Molecular and Morphological Analyses for Delimiting Species Boundaries: The Case of Sclerodermus cereicollis Kieffer, 1904 (Hymenoptera: Bethylidae) †
Abstract
1. Introduction
2. Materials and Methods
2.1. Molecular Protocol
2.2. Species Delimitation Methods
3. Results
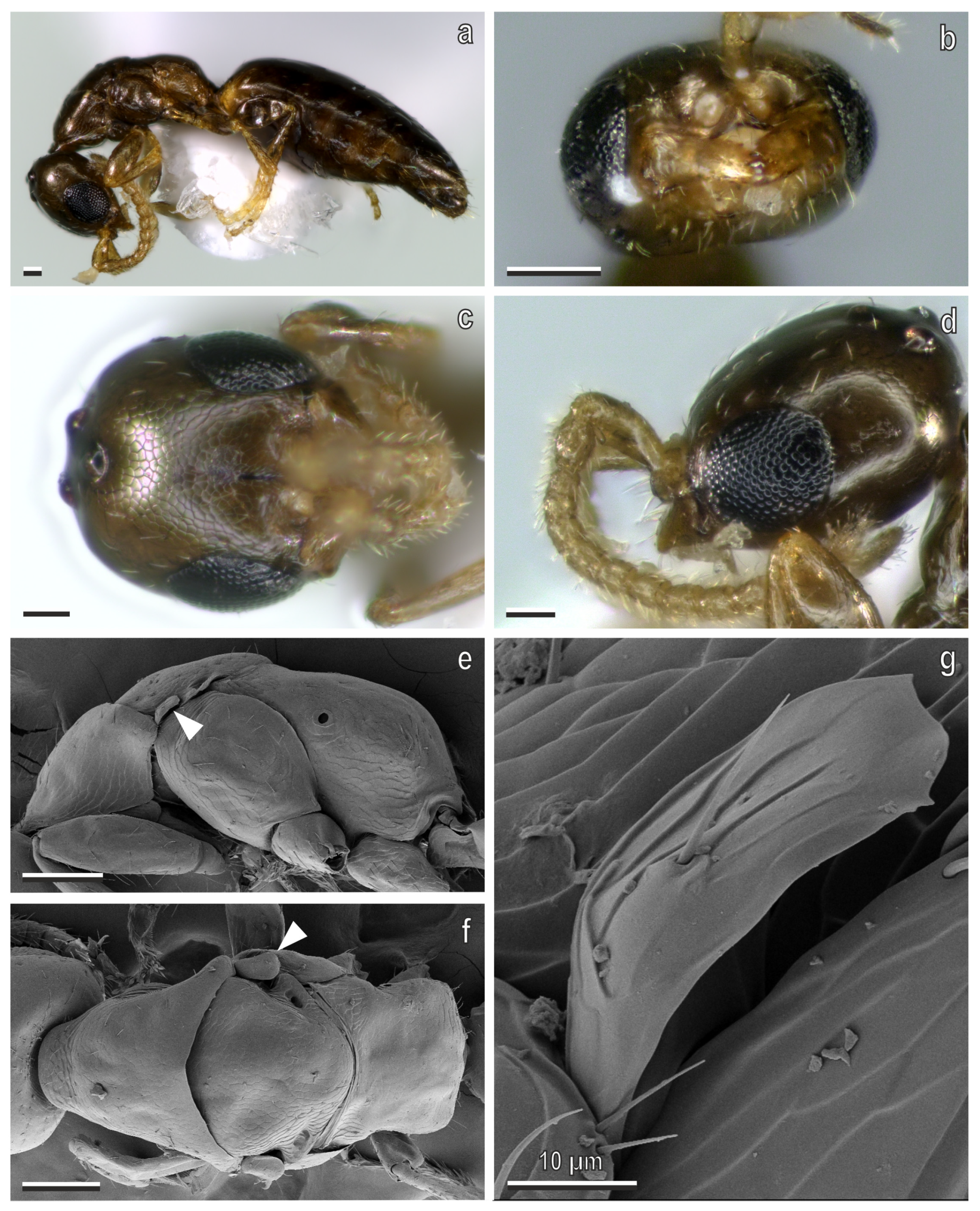
3.1. Sclerodermus cereicollis Kieffer, 1904
3.2. Sclerodermus annobonensis Masini, Colombo & Azevedo sp. nov.
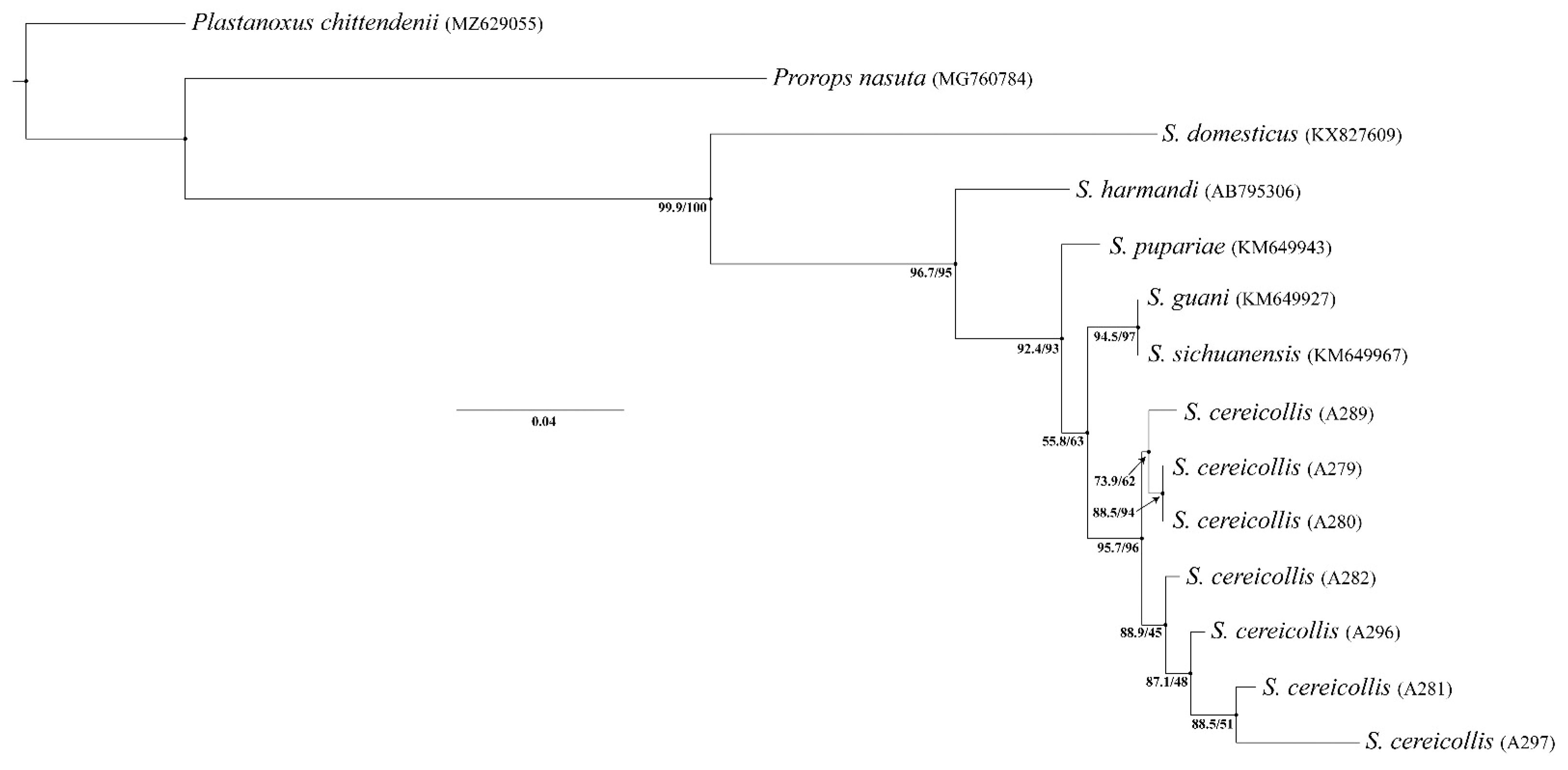
3.3. Molecular Analyses
4. Discussion
4.1. Taxonomic Identity of Sclerodermus cereicollis
4.2. Genetic Evidence for Conspecificity of European Syntypes and Field Specimens
4.3. Taxonomic Identity of Sclerodermus annobonensis Masini, Colombo & Azevedo sp. nov.
- 1.
- Head and metapectal–propodeal disc barrel-shaped (Figure 15b) … S. wollastonii Westwood, 1881
- 2.
- Metapectal–propodeal disc with sides slightly divergent posterad (Figure 10g) … 3
- 3.
