Abstract
Moths are among the most diverse and abundant animals. They have evolved a suite of acoustic defenses, such as producing ultrasound to guard against predation by low-duty-cycle bats. Recently, we found that the ultrasonic clicks of several moth species can also help decrease the capture success of high-duty-cycle bats. We tested whether moth clicks could advertise moth toxicity and/or jam the echolocation calls of high-duty-cycle bats. We examined the acoustic response of a hawkmoth (Cechenena minor) and a tiger moth (Creatonotos transiens) to free-flying horseshoe bats, Rhinolophus osgoodi. C. minor and C. transiens produced anti-bat ultrasonic clicks with an average duty cycle of 22.3% and 4.9%, respectively. The clicks of C. minor overlapped spectrally with the echolocation calls of R. osgoodi. C. minor occasionally emitted clicks immediately after the end of bat feeding buzzes. The clicks of C. transiens did not overlap spectrally with the echolocation calls of R. osgoodi. More than half the click sequences of C. transiens were produced before the emergence of bat feeding buzzes and did not overlap temporally with the feeding buzzes. Moreover, we found that C. minor was palatable and C. transiens was unpalatable to R. osgoodi. These results suggest that C. minor might be capable of jamming R. osgoodi and might also use ultrasonic clicks as warning signals. The clicks of C. transiens probably function to advertise moth toxicity. This study highlights the diversity of moth anti-bat sounds and illustrates the potential mechanisms of moth acoustic defense against high-duty-cycle bats.
1. Introduction
Anti-predator behaviors of prey can influence the population dynamics of predator–prey systems [1,2,3]. Uncovering the mechanisms of prey defense against predators is crucial to improving our understanding of how prey and predators interact and how predator–prey interactions have shaped population cycles and biological diversity [4,5,6,7,8]. Moths are among the most diverse and abundant animals [9]. The biosonar arms race between moths and echolocating bats is a classic example of predator–prey interactions [10,11,12]. Most bat species have evolved echolocation to hunt prey in the darkness [13]. Many moths counter with ultrasound-sensitive ears and ultrasound production [12,14]. Ultrasound production represents one of the most common anti-bat strategies in moths, such that 20% of moths may be capable of producing anti-bat ultrasonic clicks [15]. Laboratory behavioral studies have shown that moth ultrasonic clicks can help moths avoid being eaten by foraging bats [16,17,18,19,20,21].
Three general hypotheses are proposed to explain the acoustic interactions between clicking moths and echolocating bats. Moth ultrasonic clicks might startle the bat (the startling hypothesis) [22,23], jam the echolocation calls of the bat (the jamming hypothesis) [5,16,24], and/or advertise moth toxicity (the aposematism hypothesis) [25,26,27]. The startling hypothesis argues that moth clicks elicit the bat’s startle reflex, allowing the moths to gain a momentary advantage to escape [22,23]. Some studies have shown that the startle-like effects of moth clicks are effective for naive bats but ephemeral for adult free-flying bats [22,23]. Naive big brown bats (Eptisicus fuscus) initially had a low proportion of direct and close-range attacks on clicking tiger moths (Bertholdia trigona) and habituated after up to 40 presentations of the moth clicks over two nights [23]. Adult big brown bats quickly habituated to the ultrasonic clicks of Cycnia tenera and ignored the moth clicks after fewer than three presentations in a single night [22]. The startling effect is unlikely to be effective under natural conditions for long periods [12]. Sonar jamming is an important mechanism for some tiger moths and hawkmoths that produce ultrasound for defense against attacking bats [5,16,24]. Moth clicks with high-duty cycles (>20%) are expected to effectively interfere with bat echolocation calls [24,28]. The duty cycle of a periodic sound refers to the ratio of signal duration to signal period. However, Fernández and colleagues did not find evidence for a duty-cycle threshold of tiger moth signals for sonar jamming [29]. A moth cannot be unambiguously classified as an aposematic or sonar jammer based solely on the duty cycles of its clicks [29]. Sonar jamming should be most effective when the clicks are produced during the terminal phase of a bat’s attack ([17], but see [30]). Many arctiines use ultrasonic clicks to advertise their toxicity (i.e., acoustic aposematism), and some bats learn to associate moth clicks with distastefulness [25,26,27,29,31,32]. Some palatable moths produce clicks to deceive their predators by mimicking unpalatable models (i.e., acoustic mimicry) [17,32]. Moth clicks serving as a function of toxicity advertisement are expected to be produced early to give attacking bats time to process the meaning of the signal [12], but Barber and Conner have found no significant difference in the timing of clicks between HDC and LDC moths [30].
