Effect of Antiparasitic Management of Cattle on the Diversity and Functional Structure of Dung Beetle (Coleoptera: Scarabaeidae) Assemblages in the Colombian Caribbean
Abstract
1. Introduction
2. Material and Methods
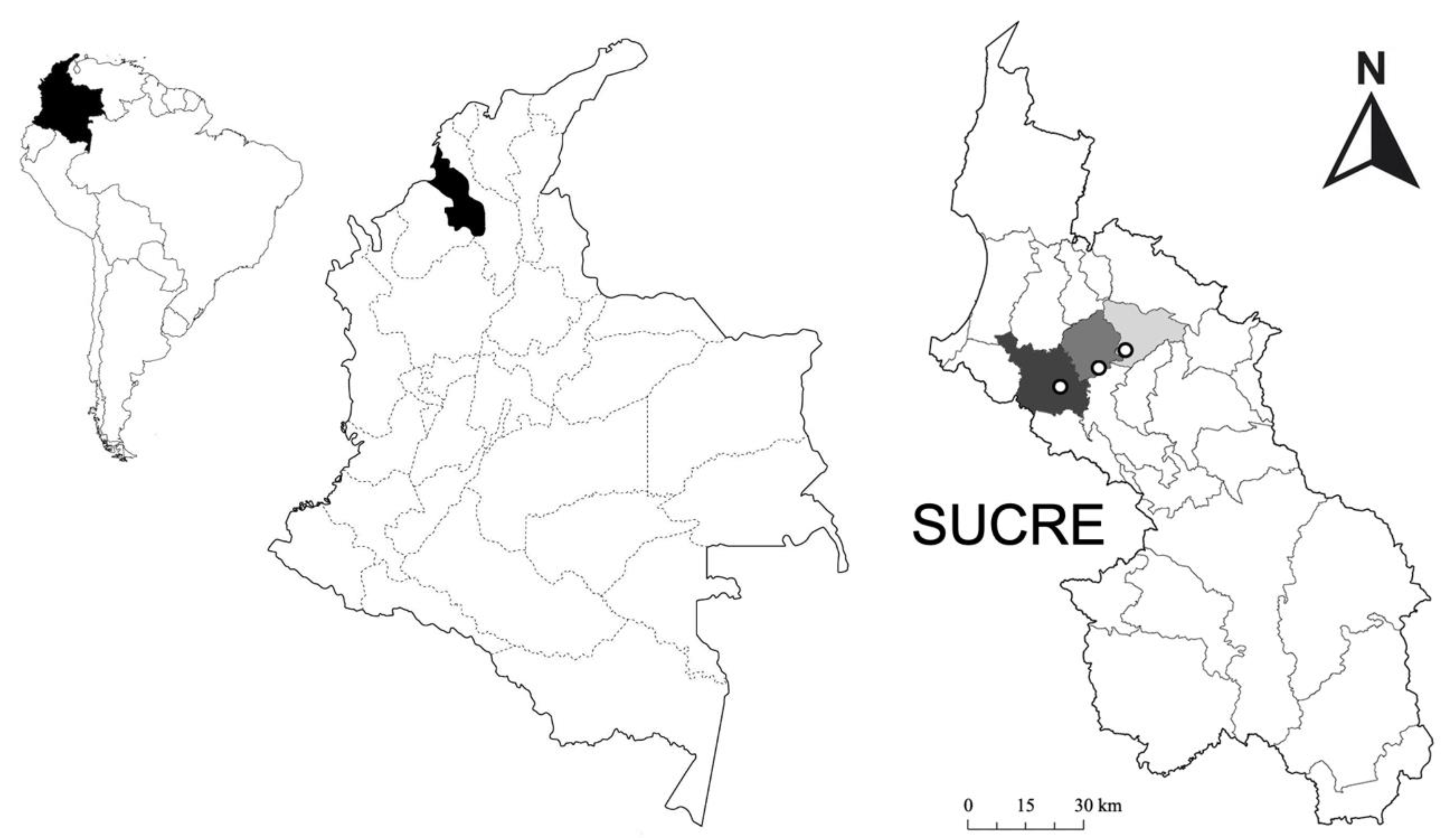
2.1. Study Area
2.2. Livestock Management
2.3. Sampling and Experimental Design
2.4. Dung Beetles’ Diversity, Identification, and Biomass
2.5. Data Analysis
3. Results
3.1. Seasonal Differences in Dung Beetle Assemblages
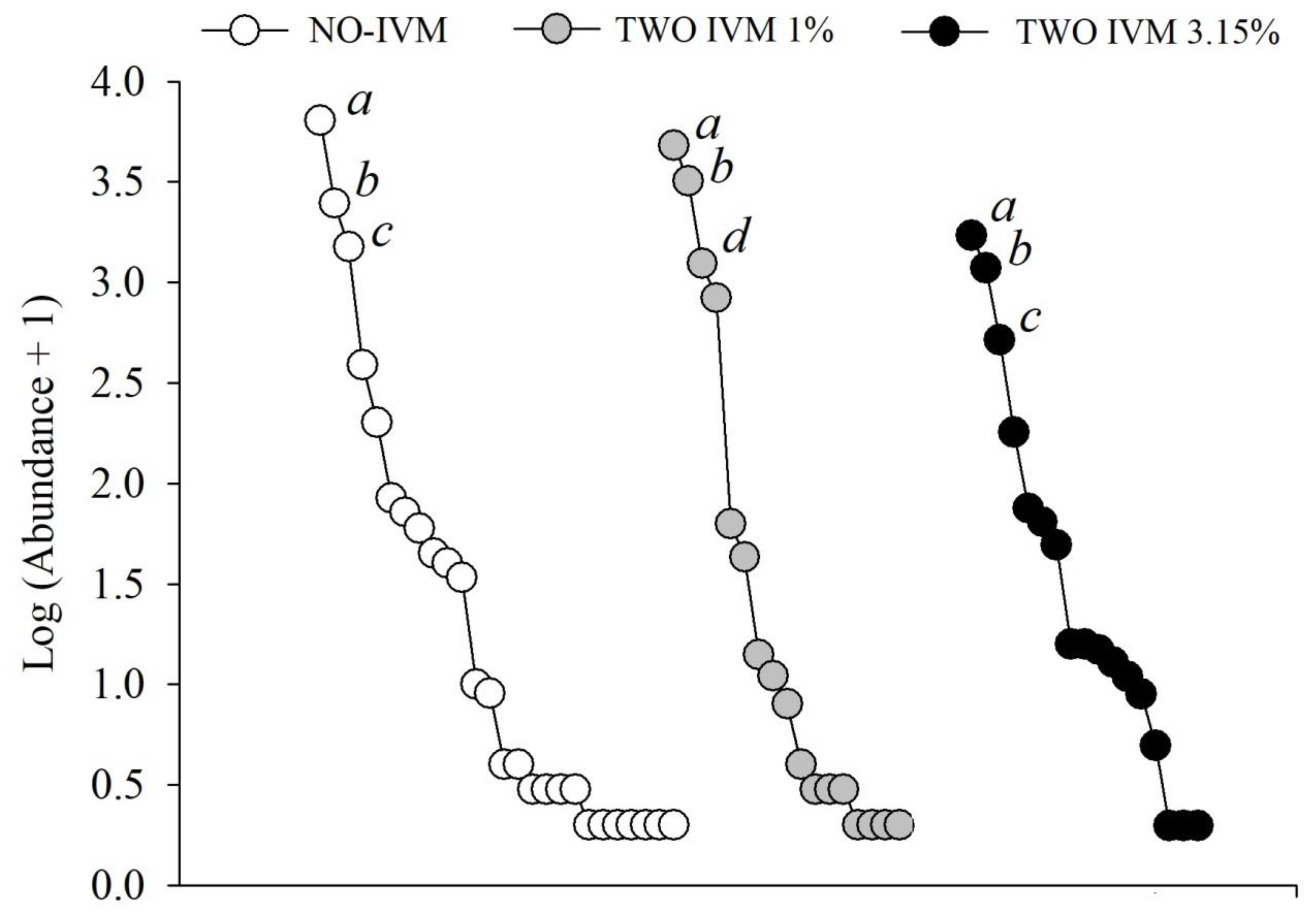
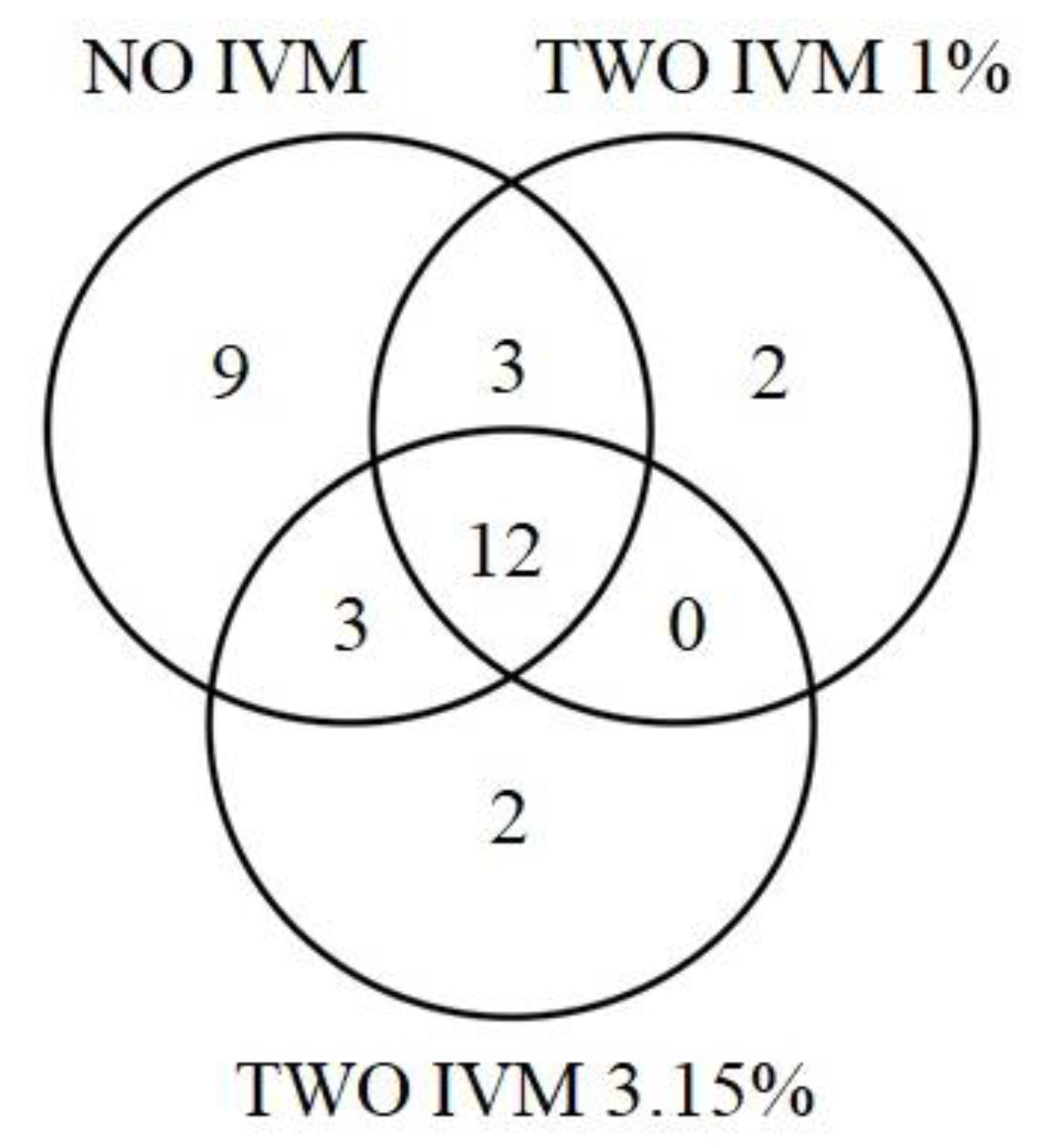
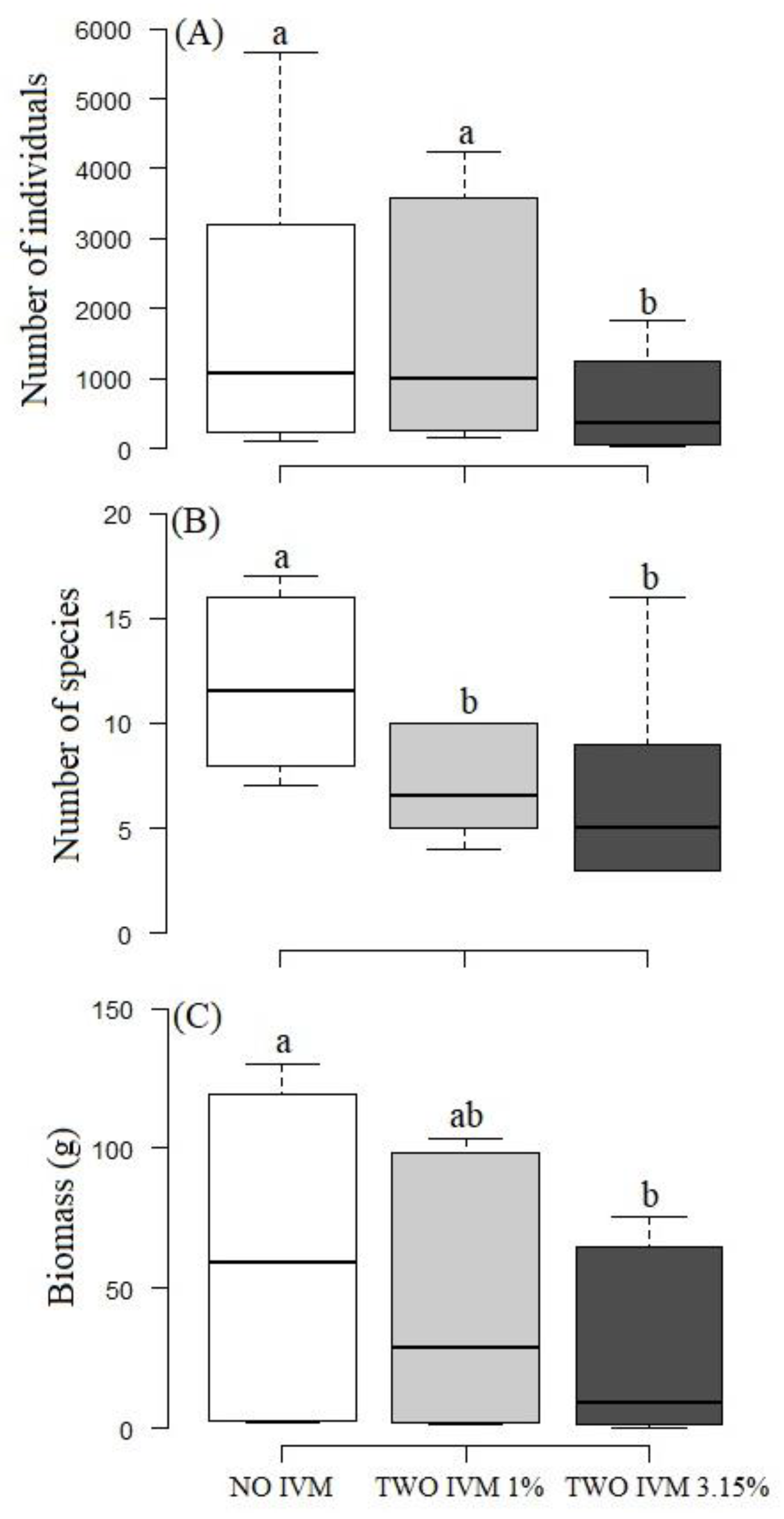
3.2. Differences in Dung Beetle Assemblages between Livestock Treatments
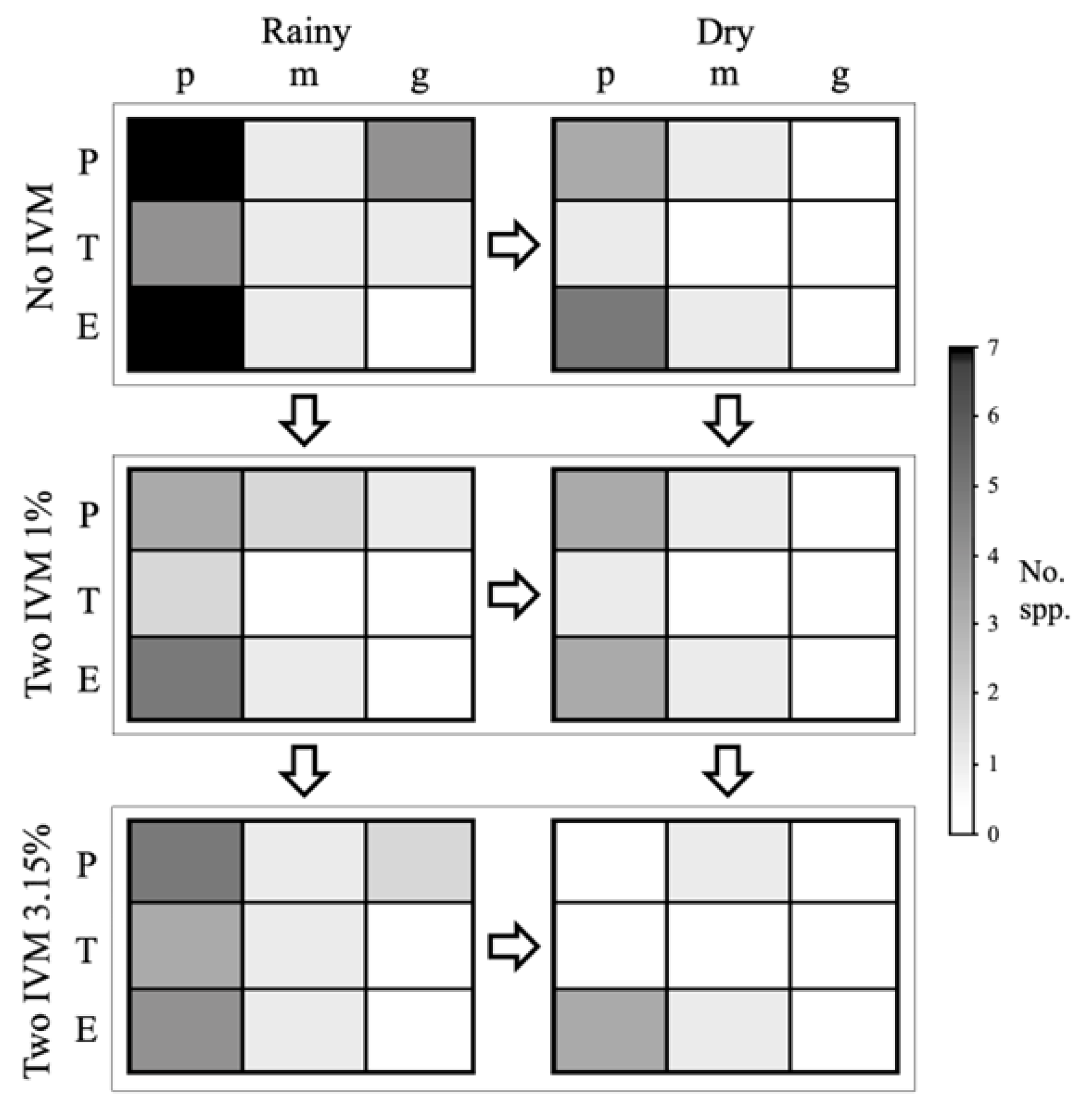
3.3. Differences in Dung Beetle Relocation Guilds between Livestock Treatments
4. Discussion
4.1. Dung Beetles’ Assemblage Analysis in Tropical Dry Forests
4.2. Dung Beetles’ Assemblage Structure according to Seasonality
4.3. Negative Effects of Livestock Management and Antiparasitic Use
5. Conclusions and Recommendations
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hernández, B.; Maes, J.; Harvey, C.; Vílches, S.; Medina, A.; Sánchez, D. Abundancia y diversidad de escarabajos coprófagos y mariposas diurnas en un paisaje ganadero en el departamento de Rivas, Nicaragua. Agrofor. Am. 2003, 10, 93–102. [Google Scholar]
- Flota-Bañuelos, C.; López-Collado, J.; Vargas-Mendoza, M.; Fajersson, P.; González-Hernández, H.; Martínez-Morales, I. Efecto de la Ivermectina en la dinámica espacio-temporal de escarabajos estercoleros en Veracruz, Mexico. Trop. Subtrop. Agroecosyst. 2012, 15, 227–239. [Google Scholar]
- FEDEGAN. Ganadería Colombiana Hoja de Ruta 2018–2022; FEDEGAN: Bogotá, Colombia, 2018; p. 126. [Google Scholar]
- ICA (Instituto Colombiano Agropecuario). Censos Pecuarios Nacional: Bogotá, Colombia. 2022. Available online: https://www.ica.gov.co/areas (accessed on 1 December 2022).
- IGAC (Instituto Geográfico Agustín Codazzi). XVIII Congreso Nacional de la Ciencia del Suelo; IGAC (Instituto Geográfico Agustín Codazzi): Villa de Leyva, Colombia, 2016. [Google Scholar]
- Doube, B.M. A functional classification for analysis of the structure of dung beetle assemblages. Ecol. Èntomol. 1990, 15, 371–383. [Google Scholar] [CrossRef]
- O´Hea, N.; Kirwan, L.; Finn, J. Experimental mixtures of dung fauna effect dung decomposition through complex effects of species interactions. Oikos 2010, 119, 1081–1088. [Google Scholar] [CrossRef]
- Lumaret, J.-P.; Kadiri, N. The influence of the first wave of colonizing insects on cattle dung dispersal. Pedobiologia 1995, 39, 506–517. [Google Scholar]
