4.2. Delimitation of Species Groups in Cardiocondyla on a Worldwide Scale
The morphological attempt to delimit within-genus species groups presented below performs without severe conflicts in the majority of cases but delivers also controversial indications. For instance, the shape of postpetiolar sternite is normally a very useful character. However, in Cardiocondyla nivalis Mann and C. allonivalis n.sp.—closely related species sharing a multitude of characters—the sternite is flat in the former but clearly bicuspidate in the latter. The same problem occurs in C. yemeni Seifert 2003. It shows a bidentate postpetiolar sternite but should belong to the C. minutior group in which the sternite is completely flat in the eight known species. Beginning with the tropical and subtropical groups, ending with those from the temperate zone and also postulating monotypical (single-species) clades, I propose the following grouping.
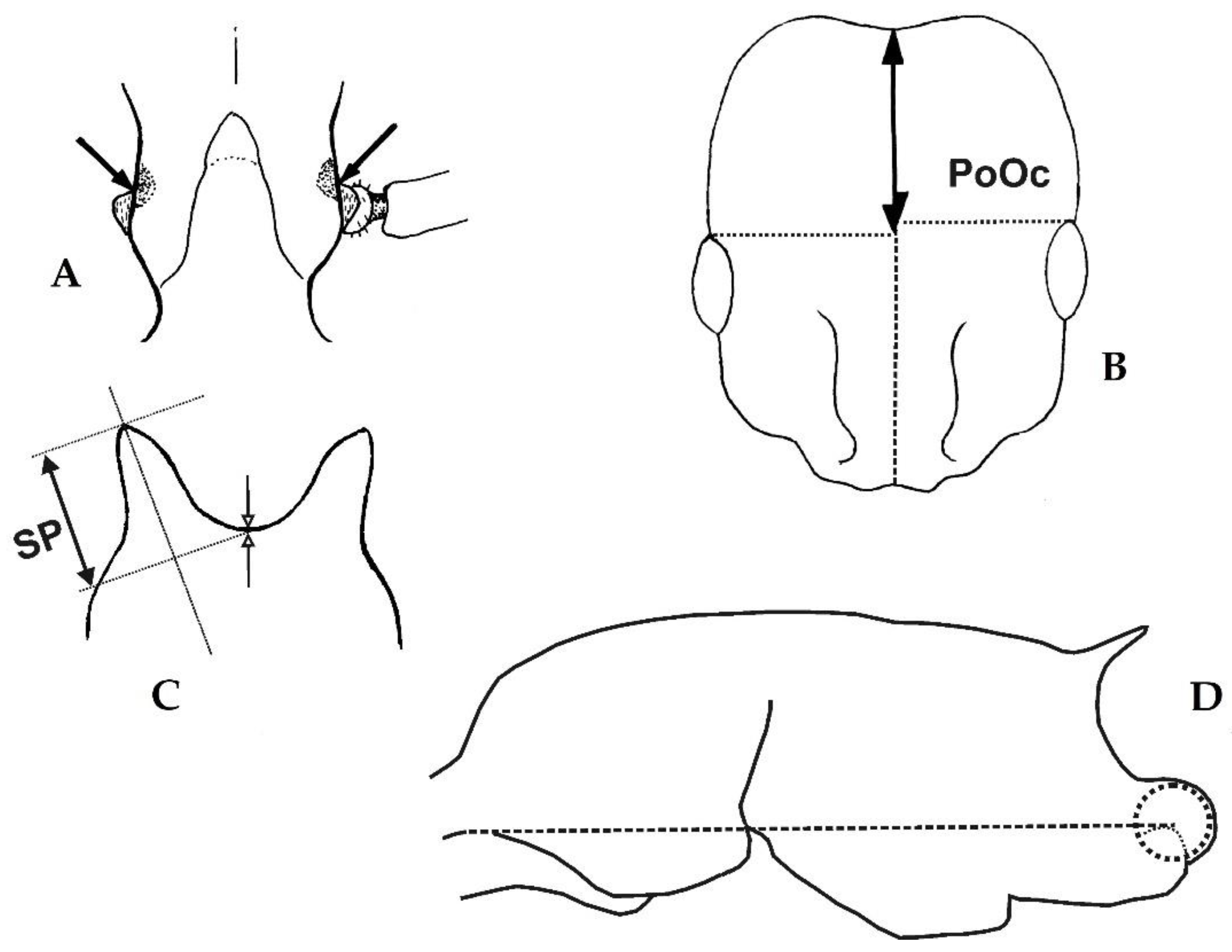
Head elongated with rather high postocular index (CL/CW 1.182–1.263, PoOc/CL 0.463–0.490) and a narrow frons (FRS/CS 0.221–0.246). Metanotal depression weak or absent (MGr/CS 0–1.25%), propodeal spines short (SP/CS 0.089–0.133). Postpetiole with a flat sternite and low (PpH/CS 0.230–0.274). Pubescence on first gaster tergite rather long and dense (PLG/CS 6.6–8.3%, sqPDG 2.8–4.0). Eight species with distributional center in the Orientalic and Australasian region of which two are worldwide tramp species.
C. minutior Forel, 1899,
C. goa Seifert, 2003,
C. tjibodana Karavajev, 1935,
C. schulzi n.sp.,
C. carbonaria Forel, 1907,
C. opaca Seifert, 2003,
C. britteni Crawley, 1920,
C. parvinoda Forel, 1902. Analyzed in this paper on species level.
Figure 2,
Figure 3,
Figure 4,
Figure 5,
Figure 6,
Figure 7,
Figure 8 and
Figure 9;
Table 1.
Head elongated with rather high postocular index (CL/CW 1.227–1.246, PoOc/CL 0.454–0.472) and a very narrow frons (FRS/CS 0.200–0.227). Metanotal depression well developed (MGr/CS 2.33–3.22%), Propodeal spines relatively short (SP/CS 0.128–0.207). Postpetiole rather high (PpH/CS 0.292–0.335) and with a bulging sternite that does not show any lobes, cusps or dents. Pubescence on first gaster tergite rather long and dense (PLG/CS 6.7–7.6%, sqPDG 3.8–4.3). At least two species with probably Afrotropical origin, one of these with worldwide tramp species properties. Described species: C. emeryi Forel, 1881, C. neferka Bolton, 1982. Not analyzed in this paper on species level.
Head rather short with comparably low postocular index (CL/CW 1.099–1.189, PoOc/CL 0.415–0.440) and a variably wide frons (FRS/CS 0.219–0.282). Scape short to very short (SL/CS 0.682–0.825). Metanotal depression well developed (MGr/CS 3.25–3.65%). Propodeal spines rather short (SP/CS 0.184–0.201). Postpetiole higher (PpH/CS 0.276–0.337) with a bilobate or bicuspidate sternite. Pubescence on first gaster tergite variable. Five described and two undescribed species with origin in the tropics of the Old World, two of these occur as worldwide tramp species. Described species: C. wroughtonii (Forel, 1890), C. obscurior Wheeler, 1929, C. yemeni Collingwood & Agosti, 1996, C. nana Seifert, 2003, C. shagrinata Seifert, 2003. Undescribed species: Cardiocondyla sp. code HEIN, Cardiocondyla sp. code AFRI. Not analyzed in this paper on species level.
Dorsal mesosomal profile evenly convex with absent or only suggested metanotal depression (MGr/CS 0–0.52%) and in dorsal view without pronotal corners. Propodeal spines well-developed (SP/CS 0.208–0.400). Postpetiolar sternite conspicuously bilobate or bicuspidate. Furthermore characterized by short head with rather low postocular index (CL/CW 1.051–1.133, PoOc/CL 0.408–0.436) and a wide frons (FRS/CS 0.242–0.337). The clearest differences to the
C. thoracica and
C. wheeleri group are the conspicuously bilobate or bicuspidate postpetiolar sternite, the evenly convex dorsal mesosomal profile and the absence of any pronotal corners. Six species in the East Orientalic and Australasian zone, no tramp species known:
C. argentea n.sp.,
C. semiargentea n.sp.,
C. argyrotricha n.sp.,
C. latifrons n.sp.,
C. micropila n.sp.,
C. pirata Seifert et al., 2013. Analyzed in this paper on species level.
Figure 10,
Figure 11,
Figure 12,
Figure 13,
Figure 14 and
Figure 15;
Table 2.
Very small (CS 357–402 µm). With exception of the dark brown to jet black gaster, whole body whitish or whitish-yellowish. Metanotal groove notable (MGr/CS 1.6–2.6%) and overall dorsal mesosomal profile more straight. Frons wider than in the former three species groups (FRS/CS 0.290–0.314). Head and scape rather short (CL/CW 1.095–1.149, SL/CS 0.787–0.857). Two species of Australasian-Polynesian origin:
C. nivalis Mann, 1919,
C. allonivalis n.sp. Analyzed in this paper on species level.
Figure 16 and
Figure 17;
Table 3.
Pronotal corners in dorsal view developed but blunt. Dorsal mesosomal profile not evenly convex with a small metanotal depression (MGr/CS 1.4–3.0%). Postpetiolar sternite flat or shallowly convex and without any prominent structures. Spine base very broad and spines rather long (SPBA/CS 0.341–0.374, SP/CS 0.296–0.418). Four Australasian species, all occurring east of the Wallace Line, no tramp species known:
C. wheeleri Viehmeyer, 1914,
C. excavata n.sp.,
C. goroka n.sp.,
C. nigrocerea Karavajev, 1935. Analyzed in this paper on species level.
Figure 18,
Figure 19,
Figure 20 and
Figure 21;
Table 4.
Pronotal corners in dorsal and lateral view very prominent and sharp. Dorsal mesosomal profile not evenly convex and with an absent to well-developed metanotal depression (MGr/CS 0–7.6%). Postpetiolar sternite differently shaped but not distinctly bilobate or bicuspidate, without any prominent structures. Spine base narrower and spines shorter than in the
C. wheeleri group (SPBA/CS 0.237–0.368, SP/CS 0.230–0.365). Three Australasian species, all occurring east of the Wallace Line, no tramp species known:
C. thoracica (Smith, 1859),
C. paradoxa Emery, 1897,
C. subspina n.sp. Analyzed in this paper on species level.
Figure 22,
Figure 23 and
Figure 24;
Table 4.
Table 1.
Measurements of workers of the Cardiocondyla minutior group. Data are given in the sequence arithmetic mean ± standard deviation [minimum, maximum]. For characters with fewer data than indicated in the head of the columns, the number of measurements is given after the square brackets.
Table 1.
Measurements of workers of the Cardiocondyla minutior group. Data are given in the sequence arithmetic mean ± standard deviation [minimum, maximum]. For characters with fewer data than indicated in the head of the columns, the number of measurements is given after the square brackets.
| | minutior
(n = 92) | goa
(n = 57) | tjibodana
(n = 65) | schulzi n.sp.
(n = 4) | opaca
(n = 1) | carbonaria
(n = 1) | britteni
(n = 1) | parvinoda
(n = 17) |
|---|
| CS [µm] | 422 ± 14
[379,450] | 413 ± 18
[380,466] | 406 ± 17
[379,448] | 432 ± 10
[418,441] | 424 | 434 | 402 | 501 ± 21
[452,534] |
| CL/CW | 1.263 ± 0.024
[1.211,1.324] | 1.239 ± 0.033
[1.180,1.319] | 1.258 ± 0.019
[1.220,1.312] | 1.187 ± 0.015
[1.167,1.199] | 1.253 | 1.282 | 1.249 | 1.182 ± 0.019
[1.146,1.221] |
| SL/CS | 0.754 ± 0.011
[0.706,0.776] | 0.760 ± 0.015
[0.724,0.790] | 0.761 ± 0.012
[0.730,0.791] | 0.780 ± 0.002
[0.777,0.782] | 0.836 | 0.789 | 0.741 | 0.831 ± 0.011
[0.808,0.852] |
| PoOc/CL | 0.475 ± 0.007
[0.463,0.495] | 0.480 ± 0.010
[0.464,0.502] | 0.480 ± 0.008
[0.458,0.495] | 0.463 ± 0.005
[0.458,0.469] | 0.468 | 0.474 | 0.482 | 0.468 ± 0.009
[0.455,0.488] |
| EYE | 0.232 ± 0.006
[0.215,0.245] | 0.230 ± 0.006
[0.217,0.244] | 0.238 ± 0.005
[0.227,0.249] | 0.228 ± 0.010
[0.220,0.242] | 0.235 | 0.243 | 0.226 | 0.219 ± 0.007
[0.207,0.238] |
| dFOV | 14.6 ± 1.2
[12.5,18.0] | 14.8 ± 1.0
[12.7,16.9] | 14.5 ± 0.95
[12.7,17.2] | 16.4 ± 0.54
[15.7,17.0] | 12.6 | 15.0 | 7.0 | 18.4 ± 1.0
[17.0,20.1] |
| FRS/CS | 0.246 ± 0.006
[0.225,0.261] | 0.238 ± 0.008
[0.215,0.254] | 0.239 ± 0.006
[0.226,0.259] | 0.237 ± 0.006
[0.232,0.243] | 0.221 | 0.233 | 0.242 | 0.241 ± 0.004
[0.234,0.250] |
| FL/FR | 1.062 ± 0.027
[1.003,1.174] 44 | 1.088 ± 0.031
[1.011,1.157] 33 | 1.072 ± 0.021
[1.031,1.111] 41 | 1.113 ± 0.022
[1.086,1.139] | 1.033 | 1.010
(photo) | 1.057
(photo) | 1.122 ± 0.022
[1.085,1.150] 9 |
| SPBA/CS | 0.288 ± 0.009
[0.262,0.318] | 0.281 ± 0.013
[0.244,0.314] | 0.284 ± 0.011
[0.247,0.303] | 0.262 ± 0.006
[0.253,0.265] | 0.254 | 0.306 | 0.283 | 0.263 ± 0.010
[0.243,0.282] |
| SP/CS | 0.133 ± 0.008
[0.114,0.156] | 0.121 ± 0.009
[0.096,0.139] | 0.132 ± 0.009
[0.113,0.152] | 0.114 ± 0.009
[0.104,0.123] | 0.092 | 0.136 | 0.089 | 0.124 ± 0.008
[0.104,0.135] |
| PeW/CS | 0.283 ± 0.011
[0.264,0.325] | 0.274 ± 0.013
[0.252,0.323] | 0.266 ± 0.012
[0.243,0.309] | 0.247 ± 0.004
[0.242,0.250] | 0.245 | 0.297 | 0.268 | 0.256 ± 0.011
[0.241,0.279] |
| PpW/CS | 0.475 ± 0.014
[0.446,0.509] | 0.473 ± 0.013
[0.445,0.510] | 0.445 ± 0.011
[0.423,0.474] | 0.425 ± 0.002
[0.423,0.427] | 0.409 | 0.482 | 0.473 | 0.402 ± 0.010
[0.384,0.421] |
| PeH/CS | 0.332 ± 0.012
[0.308,0.368] | 0.336 ± 0.011
[0.315,0.364] | 0.337 ± 0.010
[0.318,0.365] | 0.278 ± 0.006
[0.273,0.284] | 0.283 | 0.348 | 0.333 | 0.305 ± 0.007
[0.390,0.317] |
| PpH/CS | 0.274 ± 0.011
[0.223,0.294] | 0.263 ± 0.008
[0.244,0.284] | 0.261 ± 0.008
[0.243,0.282] | 0.240 ± 0.002
[0.238,0.242] | 0.251 | 0.265 | 0.272 | 0.230 ± 0.007
[0.220,0.243] |
| sqPDG | 3.23 ± 0.20
[2.82,3.71] | 3.38 ± 0.22
[2.99,3.90] | 3.17 ± 0.23
[2.73,3.76] | 3.76 ± 0.22
[3.49,4.01] | 3.31 | 3.01 | 4.25 | 3.62 ± 0.25
[3.23,4.07] |
PLG/CS
[%] | 7.90 ± 0.43
[6.99,9.15] | 7.13 ± 0.50
[6.19,8.43] | 7.61 ± 0.41
[6.70,8.54] | 7.36 ± 0.07
[7.28,7.44] | 7.47 | 8.30 | 6.61 | 8.08 ± 0.67
[6.56,9.52] |
MGr/CS
[%] | 0.39 ± 0.45
[0.0,1.9] | 0.55 ± 0.43
[0.0,1.4] | 0.61 ± 0.44
[0.0,1.8] | 0.11 ± 0.15
[0.0,0.3] | 0.00 | 0.90 | 0.00 | 1.25 ± 0.59
[0.4,2.9] |
Table 2.
Measurements of workers of the
Cardiocondyla argentea group. Explanation of data arrangement as in
Table 1.
Table 2.
Measurements of workers of the
Cardiocondyla argentea group. Explanation of data arrangement as in
Table 1.
| | argentea
n.sp. (n = 11) | semiargentea
n.sp. (n = 13) | argyrotricha
n.sp. (n = 23) | latifrons
n.sp. (n = 61) | micropila
n.sp. (n = 70) | pirata
(n = 6) |
|---|
| CS [µm] | 491 ± 24
[464,531] | 439 ± 16
[413,468] | 512 ± 21
[476,556] | 410 ± 12
[382,436] | 395 ± 18
[358,457] | 397 ± 4
[392,402] |
| CL/CW | 1.051 ± 0.008
[1.038,1.065] | 1.131 ± 0.019
[1.102,1.170] | 1.133 ± 0.023
[1.094,1.182] | 1.102 ± 0.017
[1.060,1.145] | 1.120 ± 0.022
[1.061,1.163] | 1.132 ± 0.010
[1.120,1.147] |
| SL/CS | 0.826 ± 0.017
[0.802,0.853] | 0.830 ± 0.016
[0.794,0.854] | 0.859 ± 0.018
[0.823,0.892] | 0.822 ± 0.009
[0.796,0.847] | 0.826 ± 0.014
[0.785,0.858] | 0.807 ± 0.005
[0.800,0.816] |
| PoOc/CL | 0.420 ± 0.007
[0.409,0.432] | 0.436 ± 0.009
[0.420,0.453] | 0.408 ± 0.007
[0.389,0.419] | 0.414 ± 0.007
[0.398,0.429] | 0.423 ± 0.010
[0.401,0.443] | 0.408 ± 0.005
[0.404,0.416] |
| EYE | 0.211 ± 0.006
[0.204,0.218] | 0.225 ± 0.008
[0.214,0.239] | 0.216 ± 0.004
[0.205,0.223] | 0.230 ± 0.004
[0.222,0.238] | 0.232 ± 0.008
[0.211,0.247] | 0.226 ± 0.002
[0.223,0.229] |
| dFOV | 14.3 ± 2.3
[11,17.8] | 13.9 ± 1.2
[10.9,15.4] | 14.5 ± 1.1
[13.1,16.7] | 17.0 ± 1.3
[14.3,20.0] | 17.4 ± 1.1
[15.1,19.4] | 8.6 ± 0.3
[8.2,9.0] |
| FRS/CS | 0.311 ± 0.005
[0.304,0.319] | 0.274 ± 0.007
[0.261,0.285] | 0.302 ± 0.007
[0.292,0.319] | 0.337 ± 0.007
[0.317,0.352] | 0.331 ± 0.011
[0.309,0.357] | 0.242 ± 0.006
[0.233,0.250] |
| FL/FR | 1.045 ± 0.013
[1.025,1.060] 5 | 1.046 ± 0.023
[1.002,1.072] 9 | 1.028 ± 0.017
[1.002,1.058] 19 | 1.009 ± 0.011
[1.000,1.036] 19 | 1.007 ± 0.013
[1.000,1.042] 19 | 1.101 ± 0.037
[1.034,1.139] |
| SPBA/CS | 0.392 ± 0.011
[0.376,0.414] | 0.362 ± 0.013
[0.342,0.386] | 0.369 ± 0.019
[0.333,0.403] | 0.357 ± 0.016
[0.321,0.390] | 0.351 ± 0.020
[0.303,0.388] | 0.373 ± 0.005
[0.364,0.378] |
| SP/CS | 0.258 ± 0.014
[0.236,0.273] | 0.273 ± 0.017
[0.242,0.300] | 0.267 ± 0.016
[0.239,0.303] | 0.400 ± 0.021
[0.343,0.448] | 0.397 ± 0.034
[0.294,0.476] | 0.208 ± 0.004
[0.204,0.214] |
| PeW/CS | 0.300 ± 0.008
[0.285,0.313] | 0.290 ± 0.012
[0.267,0.301] | 0.303 ± 0.011
[0.286,0.325] | 0.319 ± 0.010
[0.291,0.349] | 0.310 ± 0.025
[0.269,0.362] | 0.359 ± 0.004
[0.355,0.367] |
| PpW/CS | 0.413 ± 0.011
[0.394,0.428] | 0.422 ± 0.015
[0.399,0.449] | 0.423 ± 0.014
[0.397,0.449] | 0.460 ± 0.012
[0.428,0.490] | 0.482 ± 0.017
[0.446,0.531] | 0.468 ± 0.004
[0.462,0.475] |
| PeH/CS | 0.335 ± 0.008
[0.326,0.354] | 0.332 ± 0.007
[0.321,0.346] | 0.336 ± 0.010
[0.315,0.353] | 0.363 ± 0.010
[0.341,0.391] | 0.362 ± 0.017
[0.332,0.422] | 0.343 ± 0.006
[0.335,0.354] |
| PpH/CS | 0.254 ± 0.009
[0.234,0.261] | 0.258 ± 0.010
[0.245,0.277] | 0.252 ± 0.009
[0.231,0.273] | 0.301 ± 0.012
[0.253,0.327] | 0.326 ± 0.015
[0.290,0.359] | 0.320 ± 0.008
[0.308,0.328] |
| sqPDG | 4.12 ± 0.43
[3.42,4.90] | 4.10 ± 0.15
[3.85,4.29] | 3.99 ± 0.20
[3.63,4.30] | 5.17 ± 0.41
[4.46,6.03] | 6.28 ± 0.72
[5.08,8.25] | 3.92 ± 0.13
[3.74,4.12] |
PLG/CS
[%] | 6.08 ± 0.51
[5.21,6.75] | 5.87 ± 0.41
[4.97,6.71] | 6.05 ± 0.31
[5.40,6.56] | 4.56 ± 0.42
[3.84,5.87] | 3.18 ± 0.52
[2.14,4.20] | 7.21 ± 0.26
[6.87,7.54] |
MGr/CS
[%] | 0.04 ± 0.12
[0.0,0.4] | 0.00 ± 0.00
[0.0,0.0] | 0.43 ± 0.40
[0.0,1.4] | 0.31 ± 0.25
[0.0,1.0] | 0.52 ± 0.40
[0.0,1.5] | 0.00 ± 0.00
[0.0,0.0] |
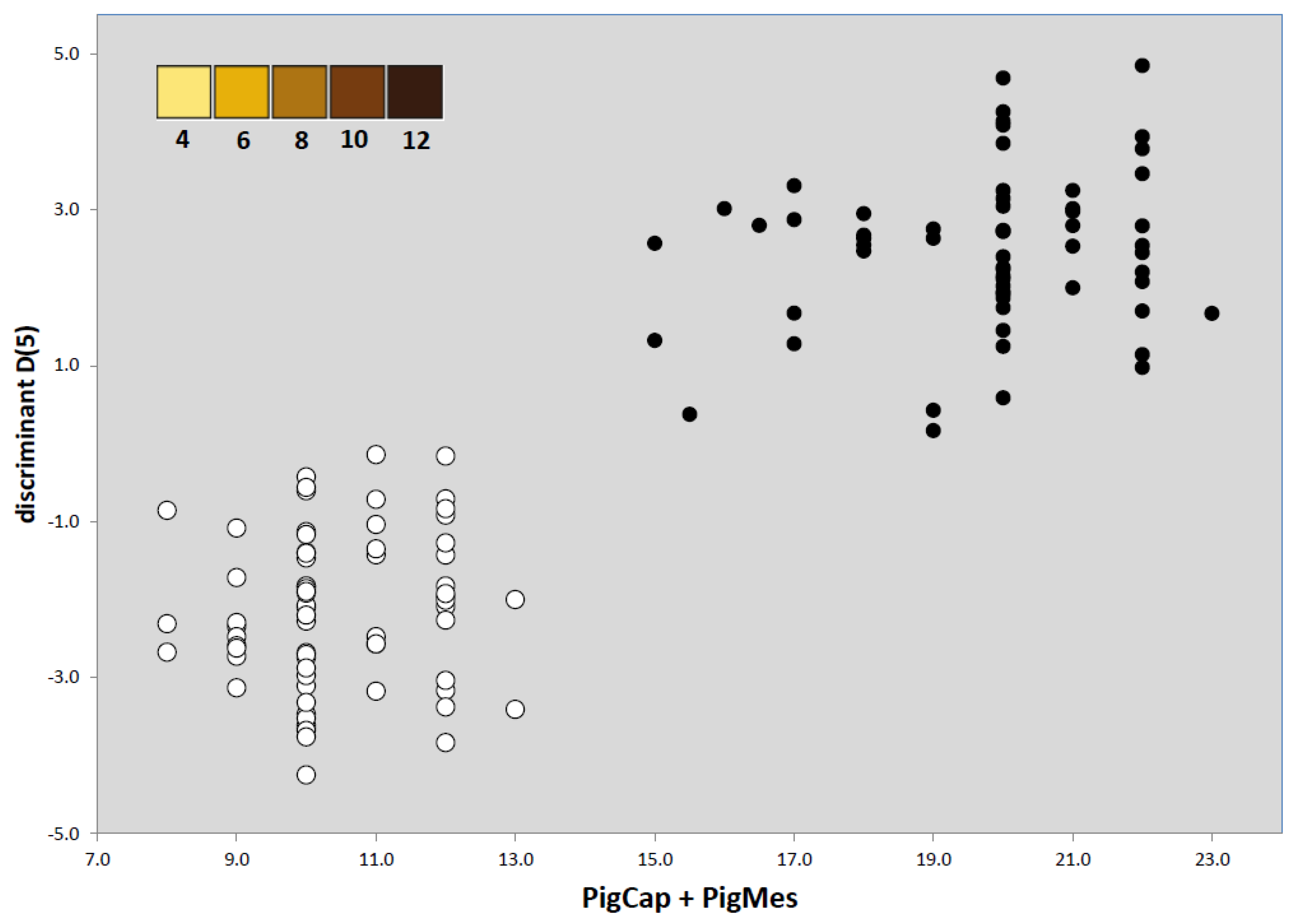
| PigCap | | | | 9.75 ± 0.99
[7,11] | 5.43 ± 0.81
[4,8] | |
| PigMes | | | | 10.07 ± 0.85
[8,12] | 5.14 ± 0.79
[4,8] | |
Table 3.
