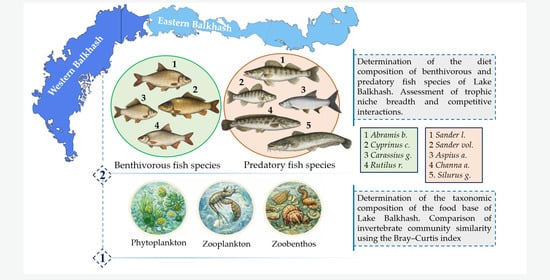
1. Introduction
The dynamics of forage organism communities (phytoplankton, zooplankton, and benthos) in lake ecosystems are influenced by a complex interplay of natural–climatic and anthropogenic factors [
1]. Within these systems, these organisms constitute the trophic foundation that supports the functioning and productivity of higher-level consumers, particularly fish communities [
2,
3]. However, existing studies in this field are generally limited to the analysis of individual invertebrate groups or isolated components of fish food chains, lacking an integrated assessment of the entire forage base of the ecosystem [
4,
5,
6].
Fish represent key functional components of lake food webs, occupying a wide spectrum of trophic niches and facilitating the transfer of matter and energy from basal resources to higher trophic levels of the ecosystem [
7]. Understanding trophic relationships among fish species, the nature of interspecific and intraspecific competition, as well as the composition and structure of the forage base, provides a scientific basis for the development of management measures aimed at conserving fish resources and, where necessary, improving ecosystem feeding conditions [
8].
For Lake Balkhash (Kazakhstan), an ecosystem characterized by a long history of the formation and transformation of both invertebrate and fish communities, research in this field remains highly relevant under current environmental change driven by climatic factors, hydrological fluctuations, anthropogenic pollution, and shifting fishing pressure [
9,
10,
11]. Large-scale introductions of forage invertebrates into Lake Balkhash during the 20th century contributed to increased fish productivity. However, subsequent interruptions and insufficient follow-up studies and management measures may have led to reduced taxonomic diversity and structural simplification of forage communities [
12,
13,
14]. Stocking efforts in Eastern Balkhash with the polychaete
Nereis diversicolor (O.F. Müller) and the bivalve
Abra ovata (Philippi) have been reported by the Balkhash branch of the Research and Production Center for Fisheries [
15]. Such transformations—manifested as both enrichment and regression of species diversity—can substantially influence the formation and functioning of fish trophic chains, ultimately affecting fish productivity, the structure of commercial stocks, and shifts in dominant species within the ichthyocenosis.
Monitoring changes in the species diversity of forage invertebrates allows for the assessment of competition intensity for food resources [
16]. When competition for shared resources arises, spatial redistribution of organisms may occur, accompanied by altered habitat use and potential trophic niche overlap not previously observed within the community structure [
17]. Given the large area of Lake Balkhash (16.4 thousand km
2) and its pronounced spatial heterogeneity in physicochemical parameters—particularly salinity differences between the western and eastern basins—analysis of the forage base and fish trophic relationships is essential to identify inter-basin differences in dietary structure and food-web organization. The salinity of the lake increases sharply from west to east. In the western part of Lake Balkhash, mineralization values range from 680 to 2200 mg/dm
3 (0.68–2.2 g/L), whereas in the eastern part they reach 4900–5600 mg/dm
3 (4.9–5.6 g/L) [
18].
In the present study, a comprehensive assessment of species composition and structure of the forage base was conducted across three trophic levels: phytoplankton, zooplankton, and zoobenthos. Based on these data, fish diets were analyzed, with particular attention to contemporary trophic linkages involving non-native species and the degree and nature of their competitive interactions. This integrative approach enabled evaluation of food-web functioning as a unified system and clarified the role of different forage components in maintaining the trophic structure of fish communities in the western and eastern parts of Lake Balkhash.
3. Results
3.1. Hydrochemical Characteristics
In 2025, the water depth at the sampling stations in the western part of Lake Balkhash ranged from 1.4 to 7.7 m, whereas in the eastern part it varied from 3.0 to 5.3 m. Water transparency in the western basin ranged from 10 to 140 cm. Toward the eastern part of the lake, an increase in transparency was observed with increasing depth, varying from 40 to 350 cm.
During the study period, the water temperature in Lake Balkhash ranged between 23.2 and 26.5 °C. The average hydrochemical parameters of Lake Balkhash water are presented in
Table 4.
The pH values were 8.61 ± 0.05 in the western basin and 8.44 ± 0.05 in the eastern basin. The concentration of dissolved oxygen was slightly lower in the western part of the lake, reaching 8.04 ± 0.06 mg/L.