- Head with sides nearly straight and strongly convergent anterad (Figure 15a) … S. pictiventris Kieffer, 1921
4.4. Comparative Morphology of Afrotropical Sclerodermus Species
4.4.1. Micropterous Forms
4.4.2. Macropterous Forms
4.5. Morphological Variability, Wing Polymorphism, and Taxonomic Implications in Sclerodermus
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| BL | body length |
| COI | cytochrome c oxidase I |
| DAO | diameter of anterior ocellus |
| DPO | diameter of posterior ocellus |
| EL | eye length |
| FWL | forewing length |
| LH | length of head |
| MCSN | Museo Civico di Storia Naturale Giacomo Doria, Genoa, Italy |
| OOL | ocello–ocular line |
| UFES | Universidade Federal do Espírito Santo, Vitória, Brazil |
| UFMG | Universidade Federal de Minas Gerais, Belo Horizonte, Brazil |
| VOL | vertex–ocular line |
| WH | width of head |
| WOT | width of ocellar triangle |
References
- Azevedo, C.O.; Alencar, I.D.; Ramos, M.S.; Barbosa, D.N.; Colombo, W.D.; Vargas, J.M.; Lim, J. Global guide of the flat wasps (Hymenoptera, Bethylidae). Zootaxa 2018, 4489, 1–294. [Google Scholar] [CrossRef]
- Benoit, P.L.G. Un nouveau Sclerodermus vulnérant pur l’homme en Afrique Centrale (Hymenoptera-Bethylidae). Bull. Ann. Soc. R. Entomol. Belg. 1957, 93, 42–46. [Google Scholar]
- Bridwell, J.C. Some Notes on Hawaiian and other Bethylidae (Hymenoptera) with the Description of a New Genus and Species. 2nd Paper. Proc. Hawaii. Entomol. Soc. 1920, 4, 291–314. [Google Scholar]
- Wu, H.; Wang, X.Y.; Li, M.L.; Yang, Z.Q.; Zeng, F.X.; Wang, H.Y.; Bai, L.; Liu, S.J.; Sun, J. Biology and mass rearing of Sclerodermus pupariae Yang et Yao (Hymenoptera: Bethylidae), an important ectoparasitoid of the emerald ash borer, Agrilus planipennis (Coleoptera: Buprestidae) in China. Acta Entomol. Sin. 2008, 51, 46–54. [Google Scholar]
- Yang, Z.Q.; Wang, X.Y.; Yao, Y.X.; Gould, J.R.; Cao, L.M. A new species of Sclerodermus (Hymenoptera: Bethylidae) parasitizing Agrilus planipennis (Coleoptera: Buprestidae) from China, with a key to Chinese species in the genus. Ann. Entomol. Soc. Am. 2012, 105, 619–627. [Google Scholar] [CrossRef]
- Yang, Z.; Wang, X.; Wang, B.; Sun, J. Recent advances in biological control of important native and invasive forest pests in China. Biol. Control China 2014, 68, 117–128. [Google Scholar] [CrossRef]
- Abdi, M.K.; Lupi, D.; Jucker, C.; Hardy, I.C. Kinship effects in quasi-social parasitoids I: Co-foundress number and relatedness affect suppression of dangerous hosts. Biol. J. Linn. Soc. 2020, 130, 627–641. [Google Scholar] [CrossRef]
- Abdi, M.K.; Hardy, I.C.; Jucker, C.; Lupi, D. Kinship effects in quasi-social parasitoids II: Co-foundress relatedness and host dangerousness interactively affect host exploitation. Biol. J. Linn. Soc. 2020, 130, 642–660. [Google Scholar] [CrossRef]
- Malabusini, S.; Lupi, D. Exploring the Biology of Quasi-Social Idiobiont Parasitoids in the Genus Sclerodermus (Hymenoptera: Bethylidae). Insects 2024, 15, 880. [Google Scholar] [CrossRef]