The knowledge of the mechanisms of moth clicks in defense against bat predation is gained from studies on the interactions between clicking moths and low-duty-cycle (LDC) bats. LDC bats, including most echolocating bat species (~1000 species), avoid forward-masking effects (an acoustic signal cannot be perceived due to the presence of a preceding signal) by separating echolocation pulse and echo in time. High-duty-cycle (HDC) bats, including ~200 species in the families Rhinolophidae and Hipposideridae and the mormoopid Pteronotus parnellii, separate pulse and echo information in frequency [33]. The echolocation calls produced by LDC bats are short (typically < 5 ms), with a duty cycle of <25%. The echolocation calls of HDC bats are characterized by high-frequency, long constant-frequency calls (typically 20–50 ms, 50–120 kHz), with a duty cycle higher than 25%. LDC bats and HDC bats differ from each other in echolocation call traits and strategies [33,34]. However, a study on the effectiveness of ultrasound production in moths against predation by HDC bats is lacking. It is unknown whether the three hypotheses could explain acoustic interactions between clicking moths and HDC bats.
Recently, we found that ultrasound production in several moth species decreased the capture success of an HDC bat species, Rhinolophus osgoodi (unpublished data). Moths producing anti-bat clicks had a survival advantage over silent moth species when attacked by the horseshoe bats in the laboratory. In the present study, we examined how ultrasound production in the moths Cechenena minor and Creatonotos transiens decreases the capture success of R. osgoodi. We hypothesized that ultrasonic moth clicks jam the sonar of bats (the jamming hypothesis) or warn the bat of the moth’s distastefulness (the aposematism hypothesis). If the moth clicks serve a jamming function, it is expected that (1) moths would produce ultrasonic clicks with high-duty cycles (>20%) in response to foraging R. osgoodi, and that (2) moth clicks would be produced during the terminal phase of a bat’s attack. If the moth clicks serve as an acoustic warning of toxicity, it is expected that (1) the moths were unpalatable to the bats, and that (2) moth clicks would be produced before the terminal phase of a bat’s attack. We examined the acoustic response of C. minor and C. transiens to free-flying R. osgoodi. We analyzed the acoustic parameters of moth clicks and bat echolocation calls and quantified the time of the emergence of moth clicks. We also tested the palatability of the moths to the bats.
2. Materials and Methods
2.1. Animals and Housing
We utilized a mist net to capture six adult male R. osgoodi from a cave in Hunan province, China in August 2022. The bats were kept together in a cage (0.5 m × 0.5 m × 0.5 m) placed in the cave during the day. We fed the bats mealworms (Tenebrio molitor larvae) and fresh moths each night. We collected one arctiine moth species, Creatonotos transiens, and one species of Sphingidae, Cechenena minor after sunset (19.30–21.00 p.m.), employing a 6 W 12 V portable heath moth trap (Natural History Book Service, Totnes, UK). The two moth species were relatively more abundant in the bat foraging habitats than other moth species capable of producing anti-bat ultrasonic clicks. The moths were kept in a mesh cage (0.3 m × 0.3 m × 0.4 m). The bats and moths were transferred to a temporary recording room (4 m × 5 m × 2.8 m) near the cave for experiments at night. The walls, ceiling, and floor of the recording room were covered with sound-absorbing foam. After all experiments were finished, we released the bats into the cave. They were released in a healthy state, and none was injured. This study was approved by the Science and Technology Ethics Committee of Northeast Normal University, China (permit ID: NENU-2022-0308).