The concentrations of nutrients in the water did not exceed the regulatory standards established for fishery water bodies [
23]. The spatial distribution of nutrient ions across the lake was relatively uniform. For example, nitrite concentrations in the western and eastern basins of Lake Balkhash were 0.04 ± 0.03 mg/L and 0.03 ± 0.04 mg/L, respectively.
The permanganate oxidizability increased from west to east. The average values of permanganate oxidizability were 5.30 ± 0.70 mg O/L in the western basin and 10.10 ± 0.30 mg O/L in the eastern basin.
A similar pattern was observed for water mineralization. In the western basin, the average mineralization was 1727.00 ± 120.00 mg/L, whereas significantly higher values were recorded in the eastern part of the lake, reaching 4068.00 ± 137.00 mg/L.
3.2. Phytoplankton
In 2025, a total of 148 phytoplankton taxa were recorded in Lake Balkhash, representing six divisions: Cyanobacteria, Bacillariophyta, Chlorophyta, Charophyta, Dinoflagellata, and Euglenophyta (
Table 5). Overall, 110 taxa were identified in the western area and 112 in the eastern one, indicating comparable levels of species richness between the two areas.
No taxa exhibited 100% occurrence in both areas. The highest frequency of occurrence was recorded for Snowella lacustris (59% in the western areaand 94% in the eastern area). Widely distributed taxa (≥50% occurrence in at least one area) included Ulnaria ulna (53% in the western area), Lindavia comta (41% and 65% in the western and eastern areas, respectively), Oocystis lacustris (35% and 53%), Gomphosphaeria aponina (65% in the eastern area), Glenodinium sp. (53% in the western area), Trachelomonas sp. (59% and 29%), and Mucidosphaerium pulchellum (47% in both areas, according to the table data). These taxa consistently showed high frequencies of occurrence within their respective areas. The majority of the recorded taxa (over 60%) occurred at frequencies of 6–24% and can therefore be classified as rare or locally distributed. This group includes numerous representatives of the genera Navicula, Nitzschia, Cymbella, and Pinnularia, as well as several green algae and dinoflagellates.
In terms of taxonomic composition, Bacillariophyta exhibited the highest diversity (76 taxa in the western area and 59 in the eastern area). Cyanobacteria comprised 37 and 27 taxa, respectively; Chlorophyta comprised 21 and 18 taxa; Charophyta comprised 3 and 2 taxa; Dinoflagellata comprised 6 and 4 taxa; and Euglenophyta comprised 5 and 2 taxa.
Overall, the phytoplankton communities of the western and eastern areas were characterized by similar species richness, the absence of taxa with ubiquitous (100%) occurrence, and a predominance of species with low frequencies of occurrence, reflecting spatial heterogeneity in community structure.
The dendrogram illustrates the very high similarity in phytoplankton species composition among 38 sites in Lake Balkhash, with all branches representing ≥90% similarity (
Figure 2).
Freshwater and slightly brackish sites (e.g., BB-WB—Bukhta B, ST-WB—Shubartyubek, TS-WB—Tasaral) form distinct clusters, indicating nearly identical species composition.
Transitional sites (e.g., MS-WB—Maly Saryshagan, OLEB—Orlinaya, UTEB—Ultarakhty) link several clusters, also demonstrating very high similarity. Highly saline or more isolated sites (e.g., ZHEB—Zhalanash, KGEB—Karashygan, TPEB—Tolepshapkan) form separate branches, yet still maintain ≥90% similarity, reflecting unique but closely related communities.
Overall, the dendrogram indicates that all studied sites of Lake Balkhash share a very high degree of similarity in phytoplankton composition, highlighting the dominance of similar species throughout the water body.
3.3. Zooplankton
In 2025, a total of 56 zooplankton taxa were recorded in Lake Balkhash, including Rotifera, Cladocera, and Copepoda (
Table 6). In the western area, 39 taxa were recorded, and in the eastern area, 36 taxa, indicating comparable species richness across the two areas. No taxa were observed with 100% occurrence in both areas. The highest frequencies were recorded for
Arctodiaptomus salinus and
Mesocyclops leuckarti (94–100%),
Diaphanosoma lacustris (82–88%), and
Thermocyclops crassus (82–88%).
Daphnia (Daphnia) galeata occurred at 47% in the eastern area, while
Harpacticoida gen. sp. appeared in 29% of samples in both areas. Rotifers, such as
Euchlanis dilatata dilatata,
Habrotrocha bidens, and
Trichotria pocillum pocillum, were recorded at intermediate frequencies (12–18%).
