Bleaching Impacts on the Last Remaining Acropora-dominated Reefs in the United Arab Emirates
Abstract
1. Introduction
2. Materials and Methods
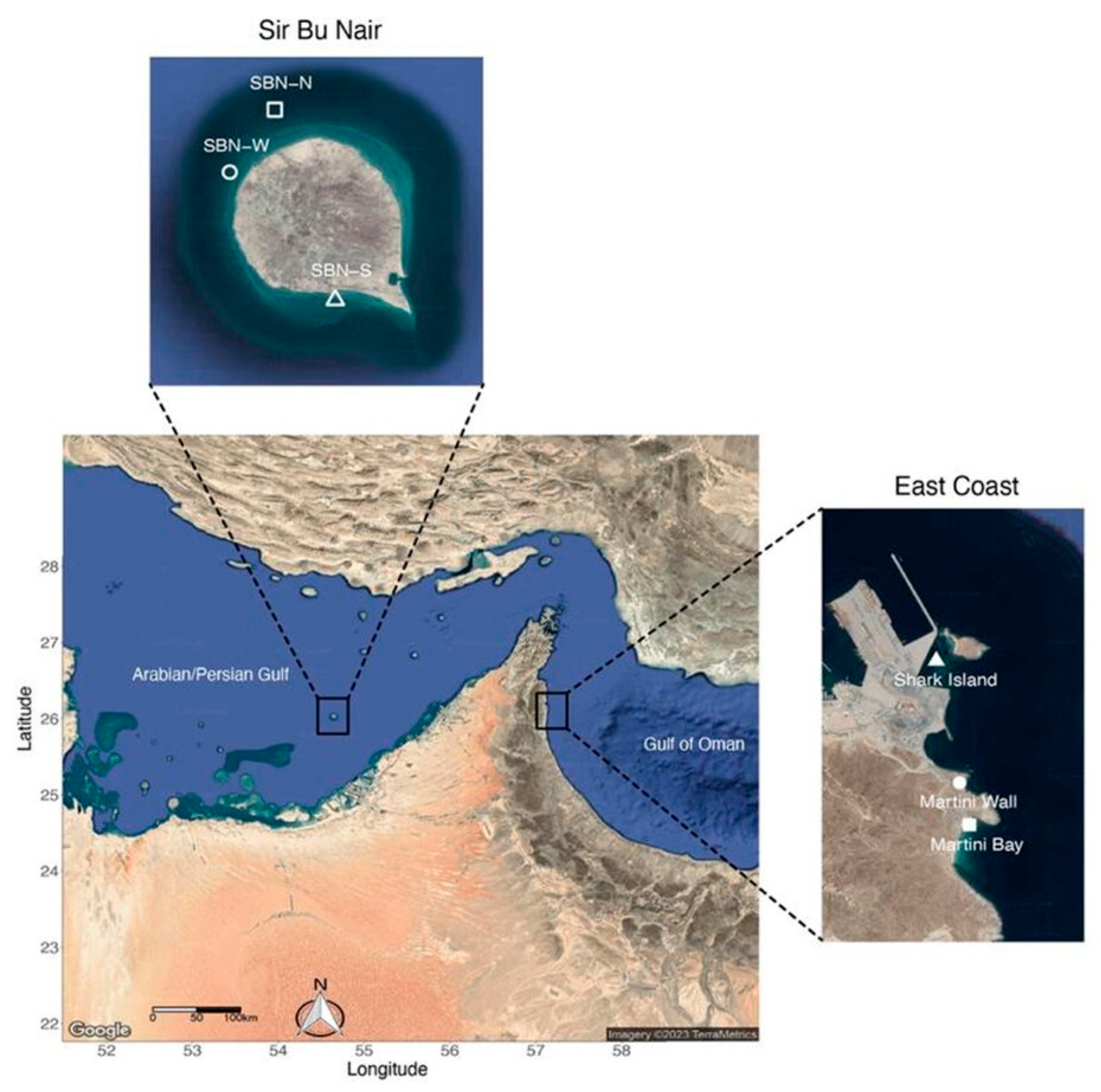
2.1. Study Sites
2.2. Data Collection
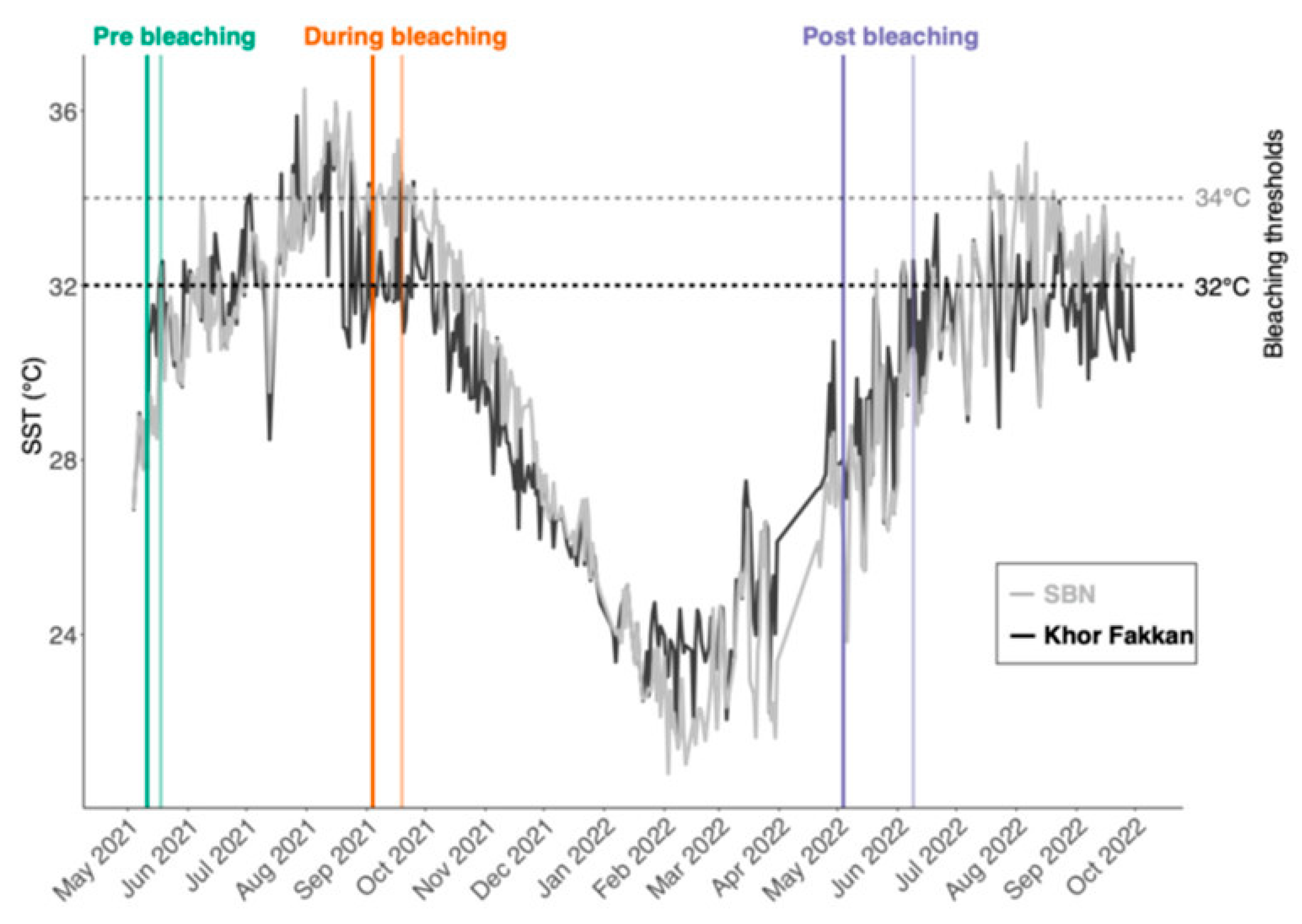
2.3. Temperature Data
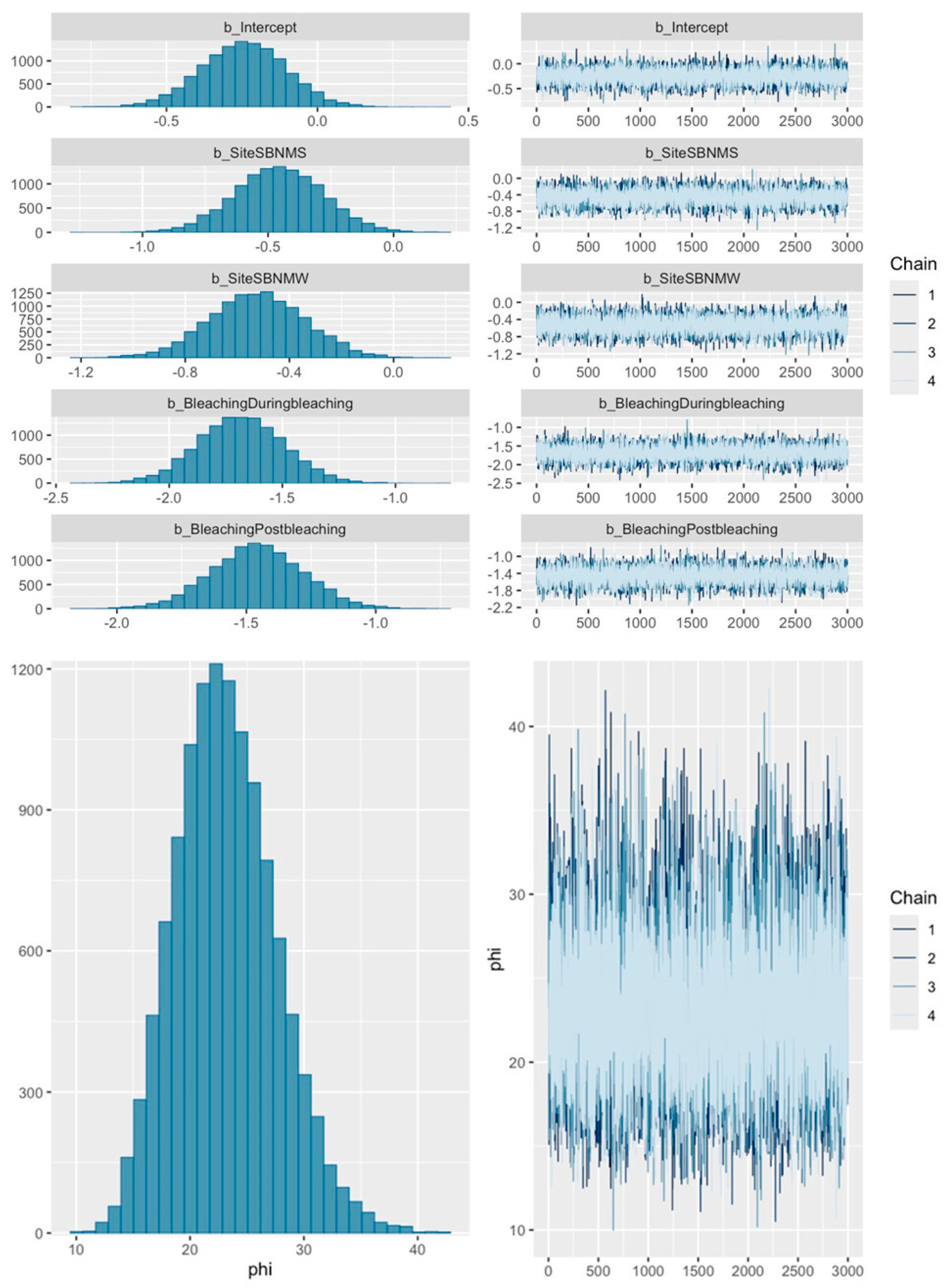
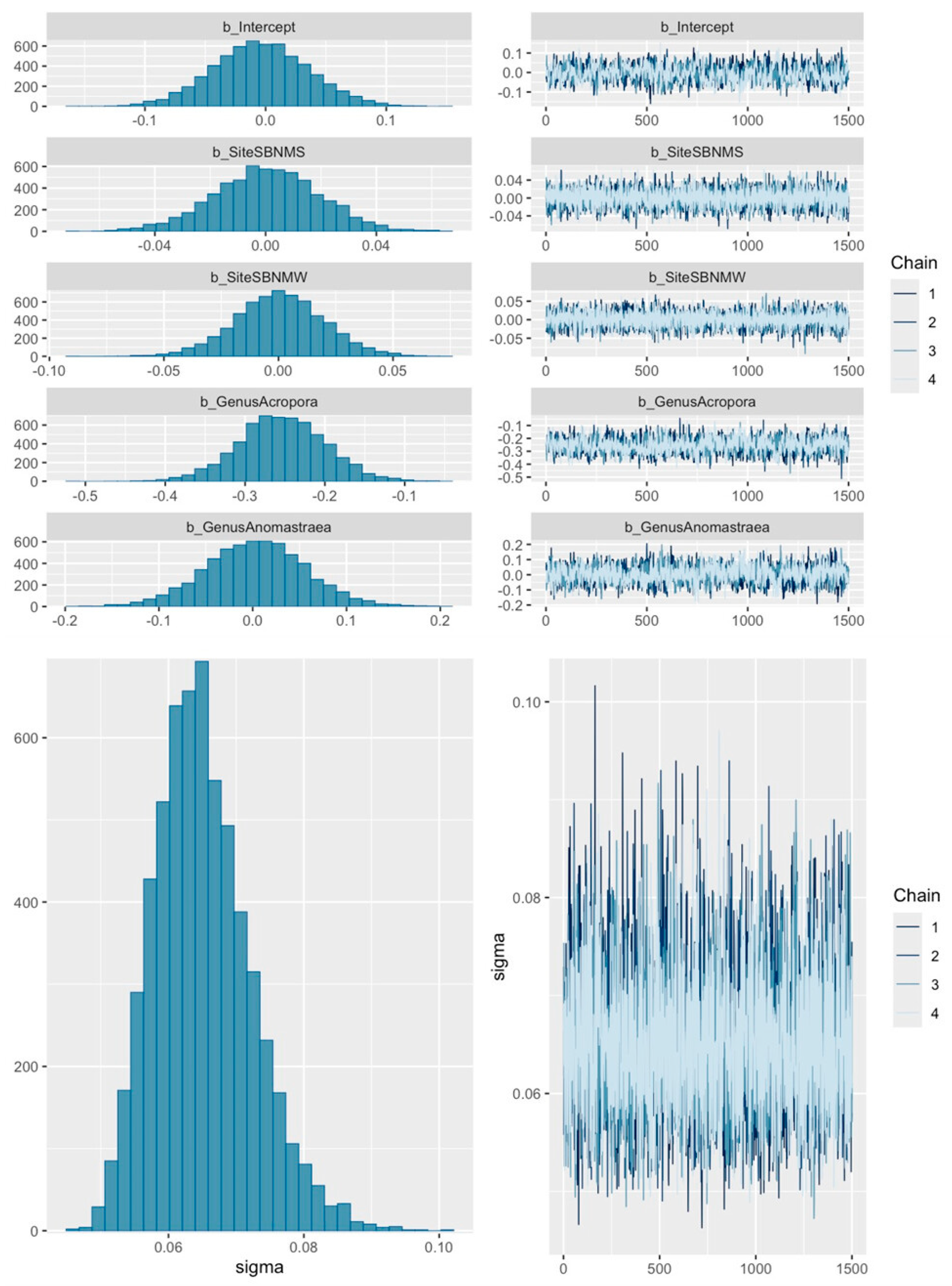
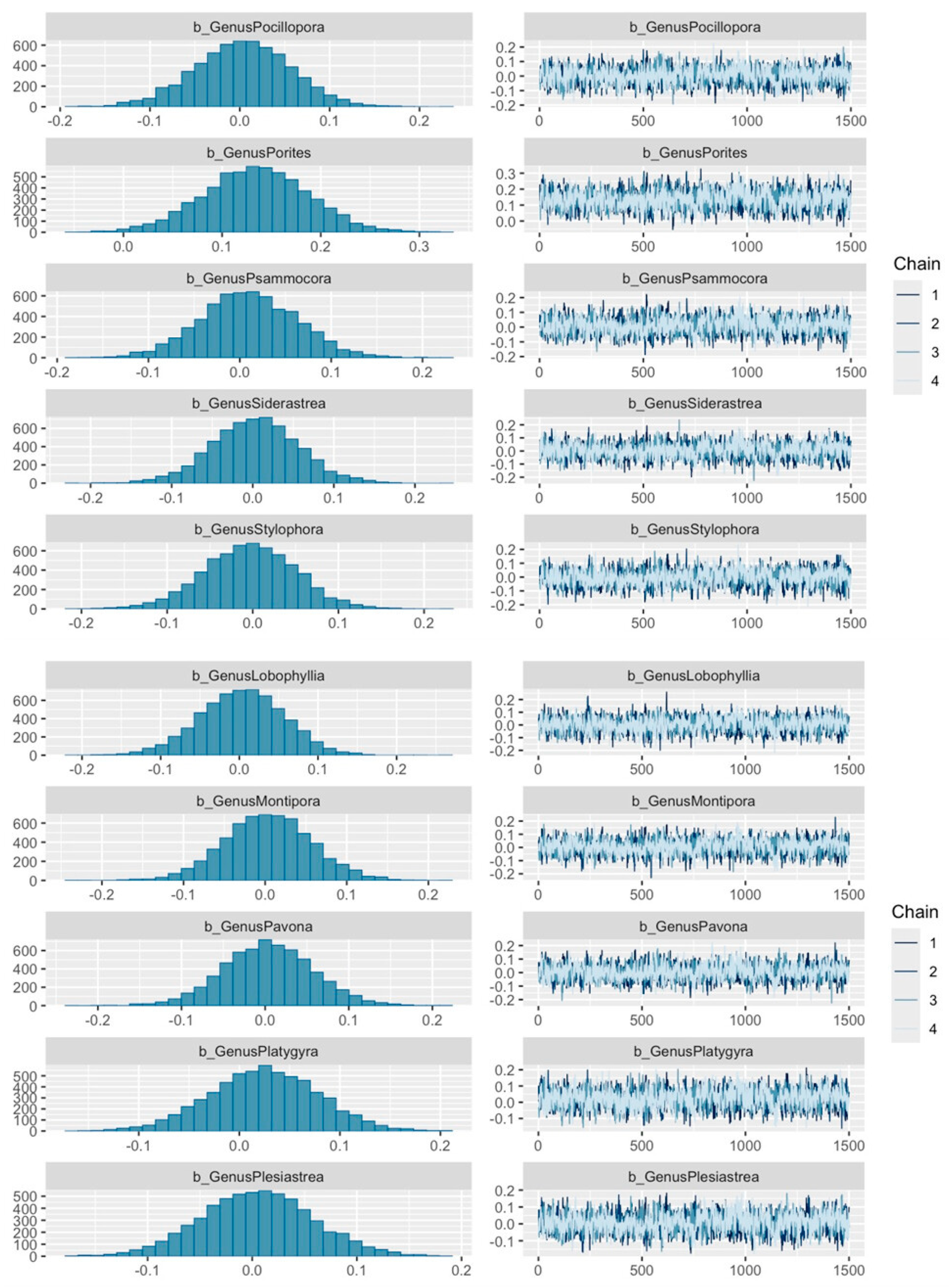
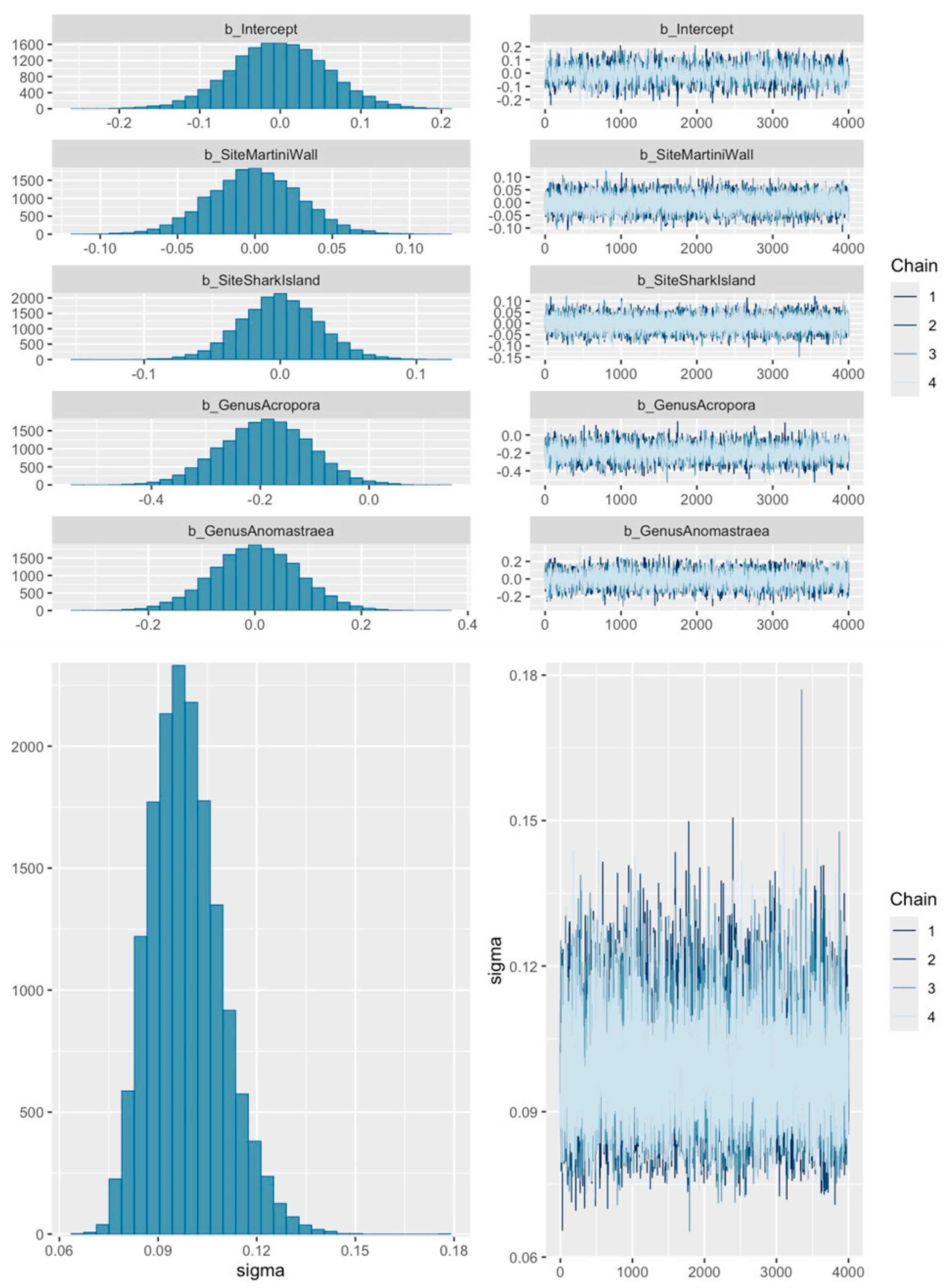
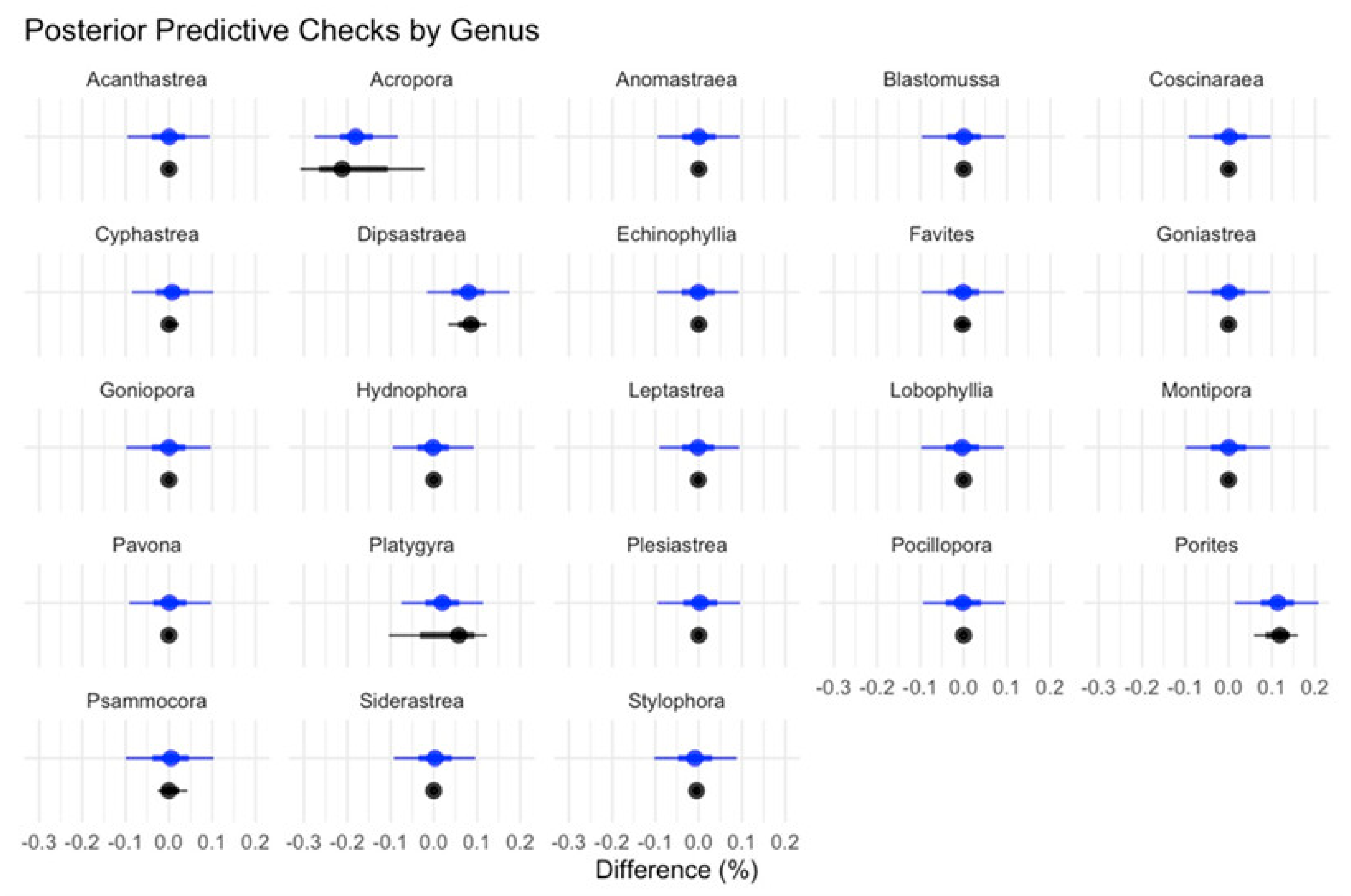
2.4. Analyses
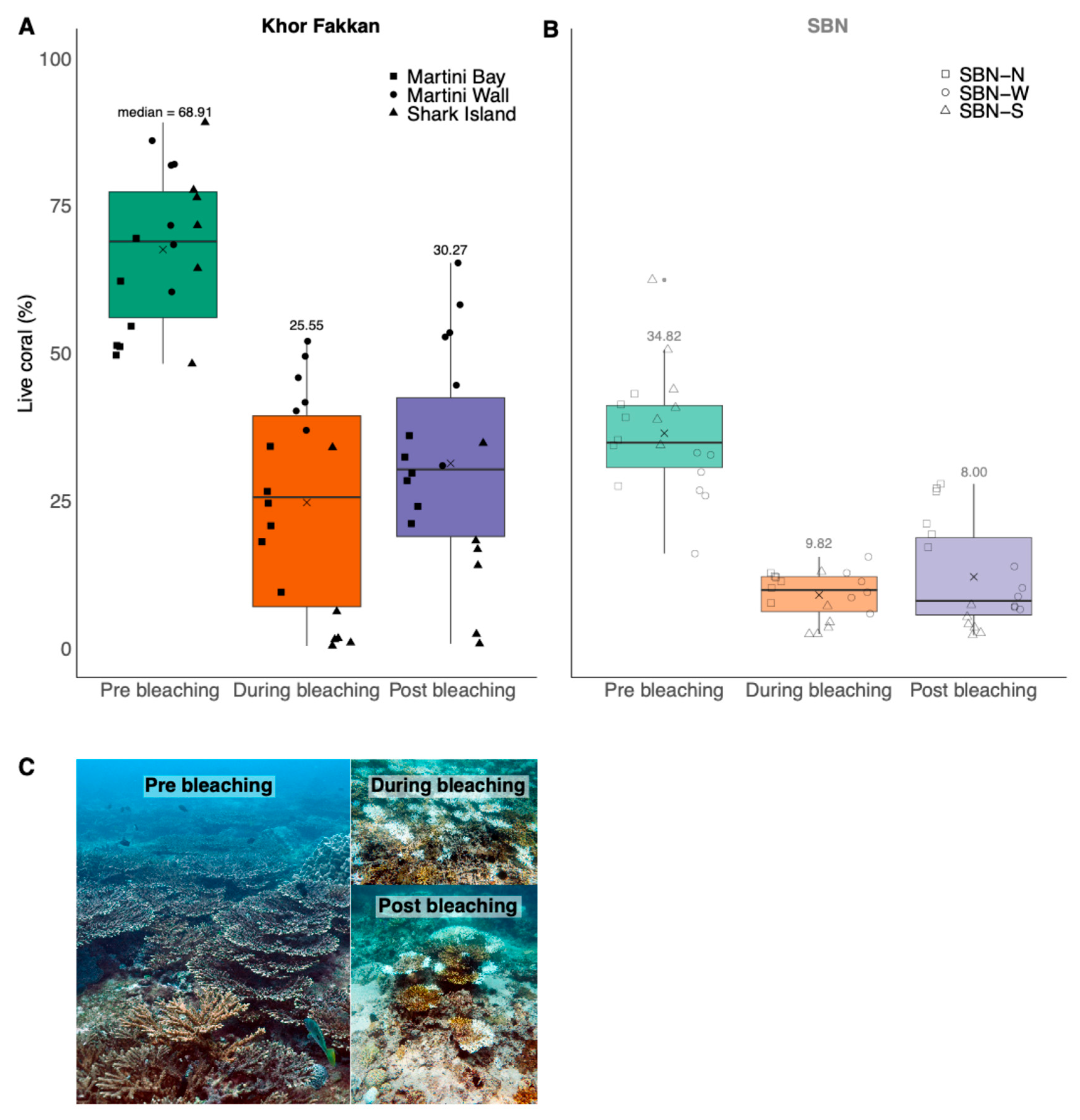
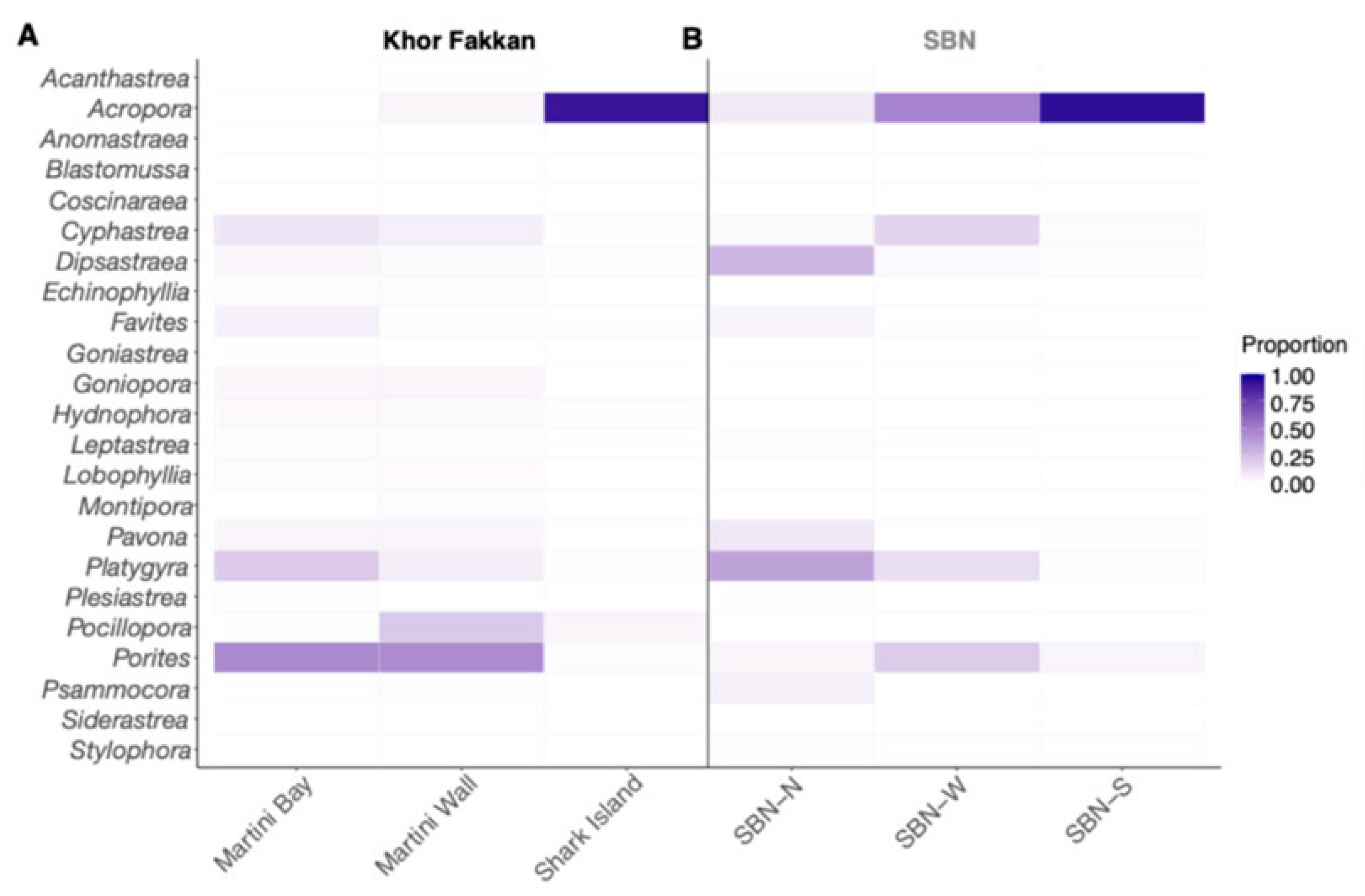
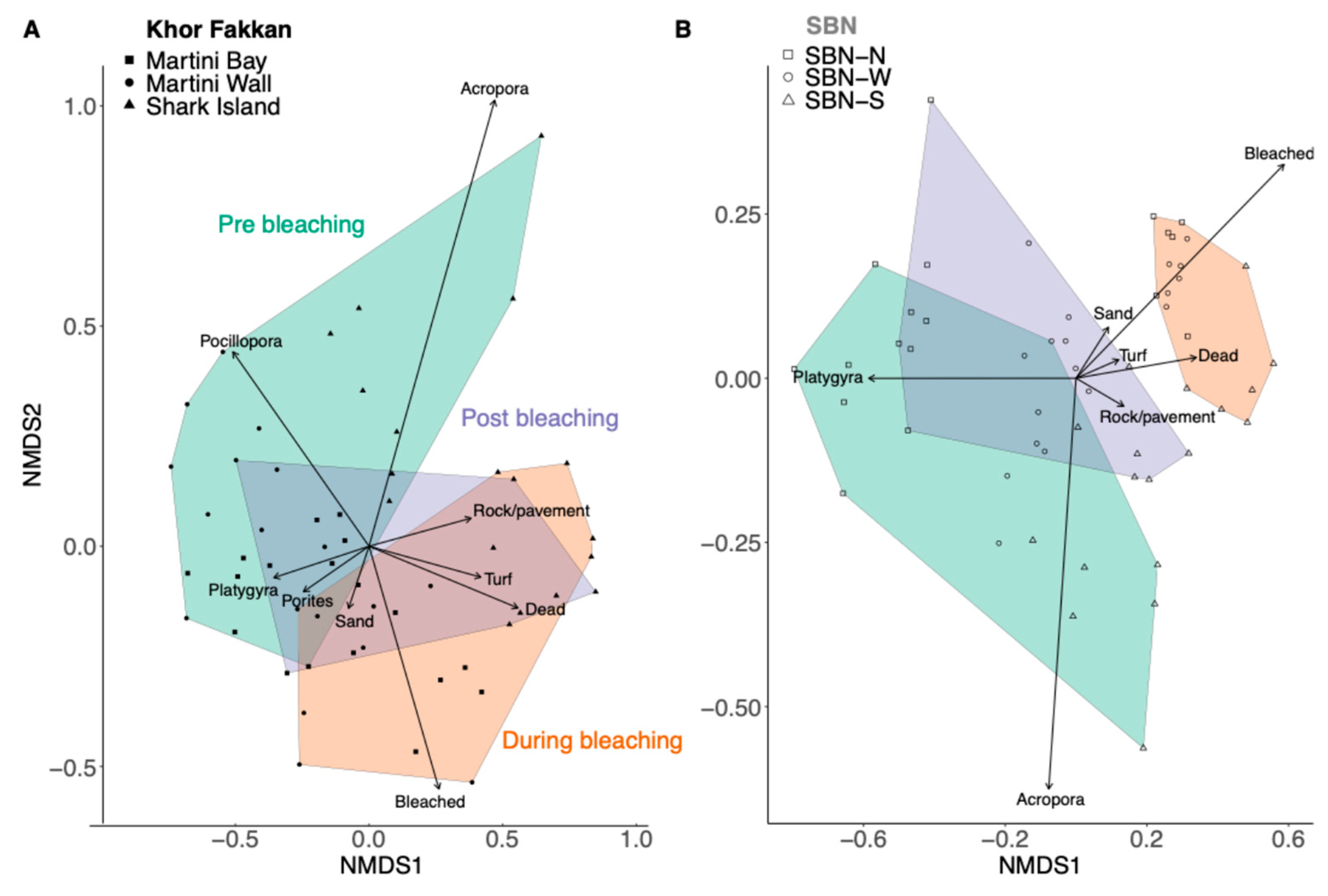
3. Results
4. Discussion
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| SBN | Sir Bu Nair |
| KF | Khor Fakkan |
| UAE | United Arab Emirates |
| HAB | Harmful algal bloom |
| DHW | Degree heating weeks |
| SST | Sea surface temperature |
| PERMANOVA | Permutational multivariate analysis of variance |
| SIMPER | Similarity percentage analysis |
| PERMDISP | Permutational multivariate analysis of dispersion |
| CI | Confidence interval |
| SE | Standard error |
| nMDS | Non-metric multidimensional scaling |
Appendix A
| Site | Transect | Date | % Live Coral |
|---|---|---|---|
| MartiniBay | A | 11 May 2021 | 62.2 |
| MartiniBay | B | 11 May 2021 | 69.5 |
| MartiniBay | C | 11 May 2021 | 51.3 |
| MartiniBay | D | 11 May 2021 | 51.1 |
| MartiniBay | E | 11 May 2021 | 54.5 |
| MartiniBay | F | 11 May 2021 | 49.6 |
| MartiniBay | A | 4 September 2021 | 9.5 |
| MartiniBay | B | 4 September 2021 | 18.0 |
| MartiniBay | C | 4 September 2021 | 20.7 |
| MartiniBay | D | 4 September 2021 | 24.5 |
| MartiniBay | E | 4 September 2021 | 26.5 |
| MartiniBay | F | 4 September 2021 | 34.2 |
| MartiniBay | A | 4 May 2022 | 36.0 |
| MartiniBay | B | 4 May 2022 | 29.6 |
| MartiniBay | C | 4 May 2022 | 28.4 |
| MartiniBay | D | 4 May 2022 | 21.1 |
| MartiniBay | E | 4 May 2022 | 32.4 |
| MartiniBay | F | 4 May 2022 | 24.0 |
| MartiniWall | A | 11 May 2021 | 71.6 |
| MartiniWall | B | 11 May 2021 | 86.0 |
| MartiniWall | C | 11 May 2021 | 82.0 |
| MartiniWall | D | 11 May 2021 | 81.8 |
| MartiniWall | E | 11 May 2021 | 68.4 |
| MartiniWall | F | 11 May 2021 | 60.4 |
| MartiniWall | A | 4 September 2021 | 36.9 |
| MartiniWall | B | 4 September 2021 | 45.8 |
| MartiniWall | C | 4 September 2021 | 52.0 |
| MartiniWall | D | 4 September 2021 | 40.2 |
| MartiniWall | E | 4 September 2021 | 49.5 |
| MartiniWall | F | 4 September 2021 | 41.6 |
| MartiniWall | A | 4 May 2022 | 44.5 |
| MartiniWall | B | 4 May 2022 | 65.3 |
| MartiniWall | C | 4 May 2022 | 53.5 |
| MartiniWall | D | 4 May 2022 | 30.9 |
| MartiniWall | E | 4 May 2022 | 52.7 |
| MartiniWall | F | 4 May 2022 | 58.2 |
| SBN-N | A | 18 May 2021 | 34.4 |
| SBN-N | B | 18 May 2022 | 27.5 |
| SBN-N | C | 18 May 2022 | 35.3 |
| SBN-N | D | 18 May 2022 | 43.1 |
| SBN-N | E | 18 May 2022 | 39.1 |
| SBN-N | F | 18 May 2022 | 41.3 |
| SBN-N | A | 19 September 2021 | 12.0 |
| SBN-N | B | 19 September 2021 | 12.2 |
| SBN-N | C | 19 September 2021 | 11.3 |
| SBN-N | D | 19 September 2021 | 10.2 |
| SBN-N | E | 19 September 2021 | 7.6 |
| SBN-N | F | 19 September 2021 | 12.7 |
| SBN-N | A | 9 June 2022 | 27.1 |
| SBN-N | B | 9 June 2022 | 27.8 |
| SBN-N | C | 9 June 2022 | 21.1 |
| SBN-N | D | 9 June 2022 | 19.3 |
| SBN-N | E | 9 June 2022 | 17.1 |