- Beynon, S.A.; Mann, D.J.; Slade, E.M.; Lewis, O.T. Species-rich dung beetle communities buffer ecosystem services in perturbed agro-ecosystems. J. Appl. Ecol. 2012, 49, 1365–1372. [Google Scholar] [CrossRef]
- Correa, C.M.A.; Braga, R.F.; Louzada, J.; Menéndez, R. Dung beetle diversity and functions suggest no major impacts of cattle grazing in the Brazilian Pantanal wetlands. Ecol. Èntomol. 2019, 44, 524–533. [Google Scholar] [CrossRef]
- Martínez, I.; Cruz, M.; Montes de Oca, E.; Suárez, T. La Función de los Escarabajos del Estiércol en los Pastizales Ganaderos, 1st ed.; Secretaría de Educación de Veracruz: México City, México, 2011; p. 49.
- Brown, J.; Scholtz, C.H.; Janeau, J.-L.; Grellier, S.; Podwojewski, P. Dung beetles (Coleoptera: Scarabaeidae) can improve soil hydrological properties. Appl. Soil Ecol. 2010, 46, 9–16. [Google Scholar] [CrossRef]
- Irshad, M.; E Eneji, A.; Hussain, Z.; Ashraf, M. Chemical characterization of fresh and composted livestock manures. J. Soil Sci. Plant Nutr. 2013, 13, 115–121. [Google Scholar] [CrossRef]
- Sommer, C.; Bibby, B.M. The influence of veterinary medicines on the decomposition of dung organic matter in soil. Eur. J. Soil Biol. 2002, 38, 155–159. [Google Scholar] [CrossRef]
- Badenhorst, J.; Dabrowski, J.; Scholtz, C.H.; Truter, W.F. Dung beetle activity improves herbaceous plant growth and soil properties on confinements simulating reclaimed mined land in South Africa. Appl. Soil Ecol. 2018, 132, 53–59. [Google Scholar] [CrossRef]
- Lobo, J.; Veiga, C. Interés ecológico de la fauna coprófaga en pastos de uso ganadero. Ecología 1990, 4, 313. [Google Scholar]
- Nichols, E.; Spector, S.; Louzada, J.; Larsen, T.; Amezquita, S.; Favila, M.; The Scarabaeinae Research Network. Ecological functions and ecosystem services provided by Scarabaeinae dung beetles. Biol. Conserv. 2008, 141, 1461–1474. [Google Scholar] [CrossRef]
- Verdú, J.R.; Lobo, J.M.; Sánchez-Piñero, F.; Gallego, B.; Numa, C.; Lumaret, J.-P.; Cortez, V.; Ortiz, A.J.; Tonelli, M.; García-Teba, J.P.; et al. Ivermectin residues disrupt dung beetle diversity, soil properties and ecosystem functioning: An interdisciplinary field study. Sci. Total Environ. 2018, 618, 219–228. [Google Scholar] [CrossRef]
- Braga, R.F.; Korasaki, V.; Andresen, E.; Louzada, J. Dung beetle community and functions along a habitat-disturbance gradient in the amazon: A rapid assessment of ecological functions associated to biodiversity. PLoS ONE 2013, 8, e57786. [Google Scholar] [CrossRef] [PubMed]
- Sands, B.; Wall, R. Sustained parasiticide use in cattle farming affects dung beetle functional assemblages. Agric. Ecosyst. Environ. 2018, 265, 226–235. [Google Scholar] [CrossRef]