Most taxa, including Anuraeopsis fissa fissa, Brachionus plicatilis, Eosphora najas, Keratella cochlearis, Acroperus costata, Chydorus sphaericus, Bosmina longirostris, and others, were observed rarely, with frequencies of 6–18%, indicating localized occurrence.
Overall, the zooplankton of Lake Balkhash in 2025 exhibited similar species richness in both areas, absence of taxa with ubiquitous occurrence, and a predominance of taxa with low frequency, reflecting spatial heterogeneity in the community.
The dendrogram reveals very high similarity in zooplankton species composition among 38 sampling stations in Lake Balkhash, with all clusters exhibiting Bray–Curtis similarity values above 90% (
Figure 3). Several sites, such as UAEB, KTWB, and SHWB, form tight clusters reflecting nearly identical species assemblages. Transitional stations (e.g., AHEB, MAVB, OREB) link clusters while maintaining similarity above 90%. Even the most distinct stations display similarity close to or exceeding 90%, indicating a largely homogeneous zooplankton community across the lake. Overall, the dendrogram highlights strong spatial uniformity in zooplankton species composition, suggesting widespread distribution of similar species throughout Lake Balkhash.
3.4. Zoobenthos
In 2025, a total of 15 zoobenthos taxa were recorded in Lake Balkhash, including representatives of Mollusca, Vermes, Crustacea, and Chironomidae (
Table 7). In the western area, 10 taxa were recorded, and in the eastern area, 12 taxa were recorded, indicating comparable species richness between the two areas.
No taxa were observed with 100% occurrence in both areas. Among the most frequently recorded taxa were Oligochaeta gen. sp. (82% in the western area, 35% in the eastern area), Corophium curvispinum (41% in the western area), and Chironomus gr. plumosus (12% in the western area, 47% in the eastern area). Hypania invalida occurred at 35% and 12% in the western and eastern areas, respectively, while Harnischia fuscimanus was recorded at 41% in the western area.
Other taxa, including Monodacna colorata, Lymnaea ovata, Hypaniolla kowalevskii, Gammarus lacustris, Niphargoides robustoides, Paramysis ullskyi, Tanypus punctipennis, and Tabanidae (Tabanus and Chrysops), were recorded at low frequencies (6–24%), indicating localized occurrence.
Overall, the zoobenthos community in Lake Balkhash in 2025 exhibited similar species richness in both areas, the absence of taxa with ubiquitous occurrence, and a predominance of taxa with low frequency, reflecting spatial heterogeneity in benthic communities (
Figure 4).
The dendrogram based on Bray–Curtis similarity shows a high degree of resemblance in zoobenthos species composition among the 38 sampling stations of Lake Balkhash (
Figure 4). All stations cluster at similarity levels above 90%, indicating low spatial variability in benthic community structure across the lake.
Several stations form very tight clusters, reflecting nearly identical zoobenthic assemblages and minimal differences in species composition. Transitional stations connect the main clusters while still maintaining similarity values above 90%, suggesting gradual spatial changes rather than distinct ecological boundaries.
Even the most dissimilar stations exhibit similarity values close to 90%, confirming the overall homogeneity of the zoobenthos community. In general, the dendrogram highlights strong spatial uniformity and a widespread distribution of similar benthic species throughout Lake Balkhash.
3.5. Diet Analysis
3.5.1. Diet Composition of Benthivorous Fish Species
The diet of benthivorous fish species in Lake Balkhash differed quantitatively between the western and eastern basins: 18 food components were identified in the western part and 16 in the eastern part. The proportional contribution of individual food items varied depending on the species considered.
Figure 5 presents the composition of dietary components of benthivorous (“non-predatory”) fish species.
The diet of common carp (C. carpio) in the western part of Lake Balkhash consisted primarily of mollusks (30.71%), detritus (15.03%), macrophytes (13.57%), chironomid larvae (8.93%), insect larvae (5.99%), mysids (3.23%), gammarids (1.71%), and unidentified digested remains (16.50%). Other food components each accounted for less than 1% of the total diet composition.
The diet of roach (R. caspicus) was dominated by macrophytes (66.04%), followed by mysids (12.16%), corophiids (5.38%), chironomid larvae (10.31%), and detritus (5.70%).
In crucian carp (C. gibelio), the diet consisted almost entirely of plant material (97.51%). The occurrence of zooplankton (1.81%), chironomid larvae (0.55%), and fish eggs (0.13%) was minimal and may be considered incidental for this species.