2.2. Acoustic Interaction between Bats and Moths
We examined the acoustic response of the moths to attacking bats to record moth anti-bat clicks and quantify the time of the emergence of moth clicks. We tethered an individual of each moth species in flight in the recording room after dark. Each one was tethered by a nylon thread with a diameter of 0.06 m and a length of 1 m, and the other end was fixed to the ceiling of the recording room. We recorded the acoustic response of the tethered moth to a free-flying R. osgoodi using a microphone (UltraSoundGate CM16/CMPA, Avisoft Bioacoustics, Berlin, Germany) connected to an ultrasound recording interface (UltraSoundGate 116 Hm) at a sample rate of 250 kHz at 16 bits/sample. The microphone was placed at a 30 cm distance from the tethered moth at rest. One of the six bats was randomly selected each night and could fly freely and attack the tethered moth in the recording room. We recorded their behavioral interactions using two digital video cameras (Sony FDR-AX60, Sony, Tokyo, Japan). The moth’s behavioral response was recorded by one camera placed 1.5 m from the moth. The foraging behavior of the bat was recorded by the other camera placed in a corner of the recording room. In each trial, except for the tethered moth, no other moths were in the recording room. Occasionally, the tethered moths produced ultrasonic clicks when being tethered and touched by the experimenter. After one or two minutes, the moths usually kept silent. We released a bat into the recording room after the tethered moth kept silent for more than three minutes. The moths produced clicks only when they were touched by the experimenter and when foraging bats were present and approached them.
2.3. Sound Analysis of Moth Ultrasonic Clicks and Bat Echolocation Calls
We measured the dominant frequency (the frequency of maximum amplitude), the maximum frequency, the minimum frequency, and the duty cycle of moth anti-bat clicks and bat echolocation calls. The duty cycle was calculated following the methods reported by Barber and Conner (2006) [30] and Kawahara and Barber (2015) [24]. In brief, the number of moth clicks in a 100 ms window was multiplied by the average click duration of the modulation cycle, and this value was then divided by 100 to obtain the duty cycle. We measured the dominant frequency using a Hamming window (a type of raised cosine window that is useful in obtaining a smooth spectral representation; FFT 4096, frequency resolution 61 Hz) in Avisoft SASLab Pro and measured the maximum and minimum frequency as the upper and lower frequencies that were −30 dB from the dominant frequency. Temporal parameters were measured from the oscillograms. In each moth-click sequence (a sequence of clicks with short inter-click silence intervals), we selected 8–12 high-quality clicks for sound analysis. These were produced during the period between the bat taking off from the walls and the end of moth capture. Low-quality or overloaded clicks were not utilized for acoustic analysis. The high-quality echolocation calls (signal-to-noise ratio >40 dB) emitted by bats when attacking the tethered moth were adopted for sound analysis. Bats typically produced two to three call sequences during the attacking period, and each call sequence included 12–22 calls. A mean value of each frequency parameter was calculated for each click/call sequence. A total of 46 click sequences of 46 moths and 15 echolocation call sequences of five bats were selected for sound analysis (Table 1; see the Section 3).

Table 1.
Acoustic parameters of moth ultrasonic clicks and bat echolocation calls. Abbreviations: fdom, dominant frequency (kHz); fmax, maximum frequency (kHz); fmin, minimum frequency (kHz); DC, Duty cycle; n, the number of click/call sequences; SD, standard deviation.
2.4. Timing of Ultrasound Production in Moths
We calculated the time difference between the emergence of a moth click and the echolocation “feeding buzz” of an attacking bat. The horseshoe bats shortened the duration and inter-pulse intervals of their echolocation calls, similar to the “feeding buzz” of some low-duty-cycle bats, in the terminal phase prior to capturing an insect [35]. The feeding buzz of R. osgoodi was distinctly marked by the decrease in pulse duration (typically 8–16 ms) and inter-pulse intervals (typically 2–3 ms), which were shorter than the duration (typically 30–60 ms) and inter-pulse intervals (typically 20–30 ms) of echolocation pulses during the approach phase, before the onset of the terminal buzz. We determined the start of a feeding buzz based on the rapid decrease in pulse duration and inter-pulse intervals.
2.5. Moth Palatability
We examined the palatability of C. minor and C. transiens to R. osgoodi, following the reported literature methods [18]. We held the abdomen of the moths with forceps and allowed them to beat their wings 1–3 cm in front of the bat’s mouth. Each bat was fed with one moth. A moth that was rejected by a bat would not be fed to another bat. Six R. osgoodi were selected to examine the palatability of the two moth species. A moth was considered eaten if the bat consumed the head, thorax, and abdomen. We considered a moth species palatable to a bat species if the former was eaten by more than three of the six bats. If a bat rejected a moth, we fed the bat a mealworm and then presented the moth a second time. The moth was considered undesirable if the bat repeatedly rejected it. A moth species was considered unpalatable to a bat species if it was rejected by more than three individuals of the bat species.