- Coop, R.; Kyriazakis, I. Nutrition-parasite interaction. Vet. Parasitol. 1999, 84, 187–204. [Google Scholar] [CrossRef]
- Lumaret, J.-P.; Martínez, I. El impacto de productos veterinarios sobre insectos coprófagos: Consecuencias sobre la degradación del estiércol en pastizales. Acta Zoológica Mex. 2005, 21, 137–148. [Google Scholar] [CrossRef]
- Floate, K.D. Endectocide use in cattle and fecal residues: Environmental effects in Canada. Can. J. Vet.-Res. 2006, 70, 1–10. [Google Scholar]
- Lumaret, J.-P.; Errouissi, F. Use of anthemilthicides in herbivores and of risks for the non target fauna of pastures. Vet. Res. 2002, 33, 547–562. [Google Scholar] [CrossRef]
- Campbell, W. Ivermectin and Abamectin; Springer Science Business Media: Berlin/Heidelberg, Germany, 1989. [Google Scholar]
- Jackson, H. Ivermectin as a systemic insecticide. Parasitol. Today 1989, 5, 146–156. [Google Scholar] [CrossRef] [PubMed]
- Molinari, G. Ivermectinas: Evaluación de su Efecto Deletéreo Mediante Ensayos de Genotoxicidad. Ph.D. Thesis, Facultad de Ciencias Naturales y Museo, Universidad Nacional de la Plata, La Plata, Argentina, 2010. [Google Scholar]
- Wohde, M.; Blanckenhorn, W.U.; Floate, K.D.; Lahr, J.; Lumaret, J.-P.; Römbke, J.; Scheffczyk, A.; Tixier, T.; Düring, R.-A. Analysis and dissipation of the antiparasitic agent ivermectin in cattle dung under different field conditions. Environ. Toxicol. Chem. 2016, 35, 1924–1933. [Google Scholar] [CrossRef] [PubMed]
- González Canga, A.; Sahagún Prieto, A.M.; José Diez Liébana, M.; Martínez, N.F.; Vega, M.S.; Vieitez, J.J.G. The pharmacokinetics and metabolism of ivermectin in domestic animal species. Vet. J. 2009, 179, 25–37. [Google Scholar] [CrossRef] [PubMed]
- Floate, K.D. Endectocide residues affect insect attraction to dung from treated cattle: Implications for toxicity tests. Med. Veter-Èntomol. 2007, 21, 312–322. [Google Scholar] [CrossRef] [PubMed]
- Martínez, I.M.; Lumaret, J.-P.; Zayas, R.O.; Kadiri, N. The effects of sublethal and lethal doses of ivermectin on the reproductive physiology and larval development of the dung beetle Euoniticellus intermedius (Coleoptera: Scarabaeidae). Can. Èntomol. 2017, 149, 461–472. [Google Scholar] [CrossRef]
- Verdú, J.R.; Cortez, V.; Ortiz, A.J.; González-Rodríguez, E.; Martínez-Pinna, J.; Lumaret, J.-P.; Lobo, J.M.; Numa, C.; Sánchez-Piñero, F. Low doses of ivermectin cause sensory and locomotor disorders in dung beetles. Sci. Rep. 2015, 5, 13912. [Google Scholar] [CrossRef]