The diet of common bream (A. brama) included six principal components: corophiids (33.76%), mysids (24.87%), mollusks (18.09%), detritus (11.82%), oligochaetes (9.18%), and macrophytes (2.28%).
In the eastern part of Lake Balkhash, the diet of common carp (C. carpio) showed an increased contribution of plant material (57.4%), followed by detritus (16.54%), chironomid larvae, algae (4.63%), mysids (2.58%), and corophiids. Digested remains accounted for 6.38%, while mayflies, caddisflies, ostracods, and periphyton each contributed less than 1%.
In roach (R. caspicus), the proportion of plant material in the eastern basin (37.91%) was lower than in the western part. Mysids comprised 25.35% of the diet, cestodes 19.29%, and chironomid larvae 14.17%, whereas the remaining components were present in minor proportions.
The diet of crucian carp (C. gibelio) remained dominated by higher aquatic vegetation (94.06%), with smaller contributions from zooplankton (3.95%), chironomid larvae (1.87%), and fish eggs (0.13%). In common bream (A. brama), mysids represented approximately 65.57% of the diet. In the eastern basin, bream also consumed chironomid larvae (17%), detritus (6.09%), corophiids (5.89%), and cestodes (3.05%).
Interspecific comparison using the Bray–Curtis index revealed differences in the consumption of dietary components among species (
Table 7).
The diet of crucian carp (C. carassius) remained dominated by higher aquatic vegetation (94.06%), with smaller contributions from zooplankton (3.95%), chironomid larvae (1.87%), and fish eggs (0.13%). In common bream (A. brama), mysids represented approximately 65.57% of the diet. In the eastern basin, bream also consumed chironomid larvae (17%), detritus (6.09%), corophiids (5.89%), and cestodes (3.06%).
Interspecific comparison using the Bray–Curtis index revealed differences in the consumption of dietary components among species (
Table 8).
Moderate trophic similarity among benthivorous fish species from the western part of Lake Balkhash was observed between roach and crucian carp (BC = 0.33), common carp and common bream (BC = 0.64), and common carp and roach (BC = 0.68). Pronounced differences in dietary composition (BC > 0.70) were recorded for crucian carp and common carp, common bream and roach, common carp and crucian carp, as well as common bream and crucian carp.
In the eastern part of Lake Balkhash, trophic similarity among benthivorous fish species remained generally moderate; however, marked dietary differentiation was observed for common bream compared to the other species.
3.5.2. Diet Composition of Predatory Fish Species
The diet of predatory fish species in Lake Balkhash is characterized by a mixed trophic structure. Digested remains of animal origin were detected in the stomach contents of all examined predatory species, indicating pronounced ichthyophagy and the consumption of other macrozoobenthic organisms.
The most diverse diet was recorded in pikeperch (
S. lucioperca) from the western part of the lake, where 10 food components were identified. The diet was dominated by digested fish remains (39.2%), reflecting a high degree of specialization toward piscivory. Substantial proportions were also represented by roach (
R. caspicus) (19.05%), crucian carp (
Carassius spp.) (17.3%), common bream (
A. brama) (9.53%), and freshwater shrimp remains (7.72%).
Figure 6 shows the composition of dietary components of predatory fish species in Lake Balkhash.
The diet of Volga pikeperch (S. volgensis), a closely related species to pikeperch, was dominated by freshwater shrimp (37.6%), mysids (23.6%), and unidentified fish remains (20.04%). The diet of this species comprised six food components in total.
In asp (A. aspius), only four dietary components were recorded, with digested fish remains (37.46%) and mysids (35.15%) constituting the largest proportions. Common bream (A. brama) and roach (R. caspicus) were also identified in the diet. The consumption of bream and roach appears consistent with their ecological characteristics, as these species inhabit not only near-bottom layers but also actively migrate within the pelagic zone, particularly during early ontogenetic stages.
In the diets of wels catfish (S. glanis) and snakehead (C. argus), remains of pikeperch accounted for 31.68% and 9%, respectively, while crucian carp represented 25.05% and 29.59%. Roach consumption was higher in snakehead (14.23%) compared to catfish (3.02%).
Significant dietary differences among predatory fish species from the western part of Lake Balkhash were observed between snakehead and pikeperch (BC = 0.76), Volga pikeperch and asp (BC = 0.76), and catfish and snakehead (BC = 0.74). The greatest dietary similarity was recorded between snakehead and both asp and catfish (BC = 0.26).
Figure 7 illustrates trophic similarities and differences among predatory fish species based on Bray–Curtis index calculations.