2.6. Statistical Analysis
The normality of the data distribution was tested by the Shapiro–Wilk test. The homogeneity of the data was tested utilizing Bartlett’s test prior to statistical analysis. We performed the Kruskal–Wallis rank sum tests with a multiple comparison (the Dunn’s Kruskal–Wallis test) to evaluate whether the acoustic parameters of moth clicks differed significantly from those of bat echolocation calls. Moreover, we utilized Wilcoxon rank sum exact tests to test for differences in maximum and minimum frequency between the moth species and the bats. All the statistical analyses were performed in R version 4.1.3.
3. Results
3.1. Acoustic Characteristics of Moth Ultrasonic Clicks and Bat Echolocation Calls
We recorded anti-bat clicks of 23 C. minor and 23 C. transiens. There was a significant difference in the dominant frequencies among moths and bats (Kruskal–Wallis rank sum test: χ2 = 25.99, df = 2, p < 0.001; Table 1; Figure 1 and Figure 2; Supplementary File). The average dominant frequencies of the clicks of the two moth species were significantly lower than those of the echolocation calls of R. osgoodi (Dunn Kruskal–Wallis multiple comparisons: C. minor vs. R. osgoodi, Z = −2.81, Padj = 0.008; C. transiens vs. R. osgoodi, Z = −5.09, Padj < 0.001). The average maximum frequency of the clicks of C. minor did not differ significantly from that of the echolocation calls of R. osgoodi (Wilcoxon rank sum exact test: W = 178, p = 0.883). The clicks of C. transiens did not overlap spectrally with the echolocation calls of R. osgoodi. The maximum frequency of the clicks of C. transiens was significantly lower than the minimum frequency of the echolocation calls of R. osgoodi (Wilcoxon rank sum exact test: W = 0, p < 0.001). C. minor and C. transiens produced ultrasonic clicks with an average high-duty cycle of 22.3% (range: 9.5–28.4%) and 4.9% (range: 2.1–9.0%), respectively.
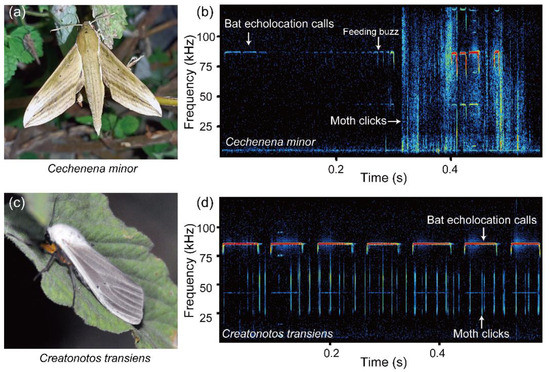
Figure 1.
Examples of echolocation calls of Rhinolophus osgoodi and anti-bat ultrasonic clicks. (a,c) The photos of Cechenena minor and Creatonotos transiens, which are capable of producing anti-bat ultrasonic clicks. (b,d) Examples of acoustic interactions between the clicking moths and echolocating bats. (b) C. minor emitted anti-bat clicks immediately after the end of the bat feeding buzz. (d) C. transiens emitted clicks before the emergence of the bat feeding buzzes.
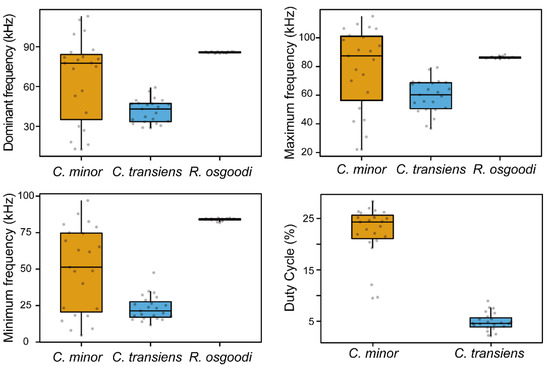
Figure 2.
Box plots of the acoustic parameters of moth ultrasonic clicks and bat echolocation calls. Graph shows the data as jittered dots. Each dot represents the value for one click/call sequence. The number of samples per condition is: C. minor = 23, C. transiens = 23, R. osgoodi = 15. The box indicates the interquartile range (IQR), the whiskers show the range of values that are within 1.5 × IQR, and a horizontal line indicates the median.