| SBN-N | F | 9 June 2022 | 26.5 |
| SBN-S | A | 18 May 2022 | 50.5 |
| SBN-S | B | 18 May 2022 | 43.8 |
| SBN-S | C | 18 May 2022 | 62.4 |
| SBN-S | D | 18 May 2022 | 38.7 |
| SBN-S | E | 18 May 2022 | 40.7 |
| SBN-S | F | 18 May 2022 | 34.4 |
| SBN-S | A | 19 September 2021 | 2.4 |
| SBN-S | B | 19 September 2021 | 2.4 |
| SBN-S | C | 19 September 2021 | 4.4 |
| SBN-S | D | 19 September 2021 | 7.1 |
| SBN-S | E | 19 September 2021 | 12.9 |
| SBN-S | F | 19 September 2021 | 3.5 |
| SBN-S | A | 9 June 2022 | 5.3 |
| SBN-S | B | 9 June 2022 | 7.23 |
| SBN-S | C | 9 June 2022 | 4.0 |
| SBN-S | D | 9 June 2022 | 2.5 |
| SBN-S | E | 9 June 2022 | 3.5 |
| SBN-S | F | 9 June 2022 | 2.2 |
| SBN-W | A | 18 May 2022 | 16.0 |
| SBN-W | B | 18 May 2022 | 26.7 |
| SBN-W | C | 18 May 2022 | 25.8 |
| SBN-W | D | 18 May 2022 | 32.7 |
| SBN-W | E | 18 May 2022 | 33.1 |
| SBN-W | F | 18 May 2022 | 29.8 |
| SBN-W | A | 19 September 2021 | 11.3 |
| SBN-W | B | 19 September 2021 | 5.8 |
| SBN-W | C | 19 September 2021 | 12.7 |
| SBN-W | D | 19 September 2021 | 8.5 |
| SBN-W | E | 19 September 2021 | 9.5 |
| SBN-W | F | 19 September 2021 | 15.5 |
| SBN-W | A | 9 June 2022 | 6.9 |
| SBN-W | B | 9 June 2022 | 8.7 |
| SBN-W | C | 9 June 2022 | 10.2 |
| SBN-W | D | 9 June 2022 | 6.5 |
| SBN-W | E | 9 June 2022 | 7.1 |
| SBN-W | F | 9 June 2022 | 13.8 |
| SharkIsland | A | 11 May 2021 | 48.2 |
| SharkIsland | B | 11 May 2021 | 76.4 |
| SharkIsland | C | 11 May 2021 | 89.1 |
| SharkIsland | D | 11 May 2021 | 64.4 |
| SharkIsland | E | 11 May 2021 | 71.6 |
| SharkIsland | F | 11 May 2021 | 77.6 |
| SharkIsland | A | 4 September 2021 | 1.5 |
| SharkIsland | B | 4 September 2021 | 0.9 |
| SharkIsland | C | 4 September 2021 | 6.2 |
| SharkIsland | D | 4 September 2021 | 34.0 |
| SharkIsland | E | 4 September 2021 | 1.6 |
| SharkIsland | F | 4 September 2021 | 0.4 |
| SharkIsland | A | 4 May 2022 | 16.7 |
| SharkIsland | B | 4 May 2022 | 2.4 |
| SharkIsland | C | 4 May 2022 | 0.7 |
| SharkIsland | D | 4 May 2022 | 14.0 |
| SharkIsland | E | 4 May 2022 | 34.7 |
| SharkIsland | F | 4 May 2022 | 18.2 |
| Khor Fakkan | SBN | |||||
|---|---|---|---|---|---|---|
| Pre-Bleaching | During Bleaching | Post-Bleaching | Pre-Bleaching | During Bleaching | Post-Bleaching | |
| All coral | ||||||
| Mean (%) | 67.54 | 24.67 | 31.29 | 36.41 | 8.99 | 12.05 |
| SE | 3.09 | 4.25 | 4.30 | 2.43 | 0.94 | 2.09 |
| Median (%) | 68.91 | 25.55 | 30.27 | 34.82 | 9.82 | 8.00 |
| IQR | 21.32 | 32.36 | 23.50 | 10.59 | 6.00 | 13.14 |
| Acropora | ||||||
| Mean (%) | 22.49 | 2.01 | 2.82 | 19.40 | 0.02 | 1.74 |
| SE | 7.49 | 1.69 | 1.52 | 4.33 | 0.02 | 0.41 |
| Median (%) | 1.36 | 0.00 | 0.00 | 11.55 | 0.00 | 1.27 |
| IQR | 56.77 | 0.14 | 0.14 | 27.77 | 0.00 | 3.00 |
| Priors for live coral models | Intercept | student_t (3, 0, 2.5) |
| b | normal (0, 10) | |
| phi | gamma (2, 0.1) | |
| Priors for proportional contribution of coral genera models | Intercept | student_t (3, 0, 2.5) |
| b | normal (0, 1) | |
| sigma | student_t (3, 0, 2.5) |
| Contribution to Average Between-Group Dissimilarity | SD | Average to SD Ratio | Average Post-Bleaching | Average Pre-Bleaching | Cumulative Contribution | p | |
|---|---|---|---|---|---|---|---|
| Acropora | 0.12 | 0.15 | 0.79 | 2.82 | 22.49 | 0.20 | 0.002 |
| Porites | 0.08 | 0.06 | 1.42 | 16.49 | 19.80 | 0.34 | 0.053 |