The diet of predatory fish in the eastern part of Lake Balkhash shows similarities in the consumption of certain components recorded in the western basin. For example, pikeperch (S. lucioperca) consumed up to 10 food components, with roach, common bream, freshwater shrimp, and digested fish remains forming the bulk of the diet. The contribution of bream and roach increased to 26.47% and 24.33%, respectively.
In Volga pikeperch (S. volgensis), digested fish remains accounted for 45.69% of the diet. Other components were more evenly distributed, including roach (23.15%), crucian carp (12.87%), freshwater shrimp (7.1%), and mysids (5.35%). Notably, unlike other predatory species, the diet of S. volgensis included the endemic Balkhash perch (Perca schrenkii Kessler, 1874). The population of this endemic species has declined markedly following the introduction and subsequent expansion of predatory fish, accompanied by a reduction in the proportion of native ichthyofauna. Its occurrence in the diet of Volga pikeperch may therefore represent an additional trophic pressure on the endemic component of the ichthyocenosis.
Crucian carp constituted the dominant prey item in the diet of wels catfish (S. glanis), accounting for 51.55%, while other components were relatively evenly distributed. As a benthic predator, catfish likely consumes crucian carp due to its high abundance in bottom habitats and spatial overlap in these biotopes.
In snakehead (C. argus), digested fish remains predominated (63.73%). Aquatic beetles (Coleoptera) contributed 12.28%, while crucian carp (18.85%) and bream (5.14%) were recorded in smaller proportions.
Dietary similarities and differences for both basins of Lake Balkhash are presented in
Figure 7.
The highest trophic similarity was identified between pikeperch (S. lucioperca) inhabiting the two basins of the lake, which is expected for the same species (BC ≤ 0.30). In addition, the closely related Volga pikeperch (S. volgensis) from the eastern basin exhibited dietary similarity with pikeperch from the western basin, suggesting partial trophic niche overlap.
In the eastern part of Lake Balkhash, moderate trophic similarity (BC within intermediate values) was observed among most of the studied species. An exception was recorded for snakehead (C. argus) and asp (A. aspius), which demonstrated pronounced differences in dietary composition (BC ≥ 0.70), indicating trophic differentiation and reduced niche overlap between these species.
3.5.3. Assessment of Trophic Niche Breadth
The diversity of dietary components identified in this study indicates varying degrees of trophic specialization among the examined fish species of Lake Balkhash. Calculation of trophic niche breadth indices enabled an integrated assessment of the feeding strategies of introduced species within this ecosystem.
Table 9 presents the niche breadth index values for species inhabiting the western and eastern parts of Lake Balkhash.
The values of Levin’s niche breadth index for the studied fish species did not exceed Bi < 0.30, allowing the introduced species of Lake Balkhash to be classified as trophically narrow specialists. These results are consistent with the analysis of dietary composition presented in
Section 3.5.2 and further confirm the identified patterns of trophic specialization.
No substantial differences in niche breadth were detected among most species, likely reflecting their taxonomic relatedness and the consumption of similar dominant food components. An exception was observed for crucian carp, for which no significant variation between basins was recorded, as its diet was largely restricted to macrophytes. Pikeperch exhibited the most diverse diet, with slightly higher niche breadth values (although still Bi < 0.30) due to the consumption of approximately 9–10 food components.
The predominance of a limited number of dominant prey groups may entail ecological risks, including increased sensitivity to environmental fluctuations, reduced capacity for trophic switching, and intensified interspecific competition. It should be noted that the present dietary analysis is based primarily on summer data, which constrains the interpretation of trophic patterns. Seasonal variation in resource availability and trophic interactions may alter the spectrum of consumed prey and allow partial substitution among food items. Therefore, further multi-seasonal studies are recommended to provide a more comprehensive assessment of trophic niche breadth and plasticity in the studied species.
3.5.4. Assessment of Niche Overlap Using Pianka’s Index
Analysis of niche overlap among benthivorous fish species in the western part of Lake Balkhash (
Table 10) revealed the presence of interspecific competition. A particularly high level of overlap was recorded between crucian carp and roach (Ojk = 0.97), indicating pronounced competition for shared food resources. For the remaining species pairs, trophic overlap was moderate and did not exceed Pianka’s index values of Ojk ≤ 0.46.
It is likely that in the western basin of Lake Balkhash, the relatively narrow trophic specialization of the studied species, combined with sufficient availability of food resources during the summer period, provided favorable feeding conditions. This may have contributed to a reduction in the overall intensity of interspecific competition for dietary resources.