- Tang, X.; Meng, L.; Kapranas, A.; Xu, F.; Hardy, I.C.W.; Li, B. Mutually beneficial host exploitation and ultra-biased sex ratios in quasisocial parasitoids. Nat. Commun. 2014, 5, 4942. [Google Scholar] [CrossRef]
- Stingeni, L.; Hansel, K.; Casciola, G.; Bianchi, L.; Tramontana, M.; Marietti, R.; Zampetti, S.; Napoli, F.; Miñón Llera, G.; Biancolini, F.; et al. Human ectoparasitosis by mites of the genus Pyemotes Amerling 1861 (Acarina: Pyemotidae). Ital. J. Dermatol. Venerol. 2023, 158, 4–14. [Google Scholar] [CrossRef] [PubMed]
- Asahina, S. On a remarkable case of the biting of a parasitic wasp, Sclerodermus nipponensis Yuasa in Tokyo (Hymenoptera, Bethylidae). Jpn. J. Med. Sci. Biol. 1953, 6, 197–199. [Google Scholar] [CrossRef]
- Ayala, L. On an unusual parasitic dermatosis due to Sclerodermus brevicornis. Minerva Dermatol. 1967, 42, 593–597. [Google Scholar] [PubMed]
- Bequaert, J. Un Hyménoptère Béthylide qui pique l’homme au Congo. Ann. Soc. Belg. Méd. Trop. 1924, 4, 163–165. [Google Scholar]
- Bernard, F.; Jacquemin, P. Effets des piqures de Scleroderma (Hyménoptères Bethylidae) et revision des espèces nord-africaines. Bull. Soc. Hist. Nat. Afr. Nord. 1948, 39, 160–167. [Google Scholar]
- Marhic, E. Liste preliminaire des Bethylidae de France (Hymenoptera: Chrysidoidea). Osmia 2022, 10, 13–24. [Google Scholar] [CrossRef]
- Masini, P.; Stingeni, L.; Salerno, G.; Saitta, V.; Azevedo, C.O.; Rebora, M.; Ornielli, M.; Hansel, K.; Bianchi, L.; Casciola, G. Human ectoparasitoses by flat wasps of the genera Sclerodermus and Cephalonomia (Hymenoptera: Bethylidae). Clin. Exp. Dermatol. 2025, 50, 1508–1521. [Google Scholar] [CrossRef]
- Mazza, G.; Inghilesi, A.F.; Materassi, V.; Mazza, U.; Milanesi, N.; Cervo, R. Infestazioni da Sclerodermus domesticus Klug, 1809 e problematiche associate (Hymenopera: Behtylidae). Onychium 2014, 10, 183–188. [Google Scholar]
- Patton, W.S. Insects, Ticks, Mites and Venomous Animals of Medical and Veterinary Importance. Part II. Public Health. Am. J. Public Health Nations Health 1931, 21, 701. [Google Scholar]
- Walton, G.A. A minute bethylid wasp of medical interest. Proc. R. Entomol. Soc. Lond. Ser. A General Entomol. 1948, 23, 32–33. [Google Scholar] [CrossRef]
- Westwood, J.O. XXXII. Monograph upon the Hymenopterous Genus Scleroderma. Trans. R. Entomol. Soc. Lond. 1839, 2, 164–172. [Google Scholar] [CrossRef]
- Westwood, J.O. VIII. Observations on the Hymenopterous genus Scleroderma, Klug, and some allied groups. Trans. R. Entomol. Soc. Lond. 1881, 29, 117–140. [Google Scholar] [CrossRef]
- Masini, P.; Piersanti, S.; Lupi, D.; Salerno, G.; Rebora, M. Antennal chemoreceptors in the European ectoparasitoid Sclerodermus cereicollis (Hymenoptera: Bethylidae). Microsc. Res. Tech. 2024, 87, 2275–2291. [Google Scholar] [CrossRef]
- Lupi, D.; Favaro, R.; Jucker, C.; Azevedo, C.O.; Hardy, I.C.; Faccoli, M. Reproductive biology of Sclerodermus brevicornis, a European parasitoid developing on three species of invasive longhorn beetles. Biol. Control 2017, 105, 40–48. [Google Scholar] [CrossRef]