| Rock/pavement | 0.07 | 0.06 | 1.17 | 20.47 | 7.92 | 0.47 | 0.003 |
| Turf | 0.06 | 0.05 | 1.22 | 20.37 | 11.51 | 0.57 | 1.000 |
| Pocillopora | 0.03 | 0.05 | 0.74 | 1.44 | 6.53 | 0.62 | 0.004 |
| Platygyra | 0.03 | 0.03 | 1.16 | 6.66 | 5.80 | 0.68 | 0.128 |
| Sand | 0.03 | 0.02 | 1.33 | 7.37 | 6.67 | 0.73 | 0.973 |
| Dead | 0.03 | 0.03 | 0.89 | 6.27 | 0.62 | 0.78 | 0.335 |
| Contribution to Average Between-Group Dissimilarity | SD | Average to SD Ratio | Average Post-Bleaching | Average Pre-Bleaching | Cumulative Contribution | p | |
|---|---|---|---|---|---|---|---|
| Turf | 0.10 | 0.04 | 2.21 | 42.68 | 23.39 | 0.23 | 0.001 |
| Acropora | 0.09 | 0.09 | 1.02 | 1.74 | 19.40 | 0.45 | 0.001 |
| Sand | 0.04 | 0.03 | 1.41 | 18.51 | 13.82 | 0.56 | 0.548 |
| Rock/Pavement | 0.04 | 0.02 | 1.47 | 13.46 | 13.92 | 0.64 | 0.024 |
| Platygyra | 0.03 | 0.03 | 0.99 | 2.62 | 5.84 | 0.71 | 0.003 |
| Region | Site | Transect | Date | Bleaching Period | nMDS1 | nMDS2 |
|---|---|---|---|---|---|---|
| KFakkan | MartiniBay | A | 4 May 2022 | Post-bleaching | −0.04093 | −0.08456 |
| KFakkan | MartiniBay | A | 4 September 2021 | During-bleaching | 0.42163 | −0.32773 |
| KFakkan | MartiniBay | A | 11 May 2021 | Pre-bleaching | −0.22845 | −0.26787 |
| KFakkan | MartiniBay | B | 4 May 2022 | Post-bleaching | −0.10945 | 0.07302 |
| KFakkan | MartiniBay | B | 4 September 2021 | During-bleaching | 0.35967 | −0.27389 |
| KFakkan | MartiniBay | B | 11 May 2021 | Pre-bleaching | −0.50130 | −0.19517 |
| KFakkan | MartiniBay | C | 4 May 2022 | Post-bleaching | −0.13797 | −0.03872 |
| KFakkan | MartiniBay | C | 4 September 2021 | During-bleaching | 0.17651 | −0.46702 |
| KFakkan | MartiniBay | C | 11 May 2021 | Pre-bleaching | −0.67839 | −0.06111 |
| KFakkan | MartiniBay | D | 4 May 2022 | Post-bleaching | −0.09112 | 0.01320 |
| KFakkan | MartiniBay | D | 4 September 2021 | During-bleaching | 0.26743 | −0.30520 |
| KFakkan | MartiniBay | D | 11 May 2021 | Pre-bleaching | −0.46953 | −0.03019 |
| KFakkan | MartiniBay | E | 4 May 2022 | Post-bleaching | −0.19584 | 0.05966 |
| KFakkan | MartiniBay | E | 4 September 2021 | During-bleaching | 0.09791 | −0.15031 |
| KFakkan | MartiniBay | E | 11 May 2021 | Pre-bleaching | −0.49088 | −0.07052 |
| KFakkan | MartiniBay | F | 4 May 2022 | Post-bleaching | −0.30456 | −0.29475 |
| KFakkan | MartiniBay | F | 4 September 2021 | During-bleaching | −0.05755 | −0.24144 |
| KFakkan | MartiniBay | F | 11 May 2021 | Pre-bleaching | −0.37077 | −0.04372 |
| KFakkan | MartiniWall | A | 4 May 2022 | Post-bleaching | −0.16605 | −0.00019 |
| KFakkan | MartiniWall | A | 4 September 2021 | During-bleaching | 0.38643 | −0.53470 |
| KFakkan | MartiniWall | A | 11 May 2021 | Pre-bleaching | −0.60253 | 0.07185 |
| KFakkan | MartiniWall | B | 4 May 2022 | Post-bleaching | −0.34583 | 0.17246 |
| KFakkan | MartiniWall | B | 4 September 2021 | During-bleaching | −0.02103 | −0.23050 |
| KFakkan | MartiniWall | B | 11 May 2021 | Pre-bleaching | −0.68149 | 0.32139 |
| KFakkan | MartiniWall | C | 4 May 2022 | Post-bleaching | 0.01809 | −0.13257 |
| KFakkan | MartiniWall | C | 4 September 2021 | During-bleaching | −0.25751 | −0.49608 |
| KFakkan | MartiniWall | C | 11 May 2021 | Pre-bleaching | −0.74207 | 0.17668 |
| KFakkan | MartiniWall | D | 4 May 2022 | Post-bleaching | 0.23084 | −0.08640 |
| KFakkan | MartiniWall | D | 4 September 2021 | During-bleaching | −0.23995 | −0.37852 |