3.2. Timing of Ultrasound Production in Moths
The time difference between the emergence of moth clicks and the bat feeding buzz was calculated for 18 C. minor and 19 C. transiens. The ultrasonic clicks of several moths had a low signal-to-noise ratio and were not used for calculating the timing of ultrasound production. In C. minor, 44.4% of click sequences (8 out of 18) were produced before (869 ± 399 ms) the emergence of the feeding buzzes of the attacking bats, and 50% (9 out of 18) of click sequences overlapped temporally with the feeding buzzes of R. osgoodi. C. minor occasionally (4 out of 18) emitted anti-bat clicks immediately after the end of the bat feeding buzz (Figure 1). In C. transiens, 63.2% (12 out of 19) of click sequences were produced before (1511 ± 1121 ms) the emergence of the feeding buzzes of attacking bats, and 57.9% (11 out of 19) of the click sequences did not overlap temporally with the bat feeding buzzes. It took about one second for the bats to fly from the walls to the position of the tethered moths. Ten of 19 C. transiens emitted clicks 1.2–3.5 s before the emergence of the bat feeding buzzes, i.e., before the bats took off from the walls of the recording room.
3.3. Moth Palatability
C. minor was palatable and C. transiens was unpalatable to R. osgoodi. All six R. osgoodi ate the head, thorax, and abdomen, except for the wings, of C. minor. The bats usually bit and consumed the head and thorax of the moths first, and then consumed the abdomen. R. osgoodi did not consume any parts of the body of C. transiens. The bats initially bit but then spat out the head, thorax, or abdomen of C. transiens; after that, they did not bite the moths again. Even if we put C. transiens into the bats’ mouths with forceps, the bats refused to consume the moths. By comparison, when we fed the bats mealworms, they quickly consumed them.
4. Discussion
The goal of this study was to examine whether the jamming hypothesis and the aposematism hypothesis could explain how ultrasound production in C. minor and C. transiens decreased the capture success of HDC bats. We found that the anti-bat clicks of C. minor were at high-duty cycles (22.3%) and those of C. transiens were at low-duty cycles (4.9%). The duty cycles of the clicks of C. minor are relatively lower than those of clicks of some moth species, for example, Carales arizonensis (38%) and Bertholdia trigona (46%) [28,29,36], which can jam bat echolocation. The moth species we studied produced ultrasonic clicks of a broad bandwidth (C. minor, 49.7–77.8 kHz; C. transiens, 23.3–59.3 kHz), which overlap spectrally with the echolocation calls of most low-duty-cycle bats (e.g., Myotis dasycneme: 29.4–73.2 kHz; Myotis daubentonii: 32.9–90.1 kHz; Eptesicus fuscus; 20–50 kHz [37,38]). In more than 57% of cases, C. transiens produced anti-bat clicks before the emergence of bat feeding buzzes.
Moth anti-bat clicks at high-duty cycles (>20%) and produced during the terminal phase of a bat’s attack are expected to serve a jamming function [17,24,28]. In this study, C. minor produced anti-bat clicks at high-duty cycles, suggesting that this moth species might be capable of reducing the probability of being captured by R. osgoodi through sonar jamming. In half the cases, C. minor did not produce anti-bat clicks during the bat buzz phase. Barber and Conner (2006) concluded that the timing of moth acoustical response to bat echolocation calls is not a diagnostic parameter of a jamming function for clicking moths [30]. In C. transiens, anti-bat clicks were at low-duty cycles. Moreover, we found that the clicks of C. transiens did not overlap spectrally with the echolocation calls of R. osgoodi. Several laboratory studies have shown that horseshoe bats exhibit more marked changes in echolocation signal parameters in response to interfering sounds that overlap spectrally with the echolocation signals than to non-overlapping interfering sounds [39,40]. The degree of spectral overlap between moth clicks and bat echolocation signals might be a crucial factor affecting the degree to which moth clicks interfere with the echolocation calls of foraging bats. The ultrasonic clicks of C. transiens might not be capable of jamming high-frequency echolocation calls of bat species such as R. osgoodi. Some studies have shown that echolocating bats adjust their vocalization in response to noise that does not overlap spectrally with their echolocation signals [39,41]. Non-overlapping noise can also reduce bat foraging efficiency [42]. We do not rule out the possibility that the clicks of C. transiens were sources of noise, decreasing the capture success rates of R. osgoodi.