Measurements of workers of the
Cardiocondyla nivalis,
C. sima and
C. sulcata groups. Explanation of data arrangement as in
Table 1.
Table 3.
Measurements of workers of the
Cardiocondyla nivalis,
C. sima and
C. sulcata groups. Explanation of data arrangement as in
Table 1.
| | allonivalis
n.sp. (n = 4) | nivalis
(n = 7) | papuana
(n = 5) | sima
(n = 7) | sulcata
n.sp. (n = 3) |
|---|
| CS | 380 ± 17
[361,402] | 376 ± 12
[357,393] | 542 ± 16
[515,556] | 538 ± 21
[504,574] | 415 ± 11
[403,428] |
| CL/CW | 1.137 ± 0.016
[1.115,1.148] | 1.127 ± 0.021
[1.095,1.149] | 1.177 ± 0.013
[1.167,1.196] | 1.119 ± 0.026
[1.075,1.157] | 1.098 ± 0.006
[1.093,1.108] |
| SL/CS | 0.808 ± 0.019
[0.787,0.824] | 0.836 ± 0.013
[0.818,0.857] | 0.954 ± 0.012
[0.943,0.968] | 0.954 ± 0.015
[0.937,0.981] | 0.787 ± 0.013
[0.769,0.805] |
| PoOc/CL | 0.440 ± 0.005
[0.434,0.444] | 0.440 ± 0.010
[0.420,0.453] | 0.413 ± 0.013
[0.399,0.429] | 0.414 ± 0.006
[0.406,0.423] | 0.451 ± 0.006
[0.444,0.458] |
| EYE | 0.242 ± 0.002
[0.240,0.244] | 0.238 ± 0.006
[0.230,0.247] | 0.213 ± 0.002
[0.209,0.215] | 0.225 ± 0.007
[0.215,0.235] | 0.241 ± 0.002
[0.238,0.242] |
| dFOV | 15.5 ± 0.8
[ 14.8,16.5] | 13.7 ± 0.7
[12.9,14.8] | 9.1 ± 6.7
[0,16.1] | 15.7 ± 2.2
[13.0,18.7] | 17.8 ± 0.9
[16.6,19.0] |
| FRS/CS | 0.299 ± 0.010
[0.290,0.314] | 0.281 ± 0.009
[0.271,0.295] | 0.327 ± 0.003
[0.325,0.330] | 0.345 ± 0.009
[0.333,0.357] | 0.246 ± 0.010
[0.237,0.261] |
| FL/FR | 1.017 ± 0.021
[1.000,1.046] | 1.010 ± 0.011
[1.000,1.027] 6 | 1.111 ± 0.034
[1.087,1.135] 2 | 1.124 ± 0.037
[1.083,1.154] 3 | 1.000 ± 0.000
[1.000,1.000] 2 |
| SPBA/CS | 0.309 ± 0.005
[0.306,0.316] | 0.309 ± 0.010
[0.300,0.329] | 0.319 ± 0.009
[0.309,0.332] | 0.341 ± 0.012
[0.321,0.357] | 0.275 ± 0.006
[0.265,0.281] |
| SP/CS | 0.185 ± 0.009
[0.178,0.197] | 0.200 ± 0.009
[0.189,0.217] | 0.214 ± 0.009
[0.202,0.222] | 0.209 ± 0.009
[0.192,0.219] | 0.172 ± 0.005
[0.165,0.180] |
| PeW/CS | 0.319 ± 0.011
[0.304,0.329] | 0.312 ± 0.015
[0.294,0.338] | 0.270 ± 0.016
[0.253,0.294] | 0.282 ± 0.019
[0.265,0.320] | 0.229 ± 0.009
[0.218,0.242] |
| PpW/CS | 0.470 ± 0.013
[0.452,0.482] | 0.483 ± 0.018
[0.457,0.502] | 0.467 ± 0.020
[0.450,0.498] | 0.449 ± 0.017
[0.426,0.489] | 0.399 ± 0.008
[0.390,0.411] |
| PeH/CS | 0.363 ± 0.012
[0.352,0.374] | 0.349 ± 0.011
[0.334,0.362] | 0.314 ± 0.006
[0.304,0.321] | 0.341 ± 0.010
[0.323,0.352] | 0.304 ± 0.003
[0.301,0.307] |
| PpH/CS | 0.356 ± 0.006
[0.348,0.361] | 0.327 ± 0.013
[0.310,0.341] | 0.301 ± 0.019
[0.280,0.331] | 0.312 ± 0.010
[0.299,0.326] | 0.282 ± 0.005
[0.278,0.291] |
| sqPDG | 4.36 ± 0.55
[3.91,5.08] | 4.94 ± 0.20
[4.64,5.13] | 3.80 ± 0.36
[3.39,4.16] | 4.10 ± 0.44
[3.44,4.93] | 5.41 ± 0.30
[5.22,5.93] |
PLG/CS
[%] | 6.44 ± 0.66
[5.79,7.23] | 5.79 ± 0.70
[5.04,6.72] | 6.59 ± 0.40
[5.93,6.93] | 5.05 ± 0.50
[4.52,6.08] | 6.44 ± 0.39
[6.09,7.07] |
MGr/CS
[%] | 2.15 ± 0.40
[1.56,2.40] | 2.16 ± 0.34
[1.60,2.61] | 1.01 ± 0.470
[0.3,1.6] | 0.19 ± 0.22
[0.0,0.5] | 6.00 ± 0.40
[5.5,6.6] |
Table 4.
Measurements of workers of the
C. thoracica and
C. wheeleri group. Explanation of data arrangement as in
Table 1.
Table 4.
Measurements of workers of the
C. thoracica and
C. wheeleri group. Explanation of data arrangement as in
Table 1.
| | paradoxa
(n = 18) | thoracica
(n = 14) | subspina
n.sp. (n = 2) | wheeleri
(n = 2) | excavata
n.sp. (n = 3) | goroka
n.sp. (n = 3) | nigrocerea
(n = 5) |
|---|
| CS | 583 ± 23
[545,626] | 479 ± 24
[436,514] | 434 ± 7
[430,439] | 515 ± 8
[509,520] | 491 ± 9
[484,500] | 590 ± 4
[586,594] | 457 ± 4
[451,460] |
| CL/CW | 1.170 ± 0.020
[1.128,1.215] | 1.098 ± 0.022
[1.064,1.126] | 1.182 ± 0.013
[1.173,1.191] | 1.152 ± 0.016
[1.141,1.163] | 1.107 ± 0.004
[1.104,1.111] | 1.145 ± 0.013
[1.130,1.155] | 1.166 ± 0.009
[1.152,1.175] |
| SL/CS | 0.978 ± 0.013
[0.948,0.997] | 0.876 ± 0.012
[0.856,0.894] | 0.812 ± 0.006
[0.808,0.816] | 0.870 ± 0.001
[0.870,0.871] | 0.866 ± 0.004
[0.863,0.871] | 0.870 ± 0.011
[0.859,0.880] | 0.816 ± 0.004
[0.812,0.820] |
| PoOc/CL | 0.436 ± 0.007
[0.424,0.446] | 0.452 ± 0.010
[0.435,0.465] | 0.465 ± 0.003
[0.463,0.467] | 0.446 ± 0.004
[0.443,0.449] | 0.445 ± 0.002
[0.443,0.447] | 0.432 ± 0.002
[0.430,0.433] | 0.426 ± 0.008
[0.414,0.437] |
| EYE | 0.229 ± 0.005
[0.218,0.238] | 0.240 ± 0.006
[0.232,0.251] | 0.223 ± 0.006
[0.219,0.227] | 0.220 ± 0.001
[0.220,0.221] | 0.238 ± 0.002
[0.237,0.240] | 0.225 ± 0.003
[0.222,0.228] | 0.224 ± 0.004
[0.220,0.231] |
| dFOV | 18.0 ± 1.3
[16.3,20.0] | 9.8 ± 1.9
[6.8,13.2] | 16.0 ± 0.0
[16,16] | 18.6 ± 1.5
[17.5,19.6] | 16.9 ± 1.5
[18.0,18.6] | 16.3 ± 1.2
[15.0,17.3] | 15.4 ± 1.8
[13,17] |
| FRS/CS | 0.258 ± 0.011
[0.242,0.276] 17 | 0.286 ± 0.007
[0.271,0.297] | 0.290 ± 0.001
[0.289,0.290] | 0.287 ± 0.001
[0.286,0.288] | 0.277 ± 0.002
[0.276,0.279] | 0.283 ± 0.005
[0.279,0.288] | 0.272 ± 0.010
[0.259,0.282] |
| FL/FR | 1.036 ± 0.016
[1.012,1.056] 8 | 1.005 ± 0.012
[1.000,1.040] 12 | 1.000 | 1.023 ± 0.001
[1.022,1.024] | 1.015 ± 0.015
[1.000,1.030] | 1.035 | 1.031 ± 0.013
[1.022,1.046] |
| SPBA/CS | 0.255 ± 0.012
[0.237,0.276] | 0.333 ± 0.015
[0.315,0.368] | 0.310 ± 0.001
[0.309,0.310] | 0.366 ± 0.004
[0.364,0.369] | 0.346 ± 0.006
[0.341,0.353] | 0.370 ± 0.007
[0.362,0.374] | 0.347 ± 0.005
[0.342,0.350] |
| SP/CS | 0.283 ± 0.025
[0.230,0.313] | 0.335 ± 0.014
[0.306,0.365] | 0.268 ± 0.010
[0.261,0.275] | 0.404 ± 0.021
[0.389,0.418] | 0.380 ± 0.004
[0.376,0.383] | 0.332 ± 0.016
[0.323,0.351] | 0.314 ± 0.012
[0.296,0.326] |
| PeW/CS | 0.233 ± 0.014
[0.208,0.258] | 0.286 ± 0.021
[0.259,0.326] | 0.245 ± 0.008
[0.239,0.251] | 0.284 ± 0.006
[0.280,0.288] | 0.257 ± 0.003
[0.254,0.260] | 0.284 ± 0.005
[0.280,0.290] | 0.270 ± 0.013
[0.253,0.282] |
| PpW/CS | 0.456 ± 0.026
[0.406,0.497] | 0.525 ± 0.015
[0.508,0.562] | 0.457 ± 0.000
[0.457,0.457] | 0.557 ± 0.006
[0.553,0.561] | 0.536 ± 0.009
[0.530,0.546] | 0.524 ± 0.013
[0.513,0.539] | 0.541 ± 0.023
[0.515,0.561] |
| PeH/CS | 0.275 ± 0.018
[0.239,0.297] | 0.349 ± 0.012
[0.333,0.378] | 0.344 ± 0.003
[0.342,0.346] | 0.344 ± 0.002
[0.343,0.346] | 0.336 ± 0.009
[0.325,0.342] | 0.351 ± 0.008
[0.344,0.359] | 0.379 ± 0.005
[0.374,0.386] |
| PpH/CS | 0.310 ± 0.013
[0.295,0.334] | 0.270 ± 0.007
[0.260,0.282] | 0.307 ± 0.006
[0.303,0.311] | 0.273 ± 0.006
[0.269,0.277] | 0.256 ± 0.011
[0.244,0.267] | 0.280 ± 0.009
[0.272,0.289] | 0.244 ± 0.008
[0.237,0.257] |
| sqPDG | 8.04 ± 1.12
[6.43,9.95] | 6.33 ± 0.44
[5.77,7.14] | 6.54 ± 0.42
[6.24,6.83] | 5.71 ± 0.04
[5.68,5.74] | 5.80 ± 0.34
[5.51,6.18] | 5.34 ± 0.33
[4.97,5.60] | 5.97 ± 0.43
[5.55,6.67] |
PLG/CS
[%] | 3.28 ± 0.22
[2.96,3.70] | 4.07 ± 0.45
[3.48,4.73] | 4.21 ± 0.08
[4.15,4.27] | 4.44 ± 0.08
[4.38,4.50] | 4.68 ± 0.33
[4.33,4.99] | 4.25 ± 0.60
[3.61,4.80] | 4.03 ± 0.33
[3.82,4.59] |
MGr/CS
[%] | 6.22 ± 0.73
[5.1,7.6] | 0.63 ± 0.42
[0.0,1.4] | 1.62 ± 0.51
[1.3,2.0] | 2.68 ± 0.03
[2.7,2.7] | 1.75 ± 0.62
[1.4,2.5] | 2.89 ± 0.11
[2.8,3.0] | 0.90 ± 0.93
[0.3,2.6] |
Monotypical group. Metanotal groove very deep and with an extremely steep posterior slope that forms with the anterior slope a distinct angle of 90–100°. Petiole with a very long peduncle, in dorsal view more than twice as long than wide and with a small circular node, in profile the node is low with a convex dorsum. Postpetiolar sternite almost flat, only with suggested and very rounded anterolateral corners. Only a single Orientalic species known from Malaysia:
Cardiocondyla sulcata n.sp. Analyzed in this paper on species level.
Figure 25;
Table 3.
Antennae with 11 segments instead of 12 in all other species of the genus. Collective length of the three apical antennal segments 57–63% of whole funiculus length, in other species shorter. Anterior part of postpetiole in dorsal view conic. Petiole with a very long peduncle and well-developed node. Scape very long in terms of the genus (SL/CS 0.937–0.981). At least two polymorphic Australasian species, no tramp species known:
C. sima Wheeler, 1935,
C. papuana Reiskind, 1965. Analyzed in this paper on species level.
Figure 26 and
Figure 27;
Table 3.
Monotypical group. Head, mesosoma, waist and gaster on whole exposed surface with a rich silvery glinting pubescence having flattened hairs. Dorsal profile of mesosoma from anterior part of pronotum to caudal part of propodeum continuously convex, the latter without any suggestions of spines or dents. Petiole extremely slender, with a very long peduncle and a long and narrow node with a feebly convex dorsal profile. Postpetiole narrow and low, distinctly longer than wide, in dorsal view its anterior half conically narrowing frontad. Only a single Afrotropical species known: Cardiocondyla monardi Santschi, 1930. Not further considered in this paper.
Monotypical group. Assessment based on evaluation of images CASENT09011751 of the holotype in
www.antweb.org. The most eye-catching character is petiole shape in lateral view which is not seen in any Afrotropical
Cardiocondyla species. Overall, this slender ant shows similarity to members of the
Cardiocondyla nuda and
C. shuckardi group but it differs from these by a much narrower frons and lower petiole and a more approached spine base. The ratio FRS*PeH/SL is 3.40% in the holotype of
C. longinoda but 5.34 ± 0.48 [3.99, 6.88]% in 759 individuals of the
C. nuda and
C. shuckardi group. The slenderness of
C. longinoda is also expressed by the 2nd component of a PCA considering the characters CS, SL, FRS, SPBA and PeH which is −3.80 in the holotype of
C. longinoda but 0.005 ± 0.991 [−2.36, 3.59] in 759 individuals of the
C. nuda and
C. shuckardi group. Only a single Afrotropical species known:
Cardiocondyla longinoda Rigato, 2002. Not further considered in this paper.
Dorsal profile of promesonotum and of propodeum frontal of spine base feebly but continuously convex—as result, the always developed and rather deep metanotal depression is formed by very shallow anterior and posterior slopes (MGr/CS 1.6–6.3%). Propodeal spines always short (SP/CS 0.029–0.088), reduced do blunt dents or obtusely angled corners. Petiole node in dorsal view always globular. Postpetiole rather narrow (PpW/CS 0.399–0.490), its sides in dorsal view rounded, outlines of postpetiole thus never not forming a hexagon. About nine species occurring in Africa, the Arab Peninsula and Iran of which one species was introduced to the Caribbean and Hawaii: Cardiocondyla shuckardi Forel, 1891, C. venustula Wheeler, 1908, C. fajumensis Forel, 1913, C. globinodis Stitz, 1923, C. sekhemka Bolton, 1982, C. zoserka Bolton, 1982, C. melana Seifert, 2003, C. unicalis Seifert, 2003. Not analyzed in this paper on species level.
Promesonotal and anterior propodeal profiles usually not forming evenly convex curvatures—as result, metanotal depression, if present at all, not as wide and with steeper anterior and posterior slopes. Propodeal spines short, appearing in lateral view as smaller angles of 60–95°. Postpetiole in dorsal aspect frequently with angulate-convex sides—the outlines of postpetiole thus resembling a hexagon. Eyes small (EYE/CS 0.231 ± 0.05), postocular index rather large (PoOc/CL 0.454 ± 0.013) and postpetiole rather narrow (PpW/CS 0.487 ± 0.030). Basic type of sculpture on paramedian vertex and mesosoma microreticulate, though varying in strength. Eight species from the tropical to south temperate zones of the Old World, with one species introduced to the Americas: C. nuda (Mayr 1866), C. mauritanica Forel, 1890, C. atalanta Forel, 1915, C. strigifrons Viehmeyer, 1922, C. kagutsuchi Terayama, 1999, C. paranuda Seifert, 2003, C. compressa Seifert et al. 2017, C. itsukii Seifert et al. 2017. Not analyzed in this paper on species level.