- Lumaret, J.-P.; Galante, E.; Lumbreras, C.; Mena, J.; Bertrand, M.; Bernal, J.L.; Cooper, J.F.; Kadiri, N.; Crowe, D. Field effects of Ivermectin residues on dung beetles. J. Appl. Ecol. 1993, 30, 428. [Google Scholar] [CrossRef]
- Floate, K. Does a repellent effect contribute to reduced levels of insect activity in dung from cattle treated with Ivermectin? Bull. Entomol. Res. 1998, 88, 291–297. [Google Scholar] [CrossRef]
- Correa, C.M.A.; Ferreira, K.R.; Abot, A.R.; Louzada, J.; Vaz-De-Mello, F.Z. Ivermectin impacts on dung beetle diversity and their ecological functions in two distinct Brazilian ecosystems. Ecol. Èntomol. 2022, 47, 736–748. [Google Scholar] [CrossRef]
- Ambrožová, L.; Sládeček, F.X.J.; Zítek, T.; Perlík, M.; Kozel, P.; Jirků, M.; Čížek, L. Lasting decrease in functionality and richness: Effects of ivermectin use on dung beetle communities. Agric. Ecosyst. Environ. 2021, 321, 107634. [Google Scholar] [CrossRef]
- Floate, K.D.; Wardhaugh, K.G.; Boxall, A.B.; Sherratt, T.N. Fecal residues of veterinary parasiticides: Nontarget effects in the pasture environment. Annu. Rev. Èntomol. 2005, 50, 153–179. [Google Scholar] [CrossRef]
- Pecenka, J.R.; Lundgren, J.G. Effects of herd management and the use of ivermectin on dung arthropod communities in grasslands. Basic Appl. Ecol. 2019, 40, 19–29. [Google Scholar] [CrossRef]
- Rosales, M.C.; Martinez, I.; López-Collado, J.; Vargas-Mendoza, M.; González-Hernández, H.; Fajersson, P. Effect of ivermectin on the survival and fecundity of Euoniticellus intermedius (Coleoptera: Scarabaeidae). Rev. Biol. Trop. 2012, 60, 333–345. [Google Scholar]
- Basto-Estrella, G.S.; Rodríguez-Vivas, R.I.; Delfín-González, H.; Reyes-Novelo, E. Dung beetle (Coleoptera: Scarabaeinae) diversity and seasonality in response to use of macrocyclic lactones at cattle ranches in the mexican neotropics. Insect Conserv. Divers. 2013, 7, 73–81. [Google Scholar] [CrossRef]
- Villada-Bedoya, S.; Chávez-Ríos, J.R.; Montoya, B.; Castelán, F.; Córdoba-Aguilar, A.; Escobar, F.; González-Tokman, D. Heat shock proteins and antioxidants as mechanisms of response to ivermectin in the dung beetle Euoniticellus intermedius. Chemosphere 2020, 269, 128707. [Google Scholar] [CrossRef] [PubMed]
- Tovar, H.; Noriega, J.A.; Caraballo, P. Efecto de la ivermectina sobre la estructura del ensamblaje de escarabajos coprófagos (Coleoptera: Scarabaidae: Aphodiinae-Scarabaeinae) en las sabanas colombianas de la región Caribe. Actual. Biol. 2016, 38, 157–166. [Google Scholar]
- Clavijo, J.; Barrera, R. Geología de las Planchas 44 Sincelejo y 52 Sahagún; Ministerio de Minas y Energía, Instituto de Investigación e Información Geocientífica, Minero-Ambiental y Nuclear, INGEOMINAS: Cartagena, Colombia, 2001; 63p.