Some tiger moths use ultrasonic clicks to advertise their distastefulness [25,26,27]. In our study, C. transiens was unpalatable to R. osgoodi. More than half the click sequences of C. transiens were produced before the emergence of bat feeding buzzes and did not overlap temporally with the feeding buzzes. These results suggest that the clicks of C. transiens might serve as an acoustic warning of toxicity. C. minor was palatable to the horseshoe bats. The anti-bat clicks of C. minor were not the honest signals of distastefulness. C. minor occasionally emitted anti-bat clicks immediately after the end of the bat feeding buzzes; these clicks cannot serve as a jamming function. We used wild adult bats for the bat--moth interactions. It seems unlikely that the clicks produced after the bat buzz phase decrease the capture success of R. osgoodi by startling the bats [12]. After catching a moth, a bat normally spends several tens of milliseconds transferring the prey from its wing membranes to its mouth; the moth can use the time to offer its acoustic warning [30]. The clicks of C. minor might also serve as an acoustic warning.
In conclusion, our results showed that C. minor produced anti-bat clicks at high-duty cycles. The clicks of C. minor might be capable of jamming the echolocation calls of R. osgoodi. C. minor was palatable to R. osgoodi. C. minor might also use ultrasonic clicks as warning signals of distastefulness. C. transiens produced anti-bat clicks at low-duty cycles, and the clicks of C. transiens did not overlap spectrally with the echolocation calls of R. osgoodi. The clicks of C. transiens might not be capable of jamming the echolocation calls of R. osgoodi. C. transiens was unpalatable to R. osgoodi and probably used ultrasonic clicks to advertise its distastefulness. Future work would benefit from collecting more evidence to support these conclusions. This study may aid in a better understanding of how ultrasound production in moths helps the prey avoid being captured by horseshoe bats. Both the aposematism and jamming hypotheses, which have been proposed for the acoustic interactions between moths and LDC bats [5,16,24], can explain the influences of moth clicks on the capture success of horseshoe bats. This study also highlights the diversity of moth anti-bat behaviors and potentially provides new insights into the biodiversity and conservation of bats. It would be interesting for future studies to investigate how moth clicks might jam the echolocation calls of HDC bats.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/d15070804/s1, Supplementary File: Examples of acoustic interactions between click moths and Rhinolophus osgoodi.
Author Contributions
A.L. and Y.H. designed the experiments; Y.H., J.L. and M.Z. performed the research, Y.H. and A.L. analyzed the data and wrote the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
This project was supported by the National Natural Science Foundation of China [grant number: 31872681, 32071492, and 32271558].
Institutional Review Board Statement
This study was approved by the Science and Technology Ethics Committee of North-east Normal University, China (permit ID: NENU-2022-0308).
Data Availability Statement
The data presented in this study are available in this article.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Hassell, M.P.; May, R.M. From individual behaviour to population dynamics. In Behavioural Ecology: Ecological Consequences of Adaptive Behaviour; Sibly, R.M., Smith, R.H., Eds.; Blackwell: Oxford, UK, 1985; pp. 3–32. [Google Scholar]
- Parker, G.A.; Hammerstein, P. Game Theory and Animal Behaviour. In Evolution: Essays in Honour of John Maynard Smith, Harvey, P., Slatkin, M., Greenwood, P., Eds.; Cambridge University Press: Cambridge, UK, 1985; pp. 73–94. [Google Scholar]