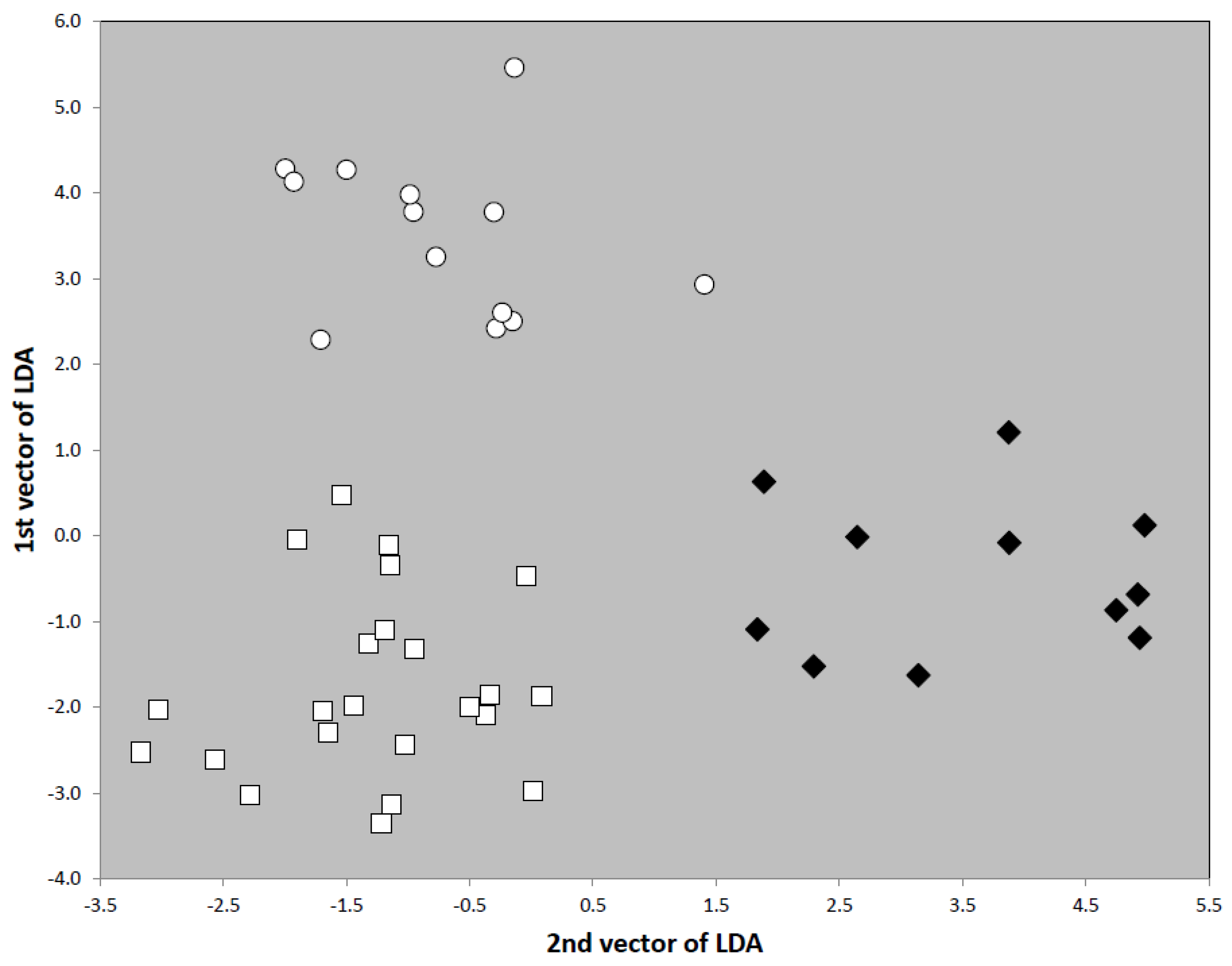
Note: There are similarities to the C. shuckardi group questioning if a separation in different groups by subtle differences in mesosoma profile and postpetiole shape are reasonable. Yet, the morphometric separation of the C. shuckardi and C. nuda group is clear. Individual workers of seven available species in the C. shuckardi group and of all eight species of the C. nuda group were correctly classified by a linear discriminant function in 98.8% of the cases. With all linear measures in mm and sqPDG as square root of the µm value, the discriminant 195.98∗MGr + 0.29∗sqPDG − 9.636∗CW − 24.69∗FRS + 48.59∗SPBA − 71.61∗SP + 58.30∗PeW − 77.64∗PpW + 56.53∗PpH + 2.367 was −1.343 ± 0.973 [−4.100, 1.430] in 540 workers of the C. nuda group and 3.342 ± 1.065 [0.465, 6.606] in 217 workers of C. shuckardi group.
True foveolae on vertex completely absent. Instead the bases of pubescence hairs are placed in the center of flat tubercles or flat pits of small diameter, giving a finely punctate surface appearance at lower magnifications, dFov 8.13 ± 0.78 [5.0, 10.0]. Frons very wide, FRS/CS 0.320 ± 0.015 [0.269, 0.353]. Propodeal spines reduced to blunt dents, SP/CS 0.070 ± 0.013 [0.027, 0.096]. Metanotal depression deep, MGr/CS 3.57 ± 0.83 [1.63, 6.16]%; all numeric data from 153 worker individuals of five species. SE Europe across Asia Minor eastwards to Tibet and Mongolia with four described and one undescribed species: C. stambuloffii Forel, 1892, C. koshewnikovi Ruzsky, 1902, C. gibbosa Kuznetzov-Ugamsky, 1927, C. tibetana Seifert, 2003 and Cardiocondyla sp. code ROLA. Not analyzed in this paper on species level.
Comment: The members of the C. batesii, C. elegans and C. ulinanini groups as they are proposed below are morphologically similar. This grouping is confirmed by an LDA considering all morphometric characters with positive checks in wild-card runs for rare species with very few samples available.
Eye very large (EYE/CS 0.263 ± 0.08), postocular distance very small (PoOc/CL 0.368 ± 0.016), head rather long (CL/CW 1.176 ± 0.023), metanotal depression always present (MGr/CS 3.51 ± 0.82%), propodeal spines short (SP/CS 0.100 ± 0.013), pubescence on first gaster tergite rather short (PLG/CS 5.50 ± 0.43%), postpetiolar sternite completely flat, without any protrusions. Nine species from the southern zone of West Palaearctic, tramp species unknown: Cardiocondyla batesii Forel, 1894, C. nigra Forel, 1905, C. kushanica Pisarski, 1967, C. brachyceps Seifert, 2003, C. opisthopsis Seifert, 2003, C. rugulosa Seifert, 2003, C. semirubra Seifert, 2003, C. tenuifrons Seifert, 2003 and Cardiocondyla sp. code VERD. Not analyzed in this paper on species level.
Eye large (EYE/CS 0.250 ± 0.006), postocular distance small (PoOc/CL 0.395 ± 0.009), head rather short (CL/CW 1.156 ± 0.017), metanotal depression always present (MGr/CS 4.50 ± 0.76%), propodeal spines short (SP/CS 0.116 ± 0.013), pubescence on first gaster tergite very long (PLG/CS 7.84 ± 0.51%), postpetiolar sternite without any protrusions. Three described and one undescribed species from the southern zone of the West Palaearctic: C. elegans Emery, 1869; C. dalmatica Soudek, 1925, C. brachyceps Seifert, 2003 and Cardiocondyla sp. code DAOI. Not analyzed in this paper on species level.
Eye large (EYE/CS 0.248 ± 0.012), postocular distance larger (PoOc/CL 0.407 ± 0.031), head longer (CL/CW 1.160 ± 0.022), metanotal depression always present (MGr/CS 3.85 ± 0.84%), propodeal spines short (SP/CS 0.114 ± 0.015), pubescence on first gaster tergite rather long (PLG/CS 6.55 ± 1.06%), postpetiolar sternite without any protrusions. Seven described and one undescribed species from the southern zone of the West and Central Palaearctic: C. ulianini Emery, 1889; C. bulgarica Forel, 1892, C. sahlbergi Forel, 1913, C. gallilaeica Seifert, 2003, C. israelica Seifert, 2003, C. littoralis Seifert, 2003 and Cardiocondyla sp. code CASP. Not analyzed in this paper on species level.
4.3. Key to the Workers of the Considered Species Groups
The key below considering the Orientalic and Australasian faunal regions would have to encompass not only the 26 species of the seven species groups treated here but also 15 species from other species groups occurring here. Several of the latter are world-wide tramp species of African origin. For reasons given in the introduction, I will not consider these accessory species here. Users should firstly look at the diagnoses of the species groups given above and simultaneously compare the images of the species which are given for each species in three visual positions and in order of relatedness. This will allow a good entry into the issue. 1a Antennae with 11 segments. Collective length of the three apical antennal segments 57–63% of whole funiculus length. Anterior part of postpetiole in dorsal view conic. Petiole with a very long peduncle and well-developed node. ..........................................................................................................................2.
1b Antennae with 12 segments. Collective length of the four apical antennal segments 57–63% of whole funiculus length. Anterior part of postpetiole in dorsal not conic.................................................................................................................................................…3.
2a Occurring west of the Wallace Line. Head shorter, CL/CW 1.057–1.157. Petiole node higher, (PeH/CS 0.323–0.352), in lateral view more produced frontad, with concave anterior profile, the dorsal profile and caudal slope form a continuous convexity (
Figure 26B). Pubescence on 1st gaster tergite shorter, PLG/CS 4.52–6.08% (
Figure 26).
sima.
2b Occurring east of the Wallace Line. Head longer (CL/CW 1.167–1.196). Petiole node lower (PeH/CS 0.304–0.321, in lateral view slightly produced caudad (
Figure 27B). Pubescence on 1st gaster tergite longer, PLG/CS 5.93–6.93% (
Figure 27).
papuana.
3a Metanotal depression very deep (MGr/CS 5.5–6.6%) and with an extremely steep posterior slope that forms with the anterior slope a distinct angle of 90–100° (
Figure 25B). Scape very short (SL/CS 0.769–0.805), petiole very narrow (PeW/CS 0.218–0.242). Postpetiolar sternite almost flat, only with suggested and very rounded anterolateral corners (
Figure 25).
sulcata n.sp.
3b Metanotal depression less developed or absent, if similarly deep than scape much longer (SL/CS > 0.85) or petiole much wider (PeW/CS > 0.25). ............................................4.
4a Pronotal corners in dorsal view very prominent, sharp, forming a distinct angle of 70–80° (
Figure 22C,
Figure 23C and
Figure 24C). Spine base narrower (SPBA/CS 0.237–0.368). …………
5.
4b Pronotal corners in dorsal view developed but blunt (
Figure 13C,
Figure 14C,
Figure 18C,
Figure 19C,
Figure 20C and
Figure 21C). Spine base very broad (SPBA/CS 0.322–0.388). ........................................................................................................................................................
7.
4c Pronotal corners in dorsal view absent. ............................................................................12.
5a Scape extremely long, SL/CS > 0.92. Metanotal groove deep, MGr/CS > 4%. Petiole very elongated and very low, PeH/CS < 0.31 (
Figure 23).
paradoxa.
5b Scape much shorter, SL/CS < 0.92. Metanotal groove shallower or absent, MGr/CS < 4%. Petiole not extremely elongated and higher, PeH/CS > 0.31. .......................................6.
6a Dorsal surface of head shining, with scattered foveolae the distance of which is much larger than their diameter. Anterior clypeal margin deeply excavated (
Figure 22A). Anterior postpetiolar sternite in lateral view not with a sharp corner (
Figure 22B). Head blackish brown (
Figure 22).
thoracica.
6b Dorsal surface of head not shining, with more densely packed foveolae the distance of which is as large or smaller than their diameter. Anterior clypeal margin not deeply excavated (
Figure 24A). Anterior postpetiolar sternite in lateral view with a sharp corner (
Figure 24D). Head yellowish (
Figure 24).
subspina n.sp.
7a Vertex and dorsal mesosoma with very deep foveolae many of which show an inner structure reminiscent of a three- or four-leafed cloverleaf (
Figure 13D and
Figure 14D). Frons wider, FRS/CS 0.309–0.357. Postpetiole narrower, PpW/CS 0.428–0.531. ........................................................................................................................................................
8.
7b Vertex and dorsal mesosoma with shallower, bicoronate foveolae lacking an inner structure reminiscent of a three- or four-leafed cloverleaf. Frons narrower, FRS/CS 0.259–0.289. Postpetiole wider, PpW/CS 0.513–0.569. .......................................................................9.
8a Whole body rather concolorous dirty to blackish brown. With all measurements in mm, discriminant 39.855∗CW + 32.809∗SPBA − 129.082∗PpH + 505.05∗PLG − 11.363 > 0 [error 0% in 61 individuals] (
Figure 13).
latifrons n.sp.
8b Whole body concolorous yellowish. Discriminant < 0 [error 0% in 70 individuals] (
Figure 14).
micropila n.sp.
9a Petiole very high (PeH/CS > 0.365), its node in profile nearly rectangular, with a long and rather straight dorsal profile. Dorsal mesosomal profile from pronotum to spine tips rather continuously straight (
Figure 21B). Scape short, SL/CS < 0.84, ratio SL/PeH∗PpH < 0.58 (
Figure 21).
nigrocerea.
9b Petiole lower (PeH/CS < 0.365), not quasi rectangular in profile with the dorsum of its node convex. Dorsal mesosomal profile from pronotum to spine tips not continuously straight. Scape longer, SL/CS > 0.84, ratio SL/PeH∗PpH > 0.58. ........................................10.
10a Anterior clypeal margin strongly concave (
Figure 19A). Head short, CL/CW 1.104–1.111, spine base narrower, SPBA/CS 0.341–0.353. Central vertex only foveolate, not with fine longitudinal carinulae (
Figure 19).
excavata n.sp.
10b Anterior clypeal margin not concave (
Figure 18A and
Figure 20A). Head longer, CL/CW 1.130–1.155. Spine base broader, SPBA/CS 0.362–0.375. Central vertex in addition to abundant foveolae finely longitudinally carinulate. ...................................................................
11.
11a Propodeal spines longer, SP/CS 0.389–0.418. Body size smaller, CS < 0.55 mm. Anterior face of petiole in profile feebly concave (
Figure 18).
wheeleri.
11b Propodeal spines shorter, SP/CS 0.323–0.351. Body size larger, CS > 0.55 mm. Anterior face of petiole in profile more concave (
Figure 20).
goroka n.sp.
12a Mesosoma thickset and hunchbacked, its dorsal profile strongly and evenly convex (
Figure 10B), ratio mesosoma length without neck shield against mesosoma height 1.95–2.12. Postpetiolar sternite bilobate or bicuspidate (
Figure 11D and
Figure 12D). .......................................................................................................................................................
13.
12b Mesosoma more slender and not hunchbacked, its dorsal profile not strongly and evenly convex, ratio mesosoma length without neck shield against mesosoma height 2.30–2.67. Postpetiolar sternite flat, not bilobate or bicuspidate (except for C. allonivalis n.sp.). .......................................................................................................................................................16.
13a Lateral head at horizontal level of eye with an longitudinal, dark brown ribbon that is as broad as the eye; this ribbon is flanked below and above by broad bands without any pigment. Petiole without any pigmentation (appearing whitish in dead, dry specimens). Propodeal spines shorter, SP/CS < 0.235, frons narrower FRS/CS < 0.260 (
Figure 15).
pirata.
13b Lateral head without ribbons and bands. Petiole at least with light yellowish pigmentation. Propodeal spines longer, SP/CS > 0.235, frons broader, FRS/CS > 0.260…………14.
14a Head very short, CL/CW 1.038–1.065. Dorsum of petiole node in profile semicircular (
Figure 10B). Silvery pubescence hairs on head flat (
Figure 10).
argentea n.sp.
14b Head longer, CL/CW 1.094–1.182. Dorsum of petiole node in profile broadly convex, not semicircular (
Figure 11B and
Figure 12B). ......................................................................................................................................................
15.
15a Mesosoma in lateral view more humpbacked and thickset, without any suggestion of a metanotal depression (
Figure 11B). Frons narrower, FRS/CS 0.261–0.285. Overall head shape in dorsal view more rectangular (
Figure 11A). Occurring east of the Wallace Line (
Figure 11).
semiargentea n.sp.
15b Mesosoma in lateral view less humpbacked and slender, sometimes with a suggestion of a metanotal depression (
Figure 12B). Frons wider, FRS/CS 0.292–0.319. Overall head shape in dorsal view more oval (
Figure 12A). Occurring west of the Wallace Line (
Figure 12).
argyrotricha n.sp.
16a Frons wider, FRS/CS 0.271–0.314. Propodeal spines longer, SP/CS 0.178–0.217. Postpetiole much higher, PpH/CS 0.310–0.361. .............................................................................17.
16b Frons narrower, FRS/CS 0.221–0.261. Propodeal spines shorter, SP/CS 0.089–0.156. Postpetiole much lower, PpH/CS 0.223–0.294. ......................................................................18.
17a Postpetiolar sternite in profile flat without any anterolateral corners (
Figure 16).
nivalis.
17b Postpetiolar sternite in profile strongly bulging; this bulge is formed by bilateral lobes strongly protruding compared to median level of sternite; these lobes appear as corners in anterolateral view (
Figure 17).
allonivalis n.sp.
18a Absolute scape length much larger, SL 385–443 µm (
Figure 9).
parvinoda.
18b Absolute scape length much smaller, SL 270–354 µm. .................................................19.
19a Posterior part of vertex glabrous with small foveolae of 7 µm mean diameter and distance between foveolae larger than their diameter. Mesosoma shining with very weak reticulate microsculpture and propodeal spines reduced to blunt dents (
Figure 8).
britteni.
19b Posterior part of vertex matt, with strongly developed microsculpture and foveolae of at least 12 µm mean diameter. Mesosoma not shiny, with well developed reticulate microsculpture and sharper, longer propodeal spines. ........................................................20.
20a Petiole very low (PeH/CS 0.273–0.284), in profile with a flat convex node (
Figure 5B). Head short, CL/CW 1.167–1.199. First gaster tergite shiny, its basal part may show a barely visible microreticulum (
Figure 5).
schulzi n.sp.
20b Petiole higher (PeH/CS 0.283–0.368), in profile with a more developed node. If petiole shape similar (opaca), then CL/CW much larger and surfaces of gaster tergites completely matt, very finely and densely longitudinally shagreened. .....................................21.
21a Scape long, SL/CS 0.836. Petiole low, PeH/CS 0.283. All body surfaces completely matt because of a dense carinulate-foveolate microsculpture (
Figure 7).
opaca.
21b Scape much shorter, SL/CS 0.725–0.790. Petiole higher, PeH/CS 0.308–0.368. …………………………………………………………………………..……………………….22.
22a Exposed surfaces of gaster tergites completely matt, very finely and densely shagreened (
Figure 6).
carbonaria.
Determination of these three species only possible by linear discriminant analysis (LDA). It is recommended to record the following characters in mm: CL, CW, SL, EYE, FRS, SP, PpW, PeH, PpH and PLG and then running the test specimens as wild-cards in a LDA against the data given in of
Supplementary Information Table S1. The total classification error in 210 worker individuals in
Table S1 is 2.4%.
Figure 2.
Cardiocondyla minutior: head in dorsal view (A), lateral view (B), dorsal view (C).
Figure 2.
Cardiocondyla minutior: head in dorsal view (A), lateral view (B), dorsal view (C).
Figure 3.
Cardiocondyla goa, holotype: head in dorsal view (A), lateral view (B), dorsal view (C).
Figure 3.
Cardiocondyla goa, holotype: head in dorsal view (A), lateral view (B), dorsal view (C).
Figure 4.
Cardiocondyla tjibodana: head in dorsal view (A), lateral view (B) dorsal view (C).
Figure 4.
Cardiocondyla tjibodana: head in dorsal view (A), lateral view (B) dorsal view (C).
Figure 5.
Cardiocondyla schulzi, holotype: head in dorsal view (A), lateral view (B), dorsal view (C).
Figure 5.
Cardiocondyla schulzi, holotype: head in dorsal view (A), lateral view (B), dorsal view (C).
Figure 6.
Cardiocondyla carbonaria, holotype (specimen CASENT0908337 from
www.antweb.org, photo Will Ericson): head in dorsal view (
A); lateral view (
B); dorsal view (
C).
Figure 6.
Cardiocondyla carbonaria, holotype (specimen CASENT0908337 from
www.antweb.org, photo Will Ericson): head in dorsal view (
A); lateral view (
B); dorsal view (
C).
Figure 7.
Cardiocondyla opaca, holotype: head in dorsal view (A), lateral view (B), dorsal view (C).
Figure 7.
Cardiocondyla opaca, holotype: head in dorsal view (A), lateral view (B), dorsal view (C).
Figure 8.
Cardiocondyla britteni, holotype (specimen CASENT0901496 from
www.antweb.org, photo Ryan Perry): head in dorsal view (
A), lateral view (
B), dorsal view (
C).
Figure 8.
Cardiocondyla britteni, holotype (specimen CASENT0901496 from
www.antweb.org, photo Ryan Perry): head in dorsal view (
A), lateral view (
B), dorsal view (
C).
Figure 9.
Cardiocondyla parvinoda: head in dorsal view (A), lateral view (B), dorsal view (C).
Figure 9.
Cardiocondyla parvinoda: head in dorsal view (A), lateral view (B), dorsal view (C).
Figure 10.
Cardiocondyla argentea n.sp. paratype: head in dorsal view (A), lateral view (B), dorsal view (C), vertex median of the eye (D).
Figure 10.
Cardiocondyla argentea n.sp. paratype: head in dorsal view (A), lateral view (B), dorsal view (C), vertex median of the eye (D).
Figure 11.
Cardiocondylasemiargentea n.sp.: paratype, head in dorsal view (A), lateral view (B), dorsal view (C) waist in frontolateral view (D).
Figure 11.
Cardiocondylasemiargentea n.sp.: paratype, head in dorsal view (A), lateral view (B), dorsal view (C) waist in frontolateral view (D).
Figure 12.
Cardiocondyla argyrotricha n.sp. holotype: head in dorsal view(A), lateral view (B), dorsal view (C), waist in lateral view (D).
Figure 12.
Cardiocondyla argyrotricha n.sp. holotype: head in dorsal view(A), lateral view (B), dorsal view (C), waist in lateral view (D).
Figure 13.
Cardiocondyla latifrons n.sp.: head in dorsal view (A), lateral view (B), dorsal view (C), vertex median of the eye (D).
Figure 13.
Cardiocondyla latifrons n.sp.: head in dorsal view (A), lateral view (B), dorsal view (C), vertex median of the eye (D).
Figure 14.
Cardiocondyla micropila n.sp., holotype: head in dorsal view (A), lateral view (B), dorsal view (C), vertex median of the eye (D).
Figure 14.
Cardiocondyla micropila n.sp., holotype: head in dorsal view (A), lateral view (B), dorsal view (C), vertex median of the eye (D).
Figure 15.
Cardiocondyla pirata holotype: head in dorsal view (A), lateral view (B), dorsal view (C).
Figure 15.
Cardiocondyla pirata holotype: head in dorsal view (A), lateral view (B), dorsal view (C).
Figure 16.
Cardiocondyla nivalis: head in dorsal view (A), lateral view (B), dorsal view (C), vertex median of the eye (D).
Figure 16.
Cardiocondyla nivalis: head in dorsal view (A), lateral view (B), dorsal view (C), vertex median of the eye (D).
Figure 17.
Cardiocondyla allonivalis n.sp., holotype: head in dorsal view (A), lateral view (B), dorsal view (C), waist in frontolateral view (D).
Figure 17.
Cardiocondyla allonivalis n.sp., holotype: head in dorsal view (A), lateral view (B), dorsal view (C), waist in frontolateral view (D).
Figure 18.
Cardiocondyla wheeleri, syntype: head in dorsal view (A), lateral view (B), dorsal view (C).
Figure 18.
Cardiocondyla wheeleri, syntype: head in dorsal view (A), lateral view (B), dorsal view (C).
Figure 19.
Cardiocondyla excavata n.sp., holotype: head in dorsal view (A), lateral view (B), dorsal view (C).
Figure 19.
Cardiocondyla excavata n.sp., holotype: head in dorsal view (A), lateral view (B), dorsal view (C).
Figure 20.
Cardiocondyla goroka n.sp., paratype: head in dorsal view (A), lateral view (B), dorsal view (C).
Figure 20.
Cardiocondyla goroka n.sp., paratype: head in dorsal view (A), lateral view (B), dorsal view (C).
Figure 21.
Cardiocondyla nigrocerea: head in dorsal view: (A), lateral view (B), dorsal view (C).
Figure 21.
Cardiocondyla nigrocerea: head in dorsal view: (A), lateral view (B), dorsal view (C).
Figure 22.
Cardiocondyla thoracica: head in dorsal view (A), lateral view (B), dorsal view (C), vertex median of the eye (D).
Figure 22.
Cardiocondyla thoracica: head in dorsal view (A), lateral view (B), dorsal view (C), vertex median of the eye (D).
