge, G.; Leal, F.; Duval, M.F. Germplasm resources of pineapple. Hortic. Rev. 1997, 21, 133–175. [Google Scholar]
- Beauman, F. The Pineapple. King of fruits; Chatto & Windus: London, UK, 2005; p. 315. [Google Scholar]
- Coppens d'Eeckenbrugge, G.; Duval, M.-F. The domestication of pineapple: context and hypotheses. Pineapple News 2009, 16, 15–27. [Google Scholar]
- Duval, M.-F.; Buso, G.C.; Ferreira, F.R.; Bianchetti, L. de B.; Coppens d’Eeckenbrugge, G.; Hamon, P.; Ferreira, M.E. Relationships in Ananas and other related genera using chloroplast DNA restriction site variation. Genome 2003, 46, 990–1004. [Google Scholar] [CrossRef]
- Leal, F.; Medina, E. Some wild pineapples in Venezuela. J. Bromeliad Soc. 1995, 45, 152–158. [Google Scholar]
- Duval, M.F.; Coppens d'Eeckenbrugge, G.; Ferreira, F.R.; Cabral, J.R.S.; Bianchetti, L. de B. First results from joint EMBRAPA-CIRAD Ananas germplasm collecting in Brazil and French Guyana. Acta Hortic. 1997, 425, 137–144. [Google Scholar]
- Leal, F.; Antoni, M.G. Especies del género Ananas: origen y distribución geográfica. Rev. Fac. Agron. Univ. Cent. Venez. 1981, 29, 5–12. [Google Scholar]
- Bertoni, M.S. Contribution à l’étude botanique des plantes cultivées. I. Essai d’une monographie du genre Ananas. Anal. Cient. Parag. (Serie II) 1919, 4, 250–322. [Google Scholar]
- García, M.L. Etude taxinomique du genre Ananas. Utilisation de la variabilité enzymatique. Doctoral thesis, Université des Sciences et Techniques du Languedoc, Montpellier, France, 1988.
- Duval, M-F.; Noyer, J-L.; Perrier, X.; Coppens d’Eeckenbrugge, G.; Hamon, P. Molecular diversity in pineapple assessed by RFLP markers. Theor. Appl. Genet. 2001, 102, 83–90. [Google Scholar] [CrossRef]
- Coppens d’Eeckenbrugge, G.; Leal, F. Morphology, anatomy and taxonomy. In The Pineapple: Botany, Production and Uses; Bartholomew, D.P., Paull, R.E., Rohrbach, K.G., Eds.; CAB International: Oxford, UK, 2003; pp. 13–32. [Google Scholar]
- Coppens d'Eeckenbrugge, G.; Duval, M.-F.; Van Miegroet, F. Fertility and self-incompatibility in the genus Ananas. Acta Hortic. 1993, 334, 45–51. [Google Scholar]
- Schultes, R.E. Ethnobotanical conservation and plant diversity in the Northwest Amazon. Diversity 1991, 7, 69–72. [Google Scholar]
- Cristancho, S.; Vining, J. Culturally defined keystone species. Res. Hum. Ecol. 2004, 11, 153–164. [Google Scholar]
- Callen, E.O. Analysis of the Tehuacan coprolites. In The Prehistory of the Tehuacan Valley; Byers, D.S., Ed.; Robert S. Peabody Foundation by the University of Texas Press: Austin, TX, USA, 1967; Volume 1; pp. 261–289. [Google Scholar]
- Pennington, T.D. The Genus Inga: Botany; The Royal Botanic Gardens: Kew, London, UK, 1997; p. 844. [Google Scholar]
- Piperno, D.R.; Dillehay, T.D. Starch grains on human teeth reveal early broad crop diet in northern Peru. Proc. Natl. Acad. Sci. USA 2008, 105, 19622–19627. [Google Scholar] [CrossRef]
- Koptur, S. Outcrossing and pollinator limitation on fruit set: breeding systems of Neotropical Inga trees (Fabaceae: Mimosoideae). Evolution 1984, 38, 1130–1143. [Google Scholar] [CrossRef]