- Malabusini, S.; Hardy, I.C.; Jucker, C.; Savoldelli, S.; Lupi, D. How many cooperators are too many? Foundress number, reproduction and sex ratio in a quasi-social parasitoid. Ecol. Entomol. 2022, 47, 566–579. [Google Scholar] [CrossRef]
- Malabusini, S.; Hardy, C.V.; Jucker, C.; Guanzani, G.; Savoldelli, S.; Lupi, D. Reproductive performance effects of rearing the quasi-social parasitoid, Sclerodermus brevicornis (Hymenoptera: Bethylidae), on a factitious host. J. Insect Sci. 2023, 23, 7. [Google Scholar] [CrossRef]
- Masini, P.; Austeri, L.; Rebora, M.; Piersanti, S.; de Francesco, F.; Salerno, G. Olfactory cues in the host-location of the European ecto-parasitoids Sclerodermus cereicollis and Sclerodermus domesticus (Hymenoptera: Bethylidae). J. Stored Prod. Res. 2024, 109, 102441. [Google Scholar] [CrossRef]
- Azevedo, C.O.; Vargas, J.M.; Colombo, W.D. Synopsis of world Discleroderma Kieffer (Hymenoptera, Bethylidae). Zootaxa 2020, 4742, 467–480. [Google Scholar] [CrossRef] [PubMed]
- Vargas R., J.M.; Colombo, W.D.; Azevedo, C.O. Revisited phylogeny of Scleroderminae (Hymenoptera: Bethylidae) reveals a plastic evolutionary history. Arthropod Syst. Phylogeny 2020, 78, 217–243. [Google Scholar] [CrossRef]
- Lanes, G.; Azevedo, C.O. Phylogeny and Taxonomy of Sclerodermini (Hymenoptera, Bethylidae, Epyrinae). Insect Syst. Evol. 2008, 39, 55–86. [Google Scholar] [CrossRef]
- Colombo, W.D.; Azevedo, C.O. Revalidation of the polymorphic genus Acephalonomia (Hymenoptera: Bethylidae) and description of a new species from Micronesia. Eur. J. Entomol. 2020, 117, 235–242. [Google Scholar] [CrossRef]
- Wang, X.; Wei, K.; Yang, Z.; Jennings, D.E.; Duan, J.J. Effects of biotic and abiotic factors on phenotypic partitioning of wing morphology and development in Sclerodermus pupariae (Hymenoptera: Bethylidae). Sci. Rep. 2016, 6, 26408. [Google Scholar] [CrossRef]
- Skvarla, M.J. A review of Sclerodermus Latreille, 1809 (Hymenoptera: Bethylidae) infestations and report of the first case in North America North of Mexico. J. Med. Entomol. 2018, 55, 752–756. [Google Scholar] [CrossRef]
- Abdi, M.K.; Lupi, D.; Hardy, I.C. Co-foundress confinement elicits kinship effects in a naturally sub-social parasitoid. J. Evol. Biol. 2020, 33, 1068–1085. [Google Scholar] [CrossRef]
- Jucker, C.; Hardy, C.V.; Malabusini, S.; de Milato, S.; Zen, G.; Savoldelli, S.; Lupi, D. Factors affecting the reproduction and mass-rearing of Sclerodermus brevicornis (Hymenoptera: Bethylidae), a natural enemy of exotic flat-faced longhorn beetles (Coleoptera: Cerambycidae: Lamiinae). Insects 2020, 11, 657. [Google Scholar] [CrossRef] [PubMed]
- Azevedo, C.O.; Colombo, W.D. Synopsis of the Neotropical Sclerodermus Latreille (Hymenoptera, Bethylidae) with description of a new species attacking human beings. Zootaxa 2022, 5124, 501–519. [Google Scholar] [CrossRef]
- Kieffer, J.J. Description de un nouveaux Dryniinae et Bethylinae du musée civic de génes. Ann. Mus. Civ. Storia Nat. Genova 1904, 1, 351–412. [Google Scholar]