Figure 23.
Cardiocondyla paradoxa: head in dorsal view (A), lateral view (B), dorsal view (C).
Figure 23.
Cardiocondyla paradoxa: head in dorsal view (A), lateral view (B), dorsal view (C).
Figure 24.
Cardiocondyla subspina n.sp., paratype: head in dorsal view (A), lateral view (B), dorsal view (C), waist in lateral view (D).
Figure 24.
Cardiocondyla subspina n.sp., paratype: head in dorsal view (A), lateral view (B), dorsal view (C), waist in lateral view (D).
Figure 25.
Cardiocondyla sulcata n.sp., paratype: head in dorsal view (A), lateral view (B), dorsal view (C). (85).
Figure 25.
Cardiocondyla sulcata n.sp., paratype: head in dorsal view (A), lateral view (B), dorsal view (C). (85).
Figure 26.
Cardiocondyla sima: head in dorsal view (A), lateral view (B), dorsal view (C).
Figure 26.
Cardiocondyla sima: head in dorsal view (A), lateral view (B), dorsal view (C).
Figure 27.
Cardiocondyla papuana: head in dorsal view, (A), lateral view (B), dorsal view (C).
Figure 27.
Cardiocondyla papuana: head in dorsal view, (A), lateral view (B), dorsal view (C).
4.4. Treatment by Species
The reasons for identification of a taxon are given in square brackets after taxonomic name, author and year.
4.4.1. Cardiocondyla minutior Forel, 1899
Cardiocondyla nuda var. minutior Forel, 1899 [type investigation]
This taxon has been described from Hawaii. Investigated were three syntype workers labelled “C. nuda Mayr v. minutior type Forel, Hawai” and Molokai Mts., 3000 ft. Perkins 1893”; depository MHN Genève.
Cardiocondyla tsukuyomi Terayama, 1999 [junior synonym, type investigation]
This taxon has been described from the island of Ada in the Okinawa Archipelago. Investigated were six paratype workers from the holotype nest labelled “VI 1988 K. Yamauchi leg., Ada, Okinawa-jima Okinawa Pref.” and “
Cardiocondyla tsukuyomi Terayama, 1999”; depository SMN Görlitz. The synonymization with
C. minutior has already been stated by Seifert [
9]. For a more comprehensive argumentation see below under taxonomic comments and clustering results.
Cardiocondyla breviscapus Seifert, 2003 [junior synonym, type investigation]
This taxon has been described from India. Investigated were the holotype and two paratype workers labelled “INDIA: Coimbatore 25.ix.79 J. Noyes” and “Holotype
Cardiocondyla breviscapus Seifert” or “Paratypes
Cardiocondyla breviscapus Seifert”; depository BMNH London. The introduction of this taxon was due to insufficient knowledge of intraspecific variability in the year 2003. The recent exploratory and hypothesis-driven data analyses performed in a larger sample indicate a synonymy with
C. minutior (see
Section 4.4.1 under taxonomic comments and clustering results).
All material examined. Including those single-specimens samples with wild-card LDA classifications having posterior probabilities
p > 0.95, numeric phenotypical data were available in 40 samples (largely nest samples) with 92 workers. For details see
Supplementary Information Tables S1 and S2. This material originated from Hawaii (3 samples), New Zeeland (1), the Society Islands (2), Ecuador (1), Brazil (2), Florida (5), Puerto Rico (3), Trinidad and Tobago (5), Egypt (2), India (2), Sri Lanka (2), Malaysia (1), and smaller Japanese Pacific islands (11).
Geographic range. Worldwide distributed in the tropical zone but so far not confirmed for Africa. According to determined vouchers, distributed between 19° S and 33° N. The verified findings are indicated in the previous paragraph. Hypothesizing the Indo-Malayan region as natural range, it is the species with the largest tramp species potential within the
C. minutior group, having colonized the Caribbean and diverse Pacific islands. It is intriguing that some 72% of all samples are from the latter two areas whereas only 16% are from the Indo-Malayan region (see also Wetterer [
25]). There are no reports on occurrence in greenhouses of the temperate zone.
Diagnosis: Worker (
Table 1,
Figure 2, key; pictures CASENT0908344 (type
minutior), CASENT0901759 (holotype
breviscapa), ANTWEB1041246 (paratype
breviscapa), ANTWEB1041253 (type
tsukuyomi) in
www.antweb.org): Small size, CS 422 µm. Head elongated, CL/CW 1.263. Postocular distance very large, PoOc/CL 0.475. Scape short, SL/CS 0.754. Eye rather small, EYE/CS 0.232, with notable micropilae, the longest measuring 6–10 µm. Occipital margin straight or very weakly concave. Frontal carinae slightly converging immediately caudal of FRS level (FL/FR 1.062). Dorsal profile of mesosoma rather straight or weakly convex, metanotal groove only suggested or entirely absent (MGr/CS 0.39%). Spines rather short and acute (SP/CS 0.133), their axis in profile deviating by 40–45° from longitudinal axis of mesosoma. Petiole rather wide and high (PeW/CS 0.283, PeH/CS 0.332), in profile with concave anterior face and rounded node that is in dorsal view circular and as long as wide. Postpetiole rather wide and very low (PpW/CS 0.475, PpH/CS 0.274), its sternite completely flat, without any anteroventral bulge; in dorsal view with angulate-convex sides and straight anterior margin. Paramedian vertex with deeply impressed, flat-bottomed foveolae of 13–18 µm diameter in dense honey-comb arrangement (if not displaced by longitudinal carinulae). Foveolae with an inner corona (margin of a flat tubercle) of 7–8 µm diameter. Mesosoma on whole surface sculptured. Waist segments with shallower and finer reticulum than on mesosoma, nodes sometimes slightly shiny. First gaster tergite with fine microreticulum that may be obscured by polluted surfaces. Pubescence on whole body long and dense, PLG/CS 7.9%, sqPDG 3.23. Color of head, mesosoma, and waist varying considerably from dirty yellowish to dark dirty brown, gaster dark to blackish brown.
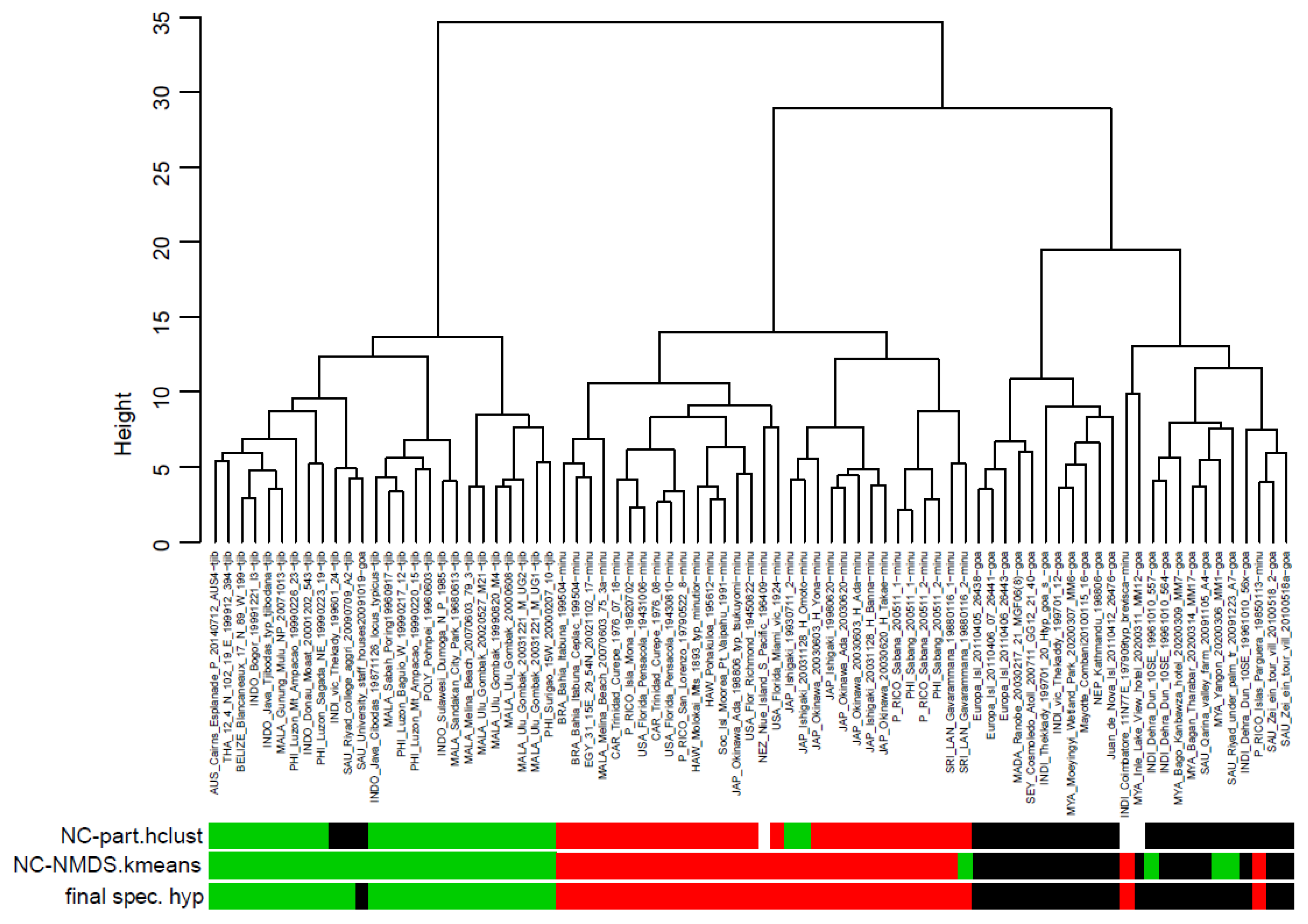
Taxonomic comments and clustering results.
C. minutior is phenotypically extremely similar to
C. goa and
C. tjibodana. Yet, the three taxa can be clustered by the exploratory data analyses (EDAs) NC-Ward, NC-part.hclust and NC-NMDS.kmeans using the 15 characters CS, CL/CW, PoOc/CL, SL/CS, FRS/CS, EYE/CS, MGr/CS, SP/CS, SpBa/CS, PeW/CS, PeH/CS, PpW/CS, PpH/CS, sqPDG and PLG/CS. Excluding single-specimen samples, a total of 79 samples with 194 worker specimens was available for analysis in these taxa. The EDA clustering was checked by running a controlling linear discriminant analysis (LDA) with species hypotheses allocated when the three EDAs agreed in classification but samples run as wild-cards when the EDAs classified ambiguously. The final species hypothesis established by the LDA determined the error on the sample level as 3.7% for NC-Ward and 6.2% for both NC-part.hclust and NC-NMDS.kmeans (
Figure 28). Running the LDA with the final species hypothesis assigned to all 194 workers specimens, the classification error on the individual level was 3.6%. Naming the clusters in
Figure 28 from left to right as clusters 1, 2 and 3 and running the specimens of the type series as wild-cards in the LDA, the following posterior probabilities were found on the sample level. The holotype series of
C. tjibodana was allocated to cluster 1 with
p = 0.9992. Allocation to cluster 2 was given with
p = 0.9989 in the holotype series of
C. minutior, with
p = 0.9799 in the holotype series of
C. tsukuyomi and with
p = 0.8254 in the holotype series of
C. breviscapus, hence the latter two taxa should represent junior synonyms of
C. minutior. Allocation to cluster 3 was established with
p = 0.9984 in the holotype of
C. goa and with
p = 0.9997 in the paratype series of
C. goa.
Figure 28.
Separation of nest samples of Cardiocondyla tjibodana (green bars), C. minutior (red bars) and C. goa (black bars) by NC-Ward, NC-part.hclust and NC-NMDS.kmeans clustering. White bars indicate outliers in NC-part.hclust. The mean classification error of the three exploratory data analyses relative to the controlling linear discriminant analysis is 5.4%. Fifteen phenotypic characters were considered.
Figure 28.
Separation of nest samples of Cardiocondyla tjibodana (green bars), C. minutior (red bars) and C. goa (black bars) by NC-Ward, NC-part.hclust and NC-NMDS.kmeans clustering. White bars indicate outliers in NC-part.hclust. The mean classification error of the three exploratory data analyses relative to the controlling linear discriminant analysis is 5.4%. Fifteen phenotypic characters were considered.
A mean error of the three EDAs on the sample level of 5.4% is larger than the threshold of 4% as it was recommended by Seifert [
8]. This might be a consequence of the small mean sample size (only 2.5 workers per supposed nest sample) and of a rather weak character system not sufficient to allow a better resolution. The phenotypical clustering is supported by differences in geographic distribution between the three taxa. Anyway, the critical error rate of the EDAs indicates a taxonomic conflict and the necessity to check the phenotypical clusters by an adequate system of nuclear DNA markers.
Biology. K. Yamauchi (pers. comm. 2001) reported for Okinawa nesting in shallow soil in open, disturbed areas with bare or weakly herbaceous ground. In Brazil, Tobago, and other areas, whereto the species has accidentally been introduced, nests can also be found in the soil, e.g., in open patches in gardens and plantations (Heinze pers. comm.). The Japanese population of
C. minutior is reported to have a karyotype of 2n = 30 [
26]. Both winged and ergatoid males occur. Winged males disperse, typically after mating within their natal colony. Ergatoid males are not known to disperse from their natal colony. Instead, they fight other ergatoid males, typically resulting in the death of all but one resident male [
27]. As any
Cardiocondyla species with tramp species potential,
C. minutior is not known to have significant ecological impacts and it seems unlikely that it will ever become a significant pest after further range expansion. Ward [
28] reported that
C. minutior was able to survive in sites dominated by the Argentine Ant
Linepithema humile.
4.4.2. Cardiocondyla goa Seifert, 2003
Cardiocondyla goa Seifert, 2003 [type investigation]
This taxon has been described from India. Investigated were one holotype worker labelled “Indien_02: Kerala, Periyar Sanctuary, vic. Thekkady, 20, 500–1000 m H, 02.05.01.1997, leg. A.Schulz, K.Vock”; three paratype workers labelled “Indien_02: Kerala, Periyar Sanctuary, vic. Thekkady, 12, 500–1000 m H, 02.05.01.1997, leg. A.Schulz, K.Vock”; depository SMN Görlitz.
All material examined. Including single-specimens samples with wild-card LDA classifications having posterior probabilities
p > 0.95, numeric phenotypical data were available in 26 samples (largely nest samples) with 57 workers. For details see
Supplementary Information Tables S1 and S2. This material originated from Saudi Arabia (5 samples), the Malagasy region (6), the Seychelles (2), India (5), Nepal (3) and Myanmar (5).
Geographic range. The verified findings given in the previous paragraph indicate a much smaller range than in C. minutior and a weaker tramp species potency. The available data suggest range borders along 23° S, 30° N, 40° E and 97° E and occurrence in strictly tropical climate. It was found in Nepal up to 1500 m.
Diagnosis: Worker (
Table 1,
Figure 3, key; picture CASENT0196173 in
www.antweb.org): Small size, CS 413 µm. Head elongated but less than in
C. minutior, CL/CW 1.239. Postocular distance very large, PoOc/CL 0.480. Scape short, SL/CS 0.760. Eye rather small, EYE/CS 0.230, with notable micropilae, the longest measuring 6–10 µm. Occipital margin straight or very weakly concave. Frontal carinae slightly converging immediately caudal of FRS level (FL/FR 1.088). Dorsal profile of mesosoma rather straight or weakly convex, metanotal groove only suggested or entirely absent (MGr/CS 0.55%). Spines shorter than in
C. minutior and rather acute (SP/CS 0.121), their axis in profile deviating by 40–45° from longitudinal axis of mesosoma. Petiole rather wide and high (PeW/CS 0.274, PeH/CS 0.336), in profile with concave anterior face and rounded node that is in dorsal view circular and as long as wide. Postpetiole rather wide and very low (PpW/CS 0.473, PpH/CS 0.263), its sternite completely flat, without any anteroventral bulge; in dorsal view with angulate-convex sides and straight anterior margin. Paramedian vertex with deeply impressed, flat-bottomed foveolae of 13–18 µm diameter in dense honey-comb arrangement (if not displaced by longitudinal carinulae). Foveolae with an inner corona (margin of a flat tubercle) of 7–8 µm diameter. Mesosoma on whole surface sculptured. Waist segments with shallower and finer reticulum than on mesosoma, nodes sometimes slightly shiny. First gaster tergite with fine microreticulum that may be obscured by polluted surfaces. Pubescence on whole body long and dense but shorter than in
C. minutior, PLG/CS 7.13%, sqPDG 3.23. Color of head, mesosoma, and waist varying considerably from dirty yellowish to dark dirty brown, gaster dark to blackish brown.
Taxonomic comments and clustering results. Considering character by character, there are only subtle morphological differences to
C. minutior and
C. tjibodana but a sufficient separation is possible by discriminant analysis (see
Section 4.4.1).
Biology. Colonies live in small nests in the ground, frequently in disturbed areas near paths or gardens. They contain multiple fertile queens as in the related species
C. minutior. Both ergatoid males and winged males have been observed for considerable time staying in the natal nest to mate with female sexuals and it was suggested that winged males have lost their function as an exclusive disperser morph [
29].
4.4.3. Cardiocondyla tjibodana Karavajev, 1935
Cardiocondyla tjibodana Karavajev, 1935 [type investigation]
This taxon has been described from Cibodas (Tjibodas)/Java. Investigated were two syntype workers labelled “Tjibodas, Java”, “5375. Coll. Karavaievi” and “Cardiocondyla tjibodana Karavaiev typ.”; depository SIZ Kiev. Comment: The two specimens fully match the description of Karavajev. However, the number of specimens is in disagreement with the original description that states: “…Tjibodas, W. Java, W. Karawajew, Nr.5375, 1 Arbeiter…” and “… Küste von Bantam, Java, an der Sundastrasse, gegenüber Meeuwen Eiland, 7.I.1913, Nr.5376, 1 Arbeiter…”. Sample No. 5376 was not seen. In case of its discovery and if representing a different species, a lectotype of C. tjibodana should be fixed in a specimen of sample 5375.
All material examined. Including single-specimens samples with wild-card LDA classifications having posterior probabilities
p > 0.95, numeric phenotypical data were available in 31 samples (largely nest samples) with 65 workers. For details see
Supplementary Information Tables S1 and S2. This material originated from Belize (1 sample), Saudi Arabia (2), India (1), Thailand (1) Malaysia (9), Indonesia (6), Philippines (5), Mariana Islands (2), Pohnpei and Solomon Islands (3), and Australia (1).
Geographic range. The bulk of findings is from the Indo-Malayan and Polynesian region. The remote findings from Belize and Saudi Arabia indicate tramp species properties but much less expressed than in C. minutior.
Diagnosis: Worker (
Table 1,
Figure 4, key; pictures CASENT0178362, CASENT0916972 (type) in
www.antweb.org): Small size, CS 406 µm. Head elongated, CL/CW 1.258. Postocular distance very large, PoOc/CL 0.480. Scape short, SL/CS 0.761. Eye slightly larger than in
C. minutior and
C. goa, small, EYE/CS 0.232, with notable micropilae, the longest measuring 6–10 µm. Occipital margin straight or very weakly concave. Frontal carinae slightly converging immediately caudal of FRS level (FL/FR 1.072). Dorsal profile of mesosoma rather straight or weakly convex, metanotal groove only suggested or entirely absent (MGr/CS 0.61%). Spines rather short and acute (SP/CS 0.132), their axis in profile deviating by 40–45° from longitudinal axis of mesosoma. Petiole slightly narrower than in
C. minutior and
C. goa but similarly high (PeW/CS 0.266, PeH/CS 0.337), in profile with concave anterior face and rounded node that is in dorsal view circular and as long as wide. Postpetiole significantly narrower than in
C. minutior and
C. goa but similarly high (PpW/CS 0.445, PpH/CS 0.261), its sternite completely flat, without any anteroventral bulge; in dorsal view with angulate-convex sides and straight anterior margin. Paramedian vertex with deeply impressed, flat-bottomed foveolae of 13–18 µm diameter in dense honey-comb arrangement (if not displaced by longitudinal carinulae). Foveolae with an inner corona (margin of a flat tubercle) of 7–8 µm diameter. Mesosoma on whole surface sculptured. Waist segments with shallower and finer reticulum than on mesosoma, nodes sometimes slightly shiny. First gaster tergite with fine microreticulum that may be obscured by polluted surfaces. Pubescence on whole body long and dense, PLG/CS 7.61%, sqPDG 3.17. Color of head, mesosoma, and waist varying considerably from dirty yellowish to dark dirty brown, gaster dark to blackish brown.
Taxonomic comments and clustering results. The differences in waist measures allow a rather good separation from
C. minutior and
C. goa (see
Section 4.4.1).
Biology. Unknown.
4.4.4. Cardiocondyla schulzi n.sp.
Etymology: Named after the collector Andreas Schulz.
Type material:
Holotype plus one paratype labelled “Indien_02: Kerala, Periyar Sanctuary, vic. Thekkady, 30, 500–1000 mH, 02.-05.1997, Leg. A. Schulz, K. Vock 30” and “Holotype (top) & paratype Cardiocondyla schulzi Seifert”; two paratype workers labelled “Indien_02: Kerala, Periyar Sanctuary, vic. Thekkady, 500–1000 mH, 02.-05.1997, Leg. A. Schulz, K. Vock X” and “Paratypes Cardiocondyla schulzi Seifert; both samples stored in SMN Görlitz.
All material examined. Only the two type samples were available, these are probably no nest samples but apparently from two different spots.
Geographic range. The collecting site in South India is situated at approximately 9.60° N, and 77.20° E.
Diagnosis: Worker (
Table 1,
Figure 5, key): Small size, CS 432 µm. Head much less elongated than in related species, CL/CW 1.187. Postocular distance large, PoOc/CL 0.463. Scape rather short, SL/CS 0.780. Eye medium sized, EYE/CS 0.228, with notable micropilae, the longest measuring 6–9 µm. Occipital margin weakly concave. Frontal carinae notably converging immediately caudal of FRS level (FL/FR 1.113). Dorsal profile of promesonotum rather straight, metanotal groove absent or barely visible (MGr/CS 0.11%). Spines short and triangular (SP/CS 0.113), their axis in profile deviating by 40–45° from longitudinal axis of mesosoma. Petiole narrow (PeW/CS 0.247), in profile much lower than in related species (PeH/CS 0.278), with straight to feebly concave anterior face and a flat convex node that is in dorsal view slightly longer than wide. Postpetiole significantly narrower and lower than in most related species except
C. opaca (PpW/CS 0.425, PpH/CS 0.240), its sternite completely flat, without anteroventral bulge; in dorsal view with convex sides and slightly concave anterior margin. Paramedian vertex with deeply impressed, flat-bottomed foveolae of 16–17 µm diameter in dense honey-comb arrangement. Foveolae with an inner corona (margin of a flat tubercle) of 7–8 µm diameter. Median vertex with few longitudinal carinulae. Mesosoma on whole surface sculptured. Waist segments with shallower and finer reticulum than on mesosoma, nodes sometimes slightly shiny. Basal part of first gaster tergite without or only barely visible microreticulum. Pubescence on whole body rather long and dense (PLG/CS 7.36%, sqPDG 3.76). Color of head, mesosoma, and waist concolorous dark brown.