| KFakkan | MartiniWall | D | 11 May 2021 | Pre-bleaching | −0.41074 | 0.26625 |
| KFakkan | MartiniWall | E | 4 May 2022 | Post-bleaching | −0.49749 | 0.19518 |
| KFakkan | MartiniWall | E | 4 September 2021 | During-bleaching | −0.26770 | −0.14489 |
| KFakkan | MartiniWall | E | 11 May 2021 | Pre-bleaching | −0.54842 | 0.43880 |
| KFakkan | MartiniWall | F | 4 May 2022 | Post-bleaching | −0.40183 | 0.03668 |
| KFakkan | MartiniWall | F | 4 September 2021 | During-bleaching | −0.19207 | −0.15954 |
| KFakkan | MartiniWall | F | 11 May 2021 | Pre-bleaching | −0.68194 | −0.16577 |
| KFakkan | SharkIsland | A | 4 May 2022 | Post-bleaching | 0.52493 | −0.16985 |
| KFakkan | SharkIsland | A | 4 September 2021 | During-bleaching | 0.70057 | −0.10429 |
| KFakkan | SharkIsland | A | 11 May 2021 | Pre-bleaching | 0.10343 | 0.26061 |
| KFakkan | SharkIsland | B | 4 May 2022 | Post-bleaching | 0.56568 | −0.14220 |
| KFakkan | SharkIsland | B | 4 September 2021 | During-bleaching | 0.46473 | −0.00380 |
| KFakkan | SharkIsland | B | 11 May 2021 | Pre-bleaching | −0.02345 | 0.35376 |
| KFakkan | SharkIsland | C | 4 May 2022 | Post-bleaching | 0.85125 | −0.02916 |
| KFakkan | SharkIsland | C | 4 September 2021 | During-bleaching | 0.74146 | 0.18808 |
| KFakkan | SharkIsland | C | 11 May 2021 | Pre-bleaching | 0.64136 | 0.93526 |
| KFakkan | SharkIsland | D | 4 May 2022 | Post-bleaching | 0.54146 | 0.15496 |
| KFakkan | SharkIsland | D | 4 September 2021 | During-bleaching | 0.48121 | 0.16913 |
| KFakkan | SharkIsland | D | 11 May 2021 | Pre-bleaching | 0.53895 | 0.56185 |
| KFakkan | SharkIsland | E | 4 May 2022 | Post-bleaching | 0.08567 | 0.16556 |
| KFakkan | SharkIsland | E | 4 September 2021 | During-bleaching | 0.83698 | 0.01648 |
| KFakkan | SharkIsland | E | 11 May 2021 | Pre-bleaching | −0.03806 | 0.54061 |
| KFakkan | SharkIsland | F | 4 May 2022 | Post-bleaching | 0.07553 | 0.10322 |
| KFakkan | SharkIsland | F | 4 September 2021 | During-bleaching | 0.82919 | −0.12683 |
| KFakkan | SharkIsland | F | 11 May 2021 | Pre-bleaching | −0.14605 | 0.48279 |
| SBN | SBN-N | A | 9 June 2022 | Post-bleaching | −0.50038 | 0.05314 |
| SBN | SBN-N | A | 18 May 2022 | Pre-bleaching | −0.65776 | −0.17509 |
| SBN | SBN-N | A | 19 September 2021 | During-bleaching | 0.29908 | 0.23780 |
| SBN | SBN-N | B | 9 June 2022 | Post-bleaching | −0.46689 | 0.04453 |
| SBN | SBN-N | B | 18 May 2022 | Pre-bleaching | −0.64264 | 0.02034 |
| SBN | SBN-N | B | 19 September 2021 | During-bleaching | 0.27225 | 0.21527 |
| SBN | SBN-N | C | 9 June 2022 | Post-bleaching | −0.42271 | 0.08718 |
| SBN | SBN-N | C | 18 May 2022 | Pre-bleaching | −0.56734 | 0.17420 |
| SBN | SBN-N | C | 19 September 2021 | During-bleaching | 0.25973 | 0.22167 |
| SBN | SBN-N | D | 9 June 2022 | Post-bleaching | −0.42031 | 0.17261 |
| SBN | SBN-N | D | 18 May 2022 | Pre-bleaching | −0.79580 | 0.01437 |
| SBN | SBN-N | D | 19 September 2021 | During-bleaching | 0.21849 | 0.24682 |
| SBN | SBN-N | E | 9 June 2022 | Post-bleaching | −0.40966 | 0.42363 |
| SBN | SBN-N | E | 18 May 2022 | Pre-bleaching | −0.46579 | 0.10098 |
| SBN | SBN-N | E | 19 September 2021 | During-bleaching | 0.31492 | 0.06407 |
| SBN | SBN-N | F | 9 June 2022 | Post-bleaching | −0.47427 | −0.07923 |
| SBN | SBN-N | F | 18 May 2022 | Pre-bleaching | −0.65548 | −0.03642 |