- Brodie, A.W.; Labarta-Cávarri, R.A.; Weber, J.C. Tree Germoplasm Management and Use On-farm in the Peruvian Amazon: A Case Study from the Ucayali Region, Peru; Overseas Development Institute: London, UK, International Center for Research in Agroforestry: Nairobi, Kenya; 1997. [Google Scholar]
- Sotelo Montes, C.; Weber, J.C. Priorización de especies arbóreas para sistemas agroforestales en la selva baja del Perú. Agrofor. Am. 1997, 4, 12–17. [Google Scholar]
- Labarta, R.A.; Weber, J.C. Valorización econômica de bienes tangibles de cinco especies arbóreas agroforestales em la Cuenca Amazónica Peruana. Rev. Flor. Centroamer. 1998, 23, 12–21. [Google Scholar]
- Hollingsworth, P.M.; Dawson, I.K.; Goodall-Copestake, W.P.; Richardson, J.E.; Weber, J.C.; Sotelo Montes, C.; Pennington, R.T. Do farmers reduce genetic diversity when they domesticate tropical trees? A case study from Amazonia. Mol. Ecol. 2005, 14, 497–501. [Google Scholar] [CrossRef]
- Doebley, J.F.; Gaut, B.S.; Smith, B.D. The molecular genetics of crop domestication. Cell 2006, 127, 1309–1321. [Google Scholar] [CrossRef]
- Dawson, I.K.; Hollingsworth, P.M.; Doyle, J.J.; Kresovich, S.; Weber, J.C.; Montes, C.S.; Pennington, T.D.; Pennington, R.T. Origins and genetic conservation of tropical trees in agroforestry systems: a case study from the Peruvian Amazon. Conserv. Genet. 2008, 9, 361–372. [Google Scholar] [CrossRef]
- Atroch, A.L.; Nascimento Filho, F.J.; Ângelo, P.C.S.; Freitas, D.V.; Sousa, N.R.; Clement, C.R. Domesticação e melhoramento do guaranazeiro. In Domesticação e Melhoramento de Plantas: Espécies Amazônicas; Borém, A., Lopes, M.T.G., Clement, C.R., Eds.; Editora da Universidade Federal de Viçosa: Viçosa, Minas Gerais, Brazil, 2009; pp. 337–365. [Google Scholar]
- Bettendorff, J.F. Crónica da Missão dos Padres da Companhia de Jesus no Estado do Maranhão; Fundação Cultural do Pará Tancredo Neves, Secretaria de Estado da Cultura: Belém, Brazil, 1990. [Google Scholar]
- Monteiro, M.Y. Antropogeografia do Guaraná; Instituto Nacional de Pesquisas da Amazônia: Manaus, Amazonas, Brazi, 1965; Cadernos da Amazônia, Volume 6; pp. 1–84. [Google Scholar]
- Pereira, N. Os Índios Maués; Organização Simões: Rio de Janeiro, Brazil, 1954. [Google Scholar]
- Freitas, D.B.; Carvalho, C.R.; Nascimento Filho, F.J.; Astolfi Filho, S. Karyotype with 210 chromosomes in guaraná (Paullinia cupana ‘Sorbilis’). J. Plant Res. 2007, 120, 399–404. [Google Scholar] [CrossRef]
- Stebbins, G.L. Polyploidy, hybridization, and the invasion of new habitats. Ann. Mo. Bot. Gard. 1985, 72, 824–832. [Google Scholar] [CrossRef]
- Sousa, N.R. Variabilidade Genética e Estimativas de Parâmetros Genéticos em Germoplasma de Guaranazeiro; Doctoral Dissertation, Dept. Agronomia, Universidade Federal de Lavras: Lavras, Minas Gerais, Brazil, 2003; p. 99. [Google Scholar]
- Piperno, D.R.; Pearsall, D.M. The Origins of Agriculture in the Lowland Neotropics; Academic Press: San Diego, CA, USA, 1998; p. 400. [Google Scholar]
- Urban, G. A história da cultura brasileira segundo as línguas nativas. In História dos Índios no Brasil, 2nd ed.; Carneiro da Cunha, M., Org.; Companhia das Letras: São Paulo, Brazil, 2002; pp. 87–102. [Google Scholar]
- Gina Giovanna Frausin Bustamante, Universidade Federal do Amazonas, Tropical Agronomy Post-Graduate Program. personal communication to Charles R.