Taxonomic comments and clustering results. Cardiocondyla schulzi n.sp. differs from C. goa, C. minutior and C. tjibodana by the very low CL/CW, PeH/CS and PeW/CS. With all measurements given in mm, it is clearly separable by a discriminant D(2) = 198.1∗PeH − 72.43∗CL + 9.013 with D(2) being −1.107 ± 0.478 [−1.780, −0.676] in four workers of C. schulzi n.sp. and 3.098 ± 1.006 [0.685, 5.959] in 210 individual workers of the other three species.
Biology. Unknown.
4.4.5. Cardiocondyla carbonaria Forel, 1907
Cardiocondyla carbonaria Forel, 1907 [type investigation]
Type material. This taxon has been described on at least two type specimens from India. Directly investigated was the type worker labelled by Forel “C.carbonaria, € type Forel\ India or. Biró 1902\Matheran 800 m”, MHN Genève. A type specimen with equal locality labels, stored in Hungarian Natural History Museum Budapest, was only examined on the basis of images in
www.antweb.org (CASENT0922222).
All material examined. Only the two type specimens are known.
Geographic range. The collecting site is east of Mumbai at approximately 18.99° N, 73.27° E and 770 m.
Diagnosis: Worker (
Table 1,
Figure 6, key; pictures CASENT0922222, CASENT0908337 in
www.antweb.org): Small size, CS 434 µm. Head much elongated, CL/CW 1.282. Postocular distance large, PoOc/CL 0.474. Scape moderately long, SL/CS 0.789. Eye large (EYE/CS 0.243), with notable micropilae, the longest measuring 15 µm. Occipital margin straight or very weakly concave. Frontal carinae nearly parallel immediately caudal of FRS level (FL/FR 1.010). Dorsal profile of mesosoma almost straight, with only weak metanotal depression (MGr/CS 0.90%). Spines in profile view acute, triangular in profile and of medium length (SP/CS 0.136), their axis in profile deviating by 40–45° from longitudinal axis of mesosoma, their bases more distant than in related species (SPBA/CS 0.306). Petiole wider and higher than on average observed in related species (PeW/CS 0.297, PeH/CS 0.337), in profile with rather a short peduncle, a concave frontal face and convex dorsal node profile; the node is in dorsal view as long as wide. Postpetiole wider than on average seen in related species but similarly low (PpW/CS 0.482, PpH/CS 0.265), its sternite very flat, anterolaterally with a short and curved costa on each side; in dorsal aspect distinctly wider than long, with convex sides and straight anterior margin. Clypeus posteriorly shagreened, with six longitudinal, curved carinae. Frontal lobes shagreenate-microfoveolate. Median area of vertex longitudinally carinulate, intercarinular spaces foveolate. Paramedian and lateral areas of vertex with deeply impressed, closely adjacent foveolae of 14–16 µm diameter that usually show a flat inner tubercle of 8–9 µm diameter. Dorsal area of mesosoma finely shagreenate-foveolate with few short, longitudinal microcarinae; lateral area of mesosoma densely reticulate. Petiole surface microreticulate-shagreened, the dorsum more shagreened. Exposed surfaces of tergites completely matt, very finely and densely shagreened. Pubescence on whole body long and dense, PLG/CS 8.30%, sqPDG 3.01. Whole body blackish brown except for light-yellowish antennae, trochanter, distal ends of femora, tibiae, and tarsi.
Taxonomic comments and clustering results. Though being in no morphometric character outside the range of extremes of Cardiocondyla goa, minutior and tjibodana, C. carbonaria clearly differs from these species as well as from C. schulzi n.sp. and C. britteni by a perfectly matt surface of head, mesosoma, waist and dorsum of gaster, caused by a very dense and fine microsculpture and shagrination. The best differences from C. opaca, which is rather similar in microsculpture, are the much larger SPBA/CS, PeW/CS, PpW/CS and PeH/CS. The unique microsculpture of the gaster tergites separate C. carbonaria and C. opaca from every species of the group.
Biology. Unknown.
4.4.6. Cardiocondyla opaca Seifert, 2003
Cardiocondyla opaca Seifert, 2003 [type investigation]
Type material. Holotype worker labelled “Indien_04: Goa, distr. Canacona, Cortigao Sanctuary, 100 mH, 06.-10.01.1997, leg. A.Schulz, K.Vock 33”, depository SMN Görlitz.
All material examined. Only the single type specimen is known.
Geographic range. The collecting site is in the Cotigao (Cortigao) Sanctuary/SW India at approximately 15.00° N, 74.15° E and 100 m.
Diagnosis: Worker (
Table 1,
Figure 7, key): Small size, CS 424 µm. Head much elongated, CL/CW 1.253. Scape much longer than in any other species of the
C. minutior group, SL/CS 0.836. Postocular distance large, PoOc/CL 0.468. Eye medium-sized, EYE 0.235. Eye setae well-developed, the longest 13 µm. Occipital margin and anterior clypeal margin slightly concave in median third. Frons very narrow, FRS/CS 0.221, frontal carinae immediately behind FRS level almost parallel (FL/FR 1.033). Dorsal profile of mesosoma rather straight, metanotal depression absent. Spines triangular and short (SP/CS 0.092); their bases more approached than in related species (SPBA/CS 0.254). Petiole low (PeH/CS 0.283), with a rather long peduncle, its dorsal profile evenly curved. Postpetiole narrower than in any related species except
C. parvinoda and very low (PpW/CS 0.409) PpH/CS 0.251); its sternite very flat, anterolaterally with a short, curved costa on each side; as long as wide, with angulate-convex sides and straight anterior margin. Head and dorsal mesosoma with striking pubescence, standing from body surface at angle of 25–30°. Whole body including gaster with perfectly matt surface, caused by a very dense and fine microsculpture of differing structure. Clypeus densely shagreened, with five longitudinal curved carinulae. Frontal lobes shagreened, in lateral parts with few short longitudinal carinulae. Frontal triangle narrow and smooth. Whole head densely longitudinally shagreened. On paramedian and lateral area of vertex, obscured within dense shagreen, foveolae of 12–13 µm diameter are present, that have an inner corona of 7 µm diameter; foveolar interspaces slightly smaller than foveolar diameter. Dorsal area of promesonotum with few foveolae of 8–9 µm diameter, obscured within dense shagreen. Remaining mesosoma fully and strongly microreticulate. Petiole coarsely microreticulate, its dorsum more shagreened. Postpetiole in dorsal aspect completely shagreened. Exposed surfaces of tergites completely mat, very finely and densely longitudinally shagreened; tergite pubescence long and dense (PLG/CS 7.47%, sqPDG 3.31). Whole body blackish except for light-brown antennae, trochanter, and distal ends of femora, and whitish yellow tibiae and tarsi.
Taxonomic comments. In addition to the most different microsculpture and surface appearance, Cardiocondyla opaca is clearly separate from C. goa, C. minutior and C. tjibodana by much larger SL/CS as well as much smaller PpW/CS and PeH/CS—all these data are outside the range of extremes in the former three species. Cardiocondyla opaca strongly differs from C. schulzi n.sp. by surface structure and much larger CL/CW and SL/CS and from C. carbonaria by much smaller SPBA/CS, PeW/CS, PpW/CS and PeH/CS.
Biology. Unknown.
4.4.7. Cardiocondyla britteni Crawley, 1920
Cardiocondyla britteni Crawley, 1920 [type investigation]
Type material. 1 type worker labelled “Cardiocondyla britteni Crawley” and “West Didsbury 12-5-1919 B.h.H. Butter Beans”, depository UM Oxford.
All material examined. Only the single type specimen is known.
Geographic range. The type locality is West Didsbury to where it has apparently been imported via the Liverpool harbor with “Butter Beans”. Crawley [
30] wrote that “… It is highly probable that the ant was imported with the beans, in which case its place of origin would be tropical America…” The vernacular name Butter Beans is ambiguous but is most frequently referred to
Lablab purpureus that has is natural origin most probably in SE Africa but its main cultivation areas in India [
31]. As no species of the
C. minutior group has been found so far to be native in America or Africa, there is a good probability that the geographic origin of
C. britteni is India or the Oriental region.
Diagnosis: Worker (
Table 1,
Figure 8, key; picture CASENT0901496 in
www.antweb.org): Small size, CS 402 µm. Head much elongated CL/CW 1.249. Scape rather short, SL/CS 0.741. Postocular distance very large, PoOc/CL 0.482. Anterior clypeal margin convex. Occipital margin weakly concave. Eye small, EYE 0.226, with micropilae sparser and shorter than in
C. minutior. Dorsal profile of mesosoma evenly convex and metanotal groove entirely absent. Spines very short (SP/CS 0.089), almost triangular and rather blunt. Petiole rather narrow (PeW/CS 0.268), in profile with a concave frontal face and convex node dorsum; the node in dorsal view almost globular. Postpetiole rather wide (PpW/CS 0.473) and moderately low (PpH/CS 0.272), its sternite very flat, without any prominent structures and in dorsal view with straight frontal margin and angulate-convex sides. Pubescence on whole body shorter and less dense than in
C. minutior (PLF/CS 6.61%, sqPDG 4.25). Clypeus smooth. Frontal laminae and a small area caudal of them finely longitudinally carinulate. Dorsum of vertex, mesosoma, and waist in overall impression distinctly shiny. Vertex with very shallow and small foveolae of only 4–10 µm diameter; foveolar interspaces brilliantly shiny and much wider than foveolar diameter, occasionally with fragments of a very fine perifoveolar microreticulum. Surface structure of dorsal promesonotum similar to vertex. Whole propodeum as well as meso- and metapleurae weakly reticulate. Lateral area of pronotum smooth, finely microreticulate. Petiolar and postpetiolar nodes shiny, with very fine microreticulum. Dorsum of gaster smooth and with fragments of very fine microreticulum. Antennal club, gaster, propodeum, meta- and mesopleurae dark to blackish brown. Vertex, promesonotum, waist, and appendages light-yellowish brown.
Taxonomic comments. By morphometry and body shape, Cardiocondyla britteni is undoubtedly a member of the C. minutior group. It differs from all other species of this group generally by the reduced sculpture on all body surfaces and in detail by the much smaller and shallower foveolae on vertex, the shiny head and dorsal areas of mesosoma, the shorter spines, and the larger sqPDG.
Biology. Unknown.
4.4.8. Cardiocondyla parvinoda Forel, 1902
Cardiocondyla parvinoda Forel, 1902 [type investigation]
Type material. The taxon has been described from Poona/India. Investigated were 2 syntype workers labelled “C. parvinoda, Forel type, Poona (Wroughton) XXIII/13”, MHN Genève; 1 syntype worker labelled “C. parvinoda, type Forel, Poona XXIII/13 (Wroughton)”, NHM Basel. 1 worker syntype labelled “Poona, Wroughton, C. parvinoda Forel type”, NHM Wien. 1 worker syntype labelled by Stitz “Paona, Wroughton” and “Cardiocondyla parvinoda For.”, ZM Berlin.
All material examined. Investigated were 6 samples (largely nest samples) with 17 workers. For details see
Supplementary Information Tables S1 and S2. This material originated from India (1 sample), Thailand (4) and the Philippines (1). An additional specimen from Calcutta/India, 1902, coll. Bingham, depicted in
www.antweb.org under CASENT0281807, was confirmed of belonging to
C. parvinoda by photo evaluation.
Geographic range. So far known only from the north of the Oriental region between 14 and 23°N and 73 and 121°E. It was found from the lowlands up to about 1300 m.
Diagnosis: Worker (
Table 1,
Figure 9, key; pictures CASENT0908346 and CASENT0901496 in
www.antweb.org): By far the largest species of the
C. minutior group, CS 501 µm. Head comparatively short CL/CW 1.182. Postocular distance rather small, PoOc/CL 0.468. Scape much longer than in most species of the
C. minutior group, SL/CS 0.831. Eyes small and with numerous micropilae of 13 µm maximum length, EYE/CS 0.219. Occipital margin straight. Anterior clypeal margin concave, clypeal sides each with 2–3 longitudinal rugae or costae that are stronger than in other species. Frons rather wide FRS/CS 0.241 and frontal carinae more diverging frontad than in related species, FL/FR 1.122. Dorsal profile of mesosoma rather straight, metanotal depression absent or weak MGr/CS 1.25%. Spines triangular and sharp, the axis in lateral view deviating from longitudinal axis of mesosoma by about 50°, SP/CS 0.124; spine bases more approached, SPBA/CS 0.263. Petiole rather low and narrow (PeH/CS 0.305, PeW/CS 0.256), with a rather long peduncle, dorsal node profile convex with a straight central portion. Postpetiole narrower than in any related species (PpW/CS 0.402) and extremely low (PpH/CS 0.230); postpetiolar sternite completely flat, without any prominent structures. Frontal laminae and anteromedian vertex longitudinally carinulate. Lateral vertex with deeply impressed, densely-packed, circular foveolae of 19–20 µm mean diameter which show a inner corona of 9–11 µm diameter. Dorsal mesosoma very shiny, but with scattered and shallow foveolae and fine irregular rugosity. Lateral mesonotum and lateral propodeum longitudinally carinulate. Meso- and metapleurae more strongly and densely longitudinally carinulate-rugulose. Both waist segments glabrous except for small hair base pits. Tergite pubescence long and rather dense, PLG/CS 8.08, sqPDG 3.62. Color variable. Dorsal head yellowish brown to dark brown; mesosoma and petiole yellow, yellowish brown or dark brown; postpetiole slightly darker; gaster dark brown.
Taxonomic comments. With exception of its larger size Cardiocondyla parvinoda shows any character typical for the C. minutior group. The combination of extremely small postpetiole measurements with a moderately elongated head, short spines and the shiny dorsal mesosoma allows a separation from any known species worldwide.
Biology. Unknown.
4.4.9. Cardiocondyla argentea n.sp.
Etymology: from argentum (Lat.) silver; named because of the silvery shine of the flattened cephalic and mesosomal pubescence hairs.
Type material:
Holotype worker and 5 paratype workers labelled “Banggai Arch., Potil Kecil 1°28′ S–123°34′ E 12–19.ii.1980\M.J.D.Brendell B.M. 1980-280”, BMNH London; 2 worker and 1 gyne paratype with identic labelling, SMN Görlitz; 3 worker paratypes labelled “INDONESIA: N.C.Seram\Manusela N.P., Wae Mual Plain 25.vii-9.ix.1987\Op. Raleigh M.J.D.Brendell. B.M. 1987-262”, BMNH London; 3 paratype workers labelled “Indonesia-05, Sulawesi, Prov. Minasha, Dumoga Bone N.P., E-side, vic. Duluduo, 0°35′ N 124°54′ E, 100 mH, 04.-09.12.2000, Leg. A Schulz”, SMN Görlitz.
Geographic range. So far only found east of the Wallace Line in North and Central Sulawesi as well as Maluku from sea level to 800 m.
Diagnosis: Worker (
Figure 10,
Table 2): Medium-sized, CS 491 µm. Head extremely short, CL/CW 1.051. Postocular distance rather small, PoOc 0.420. Scape moderately long, SL/CS 0.826. Eye small, EYE 0.211. Anterior clypeal margin between the level of frontal carinae concave. Occipital margin slightly concave or straight. Frontal carinae converging immediately posterior of the FRS level, FRS/CS 0.311. Mesosoma extremely thickset and short; its length without neck shield only 1.1 of CL. Dorsal profile of mesosoma hump-backed, strongly and evenly convex, without any metanotal groove. Spines of medium length (SP/CS 0.258), acute, in lateral view feebly upcurved and their axis deviating by 25° from longitudinal axis of mesosoma, spine bases widely distant, SPBA/CS 0.392. Petiole in lateral view high (PeH/CS 0.335), with a short peduncle a concave frontal and convex caudodorsal profile; the node in dorsal view wider than long, PeW/CS 0.300. Postpetiole in dorsal view narrow (PpW/CS 0.413), extremely short and with a concave anterior margin; in lateral view very low (PpH/CS 0.254), with a semicircular ventral profile that is produced by prominent bilateral lobes of the sternite which strongly protrude compared to median level. Whole surface of head and mesosoma densely foveolate-microreticulate, thus appearing at lower magnifications perfectly matt. The foveolae on vertex and dorsal pronotum are larger with a foveolar diameter of 11–15 µm but are not salient because of partial covering by pubescence. Petiole and postpetiole microreticulate and matt; postpetiole in the holotype series mildly shiny and weakly microcorrugated. First gaster tergite shiny, with a delicate, fragmentary microreticulum. Cephalic and mesosomal pubescence hairs flattened and appearing silvery (
Figure 10D), their larger diameter 4 µm (normal in
Cardiocondyla are 1.7–2 µm), the smaller diameter about 2.5 µm. Gastral pubescence not flatted, rather long and dense (PLG/CS 6.08%, sqPDG 4.12). Whole body rather concolorous dark to medium brown.
Taxonomic comments. There are three similar Indo-Malayan species sharing a silvery pubescence, a thickset, hump-backed mesosoma and an extremely low postpetiole with prominent bilateral sternal lobes:
Cardiocondyla argentea n.sp.,
Cardiocondyla semiargentea n.sp. and
Cardiocondyla argyrotricha n.sp. The former two species occur east of the Wallace Line and the latter one west of it. These species can be separated by a PCA and LDA. The PCA was run considering the 15 characters CS, CL/CW, SL/CS, SPBA/CS, PeW/CS, PpW/CS, SP/CS, FRS/CS, EYE/CS, PeH/CS, PpH/CS, sqPDG, PLG/CS, PoOc/CL and MpGr. The first three components of this PCA where then used as variables in a LDA in order to avoid an overfitting of the discriminant function due to low class size (only 11 specimens in the smallest class). Both the LDA and the leave-one-out cross-validation LDA confirmed all 47 species hypotheses (
Figure 29).
Biology. Unknown but the biology should be, at least regarding male morphology and behavior, similar to the situation in the well studied, closely related
Cardiocondyla argyrotricha n.sp. (see
Section 4.4.11).
Figure 29.
Separation of worker individuals of Cardiocondyla argentea n.sp. (black rhombs), C. argyrotricha n.sp. (white squares) and C. semiargentea n.sp. (white dots) by a linear discriminant analysis (LDA). In order to avoid character overfitting, the LDA was run using the first three components of a principal component analysis considering 15 morphometric characters.
Figure 29.
Separation of worker individuals of Cardiocondyla argentea n.sp. (black rhombs), C. argyrotricha n.sp. (white squares) and C. semiargentea n.sp. (white dots) by a linear discriminant analysis (LDA). In order to avoid character overfitting, the LDA was run using the first three components of a principal component analysis considering 15 morphometric characters.
4.4.10. Cardiocondyla semiargentea n.sp.
Etymology: from “semi” (Lat.) partly and “argentum” (Lat.) silver); because of the less developed silvery pubescence on head and mesosoma compared to Cardiocondyla argentea n.sp.
Type material. Holotype worker labelled “INDONESIA: SULAWESI UTARA, Danau Mooat, 1200 m\nr Kotamobagu 23. x. 1985”, BMNH London; 2 worker paratypes with identic labelling, SMN Goerlitz; 1 paratype worker labelled “Indonesia_04, Sulawesi, Prov. Minasha, vic. Gunung Ambang, Donau Moaat, 0°44′ N, 124°27′ E, 800–1000 mH, 02.12.2000 543 Leg. A.Schulz”, SMN Görlitz; 2 worker paratypes labelled “INDONESIA: SULAWESI UTARA, Dumoga-Bone N.P. 9–16 May 1985”, BMNH London; 1 worker paratype with identic labelling, SMN Görlitz. 1 worker paratype labelled “INDONESIA: SULAWESI UTARA, Dumoga-Bone N.P.\Fog 5 400 m. 11.ii.85 BMNH Plot C”, BMNH London.
Geographic range. So far only found east of the Wallace Line in North Sulawesi from 385 to 1150 m above sea level.
Diagnosis: Worker (
Figure 11,
Table 2): Smaller than
C. argentea n.sp., with much longer head and larger postocular distance, CS 439 µm, CL/CW 1.131, PoOc/CL 0.436. Scape moderately long, SL/CS 0.830. Eye larger than in
C. argentea, EYE 0.225. Anterior clypeal margin between the level of frontal carinae rather straight or slightly concave. Occipital margin slightly concave. Frontal carinae more approached than in
C. argentea n.sp., FRS/CS 0.274, and immediately posterior of the FRS level slightly converging. Mesosoma extremely short; its length without neck shield only 1.04 of CL. Dorsal profile of mesosoma strongly and evenly convex. Spines of slightly longer than in
C. argentea n.sp. (SP/CS 0.273), acute, their bases more approached (SPBA/CS 0.362) than in
C. argentea n.sp.; in lateral view straight and their axis deviating by 20° from longitudinal axis of mesosoma. Petiole rather high and moderately wide (PeH/CS 0.332, PeW/CS 0.290); in lateral view in lateral view with a short peduncle, a rather high node, a concave frontal and convex caudodorsal profile; the node in dorsal view as wide or slightly wider than long. Postpetiole narrow, very low (PpW/CS 0.422, PpH/CS 0.258) and extremely short; in dorsal view with a strongly concave anterior margin; in lateral view with a semicircular ventral profile that is produced by prominent bilateral lobes of the sternite which strongly protrude compared to median level (
Figure 11D). Whole surface of head and mesosoma densely foveolate-microreticulate, thus appearing at lower magnifications perfectly matt. Foveolae on vertex and dorsal pronotum with 11–15 µm diameter. Petiole microreticulate and matt; dorsal postpetiole mildly shiny and weakly microreticulate. First gaster tergite smooth and shiny without a microreticulum. Cephalic and mesosomal pubescence hairs silvery but, in contrast to
C. argentea n.sp., not clearly flattened. Gastral pubescence rather long and dense (PLG/CS 5.87%, sqPDG 4.10). Strong color polymorphism. The darkest morph with whole body dark to medium brown, petiole slightly lighter with yellowish tinge. Intermediate morph with head and mesosoma dark to medium brown but spine tips, petiole, frontal laminae and clypeus contrastingly yellowish. The extremely bicolored morph has head and antennae light yellow; tibiae, tarsae, spine tips, petiole and postpetiole yellowish white; mesosoma blackish, femora and gaster blackish brown.
Taxonomic comments. The strikingly bicolored morph shows no obvious structural differences to the dark and intermediate morphs and is considered here as a mutant. For separation from the closely related species
C. argentea n.sp. and
C. argyrotricha n.sp. see
Section 4.4.9.