| SBN | SBN-N | F | 19 September 2021 | During-bleaching | 0.22703 | 0.12624 |
| SBN | SBN-S | A | 9 June 2022 | Post-bleaching | 0.15020 | 0.01745 |
| SBN | SBN-S | A | 18 May 2022 | Pre-bleaching | 0.02372 | −0.28821 |
| SBN | SBN-S | A | 19 September 2021 | During-bleaching | 0.47932 | 0.17071 |
| SBN | SBN-S | B | 9 June 2022 | Post-bleaching | 0.00528 | −0.07488 |
| SBN | SBN-S | B | 18 May 2022 | Pre-bleaching | −0.12217 | −0.24707 |
| SBN | SBN-S | B | 19 September 2021 | During-bleaching | 0.31285 | −0.01554 |
| SBN | SBN-S | C | 9 June 2022 | Post-bleaching | 0.20635 | −0.15444 |
| SBN | SBN-S | C | 18 May 2022 | Pre-bleaching | 0.19007 | −0.56324 |
| SBN | SBN-S | C | 19 September 2021 | During-bleaching | 0.55732 | 0.02324 |
| SBN | SBN-S | D | 9 June 2022 | Post-bleaching | 0.31806 | −0.11424 |
| SBN | SBN-S | D | 18 May 2022 | Pre-bleaching | −0.00867 | −0.36275 |
| SBN | SBN-S | D | 19 September 2021 | During-bleaching | 0.41066 | −0.04732 |
| SBN | SBN-S | E | 9 June 2022 | Post-bleaching | 0.16554 | −0.15057 |
| SBN | SBN-S | E | 18 May 2022 | Pre-bleaching | 0.23022 | −0.28422 |
| SBN | SBN-S | E | 19 September 2021 | During-bleaching | 0.49717 | −0.01786 |
| SBN | SBN-S | F | 9 June 2022 | Post-bleaching | 0.17292 | −0.11533 |
| SBN | SBN-S | F | 18 May 2022 | Pre-bleaching | 0.22206 | −0.34363 |
| SBN | SBN-S | F | 19 September 2021 | During-bleaching | 0.48369 | −0.06708 |
| SBN | SBN-W | A | 9 June 2022 | Post-bleaching | −0.00112 | 0.01482 |
| SBN | SBN-W | A | 18 May 2022 | Pre-bleaching | −0.06942 | 0.05576 |
| SBN | SBN-W | A | 19 September 2021 | During-bleaching | 0.31357 | 0.21244 |
| SBN | SBN-W | B | 9 June 2022 | Post-bleaching | −0.13362 | 0.20535 |
| SBN | SBN-W | B | 18 May 2022 | Pre-bleaching | −0.14590 | 0.03361 |
| SBN | SBN-W | B | 19 September 2021 | During-bleaching | 0.29087 | 0.15206 |
| SBN | SBN-W | C | 9 June 2022 | Post-bleaching | −0.02964 | 0.05658 |
| SBN | SBN-W | C | 18 May 2022 | Pre-bleaching | −0.10662 | −0.05202 |
| SBN | SBN-W | C | 19 September 2021 | During-bleaching | 0.26281 | 0.17372 |
| SBN | SBN-W | D | 9 June 2022 | Post-bleaching | −0.02082 | 0.09284 |
| SBN | SBN-W | D | 18 May 2022 | Pre-bleaching | −0.21795 | −0.25139 |
| SBN | SBN-W | D | 19 September 2021 | During-bleaching | 0.25482 | 0.10872 |
| SBN | SBN-W | E | 9 June 2022 | Post-bleaching | 0.03573 | −0.02000 |
| SBN | SBN-W | E | 18 May 2022 | Pre-bleaching | −0.08847 | −0.11159 |
| SBN | SBN-W | E | 19 September 2021 | During-bleaching | 0.29504 | 0.17115 |
| SBN | SBN-W | F | 9 June 2022 | Post-bleaching | −0.11111 | −0.10004 |
| SBN | SBN-W | F | 18 May 2022 | Pre-bleaching | −0.19426 | −0.14889 |
| SBN | SBN-W | F | 19 September 2021 | During-bleaching | 0.25906 | 0.12972 |





References
- Foster, K.; Foster, G.; Tourenq, C.; Shuriqi, M. Shifts in coral community structures following cyclone and red tide disturbances within the Gulf of Oman (United Arab Emirates). Mar. Biol. 2011, 158, 955–968. [Google Scholar] [CrossRef]
- Burt, J. Coral reefs of the Emirates. In A Natural History of the Emirates; Burt, J., Ed.; Springer Nature: Cham, Switzerland, 2023; pp. 325–351. [Google Scholar] [CrossRef]
- Burt, J. From Phoenix to Sisyphus: Climate change impacts and intervention strategies for Arabian Gulf coral reefs. J. Coast. Res. 2024, 113, 839–845. [Google Scholar] [CrossRef]