- Holdridge, L. Ecología Basada en Zonas de Vida, 1st ed.; Jiménez, H., Translator; Centro Científico Tropical IICA: San José, Costa Rica, 1987; p. 216. [Google Scholar]
- Navarro, I.; Roman, K.; Gomez, H.; Peréz, A. Listado de escarabajos coprófagos (Coleoptera: Scarabaeidae: Scarabaeinae) de La Serrania de Coraza, Sucre (Colombia). Rev. Colomb. Cienc. Anim. 2011, 3, 262–268. [Google Scholar] [CrossRef]
- Navarro, I.; Roman, K.; Gomez, H.; Peréz, A. Variación estacional en escarabajos coprófagos (Coleoptera: Scarabaeidae: Scarabaeinae) de la serranía de Coraza, Sucre (Colombia). Rev. Colomb. Cienc. Anim. 2011, 3, 102–110. [Google Scholar] [CrossRef]
- Mora-Aguilar, E.F.; Arriaga-Jiménez, A.; Correa, C.M.A.; da Silva, P.G.; Korasaki, V.; López-Bedoya, P.A.; Hernández, M.I.M.; Pablo-Cea, J.D.; Salomão, R.P.; Valencia, G.; et al. Toward a standardized methodology for sampling dung beetles (Coleoptera: Scarabaeinae) in the Neotropics: A critical review. Front. Ecol. Evol. 2023, 11, 1096208. [Google Scholar] [CrossRef]
- Larsen, T.H.; Forsyth, A. Trap spacing and transect design for dung beetle biodiversity studies. Biotropica 2005, 37, 322–325. [Google Scholar] [CrossRef]
- Pérez-Cogollo, L.; Rodríguez-Vivas, R.; Reyes-Novelo, E.; Delfín-González, H.; Muñoz-Rodríguez, D. Survival and reproduction of Onthophagus landolti (Coleoptera: Scarabaeidae) exposed to ivermectin residues in cattle dung. Bull. Èntomol. Res. 2016, 107, 118–125. [Google Scholar] [CrossRef] [PubMed]
- Edmonds, W.D. Revision of Phanaeus Macleay, a New World genus of scarabaeine dung beetles (Coleoptera: Scarabaeidae, Scarabaeinae). Contrib. Sci. 1994, 443, 1–105. [Google Scholar] [CrossRef]
- Genier, F. A revision of the Neotropical genus Ontherus Erichson (Coleoptera: Scarabaeidae, Scarabaeinae). Mem. Entomol. Soc. Can. 1996, 128, 3–170. [Google Scholar] [CrossRef]
- González, F.A.; Molano, F.; Medina, C. Los subgéneros Calhyboma, Hybomidium y Telhyboma (Coleoptera: Scarabaeidae: Scarabaeinae: Deltochilum) en Colombia. Rev. Colomb. Entomol. 2009, 35, 253–274. [Google Scholar] [CrossRef]
- Edmonds, W.; Zídek, J. A taxonomic review of the Neotropical genus Coprophanaeus Olsoufieff, 1924 (Coleoptera: Scarabaeidae, Scarabaeinae). Insecta Mundi 2010, 129, 1–111. [Google Scholar]
- Camero, E. Los escarabajos del género Eurysternus Dalman, 1824 (Coleoptera: Scarabaeidae) de Colombia. Bol. Soc. Entomol. Aragonesa 2010, 46, 147–179. [Google Scholar]
- Vaz-De-Mello, F.; Edmonds, W.D.; Ocampo, F.C.; Schoolmeesters, P. A multilingual key to the genera and subgenera of the subfamily Scarabaeinae of the New World (Coleoptera: Scarabaeidae). Zootaxa 2011, 2854, 1–73. [Google Scholar] [CrossRef]
- Cartwright, O.L. Two new species of Cartwrightia from Central and South America (Coleoptera: Scarabaeidae: Aphodiinae). Proc. U. S. Natl. Mus. 1967, 124, 1–8. [Google Scholar] [CrossRef]
- Dellacasa, G.; Bordat, P.; Dellacasa, M. A revisional essay of world genus-group taxa of Aphodiinae (Coleoptera: Aphodiidae). Mem. Della Soc. Entomol. Ital. Genova 2001, 79, 1–482. [Google Scholar]
- Dellacasa, M.; Gordon, R.D.; Dellacasa, G. Aphodiinae described or recorded by Bates in Biologia Centrali-Americana (Coleoptera Scarabaeoidea: Aphodiidae). Acta Zool. Mex. 2002, 86, 155–223. [Google Scholar] [CrossRef]
- Stebnicka, Z.T. The Genus Ataenius Harold, 1867 (Coleoptera: Scarabaeidae) of New World. Iconography; Institute of Systematics and Evolution of Animals: Kraków, Poland; Polish Academy of Sciences: Warsaw, Poland, 2007. [Google Scholar]
- Halffter, G.; Edmonds, W. The Nesting Behavior of Dung Beetles (Scarabaeinae): An Ecological and Evolutive Approach; Instituto de Ecología: Mexico City, Mexico, 1982; p. 176. [Google Scholar]
- Noriega, J.A.; March-Salas, M.; Castillo, S.; García-Q, H.; Hortal, J.; Santos, A.M.C. Human perturbations reduce dung beetle diversity and dung removal ecosystem function. Biotropica 2021, 53, 753–766. [Google Scholar] [CrossRef]
- Chao, A.; Gotelli, N.J.; Hsieh, T.C.; Sander, E.L.; Ma, K.H.; Colwell, R.K.; Ellison, A.M. Rarefaction and extrapolation with Hill numbers: A framework for sampling and estimation in species diversity studies. Ecol. Monogr. 2014, 84, 45–67. [Google Scholar] [CrossRef]
- Hsieh, T.C.; Ma, K.H.; Chao, A. iNEXT: An R package for rarefaction and extrapolation of species diversity (hill numbers). Methods Ecol. Evol. 2016, 7, 1451–1456. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2022; Available online: https://www.eea.europa.eu/data-and-maps/indicators/oxygen-consuming-substances-in-rivers/r-development-core-team-2006 (accessed on 1 December 2022).
- Crawley, M.J. Statistical Computing–An Introduction to Data Analysis Using S-Plus; John Wiley Sons: London, UK, 2013. [Google Scholar]
- Dufrene, M.; Legendre, P. Species assemblages and indicators species: The need for a flexible asymmetrical approach. Ecol. Monogr. 1997, 67, 345–366. [Google Scholar] [CrossRef]
- Correa, C.M.A.; Peres, N.D.; Holdbrook, R. Patterns of alimentary resource use by dung beetles in introduced Brazilian pastures: Cattle versus sheep dung. Èntomol. Sci. 2020, 23, 271–279. [Google Scholar] [CrossRef]
- Cáceres, M.D.; Jansen, F.; Dell, N. Package ‘Indicspecies’. 2022. Available online: https://cran.r-project.org/web/packages/indicspecies/indicspecies.pdf (accessed on 15 December 2022).
- Locarno, L.C.P.; Schoolmeesters, P. Small dung beetles of Colombia (Coleoptera Scarabaeoidea Aphodiinae) I: Preliminary catalog and key for registered species. Bol. Cient. Mus. Hist. Nat. Univ. Caldas 2019, 23, 279–302. [Google Scholar] [CrossRef]
- Miles, L.; Newton, A.C.; DeFries, R.S.; Ravilious, C.; May, I.; Blyth, S.; Kapos, V.; Gordon, J.E. A global overview of the conservation status of tropical dry forests. J. Biogeogr. 2006, 33, 491–505. [Google Scholar] [CrossRef]
- Portillo-Quintero, C.; Sanchez-Azofeifa, A.; Calvo-Alvarado, J.; Quesada, M.; Santo, M.M.D.E. The role of tropical dry forests for biodiversity, carbon and water conservation in the neotropics: Lessons learned and opportunities for its sustainable management. Reg. Environ. Chang. 2014, 15, 1039–1049. [Google Scholar] [CrossRef]
- IAVH (Instituto de Investigación en Recursos Biológicos Alexander Von Humboldt). El Bosque Seco Tropical (Bs-T) en Colombia; Informe GEMA Programa de Inventario de la Biodiversidad, Instituto Alexander von Humboldt: Bogotá, Colombia, 1998. [Google Scholar]
- Pizano, C.; García, H. El Bosque Seco Tropical en Colombia; Instituto de Investigación de Recursos Biológicos Alexander von Humboldt (IAvH): Bogotá, Colombia, 2014; 353p. [Google Scholar]