- Wadt, L.H.; Kainer, K.A. Domesticação e melhoramento de castanheira. In Domesticação e Melhoramento—Espécies Amazônicas; Borém, A., Lopes, M.T.G., Clement, C.R., Eds.; Editora da Universidade Federal de Viçosa: Viçosa, Minas Gerais, Brazil, 2009; pp. 297–318. [Google Scholar]
- Mori, S.A.; Prance, G.T. Taxonomy, ecology, and economic botany of the Brazil nut (Bertholletia excelsa Humb. & Bonpl.: Lecythidaceae). In New Directions in the Study of Plants and People; Prance, G.T., Balick, M.J., Eds.; The New York Botanical Garden: Bronx, NY, 1990; Advances in Economic Botany; Volume 8, pp. 130–150. [Google Scholar]
- Müller, H.A.; Rodrigues, A.A.; Müller, A.A.; Müller, N.R.M. Castanha-do-Brasil. Resultados de Pesquisa; EMBRAPA, Centro do Pesquisas Agropecuário do Trópico Umido: Belém, Brazil, 1980. [Google Scholar]
- Nelson, B.W.; Absy, M.L.; Barbosa, E.M.; Prance, G.T. Observations on flower visitors to Bertholletia excelsa H.B.K. and Couratari tenuicarpa A.C.SM. (Lecythidaceae). Acta Amazonica 1985, 15 Suppl, 225–234. [Google Scholar]
- Bruce, W. Nelson, Instituto Nacional de Pesquisas da Amazônia, personal communication, cited in Clement, C.R. Brazil nut. In Selected Species and Strategies to Enhance Income Generation from Amazonian Forests; Clay, J.W., Clement, C.R., Eds.; Food and Agriculure Organization: Rome, Italy, 1993; FO: Misc/93/6 Working Paper; pp. 115–127. [Google Scholar]
- Lemes, M.R.; Dick, C.W.; Gribel, R. Filogeografia e estrutura genética de populações de espécies florestais: implicações para conservação e manejo. In Os Avanços da Botânica no Início do Século XXI; Mariath, J.E.A., Santos, R.P., Eds.; Sociedade Brasileira de Botânica: Porto Alegre, Rio Grande do Sul, Brazil, 2006; pp. 120–123. [Google Scholar]
- Gribel, R.; Lemes, M.R.; Bernardes, L.G.; Pinto, A.E.; Shepard, G.H., Jr. Phylogeography of the Brazil-Nut Tree (Bertholletia excelsa, Lecythidaceae): Evidence of Human Influence on the Species’ Distribution; Association for Tropical Biology and Conservation: Morelia, Mexico, 2007; p. 281. [Google Scholar]
- Lemes, M.R.; Gribel, R.; Proctor, J.; Grattapaglia, D. Population structure of mahogany (Swietenia macrophylla King, Meliaceae) across the Brazilian Amazon, based on variation at microsatellite loci: implications for conservation. Mol. Ecol. 2003, 12, 2875–2883. [Google Scholar] [CrossRef]
- Buckley, D.P.; O’Malley, D.M.; Apsit, V.; Prance, G.T.; Bawa, K.S. Genetics of Brazil nut (Bertholletia excelsa Humb. & Bonpl.: Lecythidaceae). 1. Genetic variation in natural populations. Theor. Appl. Genet. 1988, 76, 923–928. [Google Scholar] [CrossRef]
- Kanashiro, M.; Harris, S.A.; Simons, A. RAPD diversity in Brazil nut (Bertholletia excelsa Humb. & Bonpl., Lecythidaceae). Silvae Genet. 1997, 46, 219–223. [Google Scholar]