- Marannino, P.; de Lillo, E. The peach flatheaded rootborer, Capnodis tenebrionis (L.), and its enemies. IOBC WPRS Bull. 2007, 30, 197–200. [Google Scholar]
- Gordh, G.; Moczar, L. A catalog of the world Bethylidae (Hymenoptera: Aculeata). Mem. Am. Entomol. Inst. 1990, 46, 1–364. [Google Scholar]
- Coyne, J.A.; Orr, H.A. Speciation; Sinauer Association: Sunderland, MA, USA, 2004. [Google Scholar]
- Harris, R.A. A glossary of surface sculpturing. Occas. Pap. Bur. Entomol. Calif. Dep. Agric. 1979, 28, 1–31. [Google Scholar]
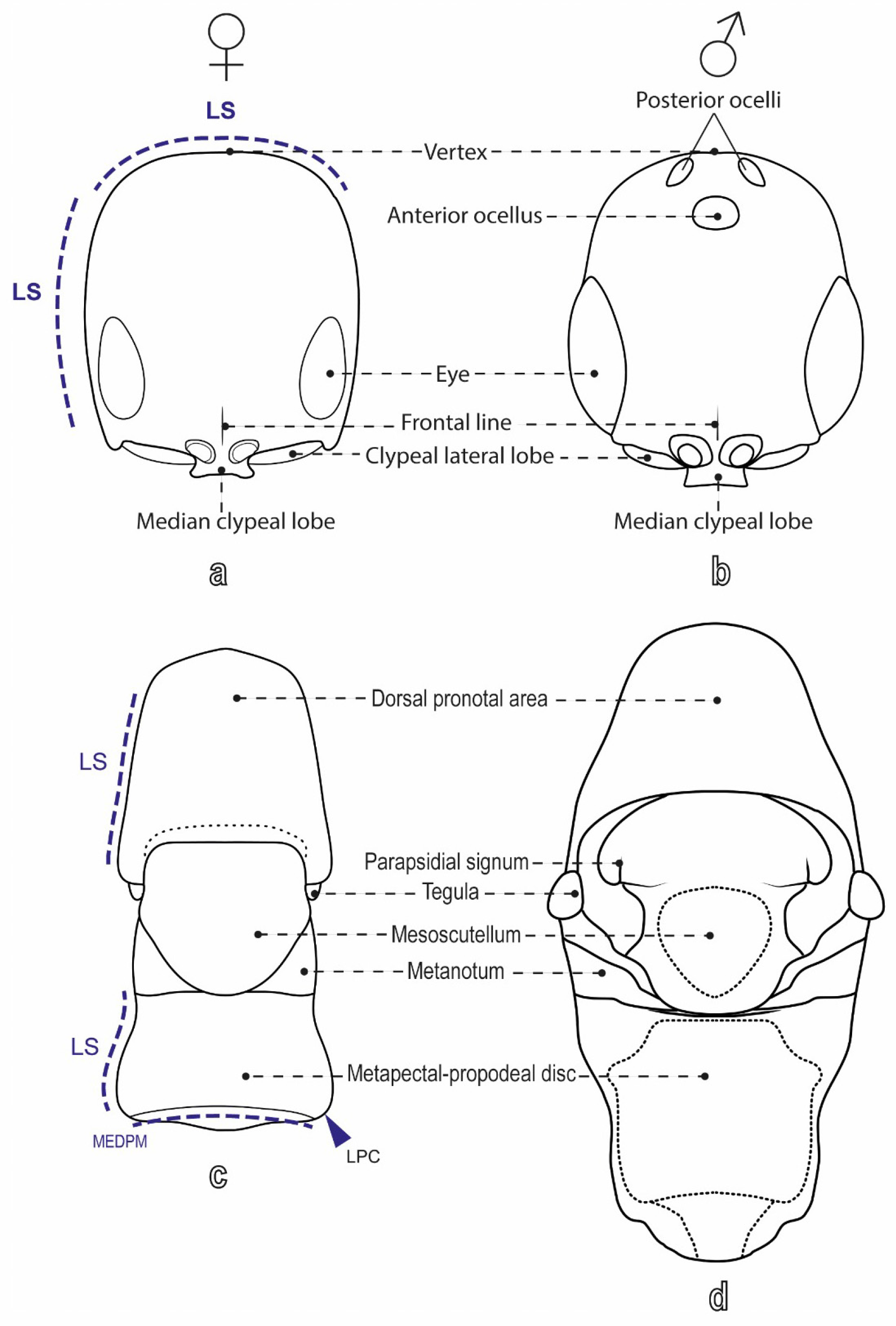
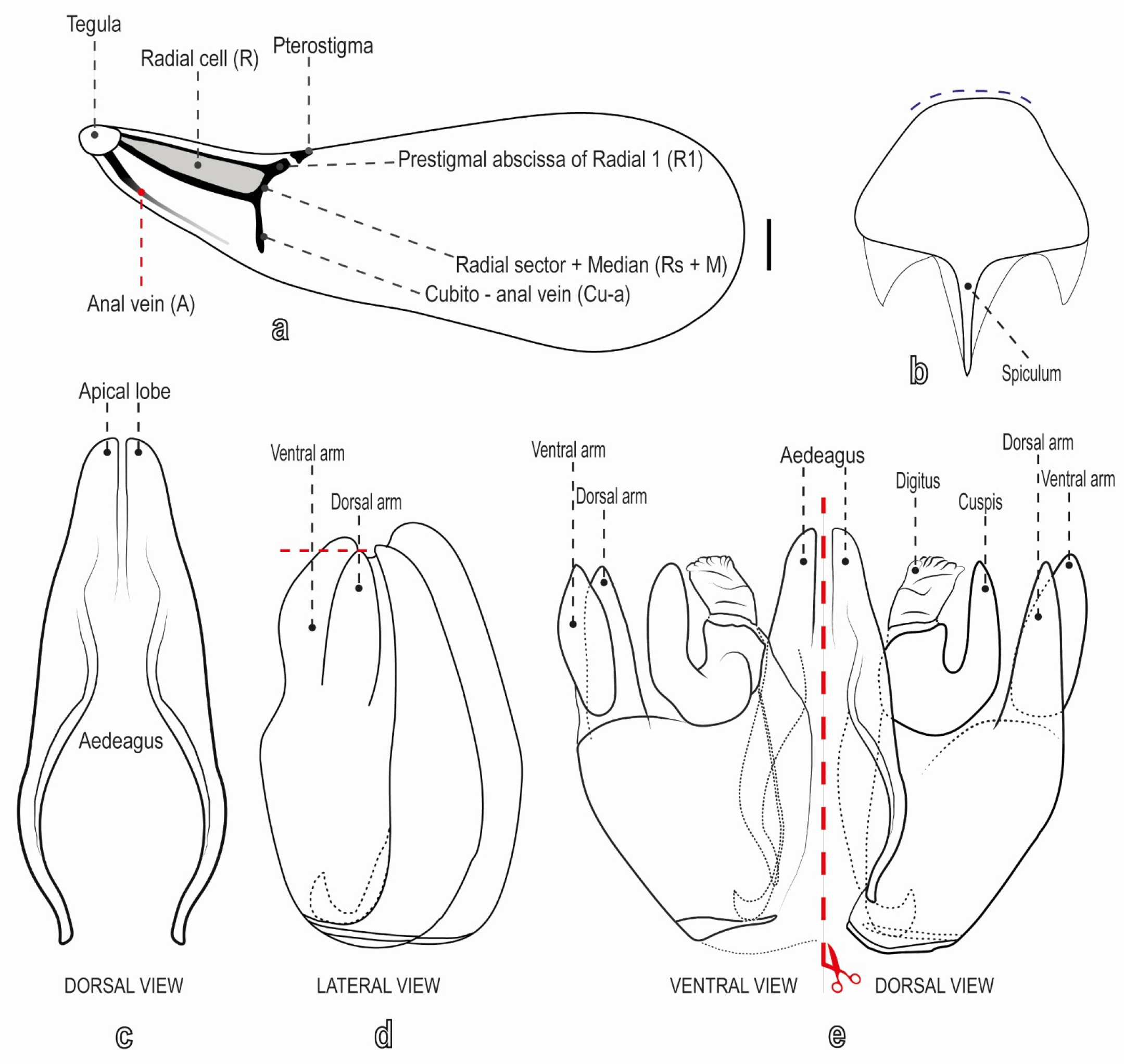
- Lanes, G.O.; Kawada, R.; Azevedo, C.O.; Brothers, D.J. Revisited morphology applied for systematics of flat wasps (Hymenoptera, Bethylidae). Zootaxa 2020, 4752, 1–127. [Google Scholar] [CrossRef] [PubMed]
- Kawada, R.; Buffington, M.L. A scalable and modular dome illumination system for scientific microphotography on a budget. PLoS ONE 2016, 11, e0153426. [Google Scholar] [CrossRef]
- Martinelli, A.B.; Waichert, C.; Barbosa, D.N.; Fagundes, V.; Azevedo, C.O. The use of proteinase K to access genitalia morphology, vouchering and DNA extraction in minute wasps. An. Acad. Bras. Ciênc. 2017, 89, 1629–1633. [Google Scholar] [CrossRef][Green Version]
- Folmer, O.; Black, M.; Hoeh, W.; Lutz, R.; Vrijenhoek, R. DNA primers for amplification of mitochondrial cytochrome c oxidase subunit I from diverse metazoan invertebrates. Mol. Mar. Biol. Biotechnol. 1994, 3, 294–299. [Google Scholar]
- Kumar, S.; Stecher, G.; Tamura, K. MEGA7: Molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol. Biol. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef]
- Altschul, S.F.; Gish, W.; Miller, W.; Myers, E.W.; Lipman, D.J. Basic local alignment search tool. J. Mol. Biol. 1990, 215, 403–410. [Google Scholar] [CrossRef]
- Altschul, S.F.; Madden, T.L.; Schäffer, A.A.; Zhang, J.; Zhang, Z.; Miller, W.; Lipman, D.J. Gapped BLAST and PSIBLAST: A new generation of protein database search programs. Nucleic Acids Res. 1997, 25, 3389–3402. [Google Scholar] [CrossRef]
- Katoh, K.; Standley, D.M. MAFFT multiple sequence alignment software version 7: Improvements in performance and usability. Mol. Biol. Evol. 2013, 30, 772–780. [Google Scholar] [CrossRef]
- Minh, B.Q.; Schmidt, H.A.; Chernomor, O.; Schrempf, D.; Woodhams, M.D.; Haeseler, A.V.; Lanfear, R. IQ-TREE 2: New models and efficient methods for phylogenetic inference in the genomic era. Mol. Biol. Evol. 2020, 37, 1530–1534. [Google Scholar] [CrossRef] [PubMed]
- Minh, B.Q.; Nguyen, M.A.T.; Haeseler, A.V. Ultrafast approximation for phylogenetic bootstrap. Mol. Biol. Evol. 2013, 30, 1188–1195. [Google Scholar] [CrossRef] [PubMed]
- Guindon, S.; Dufayard, J.F.; Lefort, V.; Anisimova, M.; Hordijk, W.; Gascuel, O. New algorithms and methods to estimate maximum-likelihood phylogenies: Assessing the performance of PhyML 3.0. Syst. Biol. 2010, 59, 307–321. [Google Scholar] [CrossRef]