Biology. Unknown but the biology should be, at least regarding male morphology and behavior, similar to the situation in the well studied, closely related
Cardiocondyla argyrotricha n.sp. (see
Section 4.4.11).
4.4.11. Cardiocondyla argyrotricha n.sp.
Etymology: from “άργυρος“ (Greek) silver and “τρίχα“ (Greek) hair; because of the silvery pubescence on head and mesosoma.
Type material:
Holotype worker plus three paratype workers labelled “MAL: 1.7294° N, 110.4472° E, Baku NP Strand, 0 m, Frohschammer 2007.05.24–16”; four paratype workers labelled “MAL: 1.7294° N, 110.4472° E, Baku NP Strand, 0 m, in Stein, Frohschammer 2007.05.24-18”; four paratype workers labelled “MAL: 1.7294° N, 110.4472° E, Baku NP Strand, 0 m, in Stein, Frohschammer 2007.05.24–19”; all deposited in SMN Görlitz.
All material examined. Numeric phenotypical data were available in 11 samples with 23 workers. The material originated from Malaysia (8 samples) and the Philippines (3). For details see
Supplementary Information Tables S1 and S2.
Geographic range. So far known from six localities west of the Wallace Line on Tioman island, Borneo and Luzon from sea level up to 380 m.
Diagnosis: Worker (
Figure 12, pictures CASENT0280593 in
www.antweb.org;
Table 2): Largest species of the
C. argentea group, CS 512 µm. Compared to
C. argentea n.sp., head much more elongated (CL/CW 1.133) but postocular distance smaller (PoOc/CL 0.408). Scape longer than in any related species, SL/CS 0.859. Eye small, EYE 0.216. In position with CL in visual plane, anterior clypeal margin between the level of frontal carinae straight to slightly concave and occipital margin straight or very slightly concave. Frontal carinae wide, slightly converging immediately posterior of the FRS level, FRS/CS 0.302. Mesosoma thickset and short; its length without neck shield only 1.1 of CL. Dorsal profile of mesosoma hump-backed, strongly and evenly convex, without any metanotal groove. Spines rather long (SP/CS 0.267), acute, their axis in lateral view deviating by 30° from longitudinal axis of mesosoma, spine bases widely distant, SPBA/CS 0.369. Petiole in lateral view high (PeH/CS 0.336), with a short peduncle, a concave frontal and convex caudodorsal profile; the node in dorsal view wider than long, PeW/CS 0.303. Postpetiole in dorsal view narrow (PpW/CS 0.423), extremely short and with a straight or slightly concave anterior margin; in lateral view very low (PpH/CS 0.252), with a semicircular ventral profile that is produced by prominent bilateral lobes of the sternite which strongly protrude compared to median level (
Figure 12D). Whole surface of head and mesosoma densely foveolate-reticulate, thus appearing at lower magnifications perfectly matt. Foveolae on vertex or dorsal mesosoma badly visible due to superimposed pubescence and disguising inner structure that is reminiscent of a tree- or four-leafed clover leaf, foveolar diameter on vertex 11–15 µm. Petiole and postpetiole foveolate-microreticulate and matt. First gaster tergite shiny, with a delicate, fragmentary microreticulum. Cephalic and mesosomal pubescence hairs flattened and appearing silvery, their larger diameter about 4 µm, the smaller one about 2.5 µm. Gastral pubescence not flatted, rather long and dense (PLG/CS 6.05%, sqPDG 3.99). Whole body rather concolorous dark to medium brown.
Taxonomic comments. For separation from the closely related species
C. argentea n.sp. and
C. semiargentea n.sp. see
Section 4.4.9.
Biology. Schmidt et al. [
32], Frohschammer [
33] and Schmidt & Heinze [
34] reported the species to be functionally monogynous, to show intranidal mating, and to develop only wingless ergatoid males with sickle-shaped mandibles which fight to monopolize mating. Once the dominant queen has died, the first eclosing gyne will mate and prevent younger gynes from becoming fertile [
35]. The mated gynes perform short-range dispersal on the ground or more rarely a wider dispersal on the wing. As a minimalistic colony founding strategy, an uninseminated gyne taking along some workers can establish a normal colony by mating with the first male, developing from her own haploid eggs [
32].
4.4.12. Cardiocondyla latifrons n.sp.*
Etymology: from Latin, meaning broad frons, because of the widely distant frontal carinae.
* The name “
Cardiocondyla latifrons” was first published by Oettler et al. [
1] and Heinze & Weber [
36]. According to the provisions of ICZN, this name was not made available with the date of these publications.
Type material. Holotype worker and 2 worker paratypes labelled “MALAYSIA Neg.Sembilan Pasoh For.Res. xi.1994\M.Brendell K.Jackson S.Lewis”, SMN Görlitz. 3 worker paratypes labelled “MALAYSIA: Ulu Gombak 2000.06.02-M3 coll. Yamauchi”, BMNH London; 2 paratypes with same labelling in SMN Görlitz. 6 worker, 3 gyne, 2 ergatoid male paratypes labelled “MALAYSIA: Ulu Gombak 2000.06.03-1 K.Yamauchi” and “MALAYSIA: Ulu Gombak 2000.06.03-2 K.Yamauchi”, SMN Görlitz. 4 workers and 1 gyne paratypes labelled “Tacloban, Leyte, P.I. XII-10-44 E.S. Ross”, CAS San Francisco.
All material examined. Numeric phenotypical data were available in 31 samples with 61 workers. The material originated from Indonesia (2 samples), Malaysia (15), the Philippines (4), Singapore (1) and Thailand (9). For details see
Supplementary Information Tables S1 and S2.
Geographic range. With exception of a site in North Sulawesi only known from west of the Wallace Line. Widely distributed and abundant over Indochina, Malaysia, Indonesia and the Philippines from sea level up to 1000 m.
Diagnosis: Worker (
Figure 13, images of paratype specimen CASENT0103274 in
www.antweb.org,
Table 2): Very small, CS 410 µm. Head short (CL/CW 1.102), with notably concave occipital margin. Postocular distance relatively small, PoOc/CL 0.414. Scape short SL/CS 0.822. Eye rather small, EYE/CS 0.230. In position with CL in visual plane, anterior clypeal margin between the level of frontal carinae strongly concave and not forming a sharp edge; instead this concavity appears in frontal view as a small area, the lower margin of which is marked by the three clypeal macrosetae. In dorsal view, the lower margin is usually concealed under the upper margin that is the anterior reference point to measure CL. Frontal carinae very widely separated (FRS/CS 0.337) and almost parallel immediately posterior of the FRS level. Dorsal mesosomal profile strongly convexly vaulted but with a contrasting small plateau on anterior pronotal dorsum. Pronotal corners in dorsal view weak. Spines very long (SP/CS 0.400, in dorsal view slightly diverging, in lateral view very feebly downcurved and their axis deviating by 25° from longitudinal axis of mesosoma. Petiole in lateral view very high (PeH/CS 0.363), with a short peduncle and a long, convex dorsal node profile that steeply slopes down to the caudal cylinder; petiole in dorsal view rather wide (PeW/CS0.319), the node slightly longer than wide, narrowing frontad. Postpetiole in dorsal view moderately wide (PpW/CS 0.460), and with a concave anterior margin; in lateral view comparatively high (PeH/CS 0.363), its sternite with pronounced anterolateral corners that are formed by bilateral lobes which strongly protrude compared to anteromedian level. Whole surface of head and mesosoma densely foveolate, thus appearing at lower magnifications perfectly matt. Vertex with deep, bicoronate foveolae of 15–20 µm diameter which usually show an inner structure reminiscent of a three- or four-leafed cloverleaf (
Figure 13D); such foveolae are on mesosoma restricted to pronotum and dorsal mesonotum, the foveolae on remaining mesosoma and waist are smaller and usually without cloverleaf-structure. First gaster tergite smooth and shiny and with a delicate microreticulum. Pubescence on 1st gaster tergite short and dilute (PLG/CS 4.56%, sqPDG 5.17), in anterior part directed caudad and longer than on central surface where the shorter hairs continuously change direction to caudomediad or mediad. Rather concolorous dirty to blackish brown.
Taxonomic comments.
Cardiocondyla latifrons n.sp. and
C. micropila n.sp. (see
Section 4.4.13) are hypothesized here as different, but closely related species. This stands in contrast to the view of Yamauchi et al. [
12] who considered them as intraspecific color morphs because of fertile hybridization under laboratory conditions and only weak fitness reduction in crosses of F1 hybrid gynes with F1 hybrid males. Yet, fertile hybridization under arranged, artificial conditions is no ultimate indication for conspecificity. Using this criterion, many well-recognized species of different eumethazoan groups would have to be synonymized [
8]. Deciding is what happens in the natural context. There are four arguments to treat them as different species.
(1) The separation of the two taxa based on the 5 morphometric characters CS, sqPDG, PLG/CS, EYE/CS and SPBA/CS is very strong. The exploratory data analyses NC-Ward, NC-part.hclust, NC-part.kmeans and NC-NMDS.kmeans as well as the PCA separated the 65 nest samples of the two taxa with an error of 0% (
Figure 30). Furthermore, the classification of the 131 worker individuals using the same character set was confirmed by both a LDA and LOOCV-LDA with 99.2%.
Figure 30.
Separation of 65 nest samples of Cardiocondyla micropila n.sp. (black bars) and C. latifrons n.sp. (grey bars) by NC-Ward, NC-part.hclust, NC-part.kmeans, NC-NMDS.kmeans clustering and by a principal component analysis considering the morphometric characters CS, sqPDG, PLG/CS, EYE/CS and SPBA/CS.
Figure 30.
Separation of 65 nest samples of Cardiocondyla micropila n.sp. (black bars) and C. latifrons n.sp. (grey bars) by NC-Ward, NC-part.hclust, NC-part.kmeans, NC-NMDS.kmeans clustering and by a principal component analysis considering the morphometric characters CS, sqPDG, PLG/CS, EYE/CS and SPBA/CS.
(2) This separation by five morphometric characters is strongly supported by clear differences in pigmentation of the 61 and 70 worker individuals of
C. latifrons n.sp. and
C. micropila n.sp. (
Figure 31). If considering the two taxa as intraspecific color morphs with dominant–recessive inheritance, we have to demand that genes directing coloration form a linkage group with genes directing body shape and pubescence characters. This appears unlikely at the first hand but is not impossible and should be checked by whole genome analyses.
Figure 31.
Separation of 131 worker individuals of Cardiocondyla micropila n.sp. (white dots) and C. latifrons n.sp. (black dots) by a linear discriminant considering the morphometric characters CS, sqPDG, PLG/CS, EYE/CS and SPBA/CS and the sum of pigmentation scores of head (PigCap) and mesosoma (PigMes). The color chart upper left visualizes the scores.
Figure 31.
Separation of 131 worker individuals of Cardiocondyla micropila n.sp. (white dots) and C. latifrons n.sp. (black dots) by a linear discriminant considering the morphometric characters CS, sqPDG, PLG/CS, EYE/CS and SPBA/CS and the sum of pigmentation scores of head (PigCap) and mesosoma (PigMes). The color chart upper left visualizes the scores.
(3) Syntopic occurrence of both taxa with nesting at the same spot a few hand spans apart has been repeatedly observed by several observers. Performing a very thorough sampling, Sabine Frohschammer collected 19 samples of C. latifrons n.sp. and 57 of C. micropila n.sp. in six localities and reported the workers within the nest populations to be either “all blackish” or “all yellow” (data file provided by Frohschammer in 2008).
(4) Both species are polygynous and mate exclusively intranidally. The number of queens in four localities collected by S. Frohschammer was 3.06 ± 4.41 [0, 27] n = 64 in C. latifrons n.sp. and 6.95 ± 7.40 [0, 34] n = 61 in C. micropila n.sp. If really belonging to the same species, there would be a high probability in a polygynous species to accept mated gynes of both color morphs, to develop nests with mixed populations of yellow and blackish ants and then to produce hybrids by intranidal mating. Whereas F1 hybrids in Yamuchi’s crossbreeding experiments were in fact mainly yellowish (indicating dominant inheritance of yellow over black), there were many specimens among the F2 hybrids with mixed coloration—i.e., head and mesosoma yellowish but gaster blackish. However, there were no mixed nest populations or distinctly bicolored individuals observed in more than 150 nest samples collected by six different collectors throughout the Indo-Malayan region.
In conclusion, both taxa behave like good species which should rarely or never hybridize when embedded in a natural context. Acceptance of mated
C. micropila n.sp. or
C. latifrons n.sp. gynes in nests of the other species is apparently inhibited—probably because the recognition cues (cuticular hydrocarbons) are mutually exclusive and the invading heterospecific gynes cannot overcome the defense mechanisms of the worker force. The case appears to be a good example that strong prezygotic isolation may impair the development of postzygotic isolation. The ant genus
Myrmoxenus Ruzsky 1902 is another example with strong prezygotic isolation in nature and at least partially successful hybridization in captivity [
37,
38]. An extreme situation is found in the bird family Anatidae in which highly discriminative mate recognition by optical and behavioral signals and a long period of pair formation before the onset of oviposition result in a strong prezygotic isolation in nature whereas fertile interspecific crossings are easily achieved in captivity even across genus borders [
39,
40].
Biology.
C. latifrons n.sp. is one of the most abundant species of the
C. argentea group and often found in disturbed areas, in ecotones such as wood margins but also in primary and secondary forest. Nests have been discovered in rock crevices, under stones or the bark of fallen trees. It is polygynous and shows intranidal mating, development of only wingless ergatoid males with sickle-shaped mandibles which fight to monopolize mating [
12]. In small colonies without a mature queen, Heinze & Weber [
36] observed that virgin gynes violently fought for inheritance of the nest site and its workers and about 50% of the young queens were killed by queen aggression and subsequent attacks by workers. This represents a novel case of direct killing of unmated sisters or half sisters in ants.
4.4.13. Cardiocondyla micropila n.sp.*
Etymology: from micro (Lat.) meaning small and pilus (Lat.) meaning hair, because of the very short and thin pubescence.
* Following an earlier provisional naming in the author’s data files, this species has been named by other authors in two non-taxonomic, sociobiological papers as “
Cardiocondyla microseta” [
1,
34]. According to the provisions of ICZN, this name was not made available with these publications. This provisional naming was later changed by me to “micropila” because “microseta” would have meant “small strong hairs” or “small bristles” which is not adequate. A return to “microseta” would also mean the expense of changing specimen labels in two remotely-housed collections.
Type material. Holotype worker (on separate pin) and 3 paratype workers labelled “MAL: 2.7825° N, 104.1309° E, Tioman, Gunung Kajang, 200 m, Primärwald, in Stein, Frohschammer 2007.06.06-118”, depository SMN Görlitz; 3 paratype workers labelled “MAL: 2.7786° N, 104.1278° E, Tioman, Primärwald, in Stein, Frohschammer 2007.06.03-99”, depository SMN Görlitz.
All material examined. Numeric phenotypical data were available in 34 samples with 70 workers. The material originated from Brunei (5 samples), Malaysia (19), the Philippines (7), Singapore (1) and Thailand (2). For details see
Supplementary Information Tables S1 and S2.
Geographic range. Only known from west of the Wallace Line and widely distributed and abundant over Indochina, Malaysia and the Philippines from sea level up to 1000 m. In contrast to C. latifrons n.sp. so far not known from Java and Sulawesi and much less abundant than the latter species in Thailand.
Diagnosis: Worker (
Figure 14,
Table 2): Very small, CS 395 µm. Head shorter than in
C. latifrons n.sp. (CL/CW 1.120), with notably concave occipital margin. Postocular distance relatively small, PoOc/CL 0.423. Scape short SL/CS 0.826. Eye rather small, EYE/CS 0.232. In position with CL in visual plane, anterior clypeal margin between the level of frontal carinae strongly concave and not forming a sharp edge; instead this concavity appears in frontal view as a small area, the lower margin of which is marked by the three clypeal macrosetae. In dorsal view, the lower margin is usually concealed under the upper margin that is the anterior reference point to measure CL. Occipital margin notably concave. Frontal carinae very widely separated (FRS/CS 0.331) and almost parallel immediately posterior of the FRS level. Dorsal mesosomal profile strongly convexly vaulted but with a contrasting small plateau on anterior pronotal dorsum. Pronotal corners in dorsal view weak. Spines very long (SP/CS 0.397), in dorsal view slightly diverging, in lateral view straight or slightly downcurved, their axis deviating by 30–35° from longitudinal axis of mesosoma. Petiole rather wide (PeW/CS 0.310), its node in dorsal view as long or slightly longer than wide, narrowing frontad. Petiole in lateral view with a short peduncle and a convex dorsal node profile that steeply slopes down to the caudal cylinder and high (PeH/CS 0.362). Postpetiole in dorsal view wider than in
C. latifrons n.sp. (PpW/CS 0.460) and with a concave anterior margin; in lateral view higher than in
C. latifrons n.sp. (PpH/CS 0.326), its sternite with pronounced anterolateral corners that are formed by bilateral lobes which strongly protrude compared to anteromedian level. Whole dorsum of head and mesosoma foveolate-reticulate, thus appearing at lower magnifications perfectly matt. Vertex, with deep foveolae of 15–20 µm diameter which often show an inner structure reminiscent of a three- or four-leafed cloverleaf (
Figure 14D); such foveolae are on mesosoma restricted to pronotum and dorsal mesonotum, the foveolae on remaining mesosoma and waist are smaller and usually without cloverleaf-structure. Postpetiole less strongly sculptured, frequently rather shiny. First gaster tergite smooth and shiny and with a delicate microreticulum. Pubescence on 1st gaster tergite much shorter than in
C. latifrons n.sp. (PLG/CS 3.18, sqPDG 6.28). All body parts concolorous light yellowish or orange.
Taxonomic comments. For separation from
Cardiocondyla latifrons n.sp. see
Section 4.4.12.
Biology. Habitat selection, overall abundance and social structure correspond to the situation in C. latifrons n.sp. Queen-queen aggression, male behavior and morphology are also similar. According to the data of Frohschammer, the number of queens in the polygynous nests is higher than in C. latifrons n.sp.
4.4.14. Cardiocondyla pirata Seifert & Frohschammer, 2013
Cardiocondyla pirata Seifert & Frohschammer, 2013 [type investigation]
Type material. The taxon has been described from Los Banos/Philippines. Investigated were the holotype worker labelled “PHI: 14.1643° N, 121.2375° E, Los Banos, University Park, 58 m, Hortarium, Frohschammer 2008.07.23 #39”; 4 paratype workers, 3 dealate gynes and 1 ergatoid male from the holotype nest labelled “PHI: 14.1643° N, 121.2375° E, Los Banos, University Park, 58 m, Hortarium, Frohschammer 2008.07.23 #39”; 3 paratype workers labeled “PHI: 14.1643° N, 121.2375° E, Los Banos, University Park, 58 m, in hole of a stone at riverside, Frohschammer 2008.07.23 #32”; all this material in SMN Görlitz.
Geographic range. Only known from the type locality.
Diagnosis: Worker (
Figure 15,
Table 2): Very small size, CS 397 µm. Head rather long (CL/CW 1.132), with notably concave occipital margin. Postocular distance relatively small, PoOc/CL 0.408. Scape very short, SL/CS 0.807. Eye rather small, EYE/CS 0.238. With maximum CL and CW in visual plane, outlines of head roughly heart-shaped, with strongly concave posterior margin and an almost straight anterior clypeal margin (a distinct concavity appears after a tilt to frontodorsal viewing position when the three clypeal macrosetae become fully visible). Frontal carinae much less distant than in any related species (FRS/CS 0.242), subparallel and slightly diverging frontal of the FRS level. Mesosoma thickset, its dorsal profile evenly convex. Anterior pronotum in dorsal view rounded, without pronounced corners. Propodeal spines much shorter than in any related species (SP/CS 0.208), in dorsal view slightly diverging, in lateral view straight and with their axis deviating by 40° from longitudinal axis of mesosoma. Petiole in lateral view rather massive and high (PeH/CS 0.343), higher than postpetiole, with a short peduncle, a slightly concave anterior profile and a convex dorsal node that steeply slopes down to the caudal cylinder; petiole in dorsal view very wide (PeW/CS 0.359), its node semiglobular and slightly wider than long. Postpetiole moderately wide and rather high (PeW/CS 0.468, PpH/CS 0.320), in dorsal view with a straight or slightly concave anterior margin, rounded sides and much wider than long; its sternite with pronounced anterolateral corners that are formed by bilateral lobes which strongly protrude compared to anteromedian level. Whole surface of head, mesosoma and petiole with a very fine (mesh diameter on vertex only 8–9 μm) but deeply sculptured reticulum, thus appearing at lower magnifications perfectly matt. Postpetiole less deeply sculptured. Scapes, coxae, femora and tibiae with fine microreticulum and appearing matt at lower magnifications. First gaster tergite very finely microreticulate-shagreened, also appearing matt at lower magnifications. All cuticular surfaces including those of the appendages with decumbent, dilute pubescence. Pubescence on 1st gaster tergite much longer and denser than in any related species [PLG/CS 7.21%, sqPDG 3.92], on anterior surface directed caudad and on posterior one caudomediad. Lateral head at horizontal level of eye with an extended, longitudinal, dark brown ribbon that is as broad as the eye; this ribbon is flanked below and above by broad bands without any pigment (as result appearing whitish). Vertex, scape, postpetiole, gaster, procoxae, tibiae and femora except their proximal and distal portions light yellowish brown. Mesosoma light orange brown. Petiole, meso- and metacoxae, clypeus, spines, funiculus as well as proximal and distal portions of femora without pigmentation (appearing whitish in dead, dry specimens).
Taxonomic comments.
Cardiocondyla pirata n.sp. shows all basic characters of the
C. argentea n.sp. group but it deviates in a number of characters. It cannot be confused with any ant worldwide because of its unique, unmistakable pigmentation pattern. This clear identification is supported by diagnostic structural and shape characters: there is no overlap with the five other species of the
C. argentea n.sp. group in the characters FRS/CS, SP/CS and PLG/CS and there is little overlap in PeW/CS and sqPDG (
Table 2).
Biology. The only complete colony collected in the field consisted of three dealate queens, 15 workers and brood and produced in the lab over 20 female sexuals and one ergatoid male, but thereafter died. Hence, there are no long-term observations on the life history of this interesting species. Considering the situation in other members of the
Cardiocondyla argentea n.sp. group [
1,
41], it is highly probable that there are only ergatoid males which are long-lived, mate always inside the nest and try to kill rivals using their sickle-shaped mandibles in order to monopolize the matings. Nests should contain 1–4 queens. The adaptive significance of the extraordinary pigmentation pattern of these tiny ants remains a puzzle (see discussion in [
42]).