- Riegl, B. Effects of the 1996 and 1998 positive sea-surface temperature anomalies on corals, coral diseases and fish in the Arabian Gulf (Dubai, UAE). Mar. Biol. 2002, 140, 29–40. [Google Scholar] [CrossRef]
- Burt, J.; Bartholomew, A.; Usseglio, P. Recovery of corals a decade after a bleaching event in Dubai, United Arab Emirates. Mar. Biol. 2008, 154, 27–36. [Google Scholar] [CrossRef]
- Riegl, B.; Purkis, S. Coral population dynamics across consecutive mass mortality events. Glob. Change Biol. 2015, 21, 3995–4005. [Google Scholar] [CrossRef]
- Burt, J.; Paparella, F.; Al-Mansoori, N.; Al-Mansoori, A.; Al-Jailani, H. Causes and consequences of the 2017 coral bleaching event in the southern Persian/Arabian Gulf. Coral Reefs 2019, 38, 567–589. [Google Scholar] [CrossRef]
- Bleuel, J.; Pennino, M.; Longo, G. Coral distribution and bleaching vulnerability areas in Southwestern Atlantic under ocean warming. Sci. Rep. 2021, 11, 12833. [Google Scholar] [CrossRef]
- Mellin, C.; Brown, S.; Cantin, N.; Klein-Salas, E.; Mouillot, D.; Heron, S.; Fordham, D. Cumulative risk of future bleaching for the world’s coral reefs. Sci. Adv. 2024, 10, eadn9660. [Google Scholar] [CrossRef]
- McClanahan, T.; Darling, E.; Maina, J.; Muthiga, N.; D’Agata, S.; Leblond, J.; Arthur, R.; Jupiter, S.; Wilson, S.; Mangubhai, S.; et al. Highly variable taxa-specific coral bleaching responses to thermal stresses. Mar. Ecol. Prog. Ser. 2020, 648, 135–151. [Google Scholar] [CrossRef]
- van Woesik, R.; Shlesinger, T.; Grottoli, A.; Toonen, R.; Vega Thurber, R.; Warner, M.; Marie Hulver, A.; Chapron, L.; McLachlan, R.; Albright, R.; et al. Coral-bleaching responses to climate change across biological scales. Glob. Change Biol. 2022, 28, 4229–4250. [Google Scholar] [CrossRef] [PubMed]
- Baum, J.; Claar, D.; Tietjen, K.; Magel, J.; Maucieri, D.; Cobb, K.; McDevitt-Irwin, J. Transformation of coral communities subjected to an unprecedented heatwave is modulated by local disturbance. Sci. Adv. 2023, 9, eabq5615. [Google Scholar] [CrossRef]
- Gonzalez, K.; Daraghmeh, N.; Lozano-Cortés, D.; Benzoni, F.; Berumen, M.; Carvalho, S. Differential spatio-temporal responses of Red Sea coral reef benthic communities to a mass bleaching event. Sci. Rep. 2024, 14, 24229. [Google Scholar] [CrossRef]
- Carpenter, K.; Abrar, M.; Aeby, G.; Aronson, R.; Banks, S.; Bruckner, A.; Chiribog, A.; Cortes, J.; Delbeek, J.; DeVantier, L.; et al. One-third of reef-building corals face elevated extinction risk from climate change and local impacts. Science 2008, 321, 560–563. [Google Scholar] [CrossRef]
- Watt-Pringle, R.; Smith, D.; Ambo-Rappe, R.; Lamont, T.; Jompa, J. Suppressed recovery of functionally important branching Acropora drives coral community composition changes following mass bleaching in Indonesia. Coral Reefs 2022, 41, 1337–1350. [Google Scholar] [CrossRef]
- Vytopil, E.; Willis, B. Epifaunal community structure in Acropora spp. (Scleractinia) on the Great Barrier Reef: Implications of coral morphology and habitat complexity. Coral Reefs 2001, 20, 281–288. [Google Scholar] [CrossRef]
- Bauman, A.; Feary, D.; Heron, S.; Pratchett, M.; Burt, J. Multiple environmental factors influence the spatial distribution and structure of reef communities in the northeastern Arabian Peninsula. Mar. Pollut. Bull. 2013, 72, 302–312. [Google Scholar] [CrossRef] [PubMed]