- Whitmore, T. Tropical forest disturbance, disappearances, and species loss. In Tropical Forest Remnants: Ecology, Management, and Conservation of Fragmented Communities; Laurance, W.F., Bierregaard, R.O., Eds.; The University of Chicago Press: Chicago, IL, USA, 1997; pp. 3–12. [Google Scholar]
- Harvey, C.; Sáenz, J. Evaluación y Conservación de Biodiversidad en Paisajes Fragmentados de Mesoamérica; Harvey, C.A., Sáenz-Méndez, J.C., Eds.; INBio, CATIE: San José, Costa Rica, 2008; pp. 9–11. [Google Scholar]
- Slade, E.M.; Mann, D.J.; Lewis, O.T. Biodiversity and ecosystem functions of tropical forest dung beetles under contrasting logging regimes. Biol. Conserv. 2011, 144, 166–174. [Google Scholar] [CrossRef]
- López-Bedoya, P.A.; Bohada-Murillo, M.; Ángel-Vallejo, M.C.; Audino, L.D.; Davis, A.L.V.; Gurr, G.; Noriega, J.A. Primary forest loss and degradation reduces biodiversity and ecosystem functioning: A global meta-analysis using dung beetles as an indicator taxon. J. Appl. Ecol. 2022, 59, 1572–1585. [Google Scholar] [CrossRef]
- Amell-Caez, Y.; Decastro-Arrazola, I.; Garcia, I. Spatial diversity of dung beetle assemblages (Coleoptera: Scarabaeidae: Scarabaeinae) in five ecoregions from Sucre, Colombian Caribbean coast. Rev. Colomb. Entomol. 2019, 45, 1–10. [Google Scholar]
- Noriega, J.A.; Solis, C.; García, H.; Murillo-Ramos, L.; Renjifo, J.; Olarte, J. Sinopsis de los escarabajos coprófagos (Coleoptera: Scarabaeinae) del caribe colombiano. Caldasia 2013, 35, 465–477. [Google Scholar]
- Taboada-Verona, C.; Sermeño-Correa, C.; Sierra-Serrano, O.; Noriega, J.A. Checklist of the superfamily Scarabaeoidea (Insecta, Coleoptera) in an urban area of the Caribbean Colombia. Check List 2019, 15, 579–594. [Google Scholar] [CrossRef]
- Bohórquez, J.; Montoya, J. Abundancia y preferencia trófica de Dichotomius belus (Coleoptera: Scarabaeidae) en la reserva forestal de Colosó, Sucre. Bol. Mus. Entomol. Univ. Val. 2009, 10, 1–7. [Google Scholar]
- Navarro, L.; Roman, K.; Gomez, H.; Pérez, A. Primer registro de Digitonthophagus gazella (Fabricius, 1787) para el departamento de Sucre, Colombia. Rev. Colomb. Cienc. Anim.-RECIA 2009, 1, 60–64. [Google Scholar] [CrossRef]
- Solis, C.; Noriega, J.A.; Herrera, G. Escarabajos coprófagos (Coleoptera: Scarabaeinae) en tres bosques seco del departamento del Atlántico-Colombia. Bol. Mus. Entomol. Univ. Val. 2011, 12, 33–41. [Google Scholar]
- Rangel-Acosta, J.; Martínez-Hernandez, N. Comparación de los ensamblajes de escarabajos copronecrófagos (Scarabaeidae: Scarabaeinae) entre fragmentos de bosque seco tropical y la matriz adyacente en el departamento del Atlántico-Colombia. Rev. Mex. Biodivers. 2017, 88, 389–401. [Google Scholar] [CrossRef]
- Rangel-Acosta, J.; Solano-Torres, J.; Martínez-Hernández, N. Variación temporal y vertical de los escarabajos coprófagos (Scarabaeidae: Scarabaeinae) en dos fragmentos de bosque seco tropical en el departamento del Atlántico-Colombia. Bol. Cient. Mus. Hist. Nat. 2018, 22, 179–198. [Google Scholar] [CrossRef]
- Ortega-Echeverría, C.; Navas, G.; Noriega, J. Estacionalidad del ensamblaje de escarabajos coprófagos (Coleoptera: Scarabaeinae) del jardín botánico de Cartagena “Guillermo Piñeres” Bolívar-Colombia. Caldasia 2019, 41, 124–138. [Google Scholar] [CrossRef]
- Rangel-Acosta, J.; Martínez-Hernández, N.; Yonoff-Zapat. Respuesta de los escarabajos coprófagos (Scarabaeidae: Scarabaeinae) a la modificación del hábitat causada por un incendio forestal en la Reserva Bijibana, Atlántico-Colombia. Rev. Mex. Biodivers. Rev. Mex. Biodivers. 2020, 91, e912879. [Google Scholar] [CrossRef]
- Noriega, J.A.; Zapata-Prisco, C.; García, H.; Hernández, E.; Hernández, J.; Martínez, R.; Santos-Santos, J.; Pablo-Cea, J.; Calatayud, J. Does ecotourism impact biodiversity? An assessment using dung beetles (Coleoptera: Scarabaeinae) as bioindicators in a tropical dry forest natural park. Ecol. Indic. 2020, 117, 106580. [Google Scholar] [CrossRef]
- Tscharntke, T.; Tylianakis, J.M.; Rand, T.A.; Didham, R.K.; Fahrig, L.; Batáry, P.; Bengtsson, J.; Clough, Y.; Crist, T.O.; Dormann, C.F.; et al. Landscape moderation of biodiversity patterns and processes—Eight hypotheses. Biol. Rev. 2012, 87, 661–685. [Google Scholar] [CrossRef] [PubMed]
- Gascon, C.; E Lovejoy, T.; Bierregaard, R.O., Jr.; Malcolm, J.R.; Stouffer, P.C.; Vasconcelos, H.L.; Laurance, W.F.; Zimmerman, B.; Tocher, M.; Borges, S. Matrix habitat and species richness in tropical forest remnants. Biol. Conserv. 1999, 91, 223–229. [Google Scholar] [CrossRef]
- Mendenhall, C.D.; Sekercioglu, C.H.; Brenes, F.O.; Ehrlich, P.R.; Daily, G.C. Predictive model for sustaining biodiversity in tropical countryside. Proc. Natl. Acad. Sci. USA 2011, 108, 16313–16316. [Google Scholar] [CrossRef] [PubMed]
- Nichols, E.; Larsen, T.; Spector, S.; Davis, A.; Escobar, F.; Favila, M.; Vulinec, K.; The Scarabaeinae Research Network. Global dung beetle response to tropical forest modification and fragmentation: A quantitative literature review and meta-analysis. Biol. Conserv. 2007, 137, 1–19. [Google Scholar] [CrossRef]
- Edwards, F.A.; Finan, J.; Graham, L.K.; Larsen, T.H.; Wilcove, D.S.; Hsu, W.W.; Chey, V.; Hamer, K.C. The impact of logging roads on dung beetle assemblages in a tropical rainforest reserve. Biol. Conserv. 2017, 205, 85–92. [Google Scholar] [CrossRef]
- Martínez-Hernández, N.; Atencia, S.; Cerpa, M.; Murillo, T.; Mejía, C. Composición y estructura de la fauna de escarabajos (Insecta: Coleoptera) atraídos por trampas de luz en la reserva ecológica de Luriza, Atlántico, Colombia. Bol. Soc. Entomol. Aragonesa (S.E.A.) 2010, 47, 373–381. [Google Scholar]
- Delgado-Gómez, P.; Lopera, A.; Rangel Ch, J.O. Variación Espacial del Ensamblaje de Escarabajos Coprófagos (Scarabaeidae: Scarabaeinae) en Remanentes de Bosque Seco en Chimichagua (Cesar, Colombia); Colombia diversidad biótica XII: La región Caribe de Colombia; En Rangel, J.O., Ed.; Universidad Nacional de Colombia: Bogotá, Colombia, 2012; pp. 833–849. [Google Scholar]
- Clavijo-Bustos, J.; Lopera-Toro, A.; Noriega, J.A. On the presence of Ataenius crenulatus Schmidt, 1910 in Colombia. Coleopt. Bull. 2023, in press. [Google Scholar]
- Noriega, J.A.; Solis, C.; Escobar, F.; Realpe, E. Escarabajos coprófagos (Coleoptera: Scarabaeidae) de la provincia de la Sierra Nevada de Santa Marta. Biota Colomb. 2007, 8, 77–86. [Google Scholar]
- Janzen, D.H. Seasonal change in abundance of large nocturnal dung beetles (Scarabaeidae) in a Costa Rican deciduous forest and adjacent horse pasture. Oikos 1983, 41, 274–283. [Google Scholar] [CrossRef]
- Breytenbach, W.; Breytenbach, G. Seasonal patterns in dung feeding Scarabaeidae in the Southern Cape. J. Entomol. Soc. South. Afr. 1986, 49, 359–366. [Google Scholar]