- Rambaut, A. Figtree v1.4.3 [Computer Software]. Available online: https://tree.bio.ed.ac.uk/software/figtree/ (accessed on 2 March 2025).
- Kimura, M. A simple method for estimating evolutionary rates of base substitutions through comparative studies of nucleotides sequences. J. Mol. Evol. 1980, 16, 111–120. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Yang, Z.; Wang, X.; Hou, Y. Molecular identification of sibling species of Sclerodermus (Hymenoptera: Bethylidae) that parasitize buprestid and cerambycid beetles by using partial sequences of mitochondrial DNA cytochrome oxidase subunit 1 and 28S ribosomal RNA gene. PLoS ONE 2015, 10, e0119573. [Google Scholar] [CrossRef]
- Colombo, W.D.; Waichert, C.; Azevedo, C.O. Molecular and Morphological Species Delimitation of Epyrinae (Hymenoptera: Bethylidae) from Papua New Guinea. In Insects of Mount Wilhelm, Papua New Guinea, 2nd ed.; Robillard, T., Legendre, F., Villemant, C., Leponce, M., Eds.; Muséum National d’Histoire Naturelle: Paris, France, 2020; pp. 209–319. [Google Scholar]
- Liao, H.; Terayama, M.; Eguchi, K. Revision of Taiwanese and Ryukyuan species of Eleganesia Alencar & Azevedo, 2018 (Hymenoptera, Bethylidae). Zool. Anz. 2021, 294, 62–79. [Google Scholar] [CrossRef]
- Marques, W.J., Jr.; Colombo, W.D.; Azevedo, C.O. Insights into the systematics of Afrotropical Laelius (Hymenoptera, Bethylidae): Combining molecular and morphological data to associate dimorphic species. Zool. Anz. 2023, 306, 90–107. [Google Scholar] [CrossRef]
- Lupi, D.; Bernardo, U.; Bonsignore, C.P.; Colombo, M.; Dindo, M.L.; Faccoli, M.; Ferracini, C.; Gualtieri, L.; Marullo, R.; Mazzon, L.; et al. Insects and globalization: Sustainable control of exotic species in Italian agro-forestry ecosystems. Landscape management for functional biodiversity. IOBC WPRS Bull. 2014, 100, 87–90. [Google Scholar]
- Abdi, M.K.; Jucker, C.; De Marchi, B.; Hardy, I.C.; Lupi, D. Performance of Sclerodermus brevicornis, a parasitoid of invasive longhorn beetles, when reared on rice moth larvae. Entomol. Exp. Appl. 2021, 169, 64–78. [Google Scholar] [CrossRef]
- de Queiroz, K. Species concepts and species delimitation. Syst. Biol. 2007, 56, 879–886. [Google Scholar] [CrossRef]
- Pannebakker, B.A.; Beukeboom, L.W.; Van Alphen, J.J.; Brakefield, P.M.; Zwaan, B.J. The genetic basis of male fertility in relation to haplodiploid reproduction in Leptopilina clavipes (Hymenoptera: Figitidae). Genetics 2004, 168, 341–349. [Google Scholar] [CrossRef] [PubMed]
- Kieffer, J.J. Nouveaux Bethylides du Soudan Égyptien. Ann. Soc. Sci. Bruxelles. 1921, 40, 114. [Google Scholar]
- Hu, Z.; Zhao, X.; Li, Y.; Liu, X.; Zhang, Q. Maternal care in the parasitoid Sclerodermus harmandi (Hymenoptera: Bethylidae). PLoS ONE 2012, 7, e51246. [Google Scholar] [CrossRef]
- Leung, K.; Beukeboom, L.W.; Zwaan, B.J. Inbreeding and outbreeding depression in wild and captive insect populations. Ann. Rev. Entomol. 2025, 70, 271–292. [Google Scholar] [CrossRef] [PubMed]