4.4.15. Cardiocondyla nivalis Mann, 1919
Cardiocondyla nivalis Mann, 1919 [description, zoogeography]
Type material. The taxon has been described from the village Pamua on the island San Cristobal (10.411° S, 161.747° E). Type specimens were not seen. The separation from
Cardiocondyla allonivalis n. sp. is based on allopatric occurrence and W.M. Mann’s description of postpetiole shape. Mann stated “… postpetiole in profile longer than high and convex above…” [
43]. This indicates that the postpetiolar sternite is not bulging in contrast to the bulging bilobate structure seen in
C. allonivalis n. sp.
Geographic range. The species is known so far only from four islands of the Solomon group: Vella Lavela (7.76° S, 156.66° E), Guadalcanal (9.65° S, 160.21° E), Renell (11.71° S, 160.36° E) and San Cristobal (10.41° S, 161.75° E).
Diagnosis: Worker (
Figure 16,
Table 3): Very small size, CS 376 µm. Head moderately long (CL/CW 1.127); with maximum CL and CW in same focal level, its posterior margin slightly concave and anterior clypeal margin strongly concave. Postocular distance large, PoOc/CL 0.440. Scape short, SL/CS 0.836. Eye rather small, EYE/CS 0.238. Frons rather narrow with short and parallel frontal carinae, FRS/CS 0.281. Mesosoma slender; dorsal profile of promesonotum and propodeum only weakly convex (nearly straight) but with a notable metanotal depression. Anterior pronotum in dorsal view rounded, without pronounced corners. Propodeal spines rather short (SP/CS 0.200), in dorsal view only very slightly diverging, in lateral view straight and their axis deviating 20° from longitudinal axis of mesosoma. Petiole in lateral view rather massive and high (PeH/CS 0.349), higher than postpetiole, with a short peduncle, a concave anterior margin and a convex dorsocaudal profile; petiole in dorsal view wide (PeW/CS 0.312), its node semiglobular and slightly longer than wide. Postpetiole moderately wide and high (PpW/CS 0.483, PpH/CS 0.327), in dorsal view much wider than long and with a concave anterior and straight to slightly convex posterior margin when these margins are adjusted to the same focal level; postpetiolar sternite flat without any anterolateral corners. Whole surface of dorsal head with circular foveolae that show a flat central tubercle, mean dFov 14.7 µm; the interspaces between foveolae about as large as foveolar diameter and finely longitudinally shagreenate (
Figure 16D). Microsculpture on dorsum of mesosoma similar to that on vertex but foveolar diameter smaller; lateral mesosoma microreticulate. Petiole very finely microreticulate. Microsculpture on postpetiole still much finer, the dorsal part as a consequence slightly shiny. Basal part of 1st gaster tergite very finely microreticulate but appearing rather shiny at lower magnifications. Pubescence on 1st gaster tergite of medium length and rather dilute (PLG/CS 5.79%, sqPDG 4.94]. Gaster jet black and all remaining body parts excluding the eyes snowy white or very pale yellowish brown.
Taxonomic comments. Cardiocondyla nivalis is a morphologically rather outstanding species found over a length of 600 km on a series of islands which remained separated during the Pleistocene sea level depressions.
Biology. Unknown.
4.4.16. Cardiocondyla allonivalis n.sp.
Etymology: from άλλο (Greek.) meaning “other” and nivalis (Lat.) meaning “snowy”; because of the overall similarity to Cardiocondyla nivalis.
Type material. Holotype and one paratype worker on separate pins, both labelled “PNG: 5.25° S, 145.267° N, Wanang, Sogeram riv. 95 m, Bait trap—ground—D4, coll. Janda 2007.10.23”; depository SMN Görlitz.
All material examined. Examined were four samples with five workers from Papua New Guinea—three samples through direct stereomicroscopic evaluation and one sample through photo evaluation. For details see
Supplementary Information Tables S1 and S2.
Geographic range. The species is so far only known from sea level up to 350 m in four sites in Papua New Guinea: Cape Wom (3.533° S, 143.583° E); Wanang (5.250° S, 145.267° E), Goldie River (9.30° S, 147.42° E) and Popondetta (8.77° S, 148.24° E).
Diagnosis: Worker (
Figure 17,
Table 3, pictures CASENT0914964 in ww.antweb.org): Very small size, CS 380 µm. Head moderately long (CL/CW 1.137); with maximum CL and CW in visual plane, its posterior margin straight or very slightly concave and anterior clypeal margin slightly concave. Postocular distance large, PoOc/CL 0.440. Scape short, SL/CS 0.808. Eye rather small, EYE/CS 0.242. Frons rather narrow with short and almost parallel frontal carinae, FRS/CS 0.299. Mesosoma slender; dorsal profile of promesonotum and propodeum slightly convex but with a notable metanotal depression. Anterior pronotum in dorsal view rounded, without pronounced corners. Propodeal spines short (SP/CS 0.185), in dorsal view slightly diverging, in lateral view nearly straight and their axis deviating 26–28° from longitudinal axis of mesosoma. Petiole in lateral view more massive and higher than in
C. nivalis (PeH/CS 0.363), only slightly higher than postpetiole, with a short peduncle, a concave anterior margin, a weakly convex dorsal profile of node that more or less linearly slopes down to caudal cylinder. Petiole in dorsal view wide (PeW/CS 0.319), its node semiglobular and slightly wider than long. Postpetiole moderately wide and but distinctly higher than in
C. nivalis (PpW/CS 0.470, PpH/CS 0.356), in dorsal view much wider than long and with a concave anterior and slightly convex posterior margin when these margins are adjusted to the same focal level; profile of postpetiolar sternite in contrast to
C. nivalis strongly bulging; this bulge is formed by bilateral lobes which strongly protrude compared to median level of sternite; these lobes appear as corners in anterolateral view (
Figure 17D). Whole surface of dorsal head with circular foveolae that show a flat central tubercle, mean dFov 15.5 µm; the interspaces between foveolae on vertex smaller than foveolar diameter and delicately longitudinally striate. Microsculpture on dorsum of mesosoma weaker than on vertex, microreticulate-corrugated and without foveolae; lateral mesosoma microreticulate. Petiole laterally finely microreticulate, dorsally more microcorrugated. Postpetiole finely microcorrugated and in overall appearance matt. Basal part of 1st gaster tergite very finely microreticulate but appearing rather shiny at lower magnifications. Pubescence on 1st gaster tergite of medium length and rather dilute (PLG/CS 6.44%, sqPDG 4.36]. Gaster jet black and all remaining body parts excluding the eyes very pale yellowish brown.
Taxonomic comments. Cardiocondyla nivalis and C. allonivalis n.sp. are morphometrically rather similar. Yet they appear to be allopatric and are easily separable by the shape of postpetiolar sternite which is flat in the former but shows in the latter a bulging lobe on each side of the sternite.
Biology. One nest was found by P.S. Ward in soil of a semi-dry littoral forest.
4.4.17. Cardiocondyla wheeleri Viehmeyer, 1914
Cardiocondyla wheeleri Viehmeyer, 1914 [type investigation]
Type material. The taxon has been described from the Rawlinson Mountains in Papua New Guinea. Investigated were 2 syntype workers labelled by H.Stitz “D.N.Guinea. Rawlinsongeb. Coll. Viehm.“ and “Cardiocondyla wheeleri Viehm”, ZM Berlin.
Geographic range. The species is so far known only from the type locality in Papua New Guinea (6.47° S, 147.10° E, 1800 m).
Diagnosis: Worker (
Figure 18,
Table 4): Medium-sized, CS 515 µm. Head rather long (CL/CW 1.152); with head adjusted in measuring position for CL, its posterior margin straight and median third of anterior clypeal margin only very feebly concave; the maximum depth of clypeal concavity is only 1.0–2.1% CL. Postocular distance large, PoOc/CL 0.446. Scape long, SL/CS 0.870. Eye small, EYE/CS 0.220. Frons rather narrow (FRS/CS 0.287), frontal carinae immediately caudal of the FRS level slightly converging (FL/FR 1.023). Metanotal depression moderately deep and with shallow slopes (MGR/CS 2.68%), dorsal profile of promesonotum and propodeum convex. Pronotal corners in dorsal view prominent, but blunt and forming an angle of 90–100°. Dorsal plane of mesosoma rather continuously narrowing from pronotal corners caudal to metanotal groove; there is only a weak convexity at the mesonotal level. Propodeal spines strong and extremely long (SP/CS 0.404), with a very large basal distance(SPBA/CS 0.366), in lateral view downcurved and their average orientation almost parallel to longitudinal mesosomal axis. Petiole rather high (PeH/CS 0.344); in lateral view with a very short peduncle, a weakly concave frontal and broadly convex dorsal profile, in dorsal view moderately wide (PeW/CS 0.284) and its node longer than wide. Postpetiole in dorsal view broad (PpW/CS 0.557), with semicircularly rounded sides and clearly excavated anterior margin, in lateral view much lower than petiole (PpH/CS 0.273), its sternite shallowly convex and without any prominent structures. Central vertex finely longitudinally carinulate-rugulose; paramedian and lateral vertex longitudinally rugulose-foveolate, foveolae rather shallow, often bicoronate, not eye-catching, with 18–20 µm diameter, interspaces wider than foveolar diameter. All interspaces between microsculptural elements on dorsal head including frontal laminae and clypeus perfectly matt (but surfaces are polluted, so perhaps in clean condition slightly shiny?). Mesosoma matt, entirely microreticulate-corrugated. Petiole rather matt, microreticulate. Postpetiole in dorsal view moderately shiny and finely microreticulate. Gaster tergites moderately shiny, with a fine microreticulum (which are really the margins of clinker-like surface structures. Tergite pubescence short and dilute (PLG/CS 4.44%, sqPDG 5.71). Whole body except of the dark brown gaster and the infuscated antennal club yellowish.
Taxonomic comments. For differences to Cardiocondyla excavata n.sp. and C. goroka n.sp. see below.
Biology. Unknown.
4.4.18. Cardiocondyla excavata n.sp.
Etymology: from excavare (Lat.), meaning “excavate”; because of the deeply concave anterior clypeal margin.
Type material. Holotype and two paratype worker on separate pins, both labelled “PNG: 6.33° S, 145.86° E, 1670 m, Kainantu-4.8 km S, edge of mixed oak forest, in log, A.G.E. Emerson 1963.01.04”; depository SMN Görlitz.
Geographic range. The species is so far known only from the type locality in Papua New Guinea (6.33° S, 145.86° E, 1670 m)
Diagnosis: Worker (
Figure 19,
Table 4): Medium-sized, CS 491 µm. Head distinctly shorter than in
C. wheeleri, CL/CW 1.107; with head adjusted in measuring position for CL, its posterior margin straight to slightly concave and median third of anterior clypeal margin in contrast to
C. wheeleri deeply concave; in caudodorsal view, the maximum depth of clypeal concavity is 2.9–3.3% CL. Postocular distance large, PoOc/CL 0.445. Scape rather long, SL/CS 0.866. Eye larger than in
C. wheeleri, EYE/CS 0.238. Frons narrow (FRS/CS 0.277), frontal carinae immediately caudal of the FRS level parallel or slightly converging. Metanotal depression shallower than in
C. wheeleri (MGr/CS 1.75%), dorsal profile of promesonotum and propodeum more linear than in
C. wheeleri. Pronotal corners in dorsal view prominent, but blunt and forming an angle of about 90°. Dorsal plane of mesosoma rather continuously narrowing from pronotal corners caudal to metanotal groove; there is no or only a weak convexity at the mesonotal level. Propodeal spines strong but shorter than in
C. wheeleri and their bases more approached (SP/CS 0.380, SPBA/CS 0.346), in lateral view downcurved with their mean orientation deviating by about 15° from longitudinal mesosomal axis. Petiole rather high (PeH/CS 0.336); in lateral view with a very short peduncle, a weakly concave frontal and broadly convex dorsal profile, in dorsal view narrower than in
C. wheeleri, PeW/CS 0.257, the node longer than wide. Postpetiole less broad than in
C. wheeleri, PpW/CS 0.536; in dorsal view, when both margins are adjusted to same focal level, with semicircularly rounded sides and strongly concave anterior and straight posterior margin; in lateral view much lower than petiole (PpH/CS 0.256), its sternite shallowly convex and without any prominent structures. Whole vertex with bicoronate foveolae of 16–18 µm diameter, the interspaces as large or smaller than foveolar diameter and with rather prominent reticulate microsculpture. Dorsal mesosoma reticulate, with scattered bicoronate foveolae, lateral mesosoma reticulate. Petiole matt, microreticulate. Postpetiole in dorsal view shiny and very delicately microreticulate. First gaster tergite shiny, on whole surface very delicately microreticulate. Tergite pubescence short and dilute (PLG/CS 4.68%, sqPDG 5.80). Whole body with exception of the dark brown gaster yellowish.
Taxonomic comments. The strongly concave clypeus, the shorter head, the larger eye and the narrower waist segments allow to hypothesize Cardiocondyla excavata n.sp. as heterospecific from C. wheeleri. There is probably some geographic isolation as the type localities are mountain regions separated by a deep river valley.
Biology. The type nest was found in a log at the margin of a mixed oak forest.
4.4.19. Cardiocondyla goroka n.sp.
Etymology: from the name of the holotype locality.
Type material. Holotype worker labelled “PAPUA N.G. Rihona, betw. Goroka and Markham Riv.\1980 R.I.Vane-Wright”, depository BMNH London; one paratype worker labelled “NEW GUINEA: Morobe district, Wau, Mt. Kaindi, alt. 2200m 25-II-1973 Thomas W. Davies”, depository CAS San Francisco; one paratype worker labelled “ PNG: 7.356° S, 146.678° E, Wau: Mt. Kaindi, 2200 m, B.B. Lowery 1967.12.13”, depository SMN Görlitz.
Geographic range. The three sites in Papua New Guinea are less than 190 km apart: The holotype type locality is situated at about 6.083° S, 145.367° E, 1500 m, the paratype locality at 7.3564° S, 146.6781° E, 2200 m and Keglsugl, the locality of the photo evaluated specimen, at 5.833° S, 145.100° E, 2600 m.
Diagnosis: Worker (
Figure 20, images of specimen CASENT0914971 in
www.antweb.org,
Table 4): much larger than
C. wheeleri and
C. excavata n.sp., CS 590 µm. Head rather short, CL/CW 1.145, occipital margin straight or slightly excavated, anterior clypeal margin slightly excavated, depth of excavation 0.7–1.0% CL. Postocular distance large, PoOc/CL 0.432. Scape rather long, SL/CS 0.870. Eye small, EYE/CS 0.225. Frons rather narrow (FRS/CS 0.283), frontal carinae immediately caudal of the FRS level parallel or weakly converging caudad and then diverging. Pronotal corners in dorsal view prominent, but blunt, forming an angle of 90–95°. Dorsal plane of mesosoma not continuously narrowing from pronotal corners caudal to metanotal groove, margin caudal of pronotal corners first concave and then convex; in contrast to
C. wheeleri, there is a conspicuous convexity at the mesonotal level. Metanotal depression moderately deep and with shallow slopes. Spines strong, straight or downcurved but shorter than in
C. wheeleri and
C. excavata n.sp. (SP/CS 0.332) and with a large basal distance (SPBA/CS 0.370); in lateral view more erected than in
C. wheeleri and
C. excavata n.sp., chord of spine base and tip deviating by 17–22° from longitudinal mesosomal axis. Petiole rather wide and high (PeW/CS 0.284, PeH/CS 0.351), the peduncle longer and the anterior profile more concave than in
C. wheeleri, dorsal profile broadly convex; the node in dorsal view globular. Postpetiole in dorsal view broad (PpW/CS 0.524) but relatively narrower than in
C. wheeleri, with semicircularly rounded sides and strongly concave anterior margin; in lateral view much lower than petiole (PpH/CS 0.280), its sternite shallowly convex and without any prominent structures. Whole dorsal head in overall impression matt. A narrow stripe on median vertex, frontal laminae, and posterior half of clypeus longitudinally carinulate. Paramedian and lateral vertex with bicoronate foveolae of 15–18 µm diameter which are deeper and more eye-catching than in
C. wheeleri; foveolae embedded between longitudinal rugulae, foveolar interspaces smaller or as wide as foveolar diameter. Mesosoma matt, entirely microreticulate-foveolate-corrugated. Petiole moderately shiny and microreticulate. Postpetiole dorsally moderately shiny and finely microreticulate. Gaster tergites moderately shiny, delicately microreticulate. Tergite pubescence short and denser than in
C. wheeleri (PLG/CS 4.25%, sqPDG 5.34). Whole body except of the blackish gaster and the infuscated antennal club yellowish.
Taxonomic comments. C. goroka n.sp. is hypothesized here as a sister species of C. wheeleri. Considering the situation in Cardiocondyla species worldwide, the large difference in absolute body size to C. wheeleri and C. excavata n.sp. is unlikely to be explained by intraspecific variance. Furthermore the differing dorsal aspect of mesosoma, spine orientation and length seem better explained by heterospecificity than by intraspecific variation.
Biology. One forager was collected by P.S. Ward in a cleared grassy area.
4.4.20. Cardiocondyla nigrocerea Karavajev, 1935
Cardiocondyla nigrocerea Karavajev, 1935 [type investigation]
Type material. The taxon has been described from Ambon/Indonesia. Investigate were one worker syntype labelled “Amboina Karavaiev\2745. Coll. Karavaievi\Cardiocondyla nigro-cerea Karav. typ.” and a second worker syntype labelled ”2745. Coll. Karavaievi\Cardiocondyla nigrocerea sp.n. Ambon”; both deposited in SIZ Kiev.
Geographic range. Only known so far from the Indonesian islands of Ambon (3.72° S, 128.15° E, 120 m) and Obi (1.388° S, 127.601° E, 33 m).
Diagnosis: Worker (
Figure 21, images of specimens CASENT0280592 and CASENT0916975 in ww.antweb.org,
Table 4): Smaller than
C. wheeleri, CS 457 µm. Head moderately elongated, CL/CW 1.166. Outlines of posterior head in dorsal view rather rectangular, postocular distance moderate (PoOc 0.426), occipital margin slightly concave. Anterior clypeal margin between the level of frontal carinae concave. Scape short, SL/CS 0.816. Eye small, EYE/CS 0.224. Frons rather narrow (FRS/CS 0.272), frontal carinae immediately posterior of the FRS level almost parallel. Mesosoma in dorsal view concavely narrowing from the pronounced, but rather blunt pronotal corners caudad to anterior propodeum; dorsal mesosomal profile forming an almost straight line from anterior pronotum to spine tips, metanotal depression shallow (MGr/CS 0.90%. Spines moderately long and with rather large basal distance (SP/CS 0.314, SPBA/CS 0.347), in dorsal view their basal parts only weakly diverging and then weakly incurving caudad, in lateral view spine axis deviating by only 5° from longitudinal mesosomal axis—i.e., quasi parallel. Petiole very high (PeH/CS 0.379), disproportionately higher than postpetiole (PeH/PpH 1.502–1.582); in lateral view with a very short peduncle and a massive, roughly square-shaped node; petiole node in dorsal view clearly longer than wide, oval, narrowing frontad and above forming a keel. Postpetiole in dorsal view much wider than petiole (PpW/CS 0.541), with convex sides and deeply concave anterior margin; in lateral view much lower than petiole (PpH/CS 0.244), its sternite flat and without any prominent structure. Vertex with bicoronate foveolae of 13–17 µm diameter, the interspaces roughly as wide as foveolar diameter and with longitudinal microrugulae. Mesosoma in overall surface appearance matt, its dorsum finely corrugated-shagreened and its lateral surfaces finely microreticulate. Lateral surfaces of petiole finely microreticulate. Dorsum of postpetiole shiny, only with a very delicate microreticulum. First gaster tergite shiny but on its whole surface delicately microreticulate. Tergite pubescence short and dilute (PLG/CS 4.46%, sqPDG 5.97). Whole body light yellowish-grey except for the contrastingly blackish brown gaster.
Taxonomic comments. Due to the unique combination of mesosomal and waist shape, Cardiocondyla nigrocerea is not to confuse with any species worldwide. Waist measurements alone are diagnostic: the ratio PeH/PpH∗PpW/PeW is 1.928 ± 0.248 [1.372, 2.908] in 2548 workers of any other species but 3.108 ± 0.069 [2.986, 3.151] in five workers of C. nigrocerea.
Biology. Karavajev found his specimens on the island of Ambon under the bark of a rotten, fallen tree trunk. The biology of the population from Obi has been studied by Heinze et al. [
41]: The nests were found in a secondary rain forest along a river and consisted of small chambers about 1–2 cm deep in the soil and had a single tiny entrance hole. Each nest contained a single dealate queen, several winged female sexuals, approximately 20 to 80 workers, and brood. One colony had an ergatoid male. The males are ergatoid and have sickle-shaped mandibles which they use to grab and kill rival males. Males reared in the laboratory were frequently moving on the brood pile apparently patrolling and antennating it for the presence of freshly eclosed female sexuals or rival males. They grasped young males with their sickle-shaped mandibles, held it for several minutes and besmeared it with secretions from the tips of their gasters. The attacked males died either directly from the attack of the dominant male or from the attack by workers whose aggression was elicited by the applied secretion.
4.4.21. Cardiocondyla thoracica (Smith, 1859)
Myrmica thoracica Smith, 1859 [photos of type specimen, description]
Type material. The taxon has been described from Aru/Indonesia. A syntype worker labelled “Myrmica thoracica Smith, TYPE. J.P.L.S.Lond. v.iii,1859,p.131”, “Aru” and “thoracica Sm.”, deposited in OXUM Oxford and depicted under specimen identifier CASENT0901412
www.antweb.org was investigated by photo evaluation.
All material examined. Eight samples with 14 workers were available for direct microscopic investigation and two additional samples were evaluated by photo evaluation (CASENT0901412 and CASENT0914970 in
www.antweb.org). They originated from Indonesia (2 samples), Papua New Guinea (2) and north Queensland (6). For details see
Supplementary Information Tables S1 and S2.
Geographic range. Only found east of the Wallace line between 2° S and 13° S, 129° E and 147° E and from sea level up to 750 m. The range includes Indonesia (Seram Solea and Aru), Papua New Guinea and the region of the coastal forest of northernmost Queensland/Australia.