- Ives, A.R.; Dobson, A.P. Antipredator behavior and the population dynamics of simple predator-prey systems. Am. Nat. 1987, 130, 431–447. [Google Scholar] [CrossRef]
- Sih, A.; Christensen, B. Optimal diet theory: When does it work, and when and why does it fail? Anim. Behav. 2001, 61, 379–390. [Google Scholar] [CrossRef]
- Corcoran, A.J.; Barber, J.R.; Conner, W.E. Tiger moth jams bat sonar. Science 2009, 325, 325–327. [Google Scholar] [CrossRef]
- Mark, C.J.; O’Hanlon, J.C.; Holwell, G.I. Camouflage in lichen moths: Field predation experiments and avian vision modelling demonstrate the importance of wing pattern elements and background for survival. J. Anim. Ecol. 2022, 91, 2358–2369. [Google Scholar] [CrossRef]
- Hounslow, J.L.; Jewell, O.J.D.; Fossette, S.; Whiting, S.; Tucker, A.D.; Richardson, A.; Edwards, D.; Gleiss, A.C. Animal-borne video from a sea turtle reveals novel anti-predator behaviors. Ecology 2021, 102, e03251. [Google Scholar] [CrossRef]
- Karakoç, C.; Clark, A.T.; Chatzinotas, A. Diversity and coexistence are influenced by time-dependent species interactions in a predator–prey system. Ecol. Lett. 2020, 23, 983–993. [Google Scholar] [CrossRef]
- Van Nieukerken, E.J. Order Lepidoptera Linnaeus, 1758. Zootaxa 2011, 3148, 212–221. [Google Scholar]
- Miller, L.A.; Surlykke, A. How some insects detect and avoid being eaten by bats: Tactics and countertactics of prey and predator. BioScience 2001, 51, 570–581. [Google Scholar] [CrossRef]
- Waters, D.A. Bats and moths: What is there left to learn? Physiol. Entomol. 2003, 28, 237–250. [Google Scholar] [CrossRef]
- Conner, W.E.; Corcoran, A.J. Sound strategies: The 65-million-year-old battle between bats and insects. Annu. Rev. Entomol. 2012, 57, 21–39. [Google Scholar] [CrossRef]
- Jones, G.; Teeling, E.C. The evolution of echolocation in bats. Trends Ecol. Evol. 2006, 21, 149–156. [Google Scholar] [CrossRef] [PubMed]
- ter Hofstede, H.M.; Ratcliffe, J.M. Evolutionary escalation: The bat–moth arms race. J. Exp. Biol. 2016, 219, 1589–1602. [Google Scholar] [CrossRef] [PubMed]
- Barber, J.R.; Plotkin, D.; Rubin, J.J.; Kawahara, A.Y. Anti-bat ultrasound production in moths is globally and phylogenetically widespread. Proc. Natl. Acad. Sci. USA. 2022, 119, e2117485119. [Google Scholar] [CrossRef]
- Fullard, J.H.; Fenton, B.M.; Simmons, J.A. Jamming bat echolocation: The clicks of arctiid moths. Can. J. Zool. 1979, 57, 647–649. [Google Scholar] [CrossRef]
- Fullard, J.H.; Simmons, J.A.; Saillant, P.A. Jamming bat echolocation: The dogbane tiger moth Cycnia tenera times its clicks to the terminal attack calls of the big brown bat Eptesicus fuscus . J. Exp. Biol. 1994, 194, 285–298. [Google Scholar] [CrossRef] [PubMed]
- Corcoran, A.J.; Hristov, N.I. Convergent evolution of anti-bat sounds. J. Comp. Physiol. A 2014, 200, 811–821. [Google Scholar] [CrossRef] [PubMed]
- Barber, J.R.; Kawahara, A.Y. Hawkmoths produce anti-bat ultrasound. Biol. Lett. 2013, 9, 20130161. [Google Scholar] [CrossRef] [PubMed]
- Barber, J.R.; Chadwell, B.A.; Garrett, N.; Schmidt-French, B.; Conner, W.E. Naive bats discriminate arctiid moth warning sounds but generalize their aposematic meaning. J. Exp. Biol. 2009, 212, 2141–2148. [Google Scholar] [CrossRef] [PubMed]
- Corcoran, A.J.; Conner, W.E. Sonar jamming in the field: Effectiveness and behavior of a unique prey defense. J. Exp. Biol. 2012, 215, 4278–4287. [Google Scholar] [CrossRef]
- Bates, D.L.; Fenton, M.B. Aposematism or startle? Predators learn their responses to the defenses of prey. Can. J. Zool. 1990, 68, 49–52. [Google Scholar] [CrossRef]