- Vayssade, C.; De Fazio, C.; Quaglietti, B.; Auguste, A.; Ris, N.; Fauvergue, X. Inbreeding depression in a parasitoid wasp with single-locus complementary sex determination. PLoS ONE 2014, 9, e97733. [Google Scholar] [CrossRef] [PubMed]
- Malabusini, S.; Hardy, I.C.W.; Jucker, C.; Savoldelli, S.; Lupi, D. Multiple foundresses and multiple hosts: The influences of kinship and host quality on group reproduction in a quasi-social parasitoid. Ecol. Entomol. 2024, 49, 254–265. [Google Scholar] [CrossRef]











| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | ||
| 1 | P. chittendenii (MZ629055) | |||||||||||||
| 2 | P. nasuta (MG760784) | 0.18 | ||||||||||||
| 3 | S. domesticus (KX827609) | 0.24 | 0.28 | |||||||||||
| 4 | S. guani (KM649927) | 0.20 | 0.29 | 0.20 | ||||||||||
| 5 | S. harmandi (AB795306) | 0.23 | 0.29 | 0.18 | 0.07 | |||||||||
| 6 | S. pupariae (KM649943) | 0.21 | 0.32 | 0.19 | 0.03 | 0.06 | ||||||||
| 7 | S. sichuanensis (KM649967) | 0.20 | 0.29 | 0.20 | 0.00 | 0.07 | 0.03 | |||||||
| 8 | S. cereicollis (A279) | 0.23 | 0.29 | 0.19 | 0.03 | 0.07 | 0.03 | 0.03 | ||||||
| 9 | S. cereicollis (A280) | 0.23 | 0.29 | 0.19 | 0.03 | 0.07 | 0.03 | 0.03 | 0.00 | |||||
| 10 | S. cereicollis (A281) | 0.24 | 0.30 | 0.20 | 0.05 | 0.08 | 0.04 | 0.05 | 0.02 | 0.02 | ||||
| 11 | S. cereicollis (A282) | 0.23 | 0.29 | 0.19 | 0.03 | 0.07 | 0.03 | 0.03 | 0.01 | 0.01 | 0.02 | |||
| 12 | S. cereicollis (A289) | 0.23 | 0.29 | 0.19 | 0.03 | 0.07 | 0.03 | 0.03 | 0.01 | 0.01 | 0.02 | 0.01 | ||
| 13 | S. cereicollis (A296) | 0.23 | 0.29 | 0.19 | 0.04 | 0.08 | 0.04 | 0.04 | 0.01 | 0.01 | 0.02 | 0.01 | 0.02 | |
| 14 | S. cereicollis (A297) | 0.25 | 0.31 | 0.19 | 0.05 | 0.08 | 0.05 | 0.05 | 0.03 | 0.03 | 0.02 | 0.02 | 0.03 | 0.02 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Masini, P.; Salerno, G.; Rebora, M.; Lupi, D.; Colombo, W.D.; Azevedo, C.O. Molecular and Morphological Analyses for Delimiting Species Boundaries: The Case of Sclerodermus cereicollis Kieffer, 1904 (Hymenoptera: Bethylidae). Diversity 2025, 17, 611. https://doi.org/10.3390/d17090611
Masini P, Salerno G, Rebora M, Lupi D, Colombo WD, Azevedo CO. Molecular and Morphological Analyses for Delimiting Species Boundaries: The Case of Sclerodermus cereicollis Kieffer, 1904 (Hymenoptera: Bethylidae). Diversity. 2025; 17(9):611. https://doi.org/10.3390/d17090611
Chicago/Turabian StyleMasini, Paolo, Gianandrea Salerno, Manuela Rebora, Daniela Lupi, Wesley D. Colombo, and Celso O. Azevedo. 2025. "Molecular and Morphological Analyses for Delimiting Species Boundaries: The Case of Sclerodermus cereicollis Kieffer, 1904 (Hymenoptera: Bethylidae)" Diversity 17, no. 9: 611. https://doi.org/10.3390/d17090611
APA StyleMasini, P., Salerno, G., Rebora, M., Lupi, D., Colombo, W. D., & Azevedo, C. O. (2025). Molecular and Morphological Analyses for Delimiting Species Boundaries: The Case of Sclerodermus cereicollis Kieffer, 1904 (Hymenoptera: Bethylidae). Diversity, 17(9), 611. https://doi.org/10.3390/d17090611