- Scott Mori. The New York Botanical Gardens, personal communication, cited in Clement, C.R. Brazil nut. In Selected Species and Strategies to Enhance Income Generation from Amazonian Forests; Clay, J.W., Clement, C.R., Eds.; Food and Agriculure Organization: Rome, Italy, 1993; FO: Misc/93/6 Working Paper; pp. 115–127. [Google Scholar]
- Souza, A.G.C.; Alves, R.M.; Sousa, N.R.; Souza, M.G. Domesticação e melhoramento do cupuaçuzeiro. In Domesticação e Melhoramento—Espécies Amazônicas; Borém, A., Lopes, M.T.G., Clement, C.R., Eds.; Editora da Universidade Federal de Viçosa: Viçosa, Minas Gerais, Brazil, 2009; pp. 319–332. [Google Scholar]
- Clement, C.R.; Venturieri, G.A. Bacuri and Cupuassu. In Fruits of Tropical and Subtropical Origin; Nagy, S., Shaw, P.E., Wardowski, W.F., Eds.; Florida Science Source: Lake Alfred, FL, USA, 1990; pp. 178–192. [Google Scholar]
- Alves, R.M.; Sebbenn, A.M.; Artero, A.S.; Clement, C.R.; Figueira, A. High levels of genetic divergence and inbreeding in populations of cupuassu (Theobroma grandiflorum). Tree Genet. Genomics 2007, 3, 289–298. [Google Scholar] [CrossRef]
- Daniel, J. Tesouro Descoberto no Máximo Rio Amazonas; Contraponto: Rio de Janeiro, Brazil, 2004; Volume 1; p. 597. [Google Scholar]
- Balée, W. Footprints of the Forest: Ka’apor Ethnobotany—The Historical Ecology of Plant Utilization by an Amazonian People; Columbia University Press: NY, USA, 1994; p. 396. [Google Scholar]
- Ducke, A. Plantas de cultura precolombiana na Amazônia brasileira. Bol. Téc. Inst. Agron. Norte 1946, 8, 1–24. [Google Scholar]
- Guimarães, P.R., Jr.; Galetti, M.; Jordano, P. Seed dispersal anachronisms: rethinking the fruits extinct megafauna ate. PLoS ONE 2008, 3, e1745. [Google Scholar] [CrossRef]
- Roosevelt, A.; Costa, M.L.; Machado, C.L.; Michab, M.; Mericer, N.; Valladas, H.; Feathers, J.; Barnett, W.; Silveira, M.I.; Henderson, A.; Silva, J.; Chernoff, B.; Reese, D.S.; Holman, J.A.; Toth, N.; Shick, K. Paleoindian cave dwellers in the Amazon: the peopling of the Americas. Science 1996, 272, 373–384. [Google Scholar]
- Oliver, J.R. The archaeology of agriculture in ancient Amazonia. In Handbook of South American Archaeology; Silverman, H., Isbell, W., Eds.; Springer: New York, NY, USA, 2008; pp. 185–216. [Google Scholar]
- Roosevelt, A.; Housley, R.A.; Silveira, M.I.; Maranca, S.; Johnson, R. Eighth millennium pottery from a prehistoric shell midden in the Brazilian Amazon. Science 1991, 254, 1621–1624. [Google Scholar]
- Lathrap, D. Our father the cayman, our mother the gourd: Spinden revisited, or a unitary model for the emergence of agriculture in the New World. In Origins of Agriculture; Reed, C.A., Ed.; Mouton: The Hague, Holand, 1977; pp. 713–751. [Google Scholar]
- Clement, C.R. Fruit trees and the transition to food production in Amazonia. In Time and Complexity in the Neotropical Lowlands: Studies in Historical Ecology; Balée, W., Erickson, C.L., Eds.; Columbia University Press: NY, USA, 2006; pp. 165–185. [Google Scholar]