- Corcoran, A.J.; Barber, J.R.; Hristov, N.I.; Conner, W.E. How do tiger moths jam bat sonar? J. Exp. Biol. 2011, 214, 2416–2425. [Google Scholar] [CrossRef] [PubMed]
- Kawahara, A.Y.; Barber, J.R. Tempo and mode of antibat ultrasound production and sonar jamming in the diverse hawkmoth radiation. Proc. Natl. Acad. Sci. USA. 2015, 112, 6407–6412. [Google Scholar] [CrossRef] [PubMed]
- Dunning, D.C. Warning sounds of moths. Z. fÜR Tierpsychol. 1968, 25, 129–138. [Google Scholar] [CrossRef]
- Dunning, D.C.; Acharya, L.; Merriman, C.B.; Ferro, L.D. Interactions between bats and arctiid moths. Can. J. Zool. 1992, 70, 2218–2223. [Google Scholar] [CrossRef]
- Hristov, N.I.; Conner, W.E. Sound strategy: Acoustic aposematism in the bat–tiger moth arms race. Naturwissenschaften 2005, 92, 164–169. [Google Scholar] [CrossRef]
- Corcoran, A.J.; Conner, W.E.; Barber, J.R. Anti-bat tiger moth sounds: Form and function. Curr. Zool. 2010, 56, 358–369. [Google Scholar] [CrossRef]
- Fernández, Y.; Dowdy, N.J.; Conner, W.E. High duty cycle moth sounds jam bat echolocation: Bats counter with compensatory changes in buzz duration. J. Exp. Biol. 2022, 225, jeb244187. [Google Scholar] [CrossRef]
- Barber, J.R.; Conner, W.E. Tiger moth responses to a simulated bat attack: Timing and duty cycle. J. Exp. Biol. 2006, 209, 2637–2650. [Google Scholar] [CrossRef] [PubMed]
- Dowdy, N.J.; Conner, W.E. Acoustic aposematism and evasive action in select chemically defended arctiine (Lepidoptera: Erebidae) species: Nonchalant or not? PLoS ONE 2016, 11, e0152981. [Google Scholar] [CrossRef]
- Barber, J.R.; Conner, W.E. Acoustic mimicry in a predator–prey interaction. Proc. Natl. Acad. Sci. USA. 2007, 104, 9331–9334. [Google Scholar] [CrossRef]
- Fenton, M.B.; Faure, P.A.; Ratcliffe, J.M. Evolution of high duty cycle echolocation in bats. J. Exp. Biol. 2012, 215, 2935–2944. [Google Scholar] [CrossRef] [PubMed]
- Schnitzler, H.-U.; Denzinger, A. Auditory fovea and Doppler shift compensation: Adaptations for flutter detection in echolocating bats using CF-FM signals. J. Comp. Physiol. A 2011, 197, 541–559. [Google Scholar] [CrossRef]
- Schnitzler, H.-U.; Kalko, E.K.V. Echolocation by insect-eating bats. BioScience 2001, 51, 557–569. [Google Scholar] [CrossRef]
- Fernández, Y.; Dowdy, N.J.; Conner, W.E. Extreme duty cycles in the acoustic signals of tiger moths: Sexual and natural selection operating in parallel. Integr. Org. Biol. 2020, 2, obaa046. [Google Scholar] [CrossRef] [PubMed]
- Siemers, B.M.; Schnitzler, H.-U. Echolocation signals reflect niche differentiation in five sympatric congeneric bat species. Nature 2004, 429, 657–661. [Google Scholar] [CrossRef] [PubMed]
- Surlykke, A.; Moss, C.F. Echolocation behavior of big brown bats, Eptesicus fuscus, in the field and the laboratory. J. Acoust. Soc. Am. 2000, 108, 2419–2429. [Google Scholar] [CrossRef]
- Hage, S.R.; Jiang, T.; Berquist, S.W.; Feng, J.; Metzner, W. Ambient noise induces independent shifts in call frequency and amplitude within the Lombard effect in echolocating bats. Proc. Natl. Acad. Sci. USA. 2013, 110, 4063. [Google Scholar] [CrossRef] [PubMed]
- Hage, S.R.; Jiang, T.; Berquist, S.W.; Feng, J.; Metzner, W. Ambient noise causes independent changes in distinct spectro-temporal features of echolocation calls in horseshoe bats. J. Exp. Biol. 2014, 217, 2440–2444. [Google Scholar] [CrossRef]
- Luo, J.; Goerlitz, H.R.; Brumm, H.; Wiegrebe, L. Linking the sender to the receiver: Vocal adjustments by bats to maintain signal detection in noise. Sci. Rep. 2015, 5, 18556. [Google Scholar] [CrossRef]
- Luo, J.; Siemers, B.M.; Koselj, K. How anthropogenic noise affects foraging. Glob. Chang. Biol. 2015, 21, 3278–3289. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).