In the eastern part of Lake Balkhash, interspecific competition increased among all examined benthivorous fish species. The highest trophic overlap was observed between roach and common carp (Ojk = 0.76), crucian carp and common carp (Ojk = 0.95), and crucian carp and roach (Ojk = 0.74). Competition involving common bream was generally moderate or minimal when compared with other species, except for roach (Ojk = 0.58). Notably, trophic overlap between common carp and common bream remained low (Ojk = 0.12), although competition between carp and other species was more pronounced.
The observed competitive patterns in the eastern basin, particularly among crucian carp, common carp, and roach, are likely associated with the seasonal abundance of macrophytes and mysids. In contrast, common bream appears to engage less frequently in direct competitive interactions with common carp due to its substantial consumption of mysids and chironomid larvae, and comparatively lower reliance on macrophytes.
Competition among predatory fish species in the western part of Lake Balkhash was recorded among four species (
Table 11). Competitive pressure exerted by Volga pikeperch was comparatively weaker. Strong niche overlap was identified between snakehead and pikeperch (Ojk = 0.92), as well as between snakehead and wels catfish (Ojk = 0.87). The consumption of crucian carp (
Carassius spp.) as a major prey item by both catfish (
S. glanis) and snakehead (
C. argus) suggests the formation of potential competitive interactions between these species. At the same time, the absence of roach (
R. caspicus) in the diet of snakehead is noteworthy, despite the similarity of their occupied habitats.
This pattern may reflect reduced availability of roach due to intensive predation by other piscivorous species, thereby limiting its inclusion in the snakehead diet. Consequently, snakehead appears to shift toward more accessible prey items, indicating a degree of trophic plasticity under conditions of interspecific competition.
In contrast to the western basin of Lake Balkhash, the eastern part exhibited increased trophic competition involving pikeperch, with overlap values of Ojk = 0.73 in relation to one species and Ojk = 0.76 in relation to asp. At the same time, trophic overlap between pikeperch and asp also increased (Ojk = 0.65). In other species pairs, competition remained moderate compared with the aforementioned groups.
4. Discussion
The comparative analysis of phytoplankton, zooplankton, and zoobenthos communities suggests that Lake Balkhash is currently undergoing a phase of structural reorganization driven by ongoing hydrological and climatic variability. The observed patterns indicate increasing biodiversity among primary producers and relative stabilization at higher trophic levels, reflecting a complex ecosystem response to long-term environmental fluctuations [
55,
56].
Phytoplankton diversity has increased markedly over the past two decades. In 2004, 91 species were recorded, with Chlorophyta dominating in the western area and Bacillariophyta forming the basis of diversity in the eastern area [
18]. By 2025, total richness had risen to 148 taxa, with a more balanced distribution between the two areas. Diatoms became the most diverse division, while cyanobacteria continued to play a significant ecological role. Notably,
Snowella lacustris remained one of the most widespread taxa across both periods, whereas species such as
Lindavia comta,
Ulnaria ulna, and
Oocystis lacustris became more prominent in 2025.
Despite the substantial increase in taxonomic richness, the absence of taxa with ubiquitous occurrence and the predominance of low-frequency species indicate persistent spatial heterogeneity within the lake. These patterns likely reflect the influence of strong environmental gradients between the western and eastern basins, including differences in salinity, hydrological connectivity, and nutrient availability. The observed diversification of phytoplankton communities may therefore be associated with improved hydrological conditions in recent years, while basin-scale environmental contrasts continue to structure community composition.
In contrast to phytoplankton, zooplankton communities demonstrate relatively stable species richness but noticeable internal restructuring. The total number of taxa remained similar between 2021–2023 (59 taxa) and 2025 (56 taxa), although the relative contribution of major groups changed [
55]. In particular, the proportional contribution of Rotifera declined, whereas Cladocera and Copepoda increased. Core taxa such as
Arctodiaptomus salinus,
Mesocyclops leuckarti,
Thermocyclops crassus,
Diaphanosoma lacustris, and
Daphnia galeata remained persistent and exhibited high frequencies in 2025, indicating a relatively stable functional core of the zooplankton community.
The decline in rotifer diversity together with the increased importance of crustacean taxa may reflect shifts in trophic structure, salinity conditions, or hydrological stability. Such changes are consistent with the general tendency for larger crustacean zooplankton to dominate under conditions of improved water balance and reduced environmental instability. As in phytoplankton communities, none of the zooplankton taxa demonstrated complete ubiquity across the lake, further emphasizing spatial patchiness as a persistent ecological feature.