- Richlen, M.; Morton, S.; Jamali, E.; Rajan, A.; Anderson, D. The catastrophic 2008–2009 red tide in the Arabian Gulf region, with observations on the identification and phylogeny of the fish-killing dinoflagellate Cochlodinium polykrikoides. Harmful Algae 2010, 9, 163–172. [Google Scholar] [CrossRef]
- Pratchett, M.; Baird, A.; Bauman, A.; Burt, J. Abundance and composition of juvenile corals reveals divergent trajectories for coral assemblages across the United Arab Emirates. Mar. Pollut. Bull. 2017, 114, 1031–1035. [Google Scholar] [CrossRef]
- Bejarano, I.; Orenes-Salazar, V.; Bento, R.; García-Charton, J.; Mateos-Molina, D. Coral reefs at Sir Bu Nair Island: An offshore refuge of Acropora in the southern Arabian Gulf. Mar. Pollut. Bull. 2022, 178, 113570. [Google Scholar] [CrossRef]
- Cavalcante, G.; Vieira, F.; Bento, R.; Bartholomew, A.; Burt, J. Oceanographic variability in Sir Bu Nair Island (Arabian/Persian Gulf) based on in situ observations. J. Coast. Res. 2024, 113, 410–414. [Google Scholar] [CrossRef]
- Bento, R.; Cavalcante, G.; Mateos-Molina, D.; Riegl, B.; Bejarano, I. Recruitment and larval connectivity of a remnant Acropora community in the Arabian Gulf, United Arab Emirates. Coral Reefs 2021, 40, 1889–1898. [Google Scholar] [CrossRef]
- Harris, C.; Schroder, T. UAE Underwater Explorer; Explorer Publishing: Dubai, United Arab Emirates, 2001. [Google Scholar]
- Beijbom, O.; Edmunds, P.; Roelfsema, C.; Smith, J.; Kline, D.; Neal, B.; Dunlap, M.; Moriarty, V.; Fan, T.; Tan, C.; et al. Towards automated annotation of benthic survey images: Variability of human experts and operational modes of automation. PLoS ONE 2015, 10, e0130312. [Google Scholar] [CrossRef] [PubMed]
- Burt, J.; Al-Harthi, S.; Al-Cibahy, A. Long-term impacts of coral bleaching events on the world’s warmest reefs. Mar. Environ. Res. 2011, 72, 225–229. [Google Scholar] [CrossRef] [PubMed]
- Oladi, M.; Rouzbehani, S.; Ahmadzadeh, F.; Ghazilou, A. Dynamics of Dipsastraea pallida-symbiont association following bleaching events across the northern Persian Gulf and Gulf of Oman. Symbiosis 2021, 84, 141–149. [Google Scholar] [CrossRef]
- Riegl, B.; Purkis, S. Coral reefs of the Gulf: Adaptation to climatic extremes in the world’s hottest sea. In Coral Reefs of the Gulf: Adaptation to Climatic Extremes; Riegl, B., Purkis, S., Eds.; Springer: Dordrecht, The Netherlands, 2012; pp. 1–4. [Google Scholar] [CrossRef]
- Bürkner, P. brms: An R package for Bayesian multilevel models using Stan. J. Stat. Softw. 2017, 80, 1–28. [Google Scholar] [CrossRef]
- Douma, J.; Weedon, J. Analysing continuous proportions in ecology and evolution: A practical introduction to beta and Dirichlet regression. Methods Ecol. Evol. 2019, 10, 1412–1430. [Google Scholar] [CrossRef]
- Lemoine, N. Moving beyond noninformative priors: Why and how to choose weakly informative priors in Bayesian analyses. Oikos 2019, 128, 912–928. [Google Scholar] [CrossRef]
- Conn, P.; Johnson, D.; Williams, P.; Melin, S.; Hooten, M. A guide to Bayesian model checking for ecologists. Ecol. Monogr. 2018, 88, 526–542. [Google Scholar] [CrossRef]
- Oksanen, J.; Kindt, R.; Legendre, P.; O’Hara, B.; Simpson, G.; Solymos, P.; Stevens, M.; Wagner, H. The Vegan Package Version 1.15-0. Available online: https://vegan.r-forge.r-project.org (accessed on 6 December 2024).