- van der Hammen, T.; Hooghiemstra, H. Neogene and Quaternary history of vegetation, climate, and plant diversity in Amazonia. Quaternary Sci. Rev. 2000, 19, 725–742. [Google Scholar] [CrossRef]
- Mayle, F.E.; Beerling, D.J.; Gosling, W.D.; Bush, M.B. Responses of Amazonian ecosystems to climatic and atmospheric carbon dioxide changes since the Last Glacial Maximum. Philos. Trans. R. Soc. London B. 2004, 359, 499–514. [Google Scholar] [CrossRef]
- Beerling, D.J.; Mayle, F.E. Contrasting effects of climate and CO2 on Amazonian ecosystems since the last glacial maximum. Glob. Change Biol. 2006, 12, 1977–1984. [Google Scholar] [CrossRef]
- Erickson, C. Historical ecology and future explorations. In Amazonian Dark Earths—Origin, Properties, and Management; Lehmann, J., Kern, D., Glaser, B., Woods, W., Eds.; Kluwer Academic Publ.: Dordrecht, Holland, 2003; pp. 455–500. [Google Scholar]
- Miller, E.T. Arqueologia nos Empreendimentos Hidroeléctricos da Eletronorte: Resultados Preliminares; Eletronorte: Brasília, DF, Brazil, 1992. [Google Scholar]
- Tudge, C. Neanderthals, Bandits and Farmers: How Agriculture Really Began; Yale University Press: New Haven, CT, USA, 1998; p. 53. [Google Scholar]
- Heckenberger, M.; Neves, E.G. Amazonian archaeology. Ann. Rev. Anthrop. 2009, 38, 251–266. [Google Scholar] [CrossRef]
- Dickau, R.; Ranere, A.J.; Cooke, R.G. Starch grain evidence for the preceramic dispersals of maize and root crops into tropical dry and humid forests of Panama. Proc. Natl. Acad. Sci. USA 2007, 104, 3651–3656. [Google Scholar] [CrossRef]
- Heckenberger, M.J.; Russell, J.C.; Toney, J.R.; Schmidt, M.J. The legacy of cultural landscapes in the Brazilian Amazon: implications for biodiversity. Philos. Trans. R. Soc London B. 2007, 362, 197–208. [Google Scholar] [CrossRef]
- Dorofeyev, V.F. Origin and Geography of Cultivated Plants; Cambridge University Press: Cambridge, UK, 1992. [Google Scholar]
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Clement, C.R.; De Cristo-Araújo, M.; Coppens D’Eeckenbrugge, G.; Alves Pereira, A.; Picanço-Rodrigues, D. Origin and Domestication of Native Amazonian Crops. Diversity 2010, 2, 72-106. https://doi.org/10.3390/d2010072
Clement CR, De Cristo-Araújo M, Coppens D’Eeckenbrugge G, Alves Pereira A, Picanço-Rodrigues D. Origin and Domestication of Native Amazonian Crops. Diversity. 2010; 2(1):72-106. https://doi.org/10.3390/d2010072
Chicago/Turabian StyleClement, Charles R., Michelly De Cristo-Araújo, Geo Coppens D’Eeckenbrugge, Alessandro Alves Pereira, and Doriane Picanço-Rodrigues. 2010. "Origin and Domestication of Native Amazonian Crops" Diversity 2, no. 1: 72-106. https://doi.org/10.3390/d2010072
APA StyleClement, C. R., De Cristo-Araújo, M., Coppens D’Eeckenbrugge, G., Alves Pereira, A., & Picanço-Rodrigues, D. (2010). Origin and Domestication of Native Amazonian Crops. Diversity, 2(1), 72-106. https://doi.org/10.3390/d2010072