Long-term zoobenthos data (1975–2024) clearly demonstrate the sensitivity of benthic communities to hydrological fluctuations. Periods of minimal diversity, such as the 7–9 taxa recorded during the 1990s, coincided with declining water levels and increased evaporation. In contrast, the peak recorded in 2021 (35 taxa) corresponded to improved water balance within the basin [
56]. In 2025, 15 taxa were recorded, representing an intermediate level of diversity.
The continued prominence of tolerant taxa such as Hypania invalida and the Chironomus plumosus group indicates that benthic communities still experience certain environmental constraints, although present conditions appear considerably more favorable than during previous periods of hydrological stress. Comparable species richness between the western and eastern areas, together with the absence of ubiquitous taxa, further supports the conclusion that spatial heterogeneity remains a fundamental characteristic of the benthic community structure.
Taken together, these results indicate that Lake Balkhash is currently experiencing a gradual process of ecological restructuring rather than a simple linear recovery. The pronounced increase in phytoplankton diversity likely reflects improved hydrological inputs and the expansion of available ecological niches. In contrast, zooplankton and zoobenthos communities appear to respond primarily through structural reorganization and stabilization of functionally important taxa rather than through a direct increase in overall species richness.
The persistence of spatial heterogeneity across all trophic levels suggests that hydrological and physicochemical gradients between the western and eastern basins remain key drivers of ecosystem organization. These gradients, which are strongly influenced by variations in inflow from the Ili River and regional climatic conditions, continue to shape the distribution and interactions of aquatic communities within the lake.
Overall, the 2025 data do not indicate a complete ecological recovery to historical maxima but rather reflect the establishment of a relatively stable and moderately resilient ecosystem state shaped by long-term variability in water balance and climate-related pressures.
Prior to the implementation of acclimatization measures, the ichthyofauna of Lake Balkhash consisted predominantly of Balkhash perch (
Perca schrenkii Kessler, 1874), Balkhash marinka (
Schizothorax argentatus Kessler, 1874), Ili marinka (
Schizothorax pseudaksaiensis Herzenstein, 1889), and spotted loach (
Triplophysa strauchii (Kessler, 1874)). At that time, the trophic network was characterized by relatively simple interactions and a low level of food competition. Stability within the fish community was largely maintained through trophic interactions between the dominant predator—Balkhash perch—and its prey, primarily juveniles of native fish species inhabiting the middle reaches of tributary rivers [
49].
A major transformation of trophic relationships began following the introduction of pikeperch and several invertebrate species in 1958 [
13], which resulted in the restructuring of food chains and increased interspecific interactions. Analyses of pikeperch feeding ecology revealed pronounced dietary shifts during the process of naturalization, reflecting a redistribution of predation pressure on native species [
57,
58]. For example, in 1963, Balkhash perch comprised 45–73% of the pikeperch diet. Subsequently, during 1964–1969, a sharp decline in marinka abundance was recorded, likely associated with intensified predation by pikeperch [
59].
Competition for food resources has been documented among benthivorous fish species, particularly between common carp and bream exploiting similar benthic prey [
60]. At the same time, pronounced qualitative and quantitative differences in zoobenthos structure have been identified between the western and eastern basins of the lake. In the western basin, carp exhibits a mixed feeding strategy involving zoobenthos, macrophytes, and detritus, with molluscs dominating the diet (20–95% of total volume). In contrast, amphipods (Corophiidae) and insect larvae, mainly chironomids, predominate in carp diets in the eastern basin [
59].
Similar trophic spectra of benthivorous fish have been reported in other lakes and reservoirs of Kazakhstan as well as in hydrologically comparable ecosystems outside the region [
61,
62,
63]. Lake Balkhash and the Alakol lake system are frequently considered ecological analogs [
64]. Comparable feeding patterns among carp, roach, and bream have also been documented in Kapshagay Reservoir [
65].
Analysis of trophic niche overlap among benthivorous fish in the western basin of Lake Balkhash indicates the presence of interspecific competition, although its overall intensity remains moderate. The most pronounced overlap was observed between crucian carp and roach (Ojk = 0.97), suggesting a high potential for competitive interactions. For other species pairs, Pianka’s index did not exceed 0.46, indicating relatively limited trophic similarity. Such patterns may be associated with trophic specialization and the relatively high availability of food resources during the summer period.
In the eastern basin, trophic overlap among benthivorous fish species appears to be higher. The greatest similarity was observed among crucian carp, common carp, and roach, suggesting increased competition for available resources. In contrast, bream demonstrated relatively low overlap with most species, except for moderate similarity with roach. This trophic differentiation may be explained by the stronger reliance of bream on mysids and chironomid larvae, which reduces competition for macrophyte resources.