- R Core Team. R: A Language and Environment for Statistical Computing (4.3.0). Available online: https://www.R-project.org (accessed on 4 December 2024).
- Wickham, H. ggplot2: Elegant Graphics for Data Analysis; Springer Nature: Cham, Switzerland, 2016. [Google Scholar]
- Kahle, D.; Wickham, H. ggmap: Spatial visualization with ggplot2. R J. 2013, 5, 144. [Google Scholar] [CrossRef]
- Gerwing, T.; Gerwing, A.; Macdonald, T.; Cox, K.; Juanes, F.; Dudas, S. Assessing the relationship between community dispersion and disturbance in a soft-sediment ecosystem. Mar. Ecol. 2018, 39, 12505. [Google Scholar] [CrossRef]
- van Woesik, R.; Kratochwill, C. A global coral-bleaching database, 1980–2020. Sci. Data 2022, 9, 20. [Google Scholar] [CrossRef]
- Goebeler, N.; Norkko, A.; Norkko, J. Ninety years of coastal monitoring reveals baseline and extreme ocean temperatures are increasing off the Finnish coast. Commun. Earth Environ. 2022, 3, 215. [Google Scholar] [CrossRef]
- NOAA National Environmental Satellite, Data and Information Service. Satellites & Bleaching. Available online: https://coralreefwatch.noaa.gov/product/5km/tutorial/crw10a_dhw_product.php (accessed on 10 August 2025).
- Giorgi, A.; Monti, M.; Radawski, J.; Olson, J. Long term benthic survey demonstrates a shift in the composition of benthic reef communities at shallow sites in Roatán, Honduras. Biodivers. Conserv. 2022, 31, 1689–1708. [Google Scholar] [CrossRef]
- Jones, N.; Leinbach, S.; Gilliam, D. Interspecific variation in demographics reveals ecological winners and losers in a highly disturbed coral reef system. Coral Reefs 2025, 1–15. [Google Scholar] [CrossRef]
- Coles, S. Coral species diversity and environmental factors in the Arabian Gulf and the Gulf of Oman: A comparison to the Indo-Pacific region. Atoll Res. Bull. 2003, 507, 1–19. [Google Scholar] [CrossRef]
- Sawall, Y.; Harris, M.; Lebrato, M.; Wall, M.; Feng, E. Discrete pulses of cooler deep water can decelerate coral bleaching during thermal stress: Implications for artificial upwelling during heat stress events. Front. Mar. Sci. 2020, 7, 502381. [Google Scholar] [CrossRef]
- Morais, J.; Tebbett, S.; Morais, R.; Bellwood, D. Natural recovery of corals after severe disturbance. Ecol. Lett. 2024, 27, e14332. [Google Scholar] [CrossRef]
- Holbrook, S.; Adam, T.; Edmunds, P.; Schmitt, R.; Carpenter, R.; Brooks, A.; Lenihan, H.; Briggs, C. Recruitment drives spatial variation in recovery rates of resilient coral reefs. Sci. Rep. 2018, 8, 7338. [Google Scholar] [CrossRef] [PubMed]
- Tebbett, S.; Morais, J.; Bellwood, D. Spatial patchiness in change, recruitment, and recovery on coral reefs at Lizard Island following consecutive bleaching events. Mar. Environ. Res. 2022, 173, 105537. [Google Scholar] [CrossRef] [PubMed]
- Darling, E.; Alvarez-Filip, L.; Oliver, T.; Mcclanahan, T.; Côté, I. Evaluating life-history strategies of reef corals from species traits. Ecol. Lett. 2012, 15, 1378–1386. [Google Scholar] [CrossRef]
- Riegl, B.; Purkis, S. Model of coral population response to accelerated bleaching and mass mortality in a changing climate. Ecol. Model. 2009, 220, 192–208. [Google Scholar] [CrossRef]
- Foster, K.; Foster, G.; Al-Harthi, S. Coral assemblages in the southeastern Arabian Gulf (Qatar and Abu Dhabi, UAE): Various stages of Acropora recovery a decade after recurrent elevated temperature anomalies. Open J. Mar. Sci. 2013, 3, 28–39. [Google Scholar] [CrossRef]
- Thomas, L.; Şahin, D.; Adam, A.; Grimaldi, C.; Ryan, N.; Duffy, S.; Underwood, J.; Kennington, W.; Gilmour, J. Resilience to periodic disturbances and the long-term genetic stability in Acropora coral. Commun. Biol. 2024, 7, 410. [Google Scholar] [CrossRef]
- Bento, R.; Feary, D.; Hoey, A.; Burt, J. Settlement patterns of corals and other benthos on reefs with divergent environments and disturbances histories around the northeastern Arabian Peninsula. Front. Mar. Sci. 2017, 4, 305. [Google Scholar] [CrossRef]
- Howells, E.; Ketchum, R.; Bauman, A.; Mustafa, Y.; Watkins, K.; Burt, J. Species-specific trends in the reproductive output of corals across environmental gradients and bleaching histories. Mar. Pollut. Bull. 2016, 105, 532–539. [Google Scholar] [CrossRef]
- Mansour, S. Geospatial modeling of environmental hazards to coral reefs in the Oman Sea. Coral Reefs 2020, 39, 555–575. [Google Scholar] [CrossRef]
- Hoegh-Guldberg, O. Climate change, coral bleaching and the future of the world’s coral reefs. Mar. Freshw. Res. 1999, 50, 839–866. [Google Scholar] [CrossRef]
- Howells, E.; Abrego, D.; Liew, Y.; Burt, J.; Meyer, E.; Aranda, M. Enhancing the heat tolerance of reef-building corals to future warming. Sci. Adv. 2021, 7, eabg6070. [Google Scholar] [CrossRef]
- Darling, E.; McClanahan, T.; Côté, I. Combined effects of two stressors on Kenyan coral reefs are additive or antagonistic, not synergistic. Conserv. Lett. 2010, 3, 122–130. [Google Scholar] [CrossRef]
- Hunt, C.; Harvey, J.; Miller, A.; Johnson, V.; Phongsuwan, N. The Green Fins approach for monitoring and promoting environmentally sustainable scuba diving operations in South East Asia. Ocean. Coast. Manag. 2013, 78, 35–44. [Google Scholar] [CrossRef]
- Toyoshima, J.; Nadaoka, K. Importance of environmental briefing and buoyancy control on reducing negative impacts of SCUBA diving on coral reefs. Ocean. Coast. Manag. 2015, 116, 20–26. [Google Scholar] [CrossRef]
- Roche, R.; Harvey, C.; Harvey, J.; Kavanagh, A.; McDonald, M.; Stein-Rostaing, V.; Turner, J. Recreational diving impacts on coral reefs and the adoption of environmentally responsible practices within the SCUBA diving industry. Environ. Manag. 2016, 58, 107–116. [Google Scholar] [CrossRef]
- Seveso, D.; Stahl, J.; Landes, A. An outbreak of Acanthaster spp. in the Gulf of Oman (United Arab Emirates). Mar. Biodivers. 2024, 54, 62. [Google Scholar] [CrossRef]
- Westcott, D.; Fletcher, C.; Kroon, F.; Babcock, R.; Plagányi, E.; Pratchett, M.; Bonin, M. Relative efficacy of three approaches to mitigate Crown-of-Thorns Starfish outbreaks on Australia’s Great Barrier Reef. Sci. Rep. 2020, 10, 12594. [Google Scholar] [CrossRef] [PubMed]
- Selig, E.; Bruno, J. A global analysis of the effectiveness of marine protected areas in preventing coral loss. PLoS ONE 2010, 5, e9278. [Google Scholar] [CrossRef]
- Ban, N.; Adam, V.; Almany, G.; Ban, S.; Cinner, J.; McCook, L.; Mills, M.; Pressey, R.; White, A. Designing, implementing and managing marine protected areas: Emerging trends and opportunities for coral reef nations. J. Exp. Mar. Biol. Ecol. 2011, 408, 21–31. [Google Scholar] [CrossRef]
- Van Lavieren, H.; Klaus, R. An effective regional marine protected area network for the ROPME sea area: Unrealistic vision or realistic possibility? Mar. Poll. Bull. 2013, 72, 389–405. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hadj-Hammou, J.; Bartholomew, A.; Bento, R.C.; Mohamed, F.A.; Cavalcante, G.H.; Burt, J.A. Bleaching Impacts on the Last Remaining Acropora-dominated Reefs in the United Arab Emirates. Diversity 2025, 17, 610. https://doi.org/10.3390/d17090610
Hadj-Hammou J, Bartholomew A, Bento RC, Mohamed FA, Cavalcante GH, Burt JA. Bleaching Impacts on the Last Remaining Acropora-dominated Reefs in the United Arab Emirates. Diversity. 2025; 17(9):610. https://doi.org/10.3390/d17090610
Chicago/Turabian StyleHadj-Hammou, Jeneen, Aaron Bartholomew, Rita C. Bento, Fatima A. Mohamed, Geórgenes H. Cavalcante, and John A. Burt. 2025. "Bleaching Impacts on the Last Remaining Acropora-dominated Reefs in the United Arab Emirates" Diversity 17, no. 9: 610. https://doi.org/10.3390/d17090610
APA StyleHadj-Hammou, J., Bartholomew, A., Bento, R. C., Mohamed, F. A., Cavalcante, G. H., & Burt, J. A. (2025). Bleaching Impacts on the Last Remaining Acropora-dominated Reefs in the United Arab Emirates. Diversity, 17(9), 610. https://doi.org/10.3390/d17090610