- Rougon, D.; Rougon, C. Dung beetles of the Sahel Region: 230–254. In Dung Beetle Ecology; Hanski, I.Y., Cambefort, Eds.; Princeton University: Princeton, NJ, USA, 1991. [Google Scholar]
- Escobar, F. Estudio de la comunidad de coleópteros coprófagos (Scarabaeidae) en un remante de bosque seco al norte del Tolima, Colombia. Caldasia 1997, 19, 419–430. [Google Scholar]
- Otavo, S.E.; Parrado-Rosselli, A.; Noriega, J.A. Superfamilia Scarabaeoidea (Insecta: Coleoptera) como elemento bio-indicador de perturbación antropogénica en un Parque Nacional amazónico. Rev. Biol. Trop. 2013, 61, 735–752. [Google Scholar] [CrossRef] [PubMed]
- Casas, C.; Pineda, N.; Monro, D.; Realpe, E.; Noriega, J. Variación estacional de la biomasa de un ensamble de escarabajos coprófagos (Coleoptera: Scarabaeinae) en un pastizal altoandino. Acta Biol. Colomb. 2021, 26, 318–326. [Google Scholar] [CrossRef]
- Andresen, E.; Feer, F. The role of dung beetles as secondary seed dispersers and their effect on plant regeneration in tropical rainforests. In Seed Fate: Predation, Dispersal and Seedling Establishment; Forget, P.-M., Lambert, J., Hulme, P., Wall, S.V., Eds.; CABI: Wallingford, UK, 2005; pp. 431–449. [Google Scholar]
- Horgan, F.G. Dung beetle assemblages in forests and pastures of El Salvador: A functional comparison. Biodivers. Conserv. 2008, 17, 2961–2978. [Google Scholar] [CrossRef]
- Escobar, F.; Chacón, P. Distribución espacial y temporal en un gradiente de sucesión de la fauna de coleopteros coprófagos (Scarabaeinae, Aphodiinae) en un bosque tropical montano, Nariño—Colombia. Rev. Biol. Trop. 2000, 48, 961–975. [Google Scholar]
- Halffter, G.; Montes de Oca, T. Daily and seasonal activities of a guild of the coprofagus, burrowing beetle (Coleoptera Scarabainae) in tropical grassland. Trop. Zool. 1995, 8, 159–180. [Google Scholar]
- Anduaga, S.; Huerta, C. Importance of dung incorporation activity by three species of coprophaneus beetles (Coleoptera: Scarabaeidae: Scarabaeinae) macrofauna in pastureland on “La Michilía” Biosphere Reserve in Durango, México. Environ. Entomol. 2007, 36, 555–559. [Google Scholar]
- Edwards, P. Seasonal variation in the dung of African mammals and consequences for coprophagous insects. Funct. Ecol. 1991, 5, 617–628. [Google Scholar] [CrossRef]
- Martínez, I.M.; Dellacasa, M.; Lumaret, J.-P.; Dellacasa, G. Phenology and reproductive cycles in Mexican aphodiine dung beetles (Coleoptera: Scarabaeidae: Aphodiinae: Aphodiini). Ann. Soc. Entomol. Fr. 2022, 58, 173–185. [Google Scholar] [CrossRef]
- Chown, S.; Klok, C. The ecological implications of physiological diversity in dung beetles. In Ecology and Evolution of Dung Beetles; Simmons, L.W., Rids-dill-Smith, T.J., Eds.; Blackwell Publishing Ltd.: Hoboken, NJ, USA, 2011; pp. 200–219. [Google Scholar]
- Martin-Piera, F.; Lobo, J.M. A comparative discussion of trophic preferences in dung beetle communities. Misc. Zool. 1996, 19, 13–31. [Google Scholar]
- Noriega, J.A.; Floate, K.D.; Génier, F.; Reid, C.A.M.; Kohlmann, B.; Horgan, F.G.; Davis, A.L.V.; Forgie, S.A.; Aguilar, C.; Ibarra, M.G.; et al. Global distribution patterns provide evidence of niche shift by the introduced African dung beetle Digitonthophagus gazella. Entomol. Exp. Appl. 2020, 168, 766–782. [Google Scholar] [CrossRef]
- Krebs, C.J. Ecology: The Experimental Analysis of Distribution and Abundances, 6th ed.; Pearson: Essex, UK, 2014; 653p. [Google Scholar]
- Martínez, N.; García, H.; Pulido, L.; Ospino, D.; Narváez, J. Escarabajos coprófagos (Coleoptera: Scarabaeinae) de la vertiente noroccidental, Sierra Nevada de Santa Marta, Colombia. Neotrop. Entomol. 2009, 38, 708–715. [Google Scholar] [CrossRef] [PubMed]
- Raine, E.H.; Slade, E.M. Dung beetle mammal associations: Methods, research trends and future directions. Proc. R. Soc. B 2019, 286, 20182002. [Google Scholar] [CrossRef] [PubMed]
- Amézquita, S.; Favila, M.E. Removal rates of native and exotic dung by dung beetles (Scarabaeidae: Scarabaeinae) in a fragmented Tropical Rain Forest. Environ. Èntomol. 2010, 39, 328–336. [Google Scholar] [CrossRef] [PubMed]
- Slade, E.M.; Kirwan, L.; Bell, T.; Philipson, C.D.; Lewis, O.T.; Roslin, T. The importance of species identity and interactions for multifunctionality depends on how ecosystem functions are valued. Ecology 2017, 98, 2626–2639. [Google Scholar] [CrossRef]
- Tonelli, M.; Verdú, J.R.; Zunino, M. Effects of the progressive abandonment of grazing on dung beetle biodiversity: Body size matters. Biodivers. Conserv. 2017, 27, 189–204. [Google Scholar] [CrossRef]
- Barraza, J.; Montes, J.; Martínez, N.; Deloya, C. Ensamblaje de escarabajos coprófagos (Scarabaeidae: Scarabaeinae) del Bosque Tropical Seco, Bahía Concha, Santa Marta (Colombia). Rev. Colomb. Entomol. 2010, 36, 285–291. [Google Scholar] [CrossRef]
- Escobar, F. Excremento, Coprófago y Deforestación en Bosque de Montaña al Suroriente de Colombia. Bachelor’s Thesis, Universidad del Valle, Cali, Colombia, 1994. [Google Scholar]
- Amat-Garcia, G.; Lopera, A.; Amézquita, S. Patrones de distribución de escarabajos coprófagos (Coleoptera: Scarabaeidae) en relictos del bosque altoandino, cordillera oriental de Colombia. Caldasia 1997, 19, 191–204. [Google Scholar]
- González-Tokman, D.; Martínez, M.I.; Villalobos-Ávalos, Y.; Munguía-Steyer, R.; Ortiz-Zayas, M.D.R.; Cruz-Rosales, M.; Lumaret, J.-P. Ivermectin alters reproductive success, body condition and sexual trait expression in dung beetles. Chemosphere 2017, 178, 129–135. [Google Scholar] [CrossRef]
- Verdú, J.R.; Cortez, V.; Ortiz, A.J.; Lumaret, J.-P.; Lobo, J.M.; Sánchez-Piñero, F. Biomagnification and body distribution of ivermectin in dung beetles. Sci. Rep. 2020, 10, 9073. [Google Scholar] [CrossRef] [PubMed]
- Errouissi, F.; Alvinerie, M.; Galtier, P.; Kerboeuf, D.; Lumaret, J.-P. The negative effects of the residues of ivermectin in cattle dung using a sustained-release bolus on Aphodius constans (Duft.) (Coleoptera: Aphodiidae). Vet. Res. 2001, 32, 421–427. [Google Scholar] [CrossRef] [PubMed]
- Dadour, I.R.; Cook, D.F.; Hennessy, D. Reproduction and survival of the dung beetle Onthophagus binodis (Coleoptera: Scarabaeidae) exposed to Abamectin and Doramectin residues in cattle dung. Environ. Èntomol. 2000, 29, 1116–1122. [Google Scholar] [CrossRef]
- Martínez, I.M.; Lumaret, J.-P.; Cruz, M.R. Suspected side effects of a herbicide on dung beetle populations (Coleoptera: Scarabaeidae). C.R. L’académie Sci. Paris Life Sci. 2001, 324, 989–994. [Google Scholar] [CrossRef] [PubMed]
- Tonelli, M.; Verdú, J.R.; Morelli, F.; Zunino, M. Dung beetles: Functional identity, not functional diversity, accounts for ecological process disruption caused by the use of veterinary medical products. J. Insect Conserv. 2020, 24, 643–654. [Google Scholar] [CrossRef]