The feeding ecology of predatory fish species—including pikeperch, snakehead, asp, wels catfish, and Volga pikeperch—is strongly influenced by habitat structure, prey availability, and ontogenetic behavioral traits. Spatial niche differentiation among predators appears to reduce direct competition: asp typically occupies surface waters, pikeperch deeper zones, wels catfish benthic habitats, and snakehead vegetated shallow areas [
66]. Ontogenetic development is known to influence feeding strategies in fish through changes in morphology, prey capture ability, and energetic requirements. In the present study, ontogenetic effects on the consumption of specific dietary components were observed in predatory species such as
Sander lucioperca and
Aspius aspius. Differences in diet composition were detected among individuals of different age groups inhabiting the western and eastern basins of Lake Balkhash. For instance, pike-perch from the western basin (mean age 3.6 ± 0.2 years) consumed approximately ten dietary components. Their diet was dominated by digested fish remains (about 40%), while other prey items included roach, crucian carp, bream, and shrimp. In contrast, individuals from the eastern basin (mean age 4.2 ± 0.2 years) consumed largely similar prey categories, but the proportion of crucian carp was lower (3.78%), and the share of digested fish remains reached 23.48%, which is nearly two times lower than in the western basin.
Age-related differences in diet composition were also observed in asp. Individuals from the western and eastern parts of the lake (7 ± 0.3 and 4 ± 0.2 years, respectively) exhibited notable differences in feeding patterns. In the eastern basin, the diet included significant proportions of mysids (30.19%) and cestodes (5.84%). In contrast, in the western basin the diet was dominated by fish prey, and cases of cannibalism involving juvenile conspecifics were recorded (35.15%).
Overall, these results suggest that ontogenetic variation contributes to shifts in trophic resource use in predatory fish species. Changes in age structure appear to influence the composition of consumed prey and may result in size-dependent trophic strategies within the fish community of Lake Balkhash.
In the western basin of Lake Balkhash, the most pronounced trophic overlap occurs among C. argus, S. lucioperca, and S. glanis, whereas S. volgensis demonstrates relatively distinct feeding preferences. In the eastern basin, niche overlap increases among pike-perch, snakehead, and A. aspius, while trophic relationships among other species combinations indicate the persistence of competitive interactions.
5. Conclusions
1. The 2025 assessment of phytoplankton, zooplankton, and zoobenthos communities in Lake Balkhash indicates a structurally reorganized yet relatively stable ecosystem under ongoing hydrological and climatic variability. Phytoplankton exhibited a substantial increase in taxonomic richness, whereas zooplankton and zoobenthos showed compositional restructuring with increased contributions of ecologically tolerant taxa. The absence of ubiquitous species and the predominance of low-frequency taxa across all trophic groups reflect persistent spatial heterogeneity. Overall, the lake demonstrates moderate ecological resilience but has not fully recovered to historical maximum diversity levels.
2. Benthivorous fish species in Lake Balkhash are characterized predominantly by a moderate degree of trophic similarity, with certain species pairs exhibiting pronounced trophic differentiation. In the western basin, a mosaic pattern of niche overlap is observed, where some species show partial overlap in resource use, whereas others display substantial differences in diet composition.
In the eastern basin, trophic similarity generally remains moderate; however, common bream (A. brama) demonstrates a more distinct trophic position compared with other benthivorous species.
3. Predatory fish species of Lake Balkhash also exhibit overall moderate trophic similarity, while maintaining intraspecific dietary stability in pikeperch (S.lucioperca). Partial niche overlap is observed between pikeperch and Volga pikeperch (Sander volgensis), whereas snakehead (C. argus) and asp (A. aspius) show marked trophic differentiation, indicating the development of distinct feeding strategies.
4. Introduced fish species are characterized by narrow trophic niche breadth (Bi < 0.30), reflecting pronounced dietary specialization. No substantial interspecific differences were detected, except for pikeperch, which exhibited a slightly broader range of consumed prey items. These findings suggest limited trophic plasticity during the summer period and highlight the need for multi-seasonal studies to achieve a more comprehensive evaluation of their ecological strategies.
5. In the western basin, trophic overlap among species is maintained, with the strongest overlap observed between crucian carp and roach. In the eastern basin, dietary similarity generally increases, while bream retains a relatively distinct trophic position. Among predatory species, the greatest niche overlap in the western basin occurs between snakehead, pikeperch, and catfish, whereas in the eastern basin trophic similarity increases between pikeperch, snakehead, and asp. Overall, the trophic relationships indicate the persistence of competitive interactions among species.