Diagnosis: Worker (
Figure 22, images of specimens CASENT0914970 in
www.antweb.org;
Table 4): Rather small, CS 479 µm. Head short, CL/CW 1.098. Posterior margin of vertex slightly convex. Postocular head sides convex and less converging than in
paradoxa, postocular distance moderate (PoOc 0.452). Anterior clypeal margin deeply and broadly excavated, whole area of median clypeus deeply concave. Scape rather long, SL/CS 0.876. Eye rather large, EYE 0.240. Frons wide (FRS/CS 0.286), frontal carinae posterior of the FRS level almost parallel or slightly diverging caudad; planes of frontal laminae sloping mediad by 40°, frontal carinae thus much elevating above the level of posterior clypeus. Mesosoma much less slender than in
paradoxa, in dorsal view with very prominent pronotal corners each forming an sharp angle of 70°, their distance as wide as posterior head, dorsal mesosoma continuously and roughly linearly narrowing from the wide pronotal corners caudad to spine base. Dorsal mesosomal profile with exception of the angle produced by the corners evenly convex; metanotal depression fully absent or only suggested (MGr/CS 0.63%). Spines sharp and long (SP/CS 0.335), with rather wide basal distance (SPBA/CS 0.333), and straight or very feebly downcurved; their axis in in lateral view usually deviating less than 20° from the longitudinal mesosomal axis. Petiole in lateral view high (PeH/CS 0.349), with a short peduncle, a concave anterior and convex dorsocaudal profile. Petiole node in dorsal view longer than wide and narrowing frontad but whole petiole rather wide (PeW/CS 0.286). Postpetiole low (PpH/CS 0.270), in dorsal view wide (PpW/CS 0.525) and with a strongly concave anterior margin. Postpetiolar sternite flat, its anterolateral portions each with a short, upcurved, and shallow carina. Head in overall appearance smooth and very shiny, median and paramedian vertex with small unstructured foveolae of 6–7 µm diameter and 17–21 µm nearest-neighbor distance, on more lateral areas of vertex, in particular the surface frontomedial of the eye, the foveolae are larger and with a structured bottom: 13–19 µm diameter and a central accessory foveola of 5–6 µm diameter (
Figure 22D); foveolar interspaces very shiny but with very delicate microreticular structures. Posterior and anteromedian clypeus smooth, in its anterolateral parts rugulose. Frontal laminae in lateral parts rugulose and with well-defined foveolae of 7–13 µm diameter. Dorsal mesosoma, smooth with scattered foveolae of 5–7 µm diameter, lateral mesosoma and waist microreticulate but shiny. 1st gaster tergite in overall impression shiny but with a well-developed microreticulum and a rather short, dilute pubescence (PLG/CS 4.21%, sqPDG 6.54). Whole head capsule and gaster blackish brown. Appendages, mesosoma and waist yellow.
Taxonomic comments.
Cardiocondyla thoracica appears rather monomorphous along those 2000 km of its multi-island range and appears unmistakable. It is considered to be of Indonesian origin [
44] and to have spread south during the last ice age when Northern Australia was connected to Papua New Guinea.
Biology. P.S. Ward found a nest in a rotten log in a New Guinean rainforest. Heinze et al. [
45] described the biology of the population from northern Queensland as follows. Nests are composed of small chambers and were found 1–3 cm deep in red, sandy soil on sun exposed, sparsely vegetated patches between the forest and a roadside ditch. Another colony was found nesting in a cavity of a fallen tree in a streambed. Ten colonies collected in the field contained between seven and approximately 80 workers (median 40) and one to four mated queens which did not show mutual aggression. In three nests a single wingless male was present. Males are exclusively ergatoid and have sickle-shaped mandibles. They appear to widely ignore adult competitors, but grab defenseless, young rivals during or immediately after emergence and besmear them with hindgut secretion. This fluid elicits deadly worker aggression against the besmeared individual.
4.4.22. Cardiocondyla paradoxa Emery, 1897
Cardiocondyla paradoxa Emery, 1897 [description]
The published type locality is “Friedrich-Wilhelmshafen” (=Madang/Papua New Guinea, 5.22° S, 145.79° E). Both the two drawings and the verbal description provided in the original description allow a clear identification of this extremely shaped ant. A worker labelled “Cardiocondyla paradoxa” (Emery’s handwriting], “N. Guinea Biró 1899”, “Sattelberg Huon-Golf” and “TYPUS”, deposited in MCSN Genova and depicted under specimen identifier CASENT0904465 in
www.antweb.org cannot be considered as type according to collecting date.
Cardiocondyla brevispinosa (Donisthorpe 1948) [junior synonym, type investigation]
This taxon has been described under the name Pheidole brevispinosa from Mafin Bay/Irian Jaya (=“Dutch New Guinea”). Investigated were two type workers from BMNH London, labelled “Maffin Bay, Dutch N. Guinea, VIII-I-44 E.S: Ross Coll.” The labelling of these is not fully coincident with the data published by Donisthorpe: “Described from four workers, Maffin Bay, Dutch New Guinea, June 10, 1944 E.S.Ross Coll.” Due to the extreme characters developed in this species, the synonymy is obvious.
All material examined. Available for direct microscopic investigation were 12 samples with 18 workers. One additional sample was evaluated by photo evaluation (CASENT0914969 in
www.antweb.org). They originated from Indonesia (2 samples) and Papua New Guinea (11). For details see
Supplementary Information Tables S1 and S2.
Geographic range. Only known from New Guinea between 1° S and 8° S, 138° E and 146° E and from sea level up to 1800 m.
Diagnosis: Worker (
Figure 23, images of specimens CASENT0914969 in
www.antweb.org;
Table 4): Large, CS 583 µm. Head much elongated, CL/CW 1.170. Postocular head sides strongly converging, postocular distance rather small (PoOc 0.436). Anterior clypeal margin deeply and broadly excavated, depth of excavation 1.7–4.3% CL. Anterior clypeal margin deeply and broadly excavated. Scape very long, SL/CS 0.978. Eye rather small, EYE 0.229. Frons narrow (FRS/CS 0.258); planes of frontal laminae sloping mediad by 40°, frontal carinae thus much elevating above the level of posterior clypeus. Mesosoma slender and in dorsal view with very prominent and sharp pronotal corners each forming an angle of 72–85°. Dorsal mesosomal profile in overall impression rather straight but metanotal depression very deep (MGr/CS 6.22%). Spines moderately long, in dorsal view with a rather low basal distance and slightly incurved (SP/CS 0.283, SPBA/CS 0.255); spine axis in lateral view weakly diverging from longitudinal mesosomal axis. Petiole very narrow (PeW/CS 0.233) and extremely elongated, in dorsal view 3–4 fold longer than wide; in lateral view very low (PeH/CS 0.275), the node much elongated and with shallowly convex dorsal profile. Postpetiole moderately wide (PpW/CS 0.456) but rather high (PeH/CS 0.310), with a prominent ventral bulge and in dorsal view with a strongly concave anterior margin. Head in overall appearance smooth and very shiny, with foveolae of 16–21 µm diameter and 30–70 µm nearest-neighbor distance; foveolar interspaces very shiny but with fine fragments of microreticular structures. Clypeus smooth. Frontal laminae smooth except for few well-defined foveolae. Mesosoma, waist, and gaster tergites almost glabrous but with a very delicate microreticulum. Tergite pubescence extremely short and very dilute (PLG/CS 3.28%, sqPDG 8.04). Whole head dark brown but with a warm tinge; antennae light yellowish brown; mesosoma, waist and whole legs yellow, gaster dark brown. A deviating color morph has a dark brown head and mesosoma, and the waist, legs and gaster medium brown.
Taxonomic comments. Cardiocondyla paradoxa is an unmistakable member of the C. thoracica group. The disagreements between the labelling of putative type specimens of C. paradoxa and C. brevispinosa and the statements in the original descriptions are obvious. However, C. paradoxa and brevispinosa are taxa with such extreme characters that an identification and synonymization by the original description alone seems to have a low risk of error.
Biology. It is a rain forest species often collected from leaf litter and foraging on ground. According to Wilson [
46] it digs its nests to about 20 cm in the soil and constructs a single entrance gallery concealed by leaf litter.
4.4.23. Cardiocondyla subspina n.sp.
Etymology: because of the dent on anteromedian postpetiolar sternite.
Type material. Holotype worker labelled “INDONESIA: N.C.SERAM\Manusela N.P. Wae Mual Plain 25.vii-9.ix.1987\Op.Raleigh M.J.D.Brendell B.M.1987-262\fog. Card. B”, BMNH London; 1 paratype worker with identic labelling, SMN Görlitz.
Geographic range. Precise geographic coordinates of the type locality Wae Mual Plain, an alluvial forest in Manusela National Park, were not retrievable at the first hand. It is assumed to be at approximately 3° S, 129.5° E and below 500 m.
Diagnosis: Worker (
Figure 24,
Table 4): Small-sized, CS 434 µm. Head long, CL/CW 1.182. Postocular head sides only weakly converging; in adjustment to measure CL, hind margin of head very feebly concave and anterior clypeal margin slightly concave, postocular distance very large, PoOc 0.465. Scape short, SL/CS 0.812. Eye rather small, EYE 0.223. Frontal carinae widely distant (FRS/CS 0.290), immediately posterior of the FRS level almost parallel. Planes of frontal laminae sloping mediad by 40°, frontal carinae thus much elevating above the level of posterior clypeus. Mesosoma in dorsal view with very pronounced anterolateral pronotal corners forming a sharp angle of 75°, their outer distance a little shorter than width of posterior head, dorsal mesosoma continuously and almost linearly narrowing from the wide pronotal corners caudad to spine base. Metanotal depression with wide slopes but rather shallow (MGr/CS 1.62%). Spines rather short (SP/CS 0.268), with rather wide basal distance (SPBA/CS 0.310) and straight; their axis in lateral view deviating by only 8° from the longitudinal mesosomal axis. Petiole in lateral view high (PeH/CS 0.344) and with a short peduncle, a concave anterior and convex dorsocaudal profile; in dorsal view 2.3fold longer than wide, rather narrow (PeW/CS 0.245) and with rather straight sides. Petiole node much longer than wide and in caudodorsal view with a wedge-shaped dorsum. Postpetiole comparatively narrow (PpW/CS 0.457), in dorsal view with a strongly concave anterior margin and convex sides; postpetiole high (PpH/CS 0.307), its sternite rather flat but with a sharp anterior edge which appears in profile as a distinct anteromedian dent (
Figure 24D). Frontal laminae and clypeus corrugated-foveolate but with small shiny surface areas. Paramedian and lateral vertex with flat-bottomed but well-demarcated foveolae of 15–17 µm diameter, which show a flat inner tubercle of 6–8 µm diameter. Foveolar interspaces at least as wide as foveolar diameter and finely corrugated. Whole mesosomal and petiolar surface reticulate, reticulation on postpetiole shallower. 1st gaster tergite in overall impression shiny but with a well-developed microreticulum and with rather short and dilute pubescence (PLG/CS 4.21%, sqPDG 6.54). Whole ant yellow except the blackish brown gaster.
Taxonomic comments. Cardiocondyla subspina can be grouped together with C. paradoxa and C. thoracica based on similarities in shape of mesosoma and frontal laminae.
Biology. Unknown. The presence in a fogging sample could suggest arboreal foraging.
4.4.24. Cardiocondyla sulcata n.sp.
Derivatio nominis: from Latin, meaning “grooved”, because of the very pronounced metanotal groove.
Type material. Holotype worker and 2 paratype workers labelled “MALAYSIA Neg.Sembilan Pasoh For.Res. iii-iv.1994\\Fog sample M.Brendel K.Jackson L.Ficken\\2-15”, BMNH London; 3 paratypes, 2 workers and 1 gyne, with identic labelling in SMN Goerlitz.
Geographic range. The only known site is the Pasoh Forest Reserve (2.982° N, 102.313° E, 130 m) in Malaysia.
Diagnosis: Worker (
Figure 25,
Table 3): Very small-sized, CS 415 µm. Head short, CL/CW 1.098. Postocular head sides convex and slightly converging; hind margin of vertex and anterior clypeal margin between the level of frontal carinae straight, postocular distance large, PoOc 0.451. Scape very short, SL/CS 0.787. Eye rather large, EYE 0.241. Frontal carinae very approached (FRS/CS 0.246), at FRS level diverging caudad. Anterior pronotum in dorsal view circularly rounded. Metanotal groove very deep (MGr/CS 6.00%) and with an extremely steep posterior slope that forms with the anterior slope a distinct angle of 90–100°. Spines with very low basal distance (SPBA/CS 0.275), rather short (SP/CS 0.172) and acute; in lateral view steep, deviating from longitudinal mesosomal axis by 35–45°. Petiole extremely narrow and rather low (PeW/CS 0.229, PeH/CS 0.304), with a very long peduncle; in dorsal view more than twice as long than wide, and with a small circular node, in profile the node is low with a convex dorsum. Postpetiole extremely narrow and very low (PpW/CS 0.399, PpH/CS 0.282); in dorsal view with convex sides and a rather narrow, strongly concave anterior margin, postpetiolar sternite almost flat, only with suggested and very rounded anterolateral corners. Whole dorsal head with very shallow microsculpture. Clypeus rather smooth, in posterior third shiny. Frontal laminae rather smooth, with fragments of microfoveolae or rugulae. Vertex with irregular, sometimes bicoronate, foveolae of 17–20 µm diameter and often irregular margins; the interspaces smaller than foveolar diameter, rather smooth with irregular fragmentary microsculpture. Whole mesosoma microreticulate. Petiole microreticulate, a small central spot of dorsal petiole node and whole postpetiolar tergite smooth and shiny. Gaster tergite smooth and shiny, with scattered fragments of very fine microreticular structures and relatively short and dilute pubescence (PLG/CS 6.44%, sqPDG 5.41). All body parts concolorous yellowish.
Taxonomic comments. Cardiocondyla sulcata n.sp. has a multitude of unique characters and is not to confuse with any known species. It could represent a monotypical clade because morphology does not give suggestions on related species.
Biology. Unknown. The presence in a fogging sample could suggest arboreal foraging.
4.4.25. Cardiocondyla sima Wheeler, 1935
Cardiocondyla (Prosopidris) sima Wheeler, 1935 [types investigated]
This taxon has been described from the Philippines. Investigated were 5 worker syntypes, labelled “Dansalan, Lanao 12/21/25 L.M.Morato”, “Gift of W.M.Wheeler”, “Syntypes of Cardiocondyla sima Wheeler S P Cover 5-00” and “M.C.Z. Cotype 20798”; depository MCZ Cambridge.
All material examined. Four samples with 7 workers were available for direct microscopic investigation—two samples from the Philippines and one each from Malaysia and Thailand. For details see
Supplementary Information Tables S1 and S2.
Geographic range. Only known from west of the Wallace Line between 4°N and 11°N, 98°E and 125°E and from sea level up to 650 m. The range stretches from the Peninsula Malacca in the west to the Philippine islands Mindanao and Leyte in the east.
Diagnosis: Worker (
Figure 26, images of specimen CASENT0913595 in
www.antweb.org,
Table 3): Medium-sized, CS 538 µm. Head short, CL/CW 1.119. Postocular distance rather low, PoOc 0.414. Overall head shape in dorsal view rather quadratic with excavated occipital margin (0.9, 2.0, 2.7% CL). Anteromedian vertex in profile slightly bulging. Anterior clypeal margin at level of anterior frontal carinae slightly concave. Antennae 11-segmented, antennal club 3-segmented and very long, its length 57–60% of whole funicular length. Scape extremely long, SL/CS 0.954. Eyes rather small in basal area but much protruding, with scattered or without micropilae, EYE 0.224, moderately elongated EL/EW 1.44. Frons very wide, FRS/CS 0.345, frontal carinae from FRS level caudad slightly converging, planes of frontal laminae sloping mediad by 40°, frontal carinae thus much elevating above the level of posterior clypeus. Mesosoma in lateral view compact, higher than in other species, its dorsal profile ± convex and metanotal depression almost absent (MGr/CS 0.19%). Spines with a very big basal distance (SPBA/CS 0.341), acute and rather long (SP/CS 0.209), their axis deviating by 25–30° from longitudinal mesosomal axis. Petiolar peduncle long. Petiole node higher than in
C. papuana (PeH/CS 0.341), in lateral view in contrast to
C. papuana more produced frontad, with concave anterior profile, the dorsal profile and caudal slope form a continuous convexity; petiole rather narrow (PeW/CS 0.282), its node in dorsal view distinctly wider than long. Postpetiole narrow and rather low (PpW/CS 0.449, PpH/CS 0.312), in dorsal view distinctly wider than long, the anterior sides almost conically converging to a narrow and straight anterior margin the width of which is restricted to the junction with petiole. Whole vertex with densely packed bicoronate foveolae of 13–17 µm µm diameter, the interspaces between foveolae smaller than foveolar diameter and with longitudinal rugulae; in the type sample foveolae on median and paramedian vertex sparse, the wide interspaces here shagreened-corrugated; longitudinal sculptural elements on vertex completely absent. Clypeus with densely packed bicoronate foveolae, the narrow interspaces with rugulae; in the type sample without foveolae and slightly shagreenate-corrugated with suggested rugulae. Whole mesosoma smooth and mildly shiny, finely microreticulate-microrugulose; dorsal mesosoma accessorily with scattered and very shallow foveolae of 4–9 µm diameter. Waist segments smooth and rather shiny, but finely microreticulate. Surface of dorsal gaster sclerites brilliantly shiny, but with traces of a microreticulum. Pubescence on 1st gaster tergite shorter than in
C. papuana and rather dilute (PLG/CS 5.05%, sqPDG 4.10). Whole ant concolorous light yellow.
Taxonomic comments. Reiskind [
47] separated
Cardiocondyla sima and its sister species
C. papuana from
Cardiocondyla by raising Wheeler’s subgenus
Prosopidris [
48] to generic level. Later, Bolton [
23] and Kugler [
49] did not see enough arguments to maintain a genus
Prosopidris. Here, I maintain the subgeneric status but can confirm that
C. sima and
C. papuana share some characters which are not known so far in other
Cardiocondyla species: In the female castes, they have only 11 antennal segments instead of 12 and a bigger antennal club. The length of the three antennal club segments is 57–63% of whole funiculus length. The same ratio is achieved by the 3 antennal club segments plus the preceding segment in the
Cardiocondyla species with 12 antennal segments. It seems as if the former funiculus segments 7 and 8 have fused in
Prosopidris to form a bigger antennal club. Another unique character of the
Prosopidris workers is the conic anterior part of postpetiole in dorsal view.
The single specimen from the island of Leyte is larger, longer-headed and has wider waist segments, much wider and more deeply impressed foveolae on lateral and paramedian vertex, longer pubescence of 1st gastral tergite, and a differing microsculpture on first gaster tergite. It could be separate species but I refrain here from describing it taxonomically.
Biology. Unknown. Specimens were sifted from leaf litter and rotten wood. Concluded from the situation in Cardiocondyla papuana the males should be ergatoid with sickle-shaped mandibles.
4.4.26. Cardiocondyla papuana (Reiskind, 1965)
Prosopidris papuana Reiskind, 1965 [types investigated]
This taxon has been described from Papua New Guinea. Investigated were the holotype and three paratype workers labelled “Bisianumu nr. Sogeri PAPUA 500 m”, “Mar15/20-55 E.O.Wilson rain forest”, “M.C.Z. Type 31156”, and “Holotype Prosopidris papuana Reiskind S.P.Cover 5-00”; the paratypes with the same labelling except for “Paratypes Prosopidris papuana Reiskind S.P.Cover 5-00”; depository MCZ Cambridge.
Geographic range. The three known sites Bisianumu (9.417° S, 147.426° E, 480 m), Bulolo (7.76° S, 147.59° E, 680 m) and Port Moresby (9.25° S, 147.08° E, 9 m) on the Huon peninsula have a maximum distance of 180 km.
Diagnosis: Worker (
Figure 27,
Table 3): Note: Future investigations have to show if the differences between the types and the specimens from Bulolo and Port Moresby which are stated below are taxonomically significant. At the current stage, I consider these as expression of intraspecific polymorphism.Medium-sized, CS 542 µm. Head longer than in C. sima, CL/CW 1.177. Postocular distance rather low, PoOc 0.413. Overall head shape in dorsal view in the types more quadratic with notably excavated hind margin, in the Bulolo and Port Moresby specimens less quadratic and weakly excavated. Anterior clypeal margin in the types evenly convex, in the Bulolo and Port Moresby specimens between the level of anterior frontal carinae straight. Anteromedian vertex in profile slightly bulging. Scape extremely long, SL/CS 0.954. Antennae 11-segmented, antennal club 3-segmented and very long, its length 60–63% of whole funicular length. Eye small (EYE/CS 0.213), in the types with scattered micropilae of 10–14 µm maximum length, in the Bulolo and Port Moresby specimens without micropilae and elongated (EL/EW 1.53). Frons wide, FRS/CS 0.327, frontal carinae immediately posterior of the FRS level converging, planes of frontal laminae sloping mediad by 40°, frontal carinae thus much elevating above the level of posterior clypeus. Mesosoma in lateral view compact, higher than in other species, its dorsal profile ± convex; in the Bulolo and Port Moresby specimens without and the types with a very wide and shallow metanotal depression (MGr/CS overall 1.01%). Spines with a moderate basal distance (SPBA/CS 0.319), acute and moderately long (SP/CS 0.214); spine axis in lateral view deviating in the types by 25° from longitudinal mesosomal axis, in the Bulolo and Port Moresby specimens by 40°. Petiolar peduncle long. Petiole low (PeH/CS 0.314), its node lower than in C. sima and in lateral view slightly produced caudad, with an almost linear frontal, semicircular dorsal, and slightly convex caudal profile. Petiole low (PeH/CS 0.314), in the types narrow (PeW/CS 0.259), in the Bulolo and Port Moresby specimens wider (PeW/CS 0.286). Petiole node in dorsal view distinctly wider than long. Postpetiole rather low (PpH/CS 0.301), in the types narrow (PpW/CS 0.454), in the Bulolo and Port Moresby specimens wider (PpW/CS 0.485). Postpetiole in dorsal view distinctly wider than long, with angularly-rounded sides that converge to a narrow and straight anterior margin the width of which is restricted to the junction with petiole. In the types, whole vertex including frontal laminae homogenously and finely shagreened-corrugated, other sculptural elements are completely absent except for few suggested foveolae of 10–13 µm diameter; in the Bulolo and Port Moresby specimens paramedian and lateral vertex with deeply impressed, rather densely-packed, bicoronate foveolae of 13–16 µm diameter and median vertex with shagreened surface and rather densely packed and slightly smaller bicoronate foveolae. Clypeus in the types slightly longitudinally rugulose, in the Bulolo and Port Moresby specimens matt with about 13 longitudinal rugulae. Whole mesosoma microreticulate-shagreened; in the Bulolo and Port Moresby specimens with scattered, shallow and small foveolae. Petiole rather shiny or matt, finely microreticulate; postpetiole more matt. Surface of first gaster tergite moderately shiny or almost matt and finely microreticulate-corrugated, in the Bulolo and Port Moresby specimens the hairs in anterior part are based in pits. Pubescence on dorsal gaster in the types longer and more dense (PLG/CS 6.80%, sqPDG 3.56) and in the Bulolo and Port Moresby specimens shorter and more dilute (PLG/CS 6.26%, sqPDG 4.13). Whole ant concolorous light yellow.
Taxonomic comments. Cardiocondyla sima and C. papuana are zoogeographically separated by the Wallace Line. The latter species differs from the former by petiole profile, larger CL/CW, smaller PeH/CS and larger PLG/CS.
Biology. The males are ergatoid and have long, sickle-shaped mandibles [
47].