- Errouissi, F.; Lumaret, J.-P. Field effects of faecal residues from ivermectin slow-release boluses on the attractiveness of cattle dung to dung beetles. Med Vet.-Èntomol. 2010, 24, 433–440. [Google Scholar] [CrossRef] [PubMed]
- Römbke, J.; Coors, A.; Alonso Fernández, A.; Förster, B.; Fernández, C.; Jensen, J.; Lumaret, J.P.; Porcel Cots, M.A.; Liebig, M. Effects of the parasiticide ivermectin on the structure and function of dung and soil invertebrate communities in the field (Madrid, Spain). Appl. Soil Ecol. 2010, 45, 284–292. [Google Scholar] [CrossRef]
- Bernal, J.L.; Del Nozal, M.J.; Salas, M.; Galante, E.; Lumaret, J.P. Determination of residual Ivermectin in cattle dung following subcutaneous injection. J. Liq. Chromatogr. 1994, 17, 2429–2444. [Google Scholar] [CrossRef]
- Nervo, B.; Tocco, C.; Caprio, E.; Palestrini, C.; Rolando, A. The effects of body mass on dung removal efficiency in dung beetles. PLoS ONE 2014, 9, e107699. [Google Scholar] [CrossRef]
- Laliberté, E.; Wells, J.A.; DeClerck, F.; Metcalfe, D.J.; Catterall, C.P.; Queiroz, C.; Aubin, I.; Bonser, S.P.; Ding, Y.; Fraterrigo, J.M.; et al. Land-use intensification reduces functional redundancy and response diversity in plant communities. Eco. Lett. 2010, 13, 76–86. [Google Scholar] [CrossRef]
- Anziani, O.; Suarez, V.; Guglielmone, A.; Wanker, O.; Grande, H.; Coles, G. Resistance to benzimidazole and avermectin anthelmintics in cattle nematodes in Argentina. Vet. Parasitol. 2004, 122, 303–306. [Google Scholar] [CrossRef] [PubMed]
- Geurden, T.; Chartier, C.; Fanke, J.; di Regalbono, A.F.; Traversa, D.; von Samson-Himmelstjerna, G.; Demeler, J.; Vanimisetti, H.B.; Bartram, D.J.; Denwood, M.J. Anthelmintic resistance to ivermectin and moxidectin in gastrointestinal nematodes of cattle in Europe. Int. J. Parasitol. Drugs Drug Resist. 2015, 5, 163–171. [Google Scholar] [CrossRef] [PubMed]

| SubFam/ Tribe | Species | FG | Livestock Management | Total (%) | |||||
|---|---|---|---|---|---|---|---|---|---|
| No-IVM | Two-IVM 1% | Two-IVM 3.15% | |||||||
| Ll | S | Ll | S | Ll | S | ||||
| Aphodiinae | |||||||||
| Aphodiini | Nialaphodius nigrita (Fabricius, 1801) | Ep | 55 | 334 | 6 | 1241 | 4 | 177 | 1817 (7.1) |
| Xenoheptaulacus tricostatus (Harold, 1869) | Ep | 2 | 0 | 0 | 0 | 0 | 0 | 2 (0.0) | |
| Eupariini | Ataenius crenulatus Schmidt, 1910 | Ep | 6235 | 160 | 4755 | 43 | 1712 | 15 | 12,920 (50.8) |
| Ataenius complicatus Harold, 1869 | Ep | 57 | 2 | 7 | 0 | 15 | 0 | 81 (0.3) | |
| Cartwrightia cartwrighti Cartwright, 1967 | Ep | 2 | 1 | 1 | 0 | 0 | 0 | 4 (0.0) | |
| Scarabaeinae | |||||||||
| Ateuchini | Ateuchus sp. 1 | Pp | 2 | 0 | 0 | 0 | 1 | 0 | 3 (0.0) |
| Uroxys sp. 1 | Pp | 0 | 3 | 0 | 0 | 0 | 0 | 3 (0.0) | |
| Uroxys sp. 2 | Pp | 1 | 0 | 0 | 1 | 0 | 0 | 2 (0.0) | |
| Coprini | Canthidium sp. 1 | Pp | 0 | 0 | 0 | 0 | 1 | 0 | 1 (0.0) |
| Dichotomius agenor (Harold, 1869) | Pg | 84 | 0 | 62 | 0 | 10 | 0 | 156 (0.6) | |
| Dichotomius coenosus (Erichson, 1848) | Pg | 2 | 0 | 0 | 0 | 0 | 0 | 2 (0.0) | |
| Deltochilini | Canthon juvencus Harold, 1868 | Tp | 0 | 0 | 0 | 1 | 0 | 0 | 1 (0.0) |
| Canthon lituratus (Germar, 1813) | Tp | 33 | 0 | 2 | 0 | 15 | 0 | 50 (0.2) | |
| Canthon mutabilis Lucas, 1859 | Tp | 43 | 1 | 10 | 0 | 75 | 0 | 129 (0.5) | |
| Canthon septemmaculatus (Latreille, 1812) | Tm | 9 | 0 | 0 | 0 | 64 | 0 | 73 (0.3) | |
| Canthon sp. 1 | Tp | 0 | 0 | 0 | 0 | 1 | 0 | 1 (0.0) | |
| Deltochilum guildingii (Westwood, 1835) | Tg | 1 | 0 | 0 | 0 | 0 | 0 | 1 (0.0) | |
| Pseudocanthon xanthurus (Blanchard, 1846) | Tp | 1 | 0 | 0 | 0 | 0 | 0 | 1 (0.0) | |
| Sylvicanthon aequinoctialis (Harold, 1868) | Tp | 1 | 0 | 0 | 0 | 0 | 0 | 1 (0.0) | |
| Demarziellini | Trichillidium pilosum (Robinson, 1948) | Pp | 1 | 0 | 0 | 0 | 0 | 0 | 1 (0.0) |
| Oniticellini | Eurysternus impressicollis Castelnau, 1840 | Ep | 188 | 12 | 34 | 8 | 48 | 1 | 291 (1.1) |
| Eurysternus mexicanus Harold, 1869 | Em | 67 | 4 | 11 | 2 | 13 | 1 | 98 (0.4) | |
| Onthophagini | Digitonthophagus gazella (Fabricius, 1787) | Pm | 1453 | 54 | 808 | 30 | 502 | 18 | 2865 (11.3) |
| Onthophagus bidentatus Drapiez, 1819 | Pp | 2 | 0 | 0 | 2 | 4 | 0 | 8 (0.0) | |
| Onthophagus buculus Mannerheim, 1829 | Pp | 39 | 0 | 2 | 0 | 12 | 0 | 53 (0.2) | |
| Onthophagus clypeatus Blanchard, 1846 | Pp | 4 | 4 | 1 | 2 | 0 | 0 | 11 (0.0) | |
| Onthophagus marginicollis Harold, 1880 | Pp | 2469 | 6 | 3192 | 0 | 1188 | 0 | 6855 (26.9) | |
| Phanaeini | Coprophanaeus gamezi Arnaud, 2002 | Pg | 1 | 0 | 0 | 0 | 0 | 0 | 1 (0.0) |
| Diabroctis cadmus (Harold, 1868) | Pg | 1 | 0 | 0 | 0 | 8 | 0 | 9 (0.0) | |
| Phanaeus hermes Harold, 1868 | Pm | 0 | 0 | 1 | 0 | 0 | 0 | 1 (0.0) | |
| Abundance | 10,753 | 581 | 8892 | 1330 | 3673 | 212 | 25,441 | ||
| Richness | 25 | 11 | 14 | 9 | 17 | 5 | 30 | ||
| Biomass (g) | 364.32 | 10.14 | 253.26 | 9.63 | 156.4 | 3.24 | 797.22 | ||
| Livestock Management | Richness | Abundance | Biomass (g) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Guild | |||||||||
| P | E | T | P | E | T | P | E | T | |
| No-IVM | 13 | 7 | 6 | 4126 | 7119 | 89 | 331.8 | 37.3 | 5.4 |
| Two-IVM 1% | 8 | 6 | 3 | 4101 | 6108 | 13 | 239.9 | 22.6 | 0.4 |
| Two-IVM 3.15% | 8 | 5 | 4 | 1744 | 1986 | 155 | 136.9 | 9.6 | 13.3 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tovar, H.L.; Correa, C.M.A.; Lumaret, J.-P.; López-Bedoya, P.A.; Navarro, B.; Tovar, V.; Noriega, J.A. Effect of Antiparasitic Management of Cattle on the Diversity and Functional Structure of Dung Beetle (Coleoptera: Scarabaeidae) Assemblages in the Colombian Caribbean. Diversity 2023, 15, 555. https://doi.org/10.3390/d15040555
Tovar HL, Correa CMA, Lumaret J-P, López-Bedoya PA, Navarro B, Tovar V, Noriega JA. Effect of Antiparasitic Management of Cattle on the Diversity and Functional Structure of Dung Beetle (Coleoptera: Scarabaeidae) Assemblages in the Colombian Caribbean. Diversity. 2023; 15(4):555. https://doi.org/10.3390/d15040555
Chicago/Turabian StyleTovar, Hernando L., César M. A. Correa, Jean-Pierre Lumaret, Pablo A. López-Bedoya, Blas Navarro, Valentina Tovar, and Jorge Ari Noriega. 2023. "Effect of Antiparasitic Management of Cattle on the Diversity and Functional Structure of Dung Beetle (Coleoptera: Scarabaeidae) Assemblages in the Colombian Caribbean" Diversity 15, no. 4: 555. https://doi.org/10.3390/d15040555
APA StyleTovar, H. L., Correa, C. M. A., Lumaret, J.-P., López-Bedoya, P. A., Navarro, B., Tovar, V., & Noriega, J. A. (2023). Effect of Antiparasitic Management of Cattle on the Diversity and Functional Structure of Dung Beetle (Coleoptera: Scarabaeidae) Assemblages in the Colombian Caribbean. Diversity, 15(4), 555. https://doi.org/10.3390/d15040555








